Abstract
Introduction:
This study performed motor unit number estimation (MUNE) of the abductor hallucis (AH) muscle from 16 healthy control subjects based on the compound muscle action potential (CMAP) scan.
Methods:
Muscle responses to electrical stimuli ranging from sub-threshold to supramaximal intensity were recorded and MUNE was determined from a model of the responses (MscanFit program).
Results:
The average CMAP amplitude and MUNE of the AH for the right and left sides combined were 19.6 ± 0.75 mV and 127 ± 5 (mean ± standard error), respectively.
Discussion:
Findings of the study provide useful information about the motor unit number of the AH.
Keywords: motor unit number estimation (MUNE), compound muscle action potential (CMAP), CMAP scan, abductor hallucis (AH), muscle
Introduction
Motor unit number estimation (MUNE) is an electrophysiological technique to estimate the number of motor units in a muscle. Most MUNE techniques including the original incremental method and subsequent approaches (e.g. multiple point stimulation, F wave, spike triggered averaging) sample from a small proportion of the unit population1. A relatively new MUNE method that samples from a larger number of units is used to record a detailed compound muscle action potential (CMAP) scan followed by model simulation of the responses2 . In addition, the recording and modeling are automated, thereby reducing subjectivity in the estimation process and minimizing recording time.
The abductor hallicus (AH) muscle abducts and flexes the great toe and contributes to the maintenance of upright posture3. It is often used in nerve conduction studies since the tibial nerve is readily accessible4,5. However, studies of the estimated number of AH motor units are rather limited6,4,7,8. In one study of 11 healthy controls, the number of AH motor units was estimated by counting the number of step increments in the CMAP over the entire stimulus intensity range7. They found that the AH contained only 43 motor units on average, much less than the estimated number in other muscles of similar size9. However, Tilki and colleagues using the incremental MUNE method in 30 healthy adults found that the AH contained 98 motor units on average6. The reason for this difference in the estimated number of motor units is uncertain.
In this study, we applied CMAP scan and model simulation2 to estimate the number of motor units in the AH, as well as the reliability of the estimation, in healthy subjects.
Methods
Subjects
Sixteen healthy subjects (6 female) between 25 and 44 years of age (mean±std: 34±6 years) were recruited for the study. None had any known sensorimotor abnormalities, obvious atrophy of the foot muscles or neuropathy based on a medical history and examination, as well as measurement of tibial nerve conduction velocity, performed by a physician experienced in electrodiagnostic studies. The right leg was dominant in all subjects except two, based on which leg they preferred to kick a ball. They submitted written consent approved by the Committee for the Protection of Human Subjects at University of Texas Health Science Center at Houston and TIRR Memorial Hermann Hospital.
Experimental set-up
Subjects were supine on an ultrasound table (Biodex Medical System, NY) with hip and knee fully extended and the ankle in a neutral position. They remained relaxed and awake during all procedures. After abrading the skin with prep pads, the active recording electrode (Ag-AgCl, 11 mm in diameter) was positioned 1 cm below and proximal to the navicular tubercle, and secured with tape. The reference and ground electrodes were positioned on the metatarsophalangeal joint of the big toe and the medial malleolus, respectively. The tibial nerve was stimulated (200 µs pulse width) with a bar electrode posterior to the medial malleolus.
The CMAP scan was performed using the UltraPro S100 system (Natus Neurology, Inc., WI). The protocol included an automatic search of two stimulation thresholds followed by the definitive scan. The thresholds were defined as the intensity to evoke the lowest-threshold motor unit and the maximum CMAP. The stimulating site was selected to reduce the maximal intensity and subject discomfort. After the thresholds were established, the stimulating range for the CMAP scan was set from 1 mA beyond the CMAP maximum current to 2 mA below the lowest threshold, thereby accounting for the inherent variability in axonal thresholds.
The CMAP scan was performed automatically by delivering repeated stimuli from the highest intensity (supramaximal stimulation) to the lowest intensity (subthreshold for the lowest-threshold motor unit). The intensity decreased linearly in 1000 steps at 2 Hz, and took about 10 min to complete. In addition, 7 subjects were re-tested bilaterally 1 to 30 days later to determine the reliability of the CMAP scan measurements. All CMAP scans were performed by the same experimenter. Test order was randomized.
Data analysis
The CMAP amplitude, measured from baseline to negative peak, and corresponding stimulus intensity from 1000 stimuli were imported to the MScanFit program for MUNE calculation. The software was created by Prof. H Bostock and can be installed in most computers2.
The intraclass correlation coefficient (ICC), the standard error of measurement (SEM) and the smallest real difference (SRD) were calculated to assess the relative and absolute reliability of the MUNE and CMAP measurements10,11. SEM% and SRD% represent the percentages of the means of all observations. It is suggested that ICC>0.75, SEM% or SRD% <10% indicates excellent reliability. The coefficient of variation (COV) was computed12.
All data were screened for outliers and normality using SPSS (IBM, New York). Paired t-test was applied to compare the differences of CMAP, mean motor unit action potential (MUAP), and MUNE between dominant and non-dominant sides. All data are presented as mean±standard error unless specified.
Results
Calculation of MUNE
The mean stimulus currents for the lowest threshold and maximal CMAP responses were 10.23±0.87 mA and 30.48±1.02 mA. All subjects tolerated the procedures well and completed the CMAP scans. A stimulus-response curve recorded on two different days in 1 subject is shown in Fig. 1, and the corresponding MUNEs were 109 and 108. In general, the CMAP amplitude decreased smoothly, similar to the sigmoid function, with a reduction in stimulation intensity over the steep portion of the plot.
Figure 1.
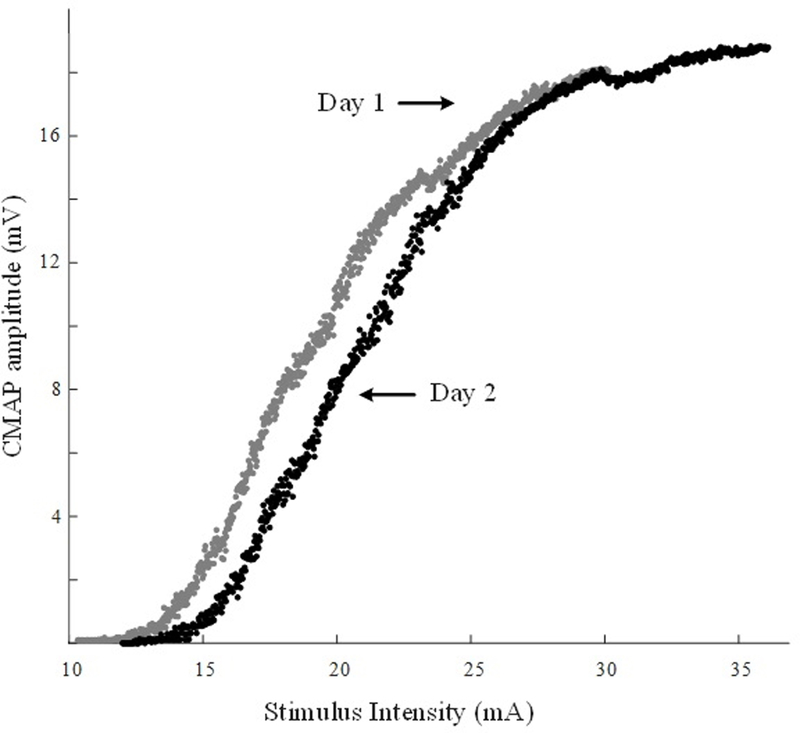
Examples of compound muscle action potential (CMAP) scan from the same subject on two different days. Stimulating ranges: 10.25 mA - 29.75 mA (day 1), 12 mA - 35.16 mA (day 2). Maximal CMAP amplitude: 18.1 mV (day 1), 18.78 mV (day 2). Mean motor unit size: 166.06 µV (day 1), 173.39 µV (day 2).
CMAP scans were recorded from AH muscles bilaterally in 14 subjects and unilaterally in 2. The AH CMAP amplitude, MUAP, and MUNE were not different between sides (CMAP: non-dominant: 19±1.01 mV, dominant: 20.27±1.13 mV, p=0.13; MUAP: non-dominant: 157.16±9.87 µV, dominant: 165.89±14 µV, p=0.3; MUNE: non-dominant: 126±8, dominant: 127±7, p=0.78). Therefore, the data from both sides were pooled. The mean values for CMAP, MUAP and MUNE, across 30 muscles were 19.64±0.75 mV (range: 9.9–28.2 mV), 160±7.79 µV (95.9–290 µV), and 127±5 (79–189), respectively.
Reliability test
Mean values for CMAP and MUNE were not different between the two test days (CMAP: p>0.51; MUNE: p>0.91). The ICCs, SEM% and SRD% suggested good measurement reliability (Table 1)
Table 1.
Reliability analysis
| CMAP |
MUNE |
|
|---|---|---|
| Test_1 | 20.04 ± 0.79 (mV) | 124 ± 6 |
| Test_2 | 20.43 ± 1.06 (mV) | 125 ± 8 |
| ICC | 0.89 | 0.93 |
| SEM% | 5.53% | 5.19% |
| SRD% | 4.98% | 3.7% |
| COV | 18.6±2.57% | 13.39±4.02% |
CMAP: compound muscle action potential; MUNE: motor unit number estimation
ICC: intraclass correlation coefficient; SEM: standard error of measurement; SRD: smallest real difference; COV: coefficient of variation
Discussion
We found that the number of the AH motor units was slightly larger than the mean of 98 units reported by Tilki and co-workers6 who used incremental MUNE. Our estimate is consistent with the modestly lower estimate of approximately 100 motor units in individual hand muscles, which likely have a slightly smaller cross-sectional area than the AH9. In addition, AH MUNE (incremental technique) after spinal cord injury ranged between 80 and 130 in 4 people, whereas values ranged from 5 to about 40 units in 8 others due to motor unit loss4. Our study included a repeat test in 7 of the 16 participants. The average MUNE showed no significant difference between the test and retest. Calculation of the absolute and relative reliability parameters also confirmed high measurement reliability.
The MUNE values of the present study and those of Tilki6 are 2 to 3 times as large as a previous report of 43 motor units7. The subjects were of similar age in all 3 studies (Johns and Fuglevand: 25–43 years, Tilki: 40±11 years, and ours: 34±6 years), so age cannot account for the different motor unit counts. The difference may result from different data collection and MUNE techniques. In the study by Johns and Fuglevand7, a less conventional setup was used, involving small recording electrodes (4 mm in diameter) and a shorter inter-electrode distance (2 cm). This may affect CMAP amplitude and lead to smaller motor unit counts. Unfortunately, the CMAP maximum was not reported in their study. In addition, the relatively larger stimulus pulse width (1 ms) applied in the previous study7 may increase the chances of recruiting multiple motor units with overlapping thresholds and account for a lower MUNE. A CMAP scan (~30 stimuli) is also used in statistical MUNE for selecting appropriate stimulus intensity13. The method assumes a Poisson distribution of motor unit size1,14 and generates lower MUNE compared with other methods15,1. The technique in this study uses more finely-graded stimuli to sample the entire motor neuron pool and implements a preliminary model to produce all individual units’ size. This approach may reduce the sampling bias introduced by using a small portion of the unit population for MUNE, and could be an additional reason for our higher counts7. The advantages of the present approach include automation of data collection and analysis which minimizes experimenter bias, and excellent reliability16. However, CMAP scan requires intensive stimulation which may limit the test to participants with good tolerance. Further study is needed to determine whether similar results can be achieved using a smaller number of stimuli.
Acknowledgement
We would like to thank Dr. H. Bostock for the help in data analysis. This study was supported in part by the National Institutes of Health of the U.S. Department of Health and Human Services (Grant #: R21 NS093727) and TIRR Rehabilitation Innovations Pilot Grant, and also in part by the Guangzhou Science and Technology Program (No. 201704030039).
Abbreviations:
- MUNE
Motor unit number estimation
- CMAP
compound muscle action potential
- AH
abductor halluces
- ICC
intraclass correlation coefficient
- SEM
standard error of measurement
- SRD
smallest real difference
- COV
coefficient of variation
- MUAP
motor unit action potential
Footnotes
Ethical Publication Statement
We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
Disclosure of Conflicts of Interest
None of the authors has any conflict of interest to disclose.
Reference
- 1.Gooch CL, Doherty TJ, Chan KM, Bromberg MB, Lewis RA, Stashuk DW, Berger MJ, Andary MT, Daube JR. Motor unit number estimation: a technology and literature review. Muscle Nerve 2014;50(6):884–893. [DOI] [PubMed] [Google Scholar]
- 2.Bostock H Estimating motor unit numbers from a CMAP scan. Muscle Nerve 2016;53(6):889–896. [DOI] [PubMed] [Google Scholar]
- 3.Kelly LA, Kuitunen S, Racinais S, Cresswell AG. Recruitment of the plantar intrinsic foot muscles with increasing postural demand. Clin Biomech (Bristol, Avon) 2012;27(1):46–51. [DOI] [PubMed] [Google Scholar]
- 4.Hunter J, Ashby P. Secondary changes in segmental neurons below a spinal cord lesion in man. Arch Phys Med Rehabil 1984;65(11):702–705. [PubMed] [Google Scholar]
- 5.Van De Meent H, Hosman AJ, Hendriks J, Zwarts M, Group E- SS, Schubert M. Severe degeneration of peripheral motor axons after spinal cord injury: a European multicenter study in 345 patients. Neurorehabil Neural Repair 2010;24(7):657–665. [DOI] [PubMed] [Google Scholar]
- 6.Erdem Tilki H, Coskun M, Unal Akdemir N, Incesu L. Axon count and sympathetic skin responses in lumbosacral radiculopathy. J Clin Neurol 2014;10(1):10–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Johns RK, Fuglevand AJ. Number of motor units in human abductor hallucis. Muscle Nerve 2011;43(6):895–896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Neuwirth C, Nandedkar S, Stalberg E, Barkhaus PE, Carvalho M, Furtula J, van Dijk JP, Baldinger R, Castro J, Costa J, Otto M, Sandberg A, Weber M. Motor Unit Number Index (MUNIX): reference values of five different muscles in healthy subjects from a multi-centre study. Clinical neurophysiology : official journal of the International Federation of Clinical Neurophysiology 2011;122(9):1895–1898. [DOI] [PubMed] [Google Scholar]
- 9.McComas AJ. 1998 ISEK Congress Keynote Lecture: Motor units: how many, how large, what kind? International Society of Electrophysiology and Kinesiology. J Electromyogr Kinesiol 1998;8(6):391–402. [DOI] [PubMed] [Google Scholar]
- 10.Weir JP. Quantifying test-retest reliability using the intraclass correlation coefficient and the SEM. J Strength Cond Res 2005;19(1):231–240. [DOI] [PubMed] [Google Scholar]
- 11.Chuang LL, Lin KC, Wu CY, Chang CW, Chen HC, Yin HP, Wang L. Relative and absolute reliabilities of the myotonometric measurements of hemiparetic arms in patients with stroke. Arch Phys Med Rehabil 2013;94(3):459–466. [DOI] [PubMed] [Google Scholar]
- 12.Nandedkar SD, Barkhaus PE, Stalberg EV. Reproducibility of MUNIX in patients with amyotrophic lateral sclerosis. Muscle Nerve 2011;44(6):919–922. [DOI] [PubMed] [Google Scholar]
- 13.Daube JR. Estimating the number of motor units in a muscle. J Clin Neurophysiol 1995;12(6):585–594. [DOI] [PubMed] [Google Scholar]
- 14.Shefner JM, Gooch CL. Motor unit number estimation. Phys Med Rehabil Clin N Am 2003;14(2):243–260. [DOI] [PubMed] [Google Scholar]
- 15.Blok JH, van Dijk JP, Drenthen J, Maathuis EM, Stegeman DF. Size does matter: the influence of motor unit potential size on statistical motor unit number estimates in healthy subjects. Clin Neurophysiol 2010;121(10):1772–1780. [DOI] [PubMed] [Google Scholar]
- 16.Jacobsen AB, Bostock H, Fuglsang-Frederiksen A, Duez L, Beniczky S, Moller AT, Blicher JU, Tankisi H. Reproducibility, and sensitivity to motor unit loss in amyotrophic lateral sclerosis, of a novel MUNE method: MScanFit MUNE. Clin Neurophysiol 2017. [DOI] [PubMed]


