We congratulate Abou-El-Ardat and colleagues,1 who studied a series of multifocal isocitrate dehydrogenase (IDH) wild-type glioblastomas (GBMs) from 6 patients and proved their monoclonal origin. On the contrary, we have come across a case of a synchronous occurrence of a high-grade and a low-grade glioma (LGG) with a discordant IDH mutational status, suggesting an independent origin.
A 48-year-old female patient with no previous history of cancer was admitted to our institution because of 2 synchronous lesions diagnosed as a glioblastoma in the left temporal lobe and an LGG in the right frontal lobe. The enhancing temporal lesion was surgically removed and the histological and molecular examination by immunohistochemistry and IDH1/2 sequencing demonstrated an IDH wild-type glioblastoma. The patient started standard treatment with radiotherapy and temozolomide. After 1 year, due to stable disease, a second surgery for the non-enhancing lesion in the left frontal lobe was performed. The histological diagnosis was of an IDH1-mutant diffuse astrocytoma (p.R132H; the mutational profiling of both cases were performed by immunohistochemistry and Sanger sequencing). The tumor showed neither loss of nuclear expression of alpha thalassemia/mental retardation syndrome X-linked protein nor 1p/19q codeletion. Methylation of O6-methylguanine-DNA methyltransferase was detected by pyrosequencing in both lesions. Fluorescence in situ hybridization (FISH) analyses showed monosomy of chromosome 10 and homozygous deletion of 9p21 (cyclin-dependent kinase inhibitor 2A gene [CDKN2A]) only in the glioblastoma; neither gain of chromosome 7 nor amplification of the epidermal growth factor receptor gene region was identified in the tumors. Monosomy of chromosome 10 may be acquired late in the evolution of a subgroup of LGGs, but gain of chromosome 7 and loss of chromosome 10 are considered early molecular events in most IDH wild-type glioblastomas (non‒glioma cytosine-phosphate-guanine island methylator phenotype glioblastoma), and even though they coexist in most cases, it is known that there are rare cases of IDH wild-type glioblastoma in which one or the other is lacking and in which the pathways affected by the abnormality that is lacking, either 7 gain or 10 loss, are impaired by alterations of other chromosomes.2,3
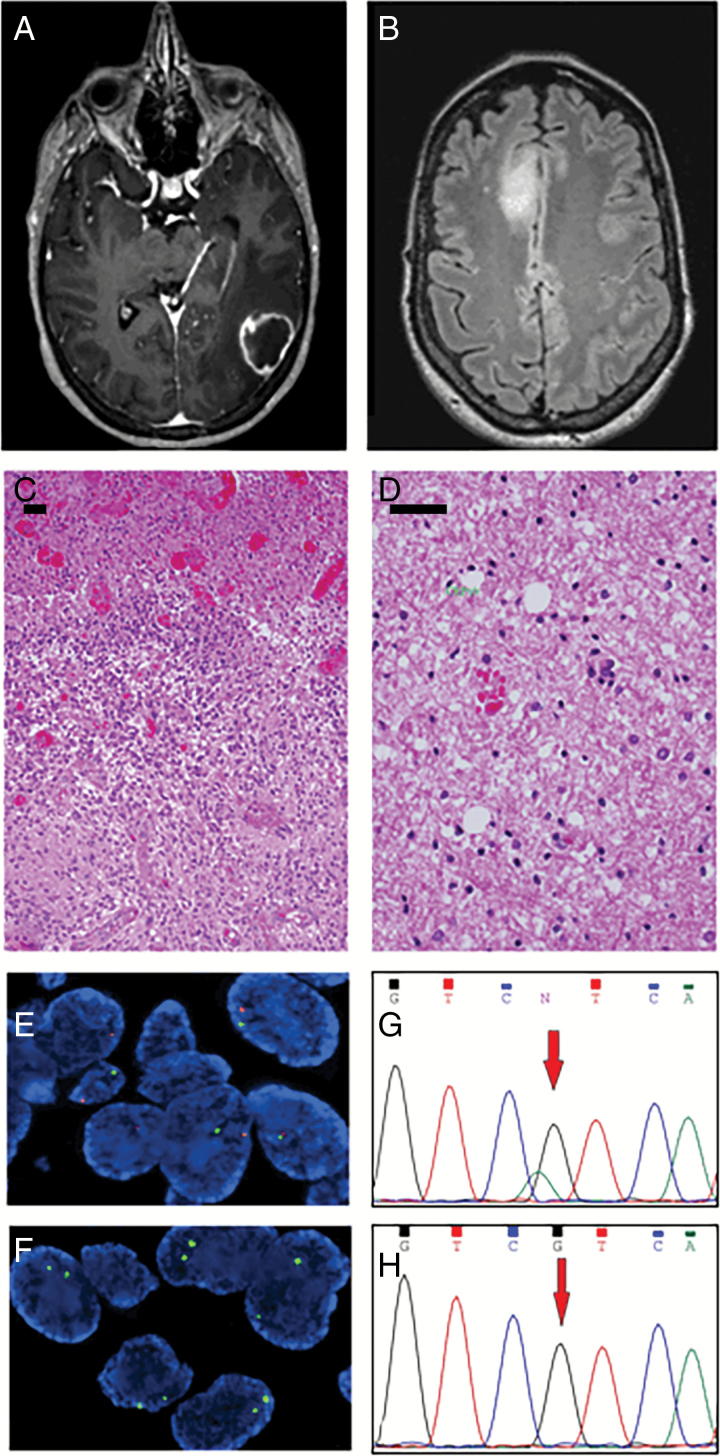
Fig. 1.
(A, C) Radiological and pathological characteristics of the IDH wild-type GBM and (B, D) IDH-mutant diffuse astrocytoma. Scale bars (C, D) = 100 μm. (E) FISH analyses showing monosomy of chromosome 10 (Vysis LSI phosphatase and tensin homolog in orange and chromosome 10 enumeration probe [CEP10] in green) and (F) homozygous deletion of 9p21 (CDKN2A) (Vysis CDKN2A in orange and CEP9 in green) in the IDH wild-type GBM. (G) Sanger sequencing of IDH-mutant diffuse astrocytoma and (H) IDH wild-type GBM sample.
Both the lesions carried telomerase reverse transcriptase (TERT) promoter mutation (228C>T). TERT promoter mutation can rarely be associated with IDH mutation in cases of LGGs which do not carry 1p/19q codeletion.4 The timing of TERT mutation is still debated, but some studies suggest that it may precede 7 gain/10 loss, 1p/19q codeletion, and even IDH mutation.3 In this perspective, it is tempting to speculate that, in our case, the 2 lesions could have stemmed from a common TERT-mutated precursor. However, considering the overall high frequency of TERT promoter mutation in gliomas, it may be argued that its occurrence in both lesions could be merely coincidental.
Expression of DNA mismatch repair (MMR) proteins was investigated using immunohistochemistry for mutS homolog (MSH)2, mutL homolog 1, MSH6, and postmeiotic segregation increased 2 proteins and both lesions resulted MMR proficient. Microsatellite instability was also investigated by fluorescent PCR and fragment analysis of BAT-25, BAT-26, NR-21, NR-24, and NR-27 mononucleotide markers on both tumors showing microsatellite stability in agreement with immunohistochemistry analysis.
A similar case with 2 different genetic pathways involved in the development of 2 distinct malignant gliomas was recently described for a pediatric patient with constitutional MMR deficiency syndrome having IDH wild-type glioblastoma and IDH1-mutant anaplastic astrocytoma.5
On the contrary, we described the first case of an adult patient without MMR deficiency showing the simultaneous presence of 2 histologically different glial tumors. Moreover, the different site of onset and the discordant status of IDH1 suggest that the 2 tumor lesions may not be linked to each other and may be characterized by a different molecular landscape. Hence, we consider that in the case of synchronous glial tumors, especially those with different radiological characteristics, molecular characterization of each lesion is warranted.
Funding
The authors declare no conflict of interest and no funding received.
References
- 1. Abou-El-Ardat K, Seifert M, Becker K et al. Comprehensive molecular characterization of multifocal glioblastoma proves its monoclonal origin and reveals novel insights into clonal evolution and heterogeneity of glioblastomas. Neuro Oncol. 2017;19(4):546–557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Ozawa T, Riester M, Cheng YK. et al. Most human non-GCIMP glioblastoma subtypes evolve from a common proneural-like precursor glioma. Cancer Cell. 2014;26(2):288–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Barthel FP, Wesseling P, Verhaak RGW. Reconstructing the molecular life history of gliomas. Acta Neuropathol. 2018;135(5):649–670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Eckel-Passow JE, Lachance DH, Molinaro AM et al. Glioma groups based on 1p/19q, IDH, and TERT promoter mutations in tumors. N Engl J Med. 2015;372(26):2499–2508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Galuppini F, Opocher E, Tabori U et al. Concomitant IDH wild-type glioblastoma and IDH1-mutant anaplastic astrocytoma in a patient with constitutional mismatch repair deficiency syndrome. Neuropathol Appl Neurobiol. 2018;44(2):233–239. [DOI] [PubMed] [Google Scholar]