Abstract
Background:
Estimation of the amount of blood products required during liver transplantation can help provision of adequate blood supply, minimize transfusion-associated complications, and plan for preventive measures in high risk patients.
Objective:
To investigate independent predictors of peri-operative blood product transfusion and its impact on short-term survival of liver transplant recipients.
Methods:
In a cross-sectional study, old charts of patients who underwent liver transplantation between March 2003 and March 2013 at Namazi Hospital, Shiraz, Iran, were reviewed. The mean amount of blood product utilized during surgery and hospital stay and the related factors, including demographic characteristics, pre-transplant laboratory data, pre-transplant clinical data, operation data, and post-transplantation data were recorded.
Results:
We studied 1198 patients who underwent liver transplantation. The mean±SD amounts of red blood cells, fresh frozen plasma, and platelet transfusion during surgery and hospital stay were 2.67±3.5, 2.06±3.8, and 1.6±3.8 units, respectively. The mortality rate was significantly higher in patients who received high amounts of blood products (p<0.001). The mean amount of blood products’ utilized during operation was significantly (p<0.001) decreased from 2003 to 2013.The mean amount of packed cell usage during operation and hospital stay was significantly (p<0.001) correlated with age, technique of surgery, serum albumin level, cirrhosis, blood urea nitrogen, length of operation, and prothrombin time.
Conclusion:
Pre-operative factors may predict blood transfusion requirements in patients undergoing liver transplantation. Therefore, evaluation of patients before operation should be considered to provide adequate blood supply and minimize transfusion-associated complications. Understanding pre-operative factors associated with rate of transfusion may help us to best utilize the limited available blood resources.
Key Words: Blood loss, Liver transplantation, Blood product transfusion, Survival rate
INTRODUCTION
Liver transplantation has significantly improved the outcome of patients with acute liver failure and end-stage liver diseases (ESLD) [1, 2]. This procedure, however, requires a considerable amount of blood product transfusion because of the vascular and coagulopathic nature of the liver. Bleeding due to portal hypertension, complexity of the procedure requiring multiple anastomoses and vascular transections, predispose patients to massive hemorrhage [3]. Blood transfusion rate is considered a poor prognostic factor, which decreases patient and graft survival [4]. Poor outcome in patients may be induced by more blood loss during surgery or direct negative effects of blood transfusion such as non-infectious and infectious complications [5]. Blood transfusion increases the risk of post-operative complications, infections, longer hospital stay, respiratory complications, cytomegalovirus infection, graft failure, and patient mortality [6]. Furthermore, a previous study suggests that higher red cells (RBCs) transfusion is associated with higher level of immunosuppression, higher frequency of alloimmunization, and lower patient survival [7]. Nevertheless, it is not clear whether these complications are solely because of receiving blood products or are consequences of more complicated surgeries requiring more transfusions.
Liver transplantation has been expanded significantly during recent years highlighting the role of a good support of blood products for transfusion requirements during and after the surgery. While reservation of additional units of packed cells, platelets, plasma, and cryoprecipitate for patients who require more blood transfusion may be necessary, overestimation of the amount of blood products required may compromise blood adequacy. Therefore, a good communication between transplant team and blood bank is of paramount importance to provide enough blood products for the procedure. On the other hand, understanding pre-operative factors associated with rate of transfusion may help us to better manage the limited available blood resources [8].
Previous studies have shown a correlation between intra-operative blood loss and a number of pre-operative variables including demographic characteristics, coagulation status, and biochemical parameters [2, 3, 6]. Intra-operative blood loss has also been reported to be associated with surgical technique, surgeon’ experience, length of surgery, and hemostatic status of patient. However, other studies have failed to find any correlation [5, 7].
No transfusion guideline has so far been developed for liver transplantation. Pre-operative evaluation should include factors influencing blood requirement during the operation. Estimation of the blood products required during the procedure can help us to provide adequate blood supply, minimize transfusion-associated complications, and plan ahead of specific measures in high risk patients. Considering the importance of blood transfusion rate on patient outcome, the present study aimed to investigate the rate of blood product transfusion and its related factors in patients undergoing liver transplantation.
PATIENTS AND METHODS
A cross-sectional study was conducted between March 2003 and March 2013 at Liver Transplantation Unit, Namazi Hospital, Shiraz, Iran. All patients who underwent liver transplantation during this period were included in the study (n=1198).
The patients’ data were collected using a well-designed data collection form, including demographic characteristics (age, sex, marital status); pre-transplant laboratory data including serum aspartate aminotransferase (AST), serum alanine aminotransferase (ALT), alkaline phosphatase, bilirubin, prothrombin time (PT), blood urea nitrogen (BUN), creatinine, hemoglobin (Hb), platelet count, hepatitis B, and hepatitis C; pre-transplant clinical data (history of previous surgery, length of disease, underlying disease, ascites, encephalopathy, and cirrhosis); operation data (length of operation); length of hospital stay; and post-transplantation one-year survival. The number of units of blood products including RBC, fresh frozen plasma (FFP), and platelet transfused during surgery and hospitalization were measured and recorded.
The study protocol was approved by the Ethics Committee, Shiraz University of Medical Sciences. The study protocol was carried out in accordance with the Helsinki declaration as revised in Seoul 2008.
Statistical Analysis
Data were analyzed using SPSS® ver 22.0 for Windows® (Chicago, IL, USA). Independent-sample Student’s t test, and one-way ANOVA were used for statistical analysis. Poisson regression was used for evaluating the effect of studied variables (demographic characteristics, pre-transplant laboratory data, pre-transplant clinical data, operation data, and post-transplantation data) on the number of blood product utilized. A p value <0.05 was considered statistically significant.
RESULTS
The mean±SD age of the patients was 31.1±16.8 (range: 1–75) years. Almost two-thirds (63.2%, n=757) of patients were male; 97 (8.2%) had previous history of surgery, 129 (10.9%) had encephalopathy, 416 (35.2%) had esophageal varices, and 328 (27.3%) had jaundice. The mean±SD duration of liver disease was 52.9±56 (range: 1–420) months. The mean±SD duration of operation was 390.5±85.1 minutes. More than 80% (n=966) of recipients stayed alive after one year post-operative.
The mean amounts of blood products utilized during surgery and hospital are summarized in Table 1. The mortality rate was significantly higher in recipients who received higher amounts of blood products (Table 2).
Table 1.
The mean±SEM amount of blood products utilized during surgery and hospital stay
| Blood product (unit) | During operation | During hospital stay | Total |
|---|---|---|---|
| RBC | 2.64±3.59 | 1.51±2.90 | 4.16±4.68 |
| FFP | 2.11±3.91 | 1.20±3.12 | 3.31±5.13 |
| Platelet | 1.66±3.96 | 0.47±2.05 | 2.13±1.39 |
RBC: Red blood cell, FFP: Fresh frozen plasma
Table 2.
The outcome of patients according to the amount of blood and blood products transfusion. Values are mean±SEM amount of blood products utilized.
| Blood product (unit) | Alive patients | Dead patients | Difference (95% CI) |
|---|---|---|---|
| Total RBC | 3.74±4.23 | 5.92±5.91 | 2.18 (1.47–2.88) |
| Total FFP | 3.06±4.56 | 4.36±6.99 | 1.30 (0.51–2.08) |
| Total platelet | 1.74±4.23 | 3.85±6.87 | 2.11 (1.36–2.85) |
RBC: Red blood cell, FFP: Fresh frozen plasma, CI: Confidence interval
Poisson regression analysis revealed a significant relationship between number of units of RBC utilized and age, albumin, presence of cirrhosis, blood urea nitrogen, length of operation, and prothrombin time (Table 3). It also showed a significant relationship between the number of units of FFP utilized and age, type of surgery, length of operation, existence of encephalopathy, existence of cirrhosis, blood urea nitrogen, albumin, and prothrombin time (Table 3). There was also a significant relationship between number of platelet units utilized and age, length of operation, pre-transplantation serum albumin, pre-transplantation BUN, and prothrombin time (Table 3).
Table 3.
Estimation parameter of factors associated with blood products utilized during operation and hospital stay determined by Poisson regression
| Blood product (dependent variable) | Independent variables | Parameter estimation | Standard deviation | p value |
|---|---|---|---|---|
| Total Red blood cell | Age | 0.47 | 0.009 | <0.001 |
| Cirrhosis | 2.29 | 0.79 | 0.004 | |
| Duration of surgery | 0.02 | 0.001 | <0.001 | |
| PT | 0.05 | 0.016 | <0.001 | |
| Albumin | 0.34 | 0.09 | <0.001 | |
| BUN | 0.06 | 0.011 | <0.001 | |
| Total Fresh frozen plasma | Encephalopathy | 1.04 | 0.50 | 0.037 |
| Cirrhosis | 2.74 | 0.96 | 0.004 | |
| Duration of surgery | 0.011 | 0.002 | <0.001 | |
| ALT | 0.001 | <0.001 | 0.009 | |
| PT | 0.072 | 0.02 | <0.001 | |
| Albumin | 0.24 | 0.10 | 0.024 | |
| BUN | 0.048 | 0.013 | <0.001 | |
| Total platelet | Duration of surgery | 0.012 | 0.002 | <0.001 |
| PT | 0.039 | 0.21 | 0.059 | |
| Albumin | 0.21 | 0.11 | 0.054 | |
| BUN | 0.05 | 0.014 | <0.001 |
The mean amount of blood products utilized was not correlated with the underlying diseases.
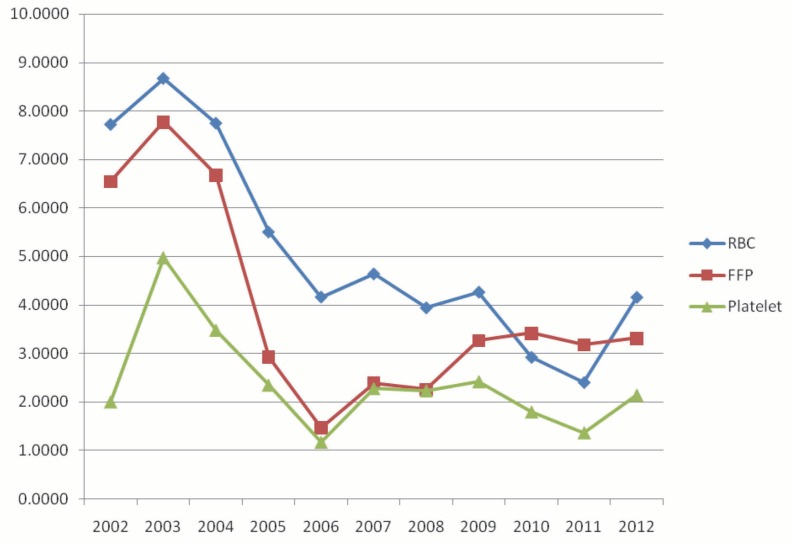
The mean amount of blood products utilized during operation significantly (p<0.001) decreased from 2003 to 2013 (Fig 1).
Figure 1.
Mean amount of blood products utilized from 2002–2011.
DISCUSSION
We found that total RBCs units transfused was associated with age, pre-transplantation serum albumin level, presence of liver cirrhosis, serum BUN, length of operation, and PT. In this study, mortality was higher in those receiving higher amounts of blood products. There was also a decreasing trend in blood products utilized between 2003 and 2013.
Liver transplantation has improved outcome of patients with ESLD and acute liver failure. Although the outcome of liver recipients has improved a lot over recent years, complications that impose significant morbidity and mortality in post-transplantation period are still prevalent [9-12]. Patients with cirrhosis may develop several complications before transplantation [13]. Therefore, a through pre-transplantation evaluation of patient should be performed to early diagnose and treat these complications [14].
Blood transfusion is one of the factors influencing graft and patient survival. Poor outcome of patients who received more blood transfusion might be attributed to more blood loss during transplantation, or adverse transfusion reaction, infectious contamination of blood products, or immune modulation of the transfused patient [15]. One-year survival rate is decreased in patients who received any amounts of FFP or more than four units of RBC [16]. However, blood transfusions are not the only variables that can influence the survival rate.
Identifying the factors associated with transfusion requirements can help us planning for sufficient blood. The lower blood products utilized might be due to using different anesthetic and surgical methods and better blood salvage techniques [17]. In our study, blood utilization has significantly decreased from 6.09 to 2.67 units. Similar finding was reported from previous studies [18]. This reduction in blood utilization may be attributed to advances in surgical and anesthetic methods, improved graft preservation and organ allocation, better patient monitoring using the piggyback technique for surgery, and better intra-operative blood salvage [19].
Previous studies found that the amount of blood usage may be associated with the severity of the disease, etiology of liver disease, pre-operative PT, history of abdominal operation, duration of surgery, factor V levels, presence of ascites, elevated serum creatinine, elevated BUN, age, MELD score, initial fibrinogen, initial central venous pressure, total anesthesia time, presence of other hematological abnormalities such as anemia and thrombocytopenia, and history of upper abdominal surgery [2, 10, 12-18]. Furthermore, portal vein hypoplasia, decreased donor liver size, use of a graft with a raw surface, poor graft-recipient body weight ratio, poor graft preservation, prolonged time of cold ischemia, and poor quality cadaveric graft might develop non-functional liver after transplantation requiring more complex surgeries needing more amounts of transfusion [16, 19].
Whether presence and degrees of coagulopathy are related to transfusion requirement is controversial. We showed that pre-operative PT was correlated with higher transfusion rate. Similar findings were reported in another study [20]. However, other studies found no association between pre-operative coagulation indices and blood transfusion requirement [21, 22].
We found that the mean RBC units used was correlated with BUN. Previous studies also showed renal function as a predictive factor for estimating blood requirement [23, 24]. Renal dysfunction may cause anemia, platelet dysfunction, defective hemostasis, and bleeding, which may cause more blood utilization during liver transplantation [25].
Previous studies indicate a positive correlation between MELD score and blood products consumption [26]. High MELD score is an indicator of multisystem dysfunction and coagulopathy [26]. In general, patients with more advanced liver disease are more prone to blood loss because of portal hypertension and defective hemostasis. However, another study shows that transfusion requirement is not related to MELD score, history of previous upper abdominal surgery, pre-operative coagulation defects, and hemoglobin level [27].
Our study revealed no correlation between hemoglobin level and blood utilization. Similar result was found in another study [21]. However, in other previous studies, a negative correlation was observed between pre-operative hemoglobin level and blood requirement [28, 29]. Another study shows that correction of hemoglobin level before operation might reduce the transfusion need during transplantation [29].
We found that FFP utilization was higher in patients with encephalopathy, cirrhosis, longer duration of surgery, elevated AST, increased PT, and lower serum albumin level. A previous study demonstrates that INR and PT, which represent patients’ hemostatic status, were positively correlated with FFP consumption, which was in agreement with the findings of the current study [30]. Another study shows that FFP consumption is negatively correlated with hemoglobin level, but this correlation was not confirmed in our study [30]. Another study shows that post-transplantation survival of patients is related to the number of FFP units transfused to patients [31]. Besides, the authors found that survival rate is more correlated with FFP transfusion compared with RBC transfusion [31]. Therefore, FFP must be transfused cautiously.
In conclusion, it seems that blood transfusion requirement in patients undergoing liver transplantation was correlated with several pre-operative factors. Therefore, decision for blood requirement must be individualized for each patient by evaluating pre-operative factors to provide adequate blood supply and minimize transfusion-associated complications.
ACKNOWLEDGMENTS
This study was financially supported by IBTO research center. Hereby, the authors would like to thank Parisa Jonghorban, Azadeh Mosallai, and Sahar Dehbidi for their assistance with data collection and entry. They are also grateful to Dr. Jamshid Jamali for data analysis. Thanks also go to Ms. Afsaneh Keivanshekouh at the Research Improvement Center of Shiraz University of Medical Sciences for improving the use of English in the manuscript.
References
- 1.Jadlowiec CC, Taner T. Liver transplantation: Current status and challenges. World J Gastroenterol. 2016;22:4438–45. doi: 10.3748/wjg.v22.i18.4438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lankarani KB, Eshraghian K, Malek-Hosseini SA, et al. Outcomes of liver transplantation for patients with acute liver failure. Arch Iran Med. 2013;16:64–7. [PubMed] [Google Scholar]
- 3.Clevenger B, Mallett SV. Transfusion and coagulation management in liver transplantation. World J Gastroenterol. 2014;20:6146–58. doi: 10.3748/wjg.v20.i20.6146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Reichert B, Kaltenborn A, Becker T, et al. Massive blood transfusion after the first cut in liver transplantation predicts renal outcome and survival. Langenbecks Arch Surg. 2014;399:429–40. doi: 10.1007/s00423-014-1181-y. [DOI] [PubMed] [Google Scholar]
- 5.Lee D, Gil W, Lee H, Lee K, Lee S, Kim S, et al. Factors affecting graft survival after living donor liver transplantation. Transplant Proc. 2004;36:2255–6. doi: 10.1016/j.transproceed.2004.08.073. [DOI] [PubMed] [Google Scholar]
- 6.Cacciarelli TV, Keeffe EB, Moore DH, et al. Effect of intraoperative blood transfusion on patient outcome in hepatic transplantation. Archives of Surgery. 1999;134:25–9. doi: 10.1001/archsurg.134.1.25. [DOI] [PubMed] [Google Scholar]
- 7.Boyd SD, Stenard F, Lee DK, et al. Alloimmunization to red blood cell antigens affects clinical outcomes in liver transplant patients. Liver Transpl. 2007;13:1654–61. doi: 10.1002/lt.21241. [DOI] [PubMed] [Google Scholar]
- 8.Donohue CI, Mallett SV. Reducing transfusion requirements in liver transplantation. World J Transplant. 2015;5:165–82. doi: 10.5500/wjt.v5.i4.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dehghani SM, Taghavi SA, Eshraghian A, et al. Hyperlipidemia in Iranian liver transplant recipients: prevalence and risk factors. J Gastroenterol. 2007;42:769–74. doi: 10.1007/s00535-007-2092-2. [DOI] [PubMed] [Google Scholar]
- 10.Dehghani SM, Nikeghbalian S, Eshraghian A, et al. New-onset diabetes mellitus presenting with diabetic ketoacidosis after pediatric liver transplantation. Pediatr Transplant. 2009;13:536–9. doi: 10.1111/j.1399-3046.2008.01117.x. [DOI] [PubMed] [Google Scholar]
- 11.Zahmatkeshan M, Geramizadeh B, Eshraghian A, et al. Transplant Proc De novo fatty liver due to vascular complications after liver. transplantation. 2011;43:615–7. doi: 10.1016/j.transproceed.2011.01.023. [DOI] [PubMed] [Google Scholar]
- 12.Lankarani KB, Eshraghian A, Nikeghbalian S, et al. New onset diabetes and impaired fasting glucose after liver transplant: risk analysis and the impact of tacrolimus dose. Exp Clin Transplant. 2014;12:46–51. doi: 10.6002/ect.2013.0047. [DOI] [PubMed] [Google Scholar]
- 13.Eshraghian A, Taghavi SA. Systematic review: endocrine abnormalities in patients with liver cirrhosis. Arch Iran Med. 2014;17:713–21. [PubMed] [Google Scholar]
- 14.Dehghani SM, Aleyasin S, Honar N, et al. Pulmonary evaluation in pediatric liver transplant candidates. Indian J Pediatr. 2011;78:171–5. doi: 10.1007/s12098-010-0216-9. [DOI] [PubMed] [Google Scholar]
- 15.Massicotte L, Beaulieu D, Roy J-D, et al. MELD score and blood product requirements during liver transplantation: no link. Transplantation. 2009;87:1689–94. doi: 10.1097/TP.0b013e3181a5e5f1. [DOI] [PubMed] [Google Scholar]
- 16.Massicotte L, Sassine M-P, Lenis S, et al. Survival rate changes with transfusion of blood products during liver transplantation. Can J Anaesth. 2005;52:148–55. doi: 10.1007/BF03027720. [DOI] [PubMed] [Google Scholar]
- 17.Frasco PE, Poterack KA, Hentz JG, Mulligan DC. A comparison of transfusion requirements between living donation and cadaveric donation liver transplantation: relationship to model of end-stage liver disease score and baseline coagulation status. Anesthesia & Analgesia. 2005;101:30–7. doi: 10.1213/01.ANE.0000155288.57914.0D. [DOI] [PubMed] [Google Scholar]
- 18.Deakin M, Gunson BK, Dunn JA, et al. Factors influencing blood transfusion during adult liver transplantation. Ann R Coll Surg Engl. 1993;75:339. [PMC free article] [PubMed] [Google Scholar]
- 19.Massicotte L, Thibeault L, Roy A. Classical Notions of Coagulation Revisited in Relation with Blood Losses, Transfusion Rate for 700 Consecutive Liver Transplantations. Semin Thromb Hemost. 2015;41:538–46. doi: 10.1055/s-0035-1550428. [DOI] [PubMed] [Google Scholar]
- 20.Massicotte L, Sassine M-P, Lenis S, Roy A. Transfusion predictors in liver transplant. Anesthesia & Analgesia. 2004;98:1245–51. doi: 10.1213/01.ane.0000111184.21278.07. [DOI] [PubMed] [Google Scholar]
- 21.Roullet S, Biais M, Millas E, et al. Risk factors for bleeding and transfusion during orthotopic liver transplantation. Ann Fr Anesth Reanim. 2011;30:349–52. doi: 10.1016/j.annfar.2011.01.008. [DOI] [PubMed] [Google Scholar]
- 22.Ramos E, Dalmau A, Sabate A, et al. Intraoperative red blood cell transfusion in liver transplantation: influence on patient outcome, prediction of requirements, and measures to reduce them. Liver Transpl. 2003;9:1320–7. doi: 10.1016/jlts.2003.50204. [DOI] [PubMed] [Google Scholar]
- 23.Hendriks H, Van der Meer J, Klompmaker I, et al. Blood loss in orthotopic liver transplantation: a retrospective analysis of transfusion requirements and the effects of autotransfusion of cell saver blood in 164 consecutive patients. Blood coagulation & fibrinolysis. 2000;11:S87–S93. doi: 10.1097/00001721-200004001-00017. [DOI] [PubMed] [Google Scholar]
- 24.Modanlou KA, Oliver DA, Grossman BJ. TRANSFUSION PRACTICE: Liver donor’s age and recipient’s serum creatinine predict blood component use during liver transplantation. Transfusion. 2009;49:2645–51. doi: 10.1111/j.1537-2995.2009.02325.x. [DOI] [PubMed] [Google Scholar]
- 25.Massicotte L, Beaulieu D, Thibeault L, et al. Coagulation defects do not predict blood product requirements during liver transplantation. Transplantation. 2008;85:956–62. doi: 10.1097/TP.0b013e318168fcd4. [DOI] [PubMed] [Google Scholar]
- 26.Liu C, Vachharajani N, Song S, et al. A quantitative model to predict blood use in adult orthotopic liver transplantation. Transfus Apher Sci. 2015;53:386–92. doi: 10.1016/j.transci.2015.07.008. [DOI] [PubMed] [Google Scholar]
- 27.Massicotte L, Beaulieu D, Roy JD, et al. MELD score and blood product requirements during liver transplantation: no link. Transplantation. 2009;87:1689–94. doi: 10.1097/TP.0b013e3181a5e5f1. [DOI] [PubMed] [Google Scholar]
- 28.Makroo R, Walia RS, Aneja S, et al. Preoperative predictors of blood component transfusion in living donor liver transplantation. Asian j transfus sci. 2013;7:140. doi: 10.4103/0973-6247.115586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nardo B, Bertelli R, Montalti R, et al. Red blood cell transfusion in liver transplantation: a case-control study. Transplant Proc. 2005;37:4389–92. doi: 10.1016/j.transproceed.2005.11.030. [DOI] [PubMed] [Google Scholar]
- 30.Ghaffaripour S, Mahmoudi H, Khosravi MB, et al. Preoperative factors as predictors of blood product transfusion requirements in orthotopic liver transplantation. Prog Transplant. 2011;21:254–9. doi: 10.1177/152692481102100311. [DOI] [PubMed] [Google Scholar]
- 31.Masior Ł, Grąt M, Krasnodębski M, et al. Prognostic Factors and Outcomes of Patients After Liver Retransplantation. Transplant Proc. 2016;48:1717–20. doi: 10.1016/j.transproceed.2016.01.055. [DOI] [PubMed] [Google Scholar]