Abstract
The heightened risk of persons with serious mental illness to contract and transmit human immunodeficiency virus (HIV) is a public health problem. Here we evaluate the interaction between psychiatric diagnosis and response to a community based-intervention targeted at treatment adherence in 236 HIV+ persons with co-occurring mental illness. To examine differential effectiveness of the intervention for categories of patient diagnosis, we reanalyzed the data after stratifying participants into two diagnostic groups: (1) participants with depressive disorders without psychosis and (2) participants with a psychotic or bipolar disorder. Outcomes included viral load and mental health quality of life (SF-12 Mental Health). We found that HIV+ persons with non-psychotic depressive disorders demonstrated a larger decrease in HIV viral load and more improvement in measures of mental health quality of life when compared to HIV+ persons with psychotic and bipolar disorders. We suggest that successful adherence interventions should be informed by psychiatric symptomatology.
Keywords: HIV, Treatment adherence, Root cause analysis, PATH+, Psychiatric diagnosis
Introduction
The co-occurrence of HIV and mental illness has been investigated since the mid-1990s. Early studies using a convenience sample of patients with serious mental illness (SMI) yielded estimates of HIV prevalence ranging from 1 to 23% [1, 2]. A second generation of prevalence data used administrative data sources, such as state HIV or AIDS registry files and found 5.7% with a diagnosis of schizophrenia [3] versus the general population prevalence rate for schizophrenia of approximately 1% [4]. Using Medicaid Claims, after controlling for a range of socio-demographic variables, Blank et al. [5], found schizophrenia spectrum disorder patients were 1.5 times as likely to have claims indicative of HIV infection and those with a major affective disorder were 3.8 times as likely to have HIV. Administrative medical records from a national sample from the Veterans Administration also indicate elevated rates of HIV in persons with serious mental illness [6].
Estimates based on representative samples are rare due to shortcomings in existing detection systems, although a recent study using the National Epidemiologic Survey on Alcohol and Related Conditions reported that, compared to their HIV-negative counterparts, men with HIV were significantly more likely to merit a range of diagnoses, including major depressive disorder/dysthymia, as well as personality disorders [7]. A 2014 study by Blank et al. [8] reported a four-fold increase in HIV infections among persons receiving mental health care in a variety of treatment settings in Philadelphia and Baltimore with 5.9% for inpatient units, 5.1% for intensive case management (ICM) programs, and 4.0% for community mental health centers with confirmed positive HIV test results.
Contrary to what might be concluded from the elevated rates of HIV infection, in the presence of good quality health care and appropriate supports, people with mental illness and HIV can have adherence rates comparable to those without mental illness, and some have found that they are less likely to discontinue anti-retroviral therapy (ART) [6, 8–10].
Poor adherence to HIV/AIDS treatment has been linked to co-morbid depression [11, 12]. However, there is convincing evidence that treatment of depression and adherence to antidepressant medication tend to improve adherence to HIV treatment and laboratory parameters of HIV disease [13, 14].
The data on the relationship between adherence to ART and comorbid bipolar disorder is more sparse and shows less consensus, with some studies demonstrating a correlation between lower adherence to ART and diagnosis of bipolar disorder [15, 16], and other studies showing no significant associations [9, 17, 18]. In addition, most studies examining the relationship between adherence to ART and comorbid schizophrenia/psychotic disorders also found no significant association [9, 16–18].
A fairly consistent finding in the literature is that adherence to psychiatric medications in individuals with a variety of psychiatric diagnoses, including depressive, bipolar and psychotic/schizophrenia spectrum disorders, is correlated to adherence to ART [9, 15, 17, 18]. There are, however, no studies that compare the effectiveness of interventions for enhancing adherence to ART between different psychiatric diagnoses or diagnostic categories. Identifying diagnostic groups that respond especially well or especially poorly to an intervention is important for intervention modification purposes and for highlighting cost-effectiveness differentials conditional on patient diagnosis [19–21].
In response to this gap in knowledge, the present study utilized data from PATH+ (Preventing AIDS Through Health for HIV+ persons) [8], an HIV treatment regimen management study characterized by elements that directly correspond to the needs of individuals with comorbid HIV and SMI, targeted at treatment adherence. The original analysis of the effects of PATH+, a nurse-led communitybased intervention to promote medication and treatment regimen management, demonstrated improvement in both biomarker and mental health-related quality of life indicators in persons with HIV/SMI. The intervention group exhibited greater reductions in mean log viral load and significant improvements in quality of life measures (i.e., SF-12 mental and physical health summary scores) as compared to an enhanced treatment-as-usual control group (eTAU) [8].
In this paper we take a different perspective on estimating the intervention effects by posing the question: did the outcomes interact with different psychiatric diagnoses of PATH+ participants? Identifying diagnostic groups that respond especially well or especially poorly to an intervention is important for intervention modification purposes and for highlighting cost-effectiveness differentials conditional on participant diagnosis [19–21]. Toward that end, the current study examined and compared participants diagnosed with depression without psychotic features with participants with a diagnosis of bipolar and/or psychotic disorders to examine a potential interaction between psychiatric diagnosis and the effectiveness of a treatment adherence intervention to reduce HIV viral load and improve quality of life. Psychosis is the cardinal feature in schizophrenia and schizoaffective disorders, but is also found in people with primary diagnoses of bipolar and major depressive disorders, and is often associated with more severe impairment. We expected better outcomes and greater improvement in adherence to ART in individuals with non-psychotic unipolar depression as compared to individuals with bipolar/psychotic disorders.
Methods and Measures
These data were collected between September 2004 and April 2009 using data from the parent study, PATH+ [8]. As described elsewhere [22, 23], the PATH+ intervention was a longitudinal randomized clinical trial using an adaptive treatment design [24] implemented through a “treatment cascade” [8, 22] to moderate the intensity and expense of the intervention to actual psychiatric and HIV adherence outcomes. The cascade was triggered by adherence rates to both antiretroviral treatment (ART) and psychiatric medications. When adherence fell below 80%, the intervention cascade was escalated until adherence was maintained equal to or above 80% for three weeks. Combined adherence to HIV and psychiatric medications was calculated weekly. Adherence was assessed by self-report and verified by pill count for both ART and psychiatric medication. If either of those criteria fell below an 80% threshold the next step in the intervention cascade was implemented. The cascade represents an increase in intensity of interventions including activation of social networks, the use of reminder beepers with alphanumeric displays in real time, and the cellular phone calls to encourage participants to follow their regimen, with the most intensive intervention level being directly observed therapy (DOT).
The PATH+ intervention group received assignment to a Nurse Health Navigator (NHN), who provided intervention services including in-home services and psychoeducation, as well as coordinated care with other service providers including infectious disease clinicians, mental health service providers (e.g., psychiatrists and case managers), substance abuse counsellors, primary care physicians, dentists, and various other providers of health services that PATH+ recipients encountered in the treatment of their co-occurring disorders. eTAU/control participants received treatment-as-usual enhanced by providing biomarker results (viral load and CD4) to treating physicians at each assessment. Oversight for the protection of human subjects was provided by the University of Pennsylvania Institutional Review Board as well as from the City of Philadelphia Department of Public Health.
Participants were recruited from HIV treatment sites throughout the city of Philadelphia. Inclusion criteria for the PATH+ study required that the participant be age 18 or older, be able to understand spoken English, have a diagnosed SMI, demonstrate sufficient competency to provide informed consent, and be HIV seropositive. These criteria were later confirmed by HIV testing and by contacting their mental health service providers. Any participant who was not currently receiving treatment for HIV was referred to the outpatient clinic at the Hospital of the University of Pennsylvania. All participants were paid $40 for each of four interviews over the 12-month study period, as well as one 24-month follow-up. A bonus of $100 was paid to participants who provided data at all study time points to incentivize provision of complete data. Eligible consenting patient participants were randomized on a 1:1 basis to the intervention and control groups. Randomization was employed to ensure that approximately equal numbers of patients were assigned to each of the two groups that were balanced with respect to observed and unmeasured baseline factors. Experimental status was blinded so that the study investigators and research staff did not know the treatment status of participants, however participants sometimes disclosed information which made it possible to identify experimental status.
Outcome Measures
Three outcome measures were collected. First, mental health quality of life was evaluated using the mental health score of the SF-12 [25]. Six items produce a mental health score normed to an average of 50. SF-12 has has been validated for use in populations with SMI, and provides a reliable and valid measurement of quality of life in samples with diagnoses across diagnostic categories that include bipolar/psychotic and depressive disorders [25–27].
Standard HIV testing yielding viral load assays and CD4 counts were provided from the clinical laboratories at the Hospital of the University of Pennsylvania. Because of the expense, HIV viral load and CD4 were measured only at baseline, 12 and 24 months. SF12 data was available at five time points (baseline, 3, 6, 12, and 24 months).
Analysis Plan
In this paper, the focus is on log HIV/SF-12 mental health quality of life data since this outcome pair showed the largest effects in the original analysis [8] (see Table 2, Model A). We first describe the demographic characteristics of the sample by diagnostic category in Table 1. For the PATH+ intervention, we compared treatment effect for participants with a primary diagnosis of non-psychotic unipolar depression, which is the largest single diagnostic category, with participants diagnosed with bipolar and/or psychotic disorder.
Table 2.
PATH+ and eTAU treatment slopes pooling both diagnostic groups
| Biomarker outcome | |
| Log HIV equation: eTAU participants α# = 2.85* β = + .053 (time metric) |
Log HIV equation: Path+ participants α# = 2.85* β = −.307*(time metric) |
| SF-12 outcome | |
| SF-12 mental health: eTAU participants α# = 40.77* β = + .28 (time metric) |
SF-12 mental health: Path+ participants α# = 40.77* β = .638*(time metric) |
Indicates intercepts fixed to equality for both experimental groups
Indicates significant at the .05 level or less, two tailed test. Goodness of fit: χ2 = 62.25, df = 58, p = .24, CFI = .98, RMSEA = .033 (CI 00-.068). N = 236
Table 1.
Characteristics of Path+ participants at baseline by diagnostic group and experimental status
| Depression* |
Bipolar/psychotic* |
|||
|---|---|---|---|---|
| PATH+ (n = 64) | eTAU (n = 48) | PATH+ (n = 63) | eTAU (n = 61) | |
| Percent male | 52 | 48 | 54 | 56 |
| Percent African American | 86 | 81 | 78 | 77 |
| Age | 42.18 [41.1 44.6] | 42.58 [40.5 44.6] | 42.71 [41.1 44.3] | 41.6 [39.4 43.8] |
| Log HIV | 2.84 [2.5 3.1] | 2.74 [2.4 3.1] | 2.87 [2.6 3.2] | 2.93 [2.6 3.2] |
| SF12: Mental | 38.19 [36.5 39.9] | 39.59 [37.5 41.7] | 41.19 [38.9 43.4] | 42.34 [40.2 44.5] |
95% confidence intervals in brackets
No significant difference between Path+ and TAU Group on all outcomes at baseline for this diagnostic group
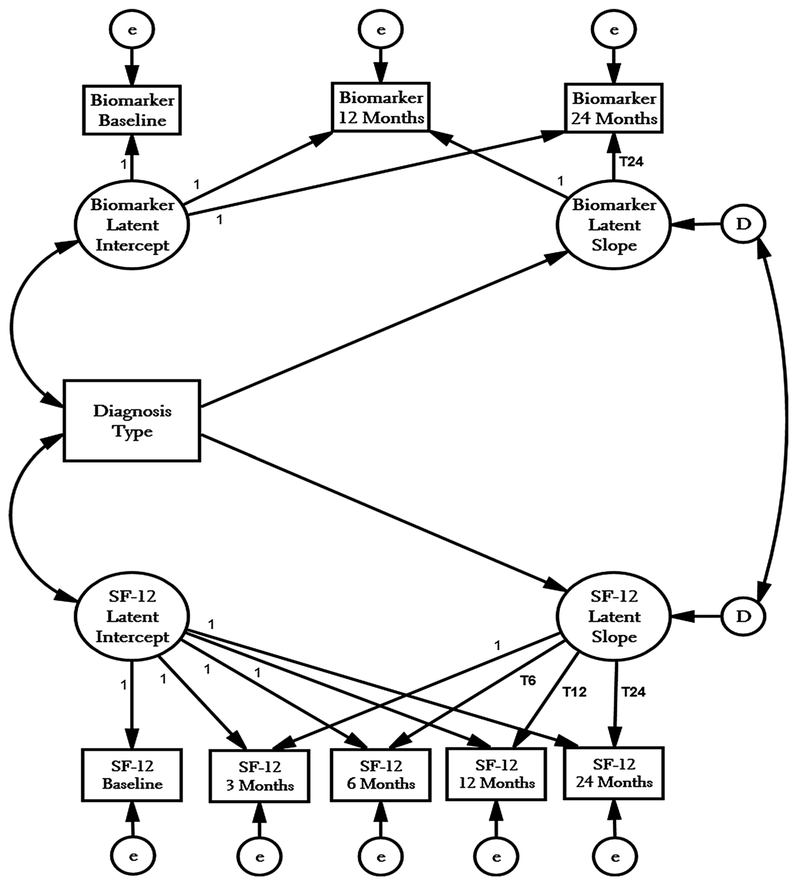
Next, we estimate the model in Fig. 1 without including the diagnostic groups to establish the pooled treatment effects, ignoring the diagnosis of the participant. To investigate subgroup differences based on diagnostic groups, we estimate two nested models stratified by PATH+ and eTAU status. The first stratified model differentiates between participants in the two diagnostic groups but constrains the slopes of the change over time to be the same for both the PATH+ and eTAU participants: this is the model of equivalent differences based on diagnostic condition in the PATH+ and eTAU experimental groups. The second stratified analysis permits the effects of the intervention to vary over both experimental and diagnostic groups to explore differences between persons with nonpsychotic unipolar depression as compared to those with bipolar and/or psychotic disorders.
Fig. 1.
Growth Curve Models for Simultaneous Analysis of HIV Bio-markers and SF-12 Mental Health Score. Constructed from equations in Table 4
Statistical Analyses Using Growth Curves
We used parallel process latent growth curve modeling because it is a data analysis strategy [28, 29] designed to detect changes in multiple outcomes over different time intervals [30, 31]. This is especially relevant here because we want to estimate treatment effect on the social and biomarker outcomes simultaneously. Growth curves typically have two different forms: unconditional and conditional. The unconditional equation predicts an individual respondent’s values of the social and biomarker outcomes as a function of [1] the initial value of outcome at the beginning of the intervention and [2] the slope of change over time. The unconditional growth model is:
| [1] |
The “i” subscript reflects individual observations, the Time Metric is the time scale, the ηi0 is the value of the latent intercept when the Time Metric is zero, the ηi1 is the regression coefficient indicating the latent slope of time for each individual, and the “t” subscript represents the ordering of the observations. Thus, the error term defines the individual (e.g., “within subject”) measurement errors of the outcomes for each observation. Because the latent parameters vary across individuals, the parameters can be treated as dependent variables in auxiliary equations that predict the initial value and slope of the outcome: the “conditional growth model” [32, 33]. Here we use the major diagnosis of the PATH+ participants as the conditional predictor of the slope and stratify the data into PATH+ and eTAU groups identify the treatment*diagnostic type interaction if present.
Estimating the Best Fitting Time Metric
For viral load in the eTAU group, the time metric is 0, 1 and 3.99. The change in viral load in the eTAU group from baseline (0) increases to 12 months (always defined as 1 standard deviation) and then accelerates to 3.99 (almost 4 times the change from baseline to 12 months) at 24 months indicating increased viremia. In the treatment group, the time metric is 0, 1 and 1.05 indicating almost no increase in viremia from 12 to 24 months. For the mental health summary score in the eTAU group, the best fitting time metric is 0, 1, .91, 1.07, and 3.53 and in the treatment group it is 0, 1, 2.53, 3.65, and 3.79. By way of illustration, taking this last metric for the mental health summary score in the PATH+ group as an example, the change from baseline to month 6 is 2.53 times as large as the change from baseline to month 3, the change from baseline to month 12 is 3.65 times as large as the change from baseline to month 3, and the change from baseline to month 24 is 3.79 times as large as the change from baseline to month 3.
Results
Participant characteristics and outcome measures by experimental status and diagnostic group at baseline are described in Table 1. PATH+ enrolled 236 HIV+ participants with SMI which yielded a total of 112 participants in unipolar depression group, and 124 participants in bipolar/ psychosis group. Sixty-four participants in the unipolar depression group had been randomized to the PATH+ intervention, and in this subgroup 52% were male, 86% were African American, and an average age of the participant was 42.18. Forty-eight participants in the unipolar depression group had been randomized to the eTAU, with 48% male, 81% African American, and an average age of 42.58. Sixty-three participants in the bipolar/psychosis group had been randomized the PATH+ intervention, with 54% male, 78% African American, and the average age of 42.71. Sixty-one participants in the bipolar/ psychosis group had been randomized to the eTAU, with 56% male, 77% African American, and an average age of 41.6.
Our initial analysis was performed by looking at three subgroups: depressive disorders without psychosis, bipolar disorder without psychosis, and psychotic disorders (schizophrenia, schizoaffective disorder, depression with psychosis, bipolar with psychosis, unspecified psychosis). Each of the last two groups did not add up to a sufficient enough number of subjects and after noting remarkably similar trends/results in the last two subgroups, we combined them into one subgroup (bipolar/psychotic) that could be compared to the first subgroup. Psychiatric diagnoses were obtained from chart reviews of enrolled participants using the last psychiatric diagnosis as the diagnosis of record. People with any bipolar diagnosis, such as bipolar type I, with or without psychotic features, bipolar type II, or any diagnosis that included psychosis, such as depression with psychotic features, schizophrenia, schizoaffective disorder, were all included in the bipolar/ psychotic disorders group. There were no statistically significant differences at baseline between the intervention and eTAU group in either diagnostic grouping on any of these demographic characteristics or study outcomes.
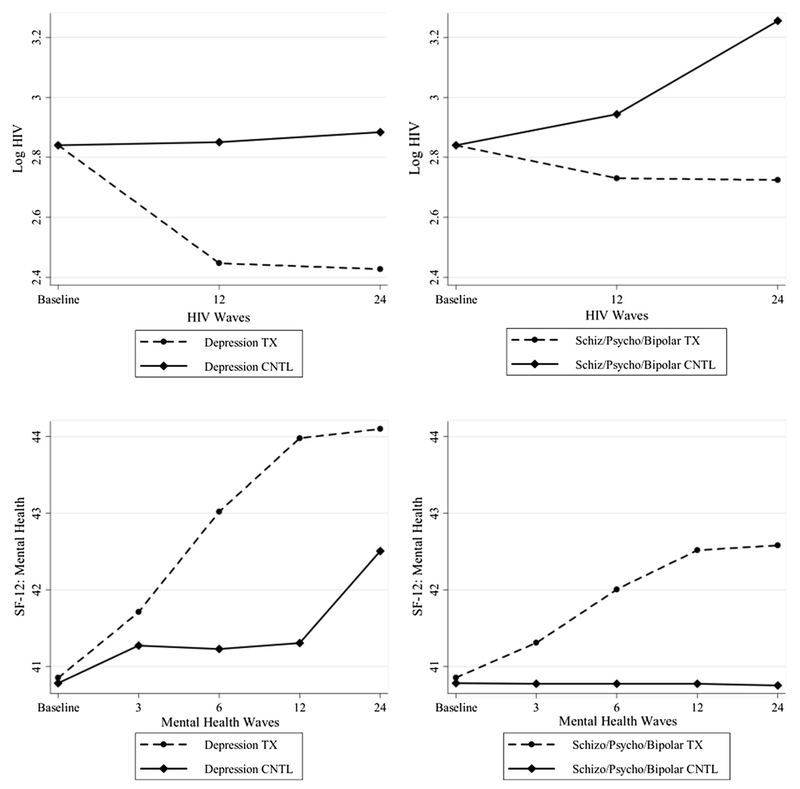
Figure 1 presents the generic analysis model. Note that the focus of the prediction is on the slopes of the two outcomes and we estimate the model for the PATH+ group and the eTAU group at the same time. Table 2 shows the outcome equations estimated for the eTAU and the PATH+ groups for all respondents. As would be expected, the eTAU group does not show a discernibly non-zero slope for either the HIV or the SF-12 outcomes, which indicates no appreciable change over time. In contrast, both the slopes changes in the PATH+ group are significant: log HIV viral load declines with time and mental health summary increases with time. This is the pattern expected and is consistent with the original results cited earlier.
Table 3 differentiates between participants in the two diagnostic groups but constrains all slopes of change over time to be the same for the PATH+ and eTAU participants: this is the test of equivalent differences based on diagnostic condition in both the PATH+ and eTAU experimental groups. HIV viral load increases significantly in the psychosis/bipolar group in both PATH+ and eTAU groups, while HIV viral load in the depressed group significantly declines. For SF-12 mental health score, all groups and conditions show a significant increase with the depressed group showing larger changes than the psychotic/bipolar group.
Table 3.
Common diagnostic group slopes in PATH+ and eTAU groups
| Biomarker outcome | |
| Log HIV equation: eTAU participants Bipolar/psychotic α# = 2.81* β = + . 088*(time metric) |
Log HIV equation: Path+ Participants Bipolar/psychotic α# = 2.81* β = + .088*(time metric) |
| Depression α# = 2.81* β = −.041(time metric) |
Depression α# = 2.81* β = −.041(time metric) |
| Change in slope due to depression: −.129* | Change in slope due to depression: −.129* |
| SF-12 outcome | |
| SF-12 mental health: eTAU participants Bipolar/psychotic α# = 40.72* β = + .366ott(time metric) |
SF-12 mental health: Path+ Participants Bipolar/psychotic α# = 40.72* β = .366ott(time metric) |
| Depression α# = 40.72* β = + .842(time metric) |
Depression α# = 40.72* β = + .842(time metric) |
| Change in slope due to depression: .476ott | Change in slope due to depression: .476ott |
Indicates intercepts fixed to equality for both experimental groups
Indicates significant at the.05 level or less, two tailed test. ott indicates one tailed test. Goodness of fit: χ2 = 140.86, df = 83, p < .01, CFI .82 , RMSEA = .077 (CI 054–.098)
Table 4 shows the results for the model that permits diagnostic group slope differences between the PATH+ and eTAU experimental groups. In the eTAU group, Log HIV significantly increases for the schizophrenia, psychosis, or bipolar participants and this increase is counter-balanced in the depressed group to bring the combined slope close to zero. The SF-12 mental health score shows no change for the schizophrenia, psychosis, or bipolar participants group and is positive for the depressed participants. In the PATH+ group, all the slopes of HIV viral load are negative and all the slopes of mental health change are positive (the SF-12 mental health slopes are statistically significant from zero). SF-12 improves for both diagnostic groups, with the unipolar depression group showing larger improvement. To better display the effects of each model, Fig. 2 plots the predicted outcomes by experimental status and diagnostic groups based on the results in Table 4. Because the results of Tables 3 and 4 are nested, we can compute the χ2 difference test [34] comparing the models with restricted versus unconstrained slopes between the diagnostic groups. The unconstrained model is a better fit, the test between Tables 4 and 3 is statistically significant: the χ2 difference is 13.87, df = 4, p < .05 (the upper 5% probability cut off for a χ2 with 4 df is 9.49 or higher).
Table 4.
Distinct diagnostic group slopes in PATH+ and eTAU groups
| Biomarker outcome | |
| Log HIV equation: eTAU participants Bipolar/psychotic α# = 2.84* β = + .104*(time metric) |
Log HIV equation: Path+ Participants Bipolar/psychotic α# = 2.84* β = − .110(time metric) |
| Depression α# = 2.84* β = + .011(time metric) |
Depression α# = 2.84* β = −.393(time metric) |
| Change in slope due to depression:−.093 | Change in slope due to depression: −.283 |
| SF-12 outcome | |
| SF-12 mental health: eTAU participants Bipolar/psychotic α# = 40.78* β = −.009(time metric) |
SF-12 mental health: Path+ Participants Bipolar/psychotic α# = 40.78* β = + .456ott(time metric) |
| Depression α# = 40.78* β = + .489(time metric) |
Depression α# = 40.78* β = + .857(time metric) |
| Change in slope due to depression: +.498 | Change in slope due to depression: +.401 |
Indicates intercepts fixed to equality for both experimental groups
Indicates significant at the .05 level or less, two tailed test. ott indicates one tailed test. Goodness of fit: χ2 = 126.99, df = 79, p < .01, CFI = .82, RMSEA = .072 (CI 048–.094). χ2 difference test between this model and the equal effects model of Table 3 is statistically significant: χ2 difference is 13.87, df = 4, p < .05 (5% area for a χ2 with 4 df is 9.49 or higher)
Fig. 2.
Growth Curves for Participant Subgroups and Experimental Status
Discussion
Subgroup analysis of PATH+ suggests that the effects of an eTAU and community-based adherence intervention for people with HIV/SMI (PATH+) are influenced by psychiatric diagnosis, with bipolar/psychotic disorders associated with poorer response to treatment in terms of both HIV viral load and mental health quality of life when compared to individuals with non-psychotic unipolar depression. Consistent with our hypothesis, viral load outcomes varied by diagnosis. For example, compared to individuals who are HIV+ with non-psychotic unipolar depression, those in the eTAU group, HIV+ with bipolar/ psychotic disorders displayed a greater increase in HIV viral load over the course of the study. In PATH+, a smaller decrease in HIV viral load was observed, but a diagnosis of bipolar or psychotic disorder still resulted in viral load values that were greater than those observed for the non-psychotic unipolar depression PATH+ subgroup. For SF-12 mental health quality of life, which is a valid measure across the spectrum of psychiatric diagnoses included in SMI category, lower scores were associated with bipolar/psychotic diagnosis compared to non-psychotic unipolar depression. For both PATH+ diagnostic subgroups, (non-psychotic unipolar depression and bipolar/ psychotic diagnoses), the PATH+ intervention demonstrated improved outcomes but that improvement was significantly greater for those with a diagnosis of non-psychotic unipolar depression. In sum, our findings demonstrate that assignment to the PATH+ experimental group resulted in lower viral loads and improved mental health quality of life across diagnostic subgroups compared to the eTAU. Yet even with the additional resources provided by the NHN, PATH+ individuals with a diagnosis of bipolar/psychotic displayed a poorer response to treatment than did PATH+ individuals with a diagnosis of non-psychotic unipolar depression. These findings suggest that among people with HIV and serious mental illness, a diagnosis of bipolar/psychotic disorder is a poorer prognostic factor for both mental health and HIV disease burden, and presents a greater challenge to improving the level of adherence to treatment. Commonly available treatments and community-based adherence interventions provide comparatively greater benefit to people diagnosed with unipolar non-psychotic unipolar depression.
Association Between Diagnostic/Clinical Features and Adherence
Our study findings are likely rooted in the participants’ psychopathological features and their effect on the person’s ability to seek out and utilize treatment available in the community as well as the ability to enable adaptive behavior change given external logistical support. Depressive illness is associated with amotivation, anergia, impairment in memory and concentration, decreased selfesteem and hopelessness, all of which can interfere with self-care and future oriented activity. Wagner et al. [35] showed that cognitive symptoms of depression -including depressed mood, loss of interest, suicidality, irritability, hopelessness, indecisiveness, poor concentration, worthlessness, and guilt- are more predictive of non-adherence to ART than vegetative symptoms of depression, such as fatigue, loss of appetite or weight, sleep disturbance, and psychomotor agitation. The same study showed that reduced depression over time leads to improved adherence. When depression lifts with psychiatric treatment, individuals are less likely to decline treatment, especially when logistical obstacles are eliminated. Without the added complication of psychosis, depressed individuals may be more likely to understand that taking HIV medication can improve health and prolong life.
Similar to depression, many characteristic features of psychosis and mania can present obstacles to treatment adherence. Certain clinical features that distinguish mania and psychosis from non-psychotic unipolar depression are unique in the way that they can influence a person’s ability and willingness to attend to healthcare needs. For those suffering from psychosis, medical providers can become the subject of paranoid delusions leading the patient to question their diagnoses and view suggested treatment as intentionally harmful or poisonous. Auditory hallucinations can include commands to refuse medication. Thought disorder can result in confusion between cause and effect, for example believing that antipsychotics cause schizophrenia and ART causes AIDS. Negative symptoms such as low motivation can interfere with all self-care activities, with adherence to medication being one of them. Cognitive impairment, which is especially severe in schizophrenia, can cause forgetfulness and misunderstanding of instructions, which also interferes with ability to adhere to treatment. Grandiose delusions can be part of both mania and non-affective psychosis and can include viewing oneself as impervious to illness or delusions of already being cured of HIV. In addition, psychotic depression can be accompanied by mood-congruent delusions of guilt and the view of oneself as hopelessly flawed and thus not worthy of help or treatment. Low insight or poor awareness of psychiatric symptoms can negatively influence attitude towards medication and is one of the major predictors of non-adherence to psychiatric treatment [36], which is in turn correlated with non-adherence to ART. Poor insight into illness occurs in about 50% of patients with schizophrenia and is less common but still significant in bipolar disorder [37]. Efficacy limitations of antipsychotics and mood stabilizers along with varying degrees of treatment resistance mean that psychiatric symptoms are likely to persist and interfere with self-care. In sum, individuals diagnosed with bipolar or psychotic illness probably face more complicated and diverse obstacles to treatment adherence that those with non-psychotic unipolar depression.
Recommendations
In order to improve treatment adherence and outcomes in HIV+ people with bipolar/psychotic disorders, more emphasis needs to be placed on addressing the unique challenges to treatment adherence associated with these psychiatric disorders. Research on correlates of adherence and adherence interventions for psychiatric medication in bipolar/psychotic disorders can be a source of ideas for designing adherence interventions for treatment of comorbid HIV infection as well as other medical conditions.
Treatment Adherence Research
A recent review by Crowe et al. [38], of eleven studies concluded that only a few interventions improved medication adherence in bipolar disorder, yet most improved clinical outcomes, raising questions about the relationship between adherence and clinical outcomes. No characteristic of the psychosocial intervention, mode of delivery, length of delivery or method of measurement appeared to differentiate studies with a positive outcome from those that showed no improvement. A review by Berk et al. [39], found that the risk factors for non-adherence in individuals with bipolar disorder differ from person to person and form predictable clusters defined by variables associated with patient, clinician, family, illness, and treatment. The same study also noted that the fostering of a collaborative therapeutic relationship was related to improved treatment outcomes. For those diagnosed with bipolar disorder, the use of psycho-education, in conjunction with shared decision making between patient and clinician, was found to be effective in improving medication adherence, especially when incorporating use of techniques to promote changes in behavior, skills and attitude [40, 41].
In 2015, El-Mallakh et al. [42] reviewed the prior ten years of research on the role of support services in promoting medication adherence in people with schizophrenia, and suggested that adherence interventions should be tailored to the specific needs of each person, stressed the importance of conducting a root-cause analysis of reasons for non-adherence in each person prior to implementation of services, and recommended addressing adherence to psychiatric medication as a primary goal with adherence to medication for medical illness as a secondary goal. More specific suggestions include interventions with technological supports and environmental cues. These can be helpful to patients with memory problems who are committed to taking medication but have adherence problems due to forgetfulness. Therapeutic support services may be more appropriate to patients with schizophrenia who are ambivalent toward taking medications and/or deny the need to take medications.
Our analyses suggest that diagnostic category can be a marker for more complex challenges to adherence, which could be ameliorated by targeted root cause analysis (RCA), a multivariate examination of the factors associated with negative outcomes, the intent of which is to maximize the benefits of future therapeutic interventions. As noted by Haynes et al. [43], a common component of successful adherence interventions entails the collection of a detailed clinical history prior to implementation of services in order to identify potential contributing factors of non-adherence with patients diagnosed with serious mental illness, a core practice of RCA.
It is also helpful to conceptualize these factors by dividing them into those that affect patients’ willingness versus ability to take medications, otherwise defined as volitional versus non-volitional factors [36]. Volitional non-adherence can be a function of lack of insight, lack of illness awareness, uncontrolled psychiatric symptoms, poor therapeutic alliance with prescribing provider, and fear of side effects and/or experience of actual side effects. Nonvolitional non-adherence factors are not generally within the proximal control of the individual and include such factors as cognitive deficits, substance use, disorganized environments, lack of health insurance or financial resources, fragmented medical care, and homelessness. Poor insight into an individual’s experience of mental and physical illness cannot be ameliorated by a focus on structural elements of the therapeutic process such as increasing accessibility to treatment or transportation. On the other hand, memory impairment or lack of health insurance cannot be fully remedied through psychoeducation or motivational interviewing.
Study Strengths and Limitations
A strength of this study is that our sample, which is characterized by complex comorbidity of HIV, mental illness, substance use and social adversity is representative of the norm for HIV clinics in the US [23]. However a number of limitations could have affected study findings. For example, our use of provider report to determine psychiatric diagnosis may not have been as reliable as if we had used a structured clinical interview (SCID). A related issue concerns active substance use by study participants which could have influenced diagnosis at the time of hospitalization since substance-induced psychiatric syndrome is often misdiagnosed as primary depression, bipolar disorder or schizophrenia spectrum disorder. Third, higher likelihood of mania and psychosis in more advanced cases of HIV disease is thought to be due to HIV infection of the central nervous system (CNS) [44, 45]. Since CNS involvement is more likely when HIV infection is poorly controlled, there may be cases in which poor adherence to ART could have placed a person at higher risk of developing psychiatric illness.
Future studies should aim for greater diagnostic precision through the use of SCID as opposed to relying on chart review or provider reports. Future research should also focus on a more nuanced understanding of complex interactions between psychiatric diagnosis, substance use, and non-adherence to psychiatric treatment and/or ART through of the use of RCA. Better knowledge of these issues could result in better outcomes for people with HIV who are comorbid with the more serious and treatment resistant forms of mental illness. In the absence of clear guidelines for individual providers faced with non-adherence to treatment in patients with HIV and mental illness, we suggest translating the strategies that have been found useful in improving adherence to psychiatric medication. As summarized in a review of literature by Julius et al. [46], these strategies include establishing and facilitating the therapeutic alliance, devoting time to specifically address medication adherence, recognizing risk factors for non-adherence, assessing patients’ motivation to adopt healthy behaviors related to adherence and involving patients’ families whenever possible.
Conclusions
Our findings strongly reinforce the awareness that serious mental illness in persons with HIV is associated with poor adherence to ART and consequently worse outcomes in HIV viral load and mental health quality of life. The main finding of this investigation is that certain mental illness diagnoses can serve as markers for barriers to treatment. In order to improve clinical outcomes for people with HIV comorbid with mental illness on the schizophrenia and bipolar spectrum, a multimodal treatment adherence intervention is required which is tailored to their unique psychiatric features and psychosocial challenges to reduce viral load. Indeed, our findings strongly suggest that psychiatric diagnosis routinely be integrated into the development and design of individualized HIV treatment adherence intervention plans. Specifically, mental health diagnoses with or without psychotic features can help inform the structure of such interventions. For example, when non-adherence is volitional, increasing the frequency and duration of contact between patient and clinician can yield improved treatment adherence and more adaptive treatment outcomes. While the proposed increase in intensity for interventions may result in proximal higher costs, these costs are offset by the potential for lower mortality for those comorbid for schizophrenia and HIV [18]. Finally, but perhaps most importantly, treatment adherence interventions have the potential to interrupt the role that co-morbid persons play as vectors of HIV transmission and of treatment resistant HIV strains. This alone may justify the higher costs associated with the delivery of expanded intensive dual HIV and psychiatric treatment services.
Acknowledgments
Funding This research was supported by Grants from National Institute on Drug Abuse (RO1-DA-015627 “HIV Prevention Program among Substance Abusing SMI”), the National Institute for Nursing Research (RO1-NR-008851 “Nursing Intervention for HIV Regimen: Adherence among SMI”), Penn Mental Health AIDS Research Center (PMHARC - P30MH097488-04) and Penn Center for AIDS Research (CFAR - P30-AI-45008-18).
Footnotes
Compliance with Ethical Standards
Conflict of interest The authors declare that they have no conflict of interest.
Ethical Approval All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent Informed consent was obtained from all individual participants included in the study.
Trial registration: clinicaltrials.gov 29 identifier NCT00264823.
References
- 1.Susser E, Valencia E, Conover S. Prevalence of HIV-infection among psychiatric patients in a New York City men’s Shelter. Am J Public Health. 1993;83(4):568–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rothbard AB, Blank MB, Staab JP, et al. Previously undetected metabolic syndrome and infectious diseases among psychiatric inpatients. Psychiatr Serv. 2009;60(4):534–7. [DOI] [PubMed] [Google Scholar]
- 3.Walkup J, Crystal S, Sambamoorthi U. Schizophrenia and major affective disorder among medicaid recipients with HIV AIDS in New Jersey. Am J Public Health. 1999;89(7):1101–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kessler RC, Birnbaum H, Demler O, et al. Prevalence and correlates of nonaffective psychosis: results from NCS-R. Biol Psychiatry. 2005;57(8):108S–9S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Blank MB, Mandell DS, Aiken L, Hadley TR. Co-occurrence of HIV and serious mental illness among Medicaid recipients. Psychiatr Serv. 2002;53(7):868–73. [DOI] [PubMed] [Google Scholar]
- 6.Himelhoch S, McCarthy JF, Ganoczy D, Medoff D, Dixon LB, Blow FC. Understanding associations between serious mental illness and HIV among patients in the VA health system. Psychiatr Serv. 2007;58(9):1165–72. [DOI] [PubMed] [Google Scholar]
- 7.Lopes M, Olfson M, Rabkin J, et al. Gender, HIV status, and psychiatric disorders: results from the National epidemiologic survey on alcohol and related conditions. J Clin Psychiatry. 2012;73(3):384–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Blank MB, Hennessy M, Eisenberg MM. Increasing quality of life and reducing HIV burden: The PATH+ intervention. AIDS Behav. 2014;18(4):716–25. doi:10.1007/s10461-013-0606x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wagner GJ, Kanouse DE, Koegel P, Sullivan G. Adherence to HIV antiretrovirals among persons with serious mental illness. AIDS Patient Care STDs. 2003;17(4):179–86. [DOI] [PubMed] [Google Scholar]
- 10.Helleberg M, Pedersen MG, Pedersen CB, Mortensen PB, Obel N. Associations between HIV and schizophrenia and their effect on HIV treatment outcomes: a nationwide population based cohort study in Denmark. Lancet HIV. 2015;2(8):e344–50. [DOI] [PubMed] [Google Scholar]
- 11.Hartzell JD, Janke IE, Weintrob AC. Impact of depression on HIV outcome in the HAART era. J Antimicrob Chemother. 2008;62(2):246–55. [DOI] [PubMed] [Google Scholar]
- 12.Springer SA, Dushaj A, Azar MM. The impact of DSM-IV mental disorders on adherence to combination antiretroviral therapy among adult persons living with HIV/aids: a systematic review. AIDS Behav. 2012;16(8):2119–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Horberg MA, Silverberg MJ, Hurley LB, et al. Effects of depression and selective serotonin reuptake inhibitor use on adherence to highly active antiretroviral therapy and on clinical outcomes in HIV-infected patients. JAIDS. 2008;47(3):384–90. [DOI] [PubMed] [Google Scholar]
- 14.Sin NL, DiMatteo MR. Depression treatment enhances adherence to antiretroviral therapy: a meta- analysis. Ann Behav Med. 2014;47(3):259–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Moore DJ, Posada C, Parikh M, et al. HIV-infected individuals with co-occurring bipolar disorder evidence poor antiretroviral and psychiatric medication adherence. AIDS Behav. 2012;16(8): 2257–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Walkup JT, Akincigil A, Chakravarty S, et al. Bipolar medication use and adherence to antiretroviral therapy among patients with HIV-AIDS and bipolar disorder. Psychiatr Serv. 2001;62(3): 313–6. [DOI] [PubMed] [Google Scholar]
- 17.Mellins CA, Havens JF, McDonnell C, et al. Adherence to antiretroviral medications and medical care in HIV-infected adults diagnosed with mental and substance abuse disorders. AIDS Care. 2009;21(2):168–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Palmer NB, Salcedo J, Miller AL, Winiarski M, Arno P. Psychiatric and social barriers to HIV medication adherence in a triply diagnosed methadone population. AIDS Patient Care STDs. 2003;17(12):635–44. [DOI] [PubMed] [Google Scholar]
- 19.Chiles JA, Lambert MJ, Hatch AL. The impact of psychological intervention on medical cost offset: a meta-analytic review. Clin Psychol Sci Pract. 1999;6(2):204–20. [Google Scholar]
- 20.Gilmer T, Stefancic A, Ettner S, Manning W, Tsemberis S. Effect of full service partnerships on homelessness, use and costs of mental health services, and quality of life among adults with serious mental illness. Arch Gen Psychiatry. 2010;67(6):645–52. [DOI] [PubMed] [Google Scholar]
- 21.Rosenheck R, Kasprow W, Frisman L, Liu-Mares W. Cost-effectiveness of supported housing for homeless persons with mental illness. Arch Gen Psychiatry. 2003;60(9):940–51. [DOI] [PubMed] [Google Scholar]
- 22.Blank MB, Eisenberg MM. Tailored treatment for HIV? persons with mental illness: the intervention cascade. J Acquir Immune Defic Syndr. 2013;63:S44–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Blank MB, Himelhoch S, Walkup J, Eisenberg MM. Treatment considerations for HIV-infected individuals with severe mental illness. Curr HIV/AIDS Rep. 2013;10(4):371–9. doi:10.1007/s11904-013-0179-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Collins L, Murphy S, Bierman K. A conceptual framework for adaptive preventive interventions. Prev Sci. 2004;5(3):185–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ware JE Jr, Kosinski M, Keller SD. A 12-item short-form health survey: construction of scales and preliminary tests of reliability and validity. Med Care. 1996;34(3):220–33. [DOI] [PubMed] [Google Scholar]
- 26.Salyers MP, Bosworth HB, Swanson JW, Lamb-Pagone J, Osher FC. Reliability and validity of the sf-12 health survey among people with severe mental illness. Med Care. 2000;38(11): 1141–50. doi:10.2307/3767812. [DOI] [PubMed] [Google Scholar]
- 27.Chum A, Skosireva A, Tobon J, Hwang S. Construct validity of the SF-12v2 for the homeless population with mental illness: an instrument to measure self-rported mental and physical health. PLoS ONE. 2016;11(3):e0148856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Curran P, Hussong A. Structural equation modeling of repeated measures data: Latent curve analysis In Moskowitz D& Hershberger S(Eds.), Modeling intraindividual variability with repeated measures data. Mahwah: Lawrence Erlbaum; 2002; pp. 59–85. [Google Scholar]
- 29.Curran P, Muthen B. The application of latent curve analysis to testing developmental theories in intervention research. Am J Community Psychol. 1999;27:567–95. [DOI] [PubMed] [Google Scholar]
- 30.Barnes G, Reifman A, Farrell M, Dintcheff B. The effects of parenting on the development of adolescent alcohol misuse: a six-wave latent growth model. J Marriage Family. 2000;62:175–86. [Google Scholar]
- 31.Fergus S, Zimmerman M, Caldwell CH. Growth trajectories of sexual risk behavior in adolescence and young adulthood. Am J Public Health. 2007;97:1096–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bollen K, Curran P. Latent curve models. New Jersey: Wiley; 2006. [Google Scholar]
- 33.Biesanz J, Deeb-Sossa N, Papadakis A, Bollen K, Curran P. The role of coding time in estimating and interpreting growth curve models. Psychol Methods. 2004;9(1):30–52. [DOI] [PubMed] [Google Scholar]
- 34.Geiser C Data analysis with mplus. New York: Guilford; 2013. [Google Scholar]
- 35.Wagner GJ, Goggin K, Remien RH, et al. A closer look at depression and its relationship to HIV antiretroviral adherence. Ann Behav Med. 2011;42(3):352–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Velligan DI, Weiden PJ, Sajatovic M, et al. The expert consensus guideline series: adherence problems in patients with serious and persistent mental illness. J Clin Psychiatry. 2009;70(Suppl 4):1–46. [PubMed] [Google Scholar]
- 37.Arango C, Amador X. Lessons learned about poor insight. Schizophr Bull. 2001;37(1):27–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Crowe M, Porter R, Inder M, Lacey C, Carlyle D, Wilson L. Effectiveness of interventions to improve medication adherence in bipolar disorder. Aust N Z J Psychiatry. 2012;46(4):317–26. doi:10.1177/0004867411428101. [DOI] [PubMed] [Google Scholar]
- 39.Berk L, Hallam KT, Colom F, et al. Enhancing medication adherence in patients with bipolar disorder. Hum Psychopharmacol. 2010;25(1):1–16. doi:10.1002/hup.1081. [DOI] [PubMed] [Google Scholar]
- 40.Vieta E Improving treatment adherence in bipolar disorder through psychoeducation. J Clin Psychiatry. 2005;66(Suppl 1):24–9. [PubMed] [Google Scholar]
- 41.Sajatovic M, Davies M, Hrouda DR. Enhancement of treatment adherence among patients with bipolar disorder. Psychiatr Serv. 2004;55(3):264–9. [DOI] [PubMed] [Google Scholar]
- 42.El-Mallakh P, Findlay J. Strategies to improve medication adherence in patients with schizophrenia: the role of support services. Neuropsychiatr Dis Treat. 2015;11:1077–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Haynes RB, Yao X, Degani A, Kripalani S, Garg A, McDonald HP. Interventions to enhance medication adherence. Cochrane Database Syst Rev. 2005;167:540–9. [DOI] [PubMed] [Google Scholar]
- 44.Mijch AM, Judd FK, Lyketsos CG, Ellen S, Cockram A. Secondary mania in patients with HIV infection: are antiretrovirals protective? J Neuropsychiatry Clin Neurosci. 1999;11(4):475–80. [DOI] [PubMed] [Google Scholar]
- 45.de Ronchi D, Faranca I, Forti P, et al. Development of acute psychotic disorders and HIV-1 infection. Int J Psychiatry Med. 2000;30(2):173–83. [DOI] [PubMed] [Google Scholar]
- 46.Julius RJ, Novitsky MA Jr, Dubin WR. Medication adherence: a review of the literature and implications for clinical practice. J Psychiatr Pract. 2009;15(1):34–44. [DOI] [PubMed] [Google Scholar]