Abstract
Candida albicans infections present a heavy burden upon public health, with only a few drugs available, while biofilms formed by C. albicans worsen this situation. Dioscin has antitumor, anti-inflammatory, and hepatoprotective effects, and this study was conducted to evaluate the effects of dioscin on the biofilm formation and development, as well as other virulence factors of C. albicans such as morphological transition, adhesion, and extracellular secreted phospholipase. Our results showed dioscin inhibits these virulence factors and has low cytotoxicity against mammalian cells. Considering protective effects of dioscin against damage on liver and kidney, dioscin may be used as a potential candidate for antifungal development.
1. Introduction
Candida albicans severely influences health of immunocompromised individuals, as this opportunistic fungal pathogen could overgrow rather than grow under immune surveillance in these hosts. This kind of uncontrollable existence causes infections in oral cavity and gastrointestinal and urogenital tract, as well as the life-threatening bloodstream infections [1]. Patients with invasive Candida infections are often associated with high mortality (about forty percent), despite antifungal therapy [2, 3]. The paucity of available drugs and the unsatisfying profiles of such drugs, such as toxicity, resistance, and low bioavailability, recapitulate the necessity to develop new antifungals [4].
As a virulence factor, biofilms formed by C. albicans are a complicated three-dimensional structure, with yeast, hyphal, and pseudohyphal cells embedded in extracellular matrix. Biofilms provide a shelter for cells within from attack by antifungals and host immune system [3]. Therefore, C. albicans infections associated with biofilms are often refractory and recurrent. Examples are biofilms on surfaces of medical devices such as catheters and joint prosthesis [3, 5]. The removal of devices often results in therapeutic failure and the fee is high [5]. This highlights again the need for development new antifungals, especially those active against biofilms.
Dioscin is a steroidal saponin that could be isolated from medicinal herbs and vegetables of Dioscorea genus (family Dioscoreaceae), such as Dioscorea zingiberensis, Dioscorea nipponica, Dioscorea panthaica, Dioscorea spongiosa, and Dioscorea cayenensis [6–10]. Dioscin has been reported to have pharmacological activities against various kinds of tumors such as gastric, colon, and gallbladder cancer and osteosarcoma [11–14]. Meanwhile, this saponin has been documented with antiviral activities against adenovirus, vesicular stomatitis virus, and hepatitis B virus [15]. Dioscin also exerts anti-inflammatory activities through TLR4/MyD88/NF-κB signaling [16]. The antihyperuricemic activity of dioscin was reported to be through enhanced excretion and decreased reabsorption of uric acid in rat model of hyperuricemia [9]. Protective effects of dioscin against liver, lung, and renal damage, as well as pulmonary fibrosis and cardiotoxicity (induced by doxorubicin), have also been reported recently [17–21]. Dioscin could also promote the regeneration of liver, which was mediated by the activation of Notch1/Jagged 1 signaling [22]. Although the antifungal activity of dioscin against C. albicans, as well as other Candida species and Saprolegnia parasitica, has been reported [7, 10, 23], the effects of dioscin on the virulence factors of C. albicans have yet not been tested. In the present study, we evaluated the antifungal effects of dioscin on the biofilm formation and development, as well as virulence factors such as adhesion, morphological transition, and extracellular enzyme production.
2. Materials and Methods
2.1. Chemicals and Strains
C. albicans SC5314, C. albicans ATCC10231, Candida glabrata ATCC2001, and Candida parapsilosis ATCC22019 were obtained from China General Microbiological Culture Collection Center (CGMCC). All these strains were maintained on YPD (yeast extract 1%, peptone 2%, dextrose 2%) medium and stored at -80°C. Before each test, a colony from fungal strains subcultured twice on YPD agar was transferred into YPD medium and incubated overnight at 30°C with a speed of 140 rpm for propagation.
Dioscin was bought from Solarbio Life Sciences. N-acetyl-D-glucosamine (GlcNAc), XTT, MTT, menadione, propidium iodide (PI), and RPMI-1640 medium were bought from Sigma.
2.2. Antifungal Susceptibility Tests
Broth microdilution methods were employed to test the antifungal activities of dioscin against fungal strains according to CLSI-M27-A3 guidelines [24]. These assays were done in triplicate and repeated three times.
2.3. Time-Killing Assay
C. albicans SC5314 cells collected from overnight cultures in YPD medium were resuspended in 1640 medium to reach a concentration of 106 cells/mL. Cell suspensions were challenged with different concentrations of dioscin at 30°C. At indicated time points, an aliquot from each treatment was taken and plated on YPD agars after serial dilution. After incubation for 24 h, numbers of colony forming units (CFUs) on YPD agars were counted. This assay was conducted in triplicate and repeated three times.
2.4. Yeast-to-Hypha Transition
1640 medium, Spider medium (nutrient broth 1%, mannitol 1%, K2HPO4, pH7.2), and GlcNAc medium (GlcNAc 0.4%, HK2PO4 0.3%, peptone 0.5%) were used to explore the effects of dioscin on the yeast-to-hyphal morphological transition. C. albicans SC5314 cell suspension at a concentration of 1 x 106 cells/mL in each medium was transferred into wells of 96-well plates and dioscin in DMSO was added to each well to obtain different final concentrations (0, 1, 2, and 4 μg/mL). After incubation at 37°C for 4 hours, the morphologies of cells exposed to different concentrations of dioscin were recorded by microscope.
2.5. Biofilm Test
Biofilms of C. albicans SC5314 were formed in 96-well plates according to the well-established methods [25]. To test the inhibitory effect of dioscin on the biofilm formation, different final concentrations of dioscin were coincubated with C. albicans cells at 37°C for 24 h, which was followed by XTT reduction assay. Meanwhile, in another 96-well plate under the same conditions, cells in each well were determined by CFU counts as mentioned above.
As for the inhibitory effect on preformed biofilms, 24-hour old biofilms in 96-well plates were washed with PBS and exposed to fresh 1640 medium containing different concentrations of dioscin for another 24 h, which was followed by XTT reduction assay. Wells with only medium were set as blank while wells containing biofilm without drug exposure were set as negative controls. These assays were performed in triplicate and repeated three times.
2.6. XTT Reduction Assay
To evaluate the viability of cells in biofilms, 100 μL of sterile XTT solution (0.5 mg/mL in PBS) supplemented with 1μM menadione was added into each well, preceded by washing with PBS to remove free-floating fungal cells. After incubation at 37°C for 2 h in dark, 70 μL of the supernatant of each well was transferred into a new 96-well plate and the optical density at 490 nm (OD490) of each well was detected by a microplate reader (VarioSkan, thermo, Germany). The viability of biofilm in wells = (OD490 treated-OD490 blank)/(OD490 negative-OD490 blank) × 100%.
2.7. Confocal Laser Scanning Microscope (CLSM) Analysis
To study the influence of dioscin on 3D structures of C. albicans biofilms, biofilms formed in the presence of dioscin were stained with Calcofluor White (CFW, 50 μg/mL) and subjected to CLSM analysis (Olympus FV1000, Japan). Z-axis scanning was employed while the number of photo-slices (step size: 2 μm) varied among different biofilms. 3D graphs of biofilms were reconstructed by Imaris 7.2.3 software (Bitplane, Switzerland).
2.8. Adhesion Test
The capacity of C. albicans SC5314 cells to adhere to polystyrene surfaces was determined by calculating viability of the residual cells in 96-well plates after exposure to dioscin and washing with PBS [26]. After cells were treated with dioscin for 1.5 h at 37°C, cells-contained wells were washed with PBS and XTT solution was added to each well to determine the viability of cells challenged with dioscin.
To investigate whether the effects of dioscin on adhesion were due to the decrease in cell numbers caused by dioscin, viable cells in each well were counted. In brief, after treatment with different concentrations of dioscin for 1.5 h as aforementioned, all cells in each well were resuspended with sterile PBS, collected by vigorous aspiration using a pipette, and serially diluted and plated on YPD agars. After incubation at 37°C for 24 h, CFUs on each agar plate were counted. These experiments were performed in triplicate and repeated three times.
2.9. Phospholipase Assay
Egg yolk emulsion agar was used to assess the effect of dioscin on phospholipase production of C. albicans [27]. Briefly, 1 μL of fungal cell suspension was added to the center of egg yolk emulsion agar supplemented with different concentrations of dioscin and plates were incubated at 37°C for 96 h to allow the precipitation zones and colonies to form on agars. The Pz value was used to quantify the enzymatic production. Pz = dcolony/dconlony+precipitation where d means the diameter of colony or precipitation.
2.10. PI Influx Assay
After incubation at 37°C for 4 h, C. albicans cells in 1640 medium (106 cells/mL) were stained with PI (10 μM) and subjected to confocal microscope analysis.
2.11. Cytotoxicity Assessment
MTT assay was employed to evaluate the toxicity of dioscin against mammalian JEG3 cells [28]. Confluent cells grown at 37°C in a humidity of 5% CO2 were challenged with different concentrations of dioscin for 24 h, followed by MTT reduction assay.
2.12. Statistical Analysis
For each assay, at least three independent tests were performed to generate results expressed as mean + standard deviations. GraphPad Prism 6.02 was used to produce figures and to calculate the significances of differences between groups (treatment vs control) through Student's t-test.
3. Results
3.1. Antifungal Susceptibility
From the results of antifungal susceptibility tests (Table 1), dioscin showed significant antifungal activities against Candida species (C. albicans, C. glabrata, and C. parapsilosis), with MICs ranging from 2 to 4 μg/mL and MFCs ranging from 4 to 8 μg/mL. The activities of dioscin were fungicidal.
Table 1.
Antifungal profiles of dioscin against Candida species.
| Fungal strains | MIC (μg/mL) | MFC (μg/mL) | MFC/MIC | Antifungal effects |
|---|---|---|---|---|
| C. albicans SC5314 | 4 | 4 | 1 | Fungicidal |
| C. albicans ATCC10231 | 4 | 4 | 1 | Fungicidal |
| C. glabrata ATCC2001 | 2 | 4 | 2 | Fungicidal |
| C. parapsilosis ATCC22019 | 4 | 8 | 2 | Fungicidal |
Because of the similar antifungal profiles of two C. albicans strains displayed, we chose the widely used standard strain SC5314 for further investigations.
3.2. Time-Kill Kinetics
As a complementary assay to antifungal susceptibility tests, time-kill assays were performed (Figure 1), and it was found that, in the first four hours, treatment with 4 μg/mL dioscin caused a reduction of 3 log10 CFU/mL compared to the initial inoculum, indicating a fungicidal effect of dioscin on C. albicans, which was consistent with antifungal susceptibility results. However, since the 4th hour, the CFU of samples treated with 4 μg/mL dioscin started to recover, indicating a short-term fungicidal effect.
Figure 1.
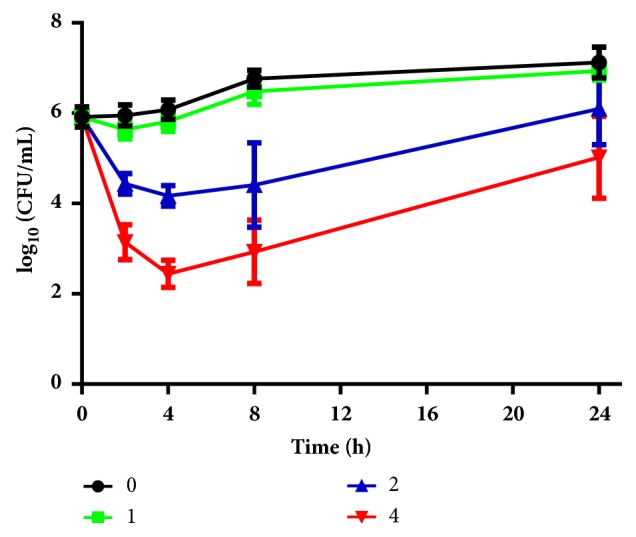
Time-kill kinetics of different concentrations of dioscin. C. albicans cell suspension with different concentrations of dioscin was grown at 30°C, 140 rpm. At indicated time points, 100 μL solution was taken from cultures and the viable cells within were determined by plating on YPD agars. CFU counts were performed after incubation for 24 h at 37°C. Assays were performed in triplicate and repeated three times.
3.3. Dioscin Inhibits Biofilm Formation and Development
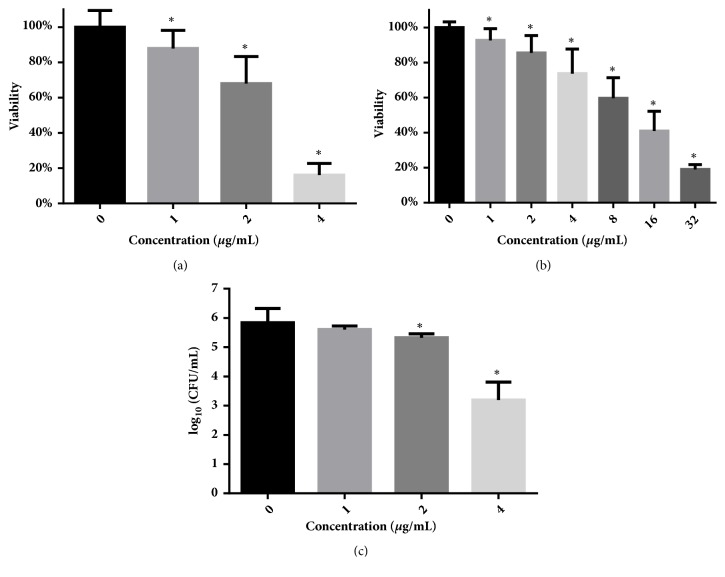
As shown in Figure 2(a), dioscin concentrations ranging from 1 to 4 μg/mL suppressed 10% ~ 80% of biofilm formation, while this concentration band could only reduce the viability of cells in mature biofilms by less than 30% (Figure 2(b)). Only at a high concentration of 16 μg/mL could this compound decrease more than 50% of cell viability of preformed biofilms (Figure 2(b)). The inhibitory effect of dioscin on biofilm formation could also be validated by confocal microscope, as biofilms that formed in the presence of increasing concentrations of dioscin showed decreased heights and proportion of filamentous cells (Figure 3). To quantify the cell numbers in biofilm after washing, CFU measurements were performed. As shown in Figure 2(c), dioscin decreased the cell numbers in biofilms formed in the presence of dioscin. This could also be confirmed by CLSM analysis in Figure 3, where, with increasing concentration of dioscin, the cell distribution in biofilms becomes sparser.
Figure 2.
Antibiofilm activities of dioscin against biofilm formation (a) and development (b) of C. albicans. The duration of treatment for both formation and development of biofilms was 24 h. Viability of biofilms treated was detected by XTT reduction assay. (c) Under biofilm-forming conditions, dioscin treatment did reduce cell numbers at sub-MIC concentrations. CFUs were counted by diluting cells in each well and plating on YPD agars. ∗ means p < 0.05.
Figure 3.
Reconstructed 3D graphs of biofilms formed under treatment with 0, 1, 2, and 4 μg/mL of dioscin for 24 h. Cells in biofilms were stained with Calcofluor White. Biofilms formed on polystyrene surfaces under dioscin exposure were recorded by confocal microscope and visualized by Imaris 7.2.3.
3.4. Dioscin Inhibits the Adhesion of C. albicans
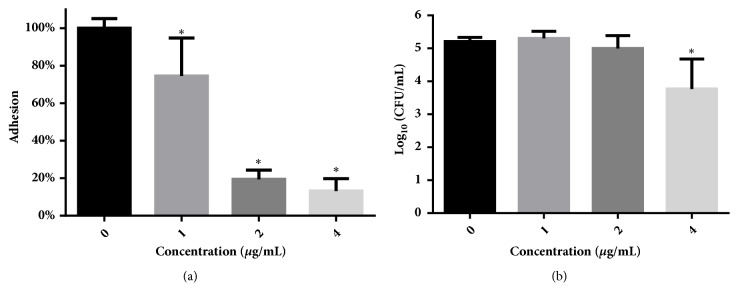
Based on our determinations that dioscin inhibited the biofilm formation, we speculated that dioscin may influence the adhesion of C. albicans cells to polystyrene surfaces since adhesion is the first step to form biofilms. To test our conjecture, we employed XTT assay to quantify the adhesion using viability of residual cells having undergone treatment with dioscin and PBS washing. As shown in Figure 4(a), dioscin inhibited the adhesion of C. albicans cells to polystyrene surfaces in a dose-dependent manner. To test whether this inhibition was due to the changes in cell numbers caused by dioscin, all cells of each group (adherent and nonadherent) were measured by CFU counts. As shown in Figure 4(b), although 4 μg/mL of dioscin reduced the viable cells significantly, 1 and 2 μg/mL of dioscin did not, suggesting that the reduced adhesion on polystyrene surfaces was not completely due to the alternations in cell numbers. In other words, dioscin did demonstrate inhibition on the adhesion of C. albicans cells on polystyrene surfaces.
Figure 4.
Dioscin inhibits the adhesion of C. albicans cells to polystyrene surfaces of 96-well plates. (a) Adhesion was calculated through comparing the residual viability of drug-treated cells left on surfaces after wash with those of drug-free controls, determined by XTT assay. (b) The inhibition of dioscin on adhesion was not completely due to the alterations in cell numbers. After 1.5 h incubation, cells in each well were collected and diluted for CFU counting. ∗, p < 0.05.
3.5. Dioscin Inhibits Yeast-to-Hyphal Transition of C. albicans
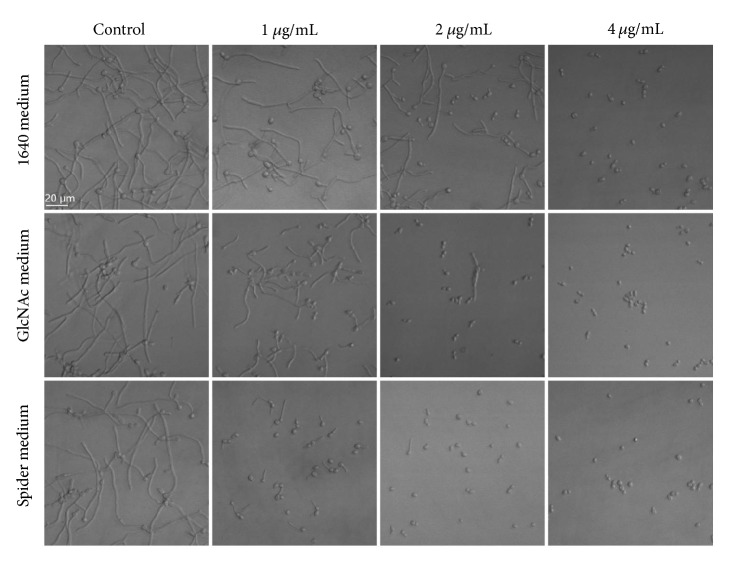
To test the inhibitory effects of dioscin on yeast-to-hyphal transition, we used three kinds of hyphal-inducing media, namely, 1640, Spider, and GlcNac medium. As shown in Figure 5, dioscin inhibited the morphological transition of C. albicans SC5314, although the extent of inhibition varied in different media.
Figure 5.
Dioscin inhibits morphological transition in different media. C. albicans cells (106 cells/mL) in 1640 medium, GlcNAc medium, and Spider medium were exposed to different concentrations of dioscin for 4 h at 37°C before the morphological changes were captures by inverted microscope.
3.6. Dioscin Inhibits the Production of Phospholipase
To investigate whether dioscin has an inhibitory effect on the production of C albicans phospholipase, egg yolk emulsion agars were employed. In this assay, a smaller Pz value means a higher enzyme production. As shown in Figure 6, 1-4 μg/mL of dioscin could decrease the production of phospholipase by C. albicans cells in a concentration-dependent manner.
Figure 6.
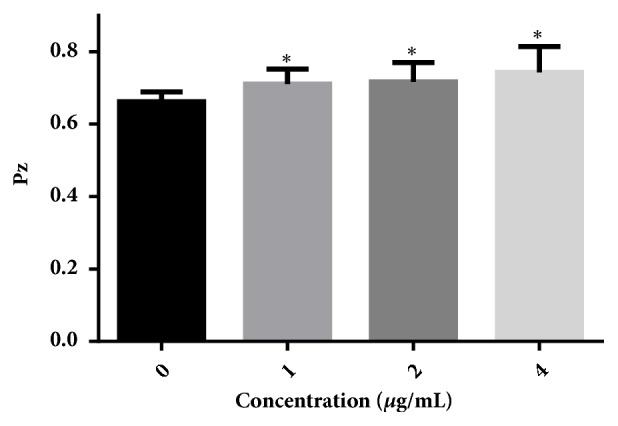
Dioscin inhibits the production of phospholipase secreted by C. albicans SC5314. In the phospholipase assay, a larger Pz value indicates less enzyme production. ∗, p < 0.05.
3.7. Dioscin Causes Damage in Cell Membrane
Cell membrane integrity was evaluated by staining with PI, a membrane-impermeable fluorescent dye that could not gain access into cell unless the plasma membrane is disrupted. As shown in Figure 7, incubation with dioscin could induce compromised cell membrane integrity in a dose-dependent manner, indicted by increased cells with red fluorescence.
Figure 7.

PI staining indicates that dioscin caused plasma membrane permeability. After being incubated with different concentrations of dioscin for 4 h, C. albicans cells were stained with PI and recorded by a confocal microscope.
3.8. Cytotoxicity of Dioscin
Cytotoxicity against human JEG3 cells was assessed using MTT assay. Although JEG3 cells treated with dioscin showed decreased viability with a half maximal inhibitory concentration (IC50) of about 13 μg/mL (as shown in Figure 8), the IC50 was higher than MICs against C. albicans cells, implying relatively low cytotoxicity.
Figure 8.
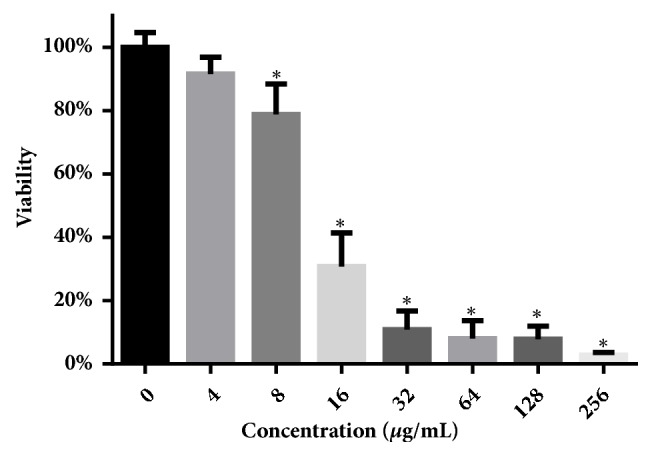
Cytotoxicity of dioscin against human JEG3 cells. After dioscin treatment for 24 h, MTT assay was performed to assess the inhibitory effect of dioscin on cell proliferation. ∗, p < 0.05.
4. Discussion
The burden on public health imposed by C. albicans infections and the limited antifungal drugs available against fungal pathogens, as well as the toxicity and drug resistance associated with conventional drugs, necessitate the development of new antifungal agents [29]. Recent years have seen increased enthusiasm in combating diseases with natural products, and dioscin is one of them [1, 21, 30]. The various protective effects of dioscin have been reported by others [18, 21], and the aim of this study was to investigate the effects of dioscin on virulence factors of C. albicans.
From antifungal susceptibility tests, we got a different MIC (4 μg/mL) against C. parapsilosis ATCC22019, compared with previously reported MIC (11.3±4.6 μg/mL) by others, which probably resulted from the different media (1640 vs YPD medium) and initial inoculum concentrations (2x103 vs 106 cells/mL) used [7].
Since saponin extract of Dioscorea panthaica Prain et Burk rhizomes which contains dioscin has been demonstrated to have antifungal effects against C. albicans, our results are consistent with these previous reports [11, 31–33]. However, 4 μg/mL of dioscin could only reduce the viable cells in the first four hours, followed by an increase in viable cells, which is different with our previous research on dioscin-containing extract [33]. At 16 μg/mL (4MIC), dioscin also showed similar curves (data not shown). This might indicate that in the extract there may exist other components with antifungal activity that synergize with dioscin, exerting a more powerful and longer fungicidal effect. The short-term fungicidal activity of dioscin was also seen in n-dodecanol, an alcohol which could induce transient fungicidal effect in Saccharomyces cerevisiae and could synergize with trans-anethole (at sub-inhibitory concentration) to produce a long-lasting fungicidal effects [34].
Although dioscin has been reported to have antifungal activity against C. albicans [7, 10], our results demonstrated for the first time that dioscin has antibiofilm activity, with IC50 for biofilm formation and development less than 4 μg/mL and 16 μg/mL, respectively. Given that azoles, such as fluconazole and voriconazole, have their BMIC90s (concentrations that inhibit 90% biofilm formation) high above 256 and 1000 μg/mL and that concentration required for AmB to inhibit biofilm formation is 8-31 times above its MIC [35], dioscin may make a promising candidate for antifungal drug development and optimization. Besides the viability analysis through XTT assay, results obtained by confocal microscope also confirmed the inhibitory effect of dioscin on biofilm formation of C. albicans. Combining results from XTT reduction, CFW staining, and CFU assays, the antibiofilm activity of dioscin may be due to the antifungal effects of dioscin against C. albicans. This is similar to the antifungal drugs caspofungin and lipid formulation of Amphotericin B, which showed efficacy on C. albicans biofilms [35].
Adhesion to abiotic or biotic surfaces is the first step to initiate the biofilm formation, thus making compounds inhibiting the adhesion attractive for antifungal development [36]. Multiple reported studies showed that compounds with antibiofilm activity also inhibit the adhesion process, such as filastatin, magnolol, honokiol, and dracorhodin perchlorate [28, 37, 38]. Dioscin has similar antiadhesion effects to those compounds.
The capacity of C. albicans to transit from yeast to hyphae phenotype has been considered as an important virulence factor during pathogenesis [39]; the influence of dioscin on the transition was investigated. In the three hyphal-inducing media, dioscin could significantly inhibit the hyphal growth. The different extent of hyphal inhibition may result from different inhibitory activity on distinct signaling pathways activated by individual stimulus [40], which need to be further validated.
As an important kind of extracellular hydrolase of C. albicans, phospholipase plays key roles in invasion and colonization by degrading components of epithelial cells. Mutants of phospholipase showed decreased colonization in major organs of infected mice, indicating attenuated virulence [39, 41]. In our research, dioscin reduced the production of phospholipase of C. albicans, consistent with other compounds with antifungal activity, such as dracorhodin perchlorate and quercetin [28, 42].
The surfactant activity of dioscin might underlie the antifungal effects through membrane-disruptive mechanism [7, 43]. In the current study, the membrane-damaging effects of dioscin have also been affirmed by increased PI influx induced by dioscin, as shown in Figure 7, which was in line with previous results published by others [7].
Considering the relatively low cytotoxicity of dioscin against human JEG3 cells, as well as the absence of obvious adverse effects in vivo via oral administration [14], dioscin may be effective against C. albicans infections in vivo, though much more need to be done.
Despite the fact that dioscin has good antifungal and antivirulence activities and relative low cytotoxicity, however, dioscin has a very low bioavailability through oral administration (approximately 0.2%) [43], which might limit the oral use of dioscin for systemic infections. Another drawback of dioscin is the poor solubility in aqueous solution, but there are already improvements designed to form copolymer micelles that facilitate the solubility [44]. Of note, the dioscin in mixed micelles demonstrated better antitumor activities against mammalian tumor cell lines, as compared to free dioscin, probably resulting from enhanced uptake by cells. The pharmacokinetic profiles have also been improved by this design, making it a potential drug-delivering system [44].
The brighter part of developing dioscin as a lead is that many analogous compounds have already been synthesized, making it more easier and more well-founded to decipher the structure-activity correlation and to optimize the chemical structures for antifungal therapy [45–47]. Another advantage of dioscin as an antifungal agent is that this compound has been used as a vital material for synthesizing other drugs such as cortisone [23], which means that dioscin could be easily used for industrial production. The third advantage of developing dioscin as an antifungal agent is safety or at least very low toxicity, considering that dioscin-containing herbs and vegetables have been consumed for centuries and that dioscin-containing DA-9801 has been approved by FDA for treating diabetic neuropathy [18, 48, 49].
5. Conclusion
In conclusion, dioscin showed significant antifungal activities against several Candida species tested through fungicidal effects. Dioscin could also inhibit the virulence factors of C. albicans, such as yeast-to-hyphal transition, extracellular phospholipase production, adhesion to abiotic surfaces, and biofilms formation. At higher concentrations, dioscin could even reduce the viability of preformed biofilms. Given the low toxicity dioscin has [18, 48, 49] and the readiness for structure-activity analysis and structural optimization [45–47], dioscin may make a promising candidate for antifungal therapies.
Acknowledgments
This work was supported by the Natural Science Foundation of Jilin Province [nos. 20180520118JH, 20170311015YY, and 20170414029GH].
Data Availability
The data used to support the findings of this study are included within the article. The more detailed data are available from the first author upon request.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Liu X., Ma Z., Zhang Ji., Yang L. Antifungal compounds against Candida Infections from Traditional Chinese Medicine. BioMed Research International. 2017;2017:12. doi: 10.1155/2017/4614183.4614183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kullberg B. J., Arendrup M. C. Invasive candidiasis. The New England Journal of Medicine. 2015;373(15):1445–1456. doi: 10.1056/nejmra1315399. [DOI] [PubMed] [Google Scholar]
- 3.Nobile C. J., Johnson A. D. Candida albicans biofilms and human disease. Annual Review of Microbiology. 2015;69(1):71–92. doi: 10.1146/annurev-micro-091014-104330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Perlin D. S., Rautemaa-Richardson R., Alastruey-Izquierdo A. The global problem of antifungal resistance: prevalence, mechanisms, and management. The Lancet Infectious Diseases. 2017;17(12):e383–e392. doi: 10.1016/S1473-3099(17)30316-X. [DOI] [PubMed] [Google Scholar]
- 5.Gulati M., Nobile C. J. Candida albicans biofilms: development, regulation, and molecular mechanisms. Microbes and Infection. 2016;18(5):310–321. doi: 10.1016/j.micinf.2016.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li J., Liang Q., Li C., Liu M., Zhang Y. Comparative transcriptome analysis identifies putative genes involved in dioscin biosynthesis in dioscorea zingiberensis. Molecules. 2018;23(2) doi: 10.3390/molecules23020454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cho J., Choi H., Lee J., Kim M.-S., Sohn H.-Y., Lee D. G. The antifungal activity and membrane-disruptive action of dioscin extracted from Dioscorea nipponica. Biochimica et Biophysica Acta. 2013;1828(3):1153–1158. doi: 10.1016/j.bbamem.2012.12.010. [DOI] [PubMed] [Google Scholar]
- 8.Tang Y., Yi T., Chen H., Zhao Z., Liang Z., Chen H. Quantitative comparison of multiple components in dioscorea nipponica and D. Panthaica by ultra-high performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry. Phytochemical Analysis. 2013;24(4):413–422. doi: 10.1002/pca.2428. [DOI] [PubMed] [Google Scholar]
- 9.Zhang Y., Jin L., Liu J., et al. Effect and mechanism of dioscin from Dioscorea spongiosa on uric acid excretion in animal model of hyperuricemia. Journal of Ethnopharmacology. 2018;214:29–36. doi: 10.1016/j.jep.2017.12.004. [DOI] [PubMed] [Google Scholar]
- 10.Sautour M., Miyamoto T., Dongmo A. Antifungal steroid saponins from dioscorea cayenensis. Planta Medica. 2004;70(1):90–92. doi: 10.1055/s-2004-815467. [DOI] [PubMed] [Google Scholar]
- 11.Dong M., Feng X.-Z., Wang B.-X., Wu L.-J., Ikejima T. Two novel furostanol saponins from the rhizomes of Dioscorea panthaica prain et burkill and their cytotoxic activity. Tetrahedron. 2001;57(3):501–506. doi: 10.1016/S0040-4020(00)01024-3. [DOI] [Google Scholar]
- 12.Li S., Li S., Cheng B., et al. Dioscin inhibits colon cancer cells’ growth by reactive oxygen species-mediated mitochondrial dysfunction and p38 and JNK pathways. Anti-Cancer Drugs. 2018;29(3):234–242. doi: 10.1097/CAD.0000000000000590. [DOI] [PubMed] [Google Scholar]
- 13.Song X., Wang Z., Liang H., et al. Dioscin induces gallbladder cancer apoptosis by inhibiting ros-mediated PI3K/AKT signalling. International Journal of Biological Sciences. 2017;13(6):782–793. doi: 10.7150/ijbs.18732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Liu W., Zhao Z., Wang Y., et al. Dioscin inhibits stem-cell-like properties and tumor growth of osteosarcoma through Akt/GSK3/β-catenin signaling pathway. Cell Death & Disease. 2018;9, article 343 doi: 10.1038/s41419-018-0363-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu C., Wang Y., Wu C., et al. Dioscin’s antiviral effect in vitro. Virus Research. 2013;172(1-2):9–14. doi: 10.1016/j.virusres.2012.12.001. [DOI] [PubMed] [Google Scholar]
- 16.Zhu S., Tang S., Su F. Dioscin inhibits ischemic stroke-induced inflammation through inhibition of the TLR4/MyD88/NF-κB signaling pathway in a rat model. Molecular Medicine Reports. 2018;17(1):660–666. doi: 10.3892/mmr.2017.7900. [DOI] [PubMed] [Google Scholar]
- 17.Li C., Lu Y., Du S., et al. Dioscin exerts protective effects against crystalline silica-induced pulmonary fibrosis in mice. Theranostics. 2017;7(17):4255–4275. doi: 10.7150/thno.20270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Qiao Y., Xu L., Tao X., et al. Protective effects of dioscin against fructose-induced renal damage via adjusting Sirt3-mediated oxidative stress, fibrosis, lipid metabolism and inflammation. Toxicology Letters. 2018;284:37–45. doi: 10.1016/j.toxlet.2017.11.031. [DOI] [PubMed] [Google Scholar]
- 19.Zheng L., Yin L., Xu L., et al. Protective effect of dioscin against thioacetamide-induced acute liver injury via FXR/AMPK signaling pathway in vivo. Biomedicine & Pharmacotherapy. 2018;97:481–488. doi: 10.1016/j.biopha.2017.10.153. [DOI] [PubMed] [Google Scholar]
- 20.Zeng H., Yang L., Zhang X., Chen Y., Cai J. Dioscin prevents LPS-induced acute lung injury through inhibiting the TLR4/MyD88 signaling pathway via upregulation of HSP70. Molecular Medicine Reports. 2018;17(5):6752–6758. doi: 10.3892/mmr.2018.8667. [DOI] [PubMed] [Google Scholar]
- 21.Zhao L., Tao X., Qi Y., Xu L., Yin L., Peng J. Protective effect of dioscin against doxorubicin-induced cardiotoxicity via adjusting microRNA-140-5p-mediated myocardial oxidative stress. Redox Biology. 2018;16:189–198. doi: 10.1016/j.redox.2018.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Xu L., Gu L., Tao X., et al. Effect of dioscin on promoting liver regeneration via activating Notch1/Jagged1 signal pathway. Phytomedicine. 2018;38:107–117. doi: 10.1016/j.phymed.2017.11.006. [DOI] [PubMed] [Google Scholar]
- 23.Liu L., Shen Y., Liu G., et al. Inhibition of dioscin on Saprolegnia in vitro. FEMS Microbiology Letters. 2015;362(24) doi: 10.1093/femsle/fnv196.fnv196 [DOI] [PubMed] [Google Scholar]
- 24.Shokri H., Sharifzadeh A. Fungicidal efficacy of various honeys against fluconazole-resistant Candida species isolated from HIV(+) patients with candidiasis. Journal de Mycologie Médicale. 2017;27(2):159–165. doi: 10.1016/j.mycmed.2017.01.004. [DOI] [PubMed] [Google Scholar]
- 25.Siles S. A., Srinivasan A., Pierce C. G., Lopez-Ribot J. L., Ramasubramanian A. K. High-throughput screening of a collection of known pharmacologically active small compounds for identification of candida albicans biofilm inhibitors. Antimicrobial Agents and Chemotherapy. 2013;57(8):3681–3687. doi: 10.1128/AAC.00680-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li Y., Chang W., Zhang M., Ying Z., Lou H. Natural product solasodine-3-O-ß-D-glucopyranoside inhibits the virulence factors of Candida albicans. FEMS Yeast Research. 2015;15(6) doi: 10.1093/femsyr/fov060. [DOI] [PubMed] [Google Scholar]
- 27.Padmavathi A. R., Bakkiyaraj D., Thajuddin N., Pandian S. K. Effect of 2, 4-di-tert-butylphenol on growth and biofilm formation by an opportunistic fungus Candida albicans. Biofouling. 2015;31(7):565–574. doi: 10.1080/08927014.2015.1077383. [DOI] [PubMed] [Google Scholar]
- 28.Yang L., Liu X., Lv L., Ma Z., Feng X., Ma T. Dracorhodin perchlorate inhibits biofilm formation and virulence factors of Candida albicans. Journal de Mycologie Médicale. 2018;28(1):36–44. doi: 10.1016/j.mycmed.2017.12.011. [DOI] [PubMed] [Google Scholar]
- 29.Salci T. P., Negri M., Abadio A. K. R., Svidzinski T. I. E., Kioshima É. S. Targeting Candida spp. to develop antifungal agents. Drug Discovery Therapy. 2018;23(4):802–814. doi: 10.1016/j.drudis.2018.01.003. [DOI] [PubMed] [Google Scholar]
- 30.Si L., Xu L., Yin L., et al. Potent effects of dioscin against pancreatic cancer via miR-149-3P-mediated inhibition of the Akt1 signalling pathway. British Journal of Pharmacology. 2017;174(7):553–568. doi: 10.1111/bph.13718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang W., Li P., Wang X., Jing W., Chen L., Liu A. Quantification of saponins in Dioscorea panthaica Prain et Burk rhizomes with monolithic column using rapid resolution liquid chromatography coupled with a triple quadruple electrospray tandem mass spectrometry. Journal of Pharmaceutical and Biomedical Analysis. 2012;71:152–156. doi: 10.1016/j.jpba.2012.07.018. [DOI] [PubMed] [Google Scholar]
- 32.Wang W., Zhao Y., Jing W., et al. Ultrahigh-performance liquid chromatography-ion trap mass spectrometry characterization of the steroidal saponins of Dioscorea panthaica Prain et Burkill and its application for accelerating the isolation and structural elucidation of steroidal saponins. Steroids. 2015;95:51–65. doi: 10.1016/j.steroids.2014.12.023. [DOI] [PubMed] [Google Scholar]
- 33.Yang L., Liu X., Zhuang X., Feng X., Zhong L., Ma T. Antifungal effects of Saponin extract from Rhizomes of Dioscorea panthaica Prain et Burk against Candida albicans. Evidence-Based Complementary and Alternative Medicine. 2018;2018:13. doi: 10.1155/2018/6095307.6095307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fujita K.-I., Ishikura T., Jono Y., et al. Anethole potentiates dodecanol's fungicidal activity by reducing PDR5 expression in budding yeast. Biochimica et Biophysica Acta (BBA) - General Subjects. 2017;1861(2):477–484. doi: 10.1016/j.bbagen.2016.09.010. [DOI] [PubMed] [Google Scholar]
- 35.Kuhn D. M., George T., Chandra J., Mukherjee P. K., Ghannoum M. A. Antifungal susceptibility of Candida biofilms: unique efficacy of amphotericin B lipid formulations and echinocandins. Antimicrobial Agents and Chemotherapy. 2002;46(6):1773–1780. doi: 10.1128/AAC.46.6.1773-1780.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cui J., Ren B., Tong Y., Dai H., Zhang L. Synergistic combinations of antifungals and anti-virulence agents to fight against Candida albicans. Virulence. 2015;6(4):362–371. doi: 10.1080/21505594.2015.1039885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fazly A., Jain C., Dehner A. C., et al. Chemical screening identifies filastatin, a small molecule inhibitor of Candida albicans adhesion, morphogenesis, and pathogenesis. Proceedings of the National Acadamy of Sciences of the United States of America. 2013;110(33):13594–13599. doi: 10.1073/pnas.1305982110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sun L., Liao K., Wang D. Effects of magnolol and honokiol on adhesion, yeast-hyphal transition, and formation of biofilm by candida albicans. PLoS ONE. 2015;10(2) doi: 10.1371/journal.pone.0117695.e0117695 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mayer F. L., Wilson D., Hube B. Candida albicans pathogenicity mechanisms. Virulence. 2013;4(2):119–128. doi: 10.4161/viru.22913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Huang G. Regulation of phenotypic transitions in the fungal pathogen Candida albicans. Virulence. 2012;3(3):251–261. doi: 10.4161/viru.20010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Theiss S., Ishdorj G., Brenot A., et al. Inactivation of the phospholipase B gene PLB5 in wild-type Candida albicans reduces cell-associated phospholipase A2 activity and attenuates virulence. International Journal of Medical Microbiology. 2006;296(6):405–420. doi: 10.1016/j.ijmm.2006.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Singh B. N., Upreti D. K., Singh B. R., et al. Quercetin sensitizes fluconazole-resistant Candida albicans to induce apoptotic cell death by modulating quorum sensing. Antimicrobial Agents and Chemotherapy. 2015;59(4):2153–2168. doi: 10.1128/AAC.03599-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Li K., Tang Y., Fawcett J. P., Gu J., Zhong D. Characterization of the pharmacokinetics of dioscin in rat. Steroids. 2005;70(8):525–530. doi: 10.1016/j.steroids.2004.11.014. [DOI] [PubMed] [Google Scholar]
- 44.Zhao J., Xu Y., Wang C., et al. Soluplus/TPGS mixed micelles for dioscin delivery in cancer therapy. Drug Development and Industrial Pharmacy. 2017;43(7):1197–1204. doi: 10.1080/03639045.2017.1304956. [DOI] [PubMed] [Google Scholar]
- 45.Deng S., Yu B., Hui Y., Yu H., Han X. Synthesis of three diosgenyl saponins: Dioscin, polyphyllin D, and balanitin 7. Carbohydrate Research. 1999;317(1-4):53–62. doi: 10.1016/S0008-6215(99)00066-X. [DOI] [PubMed] [Google Scholar]
- 46.Li M., Han X., Yu B. Synthesis of monomethylated dioscin derivatives and their antitumor activities. Carbohydrate Research. 2003;338(2):117–121. doi: 10.1016/S0008-6215(02)00443-3. [DOI] [PubMed] [Google Scholar]
- 47.Chen P., Wang P., Song N., Li M. Convergent synthesis and cytotoxic activities of 26-thio- and selenodioscin. Steroids. 2013;78(9):959–966. doi: 10.1016/j.steroids.2013.05.018. [DOI] [PubMed] [Google Scholar]
- 48.Kang K. B., Ryu J., Cho Y., Choi S.-Z., Son M., Sung S. H. Combined Application of UHPLC-QTOF/MS, HPLC-ELSD and 1H–NMR Spectroscopy for Quality Assessment of DA-9801, A Standardised Dioscorea Extract. Phytochemical Analysis. 2017;28(3):185–194. doi: 10.1002/pca.2659. [DOI] [PubMed] [Google Scholar]
- 49.Nishina A., Ebina K., Ukiya M., et al. Dioscin Derived from Solanum melongena L. "Usukawamarunasu" Attenuates α-MSH-Induced Melanogenesis in B16 Murine Melanoma Cells via Downregulation of Phospho-CREB and MITF. Journal of Food Science. 2015;80(10):H2354–H2359. doi: 10.1111/1750-3841.13068. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study are included within the article. The more detailed data are available from the first author upon request.