Abstract
The treatment of hepatitis C virus (HCV) infection by combination of direct acting antivirals (DAA), with different mode of action, has made substantial progress in the past few years. However, appearance of resistance and high cost of the therapy is still an obstacle in the achievement of the therapy, more specifically in developing countries. In this context, search for affordable antivirals with new mechanisms of action is still needed. Tea, after water, is the most popular drink worldwide. Polyphenols extracted from green tea have already shown anti-HCV activity as entry inhibitors. Here, three different theaflavins, theaflavin (TF1), theaflavin-3’-monogallate (TF2), and theaflavin-3-3’-digallate (TF3), which are major polyphenols from black tea, were tested against HCV in cell culture. The results showed that all theaflavins inhibit HCV infection in a dose-dependent manner in an early step of infection. Results obtained with HCV pseudotyped virions confirmed their activity on HCV entry and demonstrated their pan-genotypic action. No effect on HCV replication was observed by using HCV replicon. Investigation on the mechanism of action of black tea theaflavins showed that they act directly on the virus particle and are able to inhibit cell-to-cell spread. Combination study with inhibitors most widely used in anti-HCV treatment regimen demonstrated that TF3 exerts additive effect. In conclusion, theaflavins, that are present in high quantity in black tea, are new inhibitors of HCV entry and hold promise for developing in therapeutic arsenal for HCV infection.
Introduction
Hepatitis C caused by hepatitis C virus (HCV) has been called silent epidemic. The majority of infections are asymptomatic, but in 20% of cases the virus persists, leading to chronic hepatitis [1] causing liver fibrosis and cirrhosis, which is often a prelude to hepatocellular carcinoma [2]. Liver transplantation is often necessary in a portion of HCV infected patients [3]. Tremendous efforts have been expended to develop efficacious prophylactic and therapeutic treatment regimen for chronic hepatitis C. No vaccine is available due, at least in part, to the high genomic variability of HCV, which has led to the distinction of seven genotypes, most of which have multiple subtypes [4]. The therapeutic option against HCV has recently been improved with the development of HCV direct acting antivirals (DAA) like Daclatasvir, Sofosbuvir and Simeprevir, targeting viral proteins NS5A, NS5B polymerase or NS3/4A protease, respectively [5]. These approved DAA prominently increase the sustained viral response (SVR) up to ~95% in most patients, depending primarily on disease stage and the genotype of the infecting virus [5]. However, treatment with DAAs is not without limitation; it is associated with side-effects, resurgence of infection in transplant patient and high cost especially in developing countries [6,7]. Approved DAAs mainly target the virus replication leading to emergence of resistance mutations in this RNA virus genome [8]. Thus, novel combinations of low cost entry inhibitors with conventional treatment targeting different stages of the HCV life cycle, may provide a promising approach against HCV drug resistance development and infection relapse [9]. Moreover, prevention of donor liver re-infection by inhibiting viral entry into hepatocytes might be achieved using DAAs targeting entry.
Hepatitis C virus is an enveloped positive-stranded RNA virus encoding a polyprotein, co- and post-translationnally cleaved into structural and non-structural proteins [10]. Two viral glycoproteins E1 and E2 are part of the lipoviroparticle envelope. Non-structural proteins, NS2 to NS5B, are involved in replication and assembly of new virions. Actual antiviral therapy with DAA targets three non-structural proteins, the RNA-dependent RNA polymerase NS5B, a non-enzymatic protein involved in replication and assembly of HCV NS5A, and the viral protease NS3/4A, involved in polyprotein processing and essential for viral replication [11]. Virus entry into hepatocytes is a multistep process that involves attachment of the particle to glycosaminoglycans and subsequent binding to entry factors, SR-B1, CD81, Claudin-1 and Occludin [12]. After clathrin-mediated endocytosis and fusion of the viral envelope to endosomal membrane, the viral RNA is replicated, assembled and released via the secretory pathway.
In recent times, a wide variety of natural compounds have been extensively studied in terms of their antiviral activity [13]. Polyphenols are one such group of compounds with potent antiviral activities. Earlier studies of others and our group have shown that epigallocatechin-3-gallate (EGCG), a major green tea polyphenol, inhibits HCV entry by a new mechanism of action [14–16]. Recently, black tea polyphenols, theaflavins (TFs), components of black tea extract, have shown potent antiviral activities against calcivirus [17], herpes simplex virus 1 (HSV-1) [18], human immunodeficiency virus 1 (HIV-1) [19] and influenza A [20]. Theaflavins are formed by the enzymatic oxidation and decarboxylation of catechins during the manufacture of black tea [21]. The most abundant TFs are theaflavin (TF1), theaflavin-3’-monogallate (TF2) and theaflavin-3-3’-digallate (TF3). Theaflavins share many of the structural characteristics of EGCG, which prompted us to screen theaflavins against HCV. We also aim to identify compounds with enhanced anti-HCV activity than previously studied compounds to boost the development of suitable inhibitors.
Here, we report the capacity of theaflavins to inhibit HCV infection in vitro, demonstrating their activity on HCV entry. Given the abundance of theaflavins, especially in tea growing areas, and popularity of tea as a drink next only to water and as health drink, the compounds hold promise for developing in therapeutic arsenal for HCV infection.
Materials and methods
Chemicals and antibodies
DMEM, goat and fetal bovine sera were purchased from Invitrogen (Carlsbad, CA). 4’,6-diamidino-2-phenylindole (DAPI) was from Molecular Probes (Invitrogen). EGCG used as a control was from Calbiochem (Merck Chemicals, Darmstadt, Germany). Theaflavin 3’-monogallate (TF2P) and theaflavin 3,3’-digallate (TF3P) used to compare with our extracted theaflavins, TF2 and TF3, were from PhytoLab (Germany). Boceprevir was kindly provided by Philippe Halfon (Hôpital Européen, Laboratoire Alphabio, Marseille, France). Sofosbuvir (PSI-7977) and Daclatasvir (BMS-790052) were purchased from Selleckchem (Houston, USA). All other chemicals like DMSO were purchased from Sigma. Mouse anti-E1 monoclonal antibody (MAb) A4 was produced in vitro [22]. Mouse anti-GFP (Roche) and mouse anti-β tubulin monoclonal antibody (TUB 2.1) were from Sigma. Peroxidase-conjugated goat anti-mouse IgG and Cy3-conjugated goat anti-mouse IgG were from Jackson Immunoresearch (West Grove, PA).
Cells and virus strains
Human hepatoma Huh-7 cells and human embryonic kidney HEK 293T cells were grown in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with glutamax-I and 10% fetal bovine serum.
Japanese fulminant hepatitis-1 (JFH-1) HCV strain containing titer enhancing mutations [23] was used. Virus stocks were prepared by infecting Huh-7 cells with HCV JFH-1 (HCVcc) with a titer of 106 pfu/ml.
Theaflavins extraction and stock preparation
Theaflavins were extracted with ethyl acetate from prepared “crush tear curl” (CTC), black tea from Assam, India. The enriched theaflavin extract was subjected to sephadex LH-20 column (Sigma) and eluted with 40% acetone solution. The elute with distinguished reddish colours for each of TF1, TF2 and TF3 was fractionated. Three fractions of theaflavins were concentrated by a rotary evaporator and lyophilized, then recrystallized with dehydrated ethanol. The degree of purity and the mass of the three theaflavins obtained (theaflavin TF1, theaflavin-3-gallate TF2, theaflavin-3,3'-digallate TF3) were confirmed by UPLC-ESI-MS (S1 Fig). Ultra-High Performance Liquid Chromatography (UHPLC) analyses were carried out using an Acquity UPLC H-Class Waters system (Waters, Guyancourt, France) equipped with a diode array detector (DAD) and an Acquity QDa ESI-Quadrupole Mass Spectrometer. The stationary phase was a Waters Acquity BEH C18 column (2,1 x 50 mm, 1,7 μm) connected to a 0.2 μm in-line filter. The mobile phase consisted of 0.1% formic acid in water, and of 0.1% formic acid in acetonitrile. The gradient of acetonitrile was: 10% (0–1 min), 10–100% (1–7 min), 100% (7–9 min) at 0.3 ml/min. Column temperature was set at 30°C. The samples were dissolved in MeOH/H2O (50:50) at the concentration of 500 μg/ml. Injection volume was 2 μl. The ionization was performed in negative mode. Cone voltage was set at 30V. Probe Temperature was 600 °C. Capillary voltage was 0.8kV. The MS-Scan mode was used from 200 to 950 Da.
The fraction enriched in TF1, the mixed TF1, TF2 and TF3, and purified TF3 were each dissolved in DMSO to produce stock solutions at a concentration of 250 mg/ml.
Cytotoxicity assay
Huh-7 cells were seeded in 96 well plates and incubated at 37°C for 24 h. Cultures of 60–70% confluency were treated with various concentrations of theaflavins. An MTS [3-(4, 5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium, inner salt] based assay (CellTiter 96 aqueous nonradioactive cell proliferation assay, Promega) was used to evaluate cell viability treated after 24, 48 and 72 h. DMSO was used as control.
Immunofluorescence assay for anti-HCV quantification in confocal microscopy
Huh-7 cells were seeded in 96-well plates (6.000 cells/well) the day before infection. Cells were infected for 2 h with HCVcc at a multiplicity of infection (MOI) of 0.8. Then, the inoculum was removed and cells were overlaid with fresh medium. After 30 h, cells were fixed with ice-cold methanol and were processed for immunofluorescent detection of E1 envelope protein as described [24]. Nuclei were stained with 1 μg/ml DAPI. Confocal images were recorded using an automated confocal microscope InCell Analyzer 6000 (GE Healthcare Life Sciences). Each image was then processed using the Colombus image analysis software (Perkin Elmer) as described [14].
Viral RNA quantification
Huh-7 cells seeded in 12-well plates were inoculated with HCVcc for 2h. The inoculum was removed and replaced with culture medium. Forty-eight hours after infection, cells were lysed in lysis buffer from the kit NucleoSpin RNA Plus (Macherey Nagel), and total RNA extracted following manufacturer’s instructions, eluted in a final volume of 60 μl of H2O, and quantified. Ten microliters of RNA were used for cDNA synthesis using High Capacity cDNA Reverse Transcription kit (Applied Biosystems). Five microliters of cDNA were used for real-time reverse-transcription polymerase chain reaction (qRT-PCR) assay using Taqman probes as described previously [25].
Detection of E1 glycoprotein by Western blot
Huh-7 cells seeded in 12-well plates were inoculated with HCVcc for 2h. The inoculum was removed and replaced with culture medium. Forty-eight hours after infection, cells were lysed in ice cold PBS containing Triton-100 1% and protease inhibitors for 20 min. Cell lysates were collected and insoluble debris were removed by centrifugation. 20 μg of proteins were analyzed by western blotting using anti-E1 and anti-β tubulin antibodies. Peroxidase-conjugated goat anti-mouse secondary antibody (Jackson Immunoresearch) was used for the revelation using ECL western blotting substrate (Thermo Fischer Scientific).
Time of addition assay
Compounds were added before, during and after inoculation of Huh-7 cells with HCVcc. Briefly, cells were incubated in the presence or absence of compounds (pretreatment) for 1 h and subsequently replaced with virus for 1 h (inoculation). Inoculum was removed and replaced with DMEM for 28 h (post-inoculation). After 30 h cells were fixed and immunofluorescence detection was performed as described above.
Direct effect on viral particle
HCV was pre-incubated with 25 μg/ml of theaflavins for 1 h before inoculation, and then the mixture was diluted 10 times before being used for the inoculation (1 h at 37°C), leading to a final concentration of theaflavins of 2.5 μg/ml. In parallel, Huh-7 cells were inoculated with HCV in the presence of theaflavins either at 2.5 or 25 μg/ml. Importantly, the MOI was kept constant in all the conditions. Inoculum was removed and replaced with DMEM. After 30 h, immunofluorescence was performed as described above.
Entry assay with HCV pseudo-particles (HCVpp)
The luciferase-based HCV pseudotyped retroviral particles were generated as previously described [26]. Briefly, HEK-293T cells were transfected with plasmids encoding HCV envelope proteins, Gag/Pol, and firefly luciferase. The HCV envelope plasmids included 6 different genotypes (1b, 2a, 3a, 4, 5 and 6). The supernatants of the transfected cells were collected 72 h later and filtered through a 0.45-μm membrane. Vesicular stomatitis virus pseudoparticles (VSVpp) were used as a control. For the entry assay, Huh-7 cells were seeded on 96-well plates overnight and incubated with HCVpp for 2 h at 37°C in the presence or not of theaflavins. Inoculum was replaced by DMEM and kept for 48 h. HCVpp entry into Huh-7 cells was measured by luciferase activity quantification using Luciferase Assay Kit (Promega) and a Berthold luminometer.
HCV replicon and replication assay
The plasmid pSGR-JFH1 encoding a sub-genomic replicon of JFH-1 strain was obtained from Dr T Wakita [27]. A BglII and an NsiI restriction sites were inserted between codons Pro419 and Leu420 of NS5A, and the coding sequence of enhanced green fluorescent protein (EGFP) was then inserted between these two sites. This position was previously shown to accept a GFP insertion in a sub-genomic replicon of the Con1 strain [28]. The plasmid was in vitro transcribed before electroporation into Huh-7 cells. Cells that express replicon were selected for using 500 μg/mL of geneticin during 15 days and cultured in a medium containing 250 μg/mL of geneticin. Huh-7 cells stably expressing replicon were seeded in 24-well plates and incubated with the different compounds for 24, 48 and 72 h. They were lysed in ice cold lysis buffer (Tris HCl 50mM, NaCl 100 mM EDTA 2 mM Triton-100 1% SDS 0.1%) containing protease inhibitors for 20 min. Cell lysates were collected and insoluble debris were removed by centrifugation. 20 μg of proteins were analyzed by western blotting using anti-GFP and anti-β tubulin antibodies. Peroxidase-conjugated goat anti-mouse secondary antibody (Jackson Immunoresearch) was used for the revelation using ECL western blotting substrate (Thermo Fischer Scientific).
Cell-to-cell transmission assay
Huh-7 cells were infected with HCVcc at MOI of 0.01. After 2 hours of contact, the inoculum was removed and replaced with fresh DMEM containing TF1, TF2, TF3 at 25 μg/ml or DMSO along with neutralizing antibody mAb 3/11 to prevent infection via the cell culture medium. The cells were propagated for another 72 h prior to immunofluorescence staining as described above. Quantification was done in 3 independent wells by taking independent pictures of different field of each well.
Statistical analysis
The statistical test used is a Kruskal Wallis nonparametric test followed by a Dunn’s multicomparison post hoc test with a confidence interval of 95% to identify individual difference between treatments. P values < 0.05 were considered as significantly different from the control. The data were analyzed using Graph Pad Prism (Version 5.0b) by comparisons between each treated group and untreated group (DMSO control).
Results
Theaflavins inhibit HCV infection
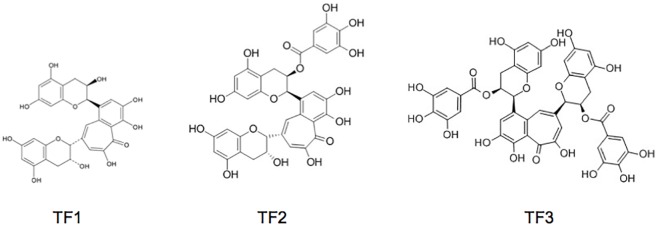
We and others have shown that EGCG, the major polyphenol present in green tea extract, was active against HCV infection [15,16,29], but no data are available on black tea polyphenols. In this context, three theaflavin derivatives (theaflavin TF1, theaflavin-3-gallate TF2 and theaflavin-3,3'-digallate TF3) were extracted from CTC Assam black tea and identified by UPLC-UV-MS (Fig 1) with retention times of 3.82, 3.92, and 3.96 min, respectively (S1 Fig). The average molecular weight of TF1 (564.49 g/mole), TF2 (716.59 g/mole) and TF3 (868.709 g/mole) was used for calculating the molar concentration of theaflavins which were generally used in micrograms in the experiments. However, the degree of purity of the compounds is not at the same level. Theaflavin-3,3'-digallate (TF3) is pure with a degree of purity superior to 97%. By contrast, TF2 which is present as two isomers, theaflavin-3-monogallate and theaflavine-3’-monogallate, is in mixture with TF1 and TF3 in the fraction purified (S1 Fig).
Fig 1. Chemical structures of theaflavins extracted from black tea.
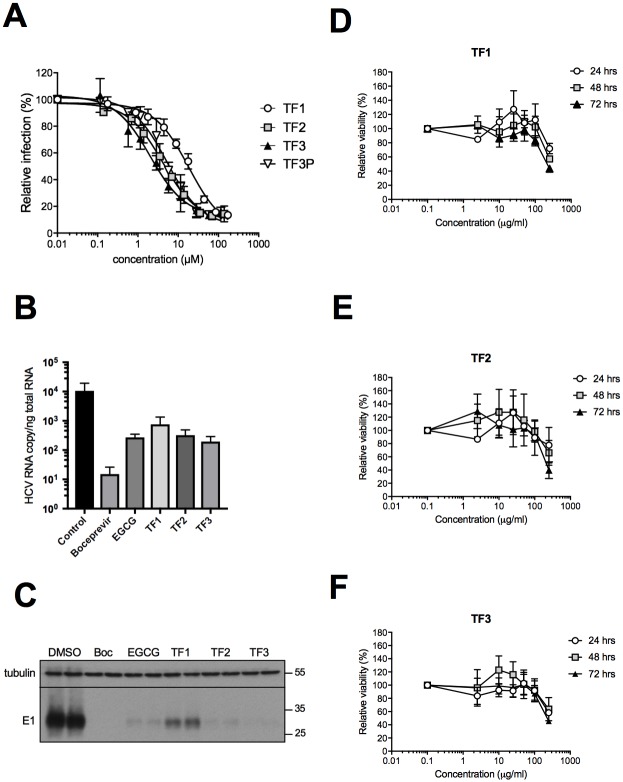
The inhibitory activity of theaflavins against HCV infectivity was carried out in dose-response experiment. Huh-7 cells were infected with HCV strain JFH1 in the presence of increasing concentrations of theaflavins, 0 to 100 μg/ml. Quantification of infected cells by immunofluorescence labeling of E1 envelope protein showed significantly decreased infectivity of HCV in the presence of each three theaflavins, with a clear dose-dependent inhibitory effect (Fig 2A). The half effective concentration (EC50) was calculated for each molecule to be 17.89, 4.08, and 2.02 μM for TF1, TF2 and TF3, respectively (Table 1). TF3 is the most active. To confirm that the activity is due to the molecule, a commercial TF3 (TF3P) was tested and its antiviral activity compared to natural TF3 extracted from black tea. Similar results were obtained with a calculated EC50 of 4.96 μM for the commercial TF3 (Fig 2A, Table 1). Because TF2 was difficult to purify and is mixed with TF1 and TF3, a commercial TF2 was also tested, TF2P, and confirmed the antiviral activity of TF2 against HCV, EC50 = 18.06 (Table 1). Antiviral activity of the different theaflavins was confirmed by the quantification of viral RNA and viral E1 glycoprotein in Huh-7 cells inoculated with HCVcc in the presence of the molecules. More than 1 log10 decrease of HCV RNA was observed after treatment with theaflavins, similar to the decrease observed with EGCG, an inhibitor of HCV entry (Fig 2B). Boceprevir, an NS3 protease inhibitor showed a higher decrease in RNA level due to its mode of action on HCV replication. Similarly, E1 envelope glycoprotein levels determined by Western blotting were also decreased in the presence of the theaflavins and the control compounds (Fig 2C). All the results confirmed that TF1 is the less active and TF3 the most active. We then tested the effect of theaflavins on cell viability by treating Huh-7 with a range of theaflavin concentrations that were used in inhibition experiments, 0 to 250 μg/ml, with three different exposure times, 24, 48 and 72 h (Fig 2D–2F). Theaflavins did not decrease cell viability up to 100 μg/ml after short (24 h) to long (72 h) exposure times, a concentration much higher than the active antiviral concentrations. The half-medium cytotoxic concentration (CC50) at 72h is estimated to be 250 μg/ml, giving a selective index (SI) of 24, 85 and 142 for TF1, TF2 and TF3, respectively (Table 1). TF3 is the most promising molecule.
Fig 2. Theaflavins inhibit HCV infection.
(A) Huh-7 cells were inoculated with HCV JFH-1 in the presence of given concentration of the different theaflavins, TF1, TF2, TF3 and a commercial TF3 (TF3P). Inoculum was removed and replaced with medium containing theaflavins. Cells were fixed at 30 h post-infection and subjected to immunofluorescence to quantify number of infected cells. Results are expressed relative to DMSO control without theaflavins (100%). Huh-7 cells were inoculated with HCV JFH-1 in the presence of the different theaflavins, TF1, TF2 and TF3 at 25 μg/ml, or EGCG at 50 μM, or boceprevir at 1nM. Inoculum was removed and replace with medium without compounds except for boceprevir. 48h after infection cells were lysed and RNA or proteins extracted. Quantification of viral RNA by qRT-PCR was performed (B). HCV envelope E1 protein was detected using specific antibody (A4) as well as ß-tubulin in cellular extracts by Western blot analysis (C). Huh-7 cells were incubated with given concentrations of TF1 (D), TF2 (E) or TF3 (F) at different concentrations during 24, 48 or 72 h. Cell viability was measured using an MTS-based assay. Optical density at 490 nm was measured at the different time points. Results are expressed as mean ± SEM (error bars) of three independent experiments performed in triplicates and relative to control conditions without compounds.
Table 1. EC50, CC50 and SI of the different theaflavins.
| EC50 | CC50a | SI | |
|---|---|---|---|
| TF1 | 17.89 μM (10.09 μg/ml) | 442.8 μM (250 μg/ml) | 24.7 |
| TF2 | 4.08 μM (2.92 μg/ml) | 348.8 μM (250 μg/ml) | 85.5 |
| TF3 | 2.02 μM (1.75 μg/ml) | 287.7 μM (250 μg/ml) | 142.4 |
| TF2P | 18.06 μM (12.92 μg/ml) | ndb | |
| TF3P | 4.96 μM (4.30 μg/ml) | ndb |
a CC50 is estimated after 72h of exposure to the cells.
b not determined
Theaflavins inhibit HCV entry and not replication
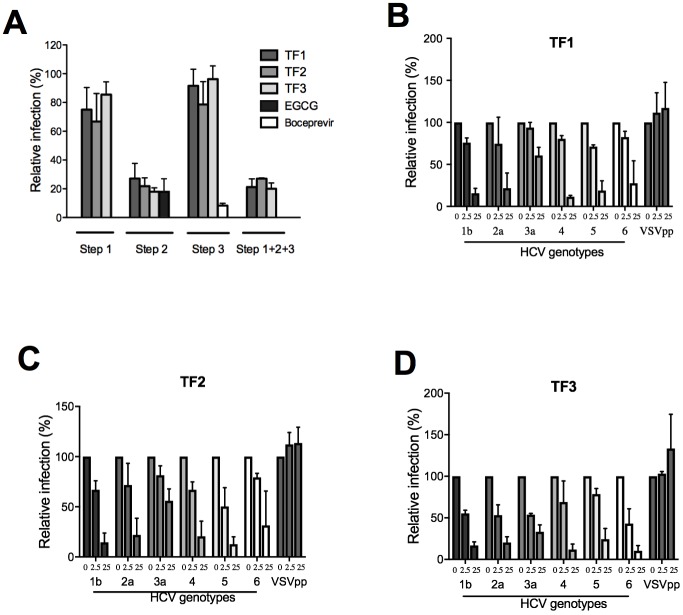
The HCV infection cycle is a multistep process that involves entry of the virus into the cell, replication of the genomic RNA and assembly/release of viral particles. To determine at which step the different theaflavins exert their action, a time of addition experiment was performed. The compounds were added before, during or after inoculation of HCV to Huh-7 cells. EGCG, an inhibitor of HCV entry, and Boceprevir, a HCV NS3 protease inhibitor, were used as controls. The result clearly demonstrates a significant inhibition of HCV only in the presence of theaflavins added during inoculation, like EGCG (Fig 3A). There was no inhibition when compounds were added to cells prior to the infection or post-infection. As expected, Boceprevir inhibits the post-inoculation step, corresponding to replication. These results, taken together, suggest that theaflavins act at an early step of the viral infectious cycle, most probably the entry step.
Fig 3. Theaflavins inhibit HCV entry in a pan-genotypic manner.
(A) Huh-7 cells were inoculated with HCV JFH-1 for 2 h and fixed at 30 h post-infection to quantify the number of infected cells. Theaflavins, at 25 μg/ml were either added for 2 h to the cells before infection (step 1), or during inoculation (step 2), or during the 28 h after inoculation (step 3), or during the 3 steps (step 1+2+3). EGCG at 50 μM or Boceprevir at 1 μM were added either during the inoculation step or post-inoculation respectively. (B-D) Huh-7 cells were inoculated with HCVpp of the given genotypes or VSVpp in the absence (0) or presence of theaflavins at 2.5 or 25 μg/ml. Cells were lysed 48h after infection and luciferase activity quantified. Results are expressed as mean ± SEM (error bars) of 3 independent experiments performed in triplicate. Data are normalized to the DMSO, which is expressed as 100% infection.
Viral pseudoparticles are good models for entry inhibitor assay and also for screening against different viral genotypes [30]. To confirm that theaflavins act at the entry step, HCVpp were produced and inoculated to Huh-7 cells in the presence of each theaflavins at active concentrations (EC50 and 10 × EC50). Envelope glycoproteins of different HCV genotypes were used, genotype 1b, 2a, 3a, 4, 5 and 6. The result shows that all the theaflavins inhibit infection of HCVpp in a dose dependent manner (Fig 3B–3D) confirming their activity on HCV entry. Moreover, the antiviral activity of each theaflavin is pan-genotypic, with an inhibition of infection for all HCV genotype tested. HCVpp of genotype 3a are less inhibited by theaflavins than HCVpp of other genotypes, even if TF3 seems more active on this genotype than TF1 or TF2.
Results presented in Fig 3A suggest that theaflavins are not active on HCV replication because no inhibitory effect was observed when the compounds were added during the 28 h post-inoculation. To confirm this data, HCV subgenomic replicon containing a GFP-tagged NS5A was used. Replicon cells were incubated with TF1, TF2 or TF3 at 25 μg/ml, or Boceprevir at 1 μM, for 72 h, and NS5A protein quantified by Western blot analysis. As expected, NS5A protein appears as two bands, P56 and P58. The result shows that the replication of the replicon is not affected by the presence of any of the theaflavins but is inhibited by Boceprevir (Fig 4). Taken together, our results show the TF1, TF2 and TF3 are inhibitors of HCV entry and not replication.
Fig 4. Theaflavins are not inhibitors of HCV replication.
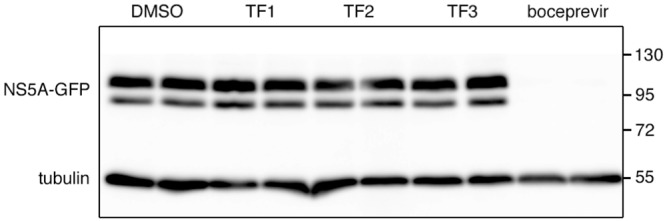
Huh-7 cells expressing a HCV replicon containing a GFP-tagged NS5A were incubated in the absence (DMSO) or presence of theaflavins at 25 μg/ml, or Bocebrevir at 1 μM, for 72 h. Cells were lysed and Western blot analysis was performed to detected NS5A-GFP and tubulin with specific antibodies. Data are representative of 3 independent experiments.
Theaflavins act directly on the viral particle
Theaflavins can inhibit HCV entry by acting either directly on the viral particle or on cellular factors. This second hypothesis seems unlikely because no effect of theaflavins on HCV infection was observed when cells were pre-treated with the molecules before infection (Fig 3A). To determine if theaflavins act directly on the viral particle, HCV viral stocks were incubated with the compounds at 25 μg/ml prior inoculation. The viral stocks containing the compounds were then diluted 10 times before inoculation leading to a final concentration of theaflavins of 2.5 μg/ml for the inoculation step. In parallel, cells were inoculated with a non-treated virus in the presence of the compounds at the two different concentrations, 2.5 and 25 μg/ml. As shown in Fig 5, the inhibitory effect of theaflavins on pre-incubated virus is similar to the one observed when virus is inoculated on cells at high concentration (25 μg/ml), demonstrating that theaflavins act directly on HCV particle before entry.
Fig 5. Theaflavins act on the viral particles prior inoculation.
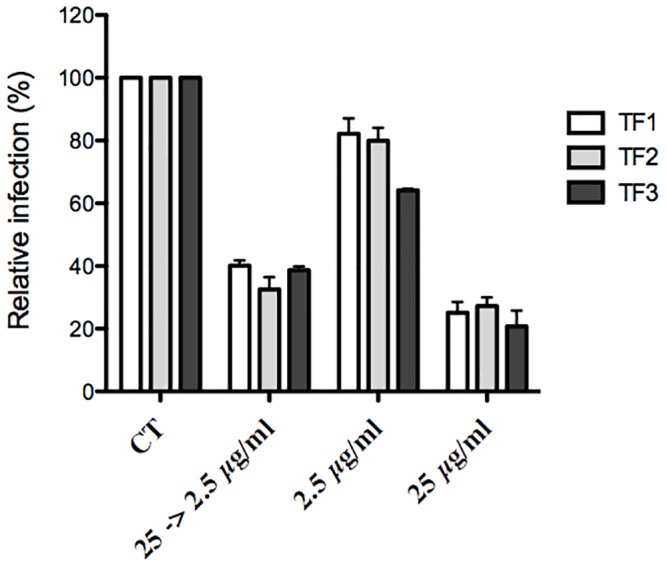
HCV virus stock was pre-incubated with 25 μg/ml of theaflavins for 1 h before inoculation, and then the mixture was diluted 10 times before being used for the inoculation, leading to a final concentration of theaflavins of 2.5 μg/ml (25 -> 2.5 μg/ml). In parallel, Huh-7 cells were inoculated with HCV in the presence of theaflavins either at 2.5 or 25 μg/ml. Cells were fixed and subjected to immunofluorescence to quantify infected cells as described above. Data are normalized to the DMSO, which is expressed as 100% infection. The results are presented as means ± SEM of three independent experiments performed in triplicate.
Theaflavins inhibit HCV cell-to-cell propagation
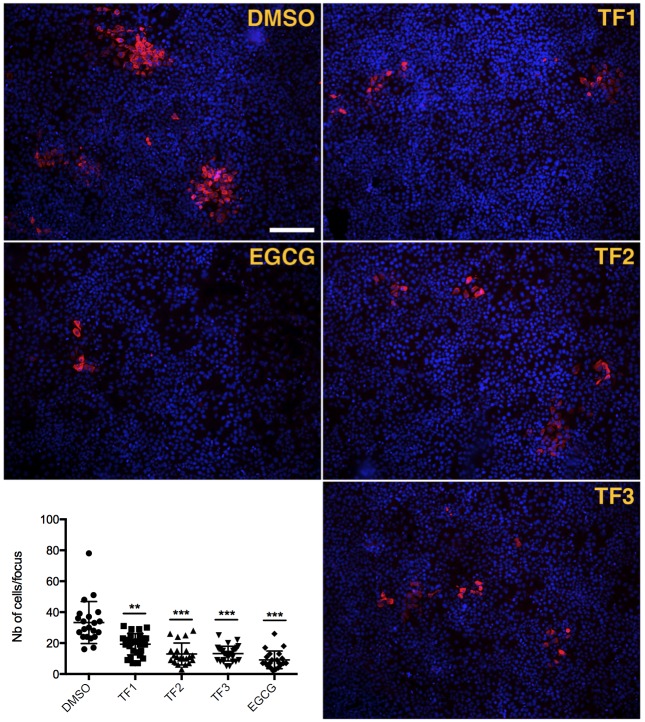
HCV infection can follow two different modes of transmission, either by direct entry into the cell via extracellular medium or via cell-to-cell spread between two neighboring cells. This last mode of transmission is resistant to neutralizing antibodies and seems to be a major way of HCV infection [31]. Therefore, it is important to identify antiviral agents able to block cell-to-cell transmission. To determine if theaflavins can inhibit HCV cell-to-cell spread, Huh-7 cells were infected with HCVcc with no inhibitor, at low MOI to visualize foci, and further incubated with theaflavins in the presence of neutralizing antibodies to prevent extracellular infection. 72 h post-infection, the number of cells in the foci were quantified. The results shows that TF1, TF2 and TF3 significantly reduce number of cells in the foci meaning reduction in cell-to-cell spread (Fig 6), like EGCG a known inhibitor of cell-to-cell transmission [15]. This shows that theaflavins are potent inhibitors of both HCV modes of entry.
Fig 6. Theaflavins inhibit cell-to-cell spread.
Huh-7 cells were inoculated with HCV JFH-1 for 2 h. Inoculum was replaced by medium containing mAb 3/11 neutralizing antibody to prevent extracellular propagation. Theaflavins at 25 μg/ml or EGCG at 50 μM were added in the medium after inoculation. Cells were fixed 72 h after infection and subjected to immunofluorescence detection of E1 HCV envelope protein revealed with a Cy3-conjugated goat anti-mouse secondary antibody (red). Nuclei were stained with DAPI (blue). Representative images of each condition are shown. Graph represents the number of cells/focus of 3 independent fields of three independent wells. Error bars represent SD. Data are representative of 3 independent experiments. Statistical analysis were performed with the Kruskal Wallis nonparametric test followed by a Dunn’s multicomparison post hoc test. **, P = 0.009; ***, P < 0.0001.
Combination of theaflavins with replication inhibitors used in hepatitis C therapy
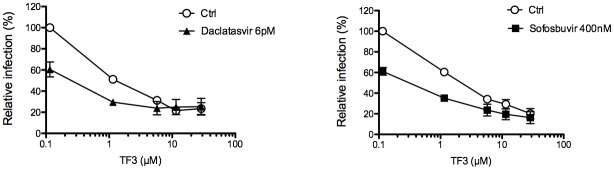
Finally, in order to determine the potential use of these compounds in antiviral therapy, we interrogated the effect of theaflavins in combination with the known DAA Sofosbuvir and Daclatasvir, two inhibitors of HCV replication that target HCV proteins NS5B and NS5A respectively [32,33]. In the experiment, TF3, the most active theaflavin, was added at different concentrations along with Sofosbuvir or Daclatasvir at fixed concentration during infection of Huh-7 cells with HCVcc. The result shows that TF3 can increase the antiviral activity of both Sofosbuvir and Daclatasvir in an additive manner (Fig 7) demonstrating that it could be use in combination with DAA used in hepatitis C therapy.
Fig 7. Theaflavins can be used in combination with DAA.
Huh-7 cells were inoculated with HCV JFH-1 in the presence of TF3 at given concentrations. Inoculum was removed and replaced by medium containing or not either Daclatasvir at 6 pM or Sofosbuvir at 400 nM. Cells were fixed at 30 h post infection and subjected to immunofluorescent detection of E1 envelope protein. Results are expressed as mean +/- SEM (error bars) of 3 independent experiments performed in triplicate. Data are normalized to DMSO, which is expressed as 100% infection.
Discussion
The present study identified new entry inhibitors of HCV, theaflavins, the major black tea polyphenols. Theaflavins inhibit all HCV genotypes and most probably induce inactivation of the viral particle before inoculation. Interestingly, they are also able to inhibit cell-to-cell spread of HCV, a major route of infection allowing evasion of the virus from neutralizing antibodies [31]. Moreover, an additive action of TF3 in combination with molecules used for hepatitis C therapy was observed.
HCV is transmitted between hepatocytes via classical cell entry but also uses direct cell-to-cell transfer to infect neighboring hepatocytes. Cell-to-cell transmission was also shown to play an important role in dissemination and maintenance of resistant variants in cell culture models [34]. HCV entry is highly orchestrated and essential in initiating viral infection and spread. This step represents a potential target for HCV inhibitors [35,36] but, up to now, no HCV entry inhibitor has been marketed, and few are in preclinical trial. The currently used therapy of DAA regimen cures more than 90% of infected patients, but the appearance of viral resistance, and recurrence of infection particularly in transplant patients is still a major limitation [37]. Entry inhibitors given along with the DAAs would be expected to exhibit a synergistic effect [35,36]. The present study supported this hypothesis when theaflavin and Sofosbuvir or Daclatasvir were added in combination and showed additive effect against HCV. This may lead to pivotal implication in therapeutic regimen of HCV especially because Sofosbuvir, in combination with other drugs, is a part of all first-line treatments for HCV, and also of some second-line treatments [37].
HCV treatment efficacy is influenced by infected viral genotype; therefore, treatment regimen is dependent on genotypes infected. Our study with HCVpp of different genotypes (1–6) showed dose-dependent inhibition of HCV by theaflavins (Fig 3B–3D). This represents an important hit for further drug development.
In recent times, a number of promising natural products with anti-HCV activities have been discovered [38–40]. They are of different origins and chemical structures and exert their antiviral effects at different levels within the virus life cycle. While green tea polyphenols have received the most attention in the past years, data presented in this study suggest that black tea theaflavins may also be potent candidates for antiviral drug development particularly as entry inhibitors against HCV. Our data show that TF3 has the most prominent effect against HCV in comparison to TF1 and TF2. TF3 (EC50 = 2.2 μM) also is more potent than EGCG (EC50 = 10.6 μM) [14].
Our results clearly indicate that all the three derivatives of theaflavin act directly on viral particles and may prevent cell surface attachment or receptor binding. The effect seems permanent, not reversible, because after a 10-fold dilution of the compound in the presence of the virus, the effect is similar to the one without dilution (Fig 5). In our earlier study cryo-electron microscopy imaging showed that EGCG and delphinidin, an anthocyanidin, have a bulging effect on HCV envelope of HCVpp [14]. We might speculate that theaflavins might have similar effect as both polyphenols shared close structural similarity. However, the exact mechanism of action of theaflavins on HCV infection needs further studies.
TF3 seems to be most promising candidate against HCV and its efficacy may be attributed to the presence of double galloyl moiety. The same has been found for HSV-1 where TF3 was reported to be most effective [18]. In contrast, the presence of the galloyl group in TF3 is not necessary for antiviral properties against calcivirus where all three theaflavin derivatives have similar efficacy [17]. To understand the role of functional groups and galloyl moiety, further study is required through the approach of combinatorial and synthetic chemistry. It is interesting to note that theaflavins and EGCG have been described to inactivate the same viruses, HSV-1, HIV-1 and influenza virus [18–20,41]. Taken together with the results presented here, it seems that these polyphenols from tea, black or green, have a very specific mode of action on enveloped viruses that could be more exploited in the context of antiviral therapy.
A future prospect of our study may be to determine the bioavailability of theaflavins in human liver. Henning et al performed study on liver tissue of mice after black tea consumption and showed that theaflavin relative absorption in liver is twice higher than EGCG [42]. TF1 concentration in mice liver is estimated at 0.4 nmol g-1 tissue, approximately 0.4 μM. In contrast, TF2 and TF3 are poorly detected. Theaflavin content may vary from one tea to another. It has been shown that Assam black tea contains more theaflavins than Darjeeling tea, and particularly TF3 (125.9 and 26.8 μg/ml respectively) [43], which could lead to higher relative absorption. Even if the tissue concentrations of theaflavins are lower than their active concentrations, their use in combination with other DAA might reduce their EC50 as shown in Fig 7. In this combination study, EC50 of TF3 is reduced in the presence of Sofosbuvir or Daclatasvir to a concentration of approximately 0.3 μM. Taken together, these results may be of crucial importance in developing a prophylactic drug for HCV risk group population.
In conclusion, the present study identified theaflavins as new entry inhibitors of HCV infection. Their pan-genotypic action and ability to inhibit cell-to-cell spread are major advantages for further evaluation for drug development, as well as their efficacy in combination with actual antiviral therapy. Moreover, their ease of extracting, availability and popularity of tea as a drink make them interesting candidate as entry inhibitors against HCV infection.
Supporting information
(A) Theaflavin (TF1) (a) chromatogram at 375 nm, Rt = 3.82 min, (b) UV spectrum, (c) mass spectrum in negative mode [M-H]- m/z 563; (B) Theaflavin-3-gallate (TF2) (a) chromatogram at 375 nm, Rt = 3.92 min, (b) UV spectrum, (c) mass spectrum in negative mode [M-H]- m/z 715; (C) Theaflavin-3,3'-digallate (a) chromatogram at 375 nm, Rt = 3.96 min, (b) UV spectrum, (c) mass spectrum in negative mode [M-H]- m/z 867.
(PDF)
Acknowledgments
The authors are thankful to Department of Health Research, Ministry of Health and Family welfare, Government of India, for providing post-doctoral overseas training fellowship to P. Chowdhury. The authors are also thankful to Director, TTRI and Secretary, TRA for their support to implement the research fellowship grant.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
PB and AV received support from the European Community (ERC-STG INTRACELLTB Grant n° 260901), the Agence Nationale de la Recherche (ANR-10-EQPX-04-01), the Feder (12001407 (D-AL) Equipex Imaginex BioMed) and the Région Nord Pas de Calais (convention n°12000080). PC received a post-doctoral overseas training fellowship from Department of Health Research, Ministry of Health and Family welfare, Government of India.
References
- 1.Bartenschlager R, Lohmann V, Penin F. The molecular and structural basis of advanced antiviral therapy for hepatitis C virus infection. Nat Rev Microbiol. 2013;11: 482–496. 10.1038/nrmicro3046 [DOI] [PubMed] [Google Scholar]
- 2.Jacobson IM, Davis GL, El-Serag H, Negro F, Trépo C. Prevalence and challenges of liver diseases in patients with chronic hepatitis C virus infection. Clin Gastroenterol Hepatol. 2010;8: 924–933; quiz e117. 10.1016/j.cgh.2010.06.032 [DOI] [PubMed] [Google Scholar]
- 3.Stepanova M, Sayiner M, de Avila L, Younoszai Z, Racila A, Younossi ZM. Long-term outcomes of liver transplantation in patients with hepatitis C infection are not affected by HCV positivity of a donor. BMC Gastroenterol. 2016;16: 137 10.1186/s12876-016-0551-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Simmonds P. The origin of hepatitis C virus. Curr Top Microbiol Immunol. 2013;369: 1–15. 10.1007/978-3-642-27340-7_1 [DOI] [PubMed] [Google Scholar]
- 5.Asselah Tarik, Boyer Nathalie, Saadoun David, Martinot-Peignoux Michele, Marcellin Patrick. Direct-acting antivirals for the treatment of hepatitis C virus infection: optimizing current IFN-free treatment and future perspectives. Liver Int. 2016;36: 47–57. 10.1111/liv.13027 [DOI] [PubMed] [Google Scholar]
- 6.Konerman MA, Lok ASF. Hepatitis C Treatment and Barriers to Eradication. Clin Transl Gastroenterol. 2016;7: e193 10.1038/ctg.2016.50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lynch SM, Wu GY. Hepatitis C Virus: A Review of Treatment Guidelines, Cost-effectiveness, and Access to Therapy. J Clin Transl Hepatol. 2016;4: 310–319. doi: 10.14218/JCTH.2016.00027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Behrens SE, Tomei L, De Francesco R. Identification and properties of the RNA-dependent RNA polymerase of hepatitis C virus. EMBO J. 1996;15: 12–22. [PMC free article] [PubMed] [Google Scholar]
- 9.Baumert TF, Jühling F, Ono A, Hoshida Y. Hepatitis C-related hepatocellular carcinoma in the era of new generation antivirals. BMC Med. 2017;15: 52 10.1186/s12916-017-0815-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lindenbach BD, Thiel H-J, Rice CM. Flaviviridae: the viruses and their replication Fields Virology 5th Edition Wolters Kluwer, Lippincott, William & Wilkins. Knipe David M., Howley Peter M.; 2007. pp. 1101–1152. [Google Scholar]
- 11.Asselah Tarik, Marcellin Patrick, Schinazi Raymond F. Treatment of hepatitis C virus infection with direct-acting antiviral agents: 100% cure? Liver Int. 2018;38: 7–13. 10.1111/liv.13673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bartosch B, Dubuisson J. Recent Advances in Hepatitis C Virus Cell Entry. Viruses. 2010;2: 692–709. 10.3390/v2030692 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kitazato K, Wang Y, Kobayashi N. Viral infectious disease and natural products with antiviral activity. Drug Discov Ther. 2007;1: 14–22. [PubMed] [Google Scholar]
- 14.Calland N, Sahuc M-E, Belouzard S, Pène V, Bonnafous P, Mesalam AA, et al. Polyphenols Inhibit Hepatitis C Virus Entry by a New Mechanism of Action. J Virol. 2015;89: 10053–10063. 10.1128/JVI.01473-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Calland N, Albecka A, Belouzard S, Wychowski C, Duverlie G, Descamps V, et al. (-)-Epigallocatechin-3-gallate is a new inhibitor of hepatitis C virus entry. Hepatol Baltim Md. 2012;55: 720–729. 10.1002/hep.24803 [DOI] [PubMed] [Google Scholar]
- 16.Ciesek S, Von Hahn T, Colpitts CC, Schang LM, Friesland M, Rg Steinmann J, et al. The Green Tea Polyphenol, Epigallocatechin-3-Gallate, Inhibits Hepatitis C Virus Entry. Hepatology. 2011;54: 1947–1955. 10.1002/hep.24610 [DOI] [PubMed] [Google Scholar]
- 17.Ohba M, Oka T, Ando T, Arahata S, Ikegaya A, Takagi H, et al. Antiviral effect of theaflavins against caliciviruses. J Antibiot (Tokyo). 2017;70: 443–447. 10.1038/ja.2016.128 [DOI] [PubMed] [Google Scholar]
- 18.de Oliveira A, Prince D, Lo C-Y, Lee LH, Chu T-C. Antiviral activity of theaflavin digallate against herpes simplex virus type 1. Antiviral Res. 2015;118: 56–67. 10.1016/j.antiviral.2015.03.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yang J, Li L, Tan S, Jin H, Qiu J, Mao Q, et al. A natural theaflavins preparation inhibits HIV-1 infection by targeting the entry step: potential applications for preventing HIV-1 infection. Fitoterapia. 2012;83: 348–355. 10.1016/j.fitote.2011.11.016 [DOI] [PubMed] [Google Scholar]
- 20.Zu M, Yang F, Zhou W, Liu A, Du G, Zheng L. In vitro anti-influenza virus and anti-inflammatory activities of theaflavin derivatives. Antiviral Res. 2012;94: 217–224. 10.1016/j.antiviral.2012.04.001 [DOI] [PubMed] [Google Scholar]
- 21.Tanaka T, Matsuo Y, Kouno I. Chemistry of secondary polyphenols produced during processing of tea and selected foods. Int J Mol Sci. 2009;11: 14–40. 10.3390/ijms11010014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dubuisson J, Hsu HH, Cheung RC, Greenberg HB, Russell DG, Rice CM. Formation and intracellular localization of hepatitis C virus envelope glycoprotein complexes expressed by recombinant vaccinia and Sindbis viruses. J Virol. 1994;68: 6147–6160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Delgrange D, Pillez A, Castelain S, Cocquerel L, Rouillé Y, Dubuisson J, et al. Robust production of infectious viral particles in Huh-7 cells by introducing mutations in hepatitis C virus structural proteins. J Gen Virol. 2007;88: 2495–2503. 10.1099/vir.0.82872-0 [DOI] [PubMed] [Google Scholar]
- 24.Rouillé Y, Helle F, Delgrange D, Roingeard P, Voisset C, Blanchard E, et al. Subcellular Localization of Hepatitis C Virus Structural Proteins in a Cell Culture System That Efficiently Replicates the Virus. J Virol. 2006;80: 2832–2841. 10.1128/JVI.80.6.2832-2841.2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Castelain S, Descamps V, Thibault V, François C, Bonte D, Morel V, et al. TaqMan amplification system with an internal positive control for HCV RNA quantitation. J Clin Virol. 2004;31: 227–234. 10.1016/j.jcv.2004.03.009 [DOI] [PubMed] [Google Scholar]
- 26.Bartosch B, Dubuisson J, Cosset F-L. Infectious Hepatitis C Virus Pseudo-particles Containing Functional E1–E2 Envelope Protein Complexes. J Exp Med. 2003;197: 633–642. 10.1084/jem.20021756 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kato T, Date T, Miyamoto M, Furusaka A, Tokushige K, Mizokami M, et al. Efficient replication of the genotype 2a hepatitis C virus subgenomic replicon. Gastroenterology. 2003;125: 1808–1817. 10.1053/j.gastro.2003.09.023 [DOI] [PubMed] [Google Scholar]
- 28.Moradpour D, Evans MJ, Gosert R, Yuan Z, Blum HE, Goff SP, et al. Insertion of Green Fluorescent Protein into Nonstructural Protein 5A Allows Direct Visualization of Functional Hepatitis C Virus Replication Complexes. J Virol. 2004;78: 7400–7409. 10.1128/JVI.78.14.7400-7409.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen C, Qiu H, Gong J, Liu Q, Xiao H, Chen XW, et al. (-)-Epigallocatechin-3-gallate inhibits the replication cycle of hepatitis C virus. Arch Virol. 2012;157: 1301–1312. 10.1007/s00705-012-1304-0 [DOI] [PubMed] [Google Scholar]
- 30.Urbanowicz RA, McClure CP, King B, Mason CP, Ball JK, Tarr AW. Novel functional hepatitis C virus glycoprotein isolates identified using an optimized viral pseudotype entry assay. J Gen Virol. 2016;97: 2265–2279. 10.1099/jgv.0.000537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Timpe JM, Stamataki Z, Jennings A, Hu K, Farquhar MJ, Harris HJ, et al. Hepatitis C virus cell-cell transmission in hepatoma cells in the presence of neutralizing antibodies. Hepatology. 2008;47: 17–24. 10.1002/hep.21959 [DOI] [PubMed] [Google Scholar]
- 32.Lam AM, Murakami E, Espiritu C, Steuer HMM, Niu C, Keilman M, et al. PSI-7851, a Pronucleotide of beta-D-2 ‘-Deoxy-2 ‘-Fluoro-2 ‘-C-Methyluridine Monophosphate, Is a Potent and Pan-Genotype Inhibitor of Hepatitis C Virus Replication. Antimicrob Agents Chemother. 2010;54: 3187–3196. 10.1128/AAC.00399-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gao M, Nettles RE, Belema M, Snyder LB, Nguyen VN, Fridell RA, et al. Chemical genetics strategy identifies an HCV NS5A inhibitor with a potent clinical effect. Nature. 2010;465: 96–U108. 10.1038/nature08960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xiao F, Fofana I, Heydmann L, Barth H, Soulier E, Habersetzer F, et al. Hepatitis C virus cell-cell transmission and resistance to direct-acting antiviral agents. PLoS Pathog. 2014;10: e1004128 10.1371/journal.ppat.1004128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zeisel MB, Fofana I, Fafi-Kremer S, Baumert TF. Hepatitis C virus entry into hepatocytes: molecular mechanisms and targets for antiviral therapies. J Hepatol. 2011;54: 566–576. 10.1016/j.jhep.2010.10.014 [DOI] [PubMed] [Google Scholar]
- 36.Colpitts CC, Baumert TF. Hepatitis C virus cell entry: a target for novel antiviral strategies to address limitations of direct acting antivirals. Hepatol Int. 2016; 1–8. 10.1007/s12072-016-9724-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li G, De Clercq E. Current therapy for chronic hepatitis C: The role of direct-acting antivirals. Antiviral Res. 2017;142: 83–122. 10.1016/j.antiviral.2017.02.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Calland N, Dubuisson J, Rouillé Y, Séron K. Hepatitis C virus and natural compounds: a new antiviral approach? Viruses. 2012;4: 2197–2217. 10.3390/v4102197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ashfaq UA, Idrees S. Medicinal plants against hepatitis C virus. World J Gastroenterol. 2014;20: 2941–2947. 10.3748/wjg.v20.i11.2941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Séron K, Sahuc M-E, Rouillé Y. Natural products and hepatitis C virus. Natural Antimicrobial Agents. Springer Nature. Jean-Michel Mérillon and Céline Riviere; 2018. p. in press.
- 41.Liu S, Lu H, Zhao Q, He Y, Niu J, Debnath AK, et al. Theaflavin derivatives in black tea and catechin derivatives in green tea inhibit HIV-1 entry by targeting gp41. Biochim Biophys Acta. 2005;1723: 270–281. 10.1016/j.bbagen.2005.02.012 [DOI] [PubMed] [Google Scholar]
- 42.Henning SM, Aronson W, Niu Y, Conde F, Lee NH, Seeram NP, et al. Tea polyphenols and theaflavins are present in prostate tissue of humans and mice after green and black tea consumption. J Nutr. 2006;136: 1839–1843. 10.1093/jn/136.7.1839 [DOI] [PubMed] [Google Scholar]
- 43.Ganguly S, G TK, Mantha S, Panda K. Simultaneous Determination of Black Tea-Derived Catechins and Theaflavins in Tissues of Tea Consuming Animals Using Ultra-Performance Liquid-Chromatography Tandem Mass Spectrometry. PLoS ONE. 2016;11: e0163498 10.1371/journal.pone.0163498 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(A) Theaflavin (TF1) (a) chromatogram at 375 nm, Rt = 3.82 min, (b) UV spectrum, (c) mass spectrum in negative mode [M-H]- m/z 563; (B) Theaflavin-3-gallate (TF2) (a) chromatogram at 375 nm, Rt = 3.92 min, (b) UV spectrum, (c) mass spectrum in negative mode [M-H]- m/z 715; (C) Theaflavin-3,3'-digallate (a) chromatogram at 375 nm, Rt = 3.96 min, (b) UV spectrum, (c) mass spectrum in negative mode [M-H]- m/z 867.
(PDF)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.