Abstract
Atherosclerosis is an autoimmune disease caused by self- and non-self-antigens contributing to excessive activation of T and B cell immune responses. These responses further aggravate vascular infiammation and promote progression of atherosclerosis and vulnerability to plaques via releasing pro-infiammatory cytokines. Regulatory T cells (Tregs) as the major immunoregulatory cells, in particular, induce and maintain immune homeostasis and tolerance by suppressing the immune responses of various cells such as T and B cells, natural killer (NK) cells, monocytes, and dendritic cells (DCs), as well as by secreting inhibitory cytokines interleukin (IL)-10, IL-35 and transcription growth factor β (TGF-β) in both physiological and pathological states. Numerous evidence demonstrates that reduced numbers and dysfunction of Treg may be involveved in atherosclerosis pathogenesis. Increasing or restoring the numbers and improving the immunosuppressive capacity of Tregs may serve as a fundamental immunotherapy to treat atherosclerotic cardiovascular diseases. In this article, we briefiy present current knowledge of Treg subsets, summarize the relationship between Tregs and atherosclerosis development, and discuss the possibilities of regulating Tregs for prevention of atherosclerosis pathogenesis and enhancement of plaque stability. Although the exact molecular mechanisms of Treg-mediated protection against atherosclerosis remain to be elucidated, the strategies for targeting the regulation of Tregs may provide specific and significant approaches for the prevention and treatment of atherosclerotic cardiovascular diseases.
Keywords: atherosclerosis, cardiovascular diseases, regulatory T cells, immune regulation, treatment strategy
Introduction
Atherosclerosis is a chronic inflammatory disease characterized by inflammatory cell activation, abnormal lipid deposition and plaque formation within the intima-media layer of large and medium-sized arteries1. Epidemiological studies have revealed that the incidences of atherosclerosis are increasing due to the improvement of living standards. The largest threat in atherosclerosis involves the rupture of instable plaques and thrombosis, which leads to cardiovascular complications such as acute coronary syndrome (ACS) and stroke. Previous studies on the pathological mechanism of atherosclerosis have largely focused on lipid metabolism disorders, infiammatory response cascades, abnormal proliferation of vascular smooth muscle cell (VSMC), endothelial cell (EC) dysfunction and foam cell formation2. However, accumulating evidence suggests that the activation of innate and adaptive immune responses is involved in the development and progression of atherosclerosis. As a result, numerous monocytes and T and B cells are recruited to lesion sites, enhancing the inflammatory response and promoting the initiation and progression of atherosclerotic lesions.
Although the most important cells in atherosclerotic lesions are macrophage-derived foam cells, T cells are extensively studied in mouse atherosclerosis models based on their immune and genetic manipulation3. Tregs are a specific subpopulation of T cells, and numerous subtypes of Tregs, including CD4+CD25+ Tregs, type 1 regulatory T cells (Tr1), type 3 helper T cells (Th3), CD4+LAP+ Tregs, CD8+CD28+ Tregs, and Tγδ Tregs, have been reported. The most typical Treg is CD4+CD25, which is crucial in immune response regulation4. CD8+CD28+ Tregs inhibit activation of naive and effector T cells and antibody production5. The function of Tγδ Tregs is similar to that of Tr1, which suppress the proliferation and pro-inflammatory cytokine secretion by naive T cells and regulate mucosal immune tolerance. In addition, these cells also regulate tumor immunity and autoimmunity4,6. Th3 mainly mediates oral tolerance and suppresses the proliferation of Th1 and Th24. CD4+LAP+ Tregs are associated with various autoimmune diseases, such as atherosclerosis and diabetes, in mouse models7.
Although cellular immune responses are detrimental to the development of atherosclerosis, all Treg types mediate immunomodulation and protect against atherosclerosis. The processes mainly involve the secretion of inhibitory cytokines, production of immunosuppressive enzymes, inhibition of cell-to-cell contact, metabolic disruption, and suppression of DC maturation and function8,9,10. The inhibitory cytokines secreted by Tregs mainly include IL-10, IL-35, and TGF-β. In addition, Tregs express several characteristic molecules related to their function, including the forkhead box P3 transcription factor (FOXP3), cytotoxic T lymphocyte-associated antigen-4 (CTLA-4), inducible co-stimulator (ICOS), lymphocyte activation gene-3 (LAG-3), CD80/CD86, and CD25. FOXP3 is a regulatory molecule for Treg maturation and maintenance of immunosuppressive functions. CTLA-4 also regulates the suppressive activity of Tregs. ICOS is required for accurate identification by Tregs. ICOS deficiency also inhibits immunosuppressive functions and decreases the numbers of Tregs. Co-stimulatory molecules CD80/CD86 and CD28 are required for Treg development and homeostasis4,11. CD25 is required for Treg survival. Substantial evidence clearly demonstrates that decreased Treg proliferation or dysfunction causes abnormal immune responses and pathologies, such as tumors, infections, autoimmune diseases, and other immune diseases12,13.
Over the past decade, the notion that Tregs regulate the development of atherosclerosis by regulating immune activation and inducing immune tolerance in atherosclerotic models and patients has received considerable attention. The focus of this review is to summarize the effects of Tregs in the pathogenesis of atherosclerosis and provide an overview of recent studies on the regulation of Tregs for the treatment of atherosclerotic disease. Understanding the pathogenesis of atherosclerosis and the development of constructive therapeutic strategies to prevent cardiovascular disease (CVD) are important.
The relationship between Tregs and development of atherosclerosis
A number of studies have demonstrated that various functional T cell subsets are closely related to the occurrence of CVD, and effector T cells are positively correlated with atherosclerosis and CVD in human and animals. Tregs are present in atherosclerotic plaques14. Additionally, evidence from animal and human studies indicate that decreased Treg numbers are beneficial to atherosclerosis processes. For instance, research reveals no evidence of atherosclerotic lesions in C57BL/6 mice or ApoE−/− mice. However, ApoE−/− mice fed a high-fat diet (HFD) exhibit reduced numbers of Tregs and an increased incidence of atherosclerosis15. Moreover, several clinical studies have also reported that plaque vulnerability and the development of ACS are associated with low levels of circulating immunosuppressive Tregs16,17 and that pro-inflammatory DC and T effector cell infiltration is increased18,19. Liu et al. demonstrated that the imbalance of Th17/Tregs also enhances plaque instability and the occurrence of ACS20. Depleting Tregs with anti-CD25 specific antibody accelerates the development of atherosclerotic lesions and increases plaque instability in ApoE−/− mice21. In addition, Zhang et al observed that Treg apoptosis was increased in non-ST-elevated ACS patients compared with chronic stable angina/chest pain syndrome patients, which may be attributable to decreased anti-apoptotic gene Bcl-2 mRNA and increased pro-apoptotic gene Bak mRNA. Moreover, oxidized low-density lipoprotein (ox-LDL) mediated the reduction in Treg numbers by promoting apoptosis22,23.
In addition to the low numbers of Tregs promoting atherosclerotic lesions, numerous studies have demonstrated that dysregulation of Tregs may contribute to the development of atherosclerosis. For example, compared with C57BL/6 mice, the inhibition of effector T cell function by Tregs isolated from ApoE−/− mice was significantly reduced. The same results were validated in ACS patients, thus reflecting defects in the immunosuppressive capacity of Tregs7. Atherosclerotic lesions also subsequently affect the plasticity of Tregs. Butcher et al found that atherosclerosis promoted the formation of an intermediately plastic Th1/Treg subset with immunosuppressive dysfunction and promoted inflammation and atherogenic T cell responses in ApoE−/− mice24. Klingenberg et al found that Treg numbers increased in coronary thrombi adjacent to culprit lesions, suggesting that circulating Tregs may migrate to atherosclerotic lesions to regulate local inflammatory responses25. Moreover, the deletion of IL-10 increased pro-inflammatory cell recruitment and plaque vulnerability in mice models. Furthermore, compared with stable patients, Treg and IL-10 levels were significantly reduced in unstable patients26.
Treg surface molecules are also associated with the development of atherosclerosis. For instance, Jia et al found that FOXP3 expression was negatively correlated with the risk of ACS17. Tregs isolated from FOXP3−/− mice lost suppressive pathogenic T cell function. Depletion of Tregs (DEREG)/LDLr−/− mice with specific depletion of FOXP3+ Tregs significantly aggravated atherosclerosis development and increased plasma atherogenic lipoprotein levels27. Ait-Oufella et al demonstrated that CD80/CD86 and CD28 deficiencies in LDLR−/− mice also promoted the development of atherosclerosis21. ICOS deficiency decreases Treg numbers and inhibits the immunosuppressive function of Tregs. Peripheral blood ICOS+ Tregs were reduced in myocardial infarction (MI) and stable angina patients28.
Collectively, these data illustrated that the numbers and immunoregulatory functions of Tregs were closely related to the development of atherosclerosis. Increasing or restoring the numbers and improving the immunosuppressive capacity of Tregs may serve as a fundamental immunotherapy to treat atherosclerotic cardiovascular diseases.
Mechanism of Treg protection against atherosclerosis
Inhibition of effector T and B cell function
Atherosclerosis involves complicated interplay between different immune cell and cytokine networks. The underlying mechanisms of Treg anti-atherosclerotic effects have been extensively investigated (Figure 1). T cells are first recruited to atheroma and mainly accumulated in unstable plaques. The majority of the atherogenic T cells include effector Th1 and Th17. Multiple studies demonstrate that Th1 promotes the migration of monocytes and T cells into atherosclerotic lesions and activate lesional antigen presenting cells (APCs) by secreting interferon-γ (IFN-γ), which enhances atherosclerosis progression and plaque vulnerability29,30. Th17 plays a vital role in atherogenesis by secreting pro-inflammatory IL-17, IL-6 and chemokines8,29. The function of Th2 remains controversial based on the change of atherosclerotic lesion state, exact site and the differences in experimental models.
Figure 1.
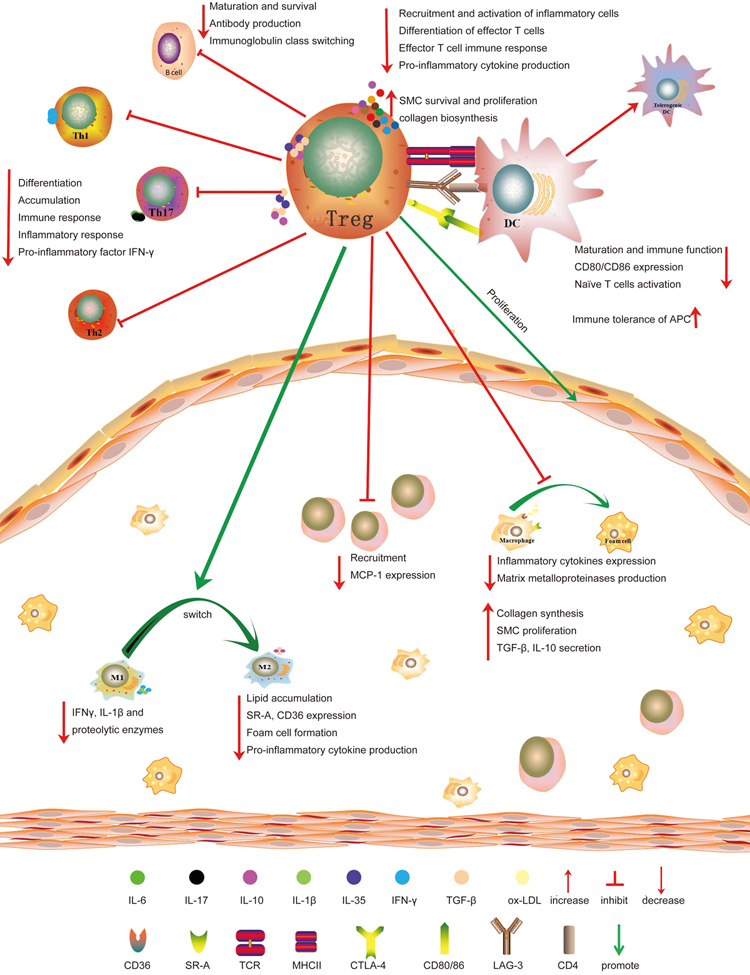
The mechanism of Tregs in the protection against atherosclerosis. Through secretion of IL-10, IL-35, and TGF-β, Tregs decrease inflammatory cell recruitment and activation, inhibit effector T cells differentiation and inflammation, and promote SMC survival and proliferation and collagen biosynthesis. Accordingly, IL-10 induces the M1 macrophage switch to the M2 phenotype, which attenuates atherosclerosis by decreasing IFN-γ, IL-1β, and proteolytic enzyme production and promoting collagen synthesis and SMC proliferation. Moreover, Tregs inhibit T cell differentiation into Th1, Th2, and Th17 subtypes and suppress the immune and inflammatory responses by inhibiting pro-inflammatory cytokine IFN-γ production. Tregs also suppress B cell maturation, antibody production and immunoglobulin class switching and modulate B cell survival by secreting granzymes and perforin. Among other pathways, Tregs inhibit the function and maturation of DCs, induce immune tolerance of APCs, and suppress naive T cell activation by CTLA-4-, TGF-β- and IL-10-mediated mechanisms. Tregs enhance plaque stability by inhibiting monocyte recruitment and promoting SMC proliferation. Furthermore, Tregs inhibit lipid and foam cell formation by decreasing the expression of CD36 and SR-A and reduce plasma VLDL, LDL, and cholesterol levels.
Tregs suppress T cell differentiation into Th1 and Th17 subtypes. Interestingly, Tregs suppress Th1 and Th17 immune responses in atherosclerosis by secreting IL-10, IL-35, and TGF-β. IL-10 is produced by Tr1, which reduces atherosclerotic lesions by inhibiting Th1 differentiation and decreasing T cell and macrophage accumulation and cytokine production in atherosclerotic plaques31. Consistently, Tr1 reduced immune responses and decreased plaque sizes and inflammation by producing IL-10 and decreasing the production of IFN-γ in ApoE−/− mice32. TGF-β attenuated atherosclerosis by inhibiting the recruitment and activation of inflammatory cells in atherosclerotic lesions and increasing the stability of plaques by promoting SMC survival and proliferation and collagen biosynthesis. However, TGF-β deficiency accelerates the development of atherosclerosis and enhances vulnerable plaques in hypercholesterolemic mice33. TGF-β overexpression increases plaque stability and prevents the progression of atherosclerosis34. IL-35 mediates the prevention of atherosclerosis by suppressing the proliferation of the T cell response, regulating the activation of naive T cells, and inhibiting the production of pro-inflammatory cytokines35. Acute myocardial infarction (AMI), unstable angina, and stable angina patients are associated with reduced IL-35 serum levels36. In addition, IL-2 is an important cytokine for T cell proliferation. Tregs reduce IL-2 levels, leading to the arrest of T cell proliferation. Therefore, Tregs protect against atherosclerosis by suppressing the function of pro-atherogenic effector T cells.
B cells are very rare within atherosclerotic plaques. However, antibodies produced by B2 cells abundantly bind within atherosclerotic plaques. Moreover, B2 cells aggravate atherogenesis by enhancing pro-inflammatory cytokine activity29. IgE was identified in human carotid atherosclerotic plaques, elevating plasma low-density lipoprotein (LDL) and increasing the risk of CVD, and in particular increasing plaque instability37. Antibody effector pathways and the classical complement pathway are activated in human atherosclerotic plaques. Transfer of B2 cells into ApoE−/− mice accelerated the progression of atherosclerosis38. Selective B cell depletion reduced atherosclerosis development and progression in ApoE−/− mice. Tregs regulate the B cell response by impairing their maturation and inhibiting antibody production and immunoglobulin class switching. Moreover, Tregs modulate survival of B cells by secreting granzymes and perforin39. Upon treatment with anti-CD20 antibody, B cell depletion attenuated atherosclerosis by suppressing effector T cell activation in LDLr−/− and ApoE−/− mice27. Therefore, regulation of B cell function is also a novel mechanism by which Tregs protect against atherosclerosis.
Modulation of dendritic cell function and maturation
DCs are specific immune cells that mediate antigen-specific immunity and tolerance and promote T cell activation. Multiple lines of evidence have demonstrated that DCs were significantly increased in atherosclerosis-prone vessels, promoted inflammatory processes within atherosclerotic lesions, and increased plaque instability29,40. These findings suggest that an inverse correlation between DCs and Tregs exists in atherosclerotic lesions. Tregs inhibit DC function and maturation by producing IL-10 and TGF-β. Other surface molecules expressed by Tregs also affect the function of DCs, such as CTLA-4 and LAG-3. Tregs downregulate CD80/CD86 expression in human and murine DCs. CTLA-4 binds to CD80/CD86, blocking the ability of DCs to activate naive T cells and inducing immune tolerance of APCs41. Several studies demonstrate that increased CTLA-4 mRNA levels were associated with increased Tregs and prevented atherosclerosis11,42,43,44. LAG-3 binds with DC surface CD4, which suppresses maturation and the immune-stimulatory capacity of DCs45.
Inhibition of macrophage inflammation and decreased plaque vulnerability
In the 1960s, macrophages were identified as the key immune cells in atherosclerotic plaques that maintain a chronic inflammation state by modulating inflammatory mediator, adhesion molecule and chemokine factor secretion and reactive oxygen and nitrogen species production46,47. Macrophages are heterogeneous cells within the atherosclerotic plaques that switch phenotypes depending on microenvironment changes. During the development of atherosclerosis, macrophages switch from the “classical” M1 phenotype to the “alternative” M2 phenotype. M1 phenotype macrophages exhibit pro-inflammatory properties and express pro-inflammatory cytokines, including IFN-γ and IL-1β, and produce proteolytic enzymes, resulting in atherosclerosis development by amplifying the inflammation response and enhancing plaque destabilization by degrading the extracellular matrix. In contrast, M2 phenotype macrophages exhibit anti-inflammatory properties and secrete TGF-β and IL-1048. Tregs promote the transformation of M1 macrophages to M2 macrophages by releasing IL-10, which is beneficial in preventing the development of atherosclerosis49. Tregs and monocyte co-cultures induce M2 phenotype production. Moreover, upon stimulation with lipopolysaccharides (LPS), the secretion of pro-inflammatory cytokines by Treg-treated monocytes was significantly reduced. The unstable plaques are attributed to lipid-filled necrotic cores, thin fibrous caps, and low ratios of SMCs to macrophages. Activated M2 macrophages enhance plaque stability by promoting collagen synthesis and SMC proliferation. These findings suggest that Tregs enhance lesion stability by inducing M2 macrophages. Tregs reduce the risk of plaque rupture by increasing SMC and collagen content and reducing pro-inflammatory cytokine release and matrix metalloproteinase (MMP) production in atherosclerotic lesions in ApoE−/− mice50.
Regulation of cholesterol metabolism and foam cell formation
Chronically high blood lipid levels activate vascular immune responses, promote the differentiation of monocytes to macrophages and phagocytosis of lipids, and ultimately promote the formation of macrophage-derived foam cells and atherosclerotic plaques51,52,53. The ox-LDL is digested by macrophages and serves as an antigen peptide presented to T cells, resulting in adaptive immune system activation and cytotoxic Th1 immune response initiation44. Therefore, regulation of macrophage cholesterol influx and efflux is of great importance in the prevention of atherosclerosis51. Numerous studies demonstrated that the development of atherosclerosis can be prevented by promoting the expression of cholesterol transporter-related protein ATP binding cassette transporters A1/G1 (ABCA1/G1) and Scavenger receptor type B class I (SR-BI) and enhancing the efflux of cholesterol in macrophages52,53,54. Studies have demonstrated that Tregs inhibit lipid accumulation in peritoneal macrophages by downregulating the expression of scavenger receptor class A (SR-A) and CD36, which inhibits foam cell formation49. Monocyte chemotactic factor-1 (MCP-1) mediates the migration of bone marrow-derived mononuclear cells to peripheral blood. Subramanian et al found that Tregs may impede monocyte recruitment into lesions by inhibiting MCP-1 expression, which also exhibits a negative effect on macrophage-derived foam cell formation and accumulation55. In addition, IL-10 reduces very low-density lipoprotein (VLDL) and LDL levels in LDLr−/− mice. DEREG mice exhibit increased plasma cholesterol and VLDL levels27. Induction of Treg proliferation in atherosclerotic mice reduces plasma cholesterol levels56.
Strategies for Tregs in the prevention and treatment of atherosclerosis
As the main cause of death worldwide, atherosclerotic plaque rupture seriously affects the health of the population and causes enormous financial burden. Over recent years, statin lipid-lowering drugs are the most common drugs used for the treatment of atherosclerotic-related diseases. Although these drugs are effective in the prevention and treatment of atherosclerosis, they have numerous undesirable effects57. Safer and more effective treatment strategies are urgently needed. Newly developed regulatory Treg biology has expanded the horizons for the treatment of autoimmune diseases and the prevention of graft versus host disease (GVHD)12,13. The development of atherosclerosis and plaque instability is mainly caused by an imbalance between effector T cells and Tregs, triggering a cascade of inflammatory reactions. Therefore, targeted treatment of atherosclerosis by modulating immunosuppressive properties and increasing the numbers of Tregs may result in significant progress, as shown in Table 1.
Table 1.
Targeted therapy of atherosclerosis by regulating regulatory T cells.
| Approaches for regulating Tregs | Effects of regulation Tregs | |
|---|---|---|
| Adoptive transfer Tregs | Tregs expansion and inhibition of effector T cells function | |
| Atherosclerosis relevant antigens (ox-LDL, HSP60, ApoB100) | Induction of antigen specific Tregs and tolerogenic DCs | |
| Pharmacological approaches | Rapamycin | Tregs expansion and T effector cell depletion |
| Mycophenolate mofeti | Inhibition of macrophages, DCs, T cells, and NKs activation and increase in the number of Tregs | |
| Vitamin D3 | Induction of Tregs and tolerogenic DCs | |
| FTY720 (fingolimod) | Tregs expansion | |
| Pioglitazone | Regulation of the balance between effector T cells and Tregs | |
| Cholesterol lowering drugs | Modulation of the ratio of Treg/effector T cell and promotion of TGFβ and IL-10 | |
| Amygdalin (vitamin B17) | Induction of Tregs expansion and increase in IL-10, TGF-β expression | |
| Application of antibodies and cytokines cholesterol | IL-2 | Selective Tregs expansion |
| Anti-CD3 antibody | Induction of Tregs expansion, reducing CD4+ T cells and lowering plasma | |
| Integrin αvβ8 | Modulation of Tregs function and inhibition of effector T cell function | |
| G-CSF | Induction of Tregs expansion and increase in IL-10 expression. | |
| Physical therapy | UVB irradiation | Improvement of the function of Tregs and regulation of effector T-cell responses |
Adoptive transfer and induced expansion of Tregs
As mentioned above, the decrease in Tregs leads to the development of atherosclerosis; thus, a potential therapeutic target involves an increase in Tregs. Recent studies have confirmed that adoptive transfer or expansion of Tregs markedly prevented the development of CVD in animal models. For example, the application of IL-2/anti-IL-2 complex-induced Treg expansion suppressed effector T cell function, reduced initial atherosclerotic lesion formation, decreased blood lipid levels, and enhanced lesion stability in LDLr−/− mice58. The administration of clonal Tregs reduces the development of atherosclerotic plaques and inflammation by reducing the atherogenic immune response and inducing IL-10 production in ApoE−/− mice59. The development of atherosclerosis may be prevented based on adoptive transfer of CD4+CD25+ Tregs in ApoE−/− mice60. Moreover, trials of adoptive transfer of Tregs have been employed for GVHD, transplantation, and autoimmunity therapy, and effective results were presented with no significant adverse effects61. Bone marrow-derived mesenchymal stem cells (BM-MSCs) exhibit immunosuppressive properties by inhibiting the activation of immune cells, such as T, B, and NK cells. Administration of BM-MSCs decreased atherosclerotic plaque size by inducing Treg expansion; improving the immunosuppressive ability of Tregs; inhibiting effector T cell proliferation; promoting anti-inflammatory cytokine TGF-β and IL-10 expression; decreasing pro-inflammatory cytokine IFN-γ, MMP-1, and hypersensitivity C reactive protein (hs-CRP) expression; and inhibiting foam cell formation by downregulation CD36 and SR-A expression in ApoE−/− mice62.
To some extent, atherosclerosis can be regarded as an autoimmune disease caused by autoimmune reactions to self-proteins, such as ox-LDL, apolipoprotein B (ApoB), and heat shock protein 60 (HSP60)63. Moreover, according to the identification of the causative antigen generation of antigen-specific Tregs has been studied in autoimmune diseases, such as rheumatoid arthritis12. Therefore, administration of atherosclerosis-relevant antigens, including ox-LDL, HSP60, and ApoB100, induced antigen-specific Tregs and tolerogenic DCs, which exhibit atheroprotective effects in mice11,64. For example, adoptive transfer of HSP60-specific Tregs inhibits atherosclerosis formation in ApoE-deficient mice65. Nasal ox-LDL- or HSP-60-induced mucosal tolerance attenuated atherosclerosis by increasing Tregs and TGF-β or IL-10 in ApoE−/− mice66. Adoptively transferred ox-LDL or ApoB100 also induced immune tolerance of DCs and inhibited atherosclerosis development by suppressing the T cell response, reducing plaque macrophage accumulation, and increasing collagen content. Recent studies have demonstrate that continuous subcutaneous injection of adjuvant-free ApoB100 peptides prevent the progression of atherosclerosis by inducing a specific Treg response in ApoE−/− mice67. Interestingly, ApoB100- and HSP60-associated auto-antigens have been defined and evaluated as therapeutic vaccines in animal models63. Notably, an ApoB100 vaccination has been developed for first-in-human clinical trials64. These data imply that regulating the number of Tregs may serve as a powerful therapeutic approach in the treatment of CVD patients. Moreover, the expansion of Tregs induced by atherosclerosis-associated vaccines may open a new path for the treatment of atherosclerosis.
Strategies for pharmacological regulation of Treg function
Recent studies have demonstrated that numerous drugs regulating Tregs have achieved significant efficacy in the treatment of atherosclerosis in animal models. For instance, mycophenolate mofetil has a strong immunosuppressive effect and inhibits the proliferation of T cells by interfering with DNA synthesis. Recent research demonstrated that mycophenolate mofetil can be used in the treatment of atherosclerotic diseases by reducing the activation of macrophages, DCs, T cells, and NKs; increasing the number of Tregs; reducing the production of MMP and cathepsins; and promoting the expression of lipid metabolism-associated genes, such as ApoE, peroxisome proliferator activated receptor (PPAR), ABCA1/G164,68. Mammalian target of rapamycin (mTOR) signaling regulates Treg differentiation and function. Rapamycin, an inhibitor of mTOR, exhibits immunosuppressive activity. induces Treg expansion and depletes T effector cells, implying that it could be employed as a new immunotherapy in T cell-mediated CVD69. Orally activated vitamin D3 is beneficial in the treatment of CVD by increasing Treg levels, inducing tolerogenic DCs, decreasing IL-12 expression, and increasing IL-10 expression in mice70. Consistent with these findings, oral FTY720 (fingolimod) ameliorates atherosclerosis by expanding Tregs, inhibiting effector T responses, and increasing TGF-ß expression in ApoE−/− mice71. Pioglitazone, a PPARγ agonist, ameliorates atherosclerosis by regulating the balance of Th1/Th2 cells, enhancing Treg response, and increasing the number of SMC and collagen content in ApoE-deficient mice72. Diet-induced hypercholesterolemia decreases the Treg/effector T cell ratio in atherosclerotic plaques, and this ration normalizes after diet reversal, suggesting that cholesterol-lowering therapies may reduce atherosclerosis by modulating the ratio of Tregs/effector T cells73. Meng et al demonstrated that simvastatin increases Treg levels and promotes TGFβ, IL-10, and FOXP3 expression in the atherosclerotic plaques of ApoE−/− mice. Atorvastatin also induces Treg expansion and promotes FOXP3 expression in humans but not in C57BL/6 mice. Moreover, simvastatin treatment in ACS patients also promotes Treg expansion, which is consistent with observations in animal studies74. Amygdalin, which is also referred to as vitamin B17, attenuates the development of atherosclerosis through regulation of lipid metabolism by decreasing plasma total cholesterol (TC), triglyceride (TG), and LDL levels; inducing Treg expansion and upregulating IL-10 and TGF-β expression in ApoE−/− mice75. Therefore, pharmacological regulation of the numbers and immunosuppressive activity of Tregs may provide valuable treatment options for atherosclerotic diseases.
Application of antibodies and cytokines to regulate Tregs
In addition to pharmacological approaches, the treatment of atherosclerosis with antibodies and cytokines has drawn increasing attention. IL-2 promotes the proliferation and differentiation of effector T cells and Tregs. However, low-dose IL-2 is effective in treating atherosclerosis via selective expansion of Tregs with significant sensitivity to IL-2. This method has been used in the clinical treatment of systemic lupus erythematosus37,76. Oral or intravenous administration of anti-CD3 antibody suppresses atherogenesis and atherosclerotic plaque formation by inducing Treg expansion and reducing CD4+ T cells in mice77. Treatment with anti-CD3 antibody and IL-2 complex also inhibits atherosclerosis78. Integrin αvβ8 mediates TGF-β activation. Thus, manipulation of integrin αvβ8 may modulate Treg function to suppress effector T cell-mediated atherosclerotic diseases79. Granulocyte Colony-Stimulating Factor (G-CSF) modulates immunity and improves immune-related diseases in animals. G-CSF also increases the numbers of Tregs and IL-10 levels and reduces IFN-γ levels in ApoE−/− mice80. Physical therapy can also play a protective role in atherosclerosis. For example, ultraviolet B irradiation attenuates the progression of atherosclerosis in atherosclerosis-prone mice by enhancing the function of Tregs and regulating effector T-cell response81.
Limitations of Tregs in atherosclerosis treatment
This review provides new insights into atherosclerotic immunopathogenesis and summarizes new therapeutic approaches related to the modulation of Tregs. Despite extensive evidence for the beneficial effects of Tregs in the treatment of animal immune diseases, there are limitations to the effects and safety related to the transfer or induction of Tregs in patients with CVD. First, transfer of Tregs is limited by low yields of amplification and purification in vitro. Second, insufficient evidence on the duration of the effects of transferred Tregs is available. Moreover, induced Tregs exhibit short survival durations after infusion, and Tregs are prone to loss of phenotype during in vitro manipulation and following reinfusion, which limits long-term treatment efficacy. For example, several studies demonstrated that induced Tregs may lose FOXP3 expression, the loss of which is accompanied by decreasing immunosuppressive ability and transformation to atherosclerotic T cells due to plasticity and instability in unique environments64,82. Third, the persistence of Tregs infusion may increase the risk of cancer and infection. Fourth, clinical studies are required to examine whether the therapeutic effects achieved in animal experiments are applicable to humans. Remarkably, because human atherosclerosis is a dynamic and complex disease, the same therapeutic strategies may have conflicting effects at different stages of atherosclerosis progression. For example, enhancing TGF-β function is not suitable for long-term treatment of CVD given its multiple targets and pleiotropic effects83. Whether IL-10 supplementation can be successfully applied to the treatment of atherosclerosis is unclear because IL-10 potentially increases the risk of cancer and lupus erythematosus84. Investigations into the precise therapeutic effects of TGF-β and IL-10 in atherosclerosis are warranted.
Several studies have demonstrated that atherosclerosis-associated antigen-induced antigen-specific Tregs reduce atherosclerosis, which can also cause antigen-dependent inflammatory responses and further aggravate the development of atherosclerosis33,85. Immune suppressive drugs, including corticosteroids and cytotoxic drugs, are effective in the treatment of atherosclerosis in animal models. However, it is not clear whether these therapies would also be beneficial in patients with cardiovascular disease. Additionally, various side effects, such as dyslipidemia, osteoporosis, growth suppression, immunosuppression, and recurrent infection, should be noted86.
Therefore, the development of safe and effective Treg-based therapies for the treatment of human atherosclerosis is challenging and meaningful. Investigations into atherosclerosis treatments should not only determine the effects of therapy on increasing Treg numbers but also enhance and stabilize Treg immune suppression functions in future clinical trials. Given recent studies on the generation of antigen-specific Treg cells via disease-associated antigens, the development of atherosclerosis-related vaccines based on atherosclerotic-associated antigens is an attractive option. It is encouraging that vaccine studies have shifted from animal experiments to clinical trials, which will lead to decisive outcomes in the treatment of atherosclerosis.
Author contribution
All authors participated in manuscript review.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No 81670401), the Scientific Research Innovation Program of post-graduate in Hu-nan Province (No CX2017B554), the Construct Program of the Key Discipline in Hu-nan Province (Basic Medicine Sciences in University of South China) and the Zhengxiang Scholar (Xiang-yang TANG) Program of the University of South China.
References
- 1.Jiang J, Mo ZC, Yin K, Zhao GJ, Lv YC, Ouyang XP, et al. Epigallocatechin-3-gallate prevents TNF-alpha-induced NF-kappaB activation thereby upregulating ABCA1 via the Nrf2/Keap1 pathway in macrophage foam cells. Int J Mol Med. 2012;29:946–56. doi: 10.3892/ijmm.2012.924. [DOI] [PubMed] [Google Scholar]
- 2.Yu XH, Jiang HL, Chen WJ, Yin K, Zhao GJ, Mo ZC, et al. Interleukin-18 and interleukin-12 together downregulate ATP-binding cassette transporter A1 expression through the interleukin-18R/nuclear factor-kappaB signaling pathway in THP-1 macrophage-derived foam cells. Circ J. 2012;76:1780–91. doi: 10.1253/circj.cj-11-1338. [DOI] [PubMed] [Google Scholar]
- 3.Ketelhuth DF, Hansson GK. Adaptive response of T and B cells in atherosclerosis. Circ Res. 2016;118:668–78. doi: 10.1161/CIRCRESAHA.115.306427. [DOI] [PubMed] [Google Scholar]
- 4.Bocian K, Kiernozek E, Domagala-Kulawik J, Korczak-Kowalska G, Stelmaszczyk-Emmel A, Drela N. Expanding diversity and common goal of regulatory T and B cells. I: Origin, phenotype, mechanisms. Arch Immunol Ther Exp (Warsz) 2017; doi:10.1007/s00005-017-0469-3. [DOI] [PMC free article] [PubMed]
- 5.Zhang H, Kong H, Zeng X, Guo L, Sun X, He S. Subsets of regulatory T cells and their roles in allergy. J Transl Med. 2014;12:125. doi: 10.1186/1479-5876-12-125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kosten IJ, Rustemeyer T. Generation, subsets and functions of inducible regulatory T cells. Antiinflamm Antiallergy Agents Med Chem. 2015;13:139–53. doi: 10.2174/1871523013666141126100019. [DOI] [PubMed] [Google Scholar]
- 7.Zhu ZF, Meng K, Zhong YC, Qi L, Mao XB, Yu KW, et al. Impaired circulating CD4+ LAP+ regulatory T cells in patients with acute coronary syndrome and its mechanistic study. PLoS One. 2014;9:e88775. doi: 10.1371/journal.pone.0088775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tselios K, Sarantopoulos A, Gkougkourelas I, Boura P. T regulatory cells: a promising new target in atherosclerosis. Crit Rev Immunol. 2014;34:389–97. doi: 10.1615/critrevimmunol.2014010802. [DOI] [PubMed] [Google Scholar]
- 9.Arce-Sillas A, Alvarez-Luquin DD, Tamaya-Dominguez B, Gomez-Fuentes S, Trejo-Garcia A, Melo-Salas M, et al. Regulatory T cells: molecular actions on effector cells in immune regulation. J Immunol Res. 2016;2016:1720827. doi: 10.1155/2016/1720827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Amin HZ, Sasaki N, Hirata KI. Regulatory T cell immunity in atherosclerosis. Acta Med Indones. 2017;49:63–8. [PubMed] [Google Scholar]
- 11.Foks AC, Lichtman AH, Kuiper J. Treating atherosclerosis with regulatory T cells. Arterioscler Thromb Vasc Biol. 2015;35:280–7. doi: 10.1161/ATVBAHA.114.303568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Komatsu N, Takayanagi H. Regulatory T cells in arthritis. Prog Mol Biol Transl Sci. 2015;136:207–15. doi: 10.1016/bs.pmbts.2015.07.021. [DOI] [PubMed] [Google Scholar]
- 13.Dasgupta A, Saxena R. Regulatory T cells: a review. Natl Med J India. 2012;25:341–51. [PubMed] [Google Scholar]
- 14.de Boer OJ, van der Meer JJ, Teeling P, van der Loos CM, van der Wal AC. Low numbers of FOXP3 positive regulatory T cells are present in all developmental stages of human atherosclerotic lesions. PLoS One. 2007;2:e779. doi: 10.1371/journal.pone.0000779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wang Z, Mao S, Zhan Z, Yu K, He C, Wang C. Effect of hyperlipidemia on Foxp3 expression in apolipoprotein E-knockout mice. J Cardiovasc Med (Hagerstown) 2014;15:273–9. doi: 10.2459/JCM.0b013e3283641b9c. [DOI] [PubMed] [Google Scholar]
- 16.Dietel B, Cicha I, Voskens CJ, Verhoeven E, Achenbach S, Garlichs CD. Decreased numbers of regulatory T cells are associated with human atherosclerotic lesion vulnerability and inversely correlate with infiltrated mature dendritic cells. Atherosclerosis. 2013;230:92–9. doi: 10.1016/j.atherosclerosis.2013.06.014. [DOI] [PubMed] [Google Scholar]
- 17.Jia L, Zhu L, Wang JZ, Wang XJ, Chen JZ, Song L, et al. Methylation of FOXP3 in regulatory T cells is related to the severity of coronary artery disease. Atherosclerosis. 2013;228:346–52. doi: 10.1016/j.atherosclerosis.2013.01.027. [DOI] [PubMed] [Google Scholar]
- 18.Niessner A, Weyand CM. Dendritic cells in atherosclerotic disease. Clin Immunol. 2010;134:25–32. doi: 10.1016/j.clim.2009.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rohm I, Atiskova Y, Drobnik S, Fritzenwanger M, Kretzschmar D, Pistulli R, et al. Decreased regulatory T cells in vulnerable atherosclerotic lesions: imbalance between pro- and anti-inflammatory cells in atherosclerosis. Mediators Inflamm. 2015;2015:364710. doi: 10.1155/2015/364710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu ZD, Wang L, Lu FH, Pan H, Zhao YX, Wang SJ, et al. Increased Th17 cell frequency concomitant with decreased Foxp3+ Treg cell frequency in the peripheral circulation of patients with carotid artery plaques. Inflamm Res. 2012;61:1155–65. doi: 10.1007/s00011-012-0510-2. [DOI] [PubMed] [Google Scholar]
- 21.Ait-Oufella H, Salomon BL, Potteaux S, Robertson AK, Gourdy P, Zoll J, et al. Natural regulatory T cells control the development of atherosclerosis in mice. Nat Med. 2006;12:178–80. doi: 10.1038/nm1343. [DOI] [PubMed] [Google Scholar]
- 22.Mor A, Luboshits G, Planer D, Keren G, George J. Altered status of CD4+CD25+ regulatory T cells in patients with acute coronary syndromes. Eur Heart J. 2006;27:2530–7. doi: 10.1093/eurheartj/ehl222. [DOI] [PubMed] [Google Scholar]
- 23.Zhang WC, Wang J, Shu YW, Tang TT, Zhu ZF, Xia N, et al. Impaired thymic export and increased apoptosis account for regulatory T cell defects in patients with non-ST segment elevation acute coronary syndrome. J Biol Chem. 2012;287:34157–66. doi: 10.1074/jbc.M112.382978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Butcher MJ, Filipowicz AR, Waseem TC, McGary CM, Crow KJ, Magilnick N, et al. Atherosclerosis-driven Treg plasticity results in formation of a dysfunctional subset of plastic IFNgamma+ Th1/Tregs. Circ Res. 2016;119:1190–203. doi: 10.1161/CIRCRESAHA.116.309764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Klingenberg R, Brokopp CE, Grives A, Courtier A, Jaguszewski M, Pasqual N, et al. Clonal restriction and predominance of regulatory T cells in coronary thrombi of patients with acute coronary syndromes. Eur Heart J. 2015;36:1041–8. doi: 10.1093/eurheartj/eht543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.George J, Schwartzenberg S, Medvedovsky D, Jonas M, Charach G, Afek A, et al. Regulatory T cells and IL-10 levels are reduced in patients with vulnerable coronary plaques. Atherosclerosis. 2012;222:519–23. doi: 10.1016/j.atherosclerosis.2012.03.016. [DOI] [PubMed] [Google Scholar]
- 27.Ait-Oufella H, Herbin O, Bouaziz JD, Binder CJ, Uyttenhove C, Laurans L, et al. B cell depletion reduces the development of atherosclerosis in mice. J Exp Med. 2010;207:1579–87. doi: 10.1084/jem.20100155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ghourbani GS, Andalib A, Hashemi M, Rezaei A. CD4+Foxp3+ Treg and its ICOS+ subsets in patients with myocardial infarction. Iran J Immunol. 2012;9:53–60. [PubMed] [Google Scholar]
- 29.Ammirati E, Moroni F, Magnoni M, Camici PG. The role of T and B cells in human atherosclerosis and atherothrombosis. Clin Exp Immunol. 2015;179:173–87. doi: 10.1111/cei.12477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Laurat E, Poirier B, Tupin E, Caligiuri G, Hansson GK, Bariety J, et al. In vivo downregulation of T helper cell 1 immune responses reduces atherogenesis in apolipoprotein E-knockout mice. Circulation. 2001;104:197–202. doi: 10.1161/01.cir.104.2.197. [DOI] [PubMed] [Google Scholar]
- 31.Potteaux S, Esposito B, van Oostrom O, Brun V, Ardouin P, Groux H, et al. Leukocyte-derived interleukin 10 is required for protection against atherosclerosis in low-density lipoprotein receptor knockout mice. Arterioscler Thromb Vasc Biol. 2004;24:1474–8. doi: 10.1161/01.ATV.0000134378.86443.cd. [DOI] [PubMed] [Google Scholar]
- 32.Mallat Z, Gojova A, Brun V, Esposito B, Fournier N, Cottrez F, et al. Induction of a regulatory T cell type 1 response reduces the development of atherosclerosis in apolipoprotein E-knockout mice. Circulation. 2003;108:1232–7. doi: 10.1161/01.CIR.0000089083.61317.A1. [DOI] [PubMed] [Google Scholar]
- 33.Robertson AK, Rudling M, Zhou X, Gorelik L, Flavell RA, Hansson GK. Disruption of TGF-beta signaling in T cells accelerates atherosclerosis. J Clin Invest. 2003;112:1342–50. doi: 10.1172/JCI18607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Frutkin AD, Otsuka G, Stempien-Otero A, Sesti C, Du L, Jaffe M, et al. TGF-[beta]1 limits plaque growth, stabilizes plaque structure, and prevents aortic dilation in apolipoprotein E-null mice. Arterioscler Thromb Vasc Biol. 2009;29:1251–7. doi: 10.1161/ATVBAHA.109.186593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Collison LW, Workman CJ, Kuo TT, Boyd K, Wang Y, Vignali KM, et al. The inhibitory cytokine IL-35 contributes to regulatory T-cell function. Nature. 2007;450:566–9. doi: 10.1038/nature06306. [DOI] [PubMed] [Google Scholar]
- 36.Lin Y, Huang Y, Lu Z, Luo C, Shi Y, Zeng Q, et al. Decreased plasma IL-35 levels are related to the left ventricular ejection fraction in coronary artery diseases. PLoS One. 2012;7:e52490. doi: 10.1371/journal.pone.0052490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sage AP, Mallat Z. Readapting the adaptive immune response - therapeutic strategies for atherosclerosis. Br J Pharmacol 2017; doi: 10.1111/bph.13700. [DOI] [PMC free article] [PubMed]
- 38.Kyaw T, Tay C, Khan A, Dumouchel V, Cao A, To K, et al. Conventional B2 B cell depletion ameliorates whereas its adoptive transfer aggravates atherosclerosis. J Immunol. 2010;185:4410–9. doi: 10.4049/jimmunol.1000033. [DOI] [PubMed] [Google Scholar]
- 39.Wang P, Zheng SG. Regulatory T cells and B cells: implication on autoimmune diseases. Int J Clin Exp Pathol. 2013;6:2668–74. [PMC free article] [PubMed] [Google Scholar]
- 40.Haddad Y, Lahoute C, Clement M, Laurans L, Metghalchi S, Zeboudj L, et al. The Dendritic Cell Receptor DNGR-1 Promotes the Development of Atherosclerosis in Mice. Circ Res. 2017;121:234–43. doi: 10.1161/CIRCRESAHA.117.310960. [DOI] [PubMed] [Google Scholar]
- 41.Gu P, Gao JF, D'Souza CA, Kowalczyk A, Chou KY, Zhang L. Trogocytosis of CD80 and CD86 by induced regulatory T cells. Cell Mol Immunol. 2012;9:136–46. doi: 10.1038/cmi.2011.62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dietrich T, Hucko T, Schneemann C, Neumann M, Menrad A, Willuda J, et al. Local delivery of IL-2 reduces atherosclerosis via expansion of regulatory T cells. Atherosclerosis. 2012;220:329–36. doi: 10.1016/j.atherosclerosis.2011.09.050. [DOI] [PubMed] [Google Scholar]
- 43.van Puijvelde GH, van Es T, van Wanrooij EJ, Habets KL, de Vos P, van der Zee R, et al. Induction of oral tolerance to HSP60 or an HSP60–peptide activates T cell regulation and reduces atherosclerosis. Arterioscler Thromb Vasc Biol. 2007;27:2677–83. doi: 10.1161/ATVBAHA.107.151274. [DOI] [PubMed] [Google Scholar]
- 44.van Puijvelde GH, Hauer AD, de Vos P, van den Heuvel R, van Herwijnen MJ, van der Zee R, et al. Induction of oral tolerance to oxidized low-density lipoprotein ameliorates atherosclerosis. Circulation. 2006;114:1968–76. doi: 10.1161/CIRCULATIONAHA.106.615609. [DOI] [PubMed] [Google Scholar]
- 45.Liang B, Workman C, Lee J, Chew C, Dale BM, Colonna L, et al. Regulatory T cells inhibit dendritic cells by lymphocyte activation gene-3 engagement of MHC class II. J Immunol. 2008;180:5916–26. doi: 10.4049/jimmunol.180.9.5916. [DOI] [PubMed] [Google Scholar]
- 46.Wilson HM. Macrophages heterogeneity in atherosclerosis - implications for therapy. J Cell Mol Med. 2010;14:2055–65. doi: 10.1111/j.1582-4934.2010.01121.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mo ZC, Xiao J, Tang SL, Ouyang XP, He PP, Lv YC, et al. Advanced oxidation protein products exacerbates lipid accumulation and atherosclerosis through downregulation of ATP-binding cassette transporter A1 and G1 expression in apolipoprotein E knockout mice. Circ J. 2014;78:2760–70. doi: 10.1253/circj.cj-14-0193. [DOI] [PubMed] [Google Scholar]
- 48.Peled M, Fisher EA. Dynamic aspects of macrophage polarization during atherosclerosis progression and regression. Front Immunol. 2014;5:579. doi: 10.3389/fimmu.2014.00579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lin J, Li M, Wang Z, He S, Ma X, Li D. The role of CD4+CD25+ regulatory T cells in macrophage-derived foam-cell formation. J Lipid Res. 2010;51:1208–17. doi: 10.1194/jlr.D000497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Meng X, Li W, Yang J, Zhang K, Qin W, An G, et al. Regulatory T cells prevent plaque disruption in apolipoprotein E-knockout mice. Int J Cardiol. 2013;168:2684–92. doi: 10.1016/j.ijcard.2013.03.026. [DOI] [PubMed] [Google Scholar]
- 51.Zhang M, Wu JF, Chen WJ, Tang SL, Mo ZC, Tang YY, et al. MicroRNA-27a/b regulates cellular cholesterol efflux, influx and esterification/hydrolysis in THP-1 macrophages. Atherosclerosis. 2014;234:54–64. doi: 10.1016/j.atherosclerosis.2014.02.008. [DOI] [PubMed] [Google Scholar]
- 52.Chen SG, Xiao J, Liu XH, Liu MM, Mo ZC, Yin K, et al. Ibrolipim increases ABCA1/G1 expression by the LXRalpha signaling pathway in THP-1 macrophage-derived foam cells. Acta Pharmacol Sin. 2010;31:1343–9. doi: 10.1038/aps.2010.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tang SL, Chen WJ, Yin K, Zhao GJ, Mo ZC, Lv YC, et al. PAPP-A negatively regulates ABCA1, ABCG1 and SR-B1 expression by inhibiting LXRalpha through the IGF-I-mediated signaling pathway. Atherosclerosis. 2012;222:344–54. doi: 10.1016/j.atherosclerosis.2012.03.005. [DOI] [PubMed] [Google Scholar]
- 54.Dong F, Mo Z, Eid W, Courtney KC, Zha X. Akt inhibition promotes ABCA1-mediated cholesterol efflux to ApoA-I through suppressing mTORC1. PLoS One. 2014;9:e113789. doi: 10.1371/journal.pone.0113789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Subramanian M, Thorp E, Hansson GK, Tabas I. Treg-mediated suppression of atherosclerosis requires MYD88 signaling in DCs. J Clin Invest. 2013;123:179–88. doi: 10.1172/JCI64617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Foks AC, Frodermann V, ter Borg M, Habets KL, Bot I, Zhao Y, et al. Differential effects of regulatory T cells on the initiation and regression of atherosclerosis. Atherosclerosis. 2011;218:53–60. doi: 10.1016/j.atherosclerosis.2011.04.029. [DOI] [PubMed] [Google Scholar]
- 57.Baigent C, Keech A, Kearney PM, Blackwell L, Buck G, Pollicino C, et al. Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet. 2005;366:1267–78. doi: 10.1016/S0140-6736(05)67394-1. [DOI] [PubMed] [Google Scholar]
- 58.Foks AC, Frodermann V, ter Borg M, Habets KL, Bot I, Zhao Y, et al. Differential effects of regulatory T cells on the initiation and regression of atherosclerosis. Atherosclerosis. 2011;218:53–60. doi: 10.1016/j.atherosclerosis.2011.04.029. [DOI] [PubMed] [Google Scholar]
- 59.Mallat Z, Gojova A, Brun V, Esposito B, Fournier N, Cottrez F, et al. Induction of a regulatory T cell type 1 response reduces the development of atherosclerosis in apolipoprotein E-knockout mice. Circulation. 2003;108:1232–37. doi: 10.1161/01.CIR.0000089083.61317.A1. [DOI] [PubMed] [Google Scholar]
- 60.Mor A, Planer D, Luboshits G, Afek A, Metzger S, Chajek-Shaul T, et al. Role of naturally occurring CD4+ CD25+ regulatory T cells in experimental atherosclerosis. Arterioscler Thromb Vasc Biol. 2007;27:893–900. doi: 10.1161/01.ATV.0000259365.31469.89. [DOI] [PubMed] [Google Scholar]
- 61.Trenado A, Fisson S, Braunberger E, Klatzmann D, Salomon BL, Cohen JL. Ex vivo selection of recipient-type alloantigen-specific CD4(+)CD25(+) immunoregulatory T cells for the control of graft-versus-host disease after allogeneic hematopoietic stem-cell transplantation. Transplantation. 2004;77:S32–34. doi: 10.1097/01.TP.0000106470.07410.CA. [DOI] [PubMed] [Google Scholar]
- 62.Wang ZX, Wang CQ, Li XY, Feng GK, Zhu HL, Ding Y, et al. Mesenchymal stem cells alleviate atherosclerosis by elevating number and function of CD4(+)CD25 (+)FOXP3 (+) regulatory T-cells and inhibiting macrophage foam cell formation. Mol Cell Biochem. 2015;400:163–72. doi: 10.1007/s11010-014-2272-3. [DOI] [PubMed] [Google Scholar]
- 63.Nilsson J, Lichtman A, Tedgui A. Atheroprotective immunity and cardiovascular disease: therapeutic opportunities and challenges. J Intern Med. 2015;278:507–19. doi: 10.1111/joim.12353. [DOI] [PubMed] [Google Scholar]
- 64.Bullenkamp J, Dinkla S, Kaski JC, Dumitriu IE. Targeting T cells to treat atherosclerosis: odyssey from bench to bedside. Eur Heart J Cardiovasc Pharmacother. 2016;2:194–9. doi: 10.1093/ehjcvp/pvw001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Yang K, Li D, Luo M, Hu Y. Generation of HSP60-specific regulatory T cell and effect on atherosclerosis. Cell Immunol. 2006;243:90–5. doi: 10.1016/j.cellimm.2007.01.002. [DOI] [PubMed] [Google Scholar]
- 66.Zhong Y, Wang X, Ji Q, Mao X, Tang H, Yi G, et al. CD4+LAP+ and CD4+CD25+Foxp3+ regulatory T cells induced by nasal oxidized low-density lipoprotein suppress effector T cells response and attenuate atherosclerosis in ApoE−/− mice. J Clin Immunol. 2012;32:1104–17. doi: 10.1007/s10875-012-9699-7. [DOI] [PubMed] [Google Scholar]
- 67.Herbin O, Ait-Oufella H, Yu W, Fredrikson GN, Aubier B, Perez N, et al. Regulatory T-cell response to apolipoprotein B100-derived peptides reduces the development and progression of atherosclerosis in mice. Arterioscler Thromb Vasc Biol. 2012;32:605–12. doi: 10.1161/ATVBAHA.111.242800. [DOI] [PubMed] [Google Scholar]
- 68.van Leuven SI, van Wijk DF, Volger OL, de Vries JP, van der Loos CM, de Kleijn DV, et al. Mycophenolate mofetil attenuates plaque inflammation in patients with symptomatic carotid artery stenosis. Atherosclerosis. 2010;211:231–6. doi: 10.1016/j.atherosclerosis.2010.01.043. [DOI] [PubMed] [Google Scholar]
- 69.Liu C, Chapman NM, Karmaus PW, Zeng H, Chi H. mTOR and metabolic regulation of conventional and regulatory T cells. J Leukoc Biol. 2015;97:837–47. doi: 10.1189/jlb.2RI0814-408R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Takeda M, Yamashita T, Sasaki N, Nakajima K, Kita T, Shinohara M, et al. Oral administration of an active form of vitamin D3 (calcitriol) decreases atherosclerosis in mice by inducing regulatory T cells and immature dendritic cells with tolerogenic functions. Arterioscler Thromb Vasc Biol. 2010;30:2495–503. doi: 10.1161/ATVBAHA.110.215459. [DOI] [PubMed] [Google Scholar]
- 71.Huang K, Li SQ, Wang WJ, Liu LS, Jiang YG, Feng PN, et al. Oral FTY720 administration induces immune tolerance and inhibits early development of atherosclerosis in apolipoprotein E-deficient mice. Int J Immunopathol Pharmacol. 2012;25:397–406. doi: 10.1177/039463201202500209. [DOI] [PubMed] [Google Scholar]
- 72.Tian Y, Yuan Z, Liu Y, Liu W, Zhang W, Xue J, et al. Pioglitazone modulates the balance of effector and regulatory T cells in apolipoprotein E deficient mice. Nutr Metab Cardiovasc Dis. 2011;21:25–32. doi: 10.1016/j.numecd.2009.07.010. [DOI] [PubMed] [Google Scholar]
- 73.Maganto-Garcia E, Tarrio ML, Grabie N, Bu DX, Lichtman AH. Dynamic changes in regulatory T cells are linked to levels of diet-induced hypercholesterolemia. Circulation. 2011;124:185–95. doi: 10.1161/CIRCULATIONAHA.110.006411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Meng X, Zhang K, Li J, Dong M, Yang J, An G, et al. Statins induce the accumulation of regulatory T cells in atherosclerotic plaque. Mol Med. 2012;18:598–605. doi: 10.2119/molmed.2011.00471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Jiagang D, Li C, Wang H, Hao E, Du Z, Bao C, et al. Amygdalin mediates relieved atherosclerosis in apolipoprotein E deficient mice through the induction of regulatory T cells. Biochem Biophys Res Commun. 2011;411:523–9. doi: 10.1016/j.bbrc.2011.06.162. [DOI] [PubMed] [Google Scholar]
- 76.Klatzmann D, Abbas AK. The promise of low-dose interleukin-2 therapy for autoimmune and inflammatory diseases. Nat Rev Immunol. 2015;15:283–94. doi: 10.1038/nri3823. [DOI] [PubMed] [Google Scholar]
- 77.Kita T, Yamashita T, Sasaki N, Kasahara K, Sasaki Y, Yodoi K, et al. Regression of atherosclerosis with anti-CD3 antibody via augmenting a regulatory T-cell response in mice. Cardiovasc Res. 2014;102:107–17. doi: 10.1093/cvr/cvu002. [DOI] [PubMed] [Google Scholar]
- 78.Kasahara K, Sasaki N, Yamashita T, Kita T, Yodoi K, Sasaki Y, et al. CD3 antibody and IL-2 complex combination therapy inhibits atherosclerosis by augmenting a regulatory immune response. J Am Heart Assoc. 2014;3:e719. doi: 10.1161/JAHA.113.000719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Worthington JJ, Kelly A, Smedley C, Bauche D, Campbell S, Marie JC, et al. Integrin alphavbeta8-mediated TGF-beta activation by effector regulatory T cells is essential for suppression of T-cell-mediated inflammation. Immunity. 2015;42:903–15. doi: 10.1016/j.immuni.2015.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Uchiyama R, Hasegawa H, Kameda Y, Ueda K, Kobayashi Y, Komuro I, et al. Role of regulatory T cells in atheroprotective effects of granulocyte colony-stimulating factor. J Mol Cell Cardiol. 2012;52:1038–47. doi: 10.1016/j.yjmcc.2011.12.016. [DOI] [PubMed] [Google Scholar]
- 81.Sasaki N, Yamashita T, Kasahara K, Fukunaga A, Yamaguchi T, Emoto T, et al. UVB exposure prevents atherosclerosis by regulating immunoinflammatory responses. Arterioscler Thromb Vasc Biol. 2017;37:66–74. doi: 10.1161/ATVBAHA.116.308063. [DOI] [PubMed] [Google Scholar]
- 82.Sakaguchi S, Vignali DA, Rudensky AY, Niec RE, Waldmann H. The plasticity and stability of regulatory T cells. Nat Rev Immunol. 2013;13:461–7. doi: 10.1038/nri3464. [DOI] [PubMed] [Google Scholar]
- 83.Flavell RA, Sanjabi S, Wrzesinski SH, Licona-Limon P. The polarization of immune cells in the tumour environment by TGFbeta. Nat Rev Immunol. 2010;10:554–67. doi: 10.1038/nri2808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Asadullah K, Sterry W, Volk HD. Interleukin-10 therapy--review of a new approach. Pharmacol Rev. 2003;55:241–69. doi: 10.1124/pr.55.2.4. [DOI] [PubMed] [Google Scholar]
- 85.Xu XH, Shah PK, Faure E, Equils O, Thomas L, Fishbein MC, et al. Toll-like receptor-4 is expressed by macrophages in murine and human lipid-rich atherosclerotic plaques and upregulated by oxidized LDL. Circulation. 2001;104:3103–8. doi: 10.1161/hc5001.100631. [DOI] [PubMed] [Google Scholar]
- 86.Buchman AL. Side effects of corticosteroid therapy. J Clin Gastroenterol. 2001;33:289–94. doi: 10.1097/00004836-200110000-00006. [DOI] [PubMed] [Google Scholar]


