Abstract
Retinal degenerative diseases, due to the lack of regeneration systems and self-renewable cells, often lead to visual impairment. Pax6 is a pleiotropic transcription factor and its expression level determines self-renewal status or differentiation of retinal cells. Here, we investigated the fate of simultaneous induction of retinal ganglion cell death and Pax6 overexpression in retro-differentiation of retinal cells and their commitment to re-enter into the cell cycle. Induction of acute retinal ganglion cell death and generation of mouse experimental model was performed by N-methyl D-aspartic acid (NMDA) injection. Recombinant AAV2 virus harboring PAX6 cDNA and reporter gene was injected into untreated and model mouse eyes. Histological analyses, including IHC and retinal flatmounts immunostaining were performed. The number of Ki67+ cells was clearly increased in model mice, presumably due to NMDA treatment and regardless of Pax6 over-expression. Unlike previous studies, Ki67+ cells were found in GCL layer and interestingly ONL cells expressed Sox2 stemness marker after NMDA cytotoxicity. The potential of retinal cells for robust Ki67 expression, after injury, and expression of Sox2, confirmed their intrinsic plasticity and made a vivid prospect for retinal regenerative medicine.
Introduction
In the human retina, due to the lack of regeneration systems and self-renewable cells, degenerative diseases often lead to a visual impairment. The vertebrate neural retina composed of six types of differentiated neurons and one type of glial cells and all of them originate in a conserved order from retinal progenitor cells (RPCs). While non-mammalian vertebrates due to the presence of RPCs, throughout their lives, have a remarkable ability to replace damaged neurons, in mammalians we have no significant retinal neurogenic source1. Our understanding of many aspects of RPCs is limited, however, because of strong relationship between cell cycle and retinal development2,3, it is reasonable to hypothesize that if we can stimulate cells to reenter to cell cycle, with respect to common ancestor, maybe all retina cell types regenerate after degenerations.
Although there are no reports on de novo neurogenesis in mammalian adults, but characterization of muller glia as intrinsic retinal stem cells4 and some findings about retinal cells plasticity5,6 can be indicative of inherent potential of retinal cells for proliferation and regeneration. Furthermore, it is reported that after induction of retinal degeneration, some cells reenter to the cell cycle7. Ooto and his colleagues showed neural regeneration after acute neurotoxic injury mediated by N-methyl D-aspartate (NMDA) in the adult mammalian retina8. Also, it has been shown some growth factors such as EGF can enforce retinal cells to proliferate9.
In recent years, successful reprogramming of adult cells by ancestral transcription factors was a milestone in cellular biology10–14. In reprogramming studies, the main goal is perturbing gene regulatory network (GRN) to achieve intended phenotype. In these strategies transcription factors are from the effective and convenient choices. The Pax6 transcription factor resides at the top of the genetic hierarchy controlling development and morphogenesis of the eye and is crucial for the development of the central nervous system (CNS), nose, pancreas, and pituitary gland15,16. This factor is one of the indicators of RPCs. Its expression level is a determinative factor in self-renewal status or differentiation of retinal cells17. It has been shown that after retinal injuries in mice, some Pax6 positive cells migrate from inner nuclear layer (INL) to outer nuclear layer (ONL); However this migration does not lead to proliferation and differentiation dislike some lower vertebrates18.
Here, we investigated perturbing GRN in inner retina layers with PAX6 overexpression, mediated by adeno-associated virus serotype-2 (AAV-2), and induction of retinal ganglion cell death to analyze expression of cell cycle marker in the mouse retina.
Results
Induction of retinal damage
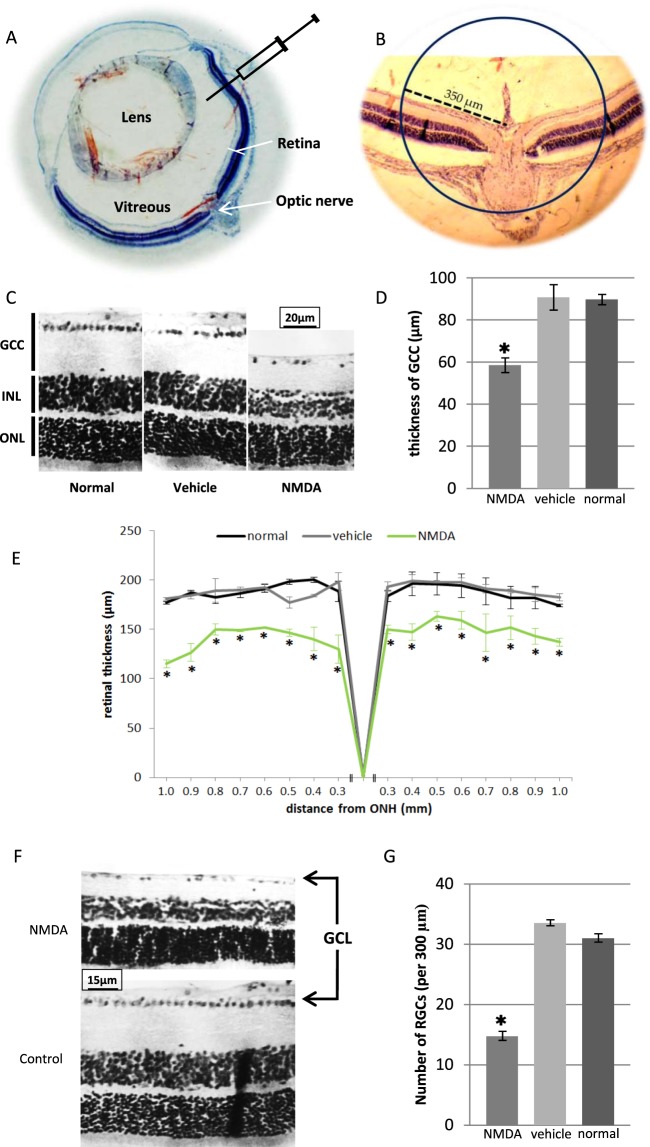
NMDA-induced RGC damage is a reliable method to generate experimental model. Generation of this model characterizes by rapid RGCs loss followed by gradual reduction in ganglion cell complex thickness (GCC: retinal nerve fiber layer, ganglion cell layer, and inner plexiform layer). The thickness maps and the quantitative thickness values of retina and GCC showed thickness changes in the NMDA-treated mice when compared with normal and vehicle-treated mice (Fig. 1C–E). ONL thickness was not affected, but INL thickness showed small changes (see Supplementary Fig. S1). The number of retinal ganglion cells was significantly decreased in treated mice (Fig. 1F,G). Normal and vehicle-treated mice were similar in all parameters.
Figure 1.
Generation of mouse experimental model by NMDA excitotoxic amino acid. (A) Schematic illustration of intravitreal injection of NMDA; All injections were performed under ora serrata by glass needle. (B) For evaluation of mice model, analyses were performed on a distinct distance of optic nerve. (C,D) Histological analyses of GCC thickness 7 days post injection. Data significantly decreased in NMDA samples versus vehicle treated and normal mice. (E) Quantitative spider plot analysis depicting significant decrease in retinal thickness in NMDA samples. (F,G) Histological analysis of number of ganglion cells 7 days post injection. Data significantly decreased in NMDA samples versus vehicle treated and normal mice. GCC: ganglion cell complex; ONL: outer nuclear layer; INL: inner nuclear layer; GCL: ganglion cell layer; ONH: optic nerve head. (*P < 0.05 versus other groups, error bar: means ± SD).
Analysis of Ki67 and Pax6 expression in normal mouse retina
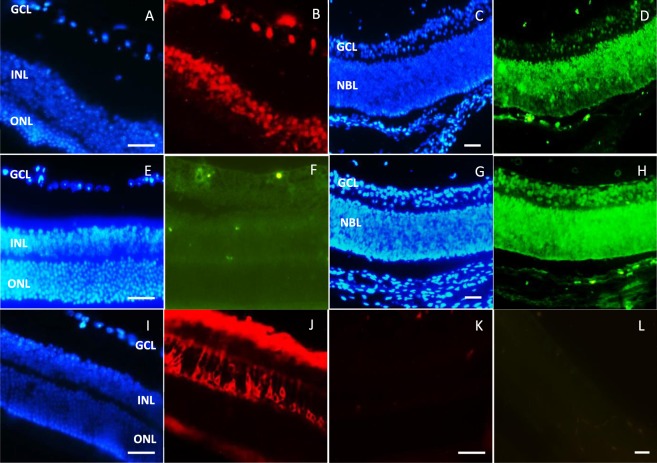
Prior to the transduction of the retina, we compared Ki67, Pax6 and PKC expression in uninjected newborn and adult mouse retina (Fig. 2). Pax6 expression was detected in all the retinal layers just after mouse birth. Its expression, as it is shown in Fig. 2C,D, determined a gradient decrease from GCL to ONL. In adult mouse, Pax6 expression is limited to inner cells of INL and GCL cells (Fig. 2A,B).
Figure 2.
Immunohistochemistry on normal mouse retina for Pax6, Ki67 and PKCα factors. (A,B) Pax6 expression in adult mouse retina; Pax6 was expressed in GCL layer and inner region of INL. (C,D) Pax6 expression in newborn (P1) mouse retina; Pax6 was expressed in all retinal layers but its expression was decreased in outer retina layer. (E,F) Ki67 expression in adult mouse retina; Ki67 was not detected in adult mouse retina. (G,H) Ki67 expression in newborn (P1) mouse retina; Ki67 was expressed in all retinal layers. (I,J) PKCα expression in adult mouse retina; PKCα was expressed in GCL layer and some cell types of INL. (K,L) Negative controls of immunofluorescences for Rhodamine and FITC-conjugated antibodies (no 1st ab.). ONL: outer nuclear layer; INL: inner nuclear layer; GCL: ganglion cell layer; NBL: neuroblastic layer. Scale bar: 50 µm.
Ki67 has no expression in adult mouse retina (Fig. 2E,F), while in newborn mouse retina all of the retinal cells expressed Ki67, clearly (Fig. 2G,H). PKCα, as serine/threonine protein kinases, is a cytoplasmic marker in bipolar and ganglion cells. Figure 2I,J, shows that PKCα is expressed in outer region of INL and ganglion cells.
Infection of mouse retina with AAV-2
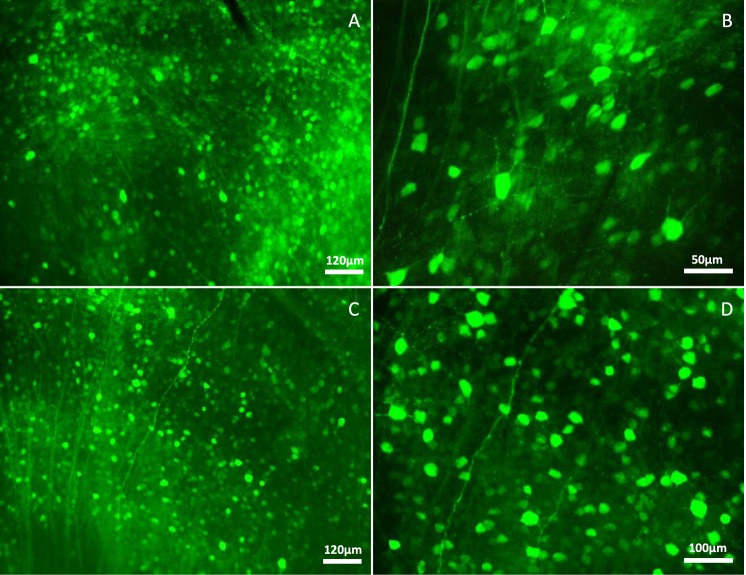
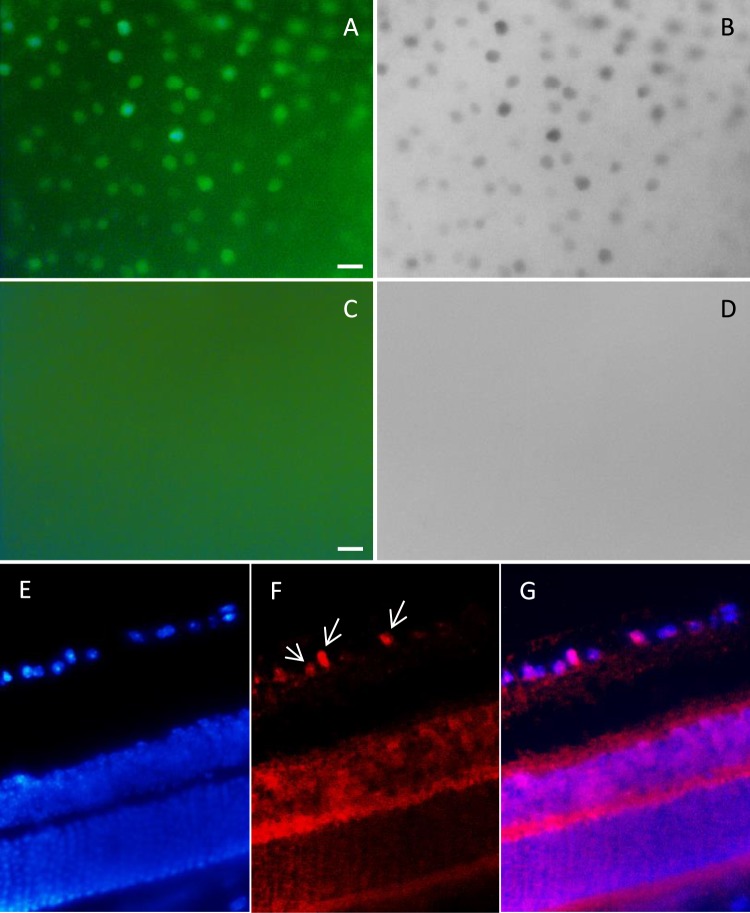
The most widely used vectors for ocular gene delivery are based on adeno-associated virus (AAV), because they elicit minimal immune responses and mediate long-term expression in a variety of non-dividing retinal cell types. Here, we used AAV serotype-2 for PAX6 delivery to the mouse retina. Viral titration was about 1012 genomic particles/ml and 108 infectious particles/ml. Radiant green cells infected with AAV2-EGFP viruses represented successful infection in experiments (Fig. 3). For in vivo retinal infection, concentration and purification of AAV paticles are the major concerns and a little decline in MOI leads to very significant decrease in infection rate19,20.
Figure 3.
Efficient EGFP expression in whole-mount retina following intravitreal injection of AAV2 viruses (after 3 weeks). (A,B) EGFP expression following injection of AAV2/EGFP viruses in different magnifications. (D,E) EGFP expression following injection of AAV2/PAX6-EGFP viruses in different magnifications. Images revealed high infection rate of the recruited AAV preparation, which is criterion for AAV grade and efficiency in transduction. (in all experiments n > 5).
Evaluation of Pax6 overexpression in normal and model mice
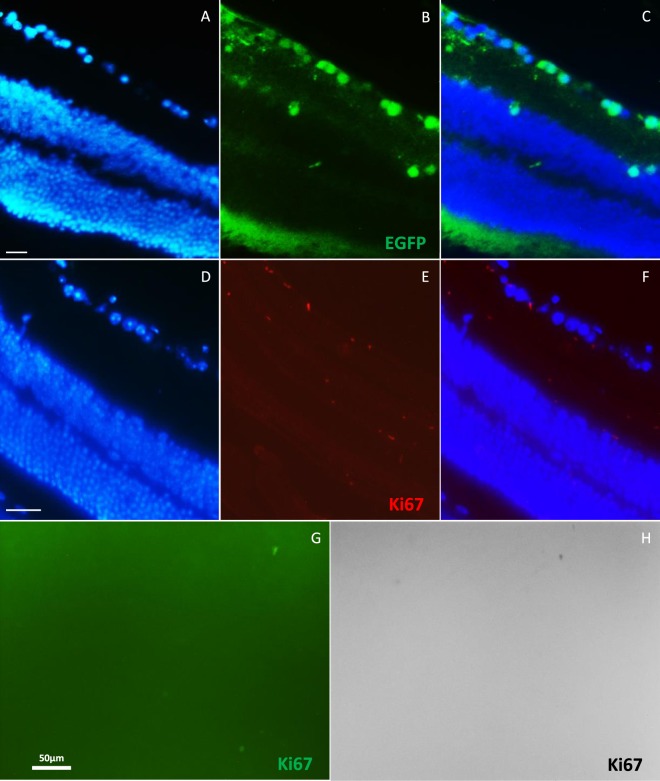
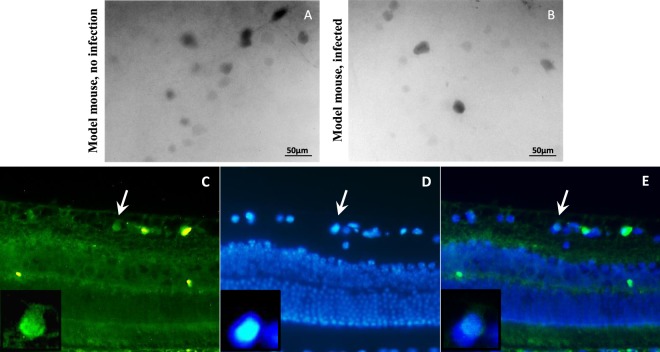
We did not observe any changes in Ki67 expression following PAX6 overexpression in both of the model mice and its controls. In normal mice, Pax6 overexpression mediated by rAAV2-Pax6 viral particles could not induce Ki67 expression. In infected retina, EGFP expression was obvious (Fig. 4A–C), however we could not detected any Ki67 expression in retinal sections (Fig. 4D–F) or flatmounts (Fig. 4G,H). In model mice, Ki67 expression was significant increased, but it was the same for the control eyes. So, Ki67 expression was independent of Pax6 overexpression and was due to NMDA cytotoxicity (Fig. 5A–D).
Figure 4.
Analysis of Ki67 expression following Pax6 overexpression that was mediated by AAV2 viruses in normal mouse retina. (A–C) Immunofluorescence of EGFP, 30 days post Pax6 overexpression. EGFP was expressed in GCL layer and some inner INL cells. (D–H) Ki67 expression was not detected in infected normal retina in sections (D–F) and flatmount (G,H); H is black and white view of G with higher contrast. n > 5, scale bar: 50 µm.
Figure 5.
Analysis of Ki67 expression following Pax6 overexpression that was mediated by AAV2 viruses in model mouse retina. (A) Immunofluorescence of Ki67 in control flatmounts (only NMDA injection, no viruses), 30 days post infection; Ki67 was expressed obviously after NMDA injection. (B) Immunofluorescence of Ki67 in flatmounts, 30 days post NMDA and viruses injection; Ki67 was expressed as the same as the controls. (C–E) Immunostaining of Ki67 on 5 µm section following injections. Ki67 was expressed in GCL and some cells in INL.
Evaluation of NMDA cytotoxicity in the mouse retina
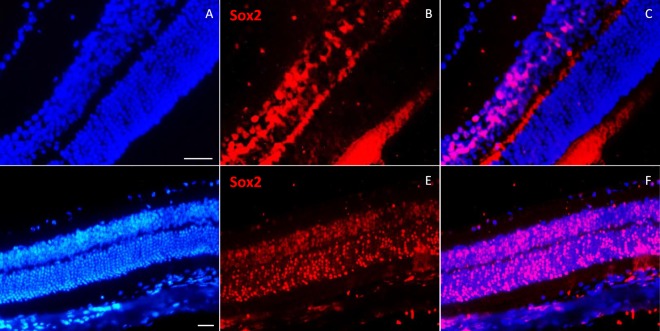
NMDA is an excitotoxic amino acid that overstimulates NMDA-receptors and leads to cell death in RGC and amacrine cells. Our results showed significant Ki67 expression, in flatmounts, followed by NMDA injection in eyes. Also, IHC analysis on retinal sections clearly showed expression of Ki67 cell cycle marker in INL and GCL layers’ cells (Fig. 5C–E). We also followed Ki67 expression after three months of damage. Ki67 revealed sustained expression without significant reduction (Fig. 6). Interestingly, after 30 days of neural injury induction by NMDA, Sox2 expression was observed in ONL layer (Fig. 7). Sox2 is a stemness marker and its expression indicates neural plasticity of photoreceptors component of retina.
Figure 6.
Immunofluorescence of Ki67, 3 months post NMDA injection. (A,B) Expression of Ki67 has been shown after 3 months on flatmounts (n = 2). (C,D) Flatmounts of vehicle injected retina as control. (E–G) Immunostaining of Ki67 on 5 µm section 3 months post NMDA injection. Ki67 was expressed in GCL (white arrows) and some cells in INL. Scale bar: 50 µm.
Figure 7.
Immunostaining of Sox2 on normal and model mouse retina sections. (A–C) In normal mouse retina, Sox2 was expressed in some INL cells, but not in other retina layers. (D–F) In NMDA injected mouse retina (after 30 days), Sox2 was obviously expressed in ONL, INL and GCL layers. n > 3, scale bar: 50 µm.
Discussion
In recent years, one of the challenging fields in biology is using of intrinsic regeneration potential in damaged organs. In fact, when it was shown that with genetic or epigenetic manipulations we can reprogram differentiated cells to stem/progenitor state, it seems more reasonable to stimulate residing, naturally dependent, cells in damaged region to regenerate the desired cells. So, many studies have been directed to evaluate differentiation potential of a variety cell types and to investigate for new therapeutic approaches. Contrary to importance of retina in many ocular diseases, there are no satisfactory numbers of studies related to research of plasticity in retinal cell types. Parts of the regenerative mechanism in non-mammalian vertebrates remain in the mammalian retina and may provide a basis to develop new strategies in patients with retinal degenerations. It seems that our knowledge about differentiation and self-renewal potential of retinal cells and also the role of genetic and epigenetic factors is not pleasing. Here, we evaluated Pax6 overexpression and NMDA cytotoxicity to stimulate proliferation in the mouse retinal cells. We realized that PAX6, as a master transcription factor, would be a valid candidate in manipulation of GRN leading to a substantial regeneration response after excitotoxic cell death stimulation in RGC layer.
Despite, well established retina regeneration in cold-blood vertebrates21 and partially in avian22,23, we have a few documents of retina regeneration in mammalians. However, some groups have reported sufficiency of few cells in INL to re-enter the cell cycle8,9. Furthermore, there are successful reports on inducing regeneration in mouse and rat retina by transcription factors and/or growth factors24–27. In model mice, Ooto and colleagues showed few cells in INL, not in other layers, have remarkable potential in entrance to cell-cycle after neurotoxic damage with NMDA8. However, our results revealed some Ki67 positive cells in GCL in addition to robust Ki67 expression in INL. Retinal ganglion cells have significant roles in visual system28 and the most common retina diseases such as glaucoma29. Here, for the first time, we showed reentrance of GCL cells to the cell cycle after NMDA treatment. Suga and his colleagues showed the degree of proliferation rate in damaged cells of the retina varies between the mouse strains7, it suggests that, the difference in the intrinsic potency of regeneration between species is very important. This issue can fully affect results of different studies, technically and theoretically. We found a considerable plasticity in GCL cells and robust Ki67 expression, even, until three months after neurotoxic damage in INL following NMDA injection.
Pax6 is a pleiotropic transcription factor that has many challenging roles in retina developments. Here, we injected intravitreally AAV serotype 2 as a delivery vehicle of Pax6 to disturb GRN in retina cells. Up-regulation of some progenitor markers including Pax6, Notch and Dll1 has been reported after NMDA damage9. Here, we investigated whether Pax6 overexpression can promote proliferation in retina. We could not detect any changes in Ki67 expression following Pax6 overexpression. However we have to consider a few points; First, intravitreal injection of AAV2 yielded robust transgene expression throughout the retina in whole-mount analysis. Retinal sections showed the expression mainly resided in RGCs and some cells in INL layer as previously reported30. However, in normal adult retina, some INL cells and ganglion cells express Pax6 intrinsically; therefore, it is possible that Pax6 overexpression with non-integrative system was not sufficient to disturb GRN. Secondly, previous studies have been shown high autoregulation of Pax6 expression31. In fact, due to important roles of expression level of this transcription factor, cells have potent mechanisms to regulate Pax6. Thirdly, in recent studies, different roles of Pax6 isoforms are established32,33. Here, we recruited dominant isoform in the retina, 5a isoform, to our analyses.
An interesting finding in our data was the detection of Sox2 expression in ONL cells after neurotoxic injury. Sox2 has a key role in retina development and is one of the factors in iPS generation10,34. This transcription factor is expressed in retinal progenitors and, in adult mammalian retina, some INL cells, including muller glia cells35. Sox2 has a complex nature in interaction with main players in retina development such as Pax636 and Wnt pathway37 and is a dose-dependent regulator of RPCs38. Previous studies in neural stem cells division demonstrated critical roles for Sox2; however there is limited knowledge about molecular mechanisms and chronic relationship in retina development. In Xenopus retina it has been demonstrated that Wnt signaling, mediated by Sox2, promotes neural destination by activating proneural gene expression and, coordinately, inhibit neural differentiation through Notch activation39. Here, we reported Sox2 expression in all retina layers including photoreceptors and GCL after 30 days of NMDA administration. Expression of Sox2 stemness marker promises high plasticity potential of retina cells and highlighted the role of microenvironment and extracellular signaling in acquisition of new phenotypes. More investigation is needed to analysis molecular pathways in retina and the cell fate machinery in reprogramming of retina cells.
Conclusion
Reports on reactivation of inactive endogenous progenitors and well established retina regeneration in non-mammalians open up new hopes for using intrinsic potential of retinal cells in healing. It seems that we need new strategies in ocular degenerative therapies. Combination of different achievements in regenerative medicine introduces novel approaches to overcome limitations in cell and gene therapies. Entrance of retinal cells to cell cycle and expression of Sox2 stemness marker after neurotoxic damage along with origination of all retina cell types from one type of proliferative cells are a vivid prospect for retinal regenerative medicine.
Materials and Methods
Animals and care
All injections were performed on 6–8 week-old NMRI mice. Animals were housed in the National Institute of Genetic Engineering and Biotechnology (NIGEB) and all experiments were performed in accordance with the ARVO (Association for Research in Vision and Ophthalmology) statement for the use of animals and protocols in ophthalmic research that were approved by the ethical committee of; “Ophthalmic Research Center, Shahid Beheshti University of Medical Sciences” and “National Institute of Genetic Engineering and Biotechnology”. The mice were maintained under normal conditions of 12 h dark/light cycle, free access to water and food and 20–25 °C temperature.
Plasmids and production of recombinant AAV virus
HEK293T cells were subjected to virus production and titration. PAX6a and IRES-EGFP (from pIRES2-EGFP) were cloned into pAAV-MCS (AAV Helper-Free System, Agilent). Triple transfection for two constructs (AAV-EGFP and AAV-PAX6/EGFP) was performed on twelve 10 cm plates by standard calcium-phosphate method. Briefly, the medium was changed 2 h before transfection. For each plates, 15 µg of each plasmids and 48 µl CaCl2 (2 M) reached to 400 µl final volume with ddH2O. This solution was added to 2× HEPES buffer saline (2× HBS: 595.7 mg HEPES, 13 mg Na2HPO4, 2H2O, 818.2 mg NaCl, 119 mg dextrose and 37 mg KCl in 50 ml ddH2O, pH 7.05) while vortexing. After 15 min incubation, DNA complexes were added to the cells. The medium was changed with 10% FBS fresh medium after 6 h. 68–72 hours later, viruses were purified and concentrated with HiTrap™ Heparin HP (GE Healthcare) and amicon columns (Millipore) as previously reported40. Viruses were aliquoted and stored at −80 °C. Viral titration was achieved with flow cytometry and qPCR. For flow cytometry, HEK293T cells were plated and infected according to standard protocol (Agilent tech., CA, USA). After 48 h, cultures were trypsinized and were washed twice with PBS. The cells were resuspended in PBS and GFP expression was analyzed (Cyflow space Partec, Germany, flowmax software). In qPCR, primers were designed for CMV promoter and standard curve were plotted. Genomic-based titration was done with Real-Time PCR.
Intraocular Injections
All experiments were done in seven groups (1: no injected, 2: PBS injected as vehicle, 3: NMDA injected, 4: EGFP viruses injected, 5: Pax6/EGFP viruses injected, 6: EGFP viruses injected 48 h after NMDA, 7: Pax6/EGFP viruses injected 72 h after NMDA). For each treatment, right eye was its control and more than six animals were applied for each group. The eyes were injected with 80 µm glass needle connected to CellTram™ oil manual microinjector (Eppendorf). Mice were anesthetized by i.p. injection of 100 mg/kg ketamine and 10 mg/kg xylazine. Before injection, a drop of tropicamide (Mydrex 0.5%) and after injection, liposic ophthalmic gel (Bausch and Lomb) and gentamicin were administrated topically. Retinal damage was induced by the injection of 2 µl NMDA (20 mM, sigma) in PBS and for other analyses, 2 µl viruses or PBS were injected intravitreally. To confirm NMDA cytotoxicity, histological analyses were performed seven days after administration. Other analyses were done thirty days after injection.
Histological Analysis of Mouse Retina
The mice were euthanized by cervical dislocation. The eyes were enucleated and after washing with PBS, fixed overnight in 4% paraformaldehyde. To evaluation of NMDA neurotoxicity, seven days after injection, three sections containing optic nerve and with maximal circumference of the eyeball were stained with hematoxylin and eosin (H and E). The number of ganglion cells and thickness of retinal layers were calculated and averaged in NMDA or PBS (vehicle) injected and normal (uninjected) eyes. To IHC analyses, the fixed tissues were washed in PBS and dehydrated in ethanol serials and xylene according to standard protocols and fixed in paraffin. 5 µm paraffin sections were subjected to IHC analyses. To whole-mount analysis, Iris and lens were removed and remained cup, fixed 10 min. in cold methanol. After three times washing, tissues were subjected to IHC. The antibodies and working dilutions in this study were as follows: mouse anti-GFP (sc-9996, 1:100), rabbit anti-Ki67 (sc-15402, 1:100), goat anti-Pax6 (sc-7750, 1:200), rabbit anti-PKC (sc-10800, 1:200), goat anti-Sox2 (sc-17320, 1:200), donkey anti-goat IgG-FITC (sc-2024, 1:200), goat anti-rabbit IgG-FITC (sc-2012, 1:200), goat anti-mouse IgG-FITC (sc-2010, 1:200), donkey anti-goat IgG-R (sc-2094, 1:100) and goat anti-rabbit IgG-R (sc-2091, 1:100).
Imaging and Analyses
H and E stained sections were photographed by light microscope and the thickness of retinal layers analyzed at a distance of 350 µm or up to 1 mm in spider plots from the optic nerve. Also, ganglion cell counts were performed at a distance between 350 and 650 µm from optic nerve. The data were averaged and presented as ± S.E.M. (n = 3). Comparisons were done by one-way ANOVA (IBM SPSS Statistics 22) and P < 0.05 was considered significant. For IHC results, An Axiophot Zeiss fluorescence microscope equipped with a 520 nm filter for the FITC and a 460 nm filter for DAPI was used. For comparison of whole-mounts and making black and white images, ImageJ software was applied.
Electronic supplementary material
Acknowledgements
The authors would like to thank Dr. Mehdi Shamsara, Dr. Morteza Daliri Joupari and Dr. Azita Parvaneh Tafreshi of the national institute of genetic engineering and biotechnology (NIGEB) for technical support.
Author Contributions
Z.S. designed and supervised the study, E.R.P. performed the experiments and wrote the manuscript, Z.S. and M.N. reviewed and edited manuscript, Z.S., E.R.P., S.S., H.A., S.J.M., and N.D. analyzed data.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-35884-5.
References
- 1.Karl MO, Reh TA. Regenerative medicine for retinal diseases: activating endogenous repair mechanisms. Trends in molecular medicine. 2010;16:193–202. doi: 10.1016/j.molmed.2010.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Andreazzoli M. Molecular regulation of vertebrate retina cell fate. Birth defects research. Part C, Embryo today: reviews. 2009;87:284–295. doi: 10.1002/bdrc.20161. [DOI] [PubMed] [Google Scholar]
- 3.Bilitou A, Ohnuma S. The role of cell cycle in retinal development: cyclin-dependent kinase inhibitors co-ordinate cell-cycle inhibition, cell-fate determination and differentiation in the developing retina. Developmental dynamics: an official publication of the American Association of Anatomists. 2010;239:727–736. doi: 10.1002/dvdy.22223. [DOI] [PubMed] [Google Scholar]
- 4.Reichenbach A, Bringmann A. New functions of Muller cells. Glia. 2013;61:651–678. doi: 10.1002/glia.22477. [DOI] [PubMed] [Google Scholar]
- 5.Stevens CB, Cameron DA, Stenkamp DL. Plasticity of photoreceptor-generating retinal progenitors revealed by prolonged retinoic acid exposure. BMC developmental biology. 2011;11:51. doi: 10.1186/1471-213X-11-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Webster, M. K. et al. Evidence of BrdU-positive retinal neurons after application of an Alpha7 nicotinic acetylcholine receptor agonist. Neuroscience, 10.1016/j.neuroscience.2017.01.029 (2017). [DOI] [PMC free article] [PubMed]
- 7.Suga A, Sadamoto K, Fujii M, Mandai M, Takahashi M. Proliferation potential of Muller glia after retinal damage varies between mouse strains. Plos one. 2014;9:e94556. doi: 10.1371/journal.pone.0094556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ooto S, et al. Potential for neural regeneration after neurotoxic injury in the adult mammalian retina. Proceedings of the National Academy of Sciences of the United States of America. 2004;101:13654–13659. doi: 10.1073/pnas.0402129101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Karl MO, et al. Stimulation of neural regeneration in the mouse retina. Proceedings of the National Academy of Sciences of the United States of America. 2008;105:19508–19513. doi: 10.1073/pnas.0807453105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 11.Zhou Q, Brown J, Kanarek A, Rajagopal J, Melton DA. In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature. 2008;455:627–632. doi: 10.1038/nature07314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ieda M, et al. Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell. 2010;142:375–386. doi: 10.1016/j.cell.2010.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Vierbuchen T, et al. Direct conversion of fibroblasts to functional neurons by defined factors. Nature. 2010;463:1035–1041. doi: 10.1038/nature08797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Qian L, et al. In vivo reprogramming of murine cardiac fibroblasts into induced cardiomyocytes. Nature. 2012;485:593–598. doi: 10.1038/nature11044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shaham O, Menuchin Y, Farhy C, Ashery-Padan R. Pax6: a multi-level regulator of ocular development. Progress in retinal and eye research. 2012;31:351–376. doi: 10.1016/j.preteyeres.2012.04.002. [DOI] [PubMed] [Google Scholar]
- 16.Ouyang J, et al. Pax6 overexpression suppresses cell proliferation and retards the cell cycle in corneal epithelial cells. Investigative ophthalmology & visual science. 2006;47:2397–2407. doi: 10.1167/iovs.05-1083. [DOI] [PubMed] [Google Scholar]
- 17.Hsieh YW, Yang XJ. Dynamic Pax6 expression during the neurogenic cell cycle influences proliferation and cell fate choices of retinal progenitors. Neural development. 2009;4:32. doi: 10.1186/1749-8104-4-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Joly S, Pernet V, Samardzija M, Grimm C. Pax6-positive Muller glia cells express cell cycle markers but do not proliferate after photoreceptor injury in the mouse retina. Glia. 2011;59:1033–1046. doi: 10.1002/glia.21174. [DOI] [PubMed] [Google Scholar]
- 19.Sarra GM, et al. Kinetics of transgene expression in mouse retina following sub-retinal injection of recombinant adeno-associated virus. Vision research. 2002;42:541–549. doi: 10.1016/S0042-6989(01)00230-9. [DOI] [PubMed] [Google Scholar]
- 20.Martin KR, Klein RL, Quigley HA. Gene delivery to the eye using adeno-associated viral vectors. Methods. 2002;28:267–275. doi: 10.1016/S1046-2023(02)00232-3. [DOI] [PubMed] [Google Scholar]
- 21.Ail D, Perron M. Retinal Degeneration and Regeneration-Lessons From Fishes and Amphibians. Current pathobiology reports. 2017;5:67–78. doi: 10.1007/s40139-017-0127-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gallina D, Todd L, Fischer AJ. A comparative analysis of Muller glia-mediated regeneration in the vertebrate retina. Experimental eye research. 2014;123:121–130. doi: 10.1016/j.exer.2013.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fischer AJ. Neural regeneration in the chick retina. Progress in retinal and eye research. 2005;24:161–182. doi: 10.1016/j.preteyeres.2004.07.003. [DOI] [PubMed] [Google Scholar]
- 24.Takeda M, et al. alpha-Aminoadipate induces progenitor cell properties of Muller glia in adult mice. Investigative ophthalmology & visual science. 2008;49:1142–1150. doi: 10.1167/iovs.07-0434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Osakada F, et al. Wnt signaling promotes regeneration in the retina of adult mammals. The Journal of neuroscience: the official journal of the Society for Neuroscience. 2007;27:4210–4219. doi: 10.1523/JNEUROSCI.4193-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wan J, Zheng H, Xiao HL, She ZJ, Zhou GM. Sonic hedgehog promotes stem-cell potential of Muller glia in the mammalian retina. Biochemical and biophysical research communications. 2007;363:347–354. doi: 10.1016/j.bbrc.2007.08.178. [DOI] [PubMed] [Google Scholar]
- 27.Jian Q, Tao Z, Li Y, Yin ZQ. Acute retinal injury and the relationship between nerve growth factor, Notch1 transcription and short-lived dedifferentiation transient changes of mammalian Muller cells. Vision research. 2015;110:107–117. doi: 10.1016/j.visres.2015.01.030. [DOI] [PubMed] [Google Scholar]
- 28.Sanes JR, Masland RH. The types of retinal ganglion cells: current status and implications for neuronal classification. Annual review of neuroscience. 2015;38:221–246. doi: 10.1146/annurev-neuro-071714-034120. [DOI] [PubMed] [Google Scholar]
- 29.Morrone LA, et al. Natural compounds and retinal ganglion cell neuroprotection. Progress in brain research. 2015;220:257–281. doi: 10.1016/bs.pbr.2015.05.004. [DOI] [PubMed] [Google Scholar]
- 30.Pang JJ, et al. Comparative analysis of in vivo and in vitro AAV vector transduction in the neonatal mouse retina: effects of serotype and site of administration. Vision research. 2008;48:377–385. doi: 10.1016/j.visres.2007.08.009. [DOI] [PubMed] [Google Scholar]
- 31.Manuel M, et al. Controlled overexpression of Pax6 in vivo negatively autoregulates the Pax6 locus, causing cell-autonomous defects of late cortical progenitor proliferation with little effect on cortical arealization. Development. 2007;134:545–555. doi: 10.1242/dev.02764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Inami W, et al. Expression of Two Classes of Pax6 Transcripts in Reprogramming Retinal Pigment Epithelium Cells of the Adult Newt. Zoological science. 2016;33:21–30. doi: 10.2108/zs150111. [DOI] [PubMed] [Google Scholar]
- 33.Sasamoto Y, et al. PAX6 Isoforms, along with Reprogramming Factors, Differentially Regulate the Induction of Cornea-specific Genes. Scientific report. 2016;6:20807. doi: 10.1038/srep20807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bhatia B, Singhal S, Tadman DN, Khaw PT, Limb GA. SOX2 is required for adult human muller stem cell survival and maintenance of progenicity in vitro. Investigative ophthalmology & visual science. 2011;52:136–145. doi: 10.1167/iovs.10-5208. [DOI] [PubMed] [Google Scholar]
- 35.Bhatia B, Singhal S, Lawrence JM, Khaw PT, Limb GA. Distribution of Muller stem cells within the neural retina: evidence for the existence of a ciliary margin-like zone in the adult human eye. Experimental eye research. 2009;89:373–382. doi: 10.1016/j.exer.2009.04.005. [DOI] [PubMed] [Google Scholar]
- 36.Inoue M, et al. PAX6 and SOX2-dependent regulation of the Sox2 enhancer N-3 involved in embryonic visual system development. Genes to cells: devoted to molecular & cellular mechanisms. 2007;12:1049–1061. doi: 10.1111/j.1365-2443.2007.01114.x. [DOI] [PubMed] [Google Scholar]
- 37.Heavner WE, Andoniadou CL, Pevny LH. Establishment of the neurogenic boundary of the mouse retina requires cooperation of SOX2 and WNT signaling. Neural development. 2014;9:27. doi: 10.1186/1749-8104-9-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Taranova OV, et al. SOX2 is a dose-dependent regulator of retinal neural progenitor competence. Genes & development. 2006;20:1187–1202. doi: 10.1101/gad.1407906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Agathocleous M, et al. A directional Wnt/beta-catenin-Sox2-proneural pathway regulates the transition from proliferation to differentiation in the Xenopus retina. Development. 2009;136:3289–3299. doi: 10.1242/dev.040451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.McClure, C., Cole, K. L., Wulff, P., Klugmann, M. & Murray, A. J. Production and titering of recombinant adeno-associated viral vectors. Journal of visualized experiments: JoVE, e3348, 10.3791/3348 (2011). [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.