Abstract
Background:
In many cases of breast cancer, the aberrant methylation of TP53 gene leads to uncontrolled cell proliferation and apoptosis inhibition. Moreover, expression of oncogenes which are under the control of P53 protein could be altered. Survivin as a conspicuous example of this category plays important roles in tumorigenesis, drug resistance and apoptosis inhibition. The present study was done to reveal the effects of Scrophularia atropatana extract on epigenetic situation of TP53 gene promoter and the expression levels of anti-apoptotic gene, survivin and its potential for production of cancer epi-drugs.
Methods:
Cytotoxic effect of dichloromethane extracts of Scrophularia plant on MCF-7 cell line was assessed in our previous study. Cell death ELISA (enzyme-linked immunosorbent assay) and TUNEL (Terminal deoxynucleotidyl transferase dUTP nick end labeling) tests were used to investigate the occurrence of apoptosis in the treated cells. Methylation Specific PCR (MSP) was employed to assess the changes in methylation status of the TP53 gene promoter. Furthermore, quantitative real time PCR was utilized to evaluate the resulting changes in TP53 and survivin genes expression.
Results:
Cell death ELISA and TUNEL assays confirmed the occurrence of apoptosis. MSP test revealed a significant change in the methylation status of TP53 promoter. QRT-PCR showed an increased TP53 gene expression in the treated cells while a significant decrease in survivin mRNA was evident.
Conclusions:
According to the outcomes, dichloromethane extract of S. atropatana returned the TP53 gene promoter hypermethylation to normal state. This plant could be a promising source for production of epi-drugs due to its apoptotic effects and reversal of TP53 epigenetic alterations.
Keywords: BIRC5, Epigenetics, P53, Scrophularia atropatana, survivin
Introduction
Despite rapid progress in science, cancer remains one of the greatest challenges to humanity. Based on statistics, almost half of all deaths in middle age are due to cancer, the incidence of which depends on factors such as genetic background (Karim-Kos et al., 2008). Meanwhile, breast cancer is responsible for a substantial part of cancer occurrences. Currently, surgery, radiotherapy and chemotherapy are widely used for treatment of this kind of cancer (Kirkwood et al., 2012). Nevertheless, due to limited effects of the present methods or their side effects, search for more effective and harmless treatments is a prominent priority. Regardless of the mentioned limitations, resistance to chemotherapy occurs in a large number of cancer patients (Gottesman et al., 2002). Logically, successful methods should be referred to the strategies which could return the factors causing tumorigenesis to their normal state.
One of the main proteins involved in cancer prevention is P53, which is encoded by the TP53 gene (Surget et al., 2014). Abnormality in the structure or function of this gene has been reported in many cancers. In addition to the direct effect on the inhibition of tumorigenesis, this protein can also neutralize the effects of anti-apoptotic proteins (di Pietro et al., 2012). One of these anti-apoptotic proteins is survivin, which prevents the programmed cell death by inhibiting caspases (Karami et al., 2013). This makes P53 as a suitable target for cancer treatment.
Epigenetic mechanisms like DNA methylation are considered as important approaches for regulation of gene expression in cells and tissues (Meng et al., 2011). Although changes in gene methylation patterns do not result in the DNA sequence alteration, they could largely affect the gene expression levels (Ghavifekr Fakhr et al., 2013). Change in the status of DNA methylation, particularly in the gene promoter area, is occurred as hyper or hypo methylation (Richards and Elgin, 2002). A decrease in P53 expression by epigenetic mechanisms, particularly DNA methylation, has been reported in 50% of human cancers (Lengauer and Issa, 1998). Fortunately, unlike genetic changes that are almost irreversible, epigenetic abnormalities are potentially rectifiable (Cai et al., 2011). This feature makes an incentive for the production and use of epi-drugs to return the malignant state to normal.
In the previous studies, we analyzed the cytotoxic effects of the extracts from Scrophularia plants on MCF-7 breast cancer cells (Orangi et al., 2016). In the present study, cell death ELISA and TUNEL (Terminal deoxynucleotidyl transferase dUTP nick end labeling) assays were performed to confirm the occurrence of apoptosis in MCF-7 cells treated by SADCM. Furthermore, after exposure to the mentioned extract, methylation pattern of TP53 gene was assessed by MSP test. Finally, changes in TP53 and survivin genes expression were evaluated by quantitative real time PCR.
Materials and Methods
Extract Preparation
S. atropatana plant was collected at the end of the growing season from East Azerbaijan province- Iran. The stems and leaves were thoroughly washed with distilled water and dried at room temperature. The extraction of dry powder was done using DCM (dichloromethane) in a Soxhlet apparatus. The resulting extracts were completely evaporated using a rotary machine at 45°C and low pressure and were stored in refrigerator. To provide the desired concentration from the plant extracts for the MCF-7 cells treatment, 0.02 g of extract was dissolved in 100 ml DMSO (Dimethyl sulfoxide) and diluted with RPMI-1640 medium. The solution was sterilized using Millipore syringe filters (0.2 μm) and stored in the refrigerator.
Cell culture
The MCF-7 breast adenocarcinoma cell line from human origin was purchased from Pasteur Institute of Iran. Cells were cultured in RPMI– 1640 medium (10.39 g/liter, Sigma) supplemented with 10% Fetal Bovine Serum (Gibco), penicillin (100U/ml) and streptomycin (100 µg/ml) (both from Sigma). The cells were then incubated at 37°C and 5% CO2.
TUNEL assay
TUNEL kit (Roche Diagnostics, Germany) was used for detection of DNA fragmentation, a prominent hallmark of apoptosis, in cells treated with SADCM.
103 of MCF-7 cells were seeded in 96 well culture plates in triplicates. 24 h later, the cells were treated with 200 and 400 mg per ml concentrations of SADCM. IC50, the concentration that gives growth inhibition by 50% was determined in previous study which its value was 200 mg per ml. The cells treated with DMSO were considered as negative control. After 24 h of treatment, TUNEL assay was carried out according to the protocol recommended by manufacturer. In summary, cells were fixed for one hour at room temperature in a solution containing 4% (w/v) of paraformaldehyde in PBS (pH 7.4). The cells then were washed 2 times with PBS (Phosphate buffered saline) and incubated with blocking solution for 10 minutes at room temperature. Subsequently, the cells were washed again with PBS buffer and incubated on ice with permeabilisation solution for 2 minutes. After washing with PBS, 50 microliters of TUNEL reaction mixture containing the TdT enzyme was added to the wells and the cells were incubated for one hour at 37°C. In this stage, the TdT enzyme adds labeled nucleotides to the 3’ end of broken DNA strands. After three times of washing with PBS, cells were incubated with 50 μl of converter-POD sterptavidin HRP (Horseradish peroxidase) solution for 30 minutes in the dark. After washing, the cells were incubated with DAB (Sigma) and analyzed by an inverted light microscope.
Cell Death Detection ELISA
A number of 104 viable cells were cultured in 96 well culture plates in triplicates. 24 h after treatment with IC50 concentrations of SADCM, the cells were assessed for apoptosis and necrosis using cell death detection ELISA kit (Roche Diagnostics GmbH, Germany). The procedure was performed according to the manufacturer’s recommendations. In brief, cells lysates (for apoptosis detection) and culture supernatants (for detection of necrosis) were incubated in a streptavidin coated microtiter plate. The simultaneous attaching of both “biotin labeled anti histone” and “anti- DNA- POD” antibodies was utilized for quantification of histone associated DNA in mono and oligonucleosome forms. After adding the appropriate substrate and color development, 100 μl of stop solution was added to each well and the absorbance were measured spectrophotometrically using an ELISA plate reader at 405 nm. The cells treated with DMSO were considered as negative control in this test.
Methylation specific PCR
MSP was employed to investigate the possible impacts of SADCM on methylation status in CpG islands of TP53 gene.
The TP53 gene sequence was obtained from NCBI databases and CpG islands in the upstream region and within the gene were analyzed using the UCSC database. Predicting the occurrence of likely promoter regions in the selected CpG islands was done using online promoter prediction software (http://rulai.cshl.edu/cgi-bin/CSHLmpd2/promExtract.pl?species=Human.) and according to the literature (Almeida et al., 2009). The required primers, including methylated (M) and unmethylated (U), were designed to assess CpG islands using MethPrimer software (http://www.urogene.org/methprimer/). Subsequently, the designated primers were blasted using the online Methblast software (http://medgen.ugent.be/methBLAST/).
DNA Extraction from MCF-7 cells was carried out using the DNeasyTM extraction kit (69581, Qiagen) based on the recommended protocol. The extracted DNA was analyzed by Eppendorf BioPhotometer spectrophotometre in 260 nm and agarose gel electrophoresis. The extracted DNA was treated with the sodium sulfite (SBS). In this process, all unmethylated cytosines were converted to uracils while methylated cytosine remained intact. For this purpose, 1 µg DNA (in a 10 microliters volume) was added to 40 microliters of nuclease free water. Then, 5.5 microliters of NaOH (2M) was added and incubated for 20 minutes at 37° C. Subsequently, 30 microliters of freshly prepared 10 mM hydroquinone and 520 microliters of sodium bisulfite (freshly prepared) were added to the reaction tube. Finally, the solution was covered with mineral oil and incubated for 16 hours at 50°C. In order to perform desulfonation, 22 microliters of 3M sodium hydroxide was added and incubated for 5 minutes at room temperature. DNA was then precipitated using ethanol- ammonium acetate method and dissolved in 30 microliters of nuclease free water.
For each studied sample, two PCR reactions, one with M primer and the other with U primer, were performed. Amplification with M primers was considered as an indicator of methylation in CpG sites within the primer sequences and PCR products resulted from U primers indicated the absence of methylation. Furthermore, amplification with both M and U primers was regarded as partial methylation. Primer sequences for Methylated-and unmethylated MSP are included in Table 1. List of ingredients used for methylated and un-methylated MSP are also provided in Table 2. Thermo cycling comprised of: one cycle of initial denaturation at 94°C for 5 min and 35 cycles of 94°C for 30 sec, 52°C for 40 sec and 72 for 40 sec. A final extension step at 72°C was also considered for 5 min. After performing PCR, electrophoresis on polyacrylamide gel with silver nitrate staining was used for detection of the amplicons.
Table 1.
Features of the Primers which Were Used in This Study
| Test type | Primers | Sequence (5’ to 3’) | Amplicon size (bp) |
|---|---|---|---|
| Methylation Specific PCR | P53 M-F | CGT CGT ATT TCG GAT TAG ATT TC | 166 |
| P53 M-R | AAA AAA ACG TAA ACG CTT CTC CG | ||
| P53 U-F | GGT TGT TGT ATT TTG GAT TAG ATT TT | 170 | |
| P53 U-R | AAA AAA AAC ATA AAC ACT TCT CAC C | ||
| qRT-PCR | Survivin-F | GGACCACCGCATCTCTACAT | 194 |
| Survivin-R | CAACGCGAAAGGAAAGACAG | ||
| β–actin-F | TCC CTG GAG AAG AGC TAC G | 131 | |
| β–actin-R | GTA GTT TCG TGG ATG CCA CA | ||
| TP53-F | TCA ACA AGA TGT TTT GCC AAC TG | 118 | |
| TP53-R | ATG TGC TGT GAC TGC TTG TAG ATG |
Table 2.
List of the Ingredients Used for Methylation Specific PCR (MSP)
| PCR ingredients | Un-methylated (µl) | Methylated (µl) |
|---|---|---|
| Input DNA | 4 | 5 |
| Taq DNA Polymerase (Fermentas) | 0.25 | 0.25 |
| PCR Buffer (Fermentas) | 2.5 | 4 |
| Primer-F (30μM) | 0.5 | 0.25 |
| Primer-R (30μM) | 0.5 | 0.25 |
| dNTPs (2.5mM, Fermentas) | 1 | 0.5 |
| MgCl2 (25 mM, Fermentas) | 2 | 2 |
| DMSO (Fermentas) | 0.5 | - |
| Nuclease free H2O | 13.75 | 12.75 |
| Total volume | 25 | 25 |
Quantitative Real Time PCR for assessment of TP53 and survivin genes expression
For this purpose, a number of 500,000 cells were cultured in 6-well plates. The cells were treated with DCM extracts of the SA plant in IC50 concentration. After 12 hours, total RNA was extracted using the RNX Plus kit (Cinnagen, Iran) according to the manufacturers’ protocol. The extracted RNA was evaluated by the Eppendorf Biophotometer and 3µg of this RNA was utilized for cDNA synthesis using the RevertAid™ (First Strand cDNA Synthesis Kit, Fermantas). CDNA synthesis was done according to the recommended protocol using random hexamer primers.
QRT- PCR was performed using the specific primers listed in Table 1. Beta actin was employed as normalizer gene. Thermo cycling started with 3 min of initial hold at 94°C and followed by 45 cycles of 94°C for 10 s, 58°C for 40 s and 72° C for 20 s. the reaction mixture used for qRT-PCR contained: 10 µl of SYBR premix (RR820L, Takara), 0.5 µl of forward and reverse primers in a concentration of 4 µM, 0.5 µl of cDNA and nuclease free water up to 20 µl. Relative expressions of TP53 and survivin mRNAs were calculated using delta-delta Ct method (Livak and Schmittgen, 2001).
Results
TUNEL Apoptosis assay
The TUNEL assay was used to confirm the occurrence of apoptosis 24 hours after the treatment with SADCM. Using this test, DNA fracturing resulted from apoptosis was appeared in brown color. Despite, brown precipitates were not observed in the untreated cells (Figure 1).
Figure 1.
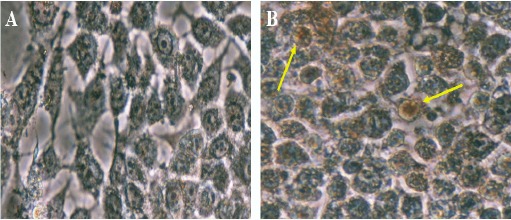
Apoptotic Effects of SADCM on MCF-7 Cells are Observed by TUNEL Assay. A, Non Treated Cells; B, MCF-7 Cells Treated with SADCM. Arrows Indicate the Apoptotic Cells.
Cell death ELISA assay for quantification of apoptosis and necrosis
The Cell death ELISA kit was utilized to evaluate the cell death induced by SADCM. Based on the results of the MTT test from previous studies, the IC50 concentration (200 mg/ml) and 400 mg/ml concentration were used for this assay. According to the results, a significant increase in apoptosis rate was detected in cells treated with the extract. By increasing the concentration from 200 to 400 micrograms, the rate of apoptosis was also increased (Figure 2).
Figure 2.
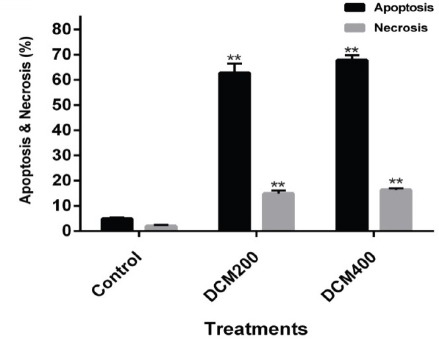
Comparison of Apoptosis and Necrosis between Untreated MCF-7 Cells and the Cells Treated with 200 and 400 mg/ml Concentrations of SADCM in 24 hours (P < 0.01).
MSP test
MSP on TP53 promoter with methylated and unmethylated primers showed that, the extract had considerable effects on the methylation status of this gene. There was a 166 bp band with the methylated primer in the untreated cells that represents the methylated status of P53 gene promoter. The intensity of the mentioned band was largely decreased after treatment with the herbal extract. This was an evident of a change from hypermethylation to hypomethylation state (Figure 3).
Figure 3.
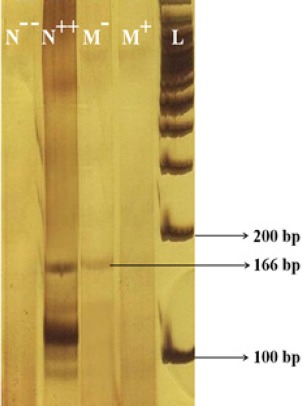
MSP with Methylated Primer Pairs. The amplicon in 166 bp size, represents a methylated status. N --, control DNA extracted from peripheral blood (neither methylated nor treated with SBS); N ++, control DNA extracted from peripheral blood (methylated using M.SssI CpG Methyltransferase enzyme and treated with SBS); M -, untreated MCF-7 cells; M +, MCF-7 cells treated with SADCM, L: 100 bp Ladder.
MSP with unmethylated primers resulted in a 170 bp band in the N -- , untreated and also treated cells). Presence of both 166 and 170 bp bands in M- and M+ groups indicates the partial methylation of TP53 gene promoter and the presence of unmethylated cytosines (Figure 4).
Figure 4.

N --, Control DNA Extracted from Peripheral Blood (neither methylated nor treated with SBS); M -, untreated MCF-7 cells; M +, MCF-7 cells treated with SADCM; L, 100 bp Ladder.
TP53 and survivin genes expression
Real time PCR was exploited for assessment of changes in TP53 and Survivin genes’ expression in MCF-7 cells treated with SADCM. Results indicated a significant increase in TP53 gene expression compared to untreated controls (Figure 5A). Moreover, a noticeable effect on the survivin gene expression was observed, where exposure to the extract was led to a significant decrease in mRNA expression (Figure 5B).
Figure 5.
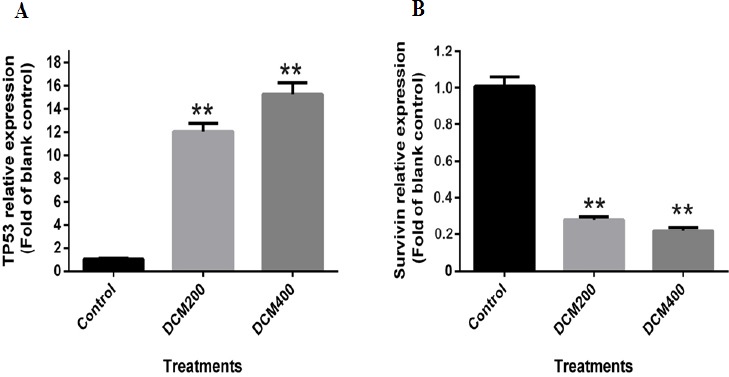
mRNA Expression Levels in Untreated Cells Versus MCF-7 Cells Treated with 200 and 400 mg/ml Concentrations of SADCM in 24 h; A, TP53 and B, survivin (P < 0.01).
Discussion
Breast cancer is a common type of cancer among women with a substantial rate of morbidity and mortality. Cancer patients usually experience different kinds of treatment strategies. However, herbal treatments because of their lower side effects have attracted a great deal of interest. Natural compounds with herbal origins such as Vinca Alkaloids, Taxanes, podophyllotoxin, Camptothecins are the most commonly used FDA approved compounds for the treatment of a wide range of cancers including breast, ovarian and lung cancers (Hosseini et al., 2015). Similarly, some derivatives from Scrophularia plants are being used in the treatment of several diseases including inflammation and infection (Uddin et al., 2011). In this regard, in previous studies, we have evaluated the cytotoxic effects of extracts and fractions derived from some Scrophularia species on a variety of cancerous cells (Hosseini et al., 2015; Orangi et al., 2016). Furthermore, the increased expression of apoptosis indicator genes including Caspase-3 and Caspase-9 besides down-regulation of anti-apoptotic genes such as Bcl-2 gene confirmed the potential of these compounds in the treatment of cancer. In the present study, we further confirmed the apoptotic effects of SADCM on MCF-7 cells by cell death ELISA and TUNEL assays. Uncovering the mechanisms by which the bioactive compounds exert their anticancer effects, could be a valuable finding for better understanding of the factors involved in the tumorigenesis and treatment process. Epigenetic changes are one of the most prominent alterations which play important roles in various types of cancer, either in the early phase or late phase. Cytosine methylation at CpG dinucleotides, especially in the promoter region of antitumor genes, is an important epigenetic change which can unusually change their transcription (Ma et al., 2010). In other words, the hyper-methylation of CpG islands in promoter region could be correlated with transcriptional silencing. The epigenetic changes are reversible alterations, which do not affect the DNA sequence. Hence, finding effective epi-drugs especially from plant origin to reverse the epigenetic changes to the normal state has recently attracted a great deal of attention (Hatzimichael and Crook, 2013). In a study, Chrysin, a flavonoid extracted from Oroxylum indicum, exhibited anticancer impacts on A375 melanoma cells via the normalization of histone acetylation and chromatin organization (Pal-Bhadra et al., 2012). In another research, Mahanin, an alkaloid from plant origin, has been used in returning hyper- methylation of the tumor suppressor gene RASSF1A in prostate cancer. Also, a reversal of epigenetic changes in colorectal carcinoma by active components extracted from Boswellia serrata plant is reported. Silencing of tumor suppressor gene TP53 is reported in several cancers (Mirza et al., 2002; Gopisetty et al., 2006; Ramezani et al., 2012) which makes it a fascinating therapeutic target. In this regard, the present study aimed to investigate the effects of the mentioned extract on methylatin status of TP53 gene promoter and consequently its expression levels by using the MSP test and qRT –PCR respectively. For this purpose, we utilized the MSP test, as a powerful technique to determine the methylation changes in this gene. Gong et al., also employed the MSP test to detect the potential of Danshen compounds on reverting the methylation status of Aurora A Gene in breast cancer (Gong et al., 2012).
According to MSP results, TP53 promoter in MCF-7 cells was in both methylated and unmethylated situation (partially methylated). Treatment with SADCM could remove the majority of methyl groups from this region and restore the methylation changes which were leading to lower gene expression to normal. Therefore, according to real time PCR results, a significant increase in TP53 mRNA expression was evident.
On the other hand, tumor formation and progression is closely associated with apoptosis impairment (Van De Voorde et al., 2012). Survivin is a protein encoded by the BIRC5 gene that is over expressed in most human cancers. This protein is a member of the apoptosis suppressor family (IAP) that inhibits apoptosis through both intrinsic and extrinsic pathways by suppressing caspases (Hosseini et al., 2015; Sah et al., 2006). Today, anti-apoptotic effect of this gene is moderated by strategies such as RNA interference methods. These studies suggest that down regulating the survivin gene leads to revival of the apoptosis pathway and a decrease of tumorigenesis (Karami et al., 2013; Montazami et al., 2015).
In addition, several studies indicated that the increased expression of TP53 gene inhibits survivin expression (Mirza et al., 2002). Accordingly, P53 protein not only could have a direct role in tumor suppression, but it could play a complementary role by overshadowing the survivin. The results of quantitative RT- PCR showed that, survivin gene expression was also reduced after treatment with the extract and this may be a consequence of P53 activity. Accordingly, SADCM could be considered as a potential source for production of cancer epi-drugs.
The apoptotic effect of SADCM on MCF-7 breast cancer cells was confirmed by cell death ELISA and TUNEL assays. According to the MSP and qRT-PCR results, this extract could drastically restore the methylation status of the TP53 gene and its expression. A decreased expression of the apoptotic gene survivin, which is expected to be influenced by P53 protein, was also evident. Consequently, SA could be considered for further investigations in production of cancer epi-drugs.
Declarations
Funding
None to declare.
Availability of data and materials
All data generated or analyzed in this study, that are relevant to the results presented in this article, are included in this article.
Kolsoum Rezaie Kahkhaie1, Mehrdad Ghavifekr Fakhr2, Dariush Shanehbandi2, Behzad Baradaran2*, Majid Farshdousti Hagh2*, Saeed Sadigh-Eteghad1, Habib Zarredar3, and Elham Safarzadeh.
Authors’ contributions
Conception and design: KRK, BB and MFH. Development of methodology: MGF, DS, and BB. Acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.): MGF, DS, BB, and KRK. Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): MFH, and SS. Writing, review, and/or revision of themanuscript: KRK, BB, MFH, HZ, and ES. Administrative, technical, or material support (i.e., reporting or organizing data, constructing databases): SS, HZ, ES, and BB. Study supervision: BB, KRK, and MFH. All authors read and approved the final manuscript.
Ethics approval and consent to participate
All patients provided written consent prior to study. The study id approved by ethical committee of Tabriz University of Medical Sciences.
Consent for publication
Not applicable.
Competing interests
The authors state no conflicts of interest.
Acknowledgements
This research was supported by Immunology Research Center, Tabriz University of Medical Sciences, Tabriz, Iran (grant number: 92/42).
References
- Almeida LO, Custodio AC, Pinto GR, et al. Polymorphisms and DNA methylation of gene TP53 associated with extra-axial brain tumors. Genet Mol Res. 2009;8:8–18. doi: 10.4238/vol8-1gmr518. [DOI] [PubMed] [Google Scholar]
- Cai FF, Kohler C, Zhang B, et al. Epigenetic therapy for breast cancer. Int J Mol Sci. 2011;12:4465–87. doi: 10.3390/ijms12074465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- di Pietro A, Koster R, Boersma-van Eck W, et al. Pro- and anti-apoptotic effects of p53 in cisplatin-treated human testicular cancer are cell context-dependent. Cell Cycle. 2012;11:4552–62. doi: 10.4161/cc.22803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghavifekr Fakhr M, Farshdousti Hagh M, Shanehbandi D, et al. DNA Methylation pattern as important epigenetic criterion in cancer. Genet Res Int. 2013;2013:317569. doi: 10.1155/2013/317569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gong Y, Li Y, Abdolmaleky HM, et al. Tanshinones inhibit the growth of breast cancer cells through epigenetic modification of Aurora A expression and function. PLoS One. 2012;7:e33656. doi: 10.1371/journal.pone.0033656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gopisetty G, Ramachandran K, Singal R. DNA methylation and apoptosis. Mol Immunol. 2006;43:1729–40. doi: 10.1016/j.molimm.2005.11.010. [DOI] [PubMed] [Google Scholar]
- Gottesman MM, Fojo T, Bates SE. Multidrug resistance in cancer:role of ATP-dependent transporters. Nat Rev Cancer. 2002;2:48–58. doi: 10.1038/nrc706. [DOI] [PubMed] [Google Scholar]
- Hatzimichael E, Crook T. Cancer epigenetics:new therapies and new challenges. J Drug Deliv. 2013;2013:529312. doi: 10.1155/2013/529312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hosseini B-A, Pasdaran A, Kazemi T, et al. Dichloromethane fractions of Scrophularia oxysepala extract induce apoptosis in MCF-7 human breast cancer cells. Bosn J Basic Med Sci. 2015;15:26–32. doi: 10.17305/bjbms.2015.1.226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Http://medgen.ugent.be/methBLAST/
- Http://rulai.cshl.edu/cgi-bin/CSHLmpd2/promExtract.pl?species=Human .
- Http://www.urogene.org/methprimer/
- Karami H, Baradaran B, Esfahani A, et al. siRNA-mediated silencing of survivin inhibits proliferation and enhances etoposide chemosensitivity in acute myeloid leukemia cells. Asian Pac J Cancer Prev. 2013;14:7719–24. doi: 10.7314/apjcp.2013.14.12.7719. [DOI] [PubMed] [Google Scholar]
- Karim-Kos HE, de Vries E, Soerjomataram I, et al. Recent trends of cancer in Europe:a combined approach of incidence, survival and mortality for 17 cancer sites since the 1990s. Eur J Cancer. 2008;44:1345–89. doi: 10.1016/j.ejca.2007.12.015. [DOI] [PubMed] [Google Scholar]
- Kirkwood JM, Butterfield LH, Tarhini AA, et al. Immunotherapy of cancer in 2012. CA Cancer J Clin. 2012;62:309–35. doi: 10.3322/caac.20132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lengauer C, Issa J-P. The role of epigenetics in cancer. Mol Med Today. 1998;4:102–3. doi: 10.1016/s1357-4310(98)01220-9. [DOI] [PubMed] [Google Scholar]
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402–8. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- Ma A-N, Huang W-L, Wu Z-N, et al. Induced epigenetic modifications of the promoter chromatin silence survivin and inhibit tumor growth. Biochem Biophys Res Commun. 2010;393:592–7. doi: 10.1016/j.bbrc.2010.02.020. [DOI] [PubMed] [Google Scholar]
- Meng HX, Hackett JA, Nestor C, et al. Apoptosis and DNA Methylation. Cancers. 2011;3:1798–820. doi: 10.3390/cancers3021798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirza A, McGuirk M, Hockenberry TN, et al. Human survivin is negatively regulated by wild-type p53 and participates in p53-dependent apoptotic pathway. Oncogene. 2002;21:2613–22. doi: 10.1038/sj.onc.1205353. [DOI] [PubMed] [Google Scholar]
- Montazami N, Kheir Andish M, Majidi J, et al. siRNA-mediated silencing of MDR1 reverses the resistance to oxaliplatin in SW480/OxR colon cancer cells. Cell Mol Biol. 2015;61:98–103. [PubMed] [Google Scholar]
- Orangi M, Pasdaran A, Shanehbandi D, et al. Cytotoxic and apoptotic activities of methanolic sub-fractions of Scrophularia oxysepala against human breast cancer cell line. Evid Based Complement Alternat Med. 2016;2016 doi: 10.1155/2016/8540640. Article ID 8540640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pal-Bhadra M, Ramaiah MJ, Reddy TL, et al. Plant HDAC inhibitor chrysin arrest cell growth and induce p21(WAF1) by altering chromatin of STAT response element in A375 cells. BMC Cancer. 2012;12:180. doi: 10.1186/1471-2407-12-180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Promoter 2.0 Prediction Server [Google Scholar]
- Ramezani F, Salami S, Omrani MD, et al. CpG island methylation profile of estrogen receptor alpha in Iranian females with triple negative or non-triple negative breast cancer:new marker of poor prognosis. Asian Pac J Cancer Prev. 2012;13:451–7. doi: 10.7314/apjcp.2012.13.2.451. [DOI] [PubMed] [Google Scholar]
- Richards EJ, Elgin SC. Epigenetic codes for heterochromatin formation and silencing:rounding up the usual suspects. Cell. 2002;108:489–500. doi: 10.1016/s0092-8674(02)00644-x. [DOI] [PubMed] [Google Scholar]
- Sah N, Khan Z, Khan G, et al. Structural, functional and therapeutic biology of survivin. Cancer Lett. 2006;244:164–71. doi: 10.1016/j.canlet.2006.03.007. [DOI] [PubMed] [Google Scholar]
- Surget S, Khoury MP, Bourdon J-C. Uncovering the role of p53 splice variants in human malignancy:a clinical perspective. OncoTargets Ther. 2014;7:57–68. doi: 10.2147/OTT.S53876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uddin SJ, Grice ID, Tiralongo E. Cytotoxic effects of Bangladeshi medicinal plant extracts. Evid Based Complement Alternat Med. 2011;2011 doi: 10.1093/ecam/nep111. Article ID 578092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van De Voorde L, Speeckaert R, Van Gestel D, et al. DNA methylation-based biomarkers in serum of patients with breast cancer. Mutat Res Rev Mutat Res. 2012;751:304–25. doi: 10.1016/j.mrrev.2012.06.001. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed in this study, that are relevant to the results presented in this article, are included in this article.
Kolsoum Rezaie Kahkhaie1, Mehrdad Ghavifekr Fakhr2, Dariush Shanehbandi2, Behzad Baradaran2*, Majid Farshdousti Hagh2*, Saeed Sadigh-Eteghad1, Habib Zarredar3, and Elham Safarzadeh.


