Abstract
Introduction
The purpose of this investigation was to: (1) to determine the reliability of rectus femoris muscle cross-sectional area and echo intensity obtained using panoramic ultrasound imaging during seated and supine lying positions before and after a 5-minute rest period and (2) to determine the influence of body position and rest period on the magnitude of rectus femoris muscle cross-sectional area and echo intensity measurements.
Methods
A total of 23 males and females (age = 21.5 ± 1.9 years) visited the laboratory on two separate occasions. During each visit, panoramic ultrasound images of the rectus femoris were obtained in both a seated and a supine position before (T1) and after a 5-minute (T2) rest period to quantify any potential changes in either muscle cross-sectional area and/or echo intensity.
Results
None of the muscle cross-sectional area or echo intensity measurements exhibited systematic variability, and the ICCs were 0.98–0.99 and 0.88–0.91, and the coefficients of variation were ≤ 3.9% and ≤ 8.2% for muscle cross-sectional area and echo intensity, respectively. Our results indicated that muscle cross-sectional area was greater in the seated than supine position, whereas echo intensity was greater in the supine position. Further, echo intensity increased in the seated position from T1 to T2.
Conclusion
Both rectus femoris muscle cross-sectional area and echo intensity may be reliably measured in either a seated or supine lying position before or after a 5-minute rest period. Aside from echo intensity in the seated position, rest period had no influence on the magnitude of muscle cross-sectional area or echo intensity. Comparison of muscle cross-sectional area values that are obtained in different body positions is ill-advised.
Keywords: Panoramic ultrasound imaging, fluid re-distribution, reproducibility, bi-articular muscle, muscle architecture
Introduction
Ultrasound (US) imaging has gained attention for its ability to provide valid,1,2 reliable,3–7 and sensitive8–13 measurements of skeletal muscle size. Recent advancements in US technology include the ability to construct two-dimensional cross-sectional images of muscles (i.e., muscle cross-sectional area; mCSA) from panoramic US scans, which has further increased the popularity of US imaging for muscle size measurements. Furthermore, when compared to magnetic resonance imaging and computerized tomography, US is more time- and cost-effective. Consequently, many studies have utilized this technology to examine acute skeletal muscle swelling in response to exercise,14,15 the effects of resistance training on skeletal muscle hypertrophy,1,12,16,17 the effects of detraining or aging on skeletal muscle atrophy,18–21 among other applications.
In addition to mCSA, a novel measurement that can be obtained from a panoramic US scan is the echo intensity (EI) of skeletal muscle. This measurement originally attracted interest as a method to noninvasively assess the composition of muscle tissue as a surrogate to muscle quality.22–24 However, EI has also been shown to be related to changes in cellular glycogen content25,26 and sensitive to tissue hydration,27 resistance training adaptations,28–30 and muscle damage.14,31 Thus, due to its sensitivity and the ease with which it can be measured, EI has also become a popular noninvasive measurement of skeletal muscle status with a wide-variety of applications.
Typically, panoramic US images of the leg extensors are performed while subjects are lying supine. Furthermore, scans are typically only taken after the subject has rested quietly for several minutes due to the suggestion that regional fluid shifts occur from the action of gravitational forces within the muscle and subcutaneous tissue,32 which may ultimately alter mCSA and EI measurements. Arroyo et al.3 compared the effect of supine rest on muscle size and EI of the vastus lateralis (VL) and did not observe changes in muscle thickness or EI across time, but did observe a time-dependent decrease in mCSA. The authors noted that “further research involving the rectus femoris is required to determine whether time of supine rest may differentially affect ultrasound measurements of the other thigh components” (p. 3). Arroyo et al.3 also did not report how rest time affected the reliability of their measurements. Finally, because of the anterior location of the rectus femoris (RF) and the ease with which it can be imaged while seated, investigators may wish to know the effect of body position (i.e., seated vs. lying prone) on the reliability and magnitude mCSA and EI measurements of the RF.
Therefore, the purpose of this investigation was to: (1) to determine the reliability of RF mCSA and EI obtained using panoramic US imaging during seated and supine positions before and after a 5-minute rest period and (2) to determine the influence of body position and rest period on the magnitude of RF mCSA and EI measurements.
Materials and methods
Participants
Twenty-three males and females between the ages of 18 and 39 volunteered to participate in this investigation (men: n = 13, age: 21.6 ± 2.2 years, weight: 85.0 ± 11.1 kg, height: 178.1 ± 7.6 cm, BMI = 26.8 ± 3.6 kg·m − 2; women: n = 10, 21.4 ± 3.6 years, 64.7 ± 7.0 kg, 165.4 ± 6.9 cm, 23.7 ± 2.3 kg·m−2). Prior to enrollment in the study, written informed consent was obtained from all participants. Following consent, each participant completed an exercise history questionnaire to quantify individual exercise habits. The participants in this study were currently performing an average of 5.9 ± 4.0 hours/week of aerobic exercise, 3.2 ± 2.1 hours/week of resistance exercise, and 2.1 ± 1.0 hours/week of recreational exercise. In addition, participants had performed aerobic, resistance, and recreational exercise for 10.0 ± 5.6 years, 5.6 ± 4.4 years, and 9.1 ± 5.4 years, respectively. All participants were instructed to refrain from engaging in any lower body physical activity 48 hours prior to testing. This investigation was approved by the University’s Institutional Review Board (IRB #: ED-16-164).
Experimental design
Participants reported to the laboratory on two occasions, separated by 48 hours, for US examination of the RF of the right leg. During each visit, panoramic US images of the RF were obtained in seated and supine lying positions before (T1) and after a 5-minute (T2) rest period. The US images were analyzed following image acquisition to quantify mCSA and EI.
Ultrasonography
Upon arrival to the laboratory at visit one, height and weight were recorded for each participant (Physician Scale 439; Detecto, Webb City, MO). Images were then obtained using a brightness mode (B-mode) ultrasound imaging device (General Electric LOGIQ S8, Wauwatosa, WI) and a multi-frequency linear-array probe (Model ML6-15-D 4-15 MHz, 50-mm field of view). To maximize acoustic coupling and reduce near-field artifacts, a generous amount of water-soluble transmission gel was applied to the skin and ultrasound probe. The ultrasound probe was positioned on the skin surface over the RF, and great care was taken to ensure that the ultrasound probe was oriented perpendicular to the RF and that consistent minimal pressure was applied to the probe to limit compression of the muscle. The image gain was set at 50 decibels (dB), the frequency at 12 megahertz (MHz), dynamic range was set at 72 dB, and the image depth was set at a range of 5–7 cm. Image depth was dependent on the tissue thickness of individual participants’ thigh but was kept consistent within individuals. Panoramic US images of the RF were taken along the axial plane at 50% of the distance from the anterior superior iliac spine to the proximal border of the patella.10 This location was marked and used on both testing days.
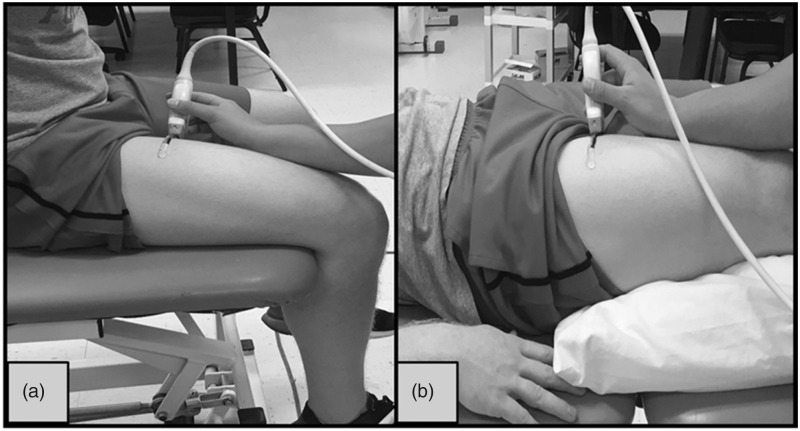
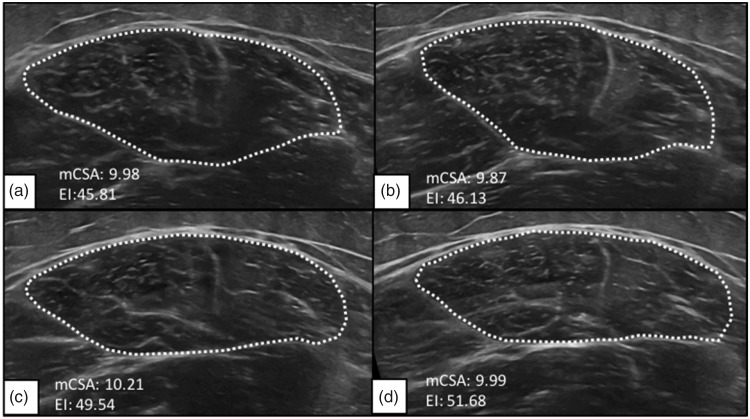
During testing, participants were examined in two different body positions: (1) seated upright on the edge of the padded plinth with their leg relaxed, hanging freely, and a 90° joint angle at the knee (Figure 1(a)), and (2) lying supine atop a padded plinth with their legs completely relaxed on top of a pillow and the foot of the measured leg braced in a custom-made foot mold that prevented external rotation of the femur (Figure 1(b)). Participants were asked to assume each position, seated and supine at two separate time points during both visit one and two. During each position and time point, three transverse panoramic images were captured from the RF (i.e., 12 total images). First, images were acquired with participants situated in the seated position (STT1) (Figure 2(a)). Directly following the acquisition of STT1 images, participants were instructed to lay supine, and images were captured immediately once set in the supine position (SPT1) (Figure 2(c)). After the SPT1 position, participants remained lying supine for 5 minutes to allow for potential fluid shifts. After the 5-minute rest period, images were obtained in the supine position for the second time (SPT2) (Figure 2(d)). Finally, participants resumed the seated position (STT2) (Figure 2(b)), and the final three images were taken. The same procedures from visit one were repeated for visit two.
Figure 1.
Two different body positions: (a) seated upright with the leg relaxed, hanging freely with 90° joint angle at the knee and (b) lying supine with the legs completely relaxed on top of a pillow.
Figure 2.
Representative ultrasound images of the rectus femoris muscle while the participant was (a and b, respectively) seated upright and (c and d, respectively) lying supine before and after the 5-minute rest period, respectively.
Ultrasound image analysis
All images were analyzed using image analysis software (ImageJ, version 1.50i) available from the National Institutes of Health (NIH, Bethesda, Maryland). To determine RF mCSA, the images were analyzed by selecting a region of interest that included as much muscle as possible without including any surrounding fascia using the polygon function in ImageJ. RF EI was quantified by analyzing the gray-scale of each individual image through the application of the standard histogram function in ImageJ to the same region of interest used to determine mCSA. The average EI was calculated as an arbitrary unit (au) value between 0 and 255 (0 = black; 255 = white).33–35 The three mCSA (cm2) and EI (au) values were then averaged for each of the body positions at T1 and T2 (i.e., SPT1, STT1, SPT2, STT2) and used for further analysis.
Statistical analysis
One-way repeated-measures analyses of variance (ANOVAs) were used to compare the means between visits for systematic variability. Test–retest reliability for mCSA and EI was examined by calculating the intraclass correlation coefficient (ICC) using model “2, k” because this model can be generalized to other testers and laboratories. The 95% confidence interval (CI) for each ICC2,k was also calculated as described previously36,37 and was used to test the null hypothesis that each ICC was equal to zero and that ICCs were equal independent of position or time.38,39 For measures of absolute reliability, the standard error of the measurement (SEM) was calculated as the square root of the mean square error term from the ANOVA table.40 The coefficient of variation (CV) was calculated as a normalized measure of the SEM by expressing the SEM relative to the grand mean.41 All data were analyzed using IBM SPSS Statistics v. 23 (IBM, Armonk, NY) and a custom written spreadsheet (Microsoft Excel, Microsoft Corporation, Redmond, WA).
Two separate, three-way within-subjects ANOVAs ((visit [visit 1 vs. visit 2] × position [seated vs. supine] × time [T1 vs. T2]) were used to analyze mCSA and EI. Significant interactions were decomposed with follow-up, lower-order ANOVAs, and Bonferroni-corrected dependent samples t-tests on the simple main effects. Significant main effects that were not involved in an interaction were analyzed with Bonferroni-corrected dependent samples t-tests on the marginal means. Partial-eta squared effect sizes () were calculated for each ANOVA. All statistical analyses were completed using IBM SPSS Statistics (v. 22; Armonk, NY) and a type I error rate was set a priori at 5%.
Results
Reliability
The grand means and test–retest reliability statistics of mCSA and EI for each position are displayed in Table 1. There was no systematic variability between visits 1 and 2 for any of the dependent variables (p > 0.05). The ICC2,k for each dependent variable was greater than zero (p ≤ 0.05). Furthermore, the ICC2,k’s were not different among position and time of measurement for either mCSA or EI, as indicated by the overlapping 95% confidence intervals. The CVs for mCSA and EI were ≤ 3.9% and ≤ 8.2%, respectively. In agreement with the ICCs, the CVs did not appear to be dependent on the position or measurement time for mCSA or EI.
Table 1.
Test–retest reliability statistics for muscle cross-sectional area (mCSA) and echo intensity for the supine and seated positions before (T1) and after (T2) resting quietly for 5 minutes.
| Body position | Grand mean | ICC2,k, 95% CI | SEM | CV (%) | p-Value | |
|---|---|---|---|---|---|---|
| mCSA | Seated T1 | 13.2 cm2 | 0.99, 0.96–0.99 | 0.36 | 2.8 | 0.70 |
| Supine T1 | 12.4 cm2 | 0.98, 0.95–0.99 | 0.54 | 4.4 | 0.95 | |
| Supine T2 | 12.4 cm2 | 0.98, 0.96–0.99 | 0.47 | 3.8 | 0.52 | |
| Seated T2 | 13.0 cm2 | 0.98, 0.96–0.99 | 0.51 | 3.9 | 0.95 | |
| Echo Intensity | Seated T1 | 35.7 au | 0.91, 0.78–0.96 | 2.90 | 8.1 | 0.10 |
| Supine T1 | 41.3 au | 0.90, 0.77–0.96 | 2.97 | 7.2 | 0.11 | |
| Supine T2 | 41.9 au | 0.91, 0.78–0.96 | 2.71 | 6.5 | 0.37 | |
| Seated T2 | 39.3 au | 0.88, 0.70–0.95 | 3.21 | 8.2 | 0.67 |
ICC2,k, 95% CI: intraclass correlation coefficient (model 2,k) and 95% confidence interval; SEM: standard error of measurement; CV: coefficient of variation; mCSA: muscle cross-sectional area; p-value: type 1 error rate for the one-way repeated-measures ANOVAs used to assess systematic variability.
Muscle cross-sectional area
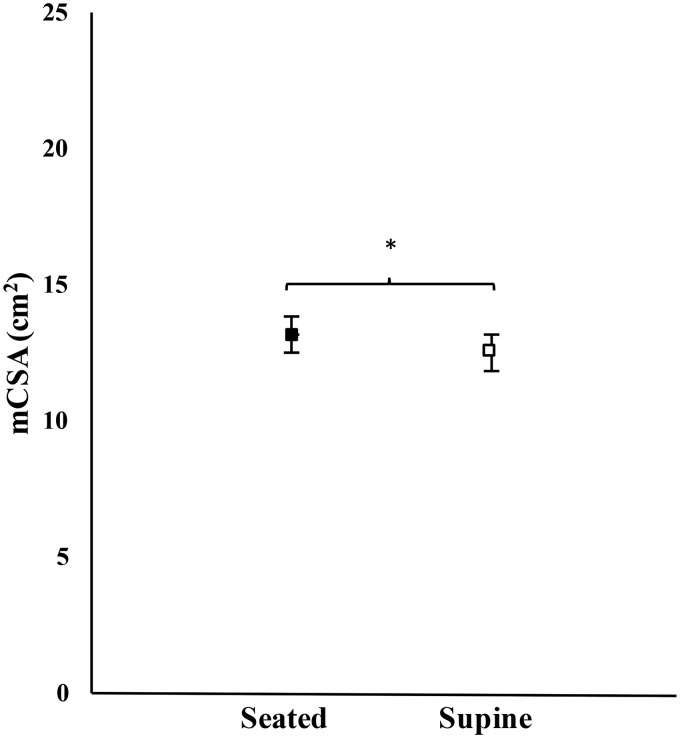
For mCSA, there was no significant three-way interaction for visit × position × time (F = 0104, p = 0.750, = 0.005), nor significant two-way interactions for visit × time (F = 1.102, p = 0.306, = 0.052), visit × position (F = 0.289, p = 0.597, = 0.014), or position × time (F = 1.090, p = 0.309, = 0.052). There was also no main effect for visit (F = 0.006, p = 0.941, < 0.001) or time (F = 4.245, p = 0.053, = 0.175). However, there was a significant main effect for position (F = 17.663, p < 0.001, < 0.469). Post hoc analyses indicated that mCSA was significantly greater during the seated versus supine positions (Bonferroni-corrected p-value, pBC < 0.001) (Figure 3).
Figure 3.
Mean (±standard error) muscle cross-sectional area (mCSA; collapsed across visit and time) of rectus femoris during the seated (closed box) and supine (open box) positions. * indicates mCSA was greater during the seated than supine position (p ≤ 0.05).
Echo intensity
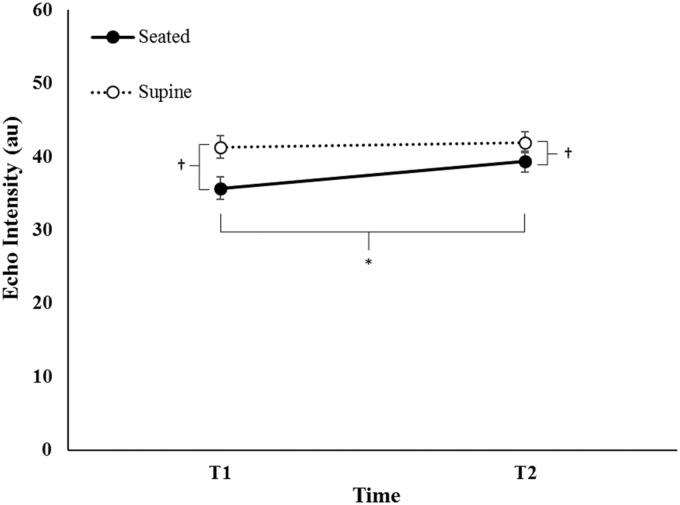
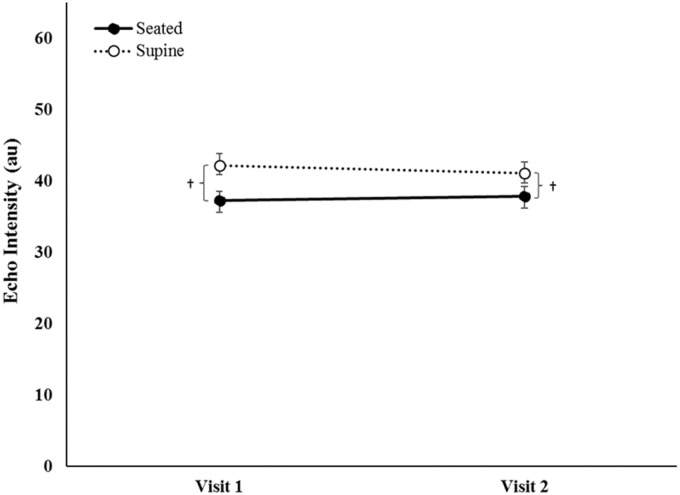
For EI, there was no significant three-way interaction for day × position × time (F = 3.365, p = 0.082, = 0.144), nor a two-way interaction for visit × time (F = 0.574, p = 0.457, = 0.028). However, there were significant two-way interactions for visit × position (F = 7.194, p = 0.014, = 0.265) and position × time (F = 21.986, p < 0.05, = 0.144). Post hoc analyses indicated that EI (collapsed across visit) significantly increased from T1 to T2 in the seated position (pBC < 0.001), but was not different from T1 to T2 in the supine position (Figure 4). EI (collapsed across visit) was greater in the supine position compared to the seated position at T1 and T2 (pBC < 0.001). Furthermore, EI (collapsed across time) was greater for the supine position during both visits (pBC < 0.001) but did not change from visit one to visit two in either the seated or supine positions (Figure 5).
Figure 4.
Mean (±standard error) echo intensity (collapsed across visit) of rectus femoris for the seated and supine positions before (T1) and after (T2) resting quietly for 5 minutes. † indicates EI was greater in the supine than seated position (p ≤ 0.05). * indicates an increase from T1 to T2 for the seated position (p ≤ 0.05)
Figure 5.
Mean (±standard error) of echo intensity (collapsed across time) of rectus femoris for the seated and supine positions during visits 1 and 2. † indicates EI is greater in the supine than seated position (p ≤ 0.05).
Discussion
We examined the influence of body position and a 5-minute rest period on the reliability and magnitude of US assessed mCSA and EI. The primary results of the present study indicated that the ICCs and CVs for mCSA ranged from 0.98 to 0.99 and 2.8% to 4.4%, respectively. The ICCs and CVs for EI ranged from 0.88 to 0.91 and 6.5% to 8.2%, respectively. There was also no systematic difference in either of the mCSA or EI measurements from visit 1 to visit 2. Furthermore, the reliability of mCSA and EI did not appear to depend on the position or the time at which the US images were taken. However, body position did influence the magnitude of RF mCSA, which was greater in the seated than supine position, and influenced EI, which was lower in the seated than supine position. In addition, EI increased in the seated position from before (STT1) to after (STT2) the 5-minute rest period. Therefore, overall, our results suggest that although ultrasound measurements of RF mCSA and EI are comparably reliable when performed in a seated or supine position before or after a 5-minute rest period, body position influenced mCSA and both the body position and rest period influenced measurements of EI.
Typically, panoramic US images of the leg extensors are performed when subjects are supine after several minutes of resting quietly. Several studies have quantified the test–re-test reliability of RF mCSA in this position,5,42–44 whereas only one to our knowledge has reported RF EI,45 and have reported high ICCs (i.e., 0.982–0.993) and low CVs (i.e., 2.8–4.4%) for these measurements. In the present study, however, we were interested in whether (1) providing a rest period before obtaining ultrasound images; and (2) the body position in which subjects were situated when images were obtained would affect the reliability of mCSA and EI measurements. Our results suggested that the RF mCSA and EI measurements obtained in this study were comparably reliable to those reported previously.5,46 Moreover, the 95% CIs for the ICCs for mCSA and EI in the present study overlapped, indicating that relative reliability was not dependent upon body position or the time at which the images were obtained. Therefore, our results agree with and extend previous studies reporting the reliability of RF mCSA and/or EI.5,46,47 RF mCSA and EI demonstrate high relative and absolute reliability independent of body position or regardless pre- or postsupine rest period.
There was a no change in mCSA from T1 to T2; however, mCSA was significantly greater in the seated when compared to the supine position. The lack of change in mCSA between T1 and T2 suggests that a prescribed rest period has very little influence on the measurement of RF mCSA. In contrast, our data suggest there was an effect of position on RF mCSA, which is likely due to the bi-articular architecture of the RF.48 Specifically, the RF originates from the anterior inferior iliac spine and the rim of the acetabulum and the fibrous capsule of the hip joint and inserts at the tibial tuberosity via the patellar ligament.49 Thus, the RF is shortened when both the hip and knee are flexed, as in the seated position, and lengthened when the hip and knee are extended, as in the supine lying position.50 Therefore, it is not surprising that RF mCSA was greater in the seated versus supine position. Given the reliability of RF mCSA measurements in the seated and supine positions, our data suggest that obtaining mCSA measurements are valid in either position, but comparison of mCSA values across positions is unnecessary. Furthermore, future studies need to examine the extent to which a change in body position would affect the other muscles of the quadriceps femoris, since these muscles are uniarticular.
Arroyo et al.3 previously reported that supine rest periods had no effect on EI of the VL. However, Arroyo et al.3 noted that future studies were needed to determine whether rest periods might influence other muscles such as the RF. Similar to Arroyo et al.,3 we did not observe a change in EI after the rest period when US images were obtained with subjects in the supine position. In contrast, we observed an increase in RF EI after the rest period when the images were obtained in the seated position. Therefore, our data suggest that body position may influence the need for a rest period before obtaining US images. Alternatively, it is possible that greater changes were observed in the seated than supine position due to the order of testing (i.e., seated, supine, supine, seated), but this seems unlikely given the minimal amount of time between the seated and supine measurements at T1 and T2. It is also possible that regional fluid shifts had a greater effect on the images obtained in the seated than supine positions, but studies are needed to further explore this effect. Overall, if images are obtained in the supine position, our data in combination with those of Arroyo et al.3 suggests that a supine rest period may not be necessary when measuring mCSA or EI of the leg extensors.
In the present study, EI was also greater in the supine than seated positions. As previously discussed with mCSA, it is likely that this is a result of joint position related changes in the shape of the RF due to its anatomy or changes to the curvature of the thigh (Figure 1). For example, EI is influenced by factors such as curvature of the limb and the angle of incidence of the US probe, which may have changed due to body position. Anecdotally, we noted that the RF appeared flatter in the images obtained in the supine than seated position. This would cause the lower boundary of the RF to be in closer proximity to the US probe and provide less opportunity for US wave attenuation. In either case, our results suggest that comparison of EI values across positions, like mCSA, is inadvisable.
The purpose of this study was twofold: (1) to determine the reliability of RF mCSA and EI obtained using panoramic US imaging during seated and supine lying positions before and after an often-recommended rest period and (2) to determine the influence of body position and rest period on the magnitude of RF mCSA and EI measurements. Overall, the results of the present study indicated that panoramic US imaging is a reliable technique for measuring mCSA and EI of the RF. Additionally, our data suggest that a 5-minute rest period has no effect on RF mCSA measurements or on EI in a supine position, though, EI was greater after the rest period in the seated position. Moreover, body position influenced both mCSA and EI, which were greater and lower, respectively, in the seated versus supine position. Therefore, reliable measurements of RF mCSA and EI may obtain in either position and before or after a prescribed rest period to allow for potential fluid shifts.32 Aside from EI in the seated position, rest period had no influence on the magnitude of mCSA or EI, and a comparison of mCSA values that are obtained in different body positions is ill-advised.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This investigation was approved by Oklahoma State University’s Institutional Review Board (IRB #: ED-16-164).
Patient consent
Written consent was obtained from the patient for publishing their photographs and images in this article. PMT gives permission to this journal to publish the photographs and images in this article.
Guarantor
Nathaniel DM Jenkins
Contributorship
PMT was a contributor to study concept and design, carried out data analysis and interpretation, and was the primary author. TWDM, MAM, RJC, and MJL helped in carrying out data acquisition and interpretation. PMT and TWDM created figures. NDMJ was the primary manuscript reviser and a substantial contributor to study concept, study design, and interpretation. All authors read, revised, and approved the final version of this manuscript. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- 1.Ahtiainen JP, Hoffren M, Hulmi JJ, et al. Panoramic ultrasonography is a valid method to measure changes in skeletal muscle cross-sectional area. Eur J Appl Physiol 2010; 108: 273–273. [DOI] [PubMed] [Google Scholar]
- 2.Hammond K, Mampilly J, Laghi FA, et al. Validity and reliability of rectus femoris ultrasound measurements: comparison of curved-array and linear-array transducers. J Rehabil Res Dev 2014; 51: 1155–1155. [DOI] [PubMed] [Google Scholar]
- 3.Arroyo E, Stout JR, Beyer KS, et al. Effects of supine rest duration on ultrasound measures of the vastus lateralis. Clin Physiol Funct Imaging 2016; 38: 155–157. [DOI] [PubMed] [Google Scholar]
- 4.Rosenberg JG, Ryan ED, Sobolewski EJ, et al. Reliability of panoramic ultrasound imaging to simultaneously examine muscle size and quality of the medial gastrocnemius. Muscle Nerve 2014; 49: 736–740. [DOI] [PubMed] [Google Scholar]
- 5.Ruas CV, Pinto RS, Lima CD, et al. Test–retest reliability of muscle thickness, echo-intensity and cross sectional area of quadriceps and hamstrings muscle groups using B-mode ultrasound. Int J Kinesiol Sports Sci 2017; 5. [Google Scholar]
- 6.Scott JM, Martin DS, Ploutz-Snyder R, et al. Reliability and validity of panoramic ultrasound for muscle quantification. Ultrasound Med Biol 2012; 38: 1656–1661. [DOI] [PubMed] [Google Scholar]
- 7.Thomaes T, Thomis M, Onkelinx S, et al. Reliability and validity of the ultrasound technique to measure the rectus femoris muscle diameter in older CAD-patients. BMC Med Imaging 2012; 12: 7–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ahtiainen JP, Walker S, Peltonen H, et al. Heterogeneity in resistance training-induced muscle strength and mass responses in men and women of different ages. Age 2016; 38: 10–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Croix MBDS, Deighan MA, Armstrong N. Assessment and interpretation of isokinetic muscle strength during growth and maturation. Sports Med 2003; 33: 727–743. [DOI] [PubMed] [Google Scholar]
- 10.Ema R, Wakahara T, Miyamoto N, et al. Inhomogeneous architectural changes of the quadriceps femoris induced by resistance training. Eur J Appl Physiol 2013; 113: 2691–2703. [DOI] [PubMed] [Google Scholar]
- 11.Roelofs EJ, Smith-Ryan AE, Melvin MN, et al. Muscle size, quality, and body composition: characteristics of division I cross-country runners. J Strength Cond Res 2015; 29: 290–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Scanlon TC, Fragala MS, Stout JR, et al. Muscle architecture and strength: adaptations to short-term resistance training in older adults. Muscle Nerve 2014; 49: 584–592. [DOI] [PubMed] [Google Scholar]
- 13.Wells AJ, Fukuda DH, Hoffman JR, et al. Vastus lateralis exhibits non-homogenous adaptation to resistance training. Muscle Nerve 2014; 50: 785–793. [DOI] [PubMed] [Google Scholar]
- 14.Buckner SL, Dankel SJ, Mattocks KT, et al. Differentiating swelling and hypertrophy through indirect assessment of muscle damage in untrained men following repeated bouts of resistance exercise. Eur J Appl Physiol 2017; 117: 213–224. [DOI] [PubMed] [Google Scholar]
- 15.Jenkins ND, Housh TJ, Bergstrom HC, et al. Muscle activation during three sets to failure at 80 vs. 30% 1RM resistance exercise. Eur J Appl Physiol 2015; 115: 2335–2347. [DOI] [PubMed] [Google Scholar]
- 16.Jenkins ND, Housh TJ, Buckner SL, et al. Neuromuscular adaptations after 2 and 4 weeks of 80% versus 30% 1 repetition maximum resistance training to failure. J Strength Cond Res 2016; 30: 2174–2185. [DOI] [PubMed] [Google Scholar]
- 17.Stock MS, Mota JA, DeFranco RN, et al. The time course of short-term hypertrophy in the absence of eccentric muscle damage. Eur J Appl Physiol 2017; 117: 989–1004. [DOI] [PubMed] [Google Scholar]
- 18.Abe T, Patterson KM, Stover CD, et al. Site-specific thigh muscle loss as an independent phenomenon for age-related muscle loss in middle-aged and older men and women. Age 2014; 36: 9634–9634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fukumoto Y, Ikezoe T, Yamada Y, et al. Age-related ultrasound changes in muscle quantity and quality in women. Ultrasound Med Biol 2015; 41: 3013–3017. [DOI] [PubMed] [Google Scholar]
- 20.Minetto MA, Caresio C, Menapace T, et al. Ultrasound-based detection of low muscle mass for diagnosis of sarcopenia in older adults. PM&R 2016; 8: 453–462. [DOI] [PubMed] [Google Scholar]
- 21.Scholten R, Pillen S, Verrips A, et al. Quantitative ultrasonography of skeletal muscles in children: normal values. Muscle Nerve 2003; 27: 693–698. [DOI] [PubMed] [Google Scholar]
- 22.Pillen S, Tak RO, Zwarts MJ, et al. Skeletal muscle ultrasound: correlation between fibrous tissue and echo intensity. Ultrasound Med Biol 2009; 35: 443–446. [DOI] [PubMed] [Google Scholar]
- 23.Reimers K, Reimers C, Wagner S, et al. Skeletal muscle sonography: a correlative study of echogenicity and morphology. J Ultrasound Med 1993; 12: 73–77. [DOI] [PubMed] [Google Scholar]
- 24.Zaidman CM, Wu JS, Kapur K, et al. Quantitative muscle ultrasound detects disease progression in Duchenne muscular dystrophy. Ann Neurol 2017; 81: 633–640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hill JC, Millan IS. Validation of musculoskeletal ultrasound to assess and quantify muscle glycogen content. A novel approach. Phys Sportsmed 2014; 42: 45–52. [DOI] [PubMed] [Google Scholar]
- 26.Nieman DC, Shanely RA, Zwetsloot KA, et al. Ultrasonic assessment of exercise-induced change in skeletal muscle glycogen content. BMC Sports Sci Med Rehabil 2015; 7: 9–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sarvazyan A, Tatarinov A, Sarvazyan N. Ultrasonic assessment of tissue hydration status. Ultrasonics 2005; 43: 661–671. [DOI] [PubMed] [Google Scholar]
- 28.Damas F, Phillips SM, Lixandrão ME, et al. Early resistance training-induced increases in muscle cross-sectional area are concomitant with edema-induced muscle swelling. Eur J Appl Physiol 2016; 116: 49–56. [DOI] [PubMed] [Google Scholar]
- 29.Jenkins ND, Miramonti AA, Hill EC, et al. Greater neural adaptations following high-vs. low-load resistance training. Front Physiol 2017; 8: 331–331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kristiansen MS, Uhrbrand A, Hansen M, et al. Concomitant changes in cross-sectional area and water content in skeletal muscle after resistance exercise. Scand J Med Sci Sports 2014; 24: e260–e268. [DOI] [PubMed] [Google Scholar]
- 31.Radaelli R, Bottaro M, Wilhelm EN, et al. Time course of strength and echo intensity recovery after resistance exercise in women. J Strength Cond Res 2012; 26: 2577–2584. [DOI] [PubMed] [Google Scholar]
- 32.Berg H, Tedner B, Tesch P. Changes in lower limb muscle cross-sectional area and tissue fluid volume after transition from standing to supine. Acta Physiol 1993; 148: 379–385. [DOI] [PubMed] [Google Scholar]
- 33.Rosenberg JG, Ryan ED, Sobolewski EJ, et al. Reliability of panoramic ultrasound imaging to simultaneously examine muscle size and quality of the medial gastrocnemius. Muscle Nerve 2014; 49: 736–740. [DOI] [PubMed] [Google Scholar]
- 34.Jenkins ND, Miller JM, Buckner SL, et al. Test–retest reliability of single transverse versus panoramic ultrasound imaging for muscle size and echo intensity of the biceps brachii. Ultrasound Med Biol 2015; 41: 1584–1591. [DOI] [PubMed] [Google Scholar]
- 35.Jenkins NDM, Miramonti AA, Hill EC, et al. Greater neural adaptations following high- vs. low-load resistance training. Front Physiol 2017; 8: 331–331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Weir JP. Quantifying test–retest reliability using the intraclass correlation coefficient and the SEM. J Strength Cond Res 2005; 19: 231–240. [DOI] [PubMed] [Google Scholar]
- 37.Shrout PE, Fleiss JL. Intraclass correlations: uses in assessing rater reliability. Psychol Bull 1979; 86: 420–428. [DOI] [PubMed] [Google Scholar]
- 38.Jenkins ND, Buckner SL, Cochrane KC, et al. Age-related differences in rates of torque development and rise in EMG are eliminated by normalization. Exp Gerontol 2014; 57: 18–28. [DOI] [PubMed] [Google Scholar]
- 39.Jenkins ND, Cramer JT. Reliability and minimum detectable change for common clinical physical function tests in sarcopenic men and women. J Am Geriatr Soc 2017; 65: 839–846. [DOI] [PubMed] [Google Scholar]
- 40.Weir JP. Quantifying test–retest reliability using the intraclass correlation coefficient and the SEM. J Strength Cond Res 2005; 19: 231–231. [DOI] [PubMed] [Google Scholar]
- 41.Hopkins WG. Measures of reliability in sports medicine and science. Sports Med 2000; 30: 1–15. [DOI] [PubMed] [Google Scholar]
- 42.Hammond K, Mampilly J, Laghi FA, et al. Validity and reliability of rectus femoris ultrasound measurements: comparison of curved-array and linear-array transducers. J Rehabil Res Dev 2014; 51: 1155–1164. [DOI] [PubMed] [Google Scholar]
- 43.Seymour JM, Ward K, Sidhu PS, et al. Ultrasound measurement of rectus femoris cross-sectional area and the relationship with quadriceps strength in COPD. Thorax 2009; 64: 418–423. [DOI] [PubMed] [Google Scholar]
- 44.Thomaes T, Thomis M, Onkelinx S, et al. Reliability and validity of the ultrasound technique to measure the rectus femoris muscle diameter in older CAD-patients. BMC Med Imaging 2012; 12: 7–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fukumoto Y, Ikezoe T, Yamada Y, et al. Skeletal muscle quality assessed from echo intensity is associated with muscle strength of middle-aged and elderly persons. Eur J Appl Physiol 2012; 112: 1519–1525. [DOI] [PubMed] [Google Scholar]
- 46.Jenkins ND, Housh TJ, Bergstrom HC, et al. Muscle activation during three sets to failure at 80 vs. 30% 1RM resistance exercise. Eur J Appl Physiol 2015; 115: 2335–2347. [DOI] [PubMed] [Google Scholar]
- 47.Fukumoto Y, Ikezoe T, Yamada Y, et al. Skeletal muscle quality assessed from echo intensity is associated with muscle strength of middle-aged and elderly persons. Eur J Appl Physiol 2012; 112: 1519–1525. [DOI] [PubMed] [Google Scholar]
- 48.Landin D, Thompson M, Reid M. Actions of two bi-articular muscles of the lower extremity: a review. J Clin Med Res 2016; 8: 489–489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Van De Graff K and Fox SI. Concepts of human anatomy and physiology. Boston, MA: McGraw-Hill; 1999.
- 50.Maffiuletti NA, Lepers R. Quadriceps femoris torque and EMG activity in seated versus supine position. Med Sci Sports Exerc 2003; 35: 1511–1516. [DOI] [PubMed] [Google Scholar]