Abstract
The role of phytochemicals as potential prodrugs or therapeutic substances against tumors has come in the spotlight in the very recent years, thanks to the huge mass of encouraging and promising results of the in vitro activity of many phenolic compounds from plant raw extracts against many cancer cell lines. Little but important evidence can be retrieved from the clinical and nutritional scientific literature, where flavonoids are investigated as major pro-apoptotic and anti-metastatic compounds. However, the actual role of these compounds in cancer is still far to be fully elucidated. Many of these phytochemicals act in a pleiotropic and poorly specific manner, but, more importantly, they are able to tune the reactive oxygen species (ROS) signaling to activate a survival or a pro-autophagic and pro-apoptosis mechanism, depending on the oxidative stress-responsive endowment of the targeted cell. This review will try to focus on this issue.
Keywords: phytochemicals, flavonoids, cancer, mitochondria, apoptosis
1. Introduction
The ability of phytochemicals to prevent cancer has been long claimed as the most outstanding potential of these plant-derived molecules, despite the fact that quite the whole bulk of these substances is made of toxic compounds [1,2,3,4]. However, the purported anticancer activity of many phenolics produced in the natural world, accounting for more than 5000 different chemically characterized compounds in edible plants [5], has been reported to fundamentally depend on their antioxidant property [6]. Notwithstanding, the ability of phytochemicals to induce the expression of the Nrf2-ARE signaling pathway [7,8,9], despite the evidence that all stressors can activate the Nrf2-ARE system [10], the potential of these plant-derived phenolic substances to promote, enhance and trigger the expression and the activity of superoxide dismutase (SOD), catalase (CAT) and glutathione peroxydase (GPX) [11], represent a fundamental hallmark to trust these substances as possible chemopreventive molecules [12,13,14,15,16]. How phytochemicals can counteract tumors is still far to be fully elucidated. Phytochemicals have a huge panoply of intracellular targets and their interaction with many of them is yet puzzling to date and particularly intriguing [17]. A first issue to be focused on is the relationship between mitochondria, reactive oxygen species (ROS) and cancer [18,19,20,21]. Plant-derived phytoestrogens, such as catechins, can induce mitochondrial biogenesis, thus restoring mitochondria function, via the induction of survival signaling systems activated during mitohormesis, such as AMPK/AAK-2, SIRT1/SIR-2.1 and FOXO/DAF-16, in C. elegans [22]. Targeting mitochondria should demonstrate also the ability of these molecules to tune cell morphogenesis and improve mitochondria function and biogenesis [23,24,25]. Actually, alterations in the mitochondrial dynamics modulate some type of tumors. For example, the dysregulated mitochondrial fusion by Mfn2 knockdowns suppresses the rate of oxygen consumption in melanoma cells, suggesting that mitochondrial dynamics, i.e., the rate of fission and fusion, modulate cell migration and progression in this type of cancer [26]. Dihydromyricetin is able to reverse mitochondrial dysfunction, which should be mediated by PGC-1α/TFAM and PGC-1α/mfn2 signaling pathways, therefore ameliorating mitochondria dynamics [27]. Mitochondria dysfunction is a typical hallmark of many cancers and the ability of phytochemicals to restore it appears quite fundamental [28,29,30]. The fìne regulation of the survival process in a cell involves a series of signaling pathways that not only encompasses the enzymatic endowment for ROS scavenging but also the complex machinery modulation of the crosstalk between mitochondria and other organelles leading to the autophagy/apoptosis balance [31,32,33].
The role of phytochemicals in this context is particularly interesting [34,35].
Phytochemicals not only may counteract cancer malignancy and progression but can induce tumor cells necroptosis, besides apoptosis [36,37]. Furthermore, the role of autophagy in cancer development has been extensively reviewed in recent years [38]. Although autophagy would lead to a suppression of tumorigenesis, some circumstances showed an opposite action on cancer [38,39]. Therefore, the ability of phytochemicals to target cellular autophagy as an approach in using the natural substances as chemopreventive compounds should be considered with particular attention, despite the many encouraging results [40,41,42]. Their activity might also target intracellular calcium signaling and endoplasmic reticulum (ER) stress [43,44], which exerts a major role in the mitochondria-mediated tuning of the many cell survival functions [45]. A role in maintaining the mitochondria–ER stress homeostasis has been recently attributed to Lon proteases (LONPs), where LONP is a protein complex made by a homo-hexameric ring-shaped structure with a serine–lysine catalytic dyad, which is highly conserved in both prokaryotic and eukaryotic organisms [46,47]. LONPs are upregulated during ER stress, via the activation of the PERK-ATF4 signaling pathway [48,49], which may be targeted by flavonoids [50,51,52]. In this perspective, plant-derived polyphenols might target many anti-oxidant cell signaling systems, which exert a major role in mitochondria biogenesis and mitochondria–ER stress homeostasis. The close interaction between mitochondria and ER may be regulated by caveolin-1, which is located at the mitochondria/ER interface where it impairs the remodeling of the mitochondria–ER relationship by making mitochondria non responsive to ER stress via the dampening of the calcium signaling [53,54]. This mechanism is counterbalanced by the PKA-DRP1-mediated signaling [54,55], which is targeted by flavonoids [56]. In cancer cells, this homeostasis can be profoundly perturbed and the activity of flavonoids can be functionally inverted with respect to the one acting on normal, non-cancerous cells [57]. Actually, tumors have a different stress response with respect to non tumoral cells, so that any therapic approach must take into account this issue [58,59].
In this review, we will attempt to elucidate the very recent novelties in the field of cancer prevention and therapy using nature-derived phytochemicals.
2. Insights on the Role of Flavonoids in Cancer
2.1. Flavonoids and Apoptosis
Table 1 summarizes some of the very recent results about the flavonoids ability in inhibiting cancer development and malignancy [60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87]. Many of these molecules act against cancer cells by promoting and activating apoptosis. The signaling pathways through which flavonoids induce apoptosis in cancerous cells are various. Besides the effect on Bax, Bcl-2 and caspases, a further possibility is represented by the inhibition of fatty acid synthase (FAS) exerted by a great number of flavonoids, such as epigallocatechin-3-gallate (EGCG), luteolin, quercetin, kaempferol, apigenin, and taxifolin, which exert their anti-lipogenic activities against many human tumors [88,89]. FAS is over-expressed in many human epithelial cancers and also in breast tumors. Its inhibition, causing the accumulation of malonyl-CoA, leads to the upregulation of ceramide levels and the inhibition of carnitine palmitoyltransferase-1, therefore inducing the expression of the pro-apoptotic genes BNP3, TRAIL and DAPK2 and causing apoptosis [90]. Interestingly, FAS inhibition causes a massive ROS upregulation, which has been reported as a key factor in promoting cancer cell apoptosis, curiously suggesting that antioxidants promote oxidative stress to kill cancerous cells [91]. Actually, flavonoids inhibit apoptosis in non cancerous cells, probably because of their induction of eustress and hence of the survival response from cells [17,92], a mechanism closely depending on the flavonoids dose [93] and their interaction with ROS as signaling molecules [17,94]. Cancer cells are characterized by a high degree of oxidative stress and ROS production, and they possess a high metabolic and peroxysomal activity, often leading to mitochondrial dysfunction and an enhanced activity of lypooxygenases, thymidine phosphorylases, oxydases and cyclooxygenases [95]. However, despite the expected need in ROS scavenging enzymes, flavonoids exert their anticancer activity very rarely with the sole increase in the anti-oxidant machinery. More complex coupled mechanisms would occur. For example, esculetin and quercetin act finely tuning the redox homeostasis in NB4 leukemia cells [96]. Paradoxically, 25 μM quercetin increased NF-κB p65 in the nucleus and reduced it in the cytosol, whereas it reduced Nrf2 in the nucleus and enhanced it in the cytosol [96]. Furthermore, esculetin, in contrast with quercetin, increases the level of the superoxide dismutase (SOD) expression [96]. A fine tuning in the cell, where flavonoids may exert an apparently contradictory action, does exist. Moreover, the hypothesis that cancer might be closely associated with an imbalance in the energy homeostass, therefore including also ROS signaling and mitochondria oscillation, has been recently addressed [17,97].
Table 1.
Some of the very recent examples of the roles exerted by flavonoids in cancer cells.
| Classification | Compound | Activity | References | |
|---|---|---|---|---|
Flavones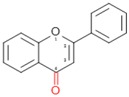
|
Apigenin | ↑ | Anticancer activity via the Wnt/β-catenin pathway and JAK-STAT. Induction of apoptsis in TRAIL-resistant cancers | [60,61,62,63,64] |
| Luteolin | ↑ | Induction of apoptosis and autophagy in ANA-1 cells via the p38, JNK and Akt signaling pathways, inhibiting Bcl-2 and beclin-1 and activating caspase-3 and caspase-8 | ||
| ↓ | Proliferation of BT474 and MCF-7 breast cancer cells | |||
| ↑ | Apoptosis in BT474 and MCF-7 breast cancer cells | |||
| ↑ | Apoptosis in ACS gastric cancer | |||
| Tangeritin | ↓ | Cell cycle in MCF7 and MDA-MB-468 breast cancer cells via the CYP1A1/CYP1B1-mediated metabolism | ||
Flavonols
|
Quercetin | ↓ | Many types of cancer via apoptosis and inhibition of cell replication | [41,65,66,67,68,69,70,71] |
| Kaempferol | ↑ | Apoptosis and autophagy in human lung cancer cells A549 via upregulation of miR-340 | ||
| ↑ | Apoptosis in HCT116, HCT15, and SW480 colorectal cancer cells | |||
| Myricetin | ↓ | Prostate cancer cell metastasis by cytotoxic activity | ||
| Fisetin | ↓ | Growth and metastasis and EMT in MDA-MB-231 and BT549 breast cancer cells | ||
| Galangin | ↓ | Proliferation of human kidney A498 cancer cells by the induction of apoptosis-targeted PI3K/Akt/mTOR signaling | ||
| Isorhamnetin | ↓ | Growth of MCF-7 breast cancer cells | ||
Flavanones (citrus fruit flavonoids)
|
Hesperetin | ↑ | Apoptosis in H522 lung cancer cells | [72,73] |
| Naringenin | ↓ | Prostate cancer metastasis via voltage-gated sodium channel blockage | ||
Flavanonols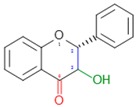
|
Taxifolin | ↓ | Mammary carcinogenesis via the LXR-mTOR/Maf1/PTEN axis and the CYP1A1- and CYP1B1-mediated cancer | [74,75] |
| Flavans (Flavanols) Green tea catechins 
|
Epigallocatechingallate (EGCG) | ↑ | Chemoprevention in glioblastoma | [76,77,78,79,80] |
| ↑ | Apoptosis in chronic myeloid leukemia by Bcr/Abl-mediated p38-MAPK/JNK and JAK2/STAT3/AKT signaling | |||
| Catechin, EGCG | ↓ | Lung tumor growth via the inhibition of programmed cell death-ligand1 (PD-L1) | ||
| Epicatechin-3-O-gallate (ECG) | ↓ | LNCaP and PC-3 prostate cancer cell growth | ||
| Epigallocatechin (EGC) | ↓ | Suppression of HPV and tumors with curcumin and resveratrol | ||
Anthocyanidins
|
Cyanidin | ↓ | Angiogenesis in breast cancer via the STAT3/VEGF pathway and miR124 mediated STAT3 downregulation | [81,82,83] |
| Delphinidin | ↑ | Apoptosis and autophagy in HER-2 positive breast cancer MDA-MB-453 and BT474 cells | ||
| ↑ | Apoptosis and EMT in human osteosarcoma cell lines via the ERK2/p38MAPK pathway | |||
Isoflavonoids
|
Genistein | ↑ | Apoptosis in Mcl1 human laryngeal cancer cells | [84,85,86,87] |
| ↓ | Proliferation of EP3-expressing melanoma | |||
| Alters epigenetic in MDA-MB-231 breast cancer cells | ||||
| Daidzein | ↑ | Apoptosis in colon cancer cells | ||
Dow arrows = inhibition; Top arrows: activation and/or promotion. Red colours in chemical formulas indicate the typical functional groups and/or chemical positions for typical attachments of substituents (isoflavonoids).
Most of flavonoids trigger the apoptotic pathways in cancer cells via their fundamental signaling pathways. The isoflavone analog phenoxodiol induces apoptosis in renal cancer by the inhibition of the Akt pathway, whereas apoptosis is induced in non cancerous cells by phosphorylation of Akt [98,99]. The induction of apoptosis in cancer cells via the PI3K/Akt signaling pathway is held by a wide spectrum of phytochemicals. Luteolin induces a caspase-dependent apoptosis in human hepatocellular carcinoma by inhibiting the Akt phosphorylation [100], and the flavonol glucoside icanin (40 μM) induces apoptosis via a ROS-mediated damage on mitochondria membrane potential by suppressing the PI3K/Akt and STAT3 signaling pathways [101]. Furthermore, baicalein, by acting on the PI3K/Akt/NF-κB, enhances the sensitivity to cisplatin of A549 lung adenocarcinoma cell line [102].Flavonoids, acting on cells via the PI3K/Akt pathway, should regulate cell survival and apoptosis at a post-mitochondrial level, i.e., downstream of the mitochondria cytochrome c release and before the activation of caspase 9 [103]. ROS have a fundamental role in the Akt-mediated signaling leading to apoptosis, as they activate the Akt/ASK1/p38MAPK pathway, causing a modulation of ASK1 dephosphorylation, which subsequently activates p38MAPK and downregulating p21 (Cip1), leading to apoptosis [104,105]. The ability of flavonoids to cause apoptosis in cancer cells via the Akt pathway may be paradoxically mediated by a rapid imbalance in the intracellular ROS homeostass or, more probably, by the complex relationship between ASK1 and NF-κB, where IKK has a major control on the ASK1-JNK axis, associating IKK with ROS and ER stress [106]. Actually, despite the previous belief about the general increase in NF-κB expression in cancer, recent reports showed that tumors differentially express specific subunits of the NF-κB pathway, suggesting the possible existence of a finely regulated tuning towards apoptosis by different flavonoids in different target cells [107]. Actually, 3,4’,7-O-trimethyl quercetin, a derivative of quercetin, induces apoptosis in ovarian cancer cell lines CRL-1978, CRL-11731, SK-OV-3, following three different target pathways, depending on the cell type [108].
The widespread knowledge about flavonoids in cancer is that these compounds are generally very able to have anti-oxidant and anti-inflammatory activities, to induce ROS-scavenging enzymes and CYP-mediated detoxyfication, to induce cell cycle arrest, apoptosis and autophagy, and to inhibit proliferation, migration and malignancy [109]. Apoptosis is generally activated by disturbing the mitochondria–ER stress balance and ROS homeostasis, accessing this machinery via the inhibition of many survival pathways. Actually, pathways by which flavonoids induce apoptosis are altogether related to cell survival processes. Glabridin causes apoptosis in oral cancer cells via the JNK1/2 pathway, which with NF-κB is involved in cell survival, apoptosis, inflammation and angiogenesis [110,111]; hesperedin induces apoptosis in endometrial carcinoma by downregulating the estrogen receptor relationship via the ERK/MAPK pathway [112]; apigenin triggers apoptosis and autophagy in hepatocellular carcinoma cells via the PI3K/Akt/mTOR signaling pathway [113]; chrysin and other fundamental components of propolis, such as the phenolic acids caffeic, ferulic and α-coumaric acid, induce a proline dehydrogenase/proline oxidase-dependent apoptosis in human tnongue squamous cell carcinoma [114]. The panoply of different targets by which flavonoids induce apoptosis may be a consequence of their modulatory role in balancing the apoptosis/necrosis ratio [115].
Research on apoptosis and autophagy caused by phytochemicals seems to be mostly polarized on certain types of cancer, i.e., breast, prostate and colon cancer and more frequently involving curcumin, flavonoids, and resveratrol [116]. Curcumin and resveratrol also induce apoptosis via the same survival pathways targeted by several flavonoids, e.g., PI3K/Akt [117,118], MAPK/JAK2/STAT3 [119,120], p38MAPK/ERK1/2/JNK [121,122], Wnt/β-catenin [123,124], NF-κB [125,126]. Besides these major pathways, phytochemicals are able to induce apoptosis in cancer cells, targeting many further signaling and biochemical mechanisms. For example, resveratrol is able to target cellular FLICE-inhibitory protein (c-FLIP), which is a master tuner in the inhibition of apoptosis with the death receptors TNF-R1, Dr5, DR4 and Fas. In lung cancer cells, resveratrol causes p-Akt and c-FLIP downregulation, inducing apoptosis along with an increase in the production of ROS and hydrogen peroxide-elicited Bid, activation of PARP and caspase 8, and downregulation of pEGFR and NF-κB protein expression [127,128].
The most common route through which flavonoids induce apoptosis in cancer cells, i.e., the PI3K/Akt pathway, is probably the major signaling mechanism leading to cell survival gene expression with different approaches, e.g., PI3K/Akt/Raf1/MEK/ERK [129,130], PI3K/Akt/mTOR [131], and PI3K/Akt/ERK1-2/NF-κB [132]. Furthermore, the flavonoid irigenin targets the TRAIL signaling pathway leading to apoptosis in gastric cancer, with the enhancement of FAS-associated protein with death domain (FADD), death receptor 5 (DR5) and Bax proapoptotic proteins [133], a way targeted also by pinostrobin [134], apigenin [135], kaempferol [136].
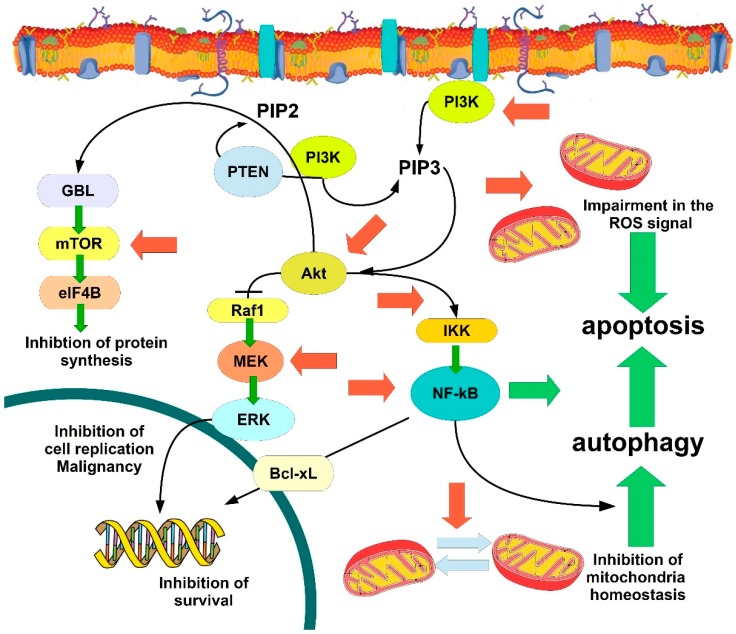
Figure 1 shows the major pathways targeted by flavonoids, coumarins and stilbenes (resveratrol) in inducing apoptosis in cell cancer.
Figure 1.
Cartoon showing the major signaling pathways targeted by phytochemicals in cancer. Phytochemicals principally act as inhibitors which red arrows indicate in the PI3K/Akt signaling pathway. Green arrows indicate the action promoted or activated by flavonoids, also following the inhibitory signaling cascades. For details and acronyms, see the text.
2.2. Autophagy and the Mitochondria–ER Stress Relatonships Leading to Cancer Cell Death
Flavonoids can target the autophagic machinery in cancer cells, so managing and ruling the survival homeostatic processes yet often leads to the apoptotic destiny of tumoral cells [116]. For example, kaempferol induces autophagy to upregulate miR-340 in human lung cancer cells [41] also via the IRE-JNK-CHOP pathway [137]. The widest activity of flavonoids on apoptosis in cancer cells via the major survival signaling pathways would raise the question about which relationship does exist between autophagy and apoptosis [138], although a fundamental link with the Akt signaling pathway has been recently highlighted [139] and some major components of the fission and fusion machinery in mitochondria, i.e., dynamin-related protein 1 (Drp1), mitofusin1/2 (Mfn1/2) and Optic Atrophy 1 (OPA1), were reported to be involved in the mitochondria-related modulation of autophagy, besides apoptosis and necroptosis [140]. Flavonoids might act in the crosstalk leading to the autophagy–survival mechanism or the autophagy–apoptosis decision [141,142,143], where a possible role is exerted by beclin-1, Bcl-2 and Bcl-xL [144,145]. Furthermore, the regulation of mitophagy and autophagy depends on acetylation (see acetylCoA) and Lys acetylation in mitochondria, accounting for a stringent control in nutrients uptake [146,147]. Autophagy can participate in cancer eradication; for example, silibinin induces MCF7 breast cancer cells death via the upregulation of beclin-1 and Atg12–Atg5 formation, the conversion of the light chain 3 (LC3)-I to LC3-II, mitochondrial leakage of the mitochondrial transmembrane potential and a decline in ATP levels, together with a great increase in ROS [148].
The role of autophagy in cancer has been extensively reviewed [149,150]. Autophagy is a key regulator of cell survival and homeostasis and past reports suggested its role as a tumor suppressor. However, a hypothesis by which autophagy leads to tumorigenesis has been related to its reduction in the stress response capability, thus leading to much more support in cell metabolism and survival processes [149]. Cancer cells are more dependent on autophagy than other cells. The abnormal increase in the metabolic demand shifts the autophagy towards mechanisms, leading to apoptosis or cell cycle arrest. Particularly for RAS-driven cancers, recent reports suggest that they are “autophagy-addicted” [151,152]. Interestingly, the flavonoid 6-C-(E-phenylethenyl)naringenin (6-CEPN), which can be found in naringenin-fortified fried beef, induces cytoprotective autophagy in colon cancer cells, although it inhibits cell growth by dampening the expression of autophagy proteins Atg7 and beclin-1. The compound 6-CEPN strongly activates RAS [153]. Further flavonoids kill cancer cells via an autophagic signaling pathway. The flavone isoorientin induces autophagy in hepatoblastoma cancer, activating ROS signaling leading to p53 expression and so concurrently activates apoptosis by the PI3K/Akt, JNK and p38MAPK signaling pathways [154]. The induction of ROS signaling to result in cancer cell apoptosis, via the initial autophagic process, has been reported also for the flavonoid wogonoside in human glioblastoma cells [155]. Interestingly, it seems that flavonoids do not exert their acknowledged antioxidant potential in cancer; however, a possible reason is that they shift the hormetic curve of survival homeostasis in cancer towards an autophagy-driven apoptotic signal, instead of an autophagy-driven survival process.
Possible sensitive targets of this “pro-oxidant” flavonoids-mediated mechanism are mitochondria, often in association with ER stress and the unfolded protein response (UPR) [156].
The prenylated flavonoid morusin induces an increase in the levels of mitochondrial calcium ions, with ER stress, induction of ROS and loss of mitochondrial membrane potential in epithelial ovarian cancer [157]. Calcium overload in mitochondria induces mitochondrial swelling and dysfunction, leading to morusin-induced paraptosis-like cell death [157]. Paraptosis is a mechanism leading to tumoral cell death, which is different from apoptosis and necrosis [158]. Some flavonoids induce paraptosis in cancer cells. For example, the polyphenol xanthohumol induces paraptosis in leukemia cells via the p38MAPK pathway [159], hesperidin, likewise morusin, causes paraptosis in HepG2 cells by inducing mitochondria calcium overload and subsequently mitochondria dysfunction [160]. Moreover, a possible finely controlled tuning of the cell survival/cell death homeostasis should explain terms such as paraptosis and necroptosis and their relationship with autophagy [161].
Flavonoids might work in the crosstalk between survival and cell death, acting either as anti-oxidant or pro-oxidant molecules, depending on the stress-responsive endowment of the perturbed cell. Certainly, the mitochondrial/ER proteasome orchestrated function may play a major role [17,53,162,163,164,165].
2.3. Epithelial Mesenchymal Transition (EMT) and Cell Cycle Arrest
Many phytochemicals, such as flavonoids, induce cancer cell death by targeting the cellular EMT pathways. Nobiletin, a hexamethoxyflavone, inhibits the EMT process initiated by hypoxia in renal cell carcinoma via the NF-κB and the Wnt/β-signaling pathways, dampening cancer migration and invasion (malignancy) [166]. The effect on EMT and therefore on cancer migration can be also associated with the induction of apoptosis [167] and even initially targeting the same signaling systems then leads to apoptosis, such as the Akt-related pathways. For example, apigenin inhibits non-small cell lung cancer metastasis by dampening the EMT signal via a CD26-Akt-Snail/Slug signaling pathway [168]. Furthermore, the PI3K/Akt signaling pathway can lead to cell cycle arrest in cancer [101]. The chalconoid cardamonin induces apoptosis and cell cycle arrest in breast cancer by downregulating Wnt3a/β-catenin induction of EMT, blocking EMT and dampening the metastatic signal [169]. Despite the attempt to induce cell survival, for example, by phosphorylating some signaling molecules, such as ERK, cancer cells can undergo cytotoxicity from flavonoids, which inhibits EMT-driven metastasis and blocks cell cycle [170]. The activity held by phytochemicals is fundamentally made of a pro-toxicant nature; normal cells have the ability to counteract their pro-oxidant potential, whereas cancer cell do not.
ROS are closely associated with EMT in cells and redox regulation in the EMT-related cancer progression is an issue of utmost interest [171,172,173]. One could think that flavonoids may even “use” their antioxidant ability to inhibit the ROS-induced EMT in cancer. However, it has been recently reported that superoxide dismutase promotes EMT, enhancing tumor metastasis, in pancreatic cancer cells via the H2O2/ERK/NF-κB signaling axis [174]. It is particularly difficult to associate flavonoids with their chemopreventive and anticancer potential by simply retrieving this relationship from their widely reported antioxidant ability. A possible master tuner is the mitochondria oscillatory balance of their biogenesis, which rules any survival control and also affects EMT [175,176,177]. ROS might exert a fundamental role in balancing signals. When cells are metabolically stressed and are driven towards mitochondria impairment, risk of mitochondria calcium overload, ER stress with UPR and so on, mild stress with ROS should work as a signal to induce survival but simply block or interrupt the autophagy, mitophagy or EMT pathway, leading to apoptosis or cell cycle arrest. When cell metabolism and bioenergetics are homeostatically controlled, mild ROS induces the survival process and restore the homeostatic balance. In the case of cancer cells, the homeostatic response is held by an unusual autophagy–survival relationship driven by ROS distress and by EMT. Mild stress (eustress), breaking down this “insane” equilibrium, should lead to cell death and EMT inhibition.
2.4. Insights into the Capability of Raw Food and Plant Extracts Containing Phytochemicals to Prevent and Counteract Cancer
Phytochemicals’ ability to counteract cancer has been particularly stressed in current in vitroand in vivoresearch [178]. The regular intake of food edible plants has been long associated with a reduction in the risk to develop cancer. A bulk of more than 200 research investigations can be retrieved from current scientific literature reporting the association between vegetables and fruit intake and the prevention of the most common and diffused cancers in humans, including the recent extensively reviewed research [179]. According to this study, the protective effect of plant-derived substances in daily diet, expressed as relative risk, was assessed for at least 128 of 156 nutritional investigations [179]. For the majority of the investigated cancers, people consuming moderate or poorly enriched diets with fruits or vegetables underwent a doubled risk to develop cancer compared to people with dietary habits including plant-derived raw foods, even considering statistics with confounders. In the case of lung cancer, at least 24 of 25 studies retrieved in the latest years reported protection from plants, after removing confounders for smoking [179]. About 28 of 29 papers reported that fruit consumption reduced the incidence of cancers in oral cavity, esophagus and larynx, 26 of 30 papers reported the fruit consumption decreased the incidence of pancreas and stomach cancer, and 28 of 38 papers revealed the decrease of bladder cancer and colorectal cancer by fruit intake [179]. Furthermore, about 11 of 13 studies’ results are positive for endometrial, cervix and ovary cancers, while the protective effect of fruits and vegetables intake was assessed also for breast cancer in a performed meta-analysis [179]. These data suggest that good cancer preventions should ask for the daily intake of plant-derived raw food [179]. Despite the many positive and encouraging results in in vitro investigations with purified, aglycone flavonoids, raw food is endowed with an additive and synergistic action of the many different phenolic components in the raw matrices, showing a better efficacy in preventing cancer [5].
Flavonoids, as a major class of phenolic compounds, exhibited highly antioxidant activity. These compounds have been connected to reduce the risk of main chronic diseases and have been recognized largely in fruits, vegetables, and other plant foods [180]. For example, indigenous individuals living in Siberian areas possess a typical socio-economical and anthropological tradition, which allow the people to use a particular kind of diet with plant raw foods. Phenolic, flavonoids, elements and organic acids content, antioxidant and cytotoxic activity of six Siberian indigenous fruits have been recently studied [181]. Cell cytotoxicity of the human prostate cancer cell line Du-145 was performed by treating the cell line with at least six different plant extracts of Siberian fruits, encompassing the concentration range of 0–100 μg/mL. Siberian apricot, Siberian mountain ash and Siberian bird cherry showed a moderate cytotoxic effect in a dose-dependent fashion. Siberian apricot extracts caused 50% of the cytotoxic effect at 25 μg/mL and almost a 100% effect at 100 μg/mL [181].
Some enzymes are particularly targeted by flavonoids contained in Siberian fruits. For example, cyclooxygenases (COXs), which play a major role in inflammatory reactions and carcinogenesis, besides modulating cell apoptosis, proliferation and the angiogenic mechanisms in tissues, are possible targets of these flavonoids. For example, anthocyanins in cherry fruits target COXs, exhibiting an anti-inflammatory activity [182]. Cherry extracts are considered as a good candidate to inhibit COX proinflammatory and pro-carcinogenetic activities. With their sweet and sour taste, cherry fruits are an acknowledged food used by Siberian to promote their good health. Cherries contain a huge amount of anthocyanins and anthcyanidins, mixed with phenolic acids. The frequent and regular intake of these fruits appears particularly beneficial in reducing the incidence of certain cancers. The study on lipid peroxidation and on the activity of COXs by flavonoids from different sweet and sour varieties of Siberian cherries assessed the role of these compunds as natural inhibitors [182]. The role of cherry anthocyanins for colon cancer prevention was confirmed in another study using a combination of dietary anthocyanin-rich extract and suboptimal dosages of sulindac in mice for 19 weeks [183]. Cherry leaves alcohol extract containing flavonoids showed a pronounced anti-inflammatory activity (52.1%), not inferior to the compared drug quercetin (50.9%) [184].
The cancer suppression activity of peach and plum extracts were also evaluated [185]. It was found that within all fractions, flavonoids and procyanidins were more potent against the three cell lines investigated in the study. Quercetin 3β-glucoside was the most bioactive compound identified in the chromatographic fractions, able to exert an anti-proliferative activity against MDA-MB-435 and MCF-10A breast cancer cell lines [185]. Growth-inhibitory effects were also reported on Caco-2, SW1116, HT29 and NCM460 cancer cell lines from extracts of plums and peaches notoriously being enriched with anthocyanins [186]. The most efficacious fraction contained flavonoids rather than anthocyanins, suggesting that a synergistic activity with phenolics in plum and peaches extracts from Siberian fruits should explicate the action on cancer cell lines previously observed [186].
Many recent research investigations have shown that phytochemicals in fruits express an orchestrated panoply of similar actions, often shown as overlapping effects, which include the regulation of the expression of detoxyfying antioxidant enzyme complexes, activation of innate immunity, modulation of gene expression, cell replication, autophagy and apoptosis and antimicrobic activity [5]. Some examples are particularly interesting for the effect of oxidation. Prunus domestica leaves extract is rich in hydroxycinnamic acids and flavonoids. The extract was tested in vitro and showed the greatest activity against the suppression of lipid peroxidation. It was found that the extract reduced the level of peroxidation products by 88.1% [187].
A prospective study, enrolling about 9959 subjects (age: 15–99 years, sex: equally distributed) and performed in Finland, reported an interesting inverse association between flavonoid assumption with diets and incidence of cancer [5].
Following a 24-year follow-up period, researchers reported that the risk of lung cancer was reduced by 50% (superior quartile of the flavonoid intake amounts). The consumption of quecetin contained in apples and onions in Hawaii was found to be inversely corelated with the incidence of lung cancer.
Moreover, the effect of onions intake resulted particularly promising in reducing squamous cell carcinoma incidence [5]. The increasing plasma levels of quercetin from ingested onions were also associated with an enhanced resistance to DNA strand breakage occurring in lymphocytes and a reduction in the level of some catabolites from oxidative stress in urine samples [5].
Further reports showed also that anthocyanins and cyanidins are able to reduce colon cancer cell lines HT29 and HCT 116 [188]. These studies reported that the 50% inhibitory concentration (IC50) values of anthocyanins and cyanidins were780 mM and 63 mM for HT29 cells and 285 mM and 85 mM for HCT116 cells, respectively, suggesting that anthocyanins and cyanidins in cherries may be promising as anticolon cancer natural compounds [188]. Actually, anthocyanidins counteract a lot of biological mechanisms, leading to cancer aggressiveness, including metastasis and chemoresistance [189].
Cyanidin is a polyphenolic pigment that can be easily found in many red berries, including blackberry, grapes, raspberry, cranberry, besides plums, apples and red cabbage and it is present also in red onions. It has a good potential benefit against cancers, thanks to its antioxidant property [178]. Cyanidin has been reported to dampen cell proliferation in cancer and to inhibit COX-2 and iNOS gene expression in colon tumors [190,191,192,193]. A study showed that cyanidin-3-glucoside inhibited the benzo[a]pyrene-7,8-diol-9,10-diol-epoxide-induced activation of the transcription factors AP-1 and NF-κB, including the phosphorylation of MEK, MKK4, Akt and MAPKs [194]. Furthermore, they also blocked the activation of the Fyn kinase signaling pathway, leading to a chemopreventive potential [194]. Further flavonoids, such as cyanidin-3-O-glucoside and cyanidin-3-O-rutinoside, as well as the ethanol extracts of raspberries, caused the inhibition of the cellular growth in highly tumorigenic rat esophagus cell lines RE-149 DHD but not in the weaker tumorigenic cell line RE-149 [195]. Further results showed that cyanidin inhibits ultraviolet light B UVB-induced COX-2 and PGE2 expression in epidermal cells by suppressing the MAPK-mediated NF-κB and AP-1 activity [195]. Furthermore, cyanidin is able to target MKK-4 MEK-1 and Raf-1 in the suppression pathway of UV-induced COX-2 [195]. Additionally, cyanidin-3-galactoside and cyanidin-3-glucoside are substrates for BRCP, while cyanidin, cyanidin-3,5-diglucuronide and cyanidin-3-rutinoside are BRCP inhibitors, although their effect on MDR1 is weak [178,196].
Catechins in green tea represent another hallmark of anticancer flavonoids. Green tea is a very widespread and commonly used beverage in the world. The majority of anticancer effects from green tea are attributed to EGCG [197,198]. EGCG is able to kill cancer cells notoriously resistant to proapoptotic stimuli and induces death through a necroptosis pathway in many cancer [189].
Tea polyphenols can prevent cancer by modulating epigenetic aberrations taking place in DNA methylation, histone modifications, and microRNAs. By altering these epimutations, they regulate chromatin dynamics and expression of those genesinducing or suppressing cancer formation [190].
Both theaflavins and thearubigins, which are very abundant polyphenolic compounds in black tea, possess a strong anticancer potential. Polyphenols in black tea inhibit cell proliferation and trigger apoptosis in Du 145 prostate cancer cells [191].
Theuse of flavonoids from raw fruits and vegetables should be encouraged with respect to nutraceutcals from purified active substances [192]. Effects of plant flavonoids on cancer can be even protective towards cancer cell lines; effects of various polyphenols, such as catechins, quercetin, flavanones, isoflavones, ellagic acid, lignans, polyphenols from red wine such as resveratrol, and curcumin, on various type of cancers, including mouth, stomach, duodenum, colon, liver, lung, mammary gland and skin, were observed [191]. This should account for the bimodal activity held by flavonoids.
The flavonol quercetin usually acts as an anticancer molecule via a process that involves the downregulation of some oncogenes (e.g., Mcl-1, Ras, MEK, P3K) or also the upregulation of some tumor suppressor genes, leading to the eradication of cancer [193]. Kaempferol, which can be found in vegetables such as broccoli, tea, grapefruit, Brussels sprouts, witchhazel and apples, has been reported as efficacious in pancreatic and lung cancer [199,200,201]. It has antiangiogenic and scavenging effects of oxygen radicals [178,202,203,204].
The flavone fisetin, which is present in plants, such as strawberries, persimmon, apples, Acacia greggii, Acacia berlandieri, Euroasiansmoketree, parrot tree, onion, cucumber and grape, is a strong antioxidant, modulating protein kinase and lipid kinase pathways [205]. Fisetin has been found to exert antitumoral actions in HCT-116 colon cancer cell lines [178]. Together with other flavones and flavonoids such as luteolin, galangin, quercetin and EGCG, fisetin induces the expression of the Nrf2 factor; moreover, it induces the phase II gene product HO-1 in human retinal pigment epithelial (RPE) cells. These cells may protect RPE cells from oxidativestress-induced death, with high efficacy and negligible toxicity [206]. Moreover, itlowered hydrogen peroxide (H2O2)-induced cell death [189,207].
The isoflavone genistein is present in plants, such as lupine, fava beans, soybeans, kudzu, and psoralea, Flemingiavestita, and coffee. It is an antioxidant and an anthelmintic, besides exerting antioxidant effects and antiangiogenic actions (blocking formation of new blood vessels), and also blocking cell replication and survival [178].
The flavanone naringenin, commonly found in citrus fruit and oranges, besides tomatoes skin and grapefruit, has a potent antioxidant property [208,209]. It suppresses the TGF-β ligand–receptor interactions [208], and TGF-β signaling, controlling a various group of cellular mechanisms in cancer, including cell proliferation, differentiation and apoptosis, as well as morin [189,208,210].
3. Conclusions and Future Remarks
The use of flavonoids and other phytochemicals in cancer research still remains a promising expectation, despite the many controversial aspects regarding the yet currently paucity in clinical results. Nutritional panels and dietary guidelines should encourage and support the introduction of plant phenolics in the daily meal composition, try to possibly reduce the amount of synthetic nutraceuticals and instead promote raw vegetables and fruit intake. Supplementation may be desirable if the composition formula is the closest possible with the raw source of the supplemented phytochemicals, a compulsory item that is very difficult to reach with optimal results. Anyway, research on flavonoids and plant extracts ensures current pharmacology to find and retrieve new drugs, able to specifically target cancer cells and to update our therapeutic endowment against malignancies. The very recent novelties in the field of polyphenol research against cancer refer to not only microRNA but also long non-coding RNA, such as glyceolinoccurring in soybean [211]. New fundamental insights in molecular biology of phytochemicals can improve our ability to investigate and use these compounds against cancer.
One fundamental question could be: why is using flavonoidsmore beneficial than using chemopreventive drugs against cancer? As a future perspective, flavonoids act as natural molecules, with surprisingly fewer side effects than chemopreventive drugs. The ability to sensitively tune fundamental mechanisms in cell survival, autophagy and apoptosis renders these compounds particularly useful in being adopted as prodrugs and also therapeutic molecules against cancer. Clinical research must be improved in this sense, in order to retrieve a more and more outstanding data collection about the excellent ability of these various natural substances to prevent and fight tumors in humans.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbrevations
| AAK | Akt-activated kinase |
| Akt | protein kinase B |
| AMPK | adenosine-monophosphate-activated proteinkinase |
| ARE | antioxidant-responsive element |
| ASK1 | apoptosis signal-regulating kinase 1 |
| Atg12–Atg5 | autophagy factor12–autophagy factor5 |
| Atg7 | autophagy factor 7 |
| Bax | Bcl-2-associated X protein |
| Bcl-2 | B-cell lymphoma 2 (an antiapoptotic protein) |
| Bid | BH3-interacting domain |
| BCRP | breast cancer resistance protein |
| BNP3 | Bcl-2-adenovirusE1B 19 kDaprotein-interactingprotein3 |
| 6-CEPN | 6-C-(E-phenylethenyl)naringenin |
| Cip1 | cyclin-inhibitor protein 1 (p21) |
| CYP | cytochrome P450 |
| DAF16 | Dauer-associated factor 16 |
| DAPK2 | death-associated protein kinase 2 |
| DRP1 | dynamin-related protein 1 |
| ERK | extracellularsignal-regulated kinase |
| FOXO | forkhead box O3 |
| HO-1 | heme oxygenase 1 |
| IKK | IκB kinase |
| iNOS | inducible nitric oxide synthase |
| IRE-JNK-CHOP | iron response element- Janus kinase-cyclophosphamide-hydroxydaunorubicin-oncovyn-prednisone) |
| JAK2 | Janus kinase 2 |
| JNK | Janus kinase |
| p38MAPK | p38 mitogen-activated protein kinase |
| MEK | a synonymof MAPK |
| MDR1 | multidrug resistant 1 |
| Mfn2 | mitofusin-2 |
| mTOR | mammalian target of rapamycin |
| NB4 | an acute promyelocytic leukemia cell line |
| NF-κB | nuclear factor kappa-light-chain-enhancer of activated B cells |
| Nrf2 | nuclear factor erythroid 2-related factor 2 |
| PARP | poly-ADP-ribose polymerase |
| PERK–ATF4 | protein kinase RNA-like endoplasmic reticulum kinase–activating transcription factor 4 |
| PGC1α | peroxisome proliferator-activated receptor gamma coactivator 1-alpha |
| PGE2 | prostaglandin E2 |
| pEGFR | phosphorylated epithelial growth factor receptor |
| PKA-PI3K | protein kinase A-phosphoinositide 3 kinase |
| Raf1 | Ras-associated factor 1 |
| SIR 2-1 | sirtuin 2-1 |
| SIRT1 | sirtuin 1 |
| Snail/Slug | transcription actors in EMT |
| STAT3 | signal transducer and activator of transcription 3 |
| TFAM | transcription factor A, mitochondrial |
| TRAIL | TNF-related apoptosis-inducing ligand |
| Wnt | wingless/integrated |
References
- 1.Chen C.Y., Kao C.L., Liu C.M. The Cancer Prevention, Anti-Inflammatory and Anti Oxidation of Bioactive Phytochemicals Targeting the TLR4 Signaling Pathway. Int. J. Mol. Sci. 2018;19:2729. doi: 10.3390/ijms19092729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chiou Y.S., Li S., Ho C.T., Pan M.H. Prevention of Breast Cancer by Natural Phytochemicals: Focusing on Molecular Targets and Combinational Strategy. Mol. Nutr. Food Res. 2018:e1800392. doi: 10.1002/mnfr.201800392. [DOI] [PubMed] [Google Scholar]
- 3.Bonam S.R., Wu Y.S., Tunki L., Chellian R., Halmuthur M.S.K., Muller S., Pandy V. What Has come out from Phytomedicines and Herbal Edibles for the Treatment of Cancer? Chem. Med. Chem. 2018;13:1854–1872. doi: 10.1002/cmdc.201800343. [DOI] [PubMed] [Google Scholar]
- 4.Galati G., O’Brien P.J. Potential toxicity of flavonoids and other dietary phenolics: Significance for their chemopreventive and anticancer properties. Free Radic. Biol. Med. 2004;37:287–303. doi: 10.1016/j.freeradbiomed.2004.04.034. [DOI] [PubMed] [Google Scholar]
- 5.Liu R.H. Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr. 2003;78(Suppl. 3):S517–S520. doi: 10.1093/ajcn/78.3.517S. [DOI] [PubMed] [Google Scholar]
- 6.Sunan W., Meckling K., Marcone M.F., Tasao R. Can phytochemical antioxidant rich foods act as anti-cancer agents? Food Res. Int. 2011;44:2545–2554. [Google Scholar]
- 7.Yang J.H., Shin B.Y., Han J.Y., Kim M.G., Wi J.E., Kim Y.W., Cho I.J., Kim S.C., Shin S.M., Ki S.H. Isorhamnetin protects against oxidative stress by activating Nrf2 and inducing the expression of its target genes. Toxicol. Appl. Pharmacol. 2014;274:293–301. doi: 10.1016/j.taap.2013.10.026. [DOI] [PubMed] [Google Scholar]
- 8.Aboonabi A., Singh I. Chemopreventive role of anthocyanins in atherosclerosis via activation of Nrf2-ARE as an indicator and modulator of redox. Biomed. Pharmacother. 2015;72:30–36. doi: 10.1016/j.biopha.2015.03.008. [DOI] [PubMed] [Google Scholar]
- 9.Costa L.G., Garrick J.M., Roquè P.J., Pellacani C. Mechanisms of Neuroprotection by Quercetin: Counteracting Oxidative Stress and More. Oxid. Med. Cell. Longev. 2016;2016:2986796. doi: 10.1155/2016/2986796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tebay L.E., Robertson H., Durant S.T., Vitale S.R., Penning T.M., Dinkova-Kostova A.T., Hayes J.D. Mechanisms of activation of the transcription factor Nrf2 by redox stressors, nutrient cues, and energy status and the pathways through which it attenuates degenerative disease. Free Radic. Biol. Med. 2015;88:108–146. doi: 10.1016/j.freeradbiomed.2015.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ighodaro Q.M., Akinloye Q.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid Alexandria. J. Med. 2017 doi: 10.1016/j.ajme.2017.09.001. in press. [DOI] [Google Scholar]
- 12.Clere N., Faure S., Martinez M.C., Andriantsitohaina R. Anticancer properties of flavonoids: Roles in various stages of carcinogenesis. Cardiovasc. Hematol. Agents Med. Chem. 2011;9:62–77. doi: 10.2174/187152511796196498. [DOI] [PubMed] [Google Scholar]
- 13.Qiu T., Wu D., Yang L., Ye H., Wang Q., Cao Z., Tang K. Exploring the Mechanism of Flavonoids Through Systematic Bioinformatics Analysis. Front. Pharmacol. 2018;9:918. doi: 10.3389/fphar.2018.00918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rauf A., Imran M., Khan I.A., Ur-Rehman M., Gilani S.A., Mehmood Z., Mubarak M.S. Anticancer potential of quercetin: A comprehensive review. Phytother. Res. 2018 doi: 10.1002/ptr.6155. [DOI] [PubMed] [Google Scholar]
- 15.Cook M.T. Mechanism of metastasis suppression by luteolin in breast cancer. Breast Cancer (Dove Med. Press) 2018;10:89–100. doi: 10.2147/BCTT.S144202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sak K. Site-specific anticancer effects of dietary flavonoid quercetin. Nutr. Cancer. 2014;66:177–193. doi: 10.1080/01635581.2014.864418. [DOI] [PubMed] [Google Scholar]
- 17.Chirumbolo S., Bjørklund G. PERM Hypothesis: The Fundamental Machinery Able to Elucidate the Role of Xenobiotics and Hormesis in Cell Survival and Homeostasis. Int. J. Mol. Sci. 2017;18:165. doi: 10.3390/ijms18010165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sabharwal S.S., Schumacker P.T. Mitochondrial ROS in cancer: Initiators, amplifiers or an Achilles’ heel? Nat. Rev. Cancer. 2014;14:709–721. doi: 10.1038/nrc3803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ralph S.J., Rodríguez-Enríquez S., Neuzil J., Saavedra E., Moreno-Sánchez R. The causes of cancer revisited: “mitochondrial malignancy” and ROS-induced oncogenic transformation—Why mitochondria are targets for cancer therapy. Mol. Asp. Med. 2010;31:145–170. doi: 10.1016/j.mam.2010.02.008. [DOI] [PubMed] [Google Scholar]
- 20.Wallace D.C. Mitochondria and cancer. Nat. Rev. Cancer. 2012;12:685–698. doi: 10.1038/nrc3365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bellot G.L., Liu D., Pervaiz S. ROS, autophagy, mitochondria and cancer: Ras, the hidden master? Mitochondrion. 2013;13:155–162. doi: 10.1016/j.mito.2012.06.007. [DOI] [PubMed] [Google Scholar]
- 22.Xiong L.G., Chen Y.J., Tong J.W., Gong Y.S., Huang J.A., Liu Z.H. Epigallocatechin-3-gallate promotes healthy lifespan through mitohormesis during early-to-mid adulthood in Caenorhabditis elegans. Redox Biol. 2018;14:305–315. doi: 10.1016/j.redox.2017.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Steib K., Schäffner I., Jagasia R., Ebert B., Lie D.C. Mitochondria modify exercise-induced development of stem cell-derived neurons in the adult brain. J. Neurosci. 2014;34:6624–6633. doi: 10.1523/JNEUROSCI.4972-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Naik P.P., Birbrair A., Bhutia S.K. Mitophagy-driven metabolic switch reprograms stem cell fate. Cell Mol. Life Sci. 2018 doi: 10.1007/s00018-018-2922-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mitra K. Mitochondrial fission-fusion as an emerging key regulator of cell proliferation and differentiation. Bioessays. 2013;35:955–964. doi: 10.1002/bies.201300011. [DOI] [PubMed] [Google Scholar]
- 26.Dal Yontem F., Kim S.H., Ding Z., Grimm E., Ekmekcioglu S., Akcakaya H. Mitochondrial dynamic alterations regulate melanoma cell progression. J. Cell Biochem. 2018 doi: 10.1002/jcb.27518. [DOI] [PubMed] [Google Scholar]
- 27.Huang Y., Chen K., Ren Q., Yi L., Zhu J., Zhang Q., Mi M. Dihydromyricetin Attenuates Dexamethasone-Induced Muscle Atrophy by Improving Mitochondrial Function via the PGC-1α Pathway. Cell Physiol. Biochem. 2018;49:758–779. doi: 10.1159/000493040. [DOI] [PubMed] [Google Scholar]
- 28.Lennon F.E., Salgia R. Mitochondrial dynamics: Biology and therapy in lung cancer. Expert Opin. Investig. Drugs. 2014;23:675–692. doi: 10.1517/13543784.2014.899350. [DOI] [PubMed] [Google Scholar]
- 29.Zhao J., Zhang J., Yu M., Xie Y., Huang Y., Wolff D.W., Abel P.W., Tu Y. Mitochondrial dynamics regulates migration and invasion of breast cancer cells. Oncogene. 2013;32:4814–4824. doi: 10.1038/onc.2012.494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Che T.F., Lin C.W., Wu Y.Y., Chen Y.J., Han C.L., Chang Y.L., Wu C.T., Hsiao T.H., Hong T.M., Yang P.C. Mitochondrial translocation of EGFR regulates mitochondria dynamics and promotes metastasis in NSCLC. Oncotarget. 2015;6:37349–37366. doi: 10.18632/oncotarget.5736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.White E., Mehnert J.M., Chan C.S. Autophagy, Metabolism, and Cancer. Clin. Cancer Res. 2015;21:5037–5046. doi: 10.1158/1078-0432.CCR-15-0490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Goldsmith J., Levine B., Debnath J. Autophagy and cancer metabolism. Methods Enzymol. 2014;542:25–57. doi: 10.1016/B978-0-12-416618-9.00002-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Poillet-Perez L., Despouy G., Delage-Mourroux R., Boyer-Guittaut M. Interplay between ROS and autophagy in cancer cells, from tumor initiation to cancer therapy. Redox Biol. 2015;4:184–192. doi: 10.1016/j.redox.2014.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hasima N., Ozpolat B. Regulation of autophagy by polyphenolic compounds as a potential therapeutic strategy for cancer. Cell Death Dis. 2014;5:e1509. doi: 10.1038/cddis.2014.467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Radogna F., Dicato M., Diederich M. Cancer-type-specific crosstalk between autophagy, necroptosis and apoptosis as a pharmacological target. Biochem. Pharmacol. 2015;94:1–11. doi: 10.1016/j.bcp.2014.12.018. [DOI] [PubMed] [Google Scholar]
- 36.Khorsandi L., Orazizadeh M., Niazvand F., Abbaspour M.R., Mansouri E., Khodadadi A. Quercetin induces apoptosis and necroptosis in MCF-7 breast cancer cells. Bratisl. Lek. Listy. 2017;118:123–128. doi: 10.4149/BLL_2017_025. [DOI] [PubMed] [Google Scholar]
- 37.Lin W., Tongyi S. Role of Bax/Bcl-2 family members in green tea polyphenol induced necroptosis of p53-deficient Hep3B cells. Tumour Biol. 2014;35:8065–8075. doi: 10.1007/s13277-014-2064-0. [DOI] [PubMed] [Google Scholar]
- 38.White E. The role for autophagy in cancer. J. Clin. Investig. 2015;125:42–46. doi: 10.1172/JCI73941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Brunelli E., Pinton G., Bellini P., Minassi A., Appendino G., Moro L. Flavonoid-induced autophagy in hormone sensitive breast cancer cells. Fitoterapia. 2009;80:327–332. doi: 10.1016/j.fitote.2009.04.002. [DOI] [PubMed] [Google Scholar]
- 40.Zheng N., Liu L., Liu W.W., Li F., Hayashi T., Tashiro S.I., Onodera S., Ikejima T. Crosstalk of ROS/RNS and autophagy in silibinin-induced apoptosis of MCF-7 human breast cancer cells in vitro. Acta Pharmacol. Sin. 2017;38:277–289. doi: 10.1038/aps.2016.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Han X., Liu C.F., Gao N., Zhao J., Xu J. Kaempferol suppresses proliferation but increases apoptosis and autophagy by up-regulating microRNA-340 in human lung cancer cells. Biomed. Pharmacother. 2018;108:809–816. doi: 10.1016/j.biopha.2018.09.087. [DOI] [PubMed] [Google Scholar]
- 42.Zhang H.W., Hu J.J., Fu R.Q., Liu X., Zhang Y.H., Li J., Liu L., Li Y.N., Deng Q., Luo Q.S., et al. Flavonoids inhibit cell proliferation and induce apoptosis and autophagy through downregulation of PI3Kγ mediated PI3K/AKT/mTOR/p70S6K/ULK signaling pathway in human breast cancer cells. Sci. Rep. 2018;8:11255. doi: 10.1038/s41598-018-29308-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kania E., Pająk B., Orzechowski A. Calcium homeostasis and ER stress in control of autophagy in cancer cells. Biomed. Res. Int. 2015;2015:352794. doi: 10.1155/2015/352794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Verfaillie T., Garg A.D., Agostinis P. Targeting ER stress induced apoptosis and inflammation in cancer. Cancer Lett. 2013;332:249–264. doi: 10.1016/j.canlet.2010.07.016. [DOI] [PubMed] [Google Scholar]
- 45.Duluc L., Soleti R., Clere N., Andriantsitohaina R., Simard G. Mitochondria as potential targets of flavonoids: Focus on adipocytes and endothelial cells. Curr. Med. Chem. 2012;19:4462–4474. doi: 10.2174/092986712803251467. [DOI] [PubMed] [Google Scholar]
- 46.Yang J., Chen W., Zhang B., Tian F., Zhou Z., Liao X., Li C., Zhang Y., Han Y., Wang Y., et al. Lon in maintaining mitochondrial and endoplasmic reticulum homeostasis. Arch. Toxicol. 2018;92:1913–1923. doi: 10.1007/s00204-018-2210-3. [DOI] [PubMed] [Google Scholar]
- 47.Polo M., Alegre F., Moragrega A.B., Gibellini L., Marti-Rodrigo A., Blas-Garcia A., Esplugues J.V., Apostolova N. Lon protease: A novel mitochondrial matrix protein in the interconnection between drug-induced mitochondrial dysfunction and endoplasmic reticulum stress. Br. J. Pharmacol. 2017;174:4409–4429. doi: 10.1111/bph.14045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hori O., Ichinoda F., Tamatani T., Yamaguchi A., Sato N., Ozawa K., Kitao Y., Miyazaki M., Harding H.P., Ron D., et al. Transmission of cell stress from endoplasmic reticulum to mitochondria: Enhanced expression of Lon protease. J. Cell Biol. 2002;157:1151–1160. doi: 10.1083/jcb.200108103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rainbolt T.K., Saunders J.M., Wiseman R.L. Stress-responsive regulation of mitochondria through the ER unfolded protein response. Trends Endocrinol. Metab. 2014;25:528–537. doi: 10.1016/j.tem.2014.06.007. [DOI] [PubMed] [Google Scholar]
- 50.Ehren J.L., Maher P. Concurrent regulation of the transcription factors Nrf2 and ATF4 mediates the enhancement of glutathione levels by the flavonoid fisetin. Biochem. Pharmacol. 2013;85:1816–1826. doi: 10.1016/j.bcp.2013.04.010. [DOI] [PubMed] [Google Scholar]
- 51.Li X.M., Liu J., Pan F.F., Shi D.D., Wen Z.G., Yang P.L. Quercetin and aconitine synergistically induces the human cervical carcinoma HeLa cell apoptosis via endoplasmic reticulum (ER) stress pathway. PLoS ONE. 2018;13:e0191062. doi: 10.1371/journal.pone.0191062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Liu C.M., Ma J.Q., Sun J.M., Feng Z.J., Cheng C., Yang W., Jiang H. Association of changes in ER stress-mediated signaling pathway with lead-induced insulin resistance and apoptosis in rats and their prevention by A-type dimeric epigallocatechin-3-gallate. Food Chem. Toxicol. 2017;110:325–332. doi: 10.1016/j.fct.2017.10.040. [DOI] [PubMed] [Google Scholar]
- 53.García-Aguilar A., Cuezva J.M. A Review of the Inhibition of the Mitochondrial ATP Synthase by IF1 in vivo: Reprogramming Energy Metabolism Inducing Mitohormesis. Front. Physiol. 2018;9:1322. doi: 10.3389/fphys.2018.01322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bravo-Sagua R., Parra V., Ortiz-Sandoval C., Navarro-Marquez M., Rodríguez A.E., Diaz-Valdivia N., Sanhueza C., Lopez-Crisosto C., Tahbaz N., Rothermel B.A., et al. Caveolin-1 impairs PKA-DRP1-mediated remodelling of ER-mitochondria communication during the early phase of ER stress. Cell Death Differ. 2018 doi: 10.1038/s41418-018-0197-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Chang C.R., Blackstone C. Drp1 phosphorylation and mitochondrial regulation. EMBO Rep. 2007;8:1088–1089. doi: 10.1038/sj.embor.7401118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cui L., Li Z., Chang X., Cong G., Hao L. Quercetin attenuates vascular calcification by inhibiting oxidative stress and mitochondrial fission. Vascul. Pharmacol. 2017;88:21–29. doi: 10.1016/j.vph.2016.11.006. [DOI] [PubMed] [Google Scholar]
- 57.Ye F., Wang H., Zhang L., Zou Y., Han H., Huang J. Baicalein induces human osteosarcoma cell line MG-63 apoptosis via ROS-induced BNIP3 expression. Tumour Biol. 2015;36:4731–4740. doi: 10.1007/s13277-015-3122-y. [DOI] [PubMed] [Google Scholar]
- 58.Herr I., Debatin K.M. Cellular stress response and apoptosis in cancer therapy. Blood. 2001;98:2603–2614. doi: 10.1182/blood.V98.9.2603. [DOI] [PubMed] [Google Scholar]
- 59.Chircop M., Speidel D. Cellular stress responses in cancer and cancer therapy. Front. Oncol. 2014;4:304. doi: 10.3389/fonc.2014.00304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ozbey U., Attar R., Romero M.A., Alhewairini S.S., Afshar B., Sabitaliyevich U.Y., Hanna-Wakim L., Ozcelik B., Farooqi A.A. Apigenin as an effective anticancer natural product: Spotlight on TRAIL, WNT/β-catenin, JAK-STAT pathways, and microRNAs. J. Cell Biochem. 2018 doi: 10.1002/jcb.27575. in press. [DOI] [PubMed] [Google Scholar]
- 61.Liao Y., Xu Y., Cao M., Huan Y., Zhu L., Jiang Y., Shen W., Zhu G. Luteolin Induces Apoptosis and Autophagy in Mouse Macrophage ANA-1 Cells via the Bcl-2 Pathway. J. Immunol. Res. 2018;2018:4623919. doi: 10.1155/2018/4623919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Dong X., Zhang J., Yang F., Wu J., Cai R., Wang T., Zhang J. Effect of luteolin on the methylation status of the OPCML gene and cell growth in breast cancer cells. Exp. Ther. Med. 2018;16:3186–3194. doi: 10.3892/etm.2018.6526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Yumnam S., Raha S., Kim S.M., Venkatarame Gowda Saralamma V., Lee H.J., Ha S.E., Heo J.D., Lee S.J., Kim E.H., Lee W.S., et al. Identification of a novel biomarker in tangeretin-induced cell death in AGS human gastric cancer cells. Oncol. Rep. 2018 doi: 10.3892/or.2018.6730. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Surichan S., Arroo R.R., Tsatsakis A.M., Androutsopoulos V.P. Tangeretin inhibits the proliferation of human breast cancer cells via CYP1A1/CYP1B1 enzyme induction and CYP1A1/CYP1B1-mediated metabolism to the product 4’ hydroxy tangeretin. Toxicol. In Vitro. 2018;50:274–284. doi: 10.1016/j.tiv.2018.04.001. [DOI] [PubMed] [Google Scholar]
- 65.Hirpara K.V., Aggarwal P., Mukherjee A.J., Joshi N., Burman A.C. Quercetin and its derivatives: synthesis, pharmacological uses with special emphasis on anti-tumor properties and prodrug with enhanced bio-availability. Anticancer Agents Med. Chem. 2009;9:138–161. doi: 10.2174/187152009787313855. [DOI] [PubMed] [Google Scholar]
- 66.Kuo W.T., Tsai Y.C., Wu H.C., Ho Y.J., Chen Y.S., Yao C.H., Yao C.H. Radiosensitization of non-small cell lung cancer by kaempferol. Oncol. Rep. 2015;34:2351–2356. doi: 10.3892/or.2015.4204. [DOI] [PubMed] [Google Scholar]
- 67.Choi J.B., Kim J.H., Lee H., Pak J.N., Shim B.S., Kim S.H. Reactive Oxygen Species and p53 Mediated Activation of p38 and Caspases is Critically Involved in Kaempferol Induced Apoptosis in Colorectal Cancer Cells. J. Agric. Food Chem. 2018;66:9960–9967. doi: 10.1021/acs.jafc.8b02656. [DOI] [PubMed] [Google Scholar]
- 68.Ye C., Zhang C., Huang H., Yang B., Xiao G., Kong D., Tian Q., Song Q., Song Y., Tan H., et al. The Natural Compound Myricetin Effectively Represses the Malignant Progression of Prostate Cancer by Inhibiting PIM1 and Disrupting the PIM1/CXCR4 Interaction. Cell Physiol. Biochem. 2018;48:1230–1244. doi: 10.1159/000492009. [DOI] [PubMed] [Google Scholar]
- 69.Li J., Gong X., Jiang R., Lin D., Zhou T., Zhang A., Li H., Zhang X., Wan J., Kuang G., et al. Fisetin Inhibited Growth and Metastasis of Triple-Negative Breast Cancer by Reversing Epithelial-to-Mesenchymal Transition via PTEN/Akt/GSK3β Signal Pathway. Front. Pharmacol. 2018;9:772. doi: 10.3389/fphar.2018.00772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zhu Y., Rao Q., Zhang X., Zhou X. Galangin induced antitumor effects in human kidney tumor cells mediated via mitochondrial mediated apoptosis, inhibition of cell migration and invasion and targeting PI3K/ AKT/mTOR signalling pathway. J. BUON. 2018;23:795–799. [PubMed] [Google Scholar]
- 71.Wu Q., Kroon P.A., Shao H., Needs P.W., Yang X. Differential Effects of Quercetin and Two of Its Derivatives, Isorhamnetin and Isorhamnetin-3-glucuronide, in Inhibiting the Proliferation of Human Breast-Cancer MCF-7 Cells. J. Agric. Food Chem. 2018;66:7181–7189. doi: 10.1021/acs.jafc.8b02420. [DOI] [PubMed] [Google Scholar]
- 72.Elango R., Athinarayanan J., Subbarayan V.P., Lei D.K.Y., Alshatwi A.A. Hesperetin induces an apoptosis-triggered extrinsic pathway and a p53- independent pathway in human lung cancer H522 cells. J. Asian Nat. Prod. Res. 2018;20:559–569. doi: 10.1080/10286020.2017.1327949. [DOI] [PubMed] [Google Scholar]
- 73.GumushanAktas H., Akgun T. Naringenin inhibits prostate cancer metastasis by blocking voltage-gated sodium channels. Biomed. Pharmacother. 2018;106:770–775. doi: 10.1016/j.biopha.2018.07.008. [DOI] [PubMed] [Google Scholar]
- 74.Haque M.W., Bose P., Siddique M.U.M., Sunita P., Lapenna A., Pattanayak S.P. Taxifolin binds with LXR (α & β) to attenuate DMBA-induced mammary carcinogenesis through mTOR/Maf-1/PTEN pathway. Biomed. Pharmacother. 2018;105:27–36. doi: 10.1016/j.biopha.2018.05.114. [DOI] [PubMed] [Google Scholar]
- 75.Haque M.W., Pattanayak S.P. Taxifolin Inhibits 7,12-Dimethylbenz(a)anthracene-induced Breast Carcinogenesis by Regulating AhR/CYP1A1 Signaling Pathway. Pharmacogn. Mag. 2018;13(Suppl. 4):S749–S755. doi: 10.4103/pm.pm_315_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Grube S., Ewald C., Kögler C., Lawson McLean A., Kalff R., Walter J. Achievable Central Nervous System Concentrations of the Green Tea Catechin EGCG Induce Stress in Glioblastoma Cells in Vitro. Nutr. Cancer. 2018:1–14. doi: 10.1080/01635581.2018.1495239. [DOI] [PubMed] [Google Scholar]
- 77.Xiao X., Jiang K., Xu Y., Peng H., Wang Z., Liu S., Zhang G. (-)-Epigallocatechin-3-gallate induces cell apoptosis in chronic myeloid leukemia by regulating Bcr/Abl-mediated p38-MAPK/JNK and JAK2/STAT3/AKT signaling pathways. Clin. Exp. Pharmacol. Physiol. 2018 doi: 10.1111/1440-1681.13037. [DOI] [PubMed] [Google Scholar]
- 78.Rawangkan A., Wongsirisin P., Namiki K., Iida K., Kobayashi Y., Shimizu Y., Fujiki H., Suganuma M. Green Tea Catechin Is an Alternative Immune Checkpoint Inhibitor that Inhibits PD-L1 Expression and Lung Tumor Growth. Molecules. 2018;23:2071. doi: 10.3390/molecules23082071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Stadlbauer S., Steinborn C., Klemd A., Hattori F., Ohmori K., Suzuki K., Huber R., Wolf P., Gründemann C. Impact of Green Tea Catechin ECG and Its Synthesized Fluorinated Analogue on Prostate Cancer Cells and Stimulated Immunocompetent Cells. Planta Med. 2018;84:813–819. doi: 10.1055/s-0044-102099. [DOI] [PubMed] [Google Scholar]
- 80.Mukherjee S., Hussaini R., White R., Atwi D., Fried A., Sampat S., Piao L., Pan Q., Banerjee P. TriCurin, a synergistic formulation of curcumin, resveratrol, and epicatechin gallate, repolarizes tumor-associated macrophages and triggers an immune response to cause suppression of HPV+ tumors. Cancer Immunol. Immunother. 2018;67:761–774. doi: 10.1007/s00262-018-2130-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ma X., Ning S. Cyanidin-3-glucoside attenuates the angiogenesis of breast cancer via inhibiting STAT3/VEGF pathway. Phytother. Res. 2018 doi: 10.1002/ptr.6201. [DOI] [PubMed] [Google Scholar]
- 82.Chen J., Zhu Y., Zhang W., Peng X., Zhou J., Li F., Han B., Liu X., Ou Y., Yu X. Delphinidin induced protective autophagy via mTOR pathway suppression and AMPK pathway activation in HER-2 positive breast cancer cells. BMC Cancer. 2018;18:342. doi: 10.1186/s12885-018-4231-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kang H.M., Park B.S., Kang H.K., Park H.R., Yu S.B., Kim I.R. Delphinidin induces apoptosis and inhibits epithelial-to-mesenchymal transition via the ERK/p38 MAPK-signaling pathway in human osteosarcoma cell lines. Environ. Toxicol. 2018;33:640–649. doi: 10.1002/tox.22548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ma C.H., Zhang Y.X., Tang L.H., Yang X.J., Cui W.M., Han C.C., Ji W.Y. MicroRNA-1469, a p53-responsive microRNA promotes Genistein induced apoptosis by targeting Mcl1 in human laryngeal cancer cells. Biomed. Pharmacother. 2018;106:665–671. doi: 10.1016/j.biopha.2018.07.005. [DOI] [PubMed] [Google Scholar]
- 85.Venza I., Visalli M., Oteri R., Beninati C., Teti D., Venza M. Genistein reduces proliferation of EP3-expressing melanoma cells through inhibition of PGE2-induced IL-8 expression. Int. Immunopharmacol. 2018;62:86–95. doi: 10.1016/j.intimp.2018.06.009. [DOI] [PubMed] [Google Scholar]
- 86.Dutta B., Park J.E., Qing I.T.Y., Kon O.L., Sze S.K. Soy-Derived Phytochemical Genistein Modifies Chromatome Topology to Restrict Cancer Cell Proliferation. Proteomics. 2018;18:e1700474. doi: 10.1002/pmic.201700474. [DOI] [PubMed] [Google Scholar]
- 87.Liang Y.S., Qi W.T., Guo W., Wang C.L., Hu Z.B., Li A.K. Genistein and daidzein induce apoptosis of colon cancer cells by inhibiting the accumulation of lipid droplets. Food Nutr. Res. 2018;62 doi: 10.29219/fnr.v62.1384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Brusselmans K., Vrolix R., Verhoeven G., Swinnen J.V. Induction of cancer cell apoptosis by flavonoids is associated with their ability to inhibit fatty acid synthase activity. J. Biol. Chem. 2005;280:5636–5645. doi: 10.1074/jbc.M408177200. [DOI] [PubMed] [Google Scholar]
- 89.Zhang J.S., Lei J.P., Wei G.Q., Chen H., Ma C.Y., Jiang H.Z. Natural fatty acid synthase inhibitors as potent therapeutic agents for cancers: A review. Pharm. Biol. 2016;54:1919–1925. doi: 10.3109/13880209.2015.1113995. [DOI] [PubMed] [Google Scholar]
- 90.Bandyopadhyay S., Zhan R., Wang Y., Pai S.K., Hirota S., Hosobe S., Takano Y., Saito K., Furuta E., Iiizumi M., et al. Mechanism of apoptosis induced by the inhibition of fatty acid synthase in breast cancer cells. Cancer Res. 2006;66:5934–5940. doi: 10.1158/0008-5472.CAN-05-3197. [DOI] [PubMed] [Google Scholar]
- 91.Li B., Espinoza I., Liu H., Lupu R. Inhibition of fatty acid synthase induces reactive oxygen species (ROS) to inhibit HER2 overexpressing beast cancer cell growth. Cancer Research; Proceedings of the 2007 98th AACR Annual Meeting; Los Angeles, CA, USA. 14–18 April 2007; Philadelphia, PA, USA: American Association for Cancer Research; 2007. Abst 4462. [Google Scholar]
- 92.Niki E. Oxidative stress and antioxidants: Distress or eustress? Arch Biochem. Biophys. 2016;595:19–24. doi: 10.1016/j.abb.2015.11.017. [DOI] [PubMed] [Google Scholar]
- 93.Murakami A. Dose-dependent functionality and toxicity of green tea polyphenols in experimental rodents. Arch. Biochem. Biophys. 2014;557:3–10. doi: 10.1016/j.abb.2014.04.018. [DOI] [PubMed] [Google Scholar]
- 94.Desikan R., Hancock J., Neill S. In: Reactive Oxygen Species as Signalling Molecules in “Antioxidants Reactive Oxygen Species in Plants”. Smirnoff N., editor. Blackwell Publl; Oxford, UK: 2005. pp. 169–195. Chapter 7. [Google Scholar]
- 95.Kumari S., Badana A.K., GMM G.S., Malla R. Reactive Oxygen Species: A Key Constituent in Cancer Survival. Biomark. Insights. 2018;13 doi: 10.1177/1177271918755391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Rubio V., García-Pérez A.I., Herráez A., Diez J.C. Different roles of Nrf2 and NF-κB in the antioxidant imbalance produced by esculetin or quercetin on NB4 leukemia cells. Chem. Biol. Interact. 2018;294:158–166. doi: 10.1016/j.cbi.2018.08.015. [DOI] [PubMed] [Google Scholar]
- 97.Khatami M. Cancer; an induced disease of twentieth century! Induction of tolerance, increased entropy and ‘Dark Energy’: Loss of biorhythms (Anabolism v. Catabolism) Clin. Transl. Med. 2018;7:20. doi: 10.1186/s40169-018-0193-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Isono M., Sato A., Asano T., Okubo K., Asano T. Evaluation of Therapeutic Potential of Phenoxodiol, a Novel Isoflavone Analog, in Renal Cancer Cells. Anticancer Res. 2018;38:5709–5716. doi: 10.21873/anticanres.12908. [DOI] [PubMed] [Google Scholar]
- 99.Chien K.J., Yang M.L., Tsai P.K., Su C.H., Chen C.H., Horng C.T., Yeh C.H., Chen W.Y., Lin M.L., Chen C.J., et al. Safrole induced cytotoxicity, DNA damage, and apoptosis in macrophages via reactive oxygen species generation and Akt phosphorylation. Environ. Toxicol. Pharmacol. 2018;64:94–100. doi: 10.1016/j.etap.2018.09.012. [DOI] [PubMed] [Google Scholar]
- 100.Im E., Yeo C., Lee E.O. Luteolin induces caspase-dependent apoptosis via inhibiting the AKT/osteopontin pathway in human hepatocellular carcinoma SK-Hep-1 cells. Life Sci. 2018;209:259–266. doi: 10.1016/j.lfs.2018.08.025. [DOI] [PubMed] [Google Scholar]
- 101.Gu Z.F., Zhang Z.T., Wang J.Y., Xu B.B. Icariin exerts inhibitory effects on the growth and metastasis of KYSE70 human esophageal carcinoma cells via PI3K/AKT and STAT3 pathways. Environ. Toxicol. Pharmacol. 2017;54:7–13. doi: 10.1016/j.etap.2017.06.004. [DOI] [PubMed] [Google Scholar]
- 102.Yu M., Qi B., Xiaoxiang W., Xu J., Liu X. Baicalein increases cisplatin sensitivity of A549 lung adenocarcinoma cells via PI3K/Akt/NF-κB pathway. Biomed. Pharmacother. 2017;90:677–685. doi: 10.1016/j.biopha.2017.04.001. [DOI] [PubMed] [Google Scholar]
- 103.Zhou H., Li X.M., Meinkoth J., Pittman R.N. Akt regulates cell survival and apoptosis at a postmitochondrial level. J. Cell Biol. 2000;151:483–494. doi: 10.1083/jcb.151.3.483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ahn J., Won M., Choi J.H., Kim Y.S., Jung C.R., Im D.S., Kyun M.L., Lee K., Song K.B., Chung K.S. Reactive oxygen species-mediated activation of the Akt/ASK1/p38 signaling cascade and p21(Cip1) downregulation are required for shikonin-induced apoptosis. Apoptosis. 2013;18:870–881. doi: 10.1007/s10495-013-0835-5. [DOI] [PubMed] [Google Scholar]
- 105.Pan J., Chang Q., Wang X., Son Y., Zhang Z., Chen G., Luo J., Bi Y., Chen F., Shi X. Reactive oxygen species-activated Akt/ASK1/p38 signaling pathway in nickel compound-induced apoptosis in BEAS 2B cells. Chem. Res. Toxicol. 2010;23:568–577. doi: 10.1021/tx9003193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Puckett M.C., Goldman E.H., Cockrell L.M., Huang B., Kasinski A.L., Du Y., Wang C.Y., Lin A., Ichijo H., Khuri F., et al. Integration of apoptosis signal-regulating kinase 1-mediated stress signaling with the Akt/protein kinase B-IκB kinase cascade. Mol. Cell Biol. 2013;33:2252–2259. doi: 10.1128/MCB.00047-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Kaltschmidt B., Greiner J.F.W., Kadhim H.M., Kaltschmidt C. Subunit-Specific Role of NF-κB in Cancer. Biomedicines. 2018;6:44. doi: 10.3390/biomedicines6020044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Ashraf A.H.M.Z., Afroze S.H., Yamauchi K., Zawieja D.C., Keuhl T.J., Erlandson L.W., Uddin M.N. Differential Mechanism of Action of 3,4′,7-O-trimethylquercetin in Three Types of Ovarian Cancer Cells. Anticancer Res. 2018;38:5131–5137. doi: 10.21873/anticanres.12835. [DOI] [PubMed] [Google Scholar]
- 109.Valko M., Rhodes C.J., Moncol J., Izakovic M., Mazur M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact. 2006;160:1–40. doi: 10.1016/j.cbi.2005.12.009. [DOI] [PubMed] [Google Scholar]
- 110.Chen C.T., Chen Y.T., Hsieh Y.H., Weng C.J., Yeh J.C., Yang S.F., Lin C.W., Yang J.S. Glabridin induces apoptosis and cell cycle arrest in oral cancer cells through the JNK1/2 signaling pathway. Environ. Toxicol. 2018;33:679–685. doi: 10.1002/tox.22555. [DOI] [PubMed] [Google Scholar]
- 111.Zhang W., Zhang Y., Ding K., Zhang H., Zhao Q., Liu Z., Xu Y. Involvement of JNK1/2-NF-κBp65 in the regulation of HMGB2 in myocardial ischemia/reperfusion-induced apoptosis in human AC16 cardiomyocytes. Biomed. Pharmacother. 2018;106:1063–1071. doi: 10.1016/j.biopha.2018.07.015. [DOI] [PubMed] [Google Scholar]
- 112.Cincin Z.B., Kiran B., Baran Y., Cakmakoglu B. Hesperidin promotes programmed cell death by downregulation of nongenomic estrogen receptor signalling pathway in endometrial cancer cells. Biomed. Pharmacother. 2018;103:336–345. doi: 10.1016/j.biopha.2018.04.020. [DOI] [PubMed] [Google Scholar]
- 113.Yang J., Pi C., Wang G. Inhibition of PI3K/Akt/mTOR pathway by apigenin induces apoptosis and autophagy in hepatocellular carcinoma cells. Biomed. Pharmacother. 2018;103:699–707. doi: 10.1016/j.biopha.2018.04.072. [DOI] [PubMed] [Google Scholar]
- 114.Celińska-Janowicz K., Zaręba I., Lazarek U., Teul J., Tomczyk M., Pałka J., Miltyk W. Constituents of Propolis: Chrysin, Caffeic Acid, p-Coumaric Acid, and Ferulic Acid Induce PRODH/POX-Dependent Apoptosis in Human Tongue Squamous Cell Carcinoma Cell (CAL-27) Front. Pharmacol. 2018;9:336. doi: 10.3389/fphar.2018.00336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Gaman L., Dragos D., Vlad A., Robu G.C., Radoi M.P., Stroica L., Badea M., Gilca M. Phytoceuticals in Acute Pancreatitis: Targeting the Balance between Apoptosis and Necrosis. Evid. Based Complement. Altern. Med. 2018;2018:5264592. doi: 10.1155/2018/5264592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Yeung A.W.K., El-Demerdash A., Berindan-Neagoe I., Atanasov A.G., Ho Y.S. Molecular Responses of Cancers by Natural Products: Modifications of Autophagy Revealed by Literature Analysis. Crit. Rev. Oncog. 2018;23:347–370. doi: 10.1615/CritRevOncog.2018027566. [DOI] [PubMed] [Google Scholar]
- 117.Zhang C., Hao Y., Wu L., Dong X., Jiang N., Cong B., Liu J., Zhang W., Tang D., De Perrot M., et al. Curcumin induces apoptosis and inhibits angiogenesis in murine malignant mesothelioma. Int. J. Oncol. 2018 doi: 10.3892/ijo.2018.4569. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Chung S.S., Dutta P., Austin D., Wang P., Awad A., Vadgama J.V. Combination of resveratrol and 5-flurouracil enhanced anti-telomerase activity and apoptosis by inhibiting STAT3 and Akt signaling pathways in human colorectal cancer cells. Oncotarget. 2018;9:32943–32957. doi: 10.18632/oncotarget.25993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Wang Y., Zhou P., Qin S., Xu D., Liu Y., Fu W., Ruan B., Zhang L., Zhang Y., Wang X., et al. The Curcumin Analogs 2-Pyridyl Cyclohexanone Induce Apoptosis via Inhibition of the JAK2-STAT3 Pathway in Human Esophageal Squamous Cell Carcinoma Cells. Front. Pharmacol. 2018;9:820. doi: 10.3389/fphar.2018.00820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Baek S.H., Ko J.H., Lee H., Jung J., Kong M., Lee J.W., Lee J., Chinnathambi A., Zayed M.E., Alharbi S.A., et al. Resveratrol inhibits STAT3 signaling pathway through the induction of SOCS-1: Role in apoptosis induction radiosensitization in head neck tumor cells. Phytomedicine. 2016;23:566–577. doi: 10.1016/j.phymed.2016.02.011. [DOI] [PubMed] [Google Scholar]
- 121.Masuelli L., Benvenuto M., Di Stefano E., Mattera R., Fantini M., De Feudis G., De Smaele E., Tresoldi I., Giganti M.G., Modesti A., et al. Curcumin blocks autophagy and activates apoptosis of malignant mesothelioma cell lines and increases the survival of mice intraperitoneally transplanted with a malignant mesothelioma cell line. Oncotarget. 2017;8:34405–34422. doi: 10.18632/oncotarget.14907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Wu X.P., Xiong M., Xu C.S., Duan L.N., Dong Y.Q., Luo Y., Niu T.H., Lu C.R. Resveratrol induces apoptosis of human chronic myelogenous leukemia cells in vitro through p38 and JNK-regulated H2AX phosphorylation. Acta Pharmacol. Sin. 2015;36:353–361. doi: 10.1038/aps.2014.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Zheng R., Deng Q., Liu Y., Zhao P. Curcumin Inhibits Gastric Carcinoma Cell Growth and Induces Apoptosis by Suppressing the Wnt/β-Catenin Signaling Pathway. Med. Sci. Monit. 2017;23:163–171. doi: 10.12659/MSM.902711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Zou Y., Yang J., Jiang D. Resveratrol inhibits canonical Wnt signaling in human MG-63 osteosarcoma cells. Mol. Med. Rep. 2015;12:7221–7226. doi: 10.3892/mmr.2015.4338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Divya C.S., Pillai M.R. Antitumor action of curcumin in human papillomavirus associated cells involves downregulation of viral oncogenes, prevention of NFkB and AP-1 translocation, and modulation of apoptosis. Mol. Carcinog. 2006;45:320–332. doi: 10.1002/mc.20170. [DOI] [PubMed] [Google Scholar]
- 126.Elshaer M., Chen Y., Wang X.J., Tang X. Resveratrol: An overview of its anti-cancer mechanisms. Life Sci. 2018;207:340–349. doi: 10.1016/j.lfs.2018.06.028. [DOI] [PubMed] [Google Scholar]
- 127.Shirley S., Micheau O. Targeting c-FLIP in cancer. Cancer Lett. 2013;332:141–150. doi: 10.1016/j.canlet.2010.10.009. [DOI] [PubMed] [Google Scholar]
- 128.Wright C., Iyer A.K.V., Yakisich J.S., Azad N. Anti-Tumorigenic Effects of Resveratrol in Lung Cancer Cells through Modulation of c-FLIP. Curr. Cancer Drug Targets. 2017;17:669–680. doi: 10.2174/1568009617666170315162932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Chin H.K., Horng C.T., Liu Y.S., Lu C.C., Su C.Y., Chen P.S., Chiu H.Y., Tsai F.J., Shieh P.C., Yang J.S. Kaempferol inhibits angiogenic ability by targeting VEGF receptor-2 and downregulating the PI3K/AKT, MEK and ERK pathways in VEGF-stimulated human umbilical vein endothelial cells. Oncol. Rep. 2018;39:2351–2357. doi: 10.3892/or.2018.6312. [DOI] [PubMed] [Google Scholar]
- 130.Semaan J., Pinon A., Rioux B., Hassan L., Limami Y., Pouget C., Fagnère C., Sol V., Diab-Assaf M., Simon A., et al. Resistance to 3-HTMC-Induced Apoptosis Through Activation of PI3K/Akt, MEK/ERK, and p38/COX-2/PGE(2) Pathways in Human HT-29 and HCT116 Colorectal Cancer Cells. J. Cell Biochem. 2016;117:2875–2885. doi: 10.1002/jcb.25600. [DOI] [PubMed] [Google Scholar]
- 131.Wani Z.A., Guru S.K., Rao A.V., Sharma S., Mahajan G., Behl A., Kumar A., Sharma P.R., Kamal A., Bhushan S., et al. A novel quinazolinone chalcone derivative induces mitochondrial dependent apoptosis and inhibits PI3K/Akt/mTOR signaling pathway in human colon cancer HCT-116 cells. Food Chem. Toxicol. 2016;87:1–11. doi: 10.1016/j.fct.2015.11.016. [DOI] [PubMed] [Google Scholar]
- 132.Ren K.W., Li Y.H., Wu G., Ren J.Z., Lu H.B., Li Z.M., Han X.W. Quercetin nanoparticles display antitumor activity via proliferation inhibition and apoptosis induction in liver cancer cells. Int. J. Oncol. 2017;50:1299–1311. doi: 10.3892/ijo.2017.3886. [DOI] [PubMed] [Google Scholar]
- 133.Xu Y., Gao C.C., Pan Z.G., Zhou C.W. Irigenin sensitizes TRAIL-induced apoptosis via enhancing pro-apoptotic molecules in gastric cancer cells. Biochem. Biophys. Res. Commun. 2018;496:998–1005. doi: 10.1016/j.bbrc.2018.01.003. [DOI] [PubMed] [Google Scholar]
- 134.Jaudan A., Sharma S., Malek S.N.A., Dixit A. Induction of apoptosis by pinostrobin in human cervical cancer cells: Possible mechanism of action. PLoS ONE. 2018;13:e0191523. doi: 10.1371/journal.pone.0191523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Kang C.H., Molagoda I.M.N., Choi Y.H., Park C., Moon D.O., Kim G.Y. Apigenin promotes TRAIL-mediated apoptosis regardless of ROS generation. Food Chem. Toxicol. 2018;111:623–630. doi: 10.1016/j.fct.2017.12.018. [DOI] [PubMed] [Google Scholar]
- 136.Zhao Y., Tian B., Wang Y., Ding H. Kaempferol Sensitizes Human Ovarian Cancer Cells-OVCAR-3 and SKOV-3 to Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand (TRAIL)-Induced Apoptosis via JNK/ERK-CHOP Pathway and Up-Regulation of Death Receptors 4 and 5. Med. Sci. Monit. 2017;23:5096–5105. doi: 10.12659/MSM.903552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Kim T.W., Lee S.Y., Kim M., Cheon C., Ko S.G. Kaempferol induces autophagic cell death via IRE1-JNK-CHOP pathway and inhibition of G9a in gastric cancer cells. Cell Death Dis. 2018;9:875. doi: 10.1038/s41419-018-0930-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Gump J.M., Thorburn A. Autophagy and apoptosis: What is the connection? Trends Cell Biol. 2011;21:387–392. doi: 10.1016/j.tcb.2011.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Noguchi M., Hirata N., Suizu F. The links between AKT and two intracellular proteolytic cascades: Ubiquitination and autophagy. Biochim. Biophys. Acta. 2014;1846:342–352. doi: 10.1016/j.bbcan.2014.07.013. [DOI] [PubMed] [Google Scholar]
- 140.Xie L.L., Shi F., Tan Z., Li Y., Bode A.M., Cao Y. Mitochondrial network structure homeostasis and cell death. Cancer Sci. 2018 doi: 10.1111/cas.13830. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Fan Y.J., Zong W.X. The cellular decision between apoptosis and autophagy. Chin. J. Cancer. 2013;32:121–129. doi: 10.5732/cjc.012.10106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Gordy C., He Y.W. The crosstalk between autophagy and apoptosis: Where does this lead? Protein Cell. 2012;3:17–27. doi: 10.1007/s13238-011-1127-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Thorburn A. Apoptosis and autophagy: Regulatory connections between two supposedly different processes. Apoptosis. 2008;13:1–9. doi: 10.1007/s10495-007-0154-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Zhou F., Yang Y., Xing D. Bcl-2 and Bcl-xL play important roles in the crosstalk between autophagy and apoptosis. FEBS J. 2011;278:403–413. doi: 10.1111/j.1742-4658.2010.07965.x. [DOI] [PubMed] [Google Scholar]
- 145.Yang J., Yao S. JNK-Bcl-2/Bcl-xL-Bax/Bak Pathway Mediates the Crosstalk between Matrine-Induced Autophagy and Apoptosis via Interplay with Beclin 1. Int. J. Mol. Sci. 2015;16:25744–25758. doi: 10.3390/ijms161025744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Webster B.R., Scott I., Traba J., Han K., Sack M.N. Regulation of autophagy and mitophagy by nutrient availability and acetylation. Biochim. Biophys. Acta. 2014;1841:525–534. doi: 10.1016/j.bbalip.2014.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Hosp F., Lassowskat I., Santoro V., De Vleesschauwer D., Fliegner D., Redestig H., Mann M., Christian S., Hannah M.A., Finkemeier I. Lysine acetylation in mitochondria: From inventory to function. Mitochondrion. 2017;33:58–71. doi: 10.1016/j.mito.2016.07.012. [DOI] [PubMed] [Google Scholar]
- 148.Jiang K., Wang W., Jin X., Wang Z., Ji Z., Meng G. Silibinin, a natural flavonoid, induces autophagy via ROS-dependent mitochondrial dysfunction and loss of ATP involving BNIP3 in human MCF7 breast cancer cells. Oncol. Rep. 2015;33:2711–2718. doi: 10.3892/or.2015.3915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Levy J.M.M., Towers C.G., Thorburn A. Targeting autophagy in cancer. Nat. Rev. Cancer. 2017;17:528–542. doi: 10.1038/nrc.2017.53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Kaczanowski S. Apoptosis: its origin, history, maintenance and the medical implications for cancer and aging. Phys. Biol. 2016;13:031001. doi: 10.1088/1478-3975/13/3/031001. [DOI] [PubMed] [Google Scholar]
- 151.Guo J.Y., Chen H.Y., Mathew R., Fan J., Strohecker A.M., Karsli-Uzunbas G., Kamphorst J.J., Chen G., Lemons J.M., Karantza V., et al. Activated Ras requires autophagy to maintain oxidative metabolism and tumorigenesis. Genes Dev. 2011;25:460–470. doi: 10.1101/gad.2016311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Guo J.Y., Xia B., White E. Autophagy-mediated tumor promotion. Cell. 2013;155:1216–1219. doi: 10.1016/j.cell.2013.11.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Zhao Y., Fan D., Ru B., Cheng K.W., Hu S., Zhang J., Li E.T., Wang M. 6-C-(E-phenylethenyl)naringenin induces cell growth inhibition and cytoprotective autophagy in colon cancer cells. Eur. J. Cancer. 2016;68:38–50. doi: 10.1016/j.ejca.2016.09.001. [DOI] [PubMed] [Google Scholar]
- 154.Yuan L., Wei S., Wang J., Liu X. Isoorientin induces apoptosis and autophagy simultaneously by reactive oxygen species (ROS)-related p53, PI3K/Akt, JNK, and p38 signaling pathways in HepG2 cancer cells. J. Agric. Food Chem. 2014;62:5390–5400. doi: 10.1021/jf500903g. [DOI] [PubMed] [Google Scholar]
- 155.Zhang L., Wang H., Cong Z., Xu J., Zhu J., Ji X., Ding K. Wogonoside induces autophagy-related apoptosis in human glioblastoma cells. Oncol. Rep. 2014;32:1179–1187. doi: 10.3892/or.2014.3294. [DOI] [PubMed] [Google Scholar]
- 156.Yi H.S., Chang J.Y., Shong M. The mitochondrial unfolded protein response and mitohormesis: A perspective on metabolic diseases. J. Mol. Endocrinol. 2018;61:R91–R105. doi: 10.1530/JME-18-0005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Xue J., Li R., Zhao X., Ma C., Lv X., Liu L., Liu P. Morusin induces paraptosis-like cell death through mitochondrial calcium overload and dysfunction in epithelial ovarian cancer. Chem. Biol. Interact. 2018;283:59–74. doi: 10.1016/j.cbi.2018.02.003. [DOI] [PubMed] [Google Scholar]
- 158.Lee D., Kim I.Y., Saha S., Choi K.S. Paraptosis in the anti-cancer arsenal of natural products. Pharmacol. Ther. 2016;162:120–133. doi: 10.1016/j.pharmthera.2016.01.003. [DOI] [PubMed] [Google Scholar]
- 159.Mi X., Wang C., Sun C., Chen X., Huo X., Zhang Y., Li G., Xu B., Zhang J., Xie J., et al. Xanthohumol induces paraptosis of leukemia cells through p38 mitogen activated protein kinase signaling pathway. Oncotarget. 2017;8:31297–31304. doi: 10.18632/oncotarget.16185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Yumnam S., Hong G.E., Raha S., Saralamma V.V., Lee H.J., Lee W.S., Kim E.H., Kim G.S. Mitochondrial Dysfunction and Ca(2+) Overload Contributes to Hesperidin Induced Paraptosis in Hepatoblastoma Cells, HepG2. J. Cell Physiol. 2016;231:1261–1268. doi: 10.1002/jcp.25222. [DOI] [PubMed] [Google Scholar]
- 161.Lalaoui N., Lindqvist L.M., Sandow J.J., Ekert P.G. The molecular relationships between apoptosis, autophagy and necroptosis. Semin. Cell Dev. Biol. 2015;39:63–69. doi: 10.1016/j.semcdb.2015.02.003. [DOI] [PubMed] [Google Scholar]
- 162.Coleman V., Sa-Nguanmoo P., Koenig J., Schulz T.J., Grune T., Klaus S., Kipp A.P., Ost M. Partial involvement of Nrf2 in skeletal muscle mitohormesis as an adaptive response to mitochondrial uncoupling. Sci. Rep. 2018;8:2446. doi: 10.1038/s41598-018-20901-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Cox C.S., McKay S.E., Holmbeck M.A., Christian B.E., Scortea A.C., Tsay A.J., Newman L.E., Shadel G.S. Mitohormesis in Mice via Sustained Basal Activation of Mitochondrial and Antioxidant Signaling. Cell Metab. 2018;28:776–786. doi: 10.1016/j.cmet.2018.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Yong C.Q.Y., Tang B.L. A Mitochondrial Encoded Messenger at the Nucleus. Cells. 2018;7:105. doi: 10.3390/cells7080105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Dandawate P.R., Subramaniam D., Jensen R.A., Anant S. Targeting cancer stem cells and signaling pathways by phytochemicals: Novel approach for breast cancer therapy. Semin. Cancer Biol. 2016;40–41:192–208. doi: 10.1016/j.semcancer.2016.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Liu F., Zhang S., Yin M., Guo L., Xu M., Wang Y. Nobiletin inhibits hypoxia-induced epithelial-mesenchymal transition in renal cell carcinoma cells. J. Cell Biochem. 2018 doi: 10.1002/jcb.27511. in press. [DOI] [PubMed] [Google Scholar]
- 167.Wang N., Zhou F., Guo J., Zhu H., Luo S., Cao J. Euxanthone suppresses tumor growth and metastasis in colorectal cancer via targeting CIP2A/PP2A pathway. Life Sci. 2018;209:498–506. doi: 10.1016/j.lfs.2018.08.052. [DOI] [PubMed] [Google Scholar]
- 168.Chang J.H., Cheng C.W., Yang Y.C., Chen W.S., Hung W.Y., Chow J.M., Chen P.S., Hsiao M., Lee W.J., Chien M.H. Downregulating CD26/DPPIV by apigenin modulates the interplay between Akt and Snail/Slug signaling to restrain metastasis of lung cancer with multiple EGFR statuses. J. Exp. Clin. Cancer Res. 2018;37:199. doi: 10.1186/s13046-018-0869-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Shrivastava S., Jeengar M.K., Thummuri D., Koval A., Katanaev V.L., Marepally S., Naidu V.G.M. Cardamonin, a chalcone, inhibits human triple negative breast cancer cell invasiveness by downregulation of Wnt/β-catenin signaling cascades and reversal of epithelial-mesenchymal transition. Biofactors. 2017;43:152–169. doi: 10.1002/biof.1315. [DOI] [PubMed] [Google Scholar]
- 170.Dogan Turacli I., Demirtas Korkmaz F., Candar T., Ekmekci A. Flavopiridol’s effects on metastasis in KRAS mutant lung adenocarcinoma cells. J. Cell Biochem. 2018 doi: 10.1002/jcb.27846. in press. [DOI] [PubMed] [Google Scholar]
- 171.Cichon M.A., Radisky D.C. ROS-induced epithelial-mesenchymal transition in mammary epithelial cells is mediated by NF-kB-dependent activation of Snail. Oncotarget. 2014;5:2827–2838. doi: 10.18632/oncotarget.1940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Fukawa T., Kajiya H., Ozeki S., Ikebe T., Okabe K. Reactive oxygen species stimulates epithelial mesenchymal transition in normal human epidermal keratinocytes via TGF-beta secretion. Exp. Cell Res. 2012;318:1926–1932. doi: 10.1016/j.yexcr.2012.05.023. [DOI] [PubMed] [Google Scholar]
- 173.Jiang J., Wang K., Chen Y., Chen H., Nice E.C., Huang C. Redox regulation in tumor cell epithelial-mesenchymal transition: Molecular basis and therapeutic strategy. Signal. Transduct. Target Ther. 2017;2:17036. doi: 10.1038/sigtrans.2017.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Li W., Cao L., Han L., Xu Q., Ma Q. Superoxide dismutase promotes the epithelial-mesenchymal transition of pancreatic cancer cells via activation of the H2O2/ERK/NF-κB axis. Int. J. Oncol. 2015;46:2613–2620. doi: 10.3892/ijo.2015.2938. [DOI] [PubMed] [Google Scholar]
- 175.Guerra F., Guaragnella N., Arbini A.A., Bucci C., Giannattasio S., Moro L. Mitochondrial Dysfunction: A Novel Potential Driver of Epithelial-to-Mesenchymal Transition in Cancer. Front. Oncol. 2017;7:295. doi: 10.3389/fonc.2017.00295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Gugnoni M., Sancisi V., Manzotti G., Gandolfi G., Ciarrocchi A. Autophagy and epithelial-mesenchymal transition: An intricate interplay in cancer. Cell Death Dis. 2016;7:e2520. doi: 10.1038/cddis.2016.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Zhang X., Cheng Q., Yin H., Yang G. Regulation of autophagy and EMT by the interplay between p53 and RAS during cancer progression (Review) Int. J. Oncol. 2017;51:18–24. doi: 10.3892/ijo.2017.4025. [DOI] [PubMed] [Google Scholar]
- 178.Wang H., Khor T.O., Shu L., Su Z.Y., Fuentes F., Lee J.H., Kong A.N. Plants vs. Cancer: A Review on Natural Phytochemicals in Preventing and Treating Cancers and Their Druggability. Anticancer Agents Med. Chem. 2012;12:1281–1305. doi: 10.2174/187152012803833026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Block G., Patterson B., Subar A. Fruit, vegetables, and cancer prevention: A review of the epidemiological evidence. Nutr. Cancer. 1992;18:1–29. doi: 10.1080/01635589209514201. [DOI] [PubMed] [Google Scholar]
- 180.Mollakhalili Meybodi N., Mohammad Mortazavian A., Bahadori Monfared A., Sohrabvandi S., Aghaei Meybodi F. Phytochemicals in Cancer Prevention: A Review of the Evidence. Iran. J. Cancer Prev. 2017;10:e7219. doi: 10.17795/ijcp-7219. [DOI] [Google Scholar]
- 181.Pasko P., Makowska-Was J., Chlopicka J., Szlosarczyk M., Tyszka-Czochara M., Dobrowolska-Iwanek J., Agnieszka Galanty A. South Siberian fruits: Their selected chemical constituents, biological activity, and traditional use in folk medicine and daily nutrition. J. Med. Plants Res. 2012;6:4698–4706. doi: 10.5897/JMPR12.874. [DOI] [Google Scholar]
- 182.Mulabagal V., Lang G.A., DeWitt D.L., Dalavoy S.S., Nair M.G. Anthocyanin content, lipid peroxidation and cyclooxygenase enzyme inhibitory activities of sweet and sour cherries. J. Agric. Food Chem. 2009;57:1239–1246. doi: 10.1021/jf8032039. [DOI] [PubMed] [Google Scholar]
- 183.Bobe G., Wang B., Seeram N.P., Nair M.G., Bourquin L.D. Dietary anthocyanin-rich tart cherry extract inhibits intestinal tumorigenesis in APC(Min) mice fedsuboptimal levels of sulindac. J. Agric. Food Chem. 2006;54:9322–9328. doi: 10.1021/jf0612169. [DOI] [PubMed] [Google Scholar]
- 184.Voronina L.M., Kravchenko G.B., Zagaiko A.L., Upyr L.V., Popovich V.P. The study of some plant extracts’ effect on the exudative phase of inflammation induced by introduction of different phlogogenes. News Pharm. 2010;1:66–68. [Google Scholar]
- 185.Noratto G., Porter W., Byrne D., Cisneros-Zevallos L. Identifying peach and plum polyphenols with chemopreventive potential against estrogen-independent breastcancer cells. J. Agric. Food Chem. 2009;57:5219–5226. doi: 10.1021/jf900259m. [DOI] [PubMed] [Google Scholar]
- 186.Lea M.A., Ibeh C., des Bordes C., Vizzotto M., Cisneros-Zevallos L., Byrne D.H., Okie W.R., Moyer M.P. Inhibition of growth and induction of differentiation of colon cancer cells by peach and plum phenolic compounds. Anticancer Res. 2008;28:2067–2076. [PubMed] [Google Scholar]
- 187.Zagayko A.L., Senyuk I.V., Lenchyk L.V., Galimullin R.M. Study of the antioxidant activity of the extract from common plum leaves. Ukr. Biopharm. J. 2014;1:25–28. [Google Scholar]
- 188.Kang S.Y., Seeram N.P., Nair M.G., Bourquin L.D. Tart cherry anthocyanins inhibit tumor development in Apc(Min) mice and reduce proliferation of human colon cancer cells. Cancer Lett. 2003;194:13–19. doi: 10.1016/S0304-3835(02)00583-9. [DOI] [PubMed] [Google Scholar]
- 189.Lefranc F., Tabanca N., Kiss R. Assessing the anticancer effects associated with food products and/or nutraceuticals using in vitro and in vivo preclinical development-related pharmacological tests. Semin. Cancer Biol. 2017;46:14–32. doi: 10.1016/j.semcancer.2017.06.004. [DOI] [PubMed] [Google Scholar]
- 190.Bag A., Bag N. Tea Polyphenols and Prevention of Epigenetic Aberrations in Cancer. J. Nat. Sci. Biol. Med. 2018;9:2–5. doi: 10.4103/jnsbm.JNSBM_46_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Pandey K.B., Rizvi S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell Longev. 2009;2:270–278. doi: 10.4161/oxim.2.5.9498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Busch C., Burkard M., Leischner C., Lauer U.M., Frank J., Venturelli S. Epigenetic activities of flavonoids in the prevention and treatment of cancer. Clin. Epigenet. 2015;7:64. doi: 10.1186/s13148-015-0095-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Russo M., Spagnuolo C., Tedesco I., Bilotto S., Russo G.L. The flavonoid quercetin in disease prevention and therapy: Facts and fancies. Biochem. Pharmacol. 2012;83:6–15. doi: 10.1016/j.bcp.2011.08.010. [DOI] [PubMed] [Google Scholar]
- 194.Lim T.G., Kwon J.Y., Kim J., Song N.R., Lee K.M., Heo Y.S., Lee H.J., Lee K.W. Cyanidin-3-glucoside suppresses B[a]PDE-induced cyclooxygenase-2 expression by directlyinhibiting Fyn kinase activity. Biochem. Pharmacol. 2011;82:167–174. doi: 10.1016/j.bcp.2011.03.032. [DOI] [PubMed] [Google Scholar]
- 195.Kim J.E., Kwon J.Y., Seo S.K., Son J.E., Jung S.K., Min S.Y., Hwang M.K., Heo Y.S., Lee K.W., Lee H.J. Cyanidin suppresses ultraviolet B-induced COX-2 expression in epidermal cells by targeting MKK4, MEK1, and Raf-1. Biochem. Pharmacol. 2010;79:1473–1482. doi: 10.1016/j.bcp.2010.01.008. [DOI] [PubMed] [Google Scholar]
- 196.Dreiseitel A., Oosterhuis B., Vukman K.V., Schreier P., Oehme A., Locher S., Hajak G., Sand P.G. Berry anthocyanins and anthocyanidins exhibit distinct affinities for the efflux transporters BCRP and MDR1. Br. J. Pharmacol. 2009;158:1942–1950. doi: 10.1111/j.1476-5381.2009.00495.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Ullah N., Ahmad M., Aslam H., Tahir M.A., Aftab M., Bibi N., Ahmad S. Green tea phytocompounds as anticancer: A review. Asian Pac. J. Trop. Dis. 2016;6:330–336. doi: 10.1016/S2222-1808(15)61040-4. [DOI] [Google Scholar]
- 198.Moreno F.S., Heidor R., Pogribny I.P. Nutritional Epigenetics and the Prevention of Hepatocellular Carcinoma with Bioactive Food Constituents. Nutr. Cancer. 2016;68:719–733. doi: 10.1080/01635581.2016.1180410. [DOI] [PubMed] [Google Scholar]
- 199.Park J.S., Rho H.S., Kim D.H., Chang I.S. Enzymatic preparation of kaempferol from green tea seed and its antioxidant activity. J. Agric. Food Chem. 2006;54:2951–2956. doi: 10.1021/jf052900a. [DOI] [PubMed] [Google Scholar]
- 200.Nöthlings U., Murphy S.P., Wilkens L.R., Henderson B.E., Kolonel L.N. Flavonols and pancreatic cancer risk: The multiethnic cohort study. Am. J. Epidemiol. 2007;166:924–931. doi: 10.1093/aje/kwm172. [DOI] [PubMed] [Google Scholar]
- 201.Cui Y., Morgenstern H., Greenland S., Tashkin D.P., Mao J.T., Cai L., Cozen W., Mack T.M., Lu Q.Y., Zhang Z.F. Dietary flavonoid intake and lung cancer—A population-based case-control study. Cancer. 2008;112:2241–2248. doi: 10.1002/cncr.23398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Gacche R.N., Shegokar H.D., Gond D.S., Yang Z., Jadhav A.D. Evaluation of selected flavonoids as antiangiogenic, anticancer, and radical scavenging agents: An experimental and in silico analysis. Cell Biochem. Biophys. 2011;61:651–663. doi: 10.1007/s12013-011-9251-z. [DOI] [PubMed] [Google Scholar]
- 203.Calderón-Montaño J.M., Burgos-Morón E., Pérez-Guerrero C., López-Lázaro M. A review on the dietary flavonoid kaempferol. Mini Rev. Med. Chem. 2011;11:298–344. doi: 10.2174/138955711795305335. [DOI] [PubMed] [Google Scholar]
- 204.Gupta S.C., Kim J.H., Prasad S., Aggarwal B.B. Regulation of survival, proliferation, invasion, angiogenesis, and metastasis of tumor cells through modulation of inflammatory pathways by nutraceuticals. Cancer Metastasis Rev. 2010;29:405–434. doi: 10.1007/s10555-010-9235-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Hsu C.P., Shih Y.T., Lin B.R., Chiu C.F., Lin C.C. Inhibitory effect and mechanisms of an anthocyanins- and anthocyanidins-rich extract from purple-shoot tea on colorectal carcinoma cell proliferation. J. Agric. Food Chem. 2012;60:3686–3692. doi: 10.1021/jf204619n. [DOI] [PubMed] [Google Scholar]
- 206.Li L., Adams L.S., Chen S., Killian C., Ahmed A., Seeram N.P. Eugenia jambolana Lam.berry extract inhibits growth and induces apoptosis of human breast cancer but not non-tumorigenic breast cells. J. Agric. Food Chem. 2009;57:826–831. doi: 10.1021/jf803407q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Lu J.N., Lee W.S., Nagappan A., Chang S.H., Choi Y.H., Kim H.J., Kim G.S., Ryu C.H., Shin S.C., Jung J.M., et al. Anthocyanins From the Fruit of VitiscoignetiaePulliat Potentiate the Cisplatin Activity by Inhibiting PI3K/Akt Signaling Pathways in Human Gastric Cancer Cells. J. Cancer Prev. 2015;20:50–56. doi: 10.15430/JCP.2015.20.1.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Rani N., Bharti S., Krishnamurthy B., Bhatia J., Sharma C., Kamal M.A., Ojha S., Arya D.S. Pharmacological Properties and Therapeutic Potential of Naringenin: A Citrus Flavonoid of Pharmaceutical Promise. Curr. Pharm. Des. 2016;22:4341–4359. doi: 10.2174/1381612822666160530150936. [DOI] [PubMed] [Google Scholar]
- 209.Mir I.A., Tiku A.B. Chemopreventive and therapeutic potential of “naringenin,” a flavanone present in citrus fruits. Nutr. Cancer. 2015;67:27–42. doi: 10.1080/01635581.2015.976320. [DOI] [PubMed] [Google Scholar]
- 210.Caselli A., Cirri P., Santi A., Paoli P. Morin: A Promising Natural Drug. Curr. Med. Chem. 2016;23:774–791. doi: 10.2174/0929867323666160106150821. [DOI] [PubMed] [Google Scholar]
- 211.Yamamoto T., Sakamoto C., Tachiwana H., Kumabe M., Matsui T., Yamashita T., Shinagawa M., Ochiai K., Saitoh N., Nakao M. Endocrine therapy-resistant breast cancer model cells are inhibited by soybean glyceollin I through Eleanor non-coding RNA. Sci. Rep. 2018;8:15202. doi: 10.1038/s41598-018-33227-y. [DOI] [PMC free article] [PubMed] [Google Scholar]