Abstract
A suite of imaging tools for detecting specific chemicals in the central nervous system could accelerate the understanding of neural signaling events critical to brain function and disease. Here, we introduce a class of nanoparticle sensors for the highly specific detection of acetylcholine in the living brain using magnetic resonance imaging. The nanosensor is composed of acetylcholine-catalyzing enzymes and pH-sensitive gadolinium contrast agents co-localized onto the surface of polymer nanoparticles, which leads to changes in T1 relaxation rate (1/T1). The mechanism of the sensor involves the enzymatic hydrolysis of acetylcholine leading to a localized decrease in pH which is detected by the pH-sensitive gadolinium chelate. The concomitant change in 1/T1 in vitro measured a 20% increase from 0 to 10 μM acetylcholine concentration. The applicability of the nanosensors in vivo was demonstrated in the rat medial prefrontal cortex showing distinct changes in 1/T1 induced by pharmacological stimuli. The highly specific acetylcholine nanosensor we present here offers a promising strategy for detection of cholinergic neurotransmission and will facilitate our understanding of brain function through chemical imaging.
Keywords: nanosensor, acetylcholine, MRI contrast agents, neurotransmitter, biosensors
Graphical Abstract

Imaging tools that enable real-time visualization of molecular neural events, namely neurotransmitter release, are highly valuable for understanding the basis of brain function and disease. Cellular-level methods such as electrophysiology1 and optical imaging2 offer recordings of neural activity with precision and high specificity, but are often limited to sampling a relatively small area of the mammalian brain. Conversely, among modern imaging techniques, magnetic resonance imaging (MRI)3 is a powerful tool that provides advantages for in vivo analysis, as it can be applied noninvasively, with unlimited tissue penetration and mapping capabilities of the whole brain. Despite these advantages, resolving target-specific detection of neurotransmitters with molecular specificity for functional neuroimaging is yet to be fully established, and only a limited number of successful studies have been implemented for in vivo monitoring in the brain. This is in part due to a low concentration of neurotransmitters in the brain as well as the low intrinsic sensitivity and resolving power of MRI. Consequently, synthesizing a highly sensitive, stable, and nontoxic probe is always a great challenge for effective application of molecular neuroimaging in the brain. Currently, a number of MRI molecular contrast agents have been explored for imaging of neurotransmitters in the brain. For example, detection of dopamine4,5 and serotonin6 has been developed from engineered forms of flavocytochrome P450-BM3 with a detectable T1 signal change. A contrast agent to detect glutamate7 has also been developed based on the displacement of mGluR5 receptor as well as agents based on a crown ether cation-binding motif chelating a gadolinium (Gd) to target glutamate, GABA, and glycine albeit with millimolar affinity.8 An additional approach, using the chemical exchange saturation transfer (CEST) effect, has been demonstrated to detect glutamate in human subjects.9,10 While there is a growing repertoire of neurotransmitter-sensitive MRI probes, there remains considerable opportunities for the development of imaging agents that can respond to neural activity with high chemical specificity and sensitivity.
In particular, the development of nanostructured sensors as MRI contrast agents is a promising direction for the detection and activity-dependent in vivo monitoring of neurotransmitters in the brain. Unlike traditional molecular organometallic chelates, these nanostructured probes offer advantages of flexibility and modularity to modify their physicochemical properties and functionalities.11 To date, MRI contrast agents have been packaged in nanoparticles in order to amplify magnetic relaxivity (r1),12 improve penetration and retention in tumor,13 and monitor enzymatic activity14 and used for theranostic applications.15 However, the development of nanoscale MRI contrast agents for the chemical imaging of neurotransmitters has yet to be realized.
In the present study, we developed a nanoparticle-sensing platform for the imaging neurotransmitter acetylcholine in living brain tissue using MRI. As an important molecular messenger, acetylcholine is involved in regulating chemical communication between cells in the brain. In particular, the cholinergic system is one of the most important modulatory neurotransmitter systems in the brain, in which both synaptic16 and volume17 transmission govern activities that depend on selective attention,18 formation of working memories,19 and cognitive behavior.20 Additionally, perturbations of the cholinergic system are implicated in schizophrenia,21 depression,22 and Alzheimer’s disease.23 Previously, direct measurement of choline using 1H NMR spectroscopy and its application in malignant breast tumors was reported.24,25 Our method differs by detecting acetylcholine directly, since it is unlikely that choline could be used a surrogate in the brain.26
The design of the nanosensor involves co-immobilizing the enzyme butyrylcholinesterase (BuChE) and pH-sensitive gadolinium contrast agents on a nanoparticle to create a pH drop triggered by the enzymatic hydrolysis of acetylcholine which was detected by the proximate contrast agents within the nanoparticle microenvironment leading to an increase in T1 relaxation rate (1/T1). The nanosensor platform presented here prevents the sensing components from diffusing away in vivo and provides a required proximity between the contrast agent and BuChE. As such, this design is well-suited for real-time imaging of acetylcholine in the living brain, in addition to filling a need in the field of MRI by providing chemical specificity to neuroimaging.
Here, we show: (1) synthesis and characterization of the nanosensor for detection of acetylcholine (ACh-MRNS); (2) response of the nanosensor to increase in acetylcholine levels with suitable sensitivity and selectivity in vitro; and (3) in vivo detection of endogenous release of acetylcholine in the rat medial prefrontal cortex (mPFC), which is known to receive dense cholinergic inputs from the basal forebrain and the hippocampal formation27 stimulated by systemic administration of clozapine.
RESULTS
Nanosensor Mechanism and Particle Characterization
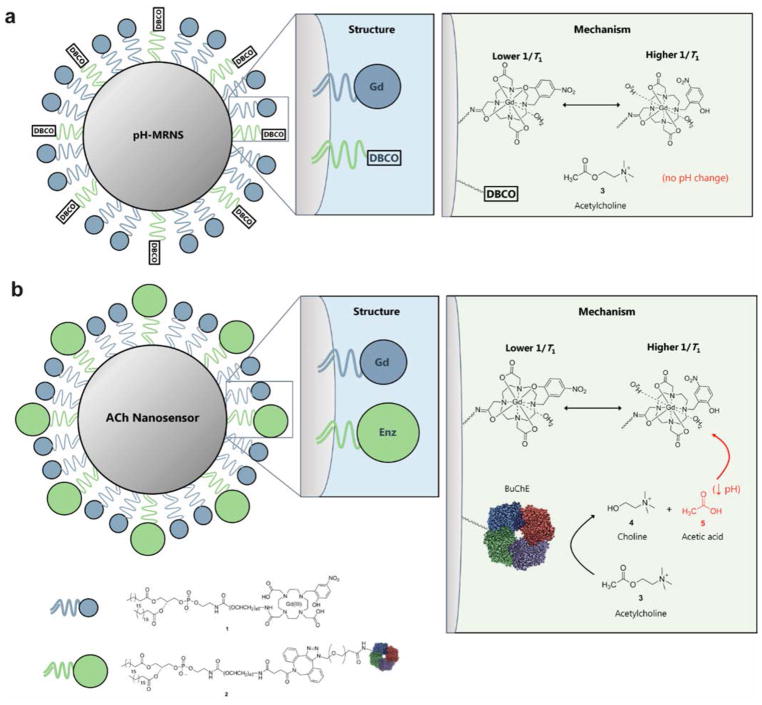
In our nanosensor design, we fabricated highly plasticized polymer nanoparticles as a platform and functionalized the surface to co-immobilize both transduction and signaling moieties (Figure 1). The mechanism is based on the enzymatic hydrolysis of acetylcholine by BuChE into choline and acetic acid, and the resulting reduction in local pH alters the water coordination28 of the pH-sensitive contrast agent leading to an increase in r1 and T1 relaxation rate (1/T1). The close proximity of the enzymes and pH-sensitive contrast agents on a nanoscale platform ensures MRI signal changes are generated by the contrast agent at the origin of the pH changes created by the enzymatic hydrolysis of acetylcholine. The H+ will be generated in the microenvironment close to the surface of the ACh-MRNS during the enzymatic hydrolysis and detected by the pH-sensitive contrast agent nearby. The localized pH changes result from a gradient as acetylcholine is consumed and H+ ions are released at a fast rate. The signal is reversible, dependent on acetylcholine concentration, and achieves a limit of detection (LOD) and sensitivity in the physiological concentration of acetylcholine (nanomolar to micromolar in the extracellular space of the brain).17
Figure 1.
Schematic of the nanosensor structure and mechanism. (A) pH-MRNS. Only pH-sensitive contrast agents were covalently conjugated to the DSPE-PEG lipids and coated on the surface of the lipophilic core. Without co-immobilized BuChE, acetylcholine will not be hydrolyzed to alter local pH. (B) ACh-MRNS. Both pH-sensitive contrast agents and BuChE were covalently conjugated to the DSPE-PEG lipids and coated on the surface of the lipophilic core. The BuChE catalyzes the hydrolysis of acetylcholine to choline and acetic acid, and the resulting drop in local pH triggers a conformational switch of the contrast agent: There is one more water molecule coordinated to one Gd(III) chelate in acidic conditions compared to its structure in basic conditions, which leads to increased T1 relaxation rate (1/T1) of the contrast agent.
We characterized two types of nanosensors intended for the in vivo study: (1) ACh-MRNS and (2) a pH-sensitive sensor (pH-MRNS) (Figure 1 and Figure S1). The ACh-MRNS contained both the pH-sensitive contrast agents and BuChE conjugated to nanoparticles for specific detection of acetylcholine; and the pH-MRNS was conjugated with pH-sensitive contrast agents only, i.e., without enzyme. The pH-MRNS served as a control due to potential interference from systemic changes in pH. The structure of the nanoparticles is similar to the optode-based nanosensors previously reported by our laboratory.29,30 The nanoparticle is composed of a core of highly plasticized polyvinyl chloride (PVC) and a coat of amphiphilic DSPE-PEG-lipid. The high molecular weight PVC (Sigma catalog no. 81392; Mw ~90000) was dissolved in bis(2-ethylhexyl)sebacate (DOS) to form a highly lipophilic nanoparticle. The surface of the particles is derivatized through the use of DSPE-PEG lipids as a biocompatible coating on the lipophilic surface (Figure S1). Separately, pH-sensitive contrast agents (compound 1) and BuChE (compound 2) were covalently linked sequentially to the corresponding DSPE-PEG-lipid derivatives (see the Materials and Methods). We synthesized the transduction element, Gd(NP-DO3A), a pH-sensitive analogue of the clinically used Gd(DOTA), based on the procedure by Woods et al.31 (Figure S2). In this structure, paramagnetic Gd(III) is chelated within a 12-member ring containing a p-nitrophenol. Under basic conditions, the probe is coordinated to a single water molecule, but under acidic conditions, two water molecules will coordinate to Gd(III), leading to increase in 1/T1 (Figure 1).
The size and surface charge of fabricated nanoparticles were characterized by dynamic light scattering (DLS) and zeta-potential in 1× PBS, pH 7.4, respectively. We measured the average size of three batches of the ACh-MRNS and pH-MRNS using DLS. The average hydrodynamic diameter of ACh-MRNS was 96 ± 26 nm (mean ± standard deviation), and the size of the pH-MRNS was 77 ± 23 nm. Parts A and B of Figure 2 display the size distribution of a single nanosensor batch, which is representative of a typical batch. The surface charge of the ACh-MRNS and pH-MRNS was −41 ± 1.9 and −29 ± 3.4 mV, respectively. The negative charge of BuChE may contribute to the difference in zeta-potential.32 We also performed transmission electron microscopy (TEM) using one batch of the ACh-MRNS as an example, and the generated image in Figure 2C indicated spherical ACh-MRNS with a size of 118 ± 25 nm and distribution shown in Figure 2D. The DSPE-PEG lipid coat was also observed in the TEM (Figure S3). The concentration of nanoparticles (2.62 ± 0.13 × 1014 particles/mL) was also identified using an nCS1 nanoparticle analyzer (Spectradyne, Torrance, CA). By combining the concentration and information from the amount of Gd bound on the surface of the nanoparticle quantified by inductively coupled mass spectrometry (ICP-MS), we estimated the density of Gd(III) on the surface of the nanoparticle to be 0.99 ± 0.27 atoms of Gd/nm2.
Figure 2.
Characterization, in vitro calibration, and selectivity of nanosensors. Dynamic light scattering (DLS) analysis showing size distribution of nanosensors, (A) ACh-MRNS, and (B) pH-MRNS. (C) TEM image of the ACh-MRNS. (D) Distribution of sizes of the ACh-MRNS from the TEM image.
The r1 of ACh-MRNS and pH-MRNS was determined as 6.36 and 6.91 mM−1 s−1 (pH 7.4), respectively, from the concentration of Gd(III) and the T1 collected from a 7 T Bruker Biospec small animal MRI scanner (Bruker Inc., Billerica, MA). In addition, the r1 of ACh-MRNS was also measured in the low field with a 60 MHz Bruker Minispec mq60 NMR analyzer (Bruker Inc., Billerica, MA) as 12.1 mM−1 s−1 (pH 7.4) at 37 °C. The r1 of both ACh-MRNS and pH-MRNS were approximately twice the value obtained from the clinically used DOTA-Gd33 and the free Gd(NP-DO3A) (Table 1), indicating suitable contrast for subsequent studies. Compared to free molecules, the density of contrast agents bound with the slower tumbling rate of the particles may restrict their internal and overall motion leading to a longer correlation time of the water-bound contrast agent (τR), which would theoretically account for the increased r1 at the same pH.34,35
Table 1.
Relaxivity (r1) of Contrast Agents Used in This Study
| contrast agent | r1 (mM−1 s−1) at pH 7.4 |
|---|---|
| DOTA-Gd | 3.2131 |
| Gd(DO3A-NP) | 3.15 ± 0.47 |
| ACh-MRNS | 6.36 ± 0.44 |
| pH-MRNS | 6.91 ± 0.65 |
Errors were calculated from SD of T1 of three independent tests using error propagation.
Sensor pH Dependence
To evaluate the pH-dependence of the r1 and the corresponding 1/T1, we suspended ACh-MRNS in PBS at pH 6, 6.5, 7, 7.4, and 8 and scanned using MRI. When the pH decreased from 8 to 6, the signal intensity (1/T1) increased by 24% (Figure S4A). The corresponding r1 of ACh-MRNS at different pH are indicated in Table 2. The extracellular pH in the brain is approximately 7.3.36 Within the physiological pH range 7.2–7.8, the 1/T1 of the ACh-MRNS changed by less than 5% (Figure S4A). The moderate response in this range means that the ACh-MRNSs are minimally affected by global pH fluctuations in the brain. To further assess the effect of pH on the response of ACh-MRNSs, we calibrated the acetylcholine response of the sensor in a background of varied pH using a 1.5 T Bruker Minispec mq60 NMR analyzer (Bruker Inc., Billerica, MA). We did not observe significant differences in the calibration against acetylcholine at pH 7.2, 7.4, and 7.8 (Figure S4B). According to the literature,37 BuChE is active between pH 6 and 8. When acetylcholine is hydrolyzed, the enzyme will remain active until the pH drops below 6, at which point the activity will be reduced. Thus, we believe that the capability of the ACh-MRNS to measure acetylcholine in this range is not effected by endogenous changes in pH. It will be important to continue to use the pH-MRNS as a control to further mitigates the risk of interference from physiological pH changes.
Table 2.
Relaxivity (r1) of ACh-MRNS at Different pH Used in This Study
| pH | r1 (mM−1 s−1) |
|---|---|
| 8 | 6.14 ± 0.57 |
| 7.4 | 6.36 ± 0.44 |
| 7 | 7.13 ± 0.32 |
| 6.5 | 7.87 ± 0.35 |
| 6 | 8.55 ± 0.56 |
Errors were calculated from SD of T1 of three independent tests using error propagation.
Sensor Calibration
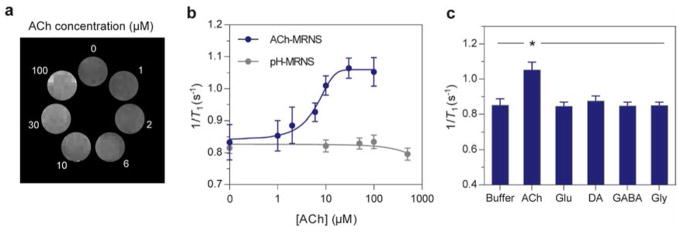
We calibrated the nanosensors by suspending nanoparticles corresponding to 0.079 mM conjugated Gd(III) with concentrations of acetylcholine varying from 0 to 100 μM in 1× PBS, pH 7.4. The ionic strength of this buffer is 162.7 mM, which is consistent with the ionic strength of artificial cerebrospinal fluid (ACSF) (152.8 mM). Since the sensors were delivered to the CSF instead of blood, we did not incorporate serum proteins to our measurements. A clear gradient in brightness was observed from the T1-weighted MR image (Figure 3A): the higher concentrations of acetylcholine led to increasingly brighter images, which represents a higher 1/T1. A calibration curve of 1/T1 as a function of concentration of acetylcholine (Figure 3B) showed an enhancement of 1/T1 by more than 10% when the concentration of acetylcholine increased from 0 to 6 μM and 20% when the concentration increased to 10 μM (blue line). Using the sigmoidal fit of the calibration curve to calculate the analytical properties of the ACh-MRNS (Figure 3B), we found a lower limit of detection (LLOD) of 2.64 μM and a sensitivity of ±4 μM (calculated from the fitted curve and error bar in Figure 3B) at an acetylcholine concentration of 10 μM. In comparison, a control study using an equivalent amount of pH-MRNS with the same concentration of Gd(III) and free BuChE (not conjugated to the particle) led to no significant change in 1/T1 when the concentration of acetylcholine was increased from 0 to 500 μM (gray). This result indicates that the amount of free enzyme was not sufficient to create T1 contrast by the pH-MRNS.
Figure 3.
Characterization, in vitro calibration, and selectivity of nanosensors. (A) Higher concentrations of acetylcholine led to brighter MR images. (B) 1/T1 of ACh-MRNS was enhanced when higher concentrations of acetylcholine were present. The ACh-MRNS (blue) were exposed to solutions of 0, 1, 2, 6, 10, 30, and 100 μM of acetylcholine. The pH-MRNS and free BuChE (gray) were exposed to solution of 0, 10, 50, 100, and 500 μM of acetylcholine. (C) The selectivity toward acetylcholine of ACh-MRNS. The nanosensors were exposed to PBS buffer or to solutions of acetylcholine (0.1 mM), glutamate (5 mM), dopamine (5 mM), GABA (5 mM), and glycine (5 mM). 1/T1 in solutions of acetylcholine was significantly higher than other groups (one-way ANOVA, *P < 0.005, for N = 3). The 1/T1 in panels B and C were calculated from an average T1 of three independent measurements. Error bars in panels B and C were calculated from standard deviation (SD) of T1 using error propagation.
Our design theory was that the nanoscale particle is a necessary component of the sensing mechanism. In short, adding the individual components without immobilization on the scaffold would not be sufficient for two reasons. First, the sensing components would diffuse away from each other in vivo and would not remain in proximity for sensing. Second, the enzyme and pH indicator are preconcentrated on the sensor, which sets up a localized pH environment that would not be seen if the enzyme was not attached to the particle. In order to validate the pH effect confined to a localized microenvironment, we measured the pH in the mixture of 0–5 mM of acetylcholine with the ACh-MRNS (Figure S5A) and pH-MRNS (with free BuChE) (Figure S5B), respectively, using fluorescein, a commonly used pH indicator. The results showed that no detectable pH changes of the bulk buffered system were detected until the level reached 5 mM of acetylcholine. Thus, we extrapolate these findings to the enhancement of 1/T1 by locally generated protons (H+) from the enzymatic hydrolysis of acetylcholine in the microenvironment at the surface of the ACh-MRNS. Similar localized pH effects have been observed in biosensors and microelectrodes, as reported by our laboratory and other groups.38–41 To confirm that the change in 1/T1 was not initiated by free Gd3+, a xylenol orange test showed that the A573/A433 did not increase when higher concentrations of acetylcholine were hydrolyzed by the ACh-MRNS and no free Gd3+ was generated in this process (Figure S6). Thus, the increase of 1/T1 was not caused by the alteration of free Gd3+.
To determine the reaction time of the enzymatic hydrolysis, we performed an Ellman’s assay which indicated that the conjugated BuChE consumed 5–100 μM of acetylcholine between 40 s and 2 min (Figure S7A,B). For MRI measurements, though, our acquisition times are longer than this 2 min time frame. For the acquisition, we used a Rapid Acquisition with Relaxation Enhancement with Variable TR (RARE-VTR) sequence to obtain the T1 in a total time of 9.5 min. This sequence was chosen as it minimizes distortion during in vivo imaging.42 During the scan, signal intensities at six different TRs were collected sequentially. The relative changes of signal intensity elicited by increasing acetylcholine levels decreased from 20% at the first TR (0–4.3 s) to 0 at last TR (236.3–570.1 s) scan sequence (Figure S8). These results indicate that the in vitro calibration mainly represented the response of ACh-MRNS to acetylcholine in the first 2 min of the scan during which the enzymatic hydrolysis of acetylcholine took place. Thus, although the 9 min scan is essential for obtaining a reliable T1, the final value correctly reflects the shorter time frame of acetylcholine hydrolysis.
Sensor Selectivity
Since ACh-MRNS is enzyme-based, we expect high specificity against other neurotransmitters (Figure 3C). To verify the selectivity, we suspended nanoparticles (corresponding to 0.079 mM conjugated Gd) in 200 μL solutions of either PBS buffer, acetylcholine, glutamate, dopamine, GABA, or glycine. By measuring 1/T1, we found that only acetylcholine solution led to a more than 20% increase compared to PBS (P = 0.0001, F value = 21.68, df = 17; ANOVA with Tukey’s posthoc test), and none of the potential interfering neurotransmitters elicited any significant difference (P > 0.5). These results are consistent with the assumption that BuChE selectively hydrolyzes acetylcholine and supports our nanosensors to selectively respond to acetylcholine.
Acetylcholine Detection in the Rat Brain in Vivo
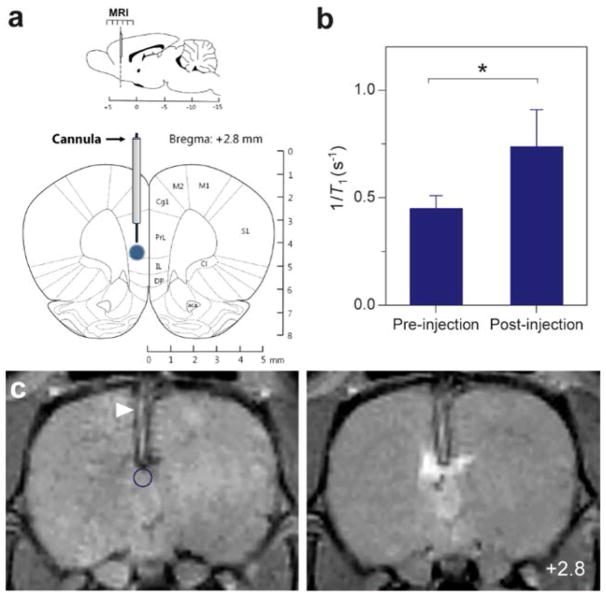
We investigated the ability of ACh-MRNS to detect endogenous acetylcholine release in the rat medial prefrontal cortex (mPFC). First, to assess the amount of contrast provided by ACh-MRNS in the living brain tissue, a 2 μL solution (47 μM of Gd(III)) of the nanosensors was injected into the mPFC through the implanted cannula of anesthetized rat subjects (Figure 4). An increase of 60% in 1/T1 was produced (Figure 4B), and a strong T1-weighted contrast was observed at the site of injection (Figure 4C). Placement of the cannula in the mPFC was verified during the anatomical MRI scans and after surgery from histological analysis (Figure S9).
Figure 4.
In vivo sensor contrast. (A) Schematic diagram of placement of cannula and ACh-MRNS infusion (blue) aimed at rat mPFC and MRI data acquired from sensor-encompassing slices. (B) ROI-averaged MR signal intensity showing increase in 1/T1 for >60% in the postnanosensor injection in comparison to preinjection slice. (C) Coronal MR image brain slice (bregma: + 2.8 mm) of pre-(left) and post-nanosensor injection (right), with circle ROI (blue) defined for analysis. Arrowhead (white) indicates the position of cannula, and the circle (blue) defines the ROI for analysis. Error bars were calculated from SD of T1 using error propagation. Diagram adapted with permission from ref 65. Copyright 2009 Academic Press.
Next, time–course changes in acetylcholine-dependent 1/T1 were acquired by stimulating the release of acetylcholine in the mPFC using a pharmacological agent, clozapine. Clozapine, an atypical antipsychotic drug, has been shown to induce a 2–3-fold increase in acetylcholine concentration from the basal level, which peaks after 30 min and is sustained for over 1 h in the rat mPFC.43 Briefly, the procedure for in vivo imaging included three consecutive scans after nanosensor delivery on a 7 T MRI scanner at 23 min intervals (MRI scan procedure: 0, 23, 46 min post nanosensor injection; Figure 5A). The experimental group (N = 6) consisted of ACh-MRNS infused through the cannula and a concurrent subcutaneous injection of clozapine into the hind of the animal. For the control group (N = 6), ACh-MRNS was delivered without clozapine treatment. T1 was quantified in regions of interest (ROIs) covering the injection sites. Each ROI volume was defined by a cylinder with a diameter of 1.2mmand a thickness of 1 mm centered at the injection site, and T1 was measured and normalized with respect to control ROIs identically placed on the contralateral side of the brain with no sensor delivery. Examination of the T1 and 1/T1 time courses in Figures 5B–D clearly displayed a difference in 1/T1 after 30 min post sensor delivery in the experimental group (blue) compared to the control group (green). For both groups, a slight decrease in 1/T1 was observed at the 23 min time period. A markedly evident 1/T1, however, was observed at 46 min time point with a significant difference of more than 13% 1/T1 in the experimental group compared to the controls (P = 0.018, Student’s t-test). Clozapine was chosen for stimulation because it is known to target the muscarinic acetylcholine receptors in the brain.44–46 In one important study using microdialysis, Ichikawa et al. found that clozapine elicited a 2.5× increase in the concentration of acetylcholine in the mPFC within 30 min after the drug was administered.43 The peak time recorded by the microdialysis is consistent with the result from the ACh-MRNS. In addition, the pattern of 1/T1 decrease over time in the control groups was consistent with the effects of particle diffusion, as indicated by a similar ~19% decrease in 1/T1 by observing the time course of similarly injected fluorescent nanoparticles in the 0.6% agarose phantoms (Figure S10). The particle diffusion was significantly slower than a molecular dye (data not shown). Previous reports show that nanoparticles can be cleared by microglia/macrophages in the CNS.47–49 According to the literature, clearance takes place in a few hours to days after the injection of nanoparticles. In the future, this factor needs to be considered when the ACh-MRNSs are used for monitoring acetylcholine in a longer-term experiment. The rate of sensor diffusion and phagocytosis is a potential factor for in vivo measurements, and we reason that the ACh-MRNS diffused at the site of injection would lead to a decrease in 1/T1, however, this effect was offset by the response of ACh-MRNS against endogenously released acetylcholine in the experiment group as observed by the significant difference detected in the scan at 46 min compared to controls.
Figure 5.
Acetylcholine detection in vivo. (A) Experimental procedure: Delivery of nanosensors (ACh-MRNS or pH-MRNS) through cannula followed by subcutaneous administration of drug (clozapine) and then three consecutive MR scans 23 min apart denoted, t = 0, 23, 46 min. (B) Coronal brain slices showing time-courses of acetylcholine detection. In the experimental group (top panels, N = 6), ACh-MRNSs were injected through the cannula with clozapine administration, while the control group (bottom panels, N = 6) comprised ACh-MRNS delivery without clozapine. For the purpose of display, the heatmap of the top layer (T1-registered map) was generated between 600 and 1600 ms and overlaid onto the image. (C) Distinct acetylcholine signal changes accompanied by a difference of 13% in 1/T1 (P = 0.018; Student’s t-test) between experimental (blue) and control groups (green) was observed at 46 min, indicating distinct detection of acetylcholine driven by local enzymatic hydrolysis by the ACh-MRNS induced by clozapine, as shown by higher 1/T1 in the experimental group. Identical MR scanning procedures were conducted in a new cohort (pH-MRNS group, gray, N = 3) to study the localized effects of enzymatic hydrolysis of acetylcholine to trigger changes in 1/T1. In this group, pH-MRNSs (nanosensors with pH-sensitive Gd and without conjugated enzymes) were delivered along with clozapine administration (gray). After 46 min, 1/T1 also showed a difference of >15% (P = 0.005) in comparison to the experimental group, showing global pH changes were not detected and induced by clozapine administration and confirming the validity of specific detection of acetylcholine driven by local enzymatic hydrolysis by the ACh-MRNS. (D) Individual data points of relative 1/T1 in the ACh-MRNS (blue, N = 6), control (green, N = 6), and pH-MRNS (gray, N = 3) group at 0, 23, 46 min postinjection were plotted. Error bars were calculated from SD of normalized T1 using error propagation.
To ascertain whether global pH changes in the brain were induced by clozapine administration and interfered with the sensor response, identical scanning procedures were performed on another cohort (N = 3). In this group, the pH-MRNSs (gray, Figure 5C) were delivered with a concurrent clozapine injection, procedurally identical to the experimental group above. A similar 1/T1 was observed after injection of nanosensors (0 and 23 min postinjection period). However, at a subsequent time point at 46 min postinjection, a difference of >15% 1/T1 was observed in comparison to the experimental group (P = 0.005) which indicates that clozapine does not cause significant acidic conditions in the brain. The individual data points of relative 1/T1 in this group are available in Figure 5D. This result suggests that ACh-MRNSs reliably detect acetylcholine levels as governed by the predicted mechanism of the nanosensor and not due to a global change in pH in the brain.
DISCUSSION
The use of MR-active nanosensors to image acetylcholine is particularly attractive for several reasons. First, the nanoscale platform incorporates all sensing components together, and hence, the close proximity between cholinesterase and pH-sensitive contrast agent creates a localized effect and facilitates specific detection of acetylcholine. Also, the nanosensors are based on a modular design that can be extended to the detection of other neurotransmitters and physiological analytes by simple substitution of enzymes or functional contrast agents.
In our studies, a 1/T1 of ~13% at 46 min indicates a micromolar increase in the acetylcholine levels, as estimated according to the in vitro calibration. This finding was comparable to other groups’ attempts to measure acetylcholine in the brain, although there is no direct method of comparison in the literature. As cited above, our studies reflect the temporal increase in acetylcholine in response to clozapine stimulation, as had been observed previously via microdialysis.43 Other examples include those that used coated microelectrodes, nicotine50 or KCl26 as local stimuli to trigger an increase of acetylcholine of up to 25 μM.51,52 However, we emphasize the difficulty of direct comparison between methods of analysis and pharmacological stimulation. Our future studies will focus on in vivo calibration and rigorous comparison as an assessment of the advantages and disadvantages of various methodologies.
In our in vivo study, we expect that nanoparticles diffuse similarly in both the experimental and control groups; hence, a sustained 1/T1 signal is attributed to the molecular changes in the brain rather than other natural variation occurring in the brain, such as pH changes, anesthetic effects, and temperature differences, as the two groups differ only by administration of the drug otherwise all other experimental conditions were identical. We did observe larger differences in the control group (Figure 5D); however, the variation becomes more pronounced at later scanning stages, i.e., 46 min, than earlier scan time points. This is observable in both control and pH-MRNS groups but evidently less in the ACh-MRNS group. The individual difference in activation of microglia and rate of diffusion between each animal may explain the variance in the control and pH-MRNS cohort. The rate of clearance due to microglia usually takes place over hours to days, and it is anticipated that it will have greater impact on longer term studies in the future. An alternative faster pulse sequence paired with higher resolution imaging could be performed to improve temporal resolution and increase the fidelity of acetylcholine detection.53,54 The current methodology demonstrates relative changes in acetylcholine levels and would require in situ calibration to be performed before quantitative results could be achieved.
Due to the difficulty of delivering our nanosensors noninvasively through the blood–brain barrier (BBB),55 which remains a key challenge for molecular neuroimaging applications in live animals, we have implemented a cannula placement for delivery of nanosensors aimed at the mPFC. For ongoing applications in animal studies, disruption of BBB using hyperosmotic shock or ultrasound methods which have been used to deliver small molecules56 and nanoparticles57 into the brain will be explored to improve probe delivery. Also, nondisruptive methods to deliver nanoparticles to the CNS have been established in preclinical studies. For instance, polymeric nanoparticles have been coated with artificial amphiphilic polymers or protein-based antibodies, peptides, or receptors to overcome the BBB via transcytosis for therapy against stroke, Alzheimer’s disease, or Parkinson’s disease.58,59 Thanks to the modular feature of the ACh-MRNS, building on these reports by modifying the surface with targeting ligands could be considered as a strategy to deliver the nanosensors. Although the challenge of crossing the BBB could limit translation of this technology to human use, there still remains substantial potential as a preclinical tool for use in animals if this barrier is not overcome. As with all measurement methods, the risk of distorting the biological environment by the removal of analyte for detection is a real possibility. In these studies, due to the size of the sensors, we believe they are located in the extrasynaptic microdomain. Thus, the volume transmission, i.e., “spillover”, acetylcholine are the species that are predominantly detected, rather than acetylcholine molecules directly involved in the synaptic cleft. Hence, the acetylcholine in the synaptic cleft will be readily available for recycling process, reducing the buffering effect by the sensors. In the future, as we strive to reduce the size of the sensors and target them to the synaptic cleft, the possible consumption of analyte may become a greater issue. Lastly, enhanced sensitivity for detection of acetylcholine can be achieved by exploring a more active enzyme such as acetylcholinesterase.60 These steps will facilitate the application of MRI and nanosensors for chemical imaging of neurotransmitters fundamental to the understanding of brain function and disease.
CONCLUSION
In summary, we have developed and characterized a neurotransmitter-sensitive MR-active nanosensor for the detection of acetylcholine in the brain. Acetylcholine is a neurotransmitter known to play a prominent role in mammalian social behaviors and neural processes that govern cognition and memory. As such, there is a considerable interest for imaging this molecule. In this study, we first demonstrated the ACh-MRNS was capable of measuring acetylcholine in low micromolar concentration by co-immobilizing cholinesterase and pH-sensitive contrast agents on a nanoparticle. We further proved that the ACh-MRNS can detect clozapine induced endogenous release of acetylcholine in the rat brain. The ACh-MRNS we report here is the first nanosensor for the detection of acetylcholine using MRI in living brain as characterized and implemented both in vitro and in vivo. Also, the modular design of sensors offers a sensing platform which can be extended to integrate different components for detection of other neurotransmitters and physiological analytes by substituting the enzymes or functional contrast agents to achieve better specificity and sensitivity.
MATERIALS AND METHODS
Materials
2-Hydroxy-5-nitrobenzyl bromide, acetic anhydride, bis(2-ethylhexyl)sebacate (DOS), butyrylcholinesterase from equine serum (EC 3.1.1.8) (BuChE), clozapine, 5,5′-dithiobis(2-nitrobenzoic acid) (DTNB), dopamine hydrochloride, fluorescein sodium salt, gadolinium(III) nitrate hexahydrate, γ-aminobutyric acid (GABA), glutamic acid, glycine, methanol, N-(3-(dimethylamino)propyl)-N′-ethylcarbodiimide hydrochloride (EDC), N,N′-(dimethylamino)-pyridine (DMAP), N,N′-dimethylformamide (DMF), N-hydroxysuccinimide (NHS), N,N,N′,N′-tetramethyl-O-(N-succinimidyl)-uroniumtetrafluoroborate (TSTU), potassium carbonate (K2CO3), triethylamine (TEA), trifluoroacetic acid (TFA), and xylenol orange tetrasodium salt were purchased from Sigma-Aldrich (St. Louis, MO). 15-Azido-4,7,10,13-tetraoxapentadecanoic acid was purchased from Alfa Aesar. DO3A tert-butyl ester (t-BOC DO3A) was purchased from Macrocyclics (Plano, TX). Phosphate-buffered saline (PBS) (1×, pH 7.4) and sterilized 0.9% saline solution were purchased from Invitrogen (Carlsbad, CA). Hydrochloric acid (1.0 N) and sodium bicarbonate were purchased from Fisher Scientific (Fair Lawn, NJ). 1,2-Distearoyl-sn-glycero-3-phosphoethanolamine-N-[amino(polyethylene glycol)-2000] (ammonium salt) (DSPE-PEG-amine), 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[dibenzocyclooctyl (polyethylene glycol)-2000] (ammonium salt) (DSPE-PEG-DBCO), 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-550] (ammonium salt) (DSPE-PEG-methoxy), and 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[poly(ethylene glycol)2000-N′-carboxyfluorescein] (ammonium salt) (DSPE-PEG-fluorescein) were purchased from Avanti Polar Lipids, Inc. (Alabaster, AL), and octadecyl rhodamine chloride (R18) was purchased from Thermo Fisher Scientific (Waltham, MA).
Synthesis of pH-Sensitive Contrast Agents
t-BOC DO3A (200 mg, 0.4 mmol) and 2-hydroxy-5-nitrolbenzyl bromide (360 mg, 2.0 mmol) were dissolved and stirred in a mixture of 2 mL of DCM and 2 mL of DMF for 1 h at room temperature. After addition of 600 mg of K2CO3, the resulting suspension was stirred overnight. Supernatant was then collected after centrifugation and reduced by vacuum, and 3 mL of TFA was added dropwise at 0 °C and then stirred overnight after it was allowed to warm to room temperature. TFA was removed by vacuum, and the residue was purified by flash chromatography to yield product (34% after two steps). 1H NMR spectra of the product (Figure S1) were compared31 to verify success of synthesis.
EDC (7.6 mg, 0.04 mmol) and NHS (4.6 mg, 0.04 mmol) in 200 μL of 0.1× PBS, pH 6 was added to pH-sensitive chelates (20 mg, 0.04 mmol) in 3 mL of 0.1× PBS (pH 6) in 10 aliquots. The solution was stirred for 30 min and then added to a solution of DSPE-PEG-amine (6 mg, 0.002 mmol) in 3 mL of 0.1× PBS (pH 7.4). After the pH was adjusted to 7.4, the reaction mixture was stirred overnight at room temperature. Gd(NO3)3·6H2O (36 mg, 0.08 mmol) in 200 μL of DI water was added to the resulting solution in five aliquots, and the pH of the reaction mixture was maintained between 4 and 6 during the addition. The resulting solution was stirred overnight at 40 °C, diluted to 24 mL with DI water, and stored at 4 °C.
Fabrication of pH-Sensitive Nanoparticles
DSPE-PEG-DBCO (0.2 mg, 0.067 μmol) in 20 μL of chloroform was dried in a glass vial before addition of 4 mL of stock solution of DSPE-PEG attached pH-sensitive contrast agent (0.33 μmol). The mixture was sonicated at 20% amplitude for 30 s to redissolve dried DSPE-PEG-DBCO using a Branson digital sonifier (Danbury, CT). Following addition of a mixture of 3 mg of PVC and 6 mg of DOS in 50 μL of THF and 80 μL of DCM, the solution was sonicated at 20% amplitude for 3 min. The resulting nanoparticle suspension was filtered using an Acrodisc syringe filter with 0.45 μm membrane (Pall Cooperation, Ann Arbor, MI), washed by DI water (1 mL × 5) in 100 kDa molecular weight cut off (MWCO) Amicon Ultra centrifugal filters (EMD Millipore, Billerica, MA), and concentrated to 0.1 mL in DI water with the same filters.
The concentrated suspension was treated with 1.5 μL of TEA, vortexed for 3 h, treated with 1 μL of acetic anhydride, and vortexed overnight. The resulting nanoparticle suspension was washed (1 mL × 5) in 100 kDa MWCO spin-filters as mentioned above and then diluted to 0.5 mL with PBS, pH 8.
Conjugation of BuChE to Nanoparticles
A solution of 15-azido-4,7,10,13-tetraoxapentadecanoic acid (30 nmol in 3 μL DMF), TSTU (1.8 mg, 6.0 μmol), and DMAP (1.4 mg, 11 μmol) were dissolved in 8 μL of DMF. The solution was vortexed for 1 h and added to solution of BuChE (0.42 nmol in 292 μL of PBS, pH 8). The mixture was vortexed for another 1 h, washed (1 mL × 5), and diluted to 0.5 mL with PBS, pH 8 in 100 kDa MWCO spin-filters as mentioned above. The modified enzyme solution and 0.5 mL nanoparticle suspension were combined and incubated for 72 h at 4 °C. The suspension was concentrated to 30 μL with 100 kDa MWCO spin-filters as mentioned above before in vitro calibration.
Particle Sizing, Zeta-Potential, and Concentration of Nanoparticle Measurements
The conjugated nanoparticles above were characterized for measurement of particle size and zeta-potential by dynamic light scattering (DLS) using a 90 Plus particle size analyzer (Brookhaven Instruments Corp.). The concentration of nanoparticles was measured using nCS1 nanoparticle analyzer (Spectradyne, Torrance, CA).
Inductively Coupled Plasma Mass Spectrometry (ICP-MS)
To identify the relaxivity (r1) of nanoparticles, we used a Bruker Aurora M90 inductively coupled plasma-mass spectrometer (Bruker Scientific Instruments, Billerica, MA) to determine the amount of Gd(III) bound to the surface of nanoparticles. Standard solutions were prepared by dissolving Gd(NO3)3 in DI water with different concentrations (0, 0.5, 5, 20, and 50 μg/L). The concentrated nanoparticle suspension from previous step was diluted by DI water in ratios of 0.2, 2, 8, and 20 μL/L, and compared with the standard solution to identify the exact amount of bound Gd(III).
Electron Microscopy
Four microliters of sample was pipetted onto a C-Flat (Protochips) holey carbon film then plunge frozen using a Gatan Cp3 CryoPlunge unit. Prepared grids were stored under liquid nitrogen until loaded for imaging in an FEI Arctica CryoFEG-TEM with autoloader. Images were collected using low-dose techniques at 200 kV. Images were analyzed using ImageJ software61 to measure diameter of nanoparticles.
To stain lipid coating of the nanoparticles, diluted nanosensors (5 μL) were placed on a 300 mesh carbon film coated copper grid (Electron Microscopy Sciences) for 1 min. The excessive liquid was removed by a piece of filter paper. The remaining sample on carbon film was stained using 5 μL of methylamine vanadate (Nanovan) for 1 min, and then the excessive liquid was blotted by a filter paper. After two rounds of staining, the images were acquired at 200 kV accelerating voltage using FEI Arctica CryoFEG-TEM.
Relaxivity Test in Low Magnetic Field
ACh-MRNS corresponding to 0.43, 0.21, 0.12. 0.11, and 0.043 mM Gd(III) was suspended in 500 μL of 1× PBS in NMR tubes and then analyzed by a 1.5 T Bruker Minispec mq60 NMR analyzer (60 MHz, Bruker Inc., Billerica, MA) at 37 °C to yield T1 of each sample. 1/T1 was plotted as a function of concentration of Gd(III), and the slope of the plotted curve is r1.
pH Calibration
We added 2 μL of a nanosensor suspension to 198 μL of PBS, pH 6, 6.5, 7, 7.4, and 8 in well plates. The well plate was scanned in a 7 T Bruker Biospec MRI scanner for small animals (Bruker Scientific Instrument, Billerica, MA). A T1-weighted Rapid Acquisition with Relaxation Enhancement with Variable TR (RARE-VTR) sequence (1 slice; 1.0 mm; TE = 12.5 ms, TR = 70, 291, 576, 976, 1651, and 5000 ms, FOV = 40 mm × 40 mm; data matrix 64 × 64) was used to generate a T1 map in about 9.5 min. The signal intensities at different TRs were also collected using Matlab code.
In Vitro Calibration
Sensor calibration was performed in a Bruker coil with an inner diameter of 7.5 cm. A Tripilot scan was initially conducted followed by addition of 2 μL of a nanosensor suspension to 198 μL solution of acetylcholine (0, 50, 100, 250, 500, and 1000 μM) in 1× PBS, pH 7.4 in well plates. The well plate was scanned in a 7 T Bruker Biospec MRI scanner for small animals (Bruker Scientific Instrument, Billerica, MA, USA). A T1-weighted Rapid Acquisition with Relaxation Enhancement with Variable TR (RARE-VTR) sequence (1 slice; 1.0 mm; TE = 12.5 ms, TR = 70, 291, 576, 976, 1651, and 5000 ms, FOV = 40 mm × 40 mm; data matrix 64 × 64) was used to generate a T1 map in about 9.5 min. The signal intensities at different TRs were also collected using Matlab code. To verify that the pH change is a local effect, we also suspended 2 μL pH sensitive nanoparticles without conjugation of enzyme (pH-MRNS) and 8.7 units free BuChE in 198 μL solution of acetylcholine (0, 10, 50, 100, and 500 μM) in PBS, pH 7.4 in well plates. The same sequence was used for the T1 map.
In Vitro Calibration at pH 7.2, 7.4, and 7.8
ACh-MRNS corresponding to 0.04 mM Gd(III) was suspended in 500 μL 1× PBS, pH 7.2, 7.4, and 7.8 with a concentration of acetylcholine varying from 0 to 100 μM in NMR tubes and then analyzed by a 1.5 T Bruker Minispec mq60 NMR analyzer (60 MHz, Bruker, Inc., Billerica, MA) at 37 °C to yield T1 of each sample.
Measurements of Overall pH Change in the Mixture of Nanosensors and Acetylcholine
Fluorescein sodium (5 μg/mL) was mixed with ACh-MRNS (0.2 mM Gd(III)) and pH-MRNS (0.2 mM Gd(III) with 8.7 units of BuChE in 100 μL 1× PBS, pH 7.4, respectively. Then 100 μL of acetylcholine solution with concentrations varying from 0 to 1 mM in 1× PBS, pH 7.4 was added to the mixture. The mixture was excited at 460 nm and read the emission at 520 nm using a SpectraMax Gemini EM plate reader.
Xylenol Orange Test
The test was performed following the procedure reported in the previous literature.62 Specifically, the ACh-MRNS corresponding to 0.1 mM conjugated Gd(III) was incubated with concentrations of acetylcholine varying from 0 to 5 mM in 100 μL HEPES buffer, pH 7.4 for 10 min and then was added with 100 μL of 0.6% (mg/mL) xylenol orange in 50 mM acetic acid buffer, pH 5.4. The absorbance at 573 and 433 nm was read using a SpectraMax Gemini EM plate reader. No significance change in A573/A433 was observed. We also plotted a work curve of the test: 100 μL of 0.6% (w/v) xylenol orange in 50 mM acetic acid buffer, pH 5.4 was added to 100 μL of Gd3(NO3)3 solution in HEPES buffer, pH 7.4 to make the final concentrations of Gd3+ varying from 0 to 100 μM. The absorbance at 573 and 433 nm was read using a SpectraMax Gemini EM plate reader.
Enzymatic Kinetics Studies
DSPE-PEG-methoxy was used to replace pH-sensitive contrast agents conjugated DSPE-PEG-amine to coat nanoparticles to avoid interference from pH-sensitive contrast agent’s absorbance at 400 nm in this study. Ellman’s assay was used to test the catalytic kinetics of conjugated and free BuChE. 8.7 units of BuChE in each form was suspended in 1 mM DTNB in 100 μL of PBS, pH 7.4 in a 96-well plate. Acetylthiocholine in 100 μL of PBS, pH 7.4, was added to the mixture to make final concentration of acetylthiocholine 0, 5, 10, 50, and 100 μM. The absorbance at 412 nm was recorded right after the addition every 5 s for 2 min using a SpectraMax Gemini EM plate reader. To study the overall change of pH led by the enzymatic hydrolysis, we suspended 2 μL of nanoparticles coated by DSPE-PEG-fluorescein and 8.7 units of azide modified BuChE in 100 μL of 1× PBS, pH 7.4. Acetylcholine in 100 μL of 1× PBS, pH 7.4, was added to the mixture to make final concentration of acetylthiocholine 0, 5, 10, 50, 100, 500, and 5000 μM. The fluorescent signal (excitation: 490 nm, emission: 520 nm, cutoff: 515 nm) was recorded right after the addition every 10 s for 10 min using a SpectraMax Gemini EM plate reader.
Selectivity Studies
Two microliters of concentrated nanosensors were suspended in 198 μL of either glutamate (5 mM), dopamine (5 mM), GABA (5 mM), or glycine (5 mM) in PBS, pH 7.4, and scanned with the same coil and sequence used for in vitro calibration. The resulting 1/T1 was compared with the 1/T1 of nanosensors in PBS, pH 7.4 and acetylcholine solution (0.1 mM in 1× PBS, pH 7.4).
Animal Care and Stereotaxic Surgery
Adult male Sprague–Dawley rats (230–300 g) were obtained from Charles River Laboratories (Wilmington, MA). The rats were maintained on a 12:12 h light/dark cycle and allowed access to food and water ad libitum. All procedures were approved by the Northeastern University Institutional Animal Care and Use Committee and were in accordance with the National Institutes of Health guidelines.
Three days prior to MRI experiments, unilateral implantation of 26-gauge plastic guide cannula (Plastics One) aimed at the medial prefrontal cortex (mPFC) was performed on animals using a stereotaxic device (Kopf Instruments) under isoflurane anesthesia. A small incision was made to expose the dorsal surface of the skull and wiped clean to reveal the position of lambda and bregma landmarks. A small hole was drilled into the skull at the coordinate position necessary to gain access to the prefrontal cortex (bregma: + 2.8 mm anterior, + 0.8 mm lateral, + 4.0 mm below the surface of the skull). The cannula was placed in the brain and anchored using plastic screws and dental acrylic. The head-wound was then sutured closed, and topical antibiotic ointment was applied to the wound area. Buprenorphine (0.5 mg/kg) was administrated to reduce pain.
In Vivo Nanosensor Injection and MRI
Animals were first anesthetized with 1–2% isoflurane and placed in a plastic positioning device and a head holder built-in with quadrature transmit/receive volume coil. Infusion of nanosensors into the mPFC was performed by lowering and placing the internal cannula attached to a 10 μL Hamilton syringe via polyethylene tubing filled with nanosensors through the guide cannula, delivering a final volume of 2 μL of nanosensors. The air was first removed prior to nanosensor delivery. The internal-injector cannula protruded 1 mm beyond the guide cannula toward the mPFC.
After delivery, MRI experiments were conducted using a 7 T Bruker Biospec 300 MHz MRI scanner for small animals (same as above for in vitro studies). The design of the positioning device and head holder coil provided complete coverage of the brain from olfactory bulbs to brain stem with excellent B1 field homogeneity. At the beginning of each imaging session, a high-resolution anatomical data set was collected using the RARE-VTR sequence (25 slices; 1.0 mm; TE = 12.5 ms, TR = 513, 800, 1400, 2200, and 6000 ms, FOV = 40mm× 40 mm, data matrix 128 × 128) to assess time-lapse nanosensor response, followed by acquisition of same sequence at multiple time-points (0, 23, 46 min postnanosensor injection). Each scan took about 23 min. For detection of drug-evoked cholinergic transients, subcutaneous injection of clozapine (20 mg/kg) dissolved in PBS, pH 6.5 into the back of rat was administered at the time of nanosensor injection, i.e., just prior to T1 scan at 0 min time point. MRI signal was quantified in regions of interest (ROIs) covering the injection sites. Each ROI volume was defined by a cylinder with a diameter of 1.2 mm in the coronal plane and a thickness of 1 mm along the rostrocaudal axis at the site of injection, centered on the tip of the internal cannula, and computing signal amplitudes normalized with respect to identical control ROIs placed on respective coordinates on the contralateral side of the brain without sensor delivery.
Histological Analysis
To verify cannula placement following MRI contrast agent injection experiments, animals were anesthetized with carbon dioxide and transcardially perfused with a solution of PBS (pH 7.4) with 1% sodium nitrite, followed by 4 wt %/vol paraformaldehyde in 0.1 M phosphate buffer (pH 7.4). Brains were removed, postfixed for 90 min in perfusion fixative, and cryoprotected in a series of 20% and 30% sucrose in 0.1 M phosphate buffer each overnight at 4 °C. Coronal sections of 40 μm thickness across a range extending ~2 mm anterior and posterior to the cannula insertion site was sectioned on a cryostat (Microm HM 550). Standard protocols were used for choline acetyltransferase (ChAT) immunohistochemical and cresyl violet (Nissl) histological staining. Briefly, for ChAT staining, free-floating sections in well-plates were first incubated in 1% H2O2/50% methanol solution for 10 min, followed by serum-blocking buffer for 60 min at room temperature. Sheep polyclonal anti-ChAT antibody (ab18736, Abcam) diluted 1:1,000 in immunobuffer containing 1% normal rabbit serum (16120107, Thermo Fisher) in PBS-0.2% Triton-X100 (PBS-T) was applied and incubated overnight at 4 °C. The sections were then incubated with horseradish peroxidase (HRP)-conjugated rabbit antisheep IgG secondary antibody (818620, Thermo Fisher) diluted 1:1000 in immunobuffer for 2 h at room temperature, and developed in 3,3-diaminobenzidine tetrahydrochloride (DAB) solution (34002, Thermo Fisher). The sections were washed between each step (3 × 5 min) in PBS-T. The sections were then mounted onto 0.5% gelatin/0.05% chrom alum coated glass slides, allowed to air-dry, dehydrated through a series of alcohols (75%, 85%, 95%, 100% twice 5 min each), cleared with xylene, and coverslipped with Permount (Fischer Scientific, Pittsburgh, PA). The sections were viewed with Olympus BX51 light microscope. Sections processed to determine nonspecific staining by following the same procedures, but with omission of the primary antibody, showed no immunohistochemical labeling.
Diffusion of the Fluorescent Nanosensors
The fluorescent nanosensors were fabricated as the pH-MRNS except for 0.001 mg R18 was incorporated to the mixture of PVC, DOS and THF. Two microliters of nanosensors containing 47 μM of Gd was delivered into a phantom made of 0.6% agarose. The follow-up imaging was performed on IVIS Lumina II (PerkinElmer) small animal imager in fluorescence mode with a 535 nm excitation filter and DSRed emission filter at 0, 2, 5, 10, 15, 20, 30, 40, 50, and 60 min post injection for 1 h. To obtain quantitative information about nanosensor diffusion, circular ROI was positioned centered near the tip of injection site and fluorescence signal amplitudes per unit area (mm2), normalized with respect to signal intensity at t = 0 was acquired using ImageJ software.61 Relative intensity from three sets of identical ROIs were obtained and averaged for the each time point.
Acetylcholine Signal Analysis and Group Sizes
Data for final analysis were extracted from 19 animals divided into three main groups: ACh-MRNS (N = 6); control (N = 6), and pH-MRNS (N = 3) groups. Only animals with correctly placed cannula tips into the mPFC, as judged after surgery from MRI scans and histological analyses, were included in the data analysis. Based on this criteria, four animals with incorrectly placed cannula were excluded from the final analysis. No randomization was conducted to determine allocation of different animal groups for the MRI scan procedure. Instead, animals were split to both experimental and control groups in each procedure. Prior to ROI image analyses, individual cases were assigned with a random number to ensure analyses were conducted in a blinded fashion.
Image and Data Analysis
All data analysis and image processing was performed with Bruker Paravision 5.1 software (Billerica, MA), Matlab (Mathworks, Natick, MA), and itk-SNAP.63 Images were reconstructed and analyzed using custom routines running in Matlab. Relaxivities were calculated from T1 obtained from itk-SNAP and concentration of Gd(III). Graphs and illustrations were compiled using Origin (OriginLab, Northampton, MA) and Illustrator (Adobe, San Jose, CA), respectively.
Statistical Analysis
All of the relaxivities, T1, sizes, and zeta-potentials collected from the in vitro experiments are average of three separate studies using different batches of ACh-MRNS or pH-MRNS. The T1 collected from the in vivo experiments are average of T1 collected from six rats in the control group, six rats in the experiment group, and three rats in pH-MRNS group using different batches of ACh-MRNS or pH-MRNS.
The differences between groups for the selectivity study was calculated using one-way analysis of variance (ANOVA) and adjusted using Tukey’s HSD for multiple comparisons. The Kolmogorov–Smirnov normality test was used to test for normality. Differences between each group obtained from in vivo data were calculated using Student’s t-test. The α level for all statistical analyses was set at 0.05. Sample size was chosen based on previous reports on MRI method development4 and drug concentrations.43 When P < 0.05, the difference was considered as significant.
Errors (Table 1 and 2, Figures 3B,C, B, and 5C,D and Figure S4) were propagated from standard deviation (SD) of T1 (σT1) or normalized T1 (σ(normalized T1) using formula derived from previous reports.64
The equation used for Tables 1 and 2 is defined as
where [Gd] = concentration of gadolinium (mM).
The equation used for Figures 3B,C and 4B is defined as
The equation used for Figures 5C,D and S4 is defined as
where normalized T1 = T1 of each animal normalized to 0 min postnanosensor injection.
Supplementary Material
Acknowledgments
This work was supported by the National Institutes of Health through Grant No. R01NS081641 (to H.A.C.). We thank K. Bardon, P. Larese-Casanova, and C. Marks for technical assistance with flash column chromatography, ICP-MS, and TEM, respectively. We also thank C. Ferris and P. Kulkarni for help with the MRI setup and pulse sequences.
Footnotes
Notes
The authors declare no competing financial interest.
The Supporting Information is available free of charge on the ACS Publications website at DOI: 10.1021/acsnano.8b01640.
Fabrication of pH-MRNS and ACh-MRNS, structure and 1H NMR spectrum of the pH-sensitive chelator, TEM image of the ACh-MRNS using NanoVan stain, in vitro nanosensor to pH dependence, pH change in the mixture of the nanosensor and acetylcholine, xylenol orange test, kinetics of BuChE, relative signal intensity at different TR, histology, and diffusion of pH-MRNS in phantom brain (Figures S1–S11) (PDF)
References
- 1.Young SH, Poo MM. Spontaneous Release of Transmitter from Growth Cones of Embryonic Neurones. Nature. 1983;305:634–637. doi: 10.1038/305634a0. [DOI] [PubMed] [Google Scholar]
- 2.Nguyen QT, Schroeder LF, Mank M, Muller A, Taylor P, Griesbeck O, Kleinfeld D. An in Vivo Biosensor for Neurotransmitter Release and in Situ Receptor Activity. Nat Neurosci. 2010;13:127–132. doi: 10.1038/nn.2469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bartelle BB, Barandov A, Jasanoff A. Molecular Fmri. J Neurosci. 2016;36:4139–4148. doi: 10.1523/JNEUROSCI.4050-15.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shapiro MG, Westmeyer GG, Romero PA, Szablowski JO, Kuster B, Shah A, Otey CR, Langer R, Arnold FH, Jasanoff A. Directed Evolution of a Magnetic Resonance Imaging Contrast Agent for Noninvasive Imaging of Dopamine. Nat Biotechnol. 2010;28:264–270. doi: 10.1038/nbt.1609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee T, Cai LX, Lelyveld VS, Hai A, Jasanoff A. Molecular-Level Functional Magnetic Resonance Imaging of Dopaminergic Signaling. Science. 2014;344:533–535. doi: 10.1126/science.1249380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brustad EM, Lelyveld VS, Snow CD, Crook N, Jung ST, Martinez FM, Scholl TJ, Jasanoff A, Arnold FH. Structure-Guided Directed Evolution of Highly Selective P450-Based Magnetic Resonance Imaging Sensors for Dopamine and Serotonin. J Mol Biol. 2012;422:245–262. doi: 10.1016/j.jmb.2012.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mishra A, Mishra R, Gottschalk S, Pal R, Sim N, Engelmann J, Goldberg M, Parker D. Microscopic Visualization of Metabotropic Glutamate Receptors on the Surface of Living Cells Using Bifunctional Magnetic Resonance Imaging Probes. ACS Chem Neurosci. 2014;5:128–137. doi: 10.1021/cn400175m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Oukhatar F, Meme S, Meme W, Szeremeta F, Logothetis NK, Angelovski G, Toth E. Mri Sensing of Neurotransmitters with a Crown Ether Appended Gd(3+) Complex. ACS Chem Neurosci. 2015;6:219–225. doi: 10.1021/cn500289y. [DOI] [PubMed] [Google Scholar]
- 9.Davis KA, Nanga RP, Das S, Chen SH, Hadar PN, Pollard JR, Lucas TH, Shinohara RT, Litt B, Hariharan H, Elliott MA, Detre JA, Reddy R. Glutamate Imaging (Glucest) Lateralizes Epileptic Foci in Nonlesional Temporal Lobe Epilepsy. Sci Transl Med. 2015;7:309ra161. doi: 10.1126/scitranslmed.aaa7095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cai K, Haris M, Singh A, Kogan F, Greenberg JH, Hariharan H, Detre JA, Reddy R. Magnetic Resonance Imaging of Glutamate. Nat Med. 2012;18:302–306. doi: 10.1038/nm.2615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Smith BR, Gambhir SS. Nanomaterials for in Vivo Imaging. Chem Rev. 2017;117:901–986. doi: 10.1021/acs.chemrev.6b00073. [DOI] [PubMed] [Google Scholar]
- 12.Andreozzi E, Wang P, Valenzuela A, Tu C, Gorin F, Dhenain M, Louie A. Size-Stable Solid Lipid Nanoparticles Loaded with Gd-Dota for Magnetic Resonance Imaging. Bioconjugate Chem. 2013;24:1455–1467. doi: 10.1021/bc300605f. [DOI] [PubMed] [Google Scholar]
- 13.Zhao J, Zhang B, Shen S, Chen J, Zhang Q, Jiang X, Pang Z. Creka Peptide-Conjugated Dendrimer Nanoparticles for Glioblastoma Multiforme Delivery. J Colloid Interface Sci. 2015;450:396–403. doi: 10.1016/j.jcis.2015.03.019. [DOI] [PubMed] [Google Scholar]
- 14.Zhang HW, Wang LQ, Xiang QF, Zhong Q, Chen LM, Xu CX, Xiang XH, Xu B, Meng F, Wan YQ, Deng DY. Specific Lipase-Responsive Polymer-Coated Gadolinium Nanoparticles for Mr Imaging of Early Acute Pancreatitis. Biomaterials. 2014;35:356–367. doi: 10.1016/j.biomaterials.2013.09.046. [DOI] [PubMed] [Google Scholar]
- 15.Park YI, Kim HM, Kim JH, Moon KC, Yoo B, Lee KT, Lee N, Choi Y, Park W, Ling D, Na K, Moon WK, Choi SH, Park HS, Yoon SY, Suh YD, Lee SH, Hyeon T. Theranostic Probe Based on Lanthanide-Doped Nanoparticles for Simultaneous in Vivo Dual-Modal Imaging and Photodynamic Therapy. Adv Mater. 2012;24:5755–5761. doi: 10.1002/adma.201202433. [DOI] [PubMed] [Google Scholar]
- 16.Gundelfinger ED, Kessels MM, Qualmann B. Temporal and Spatial Coordination of Exocytosis and Endocytosis. Nat Rev Mol Cell Biol. 2003;4:127–139. doi: 10.1038/nrm1016. [DOI] [PubMed] [Google Scholar]
- 17.Sarter M, Parikh V, Howe WM. Phasic Acetylcholine Release and the Volume Transmission Hypothesis: Time to Move On. Nat Rev Neurosci. 2009;10:383–390. doi: 10.1038/nm2635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Harris KD, Thiele A. Cortical State and Attention. Nat Rev Neurosci. 2011;12:509–523. doi: 10.1038/nrn3084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hasselmo ME, Bower JM. Acetylcholine and Memory. Trends Neurosci. 1993;16:218–222. doi: 10.1016/0166-2236(93)90159-j. [DOI] [PubMed] [Google Scholar]
- 20.Everitt BJ, Robbins TW. Central Cholinergic Systems and Cognition. Annu Rev Psychol. 1997;48:649–684. doi: 10.1146/annurev.psych.48.1.649. [DOI] [PubMed] [Google Scholar]
- 21.Lewis DA, Lieberman JA. Catching up on Schizophrenia: Natural History and Neurobiology. Neuron. 2000;28:325–334. doi: 10.1016/s0896-6273(00)00111-2. [DOI] [PubMed] [Google Scholar]
- 22.Manji HK, Drevets WC, Charney DS. The Cellular Neurobiology of Depression. Nat Med. 2001;7:541–547. doi: 10.1038/87865. [DOI] [PubMed] [Google Scholar]
- 23.Mattson MP. Pathways Towards and Away from Alzheimer’s Disease. Nature. 2004;430:631–639. doi: 10.1038/nature02621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bolan PJ, Meisamy S, Baker EH, Lin J, Emory T, Nelson M, Everson LI, Yee D, Garwood M. In Vivo Quantification of Choline Compounds in the Breast with 1h Mr Spectroscopy. Magn Reson Med. 2003;50:1134–1143. doi: 10.1002/mrm.10654. [DOI] [PubMed] [Google Scholar]
- 25.Baik HM, Su MY, Yu H, Mehta R, Nalcioglu O. Quantification of Choline-Containing Compounds in Malignant Breast Tumors by 1h Mr Spectroscopy Using Water as an Internal Reference at 1. 5 T. MAGMA. 2006;19:96–104. doi: 10.1007/s10334-006-0032-4. [DOI] [PubMed] [Google Scholar]
- 26.Mitchell KM. Acetylcholine and Choline Amperometric Enzyme Sensors Characterized in Vitro and in Vivo. Anal Chem. 2004;76:1098–1106. doi: 10.1021/ac034757v. [DOI] [PubMed] [Google Scholar]
- 27.Conde F, Maire-Lepoivre E, Audinat E, Crepel F. Afferent Connections of the Medial Frontal Cortex of the Rat. Ii. Cortical and Subcortical Afferents. J Comp Neurol. 1995;352:567–593. doi: 10.1002/cne.903520407. [DOI] [PubMed] [Google Scholar]
- 28.Caravan P, Ellison JJ, McMurry TJ, Lauffer RB. Gadolinium(Iii) Chelates as Mri Contrast Agents: Structure, Dynamics, and Applications. Chem Rev. 1999;99:2293–2352. doi: 10.1021/cr980440x. [DOI] [PubMed] [Google Scholar]
- 29.Cash KJ, Li C, Xia J, Wang LV, Clark HA. Optical Drug Monitoring: Photoacoustic Imaging of Nanosensors to Monitor Therapeutic Lithium in Vivo. ACS Nano. 2015;9:1692–1698. doi: 10.1021/nn5064858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ruckh TT, Skipwith CG, Chang W, Senko AW, Bulovic V, Anikeeva PO, Clark HA. Ion-Switchable Quantum Dot Forster Resonance Energy Transfer Rates in Ratiometric Potassium Sensors. ACS Nano. 2016;10:4020–4030. doi: 10.1021/acsnano.5b05396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Woods M, Kiefer GE, Bott S, Castillo-Muzquiz A, Eshelbrenner C, Michaudet L, McMillan K, Mudigunda SD, Ogrin D, Tircso G, Zhang S, Zhao P, Sherry AD. Synthesis, Relaxometric and Photophysical Properties of a New Ph-Responsive Mri Contrast Agent: The Effect of Other Ligating Groups on Dissociation of a P-Nitrophenolic Pendant Arm. J Am Chem Soc. 2004;126:9248–9256. doi: 10.1021/ja048299z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Teng TL, Harpst JA, Lee JC, Zinn A, Carlson DM. Composition and Molecular Weights of Butyrylcholinesterase from Horse Serum. Arch Biochem Biophys. 1976;176:71–81. doi: 10.1016/0003-9861(76)90142-9. [DOI] [PubMed] [Google Scholar]
- 33.Lee SM, Song Y, Hong BJ, MacRenaris KW, Mastarone DJ, O’Halloran TV, Meade TJ, Nguyen ST. Modular Polymer-Caged Nanobins as a Theranostic Platform with Enhanced Magnetic Resonance Relaxivity and Ph-Responsive Drug Release. Angew Chem, Int Ed. 2010;49:9960–9964. doi: 10.1002/anie.201004867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Grogna M, Cloots R, Luxen A, Jerome C, Desreux JF, Detrembleur C. Design and Synthesis of Novel Dota(Gd3+)-Polymer Conjugates as Potential Mri Contrast Agents. J Mater Chem. 2011;21:12917–12926. [Google Scholar]
- 35.Caravan P, Farrar CT, Frullano L, Uppal R. Influence of Molecular Parameters and Increasing Magnetic Field Strength on Relaxivity of Gadolinium- and Manganese-Based T1 Contrast Agents. Contrast Media Mol Imaging. 2009;4:89–100. doi: 10.1002/cmmi.267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chesler M. Regulation and Modulation of Ph in the Brain. Physiol Rev. 2003;83:1183–1221. doi: 10.1152/physrev.00010.2003. [DOI] [PubMed] [Google Scholar]
- 37.Augustinsson KL. Butyryl- and Propionylcholinesterases and Related Types of Eserine-Sensitive Esterases. In: Boyer PD, Lardy H, Myrback K, editors. The Enzymes. 2. Vol. 4. Academic Press; New York: 1960. pp. 521–540. [Google Scholar]
- 38.Hoa DT, Kumar TNS, Punekar NS, Srinivasa RS, Lal R, Contractor AQ. Biosensor Based on Conducting Polymers. Anal Chem. 1992;64:2645–2646. [Google Scholar]
- 39.Nishizawa M, Matsue T, Uchida I. Penicillin Sensor Based on a Microarray Electrode Coated with Ph-Responsive Polypyrrole. Anal Chem. 1992;64:2642–2644. doi: 10.1021/ac00045a030. [DOI] [PubMed] [Google Scholar]
- 40.Luo XL, Xu JJ, Zhao W, Chen HY. A Novel Glucose Enfet Based on the Special Reactivity of Mno2 Nanoparticles. Biosens Bioelectron. 2004;19:1295–1300. doi: 10.1016/j.bios.2003.11.019. [DOI] [PubMed] [Google Scholar]
- 41.Walsh R, Morales JM, Skipwith CG, Ruckh TT, Clark HA. Enzyme-Linked DNA Dendrimer Nanosensors for Acetylcholine. Sci Rep. 2015;5:14832. doi: 10.1038/srep14832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ferris CF, Kulkarni P, Sullivan JM, Jr, Harder JA, Messenger TL, Febo M. Pup Suckling Is More Rewarding Than Cocaine: Evidence from Functional Magnetic Resonance Imaging and Three-Dimensional Computational Analysis. J Neurosci. 2005;25:149–156. doi: 10.1523/JNEUROSCI.3156-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ichikawa J, Dai J, O’Laughlin IA, Fowler WL, Meltzer HY. Atypical, but Not Typical, Antipsychotic Drugs Increase Cortical Acetylcholine Release without an Effect in the Nucleus Accumbens or Striatum. Neuropsychopharmacology. 2002;26:325–339. doi: 10.1016/S0893-133X(01)00312-8. [DOI] [PubMed] [Google Scholar]
- 44.Fritze J, Elliger T. Pirenzepine for Clozapine-Induced Hypersalivation. Lancet. 1995;346:1034. doi: 10.1016/s0140-6736(95)91713-6. [DOI] [PubMed] [Google Scholar]
- 45.Michal P, Lysikova M, El-Fakahany EE, Tucek S. Clozapine Interaction with the M2 and M4 Subtypes of Muscarinic Receptors. Eur J Pharmacol. 1999;376:119–125. doi: 10.1016/s0014-2999(99)00341-6. [DOI] [PubMed] [Google Scholar]
- 46.Bymaster FP, Calligaro DO, Falcone JF, Marsh RD, Moore NA, Tye NC, Seeman P, Wong DT. Radioreceptor Binding Profile of the Atypical Antipsychotic Olanzapine. Neuropsychopharmacology. 1996;14:87–96. doi: 10.1016/0893-133X(94)00129-N. [DOI] [PubMed] [Google Scholar]
- 47.Harrison J, Bartlett CA, Cowin G, Nicholls PK, Evans CW, Clemons TD, Zdyrko B, Luzinov IA, Harvey AR, Iyer KS, Dunlop SA, Fitzgerald M. In Vivo Imaging and Biodistribution of Multimodal Polymeric Nanoparticles Delivered to the Optic Nerve. Small. 2012;8:1579–1589. doi: 10.1002/smll.201102648. [DOI] [PubMed] [Google Scholar]
- 48.Hutter E, Boridy S, Labrecque S, Lalancette-Hebert M, Kriz J, Winnik FM, Maysinger D. Microglial Response to Gold Nanoparticles. ACS Nano. 2010;4:2595–2606. doi: 10.1021/nn901869f. [DOI] [PubMed] [Google Scholar]
- 49.Choi J, Zheng Q, Katz HE, Guilarte TR. Silica-Based Nanoparticle Uptake and Cellular Response by Primary Microglia. Environ Health Perspect. 2010;118:589–595. doi: 10.1289/ehp.0901534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Giuliano C, Parikh V, Ward JR, Chiamulera C, Sarter M. Increases in Cholinergic Neurotransmission Measured by Using Choline-Sensitive Microelectrodes: Enhanced Detection by Hydrolysis of Acetylcholine on Recording Sites? Neurochem Int. 2008;52:1343–1350. doi: 10.1016/j.neuint.2008.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Parikh V, Kozak R, Martinez V, Sarter M. Prefrontal Acetylcholine Release Controls Cue Detection on Multiple Timescales. Neuron. 2007;56:141–154. doi: 10.1016/j.neuron.2007.08.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Parikh V, Pomerleau F, Huettl P, Gerhardt GA, Sarter M, Bruno JP. Rapid Assessment of in Vivo Cholinergic Transmission by Amperometric Detection of Changes in Extracellular Choline Levels. Eur J Neurosci. 2004;20:1545–1554. doi: 10.1111/j.1460-9568.2004.03614.x. [DOI] [PubMed] [Google Scholar]
- 53.Gao Y, Erokwu BO, DeSantis DA, Croniger CM, Schur RM, Lu L, Mariappuram J, Dell KM, Flask CA. Initial Evaluation of Hepatic T1 Relaxation Time as an Imaging Marker of Liver Disease Associated with Autosomal Recessive Polycystic Kidney Disease (Arpkd) NMR Biomed. 2016;29:84–89. doi: 10.1002/nbm.3442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Herrmann K, Erokwu BO, Johansen ML, Basilion JP, Gulani V, Griswold MA, Flask CA, Brady-Kalnay SM. Dynamic Quantitative T1Mapping in Orthotopic Brain Tumor Xenografts. Transl Oncol. 2016;9:147–154. doi: 10.1016/j.tranon.2016.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Vykhodtseva N, McDannold N, Hynynen K. Progress and Problems in the Application of Focused Ultrasound for Blood-Brain Barrier Disruption. Ultrasonics. 2008;48:279–296. doi: 10.1016/j.ultras.2008.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hynynen K, McDannold N, Sheikov NA, Jolesz FA, Vykhodtseva N. Local and Reversible Blood-Brain Barrier Disruption by Noninvasive Focused Ultrasound at Frequencies Suitable for Trans-Skull Sonications. NeuroImage. 2005;24:12–20. doi: 10.1016/j.neuroimage.2004.06.046. [DOI] [PubMed] [Google Scholar]
- 57.Muldoon LL, Sandor M, Pinkston KE, Neuwelt EA. Imaging, Distribution, and Toxicity of Superparamagnetic Iron Oxide Magnetic Resonance Nanoparticles in the Rat Brain and Intracerebral Tumor. Neurosurgery. 2005;57:785–796. doi: 10.1093/neurosurgery/57.4.785. [DOI] [PubMed] [Google Scholar]
- 58.Patel T, Zhou J, Piepmeier JM, Saltzman WM. Polymeric Nanoparticles for Drug Delivery to the Central Nervous System. Adv Drug Delivery Rev. 2012;64:701–705. doi: 10.1016/j.addr.2011.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Saraiva C, Praca C, Ferreira R, Santos T, Ferreira L, Bernardino L. Nanoparticle-Mediated Brain Drug Delivery: Overcoming Blood-Brain Barrier to Treat Neurodegenerative Diseases. J Controlled Release. 2016;235:34–47. doi: 10.1016/j.jconrel.2016.05.044. [DOI] [PubMed] [Google Scholar]
- 60.Darvesh S, Hopkins DA, Geula C. Neurobiology of Butyrylcholinesterase. Nat Rev Neurosci. 2003;4:131–138. doi: 10.1038/nrn1035. [DOI] [PubMed] [Google Scholar]
- 61.Schneider CA, Rasband WS, Eliceiri KW. Nih Image to Imagej: 25 Years of Image Analysis. Nat Methods. 2012;9:671–675. doi: 10.1038/nmeth.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Barge A, Cravotto G, Gianolio E, Fedeli F. How to Determine Free Gd and Free Ligand in Solution of Gd Chelates. A Technical Note. Contrast Media Mol Imaging. 2006;1:184–188. doi: 10.1002/cmmi.110. [DOI] [PubMed] [Google Scholar]
- 63.Yushkevich PA, Piven J, Hazlett HC, Smith RG, Ho S, Gee JC, Gerig G. User-Guided 3d Active Contour Segmentation of Anatomical Structures: Significantly Improved Efficiency and Reliability. NeuroImage. 2006;31:1116–1128. doi: 10.1016/j.neuroimage.2006.01.015. [DOI] [PubMed] [Google Scholar]
- 64.Tellinghuisen J. Statistical Error Propagation. J Phys Chem A. 2001;105:3917–3921. [Google Scholar]
- 65.Paxinos G, Watson C. The Rat Brain in Stereotaxic Coordinates. 6. Academic Press, Elsevier; 2009. Compact. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.