Abstract
In South America, the mesocarp flour of Prosopis species plays a prominent role as a food resource in arid areas. The aim of this work was the characterization of the phenolic antioxidants occurring in the pod mesocarp flour of Chilean Prosopis. Samples were collected in the Copiapo, Huasco and Elqui valleys from the north of Chile. The samples of P. chilensis flour exhibited a total phenolic content ranging between 0.82–2.57 g gallic acid equivalents/100 g fresh flour weight. The highest antioxidant activity, measured by the DPPH assay, was observed for samples from the Huasco valley. HPLC-MS/MS analysis allowed the tentative identification of eight anthocyanins and 13 phenolic compounds including flavonol glycosides, C-glycosyl flavones and ellagic acid derivatives. The antioxidant activity and the phenolic composition in the flour suggest that this ancient South American resource may have potential as a functional food.
Keywords: Prosopis chilensis, mesocarp flour, flavonoids, antioxidant, traditional food
1. Introduction
Worldwide climate change and the long-lasting drought in northern Chile suggest that attention should be paid to native species that are adapted to arid environments. In arid and semi-arid lands, pods of the trees belonging to genus Prosopis, locally known as “algarrobo” in South America, are relevant food sources [1]. They were gathered by all the pre-Columbian human groups, including those living in the south of United States of America [2], Amerindians in the Paraguayan Chaco [3,4], Argentina [5], and Chile [6,7]. Prosopis pods constitute a food source for humans and animals [8]. Different food products are prepared from Prosopis, including flour, sweets, syrup or fermented alcoholic beverages [6,8]. After the European conquest and introduction of new crops, “algarrobo” pods were used to feed cattle and sometimes used in the local cuisine. The traditional use remained in rural areas and in the Chaco phytogeographical region of South America [3,4]. The pod flour is used to prepare a kind of bread, known as “patay” in Argentina [9] or is fermented into various alcoholic beverages (“añapa”, “aloja” and “chicha”) [10].
In northern Chile and Peru, “algarrobo” pods were an important food source in pre-Hispanic times and they can be found in archeological sites and in burials [11]. In Chile, the commercialized “algarrobo” flour is mainly imported from Peru by small local businessmen. The total production of “algarrobo” flour in Peru was estimated in 1422 tons in 2011 [12]. According to Soto and Gysling [13] the cultivated area of “algarrobo” trees in Peru comprises 5140 ha, mostly located in the Tarapaca Region (3246 ha), close to the border with Chile. Data about “algarrobo” flour production in Chile is not available. The Prosopis pods mesocarp flour was investigated for its food applications [14] and has been used to prepare cookies and fried chips [10]. The Prosopis flour potential as food was revised by Felker et al. [15] and by Cardozo et al. [9].
Chilean and Argentinean Prosopis species were investigated for their alkaloid content and composition, as well as for some biological activities, including enzyme inhibition, antioxidant effect and DNA binding [9,16,17,18]. The alkaloids isolated so far occur mainly in the leaves (folioles) while the pods contained large amounts of the amino acid proline, as should be expected for plants growing in soils affected by drought and salinity [19,20,21]. Prosopis pods were also analysed in a study on the proximate composition and bioactivity of food plants consumed by Chilean Amerindians [7]. Despite its long tradition of use as food and the potential of local resources for cuisine, there is little information on the phenolic compounds that can occur in the Chilean “algarrobo” pods mesocarp flour. The Argentinean “algarrobo” pods meal [5,9] as well as the seed flour [22] was investigated. The phenolic from “algarrobo” pods syrup was described by Quispe et al. [23]. Phenolics occurring in plant foods, grains and flour are dietary constituents that have been shown to present relevant biological activities. The pod flour of the Argentinian Prosopis alba and P. nigra contains antioxidant and anti-inflammatory agents [5], are not genotoxic and can be considered safe for human consumption [9]. It has been shown that the wheat flour contains antioxidant and antiproliferative phenolics [24] while beans phenolics shows antioxidant effects and are inhibitors of the enzymes α-amylase and α-glucosidase [25]. Lentil phenolics are not only antioxidant, but also inhibit α-glucosidase and pancreatic lipase [26]. Methanolic extracts from raw and processed Kenyan native food ingredients disclosed the antioxidant and hypoglycemic potential of their phenolic constituents [27]. In continuation of our studies on native South American food resources, we now report the naturally occurring phenolic compounds and antioxidant activity of Chilean Prosopis pods mesocarp flour.
2. Results and Discussion
2.1. Prosopis Flour Characterization and Antioxidant Activity
Six “algarrobo” pods samples were collected from the longitudinal valleys of Huasco, Elqui and Copiapó in northern Chile (Figure 1). The morphological variation of the samples is shown in Figure 2. The flour yield, phenolic and flavonoid content and antioxidant activity of the flour extracts were determined and are summarized in Table 1. The Prosopis flours presented different hues according to the colour of the pods. Pods from Elqui valley showed sticky granules producing tacky flours when ground. The percent of mesocarp flour yield ranged from 36.7% flour to pod ratio for the deep purple Puquio sample, 31.8% for the Elqui valley and 3.3%–17.8% for the beige pods of P. chilensis collected in the Huasco valley, respectively. The sample from El Transito, Huasco Valley, presented a very low flour to pod ratio (3.3%). With thin pods, this sample is considered inedible by the local population [3,4,8].
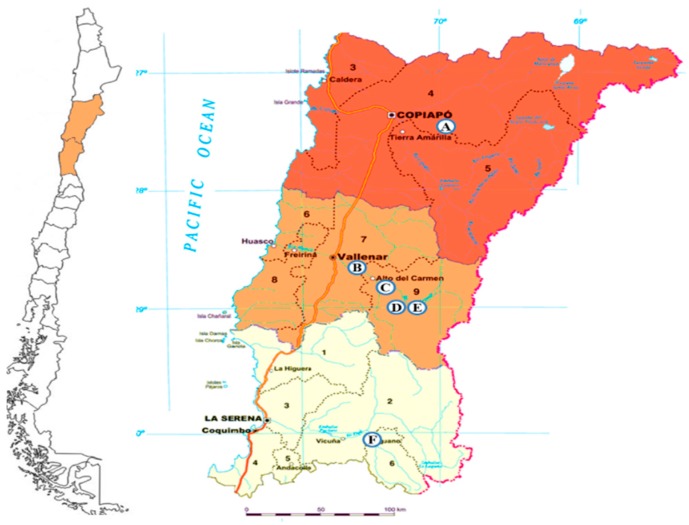
Figure 1.
Map of Chile showing the collection places of Prosopis pods. Copiapo valley: Puquio (A); Huasco valley: Alto del Carmen (B), El Transito (C); Pinte (D) and Plaza de Pinte (E); Elqui valley (F).
Figure 2.
Chilean Prosopis pods showing morphological variation according to collection place. Puquio (a); Alto del Carmen (b), El Transito (c); Pinte (d); Plaza de Pinte (e); Elqui valley (f).
Table 1.
Percent flour yield from pods, total phenolic (TP) and flavonoid (TF) content in flour weight (FW) and antioxidant activity of phenolic enriched methanol flour extract (PEFE) of Chilean Prosopis mesocarp flour.
| Sample Origin | % Flour to Pod Ratio | Flour Color | TP(g GAE/100 g FW) | TF(g QE/100 g FW) | % XAD-Retained PEFE | DPPH SC50 (µg PEFE/mL) | FRAP (mMoles TE/g PEFE) | TEAC (μM TE/g PEFE) |
|---|---|---|---|---|---|---|---|---|
| Copiapó valley | ||||||||
| Puquio | 36.7 | Light greyish tan | 2.54 ± 0.12 | n.d. a | 4.26 | 70.51 | 0.50 ± 0.01 | 267.5 |
| Huasco valley | ||||||||
| Alto del Carmen | 15.4 | Light tan | 2.57 ± 0.09 | 0.38 ± 0.07 | 0.19 | 12.07 | 3.45 ± 0.06 | 3206.6 |
| El Tránsito | 3.3 | Light tan | 1.33 ± 0.06 | 0.25 ± 0.01 | 1.63 | 52.85 | 0.65 ± 0.04 | 428.6 |
| Pinte | 15.5 | Light tan | 0.82 ± 0.03 | 0.17 ± 0.01 | 0.91 | 52.97 | 0.63 ± 0.06 | 530.5 |
| Plaza de Pinte | 17.8 | Light tan | 2.11 ± 0.10 | 0.56 ± 0.10 | 0.10 | 23.74 | 1.21 ± 0.08 | Inactive |
| Elqui valley | ||||||||
| Elqui valley | 31.8 | Pale tan | 0.89 ± 0.11 | 0.23 ± 0.03 | 2.08 | >100 | 0.36 ± 0.01 | Inactive |
| Quercetin b | 7.82 ± 0.30 | 10.77 ± 0.16 | 8157.9 |
Abbreviations. FW: Fresh flour weight; PEFE: phenolic.enriched MeOH flour extract; TP: total phenolic content; TF: total flavonoid content; Antioxidant activity: DPPH (discoloration of the free radical 1,1-diphenyl-2-picrylhydrazyl, SC50 in µg PEFE/mL), FRAP (Ferric Reducing Antioxidant Power, mMol TE/g PEFE), TEAC (Trolox-Equivalent Antioxidant Capacity, μM TE/g PEFE) were carried out in triplicate and results are expressed as mean values ± SD. a below quantification level. b Quercetin at 30 µg/mL for FRAP assay.
The range of phenolic compounds (TP) in flours was 0.82–2.57 g GAE per 100 g FFW. The higher values were from Alto del Carmen (2.75 g GAE/100 g FFW), Puquio (2.54 g GAE/100 g FFW) and Plaza de Pinte (2.11 g GAE/100 g FFW). Total flavonoid (TF) content in flour was low (0.17–0.56 g QE/100 g flour weight), and no correlation was observed between the TP and TF content. As TP and TF of the flour was low, samples were enriched in phenolics for antioxidant activity studies and phenolic profiling. The flour samples were extracted with MeOH and phenolics were retained on Amberlite XAD-7 to obtain the phenolic-enriched flour extract (PEFE). The highest PEFE was from the Copiapo Valley sample (4.26%). Lower PEFE values for the different samples of P. chilensis ranged from 0.10% to 2.08% for the Huasco and Elqui Valley samples (Table 1).
The best antioxidant activity of the PEFE, measured by the DPPH discoloration assay, was found in the Huasco Valley samples: Alto del Carmen and Plaza de Pinte (SC50 12.07 and 23.74 µg/mL, respectively). The same samples presented the highest activity in the FRAP assay, with values of 3.45 and 1.21 mM TE/g PEFE, respectively. In addition, the highest TEAC value was observed for the Alto del Carmen sample with a 3206.61 µM TE/g PEFE. There was a statistical correlation between the TP content and FRAP (r = 0.565; p < 0.05). Large variation in mesocarp flour yield and phenolic content was observed for P. chilensis. The close related species P. alba and P. nigra are common in the Chaco zone of South America and occur in the eastern Andean ranges of Argentina.
In a study by Cardozo et al. [9], TP in ethanolic extracts of P. alba and P. nigra pods flour were 0.18 and 0.19 g/100 g dry weight (DW), with flavonoids accounting for 0.01 and 0.06 g/100 g DW, respectively. When the flour was extracted with water, the TP values increased to 0.40 and 0.41 g/100 g DW and TF to 0.03 and 0.13 g/100 g DW for P. alba and P. nigra, respectively. The TP (0.82–2.57 g GAE/100 g flour) and TF content (0.17–0.56 g QE/100 flour) of the Chilean Prosopis flour samples was higher than that of the Argentinian species.
In a review on pod mesocarp flour of Prosopis species, Felker et al. [14] refer to free phenolic concentrations of 0.18 and 0.41 GAE/100 g DW for P. alba and P. nigra flour, respectively. According to the same author, TP for wheat bran ranges between 0.126–0.316 g GAE/100g DW or 0.27–0.35 g GAE/100 g DW while white wheat flour contains 0.0044–0.014 g GAE/100g DW [14].
2.2. HPLC-DAD-MS/MS Analysis
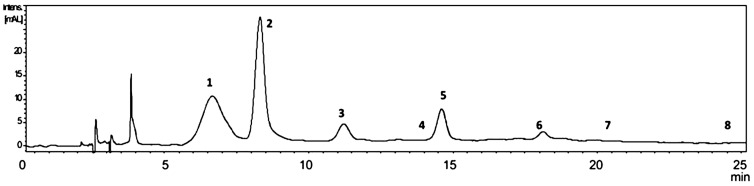
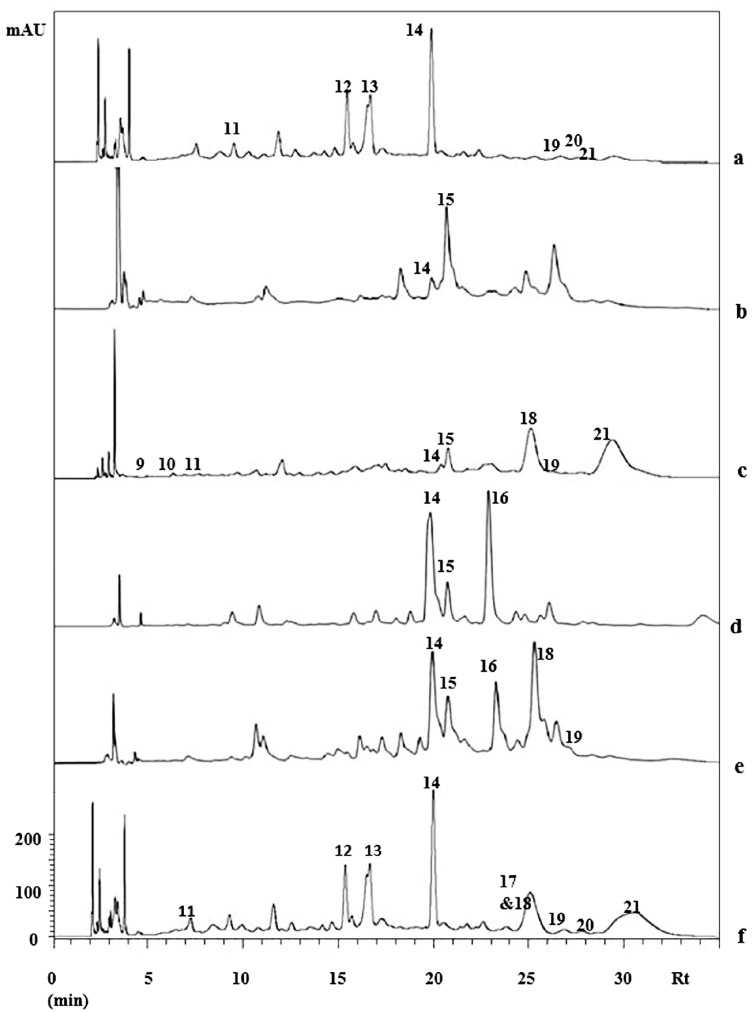
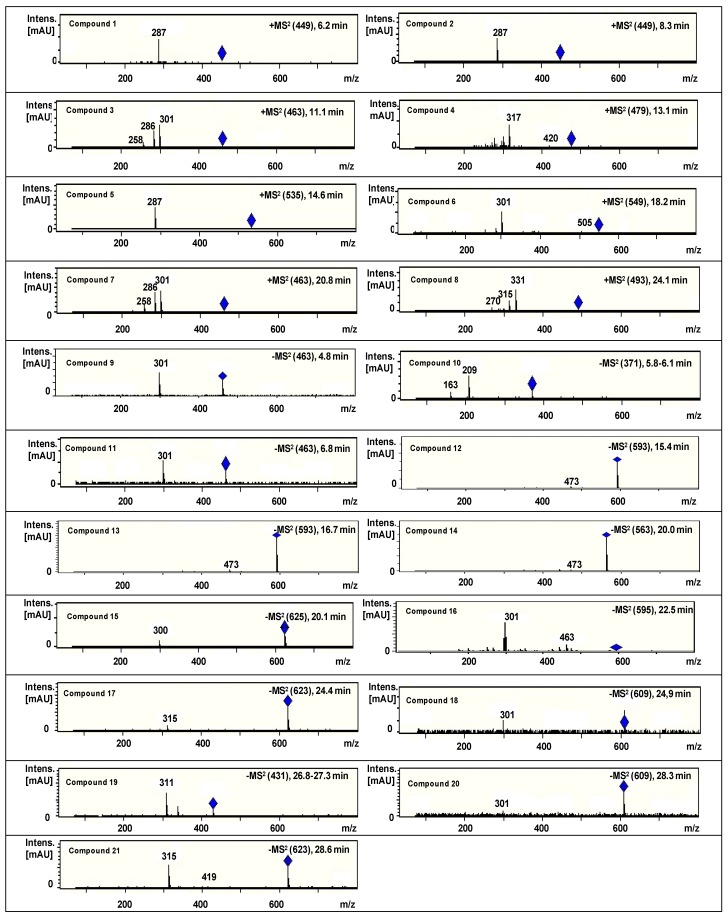
The composition of PEFE was assessed by HPLC-DAD-MS/MSn. Anthocyanins were detected in the positive ion mode, while other phenolic compounds were analyzed in the negative ion mode. The representative HPLC-DAD chromatogram of PEFE from the Puquio sample is presented in Figure 3. The HPLC-DAD chromatograms of other samples are shown in Figure 4. Extracted ion MS2 spectra of compounds 1–21 are presented in Figure 5. The tentative identification of anthocyanins (A) and phenolic compounds (B) is presented in Table 2.
Figure 3.
HPLC-DAD chromatogram of the phenolic enriched flour extract (PEFE) of Puquio showing the anthocyanins occurring in the sample. Detection: 535 nm. Compounds: 1: Cyanidin 3-hexoside; 2: Cyanidin 3-hexoside; 3: Peonidin 3-hexoside; 4: Petunidin hexoside; 5: Cyanidin malonyl hexoside; 6: Peonidin malonyl hexoside; 7: Peonidin 3-hexoside; 8: Malvidin hexoside.
Figure 4.
HPLC-DAD chromatogram of the Amberlite-retained fraction from the methanolic extract from “algarrobo” mesocarp flour. a: Puquio; b: Alto del Carmen; c: El Transito; d: Pinte; e: Plaza de Pinte; f: Elqui valley. Detection: UV, 254 nm. Compounds: 9: Ellagic acid (EA) hexoside; 10: Hydroxyferuloyl hexoside; 11: EA hexoside; 12: Vicenin II/Isomer; 13: Vicenin II/Isomer; 14: Schaftoside/isoschaftoside; 15: Quercetin (Q) dihexoside; 16: Q-hexosidepentoside; 17: Q-methyl ether rhamnoside hexoside; 18: Q-rutinoside; 19: Isovitexin; 20: Q-rhamnoside-hexoside; 21: Q-methyl ether rhamnoside hexoside.
Figure 5.
Extracted ion MS2 spectra of compounds 1–21 tentatively identified in the phenolic-enriched flour extract of Chilean Prosopis pods.
Table 2.
Tentative identification of anthocyanins and phenolics in phenolic-enriched Prosopis pod mesocarp flour extracts by HPLC-DAD-ESI-MS data. Anthocyanins (A) were detected in Puquio sample. Phenolics different than anthocyanins (B) were found in all phenolic-enriched pod mesocarp flour extracts.
A: Anthocyanins in Prosopis pods meal (Puquio sample), detection at 535 nm.
| Compound | Rt (min) | UV λmax (nm) | [M]+ (m/z) | MS/MS (m/z) | Tentative Identification |
|---|---|---|---|---|---|
| 1 | 6.2 | 516, 425 sh, 279, 224 | 449 | 287 | Cyanidin 3-hexoside |
| 2 | 8.3 | 516, 425 sh, 279, 224 | 449 | 287 | Cyanidin 3-hexoside |
| 3 | 11.1 | 517, 321 sh, 278, 225 | 463 | 301,286,258 | Peonidin 3-hexoside |
| 4 | 13.1 | - | 479 | 420,317 | Petunidin hexoside |
| 5 | 14.6 | 517, 307sh, 285, 228 | 535 | 287 | Cyanidin malonyl hexoside |
| 6 | 18.2 | 517, 320 sh, 279, 228 | 549 | 505,301 | Peonidin malonyl hexoside |
| 7 | 20.8 | 520, 319 sh, 270, 229 | 463 | 301,286,258 | Peonidin 3-hexoside |
| 8 | 24.1 | - | 493 | 331,315,270 | Malvidin hexoside |
B: Phenolics in phenolic-enriched Prosopis pod mesocarp flour extracts, detection at 254 nm.
| Compound | Rt (min) | UV λmax (nm) | MW | [M-H] ‾ and Fragment Ions | Tentative Identification |
|---|---|---|---|---|---|
| 9 | 4.8 | - | 464 | 463, 301 | Ellagic acid hexoside |
| 10 | 5.8–6.1 | 289, 228 | 372 | 371, 209, 163 | Hydroxyferulic acid hexoside |
| 11 | 6.8 | 278 | 464 | 463, 301 | Ellagic acid hexoside |
| 12 | 15.4 | 334, 270 | 594 | 593, 473 | Vicenin II/Isomer |
| 13 | 16.7 | 334, 271 | 594 | 593, 473 | Vicenin II/Isomer |
| 14 | 20.0 | 333, 270 | 564 | 563, 503, 473, 443, 383 | Schaftoside/isoschaftoside |
| 15 | 20.1 | 352, 262 | 626 | 625, 300 | Q-dihexoside |
| 16 | 22.5 | 352, 267 sh, 256 | 596 | 595, 463, 301 | Q-hexosidepentoside |
| 17 | 24.4 | 354, 268 sh, 254 | 624 | 623, 315 | Q-methyl ether rhamnoside hexoside |
| 18 | 24.9 | 355, 297sh, 267sh, 254 | 610 | 609, 301 | Q-rutinoside (rhamnoside hexoside) |
| 19 | 26.8–27.3 | - | 432 | 431, 311 | Isovitexin |
| 20 | 28.3 | 352, 265 sh, 254 | 610 | 609, 301 | Q-rhamnoside-hexoside |
| 21 | 28.6 | 349, 267 sh, 253 | 624 | 623, 419, 315 | Q-methyl ether rhamnoside hexoside |
The visible spectra of compounds 1–3, 5–7, as detected in the DAD, show maxima in the 516–520 nm range, in agreement with anthocyanidins bearing a substituent at the 3-position [28,29]. According to Ververidis et al. [30] 3-glycosylation of the anthocyanidins confers stability to the pigments, being the most common substitution pattern for this group of compounds. MS/MS experiments show loss of 162 atomic mass units (amu) for compounds 1–8, leading to a m/z ion at 287 amu for compounds 1, 2 and 5, m/z 301 for 3, 6 and 7, m/z 317 for 4 and 331 for 8. This is consistent with the occurrence of cyanidin, peonidin, petunidin and malvidin hexosides in the sample. The main compound was cyanidin 3-hexoside. A malonoyl unit was detected in compounds 5 and 6, assigned as cyanidin malonyl hexoside and peonidin malonyl hexoside, respectively.
These anthocyanins have not been previously reported from Prosopis chilensis and are responsible, at least in part, of the dark purple colour of the pods. Compounds 9 and 11 presented the same pseudo molecular ion at 463 amu and loss of a hexose, leading to the ion at m/z 301. The elution time and UV spectrum of 11 is in agreement with a phenolic compound, consistent with ellagic acid. Both compounds differ in the identity of the hexose and were tentatively identified as ellagic acid hexosides. The compound 10 showed in the MS/MS spectrum the loss of a hexoside, leading to the aglycone at m/z 209, in agreement with hydroxyferulic acid hexoside [31].
The UV spectra of compounds 15–18, 20 and 21 show maxima in the range 349–354 nm, in agreement with the Band I of flavonol substituted at the 3-O-position. MS/MS analysis showed the loss of two 162 amu fragments (hexose) for compounds 15 and one hexose and a pentose for 16.
Consecutive losses of rhamnose and hexose or rutinoside were observed for 17, 18, 20 and 21, leading to aglycones with m/z 301 or 315, in agreement with quercetin (15, 16, 18 and 20) and quercetin methyl ether (17 and 21). The compounds were identified as quercetin glycosides 15, 16, 18 and 20 and quercetin methyl ether glycosides 17 and 21. Flavone C-glycosides show characteristic UV maxima around 330 nm (Band 1) and losses of 120 amu in the MS/MS spectra [32]. Compounds 12–14 and 19 presented UV and mass spectra in agreement with this group of compounds and were assigned as vicenin II isomers (12 and 13), schaftoside or isoschaftoside (14) and isovitexin (19). The C-glycosyl flavones schaftoside, isoschaftoside and vicenin II were recently identified as constituents of Prosopis pods syrup [23].
HPLC-DAD-MS/MS analysis of the PEFE allowed the tentative identification of eight anthocyanins, two ellagic acid glycosides, six flavonol O-glycosides, four flavone C-glycosides and a hydroxyferulic acid hexoside. Anthocyanins were detected only in the sample from the Copiapo Valley (Puquio). The compounds were cyanidin, peonidin, petunidin and malvidin derivatives, suggesting similarity with P. nigra [5]. Anthocyanins have been identified in several crops, including black beans [33], black rice [34], and purple corn [35]. The anthocyanins delphinidin 3-glucoside, petunidin 3-glucoside and malvidin 3-glucoside were reported for black bean [33] and cyanidin-3-O-sambubioside, the 3-O-glucosides of delphinidin, cyanidin, pelargonidin and peonidin were described from the immature purple pods of yard-long beans [36]. Despite their widespread distribution in Nature, anthocyanins have not been reported from South American Prosopis pods until a recent study on P. alba and P. nigra mesocarp flour [5]. In the present work, anthocyanins were identified in the Prosopis sample collected in Puquio. This place was a natural corridor that allowed crossing the Andes from the Copiapo Valley to the Argentinian provinces of Catamarca, La Rioja and Tucuman through the Paso San Francisco. As this was also a pre-Columbian route to cross the Andes and there is no previous records of P. nigra in Chile, we cannot rule out three possibilities: (i) that the distribution range of the species included part of the western Andean valleys; (ii) that the species was introduced in Chile by migrants or, (iii) that the sample is a high anthocyanin variety of P. chilensis. The pods as well as the starch from P. chilensis and P. flexuosa are frequently found in archeological remains in the dry valleys of Catamarca, Argentina [11].
Main constituents in the PEFE were flavonoids. Different patterns of phenolic compounds other than anthocyanins were observed in the chromatograms of the samples (Figure 4). Similar constituents are present in the Puquio and Elqui samples with the C-glycosylflavones 12, 13 and 14 as the main compounds. In the Pinte, Plaza de Pinte and Alto del Carmen flours (Figure 4), the C-glycoside 14 occurs with the quercetin glycoside 15, but in different ratios. The Plaza de Pinte sample showed a more complex composition, with additional quercetin glycosides. The sample from El Transito presented a higher proportion of 18 and the flavonol glycoside 21. Quercetin glycosides and flavone C-glycosides are observed in the PEFE. The chemistry and morphology of the Prosopis sample from the Copiapo valley suggest a species different than P. chilensis.
The lactone 5,6-dihydro-6-propyl-2H-pyran-2-one was identified as the major volatile from Prosopis flour [37].The alkaloids phenethylamine and tryptamine were isolated from P. chilensis pods [6]. In the present work, only phenolics were identified in the PEFE. The phenolics from the pods mesocarp meal were compared with those identified in the traditional syrup “algarrobina” [23]. Common constituents were flavone C-glycosides such as vicenin II or isomer, schaftoside/ isoschaftoside and quercetin glycosides, suggesting a common trend in the phenolic compounds composition of Prosopis pods-derived products. The total flavonoid C-glycosides from Abrus mollis extract, containing mainly vicenin-II, isoschaftoside and schaftoside was shown to display strong anti-inflammatory and hepatoprotective effect in mice [38]. The C-glycoside mixture also presented antioxidant and protective effect on lipopolysaccharide-induced lipotoxicity in mice [39]. The same compounds occur in the phenolic-enriched fraction of the Prosopis flour. Vicenin-II shows anti-glycation activity [40] and is active against prostate cancer cells [41]. The structural features of flavonoids associated with antioxidant effect are known [42]. Tsuchiya [43] reported the structural relationship between flavonoids and cell-mimetic membranes and its relation with antioxidant and anti-proliferative effect on cells. However, increasing evidence indicates that the in vivo bioactive flavonoids are not necessarily the naturally occurring compounds [44]. The O-flavonoid glycosides are hydrolyzed and the aglycones are transformed into glucuronides, O-methylated derivatives and sulphates and are further degraded by the gut microflora [44].
The phenolics identified in the flour are compounds with known antioxidant properties and display also other biological effects that might have a positive impact in the health of consumers. More research work should be undertaken to fully disclose the potential of this ancient South American resource as functional food.
3. Experimental Section
3.1. Chemicals
Folin-Ciocalteau phenol reagent, 2,4,6-tri(2-pyridyl)1,3,5-triazine (TPTZ) sodium acetate, 1,1-diphenyl-2-picrylhydrazyl radical (DPPH), quercetin, gallic acid and AlCl3 were purchased from Sigma-Aldrich (St. Louis, MO, USA). 2,2’-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid (ABTS) diammonium salt, 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), potassium persulfate, sodium carbonate, FeCl3·6H2O, HPLC-grade methanol, acetonitrile and formic acid were purchased from Merck (Darmstadt, Germany). Ultrapure water was obtained using a BarnstedEasyPure water filter (Thermo Scientific, Marietta, OH, USA).
3.2. Plant Material and Sample Preparation
Algarrobo pods were collected in the Copiapo, Huasco and Elqui valleys in February 2013 (Figure 1). The samples were classified as Prosopis chilensis by Dr. Patricio Peñailillo and voucher specimen were deposited at the Herbario de la Universidad de Talca. The collection places were as follows: Copiapo Valley: road to Paso San Francisco, near Puquio (27°08′55′′S, 69°52′24′′W); Huasco Valley: Alto del Carmen (28°44′50′′S, 70°29′57′′W), El Transito (28°51′04′′S, 70°18′33′′W); road to Pinte (28°57′47′′S, 70°16′54′′W) and Plaza de Pinte (28°58′45′′S, 70°16′55′′W); Elqui Valley: 30°06′39′′S, 70°29′58′′W. Samples were transported to the lab and kept at room temperature, according to the traditional storage indications. The air-dried pods were processed in a grinder to separate the seeds from the mesocarp flour. The traditional flour preparation was followed, using a mortar and pestle. The flour was sieved and weighed to establish the pod/mesocarp flour ratio. Pods flour was extracted with MeOH under sonication (2 × 3 min each time), in 1:10 flour to MeOH w/v ratio. The MeOH solution was filtered and taken to dryness under reduced pressure to afford the crude MeOH extract. The extracts were dissolved in water, filtered and adsorbed into Amberlite XAD-7, pre-treated as described in Jiménez-Aspee et al. [45]. Phenolic compounds were desorbed from the resin using MeOH and MeOH:H2O 7:3 (v/v) and the combined extracts of each sample were taken to dryness and lyophilized. The phenolic-enriched flour extracts (PEFE) were concentrated under reduced pressure and lyophilized for its analysis.
3.3. Total Phenolic (TP) and Total Flavonoid (TF) Contents
The total phenolic (TP) and total flavonoid (TF) content was determined in the flour MeOH extract as described by Jiménez-Aspee et al. [45], with slight modifications. Stock solutions (2 mg/mL) were prepared in MeOH:H2O (1:1). For TP, the Folin-Ciocalteu method was followed. The results are expressed as g gallic acid equivalents (GAE)/100 g fresh flour weight (FFW). For TF, the AlCl3 methodology was used. TF was expressed as g quercetin equivalents (QE)/100 g FFW. Absorbance of each solution were measured by spectrophotometer (Thermo Spectronic Helios Alfa, Cambridge, UK) at 725 and 510 nm, respectively, after 15 min of incubation at room temperature.
3.4. Antioxidant Activity
The antioxidant activity of the samples was determined by three assays, as described [45,46]. PEFEs were dissolved in 50% v/v aqueous methanol at a final concentration of 300 μg/mL. Stock solutions were filtered and kept in the dark, and all analyses were performed on the same day.
DPPH discoloration assay was carried out with final concentrations of 100, 33 and 11 μg/mL. The DPPH solution was freshly prepared in methanol (20 mg/L) and mixed with the extract at the above given concentrations. Absorbance was measured at 517 nm in a universal microplate reader (Biotek Instruments Inc., ELx 800, Winooski, VT, USA). SC50 values (μg/mL), corresponding to the amount of extract that scavenges the radical concentration by 50%, were calculated using the OriginPro 8.0 software (OriginLab Corporation, Northampton, MA, USA).
For the ferric reducing antioxidant power (FRAP) assay, a 300 μg/mL extract aliquot was mixed with warm FRAP solution and left to stand in the dark for 30 min. Absorbance was read at 593 nm using a spectrophotometer (Thermo Spectronic Helios Alfa) Results are expressed as mMoles Trolox equivalents (TE)/g extract.
The Trolox-equivalent antioxidant capacity (TEAC) determinations were carried out by mixing ABTS•+ with fresh standard (1 mM Trolox) or extract (100, 150, 200, 250 and 300 µg/mL). Absorbances were read at 734 nm after 6 min of room temperature incubation using a spectrophotometer (Thermo Spectronic Helios Alfa). Results are expressed as μM Trolox equivalents/g extract.
3.5. HPLC-DAD-MS Analysis
The extracts were analysed by HPLC coupled to a diode array detector (HPLC-DAD) to set the conditions for HPLC-ESI-MS/MS studies. The HPLC system used for DAD analysis was a Shimadzu equipment (Shimadzu Corporation, Kyoto, Japan) consisting of a LC-20AT pump, a SPD-M20A UV diode array detector, CTO-20AC column oven and a LabSolution software. A MultoHigh 100 RP 18–5µm (250 × 4.6 mm) column (CS-Chromatographie Service GmbH, Langerwehe, Germany) maintained at 25 °C was used. Approximately 5 mg/mL of PEFE was filtered through a 0.45 µm filter (Waters, Milford, MA, USA) and injected into HPLC-DAD and HPLC-ESI-MS/MS. The compounds were monitored at 254, 330 and 535 nm, and UV spectra from 200 to 600 nm were recorded for peak characterization. The HPLC analysis was performed using a linear gradient solvent system as described by Quispe et al. [23]. The flow rate was 1 mL/min and the volume of injected sample was 20 µL.
The mass spectrometer consisted of a HPLC HP1100 (Agilent Technologies Inc., Santa Clara, CA, USA) connected through a split to the mass spectrometer Esquire 4000 Ion Trap LC/MS(n) system (Bruker Daltonik GmbH, Bremen, Germany). Ionization was performed at 3000 V assisted by nitrogen as nebulizing gas at 24 psi and as drying gas at 365 °C and a flow rate of 6 L/min. Negative ions were detected using full scan (m/z 20–2200) and normal resolution (scan speed 10300 m/z/s; peak with 0.6 FWHM/m/z). The trap parameters were set in ion charge control (ICC) using manufacturer default parameters, and maximum accumulation time of 200 ms. Collision induced dissociation (CID) was performed by collisions with helium background gas present in the trap and automatically controlled through Smart Frag option.
Additional mass spectrometry measurements were performed using an Agilent Series 1200 LC System (Agilent, Ramsey, MN, USA) coupled to a MicroQTOF Q II (Bruker Daltonics, Billerica, MA, USA). The HPLC system consisted in a micro vacuum degasser, binary pumps, an autosampler (40 μL sample loop), a thermostated column compartment and a diode array detector. The mass spectrometer equipped with an electrospray ion source and QTOF analyser, was used in MS and MS/MS mode for the structural analysis of phenolics. HPLC analyses were performed on a thermostated (40 °C) MultoHigh 100 RP 18–5µm (250 × 4.6 mm) column (5 μm) with a flow rate of 1.0 mL/min using a split to the detector. The solvents and ramp were the same as described for the ion trap equipment.
ESI-MS detection was performed in negative and positive ion mode with mass acquisition between 100 and 1500 Da. Nitrogen was used as drying and nebulizer gas (7 L/min and 3.5 bar, respectively), and 180 °C for drying temperature. For MS/MS experiments, fragmentation was achieved by using Auto MS2 option. DAD analyses were carried out in the range between 200 and 700 nm. The identification of phenolic compounds in “algarrobo” pods meal was carried out by comparison of the spectral properties (UV and ESI-MS and MS/MS) of the compounds with literature data.
3.6. Statistical Analysis
Determinations of TP, TF, DPPH and FRAP, were performed in triplicate and results are expressed as mean values ± SD. For the TEAC assay, a curve was plotted for each sample and a correlation coefficient with 95% confidence limit was established. To assess the relationship between the antioxidant activities and the TP and TF content, Pearson’s correlation coefficients were calculated with 95% confidence. Statistical analysis was carried out using the software SPSS 14.0 for Windows.
4. Conclusions
The main compounds in the PEFE were flavonoids. One sample contained cyanidin hexoside and other anthocyanins, being this the first report on the occurrence of anthocyanins in Chilean Prosopis pods. The phenolic composition and antioxidant properties of the Chilean Prosopis mesocarp flour supports its use as a functional food. Additional studies are required to compare the potential of the different flour sources in artisanal and commercial food products. A higher number of samples should be analyzed to have a better picture on the phenolic composition of Chilean Prosopis mesocarp flour.
Acknowledgments
We thank FONDECYT Project 1120096, PCCI12067, MINCYT (CH/11/13) “Valorización de frutos nativos sudamericanos. Metabolómica de frutos de algarrobos”, ANPCyT (PICT 1959), Programa de Investigación de Excelencia Interdisciplinaria (PIEI-QUIM-BIO), Universidad de Talca for financial support. We are grateful to Patricio Peñailillo, Herbario de la Universidad de Talca, for the identification of the plants.
Author Contributions
C.Q., M.J.P. and A.S.C. worked on the HPLC-DAD and HPLC-DAD-MS fingerprints. M.P.C.S. prepared the Prosopis flour extracts, F.J.-A. and C.T. performed the antioxidant experiments. G.S.H. and M.I.I. contributed with experiment planning, data interpretation and revised the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Sample Availability: Samples are not available from authors.
References
- 1.Fagg C., Stewart J. The value of Acacia and Prosopis in arid and semi-arid environments. J. Arid Environ. 1994;27:3–25. doi: 10.1006/jare.1994.1041. [DOI] [Google Scholar]
- 2.Felger R.S. Mesquite in Indian Cultures of South-Western North America. In: Simpson B.B., editor. Mesquite: Its Biology in Two Desert Ecosystems. 1st ed. Dowden, Hutchinson and Ross; Stroudsburg, PA, USA: 1977. pp. 150–176. [Google Scholar]
- 3.Schmeda-Hirschmann G. Plant resources used by the Ayoreo of the Paraguayan Chaco. Econ. Bot. 1994;48:252–258. doi: 10.1007/BF02862325. [DOI] [Google Scholar]
- 4.Schmeda Hirschmann G. Etnobotánica Ayoreo. Contribución al estudio de la flora y vegetación del Chaco. XI. Candollea. 1998;53:1–50. [Google Scholar]
- 5.Pérez M.J., Cuello A.S., Zampini I.C., Ordoñez R.M., Alberto M.R., Quispe C., Schmeda-Hirschmann G., Isla M.I. Polyphenolic compounds and anthocyanin content of Prosopis nigra and Prosopis alba pods flour and their antioxidant and anti-inflammatory capacity. Food Res. Int. 2014;64:762–771. doi: 10.1016/j.foodres.2014.08.013. [DOI] [PubMed] [Google Scholar]
- 6.Astudillo L., Schmeda-Hirschmann G., Herrera J.P., Cortés M. Proximate composition and biological activity of Chilean Prosopis species. J. Sci. Food Agric. 2000;80:567–573. doi: 10.1002/(SICI)1097-0010(200004)80:5<567::AID-JSFA563>3.0.CO;2-Y. [DOI] [Google Scholar]
- 7.Schmeda-Hirschmann G., Razmilic I., Gutierrez M.I., Loyola J.I. Proximate composition and biological activity of food plants gathered by Chilean Amerindians. Econ. Bot. 1999;53:177–187. doi: 10.1007/BF02866496. [DOI] [Google Scholar]
- 8.Arenas P. Etnografía y alimentación entre los Toba-Ñachilamolekek y Wichí-Lhukútas del Chaco Central. (Argentina) 1st ed. ProBiota Facultad de Ciencias Naturales y Museo, Universidad Nacional de La Plata; Buenos Aires, Argentina: 2003. [Google Scholar]
- 9.Cardozo M.L., Ordóñez R.M., Zampini I.C., Cuello A.S., Dibenedetto G., Isla M.I. Evaluation of antioxidant capacity, genotoxicity and polyphenol content of non-conventional food: Prosopis flour. Food Res. Int. 2010;43:1505–1510. doi: 10.1016/j.foodres.2010.04.004. [DOI] [Google Scholar]
- 10.Escobar B., Estévez A.M., Fuentes C., Venegas D. Use of Algarrobo (Prosopis chilensis (Mol) Stuntz) flour as protein and dietary fiber source in cookies and fried chips manufacture. Arch. Latinoam. Nutr. 2009;59:191–198. [PubMed] [Google Scholar]
- 11.Giovannetti M.A., Lema V.S., Bartoli C.G., Capparelli A. Starch grain characterization of Prosopis chilensis (Mol.) Stuntz and P. flexuosa DC, and the analysis of their archaeological remains in Andean South America. J. Archaeol. Sci. 2008;35:2973–2985. [Google Scholar]
- 12.Fuentes V. Productos Forestales no madereros. INFOR. 2013;16:1–6. [Google Scholar]
- 13.Soto D., Gysling J. Productos con oportunidades de desarrollo en Chile: Mucílago de algarrobo chileno (Prosopis chilensis) INFOR. 2009;15:255–276. [Google Scholar]
- 14.Felker P.A., Takeoka G., Dao L. Pod mesocarp flour of North and South American species of leguminous tree Prosopis (Mesquite): Composition and food applications. Food Rev. Int. 2013;29:49–66. doi: 10.1080/87559129.2012.692139. [DOI] [Google Scholar]
- 15.Felker P., Grados N., Cruz G., Prokopiuk D. Economic assessment of production of flour from Prosopis alba and P. pallida pods for human food applications. J. Arid Environ. 2003;53:517–528. doi: 10.1006/jare.2002.1064. [DOI] [Google Scholar]
- 16.Astudillo L., Jürgens K., Schmeda-Hirschmann G., Griffith G.A., Holt D.J., Jenkins P.R. DNA binding alkaloids from Prosopis alba. Planta Med. 1999;65:161–162. doi: 10.1055/s-2006-960454. [DOI] [PubMed] [Google Scholar]
- 17.Tapia A., Feresin G.E., Bustos D., Astudillo L., Theoduloz C., Schmeda-Hirschmann G. Biologically active alkaloids and a free radical scavenger from Prosopis species. J. Ethnopharmacol. 2000:241–246. doi: 10.1016/S0378-8741(00)00171-9. [DOI] [PubMed] [Google Scholar]
- 18.Schmeda-Hirschmann G., Jakupovic J. A DNA binding compound from Prosopis tamarugo pods. Bol. Soc. Chil. Quim. 2000;45:645–647. [Google Scholar]
- 19.Jain M., Jos E.M., Arora D., Sharma Y.V.R.K. Effect of proline on Triticum aestivum (wheat) under the drought conditions of salinity. J. Pharm. Res. 2013;7:506–509. doi: 10.1016/j.jopr.2013.05.002. [DOI] [Google Scholar]
- 20.Vendruscolo E.C.G., Schuster I., Pileggi M., Scapim C.A., Molinari H.B.C., Marur C.J., Vieira L.G.E. Stress-induced synthesis of proline confers tolerance to water deficit in transgenic wheat. J. Plant Physiol. 2007;164:1367–1376. doi: 10.1016/j.jplph.2007.05.001. [DOI] [PubMed] [Google Scholar]
- 21.Ortiz C.A., Bravo L.A., Pinto M., Cardemil L. Physiological and molecular responses of Prosopis chilensis under field and simulation conditions. Phytochemistry. 1995;40:1375–1382. doi: 10.1016/0031-9422(95)00411-Y. [DOI] [Google Scholar]
- 22.Cattaneo F., Sayago J.E., Alberto M.R., Zampini I.C., Ordoñez R.M., Chamorro V., Pazos A., Isla M.I. Anti-inflammatory and antioxidant activities, functional properties and mutagenicity studies of protein and protein hydrolysate obtained from Prosopis alba seed flour. Food Chem. 2014;161:391–399. doi: 10.1016/j.foodchem.2014.04.003. [DOI] [PubMed] [Google Scholar]
- 23.Quispe C., Petroll K., Theoduloz C., Schmeda-Hirschmann G. Antioxidant effect and characterization of South American Prosopis pods syrup. Food Res. Int. 2014;56:174–181. doi: 10.1016/j.foodres.2013.12.033. [DOI] [Google Scholar]
- 24.Lv J., Yu L., Lu Y., Niu Y., Liu L., Costa J., Yu L. Phytochemical compositions, and antioxidant properties, and antiproliferative activities of wheat flour. Food Chem. 2012;135:325–331. doi: 10.1016/j.foodchem.2012.04.141. [DOI] [PubMed] [Google Scholar]
- 25.Mojica L., Meyer A., Berhow M.A., González de Mejía E. Bean cultivars (Phaseolus vulgaris L.) have similar high antioxidant capacity, in vitro inhibition of α-amylase and α-glucosidase while diverse phenolic composition and concentration. Food Res. Int. 2015;69:38–48. doi: 10.1016/j.foodres.2014.12.007. [DOI] [Google Scholar]
- 26.Zhang B., Deng Z., Ramdath D.D., Tang Y., Chen P.X., Liu R., Liu Q., Tsao R. Phenolic profiles of 20 Canadian lentil cultivars and their contribution to antioxidant activity and inhibitory effects on α-glucosidase and pancreatic lipase. Food Chem. 2015;172:862–872. doi: 10.1016/j.foodchem.2014.09.144. [DOI] [PubMed] [Google Scholar]
- 27.Kunyanga C.N., Imungi J.K., Okoth M.W., Biesalski H.K., Vadivel V. Total phenolic content, antioxidant and antidiabetic properties of methanolic extract of raw and traditionally processed Kenyan indigenous food ingredients. LWT-Food Sci. Technol. 2012;45:269–276. doi: 10.1016/j.lwt.2011.08.006. [DOI] [Google Scholar]
- 28.Escribano-Bailón M.T., Santos-Buelga C., Rivas-Gonzalo J.C. Anthocyanins in cereals. J. Chromatogr. A. 2004;1054:129–141. doi: 10.1016/j.chroma.2004.08.152. [DOI] [PubMed] [Google Scholar]
- 29.Wu X., Prior R.L. Systematic identification and characterization of anthocyanins by HPLC-ESI-MS/MS in common foods in the United States: fruits and berries. J. Agric. Food Chem. 2005;53:2589–2599. doi: 10.1021/jf048068b. [DOI] [PubMed] [Google Scholar]
- 30.Ververidis F., Trantas E., Douglas C., Vollmer G., Kretzschmar G., Panopoulos N. Biotechnology of flavonoids and other phenylpropanoid-derived natural products. Part I: Chemical diversity, impact on plant biology and human health. Biotechnol. J. 2007;2:1214–1234. doi: 10.1002/biot.200700084. [DOI] [PubMed] [Google Scholar]
- 31.Harbaum B., Hubbermann E.M., Wolff C., Herges R., Zhu Z., Schwarz K. Identification of flavonoids and hydroxycinnamic acids in Pak Choi varieties (Brassica campestris L. ssp. chinensis var. communis) by HPLC-ESI-MSn and NMR and their quantification by HPLC-DAD. J. Agric. Food Chem. 2007;55:8251–8260. doi: 10.1021/jf071314+. [DOI] [PubMed] [Google Scholar]
- 32.Ferreres F., Silva B.M., Andrade P.B., Seabra R.M., Ferreira M.A. Approach to the study of C-glycosyl flavones by ion trap HPLC-PAD-ESI/MS/MS: Application to seeds of quince (Cydoniaoblonga) Phytochem. Anal. 2003;14:352–359. doi: 10.1002/pca.727. [DOI] [PubMed] [Google Scholar]
- 33.Takeoka G.R., Dao L.T., Full G.H., Wong R.Y., Harden L., Edwards R., Berrios J. Characterization of black bean (Phaseolus vulgaris, L) anthocyanins. J. Agric. Food Chem. 1997;45:3395–3400. doi: 10.1021/jf970264d. [DOI] [Google Scholar]
- 34.Kamiya H., Yanase E., Nakatsuka S. Novel oxidation products of cyanidin 3-O-glucoside with 2,2'-azobis-(2,4-dimethyl) valeronitrile and evaluation of anthocyanin content and its oxidation in black rice. Food Chem. 2014;155:221–226. doi: 10.1016/j.foodchem.2014.01.077. [DOI] [PubMed] [Google Scholar]
- 35.De Pascual-Teresa S., Santos-Buelga C., Rivas-Gonzalo J.C. LC-MS analysis of anthocyanins from purple corn cob. J. Sci. Food Agric. 2002;82:1003–1006. doi: 10.1002/jsfa.1143. [DOI] [Google Scholar]
- 36.Ha T.J., Lee M.H., Park C.H., Pae S.B., Shim K.B., Ko J.M., Shin S.O., Baek I.Y., Park K.Y. Identification and characterization of anthocyanins in yard-long beans (Vigna unguiculata ssp. sesquipedalis L.) by high-performance liquid chromatography with diode array detect ion and electrospray ionization/mass spectrometry (HPLC-DAD-ESI/MS) analysis. J. Agric. Food Chem. 2010;58:2571–2576. doi: 10.1021/jf903883e. [DOI] [PubMed] [Google Scholar]
- 37.Takeoka G.R., Wong R.Y., Dao L., Felker P. Identification of 5,6-dihydro-6-propyl-2H-pyran-2-one as the major volatile constituent in mesquite (Prosopis) flour. Food Chem. 2009;115:1025–1027. doi: 10.1016/j.foodchem.2008.12.081. [DOI] [Google Scholar]
- 38.Wang Y., Jiang Z.Z., Chen M., Wu M.J., Guo H.L., Sun L.X., Wang H., Zhang S., Wang T., Zhang L.Y. Protective effect of total flavonoid C-glycosides from Abrus mollis extract on lipopolysaccharide-induced lipotoxicity in mice. Chin. J. Nat. Med. 2014;12:461–468. doi: 10.1016/S1875-5364(14)60072-8. [DOI] [PubMed] [Google Scholar]
- 39.Chen M., Wang T., Jiang Z.Z., Shan C., Wang H., Wu M.J., Zhang S., Zhang Y., Zhang L.Y. Anti-inflammatory and hepatoprotective effects of total flavonoid C-glycosides from Abrus mollis extracts. Chin. J. Nat. Med. 2014;12:590–598. doi: 10.1016/S1875-5364(14)60090-X. [DOI] [PubMed] [Google Scholar]
- 40.Islam M.N., Ishita I.J., Jung H.A., Choi J.S. Vicenin 2 isolated from Artemisia capillaris exhibited potent anti-glycation properties. Food Chem. Toxicol. 2014;69:55–62. doi: 10.1016/j.fct.2014.03.042. [DOI] [PubMed] [Google Scholar]
- 41.Nagaprashantha L.D., Vatsyayan R., Singhal J., Fast S., Roby R., Awasthi S., Singhal S.S. Anti-cancer effects of novel flavonoid vicenin-2 as a single agent and in synergistic combination with docetaxel in prostate cancer. Biochem. Pharmacol. 2011;82:1100–1109. doi: 10.1016/j.bcp.2011.07.078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Agati G., Azzarello E., Pollastri S., Tattini M. Flavonoids as antioxidants in plants: Location and functional significance. Plant Sci. 2012;196:67–76. doi: 10.1016/j.plantsci.2012.07.014. [DOI] [PubMed] [Google Scholar]
- 43.Tsuchiya H. Structure-dependent membrane interaction of flavonoids associated with their bioactivity. Food Chem. 2010;120:1089–1096. doi: 10.1016/j.foodchem.2009.11.057. [DOI] [Google Scholar]
- 44.Spencer J.P.E., El Mohsen M.M.A., Rice-Evans C. Cellular uptake and metabolism of flavonoids and their metabolites: Implications for their bioactivity. Arch. Biochem. Biophys. 2004;423:148–161. doi: 10.1016/j.abb.2003.11.010. [DOI] [PubMed] [Google Scholar]
- 45.Jiménez-Aspee F., Quispe C., Soriano M.D.P.C., Fuentes Gonzalez J., Hüneke E., Theoduloz C., Schmeda-Hirschmann G. Antioxidant activity and characterization of constituents in Copao fruits (Eulychnia acida Phil., Cactaceae) by HPLC-DAD-MS/MSn. Food Res. Int. 2014;62:286–298. doi: 10.1016/j.foodres.2014.03.013. [DOI] [Google Scholar]
- 46.Simirgiotis M.J., Bórquez J., Schmeda-Hirschmann G. Antioxidant capacity, polyphenolic content and tandem HPLC-DAD-ESI/MS profiling of phenolic compounds from the South American berries Luma apiculata and Luma chequén. Food Chem. 2013:289–299. doi: 10.1016/j.foodchem.2013.01.089. [DOI] [PubMed] [Google Scholar]