Abstract
Background
Heart failure with preserved ejection fraction (HFpEF) is a multifactorial syndrome characterized by a limited exercising capacity. High-intensity interval training (HIIT) is an emerging strategy for exercise rehabilitation in different settings. In patients with HFpEF, HIIT subacute effects on endothelial function and blood pressure are still unknown.
Objective
To evaluate the subacute effect of one HIIT session on endothelial function and blood pressure in patients with HFpEF.
Methods
Sixteen patients with HFpEF underwent a 36-minute session of HIIT on a treadmill, alternating four minutes of high-intensity intervals with three minutes of active recovery. Brachial artery diameter, flow-mediated dilation, and blood pressure were assessed immediately before and 30 minutes after the HIIT session. In all analyses, p <0.05 was considered statistically significant.
Results
There was an increase in brachial artery diameter (pre-exercise: 3.96 ± 0.57 mm; post-exercise: 4.33 ± 0.69 mm; p < 0.01) and a decrease in systolic blood pressure (pre-exercise: 138 ± 21 mmHg; post-exercise: 125 ± 20 mmHg; p < 0.01). Flow-mediated dilation (pre-exercise: 5.91 ± 5.20%; post-exercise: 3.55 ± 6.59%; p = 0.162) and diastolic blood pressure (pre-exercise: 81 ± 11 mmHg; post-exercise: 77 ± 8 mmHg; p = 1.000) did not change significantly. There were no adverse events throughout the experiment.
Conclusions
One single HIIT session promoted an increase in brachial artery diameter and reduction in systolic blood pressure, but it did not change flow-mediated dilation and diastolic blood pressure.
Keywords: Heart Failure, Arterial Pressure, Exercise, Vasodilatation, Brachial Artery, Endothelium/function
Introduction
Heart failure with preserved ejection fraction (HFpEF) is a complex and prevalent clinical syndrome characterized by a significant limitation to exercising capacity, and pharmacological treatment has not evidenced any improvement in mortality rates in this scenario yet.1,2 Therapeutic approaches are limited and they are mainly based on symptom management and control of cardiovascular risk factors, such as high blood pressure (BP).3-5
Hypertension is associated with increased oxidative stress and vascular inflammation, closely related to endothelial dysfunction.6,7 On the other hand, attenuated endothelial function in individuals with HFpEF contributes to intolerance to exercising8-10 and it is an independent predictor of adverse cardiovascular events.11,12 As a non-pharmacological intervention, exercise training appears as a potential strategy to be included in HFpEF's therapeutic arsenal.13,14
High-intensity interval training (HIIT) has emerged as an exercise modality with a positive impact on some cardiovascular outcomes, and it is at least as effective as moderate-intensity continuous training in patients with heart failure with reduced ejection fraction.15-17 Recent meta-analyses have demonstrated that HIIT, in a long-term basis, is more effective in promoting endothelial function improvement and BP reduction in individuals with cardiovascular risk factors.18,19 In previous studies, after one single HIIT session, patients with coronary artery disease and hypertension showed increased brachial artery diameter,20,21 improved endothelial function,20 and reduced BP.21-23
It is well known that HFpEF patients have attenuated vasodilator reserve while exercising and their ventricular-arterial coupling responses are impaired.9,10,24 However, the effect of one HIIT session on endothelial function and BP in these patients is still unknown. Considering this gap in the literature, the aim of this study was to evaluate brachial artery diameter, endothelial function, and BP 30 minutes after one HIIT session in patients with HFpEF.
Methods
Study design and patients
This before-and-after (quasi-experimental) study was conducted between June 2014 and November 2015. Nineteen patients with HFpEF, according to the European Society of Cardiology criteria,25 were sequentially recruited in an outpatient cardiology clinic of a tertiary hospital in southern Brazil. Eligibility criteria were presence of signs and symptoms of heart failure, preserved ejection fraction (> 50%), diastolic dysfunction (left ventricular end-diastolic volume index < 97 mL/m2) with increased filling pressure (E/e' > 8), and in the case of E/e' < 15, at least one diagnostic criterion for HFpEF, according to the abovementioned document. Age between 40-75 years, New York Heart Association (NYHA) functional class I to III, and clinical stability under optimal drug therapy in previous 3 months, was also considered criteria for eligibility. Patients with severe lung disease, moderate-to-severe valvular disease and peripheral arterial disease were excluded. Similarly, autonomic neuropathy, unstable angina, a history of complex arrhythmias induced by stress, patients with implantable cardiac electronic devices and those with cognitive and/or limiting musculoskeletal conditions, were excluded.
Firstly, patients underwent a Doppler echocardiography with color flow mapping to confirm the diagnosis criteria for HFpEF. Then, a maximal cardiopulmonary exercise testing was performed to assess ventilatory thresholds and peak oxygen consumption, as well as heart rate response to exercise. Up to 14 days after the cardiopulmonary exercise testing, brachial artery diameter, flow-mediated dilation (FMD) and endothelium-independent dilation were assessed immediately before and 30 minutes after a HIIT session. In the same experimental session, BP and heart rate were measured at two different moments before and after exercise as described below.
Measurements and instruments
Patients' characteristics at baseline
Demographic and clinical data were collected on the first day through a questionnaire and verified in the medical records of each patient. Anthropometric data were collected at the time the questionnaire was completed.
Transthoracic echocardiogram
All echocardiographic examinations were performed using equipment Envisor C HD or HD 11 (Philips, USA) with a standard multifrequency sectorial transducer by a trained cardiologist. Images were acquired following a standardized protocol, following recommendations present in the current guidelines.25,26 Cine loops and static images of 3 consecutive beats were recorded on standard 2D, M-mode, Doppler and tissue Doppler echocardiographic views. Left ventricular ejection fraction was calculated using the Teichholz formula from the parasternal long-axis view. For patients with regional wall motion abnormalities, the Simpson rule was used to calculate the ejection fraction. Left atrium volume was measured at ventricular systole, just before mitral valve opening, and calculated from apical 4-and 2-chamber views using the biplane method of disks. Left ventricular diastolic function was evaluated with transmitral pulsed Doppler (peak E velocity, peak A velocity, E/A ratio and deceleration time) and mitral annulus tissue Doppler velocity (early diastolic velocity - e', late diastolic velocity - a').
Cardiopulmonary exercise test
The test was performed on a treadmill (General Electric T-2100, GE Healthcare, Waukesha, USA), and breath-by-breath expired gas analysis was carried out using a Cortex Metalyzer 3B system (Cortex Medical, Leipzig, Germany). Heart rate was monitored with a 12-lead electrocardiograph (Nihon Kohden Corporation, Tokyo, Japan), with electrode placement as described by Mason and Likar.27 BP was measured with a sphygmomanometer (PA 2001, P.A. MED, São Paulo, Brazil) every 3 minutes during the test and also at the physician's discretion. All tests were performed in the morning, with room temperature between 18 and 22ºC and relative humidity around 60%, and they were conducted always by the same researcher (ADS), a cardiologist with expertise in cardiopulmonary exercise testing, certified by the Department of Exercise Testing and Cardiovascular Rehabilitation of the Brazilian Society of Cardiology. An individualized ramp protocol was used as described elsewhere in this study.28 Tests were considered maximal when the respiratory quotient (R) was equal to or higher than 1.10.
Blood pressure
BP was measured with a digital device (G-Tech MA100, Shenzen, China) at four different points in time: 1) pre-assessment of endothelial function (after 15 minutes seating at rest); 2) immediately before HIIT session; 3) 5 minutes after HIIT session; 4) 30 minutes after HIIT session.
Endothelial function
Patients were instructed not to do any type of exercise, not to smoke, and not to dear or drink any caffeine or alcohol for 24 hours before the evaluation. The evaluation started after 15 minutes of seated rest in a room with temperature between 18 and 22ºC. Patients stood in the supine position with their left arm positioned comfortably. Noninvasive measurements of endothelial function were performed using a two-dimensional Philips EnVisor Ultrasound system (Philips, USA) with an electrocardiogram module and a high-frequency (7-12 MHz) vascular transducer.
An image of the brachial artery was obtained 2-5 cm from the antecubital fossa on a longitudinal plane. Artery diameter was manually measured from the anterior and posterior intimal layer. Visual inspection of single frames was performed and calipers were placed at discrete points along the long axis of the B-mode image, when means were calculated.
After measurements of brachial artery diameter were taken at baseline, a sphygmomanometer was inflated on patient's left forearm with 50 mmHg above the systolic BP, remaining there for 5 minutes. Sixty seconds after deflation of the sphygmomanometer cuff, a new image was recorded synchronized with the R wave of the electrocardiogram to identify the artery diameter, enabling FMD measurements.
After 15 minutes (for normalization), the artery diameter was measured again. Then, a dose (0.4 mg) of nitroglycerin spray was administered sublingually. After 5 minutes, another image was recorded to measure endothelium-independent dilation. These data were obtained before exercise and 30 minutes after the HIIT session.
FMD was expressed as the relative change in brachial artery diameter during the hyperemic phase, as follows: [(post-hyperemic diameter - baseline diameter) / baseline diameter] × 100.
High-intensity interval training protocol
The HIIT session was performed on a treadmill according to the protocol recommended by the European Society of Cardiology (ESC).15 The session started with an 8-minute warm-up at moderate intensity followed by four blocks of 4 minutes each at 85-95% maximal heart rate, 15 to 17 on Borg rating of perceived exertion scale,29 alternated with 3 minutes at 60-70% maximal heart rate, 11 to 13 on Borg scale. It ended with 3 minutes of cool-down at moderate intensity, totaling 36 minutes. The heart rate target zone stipulated for each block was based on the maximal heart rate reached at cardiopulmonary exercise testing and was continuously measured during training through 12-lead electrocardiographic monitoring (Nihon Kohden Corporation, Tokyo, Japan).
Statistical analysis
Data were analyzed using SPSS, version 20.0. Categorical variables are described as absolute frequencies and percentages. Continuous variables with normal distribution are described as means and standard deviations. The only variable without normal distribution (VE/VCO2 slope) was described as median and interquartile range. After meeting the assumptions of normality, the Student t-test for paired samples was used to compare means of the endothelial function variables (brachial artery diameter, FMD, and endothelium-independent dilation) pre- and post-exercise. Generalized estimating equations (GEE) were used to compare mean BP and heart rate between four different moments during the experiment. In all analyses, p <0.05 was considered statistically significant.
Results
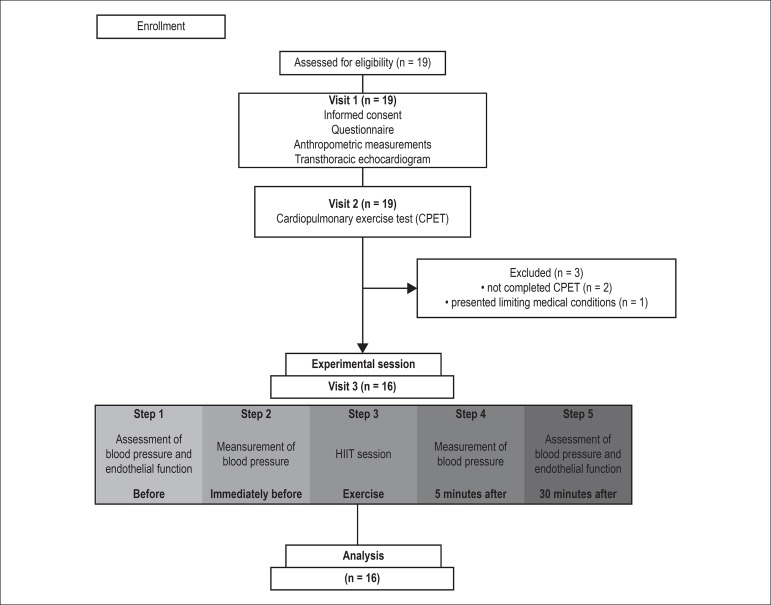
Initially nineteen patients were included in the study. After the first evaluation, two patients who did not complete the cardiopulmonary exercise testing and one who had a limiting medical condition were excluded, as shown in Figure 1.
Figure 1.
Study flow diagram.
Table 1 shows the demographic, anthropometric, and clinical characteristics of the sample.
Table 1.
Participants’ characteristics at baseline
| Characteristic | n = 16 |
|---|---|
| Female | 9 (56%) |
| Age (years) | 59 ± 7 |
| Weight (kg) | 87 ± 28 |
| Height (cm) | 159 ± 10 |
| Body mass index (kg/m2) | 34 ± 7 |
| Waist circumference (cm) | 110 ± 27 |
| Smoking | |
| Active smoker | 2 (12%) |
| Former smoker | 7 (44%) |
| NYHA functional classification | |
| II | 12 (75%) |
| III | 4 (25%) |
| Comorbidities | |
| Hypertension | 16 (100%) |
| Diabetes | 7(44%) |
| Rheumatic disease (gout) | 2 (12%) |
| Atrial fibrillation | 1 (6%) |
| CRF | 4 (25%) |
| AMI | 2 (12%) |
| Stroke | 3 (19%) |
| Medications | |
| ACEI/ARA | 16 (100%) |
| Beta-blockers | 13 (81%) |
| Diuretics | 13 (81%) |
| Calcium channel blockers | 11 (69%) |
| Statins | 10 (62%) |
| Antiplatelets | 9 (56%) |
| Vasodilators | 7 (44%) |
| Hypoglycemic drugs | 7 (44%) |
Values are described as mean ± standard deviation or absolute frequency (percentage). Former smoker: more than 1 year without smoking; NYHA: New York Heart Association; CRF: chronic renal failure; AMI: acute myocardial infarction; ACEI: angiotensin-converting enzyme inhibitors; ARA: angiotensin receptor antagonists.
All patients presented normal ejection fraction, reduced left ventricular end-diastolic volume index and increased filling pressure, as shown in table 2. However, eight patients presented 15 > E/e' > 8. Among these individuals at least one diagnostic criterion for HFpEF was confirmed. Reduced functional capacity and increased ventilatory inefficiency were identified by cardiopulmonary exercise testing. The mean peak respiratory exchange ratio > 1.1 was reach as maximality criterion as shown in table 3.
Table 2.
Echocardiographic variables
| Variables | n = 16 |
|---|---|
| LVEF (%) | 68 ± 5 |
| E/e’ | 13 ± 4 |
| LAD (cm) | 4.22 ± 0.41 |
| LVESV (ml) | 37.9 ± 9.10 |
| LVEDV (ml) | 124.41 ± 23.24 |
| LVEDVI (ml/m2) | 67.09 ± 6.35 |
| IVST (cm) | 1.15 ± 0.17 |
| PWT (cm) | 1.10 ± 0.19 |
| LVM (g) | 244.35 ± 58 |
| LVMI (g/m2) | 146.2 ± 35.84 |
| LAVI (ml/m2) | 20.81 ± 3.40 |
Values are described as mean ± standard deviation.LVEF: left ventricular ejection fraction; E/e’: early diastolic peak velocity and diastolic peak velocities of the mitral annulus ratio; LAD: left atrium diameter; LVESV: left ventricular end-systolic volume; LVEDV: left ventricular end-diastolic volume; LVEDVI: left ventricular end-diastolic volume indexed to body surface; IVST: interventricular septum thickness; PWT: posterior wall thickness; LVM: left ventricular mass; LVMI: left ventricular mass indexed by body surface; LAVI: left atrial volume indexed to body surface.
Table 3.
Cardiopulmonary exercise testing variables
| Variables | n = 16 |
|---|---|
| VO2 peak (mL.kg-1min-1) | 18.40 ± 3.16 |
| HR max. (bpm) | 125 ± 23 |
| VE/VCO2 slope | 33 ± 6 |
| PET CO2 rest (mmHg) | 33 ± 3 |
| Pulse O2 | 11.36 ± 4.45 |
| R peak | 1.16 ± 0.13 |
Values are described as mean ± standard deviation or median±interquartile range. VO2 peak: peak oxygen consumption; HR max.: maximum heart rate; VE/VCO2 slope: incline of the ventilatory equivalent of carbon dioxide; PET CO2 rest: expired pressure of carbon dioxide; O2 pulse: oxygen pulse; R peak: respiratory quotient.
All patients tolerated exercise and completed the experimental session. Exercise protocol variables are described in table 4.
Table 4.
Exercise protocol variables
| Variables | Moderate intensity | High Intensity |
|---|---|---|
| HR (bpm) | 98 ± 19 | 113 ± 24 |
| BORG | 13 ± 2 | 16 ± 2 |
| Speed (km/h) | 3 ± 0.3 | 4.9 ± 0.8 |
| Incline (%) | 0.9 ± 0.9 | 5.5 ± 1.9 |
Values are described as mean ± standard deviation. HR: heart rate; BORG: scale of perceived exertion.
One single HIIT session promoted subacute increase of 0.37 ± 0.44 mm in brachial artery diameter, as shown in Figure 2. This increase was also observed in brachial artery diameter post-hyperemia. However, when these data were used to calculate pre- and post-HIIT variation in the artery diameter, there was no difference in absolute FMD and relative FMD. Also, there was no difference in the brachial artery diameter pre-NTG (Nitrogen) and post-NTG. Similarly, there was no difference in absolute endothelium-independent dilation and relative endothelium-independent dilation after one HIIT session, as presented in table 5.
Figure 2.
Brachial artery diameter pre- and post-high-intensity interval training session. Data are expressed as mean± standard deviation. Lines represent individual values. Probability value indicates within-group significant differences.
Table 5.
Brachial artery diameters and variations pre- and post-high-intensity interval training session.
| Variables | Pre | Post | p |
|---|---|---|---|
| Brachial artery diameter (mm) | 3.96 ± 0.57 | 4.33 ± 0.69 | < 0.01 |
| Brachial artery diameter post-hyperemia (mm) | 4.19 ± 0.61 | 4.47 ± 0.66 | < 0.05 |
| Absolute FMD (mm) | 0.23 ± 0.20 | 0.13 ± 0.26 | 0.177 |
| Relative FMD (%) | 5.91± 5.20 | 3.55 ± 6.59 | 0.162 |
| Brachial artery diameter pre-NTG (mm) | 4.11 ± 0.65 | 4.16 ± 0.68 | 0.528 |
| Brachial artery diameter post-NTG (mm) | 4.57 ± 0.65 | 4.52 ± 0.64 | 0.541 |
| Absolute NTG (mm) | 0.46 ± 0.17 | 0.35 ± 0.20 | 0.106 |
| Relative NTG (%) | 11.4 ± 4.4 | 9.0 ± 5.37 | 0.117 |
Values are described as mean ± standard deviation. FMD: flow-mediated dilatation; NTG: nitroglycerin.
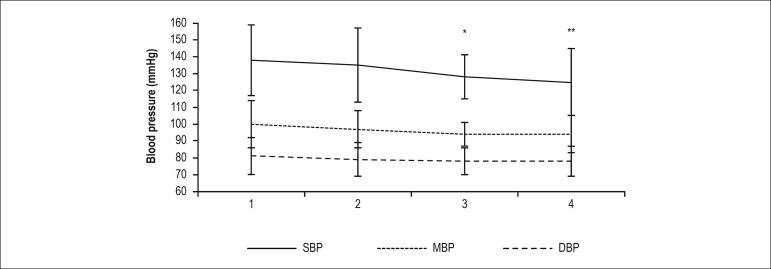
Baseline systolic and diastolic BP were 138 ± 21 mmHg and 81 ± 11 mmHg, respectively. Figure 3 shows variation in BP at four different points in time of the experiment. A significant reduction in systolic BP was observed 5 and 30 minutes after the HIIT session compared to the first measurement. There was no difference in diastolic BP and mean BP before and after the HIIT session.
Figure 3.
Variation of blood pressure pre- and post-high-intensity interval training session. Data are expressed as mean± standard deviation. Lines represent mean values:.1) pre-assessment of endothelial function; 2) immediately before HIIT session; 3) 5 minutes after HIIT session; 4) 30 minutes after HIIT session. SBP, systolic blood pressure; MBP, mean blood pressure; DBP, diastolic blood pressure. Probability value indicates within-group differences between points 3 and 1, and points 4 and 1 of SBP. *p < 0.05, **p < 0.01.
Discussion
To our knowledge, this is the first study to show that one single session of HIIT is effective in promoting a significant subacute increase in brachial artery diameter, which was accompanied by a significant reduction in systolic BP in patients with HFpEF. Borlaug et al.9 demonstrated that these individuals have global dysfunction in cardiovascular reserve, showing an impaired reduction in systemic vascular resistance and a blunted increase in blood flow while exercising. According to the authors, these phenomena are potential contributors to limited functional capacity in this situation.
Patients with HFpEF in our sample showed vasodilation after one single HIIT session suggesting that this type of exercise is a stimulus capable of promoting subacute systemic vasomotor changes, even in patients with impaired ventricular-arterial coupling9-11 and chronic vascular dysfunction.6,8,30 It is important to mention that some acute and subacute physiological responses to exercise may be clinically relevant. These responses can be superimposed after consecutive exercise sessions are carried out as a temporal summation and they may contribute to chronic adaptations of exercise training.31 Thus, successive sessions of exercise that increase blood flow, shear stress and, consequently, bioavailability of nitric oxide, may be a key mechanism for chronic adaptations in peripheral hemodynamics.32 Fu et al. found that after 12 weeks of HIIT, patients with HFpEF increased the VO2peak and improved peripheral hemodynamics, through increased blood distribution and oxygen extraction by the musculature while exercising.33
Exercise-mediated increases in shear stress have a strong and dose-dependent effect on conduit artery dilation.33 Birk et al.34 observed that vasodilation occurred in a greater extent immediately after highly intensive exercising compared to lowly intensive exercise sessions.34 However, it seems that the greater the vasodilation promoted by exercise, the lower the vasodilating response observed by occlusion immediately after the exercise session in healthy individuals.
Although there is no previous publication concerning subacute effect of an exercise session on endothelial function in patients with HFpEF, previous studies have evaluated patients with heart failure with reduced ejection fraction in a similar context.35,36 Those participants responded to a single cycling exercise session with improved forearm endothelium-dependent vasodilation (reactive hyperemia) evaluated by plethysmography up to 30 minutes after exercise.35 Currie et al.20 evaluated coronary artery disease patients after one single HIIT session and found an increase in the endothelial function after 60 minutes.20 In other experiment, the same group showed that only individuals with coronary artery disease with endothelial dysfunction presented augmentation in FMD after 15 minutes of a HIIT session.21 Interestingly, as in our experiment, in both studies the brachial artery diameter was increased.
Some evidence points out that exercising performed at submaximal intensities closer to the peak of exercise promotes a greater and longer reduction in BP after exercising than when exercising less intensively.37,38 The hypotensive effect of HIIT is already well established in the literature, but prior to this study, BP had not been evaluated in patients with HFpEF after a session of any type of exercise. In our experiment, we observed an absolute reduction of 12.7 ± 3.8 mmHg in systolic BP 30 minutes after an exercise session. On a chronic basis, this reduction may have clinical relevance, especially in the case of a syndrome whose strict control of BP pressure is crucial. Interestingly, a recent meta-analysis has demonstrated that HIIT performed at least 3 times a week for 12 weeks resulted in a significant reduction in systolic BP in overweigh/obese individuals.19
It is noteworthy that in this subgroup of individuals with HFpEF and reduced functional capacity, high-intensity exercising was well tolerated, once appropriate overload (speed and slope) was individually prescribed, always considering the target zones established based on maximal cardiopulmonary exercise test results of each individual.
Finally, in a condition characterized by exercise limitation, aerobic exercise training has a significant role and is indicated for all patients capable of performing it. In an acute and subacute setting, HIIT reduced BP and increased brachial artery diameter, suggesting that this training modality could be a beneficial alternative for individuals with HFpEF.
Limitations and future perspectives
This was a small, single-center; before-and-after study with HEpEF patients where the presence of diabetes, atherosclerosis, gout, and use of tobacco may have influenced the study outcomes. However, these characteristics represent the reality of this complex syndrome which have multiple comorbidities. We acknowledge that further studies are necessary to evaluate the effect of a HIIT session, especially after one hour, as well as the long-term efficacy of this exercise strategy as part of a cardiovascular rehabilitation program for these patients. Finally, the presence of a control group of matched individuals without HFpEF could help establishing which responses can be attributed to the syndrome under study. Likewise, comparing a HIIT session with a continuous moderate-intensity training session could help establishing the differences in hemodynamic response among these different exercise protocols.
Conclusion
One single HIIT session promoted an increase in brachial artery diameter and a reduction in systolic BP, and did not change FMD and diastolic BP 30 minutes after the exercise session.
Footnotes
Sources of Funding
Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) e Fundo de Incentivo à Pesquisa e Eventos (FIPE) do Hospital de Clínicas de Porto Alegre (HCPA).
Study Association
This article is part of the thesis of master submitted by Juliana Beust de Lima, from Universidade Federal do Rio Grande do Sul.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of the Clínicas de Porto Alegre under the protocol number 130471. All the procedures in this study were in accordance with the 1975 Helsinki Declaration, updated in 2013. Informed consent was obtained from all participants included in the study.
Author contributions
Conception and design of the research: Lima JB, Silveira AD, Zanini M, Nery RM, Stein R; Acquisition of data: Lima JB, Silveira AD, Saffi MAL, Menezes MG, Piardi DS, Ramm LDCR; Analysis and interpretation of the data: Lima JB, Saffi MAL, Menezes MG, Piardi DS, Ramm LDCR, Stein R; Statistical analysis: Lima JB; Writing of the manuscript: Lima JB, Silveira AD, Stein R; Critical revision of the manuscript for intellectual content: Lima JB, Silveira AD, Saffi MAL, Zanini M, Nery RM, Stein R.
Potential Conflict of Interest
No potential conflict of interest relevant to this article was reported.
References
- 1.Owan TE, Hodge DO, Herges RM, Jacobsen SJ, Roger VL, Redfield MM. Trends in prevalence and outcome of heart failure with preserved ejection fraction. N Engl J Med. 2006;355(3):251–259. doi: 10.1056/NEJMoa052256. [DOI] [PubMed] [Google Scholar]
- 2.Redfield MM. Heart failure with preserved ejection fraction. N Engl J Med. 2016;375(19):1868–1877. doi: 10.1056/NEJMcp1511175. [DOI] [PubMed] [Google Scholar]
- 3.McMurray JJ, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K, et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2012;33(14):1787–1847. doi: 10.1093/eurheartj/ehs104. [DOI] [PubMed] [Google Scholar]
- 4.McMurray JJ, Carson PE, Komajda M, McKelvie R, Zile MR, Ptaszynska A, et al. Heart failure with preserved ejection fraction: clinical characteristics of 4133 patients enrolled in the I-PRESERVE trial. Eur J Heart Fail. 2008;10(2):149–156. doi: 10.1016/j.ejheart.2007.12.010. [DOI] [PubMed] [Google Scholar]
- 5.WRITING COMMITTEE MEMBERS. Yancy CW, Jessup M, Bozkurt B, Butler J, Casey Jr DE, Drazner MH, et al. 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2013;128(16):e240–e327. doi: 10.1161/CIR.0b013e31829e8776. [DOI] [PubMed] [Google Scholar]
- 6.Paulus WJ, Tschöpe C. A novel paradigm for heart failure with preserved ejection fraction. J Am Coll Cardiol. 2013;62(4):263–271. doi: 10.1016/j.jacc.2013.02.092. [DOI] [PubMed] [Google Scholar]
- 7.Lim SL, Lam SPC. Breakthrough in heart failure with preserved ejection fraction: are we there yet? Korean J Intern Med. 2016;31(1):1–14. doi: 10.3904/kjim.2016.31.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Haykowsky MJ, Kitzman DW. Exercise physiology in heart failure and preserved ejection fraction. Heart Fail Clin. 2014;10(3):445–452. doi: 10.1016/j.hfc.2014.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Borlaug BA, Olson TP, Lam CS, Flood KS, Lerman A, Johnson BD, et al. Global cardiovascular reserve dysfunction in heart failure with preserved ejection fraction. J Am Coll Cardiol. 2010;56(11):845–854. doi: 10.1016/j.jacc.2010.03.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Borlaug BA, Melenovsky V, Russell SD, Kessler K, Pacak K, Becker LC, et al. Impaired chronotropic and vasodilator reserves limit exercise capacity in patients with heart failure and a preserved ejection fraction. Circulation. 2006;114(20):2138–2147. doi: 10.1161/CIRCULATIONAHA.106.632745. [DOI] [PubMed] [Google Scholar]
- 11.Akiyama E, Sugiyama S, Matsuzawa Y, Konishi M, Suzuki H, Nozaki T, et al. Incremental prognostic significance of peripheral endothelial dysfunction in patients with heart failure with normal left ventricular ejection fraction. J Am Coll Cardiol. 2012;60(18):1778–1786. doi: 10.1016/j.jacc.2012.07.036. [DOI] [PubMed] [Google Scholar]
- 12.Matsue Y, Suzuki M, Nagahori W, Ohno M, Matsumura A, Hashimoto Y, et al. Endothelial dysfunction measured by peripheral arterial tonometry predicts prognosis in patients with heart failure with preserved ejection fraction. Int J Cardiol. 2013;168(1):36–40. doi: 10.1016/j.ijcard.2012.09.021. [DOI] [PubMed] [Google Scholar]
- 13.Pandey A, Parashar A, Kumbhani JD, Agarwal S, Garg J, Kitzman D, et al. Exercise training in patients with heart failure and preserved ejection fraction: meta-analysis of randomized control trials. Circ Heart Fail. 2015;8(1):33–40. doi: 10.1161/CIRCHEARTFAILURE.114.001615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dieberg G, Ismail H, Giallauria F, Smart NA. Clinical outcomes and cardiovascular responses to exercise training in heart failure patients with preserved ejection fraction: a systematic review and meta-analysis. J Appl Physiol. 2015;119(6):726–733. doi: 10.1152/japplphysiol.00904.2014. [DOI] [PubMed] [Google Scholar]
- 15.Mezzani A, Hamm LF, Jones AM, McBride PE, Moholdt T, Stone JA, et al. Aerobic exercise intensity assessment and prescription in cardiac rehabilitation: a JOINT POSITION STATEMENT OF THE EUROPEAN ASSOCIATION FOR CARDIOVASCULAR PREVENTION AND REHABILITATION, THE AMERICAN ASSOCIATION OF CARDIOVASCULAR AND PULMONARY REHABILITATION, AND THE CANADIAN ASSOCIATION OF CARDIAC REHABILITATION. J Cardiopulm Rehabil Prev. 2012;32(6):327–350. doi: 10.1097/HCR.0b013e3182757050. [DOI] [PubMed] [Google Scholar]
- 16.Wisløff U, Støylen A, Loennechen JP, Bruvold M, Rognmo Ø, Haram PM, et al. Superior cardiovascular effect of aerobic interval training versus moderate continuous training in heart failure patients: a randomized study. Circulation. 2007;115(24):3086–3094. doi: 10.1161/CIRCULATIONAHA.106.675041. [DOI] [PubMed] [Google Scholar]
- 17.Ellingsen Ø, Halle M, Conraads V, Støylen A, Dalen H, Delagardelle C, et al. High-intensity interval training in patients with Heart Failure With Reduced Ejection Fraction. Circulation. 2017;135(9):839–849. doi: 10.1161/CIRCULATIONAHA.116.022924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ramos JS, Dalleck LC, Tjonna AE, Beetham KS, Coombes JS. The impact of high-intensity interval training versus moderate-intensity continuous training on vascular function: a systematic review and meta-analysis. Sports Med. 2015;45(5):679–692. doi: 10.1007/s40279-015-0321-z. [DOI] [PubMed] [Google Scholar]
- 19.Batacan RB, Duncan MJ, Dalbo VJ, Tucker PS, Fenning AS. Effects of high-intensity interval training on cardiometabolic health: a systematic review and meta-analysis of intervention studies. Br J Sports Med. 2017;51(6):494–503. doi: 10.1136/bjsports-2015-095841. [DOI] [PubMed] [Google Scholar]
- 20.Currie KD, Mckelvie RS, Macdonald MJ. Flow-Mediated Dilation Is Acutely Improved after High-Intensity Interval Exercise. Med Sci Sports Exerc. 2012;44(11):2057–2064. doi: 10.1249/MSS.0b013e318260ff92. [DOI] [PubMed] [Google Scholar]
- 21.Currie KD, McKelvie RS, MacDonald MJ. Brachial artery endothelial responses during early recovery from an exercise bout in patients with coronary artery disease. BioMed Res Int. 2014;2014:591918–591918. doi: 10.1155/2014/591918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ciolac EG, Guimarães GV, D'Àvila VM, Bortolotto LA, Doria EL, Bocchi EA. Acute effects of continuous and interval aerobic exercise on 24-h ambulatory blood pressure in long-term treated hypertensive patients. Int J Cardiol. 2009;133(3):381–387. doi: 10.1016/j.ijcard.2008.02.005. [DOI] [PubMed] [Google Scholar]
- 23.Carvalho RST de, Pires CMR, Junqueira GC, Freitas D, Marchi-Alves LM. Hypotensive response magnitude and duration in hypertensives: continuous and interval exercise. Arq Bras Cardiol. 2014;104(3):234–241. doi: 10.5935/abc.20140193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kawaguchi M, Hay I, Fetics B, Kass DA. Combined ventricular systolic and arterial stiffening in patients with heart failure and preserved ejection fraction implications for systolic and diastolic reserve limitations. Circulation. 2003;107(5):714–720. doi: 10.1161/01.cir.0000048123.22359.a0. [DOI] [PubMed] [Google Scholar]
- 25.Paulus WJ, Tschöpe C, Sanderson JE, Rusconi C, Flachskampf FA, Rademakers FE, et al. How to diagnose diastolic heart failure: a consensus statement on the diagnosis of heart failure with normal left ventricular ejection fraction by the Heart Failure and Echocardiography Associations of the European Society of Cardiology. Eur Heart J. 2007;28(20):2539–2550. doi: 10.1093/eurheartj/ehm037. [DOI] [PubMed] [Google Scholar]
- 26.Cheitlin MD, Armstrong WF, Aurigemma GP, Beller GA, Bierman FZ, Davis JL, et al. ACC/AHA/ASE 2003 Guideline Update for the Clinical Application of Echocardiography: Summary Article: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/ASE Committee to Update the 1997 Guidelines for the Clinical Application of Echocardiography) Circulation. 2003;108(9):1146–1162. doi: 10.1161/01.CIR.0000073597.57414.A9. [DOI] [PubMed] [Google Scholar]
- 27.Mason RE, Likar I. A new system of multiple-lead exercise electrocardiography. Am Heart J. 1966;71(2):196–205. doi: 10.1016/0002-8703(66)90182-7. [DOI] [PubMed] [Google Scholar]
- 28.Nery RM, Zanini M, de Lima JB, Bühler RP, da Silveira AD, Stein R. Tai Chi Chuan improves functional capacity after myocardial infarction: A randomized clinical trial. Am Heart J. 2015;169(6):854–860. doi: 10.1016/j.ahj.2015.01.017. [DOI] [PubMed] [Google Scholar]
- 29.Borg GA. Psychophysical bases of perceived exertion. Med Sci Sports Exerc. 1982;14(5):377–381. [PubMed] [Google Scholar]
- 30.Maréchaux S, Samson R, van Belle E, Breyne J, de Monte J, Dédrie C, et al. Vascular and microvascular endothelial function in heart failure with preserved ejection fraction. J Card Fail. 2015;22(1):3–11. doi: 10.1016/j.cardfail.2015.09.003. [DOI] [PubMed] [Google Scholar]
- 31.da Nobrega AC. The subacute effects of exercise: concept, characteristics. Exerc Sport Sci Rev. 2005;33(2):84–87. doi: 10.1097/00003677-200504000-00005. [DOI] [PubMed] [Google Scholar]
- 32.Dawson EA, Green DJ, Timothy Cable N, Thijssen DH. Effects of acute exercise on flow-mediated dilatation in healthy humans. J Appl Physiol. 2013;115(11):1589–1598. doi: 10.1152/japplphysiol.00450.2013. [DOI] [PubMed] [Google Scholar]
- 33.Fu TC, Yang NI, Wang CH, Cherng WJ, Chou SL, Pan TL, et al. Aerobic interval training elicits different hemodynamic adaptations between heart failure patients with preserved and reduced ejection fraction. Am J Phys Med Rehabil. 2016;95(1):15–27. doi: 10.1097/PHM.0000000000000312. [DOI] [PubMed] [Google Scholar]
- 34.Birk GK, Dawson EA, Batterham AM, Atkinson G, Cable T, Thijssen DH, et al. Effects of exercise intensity on flow mediated dilation in healthy humans. Int J Sports Med. 2012;34(5):409–414. doi: 10.1055/s-0032-1323829. [DOI] [PubMed] [Google Scholar]
- 35.Umpierre D, Stein R, Vieira PJ, Ribeiro JP. Blunted vascular responses but preserved endothelial vasodilation after submaximal exercise in chronic heart failure. Eur J Cardiovasc Prev Rehabil. 2009;16(1):53–59. doi: 10.1097/HJR.0b013e32831c8489. [DOI] [PubMed] [Google Scholar]
- 36.Guindani G, Umpierre D, Grigoletti SS, Vaz M, Stein R, Ribeiro JP. Blunted local but preserved remote vascular responses after resistance exercise in chronic heart failure. Eur J Prev Cardiol. 2012;19(5):972–982. doi: 10.1177/1741826711418931. [DOI] [PubMed] [Google Scholar]
- 37.Forjaz CL, Cardoso Jr CG, Rezk CC, Santaella DF, Tinucci T. Postexercise hypotension and hemodynamics: the role of exercise intensity. J Sports Med Phys Fitness. 2004;44(1):54–62. [PubMed] [Google Scholar]
- 38.Quinn TJ. Twenty-four hour, ambulatory blood pressure responses following acute exercise: impact of exercise intensity. J Hum Hypertens. 2000;14(9):547–553. doi: 10.1038/sj.jhh.1001106. [DOI] [PubMed] [Google Scholar]