Abstract
Two new alkaloids, aconicarmine (1) and aconicaramide (5), were isolated from the EtOH extract of the lateral roots of Aconitum carmichaelii, together with five known compounds: fuziline (2), neoline (3), N-ethylhokbusine B (4), 5-hydroxymethylpyrrole-2-carbaldehyde (6), and oleracein E (7). Their structures were elucidated by physical and NMR analysis. Pyrrole alkaloids were isolated from A. carmichaelii for the first time. In the in vitro assays, compounds 2 and 3 showed activity against pentobarbital sodium-induced cardiomyocytes damage by obviously recovering beating rhythm and increasing the cell viability, while compounds 5 and 7 showed moderate antibacterial activity.
Keywords: Aconitum carmichaelii, lateral root, alkaloids, bioactivities
1. Introduction
Aconitum carmichaelii Debx. (Ranunculaceae) is widely distributed and cultivated in China's Sichuan province [1]. The parent and lateral roots of A. carmichaelii, two well known traditional Chinese medicines, named “chuan wu” and “fu zi” respectively in Chinese, have been widely used in China to treat various symptoms such as cadianeuria, neuralgia, rheumatalgia, and inflammation [1,2]. Previous chemical studies of this plant have led to the isolation of more than 66 diterpenoid alkaloids, four flavonoids, a ceramide, a steroid saponin, and a pyrimidine [2,3,4,5]. In addition, 147 diterpenoid alkaloids, including a lot of lipo-alkaloids, have been reported and identified by means of LC-MS analysis [6,7]. In searching for bioactive natural products from “fu zi”, two new alkaloids, aconicarmine (1) and aconicaramide (5), were isolated from the lateral roots, together with five known compounds: fuziline (2) [8], neoline (3) [9], N-ethylhokbusine B (4) [10], 5-hydroxymethyl-pyrrole-2-carbaldehyde (6) [11], and oleracein E (7) [12]. This paper describes the isolation, structure elucidation, and bioassays of these isolates.
2. Results and Discussion
The EtOH extract of the lateral roots of A. carmichaelii was suspended in water and successively partitioned with petroleum ether, EtOAc, and n-BuOH. Separation of the n-BuOH fraction by column chromatography provided compounds 1−7 (Figure 1).
Figure 1.
Structures of compounds 1–7.
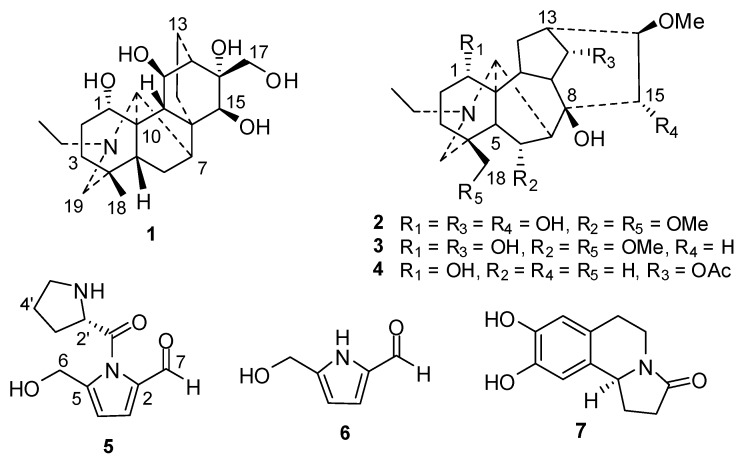
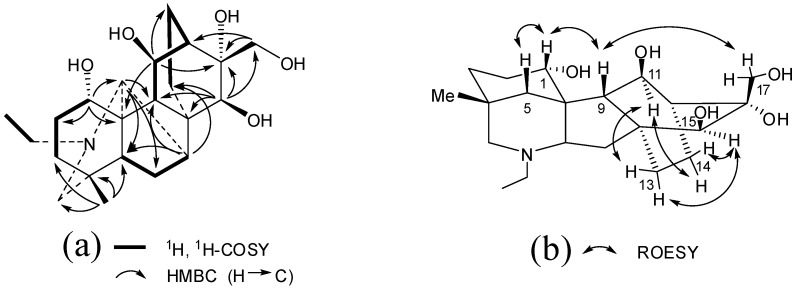
Compound 1 was obtained as colorless needles. The molecular formula C22H35NO5, with six degrees of unsaturation, was indicated by HR-ESI-MS m/z 394.2596 [M+H]+ (calcd for C22H36NO5, 394.2593) and NMR data (Table 1). The 1H-NMR spectrum displayed resonances assignable to an angular methyl group (δH 0.75, 3H, s, H-18), a N-ethyl group (δH 1.10, 3H, t, J = 7.2 Hz, H-22 and δH 2.48, 2H, q, J = 7.2 Hz, H-21), a N-methine group (δH 3.75, 1H, br s, H-20), three oxymethines (δH 3.83, 1H, s, H-15; δH 4.18, 1H, dd, J = 4.8, 10.2 Hz, H-1; and δH 4.56, 1H, d, J = 9.6 Hz, H-11), and an isolated oxymethylene group (δH 3.79, 1H, d, J = 12.0 Hz, H-17a and δH 4.03, 1H, d, J = 12.0 Hz, H-17b). In addition, it showed partially overlapped resonances ascribable to several aliphatic methylenes and methines between δH 0.99 and 2.86 ppm. The 13C-NMR and DEPT spectra of 1 revealed 22 carbon resonances corresponding to the above protonated units and four quaternary carbons (one oxygen-bearing, δC 80.3). The above-mentioned spectroscopic data suggested that 1 was a C20-diterpenoid alkaloid with an atisine-denudatine skeleton and an N-ethyl group [13]. Detailed comparison of the NMR data and the molecular composition of 1 with those of 11-epi-16α,17-dihydroxylepenine [14] indicated that compound 1 was an isomer of the latter. The 13C-NMR spectrum showed high similarity between them, except that the signal of C-11 (δC 72.3) in 1 was deshielded by 7.5 ppm compared to that of 11-epi-16α,17-dihydroxylepenine possessing an α hydroxy group at C-11. This revealed 1 was an 11β-hydroxyepimer [14], which was proved by 2D NMR experiments, including the ROESY analysis. The five OH groups could be located at C-1, C-11, C-15, C-16, and C-17, respectively, according to their HMBC correlations (Figure 2). In the ROESY spectrum, the correlations of H-11 and H-15 with H-13 and H-14, the same as those of lepenine [15], verified the OH-11 and OH-15 groups were β-oriented. Moreover, the correlations of H-1/H-5, H-1/H-9, and H-9/H-17 demonstrated the α-configuration of OH-1 and OH-16 (Figure 3). Accordingly, Compound 1 was established to be 16α,17-dihydroxylepenine and named aconicarmine.
Table 1.
1H- (400 MHz) and 13C-NMR (100 MHz) data of 1 (in CD3OD, δ in ppm, J in Hz).
| No. | δ H | δ C | No. | δ H | δ C |
|---|---|---|---|---|---|
| 1 | 4.18 dd (10.8, 6.4) | 71.3 | 12 | 1.60 m | 46.2 |
| 2 | 1.78 m, 2.40 m | 31.8 | 13 | 1.42 m, 1.92 m | 21.6 |
| 3 | 1.39 m, 1.61 m | 39.7 | 14 | 1.03 m, 1.84 m | 28.3 |
| 4 | – | 34.6 | 15 | 3.83 s | 86.4 |
| 5 | 1.37 d (8.0) | 54.3 | 16 | – | 80.3 |
| 6 | 1.26 dd (14.0, 5.2) | 24.5 | 17 | 3.79 d (12.0) | 69.2 |
| 2.86 dd (14.0, 8.0) | – | 4.03 d (12.0) | – | ||
| 7 | 2.12 d (5.2) | 43.5 | 18 | 0.75 s | 26.4 |
| 8 | – | 44.9 | 19 | 2.31 d (11.2), 2.58 d (11.2) | 58.1 |
| 9 | 1.82 d (9.6) | 51.8 | 20 | 3.75 br s | 68.5 |
| 10 | – | 52.1 | 21 | 2.48 m, 2.60 m | 52.1 |
| 11 | 4.56 d (9.6) | 72.3 | 22 | 1.10 t (7.2) | 13.7 |
Figure 2.
(a) Key 1H, 1H-COSY and HMBC correlations of aconicarmine(1); (b) Key ROESY correlations of aconicarmine(1).
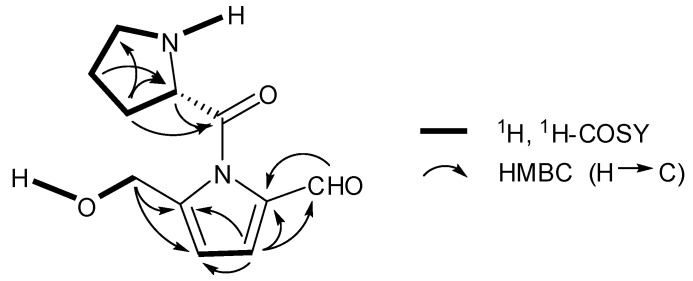
Figure 3.
Key 1H, 1H-COSY and HMBC correlations of aconicaramide(5).
Compound 5, obtained as a white powder, had the molecular formula C11H14N2O3 as indicated by HR-ESI-MS m/z 245.0906 [M+Na]+ (calcd for C11H14N2O3Na, 245.0902). The 1H-NMR spectrum of 5 displayed signals ascribed to an aldehyde group (δH 9.37, 1H, s, H-7), a pair of coupled olefinic methine protons (δH 6.19, 1H, d, J = 4.0 Hz, H-4 and δH 6.95, 1H, d, J = 4.0 Hz, H-3), an isolated oxymethylene group (δH 4.65, 2H, s, H-6), and an exchangeable proton (δH 4.39, s). These data, similar to those of 6 (Table 2), together with the carbon signals at (δC 178.8, 144.5, 133.4, 125.5, 110.0, and 56.7), indicated the presence of a pyrrole ring with the substitutions of an aldehyde and a hydroxymethyl group [16]. This was confirmed by HMBC correlations of H-3 with C-2, C-4, C-5, and C-7, H-6 with C-4 and C-5 (Figure 3).
Table 2.
1H- (400 MHz) and 13C-NMR (100 MHz) data of 5 and 6 (in CD3COCD3, δ in ppm, J in Hz).
| No. | δ H | δ C | |||
|---|---|---|---|---|---|
| 5 | 6 | 5 | 6 | ||
| 1 | – | 10.88 s | – | – | |
| 2 | – | – | 133.4 | 132.6 | |
| 3 | 6.95 d (4.0) | 6.91 d (4.0) | 125.5 | 125.7 | |
| 4 | 6.19 d (4.0) | 6.22 d (4.0) | 110.0 | 108.5 | |
| 5 | – | – | 144.5 | 142.0 | |
| 6 | 4.65 s | 4.64 s | 56.7 | 56.8 | |
| 7 | 9.37 s | 9.47 s | 178.8 | 178.1 | |
| OH-6 | 4.39 s | 4.29 s | – | ||
| 1′ | 6.69 ( s) | – | |||
| 2′ | 5.00 dd (10.8, 5.6) | 57.5 | |||
| 3′ | 1.97 m, 2.23 m | 30.1 | |||
| 4′ | 1.97 m, 2.29 m | 23.6 | |||
| 5′ | 3.31 m, 3.63 m | 42.7 | |||
| 6′ | – | 169.2 | |||
Comparison of the NMR data between 5 and 6 indicated that they differed in the presence of resonances attributable to an additional prolyl moiety (δC 23.6, 29.9, 42.7, 57.5, 169.2) in 5 [17]. In addition, almost no downfield shift of H-6 was observed in 5 as compared with that of 6, suggesting that the prolyl unit was attached to N instead of OH-6. This conjecture was refined by a 1H-1H COSY correlation observed between the exchangeable proton (δH 4.39) and H-6 (δH 4.65), as well as no HMBC correlation of H-6 with the carbonyl (C-6′).
Thus, the planar structure of 5 was established. The (S)-configuration at C-2′ was deduced by the negative specific rotation ([α]20D = −75.0), consistent with that of (S)-proline [18], but opposite that of (R)-proline [19]. Therefore, compound 5 was determined as N-(L-prolyl)-5-hydroxymethyl-1H-pyrrole-2-carbaldehyde and named aconicaramide.
The protective activities of the compounds against cardiomyocyte damage induced by pentobarbital sodium in primary cultured neonatal rat cardiomyocytes were investigated by the MTT method. The results showed that pentobarbital sodium induced a significant inhibition of MTT reduction. At concentrations of 10 μM, 1 μM, and 0.1 μM, compounds 2 and 3 increased the cell viability obviously (Table 3) and recovered beating rhythm when examined under a microscope. In addition, compound 5 showed moderate antibacterial activity against Macrococcus caseolyticus, Staphylococcus epidermidis and Staphylococcus aureus (MIC 200, 400 and 800 μg/mL, respectively), while compound 7 displayed antibacterial activity against Staphylococcus aureus, Macrococcus caseolyticus, Klebsiella pneumoniae and Streptococcus pneumoniae (MIC 50, 200, 200 and 200 μg/mL, respectively).
Table 3.
Protective effects of 2 and 3 against cardiomyocyte damage induced by pentobarbital sodium.
| Compound | Increase of the cell viability (%) | ||
|---|---|---|---|
| 10 μM | 1 μM | 0.1 μM | |
| 2 | 65.44 | 64.14 | 63.09 |
| 3 | 73.82 | 72.51 | 47.64 |
3. Experimental
3.1. General
NMR spectra were recorded on a Bruker-AV-400 spectrometer. HRESIMS were measured with Waters Synapt G2 HDMS. IR were recorded on a Vector 22 FT-IR spectrometer. UV spectra were obtained on a Shimadzu UV-260 spectrophotometer. Optical rotations were measured with a Perkin-Elmer 341 plus. Column chromatography was performed with silica gel (200–300 mesh, Yantai Institute of Chemical Technology, Yantai, China), Al2O3 (100–200 mesh, Shanghai Ludu Chemical Reagent Factory, Shanghai 200000, China), MCI gel CHP 20P (75–150 μm, Mitsubishi Chemical, Co., Japan), and Sephadex LH-20 (Amersham Pharmacia Biotech AB, Uppsala, Sweden).
3.2. Plant Material
The lateral root of A. carmichaelii was collected in July of 2010 from the culture field in Jiangyou, Sichuan postal code, China. Plant identity was verified by Prof. Min Li (Chengdu University of TCM, Sichuan, China). A voucher specimen (SFZ-0710) was deposited at the School of Pharmacy, Chengdu University of TCM, Chengdu, China.
3.3. Extraction and Isolation
The air-dried lateral roots (5 kg) of A. carmichaelii were extracted three times with 95% EtOH (30 L) for 2 h under reflux. The EtOH extract was concentrated in vacuo to yield a semi-solid (620 g), which was suspended in water and then extracted successively with petroleum ether, EtOAc and n-BuOH (5 × 2.5 L, 25 °C). The n-BuOH extract (85 g) was subjected to silica gel CC using a gradient elution of CHCl3–MeOH (50:1–1:1) to afford eleven fractions (Fractions A–K). Fraction B was further separated by Sephadex LH-20 (CHCl3–MeOH 1:1) to give five subfractions (B1–B5). The successive separation of B2 with Sephadex LH-20 (MeOH-H2O 1:1) and with PTLC (CHCl3-Me2CO 15:1) yielded 4 (5 mg), 5 (10 mg), and 6 (7 mg). B4 was fractioned by Sephadex LH-20 (petroleum ether−CHCl3−MeOH, 2:2:1) to give 7 (21 mg). Compound 3 (1.2 g) was crystallizated from Fraction D and then recrystallizated with CHCl3. Fraction G was separated by flash chromatography over MCI gel with a gradient of increasing MeOH (20%–100%) in water, to yield subfractions G1–G6. G2 was purified via Sephadex LH-20 (MeOH–H2O 1:1) followed by crystallization to yield 2 (0.9 g). Separation of G5 by chromatography over Al2O3 (CHCl3−MeOH, 2:1) and Sephadex LH-20 (MeOH–H2O 1:1) to afford 1 (170 mg).
16α,17-Dihydroxylepenine (1): Colorless needles, [α]20D = −29.8 (c = 0.10, MeOH); IR (KBr) νmax: 3421, 2927, 1459, 1383, 1064 cm−1; ESI-MS m/z 394.2 [M+H]+, 416.2 [M+Na]+; HRESI-MS: m/z 394.2596 [M+H]+ (calcd for C22H36NO5, 394.2593); 1H- and 13C-NMR data see Table 1.
N-(L-Prolyl)-5-hydroxymethyl-1H-pyrrole-2-carbaldehyde (5): White powder, [α]20D = −75.0 (c = 0.28, MeOH); UV (MeOH) λmax: 202 (4.16), 258 (4.04), 294 (4.28) nm; IR (KBr) νmax: 3309, 2941, 2872, 1656, 1488, 1449, 1328, 1296, 1032, 776 cm−1; ESI-MS m/z 245.1 [M+Na]+; HRESI-MS m/z 245.0906 [M+Na]+ (calcd for C11H14N2O3Na, 245.0902); 1H- and 13C-NMR data see Table 2.
3.4. Cardiomyocyte Protection Assay
Neonatal rat cardiomyocytes were cultured in 96-well plates with DMEM media supplemented with 15% FBS. Cultures were maintained in a 37 °C humidified incubator with 5% CO2. On the fifth day when the cardiomyocytes were in the growth with rhythmical beating, they were exposed to the medium containing pentobarbital sodium at a concentration of 8 mg/mL. After 8 min, the medium was replaced with serum free medium including compounds at concentrations of 10 μM, 1 μM, and 0.1 μM, respectively, and incubated for 24 h. Then, 10 μL of MTT solution (5 mg/mL) was added and incubated for 4 h. Absorbance was measured at both 570 nm and 655 nm, and cell viability was evaluated with the deviations between them.
3.5. Antibacterial Activity Experiments
All bacteria were obtained from clinical samples and stored in the Department of Pharmacology of Chengdu University of TCM. The in vitro antibacterial activity was determined by the standard agar dilution method, according to NCCLS (National Committee for Clinical Laboratory Standard). 2 μL of cultures of test strains at the concentration of 1 × 106 CFU/mL were inoculated on Mueller Hinton agar containing different concentrations of the test compounds. The MIC values were determined after incubation at 35–37 °C for 18–24 h.
4. Conclusions
Two new alkaloids aconicarmine (1) and aconicaramide (5) were isolated from the lateral roots of A. carmichaelii, together with five known alkaloids. Compounds 5 and 6 were the first report of pyrrole alkaloids from A. carmichaelii. Compounds 2 and 3 showed activity against pentobarbital sodium-induced cardiomyocytes damage by recovering beating rhythm and increasing the cell viability obviously. Compounds 5 and 7 showed moderate antibacterial activity.
Acknowledgments
Financial support from the National Technology R&D Program for the “Twelfth Five-Year” Plan of China (grant No. 2010BAE00406), and the 973 Project of the Ministry of Sciences and Technology (grant No. 2006CB5604705) is acknowledged.
Conflict of Interest
The authors declare no conflict of interest.
Footnotes
Sample Availability: Not availbale.
References
- 1.Jiangsu New Medical College. Dictionary of Traditional Chinese Medicine. Shanghai Science and Technology Publishing House; Shanghai, China: 1995. pp. 228–232.pp. 1191–1194. [Google Scholar]
- 2.Shen Y., Zuo A.X., Jiang Z.Y., Zhang X.M., Wang H.L., Chen J.J. Two new C20-diterpenoid alkaloids from Aconitum carmichaelii. Helv. Chim. Acta. 2011;94:122–126. doi: 10.1002/hlca.201000152. [DOI] [Google Scholar]
- 3.Jiang B.Y., Lin S., Zhu C.G., Wang S.J., Wang Y.N., Chen M.H., Zhang J.J., Hu J.F., Chen N.H., Yang Y.C., et al. Diterpenoid alkaloids from the lateral root of Aconitum carmichaelii. J. Nat. Prod. 2012;75:1145–1159. doi: 10.1021/np300225t. [DOI] [PubMed] [Google Scholar]
- 4.Shim S.H., Lee S.Y., Kim J.S., Son K.H., Kang S.S. Norditerpenoid alkaloids and other components from the processed tubers of Aconitum carmichaeli. Arch. Pharm. Res. 2005;28:1239–1243. doi: 10.1007/BF02978206. [DOI] [PubMed] [Google Scholar]
- 5.Han G.Y., Liang H.Q., Zhang W.D., Chen H.S., Wang J.Z., Liao Y.Z., Liu M.Z., Shen Q.H. Studies on the alkaloids and a new cardiac principle isolated from Jiangyou Fuzi (Aconitum carmichaelii Debx.) Nat. Prod. Res. Dev. 1997;9:30–33. [Google Scholar]
- 6.Csupor D., Wenzig E.M., Zupkó I., Wölkart K., Hohmann J., Bauer R. Qualitative and quantitative analysis of aconitine-type and lipo-alkaloids of Aconitum carmichaelii roots. J. Chromatogr. A. 2009;1216:2079–2086. doi: 10.1016/j.chroma.2008.10.082. [DOI] [PubMed] [Google Scholar]
- 7.Yue H., Pi Z., Song F., Liu Z., Cai Z., Liu S. Studies on the aconitine-type alkaloids in the roots of Aconitum Carmichaeli Debx. by HPLC/ESIMS/MSn. Talanta. 2009;77:1800–1807. doi: 10.1016/j.talanta.2008.10.022. [DOI] [PubMed] [Google Scholar]
- 8.Pelletier S.W., Mody N.V., Varughese K.I. Fuziline, A new alkaloid from the Chinese drug “Fuzi” (Aconitum carmichaeli Debx) Heterocycles. 1982;18:47–49. doi: 10.3987/S(B)-1982-01-0047. [DOI] [Google Scholar]
- 9.Khetwal K.S., Desai H.K., Joshi B.S., Pelletier S.W. Norditerpenoid alkaloids from the aerial parts of Aconitum balfourii stapf. Heterocycles. 1994;38:833–842. doi: 10.3987/COM-93-6638. [DOI] [Google Scholar]
- 10.Hikino H., Kuroiwa Y., Konno C. Structure of hokbusine A and B, diterpenic alkaloids of Aconitum carmichaeli roots from Japan. J. Nat. Prod. 1983;46:178–182. doi: 10.1021/np50026a006. [DOI] [Google Scholar]
- 11.Hiermann A., Kedwani S., Schramm H.W., Seger C. A new pyrrole alkaloid from seeds of Castanea sativa. Fitoterapia. 2002;73:22–27. doi: 10.1016/S0367-326X(01)00344-6. [DOI] [PubMed] [Google Scholar]
- 12.Xiang L., Xing D.M., Wang W., Wang R.F., Ding Y., Du L.J. Alkaloids from Portulaca oleracea L. Phytochemistry. 2005;66:2595–2601. doi: 10.1016/j.phytochem.2005.08.011. [DOI] [PubMed] [Google Scholar]
- 13.Wada K., Kawahara N. Diterpenoid and norditerpenoid alkaloids from the roots of Aconitum yesoense var. macroyesoense. Helv. Chim. Acta. 2009;92:629–637. doi: 10.1002/hlca.200800345. [DOI] [Google Scholar]
- 14.Zhang F., Peng S.L., Liao X., Yu K.B., Ding L.S. Three new diterpene alkaloids from the roots of Aconitum nagarum var. lasiandrum. Planta Med. 2005;71:1073–1076. doi: 10.1055/s-2005-873135. [DOI] [PubMed] [Google Scholar]
- 15.Batsuren D., Tunsag J., Batbayar N., Mericli A.M., Mericli F., Teng Q., Desai H.K., Joshi B.S., Pelletier S.W. Alkaloids of some mongolian Ranunculaceae species: detailed 1H and 13C-NMR studies of denudatine and lepenine. Heterocycles. 1998;49:327–341. doi: 10.3987/COM-98-S40. [DOI] [Google Scholar]
- 16.Sudhakar G., Kadam V.D., Bayya S., Pranitha G., Jagadeesh B. Total synthesis and stereochemical revision of acortatarins A and B. Org. Lett. 2011;13:5452–5455. doi: 10.1021/ol202121k. [DOI] [PubMed] [Google Scholar]
- 17.Lu D.F., Gong Y.F., Wang W.Z. Prolylprolinol catalyzed asymmetric michael addition of aliphatic aldehydes to nitroalkenes. Adv. Synth. Catal. 2010;352:644–650. doi: 10.1002/adsc.200900687. [DOI] [Google Scholar]
- 18.Fadel A., Lahrache N. An efficient synthesis of enantiomerically pure (R)-pipecolic acid, (S)-proline, And their N-alkylated derivatives. J. Org. Chem. 2007;72:1780–1784. doi: 10.1021/jo062382i. [DOI] [PubMed] [Google Scholar]
- 19.Fitzi R., Seebach D. Resolution and use in α-amino acid synthesis of imidazolidinone glycine derivatives. Tetrahedron. 1988;44:5277–5292. doi: 10.1016/S0040-4020(01)86036-1. [DOI] [Google Scholar]