Abstract
Operant conditioning of the spinal stretch reflex (SSR) or its electrical analog, the H-reflex, is a valuable experimental paradigm for studying the acquisition and maintenance of a simple motor skill. The CNS substrate of this skill consists of brain and spinal cord plasticity that operates as a hierarchy—the learning experience induces plasticity in the brain that guides and maintains plasticity in the spinal cord. This is apparent in the two components of the skill acquisition: task-dependent adaptation, reflecting brain plasticity; and long-term change, reflecting gradual development of spinal plasticity. The inferior olive, cerebellum, sensorimotor cortex, and corticospinal tract (CST) are essential components of this hierarchy. The neuronal and synaptic mechanisms of the spinal plasticity are under study. Because acquisition of this skill changes the spinal cord, it can affect other skills, such as locomotion. Thus, it enables investigation of how the highly plastic spinal cord supports the acquisition and maintenance of a broad repertoire of motor skills throughout life. These studies have resulted in the negotiated equilibrium model of spinal cord function, which reconciles the spinal cord’s long-recognized reliability as the final common pathway for behaviors with its recently recognized ongoing plasticity. In accord with this model, appropriate H-reflex conditioning in a person with spasticity due to an incomplete spinal cord injury can trigger wider beneficial plasticity that markedly improves walking. H-reflex operant conditioning appears to provide a valuable new method for enhancing functional recovery in people with spinal cord injury and possibly other disorders as well.
Introduction
Throughout life, humans acquire and maintain numerous adaptive behaviors acquired through practice, commonly referred to as skills [1]. The wide variety of skills and the complex central nervous system (CNS) changes, or plasticity, underlying them make their investigation a central problem in basic and clinical neuroscience research.
For the past 35 years, operant conditioning of the spinal stretch reflex (SSR) or its electrical analog the H-reflex has provided a valuable experimental paradigm for investigating skill acquisition and the plasticity induced by the skill acquisition process (i.e., learning). In this brief review, we introduce the SSR, the H-reflex, and the operant conditioning protocol that can gradually increase or decrease SSR or H-reflex size. We then highlight the key contributions that this paradigm has made to our understanding of the plasticity that underlies the acquisition and maintenance of a simple motor skill. We go on to review the insights it has provided as to how the spinal cord supports the acquisition and maintenance of many motor skills, and how these insights have led to the negotiated equilibrium model of spinal cord function. Finally, we discuss the use of spinal reflex operant conditioning as a new therapeutic approach that can enhance recovery of locomotion or other important skills for people with spinal cord injury (SCI) or other neuromuscular disorders.
The simplest behavior: the spinal stretch reflex (SSR)
The SSR is the simplest behavior of the mammalian CNS; it is produced primarily by a two-neuron one-synapse pathway comprised of the Ia sensory afferent fiber from the muscle, the spinal α-motoneuron that activates the muscle, and the synapse between them (Fig. 1a) [2]. The knee-jerk reflex is probably the best-known example of an SSR: a tap to the patellar ligament stretches the quadriceps muscle and excites its Ia afferents; they in turn excite its motoneurons and thereby cause the muscle to contract.
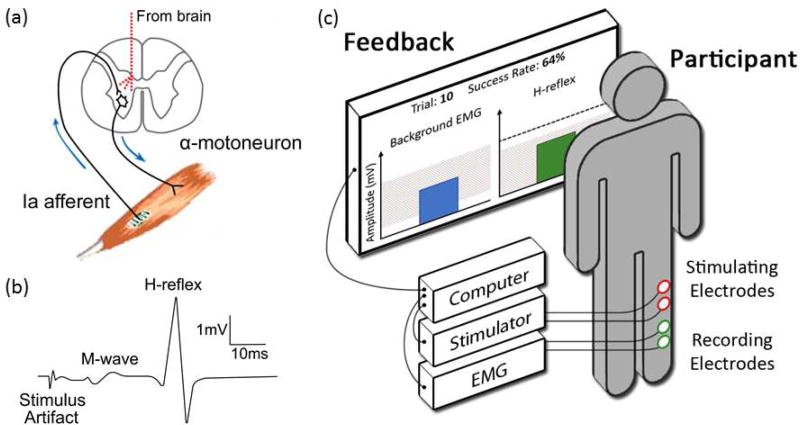
Figure 1.
(a) Main pathway of the spinal stretch reflex (SSR) and its electrical analog, the H-reflex. The pathway comprises the Ia afferent fiber from the muscle spindle, its synapse on the α-motoneuron, and the α-motoneuron itself. When the afferent is excited, it excites motoneurons innervating the muscle and its synergists. If it is excited by muscle stretch, the muscle response is the SSR; if it is excited by an electrical stimulus delivered to the nerve, the response is the H-reflex. The SSR and the H-reflex are typically measured by electromyography (EMG). While their pathway is entirely spinal, it is influenced by descending activity from the brain that affects the afferent synapse (presynaptically) and the motoneuron itself. Through this influence and the plasticity it induces in the spinal cord, the brain can gradually modify these spinal reflexes to increase rewards [11,46]. (b) Soleus EMG trace for a single trial, showing the stimulus artifact, the M-wave, and the H-reflex (modified from [13]). (c) Experimental setup for operant conditioning of the soleus H-reflex in human participants (based on experimental setups and figures in [9,12,18]). A pair of recording electrodes measures EMG activity from the soleus muscle; a pair of stimulating electrodes behind the knee elicits the H-reflex. The participant is asked to stand naturally and generate background EMG activity in the shaded range. After several seconds, a stimulus just above M-wave threshold elicits the M-wave and the H-reflex. (The threshold M-wave, or direct muscle response, results from excitation of a few large efferent fibers. Throughout the study, stimulus amplitude is continually adjusted to maintain the same M-wave size, thus ensuring that the effective stimulus strength does not change over the baseline, conditioning, and follow-up sessions.) If H-reflex size falls in the shaded range, the bar is green and the trial is a success; if it falls outside the range, the bar is red and the trial is not a success. The dashed horizontal line indicates the participant’s average H-reflex size from the control trials at the beginning of each experimental session. This illustration shows a successful down-conditioning trial; for up-conditioning the success range would be above a criterion.
It is also possible to excite the large Ia afferent fibers directly by delivering a weak electrical stimulus to the skin overlying the nerve. This elicits the H-reflex, an electrical analog of the SSR [3–5]. The weak stimulus also excites a few of the largest efferent motor fibers, leading to a direct muscle response, or M-wave. The M-wave is a purely peripheral response; due to its short pathway, it precedes the H-reflex (Fig. 1b) [4]. Both the M-wave and H-reflex are usually measured by recording electromyographic (EMG) activity from the muscle.
A simple skill: operantly conditioned change in SSR or H-reflex size
Animals and humans can modify the size of the SSR or H-reflex when they are rewarded for doing so—that is, through operant conditioning [6–11] (reviewed in [12,13]). The standard protocol consists of a sequence of conditioning trials. During each trial, the participant provides a specified level of background muscle activity, the reflex is elicited, and a reward follows immediately if the reflex (measured by EMG) satisfies a criterion value. After ~1000 trials, the reflex begins to change in the rewarded direction; and the amount of change grows gradually as conditioning trials continue over subsequent days and weeks. This modified reflex is a simple motor skill—an adaptive behavior acquired through practice [1,14].
As an example, consider the up-conditioning or down-conditioning of the H-reflex of the soleus muscle in humans (experimental setup shown in Fig. 1c) [9]. In the standard protocol, participants are asked to (1) stand with a natural posture and (2) generate a stable level of soleus muscle activity (as measured by EMG) for several seconds. If both requirements are fulfilled, an H-reflex is elicited by a pulse of electrical stimulation just above the threshold necessary to elicit an M-wave. In the control mode, no feedback on H-reflex size is provided. In the conditioning mode, visual feedback on H-reflex size is provided immediately: up-conditioning participants are visually rewarded when their H-reflex size is above a criterion; down-conditioning participants are visually rewarded when their H-reflex size is below a criterion. Over the course of several weeks (three 1-hr sessions/wk), ~80% of the participants change their H-reflex size in the rewarded direction. In the other ~20%, the H-reflex does not change. Figure 2a shows the course of H-reflex change in successful participants [9]. These participants acquire a new skill, a larger or smaller H-reflex. Furthermore, the change in H-reflex size persists for at least several months after conditioning ends (Fig. 2a, right-most data points from follow-up sessions).
Figure 2.
Operant conditioning of the human soleus H-reflex. Data and method are from [9]. There were 3 stages (each with multiple sessions) to the protocol: baseline (6 sessions), conditioning (24 sessions), and follow-up (4 sessions). Baseline and conditioning sessions were usually scheduled three times a week for ten weeks. Follow-up sessions occurred 10–14 days, one month, two months, and three months after the final conditioning session. During each conditioning or follow-up session, the participant first completed a set of 20 control trials without instruction to change H-reflex size or being provided feedback on H-reflex size. S/he then completed three 75-trial blocks of conditioning trials in which s/he was asked to increase (or decrease) H-reflex size and was provided with feedback immediately after each trial indicating whether H-reflex size met the size criterion (Fig. 1c). (a) Total impact of the conditioning protocol on H-reflex size. Average H-reflex size for conditioning trials during each session (with standard error bars) for all successful up-conditioning participants (upward triangles) and down-conditioning participants (downward triangles) over the three stages of the study. The two components of skill acquisition are shown in b and c. (b) Task-dependent adaptation appears early and remains the same thereafter. It was separated from long-term change in H-reflex size by subtracting average H-reflex size for the control trials at the beginning of the session (i.e., (c)) from average H-reflex size for the conditioning trials of the session (i.e., (a)). Thus, it indicates the change in reflex size that the participant learns to produce immediately when s/he is asked to do so. This learning occurs over the first few conditioning sessions (i.e., over the first ~1000 conditioning trials). (c) Long-term change begins later and grows gradually thereafter. It is indicated by the average H-reflex size for the control trials of each session. The figure is simplified from [9], which has full details.
This simple motor skill can be acquired by monkeys [11], mice [6], rats [7], and humans [8,9]. It is possible to operantly condition the SSR or H-reflex of arm [8,11] or leg muscles [9]. In both animals and humans, the percentage of participants in whom operant conditioning is successful is relatively consistent. In ~80% of participants the reflex changes in the rewarded direction, while in the remaining ~20% the reflex does not change.
It is generally not clear why conditioning fails to change the H-reflex in some participants, although some limited insights are available (e.g., [15]). Recent unpublished data suggest that the success rate rises well above 80% as the investigators’ skill in administering the conditioning protocol gradually improves (see [16]). One particularly interesting observation is that, in rats in which conditioning fails (i.e., H-reflex size measured in the conditioning protocol does not change), H-reflex size measured during locomotion may nevertheless change [17]. From an experimental perspective, animals in which the H-reflex does not change are a useful control population; they help in assessing whether a given anatomical or physiological change underlies successful conditioning (see The plasticity underlying this simple motor skill).
This simple skill—the acquisition of a larger or smaller SSR or H-reflex in the absence of any change in posture or in EMG activity (of the muscle that produces the reflex or in other muscles)—differs from the rapid reflex modulations associated with skills such as locomotion (e.g., [18]). In the latter instances, the rapid reflex modulations are features of a complex skill (e.g., locomotion) that has itself been acquired over a prolonged training period; the rapid reflex modulations cannot be readily produced in isolation from the complete skill.
The physical separation of the spinal cord from the brain and their connection through experimentally accessible tracts have made it possible to show that the simple skill of a larger or smaller H-reflex depends on plasticity in both structures. It has also made it possible to investigate how plasticity in the brain and spinal cord interact to produce a larger or smaller H-reflex. The key results of studies over the past 35 years are summarized in the next sections.
The two components of this simple motor skill
The acquisition of this simple motor skill has two distinct components: (1) a component (phase 1) that appears early and remains the same thereafter (Fig. 2b; task-dependent adaptation); and (2) a component (phase 2) that appears later and grows slowly thereafter (Fig. 2c; long-term change) [19]. In either biceps brachii SSR conditioning in monkeys or soleus H-reflex conditioning in humans, task-dependent adaptation appears after about 1000 trials [9,19]. Once it appears, it remains stable in magnitude and can be turned on and off at will; that is, it is present only when the participant is trying to change reflex size [9]. Thus, it functions in a manner similar to that of the long-latency transcortical responses to muscle stretch, which can also be turned on and off at will [20,21]. Long-term SSR or H-reflex change, as its name implies, takes longer to appear (e.g., 10–12 sessions (~2500 trials) in humans) and continues to grow as the training continues [9,19]. Once it appears, it is always present; and, when the reward criterion is reversed, the change reverses in the same gradual fashion [22].
The plasticity underlying this simple motor skill
Animal and human studies indicate that the two components of this simple motor skill—task-dependent adaptation and long-term change—reflect plasticity in the brain and the spinal cord, respectively [12,13]. Figure 3 summarizes our current knowledge of the brain and spinal cord structures involved in the acquisition and maintenance of this simple skill.
Figure 3.
Brain and spinal cord structures (including spinal interneurons (IN)) involved in H-reflex operant conditioning and the connections between them. The pink ellipses indicate sites of plasticity in the spinal cord. The striped pink ellipses indicate putative sites of plasticity in the brain [26]. As described in the text, this multi-site plasticity is believed to function as a hierarchy in which the plasticity in the brain induces and maintains the plasticity in the spinal cord. (Figure modified and updated from [39] and [13].)
This brain and spinal plasticity appears to operate as a hierarchy in which the plasticity in the brain guides and maintains the plasticity in the spinal cord [13,23]. The current hypothesis [24] is that the reward contingency generates inferior olive output [23,24] that produces cerebellar plasticity [25,26] that produces sensorimotor cortex plasticity [27] that produces corticospinal tract (CST) activity [28–30] that is responsible for task-dependent adaptation. Thus, plasticity in the brain is responsible for task-dependent adaptation in H-reflex size. The continued presence of this CST influence over subsequent days and weeks is thought to gradually change the spinal cord. This spinal cord plasticity is responsible for the long-term change in reflex size.
The plasticity in the spinal cord includes changes in motoneurons and interneurons within the spinal cord; these changes and their mechanisms are just beginning to be identified. In down-conditioned monkeys, for example, a positive shift in the firing threshold of soleus motoneurons largely accounts for the smaller H-reflex [31,32], as well as for the accompanying drop in motoneuron axonal conduction velocity (which is also found in down-conditioned rats [33]). These changes in firing threshold and conduction velocity do not occur in animals in which down-conditioning fails, implying that the threshold change underlies the smaller H-reflex. While the CST appears to convey the critical influence from the brain to the motoneurons that leads to the change in conduction velocity [28–30], it does not synapse directly on rat soleus motoneurons [34]; thus, its impact on the motoneurons is produced through one or more spinal interneurons. In rats, successful H-reflex down-conditioning is accompanied by increases in the number of identifiable GABAergic interneurons in the ventral horn and in the number of identifiable GABAergic terminals on soleus motoneurons [35,36]. In rats in which H-reflex down-conditioning fails, these changes do not occur; this suggests that GABAergic interneurons and terminals may convey the critical CST influence to the motoneurons. At present, there is less information concerning the possible mechanisms of up-conditioning. Up- and down-conditioning are not simply mirror images of each other; they appear to depend on different mechanisms.
The negotiated equilibrium model of spinal cord function
The growing evidence that the spinal cord changes throughout life indicates that the 19th-century model of a hard-wired spinal cord is no longer adequate. A new model is needed that reconciles the recently appreciated plasticity of the spinal cord with its long-recognized role as the final common pathway for motor behaviors. This need has provided the impetus for the negotiated equilibrium model of spinal cord function [3,12,13,37,38].
According to this new model, the substrate of each motor behavior comprises brain and spinal plasticity: the plasticity in the brain induces and maintains the plasticity in the spinal cord (Fig. 3, top). As each behavior occurs, deviations from its key features (e.g., right/left symmetry in step-cycle timing and hip height during locomotion) guide corrective changes in its substrate. All motor behaviors undergo this process concurrently. The aggregate process is a negotiation among the behaviors; they negotiate the properties of the spinal neurons and synapses that they all use. The ongoing negotiation maintains the spinal cord in an equilibrium that serves all the behaviors in the individual’s repertoire. When a new behavior expands the negotiation; compensatory plasticity preserves the key features of old behaviors. The new equilibrium serves all the behaviors in the expanded repertoire.
For example, when the new behavior of a larger or smaller H-reflex changes the spinal pathway of the reflex, it affects the kinematic and EMG details of locomotion [17,39]. At the same time, compensatory changes in other pathways preserve the key features of locomotion (e.g., right/left symmetry in step-cycle timing and hip height) [37,40]. This compensation is guided by the feedback the brain receives on the locomotor impact of the plasticity underlying the new behavior [37].
When a spinal cord injury has already impaired locomotion, the same impetus for compensatory plasticity is not present because locomotion is already abnormal. In this situation, appropriate H-reflex conditioning can actually improve locomotion [18,41–43].
Therapeutic use of this simple motor skill
The benefit of H-reflex conditioning for those with spinal cord injuries has been demonstrated in both rats and humans. In rats in which transection of the right lateral column of the spinal cord weakened the right soleus locomotor burst and produced a limp, up-conditioning of the right soleus H-reflex strengthened the burst and eliminated the limp [44]. Similarly, in humans in whom a partial spinal cord injury caused spasticity (i.e., exaggerated reflexes) that impaired locomotion, down-conditioning of the soleus H-reflex in the more impaired leg increased walking speed and reduced limping (Fig. 4) [18]. Furthermore, by targeting beneficial plasticity to an important spinal pathway and joining the ongoing negotiation among behaviors, appropriate H-reflex conditioning leads to wider plasticity that improves locomotor function in the muscles of both legs [18].
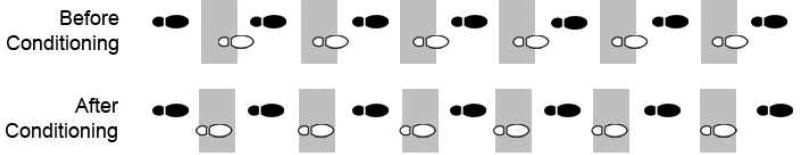
Figure 4.
Down-conditioning of the soleus H-reflex improves step-cycle symmetry in a person with spasticity due to chronic incomplete spinal cord injury. The figure shows successive step cycles before and after down-conditioning. The open footprint shows the steps of the more impaired leg (i.e., the leg in which the soleus H-reflex was down-conditioned). The filled footprint shows the steps of the other leg. The shaded areas indicate where the steps of the impaired leg should occur (i.e., midway between the steps of the other leg). Before H-reflex down-conditioning, the steps of the impaired leg are delayed; thus, the subject is limping. After H-reflex down-conditioning, the steps occur on time; the limp is not evident. (Modified from [18].)
These exciting results suggest that reflex operant conditioning protocols can provide an important new therapeutic approach. The unique specificity of this approach (it can target a specific spinal pathway and strengthen or weaken it as needed to address the functional impairment) and its ability to trigger wider beneficial plasticity suggest that it could complement conventional therapies and thereby improve functional recovery for people with spinal cord or brain injury, stroke, peripheral nerve injuries, or other chronic neuromuscular disorders [12,45].
Conclusions
Operant conditioning of the SSR or H-reflex provides a valuable experimental paradigm for investigating the acquisition and maintenance of a simple skill. The CNS substrate of this skill comprises plasticity in the brain and spinal cord that functions as a hierarchy—the learning experience creates the plasticity in the brain that in turn guides and maintains the plasticity in the spinal cord. This is apparent in the two components of the skill acquisition: task-dependent adaptation, which reflects brain plasticity; and long-term change, which reflects the gradual development of spinal plasticity. The inferior olive, cerebellum, sensorimotor cortex, and CST are essential components of this hierarchy. The spinal neuronal and synaptic mechanisms of the skill are under study. Because this skill changes the spinal cord, it affects other skills, such as locomotion. Thus, by enabling investigation of how the highly plastic spinal cord acquires and maintains a broad repertoire of motor skills throughout life, this simple skill has helped lead to the formulation of the negotiated equilibrium model of spinal cord function, which replaces the obsolete 19th-century model of a hard-wired spinal cord. In accord with this new model, appropriate H-reflex conditioning constitutes a new therapeutic approach that can enhance recovery of function in people after spinal cord injury.
Highlights.
The spinal stretch reflex (SSR) and H-reflex can be operantly conditioned.
The resulting change in SSR or H-reflex size is a simple motor skill.
The size change consists of task-dependent adaptation plus long-term change.
This skill depends on a hierarchy of brain and spinal cord plasticity.
H-reflex conditioning can enhance locomotor recovery after spinal cord injury.
Acknowledgments
We thank Dr. Amir Eftekhar, Ms. Susan Heckman, Dr. Jonathan Carp, and Ms. Jennifer Barnes for their valuable comments on the paper. We also thank Dr. Eftekhar and Dr. Aiko Thompson for their useful advice and assistance in regard to the figures.
The National Center for Adaptive Neurotechnologies is supported by the National Institute of Biomedical Imaging and Bioengineering of the NIH (Grant 1P41EB018783 (JRW)). Work in the authors’ laboratory has also been supported by NIH grants HD36020 (XYChen), NS061823 (JRW & XYChen), NS22189 (JRW), HD32571 (AWEnglish), by VA Merit Award 1 I01 BX002550 (JRW), and by the New York State Spinal Cord Injury Research Board (SCIRB).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
BIBLIOGRAPHY
- 1.Compact Oxford English dictionary. second. Oxford University Press; 1993. [Google Scholar]
- 2.Misiaszek JE. The H-reflex as a tool in neurophysiology: Its limitations and uses in understanding nervous system function. Muscle Nerve. 2003;28:144–160. doi: 10.1002/mus.10372. [DOI] [PubMed] [Google Scholar]
- 3**.Pierrot-Deseilligny E, Burke D. The circuitry of the human spinal cord: spinal and corticospinal mechanisms of movement. Cambridge University Press; 2012. Outstanding comprehensive resource for scientists and clinicians studying spinal pathways and their plasticity. Includes authoritative descriptions of methodologies, spinal pathways, and the role of spinal pathways in movement. [Google Scholar]
- 4.Tucker KJ, Tuncer M, Türker KS. A review of the H-reflex and M-wave in the human triceps surae. Hum Mov Sci. 2005;24:667–688. doi: 10.1016/j.humov.2005.09.010. [DOI] [PubMed] [Google Scholar]
- 5.Zehr PE. Considerations for use of the Hoffmann reflex in exercise studies. Eur J Appl Physiol. 2002;86:455–468. doi: 10.1007/s00421-002-0577-5. [DOI] [PubMed] [Google Scholar]
- 6.Carp JS, Tennissen AM, Chen XY, Wolpaw JR. H-reflex operant conditioning in mice. J Neurophysiol. 2006;96:1718–1727. doi: 10.1152/jn.00470.2006. [DOI] [PubMed] [Google Scholar]
- 7.Chen XY, Wolpaw JR. Operant conditioning of H-reflex in freely moving rats. J Neurophysiol. 1995;73:411–415. doi: 10.1152/jn.1995.73.1.411. [DOI] [PubMed] [Google Scholar]
- 8*.Evatt ML, Wolf SL, Segal RL. Modification of human spinal stretch reflexes: preliminary studies. Neurosci Lett. 1989;105:350–355. doi: 10.1016/0304-3940(89)90646-0. First demonstration of SSR or H-reflex operant conditioning in humans. [DOI] [PubMed] [Google Scholar]
- 9.Thompson AK, Chen XY, Wolpaw JR. Acquisition of a simple motor skill: Task-dependent adaptation plus long-term change in the human soleus H-reflex. J Neurosci. 2009;29:5784–5792. doi: 10.1523/JNEUROSCI.4326-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wolpaw JR. Operant conditioning of primate spinal reflexes: the H-reflex. J Neurophysiol. 1987;57:443–459. doi: 10.1152/jn.1987.57.2.443. [DOI] [PubMed] [Google Scholar]
- 11.Wolpaw JR, Kieffer VA, Seegal RF, Braitman DJ, Sanders MG. Adaptive plasticity in the spinal stretch reflex. Brain Res. 1983;267:196–200. doi: 10.1016/0006-8993(83)91059-4. [DOI] [PubMed] [Google Scholar]
- 12**.Thompson AK, Wolpaw JR. Operant conditioning of spinal reflexes: from basic science to clinical therapy. Front Integr Neurosci. 2014;8 doi: 10.3389/fnint.2014.00025. Succinct comprehensive review of spinal reflex operant conditioning. Includes overview of extensive methodological and mechanistic studies as well as the recent clinical studies indicating its potential therapeutic applications. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13**.Wolpaw JR. What can the spinal cord teach us about learning and memory? The Neuroscientist. 2010;16:532–549. doi: 10.1177/1073858410368314. This comprehensive review introduces the ‘negotiated equilibrium’ model of spinal cord function. [DOI] [PubMed] [Google Scholar]
- 14.Shmuelof L, Krakauer JW. Are we ready for a natural history of motor learning? Neuron. 2011;72:469–476. doi: 10.1016/j.neuron.2011.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wolpaw JR, Herchenroder PA, Carp JS. Operant conditioning of the primate H-reflex: factors affecting the magnitude of change. Exp Brain Res. 1993;97:31–39. doi: 10.1007/BF00228815. [DOI] [PubMed] [Google Scholar]
- 16.Thompson AK, Wolpaw JR. Sensorimotor Rehabilitation - At the Crossroads of Basic and Clinical Sciences. Elsevier; 2015. Targeted neuroplasticity for rehabilitation; pp. 157–172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen Y, Chen XY, Jakeman LB, Schalk G, Stokes BT, Wolpaw JR. The interaction of a new motor skill and an old one: H-reflex conditioning and locomotion in rats. J Neurosci. 2005;25:6898–6906. doi: 10.1523/JNEUROSCI.1684-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18**.Thompson AK, Pomerantz FR, Wolpaw JR. Operant conditioning of a spinal reflex can improve locomotion after spinal cord injury in humans. J Neurosci. 2013;33:2365–2375. doi: 10.1523/JNEUROSCI.3968-12.2013. First study indicating that appropriate H-reflex conditioning can improve locomotion in people with chronic incomplete spinal cord injury. The improvements were apparent to the participants in their daily lives. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wolpaw JR, O’Keefe JA. Adaptive plasticity in the primate spinal stretch reflex: evidence for a two-phase process. J Neurosci. 1984;4:2718–2724. doi: 10.1523/JNEUROSCI.04-11-02718.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hammond PH. The influence of prior instruction to the subject on an apparently involuntary neuro-muscular response. J Physiol. 1956;132:17. [PubMed] [Google Scholar]
- 21.Lee RG, Tatton WG. Motor responses to sudden limb displacements in primates with specific CNS lesions and in human patients with motor system disorders. Can J Neurol Sci. 1975;2:285–293. doi: 10.1017/s0317167100020382. [DOI] [PubMed] [Google Scholar]
- 22.Wolpaw JR. Adaptive plasticity in the primate spinal stretch reflex: reversal and redevelopment. Brain Res. 1983;278:299–304. doi: 10.1016/0006-8993(83)90259-7. [DOI] [PubMed] [Google Scholar]
- 23*.Chen XY, Wang Y, Chen Y, Chen L, Wolpaw JR. The inferior olive is essential for long-term maintenance of a simple motor skill. J Neurophysiol. 2016;116:1946–1955. doi: 10.1152/jn.00085.2016. Demonstrates the critical role of the inferior olive in maintenance of a down-conditioned H-reflex and summarizes current understanding of the hierarchy of plasticity that underlies this simple motor skill. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chen XY, Wang Y, Chen Y, Chen L, Wolpaw JR. Ablation of the inferior olive prevents H-reflex down-conditioning in rats. J Neurophysiol. 2016;115:1630–1636. doi: 10.1152/jn.01069.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen XY, Wolpaw JR. Ablation of cerebellar nuclei prevents H-reflex down-conditioning in rats. Learn Mem. 2005;12:248–254. doi: 10.1101/lm.91305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wolpaw JR, Chen XY. The cerebellum in maintenance of a motor skill: A hierarchy of brain and spinal cord plasticity underlies H-reflex conditioning. Learn Mem. 2006;13:208–215. doi: 10.1101/lm.92706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen XY, Carp JS, Chen L, Wolpaw JR. Sensorimotor cortex ablation prevents H-reflex up-conditioning and causes a paradoxical response to down-conditioning in rats. J Neurophysiol. 2006;96:119–127. doi: 10.1152/jn.01271.2005. [DOI] [PubMed] [Google Scholar]
- 28.Chen XY, Wolpaw JR. Dorsal column but not lateral column transection prevents down-conditioning of H-reflex in rats. J Neurophysiol. 1997;78:1730–1734. doi: 10.1152/jn.1997.78.3.1730. [DOI] [PubMed] [Google Scholar]
- 29.Chen XY, Carp JS, Chen L, Wolpaw JR. Corticospinal tract transection prevents operantly conditioned H-reflex increase in rats. Exp Brain Res. 2002;144:88–94. doi: 10.1007/s00221-002-1026-8. [DOI] [PubMed] [Google Scholar]
- 30.Chen XY, Wolpaw JR. Probable corticospinal tract control of spinal cord plasticity in the rat. J Neurophysiol. 2002;87:645–652. doi: 10.1152/jn.00391.2001. [DOI] [PubMed] [Google Scholar]
- 31.Carp JS, Wolpaw JR. Motoneuron plasticity underlying operantly conditioned decrease in primate H-reflex. J Neurophysiol. 1994;72:431–442. doi: 10.1152/jn.1994.72.1.431. [DOI] [PubMed] [Google Scholar]
- 32.Halter JA, Carp JS, Wolpaw JR. Operantly conditioned motoneuron plasticity: possible role of sodium channels. J Neurophysiol. 1995;73:867–871. doi: 10.1152/jn.1995.73.2.867. [DOI] [PubMed] [Google Scholar]
- 33.Carp JS, Chen XY, Sheikh H, Wolpaw JR. Operant conditioning of rat H-reflex affects motoneuron axonal conduction velocity. Exp Brain Res. 2001;136:269–273. doi: 10.1007/s002210000608. [DOI] [PubMed] [Google Scholar]
- 34.Yang H-W, Lemon RN. An electron microscopic examination of the corticospinal projection to the cervical spinal cord in the rat: lack of evidence for cortico-motoneuronal synapses. Exp Brain Res. 2003;149:458–469. doi: 10.1007/s00221-003-1393-9. [DOI] [PubMed] [Google Scholar]
- 35.Wang Y, Pillai S, Wolpaw JR, Chen XY. Motor learning changes GABAergic terminals on spinal motoneurons in normal rats. Eur J Neurosci. 2006;23:141–150. doi: 10.1111/j.1460-9568.2005.04547.x. [DOI] [PubMed] [Google Scholar]
- 36.Wang Y, Pillai S, Wolpaw JR, Chen XY. H-reflex down-conditioning greatly increases the number of identifiable GABAergic interneurons in rat ventral horn. Neurosci Lett. 2009;452:124–129. doi: 10.1016/j.neulet.2009.01.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37**.Chen Y, Chen L, Wang Y, Chen XY, Wolpaw JR. Why new spinal cord plasticity does not disrupt old motor behaviors. J Neurosci. 2017;37:8198–8206. doi: 10.1523/JNEUROSCI.0767-17.2017. Shows that removal of sensory feedback allows H-reflex operant conditioning to impair locomotion in rats. Provides critical support for the negotiated equilibrium model of spinal cord function. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wolpaw JR, Tennissen AM. Activity-dependent spinal cord plasticity in health and disease. Annu Rev Neurosci. 2001;24:807–843. doi: 10.1146/annurev.neuro.24.1.807. [DOI] [PubMed] [Google Scholar]
- 39*.Nielsen J, Crone C, Hultborn H. H-reflexes are smaller in dancers from The Royal Danish Ballet than in well-trained athletes. Eur J Appl Physiol. 1993;66:116–121. doi: 10.1007/BF01427051. Strong evidence for the role of adaptive changes in spinal reflexes in the acquisition of complex motor skills. [DOI] [PubMed] [Google Scholar]
- 40.Makihara Y, Segal RL, Wolpaw JR, Thompson AK. Operant conditioning of the soleus H-reflex does not induce long-term changes in the gastrocnemius H-reflexes and does not disturb normal locomotion in humans. J Neurophysiol. 2014;112:1439–1446. doi: 10.1152/jn.00225.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chen Y, Chen L, Liu R, Wang Y, Chen XY, Wolpaw JR. Locomotor impact of beneficial or nonbeneficial H-reflex conditioning after spinal cord injury. J Neurophysiol. 2014;111:1249–1258. doi: 10.1152/jn.00756.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen Y, Chen L, Wang Y, Wolpaw JR, Chen XY. Persistent beneficial impact of H-reflex conditioning in spinal cord-injured rats. J Neurophysiol. 2014;112:2374–2381. doi: 10.1152/jn.00422.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Manella KJ, Roach KE, Field-Fote EC. Operant conditioning to increase ankle control or decrease reflex excitability improves reflex modulation and walking function in chronic spinal cord injury. J Neurophysiol. 2013;109:2666–2679. doi: 10.1152/jn.01039.2011. [DOI] [PubMed] [Google Scholar]
- 44.Chen Y, Chen XY, Jakeman LB, Chen L, Stokes BT, Wolpaw JR. Operant conditioning of H-reflex can correct a locomotor abnormality after spinal cord injury in rats. J Neurosci. 2006;26:12537–12543. doi: 10.1523/JNEUROSCI.2198-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chen Y, Wang Y, Chen L, Sun C, English AW, Wolpaw JR, Chen XY. H-reflex up-conditioning encourages recovery of EMG activity and H-reflexes after sciatic nerve transection and repair in rats. J Neurosci. 2010;30:16128–16136. doi: 10.1523/JNEUROSCI.4578-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wolpaw JR. The complex structure of a simple memory. Trends Neurosci. 1997;20:588–594. doi: 10.1016/s0166-2236(97)01133-8. [DOI] [PubMed] [Google Scholar]