Abstract
Epilepsy is a neuronal dysfunction syndrome characterized by transient and diffusely abnormal discharges of neurons in the brain. Previous studies have shown that mutations in the syntaxin 1b (stx1b) gene cause a familial, fever-associated epilepsy syndrome. It is unclear as to whether the stx1b gene also correlates with other stimulations such as flashing and/or mediates the effects of antiepileptic drugs. In this study, we found that the expression of stx1b was present mainly in the brain and was negatively correlated with seizures in a pentylenetetrazole (PTZ)-induced seizure zebrafish model. The transcription of stx1b was inhibited by PTZ but rescued by valproate, a broad-spectrum epilepsy treatment drug. In the PTZ–seizure zebrafish model, stx1b knockdown aggravated larvae hyperexcitatory swimming and prompted abnormal trajectory movements, particularly under lighting stimulation; at the same time, the expression levels of the neuronal activity marker gene c-fos increased significantly in the brain. In contrast, stx1b overexpression attenuated seizures and decreased c-fos expression levels following PTZ-induced seizures in larvae. Thus, we speculated that a deficiency of stx1b gene expression may be related with the onset occurrence of clinical seizures, particularly photosensitive seizures. In addition, we found that berberine (BBR) reduced larvae hyperexcitatory locomotion and abnormal movement trajectory in a concentration-dependent manner, slowed down excessive photosensitive seizure-like swimming, and assisted in the recovery of the expression levels of STX1B. Under the downregulation of STX1B, BBR’s roles were limited: specifically, it only slightly regulated the levels of the two genes stx1b and c-fos and the hyperexcitatory motion of zebrafish in dark conditions and had no effect on the overexcited swimming behavior seen in conjunction with lighting stimulation. These findings further demonstrate that STX1B protein levels are negatively correlated with a seizure and can decrease the sensitivity of the photosensitive response in a PTZ-induced seizure zebrafish larvae; furthermore, STX1B may partially mediate the anticonvulsant effect of BBR. Additional investigation regarding the relationship between STX1B, BBR, and seizures could provide new cues for the development of novel anticonvulsant drugs.
Keywords: STX1B, berberine, epilepsy, photosensitive seizure, PTZ, zebrafish
Introduction
Epilepsy is a chronic neurological disease with a high prevalence characterized by spontaneous seizures, abnormal discharges of the brain, and convulsion. According to statistics, 1% of the global population suffers from epilepsy; among them, children, 1 out of 200 of whom are affected (Cowan, 2002; Poduri and Lowenstein, 2011). According to the International League Against Epilepsy 2017 Classification of Seizure Types Basic Version, three major types exist – focal onset, generalized onset, and unknown onset – in which the motor type of seizure is involved. Notably, hyperkinetic seizures have been specified as a subtype of motor onset under focal onset. Patients with motor onset usually suffer a sudden loss of consciousness and symptoms such as rigidity and convulsion. Furthermore, about 20% of epilepsy patients demonstrate other mental illnesses due to anxiety and sleep problems (Sillanpaa et al., 2016; Besag, 2018). Therefore, epilepsy is a serious social burden and a threat to patients in terms of both their physical and mental health, and often brings about great loss of property.
Photosensitive epilepsy is caused by visual stimuli with an abnormal electroencephalogram response, which is known as a photoparoxysmal response (Fisher et al., 2005). Recently, people are increasingly coming into contact with more electronic devices, such as televisions, computers, cameras, and other similar items. Unfortunately, this growth in intermittent photic stimulation has greatly increased the prevalence of epileptic seizures. Therefore, the incidence of photosensitive epilepsy is also increasing (Poleon and Szaflarski, 2017), with approximately 5% of epilepsy patients being affected (Martins da Silva and Leal, 2017). A recent study employed gene sequencing to identify the cause of the archetypal generalized photosensitive epilepsy syndrome as a chromodomain helicase DNA-binding protein 2 (CHD2) mutation and found approximately five times as many CHD2 variants in photosensitive epilepsy patients as in the controls (Galizia et al., 2015; Poleon and Szaflarski, 2017). According to another study, bromodomain-containing protein 2 might be an underlying susceptible gene for the photoparoxysmal response (Lorenz et al., 2006). However, despite the efforts of these investigations, the pathogenesis of photosensitive epilepsy is still unclear.
Syntaxin 1b (STX1B) is a soluble, N-ethylmaleimide-sensitive fusion attachment receptor (SNARE) protein located in the presynaptic membrane that mediates the fusion of the synapse vesicle and the target membrane, promotes the release of neurotransmitters, and is expressed in the central nervous system (Sollner et al., 1993; Sudhof, 2013; Zhou et al., 2013). According to previous reports, the mutation of stx1b is related to the onset of familial fever-associated epilepsy syndromes. In previous research, stx1b knockdown presented abnormal electrographic activity in zebrafish larvae under hyperthermic conditions (Schubert et al., 2014; Kearney, 2015). Clinical observation found that the presentation of myoclonic astatic epilepsy (MAE) was also related to the variant or deletion of the stx1b gene, suggesting that STX1B should closely observed in the diagnosis of MAE (Vlaskamp et al., 2016). Whether STX1B is involved in photosensitive epilepsy or not has to our knowledge, not yet been reported on.
Berberine (BBR) is a natural compound extracted from the traditional Chinese herb Coptis chinensis and has for many years been known to have a good effect on diarrhea. Studies have shown that BBR also has potential therapeutic effects in diabetes (Zhang et al., 2010), hyperlipidemia (Kong et al., 2004; Kim et al., 2009), heart disease (Lau et al., 2001; Zeng et al., 2003), and inflammation (Choi et al., 2006; Lou et al., 2011). In addition, BBR was found to have a neuroprotective effect on multiple central nervous system diseases, such as Alzheimer’s disease and epilepsy (Kulkarni and Dhir, 2010; Gao et al., 2014; Hussien et al., 2018). In one study, BBR notably improved cognitive behavior in a rat model of Alzheimer’s disease and inhibited the formation of Aβ42, a main constituent of amyloid-β plaques associated with the neurodegenerative condition (Hussien et al., 2018). Another investigation reported that BBR increased the levels of both interleukin 1β and inducible nitric oxide synthase to mediate neuroprotective properties and ameliorated spatial memory impairment in a rat model of Alzheimer’s disease (Zhu and Qian, 2006). In a kainate-induced temporal lobe seizure rat model, BBR significantly decreased the incidence of seizures (Mojarad and Roghani, 2014). Furthermore, in a pilocarpine-induced seizure rat model, BBR delayed both latency to the first seizure and time to the development of status epilepticus (Gao et al., 2014). However, few studies on the antiepileptic mechanism of BBR have been published to date. Pentylenetetrazole (PTZ) is a gamma-aminobutyric acid (GABA) receptor inhibitor (Macdonald and Barker, 1977) capable of resisting the inhibitory effect of GABA on neural activity and is often used in seizure models in rodents and zebrafish (Baraban et al., 2005; Stewart et al., 2012; Epps and Weinshenker, 2013; Grone and Baraban, 2015). A number of studies have presented zebrafish epilepsy-like seizures via PTZ induction models over the past 10 years (Baraban et al., 2005; Ellis and Soanes, 2012; Stewart et al., 2012; Gupta et al., 2014; Rahn et al., 2014; Torres-Hernandez et al., 2015; Barbalho et al., 2016). Referring to Barabans’ research (Baraban et al., 2005), we established a zebrafish seizure model using PTZ and studied the zebrafish convulsive episodes under a dark condition and lighting stimulation; using this model, the correlations of STX1B with seizures and the anticonvulsant effects of BBR were investigated. We found that BBR can promote the expression of STX1B directly or indirectly and alleviate epilepsy-like seizures, especially photosensitive seizures in PTZ-induced seizure zebrafish larvae.
Materials and Methods
Zebrafish Feeding and Care
AB wild-type line zebrafish (Danio rerio) were obtained from the College of Life Sciences and Technology of Tsinghua University in Beijing, China. The zebrafish were raised under standard laboratory conditions with a 14-h light/10-h dark cycle at a temperature of 28.5°C ± 1°C (Kimmel et al., 1995). Zebrafish embryos and larvae were incubated in the rearing water of 280 mg/L Tropical Marine Artificial Seawater Crystal (CNSIC Marine Biotechnology Co., Ltd., Tianjin, China), with a conductivity of 450 to 550 μS. This research was reviewed and approved by the Laboratory Animal Management and Animal Welfare Committee at the Institute of Medicinal Biotechnology of the Chinese Academy of Medical Sciences. The zebrafish experimental protocols complied with the Ethics of Animal Experiments guidelines set by the Institute of Medicinal Biotechnology of the Chinese Academy of Medical Sciences.
Microinjection
Two stx1b morpholino oligos and a scrambled morpholino oligo were purchased from Gene Tools, LLC (Philomath, OR, United States). The two stx1b morpholino oligo sequences were as follows: 5′-GTGCGATCCTTCATTTTTCCCCGCC-3′ (stx1b-MO1) and 5′-AAATATCTCTTGAGATGTCCGCTGC-3′ (stx1b-MO2) (Schubert et al., 2014), which are the stx1b antisense oligos used to inhibit STX1B expression by binding to STX1B initiation codon sites. The scrambled morpholino oligo with a randomized 25-base sequence designed by Gene Tools1 (Philomath, OR, United States) was used as a nonsense control for stx1b-MO. As part of the present study, 0.5 nL of 50 μM stx1b-MO1 or stx1b-MO2 was injected into each embryo of the 1–4-cell stage, and the embryos were subsequently cultivated in the rearing water as described above. STX1B overexpression was prompted via injection of 0.5 nL of pIRES2-stx1b-EGFP and pIRES2-EGFP (as a mock control) at a concentration of 60 ng/μL. The injected embryos at 5 days postfertilization (dpf) were collected for subsequent experiments.
Chemical Treatment
Berberine was obtained from the National Institutes for Food and Drug Control (Beijing, China). Valproate (VPA) (valproic acid sodium salt, P4543) and PTZ (P6500) were purchased from Sigma-Aldrich (St. Louis, MO, United States).
For the seizure model group, we essentially followed the method described by Baraban et al. (2005). Briefly, zebrafish larvae at 7 dpf were exposed to a PTZ solution at concentrations of 2, 4, and 6 mM, respectively, for 1 h and then collected for a behavioral experiment or for 2 h and then collected for in situ hybridization and western blotting experiments. Based on the results of the PTZ dose experiment, 4 mM of PTZ was used for the subsequent experiments conducted in the PTZ–seizure-related groups. Each group contained 24 larvae.
For the drug-treated groups, wild-type larvae and injected larvae at 5 dpf were exposed to BBR at concentrations of 25, 50, and 75 μM or to VPA at concentrations of 60, 120, and 240 μM for 2 days (7 dpf), respectively, after being washed three times with the normal rearing solution. Then, the larvae were exposed to 4 mM of PTZ for 1 h and collected for a behavioral experiment, or after 2 h collected for subsequent experiments including whole-mount in situ hybridization and western blotting detections for c-fos and stx1b transcription and protein levels.
Whole-Mount in situ Hybridization
Sense and antisense RNA probes of the genes c-fos and stx1b were synthesized using a digoxigenin RNA labeling kit (1175025; Roche Applied Science, Penzberg, Germany) and complementary DNA fragment templates that were amplified using reverse transcription-polymerase chain reaction and inserted into a pGEM-T plasmid. Gene c-fos primer pair sequences were as follows: 5′-AACTGTCACGGCGATCTCTT-3′ (the forward primer) and 5′-CTTGCAGATGGGTTTGTGTG (the reverse primer) (Baraban et al., 2005). Gene stx1b primer pair sequences were as follows: 5′-GCAGCACCAAACCCTGATGAAA (the forward primer) and 5′-CCTCCGATACTGGACCGCAAAA (the reverse primer). Larvae were fixed with 4% paraformaldehyde overnight at 4°C before being stored in methanol at 4°C. Procedures for whole-mount in situ hybridization were performed as described by Whitlock and Westerfield (2000).
Western Blotting
For western blot analysis, total proteins were extracted from zebrafish larvae with a RIPA lysis kit (C1053; Applygen Technologies Inc., Beijing, China), separated using 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis, and transferred to a nitrocellulose filter (T41524; PALL, Mexico). Protein blots were blocked with 5% milk in Tris-buffered saline for 1 h at room temperature, with antibodies against STX1B (1:1,000 dilution; 110 403; Synaptic Systems, Coventry, United Kingdom) and β-actin (1:2,000 dilution; A5441; Sigma-Aldrich, St. Louis, MO, United States). The blots were incubated with secondary antibodies (goat anti-mouse or goat anti-rabbit immunoglobulin G from ZSGB-BIO, Beijing, China) for 1 h and visualized by an immobilon western chemiluminescent horseradish peroxidase substrate (Millipore, Billerica, MA, United States). The western blotting was performed in parallel three times.
Behavioral Experiment
All of the zebrafish swimming activity was analyzed at 7 dpf by the ZebraLab Video-Track system version 3.3 (ViewPoint Life Science, Montreal, QC, Canada). The zebrafish larvae were individually placed into the wells of a 48-well plate (1 fish/well). Locomotor distance, velocity, and swimming tracks were separately recorded in two kinds of conditions. In the first, the larvae stayed in a dark box and their swimming actions were recorded for 20 min, during which time the data and moving tracks were collected once every 2 min, with a red trajectory indicating an abnormal swimming trajectory and an overspeed higher than 4 cm/s defined as a highly active movement and a green trajectory indicating a velocity between 0.2 and 4 cm/s defined as an active movement, respectively. The second condition involved a shift experiment between dark and light, in which the zebrafish larvae were subjected to three cycles of 5 min dark and 10 s light periods, with data collected once every 10 s.
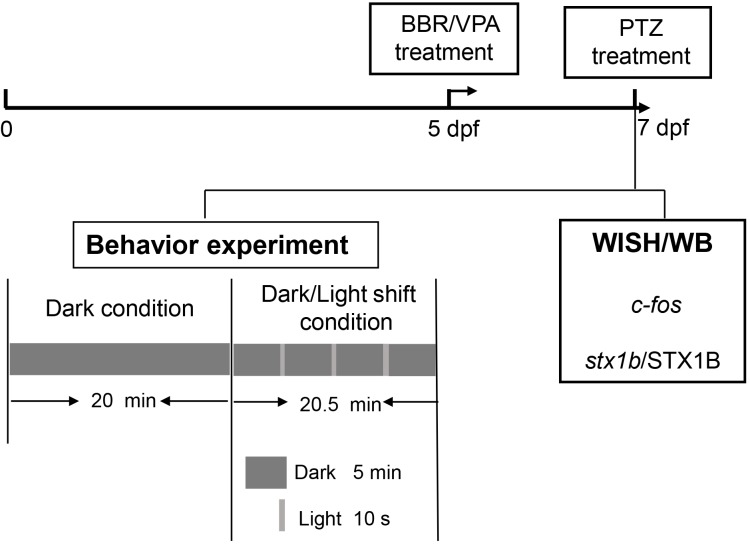
The experimental procedure and pharmacological manipulations in this study are depicted in the flowchart in Figure 1.
FIGURE 1.
A representative flowchart of the experimental procedure and methods in this study.
Statistical Analysis
All data were plotted by using GraphPad Prism 5.0 (GraphPad Software Inc., La Jolla, CA, United States). Comparisons between multiple groups were carried out using the analysis of variance test. Significance for all tests was defined at ∗/#/&/Φ/𝜃/φ /§ P < 0.05; ∗∗/##/&&/ΦΦ/𝜃𝜃/φ φ /§§ P < 0.01; and ∗∗∗/###/&&&/ΦΦΦ/𝜃𝜃𝜃/φ φ φ /§§§ P < 0.001.
Results
PTZ Induced a Zebrafish Epilepsy-Like Seizure Model and Suppressed Expression of stx1b Gene in Zebrafish Larvae Brains
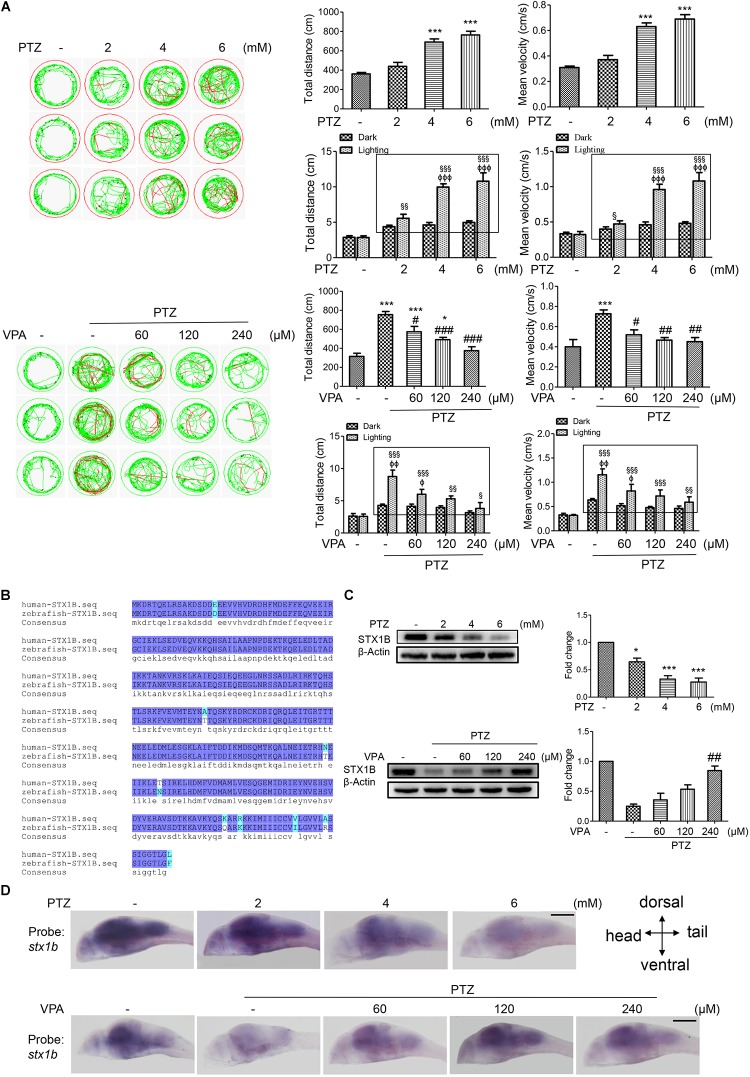
Previous research has reported that the human stx1b gene is associated with familial fever-associated epilepsy syndromes and plays a part in rescuing the function of stx1b knockdown in zebrafish (Schubert et al., 2014; Kearney, 2015). In the present study, we are interested in whether STX1B is also related to the seizures caused by PTZ and if it mediates the effects of the antiepileptic drugs VPA and BBR in zebrafish. First, we set up a zebrafish seizure model using PTZ and confirmed the model by use of a VPA. In this model, larvae swimming distance, velocity, and abnormal trajectory were significantly increased in a PTZ dose-dependent manner and were aggravated particularly under the condition of a shift between dark and light (Figure 2A). VPA showed an obvious therapeutic effect on the seizure-like swimming; specifically, the PTZ-induced larval overspeed swimming was slowed down in a VPA dose-dependent manner under both the dark condition and the dark–light shift condition (Figure 2A). Then, we compared the homology between human and zebrafish STX1B protein sequences. Each of these two STX1B proteins consist of 288 amino acids with a positive ratio of 98% and an identity ratio of 96.8%, in which only 5 amino acids are different and 4 amino acids have similar polarity (Figure 2B). Therefore, it can be speculated that both proteins may have similar biological functions. Western blotting confirmed that the STX1B protein was decreased by PTZ and increased by VPA in larvae (Figure 2C). In addition, hybridization in situ results showed that the stx1b gene was expressed mainly in the brain region and clearly downregulated by PTZ and recovered by VPA in a dose-dependent manner (Figure 2D). This overexcited behavior was inversely proportional to the STX1B level. These results indicate that the STX1B level is negatively associated with PTZ-induced seizures in zebrafish and more closely correlated with a photosensitive seizure. Based on these results, we chose a PTZ concentration of 4 mM for our PTZ-induced seizure model and a VPA dose of 120 μM as a positive control in the following experiments.
FIGURE 2.
Stx1b gene expression was suppressed in PTZ-induced seizure zebrafish larvae. (A) Seizure-like swimming was induced by PTZ in zebrafish larvae. The left track panel and the right upper histograms show the larval swimming behavior (distance and speed) during 20 min in the dark condition. The red trajectory indicates overactive movement and the green trajectory indicates active movement in the left track figure. The right upper histograms show the larval swimming distance and speed, which were recorded for 20 min in the dark condition. The right lower histograms show the larval swimming distance and speed in three cycles of 5 min dark and 10 s light periods; the open boxes show the differences in distance and velocity of the overexcited larvae between the dark and light conditions (n = 24). (B) Alignment of human and zebrafish STX1B amino acid sequences. The amino acids shown in dark blue are identical, those in shallow blue demonstrate amino acids with similar polarity, and those in white/shallow blue are different. (C) Western blotting tests indicated that STX1B protein was decreased by PTZ (Upper) and increased by VPA (Lower) in a concentration-dependent manner (n = 3). ∗P < 0.05 and ∗∗∗P < 0.001 vs. wild-type; #P < 0.05, ##P < 0.01, and ###P < 0.001 vs. PTZ model; § P < 0.05, §§ P < 0.01, and §§§P < 0.001 vs. wild-type in the light condition; ΦP < 0.01, ΦΦP < 0.01 and ΦΦΦP < 0.001 indicated light vs. dark in the same set of conditions. (D) Hybridization in situ results show STX1B gene expression in the larval brain inhibited by PTZ and rescued by VPA in a concentration-dependent manner (n = 20).
Level of stx1b Correlates Inversely With PTZ-Induced Seizure in Zebrafish Larvae
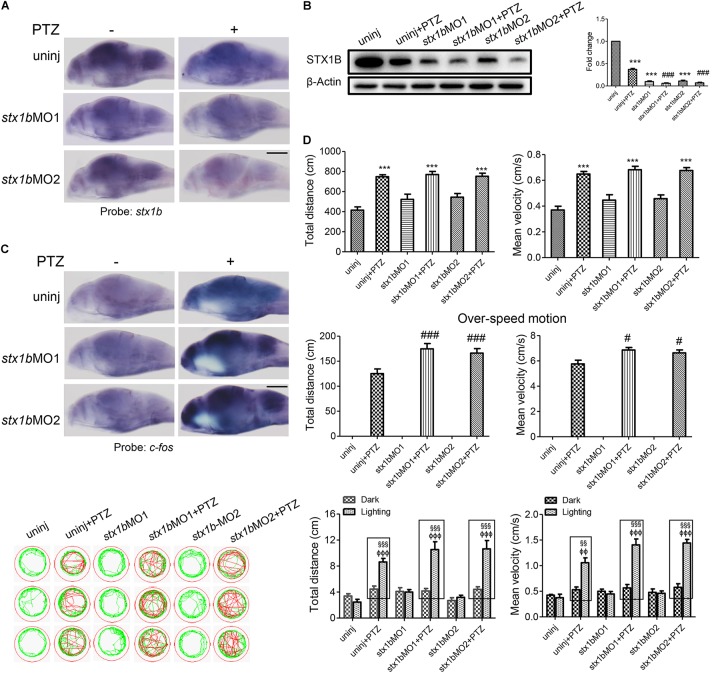
Further, we investigated whether STX1B could affect PTZ-induced seizures, especially under lighting stimulation using gene knockdown and overexpression methods. A stx1b overexpression plasmid and two stx1b morpholino oligos were separately injected into zebrafish embryos to upregulate or downregulate stx1b gene expression. When the zebrafish embryos injected with the stx1b morpholino oligos were exposed to PTZ, stx1b messenger RNA and protein levels were lower (Figures 3A,B) and the neuronal activity marker c-fos level was higher (Figure 3C) than in those larvae exposed only to PTZ or that received only a morpholino oligos injection. This suggests that the downregulation of STX1B combined with PTZ exposure worsened dysregulation of the two genes’ expression. Additionally, behavioral experiments showed that the knockdown of stx1b aggravated the abnormal swimming pathway and velocity instead of the total average velocity and distance in the dark condition and also intensified overexcited behavior under light stimulation induced by PTZ, in comparison with in the PTZ-only and morpholino oligos injection-only groups (Figure 3D). The behavior changes between the wild-type group and the groups that underwent morpholino oligos injection without PTZ induction were minor or not observed, meaning that the existence of a partial deficiency of STX1B in wild-type larvae did not affect their behavior too significantly. A scrambled MO as a nonsense control for stx1b-MO showed no effects on the expression of stx1b and c-fos and also did not change larval swimming behavior in comparison with the uninjected and PTZ-induction groups (relevant data supplied in the Supplementary Material). These results imply that the downregulation of STX1B probably promoted the onset of epilepsy-like seizures, particularly in the case of photic stimulation on the PTZ-treated zebrafish.
FIGURE 3.
Larval seizure was aggravated by downregulation of stx1b transcription in the PTZ-induced seizure model. Levels of stx1b gene transcription (n = 20) (A) and STX1B protein (n = 3) (B) were reduced and c-fos gene transcription was increased in the larval (7 dpf) brain (n = 20) (C) by stx1b morpholino oligos injection in the PTZ model, as compared with in the PTZ-only and the morpholino oligos-only injection models. The larval swimming experiment (n = 24) (D) showed that average speed and total distance were not changed, but that the abnormal pathway and overspeed were increased following 20 min in the dark condition and that photosensitive seizure was aggravated under the condition of light–dark transition with 5 min in the dark and 10 s in the light for three cycles in the PTZ plus stx1b morpholino oligos larvae, as compared with the two groups of the PTZ-only and the stx1b morpholino oligos-only injection models. The data show average speeds during the 20 min in the dark and the 10 s in the dark–light transformation; the boxes indicate the difference of locomotion distances and speeds between the light–dark transitions. Swimming tracks were recorded at 2 min in the dark condition and the red trajectory indicates overactive movement and the green trajectory indicates active movement. stx1b-MO1 and stx1b-MO2 were two morpholino oligos that bound to the stx1b messenger RNA initiate sequence with a different sequence; by using two target oligos, their inhibition effect was confirmed with each other. ∗∗∗P < 0.001 vs. wild-type; #P < 0.05 and ###P < 0.001 vs. PTZ model; §§P < 0.01 and §§§P < 0.001 vs. wild-type in the light condition; ΦΦP < 0.01 and ΦΦΦP < 0.001 indicates light vs. dark.
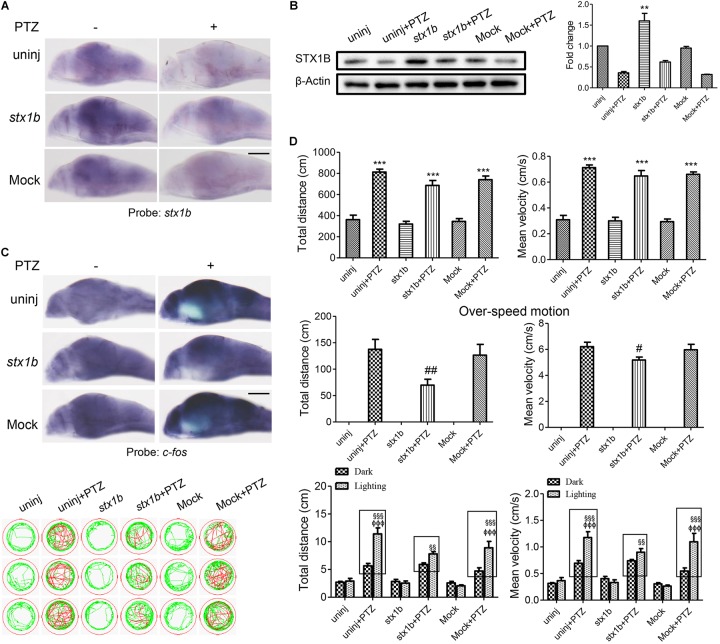
To further verify these outcomes, we constructed a stx1b overexpression vector and injected it into zebrafish embryos. As shown in Figures 4A,B, in the group of PTZ plus stx1b overexpression, the levels of stx1b messenger RNA and protein were higher than in the PTZ-treated group; in addition the expression of c-fos in the brain was significantly lower than in the PTZ-treated group (Figure 4C). Behavioral experiment results showed that the overexpression of STX1B had no significant effect on the total distance and average velocity of the PTZ-injected zebrafish, but had a notable reducing effect on abnormal trajectory and overspeed locomotion in the dark condition (Figure 4D). Moreover, the overexpression of STX1B significantly slowed down the PTZ-induced larval overexcited response in the dark–light shift condition (Figure 4D). Those results confirm that the upregulation of STX1B alleviated the seizure, including in particular a photosensitive seizure, in PTZ-treated zebrafish, suggesting that the overexpression of STX1B might have a potential protective effect in a PTZ-induced seizure model.
FIGURE 4.
Larval seizure-like behavior was reduced by increased STX1B level in a PTZ-induced seizure model. (A) Both stx1b transcription in the wild-type and PTZ models were enhanced in larval (7 dpf) brains by stx1b injection as compared with that following no injection and mock injection (n = 20). (B) Western blotting confirmed differential levels of STX1B protein among the variant groups; notably, the STX1B level was raised in the stx1b-PTZ group as compared with in the PTZ-only group (n = 3). (C) c-fos messenger RNA was decreased by STX1B overexpression in the stx1b-PTZ model versus in the PTZ model group or the mock-PTZ group (n = 20). (D) The larval swimming experiment showed that, with STX1B overexpression, average speed and total distance were not obviously changed but abnormal pathway and overspeed were significantly decreased with 20 min in the dark condition, while photosensitive seizure was inhibited under the condition of light–dark shift in the PTZ-model larvae as compared with in the two groups of the PTZ-only model and the PTZ plus mock injection model (n = 24). The rectangles indicate differential responses between light–dark transitions in the three groups of the PTZ-model larvae. ∗∗P < 0.01 and ∗∗∗P < 0.001 vs. wild-type; #P < 0.05 and ##P < 0.01 vs. PTZ model; §§ P < 0.01 and §§§ P < 0.001 vs. wild-type in the light condition; ΦΦΦP < 0.001 indicates light vs. dark.
Berberine Reduced the PTZ-Induced Seizure-Like Response by Promoting stx1b Gene Expression
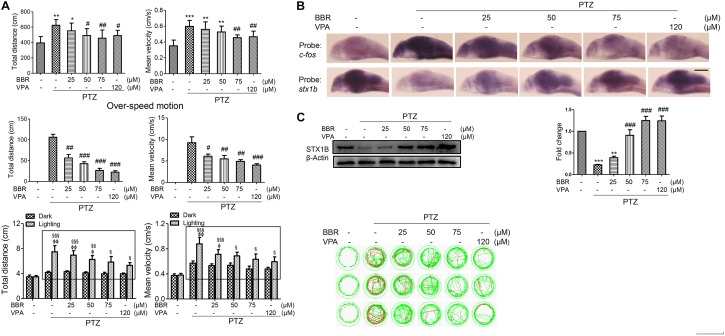
Previous studies have reported that the use of BBR significantly decreased the incidence of seizures in a seizure rat model (Mojarad and Roghani, 2014) and delayed both latency to the first seizure and time to develop status epilepticus in a pilocarpine-induced seizure rat model (Gao et al., 2014). However, few studies on the anticonvulsant mechanism of BBR have been reported at this time. In this work, we are interested in researching whether the stx1b gene correlates with the BBR anticonvulsant effect. A larval swimming experiment was first performed and the results showed that BBR reduced larval average velocity and total movement distance including abnormal swimming track and overspeed in the dark condition; in addition, BBR also more obviously alleviated a PTZ-induced overexcited response in the light stimulation condition in PTZ-induced zebrafish, in a dose-dependent manner (Figure 5A). In situ hybridization results showed that BBR inhibited the increase of the c-fos level induced by PTZ and promoted STX1B expression in a concentration-dependent manner in the brain of PTZ-treated larvae (Figure 5B). Furthermore, a western blotting test also confirmed that STX1B protein increased in a BBR concentration-dependent manner in PTZ–seizure larvae (Figure 5C). In these tests, a BBR effect that occurred at 75 μM was shown to be nearly similar to that seen with VPA at 120 μM. These results suggest that BBR probably has a therapeutic effect on PTZ-induced seizures in zebrafish. Therefore, we speculate that BBR might be able to suppress an epilepsy-like seizure by upregulating STX1B expression and also that the level of STX1B is associated with seizure outlook.
FIGURE 5.
Berberine (BBR) inhibited seizure in PTZ-model zebrafish with the increase of STX1B level. (A) A behavioral experiment showed that BBR inhibited the larval overexcited locomotion in speed and distance under the conditions of non-stimulation and dark–light cycling stimulation in PTZ-model larvae. A representative swimming trajectory (2 min) is presented (n = 24). The rectangles showed differential distances and speeds between light–dark transitions in the groups of PTZ plus BBR larvae as compared with in the PTZ-only model. (B) Hybridization in situ showed that BBR inhibited the increase of c-fos and rescued stx1b descending induced by PTZ in the larval (7 dpf) brains (n = 20). (C) Western blotting results confirmed that BBR recovered STX1B protein levels to almost normal in a dose-dependent manner (n = 3). ∗P < 0.05, ∗∗P < 0.01, and ∗∗∗P < 0.001 vs. wild-type; #P < 0.05, ##P < 0.01, and ###P < 0.001 vs. PTZ model; § P < 0.05, §§ P < 0.01, and §§§ P < 0.001 vs. wild-type in the light condition; ΦP < 0.01 and ΦΦP < 0.01 indicates light vs. dark.
STX1B Mediated the Therapeutic Effect of Berberine on PTZ-Induced Seizure in Zebrafish
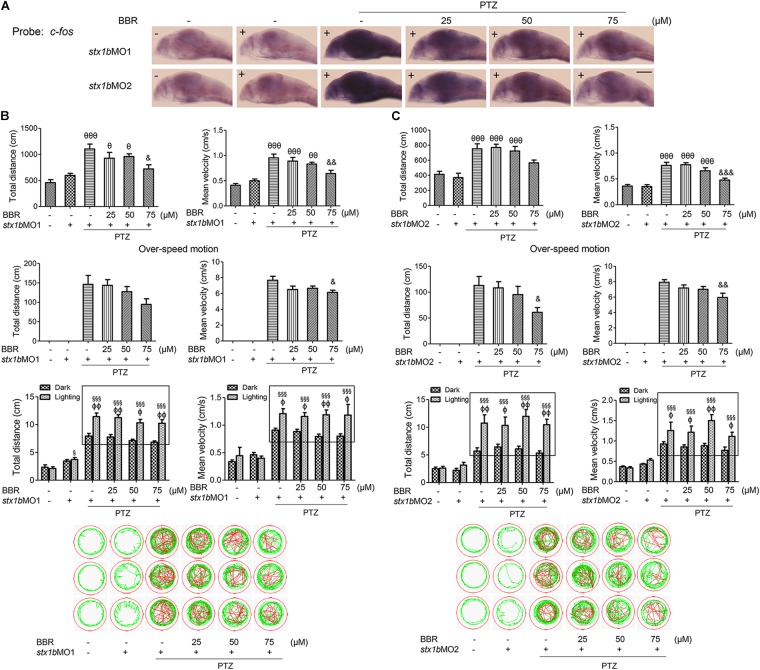
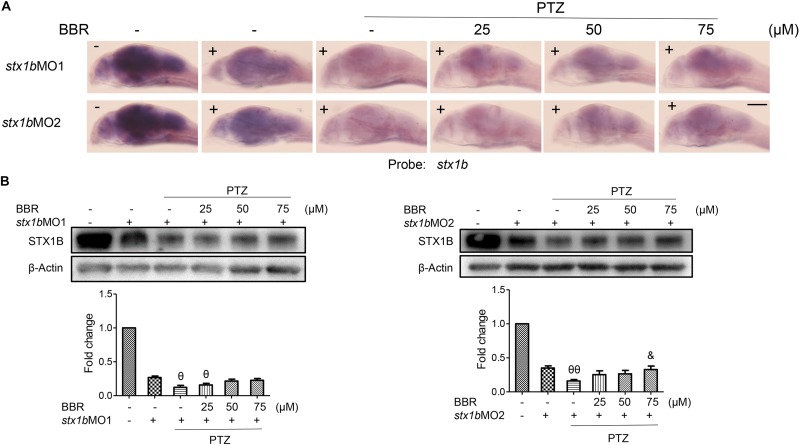
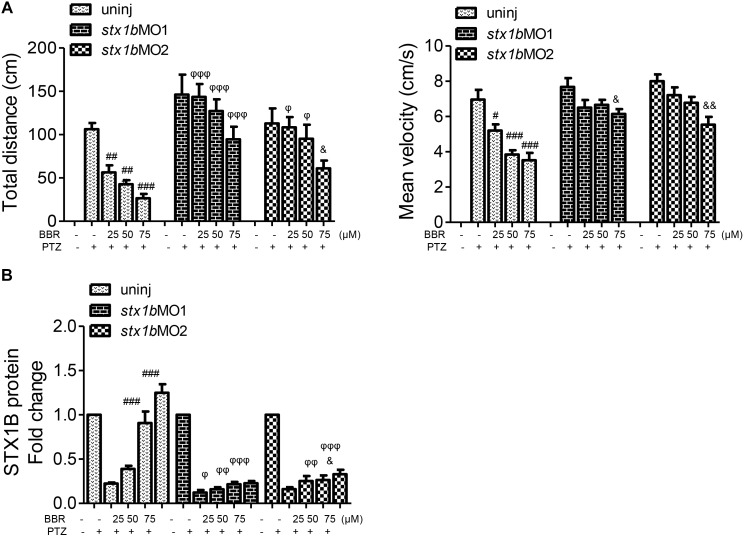
Since BBR is likely to suppress the onset of PTZ-induced seizures in zebrafish accompanying the enhancement of STX1B expression, we evaluated whether or not BBR was dependent on STX1B protein to play the anticonvulsant role in the zebrafish seizure model. In situ hybridization results showed that, under the stx1b morpholino oligos injection condition, BBR only moderately reduced the c-fos level in the brain region of PTZ-treated zebrafish (Figure 6A). Behavioral results revealed that BBR mildly attenuated the increase of the average velocity and total movement distance including the abnormal trajectory and overspeed (clonus-like convulsions) in the group of PTZ plus stx1b morpholino oligos in the dark condition (Figures 6B,C), suggesting that stx1b knockdown caused BBR inhibition action that was obviously weaker than that in PTZ-only-treated zebrafish (Figure 5A). Moreover, BBR did not prevent an overexcited response in the light stimulation condition (Figures 6B,C). Subsequently, we studied the efficiency of BBR activating STX1B expression under stx1b knockdown in the PTZ-treated larvae and found that BBR only slightly raised stx1b messenger RNA and protein levels in the PTZ plus stx1b morpholino oligos group, in which the STX1B level was lower than that in the stx1b morpholino oligos group and considerably lower than that in the normal control group (Figures 7A,B). Furthermore, a data comparative analysis was carried out between BBR with and without stx1b morpholino oligos injection and indicated that STX1B downregulation significantly weakened or even eliminated BBR efficiency for suppressing an epileptic seizure including abnormal trajectory and overspeed in the dark condition and STX1B protein levels (Figure 8) as well as photosensitive seizures (Figure 6) of the PTZ-induced seizure in zebrafish. Considering the cohesive tendency between the STX1B level variation and the larval behavior results, we infer that STX1B is an important mediator for BBR action on anticonvulsants, in particular for the inhibition of photosensitive seizures that may require proper STX1B expression.
FIGURE 6.
Downregulation of STX1B weakened the effects of BBR on anticonvulsant in the PTZ-induced seizure zebrafish model. (A) Hybridization in situ showed that there was a change in the c-fos messenger RNA level in the larval (7 dpf) brain that was induced by BBR in the PTZ plus stx1b morpholino oligos group versus in the three control groups of wild-type, stx1b morpholino oligos injection, and PTZ plus stx1b morpholino oligos (n = 20). (B,C) STX1B downregulation attenuated the efficiency of BBR inhibition on larval overexcited locomotion in terms of speed and distance under non-stimulation conditions and eliminated the action of BBR under dark–light transitions. Swimming trajectories are presented in 2 min recording charts; red tracks indicate over locomotion, while the rectangles indicate the difference between light–dark transitions (n = 24). &P < 0.05, &&P < 0.01, and &&&P < 0.001 vs. PTZ plus stx1b morpholino oligos model; 𝜃P < 0.05, 𝜃𝜃P < 0.01, and 𝜃𝜃𝜃P < 0.001 vs. stx1b morpholino oligos model; §P < 0.05 and §§§P < 0.001 vs. wild-type in the light condition; ΦP < 0.01 and ΦΦP < 0.01 indicates light vs. dark.
FIGURE 7.
Stx1b morpholino oligos injection suppressed BBR activation on STX1B expression in the PTZ-model larvae. (A) Hybridization in situ results show that a change of the stx1b messenger RNA level in the larval (7 dpf) brain was induced by BBR with stx1b morpholino oligos injection in the PTZ-model zebrafish, as compared with in the wild-type, stx1b morpholino oligos injection, and PTZ plus stx1b morpholino oligos groups (n = 20). (B) Western blotting results indicated a change of the STX1B protein level similar to the change of the stx1b messenger RNA level under the same treatments (n = 3). &P < 0.05 vs. PTZ plus stx1b morpholino oligos model; 𝜃P < 0.05 and 𝜃𝜃P < 0.01 vs. stx1b morpholino oligos model.
FIGURE 8.
Comparative analysis of epilepsy-like seizure and STX1B protein levels between BBR with and without stx1b morpholino oligos injection in the PTZ-induced seizure zebrafish. (A) Behavioral comparison indicates that the BBR effect of antiseizure was weakened by stx1b morpholino oligo injection. (B) Comparison of STX1B protein levels induced by BBR between stx1b gene knockdown and non-knockdown. Western blotting showed that levels of STX1B protein were significantly decreased by stx1b morpholino oligo injection under BBR existence. The histograms are generated from data in Figure 5 of behavior and western blotting, Figure 6 of behavior, and Figure 7 of western blotting. #P < 0.05, ##P < 0.01, and ###P < 0.001 vs. PTZ model; &P < 0.05 and &&P < 0.01 vs. PTZ plus stx1b morpholino oligos model. φ P < 0.05, φ φ P < 0.01, and φ φ φ P < 0.001 indicate differences in the comparison between uninjected and morpholino oligo injection in the PTZ model under the same concentration of BBR, respectively.
Discussion
STX1B is a synapse fusion protein that is associated with the release of neurotransmitters, and mutations of the stx1b gene lead to familial fever-associated epilepsy syndromes in humans (Sudhof, 2013; Schubert et al., 2014). Stx1b knockout mice (Stx1b-/-) demonstrated damaged glutamatergic and GABAergic synaptic transmissions (Mishima et al., 2014), while Stx1b+/- mice exhibited a reduced release of GABA and a disturbance of the dopaminergic system in the central nervous system (Fujiwara et al., 2017). GABA is an important inhibitory neurotransmitter in the brain, and the roles of GABA and its receptor in epilepsy have been widely studied (Ferando and Mody, 2012). PTZ is a regular compound used to trigger seizures in animal models that selectively blocks GABA receptor channels and weakens GABA-mediated neurotransmitter systems, causing the neurons to overexcite (Soares et al., 2017). In this study, we used PTZ to establish a zebrafish seizure model and researched STX1B functions in epilepsy-like seizures, including photosensitive seizures. We found that the stx1b gene expression decrease that accompanies epilepsy-like seizure aggravation, was induced by PTZ, and that STX1B increase and the alleviation of a seizure were observed under treatment of the anti-epilepsy drug VPA. Moreover, stx1b knockdown made zebrafish more sensitive to PTZ than just PTZ treatment did (Figure 3). This indicates that STX1B decline is closely related with PTZ-induced epileptic seizures and that STX1B might be a protein marker in a PTZ-induced seizure model for the screening of anticonvulsant drugs. The alignment of human and zebrafish STX1B protein sequences showed that the STX1B proteins have a high homology of 98% (Figure 2A) and that they possess the same structural domain as compared with syntaxin and SNARE2. Altogether, these results hint that STX1B may exert similar biological functions in zebrafish as in humans.
A photosensitive seizure is a kind of epileptic response to the visual stimuli of color and light. Triggers can include television and computer games, among many others (Martins da Silva and Leal, 2017). At present, the relationship between photosensitive epilepsy and other genes involved is not very clear, in spite of bromodomain-containing protein 2 and CHD2 being known as a likely susceptible gene in photosensitive epilepsy (Lorenz et al., 2006; Galizia et al., 2015; Poleon and Szaflarski, 2017). However, the correlation of STX1B to photosensitive epileptic seizures has not been reported until now. Photosensitive epilepsy does not only occur in a single kind of epilepsy syndrome; it has also been found in juvenile myoclonic epilepsy, eyelid myoclonia (Jeavons syndrome), and Dravet syndrome (Poleon and Szaflarski, 2017). According to a study, photosensitivity was reported to occur in approximately 31% of those with juvenile myoclonic epilepsy (Wolf and Goosses, 1986). Photosensitive epilepsy usually occurs in adolescents: it is estimated that patients between the ages of 7 and 19 years are about five times more likely than those in other age groups to demonstrate the condition (de Bittencourt, 2004). Therefore, it could be argued that photosensitive epilepsy is a serious threat to the physical and mental health of teenagers. In this study, we explore the correlation between STX1B level and photosensitive seizures under the condition of a dark–light shift in a PTZ–seizure zebrafish model. Our behavioral experiments show that PTZ treatment with stx1b knockdown made the larvae oversensitive to light stimulation, and the c-fos level (c-fos is recognized as a marker for neuronal activity, and the expression level of c-fos is positively correlated with the degree of epileptic seizure) (Baraban et al., 2005) in the zebrafish brain was significantly higher in the PTZ-treated zebrafish with stx1b knockdown than in the PTZ-only model group. In contrast, STX1B overexpression decreased larval overspeed swimming behaviors under light stimuli and suppressed the c-fos expression in the zebrafish brain, as compared with in the PTZ group. Therefore, we suppose that the STX1B protein can alleviate PTZ-induced photosensitive seizures.
Despite there being no known reports of STX1B correlating with photosensitivity, several studies have implicated CHD2 in photosensitivity and have shown that CHD2 mutation is the first identified cause of the archetypal generalized photosensitive epilepsy syndrome, with CHD2 knockdown markedly increased in the case of zebrafish larval photosensitivity (Galizia et al., 2015). According to other reports, chd2 gene mutations were described in MAE (Carvill et al., 2013; Thomas et al., 2015); at the same time, stx1b gene variants or deletions can also be involved in the etiology of MAE (Vlaskamp et al., 2016). Since both stx1b and chd2 gene mutations can lead to MAE, whether or not STX1B is also related to photosensitivity epilepsy like CHD2 is, remains a question. MAE is an epilepsy characterized by the occurrence of myoclonic–atonic seizures, while myoclonic seizures are a typical symptom in the PTZ-induced seizure model (Frye and Muscatiello, 2001; Poplawska et al., 2015). In association with our results, these studies imply that STX1B may be associated with photosensitive epilepsy. However, the relationship between STX1B and photosensitive response still needs further clinical study.
Berberine was reported to have a protection effect on neurodegenerative and neuropsychiatric disorders with respect to its antioxidant and anti-inflammatory roles (Yoo et al., 2006, 2008; Sedaghat et al., 2017). Some studies have shown that BBR antagonized N-methyl-D-aspartate-induced excitotoxicity in gerbil hippocampal neurons (Yoo et al., 2008) and inhibited morphine-induced locomotor sensitization in mice (Yoo et al., 2006). Moreover, BBR attenuated a repeated nicotine-induced behavioral sensitization by decreasing postsynaptic neuronal activation in rats (Lee et al., 2007). These findings suggest that BBR probably is involved in the inhibition of neuron-locomotion overactivity, but published reports about the action of BBR in epilepsy remain scarce. In the present study, we found that BBR alleviated the overexcitation reaction and decreased the level of c-fos induced by PTZ, yet rescued the level of stx1b transcription suppressed by PTZ. When STX1B was downregulated, BBR’s therapeutic effect on a photosensitive seizure was significantly reduced or eliminated, suggesting that BBR’s inhibitory effect on a photosensitive seizure was dependent on the presence of STX1B protein. We speculate that BBR may indirectly activate some transcription factors to enhance the expression of the stx1b gene.
In summary, PTZ induces an epilepsy-like seizure, including photosensitive seizures in zebrafish, which may be partially mediated by STX1B deficiency. Adequate STX1B levels can slow down the hyperexcitation locomotion induced by PTZ in zebrafish. BBR can suppress PTZ-induced seizures in zebrafish by raising STX1B levels. Further research on the relationship between STX1B, BBR, and seizures may provide new clues for the development of novel antiepileptic drugs.
Author Contributions
J-PZ conceived and designed the project. Y-MZ and BC performed the experiments and treated the data. J-DJ provided substantial discussion for writing the manuscript. Y-MZ and J-PZ wrote the manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Ms. Jie Meng for fish husbandry.
Funding. This work was supported by the CAMS Major Collaborative Innovation Project (No. 2016-I2M-1-011) and the Foundation for Innovative Research Groups of the National Natural Science Foundation of China (No. 81621064). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fnmol.2018.00378/full#supplementary-material
References
- Baraban S. C., Taylor M. R., Castro P. A., Baier H. (2005). Pentylenetetrazole induced changes in zebrafish behavior, neural activity and c-fos expression. Neuroscience 131 759–768. 10.1016/j.neuroscience.2004.11.031 [DOI] [PubMed] [Google Scholar]
- Barbalho P. G., Carvalho B. S., Lopes-Cendes I., Maurer-Morelli C. V. (2016). Cyclooxygenase-1 as a potential therapeutic target for seizure suppression: evidences from zebrafish pentylenetetrazole-seizure model. Front. Neurol. 7:200. 10.3389/fneur.2016.00200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Besag F. M. (2018). Epilepsy in patients with autism: links, risks and treatment challenges. Neuropsychiatr. Dis. Treat. 14 1–10. 10.2147/NDT.S120509 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carvill G. L., Heavin S. B., Yendle S. C., McMahon J. M., O’Roak B. J., Cook J., et al. (2013). Targeted resequencing in epileptic encephalopathies identifies de novo mutations in CHD2 and SYNGAP1. Nat. Genet. 45 825–830. 10.1038/ng.2646 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi B. H., Ahn I. S., Kim Y. H., Park J. W., Lee S. Y., Hyun C. K., et al. (2006). Berberine reduces the expression of adipogenic enzymes and inflammatory molecules of 3T3-L1 adipocyte. Exp. Mol. Med. 38 599–605. 10.1038/emm.2006.71 [DOI] [PubMed] [Google Scholar]
- Cowan L. D. (2002). The epidemiology of the epilepsies in children. Ment. Retard. Dev. Disabil. Res. Rev. 8 171–181. 10.1002/mrdd.10035 [DOI] [PubMed] [Google Scholar]
- de Bittencourt P. R. (2004). Photosensitivity: the magnitude of the problem. Epilepsia 45(Suppl. 1), 30–34. 10.1111/j.0013-9580.2004.451010.x [DOI] [PubMed] [Google Scholar]
- Ellis L. D., Soanes K. H. (2012). A larval zebrafish model of bipolar disorder as a screening platform for neuro-therapeutics. Behav. Brain Res. 233 450–457. 10.1016/j.bbr.2012.05.043 [DOI] [PubMed] [Google Scholar]
- Epps S. A., Weinshenker D. (2013). Rhythm and blues: animal models of epilepsy and depression comorbidity. Biochem. Pharmacol. 85 135–146. 10.1016/j.bcp.2012.08.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferando I., Mody I. (2012). GABAA receptor modulation by neurosteroids in models of temporal lobe epilepsies. Epilepsia 53(Suppl. 9), 89–101. 10.1111/epi.12038 [DOI] [PubMed] [Google Scholar]
- Fisher R. S., Harding G., Erba G., Barkley G. L., Wilkins A. Epilepsy Foundation of America Working Group. (2005). Photic- and pattern-induced seizures: a review for the epilepsy foundation of america working group. Epilepsia 46 1426–1441. 10.1111/j.1528-1167.2005.31405.x [DOI] [PubMed] [Google Scholar]
- Frye C. A., Muscatiello N. A. (2001). 3alpha,5alpha-THP in the raphe magnus attenuates PTZ-induced myoclonic seizures. Brain Res. 911 146–151. 10.1016/S0006-8993(01)02560-4 [DOI] [PubMed] [Google Scholar]
- Fujiwara T., Kofuji T., Mishima T., Akagawa K. (2017). Syntaxin 1B contributes to regulation of the dopaminergic system through GABA transmission in the CNS. Eur. J. Neurosci. 46 2867–2874. 10.1111/ejn.13779 [DOI] [PubMed] [Google Scholar]
- Galizia E. C., Myers C. T., Leu C., de Kovel C. G., Afrikanova T., Cordero-Maldonado M. L., et al. (2015). CHD2 variants are a risk factor for photosensitivity in epilepsy. Brain 138(Pt 5), 1198–1207. 10.1093/brain/awv052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao F., Gao Y., Liu Y. F., Wang L., Li Y. J. (2014). Berberine exerts an anticonvulsant effect, and ameliorates memory impairment, and oxidative stress in a pilocarpine-induced epilepsy model in the rat. Neuropsychiatr. Dis. Treat. 10 2139–2145. 10.2147/NDT.S73210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grone B. P., Baraban S. C. (2015). Animal models in epilepsy research: legacies and new directions. Nat. Neurosci. 18 339–343. 10.1038/nn.3934 [DOI] [PubMed] [Google Scholar]
- Gupta P., Khobragade S. B., Shingatgeri V. M. (2014). Effect of various antiepileptic drugs in zebrafish PTZ-seizure model. Indian J. Pharm. Sci. 76 157–163. [PMC free article] [PubMed] [Google Scholar]
- Hussien H. M., Abd-Elmegied A., Ghareeb D. A., Hafez H. S., Ahmed H. E. A., El-Moneam N. A. (2018). Neuroprotective effect of berberine against environmental heavy metals-induced neurotoxicity and Alzheimer’s-like disease in rats. Food Chem. Toxicol. 111 432–444. 10.1016/j.fct.2017.11.025 [DOI] [PubMed] [Google Scholar]
- Kearney J. A. (2015). Synaptopathies heat up: mutations in stx1b in fever-associated epilepsies. Epilepsy curr. 15 138–139. 10.5698/1535-7597-15.3.138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim W. S., Lee Y. S., Cha S. H., Jeong H. W., Choe S. S., Lee M. R., et al. (2009). Berberine improves lipid dysregulation in obesity by controlling central and peripheral AMPK activity. Am. J. Physiol. Endocrinol. Metab. 296 E812–E819. 10.1152/ajpendo.90710.2008 [DOI] [PubMed] [Google Scholar]
- Kimmel C. B., Ballard W. W., Kimmel S. R., Ullmann B., Schilling T. F. (1995). Stages of embryonic development of the zebrafish. Dev. Dyn. 203 253–310. 10.1002/aja.1002030302 [DOI] [PubMed] [Google Scholar]
- Kong W., Wei J., Abidi P., Lin M., Inaba S., Li C., et al. (2004). Berberine is a novel cholesterol-lowering drug working through a unique mechanism distinct from statins. Nat. Med. 10 1344–1351. 10.1038/nm1135 [DOI] [PubMed] [Google Scholar]
- Kulkarni S. K., Dhir A. (2010). Berberine: a plant alkaloid with therapeutic potential for central nervous system disorders. Phytother. Res. 24 317–324. 10.1002/ptr.2968 [DOI] [PubMed] [Google Scholar]
- Lau C. W., Yao X. Q., Chen Z. Y., Ko W. H., Huang Y. (2001). Cardiovascular actions of berberine. Cardiovasc. Drug Rev. 19 234–244. 10.1111/j.1527-3466.2001.tb00068.x [DOI] [PubMed] [Google Scholar]
- Lee B., Yang C. H., Hahm D. H., Lee H. J., Choe E. S., Pyun K. H., et al. (2007). Coptidis Rhizoma attenuates repeated nicotine-induced behavioural sensitization in the rat. J. Pharm. Pharmacol. 59 1663–1669. 10.1211/jpp.59.12.0008 [DOI] [PubMed] [Google Scholar]
- Lorenz S., Taylor K. P., Gehrmann A., Becker T., Muhle H., Gresch M., et al. (2006). Association of BRD2 polymorphisms with photoparoxysmal response. Neurosci. Lett. 400 135–139. 10.1016/j.neulet.2006.02.026 [DOI] [PubMed] [Google Scholar]
- Lou T., Zhang Z., Xi Z., Liu K., Li L., Liu B., et al. (2011). Berberine inhibits inflammatory response and ameliorates insulin resistance in hepatocytes. Inflammation 34 659–667. 10.1007/s10753-010-9276-2 [DOI] [PubMed] [Google Scholar]
- Macdonald R. L., Barker J. L. (1977). Pentylenetetrazol and penicillin are selective antagonists of GABA-mediated post-synaptic inhibition in cultured mammalian neurones. Nature 267 720–721. 10.1038/267720a0 [DOI] [PubMed] [Google Scholar]
- Martins da Silva A., Leal B. (2017). Photosensitivity and epilepsy: current concepts and perspectives-A narrative review. Seizure 50 209–218. 10.1016/j.seizure.2017.04.001 [DOI] [PubMed] [Google Scholar]
- Mishima T., Fujiwara T., Sanada M., Kofuji T., Kanai-Azuma M., Akagawa K. (2014). Syntaxin 1B, but not syntaxin 1A, is necessary for the regulation of synaptic vesicle exocytosis and of the readily releasable pool at central synapses. PLoS One 9:e90004. 10.1371/journal.pone.0090004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mojarad T. B., Roghani M. (2014). The anticonvulsant and antioxidant effects of berberine in kainate-induced temporal lobe epilepsy in rats. Basic Clin. Neurosci. 5 124–130. [PMC free article] [PubMed] [Google Scholar]
- Poduri A., Lowenstein D. (2011). Epilepsy genetics–past, present, and future. Curr. Opin. Genet. Dev. 21 325–332. 10.1016/j.gde.2011.01.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poleon S., Szaflarski J. P. (2017). Photosensitivity in generalized epilepsies. Epilepsy Behav. 68 225–233. 10.1016/j.yebeh.2016.10.040 [DOI] [PubMed] [Google Scholar]
- Poplawska M., Wroblewska D., Borowicz K. K. (2015). Interactions between an antidepressant reboxetine and four classic antiepileptic drugs in the mouse model of myoclonic seizures. Pharmacol. Rep. 67 1141–1146. 10.1016/j.pharep.2015.04.016 [DOI] [PubMed] [Google Scholar]
- Rahn J. J., Bestman J. E., Josey B. J., Inks E. S., Stackley K. D., Rogers C. E., et al. (2014). Novel vitamin K analogs suppress seizures in zebrafish and mouse models of epilepsy. Neuroscience 259 142–154. 10.1016/j.neuroscience.2013.11.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schubert J., Siekierska A., Langlois M., May P., Huneau C., Becker F., et al. (2014). Mutations in STX1B, encoding a presynaptic protein, cause fever-associated epilepsy syndromes. Nat. Genet. 46 1327–1332. 10.1038/ng.3130 [DOI] [PubMed] [Google Scholar]
- Sedaghat R., Taab Y., Kiasalari Z., Afshin-Majd S., Baluchnejadmojarad T., Roghani M. (2017). Berberine ameliorates intrahippocampal kainate-induced status epilepticus and consequent epileptogenic process in the rat: underlying mechanisms. Biomed. Pharmacother. 87 200–208. 10.1016/j.biopha.2016.12.109 [DOI] [PubMed] [Google Scholar]
- Sillanpaa M., Besag F., Aldenkamp A., Caplan R., Dunn D. W., Gobbi G. (2016). Psychiatric and behavioural disorders in children with epilepsy (ILAE task force report): epidemiology of psychiatric/behavioural disorder in children with epilepsy. Epileptic Disord. 18 S2–S7. 10.1684/epd.2016.0810 [DOI] [PubMed] [Google Scholar]
- Soares D. C. S., Portela J. L. R., Roos D. H., Rodrigues N. R., Gomes K. K., Macedo G. E., et al. (2017). Treatment with pentylenetetrazole (PTZ) and 4-aminopyridine (4-AP) differently affects survival, locomotor activity, and biochemical markers in Drosophila melanogaster. Mol. Cell. Biochem. 442 129–142. 10.1007/s11010-017-3198-3 [DOI] [PubMed] [Google Scholar]
- Sollner T., Whiteheart S. W., Brunner M., Erdjument-Bromage H., Geromanos S., Tempst P., et al. (1993). SNAP receptors implicated in vesicle targeting and fusion. Nature 362 318–324. 10.1038/362318a0 [DOI] [PubMed] [Google Scholar]
- Stewart A. M., Desmond D., Kyzar E., Gaikwad S., Roth A., Riehl R., et al. (2012). Perspectives of zebrafish models of epilepsy: what, how and where next? Brain Res. Bull. 87 135–143. 10.1016/j.brainresbull.2011.11.020 [DOI] [PubMed] [Google Scholar]
- Sudhof T. C. (2013). Neurotransmitter release: the last millisecond in the life of a synaptic vesicle. Neuron 80 675–690. 10.1016/j.neuron.2013.10.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas R. H., Zhang L. M., Carvill G. L., Archer J. S., Heavin S. B., Mandelstam S. A., et al. (2015). CHD2 myoclonic encephalopathy is frequently associated with self-induced seizures. Neurology 84 951–958. 10.1212/WNL.0000000000001305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres-Hernandez B. A., Del Valle-Mojica L. M., Ortiz J. G. (2015). Valerenic acid and Valeriana officinalis extracts delay onset of pentylenetetrazole (PTZ)-induced seizures in adult Danio rerio (Zebrafish). BMC Complement. Altern. Med. 15:228. 10.1186/s12906-015-0731-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vlaskamp D. R., Rump P., Callenbach P. M., Vos Y. J., Sikkema-Raddatz B., van Ravenswaaij-Arts C. M., et al. (2016). Haploinsufficiency of the STX1B gene is associated with myoclonic astatic epilepsy. Eur. J. Paediatr. Neurol. 20 489–492. 10.1016/j.ejpn.2015.12.014 [DOI] [PubMed] [Google Scholar]
- Whitlock K. E., Westerfield M. (2000). The olfactory placodes of the zebrafish form by convergence of cellular fields at the edge of the neural plate. Development 127 3645–3653. [DOI] [PubMed] [Google Scholar]
- Wolf P., Goosses R. (1986). Relation of photosensitivity to epileptic syndromes. J. Neurol. Neurosurg. Psychiatry 49 1386–1391. 10.1136/jnnp.49.12.1386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoo K. Y., Hwang I. K., Kim J. D., Kang I. J., Park J., Yi J. S., et al. (2008). Antiinflammatory effect of the ethanol extract of Berberis koreana in a gerbil model of cerebral ischemia/reperfusion. Phytother. Res. 22 1527–1532. 10.1002/ptr.2527 [DOI] [PubMed] [Google Scholar]
- Yoo K. Y., Hwang I. K., Lim B. O., Kang T. C., Kim D. W., Kim S. M., et al. (2006). Berberry extract reduces neuronal damage and N-Methyl-D-aspartate receptor 1 immunoreactivity in the gerbil hippocampus after transient forebrain ischemia. Biol. Pharm. Bull. 29 623–628. 10.1248/bpb.29.623 [DOI] [PubMed] [Google Scholar]
- Zeng X. H., Zeng X. J., Li Y. Y. (2003). Efficacy and safety of berberine for congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am. J. Cardiol. 92 173–176. 10.1016/S0002-9149(03)00533-2 [DOI] [PubMed] [Google Scholar]
- Zhang H., Kong W. J., Shan Y. Q., Song D. Q., Li Y., Wang Y. M., et al. (2010). Protein kinase D activation stimulates the transcription of the insulin receptor gene. Mol. Cell. Endocrinol. 330 25–32. 10.1016/j.mce.2010.07.022 [DOI] [PubMed] [Google Scholar]
- Zhou P., Pang Z. P., Yang X., Zhang Y., Rosenmund C., Bacaj T., et al. (2013). Syntaxin-1 N-peptide and Habc-domain perform distinct essential functions in synaptic vesicle fusion. EMBO J. 32 159–171. 10.1038/emboj.2012.307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu F., Qian C. (2006). Berberine chloride can ameliorate the spatial memory impairment and increase the expression of interleukin-1beta and inducible nitric oxide synthase in the rat model of Alzheimer’s disease. BMC Neurosci. 7:78. 10.1186/1471-2202-7-78 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.