Abstract
Research was carried out to estimate the levels of capsaicin and dihydrocapsaicin that may be found in some heat tolerant chili pepper genotypes and to determine the degree of pungency as well as percentage capsaicin content of each of the analyzed peppers. A sensitive, precise, and specific ultra fast liquid chromatographic (UFLC) system was used for the separation, identification and quantitation of the capsaicinoids and the extraction solvent was acetonitrile. The method validation parameters, including linearity, precision, accuracy and recovery, yielded good results. Thus, the limit of detection was 0.045 µg/kg and 0.151 µg/kg for capsaicin and dihydrocapsaicin, respectively, whereas the limit of quantitation was 0.11 µg/kg and 0.368 µg/kg for capsaicin and dihydrocapsaicin. The calibration graph was linear from 0.05 to 0.50 µg/g for UFLC analysis. The inter- and intra-day precisions (relative standard deviation) were <5.0% for capsaicin and <9.9% for dihydrocapsaicin while the average recoveries obtained were quantitative (89.4%–90.1% for capsaicin, 92.4%–95.2% for dihydrocapsaicin), indicating good accuracy of the UFLC method. AVPP0705, AVPP0506, AVPP0104, AVPP0002, C05573 and AVPP0805 showed the highest concentration of capsaicin (12,776, 5,828, 4,393, 4,760, 3,764 and 4,120 µg/kg) and the highest pungency level, whereas AVPP9703, AVPP0512, AVPP0307, AVPP0803 and AVPP0102 recorded no detection of capsaicin and hence were non-pungent. All chili peppers studied except AVPP9703, AVPP0512, AVPP0307, AVPP0803 and AVPP0102 could serve as potential sources of capsaicin. On the other hand, only genotypes AVPP0506, AVPP0104, AVPP0002, C05573 and AVPP0805 gave a % capsaicin content that falls within the pungency limit that could make them recommendable as potential sources of capsaicin for the pharmaceutical industry.
Keywords: capsaicin, dihydrocapsaicin, ultra-fast liquid chromatography, chili pepper, Scoville heat units
1. Introduction
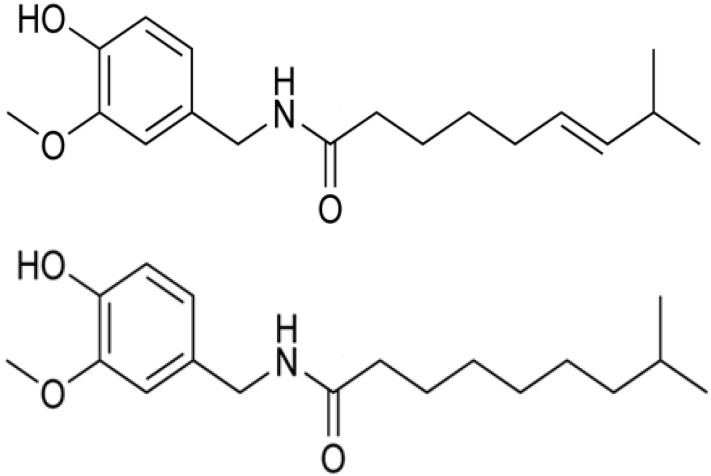
Chili pepper, which belongs to the genus Capsicum contains capsaicinoids, alkaloid compounds that produce the pungency associated with eating chilies [1]. The two major capsaicinoids are capsaicin (N-[(4-hydroxy-3-methoxypheny) methyl]-8-methyl-E-6-nonenamide) and dihydrocapsaicin (N-[(4-hydroxy-3-methoxyphenyl)methyl]-8-methyl-6-nonanamide) which comprise over 90% of the total present in the fruit [2] (Figure 1). Capsaicin is a flavourless, odourless and colourless compound found in varying amounts in peppers. Capsaicinoids are only found in the Capsicum genus and are bioactive molecules currently relevant in medical and food sciences [3,4,5] as well as in the defense weapon industry [6]. Capsaicinoids occur in the placental tissue of pepper fruits [7], and their biosynthesis depends on a complex and still not fully characterized enzymatic pathway.
Figure 1.
Structures of capsaicin (top) and dihydrocapsaicin (bottom).
Capsaicin is the active element in pepper, which accounts for its prominent pharmaceutical and antioxidant properties. Research has shown that the more the capsaicin, the hotter the pepper, and the higher the antioxidant level. It is the principal pungent and irritating constituent of hot peppers that produce the pungency associated with the eating of chilies. Capsaicin and other capsaicinoids produce a number of physiological and pharmacological effects on the cardiovascular system and gastro-intestinal track [8,9,10,11,12]. Capsaicin in peppers has been shown to slightly control appetite – at least briefly. It has also been reported to raise the body temperature [9]. That warming effect may have another benefit that may help with weight loss. The temperature at which chili peppers are grown, the position of the fruit on the plant, age of the plant and light intensity are all factors affecting the total amount of capsaicin in a given chili pepper variety. Capsaicinoid levels depend on the genotype [13] and also change during fruit development [14,15,16]. Moreover, environmental and nutritional conditions occurring during the cultivation of peppers can affect the capsaicinoid content. For instance, significant differences in pungency were found in double-haploid chili plants grown in five different plots of the same field [17], and the total capsaicinoid content in “Padrón” pepper fruits developed in summer was found to be larger than in those fruits developed in autumn [18].
The large variability in capsaicinoid content found naturally in pepper genotypes is a critical point in breeding and production. For instance, capsaicin and dihydrocapsaicin contents ranged from 2 to 6,639 mg/kg in eight different pepper genotypes [19]. Therefore, there is a requirement for analytical techniques able to determine very low amounts of capsaicinoids. These techniques should also be capable of determining amounts of the different capsaicinoid molecules, which have very similar chemical structures. These requirements are met by HPLC-MS (mass spectrometry) techniques, which have a high selectivity and sensitivity and have been used for the determination of capsaicinoids in forensic, medical, and food sciences [19,20,21,22].
The first method developed for the measurement of chili pungency was the Scoville Organoleptic Test [23]. A group of five testers assess a water-diluted chili sample and then records the hot flavor level. Serial dilution of the sample is necessary to make the pungency undetectable. A number is assigned to each hot pepper according to the dilution test and expressed it as a scale called the Scoville Organoleptic Scale assigned by Pharmacist Wilbur Scoville [23]. The heat levels vary widely from 0–500,000 Scoville heat units (SHU). They are classified as:
-
-
(0–700 SHU) non-pungent
-
-
(700–3,000 SHU) mildly pungent
-
-
(25, 000–70,000 SHU) highly pungent
-
-
(3,000–25,000 SHU) moderately pungent
-
-
(>80,000 SHU) very highly pungent [24]
However, the traditional method has been replaced by a number of instrumental methods such as Gas chromatography (GC), Gas Chromatography-Mass Spectrometry (GC-MS) and high performance liquid chromatography (HPLC) which are more reliable and accurate. Researchers need reliable, safe and standard methods that could be useful for comparing pungency levels among different samples or genotypes of chili. In this research Ultra Fast Liquid Chromatography (UFLC) was used, which is faster and simpler than conventional HPLC. The transition from LC to ultra fast LC reduces some of the limitations normally associated with LC. With HPLC, when analyzing multiple samples, each of which takes a long time to complete, the need to conduct re-analysis for whatever reason can result in product delays. However, with ultra fast liquid chromatography, an ultra high speed analysis could be achieved. This means of shortening of the time required to complete the analysis, thereby reducing the risks associated with time-sensitive analyses. This research also aims at estimating the levels of capsaicin and dihydrocapsaicin that may be found in some heat tolerant pepper varieties and to determine the degree of pungency as well as percentage of capsaicins of each of the analyzed peppers which could be used in pharmaceuticals.
2. Results and Discussion
2.1. Optimization of UFLC Separation Condition
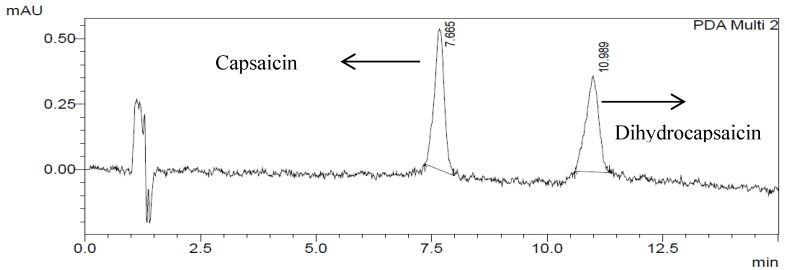
The chromatographic conditions used were optimized with the aim of obtaining the separation with of adjacent peaks with good resolution within a short analysis time. A binary mixture of 1% acetic acid (aq)—acetonitrile was used as described by the AOAC [25] official method. Under the optimal isocratic conditions, both capsaicin (retention time 7.665 min) and dihydrocapsaicin (retention time 10.989 min) were separated within 15 min (Figure 2). Since the molecular structures of both capsaicin and dihydrocapsaicin are very similar, the maximum absorption wavelengths determined by PDA are also nearly the same and found to be 280.8 and 279.6 nm. The PDA using Shimadzu LC solution software detects the absorbance at the chosen wavelengths of the capsaicinoids and simultaneously provides their absorption spectra. Identification of compounds was achieved by retention time and absorption spectrum of standard and sample. However, both compounds were detected with PDA at 280 nm.
Figure 2.
Chromatogram of capsaicin and dihydrocapsaicin (0.50 µg/g) using UV detection at 280 nm.
2.2. Method Validation
The validation and verification of the UFLC method was carried out according to international guidelines for validation of the Food and Drug Administration (FDA) and National Association of Testing Authorities (NATA):
Linearity The linearity was found to be in the range of 0.05 to 0.50 µg/g for both compounds. Standard solutions were prepared from a stock solution of capsaicin and dihydrocapsaicin using six serial dilutions at 0.50, 0.40, 0.30, 0.20, 0.10 and 0.5 µg/g (acceptable by NATA). Each solution was injected three times and the average values of the triplicate analysis were presented in Table 1. The standard solutions were run on the ultra high performance liquid chromatography and the standard curves were generated by plotting peak area against concentration. The external calibration curves (Supplementary Data) were found at r2 = 0.9999 for capsaicin and r2 = 0.9996 for dihydrocapsaicin. The values of r2 were highly significant confirming the good linearity of the method. The regression line equations were:
| Y = 18419x + 188.83 (capsaicins); Y = 15797x + 148.72 (dihydrocapsaicins) | (1) |
Table 1.
Calibration data of the UFLC method for the determination of capsaicinoids (µg/g).
| Capsaicinoids | Linear Range | R2 | Ret. Time | Average Peak Area | SD | % RSD |
|---|---|---|---|---|---|---|
| Capsaicin | 0.05–0.50 | 0.9999 | 7.665 | 4947.1 | 27.7 | 0.56 |
| Dihydrocapsaicin | 0.05–0.50 | 0.9996 | 10.989 | 4229.6 | 58.1 | 1.37 |
n = 3.
The y-intercept means that when the concentration (x) = 0, then the peak areas of capsaicin and dihydrocapsaicin would be 189 and 149, respectively. The lowest measured values in this investigation for capsaicin and dihydrocapsaicin were 1,106 and 949 respectively, which are already five times the y-intercepts. This showed that all other values would be reliable. However, in this context, y-intercepts are not relevant, since at 0 µg/g of capsaicins and dihydrocapsaicins no peak area would be detected.
To check for the significant intercepts, we calculated percentage of y-intercept by dividing its value by the response of the 100% concentration response, multiplied by 100. We obtained values within ±2.0% both for capsaicin and dihydrocapsaicin, which are associated to the correlation coefficient which were more or equal to 0.999; we therefore considered that the standard curves are linear. For capsaicin, 100% concentration response was 9,392 (y-intercept 2.0%) and for dihydrocapsaicin it was 8,097 (y-intercept 1.8%). These limits are acceptable by the international guidelines for validation of the FDA.
Expected and actual concentration responses were plotted against expected concentrations (0.5, 0.25, 0.125 and 0.0625 µg/g) using four dilution factors (0, 2, 4 and 8). They both gave an equation (y = bx). The expected stock dilutions are more concentrated than the actual concentration dilutions which indicated that the dilutions are the concentrations expected (see Supplementary Data).
Limit of Detection (LOD) and Limit of Quantitation (LOQ) The method was validated by evaluating limit of detection (LOD) and limit of quantitation (LOQ) for both capsaicinoids. LOD and LOQ were estimated at an SD/b ratio of 3 and 10, where SD and b stand for the standard deviation of the slope and intercept of the regression line, respectively. The limit of detection (LOD) was 0.045 µg/kg and 0.151 µg/kg for capsaicin and dihydrocapsaicin, respectively. The limit of quantitation (LOQ) was 0.110 µg/kg and 0.368 µg/kg for capsaicin and dihydrocapsaicin, respectively.
Reproducibility An inter-day reproducibility (n = 30; acceptable by FDA and NATA) test was performed on capsaicin and dihydrocapsaicin for the UFLC method using four different chili pepper genotypes. The average relative standard deviations of the 30 replicate analysis of the inter-day reproducibility were represented in Table 2. This showed that the UFLC method is highly reproducible.
Table 2.
Inter-day reproducibility data of the UFLC method for the determination of capsaicinoids in pepper (µg/kg).
| AVPP0705 | AVPP0002 | AVPP0805 | C05573 | |||||
|---|---|---|---|---|---|---|---|---|
| No. Sample | Cap | Dihy | Cap | Dihy | Cap | Dihy | Cap | Dihy |
| 1 | 1908 (1) | 711 | 768 | 486 | 492 | 420 | 476 | 358 |
| 2 | 1868 | 690 | 811 | 485 | 502 | 433 | 466 | 360 |
| 3 | 1798 | 750 | 798 | 501 | 472 | 390 | 456 | 371 |
| 4 | 1867 | 701 | 779 | 499 | 501 | 387 | 500 | 350 |
| 5 | 1902 | 699 | 700 | 512 | 512 | 417 | 467 | 351 |
| 6 | 1998 | 680 | 801 | 501 | 511 | 401 | 480 | 354 |
| 7 | 1798 | 712 | 822 | 512 | 499 | 413 | 456 | 348 |
| 8 | 1811 | 718 | 783 | 493 | 518 | 429 | 489 | 359 |
| 9 | 1798 | 675 | 814 | 524 | 522 | 410 | 480 | 366 |
| 10 | 1928 | 700 | 780 | 505 | 498 | 386 | 457 | 379 |
| 11 | 1788 | 690 | 764 | 516 | 519 | 427 | 470 | 367 |
| 12 | 1901 | 721 | 817 | 507 | 510 | 388 | 469 | 346 |
| 13 | 1691 | 710 | 818 | 498 | 486 | 427 | 498 | 379 |
| 14 | 1800 | 724 | 808 | 509 | 532 | 430 | 481 | 380 |
| 15 | 1860 | 691 | 802 | 481 | 520 | 379 | 465 | 356 |
| 16 | 2198 | 736 | 821 | 521 | 472 | 424 | 488 | 345 |
| 17 | 1998 | 722 | 729 | 462 | 494 | 378 | 487 | 361 |
| 18 | 1878 | 716 | 813 | 481 | 514 | 415 | 497 | 344 |
| 19 | 1754 | 702 | 794 | 474 | 512 | 411 | 476 | 368 |
| 20 | 1791 | 683 | 765 | 535 | 508 | 402 | 486 | 376 |
| 21 | 1802 | 724 | 816 | 506 | 519 | 378 | 472 | 358 |
| 22 | 1855 | 731 | 807 | 527 | 481 | 367 | 459 | 364 |
| 23 | 1868 | 734 | 818 | 508 | 496 | 432 | 469 | 361 |
| 24 | 1801 | 745 | 799 | 487 | 477 | 421 | 480 | 357 |
| 25 | 1831 | 728 | 840 | 520 | 462 | 389 | 490 | 383 |
| 26 | 1808 | 767 | 788 | 491 | 530 | 398 | 487 | 377 |
| 27 | 1798 | 771 | 801 | 522 | 505 | 435 | 477 | 354 |
| 28 | 1818 | 747 | 810 | 502 | 528 | 426 | 485 | 362 |
| 29 | 1899 | 789 | 824 | 524 | 497 | 419 | 491 | 391 |
| 30 | 1861 | 724 | 775 | 515 | 474 | 399 | 475 | 365 |
| Mean | 1855.44 | 719.71 | 795.50 | 503.47 | 502.10 | 407.70 | 477.63 | 363.00 |
| SD | 92.37 | 27.50 | 29.31 | 17.29 | 18.76 | 19.45 | 12.53 | 11.99 |
| RSD% | 4.98 | 3.82 | 3.68 | 3.43 | 3.74 | 4.77 | 2.62 | 3.30 |
(1) Values represent the mean of five replicate analysis; SD, standard deviation; RSD, relative standard deviation; Cap, capsaicin; Dihy, dihydrocapsaicin; n = 30.
Repeatability An intra-day repeatability (n = 30) test was performed on capsaicin and dihydrocapsaicin for the UFLC method using four different chili pepper genotypes. The average relative standard deviations of the 30 replicate analysis of the intra-day repeatability were represented in Table 3. The result shows that the method is highly repeatable.
Table 3.
Intra-day repeatability data of the UFLC method for the determination of capsaicinoids inpepper (μg/kg).
| AVPP0705 | AVPP0002 | AVPP0805 | C05573 | |||||
|---|---|---|---|---|---|---|---|---|
| No. Sample | Cap | Dihy | Cap | Dihy | Cap | Dihy | Cap | Dihy |
| 1 | 1778 (1) | 794 | 677 | 411 | 481 | 389 | 386 | 288 |
| 2 | 1801 | 789 | 687 | 401 | 488 | 367 | 381 | 298 |
| 3 | 1798 | 777 | 666 | 409 | 498 | 380 | 388 | 290 |
| 4 | 1890 | 698 | 657 | 418 | 468 | 381 | 387 | 295 |
| 5 | 1870 | 650 | 689 | 399 | 470 | 385 | 370 | 280 |
| 6 | 1786 | 699 | 670 | 389 | 484 | 379 | 377 | 279 |
| 7 | 1832 | 730 | 697 | 388 | 479 | 370 | 376 | 281 |
| 8 | 1799 | 786 | 678 | 390 | 480 | 377 | 369 | 286 |
| 9 | 1875 | 756 | 680 | 400 | 485 | 384 | 380 | 291 |
| 10 | 1800 | 790 | 681 | 412 | 489 | 386 | 383 | 278 |
| 11 | 1776 | 769 | 699 | 408 | 500 | 378 | 379 | 299 |
| 12 | 1854 | 798 | 657 | 403 | 496 | 390 | 385 | 296 |
| 13 | 1831 | 766 | 673 | 398 | 477 | 391 | 397 | 285 |
| 14 | 1876 | 801 | 674 | 405 | 465 | 394 | 390 | 294 |
| 15 | 1894 | 799 | 660 | 410 | 478 | 387 | 389 | 284 |
| 16 | 1799 | 800 | 664 | 409 | 473 | 369 | 375 | 287 |
| 17 | 1876 | 811 | 675 | 420 | 476 | 388 | 367 | 287 |
| 18 | 1865 | 737 | 674 | 419 | 483 | 376 | 378 | 284 |
| 19 | 1745 | 788 | 679 | 396 | 454 | 395 | 394 | 280 |
| 20 | 1789 | 776 | 672 | 386 | 495 | 385 | 392 | 283 |
| 21 | 1699 | 781 | 669 | 397 | 475 | 380 | 386 | 278 |
| 22 | 1855 | 787 | 681 | 408 | 476 | 382 | 374 | 289 |
| 23 | 1866 | 780 | 679 | 415 | 476 | 375 | 384 | 300 |
| 24 | 1886 | 769 | 665 | 414 | 497 | 367 | 382 | 276 |
| 25 | 1876 | 770 | 671 | 402 | 469 | 366 | 372 | 288 |
| 26 | 1856 | 779 | 677 | 412 | 472 | 387 | 378 | 277 |
| 27 | 1767 | 784 | 686 | 397 | 486 | 386 | 379 | 286 |
| 28 | 1803 | 793 | 676 | 386 | 465 | 388 | 383 | 281 |
| 29 | 1896 | 764 | 654 | 399 | 490 | 374 | 371 | 284 |
| 30 | 1876 | 813 | 649 | 407 | 477 | 385 | 369 | 273 |
| Mean | 1830.5 | 771.13 | 673.9 | 403.6 | 480.1 | 381.4 | 380.7 | 285.9 |
| SD | 50.76 | 36.13 | 11.76 | 9.83 | 11.03 | 8.11 | 7.87 | 7.16 |
| RSD% | 2.77 | 4.69 | 1.75 | 2.44 | 2.30 | 2.13 | 2.07 | 2.50 |
(1) Values represent the mean of five replicate analysis; SD, standard deviation; RSD, relative standard deviation; Cap, capsaicin; Dihy, dihydrocapsaicin; n = 30.
Precision and accuracy Intra-day and inter-day precision data of the UFLC method were given in Table 4, indicating that the relative standard deviations are better than 5.0% for capsaicin and 9.9% for dihydrocapsaicin. Recovery experiments were performed using the standard addition method in order to study the accuracy of the UFLC method. The recovery of the added standard to the assay samples was calculated according to [26]:
| Percentage recovery % = [(Ct − Cu)/Ca] × 100 | (2) |
where Ct is the total concentration of the analyte found, Cu is the concentration of the present analyte in the original AVPP0705, and Ca is the concentration of the pure analyte added to the original AVPP0705. The results were given in Table 4. The average recoveries obtained were quantitative (89.4%–90.1% for capsaicin, 92.4%–95.2% for dihydrocapsaicin), indicating good accuracy of the UFLC method.
Table 4.
Precision and accuracy data of the UFLC for the determination of capsaicinoids in AVPP0705.
| Component (1) | Spiked amount (µg/kg) | Intra-day (%) | Inter day (%) | Recovery (%) |
|---|---|---|---|---|
| Capsaicin | 1302 | 2.07 | 5.01 | 90.1 |
| 3009 | 4.81 | 3.27 | 89.4 | |
| Dihydrocapsaicin | 807.6 | 5.81 | 9.89 | 95.2 |
| 3541 | 5.00 | 4.63 | 92.4 |
(1) Sample weight approximately 3.0 g; Concentration of capsaicin and dihydrocapsaicin in the initial sample was 13,076 and 7,155 µg/kg, respectively; n = 3.
2.3. Analysis of Capsaicinoids in Samples
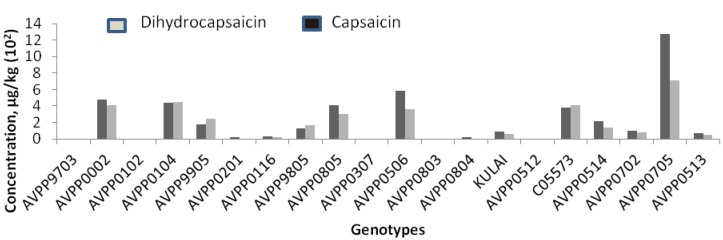
The high-speed analysis of the UFLC method was considered as providing good-efficiency analysis and to be environmentally friendly. The UFLC method was applied to determine the content of capsaicin and dihydrocapsaicin contents of twenty-one pepper genotypes and their corresponding pungency levels. The chromatograms attached (supplementary data) correspond to an extracted solution of some genotypes. From the chromatograms obtained from the studied chili peppers, the main peaks of interest identified among the capsaicinoids were capsaicin and dihydrocapsaicin. The UV absorption spectra corresponding to capsaicin and dihydrocapsaicin peaks were obtained from the photo diode array detector (PDA). The ultraviolet detection wavelength was set at 280 nm for all the capsaicinoids, because it is the maximum absorbance for both capsaicinoids. The chromatogram showed a complete separation between the two elements (capsaicin and dihydrocapsaicin) and no interference with other capsaicinoid peaks. The capsaicinoid contents are calculated and presented in Figure 3. The amount of capsaicin and dihydrocapsaicin from the chili pepper samples were found to differ significantly (p > 0.05). It ranged from 0–13,076 µg/kg and 0–7,155 µg/kg for both capsaicin and dihydrocapsaicin, respectively, as shown in Table 5. Genotype AVPP0705 was found to record the highest capsaicin content and was the highest in pungency which was significantly (p > 0.05) higher than all the other samples tested. Genotypes AVPP9703, AVPP0512, AVPP0307, AVPP0803 and AVPP0102 were found to record no capsaicin and therefore be non-pungent. Similar variation in capsaicin content of different peppers has been previously reported [27,28,29].
Figure 3.
Capsaicin and dihydrocapsaicin obtained using acetonitrile as extraction solvent.
Table 5.
The capsaicinoids content of the twenty-one chilli pepper genotype samples (µg/kg).
| Genotypes | Capsaicin | Dihydrocapsaicin | Total Capsaicinoids |
|---|---|---|---|
| AVPP0705 | 13076 | 7155 | 20231 |
| AVPP0506 | 5945 | 2999 | 8944 |
| AVPP0104 | 4283 | 4698 | 8981 |
| AVPP0002 | 4945 | 4346 | 9291 |
| C05573 | 2989 | 4280 | 7269 |
| AVPP0805 | 4230 | 3340 | 7570 |
| AVPP9905 | 2054 | 2218 | 4272 |
| AVPP0904 | 2012 | 1613 | 3625 |
| AVPP0514 | 2468 | 1470 | 3938 |
| AVPP9805 | 1248 | 1568 | 2816 |
| AVPP0702 | 1524 | 850 | 2374 |
| KULAI | 799 | 606 | 1405 |
| AVPP0513 | 892 | 553 | 1445 |
| AVPP0116 | 299 | 246 | 545 |
| AVPP0804 | 191 | ND | 191 |
| AVPP0201 | 186 | ND | 186 |
| AVPP9703 | ND | ND | ND |
| AVPP0512 | ND | ND | ND |
| AVPP0307 | ND | ND | ND |
| AVPP0803 | ND | ND | ND |
| AVPP0102 | ND | ND | ND |
n = 2.
For all the chili pepper samples, the correlation between Scoville heat unit (SHU) and the two capsaicinoids obtained was calculated as shown in Table 5 and Table 6 by using the relationship between this content (µg/kg) and its SHU rating of approximately 15 SHU equivalents to 10 µg/kg of capsaicinoids [30]. Therefore, their corresponding SHU were found in the range of 0-237,245 SHU. From these results, it is indicated that capsaicin and dihydrocapsaicin were primarily responsible for the SHU rating. Thus, the chili sample AVPP0705 gave quite a high SHU related with its higher content of the capsaicinoids. Therefore, total yields of capsaicinoids in these chili peppers ranged from 0–20,231 µg/kg. In addition, capsaicin and dihydrocapsaicin have the same trend in contents of the capsaicinoids, and in particular capsaicin was found in higher contents than dihydrocapsaicin in all samples studied except C05573, AVPP9905 and AVPP9805. Genotypes were classified into five different classes viz: very highly pungent, highly pungent, moderately pungent, mildly pungent and non-pungent as shown in Table 6. AVPP0705 recorded the highest while AVPP9703, AVPP0512, AVPP0307, AVPP0803 and AVPP0102 were recorded as non-pungent.
Table 6.
The % capsaicin content, Scoville heat units, and degree of pungency of twenty-one chilli pepper genotype samples (dry weight).
| Genotypes | % Capsaicin Content | Scoville Heat Unit | Degree of Pungency |
|---|---|---|---|
| AVPP0705 | 1.49 | 237245 | very highly pungent |
| AVPP0506 | 0.66 | 104888 | very highly pungent |
| AVPP0104 | 0.69 | 110796 | very highly pungent |
| AVPP0002 | 0.65 | 104678 | very highly pungent |
| C05573 | 0.57 | 91097 | very highly pungent |
| AVPP0805 | 0.56 | 88906 | very highly pungent |
| AVPP9905 | 0.30 | 47946 | highly pungent |
| AVPP0904 | 0.30 | 47372 | highly pungent |
| AVPP0514 | 0.28 | 44259 | highly pungent |
| AVPP9805 | 0.22 | 35769 | highly pungent |
| AVPP0702 | 0.14 | 22146 | moderately pungent |
| KULAI | 0.13 | 20564 | moderately pungent |
| AVPP0513 | 0.13 | 20566 | moderately pungent |
| AVPP0116 | 0.04 | 7170 | moderately pungent |
| AVPP0804 | 0.02 | 2767 | mildly pungent |
| AVPP0201 | 0.02 | 3020 | mildly pungent |
| AVPP9703 | 0 | 0 | non-pungent |
| AVPP0512 | 0 | 0 | non-pungent |
| AVPP0307 | 0 | 0 | non-pungent |
| AVPP0803 | 0 | 0 | non-pungent |
| AVPP0102 | 0 | 0 | non-pungent |
n = 2.
2.4. Percentage Capsaicin Content
The number of SHUs of the pepper in isolation is not the critical factor. The most important factor is the capsaicin content. All peppers used in this study, fall outside the pungency limit (0.5%–0.9%) presented by the BPC (British Pharmaceutical Codex) [31] except AVPP0506, AVPP0104, AVPP0002, C05573 and AVPP0805 that fall within the pungency limit (Table 6), hence could be recommended for oleoresin production, which is used in the formulation of certain pharmaceuticals. Despite the fact that AVPP0705 gave the highest capsaicin content, it would not be recommended for pharmaceutical Industry because the percentage capsaicin content is high (1.5%) as there have been no proof it is safe for human use [32,33]. Therefore, on the basis of capsaicin content, only AVPP0506, AVPP0104, AVPP0002, C05573 and AVPP0805 can serve as potential sources of capsaicin for use in the pharmaceutical industry.
3. Experimental
3.1. Instrument and Apparatus
Ultra-fast liquid chromatography was carried out using a Shimadzu Ultra XR (LC- 20AD × R) system (Columbia, SC, USA) equipped with a SPD-M20A prominence Diode Array detector, SK- 20A × R auto sampler and CTO- 20A column oven. Detection was conducting using a UV absorption detector. Identification of capsaicinoids was achieved through comparison of retention times of each capsaicinoids of the chilli samples with those of standard compounds.
3.2. UFLC Analytical Conditions
Column: Purospher® STAR RP-18 e (150 mm × 4.6 mm × 5 µm)
Mobile phase: 1.0% Acetic Acid aq./Acetonitrile = 1/1 (v/v)
Flow rate: 1.2 mL
Column Temp: 30 °C
Detection: SPD-M20 A at 280 nm
Injection Vol.: 2 µL
Data acquisition time: Sampling = 6.25 Hz; Time constant = 0.160 s
3.3. Samples
Twenty one genotypes of chili pepper seeds were collected from AVRDC, Taiwan, and grown under heat condition (Table 7). Whole ripe fruits were harvested and dried for capsaicinoid extraction and analysis. The extraction of capsaicin from the chili pepper samples was done using the method described by [1] and capsaicinoids levels were analyzed using ultra fast liquid chromatography. A sample for assay consisted of 5–8 fruits depending on the size of the chili fruit. The extraction and quantitation was carried out in duplicate for each genotype. The fruits were oven-dried at 60 °C 2–5 days (depending on the fruit size), grounded using laboratory mill. The grounded samples were stored in sealed plastic bags at 20 °C until processed.
Table 7.
Genotypes and their degree of heat tolerance.
| Genotypes | Degree of tolerance * |
|---|---|
| AVPP0705 | Tolerant |
| AVPP0506 | Tolerant |
| AVPP0104 | Moderately Tolerant |
| AVPP0002 | Sensitive |
| C05573 | Tolerant |
| AVPP0805 | Tolerant |
| AVPP9905 | Tolerant |
| AVPP0904 | Tolerant |
| AVPP0514 | Tolerant |
| AVPP9805 | Tolerant |
| AVPP0702 | Tolerant |
| KULAI | Moderately Tolerant |
| AVPP0513 | Tolerant |
| AVPP0116 | Tolerant |
| AVPP0804 | Tolerant |
| AVPP0201 | Tolerant |
| AVPP9703 | Sensitive |
| AVPP0512 | Tolerant |
| AVPP0307 | Tolerant |
| AVPP0803 | Tolerant |
| AVPP0102 | Moderately Tolerant |
* Data not shown.
3.4. Reagents
Analytical grade acetonitrile (99.9%) and methanol (100%) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Glacial acetic acid (99.8%) was from R & M Marketing (Essex, UK). Capsaicin (>95%) and dihydrocapsaicin (~90%) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Stock solution of each capsaicinoids to be determined was prepared by weighing accurately 50 mg and dissolving each compound in 100% methanol. These solutions were stored at 4 °C and used for the preparation of diluted standard solution in methanol.
3.5. Extraction of Capsaicinoids
For the capsaicinoid extraction, a 1:10 (g/mL) ratio of dried chili powder to acetonitrile was placed in 120 mL glass bottles. Bottles were capped and placed in an 80 °C water bath for 4 h; they were swirled manually every hour. Samples were removed from the water bath and cooled to room temperature. Two to 3 mL of supernatant was extracted and filtered (0.45 µm filter on a 5-mL disposable syringe) into a 2-mL glass sample vial, capped and stored at 4 °C until analyses [1]. A 2 µL aliquot was used for each UFLC injection. For each variety, extraction and analysis was carried out in duplicate.
3.6. Conversion to Scoville Heat Units (SHU)
Scoville Heat Units was used to calculate the heat for all samples. Scoville Heat Units are calculated in parts per million of heat (ppmH) based on sample dry weight according to the following formula [32]:
|
ppmH = [Peak area of capsaicin + (0.82) (peak area of dihydrocapsaicin)] (ppm standard) (mL acetonitrile)/(Total capsaicin peak area of standard) (g sample) |
(3) |
Conversion to Scoville Heat Units was made by multiplying ppmH by a factor of 15.
3.7. Percentage Capsaicin Content
The determination of capsaicin content was performed according to the method described by [33]:
| A divided by B times the percentage of pepper = capsaicin Content | (4) |
where A = Scoville Heat Units claimed, B = 16 Million SHUs which is the rating for 100% pure capsaicin and % Pepper = percentage of pepper claimed
3.8. Statistical Analysis
ANOVA for the capsaicin, dihydrocapsaicin, and total capsaicinoid content data for the genotypes was carried out according to the general linear model (GLM), using the SAS software package version 9.2 (SAS Institute Inc., Cary, NC, USA). Means were compared using the LSD test. To estimate the suitability of the qualitative analysis to distinguish degrees of pungency, ANOVA of capsaicin, dihydrocapsaicin, and total capsaicinoid content data for the qualitative categories was carried out. Means were compared using Duncan’s and LSD test.
4. Conclusions
The results from this experiment showed that the UFLC method can be applicable to the chili pepper variety, demonstrating excellent separation without hindrance of any interference. AVPP0705, AVPP0506, AVPP0104, AVPP0002, C05573 and AVPP0805 are the most pungent genotypes among the peppers studied. A few genotypes AVPP9703, AVPP0512, AVPP0307, AVPP0803 and AVPP0102 that recorded 0 SHU (non-detect) where found to be non-pungent. Others fall between moderately and mildly pungent genotypes. This shows that, with exception of AVPP9703, AVPP0512, AVPP0307, AVPP0803 and AVPP0102, all pepper genotypes studied can serve as potential sources of capsaicin. On the other hand, only genotypes AVPP0506, AVPP0104, AVPP0002, C05573 and AVPP0805 would be recommended as potential source of capsaicin for the pharmaceutical industries.
Acknowledgments
The authors are grateful to the Ministry of Education, Malaysia for adequate funding of the research through the Fundamental Research Grant Scheme (FRGS/1/2012/STWN03/UPM/02/2: 07-01-13-1240FR). Also, we are grateful to Haris Ahmad who helped run the UFLC system and with the data analysis.
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/1420-3049/19/5/6474/s1.
Author Contributions
All authors contributed equally to this work.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Sample Availability: Samples of the compounds are available from the authors.
References
- 1.Collins M.D., Mayer-Wasmund L., Bosland P.W. Improved method for quantifying capsaicinoids in Capsicum using high performance liquid chromatography. HortScience. 1995;30:137–139. [Google Scholar]
- 2.Peña-Alvarez A., Ramírez-Maya E., Alvarado-Suárez L.A. Analysis of capsaicin and dihydrocapsaicin in peppers and pepper sauces by solid phase microextraction–gas chromatography–mass spectrometry. J. Chromatogr. A. 2009;1216:2843–2847. doi: 10.1016/j.chroma.2008.10.053. [DOI] [PubMed] [Google Scholar]
- 3.Caterina M.J., Schumacher M.A., Tominaga M., Rosen T.A., Levine J.D., Julius D. The capsaicin receptor: A heatactivated ion channel in the pain pathway. Nature. 1997;389:816–824. doi: 10.1038/39807. [DOI] [PubMed] [Google Scholar]
- 4.Caterina M.J., Leffler A., Malmberg A.B., Marti W.J., Trafton J., Petersen-Zeitz K.R., Koltzenburg M., asbaum A.I., Julius D. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science. 2000;288:306–313. doi: 10.1126/science.288.5464.306. [DOI] [PubMed] [Google Scholar]
- 5.Chu C.J., Huang S.M., de Petrocellis L., Bisogno T., Ewing S.A., Miller J.D., Zipkin R.E., Daddario N., Appendino G., Di Marzo V., et al. N-Oleoyldopamine, a novel endogenous capsaicin-like lipid that produces hyperalgesia. J. Biol. Chem. 2003;278:13633–13639. doi: 10.1074/jbc.M211231200. [DOI] [PubMed] [Google Scholar]
- 6.Lee R.J., Yolton R.L., Yolton D.P., Schnider C., Janin M.L. Personal defense sprays: Effects and management of exposure. J. Am. Optom. Assoc. 1996;67:548–560. [PubMed] [Google Scholar]
- 7.Iwai K., Suzuki T., Fujiwake H. Formation and accumulation of pungent principle of hot pepper fruits, capsaicin, and its analogues, in Capsicum annuum var. annuum cv. Karayatsubusa at different stages of flowering. Agric. Biol. Chem. 1979;43:2493–2498. doi: 10.1271/bbb1961.43.2493. [DOI] [Google Scholar]
- 8.Iida T., Moriyama T., Kobata K. TRPV1 activation and induction of nociceptive response by a non-pungent capsaicin-like compound, capsiate. Neuropharmacology. 2003;44:958–967. doi: 10.1016/s0028-3908(03)00100-x. [DOI] [PubMed] [Google Scholar]
- 9.Backonja M.M., Malan T.P., Vanhove G.F., Tobias J.K. NGX-4010, a high-concentration capsaicin patch, for the treatment of postherpetic neuralgia: A randomized, double-blind, controlled study with an open-label extension. Pain Med. 2010;11:600–608. doi: 10.1111/j.1526-4637.2009.00793.x. [DOI] [PubMed] [Google Scholar]
- 10.Tesfaye S. Advances in the management of diabetic peripheral neuropathy. Curr. Opin. Support. Palliat. Care. 2009;3:136–143. doi: 10.1097/SPC.0b013e32832b7df5. [DOI] [PubMed] [Google Scholar]
- 11.Derry S., Lloyd R., Moore R.A., McQuay H.J. Topical capsaicin for chronic neuropathic pain in adults. Cochrane Database Syst. Rev. 2009;7:CD007393. doi: 10.1002/14651858.CD007393.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Reyes-Escogido M.L., Gonzalez-Mondragon E.G., Vazquez-Tzompantzi E. Chemical and Pharmacological Aspects of Capsaicin. Molecules. 2011;16:1253–1270. doi: 10.3390/molecules16021253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.DeWitt D., Bosland P.W. The Pepper Garden. Ten Speed Press; Berkeley, CA, USA: 1993. [Google Scholar]
- 14.Contreras-Padilla M., Yahia E.M. Changes in capsaicinoids during development, maturation, and senescence of chili peppers and relation with peroxidase activity. J. Agric. Food Chem. 1998;46:2075–2079. [Google Scholar]
- 15.Estrada B., Bernal M.A., Diaz J., Pomar F., Merino F. Fruit development in Capsicum annuum: Changes in capsaicin, lignin, free phenolics, and peroxidase patterns. J. Agric. Food Chem. 2000;48:6234–6239. doi: 10.1021/jf000190x. [DOI] [PubMed] [Google Scholar]
- 16.Estrada B., Bernal M.A., Diaz J., Pomar F., Merino F. Capsaicinoids in vegetative organs of Capsicum annuum L. in relation to fruiting. J. Agric. Food Chem. 2002;50:1188–1191. doi: 10.1021/jf011270j. [DOI] [PubMed] [Google Scholar]
- 17.Harvell K., Bosland P.W. The environment produces a significant effect on the pungency of chilis. HortScience. 1997;32:1292. [Google Scholar]
- 18.Estrada B., Diaz J., Merino F., Bernal M.A. The effect of seasonal changes on the pungency level of Padron pepper fruits. Capsicum Eggplant Newsl. 1999;18:28–31. [Google Scholar]
- 19.Garces-Claver A., Arnedo-Andre’s M.S., Abadia J., Gil-Ortega R., Alvarez-Fernandez A. Determination of capsaicin and dihydrocapsaicin in Capsicum fruits by liquid chromatographyelectrospray/time-of-flight mass spectrometry. J. Agric. Food Chem. 2006;54:9303–9311. doi: 10.1021/jf0620261. [DOI] [PubMed] [Google Scholar]
- 20.Reilly C.A., Crouch D.J., Yost G.S., Fatah A.A. Determination of capsaicin, nonivamide, and dihydrocapsaicin in blood and tissue by liquid chromatography-tandem mass spectrometry. J. Anal. Toxicol. 2002;26:313–319. doi: 10.1093/jat/26.6.313. [DOI] [PubMed] [Google Scholar]
- 21.Thompson R.Q., Phinney K.W., Welch M.J., White V.E. Quantitative determination of capsaicinoids by liquid chromatography-electrospray mass spectrometry. Anal. Bioanal. Chem. 2005;381:1441–1451. doi: 10.1007/s00216-005-3102-y. [DOI] [PubMed] [Google Scholar]
- 22.Schweiggert U., Carle R., Schieber A. Characterization of major and minor capsaicinoids and related compounds in chili pods (Capsicum frutescens L.) by high-performance liquid chromatography-atmospheric pressure chemical ionization mass spectrometry. Anal. Chim. Acta. 2006;557:236–244. [Google Scholar]
- 23.Scoville W.L. Note on Capsicum. J. Am. Pharm. Assoc. 1912;1:453–454. [Google Scholar]
- 24.Weiss E.A. Spice Crops. CABI Publishing International; New York, NY, USA: 2002. p. 411. [Google Scholar]
- 25.George W.L. Official Methods of Analysis of AOAC International. [(accessed on 20 January 2014)]. Available onlin: http://www.aoac.org/iMIS15_Prod/AOAC/Publications/Official_Methods_of_Analysis.html.
- 26.Ha J., Seo H.Y., Shim Y.S., Seo D.W., Seog H., Ito M., Nakagawa H. Determination of capsaicinoids in foods using ultra high performance liquid chromatography. Food Sci. Biotechnol. 2010;19:1005–1009. [Google Scholar]
- 27.Nwokem C.O., Agbaji E.B., Kagbu J.A., Ekanem E.J. Determination of capsaicin content and pungency level of five different peppers grown in Nigeria. NY Sci. J. 2010;3:17–21. [Google Scholar]
- 28.Othman Z.A.A., Ahmed Y.B.H., Habila M.A., Ghafar A.A. Determination of capsaicin and Dihydrocapsaicin in Capsicum Fruit samples using High Performance Liquid Chromatography. Molecules. 2011;16:8919–8929. doi: 10.3390/molecules16108919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sanatombi K., Sharma G.J. Capsaicin content and pungency of different Capsicum spp. cultivars. Not. Bot. Horti. Agrobot. Cluj. Napoca. 2008;36:89–90. [Google Scholar]
- 30.Mathur R., Dangi R.S., Das S.C., Malhotra R.C. The hottest chilli variety inIndia. Curr. Sci. India. 2000;79:287–288. [Google Scholar]
- 31.Pharmaceutical Society of Great Britain . British Pharmaceutical Codex. The Pharmaceutical Press; London, UK: 1973. [Google Scholar]
- 32.American Spice Trade Association . Official Analytical Methods of the American Spice Trade Association. 3rd ed. American Spice Trade Association; Englewood Cliffs, NJ, USA: 2004. [Google Scholar]
- 33.Pepper Enforcement. [(accessed on 20 January 2014)]. Available online: http://www.pepperenforcement.com/capsaicin.html.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.