Table 2.
HCCP-mediated formation of quinazoline thioethers from quinazolin-4(3H)-ones a. 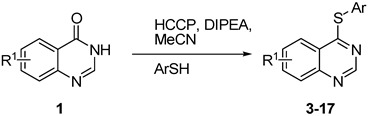
| Entry | Quinazolin-4(3H)-one | ArSH | Product | Yield (%) b | ||
|---|---|---|---|---|---|---|
| 1 |
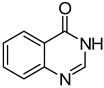
|
1a |
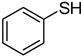
|
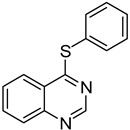
|
3 | 79 |
| 2 | 1a |
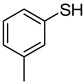
|
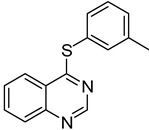
|
4 | 64 c | |
| 3 | 1a |
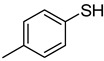
|
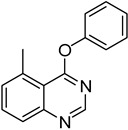
|
5 | 69 | |
| 4 | 1a |
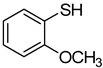
|
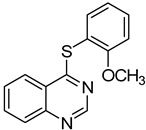
|
6 | 59 | |
| 5 | 1a |
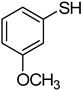
|
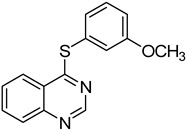
|
7 | 50 | |
| 6 | 1a |
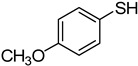
|
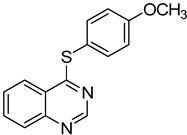
|
8 | 86 | |
| 7 | 1a |
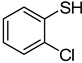
|
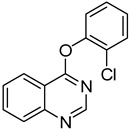
|
9 | 94 | |
| 8 | 1a |
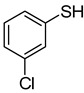
|
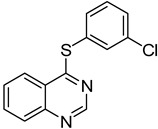
|
10 | 66 | |
| 9 | 1a |
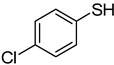
|
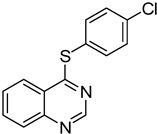
|
11 | 91 | |
| 10 |
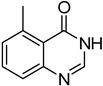
|
1b |
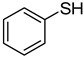
|
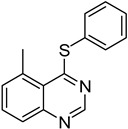
|
12 | 54 d |
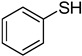
| 11 |
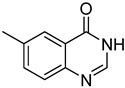
|
1c |
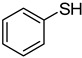
|
|
13 | 60 |
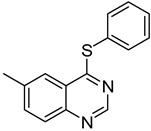
| 12 |
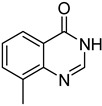
|
1d |
|
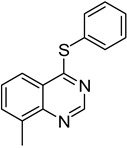
|
14 | 64 |
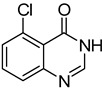
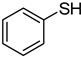
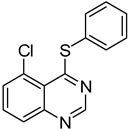
| 13 |
|
1e |
|
|
15 | 51 d |
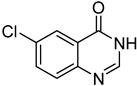
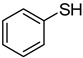
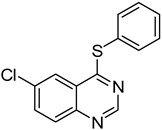
| 14 |
|
1f |
|
|
16 | 79 |
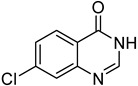
| 15 |
|
1g |
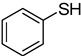
|
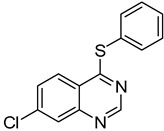
|
17 | 72 |
aReagents andConditions: 1 (0.5 mmol), HCCP (1.1 equiv.), DIPEA (5.0 equiv.), MeCN (5 mL), rt, activation time (1 h), then thiophenols (5.0 equiv.), 45 °C, 23 h; b Isolated yield; cm-CH3PhSH (6.0 equiv.); reaction time (48 h); d Activation time (20 h).
