ABSTRACT
Infertility, which affects ∼15% of the world's population, is a global public health issue recognized by the WHO. Therefore, it is of major clinical and public health importance to investigate whether modifiable lifestyle factors—such as stress, drug use, smoking, alcohol intake, and diet—may influence human fertility. A systematic review and meta-analysis of randomized clinical trials (RCTs) from the MEDLINE-PubMed database was conducted to assess the effect of nutrients, dietary supplements, or food on sperm quality parameters. In total, 28 articles were included for qualitative analysis and 15 for quantitative meta-analysis. Total sperm concentrations [expressed as mean differences (MDs); 95% CIs, in spermatozoa (spz)/mL] were increased by selenium (3.91 × 106 spz/mL; 3.08, 4.73 spz/mL), zinc (1.48 × 106 spz/mL; 0.69, 2.27 spz/mL), omega-3 (n–3) fatty acids (10.98 × 106 spz/mL; 10.25, 11.72 spz/mL), and coenzyme Q10 (CoQ10) (5.93 × 106 spz/mL; 5.36, 6.51 spz/mL). Sperm counts were increased by ω-3 fatty acids (18.70 × 106 spz/mL; 16.89, 20.51 spz/mL) and CoQ10 supplementation (10.15 × 106 spz/mL; 8.34, 11.97 spz/mL). Sperm total motility was increased by selenium (3.30%; 2.95%, 3.65%), zinc (7.03%; 6.03%, 8.03%), ω-3 fatty acids (7.55%; 7.09%, 8.01%), CoQ10 (5.30%; 4.98%, 5.62%), and carnitines (7.84%; 6.54%, 9.13%), whereas sperm progressive motility was increased only after supplementation with carnitines (7.45%; 6.24%, 8.67%). Finally, sperm morphology was enhanced by selenium (1.87%; 1.50%, 2.24%), ω-3 fatty acid (0.91%; 0.69%, 1.13%), CoQ10 (1.06%; 0.72%, 1.41%), and carnitine (4.91%; 3.68%, 6.15%) supplementation. This meta-analysis of RCTs suggests that some dietary supplements could beneficially modulate sperm quality parameters and affect male fertility. However, results must be cautiously interpreted due to the limited sample size of the meta-analyzed studies and the considerable observed interstudy heterogeneity.
The present study and the corresponding search protocol were registered at the PROSPERO registry at http://www.crd.york.ac.uk/PROSPERO as CRD42017058380.
Keywords: diet, nutrition, nutrients, food, sperm quality, male infertility, RCT
Introduction
Infertility, which affects ∼15% of the world's population, is a global public health issue recognized by the WHO (1). In the case of male fertility, a recent meta-regression analysis reported a significant worldwide decline in total sperm counts between 1973 and 2011 (2). These data strongly suggest a significant decline in male reproductive health, with crucial implications for human reproduction and perpetuation of the species. Research aimed at revealing the causes and implications of this decline is therefore urgently needed.
Investigating modifiable lifestyle factors that influence human fertility—such as stress, drug use, smoking, alcohol intake, and diet—is of major clinical and public health importance for understanding the problem. Indeed, several observational studies that explored the associations between dietary patterns, food and nutrient consumption, and sperm quality suggest that adhering to a healthy diet (e.g., the Mediterranean diet) may improve male sperm quality parameters (3). In addition to observational studies, which are important for creating new hypotheses, randomized clinical trials (RCTs) are also needed. Such trials are considered the gold standard in terms of scientific evidence if the quality of design of the interventions and the execution of the trial are high, because they enable strong conclusions to be drawn and can be used for future clinical and public health recommendations. Several RCTs have tested the effect of food and nutrients on male fertility parameters. Differences in supplements tested, study duration and design, as well as the different interventions, populations, and measured outcomes make it extremely difficult to compare these trials.
One systematic review of clinical trials recently attempted to summarize knowledge in this field (4). Unfortunately, the authors of the review merged observational studies and RCTs, did not take into account certain relevant articles, and included others that were of low quality or that contained a high risk of bias (ROB), which made it difficult to draw strong conclusions.
The aims of the present systematic review of RCTs that have tested the effect of nutrients, dietary supplements, or food on sperm quality parameters were as follows: 1) to update scientific evidence on the topic by assessing the ROB in all the articles selected, and 2) to meta-analyze the effect of similar interventions on selected endpoints.
Methods
Protocol and registration
The present study and the corresponding search protocol have been registered in the PROSPERO registry (http://www.crd.york.ac.uk/PROSPERO) as PROSPERO 2017: CRD42017058380.
Literature search strategy
A systematic, comprehensive search of the literature published between the earliest available online indexing year and October 2017 by searching the MEDLINE-PubMed database (http://www.ncbi.nlm.nih.gov/pubmed) and hand-searching the reference lists of the retrieved papers was carried out in accordance with the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (5, 6).
With the use of Medical Subject Headings and keywords, 2 search subsets were used: the first subset comprised male infertility-related keywords [fertility OR infertility OR male infertility OR male fertility OR sperm dysfunction(s) OR sperm DNA damage OR varicocele OR asthenozoospermia OR oligozoospermia OR oligoasthenozoospermia OR oligoasthenoteratozoospermia OR teratozoospermia]; and the second subset comprised nutrition and/or diet-related keywords [diet OR nutrients OR food OR food supplement OR dietary supplement OR probiotic OR nuts OR vitamin C OR vitamin E OR zinc OR antioxidants OR cereals OR meat OR vegetable OR fruit OR fishes OR legumes OR milk OR yogurt OR cheese OR seeds OR eggs OR dairy product(s) OR micronutrient(s) OR vitamins OR alcohol consumption OR l-carnitine OR n-acetylcysteine OR glutathione OR coenzyme q10 OR selenium OR fatty acids OR sugar]. The following inclusion filters were applied in the search: Classical Article, Clinical Conference, Clinical Study, Clinical Trial, Clinical Trial-Phase I, Clinical Trial-Phase II, Clinical Trial-Phase III, Clinical Trial-Phase IV, Controlled Clinical Trial, English Abstract, Journal Article, Letter, Meta-Analysis, Multicenter Study, Pragmatic Clinical Trial, Evaluation Studies, Case Reports, Congresses, Dataset, Introductory Journal Article, Abstract, Humans, English, and Male. The complete search strategy is available in Supplemental Appendix 1.
Eligibility criteria and study selection
The titles and abstracts of all the preselected articles were screened for eligibility by 2 independent researchers (AS-H and NR-E), who are specialists in male (in)fertility and human nutrition, respectively. Any discrepancies were re-evaluated together with a third author (JS-S). After primary screening (to evaluate the scope of the study), the full texts of the selected articles were obtained. Only RCT studies in which fertile/infertile men were well defined (men with or without sperm disorders, sperm DNA damage, or idiopathic infertility) were included for the qualitative analysis. The primary outcomes of the selected studies had to have referred to the following semen quality parameters: semen volume, ejaculate pH, total sperm count or concentration, sperm vitality, sperm motility (progressive or total motility), sperm morphology, acrosome resistance, sperm DNA fragmentation (SDF) or damage, sperm chromatin integrity, sperm reactive oxygen species (ROS), sperm aneuploidies, sperm function parameters, or hormonal levels. Exclusion criteria were as follows: case-control, cross-sectional, observational prospective or retrospective studies, animal or in vitro studies, review articles, studies conducted on individuals with varicocele or other fertility-related diseases, studies with drug interventions, studies with ≤15 participants per intervention, uncontrolled intervention studies, and studies with a high ROB (see the ROB section). Finally, RCTs testing the effect of food extracts, botanic extracts, or drugs have also been excluded from the present review.
Data extraction
With the use of a standardized model, the following information from each study was extracted: authors, year of publication, journal, title of the article, location of the study, age, population studied, sample size, study design (parallel or crossover), interventions, primary outcomes, and main conclusions. Data were first extracted and further checked by the researchers for discrepancies in order to minimize the possibility of errors.
ROB
Analyzing all the data extracted, the quality of the studies selected was evaluated through a ROB index based on 7 categories (7). ROB was assessed in parallel by 2 authors (AS-H and NR-E) and discrepancies were re-evaluated together with a third author (JS-S). Applying this system, the ROB of individual studies was assessed with the use of the following criteria: 1) random sequence generation (due to inadequate generation of randomized sequences); 2) allocation concealment (due to inadequate concealment of allocations before assignment); 3) blinding of participants and personnel (due to knowledge of the allocated interventions by participants and personnel during the study); 4) blinding of outcome assessment (due to knowledge of the allocated interventions by outcome assessors); 5) incomplete outcome data (due to the amount, nature, or handling of incomplete outcome data); 6) selective reporting (due to selective outcome reporting); and 7) other bias (due to problems not covered elsewhere). Studies whose mean ROB was high were considered to be of low quality and therefore excluded, whereas those whose mean ROB was low or unclear were accepted for the systematic qualitative review and quantitative analysis.
Statistical analysis
Meta-analysis was conducted through the use of Review Manager (RevMan) software version 5.3 (http://community.cochrane.org/tools/review-production-tools/revman-5) in accordance with the Cochrane guidelines (7). The difference in the change from the baseline values for the intervention and placebo/control arms was derived from each trial. However, if the change from the baseline values was not available, end-of-treatment values were used. When necessary, an imputed SD or SE for the between-treatment difference was calculated. In crossover trials, to impute SD for between-treatment differences, correlation coefficients between baseline and end-of-treatment values within each trial were derived via a published equation (8). When multiple intervention arms were present in a single trial, intervention arms were pooled to obtain a single pairwise comparison to mitigate unit‐of‐analysis error. To evaluate the differences in sperm quality parameters between the intervention and the control groups, the data were pooled through the use of the inverse variance method with the fixed effects model and the results were expressed as mean differences (MDs) with 95% CIs. Statistical significance was set at P < 0.05. Heterogeneity between the studies was evaluated via a chi-square test and the I2 index with the significance level set at P < 0.10. I2 values <50% were deemed moderate, ≥50% to <75% were deemed substantial, and ≥75% were deemed of considerable heterogeneity (7).
Results
Study characteristics
A total of 2381 articles were identified after a primary search of MEDLINE-PubMed and 1 study from other sources (Supplemental Figure 1). After analyzing every abstract (n = 2382), 2240 articles were excluded because they were beyond the scope of the present study (did not assess the effect of nutrients, supplements, or food on sperm quality parameters). A total of 142 articles were collected as full texts and their inclusion/exclusion criteria and ROB were assessed: of these articles, 110 were excluded because they did not meet the inclusion/exclusion criteria (no control group, n = 34; small sample size, n = 26; non-RCT, n = 22; did not meet primary outcome, n = 5; studies with animals or nonmale subjects, n = 3; in vitro studies, n = 2; participants with varicocele, n = 5; other fertility-related diseases, n = 8; using intervention with drugs, n = 3; 2 interventions at the same time, n = 1; and using a food extract, n = 1), and 4 were excluded because they were classified as having a high ROB. After applying all the eligibility parameters, 28 articles were included for qualitative analysis. When ≥2 studies had analyzed the same exposures and outcomes, the results were meta-analyzed. Therefore, 15 were quantitatively analyzed via a meta-analysis approach.
The articles included subjects (n = 2900) from 11 countries: Australia, England, Germany, Iran, Italy, Kuwait, Netherlands, Saudi Arabia, Scotland, Spain, and the United States. The age of the participants ranged from 18 to 52 y. There were 26 parallel-group RCTs and 2 crossover RCTs.
Qualitative analysis
Eight of the 28 articles assessed antioxidant supplements (9–16). Four articles evaluated folic acid and/or zinc (17–20), 2 articles evaluated omega-3 fatty acid supplements (21, 22), 5 articles assessed coenzyme Q10 (CoQ10) supplements (23–27), 3 assessed carnitines (28–30), and 6 assessed some other dietary supplements (31–36). All the studies evaluated had sperm parameters and quality as study outcomes (Table 1).
TABLE 1.
Summary of the RCT studies investigating the effect of nutrition on sperm quality parameters or sexual hormones1
| Reference | Location | Age (y) | Population studied | Background diet controlled during the study? | Design | Intervention | Primary outcome | Principal conclusion | ROB1 | ROB2 | ROB3 | ROB4 | ROB5 | ROB6 | ROB7 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Antioxidant | |||||||||||||||
| Haghighian et al. (16) | Iran | Intervention (32.98 ± 5.35); placebo (34.12 ± 4.79) | 44 patients with iA (23 intervention; 21 placebo) | No | Parallel | 12 wk. α-Lipoic acid (600 mg) or placebo per day | Sperm parameters (semen volume, sperm count and concentration, sperm motility, vitality, and morphology) | α-Lipoic acid improves sperm count and concentration, and motility (progressive motility). No effect on semen volume, sperm vitality, or morphology. | LB | UB | LB | LB | LB | LB | UB |
| Hawkes et al. (9) | United States | 18–45 | 42 healthy participants (22 intervention; 20 placebo) | No | Parallel | 11 mo. Se (300 µg) or placebo (Se < 1.5 µg) per day | Semen parameters (semen volume, sperm count and concentration, sperm motility, and morphology) and serum hormones (T, LH, PSA) | Se has no effect on conventional sperm parameters or serum hormones. | UB | UB | LB | LB | LB | UB | UB |
| Safarinejad and Safarinejad (10) | Iran | Intervention Se (31 ± 9); intervention NAC (32 ± 10); intervention NAC + Se (31 ± 8); placebo (31 ± 9) | 420 patients with iOAT (314 intervention [105 Se, 105 NAC, 104 NAC + Se]; 106 placebo) | No | Parallel | 6 mo. Se (200 µg), NAC (600 mg), Se + NAC (200 µg + 600 mg respectively), or placebo per day | Sperm parameters (semen volume, sperm count and concentration, sperm motility, and morphology), serum hormones (T, LH, FSH, inhibin B, PRL) | Se, NAC, or Se + NAC improve all conventional sperm parameters (semen volume, sperm count and concentration, sperm motility, and morphology), except sperm motility in NAC group. Se, NAC, or Se + NAC increase T, LH, and inhibin B but decrease FSH. No effect on PRL serum hormone. | LB | UB | LB | LB | UB | LB | UB |
| Greco et al. (15) | Italy | ND | 64 patients with idiopathic infertility (32 intervention and 32 placebo) | No | Parallel | 2 mo. Vitamin C (1 g) + vitamin E (1 g), or placebo per day | Semen parameters (sperm count and concentration, sperm motility, and morphology), sperm DNA damage | Vitamin C and E improve sperm DNA fragmentation index. No effect on conventional semen parameters. | UB | LB | LB | LB | LB | UB | UB |
| Rolf et al. (11) | Germany | Intervention (36.1 ± 5.0); placebo (35.2 ± 4.8) | 31 patients with asthenozoospermia or moderate oligoasthenozoospermia (15 intervention; 16 placebo) | No | Parallel | 8 wk. Vitamin C (1000 mg) + vitamin E (800 mg), or placebo per day | Sperm parameters (semen volume, sperm concentration, sperm motility, sperm vitality, and morphology), serum hormones (T, FSH, LH, E2) | Vitamin C + E has no effect on conventional semen parameters or hormones. | LB | LB | LB | LB | LB | LB | UB |
| Scott et al. (12) | Scotland | Se intervention (32.6 ± 1.1); Se + vitamins intervention (33.9 ± 0.9); placebo (32.9 ± 1.5) | 64 patients with asthenozoospermia (46 intervention [16 Se, 30 Se + vitamins]; 18 placebo) | No | Parallel | 3 mo. Selenium (100 µg), selenium + vitamin A (1 mg) + vitamin C (10 mg) + vitamin E (15 mg), or placebo per day | Sperm parameters (sperm concentration and motility), conception chance | Selenium improves sperm motility and chance of conception. No effect on sperm concentration. | LB | UB | LB | LB | UB | UB | UB |
| Suleiman et al. (14) | Saudi Arabia | Intervention (27–52); placebo (22–45) | 87 patients with asthenozoospermia (52 intervention and 35 placebo) | No | Parallel | 6 mo. Vitamin E (300 mg) or placebo per day | Sperm parameters (sperm motility) and pregnancy rate | Vitamin E improves sperm motility and pregnancy rate. | UB | UB | LB | LB | UB | UB | UB |
| Kessopoulou et al. (13) | England | 32 (26–49) | 30 healthy participants with high levels of ROS | No | Crossover | 3 mo. 1 mo WO, 3 mo vitamin E intervention/placebo, 1 mo WO, 3 mo vitamin E intervention/placebo. Vitamin E (600 mg) or placebo per day | Sperm parameters (semen volume, sperm concentration, sperm motility, and morphology), zona binding test, ROS | Vitamin E improves the zona binding test. No effect on ROS or conventional sperm parameters. | UB | UB | LB | LB | UB | LB | UB |
| Folic acid and/or zinc | |||||||||||||||
| Raigani et al. (19) | Iran | ND | 83 patients with iOAT (65 intervention [20 folic acid, 24 zinc sulfate, 21 folic acid + zinc sulfate]; 18 placebo) | No | Parallel | 16 wk. Folic acid (5 mg), zinc sulfate (220 mg), folic acid + zinc sulfate (5 mg + 220 mg, respectively), or placebo per day | Sperm parameters (sperm concentration, sperm motility, sperm vitality, and morphology), DNA damage, and chromatin damage | Zinc sulfate improves sperm chromatin integrity. Folic acid and/or zinc sulfate has no effect on conventional sperm parameters. | UB | UB | LB | LB | UB | LB | UB |
| Ebisch et al. (17) | Netherlands | Subfertile men 35.0 (32.3–37.0); fertile 34.0 (31.0–38.0) | 87 participants (42 intervention [24 fertile, 18 subfertile] and 45 placebo [23 fertile, 22 subfertile]) | No | Parallel | 6 mo. Folic acid (5 mg) + zinc sulfate (66 mg), or placebo per day | Semen parameters (semen volume, sperm concentration, sperm motility, and morphology), serum hormones (T, FSH, inhibin B) | Folic acid + zinc sulfate improve sperm concentration. No effect on semen volume, sperm motility or morphology, or serum hormones. | LB | UB | LB | LB | UB | LB | UB |
| Wong et al. (18) | Netherlands | Subfertile men (34.3 ± 3.9); fertile men (34.2 ± 4.2) | 193 healthy participants (99 fertile, 94 subfertile) | No | Parallel | 6 mo. Folic acid (5 mg), zinc sulfate (66 mg), folic acid + zinc sulfate (5 mg + 66 mg, respectively), or placebo per day | Sperm parameters (semen volume, sperm count and concentration, motility, and morphology) | Folic acid + zinc sulfate improves sperm concentration and morphology; folic acid improves sperm morphology in subfertile patients. Zinc improves sperm morphology in fertile patients. No effect on semen volume, sperm count, or motility. | LB | LB | LB | LB | LB | LB | UB |
| Omu et al. (20) | Kuwait | Intervention (37.8 ± 7.9); control (38.1 ± 8.2) | 97 patients with asthenozoospermia (49 intervention; 48 control) | No | Parallel | 3 mo. Zinc (500 mg) per day, or no therapy | Sperm parameters (semen volume, sperm concentration, sperm motility, and morphology), HOS, serum hormones (T, FSH, LH, PRL), pregnancy, abortion, delivered or ongoing pregnancies | Zinc improves sperm concentration, sperm motility, sperm integrity membrane (HOS), fertilizing capacity, conception, and pregnancy. | UB | UB | UB | UB | LB | LB | UB |
| ω-3 Fatty acid | |||||||||||||||
| Martínez-Soto et al. (21) | Spain | Intervention (35 ± 0.8); placebo (35.6 ± 1.0) | 57 patients attending an infertility clinic (32 intervention; 25 placebo) | No | Parallel | 10 wk. ω-3 DHA-enriched oil (990 mg DHA + 135 mg EPA) or placebo (1500 mg sunflower oil) per day | Sperm parameters (semen volume, sperm count and concentration, sperm motility, and morphology), sperm DNA fragmentation | DHA reduces the percentage of spermatozoa with DNA damage. No effect on any conventional sperm parameter. | UB | UB | LB | LB | UB | LB | UB |
| Safarinejad (22) | Iran | Intervention (32 ± 9); placebo (32 ± 10) | 211 patients with iOAT (106 intervention; 105 placebo) | No | Parallel | 32 wk. ω-3 Group (1.12 g EPA + 0.72 g DHA) or placebo per day | Sperm parameters (semen volume, sperm count and concentration, sperm motility, and morphology), serum hormones (T, LH, FSH, E2, PRL) | EPA and DHA improve sperm count and concentration, sperm motility, and morphology. No effect on semen volume or serum hormones. | UB | UB | LB | LB | UB | LB | UB |
| CoQ10 | |||||||||||||||
| Nadjarzadeh et al. (23) | Iran | Intervention (34.17 ± 4.52); placebo (34.67 ± 6.69) | 47 patients with iOAT (23 intervention; 24 placebo) | Yes | Parallel | 12 wk. CoQ10 (200 mg), or placebo (containing lactose) per day | Sperm parameters (sperm concentration, sperm motility, and morphology) | CoQ10 has no effect on conventional sperm parameters. | UB | UB | LB | LB | UB | LB | UB |
| Safarinejad et al. (24) | Iran | Intervention (31); placebo (32) | 191 patients with iOAT (96 intervention; 95 placebo) | No | Parallel | 6 mo. Ubiquinol, or reduced CoQ10 (200 mg), or placebo per day | Sperm parameters (semen volume, sperm count and concentration, sperm motility, and morphology), and serum hormones (T, LH, FSH, inhibin B, PRL) | Ubiquinol improves sperm count, concentration, and motility, increases inhibin B, and reduces LH and FSH. No effect on semen volume, sperm morphology, T, or PRL. | LB | LB | LB | LB | UB | LB | UB |
| Nadjarzadeh et al. (25) | Iran | Intervention (34.17 ± 4.52); placebo (34.67 ± 6.69) | 47 patients with iOAT (23 intervention; 24 placebo) | Yes | Parallel | 12 wk. CoQ10 (200 mg), or placebo (containing lactose) per day | Sperm parameters (semen volume, pH, sperm count and concentration, sperm motility, and morphology) | CoQ10 has no effect on conventional sperm parameters. | UB | UB | LB | LB | UB | LB | UB |
| Balercia et al. (26) | Italy | 32 (27–39) | 55 patients with iA (28 intervention; 27 placebo) | No | Parallel | 6 mo. CoQ10 (200 mg) or placebo per day | Semen parameters (sperm concentration, motility, and morphology) | CoQ10 improves sperm motility. No effect on sperm concentration or morphology. | UB | UB | LB | LB | LB | LB | UB |
| Safarinejad (27) | Iran | Intervention (28 ± 9); placebo (28 ± 10) | 194 patients with iOAT (98 intervention; 96 placebo) | No | Parallel | 6 mo. CoQ10 (300 mg) or placebo per day | Sperm parameters (semen volume, sperm count and concentration, sperm motility, and morphology), acrosome-reacted spermatozoa, and serum hormones (T, LH, FSH, inhibin B, PRL) | CoQ10 improves sperm count and concentration, sperm motility, and morphology. Se reduces FSH and LH, and increases inhibin B and acrosome-reacted spermatozoa. | LB | UB | LB | LB | LB | LB | UB |
| Carnitine | |||||||||||||||
| Balercia et al. (28) | Italy | 24–38 | 59 patients with iA (44 intervention [15 LC, 15 LAC, 14 LC + LAC]; 15 placebo) | No | Parallel | 6 mo. LC (3 g), LAC (3 g), LC + LAC (2 g + 1 g, respectively), or placebo (malic acid, sodium benzoate, sodium saccharinate dihydrate, anhydrous sodium citrate, pineapple flavoring, and demineralized water) per day | Sperm parameters (semen volume, sperm concentration, sperm motility, and morphology) | LC, LAC, or LC + LAC improve sperm motility. LAC improves sperm concentration and LC sperm morphology. No effect on semen volume. | UB | UB | LB | LB | LB | LB | UB |
| Lenzi et al. (29) | Italy | 20–40 | 56 patients with iOAT (30 intervention; 26 placebo) | No | Parallel | 6 mo. LC (2 g) + LAC (1 g), or placebo per day | Sperm parameters (semen volume, sperm concentration, motility, and morphology) | LC + LAC improve sperm motility. No effect on semen volume, sperm concentration, or morphology. | UB | UB | LB | LB | LB | LB | UB |
| Lenzi et al. (30) | Italy | 20–40 | 86 infertile patients | No | Crossover | 2 mo WO, 2 mo LC intervention/placebo, 2 mo WO, and 2 mo LC intervention/placebo. LC (2 g), or placebo per day | Sperm parameters (semen volume, sperm concentration, motility, and morphology) | LC improves sperm concentration and motility. No effect on semen volume or sperm morphology. | UB | UB | LB | LB | UB | LB | UB |
| Dietary supplements | |||||||||||||||
| Maretti and Cavallini (31) | Italy | Intervention (37; 32–42); placebo (36; 30–43) | 41 patients with iOAT (20 intervention; 21 placebo) | No | Parallel | 6 mo. Flortec (Lactobacillus paracasei B21060 5 × 109 CFUs + arabinogalactan 1243 mg + oligo-fructosaccharides 700 mg + l-glutamine 500 mg) or placebo (alimentary starch) per day | Sperm parameters (semen volume, pH, sperm count and concentration, sperm motility, and morphology), serum hormones (T, LH, FSH, E2, PRL) | Flortec improves semen volume, sperm count, sperm concentration, progressive motility, and morphology, also improves FSH, LH, and T levels. No effect on PRL and E2. | LB | UB | LB | LB | LB | LB | UB |
| Calogero et al. (32) | Italy | Intervention (28 ± 9); placebo (28 ± 10) | 194 patients with idiopathic infertility (98 intervention; 96 placebo) | No | Parallel | 3 mo. Inofolic (4 g MI + 400 µg folic acid) or placebo (400 µg folic acid alone) per day | Sperm parameters (semen volume, sperm count and concentration, sperm motility, and morphology), acrosome-reacted spermatozoa, serum hormones (T, LH, FSH, inhibin B, PRL) | MI increases sperm count and concentration, motility (progressive motility), and T levels, and decreases acrosome-reacted spermatozoa, LH, and FSH levels. No effect on semen volume, PRL, or inhibin B. | UB | UB | LB | LB | LB | UB | UB |
| Kolahdooz et al. (33) | Iran | Intervention (31.5 ± 1.1); placebo (32.1 ± 0.8) | 68 patients with idiopathic infertility (34 intervention; 34 placebo) | No | Parallel | 2 mo. Nigella sativa seed oil (5 mg of N. sativa oil) or placebo (liquid paraffin) per day | Sperm parameters (semen volume, pH, sperm concentration, motility, and morphology) | N. sativa improves semen volume and pH, sperm concentration, motility, and morphology, and semen round cells in the ejaculate (reduction of this type of cells). | UB | LB | LB | LB | UB | LB | UB |
| Robbins et al. (34) | United States | Intervention (25.6 ± 4.0); control (24.8 ± 3.7) | 107 healthy participants (55 intervention; 52 control) | Yes | Parallel | 12 wk. Walnuts (75 g) or no tree nuts consumption (control) in the context of a Westernized diet | Sperm parameters (sperm concentration, motility, vitality, and morphology), and sperm aneuploidy (X, Y, 18) | Walnuts improve sperm motility, vitality, and morphology. No effect on semen volume, sperm concentration, or aneuploidy. | LB | UB | LB | LB | LB | LB | UB |
| Safarinejad et al. (35) | Iran | Intervention (28.4 ± 5.2); placebo (28.8 ± 5.6) | 230 patients with iOAT (114 intervention; 116 placebo) | No | Parallel | 6 mo. Saffron (60 mg Crocus sativa) or placebo per day | Sperm parameters (semen volume, sperm count and concentration, sperm motility, and morphology), serum hormones (T, LH, FSH, PRL, TSH) | Saffron has no effect on conventional sperm parameters or serum hormones. | UB | UB | LB | LB | UB | LB | UB |
| Tremellen et al. (36) | Australia | Intervention (37.1 ± 5.1); placebo (35.5 ± 4.3) | 60 patients with idiopathic infertility attending an infertility clinic (n = ND) | No | Parallel | 3 mo. Menevit (lycopene 6 mg, vitamin E 400 IU, vitamin C 100 mg, zinc 25 mg, selenium 26 µg, folate 0.5 mg, garlic 1 g, palm oil) or placebo (palm oil) per day | Sperm parameters (sperm concentration, sperm motility, sperm vitality, and morphology), SDF, and fecundability parameters (cleavage stage embryo quality, oocyte fertilization rate, and pregnancy rates) | Menevit improves pregnancy rates during IVF-ICSI treatment. No effect on conventional sperm parameters. | LB | UB | LB | LB | LB | LB | UB |
1The studies are arranged by supplement compounds and from the most recent to the oldest study. The studies are classified and ordered as: antioxidant supplement studies, folic acid and/or zinc studies, ω-3 fatty acid studies, CoQ10 studies, carnitine studies, and dietary supplement studies. Age is given as mean ± SD or mean (range) where such data are available. CoQ10, coenzyme Q10; E2, estradiol; FSH, follicle-stimulating hormone; HOS, hypo-osmotic swelling; iA, idiopathic asthenozoospermia; ICSI, intra-citoplasmatic sperm injection; iOAT, idiopathic oligoasthenoteratozoospermia; IVF, in vitro fertilization; LAC, l-acetyl carnitine; LB, low risk of bias; LC, l-carnitine; LH, luteinizing hormone; MI, myoinositol; NAC, N-acetyl cysteine; ND, no data; PRL, prolactin; PSA, prostatic-specific antigen; RCT, randomized clinical trial; ROB1, random sequence generation; ROB2, allocation concealment; ROB3, blinding of participants and personnel; ROB4, blinding of outcome assessment; ROB5, incomplete outcome data; ROB6, selective reporting; ROB7, other bias; ROS, reactive oxygen species; SDF, sperm DNA fragmentation; T, testosterone; TSH, thyroid-stimulating hormone; UB, unclear risk of bias; WO, washout period.
Antioxidant supplements
The vast majority of the RCTs were conducted with the use of antioxidants or cocktails of antioxidants. The studies included in the qualitative analysis are shown in Table 1 (9–16).
Selenium supplements were tested in 3 studies (9, 10, 12). Whereas Hawkes et al. (9) reported that supplementation with 300 µg Se/d had no effect on conventional sperm parameters or serum hormones, Scott et al. (12) reported that 100 µg Se/d for 3 mo improved sperm motility and increased the chance of conception, and Safarinejad and Safarinejad (10) reported that 200 µg Se/d for 6 mo improved semen volume, total sperm count and concentration, and morphology. In the study by Scott et al. (12), adding vitamin C and vitamin E to the supplement had no synergic effect on these semen parameters. In the study by Safarinejad and Safarinejad (10), adding N-acetyl cysteine to the selenium supplement improved these parameters but also affected certain sex hormones, increasing testosterone, luteinizing hormone (LH), and inhibin B and decreasing follicle-stimulating hormone (FSH).
No effect on conventional semen parameters and sex hormones has been demonstrated with the use of vitamin E or vitamin C + E supplementation (11, 13, 15), except in 1 RCT (14) in which the administration of 300 mg vitamin E significantly improved sperm motility after 6 mo. However, an improvement in fecundity capacity was reported with the use of the zona binding test after 3 mo of vitamin E supplementation (13) in sperm DNA fragmentation indexes after 2 mo of vitamin C + E supplementation (15) and in pregnancy rates after 3 mo of vitamin E supplementation (14).
Finally, supplementation for 12 wk with 600 mg α-lipolic acid improved total sperm count, concentration, and motility (progressive motility) but had no effect on semen volume, sperm vitality, or morphology (16).
Folic acid and zinc
Four studies investigated the effects of folic acid and/or zinc supplements on different semen variables (Table 1). Although the main conclusions are controversial, some of the results are worth emphasizing. Whereas the intake of folic acid + zinc sulfate led to improvements in sperm concentration (17, 18) and morphology (18), isolated folic acid or zinc supplementation improved other sperm-related parameters. Specifically, improvements in sperm chromatin integrity indexes (19) or sperm concentration, sperm motility, sperm integrity membrane through the hypo-osmotic swelling test, fertilizing capacity, conception, and pregnancy (20) were reported after supplementation with zinc sulfate in infertile patients with idiopathic oligoasthenoteratozoospermia (iOAT) and asthenozoospermia, respectively. Also, an improvement in sperm morphology was demonstrated after supplementation with folic acid (5 mg/d) in subfertile healthy patients (18).
ω-3 Fatty acids
Two parallel-group RCTs (Table 1) evaluated the effect of ω-3 fatty acid supplementation on sperm parameters (21, 22).
Supplementation with DHA + EPA (990 mg/d and 135 mg/d, respectively) for 10 wk demonstrated no effect on sperm parameters but improved SDF (21). Although supplementation with higher amounts of DHA + EPA (0.72 g/d and 1.12 g/d, respectively) led to significant improvements in total sperm count and concentration, sperm motility, and morphology, it had no effect on semen volume or serum sex hormone concentrations (22).
CoQ10
In terms of intervention (3–6 mo in length and 200–300 mg of supplementation/d), the most homogeneous group of studies are those that used CoQ10 (see Table 1) (23–27). The 2 articles by Nadjarzadeh et al. (23, 25) are considered as 1 study.
Studies testing the effect of supplementation with CoQ10 for a moderate-to-short-term intervention period (≤3 mo) reported no effect on conventional sperm parameters (23, 25). On the other hand, RCTs that explored the effects after 6 mo of intervention reported improvements in classical sperm parameters such as sperm motility (26), total sperm count and concentration (24), and morphology (27). The 2 studies by Safarinejad et al. (24, 27) also described a peripheral increase in the inhibin-B hormone and a reduction in LH and FSH after CoQ10 supplementation. A reduction in acrosome-reacted spermatozoa in the ejaculate (an important parameter in the fecundation process) was also observed by Safarinejad (27) in 2009.
Carnitines
Three RCT studies with carnitines are summarized in Table 1 (28–30).
The administration of all types of isolated carnitines, such as l-acetyl carnitine (LAC), l-carnitine (LC), or complexes of both carnitines (LAC and LC), has been shown to increase sperm motility (28–30). Supplementation with between 2 and 3 g LC/d improved sperm concentration (30) and morphology (28). Finally, in the aforementioned study (28), 1 g LAC/d also improved sperm concentration, but no effect of carnitine intake on semen volume was reported in any of these studies.
Dietary supplements
Table 1 summarizes the effects of several dietary supplements on sperm parameters (31–36).
In idiopathic oligoasthenoteratozoospermic patients, improvements in semen volume; total sperm count, concentration, progressive motility, and morphology; and levels of FSH, LH, and testosterone were reported after 6 mo supplementation with 1 Flortec capsule/d (Lactobacillus paracasei B21060 5 × 109 CFUs/d + arabinogalactan 1243 mg/d + oligo-fructosaccharides 700 mg/d + l-glutamine 500 mg/d) (31).
After 3 mo supplementation with 4 g myoinositol/d, total sperm count and concentration, progressive motility, and testosterone levels increased and acrosome-reacted spermatozoa, LH, and FSH levels decreased (32).
Supplementation with Menevit, a complex enriched with many antioxidants (lycopene 6 mg/d, vitamin E 400 IU/d, vitamin C 100 mg/d, zinc 25 mg/d, selenium 26 µg/d, folate 0.5 mg/d, garlic 1 g/d, and palm oil), had no effect on any conventional parameter (36).
Nigella sativa was tested (33). After the intervention with this herb, improvements in several seminogram parameters in the ejaculate (including semen volume and pH, sperm concentration, motility, sperm morphology, and semen round cells) were reported.
One RCT that used saffron (Crocus sativus), an ancestral herbal remedy traditionally thought to improve semen parameters, as a supplement showed that consuming 60 mg of saffron/d for 26 wk had no effect on conventional sperm parameters or serum hormones (35).
To the best of our knowledge, the only food that has been tested in an RCT was walnuts (34). This study showed that consuming 75 g of raw walnuts/d in the context of a Western-style diet improved sperm motility, sperm vitality, and morphology in healthy individuals but that supplementation with walnuts had no effect on semen volume, sperm concentration, or aneuploidy index.
Quantitative analysis
The relatively high number of RCTs that have used selenium, zinc, folic acid, ω-3, CoQ10, and carnitines as supplements and the homogeneity between them led us to conduct a meta-analysis to test the effect of these supplements on various sperm outcomes.
Selenium
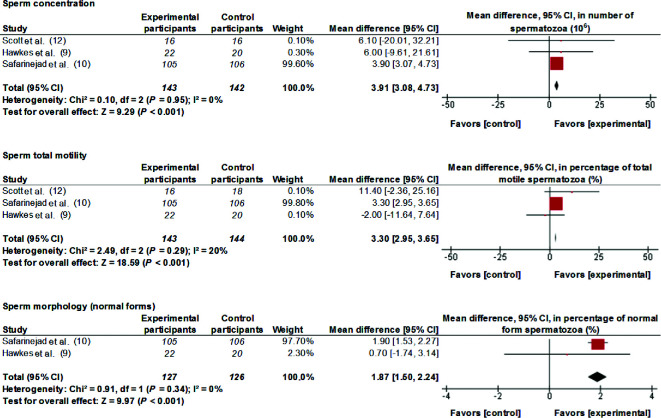
Data from 3 studies have been meta-analyzed. Supplementation of 100–300 µg Se/d for between 3 and 11 mo improved (MD; 95% CI) sperm concentration [3.91 × 106 spermatozoa (spz)/mL; 3.08, 4.73 spz/mL; P < 0.001], total motility (3.30%; 2.95%, 3.65%; P < 0.001), and morphology (1.87%; 1.50%, 2.24%; P < 0.001) (Figure 1). Interstudy heterogeneity was nonsignificant (I2 ≤ 20, P > 0.1).
FIGURE 1.
MDs and 95% CIs for the effects of selenium supplements on sperm concentration, sperm total motility, and sperm morphology. The forest plots of the studies use generic-inverse variance and a fixed-effects estimate method. The points for each study indicate the MD, the size of the boxes indicates the weight of the study, and the horizontal lines indicate the 95% CI for each study. The bold data represent the total number of participants for all studies, and the diamond represents the pooled MD. MD, mean difference.
Zinc
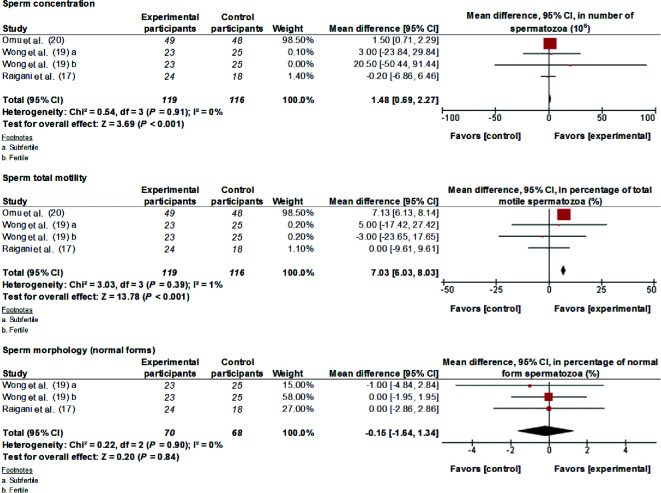
Analyzing data from 3 studies, the present study found that 66–500 mg Zn supplementation/d for 3–6 mo improved (MD; 95% CI) sperm concentration (1.48 × 106 spz/mL; 0.69, 2.27 spz/mL; P < 0.001) and total motility (7.03%; 6.03%, 8.03%; P < 0.001) (Figure 2). Interstudy heterogeneity was nonsignificant (I2 ≤ 1, P > 0.1).
FIGURE 2.
MDs and 95% CIs for the effects of zinc supplements on sperm concentration, sperm total motility, and sperm morphology. The forest plots of the studies use generic-inverse variance and a fixed-effects estimate method. The points for each study indicate the MD, the size of the boxes indicates the weight of the study, and the horizontal lines indicate the 95% CI for each study. The bold data represent the total number of participants for all studies, and the diamond represents the pooled MD. MD, mean difference.
Folic acid
Data from 2 studies with folic acid have been meta-analyzed (Supplemental Figure 2). Supplementation with 5 mg folic acid/d for 3–6 mo did not improve sperm concentration, total motility, or morphology in fertile and subfertile participants (I2 = 0, P > 0.1).
ω-3 Fatty acids
Administration of a supplement containing 1 g DHA/d and 1 g EPA/d for 10–32 wk improved (MD; 95% CI) total sperm count (18.70 × 106 spz; 16.89, 20.51 spz; P < 0.001), sperm concentration (10.98 × 106 spz/mL; 10.25, 11.72 spz/mL; P < 0.001), total motility (7.55%; 7.09%, 8.01%; P < 0.001), and morphology (0.91%; 0.69%, 1.13%; P < 0.001) (Figure 3). There was evidence of considerable and significant heterogeneity between the 2 meta-analyzed studies (I2 > 90, P < 0.001).
FIGURE 3.
MDs and 95% CIs for the effects of ω-3 fatty acid supplements on total sperm count, sperm concentration, sperm total motility, and sperm morphology. The forest plots of the studies use generic-inverse variance and a fixed-effects estimate method. The points for each study indicate the MD, the size of the boxes indicates the weight of the study, and the horizontal lines indicate the 95% CI for each study. The bold data represent the total number of participants for all studies, and the diamond represents the pooled MD. MD, mean difference.
CoQ10
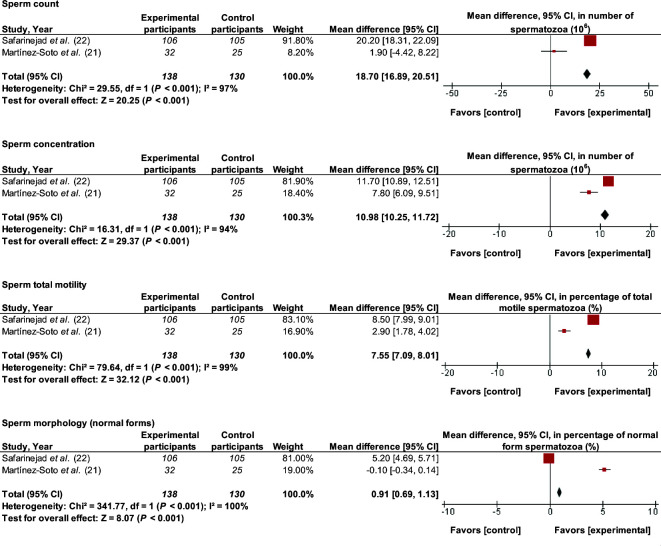
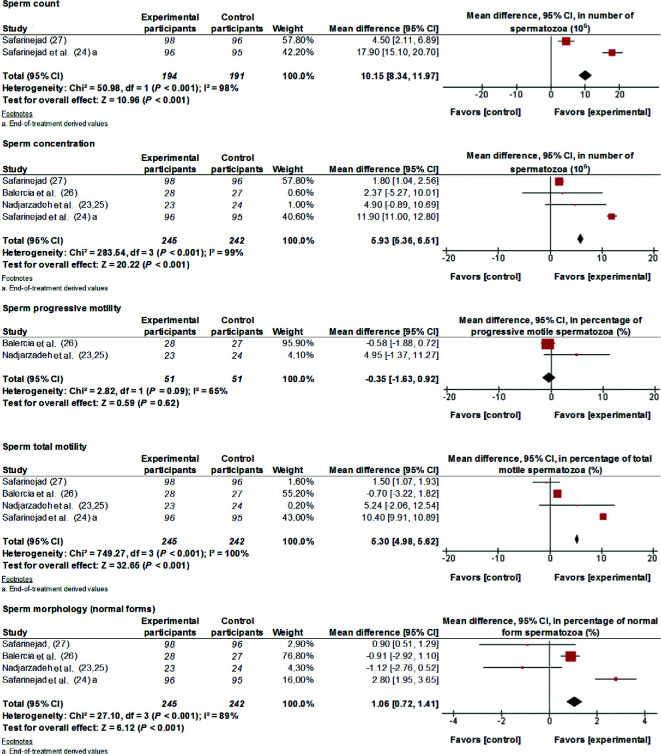
Analyzing data from 4 RCTs, the present study found that supplementation with 200–300 mg CoQ10/d for 3–6 mo improved (MD; 95% CI) total sperm count (10.15 × 106 spz; 8.34, 11.97 spz; P < 0.001), sperm concentration (5.93 × 106 spz/mL; 5.36, 6.51 spz/mL; P < 0.001), sperm total motility (5.30%; 4.98%, 5.62%; P < 0.001), and morphology (1.06%; 0.72%, 1.41%; P < 0.001) (Figure 4). The effect on sperm progressive motility had substantial interstudy heterogeneity (I2 = 65%, P = 0.09), and there was considerable interstudy heterogeneity for other sperm parameters (I2 ≥ 89%, P < 0.001). The 2 articles by Nadjarzadeh et al. (23, 25) were computed as 1 study.
FIGURE 4.
MDs and 95% CIs for the effects of coenzyme-Q10 supplements on total sperm count, sperm concentration, sperm progressive motility, sperm total motility, and sperm morphology. The forest plots of the studies use generic-inverse variance and a fixed-effects estimate method. The points for each study indicate the MD, the size of the boxes indicates the weight of the study, and the horizontal lines indicate the 95% CI for each study. The bold data represent the total number of participants for all studies, and the diamond represents the pooled MD. The 2 articles by Nadjarzadeh et al. (23, 25) are computed as 1 study. MD, mean difference.
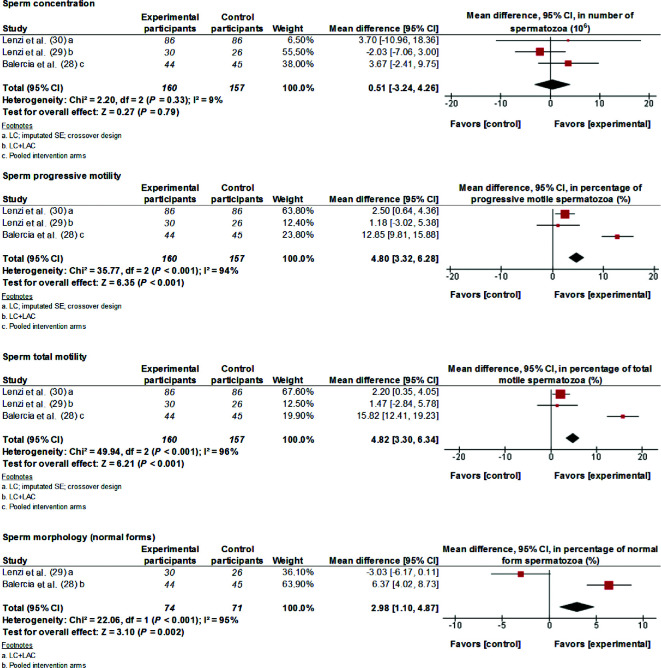
Carnitines
Data from 3 studies have been meta-analyzed. Supplementation with 3 g LC/d and 1 g LAC/d for 2–6 mo significantly improved (MD; 95% CI) sperm progressive motility (4.80%; 3.32%, 6.28%; P < 0.001), total motility (4.82%; 3.30%, 6.34%; P < 0.001), and morphology (2.98%; 1.10%, 4.87%; P = 0.002) (Figure 5). Except for sperm concentration, where homogeneity was very high (I2 = 9%, P = 0.33), there was evidence of considerable and significant heterogeneity between the studies for the motility and morphology parameters (I2 ≥ 90, P < 0.001).
FIGURE 5.
MDs and 95% CIs for the effects of carnitine (LC, LAC, or LC + LAC) supplements on sperm concentration, sperm progressive motility, sperm total motility, and sperm morphology. The forest plots of the studies use generic-inverse variance and a fixed-effects estimate method. The points for each study indicate the MD, the size of the boxes indicates the weight of the study, and the horizontal lines indicate the 95% CI for each study. The bold data represent the total number of participants for all studies, and the diamond represents the pooled MD. LAC, l-acetyl carnitine; LC, l-carnitine; MD, mean difference.
Discussion
This systematic review of RCTs provides the most wide-ranging analysis to date for the effects of nutrients, supplements, or foods on sperm quality parameters. The meta-analysis included in the review revealed a significant beneficial effect on total sperm count from supplementation with ω-3 and CoQ10; on sperm concentration from supplementation with selenium, zinc, ω-3, and CoQ10; on sperm motility from supplementation with selenium, zinc, ω-3, CoQ10, and carnitines; and on sperm morphology from supplementation with selenium, ω-3, CoQ10, and carnitines. The review suggests that some dietary supplements may help to modulate male fertility.
Different underlying mechanisms could explain these results and therefore deserve comment. Oxidative stress (OS) is identified as one of the main mediators of male infertility. It causes sperm dysfunctions and is related to increased cellular damage triggered by ROS. This occurs naturally in sperm cells because high levels of sperm motility, in the case of the hyperactivation required in zona-pellucida binding, induce ROS (37). However, high levels of ROS were also strongly correlated with sperm DNA damage and low percentages of sperm motility (38), among other sperm-related outcomes. The ROS-DNA–damage sperm motility pathway may also act in the opposite direction, i.e., DNA damage induces ROS through the H2AX (H2A histone family, member X)–Hox1 [NAD(P)H oxidase]/Rac1 (Rac Family Small GTPase 1) pathway (39). In this scenario, the equilibrium between antioxidants and ROS may be key for achieving better sperm quality (mainly in terms of sperm motility, vitality, and DNA damage). This is why most of the RCTs in the literature tested antioxidant supplements in order to balance OS. Some supplements (vitamin E and zinc) proved beneficial for increasing the live birth rate in couples with male or unexplained subfertility and some (certain carnitine supplements) proved beneficial for increasing the pregnancy rate. Other supplements had no beneficial effects in this regard (40).
The main antioxidants tested as supplements with a positive effect on sperm quality parameters were selenium and zinc. On one hand, selenium is essential for the normal spermatogenesis of mammals and plays a pivotal role in increasing glutathione peroxidase-1 expression and activity, which, in turn, destroys hydrogen peroxide molecules (41). On the other hand, zinc is also an antioxidant element with a membrane-stabilizing activity by inhibiting membrane-bound oxidative enzymes such as NAD(P) oxidase (42). A recent meta-analysis showed that the zinc content in the seminal plasma of infertile males was significantly lower than those of normal males, which indicates that zinc supplementation may significantly increase the sperm quality of infertile males (43). The present meta-analysis of RCTs in humans that used zinc and selenium as supplements reinforces this hypothesis. However, no consistent beneficial effects of other antioxidants, including folic acid, have been demonstrated.
ω-3 PUFAs are fatty acids with anti-inflammatory and antioxidant properties potentially modifying cell membrane composition and functionality. The mechanism by which ω-3 (and ω-6) PUFAs can affect spermatogenesis is their incorporation into the spermatozoa cell membrane. It has been demonstrated that the successful fertilization of spermatozoa depends on the lipid composition of the spermatozoa membrane (44). In line with this finding, the present RCT meta-analysis shows positive effects on sperm concentration after supplementation with ω-3 PUFAs. However, other RCTs conducted in large samples of participants are needed in order to definitively endorse the beneficial effect of ω-3 supplementation on sperm motility and pregnancy indicators.
CoQ10 is also an antioxidant molecule with a central role in the electron-transport system. As Balercia et al. (26) and Safarinejad (27) pointed out, CoQ10 inhibits organic peroxide formation in seminal fluid and may therefore reduce sperm-cell OS. In the last 2 decades, interest in this molecule as a supplement for treating infertile men and fecundability has grown. In a meta-analysis conducted in 2013 by Lafuente et al. (45) and in the present review, an overall improvement was shown in sperm parameters but not in live birth or pregnancy rates (45). However, high heterogeneity between the studies was reported, which indicates that more and larger studies are needed before supportive recommendations can be made.
The lack of clear effects of antioxidant supplements on sperm parameters in some of the studies included in our systematic review can be explained by the amount/dose of antioxidant used, because long-term treatments with larger amounts of phenolic or other antioxidant compounds have proven to have pro-oxidant effects. In addition, the low amount/dose of antioxidants used in these studies may have been unable to beneficially affect sperm parameters (46, 47).
Majzoub and Agarwal (48) conducted a narrative review in relation to studies that used antioxidants in iOAT and concluded that additional randomized controlled studies are required to confirm the efficacy and safety of antioxidant supplementation in the medical treatment of idiopathic male infertility, as well as the dosage required to improve semen parameters, fertilization rates, and pregnancy outcomes in iOAT.
The present study shows that carnitine supplementation also has certain beneficial effects on spermatozoa motility and morphology, although there was also considerable heterogeneity between the 3 studies meta-analyzed. LC and LAC play important roles in sperm metabolism by providing immediate available energy for use by spermatozoa, which positively affects sperm motility, the spermatogenic process, and maturation (49). In addition, carnitines are involved in the transportation of long-chain fatty acids into the mitochondrial matrix for β-oxidation and exert antioxidant activity by increasing the expression of antioxidant enzymes (28). Finally, although studies with food extracts or botanic extracts may be of potential interest for fertility modulation, these types of supplements are outside of the scope of the present review and meta-analysis, and therefore these RCTs have not been included.
Strengths and limitations
Certain limitations of the present study should be acknowledged. The search strategy was limited to the MEDLINE-PubMed database or hand-searching and did not include other databases (e.g., EMBASE). Although our search strategy included a broad number of search terms, and the use of the most relevant scientific database combined with hand-searched reference lists, it is possible (although also improbable) that not all relevant publications were identified. It is also important to point out that, because few studies were included in the meta-analysis (<10 articles meta-analyzed/group), we were unable to assess the across-studies ROB with a post hoc analysis. Also, in the present meta-analysis considerable interstudy heterogeneity was observed for most outcomes, but this could not be explored with subgroup analysis because of the few studies included. It is therefore difficult to draw strong conclusions or to make evidence-based recommendations. Unfortunately, in our analyses we did not control for the background diet and/or any dietary changes that occurred during the intervention in most of the included RCTs. Indeed, whether the changes observed in some fertility parameters can be explained by changes in the intervention diet remains to be elucidated, thus decreasing the level of scientific evidence derived from these studies. Furthermore, a potential source of bias could be derived from the fact that the studies included in this review did not report information about background medication and/or changes in medication during the trial. However, this is probably irrelevant because most of the studies were conducted in healthy young populations rarely receiving medication. The subfertile populations were heterogeneous and had different phenotypes (e.g., asthenozoospermic, oligoasthenozoospermic, or oligoasthenoteratozoospermic participants; patients with idiopathic infertility who attended infertility clinics). It is difficult, therefore, to generalize the results to other phenotypes of populations. Another limitation relates to judging the biological significance of the improvements observed in some sperm parameters because their effects on fertility need to be confirmed with other studies. Finally, in some studies, neither the manufacture reliability of the supplements used nor their bioavailability are clearly explained, making interpretation of the findings difficult.
The main strengths of the present study include its multistage design with multiauthor validation, the evaluation of ROB, and the possibility of replicating the systematic review and meta-analysis with the same system. Finally, the age range of the populations studied is quite low (18–52 y) and corresponds to the main male reproductive age.
Conclusions
This systematic review and meta-analysis of RCTs provides the most wide-ranging analysis to date of the effects of nutrients, supplements, and food on sperm quality parameters. The present study concludes that diet supplementation with certain antioxidants, especially selenium, zinc, ω-3 fatty acids, CoQ10, and carnitines, and certain foods rich in these supplements can beneficially modulate sperm quality parameters and affect male fertility. The small number of studies that have tested similar supplements, the small sample sizes included in those studies, and the high degree of interstudy heterogeneity across outcomes mean that further research may lead to a change in the effect estimates outlined in this meta-analysis. More RCTs with larger samples and clear inclusion/exclusion criteria are needed in future to test how these types of supplements affect not only sperm parameters but also fecundability.
Supplementary Material
Acknowledgments
The authors’ responsibilities were as follows—AS-H and JS-S: initiated the idea of this review and designed it; AS-H: collected the data, performed the meta-analysis, and wrote the manuscript; AS-H and NR-E: selected the data and assessed the articles; NR-E, NB-T, BV, and MB: critically reviewed the article for important intellectual content; NB-T: reviewed the meta-analysis process; JS-S: assessed the articles and helped to draft and critically review the article for important intellectual content; and all authors: read and approved the final manuscript.
Notes
Supported by the Plan Nacional de Investigación Científica, Desarrollo e Innovación Tecnológica, the Instituto de Salud Carlos III—Fondo de Investigación Sanitaria grant PI12/0153, and the Fondo Europeo de Desarrollo Regional (FEDER).
Author disclosures: AS-H, NR-E, NB-T, BV, MB, and JS-S, no conflicts of interest.
Supplemental Figures 1 and 2 and Supplemental Appendix 1 are available from the “Supplementary data” link in the online posting of the article and from the same link in the online table of contents at https://academic.oup.com/advances/.
Consorcio CIBER, M.P., CIBERobn is an initiative of the Instituto de Salud Carlos III.
Abbreviations used:
- CoQ10
coenzyme Q10
- FSH
follicle-stimulating hormone
- iOAT
idiopathic oligoasthenoteratozoospermia
- LAC
l-acetyl carnitine
- LC
l-carnitine
- LH
luteinizing hormone
- MD
mean difference
- OS
oxidative stress
- RCT
randomized clinical trial
- ROB
risk of bias
- ROS
reactive oxygen species
- spz
spermatozoa.
References
- 1. World Health Organization WHO laboratory manual for the examination and processing of human semen. Geneva: World Health Organization; 2010. [Google Scholar]
- 2. Levine H, Jørgensen N, Martino-Andrade A, Mendiola J, Weksler-Derri D, Mindlis I, Pinotti R, Swan SH. Temporal trends in sperm count: a systematic review and meta-regression analysis. Hum Reprod Update 2017;23:646–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Salas-Huetos A, Bulló M, Salas-Salvadó J. Dietary patterns, foods and nutrients in male fertility parameters and fecundability: a systematic review of observational studies. Hum Reprod Update 2017;23:371–89. [DOI] [PubMed] [Google Scholar]
- 4. Giahi L, Mohammadmoradi S, Javidan A, Sadeghi MR. Nutritional modifications in male infertility: a systematic review covering 2 decades. Nutr Rev 2016;74:118–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Goetzsche PC, Ioannidis JP, Clarke M, Devereaux P, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol 2009;62:e1–34. [DOI] [PubMed] [Google Scholar]
- 6. Moher D, Liberati A, Tetzlaff J, Altman DG, Altman D, Antes G, Atkins D, Barbour V, Barrowman N, Berlin JA, et al. . Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 2009;6:e1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. O'Connor D, Green S, Higgins JP. Cochrane handbook for systematic reviews of interventions. Version 5 Hoboken, NJ: Wiley-Blackwell; 2008. 674 p. [Google Scholar]
- 8. Elbourne DR, Altman DG, Higgins PT, Curtin F, Worthington V, Vail A. Meta-analyses involving cross-over trials: methodological issues. Int J Epidemiol 2002;31:140–9. [DOI] [PubMed] [Google Scholar]
- 9. Hawkes WC, Alkan Z, Wong K. Selenium supplementation does not affect testicular selenium status or semen quality in North American men. J Androl 2009;30:525–33. [DOI] [PubMed] [Google Scholar]
- 10. Safarinejad MR, Safarinejad S. Efficacy of selenium and/or N-acetyl-cysteine for improving semen parameters in infertile men: a double-blind, placebo controlled, randomized study. J Urol 2009;181:741–51. [DOI] [PubMed] [Google Scholar]
- 11. Rolf C, Cooper TG, Yeung CH, Nieschlag E. Antioxidant treatment of patients with asthenozoospermia or moderate oligoasthenozoospermia with high-dose vitamin C and vitamin E: a randomized, placebo-controlled, double-blind study. Hum Reprod 1999;14:1028–33. [DOI] [PubMed] [Google Scholar]
- 12. Scott R, MacPherson A, Yates RW, Hussain B, Dixon J. The effect of oral selenium supplementation on human sperm motility. Br J Urol 1998;82:76–80. [DOI] [PubMed] [Google Scholar]
- 13. Kessopoulou E, Powers HJ, Sharma KK, Pearson MJ, Russell JM, Cooke ID, Barratt CLRR. A double-blind randomized placebo cross-over controlled trial using the antioxidant vitamin E to treat reactive oxygen species associated male infertility. Fertil Steril 1995;64:825–31. [DOI] [PubMed] [Google Scholar]
- 14. Suleiman S, Eamin Ali M, Zaki Z, El-Malik E, Nasr M. Lipid peroxidation and human sperm motility: protective role of vitamin E. J Androl 1996;17:530–7. [PubMed] [Google Scholar]
- 15. Greco E, Iacobelli M, Rienzi L, Ubaldi F, Ferrero S, Tesarik J. Reduction of the incidence of sperm DNA fragmentation by oral antioxidant treatment. J Androl 2005;26:349–53. [DOI] [PubMed] [Google Scholar]
- 16. Haghighian HK, Haidari F, Mohammadi-Asl J, Dadfar M. Randomized, triple-blind, placebo-controlled clinical trial examining the effects of alpha-lipoic acid supplement on the spermatogram and seminal oxidative stress in infertile men. Fertil Steril 2015;104:318–24. [DOI] [PubMed] [Google Scholar]
- 17. Ebisch IMW, Pierik FH, De Jong FH, Thomas CMG, Steegers-Theunissen RPM. Does folic acid and zinc sulphate intervention affect endocrine parameters and sperm characteristics in men? Int J Androl 2006;29:339–45. [DOI] [PubMed] [Google Scholar]
- 18. Wong WWY, Merkus HHMW, Thomas CM, Menkveld R, Zielhuis GA, Steegers-Theunissen RP. Effects of folic acid and zinc sulfate on male factor subfertility: a double-blind, randomized, placebo-controlled trial. Fertil Steril 2002;77:491–8. [DOI] [PubMed] [Google Scholar]
- 19. Raigani M, Yaghmaei B, Amirjannti N, Lakpour N, Akhondi MM, Zeraati H, Hajihosseinal M, Sadeghi MR. The micronutrient supplements, zinc sulphate and folic acid, did not ameliorate sperm functional parameters in oligoasthenoteratozoospermic men. Andrologia 2014;46:956–62. [DOI] [PubMed] [Google Scholar]
- 20. Omu AE, Dashti H, Al-Othman S. Treatment of asthenozoospermia with zinc sulphate: andrological, immunological and obstetric outcome. Eur J Obstet Gynecol Reprod Biol 1998;79:179–84. [DOI] [PubMed] [Google Scholar]
- 21. Martínez-Soto JC, Domingo JC, Cordobilla B, Nicolás M, Fernández L, Albero P, Gadea J, Landeras J. Dietary supplementation with docosahexaenoic acid (DHA) improves seminal antioxidant status and decreases sperm DNA fragmentation. Syst Biol Reprod Med 2016;62:387–95. [DOI] [PubMed] [Google Scholar]
- 22. Safarinejad MR. Effect of omega-3 polyunsaturated fatty acid supplementation on semen profile and enzymatic anti-oxidant capacity of seminal plasma in infertile men with idiopathic oligoasthenoteratospermia: a double-blind, placebo-controlled, randomised study. Andrologia 2011;43:38–47. [DOI] [PubMed] [Google Scholar]
- 23. Nadjarzadeh A, Shidfar F, Amirjannati N, Vafa MR, Motevalian SA, Gohari MR, Nazeri Kakhki SA, Akhondi MM, Sadeghi MR. Effect of coenzyme Q10 supplementation on antioxidant enzymes activity and oxidative stress of seminal plasma: a double-blind randomised clinical trial. Andrologia 2014;46:177–83. [DOI] [PubMed] [Google Scholar]
- 24. Safarinejad MR, Safarinejad SS, Shafiei N, Safarinejad SS. Effects of the reduced form of coenzyme Q10 (ubiquinol) on semen parameters in men with idiopathic infertility: a double-blind, placebo controlled, randomized study. J Urol 2012;188:526–31. [DOI] [PubMed] [Google Scholar]
- 25. Nadjarzadeh A, Sadeghi MR, Amirjannati N, Vafa MR, Motevalian SA, Gohari MR, Akhondi MA, Yavari P, Shidfar F. Coenzyme Q10 improves seminal oxidative defense but does not affect on semen parameters in idiopathic oligoasthenoteratozoospermia: a randomized double-blind, placebo controlled trial. J Endocrinol Invest 2011;34:224–8. [DOI] [PubMed] [Google Scholar]
- 26. Balercia G, Buldreghini E, Vignini A, Tiano L, Paggi F, Amoroso S, Ricciardo-Lamonica G, Boscaro M, Lenzi A, Littarru GP. Coenzyme Q10 treatment in infertile men with idiopathic asthenozoospermia: a placebo-controlled, double-blind randomized trial. Fertil Steril 2009;91:1785–92. [DOI] [PubMed] [Google Scholar]
- 27. Safarinejad MR. Efficacy of coenzyme Q10 on semen parameters, sperm function and reproductive hormones in infertile men. J Urol 2009;182:237–48. [DOI] [PubMed] [Google Scholar]
- 28. Balercia G, Regoli F, Armeni T, Koverech A, Mantero F, Boscaro M. Placebo-controlled double-blind randomized trial on the use of L-carnitine, L-acetylcarnitine, or combined L-carnitine and L-acetylcarnitine in men with idiopathic asthenozoospermia. Fertil Steril 2005;84:662–71. [DOI] [PubMed] [Google Scholar]
- 29. Lenzi A, Sgrò P, Salacone P, Paoli D, Gilio B, Lombardo F, Santulli M, Agarwal A, Gandini L. A placebo-controlled double-blind randomized trial of the use of combined L-carnitine and L-acetyl-carnitine treatment in men with asthenozoospermia. Fertil Steril 2004;81:1578–84. [DOI] [PubMed] [Google Scholar]
- 30. Lenzi A, Lombardo F, Sgrò P, Salacone P, Caponecchia L, Dondero F, Gandini L. Use of carnitine therapy in selected cases of male factor infertility: a double-blind crossover trial. Fertil Steril 2003;79:292–300. [DOI] [PubMed] [Google Scholar]
- 31. Maretti C, Cavallini G. The association of a probiotic with a prebiotic (Flortec, Bracco) to improve the quality/quantity of spermatozoa in infertile patients with idiopathic oligoasthenoteratospermia: a pilot study. Andrology 2017;5:439–44. [DOI] [PubMed] [Google Scholar]
- 32. Calogero AE, Gullo G, La Vignera S, Condorelli RA, Vaiarelli A. Myoinositol improves sperm parameters and serum reproductive hormones in patients with idiopathic infertility: a prospective double-blind randomized placebo-controlled study. Andrology 2015;3:491–5. [DOI] [PubMed] [Google Scholar]
- 33. Kolahdooz M, Nasri S, Modarres SZ, Kianbakht S, Huseini HF. Effects of Nigella sativa L. seed oil on abnormal semen quality in infertile men: a randomized, double-blind, placebo-controlled clinical trial. Phytomedicine 2014;21:901–5. [DOI] [PubMed] [Google Scholar]
- 34. Robbins WA, Xun L, FitzGerald LZ, Esguerra S, Henning SM, Carpenter CL. Walnuts improve semen quality in men consuming a Western-style diet: randomized control dietary intervention trial. Biol Reprod 2012;87:1–8. [DOI] [PubMed] [Google Scholar]
- 35. Safarinejad MR, Shafiei N, Safarinejad S. A prospective double-blind randomized placebo-controlled study of the effect of saffron (Crocus sativus Linn.) on semen parameters and seminal plasma antioxidant capacity in infertile men with idiopathic oligoasthenoteratozoospermia. Phytother Res 2011;25:508–16. [DOI] [PubMed] [Google Scholar]
- 36. Tremellen K, Miari G, Froiland D, Thompson J. A randomised control trial examining the effect of an antioxidant (Menevit) on pregnancy outcome during IVF-ICSI treatment. Aust New Zeal J Obstet Gynaecol 2007;47:216–21. [DOI] [PubMed] [Google Scholar]
- 37. Aitken RJ, Gibb Z, Baker MA, Drevet J, Gharagozloo P. Causes and consequences of oxidative stress in spermatozoa. Reprod Fertil Dev 2016;28:1–10. [DOI] [PubMed] [Google Scholar]
- 38. Aitken RJ. Oxidative stress and the etiology of male infertility. J Assist Reprod Genet 2016;33:1691–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Kang MA, So EY, Simons AL, Spitz DR, Ouchi T. DNA damage induces reactive oxygen species generation through the H2AX-Nox1/Rac1 pathway. Cell Death Dis 2012;3:e249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Showell MG, Mackenzie-Proctor R, Brown J, Yazdani A, Stankiewicz MT, Hart RJ. Antioxidants for male subfertility. Cochrane Database Syst Rev Antioxidants 2014;1:CD007411. [DOI] [PubMed] [Google Scholar]
- 41. Schnabel R, Lubos E, Messow CM, Sinning CR, Zeller T, Wild PS, Peetz D, Handy DE, Munzel T, Loscalzo J, et al. . Selenium supplementation improves antioxidant capacity in vitro and in vivo in patients with coronary artery disease. The SElenium Therapy in Coronary Artery disease Patients (SETCAP) study. Am Heart J 2008;156:1201.e1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Prasad AS. Clinical, immunological, anti-inflammatory and antioxidant roles of zinc. Exp Gerontol 2008;43:370–7. [DOI] [PubMed] [Google Scholar]
- 43. Zhao J, Dong X, Hu X, Long Z, Wang L, Liu Q, Sun B, Wang Q, Wu Q, Li L. Zinc levels in seminal plasma and their correlation with male infertility: a systematic review and meta-analysis. Sci Rep 2016;6:e22386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Safarinejad MR, Safarinejad S. The roles of omega-3 and omega-6 fatty acids in idiopathic male infertility. Asian J Androl 2012;14:514–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Lafuente R, González-Comadrán M, Solà I, López G, Brassesco M, Carreras R, Checa MA. Coenzyme Q10 and male infertility: a meta-analysis. J Assist Reprod Genet 2013;30:1147–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Tvrdá E, Kováčik A, Tušimová E, Massányi P, Lukáč N. Resveratrol offers protection to oxidative stress induced by ferrous ascorbate in bovine spermatozoa. J Environ Sci Health A Tox Hazard Subst Environ Eng 2015;50:1440–51. [DOI] [PubMed] [Google Scholar]
- 47. Auger C, Gérain P, Laurent-Bichon F, Portet K, Bornet A, Caporiccio B, Cros G, Teissédre PL, Rouanet JM. Phenolics from commercialized grape extracts prevent early atherosclerotic lesions in hamsters by mechanisms other than antioxidant effect. J Agric Food Chem 2004;52:5297–302. [DOI] [PubMed] [Google Scholar]
- 48. Majzoub A, Agarwal A. Systematic review of antioxidant types and doses in male infertility: benefits on semen parameters, advanced sperm function, assisted reproduction and live-birth rate. Arab J Urol 2018;16:113–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Agarwal A, Said TM. Carnitines and male infertility. Reprod Biomed Online 2004;8:376–84. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.