Abstract
Background/Aims
The aim of this study was to investigate the influence of a synbiotic preparation (a mixture of six probiotics and a prebiotic) on aberrant crypt foci (ACF) formation, dysplasia, inflammation, and colitis-like lesions in experimental colon cancer in rats.
Materials and Methods
Sixty male rats were categorized into three groups of 20 animals each. Group A was administered 1,2-dimethylydrazine, 15 mg/kg body weight (BW), once a week for 2 weeks. Group B was administered 1,2-dimethylydrazine at the same dose plus synbiotic, started after the second dose of carcinogen and lasted for 5 weeks. Group C was administered synbiotic plus carcinogen from the beginning of the experiment and lasted for 7 weeks. Animals were killed at the end of week 7.
Results
At the end of the experiment, the animals that received carcinogen plus the synbiotic had 100%, whereas the animals that received only carcinogen has 70% survival. Animals of groups B and C had significantly lower percentage of inflammation, colitis-like lesions, and ACF dysplasia than animals of group A, whereas those of group C had the least pathological lesions.
Conclusion
Synbiotics seem to protect against the appearance of preneoplastic colon lesions in rats. The results of this experimental study suggest that treatment with a synbiotic preparation exerts significant antimutagenic properties against the development of preneoplastic lesions in rats.
Keywords: Colitis, experimental colon cancer, prebiotics, probiotics, synbiotics
INTRODUCTION
Strategies of colorectal cancer (CRC) prevention by means of dietary restrictions and modifications have received much attention during the last years. Calcium, selenium, and other foods and food elements have been shown to be beneficial in clinical and epidemiological studies (1,2). In contrast, experimental evidence suggests that the ingestion of probiotics (living organisms believed to have a beneficial effect on health) or prebiotics (non-digestible food ingredients that promote the growth of beneficial-resident bacteria) have a significantly positive effect on precancerous colonic lesions and CRC development (3–10). The administration of synbiotics (combination of pro -and prebiotics) could theoretically have a synergistic protective effect against the development and progression of CRC via mechanisms including the decrease of intestinal inflammation, enhancement of immune function and anti-tumorigenic activity, binding to potential food carcinogens, and reduction in bacterial enzymes that hydrolyze precarcinogenic compounds. There are actually some experimental data concerning the influence of synbiotic administration in animals as well as in humans, although few conclusive data could be drawn (11–19). Some of these probiotics have already been used in clinical trials (20). All of these strains are commercially available in various forms.
The aim of this study was to investigate the influence of the administration of a synbiotic preparation (a mixture of six probiotics and a prebiotic) on aberrant crypt foci (ACF) formation, dysplasia, inflammation, and colitis-like lesions in experimental colon cancer in rats. In this experimental model, we used a synbiotic preparation, instead of only pro -or prebiotics, because we anticipated better results from the combination, compared to the administration of only probiotics or prebiotics because of their synergistic effect on the gut microflora.
MATERIALS AND METHODS
The experiments were approved by the veterinary authorities of East Attica Region in accordance with the Greek law No 160/91 and the European Community regulations. (License for experimentation: Ref number 695, and renewal 805. Directive 609/1986-East Attica -Athens Veterinary Authority). All animal experiments conform to institutional guidelines. All co-authors are in agreement with the content of this article.
Experimental animals
Adult male Wistar rats, weighing 200–240 g, were allowed to adapt to our laboratory conditions 1 week prior to the experiment. They were housed individually in cages maintained at a constant temperature (22°C) and in a 12-h day/night cycle with ad libitum food and water. A total of 60 rats were used.
Experimental groups
Experimental animals were categorized into 3 groups of 20 animals each.
Group A received only treatment with 1,2-dimethylydrazinedihydrochloride (D161802; Sigma-Aldrich) subcutaneously in a dose of 15 mg/kg body weight (BW), once a week for 2 weeks.
Group B received treatment with 1,2-dimethylydrazine dihydrochloride 15 mg/kg BW subcutaneously once a week for 2 weeks plus synbiotic. Synbiotic administration in this group began after the second dose of carcinogen.
Group C received synbiotic plus carcinogen starting from the beginning of the experiment, for a total of 7 weeks.
Synbiotic preparation
A mixture of six probiotics namely Lactobacillus acidophilus, Bifidobacterium sp., Streptococcus thermophilus, Lactobacillus casei, L. delbrueckii subsp Bulgaricus. and Bifidobacterium longum, in a dose of 4’108 viable cells/g diet and a prebiotic (chicory fructo-oligosaccharides (FOS). All the products were provided by the pharmaceutical company Elpen Pharma, Pikermi, Greece. The proportion of bacteria in the mixture was equal. S. thermophilus is classified as a lactic acid bacterium that can be found in fermented milk products and is used in the production of yogurt along with L. delbrueckii subsp Bulgaricus. The two species are synergistic, providing folic and formic acids. The dose of 5% w/w in diet per day was administered per os to each animal (1 mL in 50 mL water/animal).
Chicory FOS are obtained through partial inulin enzymatic hydrolysis extracted with hot water from the chicory root (Cichorium intybus). FOS are oligosaccharides that belong to soluble dietary fiber that is found in several plants including chicory. They consist of linear chains of fructose units, linked by beta bonds. Dietary FOS are not cleaved by small intestinal glycosidases, thus reaching the large bowel intact. Subsequently, they are metabolized by the intestinal microbiota to form a number of metabolites, including short-chain fatty acids. FOS have important beneficial physiological effects. Therefore, they are included in various food products owing to their prebiotic effect stimulating the growth of non-pathogenic intestinal microflora (21).
All live animals were sacrificed at the end of week 7 (49th day).
Histology
For histologic evaluation, we examined two tissue samples from the large bowel of each animal, from all the groups (A, B, and C). The two samples were obtained from the proximal and distal large bowels, respectively. Tissues were fixed in 10% neutral phosphate-buffered formalin and embedded in paraffin for routine histology, which was totally blinded for the three pathologists. Multiple 5-μm sections were cut and stained with hematoxylin and eosin (H&E).
Histological criteria for ACF were those generally accepted as ACF without dysplasia (normal mucosa or hyperplastic), ACF with dysplasia, and ACF with mixed type of hyperplasia and dysplasia (22). ACF comprises a single cluster or clusters of abnormally large crypts of the colon mucosa. They can be elevated from the normal surrounding mucosa, with a dilated or slit-like opening and with a thick epithelial lining that stains darker-than-normal crypts with a large pericryptal zone (23). Crypts with dysplasia have ≥1 abnormal cytologic features viz., nuclear enlargement, stratification, pleomorphism, hyperchromasia, or increased nuclear-to-cytoplasmic ratios extending to the mucosa surface, with the absence or presence of architectural distortion (24). The location of the ACF in the distal than the proximal colon and the immunohistochemical profile and genetic alterations strongly suggests a relationship between ACF and colon cancer. Colitis-like lesions comprised crypts and eosinophilic infiltration. So, the most important features evaluated in this experiment were ACF, inflammation, and colitis-like lesions.
Statistical analysis
The statistical software Statistical Package for Social Sciences, version 17.0 (SPSS Inc.; Chicago, IL, USA), was used. Descriptive data are expressed as mean±standard deviation. In this analysis, either Pearson’s chi-squared test or Student’s t-test was applied. A difference between groups was considered to be significant at p <0.05.
RESULTS
Clinical results
At the end of the experiment, all the animals that received carcinogen and synbiotics had 100% survival compared with 70% survival of animals that received only carcinogen (p=0.001) (Figure 1).
Figure 1.

All animals that have received were alive compared with 14 of 20 animals that have received only carcinogen (p=0.001)
No significant alterations in the body weight of the animals before and at the end of the experiment were noted (Table 1).
Table 1.
Alterations in the body weights of the three groups of animals before and at the end of the experiment
| Group A (*) (carcinogen only) | Group B (carcinogen plus synbiotic) | Group C (carcinogen plus synbiotic from the beginning of the experiment) | |||
|---|---|---|---|---|---|
|
| |||||
| Start | End | Start | End | Start | End |
| 338+/−69 | 358+/−75 | 306+/−52 | 344+/−21 | 332+/−23 | 333+/−44 |
Because of the phenomenon of cannibalism appearing in the first group, the body weight calculation was possible only for the survived (14) animals.
Histological results
The most important features, concerning the histological evaluation of the whole experiment, are as follows:
In group A, normal colonic mucosa was noticed in 7.1% (1 of 14), inflammation of the bowel in 86% (12 of 14), and colitis-like lesions and ACF with dysplasia in 28.5% (4 of 14) of the animals, meaning that one or more lesions could be simultaneously found on histology.
In group B, normal colonic mucosa was noted in 25% (5 of 20), inflammation in 40% (8 of 20), colitis-like lesions in 5% (1 of 20), and ACF with dysplasia in 5% (1 of 20) of the animals.
In group C, normal colonic mucosa was noticed in 35% (7 of 20) and inflammation in 30% (6 of 20). No evidence of colitis-like lesions and/or ACF with dysplasia in any one of animals of this group was observed.
Comparison of results
Group C versus group A
Comparing group C (animals treated with carcinogen and synbiotics from the beginning of the experiment) with group A (group with carcinogen only), we observed that the animals of group C had significantly more normal-appearing crypts (p=0.044) and less inflammation (p<0.0001) in the bowel mucosa (Figure 2).
Figure 2.

Group C versus Group A: Normal-appearing crypts were found in 38% of the animals in Group C and 7% in Group A (p=0.044); inflammation was found in 28% of the animals in Group C and 86% in Group A (p<0.0001)
However, the most important feature was the complete absence of colitis-like lesions (p<0.019) and ACF with dysplasia (p<0.058) in group C as compared with group A (Figure 3).
Figure 3.

Group C versus Group A Colitis-like lesion was found in 29% (p<0.019) and ACF with dysplasia in 23% in Group A (p<0.058) None of these lesions were found in Group C
Group B versus group A
Comparing group B with group A, we observed that animals that were treated with synbiotics started after the second dose of the carcinogen had significantly more normal colonic crypts (p<0.044) and less inflammation (Figure 4).
Figure 4.

Group B versus Group A Normal mucosa was found in 23% of the animals in Group B and 7% in Group A (p<0.044) Inflammation was detected in 40% of the animals in Group B and in 86% in Group A (p<0.0001)
The percentage of colitis-like lesions and ACF with dysplasia in animals of group B was lower than those of the control group (p<0.019 and p<0.058, respectively) (Figure 5).
Figure 5.

Group B versus Group A Colitis-like lesions were detected in 5% of the animals in Group B and 29% in Group A (p<0.019). ACF with dysplasia was seen in 3% of the animals in Group B and 23% in Group A (p=0.058)
Group C versus group B
Comparing group C with group B, we found that animals that were treated with carcinogen and synbiotic from the beginning of the experiment had the least pathological features as compared to the animals that were treated with synbiotic administered after the second dose of the carcinogen. So, normal mucosa was found in 35% of the animals in group C and in 25% in group B (p=0.49). Inflammation was detected in 30% of the animals in group C and in 40% in group B (p=0.51) (Figure 6).
Figure 6.
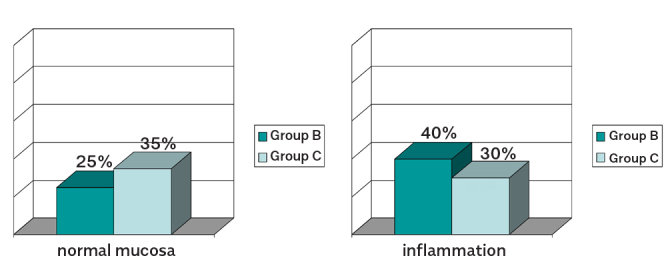
Group C versus Group B Normal mucosa was found in 35% of the animals in Group C and in 25% in Group B (p=0.0044) Inflammation was detected in 30% of the animals in Group C and in 40% in Group B (p<0.0001)
Colitis-like lesions and ACF dysplasia were detected in 5% of the animals in group B. There was no such observation in group C (Figure 7).
Figure 7.

Group C versus Group B: Colitis-like lesions were detected in 5% and ACF dysplasia in 5% of the animals in Group B There was no such observation in Group C
Other histological features
Other histological features recognized were vasculitis and ACF with hyperplasia, but the occurrence these lesions were not statistically significant. Figures 8–11 show some characteristic lesions observed in the three groups of animals.
Figure 8.

Abnormally large crypts with dilated or slit-like opening (long arrow), architectural distortion, and abnormal cytologic features such as hyperchromasia and increased nuclear to cytoplasmic ratio (short arrows); HE stain; 100×
Figure 9.

Slit-like opening of large abnormal crypts that shows cell stratification, nuclear enlargement, and increased nuclear-to-cytoplasmic ratio (thick arrows) in comparison with normal crypts (thin arrows)
Figure 10.

Abnormal crypts with architectural distortion consists of cells that shows nuclear enlargement, hyperchromasia, and increased nuclear-to-cytoplasmic ratio (long arrow) in comparison with normal crypts (short arrows)
Figure 11.

Colitis-like lesion: active changes consist of inflammatory infiltration by neutrophils, plasma cells, and crypt abscesses (neutrophils in gland lumen/arrow)
DISCUSSION
The small number of animals, the different time periods of carcinogen and symbiotic administration in groups B and C, and the fact that the present study gives no data for comparing the effect of prebiotics and probiotics alone with the combination symbiotic treatment on colonic histological architecture are some of the limitations of this study.
It has been suggested that consumption of prebiotics, such as FOS or fructans, and probiotics, such as Lactobacilli or Bifidobacteria, has been associated with reduced incidence of tumors and precancerous lesions in experimental colon cancer in rats, and combination treatment with pre -and probiotics (the so-called synbiotics) may have a synergistic effect in reducing precancerous lesions in the colon (3–19). In the present study, we investigated the influence of a synbiotic regime on experimental colon cancer by administering a mixture comprising a large number of probiotics with a prebiotic, as there are very few data available in the relevant literature.
The results showed that the animals treated with the synbiotic regime survived in greater degree compared with untreated animals. Death of 6 of the 20 animals of group A is exclusively attributable to the toxic action of dimethylydrazine, despite the fact that no obvious cancer development was noticed. Indeed, according to the available data of toxicity, minor non-lethal effects appear to occur at cumulative exposures of <100 ppm multiplied by hours, whereas at cumulative exposures or slightly greater than this level, quite notable effects have been reported [NRC (National Research Council), 1985. Emergency and Continuous Exposure Guidance Levels for Selected Airborne Contaminants, Vol. 5, pp.5–21. Washington, DC: National Academy Press]. Lethality has been demonstrated when cumulative exposures exceed the levels of 100 ppm multiplied by hours, only slightly. It is of interest that dimethylydrazine has a very narrow margin between the levels of exposure, resulting in no significant toxicity and levels of exposure causing substantial lethality. The dose used in our experiment (15 mg/kg BW, once a week for 2 weeks) exceeds these levels, thus producing a significantly high mortality rate. Treated animals exhibited significantly lower percentage of lymphoid hyperplasia, colitis-like lesions, and ACF dysplasia as compared with animals that received carcinogen without synbiotics. Another point of interest was the finding suggesting that treated animals from the first day of the experiment had the least pathological lesions as compared to animals that received synbiotics two weeks after the administration of carcinogen. The significantly lower degrees of ACF, dysplasia, inflammation, and colitis-like lesions noticed in the treated animals suggests that this combination of pre -and probiotics could offer protection from cancer development in this kind of animal.
The evidence suggesting that consumption of probiotics may have a significantly positive effect on precancerous colonic lesions and CRC development has been previously suggested (25,26,27). So, McIntosh et al. (25) showed that a strain of Lactobacillus acidophilus Delvo Pro LA-1, supplied as freeze-dried bacteria in the diet, was protective against the development of large bowel tumors in rats. O’Mahony et al. (7) showed that the modification of enteric flora in IL-10 knockout mice by probiotic Lactobacilli was associated with reduced the prevalence of colon cancer and mucosal inflammatory activity. Similar results were reported by Yamazaki et al. (26). Beneficial results in terms of exhibiting significant antimutagenic activity against colon cancer were also reported by Marotta et al. (27) using a probiotic preparation.
So far, a number of studies dealing with the effects of inulin-type fructans on ACF or tumors in the colon of rats and mice have been reported (28,29). According to the findings of a comprehensive review of 12 relevant studies involving 29 individual treatment groups, there was a significant reduction of ACF in 21 of the 24 treatment groups and of tumor incidence in all five treatment groups, Inulin-type fructans reduced tumor incidence in APC (Min) mice in two of four studies and reduced growth and metastasizing properties of implanted tumor cells in mice (four studies) (28). The effects have been reported to be associated with gut flora-mediated fermentation and production of butyrate. There are some reports claiming that the use of synbiotics exerts beneficial biological effects beyond those of the individual components. Femia et al. (15) found that the combined administration of B. lactis and L. rhamnosus with inulin enriched with oligofructose was able to further reduce the incidence of adenomas and cancers induced by azoxymethane. It is of interest that this reduction was of the same magnitude as chemopreventive agents. Rowland et al. (29), using the synbiotic combination of inulin and Bifidobacterium longum, were able to detect a reduction of preneoplastic ACF. It was also noticed that combined treatment with the two agents was more effective in reducing colonic lesions. The same results were produced by others using combination of oligofructose and bifidobacteria (14).
Apart from the animal studies, there are some recently published reports describing the results of the administration of synbiotics in humans. In a randomized, double-blind, placebo-controlled trial of a synbiotic food administered in polypectomized patients and patients with CRC, Rafter et al. (17) found that the synbiotic intervention resulted in a significant increase in bifidobacteria and lactobacilli and a decrease in Clostridium perfringens. The intervention significantly reduced colorectal proliferation and the capacity of fecal water to induce necrosis in colonic cells and improved epithelial barrier function in polypectomized patients. Also, the synbiotic consumption prevented an increased secretion of IL-2 by peripheral blood mononuclear cells in the polypectomized patients and increased the production of IFN-g in patients with CRC. In another study, it was found that administration of Lactobacillus casei for 2 years in patients with a positive history for colorectal polyps resulted in significantly lower rate of tumors bearing moderate or higher degree atypia (18). FOS could also decrease the development of premalignant CRC lesions via the stimulation of the growth of endogenous Bifidobacteria and Lactobacilli. This can be achieved because FOS are metabolized in the colon by bacteria producing the b-galactosidase (29).
Concerning the possible mechanisms of action, it seems certain that probiotics can increase the number of beneficial bacteria, whereas the prebiotics can stimulate the development of the residential beneficial intestinal bacteria. Prebiotics serve as food for the ingested probiotics, thus preventing the depletion of the existing flora. Dietary carbohydrates escaping digestion/absorption in the small bowel and prebiotics undergo fermentation in the colon and give rise to short-chain fatty acids that enhance the growth of lactobacilli and bifidobacteria. Pro -and prebiotics can decrease intestinal inflammation, enhance immune function and anti-tumorigenic activity, bind to potential food carcinogens, and decrease bacterial enzymes that hydrolyze precarcinogenic compounds such as b-glucuronidase (30). The effect of prebiotics on cell proliferation, differentiation, apoptosis, mucin production, immune function, mineral absorption, lipid metabolism, and gastrointestinal peptides has been well documented (31). Prebiotics (inulin enriched with oligofructose) and probiotics (Lactobacillus rhamnosus GG and Bifidobacterium lactis Bb12) in combination may contribute to the suppression of colon carcinogenesis by modulating the gut-associated lymphoid tissue (32). Hughes and Rowland (33) found that the mean number of apoptotic cells per crypt was significantly higher in the colon of rats that were fed oligofructose or long-chain inulin as compared to those that were fed the basal diet alone. Le Leu et al. (12) found that the acute apoptotic response to azoxymethane was significantly increased by B. lactis in synbiotic combination with resistant starch, confirming that a synbiotic exerts a pro-apoptotic effect, not shared by the individual components. It seems that restoration of the number of CD8-positive T lymphocytes may play a key role in the preventive effect of probiotics against colon carcinogenesis (26). Foo et al. (34) suggested that probiotics prevent the development of 1,2-dimethylhydrazine-induced colonic tumorigenesis through suppressed colonic mucosa cellular proliferation and increased stimulation of macrophages. Finally, the use of synbiotics as a better prophylactic strategy than the use of probiotic and prebiotic alone has recently been suggested by Verma and Shukla (35) The beneficial effect has been attributed to the greater increase in antioxidants associated with the higher degree of attenuation of dimethylhydrazine dihydrochloride-induced tumorigenesis. In our study, the reduction in inflammation and colitis-like lesions noticed in the groups of animals receiving the synbiotic regime could be attributed to the downregulation of synthesis of pro-inflammatory cytokines and suppression of NF-kB activity in mucosal cells, as it has been suggested by Štofilová et al. in their recently published experimental work on dimethylhydrazine-induced carcinogenesis (36).
In conclusion, we found that animals treated with carcinogen and synbiotics had a significantly lower percentage of inflammation, colitis-like lesions, and ACF dysplasia than those receiving only carcinogen. Animals treated with probiotics from the beginning of the experiment had the least pathological lesions. Treatment with a synbiotic preparation exerts significant antimutagenic properties against the development of preneoplastic lesions in rats.
Footnotes
Ethics Committee Approval: Ethics committee approval was received for this study from the East Attica Region in accordance with the Greek law No 160/91 and the European Community regulations.
Informed Consent: N/A.
Peer-review: Externally peer-reviewed.
Author Contributions: Concept -A.E.P., J.K.T.; Design -F.G., P.D.; Supervision -J.K.T., A.E.P.; Resources: A.E.P.; Materials -A.E.P., G.S.; Data Collection and/or Processing -R.K., G.S., M.K.; Analysis and/or Interpretation -R.K., R.D.F.G.; Literature Search -R.K., R.D.F.G.; Writing -F.G., C.V., M.K.; Critical Reviews -A.E.P., J.K.T.
Conflict of Interest: The authors have no conflicts of interest to declare.
Financial Disclosure: The authors declared that this study has received no financial support.
REFERENCES
- 1.Pericleous M, Mandair D, Caplin ME. Diet and supplements and their impact on colorectal cancer. J Gastrointest Oncol. 2013;4:409–23. doi: 10.3978/j.issn.2078-6891.2013.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tárraga López PJ, Albero JS, Rodríguez-Montes JA. Primary and secondary prevention of colorectal cancer. Clin Med Insights Gastroenterol. 2014;7:33–46. doi: 10.4137/CGast.S14039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Foo NP, Ou Yang H, Chiu HH, et al. Probiotics prevent the development of 1,2-dimethylhydrazine (DMH)-induced colonic tumorigenesis through suppressed colonic mucosa cellular proliferation and increased stimulation of macrophages. J Agric Food Chem. 2011;59:13337–45. doi: 10.1021/jf203444d. [DOI] [PubMed] [Google Scholar]
- 4.Klewicka E, Nowak A, Zduńczyk Z, Cukrowska B, Błasiak J. Protective effect of lactofermented beetroot juice against aberrant crypt foci formation and genotoxicity of fecal water in rats. Exp Toxicol Pathol. 2012;64:599–604. doi: 10.1016/j.etp.2010.12.001. [DOI] [PubMed] [Google Scholar]
- 5.Appleyard CB, Cruz ML, Isidro AA, Arthur JC, Jobin C, De Simone C. Pretreatment with the probiotic VSL#3 delays transition from inflammation to dysplasia in a rat model of colitis-associated cancer. Am J Physiol Gastrointest Liver Physiol. 2011;301:G1004–13. doi: 10.1152/ajpgi.00167.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chong ES. A potential role of probiotics in colorectal cancer prevention: review of possible mechanisms of action. World J Microbiol Biotechnol. 2014:351–74. doi: 10.1007/s11274-013-1499-6. [DOI] [PubMed] [Google Scholar]
- 7.O’Mahony L, Feeney M, O’Halloran S, et al. Probiotic impact on microbial flora, inflammation and tumor development in IL-10 knockout mice. Aliment Pharmacol Ther. 2001;15:1219–25. doi: 10.1046/j.1365-2036.2001.01027.x. [DOI] [PubMed] [Google Scholar]
- 8.Verma A, Shukla G. Probiotics Lactobacillus rhamnosus GG, Lactobacillus acidophilus suppresses DMH-induced procarcinogenic fecal enzymes and preneoplastic aberrant crypt foci in early colon carcinogenesis in Sprague Dawley rats. Nutr Cancer. 2013;65:84–91. doi: 10.1080/01635581.2013.741746. [DOI] [PubMed] [Google Scholar]
- 9.Chang JH, Shim YY, Cha SK, Reaney MJ, Chee KM. Effect of Lactobacillus acidophilus KFRI342 on the development of chemically induced precancerous growths in the rat colon. J Med Microbiol. 2012;61:361–8. doi: 10.1099/jmm.0.035154-0. [DOI] [PubMed] [Google Scholar]
- 10.Pool-Zobel BL. Inulin-type fructans and reduction Cancer risk: review of experimental and human data. Br J Nutr. 2005;93( Suppl 1):S73–90. doi: 10.1079/BJN20041349. [DOI] [PubMed] [Google Scholar]
- 11.Håkansson A, Bränning C, Molin G, et al. Blueberry husks and probiotics attenuate colorectal inflammation and oncogenesis, and liver injuries in rats exposed to cycling DSS-treatment. PLoS One. 2012;7:e33510. doi: 10.1371/journal.pone.0033510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Le Leu RK, Hu Y, Brown IL, Woodman RJ, Young GP. Synbiotic intervention of Bifidobacterium lactis and resistant starch protects against colorectal cancer development in rats. Carcinogenesis. 2010;31:246–51. doi: 10.1093/carcin/bgp197. [DOI] [PubMed] [Google Scholar]
- 13.Turunen K, Tsouvelakidou E, Nomikos T, Mountzouris KC, Karamanolis D, Triantafillidis J, Kyriacou A. Impact of beta-glucan on the faecal microbiota of polypectomized patients: a pilot study. Anaerobe. 2011;17:403–6. doi: 10.1016/j.anaerobe.2011.03.025. [DOI] [PubMed] [Google Scholar]
- 14.Gallaher DD, Khil J. The effect of synbiotics on colon carcinogenesis in rats. J Nutr. 1999;129( 7 Suppl):1483S–7S. doi: 10.1093/jn/129.7.1483S. [DOI] [PubMed] [Google Scholar]
- 15.Femia AP, Luceri C, Dolara P, et al. Antitumorigenic activity of the prebiotic inulin enriched with oligofructose in combination with the probiotics Lactobacillus rhamnosus and Bifidobacterium lactis on azoxymethane-induced colon carcinogenesis in rats. Carcinogenesis. 2002;23:1953–60. doi: 10.1093/carcin/23.11.1953. [DOI] [PubMed] [Google Scholar]
- 16.Van Loo J, Clune Y, Bennett M, Collins JK. The SYNCAN project: goals, set-up, first results and settings of the human intervention study. Br J Nutr. 2005;93( Suppl 1):S91–8. doi: 10.1079/BJN20041353. [DOI] [PubMed] [Google Scholar]
- 17.Rafter J, Bennett M, Caderni G, et al. Dietary synbiotics reduce cancer risk factors in polypectomized and colon cancer patients. Am J Clin Nutr. 2007;85:488–96. doi: 10.1093/ajcn/85.2.488. [DOI] [PubMed] [Google Scholar]
- 18.Ishikawa H, Akedo I, Otani T, et al. Randomized trial of dietary fiber and Lactobacillus casei administration for prevention of colorectal tumors. Int J Cancer. 2005;116:762–7. doi: 10.1002/ijc.21115. [DOI] [PubMed] [Google Scholar]
- 19.Van Loo J, Jonkers N. Evaluation in human volunteers of the potential anticarcinogenic activities of novel nutritional concepts: prebiotics, probiotics and synbiotics (the SYNCAN project QLK1-1999-00346) Nutr Metab Cardiovasc Dis. 2001;11(4 Suppl):87–93. [PubMed] [Google Scholar]
- 20.Ringel Yehuda, Ringel-Kulka Tamar, Palsson Olafur S. Clinical trial: Probiotic bacteria lactobacillus acidophilus NCFM and bifidobacterium lactis Bi-07 versus placebo for the symptoms of bloating in patients with functional bowel disorders -a double-blind study. J Clin Gastroenterol. 2011;45:518–25. doi: 10.1097/MCG.0b013e31820ca4d6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sabater-Molina M, Larqué E, Torrella F, Zamora S. Dietary fructooligosaccharides and potential benefits on health. J Physiol Biochem. 2009;65:315–28. doi: 10.1007/BF03180584. [DOI] [PubMed] [Google Scholar]
- 22.Cheng L, Lai MD. Aberrant crypt foci as microscopic precursors of colorectal cancer. World J Gastroenterol. 2003;9:2642–9. doi: 10.3748/wjg.v9.i12.2642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pretlow TP, Barrow BJ, Asthon WS, et al. Aberrant crypt foci and preneoplastic foci in human colonic mucosa. Cancer Res. 1991;51:1564–7. [PubMed] [Google Scholar]
- 24.Rudolph RE, Domitz JA, Lampe JW, et al. Risk factors for colorectal cancer in relation to number and size of aberrant crypt foci in humans. Cancer Epidemiol Biomarkers Prevention. 2005;14:605–8. doi: 10.1158/1055-9965.EPI-04-0058. [DOI] [PubMed] [Google Scholar]
- 25.McIntosh GH, Royle PJ, Playne MJ. A probiotic strain of L. acidophilus reduces DMH-induced intestinal tumors in male Sprague-Dawley rats. Nutr Cancer. 1999;35:153–9. doi: 10.1207/S15327914NC352_9. [DOI] [PubMed] [Google Scholar]
- 26.Yamazaki K, Tsunoda A, Sibusawa M, et al. The effect of an oral aministration of Lactobacillus casei strain shirota on azoxymethane-induced colonic aberrant crypt foci and colon cancer in rats. Oncol Rep. 2000;7:977–82. doi: 10.3892/or.7.5.977. [DOI] [PubMed] [Google Scholar]
- 27.Marotta F, Naito Y, Minelli E, et al. Chemopreventive effect of a probiotic preparation on the development of preneoplastic and neoplastic colonic lesions: an experimental study. Hepatogastroenterology. 2003;50:1914–8. [PubMed] [Google Scholar]
- 28.Pool-Zobel BL. Inulin-type fructans and reduction in colon cancer risk: review of experimental and human data. Br J Nutr. 2005;93( Suppl 1):S73–90. doi: 10.1079/BJN20041349. [DOI] [PubMed] [Google Scholar]
- 29.Rowland IR, Rumney CJ, Coutts JT, Lievense LC. Effect of Bifidobacterium longum and inulin on gut bacterial metabolism and carcinogen-induced aberrant crypt foci in rats. Carcinogenesis. 1998;19:281–5. doi: 10.1093/carcin/19.2.281. [DOI] [PubMed] [Google Scholar]
- 30.Geier MS, Butler RN, Howarth GS. Probiotics, prebiotics and synbiotics: a role in chemoprevention for colorectal cancer? Cancer Biol Ther. 2006;5:1265–9. doi: 10.4161/cbt.5.10.3296. [DOI] [PubMed] [Google Scholar]
- 31.Roy CC, Kien CL, Bouthillier L, Levy E. Short-chain fatty acids: ready for prime time? Nutr Clin Pract. 2006;21:351–66. doi: 10.1177/0115426506021004351. [DOI] [PubMed] [Google Scholar]
- 32.Roller M, Pietro Femia A, Caderni G, Rechkemmer G, Watzl B. Intestinal immunity of rats with colon cancer is modulated by oligofructose-enriched inulin combined with Lactobacillus rhamnosus and Bifidobacterium lactis. Br J Nutr. 2004;92:931–8. doi: 10.1079/BJN20041289. [DOI] [PubMed] [Google Scholar]
- 33.Hughes R, Rowland IR. Stimulation of apoptosis by two prebiotic chicory fructans in the rat colon. Carcinogenesis. 2001;22:43–7. doi: 10.1093/carcin/22.1.43. [DOI] [PubMed] [Google Scholar]
- 34.Foo NP, Ou Yang H, Chiu HH, et al. Probiotics prevent the development of 1,2-dimethylhydrazine (DMH)-induced colonic tumorigenesis through suppressed colonic mucosa cellular proliferation and increased stimulation of macrophages. J Agric Food Chem. 2011;59:13337–45. doi: 10.1021/jf203444d. [DOI] [PubMed] [Google Scholar]
- 35.Verma A, Shukla G. Synbiotic (Lactobacillus rhamnosus+Lactobacillus acidophilus+inulin) attenuates oxidative stress and colonicdamage in 1,2 dimethylhydrazine dihydrochloride-induced colon carcinogenesis in Sprague-Dawley rats: a long-term study. Eur J Cancer Prev. 2014;23:550–9. doi: 10.1097/CEJ.0000000000000054. [DOI] [PubMed] [Google Scholar]
- 36.Štofilová J, Szabadosová V, Hrčková G, et al. Co-administration of a probiotic strain Lactobacillus plantarum LS/07 CCM7766 with prebiotic inulin alleviates the intestinal inflammation in rats exposed to N,N-dimethylhydrazine. Int Immunopharmacol. 2015;24:361–8. doi: 10.1016/j.intimp.2014.12.022. [DOI] [PubMed] [Google Scholar]


