Abstract
Objective
For patients with locally advanced rectal cancer (LARC), adjuvant chemotherapy selection following surgery remains a major clinical dilemma. Here we investigated the ability of circulating tumour DNA (ctDNA) to improve risk stratification in patients with LARC.
Design
We enrolled patients with LARC (T3/T4 and/ or N+) planned for neoadjuvant chemoradiotherapy. Plasma samples were collected pretreatment, postchemoradiotherapy and 4–10 weeks after surgery Somatic mutations in individual patient’s tumour were identified via massively parallel sequencing of 15 genes commonly mutated in colorectal cancer. We then designed personalised assays to quantify ctDNA in plasma samples. Patients received adjuvant therapy at clinician discretion, blinded to the ctDNA results.
Results
We analysed 462 serial plasma samples from 159 patients. ctDNA was detectable in 77%, 8.3% and 12% of pretreatment, postchemoradiotherapy and postsurgery plasma samples. Significantly worse recurrence-free survival was seen if ctDNA was detectable after chemoradiotherapy (HR 6.6; P<0.001) or after surgery (HR 13.0; P<0.001). The estimated 3-year recurrence-free survival was 33% for the postoperative ctDNA-positive patients and 87% for the postoperative ctDNA-negative patients. Postoperative ctDNA detection was predictive of recurrence irrespective of adjuvant chemotherapy use (chemotherapy: HR 10.0; P<0.001; without chemotherapy: HR 22.0; P<0.001). Postoperative ctDNA status remained an independent predictor of recurrence-free survival after adjusting for known clinicopathological risk factors (HR 6.0; P<0.001).
Conclusion
Postoperative ctDNA analysis stratifies patients with LARC into subsets that are either at very high or at low risk of recurrence, independent of conventional clinicopathological risk factors. ctDNA analysis could potentially be used to guide patient selection for adjuvant chemotherapy.
INTRODUCTION
Colorectal cancer is a major health burden globally, with approximately 30% of cases arising within the rectum.1 The current standard of care for patients with locally advanced rectal cancer (LARC), defined by either clinical stage T3/4 or node-positive disease, consists of neoadjuvant chemoradiotherapy followed by total mesorectal excision (TME) surgery. With local recurrence now uncommon,2 distant relapse is the major cause of morbidity and mortality.3 While recommended by guidelines,4,5 the impact of adjuvant (postoperative) chemotherapy on the risk of distant relapse remains questionable. Recent individual studies6–8 and meta-analyses3,9,10 of series from the modern era have failed to demonstrate any survival benefit for adjuvant chemotherapy. However, a survival benefit from adjuvant chemotherapy might be more clearly evident if we could identify and selectively treat patients who are at the greatest risk of distant recurrence.
The pathological stage after chemoradiotherapy (the yp stage) is the best available marker of distant recurrence risk in LARC. The best outcomes are seen in the 15%–27% of patients that achieve a pathological complete response (pCR—ypT0N0), whereas the worst outcomes are seen in those with persistently involved mesorectal lymph nodes (ypN+).11–13 Clinicians in routine care are increasingly using the pathological stage to guide adjuvant therapy decisions despite the lack of conclusive data to support this approach. Improved risk stratification could further refine clinical decision-making.
Circulating tumour DNA (ctDNA) containing tumour-specific DNA mutations can be found in the cell-free component of peripheral blood in a proportion of patients with solid tumours.14,15 The detection of ctDNA after apparently curative surgery for early-stage disease has been shown to be associated with a high risk of recurrence across multiple tumour types.16–19 Here, we report on the results of a correlative biomarker study in patients with TARC, where the primary aim was to demonstrate the association between the detection of ctDNA postoperatively and the risk of recurrence.
METHODS
Study design and participants
This prospective multicentre study recruited patients with TARC treated with curative intent at 11 Australian hospitals (Australian New Zealand Clinical Trials Registry number ACTRN12612000327886). Key eligibility criteria included a diagnosis of rectal adenocarcinoma, pretreatment MRI (or endorectal ultrasound if MRI was contraindicated) staging which demonstrated locally advanced disease (cT3-4N0 or cTanyN1-2), a staging CT chest/abdomen/pelvis prior to commencing preoperative chemoradiotherapy that demonstrated no metastatic disease, an Eastern Cooperative Oncology Group performance status of 0 to 2 and planned treatment with preoperative long course fluoropyrimidine-based chemoradiotherapy to be followed by TME surgery. Patients with a previous malignancy within the last 3 years were excluded. An elevated carcinoembryonic antigen (CEA) at diagnosis was not an exclusion criterion. The use of chemotherapy after surgery was at the discretion of the treating clinician, who was blinded to the ctDNA result.
Blood samples for ctDNA and CEA analysis were collected prior to commencing chemoradiotherapy (pretreatment), 4–6 weeks following completion of chemoradiotherapy (postchemoradiotherapy) and 4–10 weeks postrectal surgery (postoperative). Postoperative blood was collected prior to commencing any adjuvant chemotherapy. At each collection time-point, at least 30 mL of blood was drawn into EDTA tubes, centrifuged twice at 1200 g and 1800 g and plasma aliquoted into 10mL tubes for storage at −80°C.
Following completion of therapy, surveillance was performed according to standard of care, which included 3-monthly clinical review and CEA assays and annual CT imaging for 3 years. Serum CEA was measured by the local diagnostic laboratory at participating site, with CEA concentrations of <5 μg/L considered normal. Pathology reports from resection specimens were reviewed to assess tumour regression following chemoradiotherapy, with pCR defined as ypT0N0. All plasma and tumour samples were sent for analysis at the Ludwig Center at Johns Hopkins.
This study was approved by the human research ethics committees at each hospital, and all participants provided written informed consent.
Circulating tumour DNA analysis
Formalin-fixed paraffin-embedded tumour tissues from the pretreatment biopsy or the resection specimen were analysed for somatic mutations in 15 genes recurrently mutated in colorectal cancer, as previously described.17 Tumour sections were macro-dissected under a dissecting microscope to ensure a neoplastic cellularity of >30%. DNA was purified with a Qiagen FFPE Kit (Qiagen cat #56494). Primers were designed and sequencing results analysed as previously described.17
For each patient, one mutation identified in the tumour tissue was assessed for its presence in the plasma. When more than one somatic mutation was identified in the tumour tissue, the mutation with the highest mutant allele fraction (MAF) relative to the MAF in normal control DNA was selected for ctDNA analysis for that patient. Ten millilitres of plasma was purified from each patient using the QIAamp Circulating Nucleic Acid kit (Qiagen cat# 55114). To distinguish ctDNA in the plasma samples from artefactual variants arising during sequencing and sample preparation steps, we used Safe-SeqS, an error-reduction technology for detection of low-frequency mutations.20 Plasma DNA was aliquoted into 12 or 24 wells of a 96-well plate, so that an average of 0.5 to 3 ng DNA was contained in each well. The DNA from each well was then amplified (15 cycles) using primers containing unique identifier sequences (UIDs), which consisted of 14 random bases with an equal probability of A, C, T and G, to allow for the distinction of each template molecule. The amplified reactions were purified with AMPure XP beads (Beckman Coulter) and eluted in Buffer EB (Qiagen). One per cent of the purified PCR product was then amplified in a second round of PCR with universal primers. The second-round PCR products were purified with AMPure and sequenced on an Illumina MiSeq instrument.
The template-specific portion of the reads was matched to reference sequences using custom scripts written in SQL and C#. Reads from a common template molecule were then grouped based on the unique identifier sequences (UIDs) that were incorporated as molecular bar codes.20 Artefactual mutations introduced during the sample preparation or sequencing steps were reduced by requiring a mutation to be present in >90% of reads. Wells with fewer than 200 UIDs as a result of poor amplification were excluded. DNA from the peripheral blood lymphocytes of healthy individuals was used as a control in each experiment to identify potential false-positive mutations.
ctDNA was classified as detectable (ctDNA positive) or undetectable (ctDNA negative) based on a permutation test that compared the mutation frequency in the sample of interest with the mutation frequencies in controls. First, the MAF, defined as the ratio between the number of supermutants and the number of UIDs for the mutation of interest, was calculated for each well with >200 UIDs. The difference in the distributions of the MAFs between the sample of interest and the controls was then statistically evaluated via an exact permutation test, using the permTS function of the R perm package (R software V.3.3.1). The one-sided test was used to avoid attributing significance to a ctDNA-negative sample that had fewer supermutants than the associated control. A P value of 0.1 was then chosen as the significance threshold to classify a sample of interest as ctDNA positive (P<0.1) or ctDNA negative. Given the lack of a gold standard, a specificity of at least 0.90 was considered desirable, and a P value significance threshold equal to 0.1 yields 0.90 specificity.17
Statistical analysis
The overall sample size was event driven, with the expectation that approximately 24 of 160 (15%) unselected patients with locally advanced rectal cancer would experience recurrence in the first 2 years. Allowing for a 20% dropout rate, a total of 200 patients were recruited. A preplanned analysis was conducted after a median follow-up of 2 years. Differences in baseline characteristics between ctDNA-positive and ctDNA-negative patients were assessed using the Fisher’s Exact test for categorical variables and Mann-Whitney (rank sum) test for continuous variables. The primary outcome measure was recurrence-free survival (RFS), measured from date of surgery to documented first recurrence or death as a result of colorectal cancer, and was censored at last follow-up or non-colorectal cancer-related death. We fitted two types of models: for univariate analyses we used the Kaplan-Meier estimator with the log-rank test. HRs were estimated by univariate Cox proportional hazard models. For the multiple variable analysis, a Cox proportional hazard model was fitted. Ties in failure times were handled using Efron’s method and the proportional hazard assumption was tested by a global test of the Schoenfeld residuals. All analyses were performed using the survival package from the R software (V.3.4.1) where P values <0.05 were considered significant.
RESULTS
Clinicopathological characteristics and ctDNA status
Patient enrolment and the study design are presented in figure 1. We enrolled 200 patients between April 2012 and December 2015. Thirty-eight patients were excluded from subsequent analysis due to the postoperative plasma samples not being collected (n=32), there being insufficient tumour tissue for mutation analysis (n=5) and non-specific imaging findings at diagnosis later shown to represent metastatic disease (n=1). Using targeted massively parallel sequencing, at least one somatic mutation was identified in the primary tumour tissue of 159/162 (98.1%) of the remaining cases. For each of these patients, we designed a personalised Safe-SeqS assay to quantify ctDNA.20 A total of 462 plasma samples were assessed using these assays. A tumour-specific mutation was detected (ctDNA positive) prior to treatment in 122/159 patients (77%), postchemoradiotherapy in 12/144 patients (8.3%) and postoperative in 19/159 patients (12%). All but one patient with a positive postoperative ctDNA had a positive ctDNA at baseline. Postoperative ctDNA detection rates were numerically higher with increasing time intervals from surgery (9%, 13% and 14% for blood samples collected at 4–6 weeks, 6–8 weeks and 8–10 weeks from surgery; online supplementary figure 1) but this was not statistically significant (P=0.72).
Figure 1.
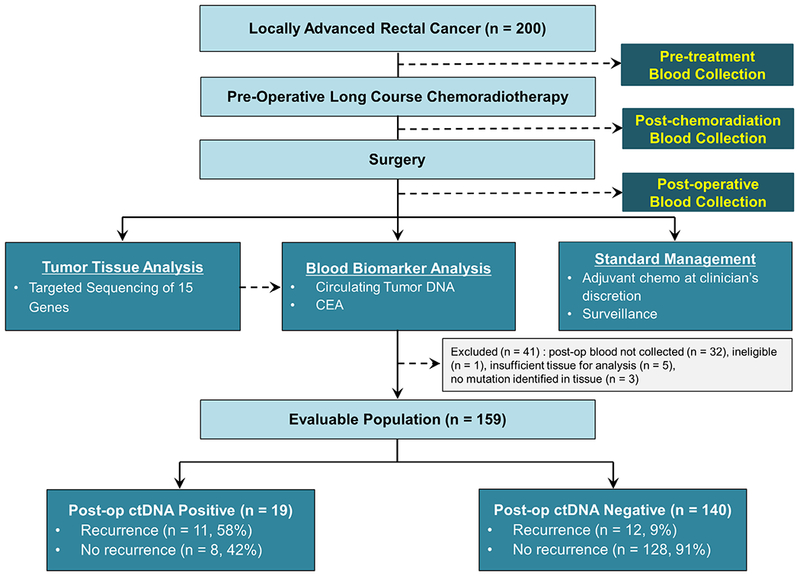
Patient enrolment, sample collections and evaluable population. CEA, carcinoembryonic antigen ; ctDNA, circulating tumour DNA; post-op, postoperative.
Baseline patient characteristics, staging information and ctDNA status pretreatment, postchemoradiotherapy and postsurgery for the 159 evaluable patients are shown in table 1. The median age was 62 years, 67% were male and 78% had clinical stage III (cTanyN1-2) disease at study entry. Following chemoradiation, 45% had a ypT3-4 tumour, 27% had persistent node-positive disease (ypN1-2) and 21% had a pathological complete response (ypT0N0). All but two patients achieved a R0 resection. The median time from completing chemoradiotherapy to surgery was 58 days. Single agent fluoropyrimidine adjuvant chemotherapy was given to 102/159 (64%) patients. Twelve of 34 (35%) patients with a pCR and 32/43 (74%) patients with ypN+disease received chemotherapy.
Table 1.
Clinicopathological characteristics and recurrence, according to ctDNA status
| Pretreatment ctDNA (n=159) |
Postchemoradiotherapy ctDNA (n=144) |
Postoperative ctDNA (n=159) |
|||||||
|---|---|---|---|---|---|---|---|---|---|
| Variable | Positive (n=122) | Negative (n=37) | P | Positive (n=12) | Negative (n=132) | P | Positive (n=19) | Negative (n=140) | P |
| Age, years | |||||||||
| Median | 63 | 59 | 0.69 | 61 | 62 | 0.97 | 59 | 63 | 0.97 |
| Range | 28–85 | 31–86 | 41–86 | 28–86 | 41–86 | 28–86 | |||
| Sex, n (%) | |||||||||
| Female | 40 (33) | 12 (32) | 1.00 | 4 (33) | 43 (33) | 1.00 | 6 (32) | 46 (33) | 1.00 |
| Male | 82 (67) | 25 (68) | 8 (67) | 89 (67) | 13 (68) | 94 (67) | |||
| Distance from anal verge (cm), n (%) | |||||||||
| 0–5 | 44 (36) | 13 (35) | 0.77 | 4 (33) | 48 (36) | 0.79 | 6 (32) | 51 (37) | 0.01 |
| > 5–10 | 55 (45) | 19 (51) | 5 (42) | 62 (47) | 5 (26) | 69 (49) | |||
| > 10 | 23 (19) | 5 (14) | 3 (25) | 22 (17) | 8 (42) | 20 (14) | |||
| Clinical disease stage, n (%) | |||||||||
| Stage II | 23 (19) | 12 (32) | 0.11 | 2 (17) | 29 (22) | 1.00 | 2 (11) | 33 (24) | 0.25 |
| Stage III | 99 (81) | 25 (68) | 10 (83) | 103 (78) | 17 (89) | 107 (76) | |||
| Pathological T stage, n (%) | |||||||||
| ypT0-2 | 65 (53) | 23 (62) | 0.36 | 5 (42) | 73 (55) | 0.38 | 5 (26) | 83 (59) | 0.01 |
| ypT3–4 | 57 (47) | 14(38) | 7 (58) | 59 (45) | 14 (74) | 57 (41) | |||
| Pathological N stage, n (%) | |||||||||
| ypN0 | 91 (75) | 25 (68) | 0.40 | 6 (50) | 97 (73) | 0.10 | 10 (53) | 106 (76) | 0.05 |
| ypN1-2 | 31 (25) | 12 (32) | 6 (50) | 35 (27) | 9 (47) | 34 (24) | |||
| Pathological complete response, n (%) | |||||||||
| Yes | 24 (20) | 10 (27) | 0.36 | 1 (9) | 28 (21) | 0.46 | 2 (11) | 32 (23) | 0.37 |
| No | 98 (80) | 27 (73) | 11 (89) | 104 (79) | 17 (89) | 108 (77) | |||
| Adjuvant chemotherapy, n (%) | |||||||||
| Yes | 40 (33) | 17 (46) | 0.17 | 4 (33) | 43 (33) | 1.00 | 11 (58) | 91 (65) | 0.61 |
| No | 82 (67) | 20 (54) | 8 (67) | 89 (67) | 8 (42) | 49 (35) | |||
| Recurrence at any site, n (%) | |||||||||
| Yes | 18 (15) | 5 (14) | 1.00 | 6 (50) | 15 (11) | 0.003 | 11 (58) | 12 (9) | <0.001 |
| No | 104 (85) | 32 (86) | 6 (50) | 117 (89) | 8 (42) | 128 (91) | |||
| Site of recurrence, n (%) | |||||||||
| Locoregional only | 3/18 (17) | 0/5 (0) | 1.00 | 0/6 (0) | 3/15 (20) | 0.53 | 1/11 (9) | 2/12 (17) | 1.00 |
| Distant±locoregional | 15/18 (83) | 5/5 (100) | 6/6 (100) | 12/15 (80) | 10/11 (91) | 10/12 (83) | |||
ctDNA, circulating tumour DNA.
No significant association was observed between pretreatment or postchemoradiotherapy ctDNA status and any clinicopathological factors. In contrast, postoperative ctDNA detection was associated with known high-risk pathological factors such as ypT3-4 and ypN1-2 stage. The relationships between prechemoradiotherapy and postchemoradiotherapy ctDNA status and pathological response are shown in table 2. There was no association between postchemoradiotherapy ctDNA status and pCR. The conversion of ctDNA status from positive at baseline to negative at 4–6 weeks after completing chemoradiotherapy was not associated with pCR (pCR vs non-pCR, 95% vs 88%, P=0.46).
Table 2.
Relationship between paired pre-CRT/post-CRT ctDNA status and pCR in 144 patients where both results are available
| pCR | Non-pCR | |
|---|---|---|
| Pretreatment ctDNA positive, n/total n (%) | ||
| Post-CRT ctDNA positive | 1/21 (5) | 11/91 (12) |
| Post-CRT ctDNA negative | 20/21 (95) | 80/91 (88) |
| Pretreatment ctDNA negative, n/total n (%) | ||
| Post-CRT ctDNA positive | 0/8 (0) | 0/24 (0) |
| Post-CRT ctDNA negative | 8/8 (100) | 24/24 (100) |
CRT, chemoradiation; ctDNA, circulating tumour DNA; pCR, pathological complete response.
ctDNA status and RFS
As of 15 May 2017, the median follow-up was 24 months (range, 1–55 months). During this period, 23 (15%) patients experienced recurrence, including 8/57 (16%) not treated with adjuvant chemotherapy and 15/102 (15%) treated with adjuvant chemotherapy. There was no difference in RFS between patients with detectable ctDNA at baseline (pretreatment) and those with no detectable ctDNA (HR 1.1; 95% CI), 0.42 to 3.0; figure 2A). In striking contrast, patients with a ctDNA-positive status after chemoradiotherapy or after surgery had an increased risk of recurrence (figure 2B,C). After chemoradiotherapy, 6 of 12 (50%) patients with positive ctDNA and 15 of 132 (11%) with negative ctDNA experienced recurrence (HR 6.6, 95% CI 2.6 to 17; P<0.001). After surgery, 11 of 19 (58%) patients with positive ctDNA and 12 of 140 (8.6%) with negative ctDNA have recurred (HR 13, 95% CI 5.5 to 31, P<0.001). The Kaplan-Meier estimates of RFS at 3 years were 50% (95% CI, 28% to 88%) and 85% (95% CI, 79% to 93%) for the postchemoradiotherapy ctDNA-positive and ctDNA-negative groups, and 33% (95% CI 16% to 72%) and 87% (95% CI 79% to 95%) for the postoperative ctDNA-positive and ctDNA-negative groups.
Figure 2.
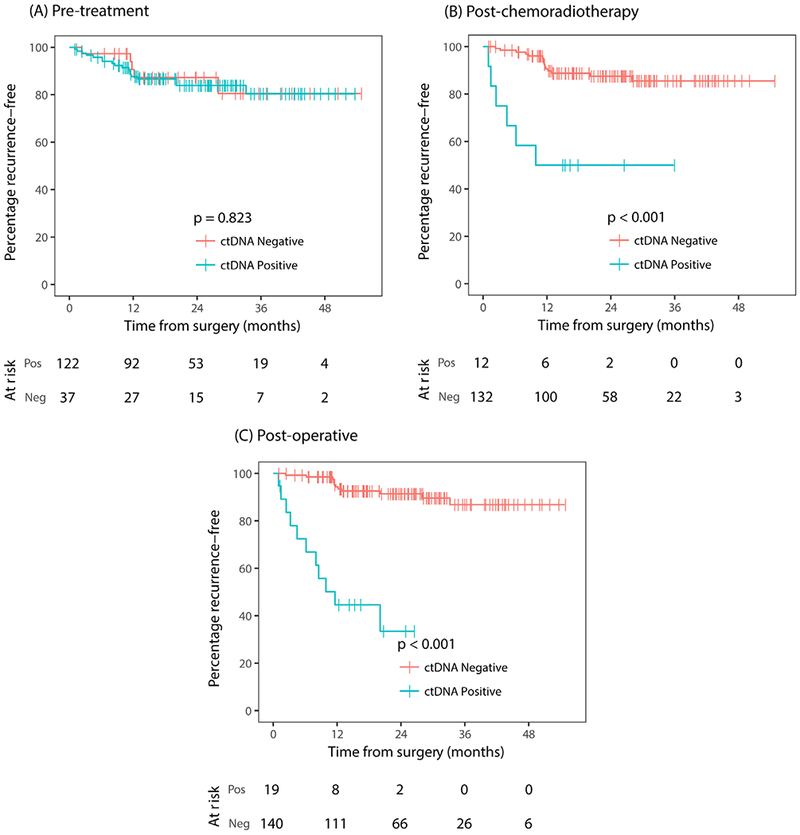
Kaplan-Meier estimates of recurrence-free survival according to ctDNA status (A) at diagnosis (pretreatment), (B) 4–6 weeks after completion of chemoradiotherapy (postchemoradiotherapy), (C) 4–10 weeks following surgery (postoperative). ctDNA, circulating tumour DNA; neg, negative; pos, positive.
We explored the association between postoperative ctDNA status and RFS stratified by chemotherapy use (figure 3A,B). Postoperative ctDNA-positive status was strongly predictive of recurrence irrespective of adjuvant chemotherapy (chemo: HR 10; 95% CI, 3.4 to 29, P<0.001; no chemo: HR 22; 95% CI 4.2 to 110, P<0.001).
Figure 3.
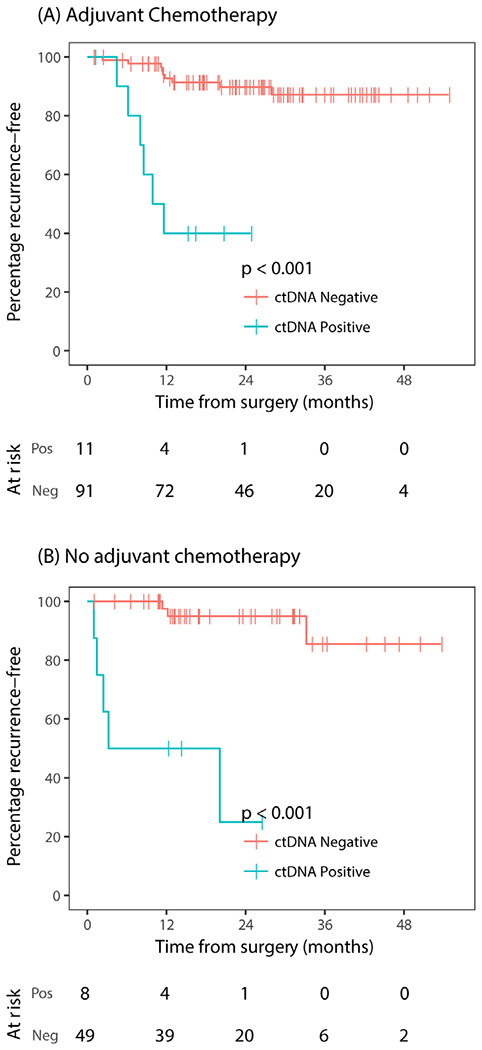
Kaplan-Meier estimates of recurrence-free survival according to postoperative ctDNA status stratified by adjuvant chemotherapy use. (A) Treated with adjuvant chemotherapy, (B) not treated with adjuvant chemotherapy. ctDNA, circulating tumour DNA; neg, negative; pos, positive.
Clinicopathological variables significantly associated with RFS in univariate analysis were sex, ypT stage, ypN stage and postoperative CEA status (table 3). A trend for association was observed for pCR status (HR 0.32; 95% CI 0.08 to 1.4; P=0.12). Post-operative ctDNA positivity remained highly predictive of recurrence among patients with pathological low risk (pCR, ypT0-2, ypN0) or high risk (ypT3-4, ypN+) disease (figure 4A–F). Specifically, postoperative ctDNA positivity was highly predictive of recurrence even after considering the two pathological variables increasingly being used in the clinic to guide adjuvant chemotherapy decisions, pCR and ypN+stage (pCR: HR 15, 95% CI 0.94 to 240, P=0.01; figure 4A; ypN+: HR 13, 95% CI 4.2 to 43, P<0.001; figure 4F).
Table 3.
Recurrence-free survival analysis by clinicopathological variables and postoperative ctDNA status
| Univariate analysis |
Multivariate analysis |
|||
|---|---|---|---|---|
| Variable | HR | P | HR | P |
| Sex: male versus female | 3.3 | 0.05 | 2.8 | 0.10 |
| ypT stage: T3–T4 versus T0–T2 | 7.2 | <0.001 | 5.2 | 0.04 |
| ypN stage: n+ versus N0 | 4.3 | <0.001 | 2.5 | 0.07 |
| pCR: no versus yes | 3.1 | 0.12 | 1.8 | 0.59 |
| Adjuvant chemotherapy: no versus yes | 1.0 | 0.97 | 0.65 | 0.39 |
| Postoperative CEA: normal versus elevated | 13 | <0.001 | 5.1 | 0.01 |
| Postoperative ctDNA: positive versus negative | 13 | <0.001 | 6.0 | <0.001 |
CEA, carcinoembryonic antigen; ctDNA, circulating tumour DNA; pCR, pathological complete response.
Figure 4.
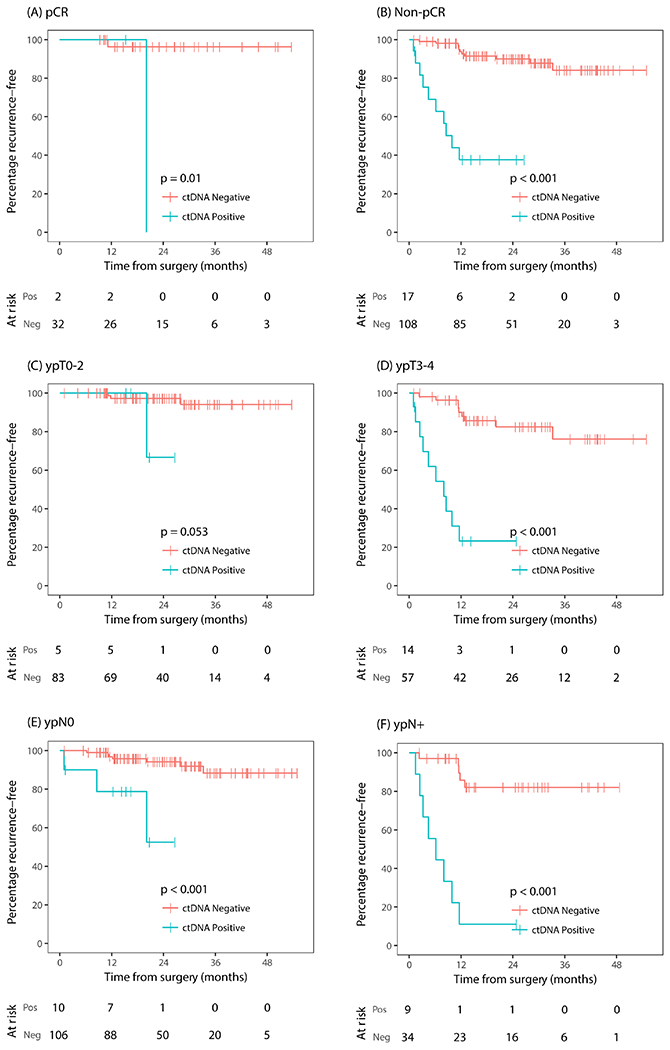
Kaplan-Meier estimates of recurrence-free survival according to postoperative ctDNA status stratified by pathological risk factors. (A) Pathological complete response, (B) pathological non-complete response, (C) pathological T0-2, (D) pathological T3-4, (E) pathological node negative disease (ypN0), (F) pathological node positive disease (ypN+). ctDNA, circulating tumour DNA; neg, negative; pos, positive.
To adjust for multiple variables in a single model, we used a Cox proportional hazard model. Postoperative ctDNA status remained the strongest independent predictor of RFS (HR 6.0; 95% CI 2.2 to 16, P<0.001; table 3), followed by ypT stage (HR 5.2, 95% CI 1.1 to 24, P=0.036) and postoperative CEA (HR 5.1, 95% CI 1.4 to 18, P=0.012). The HR for ctDNA increased to 8.7 (95% CI, 3.5 to 22) when postoperative CEA was excluded from the multivariate model. A global test did not reject the proportional hazard assumption (P=0.43).
Serial ctDNA, CEA and recurrence
Baseline (pretreatment) and postoperative CEA results were available in 155 and 154 cases, respectively. Of these, CEA was elevated in 41 patients (26%) at baseline and in 7 patients (4.5%) postsurgery. A trend for association was observed between pretreatment CEA status and RFS (HR 2.1, 95% CI 0.90 to 5.0, P=0.09). The relationships between postoperative ctDNA, postoperative CEA and recurrence status are shown in table 4. CEA was elevated postoperatively in 5 of 22 cases (23%) that recurred and in 2 of 132 cases (1.5%) that did not recur. For patients that experienced recurrence, all five cases with an elevated postoperative CEA also had detectable postoperative ctDNA, but only 5 of the 11 patients (45%) with detectable ctDNA postoperatively had an elevated CEA postoperatively. Postoperative ctDNA assessment added significant prognostic value to patients with a non-elevated postoperative CEA (HR, 8.8; 95% CI 3.2 to 24; P<0.001; figure 5).
Table 4.
Relationship between postoperative ctDNA, postoperative CEA and recurrence status in all patients
| Recurrence | No recurrence | |
|---|---|---|
| Postoperative ctDNA positive, —n/total n (%) | ||
| Postoperative CEA elevated | 5/11 (45) | 1/8 (13) |
| Postoperative CEA not elevated | 6/11 (55) | 7/8 (87) |
| Postoperative ctDNA negative, n/total n (%) | ||
| Postoperative CEA elevated | 0/11 (0) | 1/124 (1) |
| Postoperative CEA not elevated | 11/11 (100) | 123/124 (99) |
Postoperative CEA data were not available in one case with recurrence and four cases without recurrence.
CEA, carcinoembryonic antigen; ctDNA, circulating tumour DNA.
Figure 5.
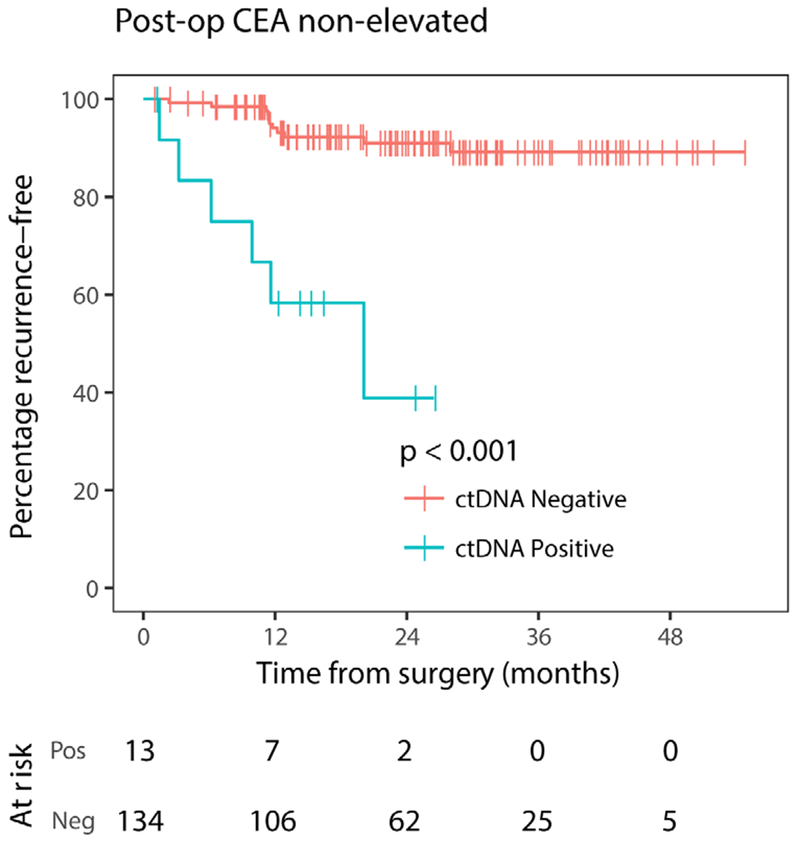
Kaplan-Meier estimates of recurrence-free survival according to postoperative ctDNA status in patients with non-elevated postoperative CEA. CEA, carcinoembryonic antigen; ctDNA, circulating tumour DNA; neg, negative; pos, positive; post-op, postoperative.
Serial ctDNA status (pretreatment, postchemoradiotherapy and postoperative) for patients with or without recurrence are shown in online supplementary figure 2. Of the 23 cases with recurrence, 17 (74%) had recurred within 12 months after surgery. Of the eight patients with ctDNA-positive status postoperatively that have not recurred to date (follow-up time: IQR 13–24 months), five had received adjuvant chemotherapy.
DISCUSSION
While multiple advances have been made in the management of LARC, patient selection for adjuvant chemotherapy after surgery, and the agent(s) of choice remain major clinical dilemmas. While guidelines currently recommend adjuvant chemotherapy in all patients with LARC, there is limited evidence in the modern era to confirm an overall survival benefit. While the use of adjuvant oxaliplatin-based treatment appears promising in patients with pathological node-positive disease, to date only an impact on disease-free survival has been demonstrated with an overall survival benefit yet to be proven.21 Better predictors of patients most likely to benefit from adjuvant chemotherapy are urgently needed.
Consistent with our recent evaluation of patients with stage II colon cancer,17 the present study confirms the promise of a novel approach to recurrence risk assessment, namely the examination of postoperative blood samples for tumour-specific DNA molecules (ctDNA) providing evidence of minimal residual disease after surgery. In the current study, we have demonstrated the ability of postoperative ctDNA analysis to identify patients with LARC at an extremely high risk of recurrence after apparently curative surgery; patients with a positive ctDNA having an estimated 3-year RFS of 33% compared with 87% in those with a negative postoperative ctDNA (HR 13; 95% CI 5.5 to 31; P<0.001). The prognostic impact of postoperative ctDNA appears even more pronounced in patients not treated with adjuvant chemotherapy (HR 22; 95% CI 4.2 to 110, P<0.001), an HR similar to that observed in our stage II colon cancer study (HR 18).
As serial plasma samples were not collected during adjuvant chemotherapy, we are unable to determine the impact of adjuvant therapy on ctDNA status. So, while we can define two subsets at high and low risk of recurrence (ctDNA detectable and ctDNA undetectable), we cannot provide evidence that the high-risk population will benefit from adjuvant chemotherapy. A major confounding factor is that chemotherapy administration in this study was at investigator discretion and is more likely to be given to high risk (74% of ypN+) than to low risk (35% of pCR) patients. That the recurrence risk is numerically lower in the higher risk patients that received chemotherapy is consistent with treatment benefit in the ctDNA-positive patients. However, the CIs are wide, and prospective randomised studies with treatment stratified based on ctDNA status are required to demonstrate any chemotherapy benefit in the high-risk group. The risk of recurrence in patients with a negative postoperative ctDNA appears low, whether or not chemotherapy was administered, and this patient subset may be able to avoid adjuvant treatment with minimal impact on recurrence risk. Again, further prospective studies are required to define the impact of withholding adjuvant chemotherapy in this low-risk subgroup.
The prognostic value of pathological risk factors (ypT and ypN stage) that we observed in our study are consistent with previous reports.12 That the association between pCR and a reduced risk of recurrence is not statistically significant is likely due to the limited sample size. Importantly, the strong prognostic impact of postoperative ctDNA was maintained when patients are stratified by pathological risk. Among patients with pathological high-risk (ypN+) disease, the detection of ctDNA identified those at very high and low risk of recurrence (3 year RFS: ctDNA positive vs ctDNA negative, 11% vs 82%; HR 13, 95% CI 4.2 to 43, P<0.001). For those with pathological low-risk disease (pCR), although postoperative ctDNA was positive in only two patients, this test was able to identify the rare patients at high risk of recurrence (HR 15, 95% CI 0.94 to 240, P=0.01). As discussed above, these analyses are confounded by the pathological risk influencing the likelihood of patients receiving chemotherapy, as clinicians were blinded to ctDNA results.
To our knowledge, this is the first study to define the prognostic significance of an elevated CEA postoperatively in LARC, seen in 7 (4.5%) of the 154 evaluable patients. As per other recent protocols,22,23 patients with LARC were not excluded from entry into our study due to an elevated CEA at diagnosis, as this can be present in patients with localised disease and normalise postsurgery. Current practice guidelines for rectal cancer treatment do not comment on postoperative CEA analysis or advise about the management of a patient with an elevated CEA after surgery. It would be of interest to examine this question in further studies, including the value of restaging to exclude distant progression prior to commencing adjuvant treatment. Notably, for those patients with a normal CEA, ctDNA detection remained associated with a high risk of recurrence (HR 8.8, 95% CI 3.2 to 24; P<0.001).
A high proportion of patients (77%) had detectable ctDNA at diagnosis, consistent with previous studies reporting detectable ctDNA in a high proportion of patients with early-stage colorectal cancer.14,24 In our study, ctDNA remained detectable in far fewer patients after chemoradiation (8%) and after surgery (12%), indicating that for the great majority, the ctDNA detected at baseline is being released from the primary tumour rather than from distant sites. This would explain why ctDNA at diagnosis is not prognostic. In other words, it is only when the primary tumour has been removed that the presence of ctDNA indicates residual micrometastatic disease, which in turn is associated with the risk of recurrence. The increase in the number of ctDNA-positive patients from postchemoradiation (8.3%) to postsurgery (12%) is consistent with micrometastatic disease progressing in the 8 to 12-week period between collecting these samples, with the increased bulk of disease now releasing sufficient ctDNA to be detectable.
The rapid decline in the number of ctDNA-positive patients from baseline to postchemoradiation reflects the substantial downstaging achieved with neoadjuvant treatment. This treatment eradicates most of the primary tumour, with the remaining neoplastic cells releasing insufficient DNA into the plasma to be detectable. The poor correlation between postchemoradiation ctDNA analysis (measured 4–6 weeks postneoadjuvant therapy) and pCR suggests that ctDNA analysis within a short interval following completion of chemoradiation cannot differentiate between minimal and no residual disease or be used to select patients for a non-operative (wait and watch) approach.25
There are potential limitations to our study, including the modest sample size and the analysis of multiple patient subsets. However, the results of this study are consistent with others that have demonstrated the potential use of ctDNA analysis as a prognostic tool.16–19,26 All of these studies highlight the potential clinical use of this biomarker as a predictor of risk of recurrence, but in all instances prospective studies demonstrating whether a change in adjuvant therapy guided by ctDNA analysis can positively impact outcomes are required before adopting this test into routine care. Such a study is now being planned for patients with LARC (Dynamic-Rectal study— ACTRN12617001560381).
Supplementary Material
Significance of this study.
What is already known on this subject?
-
▶
The survival benefit of adjuvant (postoperative) chemotherapy in patients with locally advanced rectal cancer remains debatable in the modern era, and improvement in risk stratification could refine clinical decision-making.
-
▶
Tumour-specific DNA can be detected in the peripheral blood (circulating tumour DNA) of patients with colorectal cancer and other solid tumours.
-
▶
Our previous work demonstrated that the detection of circulating tumour DNA after curative intent surgery is predictive of a very high risk of recurrence in stage II colon cancer.
What are the new findings?
-
▶
This study provides the first evidence that circulating tumour DNA analysis after curative intent surgery for locally advanced rectal cancer could stratify patients into subsets at very high risk or low risk of recurrence.
-
▶
The strong prognostic impact of postoperative circulating tumour DNA status appears to be independent of other known pathological risk factors.
-
▶
Detection of circulating tumour DNA at diagnosis, prior to any treatment, was not predictive of disease recurrence.
How might it impact on clinical practice in the foreseeable future?
-
▶
Postoperative circulating tumour DNA analysis could help clinicians identify and selectively treat patients who are at the greatest risk of distant recurrence. Given the low risk of recurrence in ctDNA-negative patients, this patient subset may be able to avoid any adjuvant treatment with minimal risk.
-
▶
This study lays the foundation for future randomised study of locally advanced rectal cancer to assess if a management approach based on circulating tumour DNA testing could improve patient outcome.
Acknowledgements
We thank Cherie Blair for expert sample management, Matthew Chapman and Jeffrey Nolan for project management and Victorian Cancer Biobank for sample processing.
Funding Australian National Health and Medical Research Council (GNT1026230), the National institute of Health (CA62924, GM07309, and P30-CA006973), Virginia and D K Ludwig Fund for Cancer Research, The John Templeton Foundation, The Conrad R Hilton Foundation, The Sol Goldman Sequencing Facility at Johns Hopkins.
Footnotes
Additional material is published online only. To view, please visit the journal online (http://dx.doi.org/10.1136/gutjnl-2017-315852).
Competing interests KK, NP, LD and BV are founders of PapGene and Personal Genome Diagnostics and members of the Scientific Advisory Boards of Morphotek and Sysmex-inostics. iK is an employee of PapGene. These companies and others have licensed patent applications on genetic technologies from Johns Hopkins, some of which result in royalty payments to KK, NP, LD, BV, and IK. The terms of these arrangements are being managed by Johns Hopkins University in accordance with its conflict of interest policies.
Ethics approval Melbourne Health.
Provenance and peer review Not commissioned; externally peer reviewed.
REFERENCES
- 1.Howlader N, Noone A, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2014. Bethesda, MD: National Cancer Institute, 2016. [Google Scholar]
- 2.Sauer R, Becker H, Hohenberger W, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 2004;351:1731–40. [DOI] [PubMed] [Google Scholar]
- 3.Breugom AJ, Swets M, Bosset JF, et al. Adjuvant chemotherapy after preoperative (chemo)radiotherapy and surgery for patients with rectal cancer: a systematic review and meta-analysis of individual patient data. Lancet Oncol 2015;16:200–7. [DOI] [PubMed] [Google Scholar]
- 4.Glynne-Jones R, Wyrwicz L, Tiret E, et al. Rectal cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 2017;28:iv22–iv40. [DOI] [PubMed] [Google Scholar]
- 5.National Comprehensive Cancer Network. Rectal Cancer (Version 3.2017).https://www.nccn.org/professionals/physician_gls/pdf/rectal.pdf. (accessed 1 Dec 2017)
- 6.Bosset JF, Calais G, Mineur L, et al. Fluorouracil-based adjuvant chemotherapy after preoperative chemoradiotherapy in rectal cancer: long-term results of the EORTC 22921 randomised study. Lancet Oncol 2014;15:184–90. [DOI] [PubMed] [Google Scholar]
- 7.Sainato A, Cernusco Luna Nunzia V, Valentini V, et al. No benefit of adjuvant fluorouracil leucovorin chemotherapy after neoadjuvant chemoradiotherapy in locally advanced cancer of the rectum (LARC): long term results of a randomized trial (I-CNR-RT). Radiother Oncol 2014;113:223–9. [DOI] [PubMed] [Google Scholar]
- 8.Breugom AJ, van Gijn W, Muller EW, et al. Adjuvant chemotherapy for rectal cancer patients treated with preoperative (chemo)radiotherapy and total mesorectal excision: a Dutch Colorectal Cancer Group (DCCG) randomized phase III trial. Ann Oncol 2015;26:696–701. [DOI] [PubMed] [Google Scholar]
- 9.Bujko K, Glynne-Jones R, Bujko M. Does adjuvant fluoropyrimidine-based chemotherapy provide a benefit for patients with resected rectal cancer who have already received neoadjuvant radiochemotherapy? A systematic review of randomized trials. Ann Oncol 2010;21:1743–50. [DOI] [PubMed] [Google Scholar]
- 10.Petrelli F, Coinu A, Lonati V, et al. A systematic review and meta-analysis of adjuvant chemotherapy after neoadjuvant treatment and surgery for rectal cancer. Int J Colorectal Dis 2015;30:447–57. [DOI] [PubMed] [Google Scholar]
- 11.Fokas E, Liersch T, Fietkau R, et al. Tumor regression grading after preoperative chemoradiotherapy for locally advanced rectal carcinoma revisited: updated results of the CAO/ARO/AIO-94 trial. J Clin Oncol 2014;32:1554–62. [DOI] [PubMed] [Google Scholar]
- 12.Maas M, Nelemans PJ, Valentini V, et al. Long-term outcome in patients with a pathological complete response after chemoradiation for rectal cancer: a pooled analysis of individual patient data. Lancet Oncol 2010;11:835–44. [DOI] [PubMed] [Google Scholar]
- 13.Park IJ, You YN, Agarwal A, et al. Neoadjuvant treatment response as an early response indicator for patients with rectal cancer. J Clin Oncol 2012;30:1770–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bettegowda C, Sausen M, Leary RJ, et al. Detection of circulating tumor DNA in earlyand late-stage human malignancies. Sci Transl Med 2014;6:224ra24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cohen JD, Javed AA, Thoburn C, et al. Combined circulating tumor DNA and protein biomarker-based liquid biopsy for the earlier detection of pancreatic cancers. Proc Natl Acad Sci U S A 2017;114:10202–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Diehl F, Schmidt K, Choti MA, et al. Circulating mutant DNA to assess tumor dynamics. Nat Med 2008;14:985–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tie J, Wang Y, Tomasetti C, et al. Circulating tumor DNA analysis detects minimal residual disease and predicts recurrence in patients with stage II colon cancer. Sci Transl Med 2016;8:346ra92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Garcia-Murillas I, Schiavon G, Weigelt B, et al. Mutation tracking in circulating tumor DNA predicts relapse in early breast cancer. Sci Transl Med 2015;7:ra133. [DOI] [PubMed] [Google Scholar]
- 19.Newman AM, Bratman SV, To J, et al. An ultrasensitive method for quantitatingcirculating tumor DNA with broad patient coverage. Nat Med 2014;20:548–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kinde I, Wu J, Papadopoulos N, et al. Detection and quantification of rare mutations with massively parallel sequencing. Proc Natl Acad Sci U S A 2011;108:9530–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhao L, Liu R, Zhang Z, et al. Oxaliplatin/fluorouracil-based adjuvant chemotherapy for locally advanced rectal cancer after neoadjuvant chemoradiotherapy and surgery: a systematic review and meta-analysis of randomized controlled trials. Colorectal Dis 2016;18:763–72. [DOI] [PubMed] [Google Scholar]
- 22.Rödel C, Graeven U, Fietkau R, et al. Oxaliplatin added to fluorouracil-based preoperative chemoradiotherapy and postoperative chemotherapy of locally advanced rectal cancer (the German CAO/ARO/AIO-04 study): final results of the multicentre, open-label, randomised, phase 3 trial. Lancet Oncol 2015;16:979–89. [DOI] [PubMed] [Google Scholar]
- 23.Gérard JP, Azria D, Gourgou-Bourgade S, et al. Clinical outcome of the ACC ORD 12/0405 PRODIGE 2 randomized trial in rectal cancer. J Clin Oncol 2012;30:4558–65. [DOI] [PubMed] [Google Scholar]
- 24.Tie J, Wang Y, Kinde I, et al. Circulating tumor DNA (ctDNA) in nonmetastatic colorectal cancer (CRC): Potential role as a screening tool. Journal of Clinical Oncology 2015;33:518.25559802 [Google Scholar]
- 25.Martens MH, Maas M, Heijnen LA, et al. Long-term outcome of an organ preservation program after neoadjuvant treatment for rectal cancer. J Natl Cancer Inst 2016;108:djw171. [DOI] [PubMed] [Google Scholar]
- 26.Pietrasz D, Pécuchet N, Garlan F, et al. Plasma circulating tumor DNA in pancreatic cancer patients is a prognostic marker. Clin Cancer Res 2017;23:116–23. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


