Abstract
Associations observed of Helicobacter pylori infection with haemoglobin levels are inconsistent. We examined associations of H. pylori sero-prevalence and serum pepsinogens (PGs), as non-invasive markers of atrophic gastritis, with haemoglobin levels. A cross-sectional study was undertaken among 654 Jewish and 937 Arab residents of Jerusalem, aged 25–78 years, randomly selected from Israel’s national population registry in age-sex and population strata. Sera were tested for H. pylori IgG, cytotoxin–associated gene A (CagA) antigen IgG antibody and PGs levels. Multivariable models were fitted to account for confounders. Participants with atrophic gastritis (PGI < 30 μg/L or a PGI: PGII < 3.0) had lower haemoglobin levels than those without: beta-coefficient −0.34 (95% CI −0.59, −0.09); in men −0.27 (95% CI −0.67, 0.12), and in women −0.43 (95% CI −0.74, −0.12). Lower haemoglobin levels were noted in persons with CagA antibody than in those H. pylori sero-negative or H. pylori-CagA sero-negative: beta-coefficient −0.14 (95% CI −0.29, 0.01). Anaemia was more common among women and men with than without atrophic gastritis: adjusted OR 2.58 (95% CI 1.48, 4.48) and 1.52 (95% CI 0.59, 3.95), respectively. In conclusion, independent of known correlates, atrophic gastritis and apparently CagA sero-positivity, a marker of H. pylori virulent strains, are associated with lower haemoglobin levels.
Introduction
Anaemia is an important public health problem, which usually results from a depletion of body iron stores. Prevalences of anaemia and iron deficiency anaemia (IDA) are increased by Helicobacter pylori infection (reviewed by Muhsen and Cohen1). H. pylori, a gram negative bacterium that colonizes the gastric mucosa and causes chronic gastritis2, is the main cause of gastric and duodenal ulcers, and an established risk factor for gastric cancer and MALT lymphoma2–5. These conditions usually develop in adulthood, although only in a subset of infected individuals2. In a recent meta-analysis we showed an increased likelihood of IDA in H. pylori infected persons vs uninfected ones: pooled odds ratio (OR) 1.72 (95% confidence interval (CI) 1.23–2.42)6. The association between H. pylori infection and all-cause anaemia was weaker: pooled OR 1.15 (95% CI 1.00–1.32)6. Currently, H. pylori eradication therapy is recommended in cases of refractory or unexplained IDA7.
Mechanisms that may explain associations of H. pylori infection with anaemia and IDA have not been fully elucidated, but pathogen-related factors might play a role. Systems of H. pylori iron regulation constantly express iron uptake, in contrast to systems of other bacteria8. H. pylori isolates from patients with IDA display a greater capability of iron uptake and of proliferation in the presence of iron compared to H. pylori isolates from non-IDA patients9. In addition, the expression of iron-repressible outer membrane proteins involved in iron acquisition differs between these groups10. Likely, some of these mechanisms enable survival of the bacteria in the hostile niche of the stomach, and also affect the host iron homeostasis. Evidence regarding the contribution of the cytotoxin-associated gene A (CagA) protein in the development of IDA remains inconclusive10–15, although this antigen was shown to be important in the pathogenesis of peptic disease and gastric cancer2,5,16. H. pylori chronic gastritis changes the physiology in the stomach17, including alterations in gastric acidity17 and ascorbic acid levels18–20, which are important in the absorption of dietary iron21,22.
Clearly, if associations of H. pylori infection with haemoglobin and other iron biomarkers is mediated by gastric inflammation, positive associations between H. pylori-related gastric pathology and anaemia are anticipated. However, the association of atrophic gastritis, a severe form of H. pylori-related gastric pathology, with haemoglobin, especially in the general population, was rarely addressed. H. pylori infection can cause atrophic body gastritis, which can lead to deficiencies in vitamin B12 and intrinsic factor, as well as hypochlorhydria; these negatively affect iron absorption23,24. Atrophic gastritis can be assessed using serum pepsinogen (PG) I and PGII, proenzymes of the digestive enzyme pepsin, which are secreted to the gastric lumen but which can also be detected in the serum25–27. With increasing severity of H. pylori gastritis, serum PGI and PGII levels are increased, but when atrophic changes occur in the corpus, PGI and the ratio of PGI: PGII decrease. More severe atrophy is related to a lower PGI: PGII ratio25,28.
The aims of the current study were to examine associations of H. pylori immunoglobulin G (IgG) sero-prevalence, CagA IgG sero-positivity and serum PGs, as non-invasive markers of atrophic gastritis, with two outcome variables: haemoglobin levels (continuous variable) and anaemia, in a population-based sample of adult men and women.
Methods
Study design and population
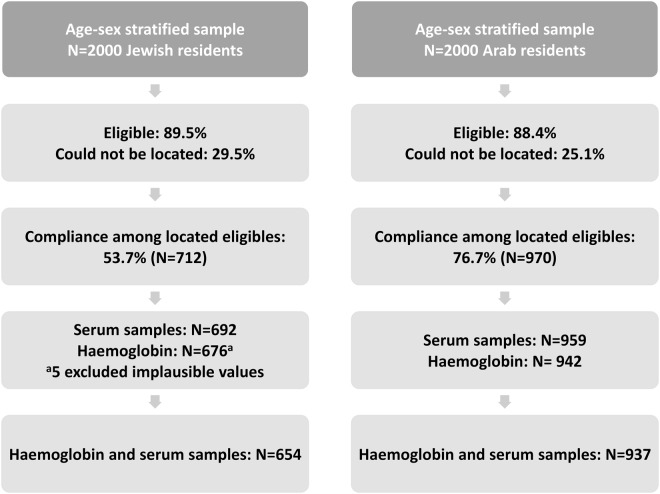
We used archived anonymized (coded) serum samples obtained in the framework of a cross-sectional study of Jewish and Arab residents of Jerusalem. Details of the study design have been reported29,30. Briefly, age-sex-stratified random samples of 2000 Arab permanent residents of East Jerusalem and 2000 Israeli Jewish residents of Jerusalem, aged 25–74 years at sampling, were drawn from the Israel national population registry. Individuals were ineligible if they were institutionalized, housebound, had a severe illness, were unable to provide informed consent, pregnant or had given birth within three months preceding study initiation. The response rates among those located were 76.7% for Arabs (N = 970) and 53.7% for Jews (N = 712)29,30. For the current study, data were available for 937 (96.6%) and 654 (91.9%) Arab and Jewish participants, respectively (Fig. 1). Data were collected by personal interviews. Information was obtained on age at enrolment (grouped as 25–44, 45–64, and 65–78 years), sex and education (classified as having an academic degree/education, high-school/some college, some high school or less). Regular smoking was defined as reported smoking of at least one cigarette daily. Weight and height were measured. Body mass index (BMI) was calculated using measured weight and height as: weight (in kilograms [kg]) divided by height2 (in meters [m]); obesity was defined as BMI ≥30 kg/m2. Blood samples were collected after a 12 hr fast, and haemoglobin levels were measured by auto-analyser. Haemoglobin levels lower than 12 g/dL in women and lower than 13 g/dL in men were employed to define anaemia. Anaemia was classified as microcytic, normocytic and macrocytic if values of mean corpuscular volume (MCV) were <80 fL, 80–100 fL and >100 fL, respectively.
Figure 1.
Sampling and enrolment.
Laboratory methods
Sera were tested for the presence of specific H. pylori IgG antibodies using enzyme-linked immunosorbent assay (ELISA) (Enzygnost® Anti-Helicobacter pylori II/IgG kit, Siemens Diagnostics Product GmbH, Marburg, Germany). Optical density values >0.250 were classified as H. pylori sero-positive following the manufacturer’s instructions. Sensitivity and specificity values of the kit are within the range of 94–98%. The detection of H. pylori IgG serum antibody using this kit was significantly correlated with the detection of H. pylori antigen in stool samples that used monoclonal antigen detection enzyme immunoassay (Spearman’s coefficient 0.70, P < 0.001) (Muhsen et al., unpublished). The presence of IgG antibody against recombinant CagA protein31 was measured in H. pylori-positive sera, employing a modified in-house ELISA protocol, as previously described31. The detection of CagA IgG serum antibody with this recombinant CagA protein has demonstrated high sensitivity (>90%) in identifying CagA strains32. Participants were classified as: a) H. pylori seronegative; b) H. pylori positive, CagA negative, if they had H. pylori IgG antibodies, but lacked CagA IgG antibodies; or c) H. pylori positive, CagA positive if they were positive for H. pylori and CagA IgG antibodies.
Concentrations of serum PGI and PGII were quantified by ELISA (Biohit Inc., Helsinki, Finland). Atrophic gastritis was defined as serum PGI levels of <30 μg/L or a PGI: PGII ratio of <3.0, as recommended by the manufacturer. Higher serum PGI and PGII levels are found in H. pylori infected vs uninfected individuals, while a lower PGI: PGII ratio is found in the former28. In our sample, H. pylori sero-status was significantly correlated with PGI level (Spearman’s coefficient 0.19, P < 0.001), PGII level (Spearman’s coefficient 0.33, P < 0.001) and PGI: PGII ratio (Spearman’s coefficient −0.23, P < 0.001). These correlations strengthened the validity of the classification of H. pylori sero-status.
Statistical analysis
Student’s t tests and one-way analysis of variance (ANOVA) were used to examine unadjusted differences in mean haemoglobin levels according to sociodemographic variables, H. pylori sero-status, atrophic gastritis and smoking. When more than two strata of a variable were compared, we used a Tukey test to account for multiple comparisons. Multivariable linear regression models with haemoglobin level as the dependent variable were fitted; from these models, we obtained beta (slope) coefficients (and 95% CIs). Categorical independent variables were included in the model as dummy variables. Chi square tests were employed to examine unadjusted associations of sociodemographic variables, H. pylori-CagA IgG antibody sero-status and atrophic gastritis, with anaemia. Multivariable logistic regression models were fitted, from which we obtained adjusted ORs and 95% CIs. Variables associated with the dependent variable (haemoglobin in linear regression and anaemia in logistic regression) in bivariate analysis with P < 0.2 were included in the multivariable analysis, in addition to age, atrophic gastritis (as measured by serum PGs) and H. pylori infection. As haemoglobin levels differ between men and women, the analyses were conducted in sex-specific strata. Interactions between population group, age, sex, H. pylori sero-status and atrophic gastritis were assessed. Data were analysed using IBM SPSS (Armonk, New York, USA) version 23 and Winpepi33.
Ethics statement
The study was approved by the Institutional Review Board of the Hadassah Medical Centre, Jerusalem, and by the ethics committee at Tel Aviv University. All participants signed an informed consent. The study was conducted in accordance with the Declaration of Helsinki ethical principles and regulation of the Ministry of Health.
Results
Information on haemoglobin and H. pylori sero-status was available for 937 and 654 Arab and Jewish participants, respectively; of these 498 (53.1%) and 348 (53.2%) were men (P = 0.9), respectively. The mean ages were 52.0 (SD 13.9) and 52.4 (13.6) years (P = 0.5) for Jews and Arabs, respectively. The mean haemoglobin level was higher among men than among women: 15.0 g/dL (SD 1.5) vs. 12.9 g/dL (SD 1.3), P < 0.001. Anaemia was evident in 206 participants: 12.9% (95% CI 11.4–14.7%). Among these, 64 (31.0%) had microcytic anaemia, 139 (67.5%) had normal MCV values and three (1.5%) had macrocytic anaemia. Anaemia was more prevalent among women (20.3% [95% CI 17.5%, 23.3%]) than men (6.5% [95% CI 5.0%, 8.3%]), P < 0.001.
Haemoglobin levels by demographic variables, H. pylori IgG sero-positivity and atrophic gastritis in men
The mean haemoglobin level decreased with age (P < 0.001). Differences between the age groups in mean haemoglobin level remained statistically significant after correction for multiple comparisons (by Tukey). A higher mean haemoglobin level was found in smokers than in non-smokers (P < 0.001). No significant difference was found in mean haemoglobin level according to education and according to H. pylori sero-status. Men with evidence of atrophic gastritis had a lower mean haemoglobin level than those without (P = 0.037) (Table 1).
Table 1.
Unadjusted mean haemoglobin levels according to demographic and behavioural variables, H. pylori sero-prevalence and atrophic gastritis by sex*.
| N | Men | P | N | Women | P | |
|---|---|---|---|---|---|---|
| Mean haemoglobin (SD) | Mean haemoglobin (SD) | |||||
| Population | 0.11 | <0.001 | ||||
| Jews | 348 | 15.1 (1.5) | 306 | 13.3 (1.2) | ||
| Arabs | 498 | 15.0 (1.4) | 439 | 12.7 (1.3) | ||
| Age (years) | <0.001† | 0.005† | ||||
| 25–44 | 297 | 15.4 (1.4) | 234 | 12.7 (1.2) | ||
| 45–64 | 358 | 15.1 (1.3) | 337 | 13.0 (1.3) | ||
| 65–78 | 191 | 14.4 (1.5) | 174 | 13.0 (1.4) | ||
| Education | 0.2† | 0.013† | ||||
| Some high school or less | 430 | 15.1 (1.5) | 418 | 12.8 (1.3) | ||
| High school certificate/some college | 200 | 14.9 (1.5) | 166 | 12.9 (1.3) | ||
| Academic education | 211 | 15.1 (1.4) | 160 | 13.2 (1.2) | ||
| Smoking | <0.001 | 0.03 | ||||
| Regular smoking ≥1 cigarette/ day | 290 | 15.4 (1.3) | 75 | 13.2 (1.4) | ||
| Other | 550 | 14.9 (1.5) | 663 | 12.9 (1.3) | ||
| Obesity | 0.9 | 0.3 | ||||
| BMI <30 kg/m2 | 615 | 15.0 (1.5) | 404 | 13.0 (1.3) | ||
| BMI ≥30 kg/m2 | 228 | 15.0 (1.4) | 341 | 12.9 (1.2) | ||
| H. pylori IgG sero-status | 0.6† | 0.012† | ||||
| Negative | 217 | 15.0 (1.6) | 189 | 12.9 (1.3) | ||
| H. pylori positive CagA negative | 388 | 15.1 (1.5) | 333 | 13.0 (1.2) | ||
| H. pylori positive CagA positive | 241 | 15.0 (1.3) | 223 | 12.7 (1.4) | ||
| Atrophic gastritis: PGI <30 μg/L or PGI: PGII <3.0 | 0.037 | 0.052 | ||||
| No | 790 | 15.0 (1.4) | 671 | 13.0 (1.2) | ||
| yes | 52 | 14.6 (1.7) | 71 | 12.5 (1.7) |
*BMI: body mass index, CagA: cytotoxin associated gene A; kg: kilogram, m: meters, PG: pepsinogen, SD: standard deviation.
†P value for the difference between the groups by one-way analysis of variance (ANOVA).
Pair comparisons by Tukey HSD Post-hoc Test – Men:
Age group: 25–44 vs 45–64 years P = 0.03; age group 24–44 vs 65–78 years P < 0.001; age group 45–64 vs 65–78 years P < 0.001.
Education: Some high school or less vs high school certificate/some college P = 0.37; some high school or less vs academic education P = 0.86, high school certificate/some college vs academic education P = 0.27. H. pylori sero-status: Negative vs H. pylori positive CagA negative P = 0.7; negative vs H. pylori positive CagA positive P = 0.9; H. pylori positive CagA negative vs H. pylori positive CagA positive P = 0.6.
Pair comparisons by Tukey HSD Post-hoc Test – Women:
Age group: 25–44 vs 45–64 years P = 0.004; age group 24–44 vs 65–78 years P = 0.026; age group 45–64 vs 65–78 years P = 0.9.
Education: Some high school or less vs high school certificate/some college P = 0.6; some high school or less vs academic education P = 0.011, some high school or less vs academic education P = 0.2. H. pylori sero-status: Negative vs H. pylori positive CagA negative P = 0.5; negative vs H. pylori positive CagA positive P = 0.2; H. pylori positive CagA negative vs H. pylori positive CagA positive P = 0.009.
On multivariable analysis, men with evidence of atrophic gastritis had non-significantly lower haemoglobin levels: beta coefficient −0.27 (95% CI −0.67, 0.12), P = 0.17 (Table 2). No statistically significant difference was found in haemoglobin levels according to CagA IgG sero-status. The associations of age and smoking with haemoglobin level persisted in this model, which also showed lower haemoglobin levels in Arab than Jewish men (Table 2). No interactions were detected between population group and atrophic gastritis (P = 0.7), population group and CagA IgG antibody sero-status (P = 0.9), and CagA IgG antibody sero-status and atrophic gastritis (P = 0.7).
Table 2.
Multiple linear regression analysis of haemoglobin levels according to demographic and behavioural variables, H. pylori sero-prevalence and serological evidence of atrophic gastritis*.
| Variable | Pooled sexes** | P | Men** | P | Women** | P |
|---|---|---|---|---|---|---|
| Beta coefficient (95% CI) | Beta coefficient (95% CI) | Beta coefficient (95% CI) | ||||
| Sex (Males vs females) | 1.98 (1.84, 2.12) | <0.001 | — | — | ||
| Age 24–44 years | Reference | Reference | Reference | |||
| Age 45–64 years | 0.02 (−0.13, 0.18) | 0.7 | −0.23 (−0.45, −0.01) | 0.04 | 0.34 (0.13, 0.56) | 0.002 |
| Age 65–78 years | −0.29 (−0.48, −0.10) | 0.002 | −0.85 (−1.12, −0.59) | <0.001 | 0.37 (0.11, 0.63) | 0.006 |
| Population group (Arabs vs Jews) | −0.36 (−0.59, −0.09) | <0.001 | −0.20 (−0.40, 0.00) | 0.05 | −0.48 (−0.69, −0.27) | <0.001 |
| Regular smoking ≥1 cigarette/ day (reference: other) | 0.48 (0.31, 0.65) | <0.001 | 0.45 (0.24, 0.66) | <0.001 | 0.29 (−0.005, 0.60) | 0.054 |
| Atrophic gastritis (PGI <30 µg/L or PGI: PGII ratio <3) (yes vs no) | −0.34 (−0.59, −0.09) | 0.009 | −0.27 (−0.67, 0.12) | 0.17 | −0.43 (−0.74, −0.12) | 0.007 |
| H. pylori positive CagA positive (yes vs no) | −0.14 (−0.29, 0.01) | 0.069 | −0.09 (−0.29, 0.13) | 0.4 | −0.15 (−0.35, 0.06) | 0.15 |
| Education: Some high school or less | −0.05 (−0.29, 0.13) | 0.5 | −0.02 (−0.26, 0.23) | 0.8 | −0.11 (−0.37, 0.15) | 0.4 |
| High school certificate/some college | −0.16 (−0.36, 0.04) | 0.12 | −0.23 (−0.51, 0.04) | 0.099 | −0.04 (−0.32, 0.25) | 0.8 |
| Academic education | Reference | Reference | Reference |
*CagA: cytotoxin associated gene A; CI: confidence intervals; PG: pepsinogen.
**Adjusted for the variables in the table. R Square 0.4 for the pooled model, 0.09 for men and 0.08 for women.
Haemoglobin levels by demographic variables, H. pylori IgG sero-positivity and atrophic gastritis in women
Lower mean haemoglobin levels were found among Arab than Jewish women. A gradient was observed in relation to education (Table 1); namely, women with some high school education or less had significantly lower haemoglobin levels than did those with an academic degree (P = 0.011 by Tukey test). The average haemoglobin level was lower in women aged 25–44 years than in women aged 45–64 years (P = 0.004 by Tukey test), and than in women aged 65–78 years (P = 0.026 by Tukey test); and it was higher in smokers than in non-smokers (P = 0.03). Women with H. pylori CagA serum IgG antibody had a lower mean haemoglobin level than H. pylori sero-positive women who were lacking serum CagA IgG antibody (P = 0.009 by Tukey test), but the level was similar to that of H. pylori negative women. Women with serological evidence of atrophic gastritis had a lower mean haemoglobin level than women without (P = 0.052) (Table 1). The differences in haemoglobin levels according to atrophic gastritis persisted in a multivariable analysis (P = 0.007); the association with CagA IgG sero-positivity did not (P = 0.15) (Table 2). No significant interaction was found between population group and atrophic gastritis (P = 0.2), population group and CagA IgG antibody sero-status (P = 0.6), and CagA sero-status and atrophic gastritis (P = 0.10).
In a pooled multivariable analysis of both sexes, men had a higher mean haemoglobin level than women. Participants with atrophic gastritis had significantly lower haemoglobin levels than those without (P = 0.009). CagA IgG sero-positivity was related to a lower mean haemoglobin level (P = 0.069) (Table 2). No significant interactions were found between sex and atrophic gastritis (P = 0.8) or by CagA IgG sero-positivity (P = 0.3).
Variables associated with anaemia
Among men, a non-significantly higher prevalence of anaemia was found among those with evidence of atrophic gastritis than those without (P = 0.13). Age was positively related to anaemia prevalence, while a lower prevalence of anaemia was found in smokers than non-smokers (P < 0.001). No difference was noted in the prevalence of anaemia according to H. pylori IgG sero-prevalence (Table 3). The results were similar in a multivariable logistic regression model that adjusted for age and H. pylori sero-positivity (Table 4). Among women, the prevalence of anaemia was higher among those who had atrophic gastritis than those without (P = 0.001) (Table 3). Anaemia prevalence was higher among women with CagA IgG serum antibody than among those who were H. pylori sero-negative, and those who lacked CagA IgG antibodies. The prevalence of anaemia was higher among Arab than Jewish women. These associations persisted in a multivariable analysis that included age and H. pylori CagA sero-positivity in addition to population group, education and smoking (Table 4).
Table 3.
Associations of sociodemographic and behavioural variables, H. pylori sero-prevalence and serological evidence of atrophic gastritis with anaemia by sex.
| Men | P* | Women | P* | |
|---|---|---|---|---|
| Anaemia/total (%) | Anaemia/total (%) | |||
| Population group | 0.11 | <0.001 | ||
| Jews | 17/348 (4.9) | 36/306 (11.8) | ||
| Arabs | 38/498 (7.6) | 115/439 (26.2) | ||
| Age, years | <0.001 | 0.062 | ||
| 25–44 | 10/297 (3.4) | 59/234 (25.2) | ||
| 45–64 | 16/358 (4.6) | 58/337 (17.2) | ||
| 65–78 | 29/191 (15.2) | 34/174 (19.5) | ||
| Education | 0.17 | 0.021 | ||
| Some high school or less | 22/430 (5.1) | 98/418 (23.4) | ||
| High school certificate/some college | 18/200 (9.0) | 32/166 (19.3) | ||
| Academic education | 13/211 (6.2) | 21/160 (13.1) | ||
| Smoking | <0.001 | 0.4 | ||
| Regular smoking ≥1 cigarette/ day | 6/291 (2.1) | 13/75 (17.3) | ||
| Other | 49/550 (8.9) | 137/663 (20.7) | ||
| Obesity | 0.9 | 0.8 | ||
| BMI <30 kg/m2 | 40/615 (6.5) | 81/404 (20.0) | ||
| BMI ≥30 kg/m2 | 15/228 (6.6) | 70/341 (20.5) | ||
| H. pylori IgG sero-status | 0.6 | 0.037 | ||
| Negative | 17/216 (7.8) | 35/189 (18.5) | ||
| H. pylori positive CagA negative | 24/338 (6.2) | 58/333 (17.4) | ||
| H. pylori positive CagA positive | 14/241(5.8) | 58/223 (26.0) | ||
| Atrophic gastritis (PGI <30 μg/L, and/or a PGI: PGII <3.0) | 0.13 | 0.001 | ||
| No | 49/789 (6.2) | 125/671 (18.6) | ||
| yes | 6/52 (11.5) | 25/71 (35.2) |
*P values were obtained by chi square test.
BMI: body mass index, CagA: cytotoxin associated gene A; kg: kilogram, m: meters, PG: pepsinogen, SD: standard deviation.
Table 4.
Multivariable logistic modelling of determinants of anaemia, stratified by sex*.
| Adjusted OR (95% CI)* | P | |
|---|---|---|
| Men** | ||
| Age, years | <0.001 (2df) | |
| 25–44 | Reference | |
| 45–64 | 1.21 (0.53, 2.81) | 0.6 |
| 65–78 | 4.63 (2.12, 10.09) | <0.001 |
| Population group: Arabs vs Jews | 2.17 (1.13, 4.16) | 0.019 |
| Atrophic gastritis (PGI <30 µg/L or PGI: PGII ratio <3) (yes vs no) | 1.52 (0.59, 3.95) | 0.3 |
| H. pylori IgG positive CagA IgG positive vs the rest | 0.89 (0.46, 1.72) | 0.7 |
| Regular smoking ≥1 cigarette/ day vs other | 0.20 (0.08, 0.53) | 0.001 |
| Education: | 0.11 (2df) | |
| Some high school or less | 0.67 (0.31,1.45) | 0.3 |
| High school certificate/some college | 1.38 (0.63–3.04) | 0.4 |
| Academic education | Reference | |
| Women** | ||
| Age, years | 0.021 (2df) | |
| 25–44 | Reference | |
| 45–64 | 0.55 (0.36, 0.85) | 0.007 |
| 65–78 | 0.60 (0.36, 1.01) | 0.056 |
| Population group: Arabs vs Jews | 2.15 (1.36, 3.38) | 0.034 |
| Atrophic gastritis (PGI <30 µg/L or PGI: PGII ratio <3) (yes vs no) | 2.58 (1.48, 4.48) | 0.001 |
| H. pylori IgG positive CagA IgG positive vs the rest | 1.40 (0.94, 2.07) | 0.09 |
| Regular smoking ≥1 cigarette/ day vs other | 0.89 (0.47, 1.69) | 0.7 |
| Education: | 0.5 (2df) | |
| Some high school or less | 1.31 (0.74, 2.34) | 0.3 |
| High school certificate/some college | 1.06 (0.56, 1.99) | 0.8 |
| Academic education | Reference | |
*CagA: cytotoxin associated gene A; CI: confidence intervals; df: degrees of freedom; OR: odd ratio; PG: pepsinogen.
Adjusted for the variables included in the table.
**For men Nagelkerke R Square = 0.157, P = 0.9 by Hosmer and Lemeshow Test.
**For women Nagelkerke R Square = 0.091, P = 0.48 by Hosmer and Lemeshow Test.
In a secondary analysis, we re-grouped the study participants according to values of the PGI: PGII ratio that correlated with gastritis severity, using the OLGA system34: most severe <3.0, moderate 3.0–6.8 and least severe >6.8. With these cut-off values, the mean haemoglobin levels were 14.6 (SD 1.6), 15.0 (SD 1.4) and 15.0 (SD 1.4), respectively, in men (P = 0.2 by ANOVA). In women, the corresponding values were 12.6 (SD 1.6), 12.7 (SD 1.3) and 13.0 (SD 1.2) (P = 0.029 by ANOVA). In a multivariable analysis, the significant gradient between PGI: PGII and haemoglobin level was maintained in women (Supplementary Table S1).
Anaemia sub-type according to H. pylori sero-status and atrophic gastritis
Overall, the prevalence of microcytic anaemia was 5.2%, 3.1% and 4.5% in persons who were H. pylori sero-negative, H. pylori sero-positive but lacking CagA IgG antibody, and H. pylori sero-positive CagA positive, respectively. The respective prevalences for normocytic anaemia were 7.6%, 8.3% and 10.3%. The prevalence of microcytic anaemia was 9.8% in persons with atrophic gastritis vs 3.6% in those without this condition. The respective prevalence for normocytic anaemia was 8.1% vs 15.4%. All three persons with macrocytic anaemia were positive for H. pylori CagA IgG but without atrophic gastritis (Supplementary Table S2).
Discussion
We examined associations of H. pylori IgG sero-prevalence, CagA IgG sero-positivity and serum PGs, as non-invasive markers of atrophic gastritis, with haemoglobin levels and anaemia, in men and women of two ethnic groups in a general population.
Serologic evidence of atrophic gastritis was associated with a higher prevalence of anaemia, particularly in women, and with lower mean haemoglobin levels (mean difference 0.34 g/dL). A similar trend, although of smaller magnitude, was found in relation to CagA IgG antibody sero-positivity (difference of 0.14 g/dL in mean haemoglobin level). These differences were more evident in women, although no significant interaction was found between sex and atrophic gastritis or between sex and CagA IgG sero-positivity. Notably, these observations were independent of age, population group, education and smoking history. A limited number of studies have addressed associations of H. pylori infection with anaemia or haemoglobin level among adults in the general population35–40. These mostly showed no significant difference between infected and uninfected individuals in mean haemoglobin levels or in the prevalence of anaemia, except for studies carried out in pregnant women39,40. None of these studies has addressed the role of CagA infection or atrophic gastritis. Our findings add a new dimension, suggesting that severe gastric inflammation, even with atrophic gastritis (as evident by serum PGs levels), rather than exposure to H. pylori per se, are involved in decreased haemoglobin levels. Thus, our results improve the risk profiling of low haemoglobin levels in relation to H. pylori infection. In dyspeptic adult patients, lower mean haemoglobin levels and a higher prevalence of anaemia were documented in H. pylori infected compared to uninfected patients41. A case-control study from the United Kingdom42 showed that adult patients referred for investigation of IDA had significantly more frequent gastric body atrophy, as demonstrated by gastric biopsy, compared to control patients with normal haemoglobin and iron levels. A gradient was observed with increased atrophy grades, whereas H. pylori infection as a main effect was not associated with IDA42. Anaemic patients with gastric body atrophy were less likely to have conditions that might be the definite cause of anaemia than were anaemic patients without atrophy. This suggests that gastric body atrophy might be a cause of anaemia in some individuals and a contributory factor in others42. Nahon el al.43 showed that patients with unexplained IDA referred for gastric tract evaluation had a higher prevalence of chronic gastritis than control patients (67% vs 47%), and than patients with atrophic gastritis (15% vs 6%) and H. pylori infection (60% vs 43%)43. Similar to our observations, Lee and colleagues44 showed in a general population sample of 2398 participants, lower haemoglobin levels and higher anaemia prevalence among persons with serological evidence of gastric atrophy (PGI: PGII ratio < 3.0, either with or without H. pylori serum antibodies) compared to those without gastric atrophy44. Collectively, these and our findings confirm the hypothesis that the association of H. pylori infection with lower haemoglobin might be mediated by H. pylori-associated inflammatory and atrophic changes affecting the gastric mucosa. Anaemia is an alarm symptom of gastric cancer that prompts invasive diagnostic techniques. Atrophic gastritis is mostly caused by H. pylori infection, and is a main precursor of gastric cancer45. A positive association between anaemia and serologic evidence of atrophic gastritis suggests that anaemia might be a marker of pre-malignant lesions in the stomach. This observation may promote early detection of gastric cancer.
Atrophic lesions in the stomach affect gastric acidity17 and ascorbic acid levels18–20, which are essential to the absorption of dietary iron21,22. Body gastric atrophy is also associated with decreases in secretion of intrinsic factor and in absorption of dietary vitamin B12, which may result in pernicious anaemia23,24. CagA is associated with an inflammatory response in the stomach, but also has the potential to induce carcinogenesis irrespective of inflammation (reviewed in31). We found a clear trend of higher prevalence of microcytic anaemia in persons with serological evidence of atrophic gastritis compared to persons without such condition, and all three persons with macrocytic anaemia tested positive for CagA IgG serum antibody.
Consistent with previous reports46–48, women aged 25–44 years had lower haemoglobin levels than did older women, thus conferring a risk for anaemia among women of child-bearing age; this is likely due to menstrual blood loss and enhanced demands related to pregnancy. In men, however, haemoglobin levels decreased with advanced age. A longitudinal study from Japan demonstrated that haemoglobin levels declined as age increased in men, while in women a decline was observed after the age of 60 years49. Age-related patterns in haemoglobin levels likely reflect the ageing process, but might also be related to a birth cohort effect, or both, as has been shown49.
Our results also confirmed previous observations of higher haemoglobin levels among smokers than non-smokers46,47,49,50. This finding apparently results from compensatory erythropoiesis stimulated by smoking-induced hypoxia.
Lower haemoglobin levels were found among Arab than Jewish patients, and Arabs tended to be more anaemic. These differences might reflect differences in socioeconomic status and dietary habits. Genetic differences may also contribute to the Arab-Jewish gap in anaemia prevalence. Consanguineous marriages are far more common in the Arab population in Israel51, as are haemoglobinopathies52.
Our study has limitations. The cross-sectional design limits causal inference regarding associations of H. pylori infection and atrophic gastritis with anaemia. Nonetheless, H. pylori infection is typically acquired in early childhood and, without treatment, persists for life. Hence, the H. pylori infection likely preceded the occurrence of anaemia, which presumably developed in adulthood. H. pylori is a main cause of atrophic gastritis, and when the latter ensues, the bacterium usually loses its niche and disappears. Hence, it is likely that some persons who were classified as H. pylori sero-negatives were actually previously infected with the bacterium. Such a scenario would likely result in underestimation of associations of H. pylori infection with haemoglobin levels and anaemia. We cannot determine whether atrophic gastritis resulted from H. pylori infection or autoimmunity. Addressing such a question is especially challenging given the overlap between these two conditions53 and the evidence showing that H. pylori might play a role in gastric autoimmunity via molecular mimicry23,54.
Our use of serum PGs as a surrogate marker to define atrophic gastritis might have limited sensitivity. However, we examined in the serum, both PGI concentration and PGI: PGII ratio; a combination of these parameters has been shown to improve the accuracy of detection. A PGI level of 25–30 µg/L or less and PGI: PGII < 3.0 are commonly used cut-off values55,56 (reviewed by Zagari et al.26) when using Biohit ELISAs, with sensitivity ranging between 71% and 90%, and high specificity of 90–98% compared to gastric biopsy56–58; this usually detects moderate to severe forms of atrophic gastritis.
Our study has a number of strengths. First, it comprises a large sample size of men and women from two general population samples. Second, persons with conditions that might induce anaemia, such as cancer, severe kidney disease, pregnancy and recent birth were excluded from the study. Third, we were able to adjust for potential confounders, which were not available in many of the previous studies that assessed the association between H. pylori infection and anaemia.
In summary, over and above known correlates of haemoglobin levels and anaemia, we found that serological evidence of atrophic gastritis was associated with lower mean haemoglobin levels, mainly in women, and a similar trend, although of smaller magnitude, was found in relation to CagA IgG antibody sero-positivity. Our results provide new insight regarding populations at risk for low haemoglobin levels in relation to H. pylori infection and its related gastritis, as measured non-invasively by serum pepsinogen levels.
Electronic supplementary material
Acknowledgements
Funding for this study was provided by the USAID MERC Program (Grant # TA-MOU-01-M21–002) (PI-JDK), by a research grant from DCURE Israel [PI-JDK], the Stanley Steyer Institute for Cancer Epidemiology and Research at Tel Aviv University, School of Public Health (PI-DC), and a MAOF scholarship awarded to KM by the Council of High Education. We are very thankful to Prof. Guillermo I Perez-Perez and Prof. Martin J Blaser from New York University School of Medicine, New York, NY, USA for providing the recombinant CagA antigen.
Author Contributions
K.M., D.C. and J.D.K. designed the study and directed its implementation, including quality assurance and control. R.S., H.N. and G.B.D. helped supervise the field activities, data collection and laboratory work. G.B.D. performed the laboratory experiments. K.M., D.C. and J.D.K. designed the study’s analytic strategy. K.M. analyzed the data and prepared the first draft of the manuscript. All authors contributed to the writing and approved the manuscript.
Data Availability
Data can be provided upon request to the corresponding author (KM).
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Daniel Cohen and Jeremy D. Kark contributed equally.
Jeremy D. Kark is deceased.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-35937-9.
References
- 1.Muhsen K, Cohen D. Helicobacter pylori infection and iron stores: a systematic review and meta-analysis. Helicobacter. 2008;13:323–340. doi: 10.1111/j.1523-5378.2008.00617.x. [DOI] [PubMed] [Google Scholar]
- 2.Cover TL, Blaser MJ. Helicobacter pylori in health and disease. Gastroenterology. 2009;136:1863–1873. doi: 10.1053/j.gastro.2009.01.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nomura A, et al. Helicobacter pylori infection and gastric-carcinoma among Japanese-Americans in Hawaii. New. Engl. J. Med. 1991;325:1132–1136. doi: 10.1056/Nejm199110173251604. [DOI] [PubMed] [Google Scholar]
- 4.Parsonnet J, et al. Helicobacter pylori infection and gastric lymphoma. N. Engl. J. Med. 1994;330:1267–1271. doi: 10.1056/NEJM199405053301803. [DOI] [PubMed] [Google Scholar]
- 5.Nomura AMY, Perez-Perez GI, Lee J, Stemmermann G, Blaser MJ. Relation between Helicobacter pylori cagA status and risk of peptic ulcer disease. Am. J. Epidemiol. 2002;155:1054–1059. doi: 10.1093/aje/155.11.1054. [DOI] [PubMed] [Google Scholar]
- 6.Hudak Lauren, Jaraisy Ameen, Haj Saeda, Muhsen Khitam. An updated systematic review and meta-analysis on the association betweenHelicobacter pyloriinfection and iron deficiency anemia. Helicobacter. 2016;22(1):e12330. doi: 10.1111/hel.12330. [DOI] [PubMed] [Google Scholar]
- 7.Malfertheiner P, et al. Management of Helicobacter pylori infection-the Maastricht V/Florence Consensus Report. Gut. 2017;66:6–30. doi: 10.1136/gutjnl-2016-312288. [DOI] [PubMed] [Google Scholar]
- 8.Van Vliet AH, et al. The role of the Ferric Uptake Regulator (Fur) in regulation of Helicobacter pylori iron uptake. Helicobacter. 2002;7:237–244. doi: 10.1046/j.1523-5378.2002.00088.x. [DOI] [PubMed] [Google Scholar]
- 9.Yokota S, et al. Enhanced Fe ion-uptake activity in Helicobacter pylori strains isolated from patients with iron-deficiency anemia. Clin Infect Dis. 2008;46:e31–33. doi: 10.1086/526784. [DOI] [PubMed] [Google Scholar]
- 10.Cardenas Victor M, Prieto-Jimenez Carmen A, Mulla Zuber D, Rivera Jose O, Dominguez Delfina C, Graham David Y, Ortiz Melchor. Helicobacter pylori Eradication and Change in Markers of Iron Stores Among Non–iron-deficient Children in El Paso, Texas: An Etiologic Intervention Study. Journal of Pediatric Gastroenterology and Nutrition. 2011;52(3):326–332. doi: 10.1097/MPG.0b013e3182054123. [DOI] [PubMed] [Google Scholar]
- 11.Berg G, Bode G, Blettner M, Boeing H, Brenner H. Helicobacter pylori infection and serum ferritin: A population-based study among 1806 adults in Germany. Am J Gastroenterol. 2001;96:1014–1018. doi: 10.1111/j.1572-0241.2001.03686.x. [DOI] [PubMed] [Google Scholar]
- 12.Miernyk K, et al. The Effect of Helicobacter pylori infection on iron stores and iron deficiency in urban Alaska Native adults. Helicobacter. 2013;18:222–228. doi: 10.1111/hel.12036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Muhsen K, et al. Helicobacter pylori infection is associated with low serum ferritin levels in Israeli Arab children-a seroepidemiologic study. J. Pediatr. Gastroentrol. Nutr. 2009;49:262–264. doi: 10.1097/MPG.0b013e31818f0a0d. [DOI] [PubMed] [Google Scholar]
- 14.Kearney DJ, Ioannou GN. The association between Helicobacter pylori seropositivity and serum markers of iron stores in the United States. Gastroenterology. 2005;128:A563–A563. [Google Scholar]
- 15.Ciacci C, et al. Helicobacter pylori impairs iron absorption in infected individuals. Digest. Liver. Dis. 2004;36:455–460. doi: 10.1016/j.dld.2004.02.008. [DOI] [PubMed] [Google Scholar]
- 16.Nomura AM, et al. Helicobacter pylori CagA seropositivity and gastric carcinoma risk in a Japanese American population. J. Infect. Dis. 2002;186:1138–1144. doi: 10.1086/343808. [DOI] [PubMed] [Google Scholar]
- 17.Levi Z, et al. Body mass index and socioeconomic status measured in adolescence, country of origin, and the incidence of gastroesophageal adenocarcinoma in a cohort of 1 million men. Cancer. 2013;119:4086–4093. doi: 10.1002/cncr.28241. [DOI] [PubMed] [Google Scholar]
- 18.Zhang ZW, et al. The relation between gastric vitamin C concentrations, mucosal histology, and CagA seropositivity in the human stomach. Gut. 1998;43:322–326. doi: 10.1136/gut.43.3.322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Park JH, et al. Correlation between Helicobacter pylori infection and vitamin C levels in whole blood, plasma, and gastric juice, and the pH of gastric juice in Korean children. J. Pediatr. Gastroentrol. Nutr. 2003;37:53–62. doi: 10.1097/00005176-200307000-00009. [DOI] [PubMed] [Google Scholar]
- 20.Rood JC, et al. Helicobacter pylori-associated gastritis and the ascorbic acid concentration in gastric juice. Nutr. Cancer. 1994;22:65–72. doi: 10.1080/01635589409514332. [DOI] [PubMed] [Google Scholar]
- 21.Betesh AL, Ana CAS, Cole JA, Fordtran JS. Is achlorhydria a cause of iron deficiency anemia? Am J Clin Nutr. 2015;102:9–19. doi: 10.3945/ajcn.114.097394. [DOI] [PubMed] [Google Scholar]
- 22.Silva B, Faustino P. An overview of molecular basis of iron metabolism regulation and the associated pathologies. Biochim. Biophys. Acta. 2015;1852:1347–1359. doi: 10.1016/j.bbadis.2015.03.011. [DOI] [PubMed] [Google Scholar]
- 23.Lahner E, Annibale B. Pernicious anemia: new insights from a gastroenterological point of view. World. J. Gastroenterol. 2009;15:5121–5128. doi: 10.3748/wjg.15.5121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Annibale B, Lahner E, Fave GD. Diagnosis and management of pernicious anemia. Curr. Gastroenterol. Rep. 2011;13:518–524. doi: 10.1007/s11894-011-0225-5. [DOI] [PubMed] [Google Scholar]
- 25.Miki K, Urita Y. Using serum pepsinogens wisely in a clinical practice. J. Dig. Dis. 2007;8:8–14. doi: 10.1111/j.1443-9573.2007.00278.x. [DOI] [PubMed] [Google Scholar]
- 26.Zagari RM, et al. Systematic review with meta-analysis: diagnostic performance of the combination of pepsinogen, gastrin-17 and anti-Helicobacter pylori antibodies serum assays for the diagnosis of atrophic gastritis. Aliment. Pharmacol. Ther. 2017;46:657–667. doi: 10.1111/apt.14248. [DOI] [PubMed] [Google Scholar]
- 27.Syrjanen K. A Panel of serum biomarkers (GastroPanel (R)) in non-invasive diagnosis of atrophic gastritis. Systematic review and meta-analysis. Anticancer. Res. 2016;36:5133–5144. doi: 10.21873/anticanres.11083. [DOI] [PubMed] [Google Scholar]
- 28.Graham DY, et al. Noninvasive versus histologic detection of gastric atrophy in a Hispanic population in North America. Clin. Gastroenterol. Hepatol. 2006;4:306–314. doi: 10.1016/j.cgh.2005.11.003. [DOI] [PubMed] [Google Scholar]
- 29.Merom D, Sinnreich R, Aboudi V, Kark JD, Nassar H. Lifestyle physical activity among urban Palestinians and Israelis: a cross-sectional comparison in the Palestinian-Israeli Jerusalem risk factor study. BMC. Public. Health. 2012;12:90. doi: 10.1186/1471-2458-12-90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kark JD, et al. Leukocyte telomere length and coronary artery calcification in Palestinians. Atherosclerosis. 2013;229:363–368. doi: 10.1016/j.atherosclerosis.2013.05.030. [DOI] [PubMed] [Google Scholar]
- 31.Blaser MJ, et al. Infection with Helicobacter pylori strains possessing cagA is associated with an increased risk of developing adenocarcinoma of the stomach. Cancer. Res. 1995;55:2111–2115. [PubMed] [Google Scholar]
- 32.Cover TL, et al. Serologic detection of infection with cagA+Helicobacter pylori strains. J. Clin. Microbiol. 1995;33:1496–1500. doi: 10.1128/jcm.33.6.1496-1500.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Abramson JH. WINPEPI updated: computer programs for epidemiologists, and their teaching potential. Epidemiol. Perspect. Innov. 2011;8:1. doi: 10.1186/1742-5573-8-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rugge M, et al. Gastritis OLGA-staging and gastric cancer risk: a twelve-year clinico-pathological follow-up study. Aliment. Pharmacol. Ther. 2010;31:1104–1111. doi: 10.1111/j.1365-2036.2010.04277.x. [DOI] [PubMed] [Google Scholar]
- 35.Milman N, Rosenstock S, Andersen L, Jorgensen T, Bonnevie O. Serum ferritin, hemoglobin, and Helicobacter pylori infection: A seroepidemiologic survey comprising 2794 Danish adults. Gastroenterology. 1998;115:268–274. doi: 10.1016/S0016-5085(98)70192-1. [DOI] [PubMed] [Google Scholar]
- 36.Santos IS, et al. Helicobacter pylori is not associated with anaemia in Latin America: results from Argentina, Brazil, Bolivia, Cuba, Mexico and Venezuela. Public. Health. Nutr. 2009;12:1862–1870. doi: 10.1017/S1368980009004789. [DOI] [PubMed] [Google Scholar]
- 37.Cardenas VM, Mulla ZD, Ortiz M, Graham DY. Iron deficiency and Helicobacter pylori infection in the United States. Am. J. Epidemiol. 2006;163:127–134. doi: 10.1093/aje/kwj018. [DOI] [PubMed] [Google Scholar]
- 38.Lynn TV, et al. Helicobacter pylori infection among non-Native educators in Alaska. Int. J. Circumpolar. Health. 2007;66:135–143. doi: 10.3402/ijch.v66i2.18244. [DOI] [PubMed] [Google Scholar]
- 39.Weyermann M, et al. Role of Helicobacter pylori infection in iron deficiency during pregnancy. Am. J. Obstet. Gynecol. 2005;192:548–553. doi: 10.1016/j.ajog.2004.08.028. [DOI] [PubMed] [Google Scholar]
- 40.Mulayim B, Celik NY, Yanik FF. Helicobacter pylori infection detected by C-14-Urea breath test is associated with iron deficiency anemia in pregnant women. J. Obstet. Gynaecol. Res. 2008;34:980–985. doi: 10.1111/j.1447-0756.2008.00822.x. [DOI] [PubMed] [Google Scholar]
- 41.Kibru, D., Gelaw, B., Alemu, A. & Addis, Z. Helicobacter pylori infection and its association with anemia among adult dyspeptic patients attending Butajira Hospital, Ethiopia. BMC. Infect. Dis. 14, 10.1186/s12879-014-0656-3 (2014). [DOI] [PMC free article] [PubMed]
- 42.Kaye PV, et al. The clinical utility and diagnostic yield of routine gastric biopsies in the investigation of iron deficiency anemia: a case-control study. Am. J. Gastroenterol. 2008;103:2883–2889. doi: 10.1111/j.1572-0241.2008.02121.x. [DOI] [PubMed] [Google Scholar]
- 43.Nahon S, et al. Helicobacter pylori-associated chronic gastritis and unexplained iron deficiency anemia: a reliable association? Helicobacter. 2003;8:573–577. doi: 10.1111/j.1523-5378.2003.00184.x. [DOI] [PubMed] [Google Scholar]
- 44.Lee SY, et al. Low Hemoglobin levels are related to the presence of gastric atrophy rather than the presence of H. pylori infection itself: a study of 2,398 asymptomatic adults. Gastroenterology. 2012;142:S474–S474. doi: 10.1016/S0016-5085(12)61799-5. [DOI] [Google Scholar]
- 45.Park YH, Kim N. Review of atrophic gastritis and intestinal metaplasia as a premalignant lesion of gastric cancer. J. Cancer. Prev. 2015;20:25–40. doi: 10.15430/JCP.2015.20.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Forrest RD, Jackson CA, Yudkin JS. The epidemiology of the haemoglobin level–a study of 1057 subjects in general practice. Postgrad. Med. J. 1987;63:625–628. doi: 10.1136/pgmj.63.742.625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Milman N, Pedersen AN. Blood haemoglobin concentrations are higher in smokers and heavy alcohol consumers than in non-smokers and abstainers-should we adjust the reference range? Ann. Hematol. 2009;88:687–694. doi: 10.1007/s00277-008-0647-9. [DOI] [PubMed] [Google Scholar]
- 48.Milman N, Byg KE, Mulvad G, Pedersen HS, Bjerregaard P. Haemoglobin concentrations appear to be lower in indigenous Greenlanders than in Danes: assessment of haemoglobin in 234 Greenlanders and in 2804 Danes. Eur. J. Haematol. 2001;67:23–29. doi: 10.1034/j.1600-0609.2001.067001023.x. [DOI] [PubMed] [Google Scholar]
- 49.Yamada M, Wong FL, Suzuki G, Study, R. E. S. A. H Longitudinal trends of hemoglobin levels in a Japanese population–RERF’s Adult Health Study subjects. Eur. J. Haematol. 2003;70:129–135. doi: 10.1034/j.1600-0609.2003.00031.x. [DOI] [PubMed] [Google Scholar]
- 50.Nordenberg D, Yip R, Binkin NJ. The effect of cigarette smoking on hemoglobin levels and anemia screening. JAMA. 1990;264:1556–1559. doi: 10.1001/jama.1990.03450120068031. [DOI] [PubMed] [Google Scholar]
- 51.Na’amnih W, et al. Continuous Decrease of Consanguineous Marriages Among Arabs in Israel. Am. J. Hum. Biol. 2015;27:94–98. doi: 10.1002/ajhb.22610. [DOI] [PubMed] [Google Scholar]
- 52.Eliakim R, Rachmilewitz EA. Hemoglobinopathies in Israel. Hemoglobin. 1983;7:479–485. doi: 10.3109/03630268309038420. [DOI] [PubMed] [Google Scholar]
- 53.Neumann WL, Coss E, Rugge M, Genta RM. Autoimmune atrophic gastritis–pathogenesis, pathology and management. Nat. Rev. Gastroenterol. Hepatol. 2013;10:529–541. doi: 10.1038/nrgastro.2013.101. [DOI] [PubMed] [Google Scholar]
- 54.D’Elios MM, Appelmelk BJ, Amedei A, Bergman MP, Del Prete G. Gastric autoimmunity: the role of Helicobacter pylori and molecular mimicry. Trends. Mol. Med. 2004;10:316–323. doi: 10.1016/j.molmed.2004.06.001. [DOI] [PubMed] [Google Scholar]
- 55.Kekki M, Samloff IM, Varis K, Ihamaki T. Serum pepsinogen I and serum gastrin in the screening of severe atrophic corpus gastritis. Scand. J. Gastroenterol. Suppl. 1991;186:109–116. doi: 10.3109/00365529109103997. [DOI] [PubMed] [Google Scholar]
- 56.Storskrubb T, et al. Serum biomarkers provide an accurate method for diagnosis of atrophic gastritis in a general population: The Kalixanda study. Scand. J. Gastroenterol. 2008;43:1448–1455. doi: 10.1080/00365520802273025. [DOI] [PubMed] [Google Scholar]
- 57.Sipponen P, et al. Serum levels of amidated gastrin-17 and pepsinogen I in atrophic gastritis: an observational case-control study. Scand. J. Gastroenterol. 2002;37:785–791. doi: 10.1080/gas.37.7.785.791. [DOI] [PubMed] [Google Scholar]
- 58.Vaananen H, et al. Non-endoscopic diagnosis of atrophic gastritis with a blood test. Correlation between gastric histology and serum levels of gastrin-17 and pepsinogen I: a multicentre study. Eur. J. Gastroenterol. Hepatal. 2003;15:885–891. doi: 10.1097/01.meg.0000059169.46867.01. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data can be provided upon request to the corresponding author (KM).