Abstract
Purpose
We describe the impact of a multifaceted program for decreasing ventilator-associated pneumonia (VAP) after implementing nine preventive measures, including selective oropharyngeal decontamination (SOD).
Methods
We compared VAP rates during an 8-month pre-intervention period, a 12-month intervention period, and an 11-month post-intervention period in a cohort of patients who received mechanical ventilation (MV) for > 48 h. The primary objective was to assess the effect on first VAP occurrence, using a Cox cause-specific proportional hazards model. Secondary objectives included the impact on emergence of antimicrobial resistance, antibiotic consumption, duration of MV, and ICU mortality.
Results
Pre-intervention, intervention and post-intervention VAP rates were 24.0, 11.0 and 3.9 VAP episodes per 1000 ventilation-days, respectively. VAP rates decreased by 56% [hazard ratio (HR) 0.44, 95% CI 0.29–0.65; P < 0.001] in the intervention and by 85% (HR 0.15, 95% CI 0.08–0.27; P < 0.001) in the post-intervention periods. During the intervention period, VAP rates decreased by 42% (HR 0.58, 95% CI 0.38–0.87; P < 0.001) after implementation of eight preventive measures without SOD, and by 70% after adding SOD (HR 0.30, 95% CI 0.13–0.72; P < 0.001) compared to the pre-intervention period. The incidence density of intrinsically resistant bacteria (to colistin or tobramycin) did not increase. We documented a significant reduction of days of therapy per 1000 patient-days of broad-spectrum antibiotic used to treat lower respiratory tract infection (P < 0.028), median duration of MV (from 7.1 to 6.4 days; P < 0.003) and ICU mortality (from 16.2 to 13.5%; P < 0.049) for patients ventilated > 48 h between the pre- and post-intervention periods.
Conclusions
Our preventive program produced a sustained decrease in VAP incidence. SOD provides an additive value.
Electronic supplementary material
The online version of this article (10.1007/s00134-018-5227-4) contains supplementary material, which is available to authorized users.
Keywords: Ventilator-associated pneumonia, Multimodal strategy, Selective oropharyngeal decontamination, Intensive care unit
Take-home message
| A multifaceted program with 9 targeted preventive measures including SOD decreased VAP rates by 85% without emergence of multi-resistant organisms. A significant reduction in median duration of mechanical ventilation and mortality was also observed. |
Introduction
Ventilator-associated pneumonia (VAP) remains a serious complication of mechanical ventilation (MV), and has an incidence between 2 and 16 episodes per 1000 ventilator-days [1], an attributable mortality of 5–13% [2], excess ICU length of stay (LOS) for survivors up to 9 days [3], and extra costs [4].
Many studies have reported decreased VAP rates associated with the use of bundles of preventive measures [5]. Fewer studies have reported compliance with preventive measures [6–16] and explored sustainability of interventions after implementation [7, 10–13, 16–19]. Except for one [19], studies have not reported results of a bundle including selective oropharyngeal decontamination (SOD).
SOD consists in the application of topical antibiotics to the oropharynx, without enteral or intravenous antibiotics. There is a significant reduction of VAP in studies which investigated decontamination with antibiotic agents [risk ratio (RR): 0.27; 95% confidence intervals (CI) 0.18–0.42] but no effect on all-cause mortality, duration of ventilation, or duration of ICU stay [20]. A recent meta-analysis including four randomized controlled trials (RCTs) demonstrated a beneficial effect of SOD on mortality [21]. Promoting bacterial resistance with SOD remains a serious concern, although a recent meta-analysis did not detect an increased incidence of antimicrobial resistant pathogens in recipients of selective decontamination compared with non-recipients [22].
The primary objective of this prospective, interventional single-center cohort study was to determine the impact of implementing a multifaceted VAP prevention program on VAP rates. Secondary objectives were to determine the impact of SOD on VAP rates, to evaluate emergence of antimicrobial resistance, and to determine the impact of the program on antibiotic consumption, duration of MV, LOS and mortality.
Methods
Setting
Geneva University Hospital is a 1900-bed hospital serving as a primary and tertiary care facility in Geneva, Switzerland. The mixed medical–surgical adult ICU has 34 beds, employs 250 nursing and medical staff members, and admits 2400 patients per year on average. The nurse–patient and nursing assistant–patient ratios are 1:2 and 1:4, respectively, with little difference across shifts or over time. Neither the structure nor the staffing of the ICU was modified during the study. As a quality improvement project, ethics committee approval was not required.
Design
This study used a quasi-experimental, uncontrolled, before-and-after design to analyze the effects of implementing a VAP prevention bundle. For the present study, the pre-intervention period comprised 8 months from January through August 2014, the intervention period 12 months from September 2014 through August 2015, and the post-intervention period (follow-up) 11 months from September 2015 through July 2016.
VAP surveillance
Surveillance of VAP was performed in our ICU since January 2014, and included all patients with MV > 48 h (endotracheal tube or tracheostomy), including the following variables for each included patient: age, sex, severity of illness as assessed using the Simplified Acute Physiology Score II [23], main reason for ICU admission, dates of ventilation, presence of VAP, date of VAP onset, Clinical Pulmonary Infection Score (CPIS) [24], diagnostic method for VAP [bronchoalveolar lavage (BAL) or mini-bronchoalveolar lavage (mini-BAL)], microorganisms identified, dates of ICU admission and discharge, and mortality.
Probable VAP was defined as the presence of (1) radiologic changes with systemic inflammation (temperature ≥ 38 °C, or leukocyte count > 12,000 or < 4000 cells/mL) with clinical pulmonary signs (i.e. purulent tracheal secretions) according to the criteria established by Hospitals in Europe Linked for Infection Control through Surveillance [25], and (2) a CPIS > 6 [24]. Definite VAP was defined by the addition of positive quantitative cultures of distal pulmonary sampling obtained by BAL (significant threshold ≥ 104 colony-forming units/mL) or mini-BAL (significant threshold ≥ 103 colony-forming units/mL). VAP was defined as early-onset when it developed in the first 4 days of MV, and as late-onset when it occurred after 4 days of MV.
Screening, data collection and reporting were conducted by the same ICU staff throughout the surveillance period and diagnosis of VAP was made independently by two investigators following a consensus meeting.
Multifaceted prevention program
A multidisciplinary task force, consisting of intensivists, ICU nurses, pharmacists, infection control and infectious diseases physicians, was created in May 2014 to develop a policy for VAP prevention. Knowledge of health-care workers (HCWs) on VAP prevention was assessed by a questionnaire in August 2014 and again in December 2015 (Supplementary Appendix). A targeted educational program of 1 h was designed for nurses, nursing assistants and physiotherapists and conducted between September 2014 and March 2015. Our multifaceted prevention program focused on nine preventive measures based on ATS/IDSA recommendations [26], meta-analyses of specific measures and monocentric studies with VAP bundle implementation. Three groups of measures were implemented throughout the intervention period: (1) good basic practices in September 2014: compliance with hand hygiene, keeping patients in a semirecumbent position (> 30°), oral hygiene three times daily with 0.2% chlorhexidine-gluconate aqueous solution, maintaining an endotracheal tube cuff pressure ≥ 25 cm H2O, daily sedation hold, daily trial of ventilation weaning, daily active mobilization; (2) new device in December 2014: continuous aspiration of subglottic secretions; (3) new practice in March 2015: SOD with colistin, tobramycin and nystatin three times per day for patients likely to be ventilated ≥ 48 h. The same measures were applied in the post-intervention period.
Adherence with the different bundle components was measured throughout the study period, and less frequently in the post-intervention period; relevant process indicators are reported in the Supplementary Appendix. Global compliance with the eight indicators (except hand hygiene compliance, measured as an aggregate data indicator) was calculated (1 point was attributed for each indicator correctly applied; sum of points was divided by the number of assessable indicators) and expressed as a percentage for each patient. A daily e-surveillance form (http://www.reallience.ch/) was used to record data. A team of five ICU and infection control personnel was trained for these evaluations. Performance feedback consisted of regular meetings, and visible displaying for HCWs of graphs documenting compliance and VAP rates.
Antimicrobial resistance
Antimicrobial resistance was evaluated throughout the project period, albeit via different methods. From January 2014 to April 2015, monthly point-prevalence studies of intestinal carriage of Gram-negative resistant bacteria were performed on all ICU patients. As of May 2015, we performed targeted screening of patients who received SOD, once the SOD was discontinued. An additional ecological analysis of all clinical or screening bacterial isolates from the ICU was performed by calculating incidence densities (per 1000 patient-days) for bacteria intrinsically resistant to either colistin or tobramycin. Detailed methods and microbiological techniques are reported in the Supplementary Appendix.
Antibiotic consumption
Total antibiotic consumption, and consumption of broad-spectrum antibiotics that are used for the treatment of lower respiratory tract infection (LRTI), were analyzed for each period (Supplementary Appendix).
Other measures
Other measures in our ICU are reported in the Supplementary Appendix.
Statistical analyses
Baseline characteristics were described by frequencies, medians and interquartile range (IQR). Groups were compared by means of the Mann–Whitney U test, the Kruskal–Wallis test, and Independent Samples Median Test, Pearson Chi-squared test or Fisher’s exact test as appropriate for continuous and categorical variables.
All VAP episodes (definite and probable) were included. We computed the number of first VAP episodes per 1000 ventilator-days at risk (until VAP diagnosis or extubation). We also computed (1) the total number of VAP episodes per 1000 ventilator-days, including all ventilation sessions, (2) the proportion of patients with VAP among patients receiving ventilation for > 48 h, and (3) the time to the first VAP episode. Only patients who received MV for > 48 h were included in the analysis.
We analyzed the effect of the intervention (with and without SOD) on the incidence of first VAP episodes by segmented regression analysis using a Poisson model [27]. In this analysis, incidence rate ratios (IRR) were computed per months before, during, and after the intervention. The model included an intercept (mean VAP rate at study initiation) and three main period effects (before–during intervention without SOD, during intervention without SOD–during intervention with SOD and during intervention with SOD–after intervention).
The risk of first VAP over time was also analyzed using a competing-risks survival approach comparing the time to first VAP, with death and ICU discharge being competing events [28]. The association between periods and VAP risk was tested using a Cox proportional cause-specific hazards model. The cumulative cause-specific hazard of VAP was estimated for each study period. We determined the empirical daily hazard rate for VAP and the hazard function from the Cox model to estimate the event rate per day. The results were presented as cause-specific hazard ratios (HR) with 95%CI.
All statistical tests were two-tailed and a P value ≤ 0.05 was considered statistically significant. Statistical analysis was performed with PASW v.23 (SPSS, Chicago, IL, USA), and STATA v.13.1 (Stata, College Station, TX, USA).
Results
Of the 6287 admissions during the study period, 2991 were followed by ICU stays > 48 h. These 2991 admissions comprised 1262 sessions of MV for > 48 h, for a total of 10,825 ventilation-days: 318 sessions of MV (3128 ventilation-days) during the pre-intervention, 507 sessions (4234 ventilation-days) during the intervention, and 437 sessions (3463 ventilation-days) during the post-intervention period.
Table 1 lists the characteristics of included patients during each period. Patients did not differ significantly with respect to age, sex, SAPS II, and main reason for ICU admission.
Table 1.
Characteristics of patients included in the study
| Variable | Pre-intervention period | Intervention period | Post-intervention period |
|---|---|---|---|
| No. of patients | 291 | 413 | 356 |
| Age, years, median (IQR) | 61.9 (48.6–73.4) | 63.8 (48.2–74.7) | 60.5 (49.4–71.2) |
| Male sex | 207 (71.1%) | 289 (70.0%) | 251 (70.5%) |
| Main reason for ICU admission: | |||
| Cardiovascular | 79 (27.1%) | 109 (26.4%) | 72 (20.2%) |
| Neurologic | 57 (19.6%) | 70 (16.9%) | 61 (17.1%) |
| Respiratory | 67 (23.0%) | 94 (22.8%) | 97 (27.2%) |
| Sepsis | 17 (5.8%) | 34 (8.2%) | 30 (8.4%) |
| Traumatology | 28 (9.6%) | 52 (12.6%) | 31 (8.7%) |
| Gastrointestinal | 31 (10.6%) | 32 (7.7%) | 43 (12.1%) |
| Metabolic | 5 (1.7%) | 9 (2.2%) | 7 (2.0%) |
| Others | 7 (2.4%) | 12 (2.9%) | 14 (3.9%) |
| Simplified acute physiology score II, median (IQR) | 53 (42–65) | 54 (41–64) | 55 (44–67) |
| Length of ICU stay, median days (IQR) | 8.7 (5.4–13.8) | 7.7 (4.7–13.12) | 8.9 (4.8–13.8) |
| 28-day mortality | 68 (23.4%) | 74 (17.9%) | 73 (20.5%) |
| ICU mortality | 47 (16.2%) | 43 (10.4%)a | 48 (13.5%)b,c |
Data are no. (%) of patients, unless otherwise indicated
IQR interquartile range
aSignificant P value between the pre-intervention and intervention periods
bSignificant P value between the pre-intervention and post-intervention periods
cSignificant P value between the three periods
Compliance with preventive measures
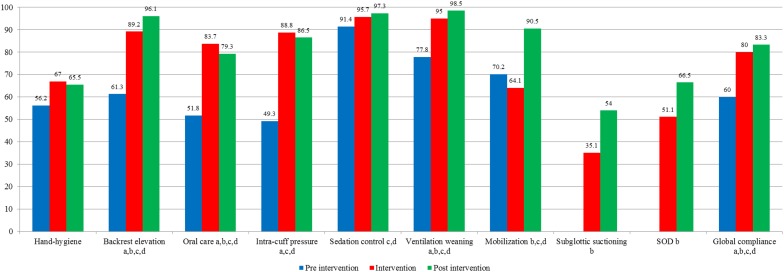
A total of 73, 1224 and 695 evaluations of hand hygiene and 223, 2042 and 632 evaluations of other preventive measures were performed for the pre-intervention, intervention and post-intervention periods, respectively (Fig. 1; e-Table 1). As shown in Fig. 1, hand-hygiene compliance was initially 56.2%, increased in the intervention period (67%) and remained stable over time (65.5%). Global compliance with all other preventive measures was initially high (60%), and increased significantly in the intervention (80%) and post-intervention (83.3%) periods. Compliance with each preventive measure increased significantly between each period and between the pre-intervention and each of the two subsequent periods, except for oral hygiene that significantly decreased in the post-intervention period compared to the intervention period (83.7 to 79.3%).
Fig. 1.
Compliance over the three periods for the nine ventilator-associated pneumonia (VAP) preventive measures, and for global compliance. Compliance was defined for each assessable preventive measures (represented as a percentage) and global compliance is expressed as median (IQRs) for all ventilator-days assessable; SOD selective oropharyngeal decontamination. a Significant P value between the pre-intervention and intervention periods, b significant P value between the intervention and post-intervention periods, c significant P value between the pre-intervention and post-intervention periods, d significant P value between the three periods
Total VAP rates
The incidence density rates were 21.4, 10.6, and 3.8 during the pre-intervention, intervention, and post-intervention periods, respectively, with significant differences between the three periods (Table 2).
Table 2.
Characteristics and outcomes of patients who received mechanical ventilation for > 48 h
| Variable | Pre-intervention period | Intervention period | Post-intervention period |
|---|---|---|---|
| Sessions of mechanical ventilation | 318 | 507 | 437 |
| Total ventilator-days | 3128 | 4234 | 3463 |
| Ventilator-days at risk | 2667 | 3920 | 3381 |
| Total VAP episodes | 67 | 45 | 13 |
| First VAP episodes | 64 (20.1%) | 43 (8.5%)a | 13(3%)b,c,d |
| Early onset | 25 (39.1%) | 22 (51.2%) | 4 (30.8%) |
| Late onset | 39 (60.9%) | 21 (48.8%) | 9 (69.2%) |
| Diagnosed by bronchoalveolar lavage | 10 (15.6%) | 6 (14%) | 4 (30.8%) |
| Diagnosed by mini-bronchoalveolar lavage | 34 (53.1%) | 34 (79.1%)a | 7 (53.8%)d |
| Probable | 28 (43.8%) | 5 (11.6%)a | 2 (15.4%)d |
| Definite | 36 (56.2%) | 38 (88.4%) | 11 (84.6%) |
| Total VAP incidence density rates per 1000 ventilator-days at risk (95% CI) | 21.4 (16.3–26.6) | 10.6 (7.5–13.7)a | 3.8 (1.7–5.8)b,c,d |
| First VAP incidence density rates per 1000 ventilator-days at risk (95% CI) | 24.0 (18.1–29.9) | 11.0 (7.7–14.2)a | 3.9 (1.8–5.9)b,c,d |
| First probable VAP incidence density rates per 1000 ventilator-days at risk (95% CI) | 10.5 (6.6–14.4) | 1.3 (0.2–2.4) | 0.6 (− 0.2 to 1.4) |
| First definite VAP incidence density rates per 1000 ventilator-days at risk (95% CI) | 13.5 (9.1–17.9) | 9.7 (6.6–12.8) | 3.3 (1.3–5.2) |
| Clinical Pulmonary Infection Score for VAP episodes, median (IQR) | 8 (7–9) | 8 (7–9) | 8 (7–8) |
| Duration of mechanical ventilation for all mechanical ventilation sessions, median days (IQR) | 7.14 (4.61–11.06) | 6.50 (4.17–9.85)a | 6.35 (4.15–9.74)c, d |
Data are no. (%) of patients, unless otherwise indicated
CI confidence interval, IQR interquartile range, VAP ventilator-associated pneumonia
aSignificant P value between the pre-intervention and intervention periods
bSignificant P value between the intervention and post-intervention periods
cSignificant P value between the pre-intervention and post-intervention periods
dSignificant P value between the three periods
First VAP rate
The proportions of patients with at least one VAP episode among patients who received ventilation for > 48 h were 20.1, 8.5, and 3% during the pre-intervention, intervention, and post-intervention periods, respectively (P < 0.001) (Table 2). The incidence density rates of first VAP episodes per 1000 ventilation-days were 24.0 during the pre-intervention period, 11.0 during the intervention period and 3.9 during the post-intervention period. VAP rates decreased by 54% in the intervention (relative risk, 0.46, 95% CI 0.31–0.67; P < 0.05) and by 84% in the post-intervention periods (relative risk, 0.16, 95% CI 0.1–0.27; P < 0.05) compared to the pre-intervention period. The median time to first VAP was 5.3 days (IQR 3.7–7.5) during the pre-intervention period 4.8 days (IQR 3.3–8.1) during the intervention period and 6.3 days (IQR 4.3–10.2) during the post-intervention period without significant differences.
The distribution of microorganisms associated with the first VAP episodes is reported in e-Table 2. There was a trend towards a decrease of Gram-negative bacilli and a relative increase of Gram-positive cocci over the study period.
Before the fifth ventilator-day, 25 (7.9% of patients receiving ventilation) first VAP episodes were identified during the pre-intervention period, 22 (4.3%) during the intervention period and 4 (0.9%) during the post-intervention period (Table 2). After the fifth ventilator-day, 39 (12.3% of patients still receiving ventilation on day 4), 21 (4.1%) and 9 (2.1%) first VAP episodes were identified during the pre-intervention, intervention and post-intervention periods, respectively. Probable VAP decreased significantly between the pre-intervention and intervention periods.
Additive effect of SOD
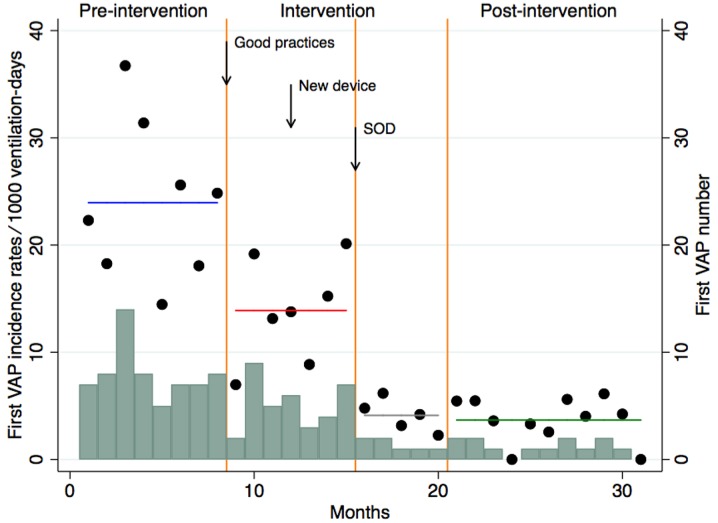
The results of the segmented regression analysis are shown in Fig. 2. Before the intervention, VAP incidence density was high and remained stable over time. The intervention without SOD was associated with a decrease of 42% in VAP rates (IRR 0.58, 95% CI 0.45–0.74). The implementation of SOD was associated with a further decrease of 70% in VAP rates (IRR 0.30, 95% CI 0.18–0.48). After the intervention, VAP incidence density was low and remained stable over time.
Fig. 2.
Segmented Poisson regression analysis, comparing incidence rates of ventilator-associated pneumonia (VAP) in the pre-intervention, the intervention period without SOD, implementation of SOD and post-intervention period according to months. SOD Selective oropharyngeal decontamination. Circles indicate first VAP incidence rates per 1000 ventilation-days according to months, horizontal lines estimated first VAP incidence rates per 1000 ventilation-days by period, blue line pre-intervention period, red line intervention period without SOD, gray line implementation of SOD period, and green line post-intervention period, vertical orange lines delimitations of time periods, bars VAP number according to months
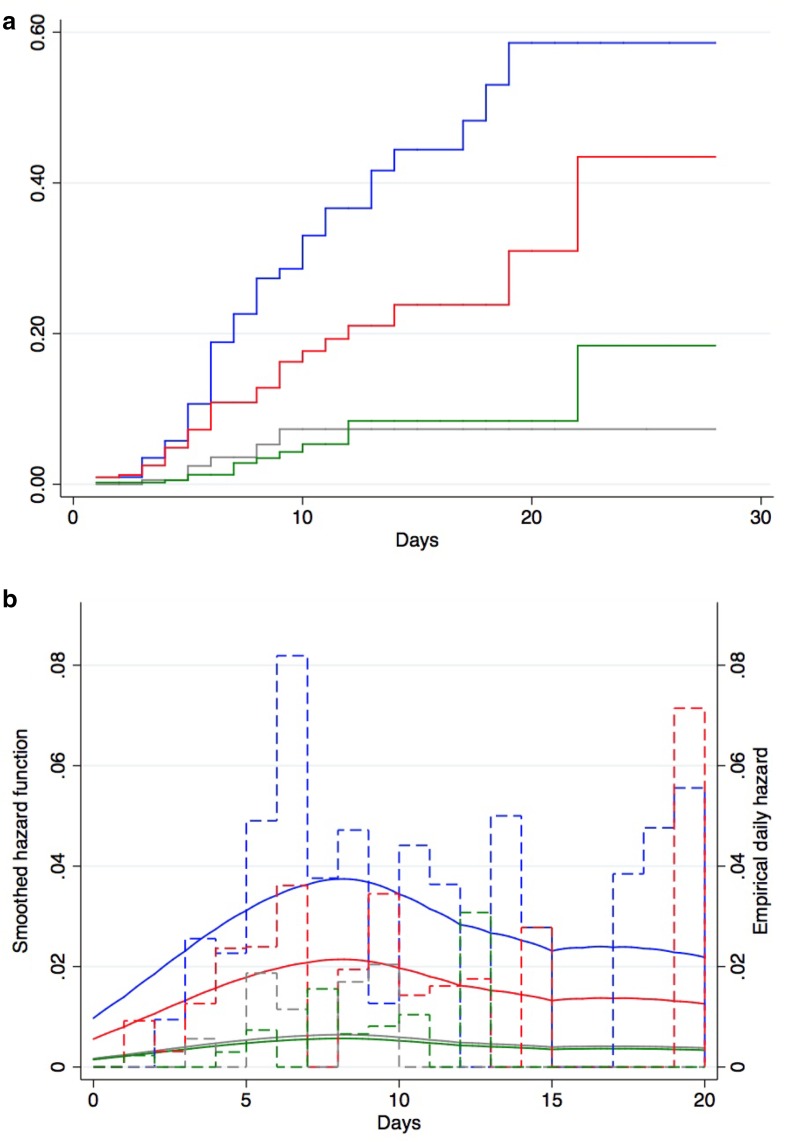
The results of the cumulative hazard of first VAP according to periods and daily hazard of first VAP are shown in Fig. 3a, b. The intervention decreased the VAP incidence rate by 56% (HR 0.44, 95% CI 0.29–0.65; P < 0.001). The intervention without SOD was associated with a significant decrease of 42% (HR 0.58, 95% CI 0.38–0.87; P < 0.001) in VAP incidence rates in comparison with baseline. SOD was associated with a significant decrease of 70% (HR 0.30, 95% CI 0.13–0.72; P < 0.001) in VAP incidence rates over time in comparison with intervention without SOD. Post-intervention was associated with a non-significant decrease of 12% (HR 0.88, 95% CI 0.33–2.32; P > 0.05) in VAP incidence rates over time in comparison with intervention with SOD. Globally, post-intervention was associated with a significant decrease of 85% (HR 0.15, 95% CI 0.08–0.28; P < 0.001) in VAP incidence rates over time in comparison with pre-intervention.
Fig. 3.
Cumulative cause-specific hazard (a) and daily hazard rates (b) of ventilator-associated pneumonia in the pre-intervention period, the intervention period without SOD, implementation of SOD and the post-intervention period using Cox proportional cause-specific hazard regression. a, b Blue lines represent the pre-intervention period, red lines the intervention period without SOD, gray lines the period after implementation of SOD, green lines the post-intervention period. b Dashed lines empirical daily hazard of VAP, solid lines smoothed hazard function
During the pre-intervention period, daily VAP hazard rates were 4.9, and 4.4% during the fifth and tenth days of MV, respectively. During the intervention without SOD, corresponding rates were 2.4 and 1.4%, respectively. During the intervention period with SOD, corresponding rates were 1.9 and 0%, respectively. During the post-intervention period, corresponding rates were 0.7 and 1.04%, respectively. The differences were statistically significant between periods only during the fifth days of MV (P = 0.03).
There was no increase in antibiotic resistance, and colistin MIC did not increase after the introduction of SOD (Supplementary Appendix).
Antibiotic consumption
Total consumption of antibiotics did not change significantly between periods. However, we observed a significant decrease in the consumption of antibiotics used for treating LRTI in the post-intervention period compared to the pre-intervention period (Supplementary Appendix and e-Fig. 4).
Other outcome measures
A significant decrease was observed between the pre-intervention and intervention periods and between the pre-intevention and post-intervention periods for the median duration of MV among included study subjects, 7.14, 6.5 and 6.35 days, respectively (Table 2), and for ICU mortality, 16.2, 10.4 and 13.5%, respectively (Table 1). The ICU LOS did not significantly differ between periods (Table 1). Knowledge of HCWs are described in the Supplementary Appendix.
Discussion
Our data suggest that the introduction of a multifaceted prevention program on VAP was associated with an important and sustained reduction in the incidence of VAP. SOD provides an additive effect.
Our initial VAP rate was higher than is usually reported in multicenter surveillance networks but similar to other settings that used comparable VAP definitions and surveillance methods, and included only patients who received MV for at least 48 h [8, 17]. Our multifaceted prevention program resulted in an 85% decrease in VAP rates. This large reduction effect, rarely described in the literature, was confirmed by using two complementary statistical approaches and was concomitant with a significant improvement in compliance with preventive measures. The risk of VAP was highest in the first few days of intubation, with a daily hazard rate of approximately 5% at day 5 of MV in the pre-intervention period and decreased with increasing duration of MV in accordance with a previous study [29]. Intervention and post-intervention periods resulted in a significant decrease of daily VAP hazard rates until the fifth day of MV. Although the difference was not statistically significant, the decrease in daily VAP hazard rates also occurred after the fifth day of MV, suggesting that the preventive measures were effective both for early and late onset VAP. In comparison with other studies, median duration of MV was shorter [7], and ICU mortality was also lower [8] in the pre-intervention period. Nevertheless, we found a significant reduction of the median duration of MV and the ICU mortality after implementation of our multifaceted program, and a significant decrease of antibiotic consumption used for LRTI treatment in the post-intervention period in comparison to the pre-intervention period. No significant difference was found for the median ICU LOS or total antibiotic consumption. Treatment of clinical sepsis and/or the “dilution effect” (antibiotics used for the treatment of suspected or proven LRTI represented overall 73% of the total antibiotic consumption) could explain these results.
We chose nine preventive measures for our bundle: hand hygiene, head-of-bed elevation, oral care with chlorhexidine, endotracheal tube cuff pressure ≥ 25 cm H2O, sedation holds, ventilation weaning, active mobilization, subglottic secretion suctioning and SOD. All have demonstrated their efficacy in reducing VAP rates, duration of MV or mortality [30, 31]. Despite a high initial global compliance with all the preventive measures (60%), global compliance increased in the post-intervention period (83.3%).
We attributed part of the success of the intervention to SOD; the intervention without SOD was associated with a significant decrease of 42% in VAP incidence rates and SOD added a further 28% in reduced VAP rates. We cannot exclude that SOD may have decreased microbiological yield in tracheal aspirates, artificially decreasing the number of probable VAP. We also cannot exclude that SOD truly “sterilized” the oropharyngeal fluid, preventing the transition from airway colonization to lung infection. A decreased rate of the incidence density of first definite VAP episodes was also observed across periods, and may favor this latter hypothesis. Accurately predicting the probability of ventilation for more than 2 days (criteria for SOD prescription in our study) was challenging in clinical practice, and this uncertainty may have lowered the number of SOD-eligible patients. Nevertheless, intensivists improved their predictive capacity during the study. Consequently, SOD compliance increased over time but the low level of compliance (51% in the intervention and 66% in the post-intervention period) could have minimized the additive value of SOD on VAP rates. Conversely, we observed a decrease in the duration of MV after the introduction of the bundle. Such a reduction could possibly be due to the decrease in VAP rates but also to better adherence to the sedation and weaning components of the multifaceted prevention program. This reduction in the duration of MV may also partially explain the decrease in the VAP rates.
Current evidence suggests that antibiotic resistance may decrease with the use of selective decontamination, although much of these data come from the Netherlands, an area with low endemic antibiotic resistance [22]. The present study was performed in a setting with a higher baseline prevalence of antimicrobial resistance [32, 33]. We did not observe an increase in antimicrobial selection pressure. No definite conclusion can be drawn about the selective pressure of SOD, as the methods of detection of emerging antimicrobial resistance were different throughout the project, and as rectal swabs were performed only once at the end of SOD administration, and not repeated over time, for example at ICU discharge. Of note, the relative proportion of Gram-positive cocci causing VAP increased in the post-intervention period, due to a substantial decrease in the proportion of Gram-negative bacilli.
Our study has several limitations. First, we did not use a randomized study design or a concurrent control group. Therefore, the exact impact of our prevention program on VAP rates may be difficult to assess. Second, our study was not blinded, and it is well known that VAP definitions carry some subjectivity. However, the methodology of VAP diagnosis did not change over time. Third, compliance with preventive measures was not evaluated for each patient and each day. Determining which individual components of our multifaceted intervention, except SOD, led to its success was not possible. Because of a possible Hawthorne effect, we cannot affirm that compliance was similar during the non-observation periods. However, the performance assessments were conducted with the same methodology, the magnitude of improvement was sufficiently high to ascertain real changes, and our program produced sustained VAP rate decreases in the long term (e-Fig. 5). Fourthly, our study was performed in a single center. Whether the implementation of our program or data collection procedures can be generalized to other ICUs remains to be determined. Finally, a possible effect of regression to the mean, due to initially high VAP rates, cannot be excluded, but is unlikely to be responsible for the totality of the decrease.
In conclusion, we have demonstrated that simple preventive measures including SOD, surveillance and feedback, and education can significantly decrease VAP rates, duration of MV, and mortality. However, long-term use of SOD implies continuous surveillance of the emergence and selection of antimicrobial resistance.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
We thank the ICU team who participated in this quality improvement project. We are also indebted to all members of the bacteriology laboratory and infection control unit who helped with this project. This study was partially supported by an unrestricted, internal grant from the Department of Anesthesiology, Intensive Care, and Pharmacology, University Hospitals of Geneva, Switzerland.
Authors’ contributions
Design and conduct of the study: C. Landelle, V. Nocquet Boyer, M. Abbas, S. Harbarth and J. Pugin. Collection of the data: C. Landelle, V. Nocquet Boyer, M. Abbas, E. Genevois, N. Abidi, S. Naimo, R. Raulais, and F. Boroli. Management, analysis and interpretation of the data: C. Landelle, V. Nocquet Boyer, M. Abbas, L. Bouchoud; H. Terrisse, J.-L. Bosson, S. Harbarth, and J. Pugin. Manuscript preparation: C. Landelle, V. Nocquet Boyer, M. Abbas, H. Terrisse, S. Harbarth, and J. Pugin. Critical revision and approval of the manuscript: All authors.
Conflicts of interest
SH reports having received a peer-reviewed research grant funded by Pfizer; he is also a temporary member of the advisory boards of DNA Electronics, Bayer and Novartis. All other authors: none to declare.
Research involving human participants
As a quality improvement project, ethics committee approval was not required.
References
- 1.Barbier F, Andremont A, Wolff M, Bouadma L. Hospital-acquired pneumonia and ventilator-associated pneumonia: recent advances in epidemiology and management. Curr Opin Pulm Med. 2013;19:216–228. doi: 10.1097/MCP.0b013e32835f27be. [DOI] [PubMed] [Google Scholar]
- 2.Melsen WG, Rovers MM, Groenwold RH, Bergmans DC, Camus C, Bauer TT, et al. Attributable mortality of ventilator-associated pneumonia: a meta-analysis of individual patient data from randomised prevention studies. Lancet Infect Dis. 2013;13:665–671. doi: 10.1016/S1473-3099(13)70081-1. [DOI] [PubMed] [Google Scholar]
- 3.Kollef MH, Hamilton CW, Ernst FR. Economic impact of ventilator-associated pneumonia in a large matched cohort. Infect Control Hosp Epidemiol. 2012;33:250–256. doi: 10.1086/664049. [DOI] [PubMed] [Google Scholar]
- 4.Hugonnet S, Eggimann P, Borst F, Maricot P, Chevrolet JC, Pittet D. Impact of ventilator-associated pneumonia on resource utilization and patient outcome. Infect Control Hosp Epidemiol. 2004;25:1090–1096. doi: 10.1086/502349. [DOI] [PubMed] [Google Scholar]
- 5.Klompas M. Ventilator-associated pneumonia: is zero possible? Clin Infect Dis. 2010;51:1123–1126. doi: 10.1086/656738. [DOI] [PubMed] [Google Scholar]
- 6.Bouadma L, Mourvillier B, Deiler V, Le Corre B, Lolom I, Regnier B, et al. A multifaceted program to prevent ventilator-associated pneumonia: impact on compliance with preventive measures. Crit Care Med. 2010;38:789–796. doi: 10.1097/CCM.0b013e3181ce21af. [DOI] [PubMed] [Google Scholar]
- 7.Rello J, Afonso E, Lisboa T, Ricart M, Balsera B, Rovira A, et al. A care bundle approach for prevention of ventilator-associated pneumonia. Clin Microbiol Infect. 2013;19:363–369. doi: 10.1111/j.1469-0691.2012.03808.x. [DOI] [PubMed] [Google Scholar]
- 8.Morris AC, Hay AW, Swann DG, Everingham K, McCulloch C, McNulty J, et al. Reducing ventilator-associated pneumonia in intensive care: impact of implementing a care bundle. Crit Care Med. 2011;39:2218–2224. doi: 10.1097/CCM.0b013e3182227d52. [DOI] [PubMed] [Google Scholar]
- 9.Sedwick MB, Lance-Smith M, Reeder SJ, Nardi J. Using evidence-based practice to prevent ventilator-associated pneumonia. Crit Care Nurse. 2012;32:41–51. doi: 10.4037/ccn2012964. [DOI] [PubMed] [Google Scholar]
- 10.Khan R, Al-Dorzi HM, Al-Attas K, Ahmed FW, Marini AM, Mundekkadan S, et al. The impact of implementing multifaceted interventions on the prevention of ventilator-associated pneumonia. Am J Infect Control. 2016;44:320–326. doi: 10.1016/j.ajic.2015.09.025. [DOI] [PubMed] [Google Scholar]
- 11.Rawat N, Yang T, Ali KJ, Catanzaro M, Cohen MD, Farley DO, et al. Two-state collaborative study of a multifaceted intervention to decrease ventilator-associated events. Crit Care Med. 2017;45:1208–1215. doi: 10.1097/CCM.0000000000002463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Berenholtz SM, Pham JC, Thompson DA, Needham DM, Lubomski LH, Hyzy RC, et al. Collaborative cohort study of an intervention to reduce ventilator-associated pneumonia in the intensive care unit. Infect Control Hosp Epidemiol. 2011;32:305–314. doi: 10.1086/658938. [DOI] [PubMed] [Google Scholar]
- 13.Caserta RA, Marra AR, Durao MS, Silva CV, Pavao dos Santos OF, Neves HS, et al. A program for sustained improvement in preventing ventilator associated pneumonia in an intensive care setting. BMC Infect Dis. 2012;12:234. doi: 10.1186/1471-2334-12-234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Marini AL, Khan R, Mundekkadan S. Multifaceted bundle interventions shown effective in reducing VAP rates in our multidisciplinary ICUs. BMJ Qual Improv Rep. 2016 doi: 10.1136/bmjquality.u205566.w2278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Okgun Alcan A, Demir Korkmaz F, Uyar M. Prevention of ventilator-associated pneumonia: use of the care bundle approach. Am J Infect Control. 2016;44:e173–e176. doi: 10.1016/j.ajic.2016.04.237. [DOI] [PubMed] [Google Scholar]
- 16.Talbot TR, Carr D, Parmley CL, Martin BJ, Gray B, Ambrose A, et al. Sustained reduction of ventilator-associated pneumonia rates using real-time course correction with a ventilator bundle compliance dashboard. Infect Control Hosp Epidemiol. 2015;36:1261–1267. doi: 10.1017/ice.2015.180. [DOI] [PubMed] [Google Scholar]
- 17.Bouadma L, Deslandes E, Lolom I, Le Corre B, Mourvillier B, Regnier B, et al. Long-term impact of a multifaceted prevention program on ventilator-associated pneumonia in a medical intensive care unit. Clin Infect Dis. 2010;51:1115–1122. doi: 10.1086/656737. [DOI] [PubMed] [Google Scholar]
- 18.Lim KP, Kuo SW, Ko WJ, Sheng WH, Chang YY, Hong MC, et al. Efficacy of ventilator-associated pneumonia care bundle for prevention of ventilator-associated pneumonia in the surgical intensive care units of a medical center. J Microbiol Immunol Infect. 2015;48:316–321. doi: 10.1016/j.jmii.2013.09.007. [DOI] [PubMed] [Google Scholar]
- 19.Alvarez-Lerma F, Palomar-Martinez M, Sanchez-Garcia M, Martinez-Alonso M, Alvarez-Rodriguez J, Lorente L, et al. Prevention of ventilator-associated pneumonia: the multimodal approach of the spanish ICU “Pneumonia Zero” program. Crit Care Med. 2018;46:181–188. doi: 10.1097/CCM.0000000000002736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li J, Xie D, Li A, Yue J. Oral topical decontamination for preventing ventilator-associated pneumonia: a systematic review and meta-analysis of randomized controlled trials. J Hosp Infect. 2013;84:283–293. doi: 10.1016/j.jhin.2013.04.012. [DOI] [PubMed] [Google Scholar]
- 21.Price R, MacLennan G, Glen J, Su DC. Selective digestive or oropharyngeal decontamination and topical oropharyngeal chlorhexidine for prevention of death in general intensive care: systematic review and network meta-analysis. BMJ. 2014;348:g2197. doi: 10.1136/bmj.g2197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Daneman N, Sarwar S, Fowler RA, Cuthbertson BH, Su DCSG. Effect of selective decontamination on antimicrobial resistance in intensive care units: a systematic review and meta-analysis. Lancet Infect Dis. 2013;13:328–341. doi: 10.1016/S1473-3099(12)70322-5. [DOI] [PubMed] [Google Scholar]
- 23.Le Gall JR, Lemeshow S, Saulnier F. A new simplified acute physiology score (SAPS II) based on a European/North American multicenter study. JAMA. 1993;270:2957–2963. doi: 10.1001/jama.1993.03510240069035. [DOI] [PubMed] [Google Scholar]
- 24.Singh N, Rogers P, Atwood CW, Wagener MM, Yu VL. Short-course empiric antibiotic therapy for patients with pulmonary infiltrates in the intensive care unit. A proposed solution for indiscriminate antibiotic prescription. Am J Respir Crit Care Med. 2000;162:505–511. doi: 10.1164/ajrccm.162.2.9909095. [DOI] [PubMed] [Google Scholar]
- 25.Suetens C, Morales I, Savey A, Palomar M, Hiesmayr M, Lepape A, et al. European surveillance of ICU-acquired infections (HELICS-ICU): methods and main results. J Hosp Infect. 2007;65:171–173. doi: 10.1016/S0195-6701(07)60038-3. [DOI] [PubMed] [Google Scholar]
- 26.American Thoracic Society, Infectious Diseases Society of America Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171:388–416. doi: 10.1164/rccm.200405-644ST. [DOI] [PubMed] [Google Scholar]
- 27.Shardell M, Harris AD, El-Kamary SS, Furuno JP, Miller RR, Perencevich EN. Statistical analysis and application of quasi experiments to antimicrobial resistance intervention studies. Clin Infect Dis. 2007;45:901–907. doi: 10.1086/521255. [DOI] [PubMed] [Google Scholar]
- 28.Wolkewitz M, Cooper BS, Palomar-Martinez M, Alvarez-Lerma F, Olaechea-Astigarraga P, Barnett AG, et al. Multilevel competing risk models to evaluate the risk of nosocomial infection. Crit Care. 2014;18:R64. doi: 10.1186/cc13821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cook DJ, Walter SD, Cook RJ, Griffith LE, Guyatt GH, Leasa D, et al. Incidence of and risk factors for ventilator-associated pneumonia in critically ill patients. Ann Intern Med. 1998;129:433–440. doi: 10.7326/0003-4819-129-6-199809150-00002. [DOI] [PubMed] [Google Scholar]
- 30.Bouadma L, Wolff M, Lucet JC. Ventilator-associated pneumonia and its prevention. Curr Opin Infect Dis. 2012;25:395–404. doi: 10.1097/QCO.0b013e328355a835. [DOI] [PubMed] [Google Scholar]
- 31.Makris D, Luna C, Nseir S. Ten ineffective interventions to prevent ventilator-associated pneumonia. Intensive Care Med. 2018;44:83–86. doi: 10.1007/s00134-017-4811-3. [DOI] [PubMed] [Google Scholar]
- 32.Olearo F, Albrich WC, Vernaz N, Harbarth S, Kronenberg A, Anresis Swiss Centre For Antibiotic Resistance. Staphylococcus aureus and methicillin resistance in Switzerland: regional differences and trends from 2004 to 2014. Swiss Med Wkly. 2016;146:w14339. doi: 10.4414/smw.2016.14339. [DOI] [PubMed] [Google Scholar]
- 33.Ammerlaan HS, Harbarth S, Buiting AG, Crook DW, Fitzpatrick F, Hanberger H, et al. Secular trends in nosocomial bloodstream infections: antibiotic-resistant bacteria increase the total burden of infection. Clin Infect Dis. 2013;56:798–805. doi: 10.1093/cid/cis1006. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.