Abstract
Background:
Recent reports of the remarkable therapeutic efficacy of 225Ac-labeled PSMA-617 for therapy of metastatic castration-resistant prostate cancer have under-lined the clinical potential of targeted alpha therapy.
Objective and Conclusion:
This review describes methods for the production of 225Ac and its daughter nuclide 213Bi and summarizes the current clinical experience with both alpha emitters with particular focus on recent studies of targeted alpha therapy of bladder cancer, brain tu-mors, neuroendocrine tumors and prostate cancer.
Keywords: Targeted alpha therapy, alpha emitter, actinium-225, bismuth-213, nuclide production, clinical application
1. INTRODUCTION
The use of alpha emitters for cancer therapy has two distinct advantages over conventional therapies. The short range of alpha radiation in human tissue corresponding to only a few cell diameters (< 0.1 mm) allows the selective killing of targeted cancer cells while sparing surrounding healthy tissue. At the same time, the high energy of alpha radiation of several MeV and its associated high linear energy transfer leads to highly effective cell killing via DNA double strand and DNA cluster breaks, which are largely independent of cell cycle and oxygenation status [1, 2]. Consequently, alpha radiation can kill cells which otherwise exhibit resistance to treatment with beta- or gamma-irradiation or chemotherapeutic drugs, and can thus offer a therapeutic option for patients resistant to conventional therapies [3-5]. Only few alpha-emitting radionuclides are suitable for clinical application in targeted alpha therapy [6]. With the exception of the approved bone-seeking alpha emitter 223Ra, by far most clinical experience is available with 225Ac (T1/2 = 9.9 d) and its short-lived daughter nuclide 213Bi (T1/2 = 46 min). Here we review methods for the production of 225Ac / 213Bi based on radiochemical extraction from 229Th sources as well as accelerator driven processes and give an overview on the existing clinical experience with special focus on recent results in the treatment of bladder cancer, glioma, neuroendocrine tumors and prostate cancer.
2. DECAY CHARACTERISTICS OF 225Ac AND 213Bi
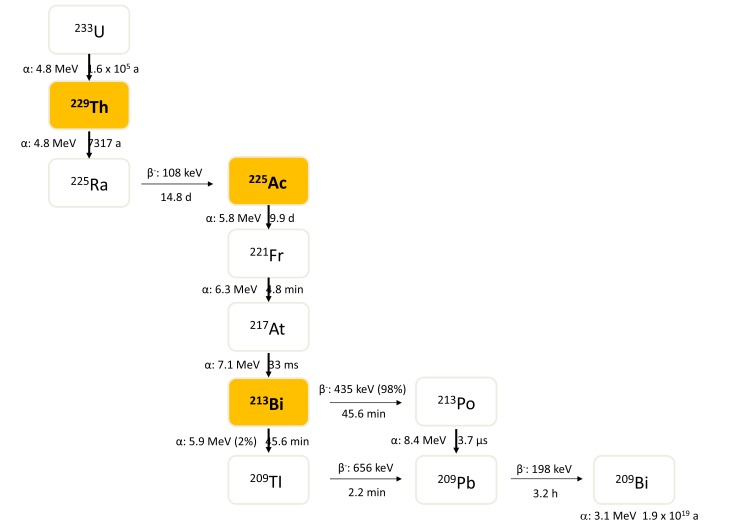
225Ac is a relatively long-lived alpha emitter with a half-life of 9.9 days [7]. It decays via a cascade of six short-lived radionuclide daughters to near stable 209Bi (T1/2 = 1.9x1019 y) (Fig. 1) [8]. The predominant decay path of 225Ac yields net four alpha particles with energies ranging from 5.8 to 8.4 MeV and associated tissue ranges of 47 to 85 µm [1]. In addition, the cascade includes two beta disintegrations of 1.6 and 0.6 MeV maximum energy. Gamma co-emissions useful for in vivo imaging are generated in the 225Ac decay path from the disintegration of 221Fr (218 keV, 11.6% emission probability) and 213Bi (440 keV, 26.1% emission probability). Its long half-life and the multiple alpha particles generated in the decay chain render 225Ac a particularly cytotoxic radionuclide.
Fig. (1).
Decay chain of 233U.
213Bi is a mixed alpha/beta emitter with a half-life of 46 min. It mainly decays via beta- emission to the ultra short-lived, pure alpha emitter 213Po (T1/2 = 4.2 µs, Eα = 8.375 MeV) with a branching ratio of 97.8% (Fig. 1). The remaining 2.2% of 213Bi decays leading to 209Tl via alpha particle emission (Eα = 5.549 MeV, 0.16%, Eα = 5.869 MeV, 2.01%). Both 213Po and 209Tl finally decay via 209Pb (T1/2 = 3.25 h, beta-) into long-lived 209Bi (T1/2 = 1.9x1019 y). The 8.375 MeV alpha particle emitted by 213Po has a path length of 85 µm in human tissue. It is contributing more than 98% of the alpha particle energy emitted per disintegration of 213Bi and can therefore be considered as mainly responsible for its cytotoxic effects. With 92.7% the majority of the total particle energy emitted per disintegration of 213Bi originates from alpha decay, while only 7.3% of decay energy is contributed by beta particle emission, including the decay of 209Pb [1]. The decay of 213Bi is accompanied by the emission of a 440 keV photon (emission probability of 26.1%) that can be detected using SPECT gamma cameras equipped with commercially available high energy collimators, allowing the monitoring of 213Bi-biodistribution and the conduction of pharmacokinetic and dosimetric studies.
3. PRODUCTION OF 225Ac AND 213Bi
3.1. Radiochemical Extraction from 229Th
For more than two decades, the main process utilized for the production of 225Ac and 213Bi has been based on radiochemical extraction from 229Th (T1/2 = 7,317 y) [9] sources originating from the decay of fissile 233U. To date, all clinical tests and the vast majority of pre-clinical research with 225Ac and 213Bi have been conducted using 225Ac / 213Bi obtained from the decay of 229Th. Sources of 229Th allowing the production of clinically relevant activities of 225Ac / 213Bi are available at the Directorate for Nuclear Safety and Security of the Joint Research Centre (JRC) of the European Commission in Karlsruhe, Germany (formerly known as Institute for Transuranium Elements) [10,11], Oak Ridge National Laboratory (ORNL), USA [12] and at the Institute of Physics and Power Engineering (IPPE) in Obninsk, Russia [13]. The 229Th sources have been obtained by separation from aged, fissile 233U originally produced by neutron irradiation of natural 232Th. More recently, the isolation of a 229Th source has also been reported from Canadian Nuclear Laboratories [14].
The production of 225Ac from 229Th at JRC Karlsruhe and ORNL has been described in detail in [10-12] and in a recent review [15]. JRC has been the first laboratory to provide 225Ac / 213Bi to clinical partners in the mid-1990's and since then, has produced approximately 13 GBq 225Ac annually for preclinical research and clinical testing performed at JRC Karlsruhe or in collaboration with a wide network of clinical partners. The maximum annual production of 225Ac at ORNL is approximately 33 GBq, while IPPE has reported a production of 22 GBq per year [13]. 225Ac produced at JRC Karlsruhe and ORNL has been extensively applied for patient treatment and found safe for administration to humans. Direct clinical application of 225Ac produced at IPPE has not been reported to date.
Overall the current worldwide production of 225Ac from 229Th amounts to approximately 68 GBq per year. This level of supply allows the conduction of pre-clinical studies and is sufficient for the treatment of several hundreds of patients per year, in particular when 225Ac-labeled ligands are used where administered activities typically range from 4 – 50 MBq per therapeutic dose. However, the current supply of 225Ac is certainly insufficient for widespread use and routine application in hospitals worldwide. Consequently, a variety of alternative methods for large-scale production of 225Ac have been investigated.
3.2. Accelerator-Based Routes
Alternative ways of producing 225Ac / 213Bi, including the irradiation of 226Ra targets using neutrons, protons, deuterons or gamma-rays and the irradiation of 232Th targets with highly energetic protons have been investigated [16], but have not yet been implemented for a regular supply of 225Ac / 213Bi.
Among these routes, the production of 225Ac by proton irradiation of 226Ra targets in a cyclotron through the reaction 226Ra(p,2n)225Ac is currently the most promising process for large-scale production of 225Ac. The maximum cross-section for the nuclear reaction has been reported as 7.1 x 102 ± 68 mb at 16.8 MeV proton energy [17]. The production can be performed with high yields in a cost-effective manner in medium-sized cyclotrons at proton energies below 20 MeV. An important advantage of the process is that no other long-lived actinium isotopes such as 227Ac (T1/2 = 21.8 y) are co-produced, and chemical purification of the irradiated targets yields 225Ac of high isotopic purity. Co-production of short-lived 226Ac (T1/2 = 29 h) and 224Ac (T1/2 = 2.9 h) according to the reactions, 226Ra(p,n)226Ac and 226Ra(p,3n)224Ac can be minimized through selection of appropriate proton energies. Furthermore, these short-lived actinium isotopes decay to low levels during the time required for target cooling and reprocessing. Although the handling of cyclotron targets made from 226Ra is technically demanding, the production of 225Ac via proton irradiation of 226Ra is the method of choice for large-scale, cost-effective production and can be expected to provide the amounts of high purity 225Ac required for widespread application in the mid-term future.
An alternative accelerator driven method for the production of 225Ac has been developed in recent years based on the irradiation of 232Th targets with high energy protons [18-23]. It has been demonstrated that spallation of 232Th with high energetic protons available at large accelerators can produce significant amounts of 225Ac and production yields in the range of several GBq have been reported for irradiations using intense proton beams and lasting 10 days [20, 21]. The spallation of 232Th simultaneously leads to a significant co-production of radionuclidic impurities that must be removed through appropriate multi-step chemical separation steps [24-26]. However, the main disadvantage of the 232Th based spallation process is the co-production of long-lived 227Ac (T1/2 = 21.8 y) at activity levels of 0.1 to 0.2% relative to the activity of 225Ac at end of bombardment [22]. Since 225Ac and 227Ac isotopes cannot be chemically separated, the implications of the 227Ac impurity for the clinical application of the 225Ac product have to be carefully evaluated. Initial studies investigating the potential toxicity and dosimetric aspects of the 227Ac impurity indicate that the consequences for patient safety might be acceptable [27]. However, issues related to the safe handling and disposal of long-lived 227Ac in hospitals might compromise the clinical acceptance of the product and still have to be addressed.
4. 225Ac / 213Bi RADIONUCLIDE GENERATORS AND LABELLING CHEMISTRY OF 225Ac AND 213Bi
225Ac / 213Bi generators have been recently reviewed in detail [28]. Briefly, 225Ac / 213Bi generators based on AG MP-50 cation exchange resin are most established and have been used for all patient studies with 213Bi to date. JRC Karlsruhe has developed a high activity generator system that allows reliable operation of the generator when loaded with activities up to 4 GBq 225Ac [29]. A key feature of this generator is the homogeneous distribution of 225Ac activity over approximately two-thirds of the generator resin in order to minimize radiolytic degradation of the organic resin and to assure reliable operation over several weeks. The generator has been successfully used for the preparation of therapeutic doses of up to 2.3 GBq 213Bi-Substance P analogue (activity at the time of injection) for locoregional treatment of brain tumors.
The labelling chemistry of 213Bi is well established. 213Bi can be stably linked to biomolecules via derivatives of DTPA (diethylene triamine pentaacetic acid) or DOTA (1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid). Derivatives of the open-chain chelating ligand DTPA are particularly suitable for 213Bi-labelling of antibodies due to their fast complexation kinetics at room temperature. As Bismuth(III) complexes of the pre-organised derivative CHX-A”-DTPA (N-[(R)-2-amino-3-(p-aminophenyl)propyl]-trans-(S,S)-cyclohexane-1,2-diamine-N,N,N’,N”,N”-pentaacetic acid) are exhibiting very high in vivo stability [30, 31], CHX-A”-DTPA is the chelate of choice for 213Bi-labelling of antibodies. Optimized protocols for 213Bi labelling of antibodies in clinical settings allow the synthesis, sterile filtration and quality control of therapeutic doses within 15 min after end of generator elution [32].
Complexes of Bi(III) with the highly pre-organized, octadentate DOTA ligand are kinetically inert and also exhibit high stability in vivo [33, 34]. A synthesis protocol based on microwave heating to 95 °C at pH 9 was developed that allows rapid 213Bi-labelling of DOTA-chelated peptides with radiochemical yields exceeding 99% and specific activities exceeding 50 MBq/nmol within less than 5 min reaction time [35]. Using this robust synthesis scheme, DOTA chelated peptides developed for peptide receptor radiotherapy, such as DOTA-Substance P targeting the neurokinin-1 receptor and the widely used somatostatin-analogs (e.g. DOTATOC, DOTATATE) can be labelled with 213Bi in a straightforward manner.
Labeling of biomolecules with 225Ac(III) is also typically performed via the DOTA chelate. Rapid synthesis can be performed via microwave heating to 95 °C for 5 min at pH 9 with radiochemical yields exceeding 99% at specific activities of 0.1 MBq /nmol [5]. For labelling of heat sensitive biomolecules at temperatures not exceeding 40 °C, slightly longer reaction times are required [36]. Recently, the 18-membered macrocycle N,N′-bis[(6-carboxy-2-pyridil)meth- yl]-4,13-diaza-18-crown-6 (H2macropa) has been investigated for chelation of 225Ac [37]. H2macropa was found to rapidly bind kBq activities of 225Ac at room temperature leading to a complex with high in vivo stability. Therefore H2macropa might offer a valuable alternative for 225Ac labelling of heat sensitive biomolecules. These highly promising results still need to be confirmed in further studies with therapeutic activities of 225Ac in the MBq range.
5. CLINICAL EXPERIENCE WITH 225Ac AND 213Bi
Table 1 gives an overview of the current clinical experience with 225Ac and 213Bi. To date more than 500 patients have received 225Ac- and 213Bi-labeled radioconjugates for therapy of leukemia [38-40], Non-Hodgkins Lymphoma [41], malignant melanoma [42-44], bladder cancer [32, 45], glioma [46-48], neuroendocrine tumors [4, 49], and prostate cancer [5, 50, 51]. All clinical tests listed in Table 1 have been conducted using 225Ac or 225Ac / 213Bi radionuclide generators produced and quality controlled at JRC Karlsruhe or ORNL. The radionuclides obtained from both sites have high radionuclidic and chemical purity, afford high labelling yields and have been found safe for administration to humans by well-trained physicians following established protocols for radiolabeling and quality control.
Table 1.
Overview of clinical experience with 225Ac- and 213Bi-labeled compounds.
| Cancer type | Radioconjugate | Patients | References |
|---|---|---|---|
| Leukemia | 213Bi-HuM195mAb | 49 | [38,39] |
| - | 225Ac-HuM195mAb | 36 | [40] |
| Lymphoma | 213Bi-anti-CD20-mAb | 12 | [41] |
| Melanoma | 213Bi-9.2.27mAb | 54 | [42-44] |
| Bladder Cancer | 213Bi-anti-EGFR-mAb | 12 | [32,45] |
| Glioma | 213Bi-Substance P | 68 | [46-48] |
| - | 225Ac-Substance P | 19 | [48] |
| Neuroendocrine tumors | 213Bi-DOTATOC | 25 | [4] |
| - | 225Ac-DOTATOC | 39 | [49] |
| Prostate cancer | 225Ac-PSMA-617 | 190 | [5,50,51] |
In view of the currently strongly increasing interest in clinical application of particularly 225Ac-labeled radiocon- jugates it is of utmost importance that 225Ac obtained from sources other than JRC or ORNL is carefully evaluated for purity and safety before administration to humans is considered by physicians. 225Ac sources of inferior purity may contain potentially toxic radionuclide impurities and may result in low radiochemical yields during radiolabelling. Appropriate purification and validation procedures must be implemented before application to humans. Synthesis and quality control of 225Ac-labeled ligands for clinical application should be performed only by well-trained staff with experience in handling and analysing 225Ac and its daughter nuclides.
5.1. Radioimmunotherapy with 213Bi and 225Ac
The pioneering first-in-human clinical investigations of 213Bi-labeled antibodies for therapy of leukemia [38,39], melanoma [42-44] and Non-Hodgkins Lymphoma [41] have been reviewed in detail previously [15]. Based on the promising results of two studies investigating the safety and therapeutic efficacy of the 213Bi-labeled anti-CD33 antibody lintuzumab for therapy of leukemia, clinical studies investigating the 225Ac labelled analogue are conducted and are summarized in a separate review in this issue.
More recently, a pilot study on the locoregional treatment of bladder cancer (carcinoma in situ) using the 213Bi-labelled anti-EGFR monoclonal antibody cetuximab has been conducted in collaboration of JRC Karlsruhe and Technical University Munich, Germany [32,45]. Carcinoma in situ (CIS) is a high-risk bladder cancer due to its tendency to invade neighbouring tissue. Standard treatment options for CIS include transurethral resection and intravesical instillation of Bacillus Calmette-Guerin (BCG). However, a significant fraction of patients eventually becomes unresponsive to BCG treatment and radical cystectomy is typically performed at this stage with an associated drastically reduced quality of life. In this pilot study of targeted alpha therapy, 12 patients scheduled for cystectomy were offered salvage treatment by intravesical instillation of 366-821 MBq 213Bi-labelled anti-EGFR monoclonal antibody. 11 out of 12 patients were treated only once in this pilot study, while one patient was treated twice. The therapy was found to be safe and without any side effects, no activity of 213Bi was detected outside the bladder. Remarkably, 3 out of 12 patients achieved complete remissions already after one (2/12) or two (1/12) treatments, thus avoiding or delaying cystectomy. These results show that locoregional targeted alpha therapy with 213Bi-anti-EGFR-MAb is a promising new option for therapy of CIS. Escalation of the administered dose and increasing the number of treatments can be expected to further enhance the therapeutic efficacy and should be investigated in a follow-up study.
5.2. Peptide Receptor Alpha Therapy with 213Bi and 225Ac
Targeted peptide receptor alpha therapy with 213Bi / 225Ac has to date been clinically tested for treatment of brain tumors, neuroendocrine tumors and prostate cancer. Important advantages of utilizing low molecular weight ligands as targeting vehicles for 213Bi and 225Ac include fast tumor uptake and rapid clearance of unbound conjugates from circulation, thus reducing haematological toxicity. Internalizing ligands are particularly advantageous for application in combination with 225Ac in order to harness the multiple alpha particles emitted in its decay chain.
5.3. 225Ac- and 213Bi-substance P analogues for Glioma Therapy
Peptide receptor alpha therapy of brain tumors has been tested for the first time in two patients as early as 1999 in collaboration of JRC Karlsruhe with University Hospital Basel [46] and subsequently, five more patients were treated in 2007/2008 [47]. The metabolically stabilized substance P analogue DOTA-[Thi8,Met(O2)11]-substance P was used as targeting vector. Substance P is the physiological ligand of the neurokinin-1 (NK-1) receptor that is consistently overexpressed in WHO grade II–IV gliomas and that has also been detected on tumour cells infiltrating the intra- and peritumoural vasculature [52]. The low molecular weight peptide (1.8 kDa) has been shown to diffuse rapidly and to be able to localise in remote satellite lesions [46]. In order to overcome the blood-brain barrier and to increase tumor uptake, administration of 213Bi-[Thi8,Met(O2)11]-substance P was conducted locoregionally via an implanted catheter system with a subcutaneous port. The treatment was found to be safe without severe adverse effects. Remarkably, all 3 early patients diagnosed with grade II gliomas still do not show signs of recurrence or neurological impairments 10 years (2/3) and 18 years (1/3) after therapy.
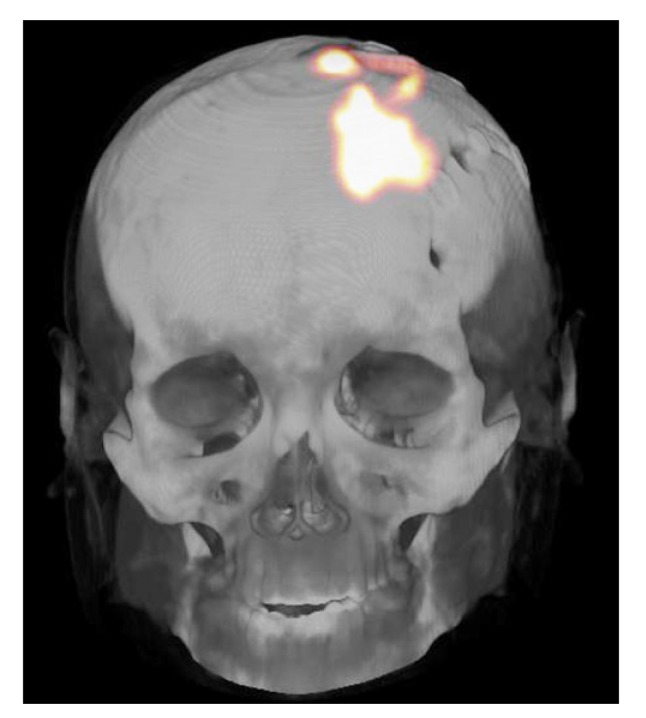
Based on the promising outcome of these initial clinical experiences with a locoregional injection of 213Bi-[Thi8, Met(O2)11]-substance P, a follow-up investigation was conducted in collaboration of JRC Karlsruhe and Medical University Warsaw [48]. The patient group consisted of 61 patients with grade II to IV gliomas, including primary and secondary recurrent glioblastoma. Patients were treated with up to 14.1 GBq 213Bi-[Thi8,Met(O2)11]-substance P, administered in up to 8 treatment cycles at two month intervals. Biodistribution was monitored via PET/CT after co-injection of 68Ga-[Thi8,Met(O2)11]-substance P. The treatment was well tolerated, no severe adverse events were observed. PET/CT imaging demonstrated high retention of the radiolabelled peptide at the tumor site (Fig. 2), less than 8% I.D. was found in the blood pool within five hours post injection. Fig. (3) shows magnetic resonance images of a patient with grade III glioma showing excellent response to locoregional treatment with 213Bi-Substance P analogue. The patient was treated with 8 cycles of 213Bi-[Thi8,Met(O2)11]-substance P with a cumulative activity of 14.1 GBq in two-month intervals. 44 months after administration of the 8th treatment cycle the patient is in excellent clinical condition. Overall analysis of the therapeutic efficacy in the 61 patients treated to date is ongoing, an interim subgroup analysis indicates prolonged survival times for grade IV patients in comparison to standard treatments [53].
Fig. (2).
68Ga-DOTATOC PET/CT image of a patient with recurrent glioblastoma after co-injection of 10 MBq 68Ga-[Thi8,Met(O2)11]-substance P and 1.7 GBq 213Bi-[Thi8,Met(O2)11]-substance P. The image taken at 1 hour post injection shows high retention of the radiolabelled compound in the tumor area, with some residual activity remaining in the catheter system.
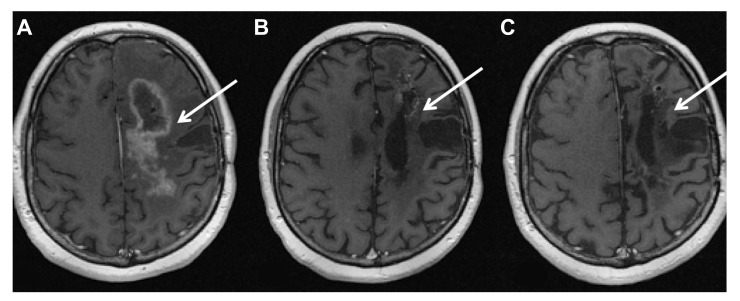
Fig. (3).
Magnetic resonance images of a patient with grade III glioma showing excellent response to locoregional treatment with 8 cycles of 213Bi-Substance P analogue with a cumulative activity of 14.1 GBq (2 months after first therapy (A); 8 months after 8th therapy (B); 33 months after 8th therapy (C)).
Widespread glioma cell infiltration into normal adjacent brain areas is the main cause for failure of glioma treatment. Although low molecular weight substance P analogues exhibit relatively rapid diffusion, the short half-life of 213Bi of 46 min might compromise the delivery of sufficient doses to remote tumor cells. In addition, high activity 225Ac/213Bi generators required for preparation of therapeutic activities in the multi GBq range are currently still very costly and in limited supply. Therapeutic application of longer-lived 225Ac could overcome these limitations, and in vitro characterisation of the 225Ac labelled derivative DOTA-[Thi8, Met(O2)11]-substance P showed promising anti-tumor efficacy [54]. Recently an initial dose escalation study investigating the intratumoral/intercavitary injection of 225Ac-DOTAGA-[Thi8,Met(O2)11]-substance P has been started [48]. To date, 19 glioma patients have been treated with activities ranging from 10 to 42 MBq 225Ac-DOTAGA-[Thi8, Met(O2)11]-substance P. The treatment has been well tolerated, analysis of therapeutic efficacy and patient recruitment is ongoing.
5.4. 225Ac- and 213Bi-DOTATOC for Therapy of Neuroendocrine Tumors
Peptide receptor alpha therapy of neuroendocrine tumors was first investigated clinically in collaboration JRC Karlsruhe and University Hospital Heidelberg. Twenty-five patients with multi-resistant neuroendocrine tumors refractory to therapy with 90Y-/177Lu-DOTATOC were treated with 213Bi-DOTATOC. Administration was performed inter-arterially into the main tumor-feeding vessel in 21 patients, while 4 patients received intravenous injections. Cumulative activities ranging from 2.6 to 21 GBq of 213Bi-DOTATOC were administered within 1-5 cycles in two-month intervals. An interim analysis of the first 8 patients showed that chronic kidney toxicity was moderate and hematoxicity was less pronounced than with preceding beta therapies [4]. Remarkably, intravenous administration of 3.3 GBq of 213Bi-DOTATOC in a patient with extensive bone marrow involvement did not lead to relevant acute haematological side effects, in accordance with the concept of selective tumor cell irradiation with the short-range alpha emitter. Although all patients reported in this investigation were in different challenging situations and had developed resistance against therapy with beta emitters, targeted alpha therapy with 213Bi-DOTATOC resulted in a high number of long-lasting anti–tumour responses, including one complete remission. The results of this investigation provided convincing clinical data demonstrating that targeted alpha therapy can offer a valuable additional treatment option to patients refractory to therapy with beta emitters. Furthermore, the study also demonstrated that targeted alpha therapy with short-range alpha radiation is also effective against solid lesions, as impressively illustrated in the SNMMI image of the year 2012 [55]. 213Bi-DOTATOC can evolve as a promising compound for therapy of neuroendocrine tumors once the supply limitations for high activity 25Ac/213Bi generators are resolved.
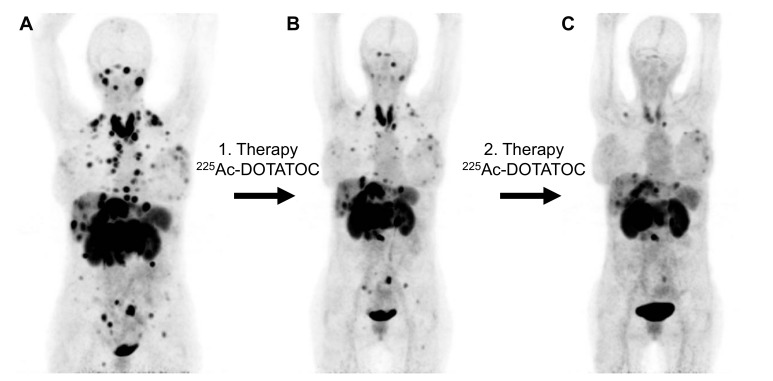
Therapy of neuroendocrine tumors using 225Ac-DOTATOC was clinically tested in a follow-up investigation with 39 patients with progressive neuroendocrine tumors [56]. The investigation was conducted as an empiric dose escalation to find the Maximum Tolerable Dose (MTD) of a single cycle and fractionation concepts. The MTD of a single cycle 225Ac-DOTATOC was considered to be 40 MBq. Multiple fractions were tolerated with 25 MBq every 4 months or 18.5 MBq every 2 months. Cumulative activities of 75 MBq were found tolerable in regard to delayed toxicity. The observed radiologic treatment response was without clear preference of a particular fractionation concept. An example of a partial response observed in a patient with a multi-resistant neuroendocrine tumor after two treatment cycles with 16 MBq (1st cycle) and 42 MBq (2nd cycle) 225Ac-DOTATOC is shown in Fig. (4).
Fig. (4).
68Ga-DOTATOC PET/CT images of a patient with a multi-resistant neuroendocrine tumor showing partial response after two treatment cycles with 16 MBq (1st cycle) and 42 MBq (2nd cycle) 225Ac-DOTATOC. (A): before therapy (Oct 2011); (B): 4 months after first therapy with 16 MBq 225Ac-DOTATOC (Feb 2012); (C): 3 months after second therapy with 42 MBq 225Ac-DOTATOC (May 2012).
5.5. 225Ac- PSMA-617 for Therapy of Prostate Cancer
The implementation of 225Ac-PSMA-617 for therapy of metastatic castration-resistant prostate cancer constitutes a major advancement in targeted alpha therapy [57]. 225Ac-PSMA-617 was first developed and characterised in vitro at JRC Karlsruhe in 2013/2014 and a microwave-assisted protocol for reliable synthesis and quality control of clinical doses was established. Based on the first reports of the promising clinical efficacy of PSMA-617 labelled with the beta emitter 177Lu and the enhanced cytotoxicity of alpha compared to beta emitters, the concept of combining 225Ac with the ligand PSMA-617 seemed highly promising. The key features of the pharmacokinetics of PSMA-617, including its fast tumor uptake, high internalization rate, extended tumor retention and rapid clearance of unbound ligand, are highly favourable for combination with an alpha emitter with a half-life of several days and multiple alpha emissions in its decay chain. Clinical testing is currently conducted in collaborations of JRC Karlsruhe with University Hospital Heidelberg, Steve Biko Academic Hospital Pretoria and Technical University Munich.
A dosimetry estimate comparing 225Ac-PSMA-617 and 213Bi-PSMA-617 demonstrated that short-lived 213Bi is an inferior choice for targeted alpha therapy with PSMA-617. 213Bi-PSMA-617 suffers from higher perfusion-dependent off-target radiation and a longer biological half-life of PSMA-617 in dose-limiting organs than the physical half-life of 213Bi [58]. To our knowledge, there are no reports to date on the clinical application of 213Bi-PSMA-617 except for a single case report and hence the subject is not discussed further [59].
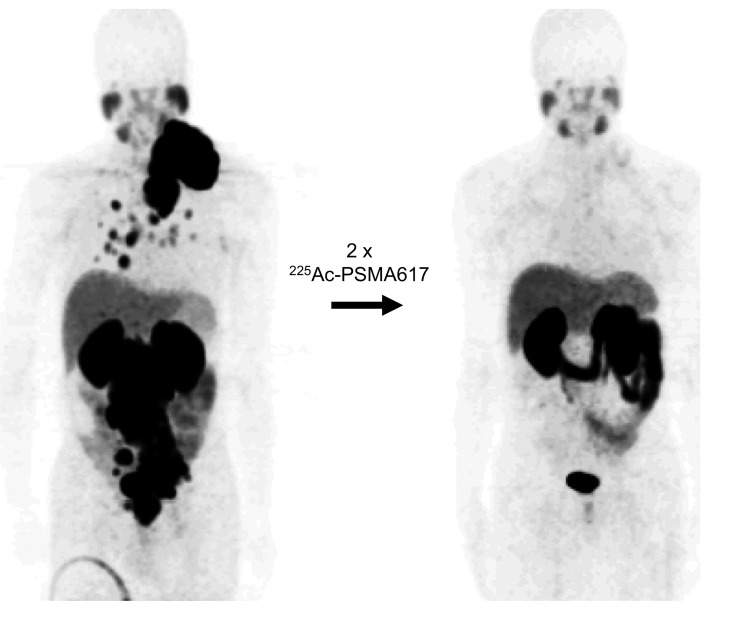
The first report of the remarkable therapeutic efficacy of 225Ac-PSMA-617 in patients with late-stage metastatic castration-resistant prostate cancer (mCRPC) presented complete remissions observed in two patients in highly challenging situations [5]. To develop a standardized treatment protocol for 225Ac-PSMA-617 therapy in advanced-stage mCRPC patients, a dosimetry estimate was calculated on the basis of time–activity curves derived from serially obtained 177Lu-PSMA-617 scans and salvage therapies empirically conducted with 50 to 200 kBq/kg of 225Ac-PSMA-617 were evaluated retrospectively regarding toxicity and treatment response. For advanced-stage patients, a treatment activity of 100 kBq/kg of 225Ac-PSMA-617 per cycle repeated every 8 weeks was found to present a reasonable trade-off between toxicity and biochemical response [50]. An interim analysis of the efficacy of this treatment protocol was conducted in a retrospectively analysed group of forty patients with mCRPC. Evaluation of PSA and radiological response demonstrated remarkable anti-tumor activity of 225Ac-PSMA-617 and swimmer-plot analysis indicated the promising duration of tumor-control, especially taking into account the unfavorable prognostic profile of the selected advanced-stage patients. Xerostomia (dry mouth syndrome) was the main reason to discontinue therapy or to refuse additional administrations, indicating that further modifications of the treatment regimen with regard to side effects might be necessary to further enhance the therapeutic range [51]. One of the excellent examples of the therapeutic efficacy of 225Ac-PSMA-617 in a patient with mCRPC that was progressive under conventional therapy is shown in Fig. (5). The patient was treated at Steve Biko Academic Hospital, Pretoria, with two cycles of 225Ac-PSMA-617 with a cumulative activity of 14 MBq. Restaging with 68Ga- PSMA PET/CT after 5 months showed a remarkable molecular imaging response. This patient also demonstrated a biochemical response with a decrease in PSA level from 1,301 to <0.05 ng/mL.
Fig. (5).
Excellent response to therapy after 2 cycles of 225Ac-PSMA-617 demonstrated by 68Ga-PSMA-11 PET/CT images of patient with metastatic castration-resistant prostate cancer before (left panel) and after (right panel), with decrease in serum PSA level from 1,301 to <0.05 ng/mL. The patient was treated at Steve Biko Academic Hospital, Pretoria, with two cycles of 225Ac-PSMA-617 with a cumulative activity of 14 MBq.
CONCLUSION
The recent reports on the remarkable therapeutic efficacy of 225Ac-PSMA-617 for the therapy of mCRPC have significantly sparked interest in the clinical application of targeted alpha therapy. The implementation of 225Ac-PSMA-617 does not only provide a promising therapeutic option for the second most frequent cancer in men, but also successfully underlines the significant potential of the concept of targeted alpha therapy as such. The clinical data recently obtained on targeted alpha therapy of neuroendocrine tumors and prostate cancer convincingly show that therapy with alpha emitters can overcome resistance to therapy with conventional drugs and also with beta emitters and can offer a valuable additional treatment option to patients that have failed established treatments. The combination of 225Ac or 213Bi with low molecular weight peptide ligands seems particularly promising due to the favourable pharmacokinetics of these ligands. The clinical experience with locoregional treatment of glioma with 225Ac- and 213Bi-labelled substance P analogues is promising and needs to be validated further. The intravesical therapy of bladder cancer with 213Bi-anti-EGFR MAb is an intriguing approach that certainly deserves further study. For all these promising novel approaches formal clinical studies are highly warranted to compare the safety and efficacy against approved therapies under controlled conditions.
To satisfy the increased clinical demand for 225Ac, accelerator driven production routes, preferably based on proton irradiation of 226Ra in medium energy cyclotrons, should be rapidly implemented. In general, the safety of 225Ac obtained from alternative sources should be carefully evaluated before its administration to humans is considered. 225Ac and 213Bi are highly cytotoxic radionuclides that can provide remarkable benefit to cancer patients. Their clinical application should be conducted by well-trained physicians, following established protocols and guidelines for radiolabeling and quality control.
Consent for Publication
Not applicable.
Acknowledgements
Declared none.
Conflict of Interest
The authors declare no conflict of interest, financial or otherwise.
References
- 1.Sgouros G., Roeske J.C., McDevitt M.R., Palm S., Allen B.J., Fisher D.R., Brill A.B., Song H., Howell R.W., Akabani G., SNM MIRD Committee Bolch, W.E.; Brill, A.B.; Fisher, D.R.; Howell, R.W.; Meredith, R.F.; Sgouros, G.; Wessels, B.W.; Zanzonico, P.B. MIRD pamphlet no. 22 (abridged): Radiobiology and dosimetry of a-particle emitters for targeted radionuclide therapy. J. Nucl. Med. 2010;51(2):311–328. doi: 10.2967/jnumed.108.058651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wulbrand C., Seidl C., Gaertner F.C., Bruchertseifer F., Morgenstern A., Essler M., Senekowitsch-Schmidtke R. Alpha-particle emitting 213Bi-anti-EGFR immunoconjugates eradicate tumor cells independent of oxygenation. PLoS One. 2013;8:e64730. doi: 10.1371/journal.pone.0064730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Friesen C., Glatting G., Koop B., Schwarz K., Morgenstern A., Apostolidis C., Debatin K.M., Reske S.N. Cancer Res. 2007;67(5):1950–1958. doi: 10.1158/0008-5472.CAN-06-3569. [DOI] [PubMed] [Google Scholar]
- 4.Kratochwil C., Giesel F.L., Bruchertseifer F., Mier W., Apostolidis C., Boll R., Murphy K., Haberkorn U., Morgenstern A. 213Bi-DOTATOC receptor-targeted alpha-radionuclide therapy induces remission in neuroendocrine tumours refractory to beta radiation: A first-in-human experience. Eur. J. Nucl. Med. Mol. Imaging. 2014;41(11):2106–2119. doi: 10.1007/s00259-014-2857-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kratochwil C., Bruchertseifer F., Giesel F.L., Weis M., Verburg F.A., Mottaghy F., Kopka K., Apostolidis C., Haberkorn U., Morgenstern A. 225Ac-PSMA-617 for PSMA targeting alpha-radiation therapy of patients with metastatic castration-resistant prostate cancer. J. Nucl. Med. 2016;57(12):1941–1944. doi: 10.2967/jnumed.116.178673. [DOI] [PubMed] [Google Scholar]
- 6.Seidl C. Radioimmunotherapy with α-particle-emitting radionuclides. Immunotherapy. 2014;6:431–458. doi: 10.2217/imt.14.16. [DOI] [PubMed] [Google Scholar]
- 7.Pommé S., Marouli M., Suliman G., Dikmen H., Van Ammel R., Jobbágy V., Dirican A., Stroh H., Paepen J., Bruchertseifer F., Apostolidis C., Morgenstern A. Measurement of the 225Ac half-life. Appl. Radiat. Isot. 2012;70:2608–2614. doi: 10.1016/j.apradiso.2012.07.014. [DOI] [PubMed] [Google Scholar]
- 8.Suliman G., Pommé S., Marouli M., Van Ammel R., Stroh H., Jobbágy V., Paepen J., Dirican A., Bruchertseifer F., Apostolidis C., Morgenstern A. Half-lives of (221)Fr, (217)At, (213)Bi, (213)Po and (209)Pb from the (225)Ac decay series. Appl. Radiat. Isot. 2013;77:32–37. doi: 10.1016/j.apradiso.2013.02.008. [DOI] [PubMed] [Google Scholar]
- 9.Varga Z., Nicholl A., Mayer K. Determination of the 229Th half-life. Phys. Rev. C. 2014;89:064310. [Google Scholar]
- 10.Apostolidis C., Molinet R., Rasmussen G., Morgenstern A. Production of Ac-225 from Th-229 for targeted alpha therapy. Anal. Chem. 2005;77:6288–6291. doi: 10.1021/ac0580114. [DOI] [PubMed] [Google Scholar]
- 11.Zielinska B., Apostolidis C., Bruchertseifer F., Morgenstern A. An improved method for the production of Ac-225/Bi-213 from Th-229 for targeted alpha therapy. Solvent Extr. Ion Exch. 2007;25(3):339–349. [Google Scholar]
- 12.Boll R.A., Malkemus D., Mirzadeh S. Production of actinium-225 for alpha particle mediated radioimmunotherapy. Appl. Radiat. Isot. 2005;62:667–679. doi: 10.1016/j.apradiso.2004.12.003. [DOI] [PubMed] [Google Scholar]
- 13.Samsonov M.D., Nerozin N.A., Podsoblyaev D.A., Prokof’ev I.V., Tkachev S.V., Khamianov S.V., Shapovalov V.V. Isolation of alpha-emitting radionuclides for nuclear medicine in JSC “SSC RF – IPPE.; Proceedings of the 10th International Symposium on Targeted Alpha Therapy; Kanazawa, Japan. May 30; 2017. [Google Scholar]
- 14.Causey P., Bureau D., Leeder K., Perron R., Hartimath S.V., Fonge H. Production of a Thorium/Actinium Generator at the Canadian Nuclear Laboratories.; Proceedings of the 10th International Symposium on Targeted Alpha Therapy; Kanazawa, Japan. May 30; 2017. [Google Scholar]
- 15.Morgenstern A., Bruchertseifer F., Apostolidis C. Targeted alpha therapy with 213Bi. Curr. Radiopharm. 2011;4(4):295–305. doi: 10.2174/1874471011104040295. [DOI] [PubMed] [Google Scholar]
- 16.Morgenstern A., Abbas K., Bruchertseifer F., Apostolidis C. Production of alpha emitters for targeted alpha therapy. Curr. Radiopharm. 2008;1:135–143. [Google Scholar]
- 17.Apostolidis C., Molinet R., McGinley J., Abbas K., Möllenbeck J., Morgenstern A. Cyclotron production of Ac-225 for targeted alpha therapy. Appl. Radiat. Isot. 2005;62:383–387. doi: 10.1016/j.apradiso.2004.06.013. [DOI] [PubMed] [Google Scholar]
- 18.Zhuikov B.L., Kalmykov S.N., Ermolaev S.V., Aliev R.A., Kokhanyuk V.M., Matushko V.L., Tananaev I.G., Myasoedov B.F. Production of 225Ac and 223Ra by irradiation of Th with accelerated protons. Radiochemistry. 2011;53(1):73–80. [Google Scholar]
- 19.Ermolaev S.V., Zhuikov B.L., Kokhanyuk V.M., Matushko V.L., Kalmykov S.N., Aliev R.A., Tananaev I.G., Myasoedov B.F. Production of actinium, thorium and radium isotopes from natural thorium irradiated with protons up to 141 MeV. Radiochim. Acta. 2012;100:223–229. [Google Scholar]
- 20.Weidner J.W., Mashnik S.G., John K.D., Hemez F., Ballard B., Bach H., Birnbaum E.R., Bitteker L.J., Couture A., Dry D., Fassbender M.E., Gulley M.S., Jackman K.R., Ullmann J.L., Wolfsberg L.E., Nortier F.M. Proton-induced cross sections relevant to production of 225Ac and 223Ra in natural thorium targets below 200MeV. Appl. Radiat. Isot. 2012;70:2602–2607. doi: 10.1016/j.apradiso.2012.07.006. [DOI] [PubMed] [Google Scholar]
- 21.Weidner J.W., Mashnik S.G., John K.D., Ballard B., Birnbaum E.R., Bitteker L.J., Couture A., Fassbender M.E., Goff G.S., Gritzo R., Hemez F.M., Runde W., Ullmann J.L., Wolfsberg L.E., Nortier F.M. 225Ac and 223Ra production via 800 MeV proton irradiation of natural thorium targets. Appl. Radiat. Isot. 2012;70:2590–2595. doi: 10.1016/j.apradiso.2012.07.003. [DOI] [PubMed] [Google Scholar]
- 22.Griswold J.R., Medvedev D.G., Engle J.W., Copping R., Fitzsimmons J.M., Radchenko V., Cooley J.C., Fassbender M.E., Denton D.L., Murphy K.E., Owens A.C., Birnbaum E.R., John K.D., Nortier F.M., Stracener D.W., Heilbronn L.H., Mausner L.F., Mirzadeh S. Large scale accelerator production of 225Ac: Effective cross sections for 78-192MeV protons incident on 232Th targets. Appl. Radiat. Isot. 2016;118:366–374. doi: 10.1016/j.apradiso.2016.09.026. [DOI] [PubMed] [Google Scholar]
- 23.Hoehr C., Bénard F., Buckley K., Crawford J., Gottberg A., Hanemaayer V., Kunz P., Ladouceur K., Radchenko V., Ramogida C., Robertson A., Ruth T., Zacchia N., Zeisler S., Schaffer P. Medical isotope production at TRIUMF- from imaging to treatment. Phys. Procedia. 2017;90:200–208. [Google Scholar]
- 24.Aliev R., Ermolaev A., Vasiliev A., Ostapenko V., Lapshina E., Zhuikov B. Isolation of medicine-applicable actinium-225 from thorium targets irradiated by medium-energy cyclotron. Solvent Extr. Ion Exch. 2014;32:468–477. [Google Scholar]
- 25.Mastren T., Radchenko V., Owens A., Copping R., Boll R., Griswold J.R., Mirzadeh S., Wyant L.E., Brugh M., Engle J.W., Nortier F.M., Birnbaum E.R., John K.D., Fassbender M.E. Simultaneous separation of actinium and radium isotopes from a proton irradiated thorium matrix. Sci. Rep. 2017;7(1):8216. doi: 10.1038/s41598-017-08506-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Radchenko V., Engle J.W., Wilson J.J., Maassen J.R., Nortier F.M., Taylor W.A., Birnbaum E.R., Hudston L.A., John K.D., Fassbender M.E. Application of ion exchange and extraction chromatography to the separation of actinium from proton-irradiated thorium metal for analytical purposes. J. Chromatogr. A. 2015;1380:55–63. doi: 10.1016/j.chroma.2014.12.045. [DOI] [PubMed] [Google Scholar]
- 27.Abergel R.J. How Biodistribution, Toxicity, and Chelation of Accelerator-produced Actinium-225 will Determine its Fate in Targeted Alpha Therapy.; Proceedings of the 10th International Symposium on Targeted Alpha Therapy; Kanazawa, Japan. May 30; 2017. [Google Scholar]
- 28.Morgenstern A., Bruchertseifer F., Apostolidis C. Bismuth-213 and actinium-225 generator performance and evolving therapeutic applications of two generator-derived alpha-emitting radioisotopes. Curr. Radiopharm. 2012;5(3):221–227. doi: 10.2174/1874471011205030221. [DOI] [PubMed] [Google Scholar]
- 29.Bruchertseifer F., Apostolidis C., Mirzadeh S., Boll R., Murphy K., Morgenstern A. Development of a High-activity 225Ac/213Bi Radionuclide Generator for Synthesis of Clinical Doses of 213Bi-labelled Biomolecules.; Proceedings of the 8th International Symposium on Targeted Alpha Therapy; Oak Ridge, USA. 2013. [Google Scholar]
- 30.Wu C., Gansow O.A., Brechbiel M.W. evaluation of methods for large scale preparation of antibody ligand conjugates. Nucl. Med. Biol. 1999;26(3):339–342. doi: 10.1016/s0969-8051(98)00112-7. [DOI] [PubMed] [Google Scholar]
- 31.Xu H., Baidoo K.E., Wong K.J., Brechbiel M.W. A novel bifunctional maleimido CHX-A” chelator for conjugation to thiol-containing biomolecules. Bioorg. Med. Chem. Lett. 2008;18(8):2679–2683. doi: 10.1016/j.bmcl.2008.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Autenrieth M.E., Seidl C., Bruchertseifer F., Horn T., Kurtz F., Feuerecker B., D’Alessandria C., Pfob C., Apostolidis C., Gschwend J.E., Schwaiger M., Scheidhauer K., Morgenstern A. Treatment of carcinoma in situ of the urinary bladder with an alpha-emitter immunoconjugate targeting the epidermal growth factor receptor: A pilot study. Eur. J. Nucl. Med. Mol. Imaging. 2018;45(8):1364–1371. doi: 10.1007/s00259-018-4003-6. [DOI] [PubMed] [Google Scholar]
- 33.Ruegg C.L., Anderson-Berg W.T., Brechbiel M.W., Mirzadeh S., Gansow O.A., Strand M. Improved in vivo stability and tumor targeting of bismuth-labeled antibody. Cancer Res. 1990;50:4221–4226. [PubMed] [Google Scholar]
- 34.Junghans R.P., Dobbs D., Brechbiel M.W., Mirzadeh S., Raubitschek A.A., Gansow O.A., Waldmann T.A. Pharmacokinetics and bioactivity of 1,4,7,10-tetra-azacylododecane n,n′,n″,n‴- tetraacetic acid (dota)-bismuth-conjugated anti-tac antibody for α-emitter (212Bi) therapy. Cancer Res. 1993;53(23):5683–5689. [PubMed] [Google Scholar]
- 35.Bruchertseifer F., Good S., Apostolidis C., Mäcke H., Morgenstern A. An improved method for Bi-213 labelling of DOTA-/DOTAGA-chelated peptides. Eur. J. Nucl. Med. Mol. Imaging. 2006;33(S2):S161. [Google Scholar]
- 36.Morgenstern A., Bruchertseifer F., Apostolidis C. 2015.
- 37.Thiele N.A., Brown V., Kelly J.M., Amor-Coarasa A., Jermilova U., MacMillan S.N., Nikolopoulou A., Ponnala S., Ramogida C.F., Robertson A.K.H., Rodríguez-Rodríguez C., Schaffer P., Williams C., Jr, Babich J.W., Radchenko V., Wilson J.J. An eighteen-membered macrocyclic ligand for actinium-225 targeted alpha therapy. Angew. Chem. Int. Ed. Engl. 2017;56(46):14712–14717. doi: 10.1002/anie.201709532. [DOI] [PubMed] [Google Scholar]
- 38.Jurcic J.G., Larson S.M., Sgouros G., McDevitt M.R., Finn R.D., Divgi C.R., Ballangrud A.M., Hamacher K.A., Ma D., Humm J.L., Brechbiel M.W., Molinet R., Scheinberg D.A. Targeted alpha particle immunotherapy for myeloid leukemia. Blood. 2002;100(4):1233–1239. [PubMed] [Google Scholar]
- 39.Rosenblat T.L., McDevitt M.R., Mulford D.A., Pandit-Taskar N., Divgi C.R., Panageas K.S., Heaney M.L., Chanel S., Morgenstern A., Sgouros G., Larson S.M., Scheinberg D.A., Jurcic J.G. Sequential cytarabine and alpha-particle immunotherapy with bismuth-213-lintuzumab (HuM195) for acute myeloid leukemia. Clin. Cancer Res. 2010;16(21):5303–5311. doi: 10.1158/1078-0432.CCR-10-0382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Berger M., Jurcic J., Scheinberg D. Efficacy of 225Ac-labeled Anti-CD33 Antibody in Acute Myeloid Leukemia (AML) Correlates with Peripheral Blast Count.; Proceedings of the 10th International Symposium on Targeted Alpha Therapy; Kanazawa, Japan. May 30 -June 1, 2017. [Google Scholar]
- 41.Schmidt D., Neumann F., Antke C., Apostolidis C., Martin S., Morgenstern A., Molinet R., Heeger S., Kronenwett R., Müller H.W., Haas R. In: Proceedings of the 4th Alpha-immunotherapy symposium. Morgenstern A., editor. Düsseldorf, Germany: ITU; 2004. http://itu.jrc.ec.europa.eu/index.php?id=205 [Google Scholar]
- 42.Allen B.J., Raja C., Rizvi S., Li Y., Tsui W., Graham P., Thompson J.F., Reisfeld R.A., Kearsley J., Morgenstern A., Apostolidis C. Intralesional targeted alpha therapy for metastatic melanoma. Cancer Biol. Ther. 2005;4(12):1318–1324. doi: 10.4161/cbt.4.12.2251. [DOI] [PubMed] [Google Scholar]
- 43.Raja C., Graham P., Abbas Rizvi S.M., Song E., Goldsmith H., Thompson J., Bosserhoff A., Morgenstern A., Apostolidis C., Kearsley J., Reisfeld R., Allen B.J. Interim analysis of toxicity and response in phase 1 trial of systemic targeted alpha therapy for metastatic melanoma. Cancer Biol. Ther. 2007;6(6):846–852. doi: 10.4161/cbt.6.6.4089. [DOI] [PubMed] [Google Scholar]
- 44.Allen B.J., Singla A.A., Rizvi S.M., Graham P., Bruchertseifer F., Apostolidis C., Morgenstern A. Analysis of patient survival in a Phase I trial of systemic targeted α-therapy for metastatic melanoma. Immunotherapy. 2011;3(9):1041–1050. doi: 10.2217/imt.11.97. [DOI] [PubMed] [Google Scholar]
- 45.Autenrieth M.E., Horn T., Kurtz F., Nguyen K., Morgenstern A., Bruchertseifer F., Schwaiger M., Blechert M., Seidl C., Senekowitsch-Schmidtke R., Gschwend J.E., Scheidhauer K. Intravesical radioimmunotherapy of carcinoma in situ of the urinary bladder after BCG failure. Urologe A. 2017;56(1):40–43. doi: 10.1007/s00120-016-0273-4. [DOI] [PubMed] [Google Scholar]
- 46.Kneifel S., Cordier D., Good S., Ionescu M.C., Ghaffari A. Hofer, Sv Kretzschmar, M.; Tolnay, M.; Apostolidis, C.; Waser, B.; Arnold, M.; Mueller-Brand, J.; Maecke, H.R.; Reubi, J.C.; Merlo, A. Local targeting of malignant gliomas by the diffusible peptidic vector 1,4,7,10-tetraazacyclododecane-1-glutaric acid-4,7,10-triacetic acid-substance P. Clin. Cancer Res. 2006;12(12):3843–3850. doi: 10.1158/1078-0432.CCR-05-2820. [DOI] [PubMed] [Google Scholar]
- 47.Cordier D., Forrer F., Bruchertseifer F., Morgenstern A., Apostolidis C., Good S., Müller-Brand J., Mäcke H., Reubi J.C., Merlo A. Targeted alpha-radionuclide therapy of functionally critically located gliomas with 213Bi-DOTA-[Thi8, Met(O2)11]-substance P: A pilot trial. Eur. J. Nucl. Med. Mol. Imaging. 2010;37(7):1335–1344. doi: 10.1007/s00259-010-1385-5. [DOI] [PubMed] [Google Scholar]
- 48.Krolicki L., Bruchertseifer F., Kunikowska J., Koziara H., Królicki B., Jakuciński M., Pawlak D., Apostolidis C., Rola R., Merlo A., Morgenstern A. Targeted alpha therapy of glioblastoma multiforme: Clinical experience with 213Bi- and 225Ac-Substance P.; Proceedings of the 10th International Symposium on Targeted Alpha Therapy; Kanazawa, Japan. May 30; 2017. [Google Scholar]
- 49.Kratochwil C. Bruchertseifer, F.; Giesel, F.; Apostolidis, C.; Haberkorn U.; Morgenstern, A. Ac-225-DOTATOC - an empiric dose finding for alpha particle emitter based radionuclide therapy of neuroendocrine tumors. J. Nucl. Med. 2015;56(S3):1232. [Google Scholar]
- 50.Kratochwil C., Bruchertseifer F., Rathke H., Bronzel M., Apostolidis C., Weichert W., Haberkorn U., Giesel F.L., Morgenstern A. Targeted Alpha Therapy of mCRPC with 225Actinium-PSMA-617: Dosimetry estimate and empirical dose finding. J. Nucl. Med. 2017;58(10):1624–1631. doi: 10.2967/jnumed.117.191395. [DOI] [PubMed] [Google Scholar]
- 51.Kratochwil C., Bruchertseifer F., Rathke H., Hohenfellner M., Giesel F.L., Haberkorn U., Morgenstern A. Targeted Alpha Therapy of mCRPC with 225Actinium-PSMA-617: Swimmer-Plot analysis suggests efficacy regarding duration of tumor-control. J. Nucl. Med. 2018;59(5):795–802. doi: 10.2967/jnumed.117.203539. [DOI] [PubMed] [Google Scholar]
- 52.Hennig I.M., Laissue J.A., Horisberger U., Reubi J.C. Substance-P receptors in human primary neoplasms: tumoral and vascular localization. Int. J. Cancer. 1995;61(6):786–792. doi: 10.1002/ijc.2910610608. [DOI] [PubMed] [Google Scholar]
- 53.Krolicki L., Bruchertseifer F., Kunikowska J., Koziara H., Królicki B., Jakuciński M., Pawlak D., Apostolidis C., Mirzadeh S., Rola R., Merlo A., Morgenstern A. Prolonged survival in secondary glioblastoma following local injection of targeted alpha therapy with 213Bi-substance P analogue. Eur. J. Nucl. Med. Mol. Imaging. 2018;45(9):1636–1644. doi: 10.1007/s00259-018-4015-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Majkowska-Pilip A., Rius M., Bruchertseifer F., Apostolidis C., Weis M., Bonelli M., Laurenza M., Krolicki L., Morgenstern A. In vitro evaluation of the novel 225Ac-DOTA-Substance P for targeted alpha therapy of glioblastoma multiforme. Chem. Biol. Drug Des. 2018;92(1):1344–1356. doi: 10.1111/cbdd.13199. [DOI] [PubMed] [Google Scholar]
- 55.SNMMI Image of the year 2012. J. Nucl. Med. 2012;53(10):23N. [PubMed] [Google Scholar]
- 56.Kratochwil C., Bruchertseifer F., Giesel F.L., Apostolidis C., Haberkorn U., Morgenstern A. Ac-225-DOTATOC - dose finding for alpha particle emitter based radionuclide therapy of neuroendocrine tumors. Eur. J. Nucl. Med. Mol. Imaging. 2015;42(S1):S36. doi: 10.1007/s00259-014-2857-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Fendler W.P., Cutler C. More α than β for prostate cancer? J. Nucl. Med. 2017;58:1709–1710. doi: 10.2967/jnumed.117.198333. [DOI] [PubMed] [Google Scholar]
- 58.Kratochwil C., Schmidt K., Afshar-Oromieh A., Bruchertseifer F., Rathke H., Morgenstern A., Haberkorn U., Giesel F.L. Targeted alpha therapy of mCRPC: Dosimetry estimate of 213Bismuth-PSMA-617. Eur. J. Nucl. Med. Mol. Imaging. 2018;45(1):31–37. doi: 10.1007/s00259-017-3817-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sathekge M., Knoesen O., Meckel M., Modiselle M., Vorster M., Marx S. (213)Bi-PSMA-617 targeted alpha-radionuclide therapy in metastatic castration-resistant prostate cancer. Eur. J. Nucl. Med. Mol. Imaging. 2017;44(6):1099–1100. doi: 10.1007/s00259-017-3657-9. [DOI] [PMC free article] [PubMed] [Google Scholar]