Abstract
Anecdotal evidence indicates that naloxone prices have risen in recent years, but limited research has examined the magnitude of these increases and potential causes. We contribute nationally representative evidence to help answer each of these questions, including wholesale pricing data from a proprietary drug sales database spanning January 2006 to February 2017. We find that all formulations of naloxone increased in price since 2006 except for Narcan Nasal Spray. These cumulative increases totaled 2281% for the 0.4 MG single-dose products, 244% for the 2 MG single-dose products, 3797% for the 4 MG multi-dose products, and 469% for the 0.4 MG Evzio auto-injector. We believe that increased demand for naloxone from the opioid epidemic may explain the more gradual price increases for the 0.4 MG single-dose and 4 MG multi-dose products prior to 2012. On the other hand, we believe that the sudden, sustained prices increases occurring for all of the products since 2012 may be the result of a drug shortage for the 0.4 MG single-dose products and the fact that each naloxone product has historically been sold by only a single competitor.
Keywords: Naloxone, Narcan, Evzio, Price, Cost, Economics
1. Introduction
Drug overdose has become the leading cause of accidental death in the United States for people ages 25 to 64, with opioids implicated in nearly two-thirds of these cases (“Ten Leading Causes of Death and Injury,”; Rudd, Aleshire, Zibbel, & Gladden, 2016). A key strategy for reducing the number of deaths due to opioid overdoses has been to expand access to the opioid antidote naloxone. The U.S. Surgeon General recently recommended that people who interact with those at elevated risk of opioid overdose obtain naloxone and learn how to administer it (Surgeon General of the United States). Likewise, many states have enacted legislation allowing naloxone to be dispensed at a pharmacy without an individual prescription (“Comprehensive Addiction and Recovery Act of 2016”; National Conference of State Legislatures; The White House n.d).
At the same time, a growing body of anecdotal evidence suggests that the price of naloxone suddenly increased by more than 50% in recent years (Goodman, 2014; Gupta, Shah, & Ross, 2016; Jacobs, 2016; “Price Soars For Key Weapon Against Heroin Overdoses, 2015”). When combined with rising insurance deductibles, (Kaiser Family Foundation and the Health Research, & Educational Trust, September 14, 2016) this increase has raised concerns among stakeholders that patients will no longer be able to afford the drug (Harm Reduction Coalition, 2015; “Senator Leahy & Leading Senators Demand Answers About Outrageous Price Hike For Lifesaving Anti-Opioid Drug”).
To make an informed decision about how to respond to these price increases, it is important to answer two questions. First, is the price of naloxone increasing, and if so, by how much? Published literature on this topic appears to be scarce. Only one study measures naloxone prices, and in doing so relies on companies’ self-reported list prices from a drug compendium (Gupta et al., 2016). Second, if the price of naloxone is increasing, why is this happening? To our knowledge, no published studies have examined the economic factors that may be behind these increases.
Our study provides nationally representative evidence to help address these two questions. We use pharmaceutical sales data from a proprietary drug sales database, based on actual wholesale purchases of naloxone, to assess whether prices for the drug have increased, and how large these increases have been. We also examine whether certain economic factors, such as raw material costs, drug shortages, increases in demand, or limited competition among naloxone manufacturers appear to be associated with price increases.
2. Materials and methods
2.1. Data source
We rely on proprietary data from IQVIA to calculate national trends in wholesale naloxone prices from January 2006 to February 2017. The IQVIA, National Sales Perspective (NSP) (IQVIA, 2017 January 2006–February 2017) measures invoice transactions between wholesalers and both retail and non-retail settings of care, such as pharmacies and hospitals (see the Database Description in the Supplemental Materials for more information); this dataset generally excludes donations and direct manufacturer sales to end buyers that do not occur through a wholesaler. IQVIA estimates that the NSP data capture 88% of wholesale purchases of prescription drugs, and applies a projection methodology to obtain national estimates (IQVIA, March, 2016).
2.2. Sample
We focus on single-ingredient naloxone hydrochloride, and divide the market into distinct formulations to examine whether price levels and trends vary between different products. We define formulations by the dosage form and the number of MG of naloxone contained in the product, equal to the strength multiplied by the size of the package. For example, a 2 mL vial of naloxone with a strength of 1 mg/mL would be counted as a 2 MG product.
The six resulting formulations include single-dose injections, which are available as 0.4 MG and 2 MG vials or syringes; multi-dose injections, which are available as 4 MG vials; nasal sprays (brand name: Narcan Nasal Spray), which are available as 4 MG devices; and auto-injectors (brand name: Evzio), which are available as 0.4 MG and 2 MG syringes.
2.3. Measures
We calculate average prices by dividing total sales in dollars, un-adjusted for inflation, by the total number of MG of naloxone sold (equal to the number of MG in the product times the number of injections, sprays, or auto-injectors sold). This allows us to compare prices across formulations that deliver different doses, and which may be priced at different levels. These prices exclude any discounts that are not recorded on a wholesaler’s invoice, such as rebates that a manufacturer provides directly to an end buyer.
3. Results
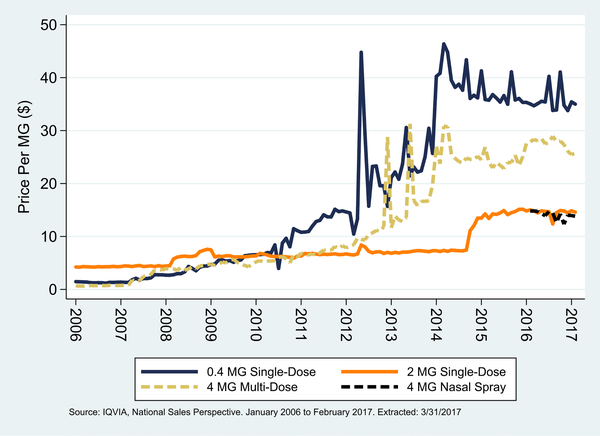
Fig. 1 shows the monthly price of naloxone for all formulations except for Evzio auto-injectors. In the latest month of data (February 2017), the 0.4 MG single-dose products cost $35.00/mg ($14.00/injection), the 2 MG single-dose products cost $14.59/mg ($29.18/ injection), the 4 MG nasal spray (Narcan Nasal Spray) cost $13.82/mg ($55.28/spray), and the 4 MG multi-dose products cost $25.72/mg ($102.88/injection). On a per MG basis, it appears that Narcan Nasal Spray priced itself comparably to the 2 MG single-dose products, which can also be used as a nasal spray when attached to a mucosal atomizer device (Harm Reduction Coalition, 2017).
Fig. 1.
Monthly price per MG of naloxone by formulation, excluding auto-injectors; January 2006 to February 2017.
In terms of price trends, each of the formulations shown in Fig. 1, besides the nasal spray, rose in price since 2006. The 0.4 MG single-dose formulations increased from $1.47/mg to $35.00/mg (2281%), the 2 MG single-dose formulations increased from $4.24/mg to $14.59/ mg (244%), and the 4 MG multi-dose formulations increased from $0.66/mg to $25.72/mg (3797%). For the 2 MG products, the increase occurred sharply in late 2014, while the other two formulations had been increasing steadily for many years until experiencing sharp price increases during 2012 to 2014. Since early 2015, however, it appears that the price of each of these products has stabilized, with smaller increases, and even in some cases decreases in price.
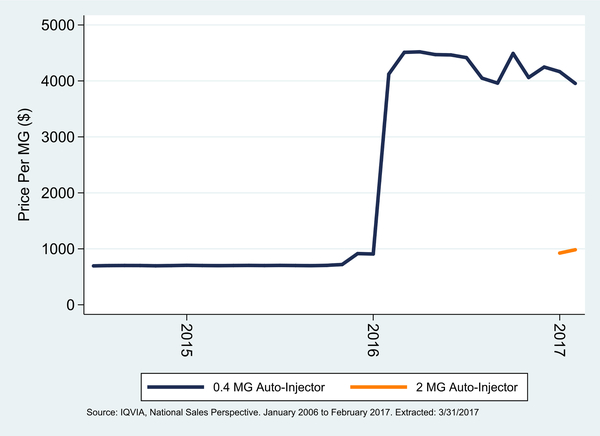
Fig. 2 shows the monthly price for naloxone auto-injectors, which are sold under the Evzio brand name. Note that this graph starts from July 2014, which was the first month of sales in the NSP data (Evzio was first approved in April 2014 (Food and Drug Administration, April 3, 2017)). Prices for these formulations were much higher. In the latest data, Evzio was priced at $3955.25/mg and $983.80/mg for the 0.4 MG and 2 MG products respectively ($1582.10 and $1967.70 per auto-injector). While the 2 MG auto-injector has only been sold since January 2017 and has had a steady price, the 0.4 MG auto-injector sharply raised its price in early 2016, with an aggregate increase from $695.36/mg to $3955.25/mg (469%) since its launch in July 2014.
Fig. 2.
Monthly price per MG of naloxone auto-injectors; July 2014 to February 2017.
For the raw data used to create both Figs. 1 and 2, see Table A in the Supplemental Materials.
4. Discussion
The price of naloxone does seem to have increased across most formulations of the drug (except for Narcan Nasal Spray). Furthermore, these increases outpaced overall inflation in pharmaceutical prices, which was 99% from January 2006 to February 2017 and 24% from July 2014 to February 2017 (Bureau of Labor Statistics, 2017). We examine four economic rationales that may explain these rising prices: three types of external shocks (raw material prices, supply disruptions, and increased demand), as well as business decisions resulting from limited competition.
4.1. Raw material prices
We reviewed internal FDA data that track the price and volume of bulk naloxone raw material imports into the United States, as self-reported by importers (“Food and Drug Administration. Office of Regulatory Affairs Reporting Analysis and Decision Support System (ORADSS),” January 2011 to September 2017). The price of naloxone raw materials has remained relatively stable since 2011 at around $0.01 to $0.02 per MG, or less than one-thousandth of the price of the finished products. This is consistent with other anecdotal evidence that naloxone raw materials are inexpensive (Jacobs, 2016).
4.2. Supply disruptions
We used archives of FDA’s public drug shortages website, maintained by the Wayback Machine, to search for naloxone shortages between August 2000 and April 2017 (Food and Drug Administration). We found two shortages listed, which appear to have been driven by issues with the 0.4 MG single-dose formulation: one from June 2010 to December 2010, and the other from February 2012 to July 2013. The second shortage appears to coincide with some of the sharp price increases for the 0.4 MG single-dose and the 4 MG multi-dose products seen in Fig. 1.
4.3. Increased demand
We evaluated surrogates for naloxone demand and found that this market is likely facing steadily increasing demand due to the opioid epidemic.
We observe that deaths involving opioid overdoses have risen steadily since 2006, from 6 per 100,000 to about 10.5 per 100,000 by 2015 (Centers for Disease Control and Prevention, 2016). In response to this trend, the Harm Reduction Coalition pledged to increase the number of doses of naloxone distributed to community groups from 140,053 doses in 2013 to 400,000 doses in 2016 (The White House; Wheeler, Jones, Gilbert, & Davidson, 2015).
However, steadily increasing demand over many years would also imply a steadily increasing price. While this was true for the 0.4 MG single-dose and 4 MG multi-dose products up until 2012, the 2 MG single-dose and 0.4 MG auto-injector experienced primarily sudden price increases, and at different points in time from each other.
4.4. Limited competition
One possible explanation for a sudden price increase is that a company finds itself with few competitors for a medically important drug and makes a business decision to raise prices.
In the naloxone market, conditions existed that may have provided firms with the leverage needed to raise prices. Each formulation was historically sold by a single company – Hospira for the 0.4 MG single-dose and 4 MG multi-dose products, Amphastar for the 2 MG single-dose product, Kaleo for the 0.4 MG and 2 MG auto-injectors, and Adapt for the 4 MG nasal spray (Rosenberg, 2015). Although these products were at least partial substitutes for one another, differences in administration meant that some users may not have been willing to switch if prices increased.
We were unable to estimate the extent to which naloxone producers could have unilaterally raised prices, since we lacked data on firms’ demand curves and production costs. However, we note that the 0.4 MG and 2 MG auto-injectors currently appear to be priced comparably to what previous research estimates as the maximum cost-effective level of a two-dose naloxone kit used by lay people – $4480/kit in 2012 dollars (Coffin & Sullivan, 2013).
While conditions in the naloxone market may have allowed for unilateral price increases, companies also appear to have had this option for many years without exercising it. It is possible that firms may have become more willing to increase prices given the increase in demand highlighted in Section 4.3, the higher launch price for Evzio, and the generally greater frequency of large price increases in the generic drug industry (United States Government Accountability Office, August, 2016).
For the list of companies holding active FDA approvals for naloxone as of April 3, 2017, see Table B in the Supplemental Materials.
5. Conclusions
The price of naloxone appears to have risen since 2006 for all formulations except for Narcan Nasal Spray. We believe that a drug shortage, increased demand, and limited competition have all contributed to these increases.
Our pricing metrics, while a useful approximation, are limited in three ways. One challenge is that they do not adjust for bulk discounts, which may lead to lower prices in hospitals and other larger settings of care than our data indicate. In addition, our metrics do not include any discounts or rebates that may be offered to institutional buyers, such as the Department of Veterans Affairs. Finally, these prices do not account for donations or direct sales by manufacturers to organizations such as schools or first responders.
Additional research is needed to further characterize the naloxone market and to identify whether price increases have the potential to affect patient access. This could include estimating the cost of manufacturing naloxone, calculating whether or how much patients or other purchasers pay out of pocket for the drug, and evaluating how willing patients are to cease use or switch to other formulations as prices increase.
Supplementary Material
HIGHLIGHTS.
All naloxone formulations except Narcan Nasal Spray increased in price since 2006.
Increases ranged from 244% to 3797% across these formulations.
Increased demand, a drug shortage, and limited competition may have contributed to price increases.
Acknowledgements
The authors would like to thank the following individuals for providing feedback on the manuscript: Peter Lurie, Douglas Throckmorton, Adam Kroetsch, Lisa Selker; and the following individuals for providing feedback on the initial analysis: Marta Wosinska, Joshua Lloyd.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
This study was carried out as part of the authors’ official duties at the FDA. The views expressed in this manuscript are the authors’ own, and do not reflect FDA’s views or policies.
Role of funding source
This study was carried out as part of the authors’ official duties at the Food and Drug Administration (FDA). The views expressed in this manuscript are the authors’ own, and do not reflect FDA’s views or policies. Any comments received from colleagues at the FDA were not required to be incorporated as a condition of submitting the manuscript.
Abbreviations:
- NSP
National Sales Perspective
- FDA
Food and Drug Administration
Footnotes
Supplementary data to this article can be found online at http://doi.org/10.1016/j.addbeh.2018.05.006.
Conflict of interest
All of the authors of this study are full-time employees at the FDA.
References
- Bureau of Labor Statistics Producer price indexes: Series WPU0638. Retrieved April 20 June 2001–March 2017, 2017.
- Centers for Disease Control and Prevention (December 16, 2016). Overview of an epidemic. Retrieved from https://www.cdc.gov/drugoverdose/data/index.html.
- Coffin PO, & Sullivan SD (2013). Cost-effectiveness of distributing naloxone to heroin users for lay overdose reversal. Annals of Internal Medicine, 158(1), 1–9. dx.doi.org/10.7326/0003-4819-158-1-201301010-00003. [DOI] [PubMed] [Google Scholar]
- Comprehensive Addiction and Recovery Act of 2016, Public Law 114–198 C.F.R. § Title IX, Section 902.
- Food and Drug Administration (2017, April 3). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. http://www.accessdata.fda.gov/scripts/cder/ob/. [Google Scholar]
- Food and Drug Administration Current drug shortages index. Retrieved from https://web.archive.org/web/20140330030227/http://www.fda.gov/Drugs/DrugSafety/DrugShortages/ucm050792.htm.
- Food and Drug Administration. Office of Regulatory Affairs Reporting Analysis and Decision Support System (ORADSS). (January 2011. to September 2017)
- Goodman DJ (2014, November 30). Naloxone, a drug to stop heroin deaths, is more costly, the police say. The New York Times. [Google Scholar]
- Gupta R, Shah ND, & Ross JS (2016). The rising price of naloxone — Risks to efforts to stem overdose deaths. New England Journal of Medicine, 375(23), 2213–2215. http://dx.doi.org/10.1056/NEJMp1609578. [DOI] [PubMed] [Google Scholar]
- Harm Reduction Coalition (2015. September). Harm Reduction Coalition Calls for Solutions to Naloxone Price Increases. Retrieved from http://harmreduction.org/overdose-prevention/calls-for-solutions-to-naloxone-price-increases/. [Google Scholar]
- Harm Reduction Coalition (2017. January). Naloxone Kit Materials. Retrieved from http://harmreduction.org/issues/overdose-prevention/tools-best-practices/od-kit-materials/. [Google Scholar]
- IQVIA (March 2016). National Sales Perspective - Sample Coverage of the Universe.
- IQVIA (2017). National Sales Perspective (NSP)™. Retrieved March 31 (January 2006 - February 2017).
- Jacobs H (2016, July 30). Big pharma is jacking up the price of the ‘antidote’ to the overdose crisis. Business Insider; Retrieved from http://www.businessinsider.com/price-of-naloxone-narcan-skyrocketing-2016-7. [Google Scholar]
- Kaiser Family Foundation and the Health Research & Educational Trust (September 14, 2016). Employer health benefits survey. Retrieved from http://kff.org/health-costs/report/2016-employer-health-benefits-survey/. [Google Scholar]
- National Conference of State Legislatures. Drug overdose immunity and good Samaritan Laws. Criminal Justice Program Retrieved from http://www.ncsl.org/research/civil-and-criminal-justice/drug-overdose-immunity-good-samaritan-laws.aspx Price Soars For Key Weapon Against Heroin Overdoses. National Public Radio. [Google Scholar]
- Rosenberg M (2015, July 1). Market structure for naloxone. Paper presented at the Exploring Naloxone Uptake and Use Food and Drug Administration. [Google Scholar]
- Rudd RA, Aleshire N, Zibbel JE, & Gladden RM (2016). Increases in drug and opioid overdose deaths - United States, 2000–2014. Morbidity and Mortality Weekly Report (MMWR), 64(50), 1378–1382. [DOI] [PubMed] [Google Scholar]
- Senator Leahy & Leading Senators Demand Answers About Outrageous Price Hike For Lifesaving Anti-Opioid Drug. [Press release]. Retrieved from https://www.leahy.senate.gov/press/senator-leahy-and-leading-senators-demand-answers-about-outrageous-price-hike_for-lifesaving-anti-opioid-drug.
- Surgeon General of the United States Surgeon general releases advisory on naloxone, an opioid overdose-reversing drug [Press release]. Retrieved from https://www.hhs.gov/about/news/2018/04/05/surgeon-general-releases-advisory-on-naloxone-an-opioid-overdose-reversing-drug.html.
- Ten Leading Causes of Death and Injury Injury Prevention & Control: Data and Statistics (WISQARS). Retrieved from http://www.cdc.gov/injury/wisqars/leadingcauses.html.
- The White House FACT SHEET: Obama administration announces public and private sector efforts to address prescription drug abuse and heroin use [press release]. Retrieved from https://www.whitehouse.gov/the-press-office/2015/10/21/fact-sheet-obama-administration-announces-public-and-private-sector.
- United States Government Accountability Office (August 2016). Part D Generic Prices Declined Overall, but Some Had Extraordinary Price Increases. Retrieved from http://www.gao.gov/assets/680/679022.pdf. [Google Scholar]
- Wheeler E, Jones TS, Gilbert MK, & Davidson PJ (2015). Opioid overdose prevention programs providing naloxone to laypersons - United States, 2014. Morbidity and Mortality Weekly Report (MMWR), 64(23), 631–635. [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.