Abstract
Candida species cause cutaneous and systemic infections with a high mortality rate, especially in immunocompromised patients. The emergence of resistance to the most common antifungal drugs, also due to biofilm formation, requires the development of alternative antifungal agents. The antimicrobial peptide VLL-28, isolated from an archaeal transcription factor, shows comparable antifungal activity against 10 clinical isolates of Candida spp. Using a fluoresceinated derivative of this peptide, we found that VLL-28 binds to the surface of planktonic cells. This observation suggested that it could exert its antifungal activity by damaging the cell wall. In addition, analyses performed on biofilms via confocal microscopy revealed that VLL-28 is differentially active on all the strains tested, with C. albicans and C. parapsilosis being the most sensitive ones. Notably, VLL-28 is the first example of an archaeal antimicrobial peptide that is active towards Candida spp. Thus, this points to archaeal microorganisms as a possible reservoir of novel antifungal agents.
Introduction
Candida species are the most prevalent opportunistic fungal pathogens worldwide. Candida spp. commonly dwell as commensal microbes colonizing the skin, oral cavities, and gastrointestinal and genital-urinary tracts of most healthy humans. When alterations in the host microbiota or in the host immune and defence system occur, Candida spp. can become pathogenic, causing numerous disorders ranging from cutaneous infections to severe systemic infections with a high mortality rate in hospitalized patients1–5.
Candida bloodstream infection is often associated with the presence of implanted medical devices, such as shunts, stents, prostheses, endotracheal tubes, and various types of catheters6–8, on which Candida species grow as a resilient biofilm capable of withstanding high antifungal concentrations. As expected, in patients with candidemia, biofilm-producing strains have been associated with increased morbidity and mortality compared to non-biofilm-producing ones9. Among Candida isolates, C. albicans represents the predominant specie, although, in recent years, an increasing incidence of fungal infections by non-albicans Candida species has been observed in hospital environments4,10–12. C. krusei, C. tropicalis, C. parapsilosis, C. glabrata have all been implicated in biofilm-associated infections13,14.
Biofilms are highly structured communities of enclosed microorganisms within a self-produced protective extracellular matrix, with biofilm-embedded cells showing properties that are distinct from planktonic cells15–17. In particular, the biofilm matrix acts as a protective barrier, making the microbial cells more resistant towards conventional antifungal therapeutics and the host immune system, as well as other environmental perturbations. Therefore, biofilm-forming Candida infections are difficult to treat, and biofilm-related (sessile) minimal inhibitory concentrations (MICs) are often extremely higher than the MICs for planktonic (non-biofilm) cells18,19. Furthermore, the variation of biofilm formation among Candida strains and/or the differential biofilm response to several antifungal classes contributes to the virulence traits. In addition to the presence of a secreted extracellular matrix, the enhanced drug resistance shown by biofilm-embedded cells is also related to (i) the local increase in cell density, (ii) the upregulation of efflux pumps, (iii) the alteration of sterols in their membranes, and iv) the activation of stress response mechanisms. This leads to the onset of persistent cells, which are a subset of metabolically dormant yeasts cells within biofilms20–23.
Because of the recalcitrance of Candida biofilms to treatment with conventional drugs, high antifungal doses in systemic therapy are needed to eradicate the infections, along with the removal of the colonized medical devices in the case of implantations24–26. Only three classes of antifungal drugs are currently being used to treat invasive candidiasis: azoles (i.e., fluconazole), echinocandins (i.e., caspofungin) and polyenes (i.e., amphotericin B)27. In recent years, the antifungal resistance of Candida spp., especially of non-albicans species, has been observed. For example, C. krusei was found to have a reduced susceptibility to fluconazole, while C. glabrata was reported to be resistant to both azoles and echinocandins28. Since amphotericin B resistance in Candida species remains extremely rare, this drug is the treatment of choice in monotherapy for life-threatening systemic candidiasis. However, with the onset of antifungal resistant pathogens, there is an increasing need to design new antimycotics and/or discover alternative agents that improve the fungicidal activity of the current antifungals.
Antimicrobial peptides (AMPs) with their extraordinary properties, such as broad-spectrum activity, rapid action and unlikely development of resistance, have become promising molecules as new antibiotics29,30. An example of such molecules is represented by cationic antimicrobial peptides (CAMPs), i.e., short and positively charged peptides with an amphipathic structure. CAMPs are active against Gram-positive and Gram-negative bacteria, as well as fungi and protozoa29–31. Regardless of their specific mechanism of action, the interaction of CAMPs with the bacterial cell membrane is the key step, which eventually leads either to the disruption of the membrane integrity or to the alteration of its electrochemical potential31,32. Interestingly, some CAMPs also exhibit toxicity towards eukaryotes, such as fungi, despite the different lipidic membrane compositions and the distinct structures of the cell wall33.
While the effect of some AMPs on biofilm formation, as well as their antimicrobial activity on biofilm-embedded cells, has already been investigated34–36, only a limited number of studies describes the effect of CAMPs on biofilm formation by fungal pathogens such as Candida37,38. Recently, we identified a cryptic CAMP-like peptide (designated as VLL-28) in the sequence of the archaeal transcription factor Stf7639, which is encoded by the hybrid plasmid–virus pSSVx infecting Sulfolobus islandicus40–42. The peptide was identified using an in silico tool developed to search for cryptic antimicrobial peptide-like sequences hidden in the primary structure of proteins32,43–46. VLL-28 displays chemical, physical and functional properties typical of CAMPs and acquires a defined structure in the presence of membrane mimetics47,48. Notably, this CAMP turned out to be toxic not only to Gram-negative and Gram-positive bacteria but also to C. albicans47. Therefore, in this study, we investigated the antifungal activity of VLL-28 towards pathogenic C. albicans and non-albicans Candida spp. isolated from blood infections. In particular, we show the in vitro ability of VLL-28 to (1) inhibit yeast cells growth in a planktonic state, (2) prevent cell adhesion, and (3) eradicate established biofilms.
Results
Antifungal activity of VLL-28 towards Candida spp. planktonic cells
MIC values for amphotericin B, anidulafungin, micafungin, caspofungin, 5-fluorocytosine, posaconazole, voriconazole, itraconazole, and fluconazole of 10 clinical isolates of Candida spp., as well as the reference strain C. albicans ATCC10231, are reported in Table S1. The antifungal activity of VLL-28, expressed as MIC and MFC values against the same panel of isolates, is shown in Table 1. Among them, the planktonic cells of C. tropicalis were the most susceptible to the peptide VLL-28 (MIC = 12.5 µM), followed by C. albicans, C. parapsilosis and C. krusei (MIC = 25 µM), while C. glabrata planktonic cells were the least sensitive, showing the highest MIC value of 50 µM. In addition, the activity of VLL-28 was also investigated in terms of minimum fungicidal concentration (MFC). The values were two-fold higher than the MIC values for all Candida species, except C. albicans 80 and C. tropicalis isolates for which the MFC values were four-fold higher than the corresponding MICs (Table 1).
Table 1.
Minimal fungicidal concentration (MFC) and minimal inhibition concentration (MIC) of amphotericin B and VLL-28 against Candida species.
| Yeasts | amphotericin B | VLL-28 | ||
|---|---|---|---|---|
| MIC μg/mL | MFC μg/mL | MIC μg/mL (μM) | MFC μg/mL (μM) | |
| C. albicans ATCC10231 | 0.25 | 1 | 88.5 (25) | 177 (50) |
| C. albicans 80 | 0.5 | 2 | 88.5 (25) | 354 (100) |
| C. albicans 81 | 0.25 | 2 | 88.5 (25) | 177 (50) |
| C. parapsilosis 3 | 0.5 | 2 | 88.5 (25) | 177 (50) |
| C. parapsilosis 10 | 0.5 | 2 | 88.5 (25) | 177 (50) |
| C. tropicalis 54 | 0.5 | 4 | 44.25 (12.5) | 177 (50) |
| C. tropicalis 2 | 0.5 | 4 | 44.25 (12.5) | 177 (50) |
| C. glabrata 28 | 0.25 | 2 | 177 (50) | 354 (100) |
| C. glabrata 34 | 0.25 | 1 | 177 (50) | 354 (100) |
| C. krusei 1 | 0.5 | 2 | 88.5 (25) | 177 (50) |
| C. krusei 14 | 1 | 2 | 88.5 (25) | 177 (50) |
In vitro biofilm formation assay
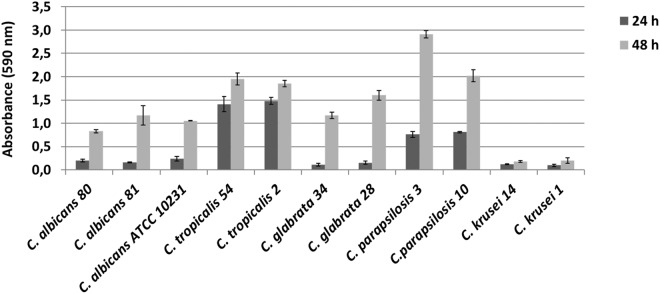
Candida species were tested for the ability to produce biofilm using the crystal violet staining method, and the results are shown in Fig. 1. All the tested isolates turned out to be biofilm producers. At 24 hours, C. glabrata 34, C. krusei 14 and 1 were weak producers, all C. albicans isolates and C. glabrata 28 were moderate producers, and all C. tropicalis and C. parapsilosis were strong producers. At 48 hours, all Candida species, with the exception of the C. krusei, were strong producers.
Figure 1.
In vitro biofilm production. Candida isolates were grown in 96-well microtitre plates and their biofilm was quantified through staining with crystal violet after 24 and 48 hours of cultivation. Each value is the average of three independent experiments in triplicate. Error bars are the standard deviations.
Cellular localization of VLL-28
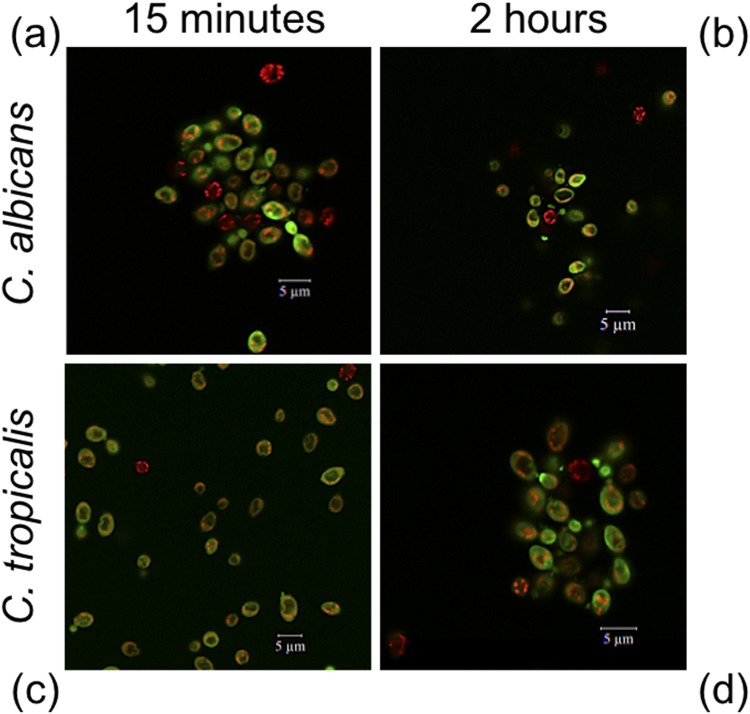
A fluorescein-labelled derivative of VLL-28 (VLL-28*) was used to study the cellular localization of the peptide in C. albicans and C. tropicalis using confocal laser scanning microscope (CLSM)49. C. albicans and C. tropicalis were chosen because these two strains are among the most sensitive to VLL-28 activity. The effectiveness of VLL-28* was found to be similar to that of the not labelled peptide50. C. albicans and C. tropicalis, pre-incubated with MitoTracker Orange, were treated with VLL-28* at the concentration of 12.5 μM and 25 μM, respectively, for 15 min and 2 h. Confocal images showed that, for both strains, the green signal (VLL-28*) was uniformly localized at the cellular surface of the treated cells after 15 min and did not overlap with the red (MitoTracker) signal (Fig. 2). This indicates that the peptide interacts with the fungal surfaces, probably binding to the negatively charged phospholipids, and that no internalization occurs.
Figure 2.
VLL-28 intracellular localization detected using confocal laser scanner microscopy. Candida cells were incubated in PBS at different times with a dose of the peptide corresponding to their MIC values. VLL-28 conjugated with FITC is coloured in green, while mitotracker is in red. Images of C. albicans and C. tropicalis cells are shown at 15 min (a,c) and 2 hours (b,d) posttreatment.
Inhibition of biofilm formation by VLL-28
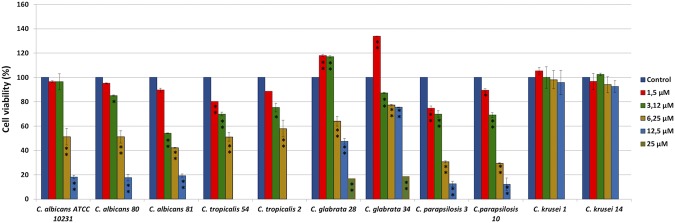
Cell adhesion either on the host cell tissues or on abiotic surfaces is the first step during Candida biofilm formation. Therefore, we investigated whether VLL-28 could prevent biofilm production by interfering with the cell viability at sub-MIC concentrations. The minimum biofilm inhibitory concentration (MBIC) was determined by quantifying the metabolic activity of the adherent cells using the XTT assay (Fig. 3). The percentage of biofilm viability was strongly reduced for C. tropicalis, C. albicans and C. parapsilosis strains, with MBIC values of 12.5 µM, and for C. glabrata strains, with MBIC values of 25 µM, while no inhibition of cell adhesion was observed for both C. krusei isolates (Fig. 3).
Figure 3.
Effect of different concentrations of VLL-28 on cell viability of Candida species. Cell viability within the biofilm was assessed by measuring the reduction of XTT. The results are reported as the percentage relative to the untreated control. Technical and biological triplicates were conducted for all experiments, and statistical significance was determined using one-way ANOVA and Dunn’s test. *P value between 0.01 and 0.001; **P value < 0.001.
Activity against preformed biofilms of Candida species
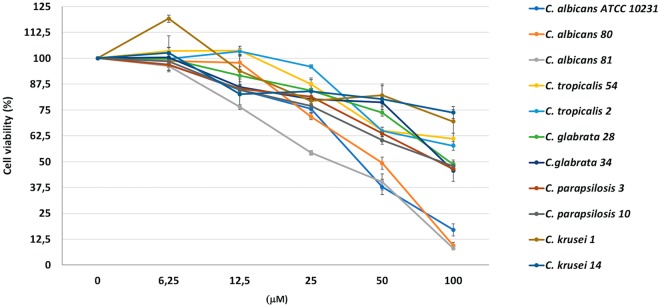
Biofilm formation in Candida confers to the embedded cells an increased resistance towards antifungal agents. Therefore, we extended our investigation to the anti-biofilm activity of VLL-28 on preformed Candida biofilms. One-day-old biofilms were exposed to increasing doses of the peptide, and its antifungal activity was quantified using the XTT reduction assay. VLL-28 effectively reduced the viability of the cells embedded in the mature biofilms, with MBEC50 and MBEC80 of 50 μM and 100 μM, respectively, for C. albicans isolates and the reference strain. In addition, VLL-28 killed 50% of the cells in the preformed biofilm (MBEC50) of C. glabrata and C. parapsilosis isolates at a concentration of 100 μM. On the other hand, VLL-28 was less effective against the mature biofilms of C. tropicalis and C. krusei isolates (Fig. 4, Table S3).
Figure 4.
Antifungal activity of VLL-28 on cells in preformed Candida biofilms. Mature biofilms were exposed to the peptide at concentrations ranging from 6.25 to 100 μM for 24 h. Each data point shows the XTT activity of the VVL-28-treated biofilm normalized to the control (untreated), which was considered to be 100%. Technical and biological triplicates were run for all experiments. Statistical significance was determined using one-way ANOVA and Dunn’s test (for P values see Table S3).
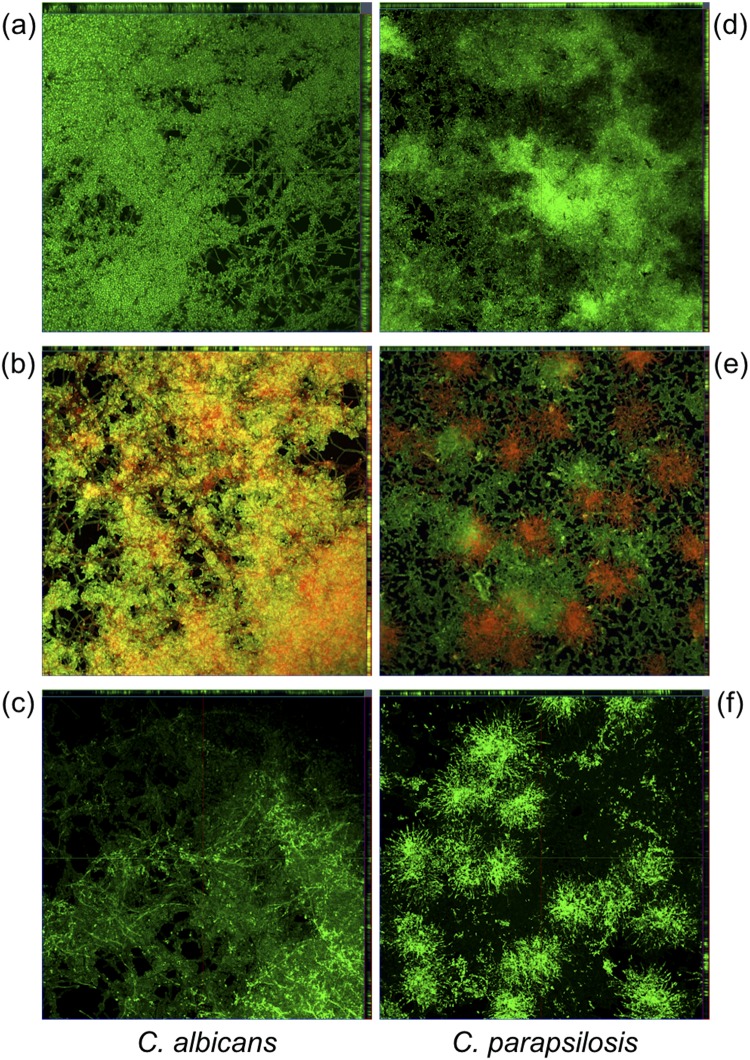
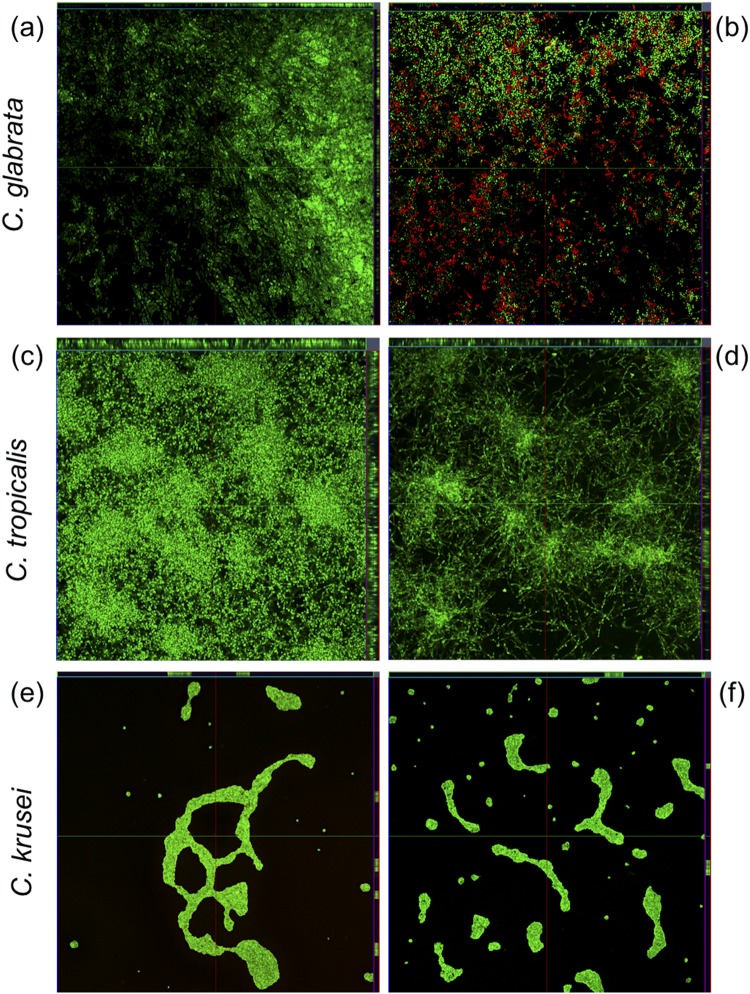
CLSM was used to investigate the effect of VLL-28 on Candida preformed biofilms. Biofilms were grown on Nunc® Lab-Tek® II chambered cover glasses and stained using the LIVE/DEAD FungaLight Yeast Viability Kit. As shown by the differential staining with SYTO9 (green fluorescence, live cells) and propidium iodide (red or yellow-red fluorescence, dead cells), VLL-28 caused the death of most of the cells embedded in the mature biofilms of C. albicans (Figs 5a,b and S1a,b) and decreased the cell viability in those of C. parapsilosis (Figs 5d,e and S1d,e) and C. glabrata (Fig. 6a,b and S2a,b). In the case of C. tropicalis biofilm, images showed a thickness reduction of approximately 30% and the appearance of regions with lower density (Figs 6c,d and S2c,d) compared to the untreated control. However, viable cells were detectable, which suggests that the peptide could have a fungistatic effect on the preformed biofilm of C. tropicalis. Regarding C. krusei, VLL-28 administration did not cause any visible change in the thicknesses and viability of its mature biofilm, which is consistent with what was observed both by CLSM imaging and by the XTT reduction assay (Figs 6e,f and S2e,f).
Figure 5.
CLSM of VLL-28 on preformed biofilm of C. albicans (a–c) and C. parapsilosis (d–f). Panels a and d show untreated biofilms stained using the LIVE/DEAD FungaLight Yeast Viability Kit. Panels b and e show biofilms treated with VLL-28 (50 µM) and stained as in a and d. Panels c and f show biofilms treated with VLL-28*.
Figure 6.
CLSM of VLL-28 on preformed biofilm of C. glabrata (a,b), C. tropicalis (c,d) and C. krusei (e,f). Panels a, c and e show untreated biofilms. Panels b, d and f show biofilms treated with VLL-28 (50 µM). All the biofilms were stained using the LIVE/DEAD FungaLight Yeast Viability Kit.
To verify whether the different effects on Candida biofilms could be traced back to the different ability of VLL-28 to penetrate the biofilm matrix, C. albicans and C. parapsilosis biofilms were treated with VLL-28* and analysed using CLSM. As shown, the distribution of VLL-28* mirrors that of the dead cells (red spots), i.e., the propidium iodide stained areas (Figs 5c,f and S1c,f).
Discussion
The high risk of invasive fungal infections within the increasing population of immunocompromised patients, along with the emergence of resistance to the most common antifungal drugs in Candida spp., also due to biofilm phenotypes, requires the development of new antifungal agents. Antimicrobial peptides might represent a very promising alternative, not only to treat planktonic Candida but also for biofilm-embedded Candida cells51.
Most of the currently used antifungal agents show a specific mechanism of action. For instance, azoles act by interfering with the biosynthesis of membrane ergosterol52,53, while echinocandins block the synthesis of cell wall 1,3 beta-glucan, and polyenes bind ergosterol and disrupt membrane integrity53,54. In contrast, amphipathic AMPs exert their antimicrobial activity by binding the cellular membrane and then increasing its permeability31,32. Unlike classic antifungal drugs, the peculiar mechanism of action of AMPs hinders the development of microbial resistance.
VLL-28 is a cryptic antimicrobial peptide derived from a transcriptional factor of Sulfolobus islandicus39. A previous study has shown the ability of VLL-28 to inhibit the growth of bacteria and fungi in planktonic form47. In this study, we investigated the antifungal and anti-biofilm activity of VLL-28 against clinical isolates of C. albicans and non-albicans species. VLL-28 exhibited fungicidal activity against almost all the planktonic Candida spp. tested, with MIC values in the range of 12.5–50 µM. These data are comparable to the MIC values reported for several other natural and artificial AMPs55–60, as reported in Table S2. VLL-28 displayed a reduced activity only towards C. glabrata, which showed the highest MIC and MFC values (Table 1). The poor susceptibility of C. glabrata to various CAMPs has already been reported61 and may reflect the unique and distinctive features of the cell wall of this pathogen62. Interestingly, VLL-28 also exhibited strong fungicidal activity towards the planktonic cells of C. krusei, which is regarded as a potentially multidrug-resistant (MDR) pathogen. This is a naturally resistant specie to fluconazole (FLC), which has shown an increased resistance to both flucytosine and amphotericin B63, as well as cross-resistance to azoles64. In addition, the clinical failure of caspofungin towards C. krusei infections has been reported in recent years65.
The results obtained with the fluorescein-labelled peptide indicate that VLL-28 interacts primarily with the fungal surfaces. In fact, no internalization was observed. This confirms that VLL-28 behaves as a typical CAMP by damaging the cell membrane and/or the cell wall, thus, greatly decreasing the likelihood of the appearance of strains resistance.
The ability of Candida spp. to adhere to epithelial or endothelial surfaces, as well as to implanted medical devices by forming resilient biofilms, is an important virulence trait that promotes the persistence of the infection. Compared with planktonic cells, Candida biofilms are characterized by increased resistance to conventional antifungal drugs, in particular to amphotericin B and fluconazole66–68. Several factors have been suggested to be involved in the resistance of biofilm to antimicrobial drugs, including the expression of conventional resistance genes, such as those coding for efflux pumps69. In addition, the production of an extracellular matrix, which is a specific mechanism of the biofilm growth mode, limits drug penetration or even binds the antifungal agent and prevents it from reaching its cellular target20,21,70. Thus, an extremely limited drug arsenal is available to treat biofilm-related Candida infections. VLL-28 effectively prevents biofilm formation by reducing the cells’ adhesion to the abiotic surfaces of all the strains tested (except for C. krusei) at a concentration of the peptide that is 50% of the MIC value. The ability of this peptide to prevent biofilm formation is particularly important for medical device implantations. In fact, the microbial colonization of indwelling medical implants with subsequent biofilm formation can lead to severe complications associated with increased morbidity and mortality, such as bloodstream infections and systemic inflammation8.
Infection by non-albicans Candida species is currently a highly active research area, as these species are becoming increasingly prevalent69. Notably, VLL-28 reduces the metabolic activity, not only of mature biofilms formed by C. albicans (80%) but also of those formed by C. glabrata and C. parapsilosis (50%), at concentrations from 2- to 4-fold higher than those needed for the planktonic state. This result is not unexpected because even conventional antifungals that exhibit anti-biofilm activity have MBECs several-fold higher than the MICs for the same isolates71–73. Furthermore, our finding is similar to those reported for several other AMPs (Table S2).
CLSM images showed a diffuse cell permeabilization in 1-day-old biofilm of C. albicans and C. glabrata treated with sub-MBEC values of VLL-28, while the effect on the mature biofilm cells of C. parapsilosis was focal. The fluorescein-labelled peptide distribution appeared to overlap permeabilized cell zones. The diffuse or focal VLL-28 toxicity may depend on the different distributions of the cells on the abiotic surfaces after 24 hours of biofilm formation. In addition, VLL-28 caused a significant decrease of the biofilm biomass of C. tropicalis, but no cell killing was observed in this case. This result may be explained by the ability of VLL-28 to affect the matrix structure/stability by interacting with one or more components, thus inducing disaggregation of the biofilm without cell killing. It is worth noting that drug diffusion to the cell community within a given biofilm can be affected not only by the overall extent of the matrix itself but also by its chemical nature74. Indeed, Al-Fattani and Douglas74 demonstrated that C. tropicalis strains could form a compact extracellular matrix containing hexosamine-rich polysaccharides, which is poorly penetrated by antifungal agents.
In conclusion, we showed the ability of VLL-28 to exert antifungal activity against planktonic cells and mature biofilm of clinical isolates of C. albicans and non-albicans species, including Candida krusei, which is intrinsically resistant to fluconazole75. These results appear relevant and deserve further study with the perspective of developing alternative and/or complementary antifungal therapies.
Materials and Methods
Yeast strains and culture conditions
The Candida species evaluated in this study included the C. albicans ATCC 10231 reference strain and eight clinical isolates (Table 1) belonging to a collection of fungal strains previously established at the Department of Molecular Medicine and Medical Biotechnology (University of Naples Federico II). Identification was performed by subcultures on chromogenic agar (Chromid agar) (Becton Dickinson) and by biochemical characterisation using the Vitek II system (Biomerieux) and was confirmed by MS MALDI-TOF (Bruker).
Susceptibility to amphotericin B, anidulafungin, micafungin, caspofungin, 5-fluorocytosine, posaconazole, voriconazole, itraconazole, and fluconazole was assessed using the Sensititre Yeast One colorimetric microdilution method (Thermofisher). All strains were stored as 15% (v/v) glycerol stocks at −80 °C. Prior to each experiment, cells were subcultured from the stocks onto Sabouraud dextrose agar (SDA) (Becton Dickinson) at 37 °C for 48 h.
Peptides
The peptide VLL-28 (VLLVTLTRLHQRGVIYRKWRHFSGRKYR) and its fluoresceinated derived form (VLLVTLTRLHQRGVIYRKWRHFSGRKYRGK*) (VLL-28*), bearing the chromophore fluorescein coupled to the last lysine residue, were synthetized and purified to 95% homogeneity by Inbios (Napoli, Italy), as confirmed by LC–MS analysis.
Determination of the minimum inhibitory concentration (MIC) and the minimum fungicidal concentration (MFC)
The antifungal activity of VLL-28 was determined using a standardized broth microdilution method (Clinical and Laboratory Standards Institute (CLSI) document M27-A2)76. Briefly, for each Candida species, the cell suspension was adjusted to 3 × 103 CFU/mL using a morpholinepropanesulfonic acid (MOPS)-buffered RPMI 1640 medium (R6504 - Sigma) supplemented with 0.2% (w/v) glucose. One hundred microlitre aliquots of these cell suspensions were dispensed into 96-well microtitre plates. Peptide stock solution was serially diluted using the same RPMI 1640 medium and added to the wells at a final concentration ranging from 3 µM to 100 µM, and the plate was incubated for 48 hours at 37 °C. Absorbance at 595 nm was measured using a microplate reader (Biorad mod 680). Amphotericin B at concentrations ranging from 0.25 to 2 µg/mL was chosen as the positive control because all the strains were sensitive to this agent according to the Sensititre Yeast One test.
The MIC was defined as the lowest concentration of the peptide that resulted in 90% growth inhibition after 48 h of incubation. The test was conducted at least three times using independent cell suspensions. The minimum fungicidal concentration (MFC) was determined by transferring 50 µl aliquots of each sample, previously treated with concentrations equal to or higher than the MIC, onto SDA plates and incubating the plates at 37 °C for 24 h. The lowest peptide concentraiton that yielded no fungal growth on agar plates was defined as the MFC.
In vitro biofilm formation assay
Biofilms of Candida spp. were formed in flat-bottomed 96-well microplates as described by Stepanovic with some modifications77. For each strain, a cell suspension in RPMI 1640 medium supplemented with 2% (w/v) glucose was adjusted to 1 × 106–5 × 106 CFU/mL as determined by cell counts using a haemocytometer Neubauer improved chamber. Plate wells were inoculated with 200 µL of standardized yeast suspension in triplicate and incubated at 37 °C for 90 minutes to allow cell adhesion. A negative control was prepared by inoculating 200 µL of a yeast suspension inactivated by boiling. After the adhesion phase, non-adherent cells were removed by thoroughly washing the wells with 0.15 M sterile phosphate-buffered saline (PBS, pH 7.2). Each well was then filled with 200 µL of fresh RPMI 1640, and the plate was incubated at 37 °C for 24 h to allow biofilm formation.
To assess biofilm formation, the culture broth was gently aspirated, and each well was washed twice with PBS and dried at 60 °C for 30 minutes. The biofilm was stained by incubation for 30 min with 50 µL of a 1% (w/v) crystal violet solution. Any excess of crystal violet was removed by washing with PBS before adding 150 µL of absolute ethanol to release the dye from the biofilm. The absorbance was measured at 590 nm using a Biophotometer (Eppendorf) and was related to the amount of biofilm produced. We used the classification introduced by Stepanović et al.77 with some modifications. The isolates tested were classified into four categories: non-adherent (NA), weakly adherent (WA), moderately adherent (MA), or strongly adherent (SA).
2,3-bis(2-methoxy-4-nitro-5-sulfo-phenyl)-2H-tetrazolium-5-carboxanilide (XTT)-reduction assay
The (XTT)-reduction assay has been used as a routine tool for the quantitative measurement of bacterial and fungal metabolic activity, growth and response to antimicrobial treatments78–82. After peptide treatment, the medium was aspirated from each well to remove floating cells, and the wells were thoroughly washed twice with PBS. The assay was conducted as described by Barra et al.80 with some modifications. Two hundred microlitres of XTT solution was added to each well, and the plate was incubated in the dark for 30 min at 37 °C. Changes in the absorbance of XTT were measured spectrophotometrically at 490 nm using a microtitre plate reader (Biorad). An XTT cell proliferation Kit II was purchased from Roche Diagnostics. Viability ratios were computed for each well with respect to their relative controls.
Confocal laser scanning microscopy
CLSM was used to illustrate the effect of peptide (50 µM) on the viability and architecture of mature (24 h) biofilms of Candida species. Biofilm-forming Candida cells were grown on Nunc® Lab-Tek® II chambered cover glasses (Sigma), and the antifungal biofilm susceptibility was assayed as described above.
Biofilms were stained with two nucleic acid dyes using the LIVE/DEAD FungaLight Yeast Viability Kit: SYTO 9 and propidium iodide (PI) (Molecular Probes). SYTO 9 penetrates both viable and nonviable cells, while PI penetrates only cells with damaged membranes (i.e., nonviable cells) and quenches the fluorescence emitted by SYTO 9. Dead and viable cells emit yellow-red and green fluorescence, respectively. Images were captured using an LSM 710 inverted confocal laser-scanning microscope (Zeiss) and analysed using CLSM Z-Stack analysis: depth measurements were taken at regular intervals across the biofilm, and three-dimensional images of mature biofilms were captured.
Cellular localization studies of VLL-28
Confocal laser-scanning fluorescence microscopy was used to study the intracellular target of the peptides49. Double staining of the C. albicans and C. tropicalis strains with FITC-labelled peptides and MitoTracker Orange (chloromethyl-H2-tetramethyl rosamine, Molecular Probes), a permanent mitochondrion-selective dye, was achieved as follows: a Candida cell suspension (200 µL; 3.2 × 106 cells/mL of PBS) was incubated with 150 nM MitoTracker Orange for 15 min at 37 °C. The cells were washed with 200 µL of PBS and treated for 15 min and 2 h with 25 µM and 12.5 µM FITC-labelled peptides for C. albicans and C. tropicalis, respectively. The cells were collected using centrifugation (5 min at 10,000 × g), suspended in 20 µL of PBS and examined by confocal microscopy using an LSM 710 confocal laser-scanning microscope equipped with a 63X objective lens.
Adhesion inhibition assay
The adhesion of Candida spp. was assayed using flat-bottomed 96-well microplates. For each isolate, 100 µL of cell suspension in RPMI medium adjusted to 1 × 106 CFU/mL was incubated with 100 µL of RPMI containing serially double-diluted peptide concentrations in order to obtain the final sub-MIC concentrations for each yeast, ranging from 1.5 µM to 25 µM. The plate was then incubated at 37 °C with a shaking rate of 100 rpm; the positive control consisted of peptide-free wells. After a 60 min adhesion phase, the medium with unbound peptide was aspirated; non-adherent cells were removed by washing the wells with PBS, and 200 µL of fresh RPMI was added. The plate was incubated further at 37 °C for 24 h, and an XTT reduction assay was performed as described below.
The adhesion inhibitory activity of the peptide is referred to as the minimum biofilm inhibitory concentration (MBIC), which is defined as the minimum peptide concentration leading to an 80% reduction of biofilm formation compared to a peptide-free control sample.
Antifungal susceptibility testing of 24 h-old Candida biofilms
Candida biofilms were produced as described above; upon mature biofilm formation, the medium was aspirated, and each well was washed twice gently with 200 µL of PBS to remove planktonic cells. Peptide aliquots (200 µL per well) ranging from 6.25 to 100 µM were added, and the plate was incubated for 24 h. Peptide-free wells were included as positive controls. The biofilm formation at 24 h and 48 h was quantified using the XTT reduction assay described below. The anti-biofilm activity of the peptide is referred to as the minimum biofilm eradication concentration (MBEC), which is defined as the minimum peptide concentration resulting in 80% disruption of the biofilm compared to a peptide-free control culture.
Statistical analysis
All experiments were performed in triplicate with the average and standard deviation calculated for all measurements. Statistical differences among the groups of data were analysed by one-way ANOVA using Prism (version 7.00 for Windows; GraphPad Software, San Diego, CA) and Dunn’s test. In all the comparisons, a P value of 0.05 or lower was considered significant.
Electronic supplementary material
Acknowledgements
This research was carried out in the frame of Programme STAR, financially supported by University of Naples Federico II and Compagnia di San Paolo (16-CSP-UNINA-007).
Author Contributions
All authors contributed to the conception and planning of the study. E.R., A.V. and S.F. conducted the experiments. M.R.C., P.C. and E.N. supervised the experimental work and analysed the data. E.N., P.C. and E.R. wrote the paper. S.F. critically reviewed the manuscript. All the authors have read and approved the final submission.
Availability of Data and Materials
The datasets used and/or analysed during the current study are available from the corresponding authors on reasonable request.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Eugenio Notomista and Maria Rosaria Catania contributed equally.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-35530-0.
References
- 1.Calderone RA, Fonzi WA. Virulence factors of Candida albicans. Trends in microbiology. 2001;9:327–335. doi: 10.1016/S0966-842X(01)02094-7. [DOI] [PubMed] [Google Scholar]
- 2.Pappas PG, et al. Guidelines for treatment of candidiasis. Clinical Infectious Diseases. 2004;38:161–189. doi: 10.1086/380796. [DOI] [PubMed] [Google Scholar]
- 3.Wenzel RP. Nosocomial candidemia: risk factors and attributable mortality. Clinical Infectious Diseases. 1995;20:1531–1534. doi: 10.1093/clinids/20.6.1531. [DOI] [PubMed] [Google Scholar]
- 4.Wisplinghoff H, et al. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clinical infectious diseases. 2004;39:309–317. doi: 10.1086/421946. [DOI] [PubMed] [Google Scholar]
- 5.Dadar Maryam, Tiwari Ruchi, Karthik Kumaragurubaran, Chakraborty Sandip, Shahali Youcef, Dhama Kuldeep. Candida albicans - Biology, molecular characterization, pathogenicity, and advances in diagnosis and control – An update. Microbial Pathogenesis. 2018;117:128–138. doi: 10.1016/j.micpath.2018.02.028. [DOI] [PubMed] [Google Scholar]
- 6.Vandecandelaere, I. & Coenye, T. In Biofilm-Based Healthcare-Associated Infections 137–155 (Springer, 2015).
- 7.Williams, C. & Ramage, G. In Biofilm-based healthcare-associated infections 11–27 (Springer, 2015).
- 8.Kojic EM, Darouiche RO. Candida infections of medical devices. Clinical microbiology reviews. 2004;17:255–267. doi: 10.1128/CMR.17.2.255-267.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tumbarello M, et al. Biofilm production by Candida species and inadequate antifungal therapy as predictors of mortality for patients with candidemia. Journal of clinical microbiology. 2007;45:1843–1850. doi: 10.1128/JCM.00131-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Montagna M, et al. Epidemiology of invasive fungal infections in the intensive care unit: results of a multicenter Italian survey (AURORA Project) Infection. 2013;41:645–653. doi: 10.1007/s15010-013-0432-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tang H-J, Liu W-L, Lin H-L, Lai C-C. Epidemiology and prognostic factors of candidemia in cancer patients. PLoS One. 2014;9:e99103. doi: 10.1371/journal.pone.0099103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pfaller MA, et al. Epidemiology and outcomes of invasive candidiasis due to non-albicans species of Candida in 2,496 patients: data from the Prospective Antifungal Therapy (PATH) registry 2004–2008. PLoS One. 2014;9:e101510. doi: 10.1371/journal.pone.0101510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Silva S, et al. Adherence and biofilm formation of non-Candida albicans Candida species. Trends in microbiology. 2011;19:241–247. doi: 10.1016/j.tim.2011.02.003. [DOI] [PubMed] [Google Scholar]
- 14.Silva S, et al. Candida glabrata, Candida parapsilosis and Candida tropicalis: biology, epidemiology, pathogenicity and antifungal resistance. FEMS microbiology reviews. 2012;36:288–305. doi: 10.1111/j.1574-6976.2011.00278.x. [DOI] [PubMed] [Google Scholar]
- 15.Kolter R. Biofilms in lab and nature: a molecular geneticist’s voyage to microbial ecology. Int Microbiol. 2010;13:7. doi: 10.2436/20.1501.01.105. [DOI] [PubMed] [Google Scholar]
- 16.Kolter R, Greenberg EP. Microbial sciences: the superficial life of microbes. Nature. 2006;441:300. doi: 10.1038/441300a. [DOI] [PubMed] [Google Scholar]
- 17.Lopez D., Vlamakis H., Kolter R. Biofilms. Cold Spring Harbor Perspectives in Biology. 2010;2(7):a000398–a000398. doi: 10.1101/cshperspect.a000398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Olsen I. Biofilm-specific antibiotic tolerance and resistance. European Journal of Clinical Microbiology & Infectious Diseases. 2015;34:877–886. doi: 10.1007/s10096-015-2323-z. [DOI] [PubMed] [Google Scholar]
- 19.Douglas LJ. Candida biofilms and their role in infection. Trends in microbiology. 2003;11:30–36. doi: 10.1016/S0966-842X(02)00002-1. [DOI] [PubMed] [Google Scholar]
- 20.Nett J, et al. Putative role of β-1, 3 glucans in Candida albicans biofilm resistance. Antimicrobial agents and chemotherapy. 2007;51:510–520. doi: 10.1128/AAC.01056-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.LaFleur MD, Kumamoto CA, Lewis K. Candida albicans biofilms produce antifungal-tolerant persister cells. Antimicrobial agents and chemotherapy. 2006;50:3839–3846. doi: 10.1128/AAC.00684-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nett JE, Sanchez H, Cain MT, Andes DR. Genetic basis of Candida biofilm resistance due to drug-sequestering matrix glucan. The Journal of infectious diseases. 2010;202:171–175. doi: 10.1086/651200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ramage G, Saville SP, Thomas DP, Lopez-Ribot JL. Candida biofilms: an update. Eukaryotic cell. 2005;4:633–638. doi: 10.1128/EC.4.4.633-638.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nobile CJ, Johnson AD. Candida albicans biofilms and human disease. Annual review of microbiology. 2015;69:71–92. doi: 10.1146/annurev-micro-091014-104330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.O’grady NP, et al. Guidelines for the prevention of intravascular catheter-related infections. American journal of infection control. 2011;39:S1–S34. doi: 10.1016/j.ajic.2011.01.003. [DOI] [PubMed] [Google Scholar]
- 26.Pappas PG, et al. Clinical practice guideline for the management of candidiasis: 2016 update by the Infectious Diseases Society of America. Clinical Infectious Diseases. 2015;62:e1–e50. doi: 10.1093/cid/civ933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.World Health Organization. Antimicrobial resistance: global report on surveillance (World Health Organization, 2014).
- 28.Pfaller MA. Antifungal drug resistance: mechanisms, epidemiology, and consequences for treatment. The American journal of medicine. 2012;125:S3–S13. doi: 10.1016/j.amjmed.2011.11.001. [DOI] [PubMed] [Google Scholar]
- 29.Haney, E. F., Mansour, S. C. & Hancock, R. E. In Antimicrobial Peptides 3–22 (Springer, 2017). [DOI] [PubMed]
- 30.Mansour SC, Pena OM, Hancock RE. Host defense peptides: front-line immunomodulators. Trends in immunology. 2014;35:443–450. doi: 10.1016/j.it.2014.07.004. [DOI] [PubMed] [Google Scholar]
- 31.Pizzo, E., Cafaro, V., Di, A. D. & Notomista, E. Cryptic Antimicrobial Peptides: identification methods and current knowledge of their immunomodulatory properties. Current pharmaceutical design (2018). [DOI] [PubMed]
- 32.Pane K, et al. Antimicrobial potency of cationic antimicrobial peptides can be predicted from their amino acid composition: Application to the detection of “cryptic” antimicrobial peptides. Journal of theoretical biology. 2017;419:254–265. doi: 10.1016/j.jtbi.2017.02.012. [DOI] [PubMed] [Google Scholar]
- 33.Swidergall M, Ernst JF. Interplay between Candida albicans and the antimicrobial peptide armory. Eukaryotic cell. 2014;13:950–957. doi: 10.1128/EC.00093-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chung PY, Khanum R. Antimicrobial peptides as potential anti-biofilm agents against multidrug-resistant bacteria. Journal of Microbiology, Immunology and Infection. 2017;50:405–410. doi: 10.1016/j.jmii.2016.12.005. [DOI] [PubMed] [Google Scholar]
- 35.Bosso A, et al. A new cryptic host defense peptide identified in human 11-hydroxysteroid dehydrogenase-1 β-like: from in silico identification to experimental evidence. Biochimica et Biophysica Acta (BBA)-General Subjects. 2017;1861:2342–2353. doi: 10.1016/j.bbagen.2017.04.009. [DOI] [PubMed] [Google Scholar]
- 36.Gaglione R, et al. Novel human bioactive peptides identified in Apolipoprotein B: Evaluation of their therapeutic potential. Biochemical pharmacology. 2017;130:34–50. doi: 10.1016/j.bcp.2017.01.009. [DOI] [PubMed] [Google Scholar]
- 37.Ordonez SR, Amarullah IH, Wubbolts RW, Veldhuizen EJ, Haagsman HP. Fungicidal mechanisms of cathelicidins LL-37 and CATH-2 revealed by live-cell imaging. Antimicrobial agents and chemotherapy. 2014;58:2240–2248. doi: 10.1128/AAC.01670-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Den Hertog AL, Van Marle J, Bolscher JG, Veerman EC, Arie V. Candidacidal effects of two antimicrobial peptides: histatin 5 causes small membrane defects, but LL-37 causes massive disruption of the cell membrane. Biochemical Journal. 2005;388:689–695. doi: 10.1042/BJ20042099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Contursi P, et al. Structural and functional studies of Stf76 from the Sulfolobus islandicus plasmid–virus pSSVx: a novel peculiar member of the winged helix–turn–helix transcription factor family. Nucleic acids research. 2014;42:5993–6011. doi: 10.1093/nar/gku215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Contursi P, Fusco S, Cannio R, She Q. Molecular biology of fuselloviruses and their satellites. Extremophiles. 2014;18:473–489. doi: 10.1007/s00792-014-0634-0. [DOI] [PubMed] [Google Scholar]
- 41.Contursi P, et al. Transcriptional analysis of the genetic element pSSVx: differential and temporal regulation of gene expression reveals correlation between transcription and replication. Journal of bacteriology. 2007;189:6339–6350. doi: 10.1128/JB.00638-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Contursi P, Cannio R, She Q. Transcription termination in the plasmid/virus hybrid pSSVx from Sulfolobus islandicus. Extremophiles. 2010;14:453–463. doi: 10.1007/s00792-010-0325-4. [DOI] [PubMed] [Google Scholar]
- 43.Contursi P, Fusco S, Limauro D, Fiorentino G. Host and viral transcriptional regulators in Sulfolobus: an overview. Extremophiles. 2013;17:881–895. doi: 10.1007/s00792-013-0586-9. [DOI] [PubMed] [Google Scholar]
- 44.Fusco S, Aulitto M, Bartolucci S, Contursi P. A standardized protocol for the UV induction of Sulfolobus spindle-shaped virus 1. Extremophiles. 2015;19:539–546. doi: 10.1007/s00792-014-0717-y. [DOI] [PubMed] [Google Scholar]
- 45.Fusco S, She Q, Fiorentino G, Bartolucci S, Contursi P. Unravelling the role of the F55 regulator in the transition from lysogeny to UV induction of Sulfolobus spindle-shaped virus 1. Journal of virology. 2015;89:6453–6461. doi: 10.1128/JVI.00363-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Prato, S., et al. Molecular modeling and functional characterization of the monomeric primase–polymerase domain from the Sulfolobus solfataricus plasmid pIT3. 275(17), 4389–4402 (2008). [DOI] [PubMed]
- 47.Notomista E, et al. The identification of a novel Sulfolobus islandicus CAMP-like peptide points to archaeal microorganisms as cell factories for the production of antimicrobial molecules. Microbial cell factories. 2015;14:126. doi: 10.1186/s12934-015-0302-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gaglione R, et al. Insights into the anticancer properties of the first antimicrobial peptide fromArchaea. Biochimica et Biophysica Acta (BBA)-General Subjects. 2017;1861:2155–2164. doi: 10.1016/j.bbagen.2017.06.009. [DOI] [PubMed] [Google Scholar]
- 49.Ruissen A, et al. Effects of histatin 5 and derived peptides on Candida albicans. Biochemical Journal. 2001;356:361. doi: 10.1042/bj3560361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pane K, et al. A new cryptic cationic antimicrobial peptide from human apolipoprotein E with antibacterial activity and immunomodulatory effects on human cells. The FEBS journal. 2016;283:2115–2131. doi: 10.1111/febs.13725. [DOI] [PubMed] [Google Scholar]
- 51.Batoni G, Maisetta G, Lisa Brancatisano F, Esin S, Campa M. Use of antimicrobial peptides against microbial biofilms: advantages and limits. Current medicinal chemistry. 2011;18:256–279. doi: 10.2174/092986711794088399. [DOI] [PubMed] [Google Scholar]
- 52.Onyewu C, Blankenship JR, Del Poeta M, Heitman J. Ergosterol biosynthesis inhibitors become fungicidal when combined with calcineurin inhibitors against Candida albicans, Candida glabrata, and Candida krusei. Antimicrobial agents and chemotherapy. 2003;47:956–964. doi: 10.1128/AAC.47.3.956-964.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mazu TK, Bricker BA, Flores-Rozas H, Ablordeppey SY. The Mechanistic Targets of Antifungal Agents: An Overview. Mini reviews in medicinal chemistry. 2016;16:555–578. doi: 10.2174/1389557516666160118112103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chen SC-A, Slavin MA, Sorrell TC. Echinocandin antifungal drugs in fungal infections. Drugs. 2011;71:11–41. doi: 10.2165/11585270-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 55.Singh K, Rani J. Sequential and Structural Aspects of Antifungal Peptides from Animals, Bacteria and Fungi Based on Bioinformatics Tools. Probiotics and antimicrobial proteins. 2016;8:85–101. doi: 10.1007/s12602-016-9212-3. [DOI] [PubMed] [Google Scholar]
- 56.Joly S, Maze C, McCray PB, Guthmiller JM. Human β-defensins 2 and 3 demonstrate strain-selective activity against oral microorganisms. Journal of clinical microbiology. 2004;42:1024–1029. doi: 10.1128/JCM.42.3.1024-1029.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Brouwer, C. P. J. M. et al. In Cohesive Journal of Microbiology & Infectious Disease, Vol. 1 (2018).
- 58.Singh K, Shekhar S, Yadav Y, Xess I, Dey S. DS6: antiCandidal, antibiofilm peptide against Candida tropicalis and exhibit synergy with commercial drug. Journal of Peptide Science. 2017;23:228–235. doi: 10.1002/psc.2973. [DOI] [PubMed] [Google Scholar]
- 59.Wang K, et al. Antimicrobial peptide protonectin disturbs the membrane integrity and induces ROS production in yeast cells. Biochimica et Biophysica Acta (BBA)-Biomembranes. 2015;1848:2365–2373. doi: 10.1016/j.bbamem.2015.07.008. [DOI] [PubMed] [Google Scholar]
- 60.Lum KY, et al. Activity of novel synthetic peptides against Candida albicans. Scientific reports. 2015;5:9657. doi: 10.1038/srep09657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Helmerhorst EJ, Venuleo C, Beri A, Oppenheim FG. Candida glabrata is unusual with respect to its resistance to cationic antifungal proteins. Yeast. 2005;22:705–714. doi: 10.1002/yea.1241. [DOI] [PubMed] [Google Scholar]
- 62.De Groot PW, et al. The cell wall of the human pathogen Candida glabrata: differential incorporation of novel adhesin-like wall proteins. Eukaryotic cell. 2008;7:1951–1964. doi: 10.1128/EC.00284-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Pfaller M, et al. Candida krusei, a multidrug-resistant opportunistic fungal pathogen: geographic and temporal trends from the ARTEMIS DISK Antifungal Surveillance Program, 2001 to 2005. Journal of Clinical Microbiology. 2008;46:515–521. doi: 10.1128/JCM.01915-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Panizo MM, Reviákina V, Dolande M, Selgrad S. Candida spp. in vitro susceptibility profile to four antifungal agents. Resistance surveillance study in Venezuelan strains. Medical mycology. 2009;47:137–143. doi: 10.1080/13693780802144339. [DOI] [PubMed] [Google Scholar]
- 65.Pelletier R, Alarie I, Lagacé R, Walsh TJ. Emergence of disseminated candidiasis caused by Candida krusei during treatment with caspofungin: case report and review of literature. Medical mycology. 2005;43:559–564. doi: 10.1080/13693780500220415. [DOI] [PubMed] [Google Scholar]
- 66.Ramage G, Wickes B, Lopez-Ribot J. Biofilms of Candida albicans and their associated resistance to antifungal agents. American clinical laboratory. 2001;20:42. [PubMed] [Google Scholar]
- 67.Fonseca E, et al. Effects of fluconazole on Candida glabrata biofilms and its relationship with ABC transporter gene expression. Biofouling. 2014;30:447–457. doi: 10.1080/08927014.2014.886108. [DOI] [PubMed] [Google Scholar]
- 68.Fernandes T, Silva S, Henriques M. Candida tropicalis biofilm’s matrix—involvement on its resistance to amphotericin B. Diagnostic microbiology and infectious disease. 2015;83:165–169. doi: 10.1016/j.diagmicrobio.2015.06.015. [DOI] [PubMed] [Google Scholar]
- 69.Mathé L, Van Dijck P. Recent insights into Candida albicans biofilm resistance mechanisms. Current genetics. 2013;59:251–264. doi: 10.1007/s00294-013-0400-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Khot PD, Suci PA, Miller RL, Nelson RD, Tyler BJ. A small subpopulation of blastospores in Candida albicans biofilms exhibit resistance to amphotericin B associated with differential regulation of ergosterol and β-1, 6-glucan pathway genes. Antimicrobial agents and chemotherapy. 2006;50:3708–3716. doi: 10.1128/AAC.00997-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kuhn D, George T, Chandra J, Mukherjee P, Ghannoum M. Antifungal susceptibility of Candida biofilms: unique efficacy of amphotericin B lipid formulations and echinocandins. Antimicrobial agents and chemotherapy. 2002;46:1773–1780. doi: 10.1128/AAC.46.6.1773-1780.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Jain N, et al. Biofilm formation by and antifungal susceptibility of Candida isolates from urine. Applied and environmental microbiology. 2007;73:1697–1703. doi: 10.1128/AEM.02439-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Prażyńska M, Bogiel T, Gospodarek-Komkowska E. In vitro activity of micafungin against biofilms of Candida albicans, Candida glabrata, and Candida parapsilosis at different stages of maturation. Folia microbiologica. 2018;63:209–216. doi: 10.1007/s12223-017-0555-2. [DOI] [PubMed] [Google Scholar]
- 74.Al-Fattani MA, Douglas LJ. Biofilm matrix of Candida albicans and Candida tropicalis: chemical composition and role in drug resistance. Journal of medical microbiology. 2006;55:999–1008. doi: 10.1099/jmm.0.46569-0. [DOI] [PubMed] [Google Scholar]
- 75.Orozco, A.S. et al. Mechanism of Fluconazole Resistance in Candida krusei. 42, 2645–2649 (1998). [DOI] [PMC free article] [PubMed]
- 76.Wayne P. National committee for clinical laboratory standards. Performance standards for antimicrobial disc susceptibility testing. 2002;12:01–53. [Google Scholar]
- 77.Stepanović S, et al. Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. Apmis. 2007;115:891–899. doi: 10.1111/j.1600-0463.2007.apm_630.x. [DOI] [PubMed] [Google Scholar]
- 78.Pulcrano G, Panellis D, De Domenico G, Rossano F. & Catania, M.R. Ambroxol influences voriconazole resistance of Candida parapsilosis biofilm. FEMS yeast research. 2012;12:430–438. doi: 10.1111/j.1567-1364.2012.00792.x. [DOI] [PubMed] [Google Scholar]
- 79.Taff HT, Nett JE, Andes DR. Comparative analysis of Candida biofilm quantitation assays. Sabouraudia. 2012;50:214–218. doi: 10.3109/13693786.2011.580016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Barra F, et al. Photodynamic and antibiotic therapy in combination to fight biofilms and resistant surface bacterial infections. International journal of molecular sciences. 2015;16:20417–20430. doi: 10.3390/ijms160920417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Khan S, Alam F, Azam A, Khan AU. Gold nanoparticles enhance methylene blue–induced photodynamic therapy: a novel therapeutic approach to inhibit Candida albicans biofilm. International journal of nanomedicine. 2012;7:3245. doi: 10.2147/IJN.S31219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Tsai P-W, Yang C-Y, Chang H-T, Lan C-Y. Human antimicrobial peptide LL-37 inhibits adhesion of Candida albicans by interacting with yeast cell-wall carbohydrates. PloS one. 2011;6:e17755. doi: 10.1371/journal.pone.0017755. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding authors on reasonable request.