Abstract
Background:
Some heavy metals (e.g., arsenic, cadmium, lead, mercury) have been associated with obesity and obesity comorbidities. The analytical approach for those associations has typically focused on individual metals. There is a growing interest in evaluating the health effects of cumulative exposure to metal mixtures.
Objectives:
We utilized our Environmental Risk Score (ERS), a summary measure to examine the risk of exposure to multi-pollutants in epidemiologic research, to evaluate the associations of cumulative exposure to a mixture of correlated heavy metals with obesity and its comorbidities including hypertension, and type-2 diabetes mellitus (T2DM) while accounting for high degree correlations and interactions among metal mixtures components.
Methods:
We examined blood and urinary markers of 18 heavy metals among 9,537 adults in NHANES 2003–2014. We randomly split data into a training set for the construction of ERS (n=6,675) and a testing set for the evaluation of its statistical performance (n=2,862). ERS of heavy metal mixtures was computed for waist circumference using adaptive elastic-net (AENET) with 189 predictors including 18 main effects, 18 squared terms, and 153 pairwise interactions of heavy metals. Regression analyses with complex survey designs were performed to assess the associations of ERS with other obesity measures, hypertension and T2DM.
Results:
7 main effects (blood lead, blood cadmium, blood mercury, and urinary markers of monomethylarsonic acid (MMA), barium, mercury and thallium), 4 squared terms (blood cadmium, urinary cadmium, urinary antimony and urinary tungsten), and 7 pairwise interactions (blood lead & urinary cadmium, blood lead & urinary MMA, blood lead & urinary uranium, urinary cadmium & urinary MMA, urinary dimethylarsinic acid (DMA) & urinary tungsten, urinary MMA & urinary cobalt, and urinary lead & urinary antimony) were selected by AENET for construction of ERS of waist circumference-related metal mixtures. An increase in ERS from 10th percentile to 90th percentile in the overall study population was significantly associated with 4.50 kg/m2 (95% CI: 4.06, 4.94) higher BMI, 4.16 mm (95% CI: 3.56, 4.76) higher skinfold thickness, and 4.11 kg (95% CI: 0.83, 7.40) higher total body fat, independent of age, sex, race/ethnicity, education, smoking status, physical activity and NHANES cycle (Ps < 0.05). Significant associations of ERS with both hypertension and T2DM were also observed (Ps < 0.05).
Conclusions:
Our study suggests that cumulative exposure to heavy metals as mixtures is associated with obesity and its related chronic conditions such as hypertension and T2DM. Additional research is needed to confirm these findings in longitudinal settings.
Keywords: Heavy metals, Mixtures, Obesity, Obesity-related Comorbidities
GRAPHICAL ABSTRACT

1. INTRODUCTION
Obesity is of public health concern worldwide(WHO, 2017). In the United States, the prevalence of obesity among adults aged 20 and over increased from 30.5% in 1999–2000 to 37.7% in 2011–2014 (Ogden et al., 2015). The health consequences of obesity are not trivial. Disease burdens such as hypertension and type-2 diabetes mellitus (T2DM) have been attributed to obesity (Must, 1999). With more than one-third of U.S. adults being obese, there is a great interest in identifying risk factors for obesity (Thayer et al., 2012). Beyond dietary factors and physical activity, previous animal and human studies suggest that heavy metals may play important roles in the etiology of obesity. A cross-sectional epidemiologic study in the general U.S. population found that barium and thallium were positively while cadmium, cobalt, cesium, and lead were inversely associated with body mass index (BMI) and/or waist circumference (WC) (Padilla et al., 2010). In the Korea National Health and Nutrition Examination Survey, blood mercury was positively associated with visceral adiposity but negatively associated with body fat percentage (J. S. Park et al., 2017; Park and Lee, 2013). Another cross-sectional study with the elderly Koreans living in coastal areas reported a positive association between blood mercury and waist-to-hip ratio (You et al., 2011). In vivo evidence suggests that perturbation of the hypothalamic dopaminergic system, endoplasmic reticulum stress, impaired adipogenesis and adipocytokines secretion might serve as potential mechanisms underlying the association of metals such as lead, arsenic, cadmium and mercury with obesity (Hou et al., 2013; Leasure et al., 2008; Tinkov et al., 2017, 2015). However, most of the previous studies, in both animal models and humans, have considered single metals as exposure variables. Little is known about potential health effects of simultaneous exposure to multiple metals and metal mixtures.
Heavy metals are widely dispersed in the environment, including soil, water, air, dust, human food chain, as well as manufacturing products (Alloway, 2013; Bosch et al., 2016; Mohod and Dhote, 2013). People could be exposed to a myriad of metals throughout their lifetime. Exposure to metal mixtures may result in effects that can depart from the mere summation of effects of single metals. A simple summation of concentrations of individual exposures may often reflect the components with the highest concentrations in the mixtures (Axelrad et al., 2009). Thus, it would be problematic in estimation of the association of cumulative exposure to pollutant mixtures with health outcomes if the lower concentration components are more potent than the higher concentration ones (Joseph M Braun et al., 2016). Additionally, complex exposure profile of pollutant mixtures may have additive, synergistic or antagonistic effects which are not detected by single pollutant approaches (S. K. Park et al., 2017a; Valeri et al., 2017). Significant synergistic toxicity of multiple metals has also been revealed by experimental studies (Preston et al., 2000; Wah Chu and Chow, 2002). Current regulatory limits of individual metals might not sufficiently protect people against toxicity of mixtures.
In 2011, the National Institute of Environmental Health Sciences (NIEHS) has set the field of combined exposures as one of the goals of the Strategic Plan (Birnbaum, 2012). Traditional toxicological studies and human health risk assessments have focused primarily on the impacts of individual metals. This could be partly due to statistical challenges such as correlations among metals, confounding by correlated co-pollutants, and lack of well-established statistical methods to identify mixtures (Joseph M. Braun et al., 2016). Recently, several methods, such as least absolute shrinkage and selection operator (LASSO) (Tibshirani, 1996), elastic-net (ENET) (Zou and Hastie, 2005), adaptive elastic-net (AENET) (Zou and Zhang, 2009), partial least squares (Wold et al., 1984), weighted quantile sum regression (Carrico et al., 2015), Bayesian model averaging (Madigan and Raftery, 1994), Bayesian kernel machine regression (Bobb et al., 2015), and Bayesian additive regression tree (Chipman et al., 2010), have been proposed to explore health effects of exposure to multiple pollutants. These approaches were compared in predicting disease endpoints associated with oxidative stress-related heavy metal mixtures using NHANES data in our recent work, among which AENET was proved satisfactory in terms of the statistical performance and epidemiologic interpretability (S. K. Park et al., 2017b).
Quantifying the impact of exposure to metal mixtures is crucial for identifying risk factors with environmental origins for obesity and its comorbidities and developing more targeted public health interventions. To the best of our knowledge, no previous study has investigated the associations of heavy metal mixtures with obesity and related disease endpoints.
The goal of the present study was to examine the associations of cumulative exposure to heavy metal mixtures with obesity and its comorbidities in the U.S. general population based on 6 cycles of the National Health and Nutrition Examination Survey (NHANES) data (from NHANES 2003–2004 to 2013–2014). To test this question, we first employed a novel statistical approach, AENET, which was proposed for analyzing high dimensional data while dealing with the collinearity problem (Zou and Zhang, 2009), to identify heavy metal mixtures including their dependencies related to WC. WC is a useful obesity measure which readily explains obesity-related health risks such as hypertension and T2DM (Janiszewski et al., 2007; Janssen et al., 2004). Then, we constructed an environmental risk score (ERS) accounting for main effects as well as pairwise interactions between metals selected by AENET, as an integrated measure of heavy metal mixtures effects on WC. The underlying idea behind the ERS is to build a predictive risk model as a weighted sum of the pollutant levels from simultaneous assessment of multiple pollutants. Weights are determined by the magnitudes (standardized regression coefficients) of the associations of each pollutant, as well as pollutant-pollutant interactions, with the outcome of interest from the AENET model. Finally, we evaluated the associations of ERS with other obesity measures and obesity-related comorbidities including hypertension and T2DM. The constructed ERS reflects predicted WC due to metal mixtures and therefore, can also be applied to other health outcomes relevant to obesity, such as hyperlipidemia, cardiovascular disease and cancer (Poirier et al., 2006).
2. METHODS
An overview of our methodology is depicted in Figure 1.
Figure 1.
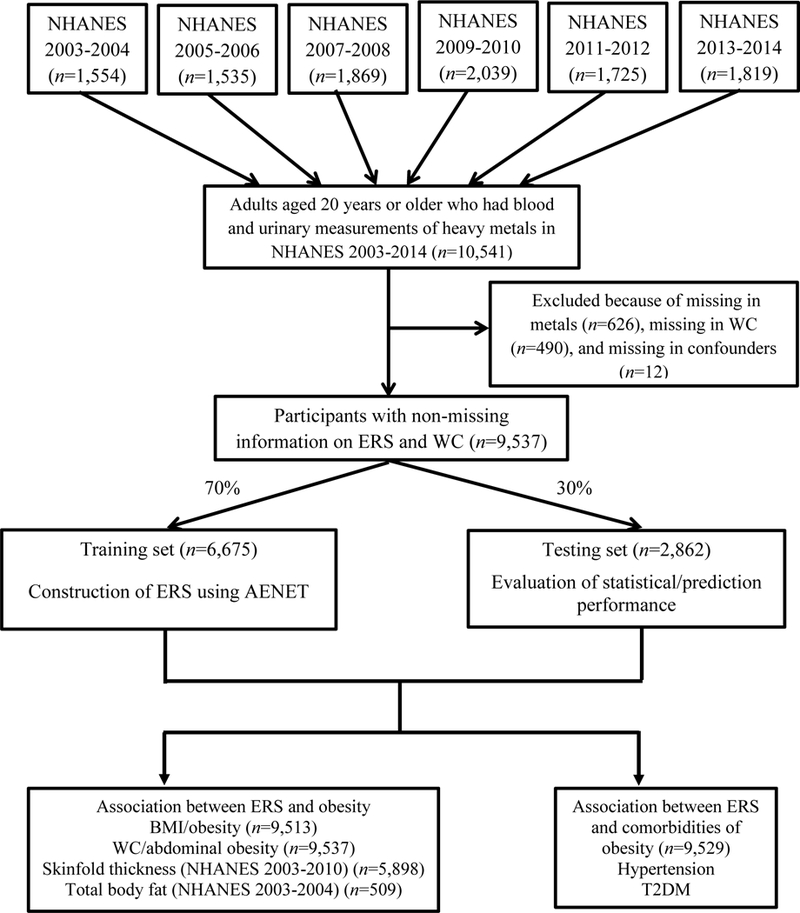
Schematic diagram of study methodology in NHANES 2003–2014. AENET, adaptive elastic-net; BMI, body mass index; ERS, environmental risk score; T2DM, type 2 diabetes mellitus; WC, waist circumference.
2.1. Study Population
The study sample consists of six continuous cycles (i.e. 2003–2004, 2005–2006, 20072008, 2009–2010, 2011–2012, and 2013–2014) of NHANES, which used a stratified multistage probability cluster design to obtain a representative sample of the civilian, noninstitutionalized U.S. population (CDC/NCHS, 2018), conducted by the Centers for Disease Control and Prevention (CDC), National Center for Health Statistics (NCHS). Two earlier cycles of NHANES (1999–2000 and 2001–2002) were not included because arsenic species were not measured in those cycles.
The participants were 10,541 adults aged 20 years or older who had urinary measurements of heavy metals. We excluded 1004 participants due to missing data in the outcome (n=470); exposures (n=626);; and core covariates (n=12) (the numbers are not mutually exclusive), leaving 9,537 eligible subjects for ERS construction in this study. To evaluate the performance of ERS in terms of its predictive ability, we randomly split the study population (n=9,537) by a ratio of 70%:30%, i.e. the training set (n=6,675) for ERS construction and the testing set (n=2,862) for validation. For other measures of adiposity in NHANES, BMI was available in all six cycles (2003–2004 to 2013–2014, n=9,513), skinfold thickness in 4 cycles (2003–2004 to 2009–2010, n=3,639) and total body fat in one cycle (2003–2004, n=509). For obesity-related comorbidities, we additionally excluded 8 participants with missing information on hypertension and T2DM.
2.2. Heavy Metal Measurements
A comprehensive set of 18 heavy metals were measured in both urine (antimony, total arsenic, arsenobetaine, monomethylarsonic acid (MMA), dimethylarsinic acid (DMA), barium, cadmium, cobalt, cesium, molybdenum, mercury, lead, thallium, tungsten, uranium: n=15) and whole blood (cadmium, lead, total mercury: n=3). All the heavy metals were analyzed with inductively coupled-plasma dynamic reaction cell-mass spectrometry (ICP-DRC-MS) by the Division of Laboratory Sciences, National Center for Environmental Health, Atlanta, Georgia (CDC/NCHS, 2018). Arsenous acid, arsenic acid, arsenocholine, beryllium and platinum in urine were not included in this study because more than 80% observations were below limits of detection (LOD). Metal concentrations below LOD were imputed using LOD divided by the square root of two by NHANES. We used these imputed values in our analyses in consistent with CDC’s National Report on Human Exposure to Environmental Chemicals (CDC, 2018).
2.3. Obesity measures
WC was measured during minimal respiration to the nearest 0.1 cm at the level of the iliac crest (CDC/NCHS, 2018). Height was measured to nearest 0.1 cm and body weight was measured to nearest 0.1 kg (CDC/NCHS, 2018). BMI was calculated as weight in kilograms divided by the square of height in meters. Triceps skinfold thickness was measured to the nearest 0.1 mm using skinfold calipers (CDC/NCHS, 2018). The measurement was taken twice on the right side of the participant’s body and averaged. Total body fat mass was measured in dual-energy X-ray absorptiometry scanning by using a Hologic QDR 4500A fan-beam densitometer (Hologic Inc, Bedford, MA) and estimated to the nearest 0.1 kg (CDC/NCHS, 2018). Abdominal obesity was defined as a WC of ≥ 102 cm for males and ≥ 88 cm for females according to the American Heart Association’s (AHA) definition (Grundy et al., 2005). We also used BMI ≥ 30 kg/m2 based on the WHO definition of obesity (WHO Expert Consultation, 2004).
2.4. Obesity Comorbidities
Blood pressures were measured three consecutive times (sometimes four times) with each participant in a seated position by using a standardized protocol that followed the AHA guidelines (CDC/NCHS, 2018). We calculated means of systolic blood pressure (SBP) and diastolic blood pressure (DBP) by averaging up to three measures after disregarding the first reading. Participants were considered to have hypertension if at least one of the following criteria were met: SBP/DBP ≥ 140/90 mmHg, self-reported physician diagnosis of hypertension, or self-reported use of hypertension medication. Prevalent T2DM was defined as self-reported physician diagnosis of diabetes, or hemoglobin A1c (HbA1c) level ≥ 6.5% (CDC/NCHS, 2018).
2.5. Covariates
Covariates were chosen a priori and included age at interview, sex, race/ethnicity (Mexican American, Other Hispanic, non-Hispanic white, non-Hispanic black, other), education (less than high school, high school, some college or higher), smoking status (never, former, current), physical activity (none, moderate, vigorous), NHANES cycles and urinary creatinine. Information on age, sex, race/ethnicity, education, smoking status and physical activity was collected using self-administered questionnaires (CDC/NCHS, 2018). We used a Directed Acyclic Graph (http://www.dagitty.net/dags.html) to show the hypothesis relations between use of metals, confounders, and WC (See Figure A.1). In our analysis, current smokers were participants who reported smoking at least 100 cigarettes during their lifetime and, at the time of the interview, reported smoking either every day or some days. Former smokers were those who had been smoked at least 100 cigarettes during their lifetime but did not smoke at the time of the interview. Never smokers were those who reported never having smoked 100 cigarettes during their lifetime. Moderate physical activity physical activity was defined as tasks that caused light sweating or a slight to moderate increase in breathing or heart rate. Vigorous physical activity was defined as tasks that caused heavy sweating or large increases in breathing or heart rate (CDC/NCHS, 2018). Urine creatinine concentration was identified as a maker of urine dilution and determined by a Jaffe rate reaction with a Beckman Synchron Analyzer (CDC/NCHS, 2018).
2.6. Statistical Analyses
Univariate statistics were calculated and examined in each NHANES survey cycle and all the cycles combined. Geometric means and standard deviations were calculated for urinary and blood measurements of metals. Spearman pairwise correlations among 18 metals were calculated and presented via a correlation-matrix heat map.
2.6.1. Construction of ERS
AENET was chosen as the primary method of selecting obesity-related heavy metals and constructing ERS. Several statistical methods have been proposed to explore the health effects of multiple pollutants in epidemiologic studies. Ordinary least squares (OLS)-based variable selection methods are commonly used but prone to over-fitting, leading to a poor performance in prediction accuracy and variance inflation with a large number of predictors, especially when predictors are highly correlated (multicollinearity) (Tibshirani, 1996). Ridge regression, as a shrinkage regression method, yields more precise estimates and better prediction accuracy than OLS, which in turn improves prediction accuracy by minimizing the ridge loss function (mean squared error plus a penalty term) (Hoerl and Kennard, 1970). However, ridge regression does not perform variable selection because it cannot shrink coefficient estimates exactly to zero (Tibshirani, 1996). LASSO, proposed by Tibshirani, shrinks coefficients towards exact zeroes, and thus promises to be a useful tool for variable selection (Tibshirani, 1996). However, with highly correlated variables as predictors, LASSO tends to select only one out of these correlated variables and ignore the others (Friedman et al., 2010). ENET is a hybrid penalized regression method that blends LASSO and ridge regression to overcome the limitation of LASSO on data with highly correlated variables (Zou and Hastie, 2005). Similar to LASSO, ENET executes variable selection, but it also has the ability to select a group of non-zero collinear variables (Zou and Hastie, 2005). Adaptive elastic net (AENET), as its name would suggest, is an adaptive version of ENET that not only deals with the collinearity problem over ENET but satisfies the asymptotic normality assumption that allows us to conduct statistical inference and hypothesis testing by providing large sample standard errors (SEs) and p-values (Zou and Zhang, 2009). Adaptive weights ensure smaller coefficients are shrunk faster to zero, whereas larger coefficients are penalized less. It should be noted that AENET performs variable selection by shrinking certain coefficients to zero but not based on p-values of coefficients (like forward selection, backward elimination, and stepwise selection). Additionally, penalized regression methods including LASSO, ENET, and AENET are different from environment-wide association study, which relies on linear regression models fitted independently for each individual pollutant and requires a correction for multiple comparisons (Agier et al., 2016). In the current analysis, 189 predictors served as candidates in the model selection, including 18 main effects, 18 squared terms, and 153 pairwise linear interactions of heavy metals. Four measures of adiposity were analyzed, including WC, BMI, skinfold thickness and total body fat, among which WC was chosen as the primary outcome for ERS construction due to data availability. WC may better predict risks of obesity-related health outcomes including hypertension, dyslipidemia, T2DM and cardiovascular disease than BMI (Janiszewski et al., 2007; Janssen et al., 2004; Menke et al., 2007; Zhu et al., 2002). Given the highly skewed distributions of WC and heavy metals, logarithmic transformations with base 10 were applied because the shapes of dose-response relationships were closer to be linear after log-transformation. We chose base 10 rather than natural log for easier interpretation of regression coefficients.
For subject i (i = 1, … , 6,675) in our study population, let Yi represent the outcome WC, (j = 1, … , 18) be the j-th metal, and Xi denote all predictors including Ei, Ei × Ei and the vector of confounders (i.e. age, sex, race/ethnicity, education, smoking status, physical activity, NHANES cycles and urinary creatinine). Then the beta coefficients from ANENT were defined by
where is the weight of j-th metal. These weights can allow coefficients of relatively less important metals to be shrunk to zeros more efficiently. By adding the penalty parameter λ1 with the sum of the absolute values of the coefficients into the ordinary least squares, we can select the metals associated with WC while shrinking some others to zero. By adding the other penalty parameter λ2 with the sum of square of regression coefficients, we can take into account correlations among metals while still performing variable selection. Both λ1 and λ2 were ascertained based on 5-fold cross-validation for minimal prediction errors. All the confounders were centered on their respective mean values and were always adjusted for (“forced”) in the selected models based on our prior knowledge (Figure A.1). The R package ‘gcdnet’ was used to implement AENET (Yang and Zou, 2015). The sample R code for construction of the ERS using AENET is available at https://github.com/XinWangUmich/Environmental-Risk-Score-AENET-. Complex survey design of NHANES was not considered in the variable selection due to difficulty in handling of survey weights automatically.
ERS was calculated using beta coefficients adopted from AENET for not only main effects but also squared terms of metal concentrations as well as all the combinations of pairwise linear interactions. The squared terms accounted for non-linear associations between metals and WC, and interaction terms implied potential departures from additive joint effects. All the terms were standardized as Z-scores for better goodness-of-fit and less collinearity issue. Then ERS was computed as a weighted sum of non-zero predictors selected from AENET by
where (j= 1,..p) is a standardized log-transformed concentration of the j-th metal. is the beta coefficient of the j-th metal. is the beta coefficient of interaction between metals k and l. And is the coefficient of squared term of the m-th metal. Note that coefficients of less important predictor terms were shrunk to zero by AENET. From this equation, the ERS for subject i can be interpreted as the predicted WC corresponding to the exposure levels of selected non-zero heavy metal predictors given all the confounders were fixed at their mean values in the population. The variables with non-zero coefficients were selected and their corresponding coefficients and P-values were estimated.
2.6.2. Assessment of ERS Performances
In order to evaluate whether the addition of ERS to other covariates could explain the extra variance of WC and improve the discriminatory capability for abdominal obesity, we randomly split the study population into a training set (n=6,675) for ERS construction and a testing set (n=2,862) for validation (Figure 1). Several metrics were calculated and compared in both datasets. First, we used linear regression for log-transformed WC and fit ERS as a continuous variable. Adjusted R2, mean squared error (MSE) and mean squared prediction error (MSPE) were calculated as indicators of goodness-of-fit in the models in both training and testing datasets. Next, we dichotomized WC at 88 cm for women and 102 cm for men as per AHA’s definition of abdominal obesity (Grundy et al., 2005), and identified the outcome as whether or not being abdominally obese. Logistic regression analyses were conducted using this binary abdominal obesity variable as the outcome and continuous ERS as the predictor. Area under the receiver operating characteristic (AUC) was computed to assess the accuracy of ERS in predicting abdominal obesity.
2.6.3. Associations of ERS with Obesity Measures and Obesity Comorbidities
Survey linear regression was used to examine the associations of ERS with other continuous obesity measures (BMI, skinfold thickness, total body mass) and blood pressures (SBP and DBP). Survey logistic regression analyses were conducted for hypertension and T2DM. All the models were adjusted for age at interview, sex, race/ethnicity, education, smoking status, physical activity, NHANES cycle and urinary creatinine. We report effect estimates (mean difference or odds ratio (OR)) comparing the 90th vs the 10th percentile of ERS and their 95% confidence intervals (CIs). All regression analyses were performed using the ‘survey’ package in R version 3.4.0 to account for the complex sampling design and weights in NHANES (CDC/NCHS, 2018).
3. RESULTS
3.1. Study Population Characteristics
Characteristics of the study population overall and by study cycle are summarized in Table 1. Participants had a mean age of 49.2 years and a standard deviation (SD) of 18.0 years. The demographics were similar across the six NHANES cycles, except oversampling of other Hispanics since 2007 (varying from 3.1% in the cycle 2005–2006 to 11.5% in the cycle 20072008), smoking and physical activity behaviors, as well as education. Data by survey cycle revealed decreasing trends in the prevalence of smokers (either current or former or both) and low education attainment (less than high school). Physical inactivity showed a sharp increase in the cycle of 2007–2008 followed by a notable decreasing trend in the later cycles. Overall, means (SDs) of WC, BMI, triceps skinfold thickness and total body fat were 98.7 (15.9) cm, 28.8 (6.6) kg/m2, 19.3 (8.4) mm, and 24.9 (11.9) kg, respectively. The prevalences of hypertension and T2DM were 40.8% and 16.0%.
Table 1.
Characteristics of study population in NHANES 2003–2014 (n=9,537).
| NHANES cycles | |||||||
|---|---|---|---|---|---|---|---|
| 2003–2004 | 2005–2006 | 2007–2008 | 2009–2010 | 2011–2012 | 2013–2014 | Overall | |
| n=1,424 | n=1,405 | n=1,631 | n=1,870 | n=1,547 | n=1,660 | n=9,537 | |
| Continuous, mean (SD) | |||||||
| Age, years | 51.1 (19.5) | 48.3 (18.8) | 50.1 (17.5) | 49.2 (17.7) | 48.1 (17.6) | 48.3 (17.1) | 49.2 (18.0) |
| Waist circumference, cm | 98.4 (14.9) | 98.3 (16.0) | 98.5 (15.1) | 99.2 (16.2) | 98.3 (16.8) | 99.1 (16.3) | 98.7 (15.9) |
| BMI, kg/m2 | 28.5 (6.1) | 28.7 (6.7) | 28.7 (6.1) | 29.1 (6.7) | 28.7 (6.9) | 29.0 (6.8) | 28.8 (6.6) |
| Skinfold thickness, mm | 19.8 (8.6) | 18.8 (8.3) | 19.3 (8.3) | 19.4 (8.4) | --a | -- | 19.3 (8.4) |
| Total body fat, kg | 24.9 (11.9) | --b | -- | -- | -- | -- | 24.9 (11.9) |
| SBP, mmHg | 126.7 (21.0) | 123.8 (19.7) | 124.3 (18.7) | 122.5 (18.7) | 122.9 (18.0) | 122.7 (17.4) | 123.7 (18.9) |
| DBP, mmHg | 70.3 (12.3) | 69.4 (12.6) | 70.6 (11.7) | 69.3 (12.1) | 71.1 (11.7) | 70.2 (11.3) | 70.1 (11.9) |
| Categorical, N (%) | |||||||
| Female | 725 (50.9) | 727 (51.7) | 823 (50.5) | 955 (51.1) | 764 (49.4) | 847 (51.0) | 4841 (50.8) |
| Race/Ethnicity | |||||||
| Mexican American | 295 (20.7) | 278 (19.8) | 284 (17.4) | 361 (19.3) | 138 (8.9) | 245 (14.8) | 1601 (16.8) |
| Other Hispanic | 32 (2.3) | 44 (3.1) | 188 (11.5) | 191 (10.2) | 168 (10.9) | 149 (9.0) | 772 (8.1) |
| Non-Hispanic White | 759 (53.3) | 729 (51.9) | 770 (47.2) | 888 (47.5) | 570 (36.9) | 724 (43.6) | 4440 (46.6) |
| Non-Hispanic Black | 288 (20.2) | 308 (21.9) | 325 (19.9) | 324 (17.3) | 395 (25.5) | 312 (18.8) | 1952 (20.5) |
| Other | 50 (3.5) | 46 (3.3) | 64 (3.9) | 106 (5.7) | 276 (17.8) | 230 (13.9) | 772 (8.1) |
| Education | |||||||
| <High school | 423 (29.7) | 394 (28.0) | 509 (31.2) | 527 (28.2) | 353 (22.8) | 343 (20.7) | 2549 (26.7) |
| High school | 380 (26.7) | 319 (22.7) | 390 (23.9) | 433 (23.2) | 306 (19.8) | 375 (22.6) | 2203 (23.1) |
| College or above | 621 (43.6) | 692 (49.3) | 732 (44.9) | 910 (48.7) | 888 (57.4) | 942 (56.8) | 4785 (50.2) |
| Smoking status | |||||||
| Never | 735 (51.6) | 732 (52.1) | 855 (52.4) | 995 (53.2) | 897 (58.0) | 948 (57.1) | 5162 (54.1) |
| Former | 392 (27.5) | 374 (26.6) | 412 (25.3) | 475 (25.4) | 355 (22.9) | 383 (23.1) | 2391 (25.1) |
| Current | 297 (20.9) | 299 (21.3) | 364 (22.3) | 400 (21.4) | 295 (19.1) | 329 (19.8) | 1984 (20.8) |
| Physical activity | |||||||
| None | 570 (40.0) | 549 (39.1) | 908 (55.7) | 1008 (53.9) | 760 (49.1) | 823 (49.6) | 4618 (48.4) |
| Moderate | 498 (35.0) | 428 (30.5) | 407 (24.9) | 507 (27.1) | 425 (27.5) | 471 (28.4) | 2736 (28.7) |
| Vigorous | 356 (25.0) | 428 (30.5) | 316 (19.4) | 355 (19.0) | 362 (23.4) | 366 (22.1) | 2183 (22.9) |
| Hypertension | 646 (45.5) | 508 (36.3) | 671 (41.2) | 756 (40.5) | 631 (40.8) | 670 (40.4) | 3882 (40.8) |
| T2DM | 209 (14.7) | 181 (12.9) | 269 (16.5) | 313 (16.8) | 279 (18.1) | 275 (16.6) | 1526 (16.0) |
Skinfold thickness was only measured in NHANES 2003–2004 to 2009–2010.
Total body fat was were only measured in NHANES 2003–2004.
3.2. Heavy Metal Concentrations and Correlations
The geometric means and geometric standard deviations of 18 heavy metal concentrations are presented in Table 2. Blood and urinary lead concentrations remarkably decreased across all 6 NHANES cycles, reflecting a reduction in environmental sources of lead in the US (Calafat, 2012). In contrast, urinary arsenobetaine concentrations increased in the latest two cycles (2011–2012 and 2013–2014), where excessive seafood consumption could serve as a major determinant among the US general population (Awata et al., 2017).
Table 2.
Geometric means and geometric standard deviations of heavy metal concentrations in NHANES 2003–2014 (n=9,537).
| NHANES cycle |
|||||||
|---|---|---|---|---|---|---|---|
| 2003–2004 n=1,424 |
2005–2006 n=1,405 |
2007–2008 n=1,631 |
2009–2010 n=1,870 |
2011–2012 n=1,547 |
2013–2014 n=1,660 |
Overall n=9,537 |
|
| In whole blood | |||||||
| Cadmium, μg/L | 0.39 (2.26) | 0.37 (2.17) | 0.39 (2.15) | 0.38 (2.14) | 0.36 (2.26) | 0.33 (2.30) | 0.37 (2.22) |
| Lead, μg/dL | 1.68 (1.92) | 1.48 (2.01) | 1.49 (1.91) | 1.31 (1.95) | 1.13 (2.01) | 1.01 (1.97) | 1.32 (2.00) |
| Total Mercury, μg/L | 0.87 (2.78) | 0.97 (2.55) | 0.90 (2.57) | 1.01 (2.59) | 0.93 (2.87) | 0.88 (2.71) | 0.93 (2.68) |
| In urine, μg/L | |||||||
| Antimony | 0.08 (1.82) | 0.07 (2.29) | 0.06 (2.16) | 0.06 (2.22) | 0.05 (1.99) | 0.04 (2.35) | 0.06 (2.21) |
| Total Arsenic | 8.95 (3.14) | 9.92 (3.23) | 8.71 (2.97) | 10.0 (3.21) | 8.75 (3.27) | 7.14 (3.05) | 8.85 (3.16) |
| Arsenobetaine | 1.79 (5.18) | 2.17 (5.82) | 1.44 (5.50) | 1.80 (6.08) | 2.71 (4.29) | 2.28 (3.84) | 1.98 (5.17) |
| MMAa | 0.82 (1.65) | 0.85 (1.57) | 0.84 (1.57) | 0.83 (1.60) | 0.79 (1.55) | 0.42 (2.42) | 0.73 (1.85) |
| DMAb | 3.83 (2.19) | 4.02 (2.19) | 3.85 (2.18) | 3.86 (2.37) | 4.10 (2.40) | 3.46 (2.18) | 3.84 (2.26) |
| Barium | 1.29 (2.66) | 1.32 (2.81) | 1.28 (2.69) | 1.30 (2.58) | 1.07 (2.68) | 0.96 (2.79) | 1.20 (2.72) |
| Cadmium | 0.29 (2.68) | 0.26 (2.73) | 0.27 (2.67) | 0.25 (2.62) | 0.23 (2.76) | 0.18 (2.95) | 0.24 (2.77) |
| Cobalt | 0.31 (2.23) | 0.37 (2.28) | 0.35 (2.16) | 0.34 (2.26) | 0.31 (2.28) | 0.37 (2.26) | 0.34 (2.25) |
| Cesium | 4.54 (2.10) | 4.64 (2.03) | 4.37 (1.97) | 4.10 (1.95) | 3.89 (2.00) | 3.92 (1.98) | 4.21 (2.01) |
| Lead | 0.70 (2.15) | 0.65 (2.43) | 0.57 (2.33) | 0.53 (2.32) | 0.41 (2.44) | 0.32 (2.51) | 0.50 (2.47) |
| Mercury | 0.44 (3.00) | 0.48 (3.01) | 0.44 (2.90) | 0.40 (2.81) | 0.36 (2.93) | 0.28 (2.77) | 0.39 (2.93) |
| Molybdenum | 38.29 (2.41) | 42.85 (2.34) | 42.08 (2.43) | 40.58 (2.37) | 36.84 (2.40) | 32.21 (2.48) | 38.59 (2.42) |
| Thallium | 0.14 (2.12) | 0.15 (2.07) | 0.14 (2.07) | 0.14 (2.05) | 0.15 (2.06) | 0.14 (2.13) | 0.14 (2.09) |
| Tungsten | 0.06 (2.59) | 0.08 (2.75) | 0.09 (2.78) | 0.07 (2.64) | 0.07 (2.70) | 0.05 (2.92) | 0.07 (2.77) |
| Uranium | 0.01 (2.22) | 0.01 (2.62) | 0.01 (2.74) | 0.01 (2.69) | 0.01 (2.42) | 0.01 (2.95) | 0.01 (2.64) |
MMA: monomethylarsonic acid
DMA: dimethylarsinic acid
Figure 2 is a heatmap showing the correlations among the 18 heavy metals using a Spearman correlation matrix. A complex exposure profile was observed among metal concentrations, with pairwise Spearman correlations ranging from slightly negative (ρ = −0.09) to strong positive correlations (ρ = 0.82). Of the 153 unique pairwise correlations, 143 were significant (Ps<0.05) after Bonferroni correction. We detected moderate to high correlations between blood lead and blood cadmium (Spearman correlation ρ = 0.35); between blood mercury and total arsenic (ρ = 0.42); between blood mercury and arsenobetaine (ρ = 0.47); and among arsenic species (ρs with a range of 0.42 to 0.82). The correlations among urinary measurements of heavy metals were generally moderate to high, with the strongest correlation between urinary cesium and thallium (ρ = 0.77).
Figure 2.

Spearman correlation matrix of metal biomarkers in NHANES 2003–2014 (n=9,537). Cadmium*, blood cadmium; Lead*, blood lead; Mercury*, blood mercury. All other metals were measured in urine. Mo, molybdenum.
3.3. Selection of Heavy Metals and ERS Construction
Table 3 summarizes the output of variable selection among main effects, squared terms and pairwise linear interactions of 18 heavy metals in relation to WC. A total of 18 variables were selected out of 189 predictor candidates: 7 main effects (blood lead (β = −0.008, 95% CI: − 0.010, −0.006), blood cadmium (β = −0.008, 95% CI: −0.010, −0.006), blood mercury (β = −0.003, 95% CI:−0.005, −0.001), urinary MMA (β = −0.010, 95% CI: −0.011, −0.008), urinary barium (β = 0.001, 95% CI: −0.001, 0.003), urinary mercury (β = −0.001, 95% CI: −0.003, 0.001), and urinary thallium (β = 0.003, 95% CI: 0.001, 0.005)); 4 squared terms (antimony (β = −0.002, 95% CI: − 0.004, 0), urinary tungsten (β = −0.001, 95% CI: −0.003, −0.001), urinary cadmium (β = −0.001, 95% CI: −0.003, 0.001) and blood cadmium (β = 0.001, 95%: −0.001, 0.003)); and 7 pairwise linear interactions (between blood lead and urinary MMA (β = 0.001, 95% CI: −0.001, 0.002), blood lead and urinary cadmium (β = −0.003, 95% CI: −0.005, −0.001), blood lead and urinary uranium (β = −0.0005, 95% CI: −0.003, 0.001), between urinary cadmium and urinary MMA (β = 0.0005, 95% CI: −0.001, 0.002), between urinary DMA and urinary tungsten (β = 0.004, 95% CI: 0.002, 0.006), between urinary MMA and urinary cobalt (β = −0.003, 95% CI: −0.005, −0.001), and between urinary lead and urinary antimony (β = −0.001, 95% CI: −0.003, 0.001)). It should be noted that beta coefficients of selected predictors were relatively small because we applied log-transformation with base 10 on both continuous WC and metal concentrations while metal concentrations were further standardized as Z-scores.
Table 3.
Selected predictors of main effects, square terms and pairwise interactions of heavy metals for waist circumference in adaptive elastic-net in NHANES 2003–2014 (n=6,675).
| Selected non-zero heavy metal predictors by AENET a |
β for 1-SD increase in log10-transformed metals (95% CI) |
P-value |
|---|---|---|
| Main effects | ||
| Blood Lead | −0.008 (−0.010, −0.006) | <.0001 |
| Blood Cadmium | −0.008 (−0.010, −0.006) | <.0001 |
| Blood Mercury | −0.003 (−0.005, −0.001) | <.0001 |
| MMAb | −0.010 (−0.011, −0.008) | <.0001 |
| Barium | 0.001 (−0.001, 0.003) | 0.09 |
| Mercury | −0.001 (−0.003, 0.001) | 0.23 |
| Thallium | 0.003 (0.001, 0.005) | 0.004 |
| Square Terms | ||
| Blood Cadmium | 0.002 (0.001, 0.003) | 0.002 |
| Cadmium | −0.002 (−0.003, −0.001) | 0.008 |
| Antimony | −0.002 (−0.003, −0.001) | <.0001 |
| Tungsten | −0.001 (−0.003, −0.001) | 0.02 |
| Pairwise Interactions | ||
| Blood Lead & MMA | 0.001 (−0.001, 0.002) | 0.52 |
| Blood Lead & Cadmium | −0.003 (−0.005, −0.001) | 0.005 |
| Blood Lead & Uranium | −0.0005 (−0.003, 0.001) | 0.59 |
| Cadmium & MMA | 0.0005 (−0.001, 0.002) | 0.50 |
| DMA & Tungsten | 0.004 (0.002, 0.006) | <.0001 |
| MMA & Cobalt | −0.003 (−0.005, −0.001) | <.0001 |
| Lead & Antimony | −0.001 (−0.003, 0.001) | 0.16 |
Heavy metal predictors were selected by adaptive elastic-net in training set (n=6,675)
MMA: monomethylarsonic acid
3.4. ERS Performances
The ERS ranged from −0.12 to 0.07 with a mean (SD) of 0 (0.02) in the training dataset (n=6,675), from −0.10 to 0.07 with a mean (SD) equal to 0 (0.02) in the testing set (n=2,682), and from −0.12 to 0.08 with a mean (SD) of 0 (0.02) in the entire dataset. Higher values in ERS indicate more susceptibility to abdominal obesity (higher WC) in relation to cumulative exposure to metal mixtures. Note that the higher ERS index does not necessarily mean higher metals exposures because the ERS was calculated as the predicted WC corresponding to the weighted selected metals concentrations. For example, AENET coefficient for urinary mercury (only the main effect was selected) equals to −0.002 (95% CI: −0.004, 0) (Table 3). Thus urinary mercury concentration negatively contributed to the ERS, which means the lower the urinary mercury concentration, the higher the ERS. A different example could be blood cadmium, that both its main effect (β = −0.007, 95% CI: −0.008, −0.005) and squared term (β = 0.001, 95%: −0.001, 0.003) were selected. Therefore, we expect that the ERS index first decreases and then increases according to blood cadmium concentration (Table 3, Figure A.2).
Table 4 provides information on the statistical performance of ERS in the prediction of log-transformed WC and abdominal obesity. ERS was significantly (p < 0.001) and strongly associated with all three outcomes after adjusting for age, sex, race/ethnicity, education, smoking status, physical activity, NHANES cycles, and urinary creatinine. When predicting log-transformed WC, adjusted R2 increased modestly in both training (0.12 to 0.18) and testing sets (0.11 to 0.16) when ERS was added to the models. Additionally, MSE and MSPE decreased slightly with the addition of ERS in the testing dataset (from 0.064 to 0.062). In the prediction of whether or not having abdominal obesity in the testing dataset, the addition of ERS to the model also offered a modest improvement in terms of AUC (from 0.71 to 0.74), suggesting that exposure to heavy metal as mixtures provides additional predictive information about abdominal obesity.
Table 4.
Evaluation of statistical performance of environmental risk scores (ERSs) in both training dataset (n=6,675) and testing dataset (n=2,862) in NHANES 2003–2014.
| Model without ERSa | Model with ERSa | |||
|---|---|---|---|---|
| Training set (n=6,675) |
Testing set (n=2,862) |
Training set (n=6,675) |
Testing set (n=2,862) |
|
| Log10-transformed waist circumference | ||||
| Percent change in outcome (95% CI)b | -- | -- | 2.1% (1.9%, 2.3%) |
1.9% (1.6%, 2.2%) |
| P-value | -- | -- | <0.0001 | <0.0001 |
| Adjusted-R2 | 0.12 | 0.11 | 0.18 | 0.16 |
| MSE/MSPE | 0.065 | 0.064 | 0.063 | 0.062 |
| Abdominal obesity: Women ≥ 88 cm, Men ≥ 102 cm in waist circumference | ||||
| Odds ratiob (95% CI) |
-- | -- | 1.71 (1.60, 1.84) |
1.57 (1.42, 1.73) |
| P-value | -- | -- | <0.0001 | <0.0001 |
| AUC | 0.71 | 0.71 | 0.75 | 0.74 |
All the models were adjusted for age, sex, race/ethinicity, education, smoking status, physical activity, NHANES cycles and urinary creatinine.
Effect estimates (percent change, β, odds ratio) are based on a standardized increment which is equivalent to one standard deviation increase in ERS.
3.5. Associations between ERS and Obesity Measures
The ERS index was also significantly associated with other obesity measures including BMI, skinfold thickness, total body fat, and binary obesity (BMI>30 vs. BMI≤30) (Ps<0.05, Table 5). Higher ERSs were significantly linked to an elevation in obesity measures and increased prevalence of obesity. More specifically, an increase in the ERS in the overall study population from −0.02 (10th percentile) to 0.02 (90th percentile), which represents the 10th to 90th percentiles of predicted WC due to the difference in exposure levels of heavy metal mixtures, was significantly associated with 4.50 kg/m2 (95% CI: 4.06, 4.94) higher BMI, 4.16 mm (95% CI: 3.56, 4.76) higher skinfold thickness, and 4.11 kg (95% CI: 0.83, 7.40) higher total body fat among US adults, with adjustment for age, sex, race/ethnicity, education, smoking status, physical activity, NHANES cycle and urinary creatinine. The adjusted OR of obesity comparing 90th vs 10th percentiles of ERS was 4.20 (95% CI: 3.51, 5.02).
Table 5.
Adjusted mean differencea in obesity measures by environmental risk score.
| Obesity measures | β, 90th vs 10th percentile (95% CI) |
P- value |
|---|---|---|
| BMIb, kg/m2 | 4.50 | <.0001 |
| (4.06, 4.94) | ||
| Skinfold thicknessc, mm | 4.16 | <.0001 |
| (3.56, 4.76) | ||
| Total body fatd, kg | 4.11 | 0.02 |
| (0.83, 7.40) | ||
| OR, 90th vs 10th percentile | P-value | |
| (95% CI) | ||
| Obesitye | 4.20 | <.0001 |
| (3.51, 5.02) | ||
All models were adjusted for age, sex, race/ethnicity, education, smoking status, physical activity, NHANES cycles and urinary creatinine. NHANES complex survey designs were also taken into account in the models.
Body mass index (BMI) was measured for all six cycles (2003–2004 to 2013–2014, n=9,513).
Skinfold thickness was measured for 4 cycles (2003–2004 to 2009–2010, n=3,639).
Total body fat was measured for (2003–2004, n=509).
Obesity was defined as BMI ≥ 30 kg/m2.
3.6. Associations between ERSs and Obesity Comorbidities
Associations of ERS with obesity comorbidities including blood pressures, hypertension and T2DM are shown in Table 6. Higher ERSs were significantly associated with elevated SBP as well as hypertension and T2DM (Ps<0.001). An increase from the 10th (−0.02) to the 90th percentile (0.02) of ERS in the overall study population was significantly associated with a 2.22 mmHg (95% CI: 0.90, 3.53) increase in SBP after controlling for age, sex, race/ethnicity, education, smoking, physical activity, NHANES cycle, and urinary creatinine. The adjusted ORs for hypertension and T2DM comparing the 90th with the 10th percentiles of the ERS were 1.55 (95% CI: 1.28, 1.88) and 3.63 (95% CI: 2.86, 4.66), respectively. No significant association was observed for DBP.
Table 6.
Adjusted effect estimatesa and 95% CIs of the associations between environmental risk score (ERS) and obesity comorbidities in NHANES 2003–2014 (n=9,529).
| Obesity Comorbidities | OR, 90th vs 10th percentile (95% CI) |
P-value |
|---|---|---|
| Hypertension | 1.55 | <0.0001 |
| (1.28, 1.88) | ||
| Type 2 diabetes mellitus | 3.63 | <0.0001 |
| (2.86, 4.66) | ||
| β, 90th vs 10th percentile | P-value | |
| (95% CI) | ||
| Systolic blood pressure, mmHg | 2.22 | 0.001 |
| (0.90, 3.53) | ||
| Diastolic blood pressure, mmHg | 0.61 | 0.13 |
| (−0.19, 1.40) | ||
All models were adjusted for age, sex, race/ethnicity, education, smoking status, physical activity, NHANES cycles and urinary creatinine. NHANES complex survey designs were also taken into account in the models.
4. DISCUSSION
To the best of our knowledge, this study is the first to assess associations of cumulative exposure to heavy metals as mixtures with obesity and obesity comorbidities. The present study created an updated ERS in relation to WC by using an advanced model selection approach (i.e. AENET) to handle statistical challenges including complex correlation structures among metals, non-linear relationships, and confounding by co-pollutants. The ERS was computed as a weighted sum of effects of selected non-zero predictors on WC as a measure of abdominal obesity. In addition to the main effects and pairwise interactions as we initially proposed in our previous study (S. K. Park et al., 2017b), we considered squared terms of metal concentrations to capture potential non-linear associations and further improve risk prediction performance. The constructed ERS was proved satisfactory in terms of reproducibility and validity in our study population. It can reflect individual susceptibility to abdominal obesity (higher WC) in relation to cumulative exposure to metal mixtures while providing modest discriminatory ability in addition to conventional obesogenic factors. Our study results indicate that cumulative exposure to metal mixtures may play a role in both obesity and its related chronic conditions such as hypertension and T2DM.
Although individual metal effects were not our primary interest, the AENET selected main effects of blood lead, blood cadmium, blood mercury and urinary metals including barium, thallium, and mercury on WC in our multipollutant approach accords with previous population-based studies. Urinary barium and thallium were noted to be positively while cadmium and lead were inversely associated with BMI and/or WC among the US general population in NHANES 1999–2002 (Padilla et al., 2010). The inverse associations between blood lead and both BMI and obesity were also observed in children, adolescents and adults in NHANES 1999–2006 (Scinicariello et al., 2013). In a national study based in South Korean adults, inverse associations of both blood lead and blood mercury with body fat percentage were observed (Park and Lee 2013). Another cross-sectional study conducted in Poland designed to examine the potential risk factors of metabolic syndrome also reported a significant and negative correlation between blood mercury and waist-to-hip ratio among a population of aging men (Rotter et al. 2015).
However, those previous epidemiological studies have focused primarily on individual metals while the complexity of mixed exposures has not been adequately captured. In the present study, the squared terms of urinary antimony, urinary tungsten, and blood/urinary cadmium as well as pairwise interactions between blood lead and urinary metals including cadmium, MMA and uranium, between urinary cadmium and urinary MMA, between urinary DMA and urinary tungsten, between urinary MMA and urinary cobalt, and between urinary lead and urinary antimony, were also identified, suggesting the possibility of interactions and nonlinear associations among multiple concurrent exposures. For example, we observed that the association between blood cadmium and WC may be non-linear because both main effect and squared term of blood cadmium were selected). This non-linear relationship between blood cadmium and WC suggests an inverse association at low-moderate cadmium levels which has been previously observed in the U.S. general population (Padilla et al., 2010), while a flattened effect at higher exposure levels (Figure A.2). This could, possibly, explain the non-significant relationships reported in other populations experienced greater cadmium body burden compared to U.S. (Lee and Kim, 2016, 2013). Such findings may also be explained as a result of complex interactions between mixture components. For example, our finding of significant interactions between blood lead and urinary cadmium, MMA and uranium on WC suggests that the effect of blood lead may change more than additively in populations with different levels of cadmium, MMA and uranium. Note that our primary goal when constructing ERS was to integrate early health effects of exposure to multiple environmental pollutants simultaneously on a common disease pathway (i.e., obesity) while accounting for high degree correlations and potential interactions among metals, to better capture the association between exposure to multiple metals as mixtures and health outcomes. Allowing for potential interactions and nonlinear associations may reconcile the context-dependent nature of mixture components effects when integrating effects of exposure to heavy metals as mixtures. Biologically based dose-response functions, such as physiologically based pharmacokinetic (PBPK) modeling, may better capture a dose-response curve for specific metals or metal-metal interactions but require more complex modeling that is hard to test and toxicological data that are often not available. Alternatively, categorizing continuously measured variables is often used in epidemiologic research to capture nonlinear dose-response associations. However, such an approach could not be the best strategy in the construction of ERS. For example, it is not statistically efficient to incorporate main effects and interaction terms in AENET if all the metals are treated as categorical variables.
In our study, when ERS was added to the conventional risk factors of obesity including age, sex, race/ethnicity, socioeconomic status (education level), physical inactivity and smoking status, modest improvements in the prediction of WC and abdominal obesity were observed as indicated by improvements in adjusted-R2 and AUC in linear regression and logistic regression models, respectively. This finding, which paralleled significant associations between ERS and all the other obesity measures including BMI, skinfold thickness and total body fat in current study, also indicated that a combined real-life exposure to heavy metals as mixtures is not only associated with one specific obesity measure – WC, but also associated with obesity, independent of other known factors.
While the underlying mechanisms are still not well understood, there is biological plausibility for heavy metals in obesity pathogenesis. Main effects of lead, mercury and MMA negatively contributed to the ERS in the current study. Several animal studies found that lead exposure may contribute to body weight loss (Donald et al., 1988; Hamir et al., 1981). The linkage between body weight and lead exposure may be through perturbation of the hypothalamic dopaminergic system. Rat pups exposed to the lowest gestational lead levels had a statistically significant increase in weight, and decreased levels of striatal dopamine and its main metabolite, 3,4-dihydroxyphenylacetic acid, compared to those exposed to the highest lead levels (Leasure et al., 2008). Mercury, as an important endocrine disruptor, has also been indicated the potential role in the pathogenesis of obesity (Tinkov et al., 2015). Decreases in adipose tissue content, adipocyte size and leptin secretion were reported in mice following mercuric chloride administration (Kawakami et al., 2012). Arsenic was also found to affect adiposity. An animal study using a mice model demonstrated that low-level inorganic arsenic and MMA could trigger endoplasmic reticulum stress and induce C/EBP homologous protein, a protein that inhibits CCAAT/enhancer-binding protein P transcriptional activity, as a result of unfolded protein response, which in turn impairs adipogenesis (Hou et al., 2013). The impact of heavy metals on obesity may also through their toxicities on the growth trajectory. Exposure to toxic metals in early pregnancy could affect the fertilized egg or zygote implantation, impair the placental growth and placental function, alter the uterine environment, dysregulate the fetal epigenome, accumulate in placental transfer cells causing a decrease in uterine blood flow, and decrease the transfer of nutrients to the fetus, and thus affect fetal growth and development (Zheng et al., 2016).
Barium and thallium were positively associated with our constructed ERS, suggesting higher WC with high exposure to these metals. One possible explanation is that these metals could promote oxidative stress, limit ATP production and increase lipid production (Mailloux et al., 2007). The reactive oxygen species generated by metals can further disrupt mitochondrial functions, alter activity of enzymes such as aconitase, and impair the tricarboxylic acid cycle via oxidative phosphorylation (Ercal et al., 2001). For other metals and metal-metal interactions, the underlying mechanisms of observed associations were less well-established.
The primary goal of epidemiologic research is to identify causal relationships. Although traditional research is often based on a priori knowledge and driven by a well-defined hypothesis, a data-driven method might be useful especially when limited information is available. The ERS in our study provides a good approximation of underlying causal relationships by capturing complex relationships and patterns among pollutants within mixtures (Shmueli, 2010). The ERS, as the prediction models, can also be used to assess the risk stratification and discrimination power for predicting specific health endpoints in future studies.
Nevertheless, our study has numerous limitations. First, the cross-sectional nature of NHANES data precludes the ability to determine chronicity of heavy metal exposures and persistence of obesity and its comorbidities. Therefore reverse causation could be an explanation for our results since chronic conditions such as diabetes may affect metal excretions in urine (Chaumont et al., 2012). Second, the ERS construction was based on WC due to data availability. More sensitive measures of obesity such as dual-energy X-ray absorptiometry scanning that directly measure body fat contents may improve risk prediction and thus better discriminate metal mixture health risk. Third, the metals used in the current study have various half-lives in human body and therefore, have different degrees of measurement error which may affect the variable selection in AENET algorithms (Sorensen et al., 2015). Most urinary metals used in this study have short half-lives and thus their urinary concentrations mainly reflect recent exposures. Such metals might not be detected to be associated with WC in AENET and could be missed in relation to other health endpoints if long-term cumulative exposure to low levels causes those health outcomes. Fourth, our models were limited to pairwise interactions including quadratic terms of the main effects. However, higher order interactions such as three-way interactions or higher than quadratic polynomial forms may exist. Due to such possible model misspecification, more complicated but important interactions may not be selected in AENET and not contribute to the corresponding ERS. It should be noted that the selected pairwise interactions in AENET reflect statistical interactions rather than biological interactions. Finally, we constructed the ERS for waist circumference as a common contributing factor to obesity and obesity comorbidities. Note that ERS was disease-specific, so it might not be able to predict chronic conditions other than obesity and its comorbidities. Future studies should explore the use of ERS in a longitudinal setting and adaptively weighted to provide a more comprehensive risk assessment for a complex chronic disease.
5. CONCLUSIONS
In conclusion, we constructed an ERS by a novel statistical approach (i.e. AENET) for assessing the associations of cumulative exposure to mixtures of 18 heavy metals with obesity and its comorbidities among US adults in NHANES 2003–2014. The ERS as a measure of integrated effects of heavy metals mixtures on WC were significantly associated with other obesity measures including BMI, skinfold thickness and total body fat as well as obesity-related chronic conditions, hypertension and T2DM, independent of other important risk factors. Our findings require confirmations in other longitudinal cohort studies but have important public health implications, given the high burden of obesity and widespread exposure to heavy metals. Future studies are also needed to examine the underlying biological mechanisms by which different metals as well as metal-metal interactions are associated with obesity.
Highlights.
Exposure to multiple metals resulted in effects that departed from mere summation of effects of single metals.
We constructed an ERS to evaluate the associations of cumulative exposure to mixtures of 18 metals with obesity and its comorbidities.
ERS was significantly associated with waist circumference, BMI, skinfold thickness and total body fat.
ERS was significantly associated with obesity comorbidities including hypertension and type-2 diabetes.
Our study suggests the potential role of exposure to heavy metals mixtures as a contributing factor to obesity and its related chronic conditions.
ACKNOWLEDGMENTS
This study was supported by grants from the National Institute of Environmental Health Sciences (NIEHS) R01-ES026578, R01-ES026964, R21-ES020811 and P30-ES017885, and by the Center for Disease Control and Prevention (CDC)/National Institute for Occupational Safety and Health (NIOSH) grant T42-OH008455 and National Science Foundation grant NSF DMS 1406712.
APPENDICES
Figure A.1.

A directed acyclic graph (DAG) for evaluation of covariate selection in the analysis of metal effects on waist circumference. Exposure is metal concentrations. Outcome is waist circumference. Factors taken into account include age at interview, sex, race/ethnicity, education, smoking status, and physical activity. The minimal and sufficient adjustment set contains all the variables included in the DAG. This figure is constructed through Directed Acyclic Graph (http://www.dagitty.net/dags.html ).
Figure A.2.

Predicted log-10 transformed waist circumference and 95% confidence band for standardized log-10 transformed blood cadmium concentration with the other metals fixed at the mean concentrations, after controlling for age, sex, race/ethnicity, education, smoking status, physical activity, NHANES cycles and urinary creatinine. Standardized log-10 transformed blood cadmium concentration in current analysis range from −2.07 to 3.98.
Footnotes
CONFLICTS OF INTEREST
The authors have declared no actual or potential conflict of interest that could inappropriately influence, or be perceived to influence our work.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- Agier L, Portengen L, Chadeau-Hyam M, Basagaña X, Giorgis-Allemand L, Siroux V, Robinson O, Vlaanderen J, González JR, Nieuwenhuijsen MJ, Vineis P, Vrijheid M, Slama R, Vermeulen R, 2016. A Systematic Comparison of Linear Regression-Based Statistical Methods to Assess Exposome-Health Associations. Environ. Health Perspect. 124, 1848–1856. https://doi.org/10.1289/EHP172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alloway BJ, 2013. Sources of Heavy Metals and Metalloids in Soils. Springer, Dordrecht, pp. 11–50. https://doi.org/10.1007/978-94-007-4470-7_2 [Google Scholar]
- Awata H, Linder S, Mitchell LE, Delclos GL, 2017. Association of Dietary Intake and Biomarker Levels of Arsenic, Cadmium, Lead, and Mercury among Asian Populations in the United States: NHANES 2011–2012. Environ. Health Perspect. 125, 314–323. https://doi.org/10.1289/EHP28 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Axelrad DA, Goodman S, Woodruff TJ, 2009. PCB body burdens in US women of childbearing age 2001–2002: An evaluation of alternate summary metrics of NHANES data. Environ. Res. 109, 368–378. https://doi.org/10.1016/j.envres.2009.01.003 [DOI] [PubMed] [Google Scholar]
- Beier EE, Maher JR, Sheu T-J, Cory-Slechta DA, Berger AJ, Zuscik MJ, Puzas JE, 2013. Heavy metal lead exposure, osteoporotic-like phenotype in an animal model, and depression of Wnt signaling. Environ. Health Perspect. 121, 97–104. https://doi.org/10.1289/ehp.1205374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birnbaum LS, 2012. NIEHS’s new strategic plan. Environ. Health Perspect. 120, a298 https://doi.org/10.1289/ehp.1205642 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bobb JF, Valeri L, Claus Henn B, Christiani DC, Wright RO, Mazumdar M, Godleski JJ, Coull BA, 2015. Bayesian kernel machine regression for estimating the health effects of multi-pollutant mixtures. Biostatistics 16, 493–508. https://doi.org/10.1093/biostatistics/kxu058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosch AC, O’Neill B, Sigge GO, Kerwath SE, Hoffman LC, 2016. Heavy metals in marine fish meat and consumer health: a review. J. Sci. Food Agric. 96, 32–48. https://doi.org/10.1002/jsfa.7360 [DOI] [PubMed] [Google Scholar]
- Braun JM, Gennings C, Hauser R, Webster TF, 2016. What Can Epidemiological Studies Tell Us about the Impact of Chemical Mixtures on Human Health? Environ. Health Perspect. 124, A6–9. https://doi.org/10.1289/ehp.1510569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun JM, Gennings C, Hauser R, Webster TF, 2016. What can epidemiological studies tell us about the impact of chemical mixtures on human health? Environ. Health Perspect. 124, A6–A9. https://doi.org/10.1289/ehp.1510569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calafat AM, 2012. The U.S. National Health and Nutrition Examination Survey and human exposure to environmental chemicals. Int. J. Hyg. Environ. Health 215, 99–101. https://doi.org/10.1016/j.ijheh.2011.08.014 [DOI] [PubMed] [Google Scholar]
- Carrico C, Gennings C, Wheeler DC, Factor-Litvak P, 2015. Characterization of Weighted Quantile Sum Regression for Highly Correlated Data in a Risk Analysis Setting. J. Agric. Biol. Environ. Stat. 20, 100–120. https://doi.org/10.1007/s13253-014-0180-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- CDC/NCHS, 2018. NHANES - Questionnaires, Datasets, and Related Documentation [WWW Document]. URL https://wwwn.cdc.gov/nchs/nhanes/Default.aspx (accessed 6.14.18).
- CDC, 2018. National Report on Human Exposure to Environmental Chemicals. Updated Tables, March 2018 [WWW Document]. URL https://www.cdc.gov/exposurereport/index.html (accessed 8.1.18).
- Chaumont A, Nickmilder M, Dumont X, Lundh T, Skerfving S, Bernard A, 2012. Associations between proteins and heavy metals in urine at low environmental exposures: evidence of reverse causality. Toxicol. Lett. 210, 345–52. https://doi.org/10.10167j.toxlet.2012.02.005 [DOI] [PubMed] [Google Scholar]
- Chipman HA, George EI, McCulloch RE, 2010. BART: Bayesian additive regression trees. Ann. Appl. Stat. 4, 266–298. https://doi.org/10.1214/09-AOAS285 [Google Scholar]
- Donald JM, Bradley M, O’Grady JE, Cutler MG, Moore MR, 1988. Effects of low-level lead exposure on 24 h activity patterns in the mouse. Toxicol. Lett. 42, 137–47. [DOI] [PubMed] [Google Scholar]
- Ercal N, Gurer-Orhan H, Aykin-Burns N, 2001. Toxic metals and oxidative stress part I: mechanisms involved in metal-induced oxidative damage. Curr. Top. Med. Chem. 1, 529–39. [DOI] [PubMed] [Google Scholar]
- Friedman J, Hastie T, Tibshirani R, 2010. Regularization paths for generalized linear models via coordinate descent. J. Stat. Softw. [PMC free article] [PubMed] [Google Scholar]
- Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, Gordon DJ, Krauss RM, Savage PJ, Smith SC, Spertus JA, Costa F, American Heart Association, National Heart, Lung, and Blood Institute, 2005. Diagnosis and Management of the Metabolic Syndrome: An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation 112, 2735–2752. https://doi.org/10.1161/CIRCULATIONAHA.105.169404 [DOI] [PubMed] [Google Scholar]
- Hamir AN, Sullivan ND, Handson PD, Wilkinson JS, Lavelle RB, 1981. Clinical signs, radiology and tissue lead distribution of dogs administered a mixture of lead chloride, lead bromide and lead sulphate. Aust. Vet. J. 57, 401–6. [DOI] [PubMed] [Google Scholar]
- Hoerl AE, Kennard RW, 1970. Ridge Regression: Biased Estimation for Nonorthogonal Problems. Technometrics 12, 55–67. https://doi.org/10.1080/00401706.1970.10488634 [Google Scholar]
- Hou Y, Xue P, Woods CG, Wang X, Fu J, Yarborough K, Qu W, Zhang Q, Andersen ME, Pi J, 2013. Association between arsenic suppression of adipogenesis and induction of CH0P10 via the endoplasmic reticulum stress response. Environ. Health Perspect. 121, 237–43. https://doi.org/10.1289/ehp.1205731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janiszewski PM, Janssen I, Ross R, 2007. Does Waist Circumference Predict Diabetes and Cardiovascular Disease Beyond Commonly Evaluated Cardiometabolic Risk Factors? Diabetes Care 30. [DOI] [PubMed] [Google Scholar]
- Janssen I, Katzmarzyk PT, Ross R, 2004. Waist circumference and not body mass index explains obesity-related health risk. Am. J. Clin. Nutr. 79, 379–84. [DOI] [PubMed] [Google Scholar]
- Kawakami T, Hanao N, Nishiyama K, Kadota Y, Inoue M, Sato M, Suzuki S, 2012. Differential effects of cobalt and mercury on lipid metabolism in the white adipose tissue of high-fat diet-induced obesity mice. Toxicol. Appl. Pharmacol. 258, 32–42. https://doi.org/10.1016/j.taap.2011.10.004 [DOI] [PubMed] [Google Scholar]
- Kawakami T, Sugimoto H, Furuichi R, Kadota Y, Inoue M, Setsu K, Suzuki S, Sato M, 2010. Cadmium reduces adipocyte size and expression levels of adiponectin and Peg1/Mest in adipose tissue. Toxicology 267, 20–26. https://doi.org/10.1016/j.tox.2009.07.022 [DOI] [PubMed] [Google Scholar]
- Leasure JL, Giddabasappa A, Chaney S, Johnson JE, Pothakos K, Lau YS, Fox DA, 2008. Low-level human equivalent gestational lead exposure produces sex-specific motor and coordination abnormalities and late-onset obesity in year-old mice. Environ. Health Perspect. 116, 355–61. https://doi.org/10.1289/ehp.10862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee B-K, Kim Y, 2016. Association of Blood Cadmium Level with Metabolic Syndrome After Adjustment for Confounding by Serum Ferritin and Other Factors: 2008–2012 Korean National Health and Nutrition Examination Survey. Biol. Trace Elem. Res. 171, 6–16. https://doi.org/10.1007/s12011-015-0499-9 [DOI] [PubMed] [Google Scholar]
- Lee B-K, Kim Y, 2013. Blood cadmium, mercury, and lead and metabolic syndrome in South Korea: 2005–2010 Korean National Health and Nutrition Examination Survey. Am. J. Ind. Med. 56, 682–692. https://doi.org/10.1002/ajim.22107 [DOI] [PubMed] [Google Scholar]
- Madigan D, Raftery AE, 1994. Model Selection and Accounting for Model Uncertainty in Graphical Models Using Occam’s Window. J. Am. Stat. Assoc. 89, 1535 https://doi.org/10.2307/2291017 [Google Scholar]
- Mailloux R, Lemire J, Appanna V, 2007. Aluminum-Induced Mitochondrial Dysfunction Leads to Lipid Accumulation in Human Hepatocytes: A Link to Obesity. Cell. Physiol. Biochem. 20, 627–638. https://doi.org/10.1159/000107546 [DOI] [PubMed] [Google Scholar]
- Menke A, Muntner P, Wildman RP, Reynolds K, He J, 2007. Measures of Adiposity and Cardiovascular Disease Risk Factors*. Obesity 15, 785–795. https://doi.org/10.1038/oby.2007.593 [DOI] [PubMed] [Google Scholar]
- Mohod CV, Dhote J, 2013. REVIEW OF HEAVY METALS IN DRINKING WATER AND THEIR EFFECT ON HUMAN HEALTH. Int. J. Innov. Res. Sci. , Eng. Technol. 2. [Google Scholar]
- Must A, 1999. The Disease Burden Associated With Overweight and Obesity. JAMA 282, 1523 https://doi.org/10.1001/jama.282.16.1523 [DOI] [PubMed] [Google Scholar]
- Ogden CL, Carroll MD, Fryar CD, Flegal KM, 2015. Key findings What was the prevalence of obesity among adults in 2011–2014? [PubMed] [Google Scholar]
- Padilla MA, Elobeid M, Ruden DM, Allison DB, 2010. An Examination of the Association of Selected Toxic Metals with Total and Central Obesity Indices: NHANES 99–02. Int. J. Environ. Res. Public Health 7, 3332–3347. https://doi.org/10.3390/ijerph7093332 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park JS, Ha KH, He K, Kim DJ, Nikonorov A, Skalny A, Suzuki S, Han J, Kim S, Park C, Oh S, Lee C, Kim K, Oh S, Kim Y, Choi W, Yoo H, Kim H, Oh S, Kim J, Lee S, Ha M, Kwon H, Park J, 2017. Association between Blood Mercury Level and Visceral Adiposity in Adults. Diabetes Metab. J. 41, 113 https://doi.org/10.4093/dmj.2017.41.2.113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park S, Lee B-K, 2013. Body Fat Percentage and Hemoglobin Levels Are Related to Blood Lead, Cadmium, and Mercury Concentrations in a Korean Adult Population (KNHANES 2008–2010). Biol. Trace Elem. Res. 151, 315–323. https://doi.org/10.1007/s12011-012-9566-7 [DOI] [PubMed] [Google Scholar]
- Park SK, Zhao Z, Mukherjee B, 2017a. Construction of environmental risk score beyond standard linear models using machine learning methods: application to metal mixtures, oxidative stress and cardiovascular disease in NHANES. Environ. Heal. 16, 102 https://doi.org/10.1186/s12940-017-0310-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park SK, Zhao Z, Mukherjee B, 2017b. Construction of environmental risk score beyond standard linear models using machine learning methods: application to metal mixtures, oxidative stress and cardiovascular disease in NHANES. Environ. Heal. 16, 102 https://doi.org/10.1186/s12940-017-0310-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poirier P, Giles T, Bray G, Hong Y, 2006. Obesity and cardiovascular disease. , Vasc. Biol. [DOI] [PubMed] [Google Scholar]
- Preston S, Coad N, Townend J, Killham K, Paton GI, 2000. Biosensing the acute toxicity of metal interactions: Are they additive, synergistic, or antagonistic? Environ. Toxicol. Chem. 19, 775–780. https://doi.org/10.1002/etc.5620190332 [Google Scholar]
- Scinicariello F, Buser MC, Mevissen M, Portier CJ, 2013. Blood lead level association with lower body weight in NHANES 1999–2006. Toxicol. Appl. Pharmacol. 273, 516–523. https://doi.org/10.1016/j.taap.2013.09.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shmueli G, 2010. To Explain or to Predict? Stat. Sci. 25, 289–310. https://doi.org/10.1214/10-STS330 [Google Scholar]
- Sørensen Ø, Frigessi A, Thoresen M, 2015. MEASUREMENT ERROR IN LASSO: IMPACT AND LIKELIHOOD BIAS CORRECTION. Stat. Sin. https://doi.org/10.2307/24311046 [Google Scholar]
- Thayer KA, Heindel JJ, Bucher JR, Gallo MA, 2012. Role of environmental chemicals in diabetes and obesity: A national toxicology program workshop review. Environ. Health Perspect. https://doi.org/10.1289/ehp.1104597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tibshirani R, 1996. Regression Shrinkage and Selection via the Lasso. J. R. Stat. Soc. Ser. B. https://doi.org/10.2307/2346178 [Google Scholar]
- Tinkov AA, Ajsuvakova OP, Skalnaya MG, Popova EV, Sinitskii AI, Nemereshina ON, Gatiatulina ER, Nikonorov AA, Skalny AV, 2015. Mercury and metabolic syndrome: a review of experimental and clinical observations. BioMetals 28, 231–254. https://doi.org/10.1007/s10534-015-9823-2 [DOI] [PubMed] [Google Scholar]
- Tinkov AA, Filippini T, Ajsuvakova OP, Aaseth J, Gluhcheva YG, Ivanova JM, Bjørklund G, Skalnaya MG, Gatiatulina ER, Popova EV, Nemereshina ON, Vinceti M, Skalny AV, 2017. The role of cadmium in obesity and diabetes. Sci. Total Environ. 601–602, 741–755. https://doi.org/10.1016/j.scitotenv.2017.05.224 [DOI] [PubMed] [Google Scholar]
- Valeri L, Mazumdar MM, Bobb JF, Claus Henn B, Rodrigues E, Sharif OIA, Kile ML, Quamruzzaman Q, Afroz S, Golam M, Amarasiriwardena C, Bellinger DC, Christiani DC, Coull BA, Wright RO, 2017. The Joint Effect of Prenatal Exposure to Metal Mixtures on Neurodevelopmental Outcomes at 20–40 Months of Age: Evidence from Rural Bangladesh. Environ. Health Perspect. 125 https://doi.org/10.1289/EHP614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wah Chu K, Chow KL, 2002. Synergistic toxicity of multiple heavy metals is revealed by a biological assay using a nematode and its transgenic derivative. Aquat. Toxicol. 61, 53–64. https://doi.org/10.1016/S0166-445X(02)00017-6 [DOI] [PubMed] [Google Scholar]
- WHO, 2017. Obesity and overweight. [Google Scholar]
- WHO Expert Consultation, 2004. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 363, 157–163. https://doi.org/10.1016/S0140-6736(03)15268-3 [DOI] [PubMed] [Google Scholar]
- Wold S, Ruhe A, Wold H, Dunn WJ III, 1984. The Collinearity Problem in Linear Regression. The Partial Least Squares (PLS) Approach to Generalized Inverses. SIAM J. Sci. Stat. Comput. 5, 735–743. https://doi.org/10.1137/0905052 [Google Scholar]
- Yang Y, Zou H, 2015. Package “gcdnet.” [Google Scholar]
- You C-H, Kim B-G, Kim J-M, Yu S-D, Kim Y-M, Kim R-B, Hong Y-S, 2011. Relationship Between Blood Mercury Concentration and Waist-to-Hip Ratio in Elderly Korean Individuals Living in Coastal Areas. J. Prev. Med. Public Heal. 44, 218–225. https://doi.org/10.3961/jpmph.2011.44.5.218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng T, Zhang J, Sommer K, Bassig BA, Zhang X, Braun J, Xu S, Boyle P, Zhang B, Shi K, Buka S, Liu S, Li Y, Qian Z, Dai M, Romano M, Zou A, Kelsey K, 2016. Effects of Environmental Exposures on Fetal and Childhood Growth Trajectories. Ann. Glob. Heal. 82, 41–99. https://doi.org/10.1016/J.AOGH.2016.01.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu S, Wang Z, Heshka S, Heo M, Faith MS, Heymsfield SB, 2002. Waist circumference and obesity-associated risk factors among whites in the third National Health and Nutrition Examination Survey: clinical action thresholds. Am. J. Clin. Nutr. 76, 743–9. [DOI] [PubMed] [Google Scholar]
- Zou H, Hastie T, 2005. Regularization and variable selection via the elastic net. J. R. Stat. Soc. Ser. [Google Scholar]
- Zou H, Zhang H, 2009. On the adaptive elastic-net with a diverging number of parameters. Ann. Stat. [DOI] [PMC free article] [PubMed] [Google Scholar]


