Loss of an enhancer element in the 3′ untranslated region of SP5G conferred day-length insensitivity to domesticated tomato cultivars and helped the species spread worldwide.
Abstract
Tomato (Solanum lycopersicum) is a major vegetable fruit grown and consumed worldwide. Modern cultivated tomatoes are derived from their wild relative, Solanum pimpinellifolium, a short-day plant that originated from the Andean region of South America. The molecular underpinnings of the regional adaptation and expansion of domesticated tomato remain largely unclear. In this study, we examined flowering time in wild and cultivated tomatoes under both long-day and short-day conditions. Using quantitative trait locus mapping in a recombinant inbred line population, we identified SELF PRUNING 5G (SP5G) as a major locus influencing daylength adaptation in tomato. Genetic diversity analysis revealed that the genomic region harboring SP5G shows signatures of a domestication sweep. We found that a 52-bp sequence within the 3ʹ untranslated region of SP5G is essential for the enhanced expression of this gene, leading to delayed flowering time in tomatoes through a promoter-enhancer interaction that occurs only under long-day conditions. We further demonstrate that the absence of the 52-bp sequence attenuates the promoter-enhancer interaction and reduces SP5G expression in cultivated tomatoes, making their flowering time insensitive to daylength. Our findings demonstrate that cis-regulatory variation at the enhancer region of the SP5G 3ʹ untranslated region confers reduced photoperiodic response in cultivated tomatoes, uncovering a regulatory mechanism that could potentially be used to manipulate flowering time in tomato through novel biotechnological approaches.
Flowering is the transition from vegetative growth to reproductive growth and depends on internal signals and external cues (Bäurle and Dean, 2006; Andrés and Coupland, 2012). Plants evolved distinct photoperiodic responses to adapt to their local environments. For long-day (LD) plants, prolonged daytime induces flowering, while short-day (SD) plants flower earlier under SD conditions. The ability of plants to respond to photoperiod requires the detection of daylength. In the LD plant Arabidopsis (Arabidopsis thaliana), CONSTANS (CO) is a major regulator of photoperiodic flowering (Samach et al., 2000; Suárez-López et al., 2001; Imaizumi and Kay, 2006; Song et al., 2015). The florigen component FLOWERING LOCUS T (FT), a CENTRORADIALIS/TERMINAL FLOWER 1/SELF-PRUNING (CETS) family member, is the final output of the photoperiodic response downstream of CO (Samach et al., 2000; Wigge et al., 2005). CO is regulated at both the transcriptional (Fornara et al., 2009) and post-transcriptional (Valverde et al., 2004; Jang et al., 2008; Liu et al., 2008) levels during the light period, allowing increased accumulation of CO protein during the daytime to induce FT, which, in turn, promotes flowering (Samach et al., 2000). In the SD plant rice (Oryza sativa), the CO homolog HEADING DATE 1 (HD1) represses the FT homolog HD3A under LD conditions but activates it under SD (Izawa et al., 2002; Hayama et al., 2003), which ultimately results in flowering. In addition, a rice-specific regulatory pathway, GRAIN NUMBER, PLANT HEIGHT, AND HEADING DATE 7 (GHD7)-EARLY HEADING DATE1 (EHD1)-HD3A/RICE FLOWERING LOCUS T1, also regulates daylength responses in rice (Song et al., 2015).
Over the course of domestication, the photoperiodic sensitivity of many crops has been modified to suit broad environmental conditions and different latitudes. The variances of several photoperiodic pathway genes, including GHD7, HD1, HD3A, and EHD1 in rice (Fujino and Sekiguchi, 2005; Xue et al., 2008; Takahashi et al., 2009; Fujino et al., 2013; Matsubara et al., 2014) and ZmCCT (a CCT domain-containing protein) in maize (Zea mays; Yang et al., 2013), resulted in the earlier flowering of these SD crops under LD conditions. As another example, the reduced photoperiodic sensitivity in wheat (Triticum aestivum) and barley (Hordeum vulgare) caused by mutation of the PHOTOPERIOD1 gene contributed greatly to the Green Revolution (Turner et al., 2005; Beales et al., 2007; Wilhelm et al., 2009). Thus, decreased daylength sensitivity plays a vital role in crop adaptation and spread.
Cultivated tomato was domesticated from the wild species Solanum pimpinellifolium, which originated in the Andean region of South America near the equator (Jenkins, 1948). Tomato was taken to Europe in the 16th century and then spread worldwide. The wild ancestors of tomato are considered SD plants, while most modern cultivars are considered day neutral plants due to the loss of or weakened photoperiodic sensitivity, rendering them adaptive to northern latitudes. In tomato, homologs of the Arabidopsis FT gene, such as SINGLE FLOWER TRUSS (SFT)(Lifschitz et al., 2006) and SELF PRUNING (SP; Pnueli et al., 1998), play important roles in controlling flowering time. However, none of them were proven to affect daylength sensitivity, suggesting that the photoperiod adaptation genes in tomato are different from those in Arabidopsis and rice. Recent studies showed that the SP5G gene, another CETS family member in tomato, acts as a flowering repressor in daylength responses (Cao et al., 2016; Soyk et al., 2017). Higher SP5G expression in Solanum galapagense causes later flowering under LD, whereas the weakened daytime induction of its transcripts in cultivated tomato results in early flowering under LD (Soyk et al., 2017). However, the detailed molecular mechanism that regulates SP5G expression in response to daylength in wild and cultivated tomatoes remains unknown.
In this study, we identified and characterized a major flowering quantitative trait locus (QTL), Q-fo1_01, from a tomato recombinant inbred line (RIL) population. Our analysis revealed that SP5G is the gene underlying this QTL for adaptation to daylength and that the genomic region around SP5G was selected during domestication. We found that the 3′ untranslated region (UTR) of SP5G acts as an enhancer to activate its expression. Moreover, a 52-bp sequence within the SP5G 3ʹ UTR is essential for the formation of an enhancer-promoter loop that regulates SP5G expression in tomato. The absence of the 52-bp sequence in many big-fruit cultivars corresponds to reduced SP5G expression, resulting in daylength insensitivity in these cultivars. Our results demonstrate that the cis-regulatory variation at the enhancer region of the SP5G 3ʹ UTR confers reduced photoperiodic response in cultivated tomatoes.
RESULTS
The Flowering Time of Cultivated Tomato Is Less Sensitive to Daylength
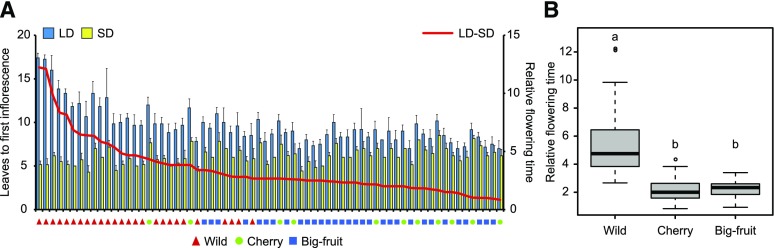
Unlike the tomato wild relative S. pimpinellifolium, which is an SD plant, many tomato cultivars are day neutral (Jenkins, 1948; Soyk et al., 2017). To better understand the daylength response of the wild progenitor and cultivated tomatoes, we measured flowering time by counting the number of leaves before the first inflorescence under LD and SD conditions in 31 big-fruit cultivars, 26 wild accessions (S. pimpinellifolium), and 11 cherry tomatoes, which are considered intermediate between the wild ancestor and cultivars (Blanca et al., 2012). Under SD conditions, most of the wild species flowered earlier than the big-fruit cultivars (Fig. 1A). The flowering time of many big-fruit tomatoes was earlier than that of wild species under LD conditions, although nearly all accessions exhibited delayed flowering as daylength increased (Fig. 1A). We defined the relative flowering time as the difference in leaf number between LD and SD. The average relative flowering time of wild species was much greater than that of the big-fruit cultivars (Fig. 1B), indicating that the wild species are more sensitive to daylength than the cultivated tomatoes.
Figure 1.
Cultivated tomatoes are less sensitive to daylength than wild species. A, Flowering time of 26 wild tomatoes, 11 cherry tomatoes, and 31 big-fruit cultivars under LD (blue bars) and SD (yellow bars) conditions. The red line denotes the relative flowering time, which was defined as the difference in leaf number before the first inflorescence between LD and SD conditions. The red triangles, green circles, and blue blocks indicate the wild species, cherry tomatoes, and big-fruit cultivars, respectively. The bars and lines indicate means and sd of six individual plants. B, Average relative flowering times of wild, cherry, and big-fruit tomatoes used in A. Different letters above the columns indicate statistically significant differences (Tukey’s honestly significant difference test, P < 0.05).
SP5G Is a Major Locus Contributing to Daylength Adaptation in Tomato
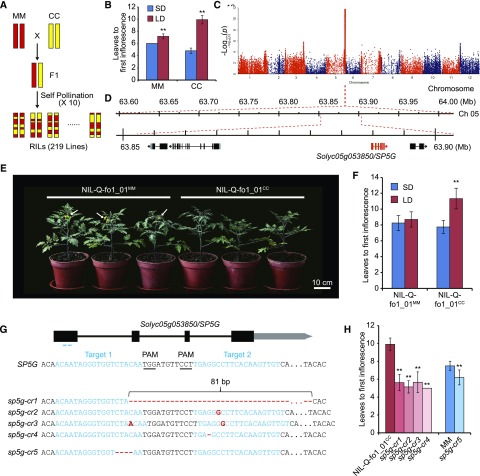
To explore the genetic basis of the difference in photoperiodic sensitivity between cultivated tomatoes and their wild relatives, we generated a stable RIL population of 219 lines after 10 selfing generations by crossing S. lycopersicum var cerasiforme (LA1310; CC) with S. lycopersicum cv Moneymaker (MM; Fig. 2A), which are sensitive and insensitive to daylength, respectively (Fig. 2B). To identify the QTLs associated with flowering time, we used whole-genome sequencing to detect single-nucleotide polymorphisms (SNPs) in the two parents and all RIL lines. On average, each independent line was sequenced with approximately 1.7-fold coverage, and about 970,729 SNPs were identified. Under LD conditions, there was one peak identified above the P value cutoff of 10−10 that was not present in the SD results (Fig. 2C; Supplemental Fig. S1). This peak region was located on the long arm of chromosome 5, spanning 420 kb and containing 51 genes (Fig. 2D).
Figure 2.
SP5G is responsible for photoperiod sensitivity in tomatoes. A, Schematic representation summarizing the construction of the RILs. MM indicates the big-fruit cv S. lycopersicum cv Moneymaker, and CC indicates the cherry tomato S. lycopersicum var cerasiforme (LA1310). The red and yellow bars indicate the chromosome regions with the MM and CC genotypes, respectively. B, Flowering time of the parental lines of the RILs under LD and SD conditions. Bars and lines indicate the means and sd of six individual plants. **, P < 0.01 (Student’s t test). C, Manhattan plots for flowering time under LD conditions. The black dashed line indicates the cutoff P value (1E-10). The red arrow indicates the main locus Q-fo1_01. D, Peak region of the Q-fo1_01 locus using the P value of 1E-10 (top) and the genomic region of difference between NIL-Q-fo1_01MM and NIL-Q-fo1_01CC (bottom). E, Appearance of the NILs of Q-fo1_01 under LD conditions. The white arrows indicate flowers. Bar = 10 cm. F, Flowering time of the NILs under LD and SD conditions. Bars and lines indicate means and sd of six individual plants. **, P < 0.01 (Student’s t test). G, SP5G mutations generated by CRISPR/Cas9 using two single-guide RNAs. Blue lines indicate the target sites of the guide RNAs. The nucleotides underlined in black indicate the protospacer-adjacent motif (PAM). The sequences of the four sp5g mutant alleles (sp5g-cr1/2/3/4) of NIL-Q-fo1_01CC and one allele (sp5g-cr5) of MM are shown. H, Flowering time of the sp5g mutant alleles compared with their controls under LD conditions. Bars and lines indicate means and sd of more than six individual plants. **, P < 0.01 (Student’s t test).
To verify that this locus contributes to the daylength sensitivity of CC, we generated near-isogenic lines (NILs; Fig. 2E) in which the Q-fo1_01 locus of the MM genotype was introgressed into CC by backcrossing six times. A total of 88 markers along 12 chromosomes were used to evaluate the background of the NILs, and both NIL-fo1_01CC and NIL-fo1_01MM had the CC genotype for nearly 95% (83 of 88) of the tested markers (Supplemental Table S1). Between NIL-fo1_01CC and NIL-fo1_01MM, the region of difference around the Q-fo1_01 locus was only about 50 kb (Fig. 2D; Supplemental Fig. S2). When evaluating the flowering time of the NILs under LD and SD conditions, we found that NILs with different genotypes at Q-fo1_01 had a similar flowering time under SD. Under LD conditions, however, NIL-fo1_01CC plants flowered much later than NIL-fo1_01MM (Fig. 2F).
One of the four predicted genes in this 50-kb region was Solyc05g053850 (Fig. 2D), which encodes the FT homolog SP5G and was reported previously to regulate the daylength response in tomato (Soyk et al., 2017). To directly test whether this gene was responsible for the flowering phenotypes observed in NIL-fo1_01CC and NIL-fo1_01MM, we mutated SP5G in NIL-fo1_01CC and MM by CRISPR/Cas9-mediated genome editing. The four null-allele sp5g mutants obtained in NIL-fo1_01CC, named sp5g-cr1/2/3/4, had a 1-bp insertion, a 2-bp insertion, a 1-bp deletion, and an 81-bp deletion, respectively; the mutant in the MM background had a 4-bp deletion (Fig. 2G). The sp5g-cr mutants in the NIL-fo1_01CC and MM backgrounds exhibited earlier flowering under LD conditions than their respective controls (Fig. 2H). These results confirmed that SP5G is the gene in the Q-fo1_01 locus that regulates daylength sensitivity in tomato.
SP5G Was Artificially Selected during Tomato Domestication
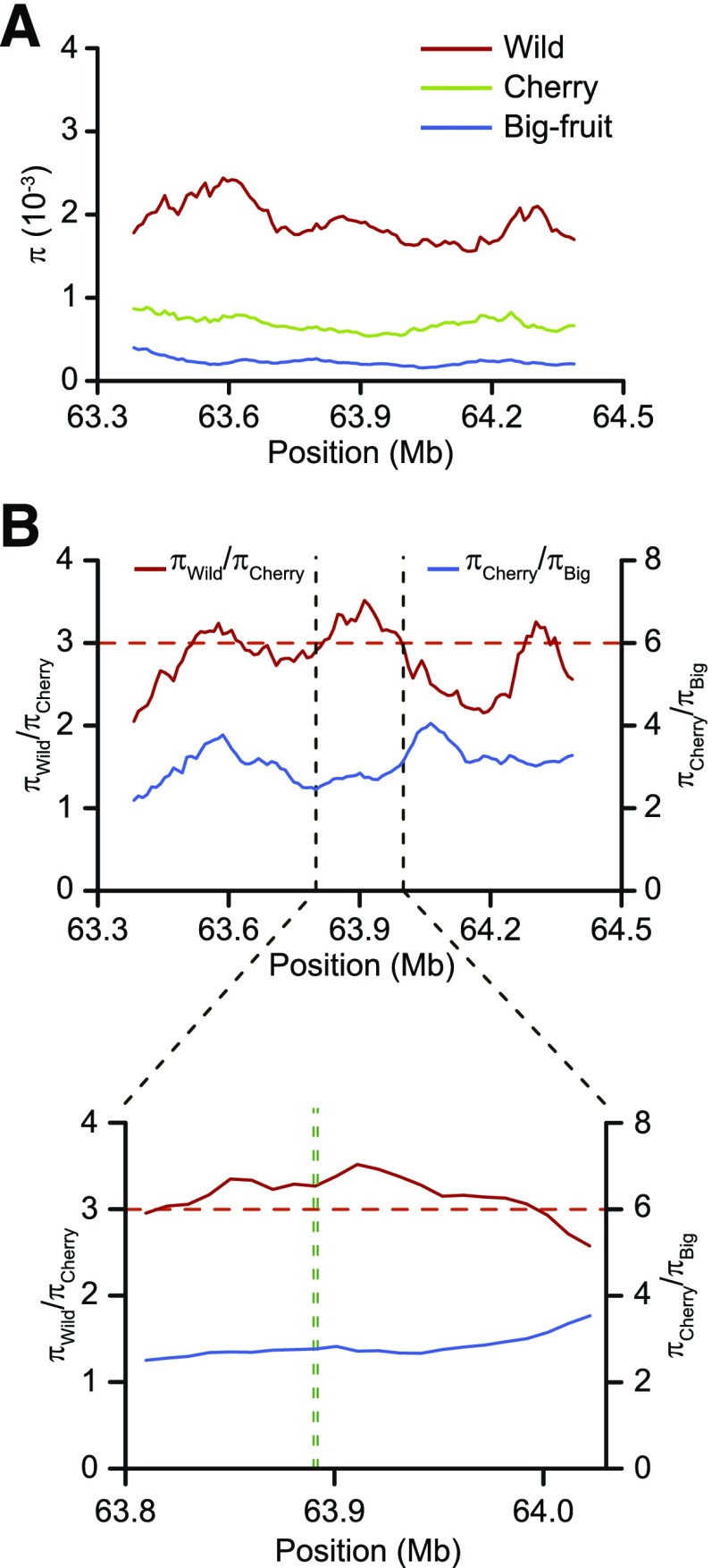
Modern cultivated tomatoes became day-neutral plants for adaptation to broad or diverse regions of cultivation. Consistently, many of the S. pimpinellifolium species were found to be more sensitive to LD in terms of flowering time compared with the big-fruit cultivars. To assess whether the altered daylength response conferred an adaptive advantage and was selected during tomato breeding, we used the published genomic sequences of 166 big-fruit S. lycopersicum varieties, 112 cherry tomatoes, and 53 S. pimpinellifolium accessions (Lin et al., 2014) to scan for signatures of selection surrounding the SP5G gene. Notably, we found that the region around SP5G had reduced nucleotide diversity (π) in cherry and big-fruit tomatoes compared with wild species (Fig. 3A). The ratio of nucleotide diversity for wild to cherry tomatoes (πwild/πcherry) in this region was above 3, which was defined previously as the cutoff for a domestication sweep (Lin et al., 2014). However, the ratio for cherry to big-fruit tomatoes was below 6.9, the cutoff for designating an improvement sweep (Lin et al., 2014). These findings suggest that SP5G was positively selected during domestication but not in tomato improvement (Fig. 3B).
Figure 3.
The genomic region around SP5G shows evidence of a domestication sweep. A, Nucleotide diversity (π) of wild species (red line), cherry tomatoes (green line), and big-fruit cultivars (blue line). B, π ratio for the wild species to the cherry tomatoes (red lines) and the cherry tomatoes to the big-fruit cultivars (blue lines) for a region on chromosome 5. The horizontal dashed red lines indicate the genome-wide top 5% ratio cutoff for candidate domestication sweeps. The vertical green dashed lines (bottom) delimit SP5G.
SP5G Expression Differs in the NILs
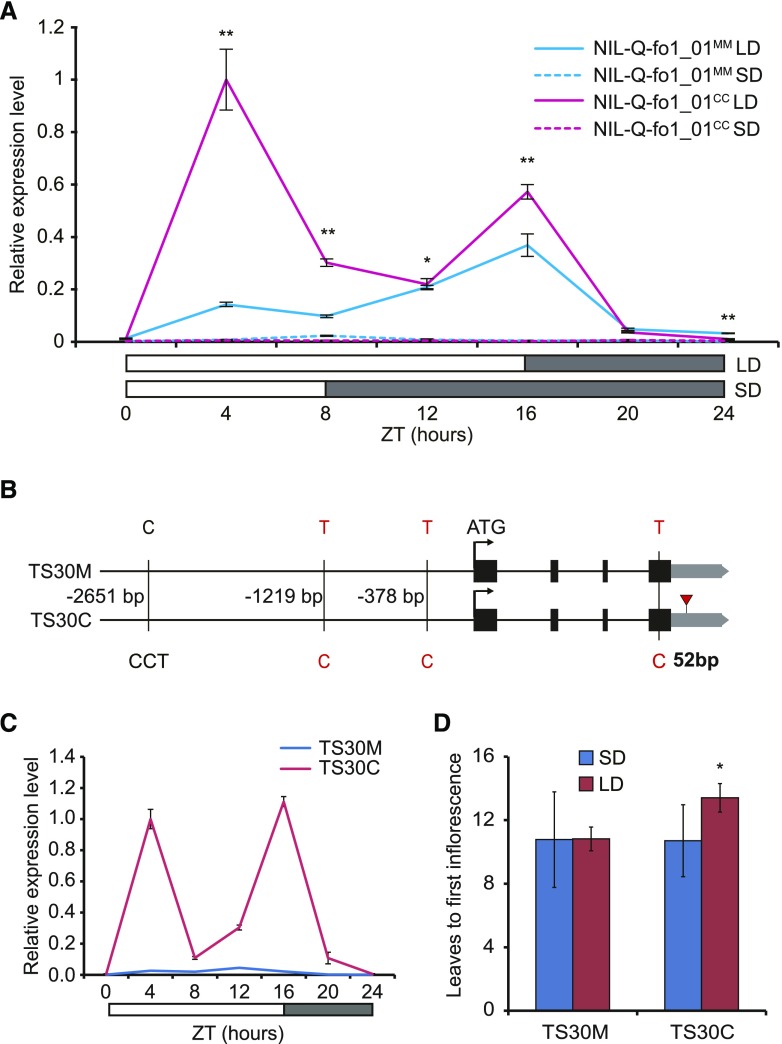
To further investigate the role of SP5G in daylength responses, its diurnal expression pattern was analyzed in the NILs exposed to different daylengths. Under LD conditions, SP5G was highly expressed at 4 h after dawn (Zeitgeber time 4 [ZT4]) in NIL-fo1_01CC and then decreased; at the end of the day, SP5G transcripts increased again and then dropped rapidly after dusk, as reported previously (Cao et al., 2016; Soyk et al., 2017; Fig. 4A). Notably, the expression peak of SP5G at ZT4 was reduced dramatically in NIL-fo1_01MM, resulting in a relatively low and stable expression from ZT0 to ZT8 in NIL-fo1_01MM (Fig. 4A). Under SD conditions, SP5G showed extremely low expression in both NIL-fo1_01CC and NIL-fo1_01MM (Fig. 4A). Given that higher SP5G expression levels result in more delayed flowering time under LD conditions in tomato (Soyk et al., 2017), our findings suggest that the differences in SP5G expression under LD conditions in these two NILs may underlie their different photoperiodic responses.
Figure 4.
SP5G expression is associated with polymorphisms between different alleles. A, Relative expression levels of SP5G over 24 h under LD (solid lines) or SD (dotted lines) conditions in 18-d-old seedlings of NIL-Q-fo1_01MM (cyan lines) and NIL-Q-fo1_01CC (magenta lines). The expression level of SP5G in NIL-Q-fo1_01CC at ZT4 under LD conditions was normalized as 1. Asterisks indicate statistically significant differences between NIL-Q-fo1_01MM and NIL-Q-fo1_01CC under LD: *, P < 0.05 and **, P < 0.01 (Student’s t test). B, Polymorphisms within the 3-kb promoter, coding sequence, and 3ʹ UTR of SP5G between TS30M and TS30C. The red triangle indicates the 52-bp sequence within the 3ʹ UTR. The polymorphisms in red indicate those that differed between the tomato groups exhibiting hypersensitivity and hyposensitivity to daylength. C, Relative expression levels of SP5G in 18-d-old seedlings of TS30M (cyan lines) and TS30C (magenta lines) under LD conditions. The expression level of SP5G in TS30C at ZT4 was normalized as 1. D, Flowering time of TS30M and TS30C under LD and SD conditions. Bars and lines indicate means and sd of more than six individual plants, and the asterisk indicates a statistically significant difference (P < 0.05, Student’s t test).
To identify the sequence variations responsible for the altered SP5G expression, we compared the SP5G genomic sequences of MM and CC along with 3 kb of the upstream promoter region. Some polymorphisms in the promoter region, three mismatches in the coding region, and a 52-bp sequence in the 3ʹ UTR that was absent in MM were identified. Among these, two SNPs in the coding region caused amino acid substitutions in the predicted SP5G protein (Supplemental Fig. S3). We further investigated the nucleotide variations among 40 tomato accessions that were either hypersensitive or hyposensitive to daylength, as described in Figure 1. In contrast to the hypersensitive group, the hyposensitive group had the MM genotype at the three coding region SNPs and lacked the 52-bp sequence in the 3ʹ UTR (Supplemental Table S2). Only three of the promoter region SNPs were identical in all of the hypersensitive tomatoes and different from the hyposensitive tomato accessions (Supplemental Table S2).
We then analyzed the SP5G sequences in all 331 accessions and identified a line, TS30, that was heterozygous in the SP5G region. We amplified and sequenced a 3-kb promoter region, coding sequence, and 3ʹ UTR of SP5G for different TS30 individuals and obtained two TS30 genotypes. TS30M had the same sequence as MM, and TS30C contained a 2-bp InDel and two SNPs in the promoter region, one nonsynonymous coding region SNP, and the 52-bp InDel in the 3ʹ UTR absent in TS30M (Fig. 4B). Except for the 2-bp InDel in the promoter, the polymorphisms of the TS30C haplotype also were identified and identical in most of the hypersensitive tomatoes. Analysis of SP5G transcript levels showed that they were much more abundant in TS30C than in TS30M under LD (Fig. 4C). Consistent with the SP5G expression levels, TS30C flowered later than TS30M under LD, but no difference was observed between them under SD (Fig. 4D). Beyond the amino acid substitution identified in the SP5G coding region, the variations in the promoter region and the 52-bp InDel in the 3ʹ UTR may contribute primarily to the regulation of SP5G expression in tomato.
The SP5G 3ʹ UTR Is an Enhancer That Regulates SP5G Expression
To investigate how these polymorphisms affect SP5G expression, we first tested the stability of the transcripts from NIL-fo1_01MM and NIL-fo1_01CC. We treated seedlings of the NILs with cordycepin C, a transcriptional inhibitor that blocks mRNA synthesis (Barrett et al., 2012; Matoulkova et al., 2012), and quantified SP5G transcripts at different time points after treatment. The degradation rates of SP5G mRNA were similar between NIL-fo1_01MM and NIL-fo1_01CC (Supplemental Fig. S4), indicating that these variants do not influence the stability of SP5G mRNA.
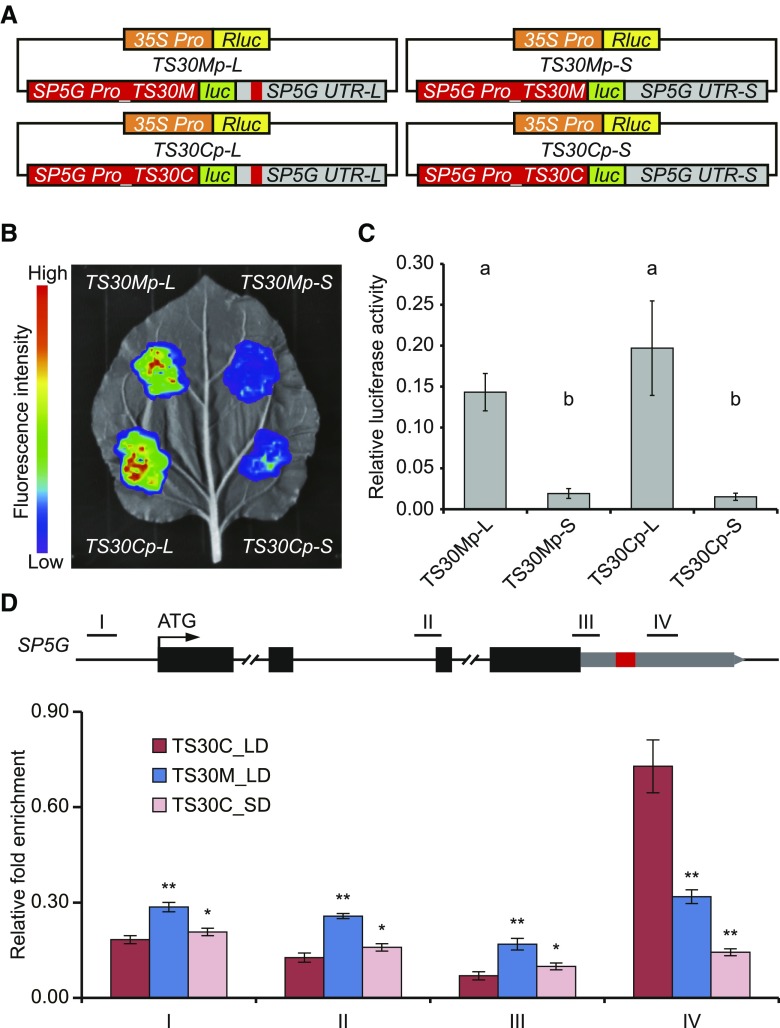
Next, we investigated whether the promoter SNPs or the 52-bp InDel affect SP5G transcription using a dual luciferase reporter assay. The SP5G 3ʹ UTR with (SP5G UTR-L) or without (SP5G UTR-S) the 52-bp sequence was fused with a firefly luciferase sequence driven by the native SP5G promoter from TS30M or TS30C. Renilla luciferase driven by the cauliflower mosaic virus 35S promoter within the same construct was used as an internal control (Fig. 5A). The constructs were expressed transiently in tobacco (Nicotiana tabacum) leaves, and luciferase activity was detected to calculate the ratio of firefly luciferase to Renilla luciferase activity as an indicator of the transcriptional efficiency of SP5G. Regardless of the promoter used (TS30M or TS30C), the ratio of firefly luciferase to Renilla luciferase activity was much higher for constructs containing the full-length 3ʹ UTR compared with constructs lacking the 52-bp sequence (Fig. 5, B and C). This finding indicated that the loss of the 52-bp sequence in the SP5G 3ʹ UTR greatly reduced its expression. The relative signal of firefly luciferase driven by the TS30C promoter was slightly higher than that driven by the TS30M promoter, but the difference was not significant when using the full-length 3ʹ UTR, suggesting that the polymorphisms in the SP5G promoter have little effect on its expression (Fig. 5C). The analysis indicated that the 3ʹ UTR is an enhancer that regulates SP5G transcripts and that the 52-bp sequence is essential for its enhancer activity. Notably, the SP5G 3ʹ UTR did not affect the activity of the 35S minimal promoter, as there was no significant difference in relative firefly luciferase activity between constructs with the full-length or truncated 3ʹ UTR driven by the 35S minimal promoter (Supplemental Fig. S5). Thus, the enhancer activity of the 3ʹ UTR appears to be specific to the SP5G native promoter.
Figure 5.
The 3ʹ UTR of SP5G with the 52-bp sequence acts as an enhancer under LD conditions. A, Schematic representation of the constructs used for the dual luciferase reporter assay. The orange and red bars indicate the 35S and SP5G promoters, respectively. The green and yellow bars indicate the coding sequences of firefly luciferase (luc) and Renilla luciferase (Rluc), respectively. The gray bar indicates the 3ʹ UTR of SP5G, and the red block within it indicates the 52-bp sequence present in the CC genotype but absent in the MM genotype. B, Photograph showing the fluorescence intensity catalyzed by firefly luciferase driven by the different combinations of two types of promoters and UTRs on the same tobacco leaf. C, Relative luciferase activity (the ratio of luc to Rluc) of the four constructs. Different letters above the bars indicate statistically significant differences (Tukey’s honestly significant difference test, P < 0.05). D, Enrichment of H3K27ac at different regions of SP5G in TS30C and TS30M. Bars and lines indicate means and se of four replicates. Asterisks indicate statistically significant differences compared with TS30C under LD: *, P < 0.05 and **, P < 0.01 (Student’s t test).
In plants, active enhancers are associated with specific histone modifications, such as H3K27 acetylation (Zhu et al., 2015; Oka et al., 2017). We examined the distribution of H3K27ac around the SP5G region by chromatin immunoprecipitation (ChIP) in TS30M and TS30C lines at ZT4 under LD conditions. Low levels of H3K27ac were observed at the transcription start site (TSS) and gene body of SP5G in TS30M and TS30C under LD conditions (Fig. 5D). However, there was a significant enrichment of H3K27ac at the SP5G 3ʹ UTR in TS30C compared with TS30M (Fig. 5D), supporting the existence of a functional enhancer at this region in TS30C. Moreover, H3K27ac abundance at the SP5G 3ʹ UTR in TS30C was reduced dramatically under SD conditions (Fig. 5D), indicating that the enhancer activity was dependent on daylength.
We then grouped the tomato accessions by the presence (noted as C) or absence (noted as M) of the 52-bp sequence within the SP5G 3ʹ UTR (Supplemental Fig. S6A). Association analysis revealed that the relative flowering time of these accessions was highly correlated with their SP5G genotype rather than their species (Supplemental Fig. S6, B and C). For example, TS433 is a wild species with the MM SP5G genotype, and this line was more insensitive to daylength than other wild lines (Supplemental Fig. S6B).
The 52-bp Sequence in the SP5G 3ʹ UTR Is Essential for Promoter-Enhancer Interaction
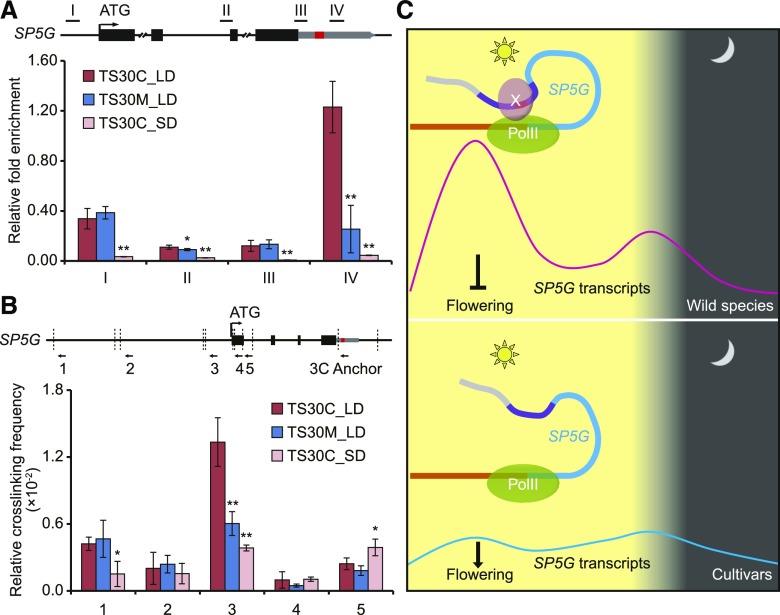
In eukaryotes, mRNA is transcribed by RNA polymerase II (PolII), which can be recruited to an enhancer region to facilitate promoter-enhancer loop formation and subsequently enhance target gene transcription (Chen et al., 2017). To detect whether PolII is recruited to the SP5G 3ʹ UTR, we performed ChIP using antibodies against PolII in TS30M and TS30C lines at ZT4 under LD conditions. Our results showed that PolII accumulation in the TSS region of SP5G was similar between the two lines (Fig. 6A). However, PolII accumulation at the SP5G 3ʹ UTR around the 52-bp region was significantly greater in TS30C than in TS30M under LD conditions (Fig. 6A), indicating that the 52-bp sequence is vital for PolII enrichment at the SP5G 3ʹ UTR. Under SD conditions, PolII accumulation was low (Fig. 6A), consistent with the finding that SP5G was hardly transcribed under SD (Fig. 4A).
Figure 6.
The 3ʹ UTR of SP5G forms a loop structure with its promoter. A, Enrichment of PolII at different regions of SP5G in TS30C and TS30M. Bars and lines indicate means and se of four replicates. *, P < 0.05 and **, P < 0.01 (Student’s t test). B, 3C analysis in TS30C and TS30M. The vertical dashed lines indicate the DpnII sites. The arrows indicate the positions and directions of primers used for 3C analysis. The black bars and gray bars on the schematic gene structure indicate coding exons and the 3ʹ UTR, respectively. The red block indicates the 52-bp sequence, which is present in TS30C but absent in TS30M. Bars and lines indicate means and se of four replicates. *, P < 0.05 and **, P < 0.01 (Student’s t test). C, Proposed model for the regulation of SP5G under LD conditions between wild and cultivated tomatoes. The thick curves indicate the genomic regions of SP5G (brown for the promoter, blue for the coding region, purple for the 3ʹ UTR, and red for the 52-bp sequence). The thin curves indicate the SP5G expression levels of wild (magenta) and cultivated (cyan) tomatoes within 1 d under LD. X indicates the possible transcription factor (TF) regulating SP5G expression.
Numerous studies have shown that an enhancer can form a loop structure with the promoter of its target gene and activate its transcription (Levine et al., 2014; Mora et al., 2016; Weber et al., 2016). To check whether a loop structure is formed between the SP5G 3ʹ UTR and promoter, we performed a chromosome conformation capture (3C) assay, a technique that detects distal DNA interactions (Dekker et al., 2002), in TS30M and TS30C at ZT4 under LD conditions. With the sequence containing the 52-bp InDel as the anchor point, we tested the cross-linking interactions between this region and various DpnII-released DNA fragments. A peak was detected between the anchor point and fragment 3 near the start codon (Fig. 6B), suggesting a physical interaction between the SP5G 3ʹ UTR and the promoter. The strength of the interaction was weakened significantly in TS30M compared with TS30C (Fig. 6B), indicating that the 52-bp sequence in the SP5G 3ʹ UTR is crucial for the promoter-enhancer interaction. Moreover, the interaction between the SP5G 3ʹ UTR and promoter was reduced dramatically under SD conditions, indicating that the regulatory role of the SP5G enhancer is dependent on daylength (Fig. 6B). Based on these observations, we propose the following model of enhancer-mediated photoperiodic sensitivity in tomato. In wild tomatoes, the SP5G 3ʹ UTR contains a functional enhancer that forms a loop with the SP5G promoter and activates the expression of the gene under LD conditions. In tomato cultivars, the enhancer is disrupted by the loss of the 52-bp sequence, resulting in weakened SP5G expression under LD conditions and consequent photoperiodic insensitivity (Fig. 6C).
DISCUSSION
In contrast to SD wild tomato species, modern cultivars have reduced photoperiodic sensitivity and are day neutral. Consistent with a recent report that altered SP5G expression contributes to the different daylength responses in tomato (Soyk et al., 2017), our QTL analysis identified SP5G as a major locus regulating daylength sensitivity in tomato that has been subject to selection during tomato domestication. We determined that a 52-bp sequence within the SP5G 3ʹ UTR is essential for the enhanced expression of SP5G under LD conditions. Moreover, the SP5G 3ʹ UTR is an enhancer capable of forming a loop structure with the SP5G promoter, as reported for other enhancers (Sakabe et al., 2012; Weber et al., 2016). The 52-bp deletion within the 3ʹ UTR in many big-fruit cultivars weakened the enhancer-promoter physical interaction, thereby decreasing SP5G expression and causing daylength insensitivity. However, PolII accumulation at the promoter and gene body of SP5G was similar in TS30C and TS30M under LD conditions, which did not reflect the different SP5G expression levels observed between these two lines. Gene expression is the output of the whole transcription cycle, including transcription initiation, elongation, and termination (Hajheidari et al., 2013). PolII carries out transcription throughout the genome and undergoes regulated pauses and releases at enhancers to resist or promote termination, effectively a mechanism for gene transcription regulation (Porrua and Libri, 2015; Henriques et al., 2018). Thus, the loss of the 52-bp sequence and the consequent reduction of PolII occupancy at the SP5G 3ʹ UTR may have led to improper transcription termination of SP5G and reduced its expression. In eukaryotes, posttranslational modifications of the C-terminal domain, the largest PolII subunit, are crucial for the regulation of multiple steps in transcription (Harlen and Churchman, 2017). In addition to the occupancy of PolII, detecting differences in the phosphorylation status of the C-terminal domain along the gene may help us better understand the transcription dynamics of SP5G.
Transcription factors (TFs) can be recruited to enhancers to facilitate promoter-enhancer interactions or mediate the interaction between PolII and DNA (Kim et al., 2010; Levine et al., 2014). Loss of the 52-bp sequence at the SP5G 3ʹ UTR enhancer may have led to the reduced occupancy of regulatory TFs potentially important for promoter-enhancer loop formation or acting with PolII to activate SP5G expression. Our observation that the decreased SP5G expression level in TS30M was much more drastic than the decreased promoter-enhancer interaction suggests that the promoter-enhancer interaction is not linearly correlated with SP5G expression. One reason may be that more TFs are required for the transcription of SP5G rather than as mediators of the promoter-enhancer loop. In fact, motifs recognized by distinct TFs were found around the 52-bp sequence, some of which participate in flowering regulation in Arabidopsis (Supplemental Table S3). Thus, the future identification of regulatory proteins that bind to the SP5G 3ʹ UTR will improve our understanding of the photoperiodic regulation of flowering in tomato.
It should be noted that, although SP5G expression was lower due to the absence of the 52-bp sequence in the 3ʹ UTR enhancer, SP5G was still induced in cultivated tomatoes under LD, which conferred a weak photoperiodic sensitivity. In agreement with the SP5G transcript levels, H3K27ac and PolII enrichment also were observed in TS30M under LD conditions. Additionally, the enhancer-promoter loop was weakened but not abolished in TS30M. These results suggest that the absence of the 52-bp sequence did not fully disrupt the enhancer activity. Consistent with this hypothesis, the SP5G knockout mutants generated by CRISPR/Cas9 in the MM background flowered earlier than MM under LD, indicating that SP5G still functions in cultivated tomatoes. We also observed that TS30H plants (heterozygous at SP5G) displayed intermediate daylength sensitivity between TS30M and TS30C under LD (Supplemental Fig. S7). These results suggest that SP5G regulates flowering time in a dose-dependent manner and that the cis-regulatory variants of SP5G cause subtle phenotypic changes by modifying the expression of SP5G. In fact, transcriptional changes causing gene dosage effects are often favored by breeders (Meyer and Purugganan, 2013). In maize, tb1 encodes a transcriptional regulator that represses branch elongation (Doebley et al., 1997). Its expression level in the wild teosinte (Zea spp.) allele is approximately half the level of the maize allele, and the difference is attributable to an upstream transposon insertion that acts as an enhancer (Clark et al., 2006; Studer et al., 2011; Zhou et al., 2011). The fw2.2 locus in tomato contributes up to 30% of the fruit weight increase observed in big-fruit cultivars versus wild tomatoes (Frary et al., 2000), and the increase may have been caused by nucleotide changes in the upstream promoter region leading to an altered expression profile (Nesbitt and Tanksley, 2002). Thus, cis-regulatory alleles provide beneficial variation for breeding in many crops. In a recent report, genome editing by CRISPR/Cas9 was used to modify the regulatory sequences of genes to obtain novel alleles affecting some important traits (Rodriguez-Leal et al., 2017). Finding novel regulatory elements of genes controlling important agronomic traits will facilitate the development of favorable alleles for breeding. In this context, our work characterizes a new target that could potentially be used to further modify tomato flowering time by manipulating the expression of SP5G through cis-regulatory variation.
MATERIALS AND METHODS
Plant Materials and Growth Conditions
RILs were derived by single-seed descent from a cross between the cultivated tomato (Solanum lycopersicum ‘Moneymaker’) and the cherry tomato accession Solanum lycopersicum var cerasiforme (LA1310). All plants were self-fertilized to the F10 generation, and a RIL population containing 219 lines was derived. For flowering time assessment, all tomato plants were grown in soil in a greenhouse. The flowering time was evaluated as the leaf number to the first inflorescence under LD conditions (Beijing, 40°N, from July to August, 14 h of light on average) and SD conditions (Beijing, 40°N, from February to March, 11.5 h of light on average). At least six individual plants of each accession were used for flowering time assessment.
Genome Sequencing, SNP Calling, and the RIL Association Study
The genomic DNA of MM, CC, and the RIL population was extracted following the standard CTAB method and prepared for sequencing on the Illumina HiSeq 2500 system with a 125-bp paired-end strategy. The sequencing depth was about 15-fold coverage for the two parental lines and about 1.7-fold coverage for the 219 RILs. The paired-end reads of MM, CC, and the RILs were mapped to the tomato reference genome (SL2.50 build; Tomato Genome Consortium, 2012) using Burrows-Wheeler Aligner version 0.7.10-r789 with default parameters (Li and Durbin, 2009). SNP calling was performed on the alignment results using the Genome Analysis Toolkit (GATK) version 3.1.1 and Picard package version 1.119 (McKenna et al., 2010) with the following steps: (1) deleting the unmapped reads, (2) deleting the duplicate reads, (3) conducting the alignment using the IndelRealigner package in GATK, and (4) SNP calling for each sample using the UnifiedGenotyper package in GATK with a minimum base quality score of 20. To ensure the quality of SNP calling in MM and CC, SNPs were filtered further with the VariantFiltration package in GATK using parameters QD < 2.0 || FS > 60.0 || MQ < 40.0 || MQRankSum < −12.5 || ReadPosRankSum < −8.0, and SNPs with DP < 10 also were removed. The remaining SNPs between MM and CC were used as the index for SNP calling in the 219 RILs. The SNPs in the 219 RILs were filtered with the Lowqual tag marked by the GATK UnifiedGenotyper package. To infer the missing genotype at an SNP site of an individual line, 20 SNPs flanking the target SNP in other lines of the population were compared with the individual line. If all of the lines having the same genotype for the flanking SNPs as the individual line had the same genotype at the target SNP, this SNP genotype was inferred/imputed to the individual line. The RIL association study was conducted using the imputed information for the SNPs of the 219 RILs by the compressed mixed linear model implemented in GAPIT (Lipka et al., 2012). The cutoff P value was set as 1E-10.
Identification of Selective Sweeps
To identify the selection region around SP5G, the SNPs near the SP5G locus in the tomato genome were obtained (corresponding to ch05: 63.3–64.5 Mb, SL2.50). We measured the level of nucleotide diversity (π) using a 100-kb window with a step size of 10 kb in wild species (wild), cherry tomatoes (cherry), and big-fruit cultivars (big). The ratios of nucleotide diversity between wild and cherry (πwild/πcherry) and cherry and big (πcherry/πbig) also were calculated. According to the strategy described in a previous study (Lin et al., 2014), the top 5% of ratios were used as the cutoff for sweeps.
RNA Stability Assay
More than five 18-d-old tomato seedlings under LD conditions were collected at ZT4 in each 50-mL tube and incubated with 16 mL of incubation buffer (1 mm PIPES, pH 6.25, 1 mm sodium citrate, 1 mm KCl, and 15 mm sucrose) at 75 rpm for 30 min at room temperature. Cordycepin C was added to a final concentration of 0.5 mm. The seedlings were incubated on a shaker at 75 rpm and harvested at 0, 20, 40, 60, 80, 100, and 120 min after treatment with cordycepin C. The materials were frozen in liquid nitrogen and stored at −80°C until use.
RNA Extraction and Quantitative PCR
RNA was extracted using TRNzol Universal reagent (Tiangen; DP424). DNA contamination was removed using the TURBO DNA-free Kit (Ambion; AM1907). Reverse transcription was performed with TransScript II First-Strand cDNA Synthesis SuperMix (Transgen; AH301) using 2 μg of total RNA. Quantitative PCR (qPCR) was performed with the KAPA SYBR FAST Universal qPCR Kit (Kapa; KK4601) on a Bio-Rad CFX-96 Real-Time PCR instrument using the following program: 3 min at 95°C followed by 40 cycles of 20 s at 95°C, 30 s at 60°C, and 20 s at 72°C. UBI3 (Solyc01g056840) was used as the internal control for reverse transcription-qPCR. The primers used for qPCR are listed in Supplemental Table S4.
Dual Luciferase Reporter Assay
For plasmid construction, the pluc-35Rluc backbone vector was obtained from pPZP211 (Hajdukiewicz et al., 1994). The nearly 1-kb SP5G 3ʹ UTR with the 52-bp sequence was amplified from LA1310 genomic DNA and integrated into pluc-35Rluc using the Infusion HD cloning kit (Clontech; 639649). For the plasmids with the short version of the 3ʹ UTR lacking the 52-bp sequence, primers XP1347 and XP1348 were used with the plasmid containing the full-length 3ʹ UTR as template. The 4.17-kb promoter of SP5G was amplified using TS30M and TS30C genomic DNA as templates, then integrated into the constructs upstream of the luc sequence using the Infusion HD cloning kit (Clontech; 639649). The primers used for the constructs are listed in Supplemental Table S4. The plasmids were transformed into Agrobacterium tumefaciens EHA105 competent cells. A single colony was cultured in Luria-Bertani medium until the OD600 value reached 1. The A. tumefaciens cells were collected by centrifugation and suspended with 10 mm MgCl2 and 150 μm acetosyringone. The cell suspensions containing the luciferase construct and the p19 plasmid were mixed at equivalent volumes and infiltrated into tobacco (Nicotiana tabacum) leaves using a syringe. The leaves were harvested and ground in liquid nitrogen at 2 d after infiltration. The activities of firefly luciferase and Renilla luciferase were measured using the Dual-Luciferase Reporter Assay System (Promega; E1910) on a Promega GLOMAX 20/20 LUMINOMETER device following the manual instructions.
ChIP and qPCR
The ChIP assay was performed using 1 g of 18-d-old seedlings collected at ZT4 under LD conditions as described previously with minor modifications (Zhang et al., 2015). In brief, the seedlings were cross-linked with formaldehyde, and the nuclei were isolated. The chromatin was sheared using a Diagenode Bioruptor Plus instrument to obtain ∼300-bp fragments. The anti-H3K27ac (Merck; 07-360) and anti-RNA PolII (Abcam; ab817) antibodies were used for immunoprecipitation. The ChIP DNA was extracted after reverse cross-linking. qPCR was performed using the KAPA SYBR FAST Universal qPCR Kit (Kapa; KK4601) on a Bio-Rad CFX-96 Real-Time PCR instrument with the following program: 3 min at 95°C followed by 50 cycles of 20 s at 95°C, 30 s at 60°C, and 20 s at 72°C. The region around the transcription start site of ACTIN (Solyc03g078400) was used as an internal control. The primers used for ChIP-qPCR are listed in Supplemental Table S4.
3C Assay
The 3C assay was performed as described previously (Hagège et al., 2007; Louwers et al., 2009). Eighteen-day-old seedlings collected at ZT4 under LD conditions were cross-linked using formaldehyde, and nuclei were isolated. The cross-linked chromatin was digested overnight using DpnII (New England Biolabs; R0543), followed by ligation with T4 DNA ligase (Thermo Fisher; EL0013) in a large volume and reverse cross-linking overnight. Ligated DNA was extracted by phenol-chloroform and precipitated with 3 volumes of ethanol. A ligated fragment of EF1α (Solyc06g005060) was amplified as the internal control to correct for differences between the templates. To eliminate the differences in primer set efficiency, 5 μg of the control plasmid (CP268) containing the genomic SP5G locus was cut with DpnII and then ligated, creating a pool of all possible ligation products for the locus of interest, consistent with the treatment of our samples. For the construction of the control plasmid, the SP5G genomic sequence from 4,800 bp upstream of the start codon to 2,869 bp downstream of the stop codon was amplified using NIL-fo1_01CC genomic DNA as a template. Three fragments of the genomic sequence were amplified separately, double digested with endonucleases, and ligated sequentially to one pEasy-blunt vector (Transgen; CB101). The primers and endonucleases used for construction are listed in Supplemental Table S4. The randomly ligated DNA products of control plasmids were diluted 100 times for qPCR. qPCR was performed with FastStart Universal SYBR Green Master (Roche; 4913914001) on a Bio-Rad CFX-96 Real-Time PCR instrument using the following program: 3 min at 95°C followed by 50 cycles of 20 s at 95°C, 30 s at 60°C, and 20 s at 72°C. The primers used for 3C are listed in Supplemental Table S4.
Accession Numbers
Sequence data from this article can be found in Sequence Read Archive under accession numbers SRP093370 and SRR2391865. The names and accession numbers of all genes mentioned in this work can be found in Supplemental Table S4.
Supplemental Data
The following supplemental materials are available.
Supplemental Figure S1. Manhattan plots for flowering time under SD conditions.
Supplemental Figure S2. Genotyping results showed the difference of nearly 50 kb between the NILs.
Supplemental Figure S3. SP5G sequence polymorphisms between MM and CC from 3 kb upstream of the start codon to the 3ʹ UTR.
Supplemental Figure S4. The mRNA stability analysis of SP5G showed no difference between NIL-Q-fo1_01MM and NIL-Q-fo1_01CC.
Supplemental Figure S5. The 3ʹ UTR of SP5G does not display enhancer activity when driven by the 35S minimal promoter.
Supplemental Figure S6. SP5G controls the photoperiodic response in tomato.
Supplemental Figure S7. Flowering time of TS30M, TS30C, and the heterozygous progeny of TS30 under LD and SD conditions.
Supplemental Table S1. Markers used for comparing the background between the NILs.
Supplemental Table S2. Polymorphisms within SP5G in 40 tomato accessions.
Supplemental Table S3. TF-binding motifs around the 52-bp sequence in the SP5G 3′ UTR.
Supplemental Table S4. List of primers.
Acknowledgments
We thank Dr. Sanwen Huang and Dr. Jianchang Gao for donating seeds of the wild, cherry, and big-fruit tomatoes. We also thank Dr. Guodong Yang for providing pPZP211 plasmids, Dr. Lin Guo for technical help on the 3C assay, and Dr. Haiyang Wang for advice on article improvement.
Footnotes
This work was supported by the National Key Research and Development Program of China (2016YFD0101007), the National Natural Science Foundation of China (31601772), and the Science and Technology Innovation Program of the Chinese Academy of Agricultural Sciences.
Articles can be viewed without a subscription.
References
- Andrés F, Coupland G (2012) The genetic basis of flowering responses to seasonal cues. Nat Rev Genet 13: 627–639 [DOI] [PubMed] [Google Scholar]
- Barrett LW, Fletcher S, Wilton SD (2012) Regulation of eukaryotic gene expression by the untranslated gene regions and other non-coding elements. Cell Mol Life Sci 69: 3613–3634 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bäurle I, Dean C (2006) The timing of developmental transitions in plants. Cell 125: 655–664 [DOI] [PubMed] [Google Scholar]
- Beales J, Turner A, Griffiths S, Snape JW, Laurie DA (2007) A pseudo-response regulator is misexpressed in the photoperiod insensitive Ppd-D1a mutant of wheat (Triticum aestivum L.). Theor Appl Genet 115: 721–733 [DOI] [PubMed] [Google Scholar]
- Blanca J, Cañizares J, Cordero L, Pascual L, Diez MJ, Nuez F (2012) Variation revealed by SNP genotyping and morphology provides insight into the origin of the tomato. PLoS ONE 7: e48198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao K, Cui L, Zhou X, Ye L, Zou Z, Deng S (2016) Four tomato FLOWERING LOCUS T-like proteins act antagonistically to regulate floral initiation. Front Plant Sci 6: 1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H, Du G, Song X, Li L (2017) Non-coding transcripts from enhancers: new insights into enhancer activity and gene expression regulation. Genomics Proteomics Bioinformatics 15: 201–207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark RM, Wagler TN, Quijada P, Doebley J (2006) A distant upstream enhancer at the maize domestication gene tb1 has pleiotropic effects on plant and inflorescent architecture. Nat Genet 38: 594–597 [DOI] [PubMed] [Google Scholar]
- Dekker J, Rippe K, Dekker M, Kleckner N (2002) Capturing chromosome conformation. Science 295: 1306–1311 [DOI] [PubMed] [Google Scholar]
- Doebley J, Stec A, Hubbard L (1997) The evolution of apical dominance in maize. Nature 386: 485–488 [DOI] [PubMed] [Google Scholar]
- Fornara F, Panigrahi KC, Gissot L, Sauerbrunn N, Rühl M, Jarillo JA, Coupland G (2009) Arabidopsis DOF transcription factors act redundantly to reduce CONSTANS expression and are essential for a photoperiodic flowering response. Dev Cell 17: 75–86 [DOI] [PubMed] [Google Scholar]
- Frary A, Nesbitt TC, Grandillo S, Knaap E, Cong B, Liu J, Meller J, Elber R, Alpert KB, Tanksley SD (2000) fw2.2: a quantitative trait locus key to the evolution of tomato fruit size. Science 289: 85–88 [DOI] [PubMed] [Google Scholar]
- Fujino K, Sekiguchi H (2005) Mapping of QTLs conferring extremely early heading in rice (Oryza sativa L.). Theor Appl Genet 111: 393–398 [DOI] [PubMed] [Google Scholar]
- Fujino K, Yamanouchi U, Yano M (2013) Roles of the Hd5 gene controlling heading date for adaptation to the northern limits of rice cultivation. Theor Appl Genet 126: 611–618 [DOI] [PubMed] [Google Scholar]
- Hagège H, Klous P, Braem C, Splinter E, Dekker J, Cathala G, de Laat W, Forné T (2007) Quantitative analysis of chromosome conformation capture assays (3C-qPCR). Nat Protoc 2: 1722–1733 [DOI] [PubMed] [Google Scholar]
- Hajdukiewicz P, Svab Z, Maliga P (1994) The small, versatile pPZP family of Agrobacterium binary vectors for plant transformation. Plant Mol Biol 25: 989–994 [DOI] [PubMed] [Google Scholar]
- Hajheidari M, Koncz C, Eick D (2013) Emerging roles for RNA polymerase II CTD in Arabidopsis. Trends Plant Sci 18: 633–643 [DOI] [PubMed] [Google Scholar]
- Harlen KM, Churchman LS (2017) The code and beyond: transcription regulation by the RNA polymerase II carboxy-terminal domain. Nat Rev Mol Cell Biol 18: 263–273 [DOI] [PubMed] [Google Scholar]
- Hayama R, Yokoi S, Tamaki S, Yano M, Shimamoto K (2003) Adaptation of photoperiodic control pathways produces short-day flowering in rice. Nature 422: 719–722 [DOI] [PubMed] [Google Scholar]
- Henriques T, Scruggs BS, Inouye MO, Muse GW, Williams LH, Burkholder AB, Lavender CA, Fargo DC, Adelman K (2018) Widespread transcriptional pausing and elongation control at enhancers. Genes Dev 32: 26–41 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imaizumi T, Kay SA (2006) Photoperiodic control of flowering: not only by coincidence. Trends Plant Sci 11: 550–558 [DOI] [PubMed] [Google Scholar]
- Izawa T, Oikawa T, Sugiyama N, Tanisaka T, Yano M, Shimamoto K (2002) Phytochrome mediates the external light signal to repress FT orthologs in photoperiodic flowering of rice. Genes Dev 16: 2006–2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jang S, Marchal V, Panigrahi KC, Wenkel S, Soppe W, Deng XW, Valverde F, Coupland G (2008) Arabidopsis COP1 shapes the temporal pattern of CO accumulation conferring a photoperiodic flowering response. EMBO J 27: 1277–1288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkins JA. (1948) The origin of the cultivated tomato. Econ Bot 2: 379–392 [Google Scholar]
- Kim TK, Hemberg M, Gray JM, Costa AM, Bear DM, Wu J, Harmin DA, Laptewicz M, Barbara-Haley K, Kuersten S, et al. (2010) Widespread transcription at neuronal activity-regulated enhancers. Nature 465: 182–187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine M, Cattoglio C, Tjian R (2014) Looping back to leap forward: transcription enters a new era. Cell 157: 13–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25: 1754–1760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lifschitz E, Eviatar T, Rozman A, Shalit A, Goldshmidt A, Amsellem Z, Alvarez JP, Eshed Y (2006) The tomato FT ortholog triggers systemic signals that regulate growth and flowering and substitute for diverse environmental stimuli. Proc Natl Acad Sci USA 103: 6398–6403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin T, Zhu G, Zhang J, Xu X, Yu Q, Zheng Z, Zhang Z, Lun Y, Li S, Wang X, et al. (2014) Genomic analyses provide insights into the history of tomato breeding. Nat Genet 46: 1220–1226 [DOI] [PubMed] [Google Scholar]
- Lipka AE, Tian F, Wang Q, Peiffer J, Li M, Bradbury PJ, Gore MA, Buckler ES, Zhang Z (2012) GAPIT: Genome Association and Prediction Integrated Tool. Bioinformatics 28: 2397–2399 [DOI] [PubMed] [Google Scholar]
- Liu LJ, Zhang YC, Li QH, Sang Y, Mao J, Lian HL, Wang L, Yang HQ (2008) COP1-mediated ubiquitination of CONSTANS is implicated in cryptochrome regulation of flowering in Arabidopsis. Plant Cell 20: 292–306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Louwers M, Splinter E, van Driel R, de Laat W, Stam M (2009) Studying physical chromatin interactions in plants using Chromosome Conformation Capture (3C). Nat Protoc 4: 1216–1229 [DOI] [PubMed] [Google Scholar]
- Matoulkova E, Michalova E, Vojtesek B, Hrstka R (2012) The role of the 3′ untranslated region in post-transcriptional regulation of protein expression in mammalian cells. RNA Biol 9: 563–576 [DOI] [PubMed] [Google Scholar]
- Matsubara K, Hori K, Ogiso-Tanaka E, Yano M (2014) Cloning of quantitative trait genes from rice reveals conservation and divergence of photoperiod flowering pathways in Arabidopsis and rice. Front Plant Sci 5: 193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, Garimella K, Altshuler D, Gabriel S, Daly M, et al. (2010) The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 20: 1297–1303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer RS, Purugganan MD (2013) Evolution of crop species: genetics of domestication and diversification. Nat Rev Genet 14: 840–852 [DOI] [PubMed] [Google Scholar]
- Mora A, Sandve GK, Gabrielsen OS, Eskeland R (2016) In the loop: promoter-enhancer interactions and bioinformatics. Brief Bioinform 17: 980–995 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nesbitt TC, Tanksley SD (2002) Comparative sequencing in the genus Lycopersicon: implications for the evolution of fruit size in the domestication of cultivated tomatoes. Genetics 162: 365–379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oka R, Zicola J, Weber B, Anderson SN, Hodgman C, Gent JI, Wesselink JJ, Springer NM, Hoefsloot HCJ, Turck F, et al. (2017) Genome-wide mapping of transcriptional enhancer candidates using DNA and chromatin features in maize. Genome Biol 18: 137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pnueli L, Carmel-Goren L, Hareven D, Gutfinger T, Alvarez J, Ganal M, Zamir D, Lifschitz E (1998) The SELF-PRUNING gene of tomato regulates vegetative to reproductive switching of sympodial meristems and is the ortholog of CEN and TFL1. Development 125: 1979–1989 [DOI] [PubMed] [Google Scholar]
- Porrua O, Libri D (2015) Transcription termination and the control of the transcriptome: why, where and how to stop. Nat Rev Mol Cell Biol 16: 190–202 [DOI] [PubMed] [Google Scholar]
- Rodriguez-Leal D, Lemmon ZH, Man J, Bartlett ME, Lippman ZB (2017) Engineering quantitative trait variation for crop improvement by genome editing. Cell 171: 470–480.E8 [DOI] [PubMed] [Google Scholar]
- Sakabe NJ, Savic D, Nobrega MA (2012) Transcriptional enhancers in development and disease. Genome Biol 13: 238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samach A, Onouchi H, Gold SE, Ditta GS, Schwarz-Sommer Z, Yanofsky MF, Coupland G (2000) Distinct roles of CONSTANS target genes in reproductive development of Arabidopsis. Science 288: 1613–1616 [DOI] [PubMed] [Google Scholar]
- Song YH, Shim JS, Kinmonth-Schultz HA, Imaizumi T (2015) Photoperiodic flowering: time measurement mechanisms in leaves. Annu Rev Plant Biol 66: 441–464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soyk S, Müller NA, Park SJ, Schmalenbach I, Jiang K, Hayama R, Zhang L, Van Eck J, Jiménez-Gómez JM, Lippman ZB (2017) Variation in the flowering gene SELF PRUNING 5G promotes day-neutrality and early yield in tomato. Nat Genet 49: 162–168 [DOI] [PubMed] [Google Scholar]
- Studer A, Zhao Q, Ross-Ibarra J, Doebley J (2011) Identification of a functional transposon insertion in the maize domestication gene tb1. Nat Genet 43: 1160–1163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suárez-López P, Wheatley K, Robson F, Onouchi H, Valverde F, Coupland G (2001) CONSTANS mediates between the circadian clock and the control of flowering in Arabidopsis. Nature 410: 1116–1120 [DOI] [PubMed] [Google Scholar]
- Takahashi Y, Teshima KM, Yokoi S, Innan H, Shimamoto K (2009) Variations in Hd1 proteins, Hd3a promoters, and Ehd1 expression levels contribute to diversity of flowering time in cultivated rice. Proc Natl Acad Sci USA 106: 4555–4560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomato Genome Consortium (2012) The tomato genome sequence provides insights into fleshy fruit evolution. Nature 485: 635–641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner A, Beales J, Faure S, Dunford RP, Laurie DA (2005) The pseudo-response regulator Ppd-H1 provides adaptation to photoperiod in barley. Science 310: 1031–1034 [DOI] [PubMed] [Google Scholar]
- Valverde F, Mouradov A, Soppe W, Ravenscroft D, Samach A, Coupland G (2004) Photoreceptor regulation of CONSTANS protein in photoperiodic flowering. Science 303: 1003–1006 [DOI] [PubMed] [Google Scholar]
- Weber B, Zicola J, Oka R, Stam M (2016) Plant enhancers: a call for discovery. Trends Plant Sci 21: 974–987 [DOI] [PubMed] [Google Scholar]
- Wigge PA, Kim MC, Jaeger KE, Busch W, Schmid M, Lohmann JU, Weigel D (2005) Integration of spatial and temporal information during floral induction in Arabidopsis. Science 309: 1056–1059 [DOI] [PubMed] [Google Scholar]
- Wilhelm EP, Turner AS, Laurie DA (2009) Photoperiod insensitive Ppd-A1a mutations in tetraploid wheat (Triticum durum Desf.). Theor Appl Genet 118: 285–294 [DOI] [PubMed] [Google Scholar]
- Xue W, Xing Y, Weng X, Zhao Y, Tang W, Wang L, Zhou H, Yu S, Xu C, Li X, et al. (2008) Natural variation in Ghd7 is an important regulator of heading date and yield potential in rice. Nat Genet 40: 761–767 [DOI] [PubMed] [Google Scholar]
- Yang Q, Li Z, Li W, Ku L, Wang C, Ye J, Li K, Yang N, Li Y, Zhong T, et al. (2013) CACTA-like transposable element in ZmCCT attenuated photoperiod sensitivity and accelerated the postdomestication spread of maize. Proc Natl Acad Sci USA 110: 16969–16974 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang S, Zhou B, Kang Y, Cui X, Liu A, Deleris A, Greenberg MV, Cui X, Qiu Q, Lu F, et al. (2015) C-terminal domains of a histone demethylase interact with a pair of transcription factors and mediate specific chromatin association. Cell Discov 1: 15003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou L, Zhang J, Yan J, Song R (2011) Two transposable element insertions are causative mutations for the major domestication gene teosinte branched 1 in modern maize. Cell Res 21: 1267–1270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu B, Zhang W, Zhang T, Liu B, Jiang J (2015) Genome-wide prediction and validation of intergenic enhancers in Arabidopsis using open chromatin signatures. Plant Cell 27: 2415–2426 [DOI] [PMC free article] [PubMed] [Google Scholar]