Abstract
Renal dysfunction results in the accumulation of various uremic toxins, including indoxyl sulphate (IS), and is a major risk factor for atrial fibrillation (AF). Experimental studies have demonstrated that IS exacerbates atrial remodelling via oxidative stress, inflammation, and fibrosis. However, its clinical impact on AF-promoting cardiac remodelling has not been described. Therefore, the purpose of this study was to clarify the relationship between basal IS levels and the 1-year outcomes after catheter ablation for the treatment of AF. Our prospective observational study included data from 125 patients with AF who underwent catheter ablation. Over a 1-year follow-up period, AF recurrence was identified in 21 patients. The 1-year AF-free survival was significantly lower in patients with high serum IS levels (≥0.65 μg/mL) than in those with low IS levels (60.1 ± 10.4% versus 85.2 ± 3.9%, P = 0.007). Univariable analysis identified that an IS concentration ≥ 0.65 μg/mL was associated with AF recurrence (hazard ratio [HR] = 3.10 [1.26–7.32], P = 0.015), and this association was maintained in multivariate analysis (HR = 3.67 [1.13–11.7], P = 0.031). Thus, in patients undergoing AF ablation, serum IS levels at baseline independently predict the recurrence of arrhythmia.
Introduction
Several clinical trials have reported a close association between atrial fibrillation (AF) and chronic kidney disease (CKD)1–5. For example, the burden of AF is increased among patients with CKD, and AF itself increases as a function of CKD severity. The relationship between renal function and AF recurrence after catheter ablation for AF was addressed in recent reports6–8. Patients with a lower estimated glomerular filtration rate (eGFR) at baseline had a greater risk of recurrence after radiofrequency catheter ablation for AF. Atrial remodelling due to CKD was thought to be responsible for the poor outcome after AF ablation in patients with CKD; however, the exact mechanism of this association remains to be elucidated.
Indoxyl sulphate (IS) is among the most representative uremic toxins derived from the metabolism of dietary protein by the gut microbiota and has been implicated in the pathogenesis of various cardiovascular diseases9–11, including AF12. Experimental studies in animal models have shown that IS can exacerbate AF via its effect on cardiac fibrosis and inflammation, with enhanced oxidative stress and reduced anti-oxidative defense13,14. These effects have been shown to induce arrhythmogenesis in pulmonary veins, sinoatrial nodes, and atria isolated from rabbit hearts15. Recently, Aoki et al.16 demonstrated, in a rat model of CKD, that IS increases the inducibility of AF, and that circulating levels of IS can be significantly attenuated by using an absorbent of uremic toxins, such as AST-120. Thus, it is plausible that IS may contribute to atrial remodelling, although there is limited clinical evidence in support of the association between IS and AF.
Therefore, the aim of our study was to investigate the relationship between IS and recurrent AF in patients after radiofrequency catheter ablation (RFCA) for AF.
Methods
Study population
This prospective observational study was conducted at the University of Tsukuba Hospital, Tsukuba, Japan, between April 2009 and March 2011. We enrolled 125 consecutive patients with non-valvular AF scheduled for RFCA for AF. Patients who had hemodynamically significant (moderate-to-severe) valvular disease, thrombus in the left atrium (LA), uncontrolled thyroid dysfunction, pre-procedural significant coronary artery stenosis, a previous myocardial infarction or cardiac surgery in the previous 3 months, contraindications to anticoagulant therapy, stage 4 or 5 CKD, or pregnancy were excluded.
All enrolled patients had symptomatic paroxysmal or persistent AF and had not responded to treatment with one or more antiarrhythmic drugs. Long-standing persistent AF was defined as any AF episode lasting for longer than 1 year. A detailed medical history regarding AF and related cardiovascular and/or systemic conditions was obtained from all patients. The CHADS2 score was calculated for each patient based on a point system, as previously described17. Transthoracic echocardiography was also performed to assess left ventricular function and the left atrial volume index (LAVI) before RFCA.
The study was in compliance with the principles outlined in the Declaration of Helsinki and was approved by Ethics Committee, University of Tsukuba Hospital. Before ablation, written informed consent was obtained from all the patients.
Study endpoint
The endpoint of this study was recurrence of AF or atrial tachycardia after a blanking period of 3 months.
Serum IS concentration
Blood samples were collected from patients just before ablation and serum was stored at −80 °C. Serum levels of IS and indole-3 acetic acid (IAA) were measured by reversed-phase high-performance liquid chromatography using a conventional octadecylsilyl silica column and a fluorescence detector, according to a previously described method, with minor modifications18. The coefficients of variation for intra-day and inter-day assays were <3%.
Classification of CKD
Classification of the CKD stage was determined using the eGFR, calculated at baseline. The eGFR was calculated using the estimation equation for Japanese patients with CKD19. This equation calculates the eGFR from serum creatinine, adjusted for age and sex, using the following formula: (eGFR [mL/min/1.73 m2] = 194 × age−0.287 × serum creatinine−1.094 × [0.739 for women]). The study population was divided into three subgroups: patients with an eGFR ≥ 90 mL/min/1.73 m2 (CKD stage 1), patients with an eGFR between 60 and 89.9 mL/min/1.73 m2 (CKD stage 2), and patients with an eGFR between 30 and 59.9 mL/min/1.73 m2 (CKD stage 3). The creatinine clearance (CrCl) was evaluated using the Cockcroft–Gault formula: CrCl (mL/min) = ([140 - age] × body weight [kg] × [0.85 if female])/(72 × serum creatinine [mg/dL]).
Catheter ablation
Procedures were performed under general anaesthesia, aiming at an anticoagulation time of 300 s. An irrigated-tip catheter was used to deliver radiofrequency energy in all patients. A circular lasso catheter was used to confirm the pulmonary vein (PV) and to map the LA area. After trans-septal puncture, PV isolation (PVI) was performed. After PVI, a bidirectional block was systematically obtained in all veins. In the case of persistent AF, complete electrical isolation of the PVs was confirmed after restoration of a sinus rhythm. If the AF was sustained or recurred despite the internal cardioversion after PVI, a stepwise approach was used, including linear ablation of the LA roof, superior vena cava isolation, and/or ablation of complex fractionated atrial electrograms.
Follow-up
Follow-up was performed at 1, 3, 6, and 12 months, or earlier if a patient developed symptoms consistent with recurrent AF. The average follow-up period was 11.7 months (range, 3.2 to 12 months). Recurrence was defined as any evidence of an episode of atrial arrhythmia lasting for more than 30 s after a 3-month blanking period.
Statistical analysis
Analyses were performed using JMP software (version 10). Values are reported as the mean ± standard deviation (SD) for continuous variables, and proportions for categorical variables. The optimal cut-off point was determined by the maximum Youden index based on receiver operating characteristic (ROC) curve analysis, and areas under the ROC curve (AUCs) were calculated. The association between serum levels of IS or IAA and other factors was evaluated using Pearson’s correlation coefficients. Differences between continuous values were assessed using an unpaired, two-tailed, t-test for normally distributed continuous variables, and a Mann–Whitney test for skewed variables. The chi-square statistic was used for testing relationships between categorical variables. The association between baseline variables and AF recurrence was evaluated using univariate and multivariate Cox proportional hazards analysis. AF-free survival curves were analysed according to the Kaplan–Meier method and compared by the log-rank test. A P-value < 0.05 was considered significant.
Results
Study population
Of the 125 consecutive patients with AF enrolled, 20 were excluded from the analysis due to missing pre-echocardiographic data (5 patients) and because of a lack of follow-up (15 patients). Relevant data of the remaining 105 patients (83.8% men, mean age: 60.0 ± 10.8 years) included in the analysis are reported in Table 1. Over the 1-year follow-up after ablation, 21 (20.0%) patients developed AF recurrence (AF in 16, uncommon atrial flutter in 1, and atrial tachycardia in 4 patients). There were no significant differences in baseline IS and IAA levels between patients who developed AF-recurrence and those who did not (IS, 0.51 ± 0.33 vs 0.43 ± 0.31 μg/mL, P = 0.27; IAA, 0.14 ± 0.05 vs 0.16 ± 0.10 μg/mL, P = 0.31, respectively). After calculating the maximum Youden index (sensitivity + specificity − 1) and the AUC, the optimal cut-off values of serum levels of IS and IAA were found to be 0.65 μg/mL and 0.17 μg/mL, respectively (Table 2).
Table 1.
Baseline characteristics.
| All (n = 105) | IS ≥ 0.65 μg/mL (n = 23) | IS < 0.65 μg/mL (n = 82) | P-value | |
|---|---|---|---|---|
| Age (years) | 60.0 ± 10.8 | 62.9 ± 11.3 | 59.2 ± 10.6 | 0.14 |
| Male sex | 88 (83.8) | 20 (87.0) | 68 (82.9) | 0.64 |
| Body mass index (kg/m2) | 23.6 ± 2.9 | 23.7 ± 3.2 | 23.6 ± 2.9 | 0.94 |
| Paroxysmal AF | 69 (65.7) | 14 (60.9) | 55 (67.1) | 0.58 |
| Long-standing AF | 19 (19.1) | 7 (30.4) | 12 (14.6) | 0.082 |
| Duration of AF history (years) | 5.5 ± 5.7 | 8.4 ± 6.9 | 4.8 ± 5.2 | 0.010 |
| Medication | ||||
| ACEIs/ARBs | 44 (41.9) | 10 (43.5) | 34 (41.5) | 0.86 |
| Statins | 33 (31.4) | 5 (21.7) | 28 (34.2) | 0.26 |
| Beta-blockers | 58 (55.2) | 19 (82.6) | 39 (47.6) | 0.003 |
| Class I AADs | 59 (56.2) | 12 (52.2) | 47 (57.3) | 0.66 |
| Class III AADs | 34 (32.4) | 10 (43.5) | 24 (29.3) | 0.20 |
| Class IV AADs | 16 (15.2) | 2 (8.7) | 14 (17.1) | 0.32 |
| Hypertension | 52 (49.5) | 13 (56.5) | 39 (47.6) | 0.45 |
| Diabetes mellitus | 14 (13.3) | 3 (13.0) | 11 (13.4) | 0.96 |
| Dyslipidaemia | 48 (45.7) | 9 (39.1) | 39 (47.6) | 0.47 |
| CHADS2 score | 0.81 ± 0.84 | 1.04 ± 0.88 | 0.74 ± 0.83 | 0.13 |
| Echocardiogram parameters | ||||
| LVEF (%) | 65.2 ± 11.2 | 66.3 ± 12.4 | 64.9 ± 10.9 | 0.62 |
| LAVI (mL/m2) | 36.3 ± 18.3 | 44.7 ± 22.0 | 33.8 ± 16.4 | 0.012 |
| eGFR (mL/min/1.73 m2) | 76.1 ± 18.5 | 68.2 ± 20.9 | 78.4 ± 17.3 | 0.019 |
| CKD stage | 0.021 | |||
| Stage 1 | 19 (18.1) | 4 (17.4) | 15 (18.3) | |
| Stage 2 | 69 (65.7) | 11 (47.8) | 58 (70.7) | |
| Stage 3 | 17 (16.2) | 8 (34.8) | 9 (11.0) | |
| BNP (pg/mL) | 78.9 ± 91.6 | 114.1 ± 111.7 | 69.5 ± 83.8 | 0.042 |
| CRP (mg/mL) | 0.097 ± 0.102 | 0.114 ± 0.111 | 0.092 ± 0.099 | 0.34 |
| IS (μg/mL) | 0.45 ± 0.31 | 0.93 ± 0.25 | 0.31 ± 0.15 | < 0.001 |
| IAA (μg/mL) | 0.16 ± 0.10 | 0.18 ± 0.07 | 0.15 ± 0.10 | 0.097 |
Values are given as mean ± SD or number (%).
AAD, anti-arrhythmic drug; AF, atrial fibrillation; ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor antagonist; BNP, B-type natriuretic peptide; CRP, C-reactive protein; eGFR, estimated glomerular filtration rate; IAA, indole-3 acetic acid; IS, indoxyl sulphate; LAVI, left atrial volume index; LVEF, left ventricular ejection fraction.
Table 2.
Optimal cut-off point based on AF recurrence.
| Median value | Range | AUC | Cut-off value | |
|---|---|---|---|---|
| IS (μg/mL) | 0.36 | 0–1.74 | 0.576 | 0.65 |
| IAA (μg/mL) | 0.13 | 0.04–0.56 | 0.522 | 0.17 |
AF, atrial fibrillation; AUC; area under the curve, IAA, indole-3 acetic acid; IS, indoxyl sulphate.
Relationship of serum IS levels with renal function and age
Serum IS levels were significantly increased in patients with CKD stage 3 (Fig. 1). However, the correlations of serum IS levels with eGFR (r = −0.295, P = 0.002) and CrCl (r = −0.263, P = 0.007) were weak (Fig. 2a,b). IS levels were also weakly correlated with age (r = 0.253, P = 0.009; Fig. 2c).
Figure 1.
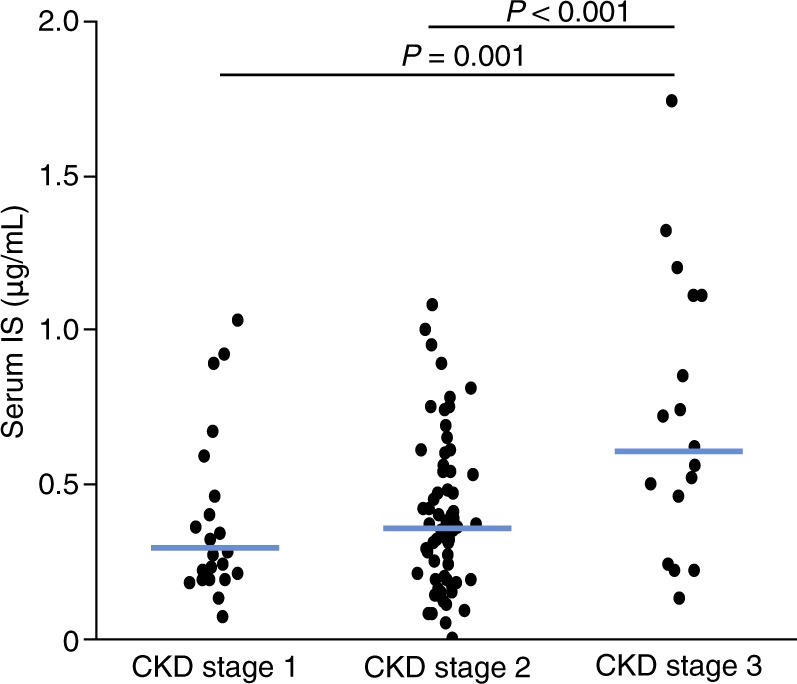
Serum levels of IS are increased in patients with reduced renal function. CKD, chronic kidney disease; IS, indoxyl sulphate.
Figure 2.
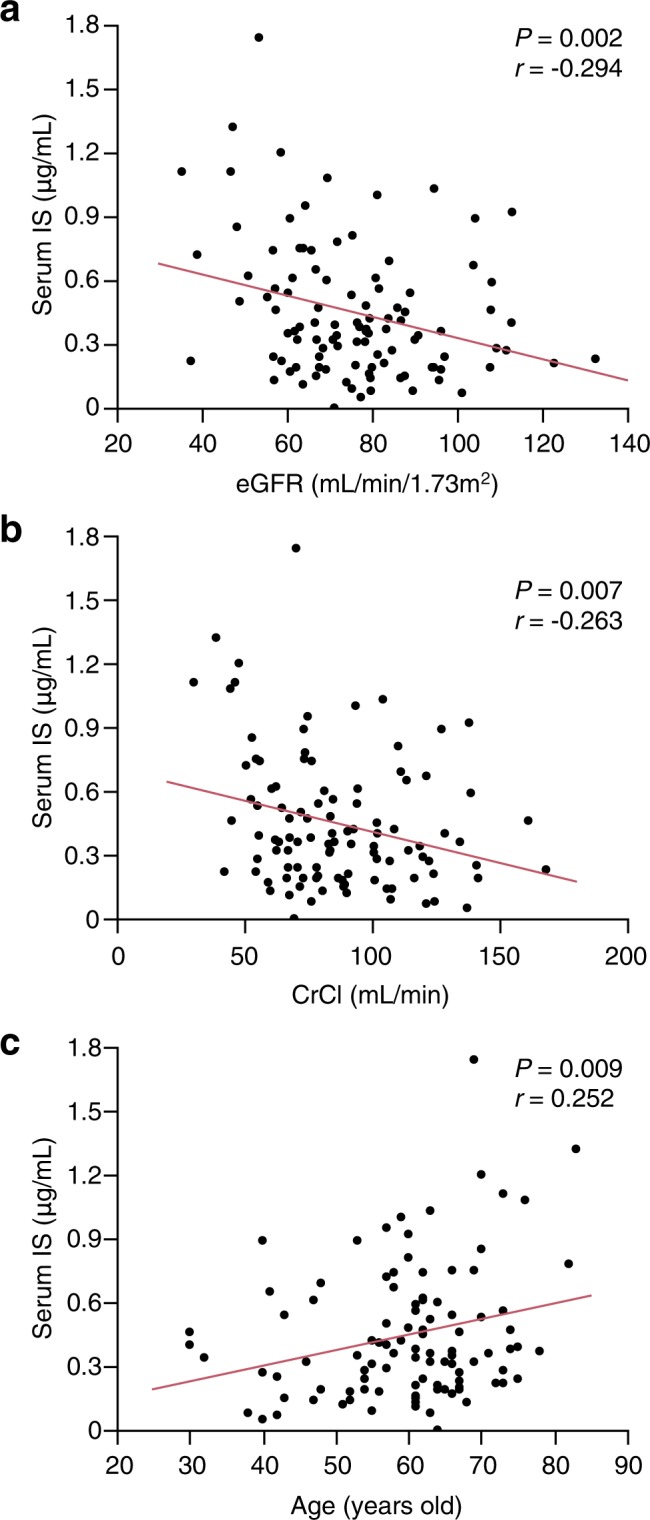
Correlations between serum levels of IS and eGFR (a), CrCl (b), or age (c). CrCl, creatinine clearance; eGFR, estimated glomerular filtration rate; IS, indoxyl sulphate.
IAA is also a uremic toxin and belongs to the family of indolic uremic solutes, such as IS. Serum levels of IAA were positively correlated with those of IS (r = 0.443, P < 0.001; Supplementary Fig. S1). In this cohort, unlike IS, IAA levels did not differ statistically significantly among patients with CKD stage 1, 2, and 3 (Supplementary Fig. S2). IAA levels were weakly correlated with eGFR (r = −0.255, P = 0.009), but not with CrCl (r = −0.102, P = 0.30) or age (r = −0.029, P = 0.77) (Supplementary Fig. S3).
Demographic and clinical characteristics of patients with high IS levels
Among the 105 patients in our study group, 23 patients (21.9%) were categorized into the high IS (≥0.65 μg/mL) and 82 patients (78.1%) into the low IS group (<0.65 μg/mL; Table 1) according to the optimal cut-off value. There were no significant differences in sex or age between the two groups. The duration after diagnosis of AF was significantly longer in the high IS group (P = 0.010). Patients with high IS levels had lower eGFR (P = 0.019), an increased LAVI (P = 0.012), an elevated BNP (P = 0.042), and were more likely to have been treated with beta-blockers before ablation (P = 0.003). After ablation, the proportion of patients taking class III anti-arrhythmic drugs (AAD) was significantly higher in the high-IS group than in the low-IS group (P = 0.004, Supplementary Table S1).
AF-free survival
The 1-year AF-free survival was significantly lower in patients with a high serum IS level than in those with a low IS level (60.1 ± 10.4% vs 85.2 ± 3.9%, P = 0.007; Fig. 3a). However, the 1-year AF-free survival did not differ (P = 0.25) between patients with high IAA levels (85.9 ± 5.8%) and those with low IAA levels (76.6 ± 5.1%) (Supplementary Fig. S4).
Figure 3.
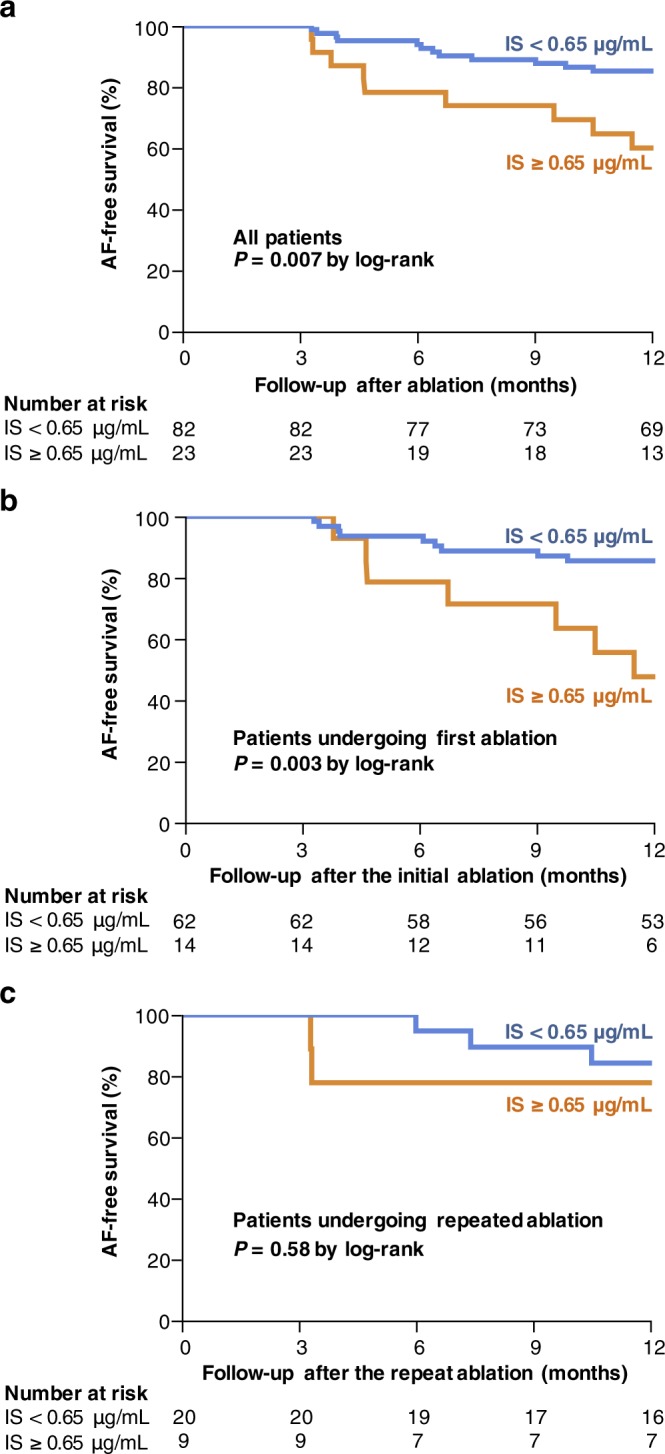
Impact of IS levels on the recurrence of AF after catheter ablation. The AF-free survival rates are shown for the whole cohort (a), patients undergoing a first AF ablation (b), and patients undergoing a repeat AF ablation (c) according to IS levels. The numbers at the bottom of the graph indicates the number of ‘at risk’ patients in each follow-up month. AF, atrial fibrillation; IS, indoxyl sulphate.
Overall, in our study group, 76 patients (72.4%) underwent a first AF ablation, with the remaining 29 (27.6%) undergoing a repeat AF ablation. Among the 29 patients with recurrent AF, recovery of electrical conduction between the PVs and the LA was identified in 26 patients (89.7%). Among patients who underwent a first AF ablation, the 1-year estimates of AF-free survival were 47.6 ± 13.8% for those with a high serum IS level and 85.5 ± 4.5% for those with a low IS level (P = 0.003; Fig. 3b). In contrast, among patients who underwent a repeat AF ablation (Fig. 3c), the 1-year AF-free survival did not significantly differ (P = 0.58) between patients with high IS levels (77.9 ± 13.9%) and those with low IS levels (84.2 ± 8.4%).
Predictive value of IS in AF recurrence
On univariate analysis, an IS level ≥ 0.65 μg/mL was associated with an increased risk of AF recurrence (hazard ratio [HR]: 3.10, 95% CI: 1.26–7.32; P = 0.015; Table 3). After adjustment for age and sex, an IS level ≥ 0.65 μg/mL remained associated with increased risk of AF recurrence (HR: 3.76, 95% CI: 1.52–9.03; P = 0.005; Table 3). In the multivariate Cox proportional hazard model, adjusted for age, sex, duration after AF diagnosis, beta-blocker use before ablation, LAVI, BNP, Class III AAD use after ablation, as well as eGFR or CKD stage, an IS level ≥ 0.65 μg/mL remained as an independent predictor of AF recurrence (HR 3.60, 95% CI: 1.12–11.0, P = 0.032; and HR 3.67, 95% CI: 1.13–11.7, P = 0.031, respectively; Table 3).
Table 3.
Impact of a level of indoxyl sulphate ≥ 0.65 μg/mL on atrial fibrillation recurrence.
| HR | 95% CI | P-value | |
|---|---|---|---|
| Unadjusted analysis | 3.10 | 1.26−7.32 | 0.015 |
| Analysis adjusted for: | |||
| Age and sex | 3.76 | 1.52−9.03 | 0.005 |
| Age, sex, duration after diagnosis of AF, beta-blocker use before ablation, LAVI, BNP, Class III AAD use after ablation, and eGFR | 3.60 | 1.12−11.0 | 0.032 |
| Age, sex, duration after diagnosis of AF, beta-blocker use before ablation, LAVI, BNP, Class III AAD use after ablation, and CKD stage | 3.67 | 1.13−11.7 | 0.031 |
AAD, anti-arrhythmic drug; AF, atrial fibrillation; BNP, B-type natriuretic peptide; CI, confidence interval; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; HR, hazard ratio; LAVI, left atrial volume index.
Discussion
In this prospective observational study, we identified high IS levels as a strong and independent predictor of AF recurrence in patients undergoing successful catheter ablation. To the best of our knowledge, this association of uremic toxins with the outcomes of AF ablation has not been described previously.
The high AF recurrence rate after ablation in patients with CKD is indicative of a possible involvement of uremic toxins in AF pathogenesis. However, no clinical studies to date have demonstrated the involvement of uremic toxins in AF recurrence. Here, we showed that patients with a higher IS levels exhibited a higher rate of AF recurrence after ablation, with serum IS being a significant predictor of AF recurrence, even after adjustment for eGFR or the stage of CKD. Thus, it seems that IS has a predictive role in AF recurrence.
In recent years, increasing attention has been paid to the relationship between IS and cardiovascular diseases among patients with CKD. Barreto et al.20 reported that a higher serum level of IS was associated with an increased overall mortality and cardiovascular-specific mortality among patients with CKD. Furthermore, Lin et al.21 indicated that IS was a valuable marker in predicting cardiovascular events in patients with advanced CKD. Other researchers have reported association of a high IS level with increased risk of LV diastolic dysfunction22,23.
In our study cohort, the IS concentration correlated poorly with eGFR (Fig. 1), which was consistent with the findings from other groups24,25. IS is a gut-derived uremic toxin and factors other than the GFR, such as renal tubular secretion, diet and intestinal absorption, and gut microbiota metabolism, may affect IS concentration26.
Recent animal studies have revealed the causative role of IS in promoting AF remodelling. Chen et al.15 reported that IS induced the occurrence of delayed after-depolarizations and burst firing in PVs isolated from rabbits. They also showed that IS-treated PV cardiomyocytes had a larger Ca2+ leak than control PV cardiomyocytes15. Aoki et al.16 showed that, in rat models of renal failure induced by 5/6 nephrectomy, administration of AST-120, which is commonly used in clinical practice as an absorbent of uremic toxins, attenuated oxidative stress, inflammation, and fibrosis in the LA, and decreased AF inducibility in vivo. In cultured atrial fibroblasts, incubation with IS upregulated the expression of oxidative stress markers NOX2/NOX4 and malondialdehyde, along with an increase in inflammatory and profibrotic signalling molecules, such as monocyte chemoattractant protein 1 (MCP-1), transforming growth factor β1 (TGF-β1), α-smooth muscle actin (α-SMA), and collagen I16. These results indicate a direct effect of IS on the progression of the AF substrate. Therefore, IS may become a novel therapeutic target for AF.
The prevalence of AF among a large population of patients with CKD was found to be 2- to 3-fold higher than that in the general population27,28. Moreover, the prevalence of AF among dialysis patients has been reported to range between 7% and 27%29–32, as compared to 0.4% to 1.0% in the general population33, indicating that haemodialysis may not eliminate the risk of AF in CKD. Therefore, non-dialyzable uremic factors were considered responsible for AF-promoting remodelling. IS is a poorly dialyzable uremic toxin due to its high protein binding. Even after haemodialysis, the serum IS levels still remain high34. Therefore, accumulating IS may directly contribute to atrial remodelling in patients with CKD.
Limitations
The study had some limitations. First, compared to patients in the low IS level group, the duration after AF diagnosis had been longer in the cohort of patients with high IS levels; this group also had a greater prevalence of beta-blocker use, increased LAVI, and worse renal function, which would be sources of possible residual confounding factors. Furthermore, our study evaluated a heterogeneous population, including patients with paroxysmal as well as persistent AF. Because of the small sample size, no separate analysis was possible between these different groups. Consequently, we cannot rule out different biomarker patterns between these two entities of AF. Second, the diagnosis of AF recurrence was based on the occurrence of symptoms and periodic and occasional ECG recordings and Holter ECG findings. Therefore, some patients with asymptomatic AF recurrence might have been missed. Third, our cohort included individuals who underwent AF ablation at our institution between 2009 and 2011. During that period, advanced technologies that are widely used today, including the contact force-sensing catheter and balloon ablation, were not approved for use in Japan. These newer technologies have been reported to increase the durability of PV isolation and reduce AF recurrence35,36. Further studies may be needed to determine whether high IS levels can predict AF recurrence in the current setting of AF ablation. Fourth, patients who had haemodynamically significant valvular heart disease or stage 4 or 5 CKD, or received dialysis were excluded from this study. Further studies are needed to determine whether high IS levels can also predict AF recurrence in those patients. Fifth, it has been reported that there were some diurnal and inter-day variations in the circulating IS concentrations37, and dietary protein intake affected the IS concentrations38. However, in this study, blood samples were collected at a single time point, but were not collected at the same time of the day for all patients, and were not standardized with respect to food intake. Finally, this study was performed in a single centre with a relatively small sample size and a short follow-up period. Therefore, further studies with more patients and a longer follow-up period may be needed to confirm our study results.
Conclusion
Serum IS levels are associated with recurrence of AF after catheter ablation. Therefore, the IS level should be considered in the prediction of recurrence after ablation.
Electronic supplementary material
Acknowledgements
We would like to thank Editage (www.editage.jp) for English language editing.
Author Contributions
F.Y. and K.T. wrote the main manuscript text and prepared the figures. M.H., J.H., S.A. and T.H. collected the follow-up data. K.D. and M.H. measured the serum levels of indoxyl sulphate and participated in redaction of the manuscript. H.Y., N.M., Y.S., N.T., K.A., A.N. and M.I. participated in redaction and extensive revision of the manuscript.
Data Availability
The datasets generated and/or analysed during the current study are available from the corresponding author upon reasonable request.
Competing Interests
A.N. received honoraria from St. Jude Medical and Japan Lifeline, and an endowment from Medtronic and DVX. K.T. and Y.S. received an endowment from St. Jude Medical. The other authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-35226-5.
References
- 1.Watanabe H, et al. Close bidirectional relationship between chronic kidney disease and atrial fibrillation: the Niigata preventive medicine study. Am. Heart J. 2009;158:629–636. doi: 10.1016/j.ahj.2009.06.031. [DOI] [PubMed] [Google Scholar]
- 2.Olesen JB, et al. Stroke and bleeding in atrial fibrillation with chronic kidney disease. N. Engl. J. Med. 2012;367:625–635. doi: 10.1056/NEJMoa1105594. [DOI] [PubMed] [Google Scholar]
- 3.Alonso A, et al. Chronic kidney disease is associated with the incidence of atrial fibrillation: the Atherosclerosis Risk in Communities (ARIC) study. Circulation. 2011;123:2946–2953. doi: 10.1161/CIRCULATIONAHA.111.020982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bansal N, et al. Incident atrial fibrillation and risk of end-stage renal disease in adults with chronic kidney disease. Circulation. 2013;127:569–574. doi: 10.1161/CIRCULATIONAHA.112.123992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lau YC, Proietti M, Guiducci E, Blann AD, Lip GYH. Atrial fibrillation and thromboembolism in patients with chronic kidney disease. J. Am. Coll. Cardiol. 2016;68:1452–1464. doi: 10.1016/j.jacc.2016.06.057. [DOI] [PubMed] [Google Scholar]
- 6.Naruse Y, et al. Concomitant chronic kidney disease increases the recurrence of atrial fibrillation after catheter ablation of atrial fibrillation: a mid-term follow-up. Heart Rhythm. 2011;8:335–341. doi: 10.1016/j.hrthm.2010.10.047. [DOI] [PubMed] [Google Scholar]
- 7.Tokuda M, et al. Relationship between renal function and the risk of recurrent atrial fibrillation following catheter ablation. Heart. 2011;97:137–142. doi: 10.1136/hrt.2010.200824. [DOI] [PubMed] [Google Scholar]
- 8.Yanagisawa S, et al. Impaired renal function is associated with recurrence after cryoballoon catheter ablation for paroxysmal atrial fibrillation: A potential effect of non-pulmonary vein foci. J. Cardiol. 2017;69:3–10. doi: 10.1016/j.jjcc.2016.07.008. [DOI] [PubMed] [Google Scholar]
- 9.Ito S, Yoshida M. Protein-bound uremic toxins: new culprits of cardiovascular events in chronic kidney disease patients. Toxins (Basel) 2014;6:665–678. doi: 10.3390/toxins6020665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lekawanvijit S, Kompa AR, Wang BH, Kelly DJ, Krum H. Cardiorenal syndrome: The emerging role of protein-bound uremic toxins. Circ. Res. 2012;111:1470–1483. doi: 10.1161/CIRCRESAHA.112.278457. [DOI] [PubMed] [Google Scholar]
- 11.Dou L, et al. The cardiovascular effect of the uremic solute indole-3 acetic acid. J. Am. Soc. Nephrol. 2015;26:876–887. doi: 10.1681/ASN.2013121283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang S-Y, Chen Y-A, Chen S-A, Chen Y-J, Lin Y-K. Uremic toxins—novel arrhythmogenic factor in chronic kidney disease-related atrial fibrillation. Acta Cardiol. Sin. 2016;32:259–264. doi: 10.6515/ACS20151116A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yisireyili M, et al. Indoxyl sulphate promotes cardiac fibrosis with enhanced oxidative stress in hypertensive rats. Life Sci. 2013;92:1180–1185. doi: 10.1016/j.lfs.2013.05.008. [DOI] [PubMed] [Google Scholar]
- 14.Lekawanvijit S, et al. Does indoxyl sulphate, a uraemic toxin, have direct effects on cardiac fibroblasts and myocytes? Eur. Heart J. 2010;31:1771–1779. doi: 10.1093/eurheartj/ehp574. [DOI] [PubMed] [Google Scholar]
- 15.CHEN W-T, et al. The uremic toxin indoxyl sulphate increases pulmonary vein and atrial arrhythmogenesis. J. Cardiovasc. Electrophysiol. 2015;26:203–210. doi: 10.1111/jce.12554. [DOI] [PubMed] [Google Scholar]
- 16.Aoki K, et al. Role of indoxyl sulphate as a predisposing factor for atrial fibrillation in renal dysfunction. J. Am. Heart Assoc. 2015;4:e002023. doi: 10.1161/JAHA.115.002023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.January CT, et al. 2014 AHA/ACC/HRS Guideline for the Management of Patients With Atrial Fibrillation: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Heart Rhythm Society. Circulation. 2014;130:e199–e267. doi: 10.1161/CIR.0000000000000041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Al Z’abi,M, Ali B, Al Toubi M. HPLC-fluorescence method for measurement of the uremic toxin indoxyl sulphate in plasma. J. Chromatogr. Sci. 2013;51:40–43. doi: 10.1093/chromsci/bms103. [DOI] [PubMed] [Google Scholar]
- 19.Matsuo S, et al. Revised equations for estimated GFR from serum creatinine in japan. Am. J. Kidney Dis. 2009;53:982–992. doi: 10.1053/j.ajkd.2008.12.034. [DOI] [PubMed] [Google Scholar]
- 20.Barreto FC, et al. Serum indoxyl sulphate is associated with vascular disease and mortality in chronic kidney disease patients. Clin. J. Am. Soc. Nephrol. 2009;4:1551–1558. doi: 10.2215/CJN.03980609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lin C-J, et al. Indoxyl sulphate predicts cardiovascular disease and renal function deterioration in advanced chronic kidney disease. Arch. Med. Res. 2012;43:451–456. doi: 10.1016/j.arcmed.2012.08.002. [DOI] [PubMed] [Google Scholar]
- 22.Shimazu S, et al. Association between indoxyl sulphate and cardiac dysfunction and prognosis in patients with dilated cardiomyopathy. Circ. J. 2013;77:390–396. doi: 10.1253/circj.CJ-12-0715. [DOI] [PubMed] [Google Scholar]
- 23.Sato B, et al. Relation of plasma indoxyl sulphate levels and estimated glomerular filtration rate to left ventricular diastolic dysfunction. Am. J. Cardiol. 2013;111:712–716. doi: 10.1016/j.amjcard.2012.11.025. [DOI] [PubMed] [Google Scholar]
- 24.Eloot S, et al. Estimated glomerular filtration rate is a poor predictor of concentration for a broad range of uremic toxins. Clin. J. Am. Soc. Nephrol. 2011;6:1266–1273. doi: 10.2215/CJN.09981110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Snauwaert E, et al. Accumulation of uraemic toxins is reflected only partially by estimated GFR in paediatric patients with chronic kidney disease. Pediatr. Nephrol. 2018;33:315–323. doi: 10.1007/s00467-017-3802-5. [DOI] [PubMed] [Google Scholar]
- 26.Vanholder RC, Eloot S, Glorieux GLRL. Future avenues to decrease uremic toxin concentration. Am. J. Kidney Dis. 2016;67:664–676. doi: 10.1053/j.ajkd.2015.08.029. [DOI] [PubMed] [Google Scholar]
- 27.Wetmore JB, et al. The prevalence of and factors associated with chronic atrial fibrillation in Medicare/Medicaid-eligible dialysis patients. Kidney Int. 2012;81:469–476. doi: 10.1038/ki.2011.416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Herzog CA, et al. Cardiovascular disease in chronic kidney disease. A clinical update from Kidney Disease: Improving Global Outcomes (KDIGO) Kidney Int. 2011;80:572–586. doi: 10.1038/ki.2011.223. [DOI] [PubMed] [Google Scholar]
- 29.Wizemann V, et al. Atrial fibrillation in hemodialysis patients: Clinical features and associations with anticoagulant therapy. Kidney Int. 2010;77:1098–1106. doi: 10.1038/ki.2009.477. [DOI] [PubMed] [Google Scholar]
- 30.Genovesi S, et al. Prevalence of atrial fibrillation and associated factors in a population of long-term hemodialysis patients. Am. J. Kidney Dis. 2005;46:897–902. doi: 10.1053/j.ajkd.2005.07.044. [DOI] [PubMed] [Google Scholar]
- 31.Fujii H, Kim J-I, Yoshiya K, Nishi S, Fukagawa M. Clinical characteristics and cardiovascular outcomes of hemodialysis patients with atrial fibrillation: a prospective follow-up study. Am. J. Nephrol. 2011;34:126–134. doi: 10.1159/000329118. [DOI] [PubMed] [Google Scholar]
- 32.Winkelmayer WC, Patrick AR, Liu J, Brookhart MA, Setoguchi S. The increasing prevalence of atrial fibrillation among hemodialysis patients. J. Am. Soc. Nephrol. 2011;22:349–357. doi: 10.1681/ASN.2010050459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fuster V, et al. ACC/AHA/ESC 2006 Guidelines for the Management of Patients With Atrial Fibrillation: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Revise the 2001 Guidelines for the Management of Patients With Atrial Fibrillation): Developed in collaboration with the European Heart Rhythm Association and the Heart Rhythm Society. Circulation. 2006;114:e257–e354. doi: 10.1161/circ.114.4.257. [DOI] [PubMed] [Google Scholar]
- 34.Miyazaki T, et al. Indoxyl sulphate stimulates renal synthesis of transforming growth factor-beta 1 and progression of renal failure. Kidney Int. Suppl. 1997;63:S211–S214. [PubMed] [Google Scholar]
- 35.Neuzil P, et al. Electrical reconnection after pulmonary vein isolation is contingent on contact force during initial treatment: results from the EFFICAS I study. Circ. Arrhythm. Electrophysiol. 2013;6:327–333. doi: 10.1161/CIRCEP.113.000374. [DOI] [PubMed] [Google Scholar]
- 36.Ücer E, et al. A RAndomized Trial to compare the acute reconnection after pulmonary vein ISolation with Laser-BalloON versus radiofrequency Ablation: RATISBONA trial. J. Cardiovasc. Electrophysiol. 2018;29:733–739. doi: 10.1111/jce.13465. [DOI] [PubMed] [Google Scholar]
- 37.Rivara Matthew B., Zelnick Leila R., Hoofnagle Andrew N., Newitt Rick, Tracy Russell P., Kratz Mario, Weigle David S., Kestenbaum Bryan R. Diurnal and Long-term Variation in Plasma Concentrations and Renal Clearances of Circulating Markers of Kidney Proximal Tubular Secretion. Clinical Chemistry. 2017;63(4):915–923. doi: 10.1373/clinchem.2016.260117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Poesen Ruben, Mutsaers Henricus A. M., Windey Karen, van den Broek Petra H., Verweij Vivienne, Augustijns Patrick, Kuypers Dirk, Jansen Jitske, Evenepoel Pieter, Verbeke Kristin, Meijers Björn, Masereeuw Rosalinde. The Influence of Dietary Protein Intake on Mammalian Tryptophan and Phenolic Metabolites. PLOS ONE. 2015;10(10):e0140820. doi: 10.1371/journal.pone.0140820. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated and/or analysed during the current study are available from the corresponding author upon reasonable request.


