Abstract
Background/aims
There is considerable interest in novel techniques to quantify choroidal blood flow (CBF) in humans. In the present study, we investigated a novel technique to measure CBF based on laser speckle flowgraphy (LSFG) in healthy subjects.
Methods
This study included 31 eyes of 31 healthy, non-smoking subjects aged between 19 and 74 years. A commercial LSFG instrument was used to measure choroidal vessel diameter (CVD) and relative flow volume (RFV) in choroidal vessels that were identified on fundus photos, an approach that was used previously only for retinal vessels. The reproducibility and the effect of isometric exercise on these parameters were investigated. The latter was compared with measurement of subfoveal CBF using laser Doppler flowmetry (LDF).
Results
Intraclass correlation coefficients for CVD and RFV were higher than 0.8 indicating excellent reproducibility. During isometric exercise, we observed an increase in ocular perfusion pressure of approximately 60% (P<0.001). The increase in RFV and CBF was lower, but also highly significant versus baseline (at minute 6 of isometric exercise: RFV 10.5%±4.2%, CBF 8.3%±3.6%; P<0.001 each) indicating choroidal autoregulation.
Conclusion
LSFG may be a novel approach to study blood flow in choroidal vessels. Data are reproducible and show good agreement with LDF data.
Trial registration number
NCT02102880, Results.
Keywords: imaging, choroid, physiology
Introduction
There is increasing interest in the quantification of choroidal blood flow (CBF), related to accumulating evidence that abnormalities in choroidal perfusion play a role in the pathogenesis of age-related macular degeneration (AMD).1–3 To image and measure CBF with optical techniques is, however, not easy because light is strongly scattered in the retinal pigment epithelium (RPE). Laser Doppler flowmetry (LDF) has been used to study blood flow in the choroid, but measurements are limited to the subfoveal area, because of the lack of retinal vessels.4 Colour Doppler imaging assesses blood velocities in the retrobulbar vessels including the posterior ciliary arteries supplying the choroid,5 but no information on volumetric blood flow can be obtained and reproducibility is poor.6 Indocyanine green angiography provides visualisation of the choroid, but the technique requires intravenous administration of a dye and extraction of quantitative blood flow data has proven difficult.7 Recently, optical coherence tomography angiography (OCTA) has attracted much interest but up to now quantification of blood flow is not possible.8 9 Quantification of blood flow in larger choroidal blood vessels was, however, achieved using Doppler OCT in a small group of healthy subjects.10
Using these technologies, several links between abnormal choroidal perfusion and AMD were established. Two longitudinal studies have shown that low CBF is a risk factor for the development of choroidal neovascularisation.11 12 In the Comparison of Age-related Macular Degeneration Treatment Trials (CATT) study, patchy choroidal filling was identified as an important risk factor for vision loss during anti-vascular endothelial growth factor (VEGF) therapy.13 A variety of OCTA provided links between choroidal vascular changes and different stages of AMD.14–17
Laser speckle flowgraphy (LSFG) is a technique that quantifies blood flow based on speckle statistics.18 This technique has been used to study optic nerve head blood flow,19 20 but when retinal areas are measured the signal contains proportions from both retinal and choroidal circulations.18 21
Materials and methods
Subjects
The study population consisted of a subgroup of healthy subjects, who participated in previously published studies.22 23 All research adhered to the guidelines set forth in the Declaration of Helsinki. Written informed consent was obtained from all participating volunteers after the nature and possible consequences of the study were explained in detail. Those subjects in whom a choroidal vessel was clearly identified were chosen for the present analysis. For reproducibility analysis, we included 21 subjects, and 10 subjects were included for the analysis during isometric exercise.
Prior to the study, all subjects underwent a comprehensive screening examination comprising medical history, physical examination, best-corrected visual acuity testing using standard ETDRS charts, slit-lamp examination including indirect funduscopy, measurement of intraocular pressure (IOP) using Goldmann applanation tonometry, measurements of systolic blood pressure (SBP) and diastolic blood pressure (DBP) with automated oscillometry and a urine pregnancy test in women with childbearing potential. The inclusion and exclusion criteria of the study were published previously.22 23 Twelve hours prior to the study, subjects were instructed to abstain from alcohol and beverages containing xanthine derivatives such as tea, coffee or cola.
Protocol
Reproducibility analysis was based on the protocol published previously.22 Briefly, three consecutive LSFG scans were acquired with a natural scotopic pupil; after pharmacological dilation of the pupil with 0.5% topical tropicamide eye drops (Mydriaticum Agepha Augentropfen; Agepha Ges.m.b.H., Vienna, Austria) and a resting period of 20 min, a second set of three consecutive LSFG measurements was acquired. The scans performed in mydriasis were used to assess reproducibility. At the end of the experiments, we performed measurements of IOP and systemic blood pressure. Experiments during isometric exercise followed the protocol described in Witwowska et al.23 A baseline recording using LSFG was performed and systemic blood pressure, pulse rate (PR) and IOP were taken while subjects were seated in a chair. Thereafter, isometric exercise was started and maintained for 6 min. This period of isometric exercise consisted of squatting in a position where the upper and the lower legs formed almost a right angle. For this purpose, the chair was carefully removed and the subjects were asked to remain in their position. LSFG measurements of blood flow, as well as readings for systemic blood pressure and PR were performed every minute throughout these experiments. In addition, a second period of isometric exercise was performed after a resting period of at least 20 min, which followed the same schedule as the first period. During the second session of isometric exercise, subfoveal CBF was, however, measured using LDF as described previously.24 25
Laser speckle flowgraphy
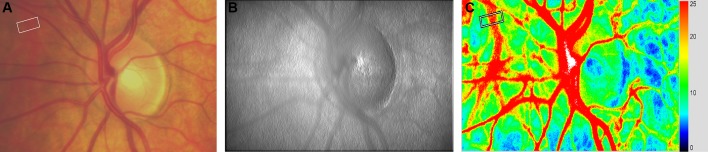
In the present study, we used a commercially available LSFG system (LSFG-NAVI; Softcare Co, Fukuoka, Japan) to study CBF. The principles of LSFG have previously been summarised in detail.18 Briefly, the device consists of a fundus camera supplied with an 830 nm diode laser and a digital charge-coupled device camera. The output parameter of LSFG is mean blur rate (MBR), which constitutes a measure of relative blood flow velocity in arbitrary units (AU) and is calculated using first order speckle statistics. One LSFG scan consists of a total of 118 images captured at a rate of 30 Hz resulting in total measurement time of approximately 4 s. The analysis software (LSFG Analyzer, Version 3.1.58; Softcare Co) produces a ‘composite map’ which colour codes perfusion at the posterior eye pole within one cardiac cycle. Recently, a technique was introduced to extract blood flow information from retinal vessels. For this purpose, a rectangular band (RB) is centred on the retinal vessel of interest. The signal from this area contains information from the blood stream within the vessel as well as from the microvasculature beneath the vessel. The signal arising from the larger vessels can automatically be separated from background MBR originating from the surrounding microvasculature by computing a threshold between MBR values. The software extracts the vessel diameter as expressed in pixels as well as the relative flow volume (RFV) in AU. In the present study, we extended this concept that was originally developed for retinal vessels to choroidal vessels (figure 1). Larger choroidal vessels were identified on fundus images and LSFG intensity maps. The vessel selection was restricted to the 21° field of view of the LSFG camera, which was centred at the optic nerve head. Afterwards, a RB having three times the width of the choroidal vessel of interest was placed over this vessel in the LSFG perfusion map and analysis was done in a similar way as previously described for retinal vessels.21 During the vessel selection, we took care to draw a RB which encompassed the choroidal vessel of interest without including any other larger vessel. The size of the RB on the first scan was saved and then exported to all the following scans for each subject.
Figure 1.
Identification of choroidal blood vessels for Laser Speckle Flowgraphy (LSFG) analysis. (A) First a choroidal vessel is identified based on a fundus photograph. The choroidal vessel of interest is shown inside a white rectangular band that is manually placed. (B) The presence of the choroidal vessel is then confirmed at the LSFG intensity image. (C) In the perfusion map, a rectangular white band is traced across the vessel which appears red because of the high intravascular blood velocity.
Laser Doppler flowmetry
Continuous measurement of CBF was performed by LDF as described previously.26 Briefly, vascularised tissue is illuminated by coherent laser light and the following haemodynamic parameters are determined from the Doppler shift power spectrum: the mean velocity of the red blood cells moving in the sampled tissue proportional to the mean Doppler frequency shift, the blood volume defined as the number of moving red blood cells proportional to the amount of Doppler shifted light and CBF calculated as the product of velocity and volume. In the present study, a laser portable confocal Doppler flowmeter was used.27
Systemic haemodynamics
SBP and DBP were measured using an automated oscillometric device. Mean arterial blood pressure (MAP) and ocular perfusion pressure (OPP) in the seated position were calculated as follows: MAP=DBP+1/3(SBP–DBP), OPP=2/3 MAP–IOP.
Statistical analysis
Descriptive data are presented as mean and SD. A repeated measures analysis of variance (ANOVA) model was used to characterise the effects of isometric exercise on the outcome parameters. To quantify reliability of measurements, intraclass correlation coefficients () were calculated using the three repeated measurements. The calculation of is based on a repeated measure ANOVA model using the variance among subjects (), the variance among measurements () and the residual error variance () and is calculated as
| . |
A of 1 means perfect reproducibility. The higher the intraclass correlation coefficient, the less is the variability of the method. In addition, the coefficients of variation were calculated. A P value of <0.05 was used as an indicator of statistical significance. All statistical analysis was carried out using CSS Statistica (Version 6.0, Tulsa, Oklahoma, USA).
Results
In a previous study on the reproducibility of LSFG in Caucasian subjects, we included a total of 80 subjects equally distributed in four age groups (group 1: 18–34 years; group 2: 35–49 years; group 3: 50–64 years; group 4: 65–80 years) each consisting of 10 male and 10 female subjects.22 For the present reproducibility analysis, a subgroup of 21 subjects was included (table 1). The subgroup of subjects in whom the effect of isometric exercise was investigated consisted of volunteers that were included in a study on the effect of squatting on optic nerve head blood flow (table 1).23
Table 1.
Demographic and baseline characteristics of subjects.
| Reproducibility study | |
| Sex (M/F) | 11/10 |
| Age (years) | 45.3±11.8 |
| Mean arterial pressure (mm Hg) | 96.4±3.5 |
| Pulse rate (beats/min) | 71.2±3.1 |
| Intraocular pressure (mm Hg) | 12.8±2.3 |
| Ocular perfusion pressure (mm Hg) | 51.2±6.1 |
| Relative flow volume (AU) | 268.3±53.1 |
| Choroidal vessel diameter (AU) | 27.8±5.5 |
| Isometric exercise study | |
| Sex (M/F) | 6/4 |
| Age (years) | 24.8±4.0 |
| Mean arterial pressure (mm Hg) | 91.8±2.7 |
| Pulse rate (beats/min) | 65.9±5.3 |
| Intraocular pressure (mm Hg) | 13.5±2.6 |
| Ocular perfusion pressure (mm Hg) | 46.8±5.9 |
| Relative flow volume (AU) | 303.5±67.1 |
| Choroidal vessel diameter (AU) | 31.3±7.0 |
| Choroidal blood flow (AU) | 33.3±4.7 |
Data are presented as means±SD.
Table 2 summarises the coefficient of variation and intraclass correlation coefficient for vessel diameters and RFV in the selected choroidal vessels. The RFV values at the three measurements were 270.2±55.3 AU, 266.1±52.7 AU and 265.6±53.9 AU, respectively. Values for CVD also showed little variability with values of 27.5±5.7 AU, 28.1±5.9 AU and 27.9±5.3 AU. According to Cicchetti, intraclass correlation coefficients of 0.75 and higher are considered as excellent reproducibility.28
Table 2.
Repeatability indices for laser speckle flowgraphy parameters
| Coefficient of variation (%) | Intraclass correlation coefficient | |
| Relative flow volume (AU) | 7.7 | 0.86 |
| Choroidal vessel diameter (AU) | 9.7 | 0.82 |
Changes in CVD, RFV and OPP during isometric exercise are presented in figure 2. The CBF and OPP data as obtained during the second period of isometric exercise are presented in figure 3. The increase in RFV, CBF and OPP was highly significant (P<0.001 each). The per cent change in OPP was, however, more pronounced than the increase in RFV and CBF indicating choroidal autoregulation. When we compared the time course of RFV and CBF, we observed a slightly higher increase with the former parameter (at minute 6 of isometric exercise: RFV: 10.5%±4.2%, CBF: 8.3%±3.6%). These data were, however, obtained during different periods of isometric exercise, but differences between the OPP increase during the two periods were small. The choroidal vessel diameter showed a tendency to decrease during isometric exercise, but this effect was not significant (at minute 6 of isometric exercise: −3.8±4.2%; P=0.08).
Figure 2.
Ocular perfusion pressure (OPP), relative flow volume (RFV) and choroidal vessel diameter (CVD) during isometric exercise. Data are expressed as per cent change from baseline. Data are presented as means±SD (n=10).
Figure 3.
Ocular perfusion pressure (OPP) and choroidal blood flow (CBF) during isometric exercise. Data are expressed as per cent change from baseline. Data are presented as means±SD (n=10).
Discussion
Herein, we present a novel approach to quantify CBF based on LSFG technology. The approach provides acceptable reproducibility and is capable of characterising changes in CBF induced by isometric exercise. Given that there is currently no other method commercially available for obtaining quantitative information on CBF, this technique may have considerable potential for understanding choroidal perfusion abnormalities in diseases of the outer retina.
The increase in CBF as observed with this technique during isometric exercise was slightly larger than the changes as obtained with LDF, but followed the same direction. In this context, several important differences between the current approach and LDF need to be mentioned which may account for these differences. On the one hand, LDF assesses the microcirculation in the subfoveal choroid, whereas the LSFG-based technique selects one larger vessel. As such LDF measures blood flow in choriocapillaris and LSFG measures blood flow in single vessels in Sattler’s or Haller’s layer.
A major advantage of the present approach is that it relies on a commercially available technique that can easily be implemented. There are, however, also several drawbacks with the technique. On the one hand, we were not able to identify choroidal vessels that were suitable for analysis in all participating subjects. On the other hand, we and others have previously shown that there may be a zero-setoff with RFV measurements when applied in retinal vessels.21 29 These conclusions were drawn based on comparison with other techniques to measure retinal blood flow such as laser Doppler velocimetry or Doppler OCT.30 31 This may also be the case when measurements of RFV are applied to choroidal vessels, but due to the absence of a gold standard technique for CBF evaluation such validation experiments cannot be performed in choroidal vessels. Since LSFG is based on a decorrelation analysis one may, however, assume that the outcome parameters do not scale linearly with blood velocity over the entire velocity range.14 Reproducibility of our approach is in the same range to that of previous techniques for the assessment of CBF6 such as pneumotonometry,32 laser interferometric measurement of fundus pulsation33 and LDF26 and superior to that of colour Doppler imaging of retrobulbar posterior ciliary arteries.6 Further studies are required to investigate what is the minimum diameter of choroidal vessels that can be studied with this approach.
The results during isometric exercise are in good agreement with previous data24 25 34–36 and support the concept that the choroid is not a passive vascular bed but rather shows characteristics of autoregulation. During isometric exercise, we observed a tendency towards vasoconstriction, which did, however, not reach the level of significance, most likely because of the small sample size and the limited pixel resolution of the employed charge-coupled device camera. This is compatible with the idea of myogenic autoregulation which has been proposed by Kiel and co-workers previously based on a set of experiments in rabbits.37–39
In conclusion, our data indicate that quantitative blood flow information of choroidal blood vessels can be extracted using a commercial LSFG system. Future studies are warranted to investigate the potential of this technique in studying CBF in diseases such as AMD in which perfusion abnormalities are assumed to play a role in the development of late stage disease.
Acknowledgments
The LSFG instrument was provided for free by Nidek. The company did, however, not influence data collection, evaluation or interpretation.
Footnotes
Contributors: GC: conceptualisation, formal analysis, investigation, methodology, visualisation, writing the original draft. KF, AMB, NL and PAW: formal analysis, methodology and investigation. KJW: investigation and project administration. MB: funding acquisition and methodology. AP-C: conceptualisation and project administration. RMW: conceptualisation, formal analysis, investigation, methodology and supervision. DS: conceptualisation, formal analysis, investigation and methodology. GG: conceptualisation, formal analysis, investigation, methodology, supervision, validation and visualisation. LS: conceptualisation, formal analysis, funding acquisition, methodology, project administration, supervision, validation, visualisation and writing the original draft. All authors contributed to writing, reviewing and editing the final manuscript.
Funding: Financial support from the Austrian Science Fund (Fonds zur Förderung der Wissenschaftlichen Forschung; www.fwf.ac.at) Grant 21406 and project KLI 340 is gratefully acknowledged.
Competing interests: None declared.
Patient consent: Obtained.
Ethics approval: Ethics Committee of the Medical University of Vienna.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1. Pemp B, Schmetterer L. Ocular blood flow in diabetes and age-related macular degeneration. Can J Ophthalmol 2008;43:295–301. 10.3129/i08-049 [DOI] [PubMed] [Google Scholar]
- 2. Feigl B. Age-related maculopathy – linking aetiology and pathophysiological changes to the ischaemia hypothesis. Prog Retin Eye Res 2009;28:63–86. 10.1016/j.preteyeres.2008.11.004 [DOI] [PubMed] [Google Scholar]
- 3. Kashani AH, Chen CL, Gahm JK, et al. Optical coherence tomography angiography: a comprehensive review of current methods and clinical applications. Prog Retin Eye Res 2017;60:66–100. 10.1016/j.preteyeres.2017.07.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Riva CE, Geiser M, Petrig BL, et al. Ocular blood flow assessment using continuous laser Doppler flowmetry. Acta Ophthalmol 2010;88:622–9. 10.1111/j.1755-3768.2009.01621.x [DOI] [PubMed] [Google Scholar]
- 5. Stalmans I, Vandewalle E, Anderson DR, et al. Use of colour Doppler imaging in ocular blood flow research. Acta Ophthalmol 2011;89:e609–e630. 10.1111/j.1755-3768.2011.02178.x [DOI] [PubMed] [Google Scholar]
- 6. Polska E, Polak K, Luksch A, et al. Twelve hour reproducibility of choroidal blood flow parameters in healthy subjects. Br J Ophthalmol 2004;88:533–7. 10.1136/bjo.2003.028480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. van Stokkum IH, Lambrou GN, van den Berg TJ. Hemodynamic parameter estimation from ocular fluorescein angiograms. Graefes Arch Clin Exp Ophthalmol 1995;233:123–30. 10.1007/BF00166603 [DOI] [PubMed] [Google Scholar]
- 8. Leitgeb RA, Werkmeister RM, Blatter C, et al. Doppler optical coherence tomography. Prog Retin Eye Res 2014;41:26–43. 10.1016/j.preteyeres.2014.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Wang J, Zhang M, Hwang TS, et al. Reflectance-based projection-resolved optical coherence tomography angiography [Invited]. Biomed Opt Express 2017;8:1536–48. 10.1364/BOE.8.001536 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Miura M, Makita S, Iwasaki T, et al. An approach to measure blood flow in single choroidal vessel using Doppler optical coherence tomography. Invest Ophthalmol Vis Sci 2012;53:7137–41. 10.1167/iovs.12-10666 [DOI] [PubMed] [Google Scholar]
- 11. Metelitsina TI, Grunwald JE, DuPont JC, et al. Foveolar choroidal circulation and choroidal neovascularization in age-related macular degeneration. Invest Ophthalmol Vis Sci 2008;49:358–63. 10.1167/iovs.07-0526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Boltz A, Luksch A, Wimpissinger B, et al. Choroidal blood flow and progression of age-related macular degeneration in the fellow eye in patients with unilateral choroidal neovascularization. Invest Ophthalmol Vis Sci 2010;51:4220–5. 10.1167/iovs.09-4968 [DOI] [PubMed] [Google Scholar]
- 13. Zhou Q, Daniel E, Grunwald JE, et al. Association between pseudodrusen and delayed patchy choroidal filling in the comparison of age-related macular degeneration treatments trials. Acta Ophthalmol 2017;95:e518–e520. 10.1111/aos.13398 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Choi W, Moult EM, Waheed NK, et al. Ultrahigh-speed, swept-source optical coherence tomography angiography in nonexudative age-related macular degeneration with geographic atrophy. Ophthalmology 2015;122:2532–44. 10.1016/j.ophtha.2015.08.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Spaide RF. Choriocapillaris flow features follow a power law distribution: implications for characterization and mechanisms of disease progression. Am J Ophthalmol 2016;170:58–67. 10.1016/j.ajo.2016.07.023 [DOI] [PubMed] [Google Scholar]
- 16. Moult EM, Waheed NK, Novais EA, et al. Swept-source optical coherence tomography angiography reveals choriocapillaris alterations in eyes with nascent geographic atrophy and drusen-associated geographic atrophy. Retina 2016;36(Suppl 1):S2–S11. 10.1097/IAE.0000000000001287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Nesper PL, Soetikno BT, Fawzi AA. Choriocapillaris nonperfusion is associated with poor visual acuity in eyes with reticular pseudodrusen. Am J Ophthalmol 2017;174:42–55. 10.1016/j.ajo.2016.10.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Sugiyama T, Araie M, Riva CE, et al. Use of laser speckle flowgraphy in ocular blood flow research. Acta Ophthalmol 2010;88:723–9. 10.1111/j.1755-3768.2009.01586.x [DOI] [PubMed] [Google Scholar]
- 19. Wang L, Cull GA, Piper C, et al. Anterior and posterior optic nerve head blood flow in nonhuman primate experimental glaucoma model measured by laser speckle imaging technique and microsphere method. Invest Ophthalmol Vis Sci 2012;53:8303–9. 10.1167/iovs.12-10911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Shiga Y, Kunikata H, Aizawa N, et al. Optic nerve head blood flow, as measured by laser speckle flowgraphy, is significantly reduced in preperimetric glaucoma. Curr Eye Res 2016;41:1447–53. 10.3109/02713683.2015.1127974 [DOI] [PubMed] [Google Scholar]
- 21. Shiga Y, Asano T, Kunikata H, et al. Relative flow volume, a novel blood flow index in the human retina derived from laser speckle flowgraphy. Invest Ophthalmol Vis Sci 2014;55:3899–904. 10.1167/iovs.14-14116 [DOI] [PubMed] [Google Scholar]
- 22. Luft N, Wozniak PA, Aschinger GC, et al. Ocular blood flow measurements in healthy white subjects using laser speckle flowgraphy. PLoS One 2016;11:e0168190 10.1371/journal.pone.0168190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Witkowska KJ, Bata AM, Calzetti G, et al. Optic nerve head and retinal blood flow regulation during isometric exercise as assessed with laser speckle flowgraphy. PLoS One 2017;12:e0184772 10.1371/journal.pone.0184772 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Fuchsjäger-Mayrl G, Luksch A, Malec M, et al. Role of endothelin-1 in choroidal blood flow regulation during isometric exercise in healthy humans. Invest Ophthalmol Vis Sci 2003;44:728–33. 10.1167/iovs.02-0372 [DOI] [PubMed] [Google Scholar]
- 25. Luksch A, Polska E, Imhof A, et al. Role of NO in choroidal blood flow regulation during isometric exercise in healthy humans. Invest Ophthalmol Vis Sci 2003;44:734–9. 10.1167/iovs.02-0177 [DOI] [PubMed] [Google Scholar]
- 26. Riva CE, Cranstoun SD, Grunwald JE, et al. Choroidal blood flow in the foveal region of the human ocular fundus. Invest Ophthalmol Vis Sci 1994;35:4273–81. [PubMed] [Google Scholar]
- 27. Geiser MH, Diermann U, Riva CE. Compact laser Doppler choroidal flowmeter. J Biomed Opt 1999;4:459–64. 10.1117/1.429960 [DOI] [PubMed] [Google Scholar]
- 28. Cicchetti DV. Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol Assess 1994;6:284–90. 10.1037/1040-3590.6.4.284 [DOI] [Google Scholar]
- 29. Luft N, Wozniak PA, Aschinger GC, et al. Measurements of retinal perfusion using laser speckle flowgraphy and doppler optical coherence tomography. Invest Ophthalmol Vis Sci 2016;57:5417–25. 10.1167/iovs.16-19896 [DOI] [PubMed] [Google Scholar]
- 30. Riva CE, Grunwald JE, Sinclair SH, et al. Blood velocity and volumetric flow rate in human retinal vessels. Invest Ophthalmol Vis Sci 1985;26:1124–32. [PubMed] [Google Scholar]
- 31. Doblhoff-Dier V, Schmetterer L, Vilser W, et al. Measurement of the total retinal blood flow using dual beam Fourier-domain Doppler optical coherence tomography with orthogonal detection planes. Biomed Opt Express 2014;5:630–42. 10.1364/BOE.5.000630 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Silver DM, Farrell RA. Validity of pulsatile ocular blood flow measurements. Surv Ophthalmol 1994;38:S72–S80. 10.1016/0039-6257(94)90049-3 [DOI] [PubMed] [Google Scholar]
- 33. Schmetterer L, Dallinger S, Findl O, et al. A comparison between laser interferometric measurement of fundus pulsation and pneumotonometric measurement of pulsatile ocular blood flow. 1. Baseline considerations. Eye 2000;14(Pt 1):39–45. 10.1038/eye.2000.9 [DOI] [PubMed] [Google Scholar]
- 34. Riva CE, Titze P, Hero M, et al. Choroidal blood flow during isometric exercises. Invest Ophthalmol Vis Sci 1997;38:2338–43. [PubMed] [Google Scholar]
- 35. Schmidl D, Boltz A, Kaya S, et al. Comparison of choroidal and optic nerve head blood flow regulation during changes in ocular perfusion pressure. Invest Ophthalmol Vis Sci 2012;53:4337–46. 10.1167/iovs.11-9055 [DOI] [PubMed] [Google Scholar]
- 36. Schmidl D, Schmetterer L, Witkowska KJ, et al. Factors associated with choroidal blood flow regulation in healthy young subjects. Invest Ophthalmol Vis Sci 2016;57:5705–13. 10.1167/iovs.16-20225 [DOI] [PubMed] [Google Scholar]
- 37. Kiel JW, Shepherd AP. Autoregulation of choroidal blood flow in the rabbit. Invest Ophthalmol Vis Sci 1992;33:2399–410. [PubMed] [Google Scholar]
- 38. Kiel JW. Choroidal myogenic autoregulation and intraocular pressure. Exp Eye Res 1994;58:529–43. 10.1006/exer.1994.1047 [DOI] [PubMed] [Google Scholar]
- 39. Kiel JW, van Heuven WA. Ocular perfusion pressure and choroidal blood flow in the rabbit. Invest Ophthalmol Vis Sci 1995;36:579–85. [PubMed] [Google Scholar]