Abstract
Microfluidic culture has the potential to revolutionize cancer diagnosis and therapy. Indeed, several microdevices are being developed specifically for clinical use to test novel cancer therapeutics. To be effective, these platforms need to replicate the continuous interactions that exist between tumor cells and non-tumor cell elements of the tumor microenvironment through direct cell-cell or cell-matrix contact or by the secretion of signaling factors such as cytokines, chemokines and growth factors. Given the challenges of personalized or precision cancer therapy, especially with the advent of novel immunotherapies, a critical need exists for more sophisticated ex vivo diagnostic systems that recapitulate patient-specific tumor biology with the potential to predicting response to immune-based therapies in real-time. Here, we present details of a method to screen for the response of patient tumors to immune checkpoint blockade therapy, first reported in Jenkins et al. Cancer Discovery 2018, with updated evaluation of murine- and patient-derived organotypic tumor spheroids (MDOTS/PDOTS), including evaluation of the requirement for 3D microfluidic culture in MDOTS, demonstration of immune-checkpoint sensitivity of PDOTS, and expanded evaluation of tumor-immune interactions using RNA-sequencing to infer changes in the tumor-immune microenvironment. We also examine some potential improvements to current systems and discuss the challenges in translating such diagnostic assays to the clinic.
Introduction
Monoclonal antibodies targeting immune checkpoints (e.g. PD-1/PD-L1 axis, CTLA-4) have demonstrated clinical activity in several malignances (Fig. 1)1, 2. Unlike molecular targeted therapies (e.g. BRAF and EGFR inhibitors), which can be employed in patients whose tumors harbor select oncogenic driver mutations (e.g. BRAF V600E, EGFR exon19del or L8585R), to date there are no reliable predictors of response for immune checkpoint inhibitors. PD-L1 expression predicts response to anti-PD-1/PD-L1 antibodies in some, but not all patients, and is increasingly recognized as an imperfect marker of activity3. Intense research efforts are now underway to identify predictive biomarkers of response, toxicity and resistance to immunotherapeutic agents.
Figure 1 – Schematic of PD-1/CTLA-4 Blockade. Schematic detailing basic steps involved in generation of tumor-specific T cells.
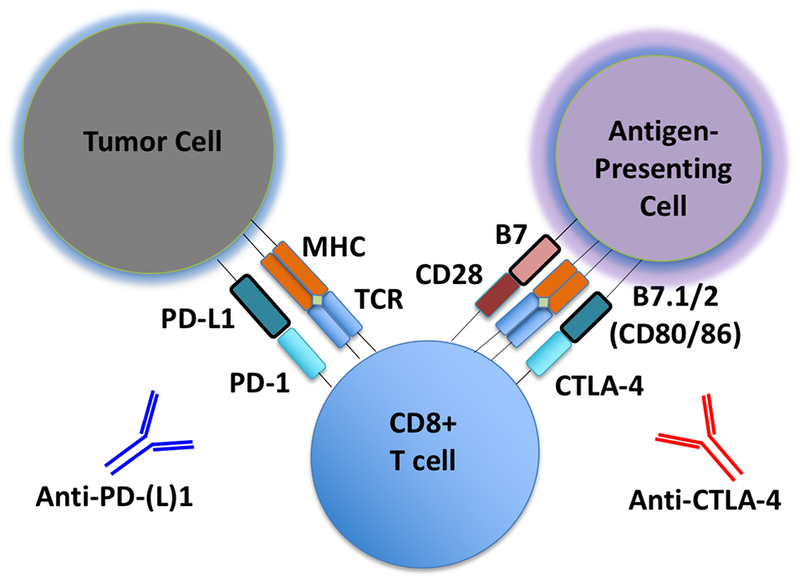
Shown is a schematic of a tumor cell, CD8 effector T cell, and an antigen-presenting cell (APC), with associated cell-cell interactions via PD-1/PD-L1 and CTLA-4/B7. Tumor-associated antigens or neo-antigens are presented by major histocompatibility complex (MHC) on APCs or tumor cells to T cells with appropriate T-cell receptor (TCR). CD28 co-activating receptor on T cells binds B7 on APCs. Anti-PD-(L)1 and anti-CTLA-4 antibodies are shown.
There is great interest in studying other immune checkpoints and novel combinations of immune modulatory agents to overcome both innate and acquired resistance to ICB4, 5. Dual immune checkpoint blockade (ICB) with combination PD-1 and CTLA-4 antibody treatment has recently shown dramatic response rates in patients with metastatic melanoma, however roughly half of patients experienced significant toxicity from the treatment regimen6, 7. Several clinical trials of combinations of immunotherapeutic agents with molecular targeted therapies (e.g. kinase inhibitors), cytotoxic chemotherapy, and/or radiation are underway, all in the effort to provide long-lasting disease control to more patients5. While the number of these ‘rational’ combination trials continues to expand, our understanding of the ‘backbone’ therapy of PD-1 or CTLA-4 blockade remains far from complete.
Mechanisms of immune escape and resistance to ICB are diverse, owing to insufficient numbers of anti-tumor T cells present, or ineffective immune response due to local (e.g. tumor or stromal derived) that impair the cytotoxic immune response8–11. Approaches to identify cellular, pathologic, immunologic, or molecular features that distinguish responding from non-responding patients have evaluated local features in the tumor microenvironment using immunohistochemical or transcriptomic evaluation of frozen or fixed tissue from biopsies, and circulating levels of select immune cells or cytokines12, 13. As biopsies from patients who have responded to ICB often demonstrate the presence of an inflammatory infiltrate within the tumor, and gene expression profiling studies have confirmed upregulation of pro-inflammatory cytokines and chemokines within tumors following PD-1 blockade, there is increasing interest in understanding the role of the tumor microenvironment in the response to ICB12, 13. Development of more sophisticated pre-clinical and ex vivo cancer models that recapitulate human tumor biology may facilitate efforts to predict response to targeted and immune-based therapies.
There is growing interest in capitalizing on the increasing availability of 3D culture systems to tackle important questions in cancer biology, especially with the recent integration of microfluidic systems. 3D-microfluidic culture systems now offer a greater level of precision for evaluating a host of complex biological phenomena in relevant model systems14. With the advent of ICB and the expanding number of novel immunotherapies and combination therapies, there is increasing interest in the use of 3D culture systems to interrogate the tumor immune microenvironment15.
Recently, we described a novel method of profiling response to PD-1 blockade using organotypic tumor spheroids cultured in collagen hydrogels suspended in a 3D microfluidic device5. Patient- or murine-derived organotypic tumor spheroids (MDOTS/PDOTS) retain autologous immune cells and respond to PD-1 blockade ex vivo16, 17. Using murine tumors from established PD-1 responsive or resistant tumors, we were able to recapitulate sensitivity and resistance to ICB ex vivo. MDOTS/PDOTS also served as a platform to test novel combination therapies in combination with PD-1 blockade, including inhibitors or TBK117 and CDK4/617, both of which enhanced response to PD-1 blockade ex vivo and subsequently in vivo efficacy was confirmed. In this paper, we will describe in detail the methods of MDOTS/PDOTS platform (“version 1.0”), and propose some novel applications and future directions.
Materials and methods
Patient Samples.
Tumor samples were collected and analyzed according to Dana-Farber/Harvard Cancer Center IRB-approved protocols. Informed consent was obtained from all patients. These studies were conducted according to the Declaration of Helsinki and approved by the MGH and DFCI IRBs.
Syngeneic Murine Models.
All animal experiments were performed in compliance with established ethical regulations and were approved by the Dana-Farber Animal Care and Use Committee. MC38 murine colon adenocarcinoma cells were generously provided by Dr. Gordon Freeman (DFCI) received under an MTA from Dr. Jeffrey Schlom of NCI (Bethesda, MD). CT26 colon carcinoma cells were purchased from ATCC (2015). Mouse tumor implantations were performed as previously described17.
Device Design and Fabrication.
MDOTS and PDOTS were evaluated using ‘3-D cell culture chip’ (DAX-1, AIM BIOTECH, https://www.aimbiotech.com/), as previously described16, 18. Microfluidic device design and fabrication using cyclic olefin polymer (COP) conducted at AIM BIOTECH. Briefly, the single layer slide format (75mm × 25mm) device or ‘chip’, consists of 3 microfluidic chambers each with a central gel channel (width 1.3 mm) flanked by two media channels (width 0.5 mm). The height of the microfluidic chambers is 0.25 mm. Media channels were designed including larger reservoirs to prevent over-aspiration (Figs. 3a–c).
Figure 3 – Microfluidic device.
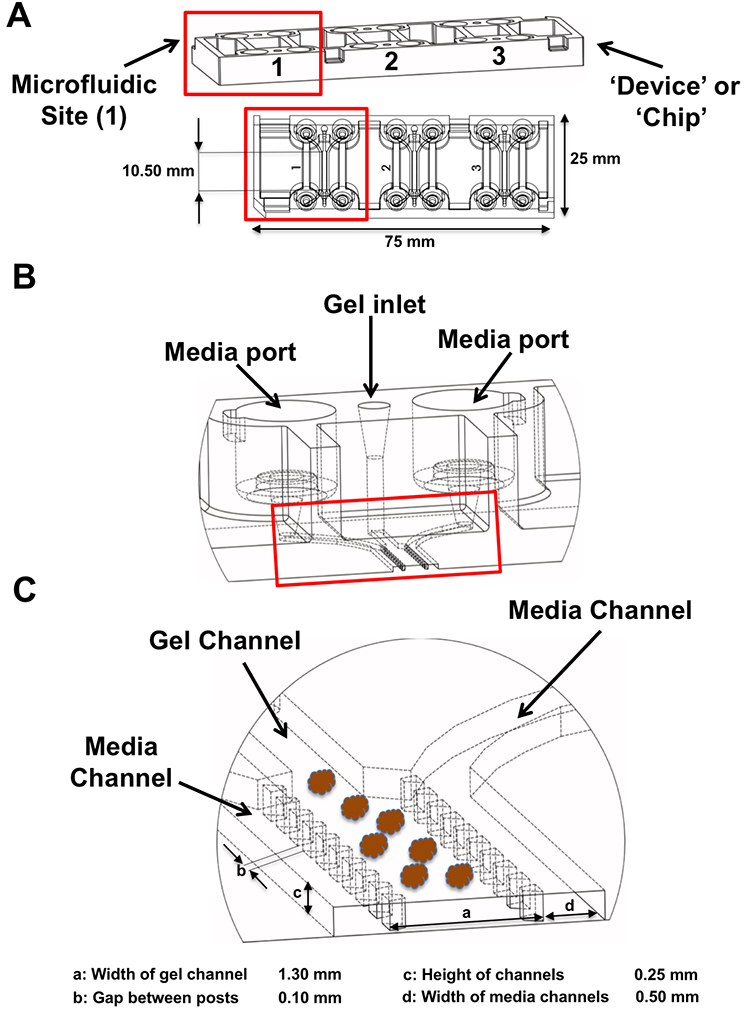
(A), the 3D cell culture chip (AIM Biotech) is shown with three independent microfluidic chambers per chip. Red rectangle identifies a single microfluidic chamber in the 3D cell culture chip. (B-C), each device contains a center gel region with posts separating the gel region from the anti-parallel side channels. Gel loading port and media ports labeled (B), along with center and side channels (C).
Preparation of MDOTS/PDOTS and Ex Vivo Microfluidic 3D Culture.
MDOTS/PDOTS (S2 fraction; 40–100 μm) containing tumor, immune, and stromal cells were prepared as described17. Briefly, fresh tumor specimens (murine and human patients) were received in media (DMEM or RPMI) on ice and minced in a standard 10cm dish using sterile forceps and scalpel. Minced tumor was resuspended in high-glucose DMEM (or RPMI, for CT26) with 100 U/mL type IV collagenase, and 15 mM HEPES (Life Technologies, Carlsbad, CA). Samples were incubated at 37°C and evaluated every 15 minutes to monitor for adequate formation of spheroids with modest release of single cells. Following digestion (range: 15–60 min), equal volumes of media were added to minced tumor specimens and samples were pelleted and resuspended in fresh media and passed over 100 μm and 40 μm filters sequentially to obtain S1 (>100 μm), S2 (40–100 μm), and S3 (<40 μm) spheroid fractions, which were subsequently transferred to ultra low-attachment tissue culture plates. An aliquot of the S2 fraction was pelleted and resuspended in type I rat tail collagen and the spheroid-collagen mixture was injected into the center gel region of the 3D microfluidic chamber (10 uL per each microfluidic chamber). After incubation for 30 minutes at 37°C in sterile humidity chambers, collagen hydrogels containing PDOTS/MDOTS were hydrated with media with or without indicated therapeutic monoclonal antibodies - MDOTS: isotype control IgG (10 μg/mL, clone 2A3) or anti-PD-1 (10 μg/mL, clone RMP1–14); PDOTS: anti-PD-1 (pembrolizumab, 250 μg/mL), anti-CTLA-4 (ipilimumab, 50 μg/mL), or combination (250 μg/mL pembrolizumab + 50 μg/mL ipilimumab), as previously shown17.
Live/Dead and Immunofluorescence Staining of MDOTS/PDOTS.
Live/Dead fluorescence staining was performed using AO/PI Staining Solution, as previously described17. Following incubation with AO/PI or Hoechst/PI (20 min, room temp, protected from light) or Hoechst/PI (45 min, 37°C, 5% CO2), images were obtained. Image capture and analysis were performed using a Nikon Eclipse 80i fluorescence microscope equipped with Z-stack (Prior), motorized stage (ProScan) and ZYLA5.5 sCMOS Camera (Andor) and NIS-Elements AR software package. Live and dead cell quantitation was performed by measuring total cell area of each dye. For direct immunofluoscence staining coupled with Live/Dead analysis, unfixed live MDOTS or PDOTS were washed with PBS and blocked with Fc receptor (FcR) blocking reagent (PDOTS, Miltenyi; MDOTS, BioLegend) for 30 minutes at room temperature. Directly conjugated antibodies for PDOTS were CD326 EpCAM- PE (clone 9C4), CD45-AlexaFluor-488 (HI30), CD8a-AlexaFluor488 (RPA-T8); for MDOTS, CD45-AlexaFluor488 or 647 (30-F11), CD8α-PE (53–6.7; BioLegend). Antibodies were diluted 1:50 in 10 μg/mL solution of Hoechst 33342 (in PBS) and loaded into microfluidic chambers for 1-hour incubation at room temperature in the dark. Spheroids were washed twice with PBS with 0.1%Tween20 followed by PBS. For viability assessment, microfluidic chambers were loaded with 1:1,000 solution of calcein AM in PBS.
Live/dead analysis in 2D conditions.
S2 fraction of CT26 tumors was split and seeded in 3-D cell culture chips parallel with flat bottom CellBind Surface 384-well plate (Corning, #3683). 2D experiment was performed as following: spheroids were loaded in wells of 384-well plate in triplicates in 2 densities of ~15 and ~30 spheroids/well in cultural media containing either IgG or αPD-1 at 10μg/mL. Plate was incubated in humidity chamber at 37°C, 5% CO2 for 5 days. At the end media was gently removed and 20μL of AOPI (Nexcelom) was loaded into each well. After 20 minute incubation in the dark at RT cells were imaged on inverted Nikon Eclipse Ti microscope equipped with Nikon DS-Qi1Mc camera using NIS-Elements software. Total area of AO-stained live cells versus Propidium Iodide-stained dead cells was quantified. CT26 tumor spheroids seeded in both high and low densities produced similar results, graph for the high density is shown.
Cytokine Profiling.
Media collected from MDOTS/PDOTS culture at the indicated time point were removed (by micropipette) and transferred to 1.5-mL Eppendorf tubes. After collection, tubes containing conditioned media were placed on dry ice before storage at −80°C. For bead-based cytokine profiling, conditioned media were thawed on ice. Four panels of multiplex ELISA assays were performed utilizing a bead-based immunoassay approach, the Thermo Fisher Immuno-Oncology Checkpoint 14-Plex Human ProcartaPlex™ Panel 1 (Cat No. EPX14A-15803–901), Bio-Rad Bio-Plex Pro Human Cytokine 27-plex panel (Cat No. m500kcaf0y), and Human Inflammation 37-plex panel (Cat No. 171AL001M), and Human Cytokine 40-plex panel (Cat No. 171AK99MR2). MDOTS/PDOTS conditioned media concentration levels (pg/mL) of each protein were derived from 5-parameter curve fitting models, and fold changes (relative to untreated or IgG control) were calculated and plotted as log2FC. Lower and upper limits of quantitation (LLOQ/ULOQ) were imputed from standard curves for cytokines above or below detection. Conditioned media from PDOTS were assayed neat. Only detectable cytokines were included in the analysis.
RNA-sequencing (RNA-seq) and CIBERSORT.
RNA-seq was performed as previously described1. For RNA-seq studies, SI-NET PDOTS were cultured in 3D cell culture chips (AIM BIOTECH). In brief, RNA lysates were prepared from SI-NET PDOTS on Day 9 using the lysis buffer from Agencourt RNAdvance kit (using 1:20 proteinase K). Conditioned media was removed (as described above), before 200 mL of lysis buffer (with proteinase K) was added to each microfluidic chamber. Devices were incubated for 25 min at 37°C, lysates were collected from each microfluidic chamber, and were transferred to RNase-free microcentrifuge tubes, and then stored at −80°C. RNA were extracted using RNAdvance Tissue kit (Beckman Coulter, Cat. No. A32649). RNA quantity and quality were assessed using Quant-iT™ RiboGreen™ RNA Assay Kit (Thermo Fisher, Cat. No. R11490) and Agilent Bioanalyzer RNA 6000 pico kit (Agilent, Cat. No. 5067–1513). RNA libraries were prepared from 10ng RNA per sample using Illumina Truseq RNA Access protocol (Illumina, Cat No. RS-301–2001). RNA-seq was performed at the DFCI Molecular Biology Core Facilities (Illumina NextSeq 500). RNA-seq data were aligned and differential expression analysis were performed using VIPER pipleline, as described19. CIBERSORT was performed as described20 (https://cibersort.stanford.edu).
Results & discussion
Sample Preparation: Basic Principles and Practical Experience -
To recapitulate the biology of the tumor immune microenvironment to model PD-1 blockade, one needs viable tumor tissue, autologous tumor-infiltrating immune cells, and an appropriate device and model extracellular matrix to permit 3-dimensional culture. When devising the workflow for PDOTS/MDOTS processing, we aimed for simplicity. As some immune cells lose viability within hours of collection, we felt it was essential to minimize the time required for tumor specimen processing. At the same time, we avoided added growth factors or cytokines to selectively support the viability or growth of lymphoid or myeloid cells as this might artificially alter biological response to ICB. Therefore, we used traditional cell culture media (DMEM or RPMI) supplemented with 10% FBS. In our initial pilot studies, this standard culture medium supported the growth of both MDOTS and PDOTS so further modification was not necessary.
Generation of MDOTS/PDOTS requires fresh tumor specimens from immune competent mouse models or patients. Several syngeneic murine cancer models have been profiled, including MC38 colon, CT26 colon, B16 melanoma, Lewis lung carcinoma, and GL261 glioblastoma. Diverse solid tumor types have been profiled to date, including primary lesions, lymphadenectomy specimens, pleural effusions, ascites fluid, and resected metastases. Baseline viability and sample size is essential for immune cell profiling and ex vivo culture. Surgical (excisional) specimens are ideal for PDOTS preparation, and are similar to the explanted murine tumors used for MDOTS preparation (Fig. 2a).
Figure 2 – MDOTS/PDOTS Workflow.
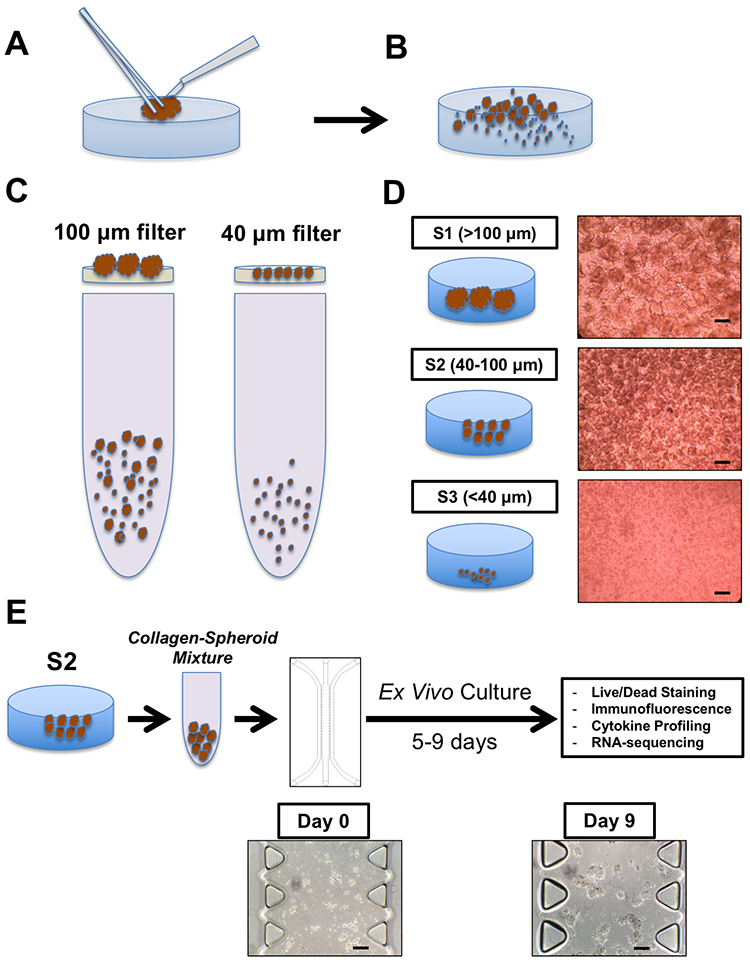
(A-B), A tumor specimen is received and subjected to physical and enzymatic dissociation (A), yielding dissociated tumor tissue (B) containing spheroids, single cells, and macroscopic tumor. (C-D), This heterogeneous mixture is then sequentially applied to 100 μm and 40 μm filters (C) to obtain three separate fractions (D), S1 (>100 μm), S2 (40–100 μm), and S3 (<40 μm). E, the S2 fraction is pelleted and resuspended in collagen to be injected into the microfluidic culture device for subsequent ex vivo culture with indicated terminal readouts. Scale bars indicate 100 μm (D, E).
While bacterial contamination was rare, all experiments were performed with antibiotics (1% penicillin-streptomycin). For primary colorectal cancer specimens (exposed to the gastrointestinal lumen and commensal bacteria), addition of metronidazole and gentamicin was required for Gram negative and anaerobic bacteria. For oropharyngeal head and neck squamous cell carcinomas, anti-fungal coverage (e.g. amphotericin B) was required to prevent contamination with Candida. Bacterial or fungal contamination was not encountered for cutaneous metastases, as the skin was sterilized prior to obtaining the excisional biopsy.
There is no minimum size cutoff, but rather sample quality, cellular viability, and immune cell composition are more important characteristics. For example, fine needle aspirates (FNAs) and core needle biopsies often yield far fewer cells than surgical (excisional) biopsies, and in our hands viability is frequently poorer from needle biopsies. On the rare occasion in which core needle biopsy yields viable cells and spheroids, there is often an insufficient number of PDOTS for extensive ex vivo profiling, thereby limiting the number of experimental conditions and replicates that are possible. On the other hand, pleural effusions and ascitic (peritoneal) fluid usually in cancer patients often generate at least 0.5–1 liter of fluid, and for most adenocarcinomas (e.g. lung, thyroid, breast) tumor cells are already aggregated with immune cells in multicellular spheroids.
With the exception of pleural effusions and peritoneal fluid (ascites) specimens, the initial step in processing involves physical and enzymatic dissociation using a limited collagenase digestion (see Materials and Methods). Type IV collagenase is used in this step as it has low tryptic activity, and it was used at a dilute concentration to avoid complete dissociation of MDOTS or PDOTS to single cells. The amount of time required to digest minced tumor tissue with type IV collagenase (in media) varies from sample to sample, and tumor to tumor. In general more fibrous tumors (e.g mesothelioma) require longer incubation times (up to 30–60 min), compared to most samples (20–30 min). Some murine tumor explants require only very brief collagenase treatment, as longer incubations have yielded an excess of single cells relative to spheroids.
Following physical and enzymatic dissociation, the minced and digested tumor specimen contains a mixture of macroscopic undigested tissue, spheroids, and single cells (Fig. 2b). Spheroids are isolated following passage of the dissociated specimen over a series of filters (100 μm and 40 xm) (Fig. 2c–d).
The second fraction (S2; 40–100 μm) is subsequently for MDOTS/PDOTS profiling and ex vivo culture (Fig. 2e). S1 and S3 fractions can be frozen or banked, or used for immune cell profiling.
3D Microfluidic Culture -
The majority of our studies to date utilized the commercially available DAX-1 3-D cell culture chip (AIM Biotech). Three self-contained microfluidic chambers are present on a single ‘3D cell culture chip’ (Fig. 3a). Following preparation of the S2 fraction, MDOTS/PDOTS are pelleted briefly and resuspended in a neutral pH collagen solution (see Materials and Methods). The collagen solution is prepared fresh for each experiment and is always prepared in excess. Pelleted MDOTS or PDOTS are initially re-suspended in a smaller volume of collagen (e.g. 100–200 μL) based on the size of the pellet, and then 10 μL is loaded into a single microfluidic chamber to confirm spheroid density and/or used for automated cell counting. Methods to accurately quantify the number of cells per spheroid (or cells per microfluidic chamber) are limited, but we will adjust the volume of collagen with a given number of spheroids to obtain approximately 10–20,000 cells per microfluidic chamber (based on automated cell counting technologies).
The collagen-spheroid mixture is kept on ice and injected into the upper and lower gel loading ports, such that the collagen-spheroid mixture extends almost half way to the opposite gel loading port, before the pipet tip is transferred to the opposite (lower) gel loading port to inject the remaining spheroids, such that the two pools of spheroids ‘meet’ in the middle of the gel region (Fig. 3a–c). After all microfluidic chambers are loaded in a similar fashion, they are transferred from the tissue culture hood into sterile humidity chambers and placed in a sterile incubator (37°C) for 30 min. During this incubation, the culture media is prepared with the various antibody or drug treatments. After the 30 min incubation is complete, each microfluidic chamber is hydrated with 250–300 μL of media (depending on the downstream application and frequency of media changes) containing the desired final concentration of the drug or antibody of interest. The chambers are then returned to the incubator in the aforementioned humidity chamber and can be cultured from 5–9 days (5–6 days routinely, especially for MDOTS which grow quickly).
MDOTS/PDOTS Readouts -
In our initial publication17, we featured five different methods of analysis of MDOTS/PDOTS: light/phase contrast microscopy, time-lapse (live) imaging, immunofluorescence microscopy, Live/Dead imaging, and secreted cytokine profiling. Much of the routine analysis of ex vivo response to PD-1 blockade focused on dynamic secreted cytokine changes using bead-based cytokine profiling and fluorescence imaging using viability dyes for quantitative live/dead assessment.
Evaluation of immune-mediated tumor cell killing by effector CD8 T cells is central to the evaluation of ex vivo profiling of PD-1 blockade. In our initial publication17, and companion study featuring PDOTS/MDOTS17, dual labeling fluorescence dyes for both qualitative and quantitative Live/Dead imaging was performed with acridine orange (AO) and propidium iodide (PI). AO/PI labeling for Live/Stain fluorescence Live/Dead staining has been in use for several decades, and are commercially available, inexpensive, and easy-to-use. Propidium iodide (PI) is a cell impermeable DNA dye that only stains cells following membrane damage (e.g. necrosis or late apoptosis) after which it binds nuclear DNA. Acridine orange (AO) is a membrane-permeable nucleic acid-binding dye. Therefore, live nucleated cells emit green fluorescent signal (AO permeable, PI excluded), whereas dead cells become permeable to PI, and pure red fluorescence is evident in dead cells due to quenching of green fluorescence by PI due to Forster (fluorescence) resonance energy transfer (FRET). Shown are MC38 MDOTS treated with isotype control IgG antibody or anti-PD-1 antibody (Fig. 4a) where treatment with the anti-PD-1 antibody results in CD8 T-cell-mediated tumor cell killing, as previously demonstrated17. Modifications to this protocol have been used with Hoechst 33342 (Ho) as a viability dye, instead of AO (Fig. 4b). Ho/PI staining can be a useful alternative for viability staining of cells with large cell size, high cytoplasm:nucleus ratio, and/or multiple cellular projections (e.g. CT26 murine colon carcinoma cells), to make threshold-setting more feasible. Quantification of Ho/PI and AO/PI fluorescence, as shown for CT26 MDOTS, demonstrates that sensitivity to PD-1 blockade is maintained in 3D microfluidic culture, but cannot be detected using MDOTS in 2D culture using 384-well plates (Fig. 4c–d).
Figure 4 – Live/Dead imaging and analysis of Murine-Derived Organotypic Tumor Spheroids.
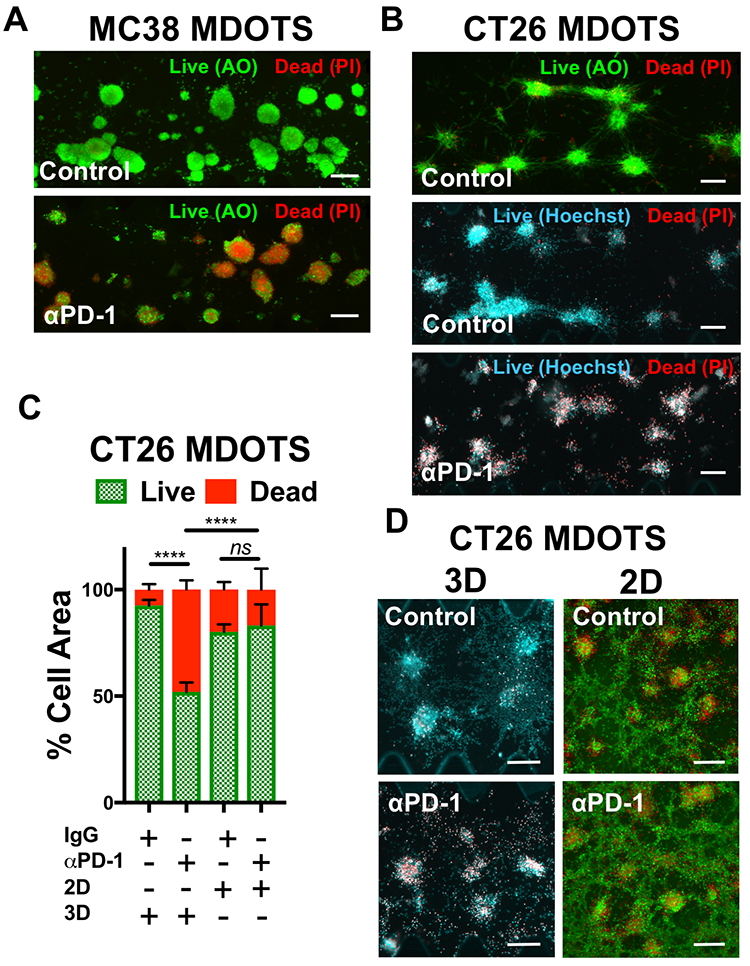
(A), acridine orange (AO) and propidium iodide (PI) staining of MC38 MDOTS on Day 6 of ex vivo culture, comparing control (isotype control IgG, 10 μg/mL) with anti-PD-1 (10 μg/mL). B, AO/PI and Hoechst/PI staining of CT26 MDOTS on Day 5 of ex vivo culture, comparing control (isotype control IgG, 10 μg/mL) with anti-PD-1 (10 μg/mL). C-D, Live/Dead analysis (C) and fluorescence images (D) of CT26 MDOTS treated with IgG or anti-PD-1 (10 μg/mL) for 5 days in 3D microfluidic culture (“3D”) compared to 384-well plates (“2D”) (3D - Ho/PI; 2D - AO/PI) (****p<0.0001, ns = not significant; Kruskal-Wallis with multiple comparisons; n≥3). Scale bars indicate 200 μm (A, B, D).
In addition to use of fluorescent dyes, direct immunofluorescence labeling can also readily be performed using MDOTS and PDOTS17. Standard immunofluorescence labeling protocols, coupled with viability dye evaluation using Calcein AM (Fig. 5a–c), can also be employed to evaluate viability of different cell types. Calcein AM is cleaved to its fluorescent form by intracellular non-specific esterases, and in combination with Hoechst can be used to quantify cellular viability. Direct immunofluorescence using cell surface proteins to delineate tumor cells and immune cells is of great utility, especially in PDOTS specimens that demonstrate great inter-sample variability (Fig. 5a–c). Shown here are PDOTS from high-grade serous carcinoma (HGSC) of the ovary and non-small cell lung carcinoma (NSCLC), where tumor cells are readily detected using EpCAM antibodies. Staining for CD8 readily identifies effector CD8+ T cells present in PDOTS, which are necessary effectors following treatment with anti-PD-(L)1 antibodies12. To date, we have focused on using direct immunofluorescence (IF) coupled with viability dyes in live, intact cells although multiplexed IF is under investigation. For example, PDOTS from patients with melanoma do not express EpCAM, and most reliable melanoma markers are intracellular proteins, which cannot be readily visualized in live, unpermeabilized cells. More sophisticated imaging, including evaluation of intracellular markers requires fixation and permeabilization, which precludes use of non-fixable viability dyes. Ideal panels would permit detection of tumor cells and immune cells (lymphoid and myeloid), expression of PD-1 and PD-L1, as well as viability in distinct cellular subsets.
Figure 5 – Fluorescence Imaging of Patient-Derived Organotypic Tumor Spheroids.
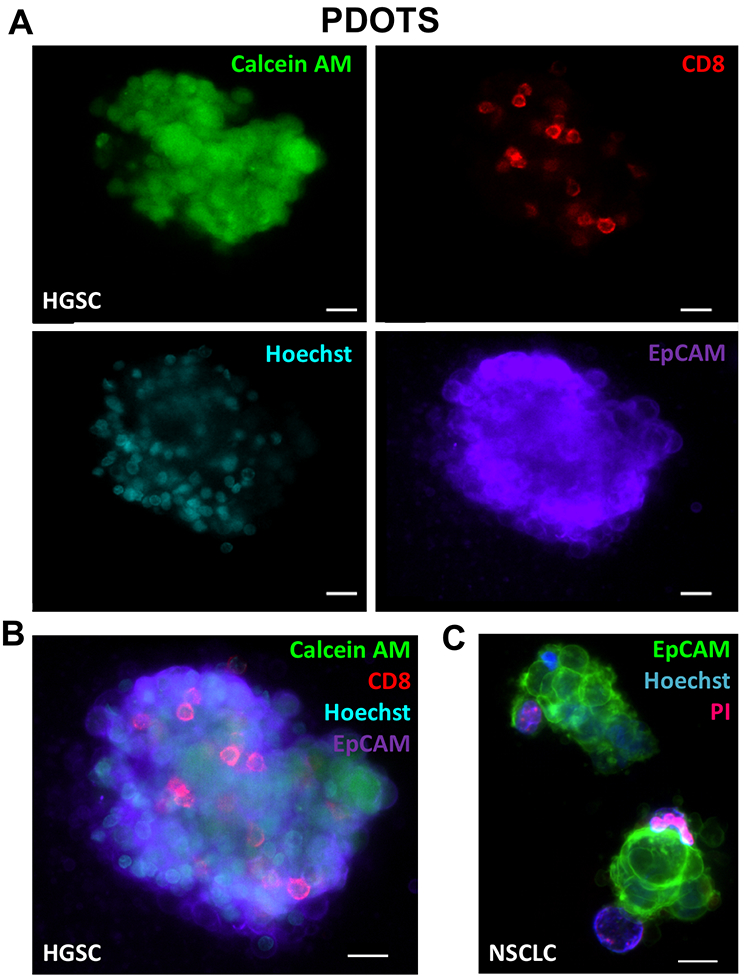
(A-B), baseline IF staining of HGSC PDOTS demonstrating viable cells (calcein AM; green), CD8 T cells (red), tumor cells (EpCAM; purple), and all nucleated cells (Hoechst; blue). C, overlay IF image of NSCLC PDOTS demonstrating EpCAM positive tumor cells (green), all nucleated cells (Hoechst; blue), and dead cells (PI; red). Scale bars indicate 20 μm (A, B, C).
Secreted cytokine profiling using multiplexed bead-based kits permits analysis of a large number of inflammatory cytokines and chemokines from a small volume of conditioned media, serum, or plasma. In profiling PDOTS or MDOTS, conditioned media is collected at indicated time points, or upon termination of ex vivo culture, and frozen (at −80°C), banked, and later batched for pooled analysis using either murine or human commercially available cytokine kits. For a given sample, routine Live/Dead analysis (Fig. 6a) can be formed after media collection and banking for bead-based cytokine profiling (Fig. 6b). In the PDOTS sample shown from a patient with a small intestinal neuroendocrine tumor (SI-NET), dual immune checkpoint blockade with αPD-1 + αCTLA-4 enhanced immune-mediated killing compared to single-agent PD-1 blockade and CTLA-4 blockade (Fig. 6a). For the PDOTS specimen featured (Fig. 6b), the evolution of cytokine and growth factor secretion is evident over time (Days 1, 3, 6, and 9), with the clearest differences between the treatment groups evident by Day 9 (e.g. IL-8, VEGF, Il-12 (p70), CCL4). A single PDOTS (or MDOTS) sample yields >200 μL of media, enough for four separate bead-based profiling runs (each requiring 50 μL of conditioned media).
Figure 6 – Ex Vivo Profiling of ICB Using PDOTS.
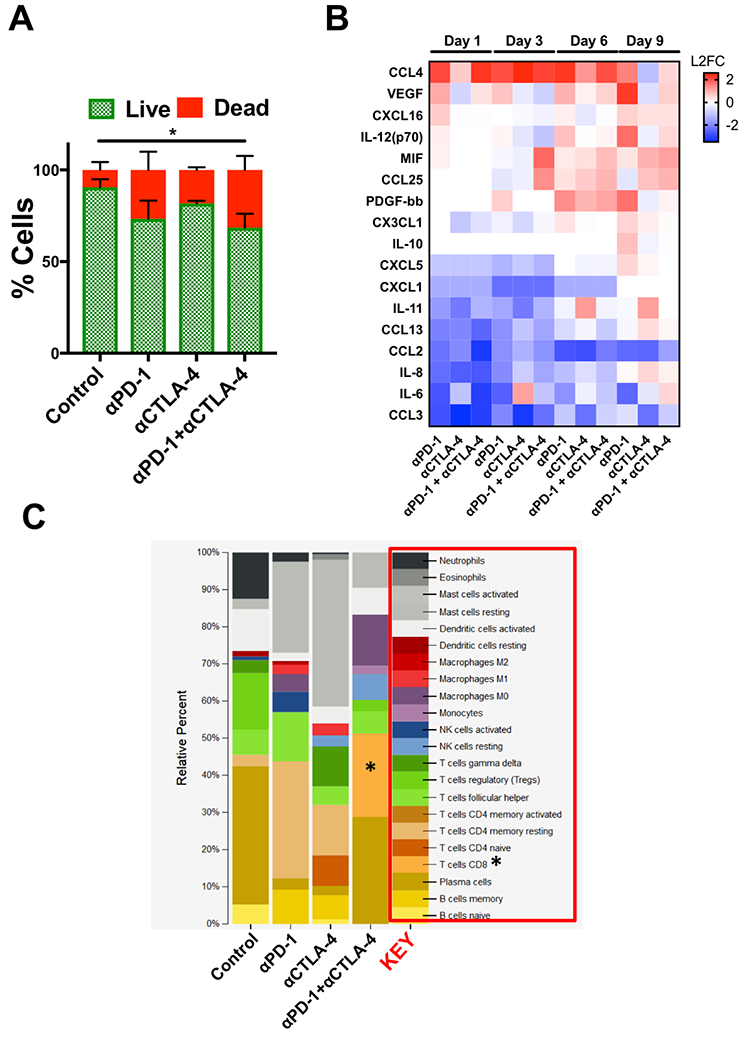
(A), AO/PI staining of SI-NET PDOTS (Day 9) treated with αPD-1, αCTLA-4, and αPD-1 + αCTLA-4 compared to untreated control PDOTS. (B), Heatmap of changes in secreted cytokines from PDOTS (SI-NET); represented as L2FC relative to untreated control at each time point. (C), Inferred changes in PDOTS immune cell populations using CIBERSORT from SI-NET PDOTS RNA-seq.
Despite the ease of using bead-based profiling to evaluate secreted cytokines and growth factors from conditioned media, most panels are not exhaustive and running multiple panels on individual samples is impractical and costly. Furthermore, for discovery efforts and pathway analysis, secretion profiling is unlikely to evaluate a sufficient number of effectors, growth factors, and chemoattractants. Lastly, molecular analysis (e.g. RNA-seq) provides a large data set that can be interrogated to infer changes in immune cell populations (e.g. CIBERSORT20). We performed a pilot study to determine if bulk RNA-sequencing (RNA-seq) could be performed on PDOTS and conducted CIBERSORT analysis to determine if ex vivo treatment with αPD-1 +/− αCTLA-4 influenced relative number of immune cells (inferred by RNA-seq). RNA was purified using a modified lysis protocol utilizing proteinase K to digest collagen gels and cellular membranes, total RNA from the mixed cell population was isolated (see Materials and Methods), and was used as input for library prep for RNA-seq. CIBERSORT was performed (as described20), and demonstrated relative expansion of CD8 T cells and M0 macrophages in PDOTS treated with dual ICB (αPD-1 + αCTLA-4) relative to control or single agent ICB-treated PDOTS. While this is an initial proof of concept experiment, it suggests that RNA-seq from PDOTS in 3D microfluidic culture is feasible, and that CIBERSORT or similar computational methods can be used to infer immune cell changes in response to ex vivo ICB.
Discussion
We have previously demonstrated that patient- and murine-derived organotypic tumor spheroids retain relevant lymphoid and myeloid subsets of immune cells, grow readily in 3D microfluidic culture in collagen hydrogels, recapitulate sensitivity and resistance to PD-1 blockade ex vivo using established immune competent murine tumor models, to test novel combination therapies using a PD-(L)1 backbone, and demonstrated the utility of PDOTS profiling to identify and nominate candidate biomarkers17. Here, we review this previously published method in greater detail with emphasis on the methods of MDOTS/PDOTS preparation and readouts, and discuss future directions with this novel technology (e.g. RNA-seq), and provide further evidence 3D microfluidic culture of MDOTS recapitulates response to PD-1 blockade which is lacking using traditional 2D culture methods.
The PDOTS/MDOTS platform offers several advantages over existing methods to evaluate tumor-immune responses to ICB (see Table 1), providing a window into the tumor immune microenvironment and enabling evaluation of acute and dynamic responses to ICB to using relevant murine models and patient samples. Unlike organoids21, 22, patient-derived xenografts (PDXs)23, and circulating-tumor cells (CTCs)24, MDOTS and PDOTS do not require days or weeks of tissue manipulation or propagation, and contain autologous, tumor-infiltrating immune cells. Importantly, MDOTS/PDOTS are not to be confused with organoids, a distinct 3D culture method developed by Hans Clevers and Toshiro Sato, which leverages native tissue stem cells to propagate normal or transformed tissue25. Organoids, like PDXs and CTCs, can be used as part of personalized medicine efforts, but lack native stromal and immune cells, thus precluding study of tumor-immune interactions. Furthermore, generation of sufficient quantity of organoids takes time, thus limiting rapid drug screening capabilities.
Table 1 –
Summary of the advantages/limitations of MDOTS/PDOTS microfluidic culture models relative to other in vivo and in vitro cancer models. Several typical references are provided for each type of culture.
| Type culture | Cancer models | Characteristics/Advantages | Limitations | ||
|---|---|---|---|---|---|
|
In vitro Ex vivo |
3D microfluidic culture of: 26–32 |
Culture Conditions |
|
Culture Limitations |
|
|
Cell line spheroids 18,29,30,32 MDOTS 16,17 |
Material & Methods |
|
Technical issues |
|
|
|
PDOTS 16,17 |
Results & Potentiality | Microfluidic devices are scalable (size, number of cells)
|
|
||
|
In vitro Ex vivo |
2D standard cell culture 31–35 |
Culture Conditions |
|
Culture Limitations |
|
|
Material & Methods |
|
|
|||
|
Results & Potentiality |
|
Technical issues | |||
|
In vitro Ex vivo |
Standard Transwell culture 36–40 |
Culture Conditions |
|
Culture Limitations |
|
|
Material & Methods |
|
Technical issues |
|
||
| Results & Potentiality |
|
||||
|
In vitro In vivo Ex vivo |
Circulating Tumor Cells (CTCs) 24,41–46 |
Culture Conditions |
|
Culture Limitations |
|
|
Material & Methods |
|
Technical issues |
|
||
| Results & Potentiality |
|
||||
|
In vitro Ex vivo |
Organoids 21,22,47–49 |
Culture Conditions |
|
Culture Limitations |
|
|
Material & Methods |
|
Technical issues |
|
||
| Results & Potentiality |
|
||||
| In vivo |
Xenografts Mouse Models 23, 31, 33, 50–54 |
Culture Conditions |
|
Culture Limitations |
|
|
Material & Methods |
|
Technical issues |
|
||
| Results & Potentiality |
|
||||
| In vivo |
Immune-Competent Mouse Models 31, 33, 54–56 |
Culture Conditions |
|
Culture Limitations |
|
|
Material & Methods |
|
Technical issues |
|
||
| Results & Potentiality |
|
MDOTS/PDOTS offer significant advantages over organoids, PDXs, and CTCs for ex vivo profiling tumor-immune responses to ICB, however there are several important limitations inherent in the current version of the platform. First, the current version of the MDOTS/PDOTS platform is currently only capable of evaluating tumor-immune interactions of immune cells that have already infiltrated the tumor. In other words, PDOTS/MDOTS “v1.0” cannot recapitulate T-cell priming (which occurs primarily in lymph nodes) or recruitment of naïve immune cells to the tumor microenvironment. Second, we have yet to perform comprehensive evaluation of tumor, stromal, and immune changes through the course of ex vivo culture, both in control conditions and in response to ICB. Thirdly, use of core needle biopsies and FNAs is challenging, and excisional specimens are preferred to yield sufficient number & quality of MDOTS/PDOTS for ex vivo profiling at present, Lastly, the influence of device dimensions, biophysical parameters, interstitial flow, hypoxia, and metabolic on tumor-immune interactions, especially cytokine production remains poorly understood and will require further investigation.
It should be noted that the size of the gel region and the size of the media channels are among the variables that may differ among different versions and types of microfluidic devices that could conceivably influence absolute and relative cytokine changes. The impact of different device parameters (e.g. device height, gel region width) on cytokine elaboration is under investigation, but remains far from complete. Identifying the physical components & dimensions of the microfluidic device that enables studying ICB will be important, especially when considering building more advanced medium- and high-throughput systems to test multiple drugs and/or antibodies in combination. An important co-variate in this analysis is the composition of the device itself. For example, the AIM microdevice is plastic, whereas other materials (e.g. polydimethylsiloxane, PDMS) used for ‘home grown’ device fabrication exhibit different properties and characteristics. This is a particularly important consideration when testing ICB in combination with small molecules (commonly prepared in DMSO), as PDMS is known to adsorb small hydrophobic molecules26, likely influencing drug delivery to MDOTS/PDOTS and ultimately drug efficacy.
While the long-term potential of MDOTS/PDOTS technology as a predictive tool is not yet clear, MDOTS/PDOTS are showing great promise in the evaluation of novel combination therapies and in the discovery of novel biomarkers. Important future directions include extending the lifetime of ex vivo cultures (e.g. tumor vascular networks), performing clinical validation studies (e.g. co-clinical trials or adaptive clinical trials using ex vivo responses to guide clinical treatment), and developing medium- and high-throughput versions of the system. With further technology development along with improved data analytics and bioinformatics, we anticipate the MDOTS/PDOTS platform form the foundation of functional precision immune-oncology efforts.
Acknowledgements
Financial support was provided by the Robert A. and Renée E. Belfer Foundation, NCI Training Grant T32CA009172–41 (R.W.J.), John R. Svenson Fellowship (R.W.J.), NCI-R01 CA190394–01 (D.A.B.), Ermenegildo Zegna Founder’s scholarship (M.C.), MIT-POLITO grant BIOMODE - Compagnia di San Paolo (M.C.), NCI-U01 CA214381–01 (R.D.K., D.A.B.) the Gloria T. Maheu and Heerwagen Family Funds for Lung Cancer Research, (D.A.B.), Expect Miracles Foundation (C.P.P.). Additional support from a Stand Up To Cancer-American Cancer Society Lung Cancer Dream Team Translational Research Grant (SU2C-AACR-DT17–15). Stand Up to Cancer is a program of the Entertainment Industry Foundation. Research grants are administered by the American Association for Cancer Research, the scientific partner of SU2C.
Footnotes
Disclosure of Potential Conflicts
A. Aref, D.A. Barbie, and R.W. Jenkins have ownership interest in pending U.S. Patent Application No. 15/540,346.. R.D. Kamm has ownership interest (including patents) in AIM Biotech, D.A. Barbie is a consultant/advisory board member for N of One.
References
- 1.Pardoll DM, Nat Rev Cancer, 2012, 12, 252–264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Topalian SL, Drake CG and Pardoll DM, Cancer Cell, 2015, 27, 450–461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Carbognin L, Pilotto S, Milella M, Vaccaro V, Brunelli M, Calio A, Cuppone F, Sperduti I, Giannarelli D, Chilosi M, Bronte V, Scarpa A, Bria E and Tortora G, PLoS One, 2015, 10, e0130142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mahoney KM, Rennert PD and Freeman GJ, Nat Rev Drug Discov, 2015, 14, 561–584. [DOI] [PubMed] [Google Scholar]
- 5.Jenkins RW, Barbie DA and Flaherty KT, Br J Cancer, 2018, 118, 9–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Larkin J, Hodi FS and Wolchok JD, N Engl J Med, 2015, 373, 1270–1271. [DOI] [PubMed] [Google Scholar]
- 7.Postow MA, Chesney J, Pavlick AC, Robert C, Grossmann K, McDermott D, Linette GP, Meyer N, Giguere JK, Agarwala SS, Shaheen M, Ernstoff MS, Minor D, Salama AK, Taylor M, Ott PA, Rollin LM, Horak C, Gagnier P, Wolchok JD and Hodi FS, N Engl J Med, 2015, 372, 2006–2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Marincola FM, Jaffee EM, Hicklin DJ and Ferrone S, Adv Immunol, 2000, 74, 181–273. [DOI] [PubMed] [Google Scholar]
- 9.Bronte V, Kasic T, Gri G, Gallana K, Borsellino G, Marigo I, Battistini L, Iafrate M, Prayer-Galetti T, Pagano F and Viola A, J Exp Med, 2005, 201, 1257–1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pitt JM, Vetizou M, Daillere R, Roberti MP, Yamazaki T, Routy B, Lepage P, Boneca IG, Chamaillard M, Kroemer G and Zitvogel L, Immunity, 2016, 44, 1255–1269. [DOI] [PubMed] [Google Scholar]
- 11.O’Donnell JS, Long GV, Scolyer RA, Teng MW and Smyth MJ, Cancer Treat Rev, 2017, 52, 71–81. [DOI] [PubMed] [Google Scholar]
- 12.Tumeh PC, Harview CL, Yearley JH, Shintaku IP, Taylor EJ, Robert L, Chmielowski B, Spasic M, Henry G, Ciobanu V, West AN, Carmona M, Kivork C, Seja E, Cherry G, Gutierrez AJ, Grogan TR, Mateus C, Tomasic G, Glaspy JA, Emerson RO, Robins H, Pierce RH, Elashoff DA, Robert C and Ribas A, Nature, 2014, 515, 568–571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Taube JM, Young GD, McMiller TL, Chen S, Salas JT, Pritchard TS, Xu H, Meeker AK, Fan J, Cheadle C, Berger AE, Pardoll DM and Topalian SL, Clin Cancer Res, 2015, 21, 3969–3976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Carvalho MR, Lima D, Reis RL, Correlo VM and Oliveira JM, Trends Biotechnol, 2015, DOI: 10.1016/j.tibtech.2015.09.009. [DOI] [PubMed] [Google Scholar]
- 15.Feder-Mengus C, Ghosh S, Reschner A, Martin I and Spagnoli GC, Trends Mol Med, 2008, 14, 333–340. [DOI] [PubMed] [Google Scholar]
- 16.Jenkins RW, Aref AR, Lizotte PH, Ivanova E, Stinson S, Zhou CW, Bowden M, Deng J, Liu H, Miao D, He MX, Walker W, Zhang G, Tian T, Cheng C, Wei Z, Palakurthi S, Bittinger M, Vitzthum H, Kim JW, Merlino A, Quinn M, Venkataramani C, Kaplan JA, Portell A, Gokhale PC, Phillips B, Smart A, Rotem A, Jones RE, Keogh L, Anguiano M, Stapleton L, Jia Z, Barzily-Rokni M, Canadas I, Thai TC, Hammond MR, Vlahos R, Wang ES, Zhang H, Li S, Hanna GJ, Huang W, Hoang MP, Piris A, Eliane JP, Stemmer-Rachamimov AO, Cameron L, Su MJ, Shah P, Izar B, Thakuria M, LeBoeuf NR, Rabinowits G, Gunda V, Parangi S, Cleary JM, Miller BC, Kitajima S, Thummalapalli R, Miao B, Barbie TU, Sivathanu V, Wong J, Richards WG, Bueno R, Yoon CH, Miret J, Herlyn M, Garraway LA, Van Allen EM, Freeman GJ, Kirschmeier PT, Lorch JH, Ott PA, Hodi FS, Flaherty KT, Kamm RD, Boland GM, Wong KK, Dornan D, Paweletz CP and Barbie DA, Cancer Discov, 2018, 8, 196–215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Deng J, Wang ES, Jenkins RW, Li S, Dries R, Yates K, Chhabra S, Huang W, Liu H, Aref AR, Ivanova E, Paweletz CP, Bowden M, Zhou CW, Herter-Sprie GS, Sorrentino JA, Bisi JE, Lizotte PH, Merlino AA, Quinn MM, Bufe LE, Yang A, Zhang Y, Zhang H, Gao P, Chen T, Cavanaugh ME, Rode AJ, Haines E, Roberts PJ, Strum JC, Richards WG, Lorch JH, Parangi S, Gunda V, Boland GM, Bueno R, Palakurthi S, Freeman GJ, Ritz J, Haining WN, Sharpless NE, Arthanari H, Shapiro GI, Barbie DA, Gray NS and Wong KK, Cancer Discov, 2018, 8, 216–233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Aref AR, Huang RY, Yu W, Chua KN, Sun W, Tu TY, Bai J, Sim WJ, Zervantonakis IK, Thiery JP and Kamm RD, Integr Biol (Camb), 2013, 5, 381–389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cejas P, Li L, O’Neill NK, Duarte M, Rao P, Bowden M, Zhou CW, Mendiola M, Burgos E, Feliu J, Moreno-Rubio J, Guadalajara H, Moreno V, Garcia-Olmo D, Bellmunt J, Mullane S, Hirsch M, Sweeney CJ, Richardson A, Liu XS, Brown M, Shivdasani RA and Long HW, Nat Med, 2016, 22, 685–691. [DOI] [PubMed] [Google Scholar]
- 20.Newman AM, Liu CL, Green MR, Gentles AJ, Feng W, Xu Y, Hoang CD, Diehn M and Alizadeh AA, Nat Methods, 2015, 12, 453–457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Clevers H, Cell, 2016, 165, 1586–1597. [DOI] [PubMed] [Google Scholar]
- 22.Dutta D, Heo I and Clevers H, Trends Mol Med, 2017, 23, 393–410. [DOI] [PubMed] [Google Scholar]
- 23.Byrne AT, Alferez DG, Amant F, Annibali D, Arribas J, Biankin AV, Bruna A, Budinska E, Caldas C, Chang DK, Clarke RB, Clevers H, Coukos G, Dangles-Marie V, Eckhardt SG, Gonzalez-Suarez E, Hermans E, Hidalgo M, Jarzabek MA, de Jong S, Jonkers J, Kemper K, Lanfrancone L, Maelandsmo GM, Marangoni E, Marine JC, Medico E, Norum JH, Palmer HG, Peeper DS, Pelicci PG, Piris-Gimenez A, Roman-Roman S, Rueda OM, Seoane J, Serra V, Soucek L, Vanhecke D, Villanueva A, Vinolo E, Bertotti A and Trusolino L, Nat Rev Cancer, 2017, 17, 254–268. [DOI] [PubMed] [Google Scholar]
- 24.Micalizzi DS, Maheswaran S and Haber DA, Genes Dev, 2017, 31, 1827–1840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sato T, Vries RG, Snippert HJ, van de Wetering M, Barker N, Stange DE, van Es JH, Abo A, Kujala P, Peters PJ and Clevers H, Nature, 2009, 459, 262–265. [DOI] [PubMed] [Google Scholar]
- 26.Halldorsson S, Lucumi E, Gomez-Sjoberg R and Fleming RM, Biosens Bioelectron, 2015, 63, 218–231. [DOI] [PubMed] [Google Scholar]
- 27.Xu X, Farach-Carson MC and Jia X, Biotechnol Adv, 2014, 32, 1256–1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Caballero D, Kaushik S, Correlo VM, Oliveira JM, Reis RL and Kundu SC, Biomaterials, 2017, 149, 98–115. [DOI] [PubMed] [Google Scholar]
- 29.Jeong SY, Lee JH, Shin Y, Chung S and Kuh HJ, PLoS One, 2016, 11, e0159013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pavesi A, Tan AT, Koh S, Chia A, Colombo M, Antonecchia E, Miccolis C, Ceccarello E, Adriani G, Raimondi MT, Kamm RD and Bertoletti A, JCI Insight, 2017, 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Aref AR and Barbie DA, Ex Vivo Engineering of the Tumor Microenvironment, Humana Press, Cham. [Google Scholar]
- 32.Canadas I, Thummalapalli R, Kim JW, Kitajima S, Jenkins RW, Christensen CL, Campisi M, Kuang Y, Zhang Y, Gjini E, Zhang G, Tian T, Sen DR, Miao D, Imamura Y, Thai T, Piel B, Terai H, Aref AR, Hagan T, Koyama S, Watanabe M, Baba H, Adeni AE, Lydon CA, Tamayo P, Wei Z, Herlyn M, Barbie TU, Uppaluri R, Sholl LM, Sicinska E, Sands J, Rodig S, Wong KK, Paweletz CP, Watanabe H and Barbie DA, Nat Med, 2018, DOI: 10.1038/s41591-018-0116-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kitajima S, Yoshida A, Kohno S, Li F, Suzuki S, Nagatani N, Nishimoto Y, Sasaki N, Muranaka H, Wan Y, Thai TC, Okahashi N, Matsuda F, Shimizu H, Nishiuchi T, Suzuki Y, Tominaga K, Gotoh N, Suzuki M, Ewen ME, Barbie DA, Hirose O, Tanaka T and Takahashi C, Oncogene, 2017, 36, 5145–5157. [DOI] [PubMed] [Google Scholar]
- 34.Terai H, Kitajima S, Potter DS, Matsui Y, Quiceno LG, Chen T, Kim TJ, Rusan M, Thai TC, Piccioni F, Donovan KA, Kwiatkowski N, Hinohara K, Wei G, Gray NS, Fischer ES, Wong KK, Shimamura T, Letai A, Hammerman PS and Barbie DA, Cancer Res, 2018, 78, 1044–1057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Domcke S, Sinha R, Levine DA, Sander C and Schultz N, Nat Commun, 2013, 4, 2126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Katt ME, Placone AL, Wong AD, Xu ZS and Searson PC, Front Bioeng Biotechnol, 2016, 4, 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Arrigoni C, Bersini S, Gilardi M and Moretti M, Int J Mol Sci, 2016, 17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Herroon MK, Diedrich JD and Podgorski I, Front Endocrinol (Lausanne), 2016, 7, 84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Roudnicky F and Hollmen M, Genom Data, 2016, 8, 1–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Guiet R, Van Goethem E, Cougoule C, Balor S, Valette A, Al Saati T, Lowell CA, Le Cabec V and Maridonneau-Parini I, J Immunol, 2011, 187, 3806–3814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lallo A, Schenk MW, Frese KK, Blackhall F and Dive C, Transl Lung Cancer Res, 2017, 6, 397–408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Krebs MG, Metcalf RL, Carter L, Brady G, Blackhall FH and Dive C, Nat Rev Clin Oncol, 2014, 11, 129–144. [DOI] [PubMed] [Google Scholar]
- 43.Pantel K and Alix-Panabieres C, Clin Chem, 2016, 62, 328–334. [DOI] [PubMed] [Google Scholar]
- 44.Pailler E, Adam J, Barthelemy A, Oulhen M, Auger N, Valent A, Borget I, Planchard D, Taylor M, Andre F, Soria JC, Vielh P, Besse B and Farace F, J Clin Oncol, 2013, 31, 2273–2281. [DOI] [PubMed] [Google Scholar]
- 45.Ross K, Pailler E, Faugeroux V, Taylor M, Oulhen M, Auger N, Planchard D, Soria JC, Lindsay CR, Besse B, Vielh P and Farace F, Expert Rev Mol Diagn, 2015, 15, 1605–1629. [DOI] [PubMed] [Google Scholar]
- 46.Pailler E, Faugeroux V, Oulhen M, Catelain C and Farace F, Transl Lung Cancer Res, 2017, 6, 444–453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Drost J and Clevers H, Nat Rev Cancer, 2018, 18, 407–418. [DOI] [PubMed] [Google Scholar]
- 48.Broutier L, Mastrogiovanni G, Verstegen MM, Francies HE, Gavarro LM, Bradshaw CR, Allen GE, Arnes-Benito R, Sidorova O, Gaspersz MP, Georgakopoulos N, Koo BK, Dietmann S, Davies SE, Praseedom RK, Lieshout R, JNM IJ, Wigmore SJ, Saeb-Parsy K, Garnett MJ, van der Laan LJ and Huch M, Nat Med, 2017, 23, 1424–1435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sachs N, de Ligt J, Kopper O, Gogola E, Bounova G, Weeber F, Balgobind AV, Wind K, Gracanin A, Begthel H, Korving J, van Boxtel R, Duarte AA, Lelieveld D, van Hoeck A, Ernst RF, Blokzijl F, Nijman IJ, Hoogstraat M, van de Ven M, Egan DA, Zinzalla V, Moll J, Boj SF, Voest EE, Wessels L, van Diest PJ, Rottenberg S, Vries RGJ, Cuppen E and Clevers H, Cell, 2018, 172, 373–386 e310. [DOI] [PubMed] [Google Scholar]
- 50.Daniel VC, Marchionni L, Hierman JS, Rhodes JT, Devereux WL, Rudin CM, Yung R, Parmigiani G, Dorsch M, Peacock CD and Watkins DN, Cancer Res, 2009, 69, 3364–3373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Morton CL and Houghton PJ, Nat Protoc, 2007, 2, 247–250. [DOI] [PubMed] [Google Scholar]
- 52.Voskoglou-Nomikos T, Pater JL and Seymour L, Clin Cancer Res, 2003, 9, 4227–4239. [PubMed] [Google Scholar]
- 53.Girotti MR, Gremel G, Lee R, Galvani E, Rothwell D, Viros A, Mandal AK, Lim KH, Saturno G, Furney SJ, Baenke F, Pedersen M, Rogan J, Swan J, Smith M, Fusi A, Oudit D, Dhomen N, Brady G, Lorigan P, Dive C and Marais R, Cancer Discov, 2016, 6, 286–299. [DOI] [PubMed] [Google Scholar]
- 54.Gengenbacher N, Singhal M and Augustin HG, Nat Rev Cancer, 2017, 17, 751–765. [DOI] [PubMed] [Google Scholar]
- 55.Gould SE, Junttila MR and de Sauvage FJ, Nat Med, 2015, 21, 431–439. [DOI] [PubMed] [Google Scholar]
- 56.Frese KK and Tuveson DA, Nat Rev Cancer, 2007, 7, 645–658. [DOI] [PubMed] [Google Scholar]


