Abstract
Efficient gene delivery technologies play an essential role in the gene functional analyses that are necessary for basic and applied researches. Mosquitoes are ubiquitous insects, responsible for transmitting many deadly arboviruses causing millions of human deaths every year. The lack of efficient and flexible gene delivery strategies in mosquitoes are among the major hurdles for the study of mosquito biology and mosquito-pathogen interactions. We found that Autographa californica multiple nucleopolyhedrovirus (AcMNPV), the type baculovirus species, can efficiently transduce mosquito cells without viral propagation, allowing high level gene expression upon inducement by suitable promoters without obvious negative effects on cell propagation and viability. AcMNPV transduces into several mosquito cell types, efficiently than in commonly used mammalian cell lines and classical plasmid DNA transfection approaches. We demonstrated the application of this system by expressing influenza virus neuraminidase (NA) into mosquito hosts. Moreover, AcMNPV can transduce both larvae and adults of essentially all blood-sucking mosquito genera, resulting in bright fluorescence in insect bodies with little or no tissue barriers. Our experiments establish baculovirus as a convenient and powerful gene delivery vector in vitro and in vivo that will greatly benefit research into mosquito gene regulation, development and the study of mosquito-borne viruses.
Introduction
Mosquitoes are primary vectors for the transmission of many human diseases such as chikungunya (CHIKV), dengue (DENV), filarial, malaria, yellow fever, and the recent Zika virus (ZIKV) outbreaks, all of which continue to pose public health risks and contribute to significant global economic losses1,2. Aedes, Culex, and Anopheles mosquito species are the most effective transmitters of deadly viruses and parasites to humans3. According to World Health Organization’s (WHO) latest report (April, 2017), malaria alone caused 429,000 deaths worldwide in 2015 and at least one million human deaths every year caused by the mosquito-borne diseases have been reported4. Despite continuous study of mosquito gene regulation and efforts to prevent mosquito-borne diseases, lack of efficient and flexible gene delivery approaches hinder investigations into virus/host interactions and mosquito biology. An efficient gene delivery system across different mosquito species into cells, larvae, and different organs of adults would obviously be an indispensable tool for such studies and have many other crucial applications in biological research.
Normally, germ line transformation technique is used to construct the stable transgenic mosquito lines to study the biological function of desired genes in the mosquitoes. However, this is a time-consuming technique and has been successful only in few mosquito species5,6. Classical in vitro plasmid transfection is a faster approach for expressing target genes, but it is associated with lower efficiency and reagent toxicity issues. Infections by viral vectors have emerged as the dominant method of choice to deliver genes in gene regulation studies. Mosquito densovirus (MDV)-mediated gene delivery has recently been developed7. However, MDVs are replication-competent and species-specific8–10, and a further drawback is the packaging limitations of DNA cargo size in MDV genomes7. Therefore, a better strategy for gene transfer in mosquitoes is greatly needed.
Baculovirus is a versatile tool for agricultural and biotechnological applications. The baculovirus expression vector system (BEVs) derived from this virus is popular for the production of engineered proteins11. This system can produce proteins with high yield and proper post-translational modifications that are suitable for various applications12. Autographa californica multiple nucleopolyhedrovirus (AcMNPV) is the type baculovirus species, which infects only lepidopteran insects and cell lines. This virus has a double-stranded, closed-circular DNA genome of 134 kb with a coding capacity of over 154 polypeptides13. In 1995, AcMNPV was found possible to transfer genes into mammalian cells and efficiently expressed by a promoter functional in the target cells6,14. It has since been successfully exploited in gene transfer applications for many mammalian cell lines, primary cells, progenitor cells, induced pluripotent (iPS) and stem cells, and is well-known as BacMam system15,16. Prior to these discoveries in the mammalian system, one study reported that AcMNPV may replicate at very low levels in Aedes aegypti mosquito cell lines, which could only be detected by a very sensitive radioisotope labeling of the viral genome17. However, no any further studies on AcMNPV in mosquitoes have been reported. Another study showed that Culex nigripalpus nucleopolyhedrovirus (CuniNPV) belongs to delta baculovirus is a pathogen of Culex mosquitoes, which are vectors of West Nile virus and other forms of encephalitis18. However, CuniNPV could only infect the genus Culex and it is not well characterized compared to AcMNPV. Also, CuniNPV is an infectious virus, and thus not suitable for the purposes of gene transfer to study gene regulations and/or the developments of the cells, larvae, and adults without significantly affecting the normal physiology of the mosquitoes. Unlike MDVs, the transgene capacity of AcMNPV is very large, probably extending beyond 100 kbp19. Baculovirus transduction into mammalian cells has been widely used to express genes driven by the mammalian promoters14,20. Since baculovirus is not a mammalian pathogen, it has been successfully adapted into a safe vector for gene delivery into mammalian cells, both in vitro and in vivo21. Baculovirus has also served as an efficient vehicle for gene transfer into Drosophila S2 cells, but not into larvae or adults22.
Here, we report the development of a baculovirus vector system for efficient gene delivery into mosquito (BacMos). This is a simple and efficient tool yet for introducing foreign genes into the cells, larvae, and adults of all tested mosquito genus that are vectors of important viral diseases. Several lines of evidence suggest that baculovirus do not replicate but efficiently transduce mosquito C6/36 cells. We first examined and compared the strength of various baculovirus-incorporated promoters in C6/36 cells, and further showed strong baculovirus-mediated foreign gene expression in larvae and in almost all adult mosquito tissues without an obvious tissue barrier. This BacMos system combines simplicity and flexibility, and serves as an alternative to existing inefficient or tedious approaches. Therefore, we believe BacMos has the potential to become a powerful tool for gene regulation analysis, which may significantly assist in advancing studies of mosquito biology and pathogen interactions.
Results
Baculovirus transduction into mosquito C6/36 cells
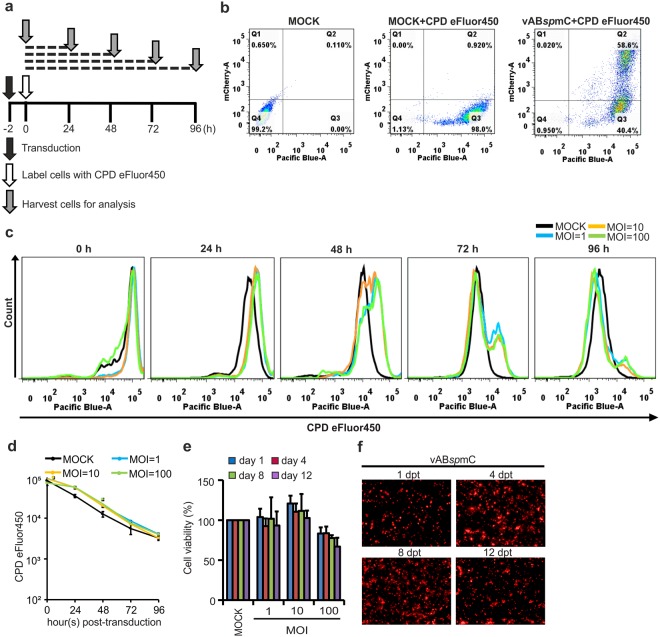
AcMNPV is currently the most commonly used baculovirus for foreign gene expression. To examine if AcMNPV can enter into mosquito host C6/36 cells and deliver an effective gene expression cassette, we utilized recombinant baculovirus (vABspmC) containing mCherry under the control of Heliothis zea nudivirus 1 (HzNV-1) virus early gene pag1 promoter23 and sv40 promoter24 (dual-promoter) that was originally designed for mammalian and insect cell systems respectively (Fig. 1a). We transduced C6/36 cells with vABspmC at MOI = 1, 10 and 100 and found mCherry fluorescence as early as 12 h (data not shown), and significant dose-dependent fluorescence intensity was detected at 48 h post-transduction (Fig. 1b). Transduced cells were then collected and analyzed by flow cytometry to determine transduction efficiency. Gating of mCherry-positive cells and quantification revealed that transduction efficiencies of 60–65% could be routinely achieved at MOI = 1, and efficiency increased to 95–100% at MOI = 100 (Fig. 1c,d). We think that mCherry could have been driven by the pag1 promoter rather than the sv40 promoter because thus far there is no evidence that the sv40 promoter is functional in mosquito hosts. It is well known that baculovirus enters insect or mammalian cells through surface GP64 proteins25. To assess GP64-mediated cell entry, baculovirus was treated with neutralizing or non-neutralizing GP64 antibodies before being transduced into C6/36 cells. We found that baculovirus treated with GP64-neutralizing antibody did not show mCherry fluorescence at 48 h post-transduction compared to a negative control (Fig. 1e), indicating that baculovirus indeed enters mosquito hosts through the GP64 surface protein. Together, these results suggest that baculovirus can successfully enter and drive transgene expression in mosquito C6/36 cells.
Figure 1.
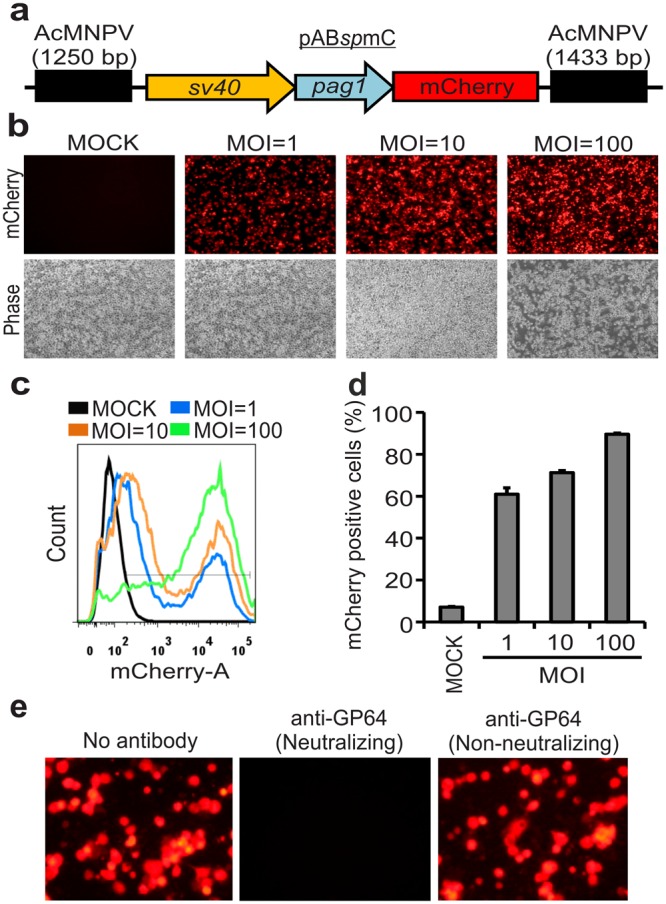
Baculovirus transduction into mosquito C6/36 cells. (a) Schematic representation of a composite expression vector for generating recombinant baculovirus. The composite transfer vector contains the sv40 and pag1 promoters, which are designed to express mCherry in mammalian and insect cells, respectively. (b) Dose-dependent entry of baculovirus. C6/36 cells were transduced with different concentrations (MOI = 1, 10, and 100) of recombinant vABspmC baculovirus. (c) Gating of mCherry-positive cells by flow cytometry, shown the efficiency of baculovirus transduction using different MOIs. (d) Quantification of mCherry-positive cells to indicate transduction efficiency. (e) GP64-mediated entry of baculovirus. C6/36 cells were treated with a baculovirus-antibody mixture (neutralizing or non-neutralizing antibodies against GP64 protein). The mCherry fluorescence images were taken at 48 h post-transduction in both panel’s (b and e). Data represent the average ± SD (standard deviation) of three biological replicates (n = 3).
Analysis of cellular proliferation and cell viability in response to baculovirus transduction
In general, the baculovirus undergoes a lytic cycle after virus infection in lepidopteran cells such as Sf9, Sf21, and High Five cells. This cycle significantly affects transcription and translation of the host genes26,27. To investigate whether C6/36 cells undergo the same lytic pathway and suffer the same cell proliferation effects in response to baculovirus, cell division was monitored by cell proliferation dye (CPD) eFluor450 dilution28. Mosquito C6/36 cells were exposed to the vABspmC virus at various MOIs, labeled with 10 µM CPD eFluor450 and cultured for up to 4 days (Fig. 2a). The cells were harvested at every 24 h interval and subjected to flow cytometry analysis to measure the dye intensities in the transduced cells (Q2 population) and mock cells (Q3 population) (Fig. 2b). We found baculovirus transduced cells showed slightly higher dye intensities at 24 and 48 h points but there is a gradual shift in the peak towards the left at later time-points reaching comparable dye intensities to mock at 96 h at all the MOIs (Fig. 2c). This data suggests that baculovirus does not significantly affect the kinetics of C6/36 cell division although there was slightly delayed in the early time points (Fig. 2d). Cell viability was analyzed at 4-day intervals by adding 10% v/v AlamarBlue to determine cell metabolic activity. There were no apparent toxicities in C6/36 cells at MOI = 1 and MOI = 10 for all exposure timeframes, but there was a mild negative effect for MOI = 100 at 12 days post-transduction (Fig. 2e). We also examined whether C6/36 cells support persistent gene expression and simultaneously analyzed the toxicity of baculovirus to C6/36 cells for up to 12 days. The mCherry expression increased as a function of time up to 8 days post baculovirus transduction before reaching saturation (Fig. 2f). Thus, BacMos expression system does not involve the baculovirus lytic cycle, no significant effect on cell proliferation kinetics and C6/36 cells can sustain baculovirus transduction for long periods of time.
Figure 2.
Effect of baculovirus transduction on cellular proliferation and toxicity. (a) Schematic diagram showing the experimental design. (b) Flow cytometry analysis of cells labeled with CPD eFluor450 dye. Representative dot plots of mock, mock + CPD eFluor450 and vABspmC + CPD eFluor450 (MOI = 1) for 24 h were shown as an example for gating of populations. (c) Representative histograms of CPD eFluor450 labeling intensity in baculovirus transduced cells (Q2 population only) and MOCK cell (Q3 population only) at the indicated time points. (d) Analysis of mean CPD eFluor450 labeling intensity in mock and baculovirus transduced cells with different MOIs at various time points. (e) Cell viability. Toxicity of baculovirus transduction into C6/36 cells at varying dosages, measured every 4 days by AlamarBlue cell viability assay. (f) Persistent baculovirus-mediated gene expression. C6/36 cells were transduced with baculovirus at MOI = 1 and expression of fluorescent protein was analyzed over several days. The mCherry fluorescence images at various time-points were taken by fluorescence microscopy. Data shown represent the averages of three biological replicates, with error bars showing standard deviations.
Replication analysis of baculovirus in mosquito C6/36 cells
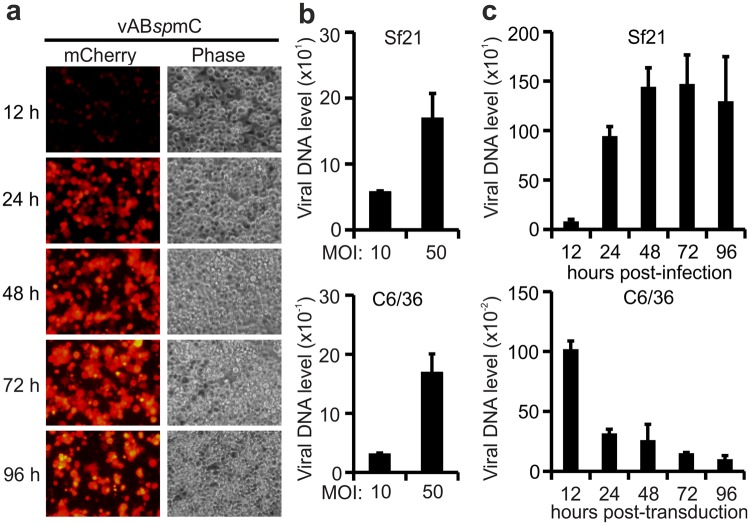
Insect Sf21 cells are a natural host and permissive for baculovirus infection and DNA replication29. Therefore, we were curious to investigate whether baculovirus could replicate in mosquito C6/36 cells. To this end, we infected Sf21 or transduced C6/36 cells with vABspmC simultaneously. Time-course mCherry fluorescence images of C6/36 cells transduced with vABspmC at MOI = 10 are shown in Fig. 3a. Then, we examined baculovirus entry efficiency in both hosts at MOI = 10 and MOI = 50. Relative amounts of intracellular viral DNA at respective MOIs 2 h post-infection/transduction were found to be similar in both hosts, suggesting that baculovirus enters with similar efficiency into C6/36 and Sf21 cells (Fig. 3b). Next, we investigated the kinetics of intracellular viral DNA accumulation as a function of time and found a gradual increase of intracellular viral DNA from 12 h to 48 h post-infection in Sf21 cells, and saturation thereafter up to 96 h post-infection. In contrast, viral DNA gradually decreased as a function of time in C6/36 cells from 12 h to 96 h post-transduction, suggesting that baculovirus likely does not replicate in mosquito C6/36 cells, at least not replicate in a detectable level (Fig. 3c).
Figure 3.
Replication analysis of baculovirus in C6/36 cells. (a) mCherry fluorescence images of baculovirus transduction. C6/36 cells were transduced with vABspmC at MOI = 10 and mCherry fluorescence images were captured by fluorescence microscopy at various time-points as shown. (b) Baculovirus entry efficiency. Baculovirus was transduced into C6/36 cells or infected with MOI = 10 and MOI = 50 for 2 h into Sf21 cells before relative entry efficiencies were quantified by qPCR. (c) Replication efficiency of baculovirus. C6/36 or Sf21 cells were transduced or infected with MOI = 10, harvested at various time-points, and intracellular viral DNA was quantified by qPCR. Amounts of viral DNA at all time-points were normalized against an internal gene control—GAPDH for Sf21 or Actin for C6/36—and further normalized against viral DNA amount after 2 h post-infection or post-transduction. Data represent the average ± SD of three biological replicates (n = 3).
It is well known that baculovirus is replication-incompetent but capable of transducing into various mammalian cells, and the BacMam system is well established for popular applications in these cells30. We wished to compare baculovirus transduction efficiency between C6/36 (BacMos) and mammalian (BacMam) hosts. Mammalian cells are usually transduced with baculovirus at high MOIs of more than several hundred to several thousand19. We transduced C6/36 and commonly used mammalian Vero-E631 and HEK-293T32,33 cells with MOIs of 1, 10 and 100, and then took mCherry fluorescence images at 48 h post-transduction (Supplementary Fig. 1a). Flow cytometry analysis showed that percentages of mCherry-positive cells increased with increasing viral dose for all three cell lines. The transduction efficiency was much higher in C6/36 cells compared to mammalian cell lines at all tested MOIs (Supplementary Fig. 1b), suggesting that mosquito C6/36 hosts are more permissive for baculovirus compared to mammalian hosts.
The most commonly used method to express foreign genes in mosquito hosts is the classical plasmid transfection approach. We compared transgene expression levels by baculovirus transduction versus plasmid DNA transfections. In order to do this, we transduced C6/36 cells with several doses of vABspmC (MOI = 1, 2 or 3) or used the plasmid pABspmC (10 kb) (DNA = 1 µg, 2 µg or 3 µg) originally inserted into the recombinant baculovirus for plasmid-based transfections (Supplementary Fig. 2a). Flow cytometry revealed that plasmid transfection efficiencies were around 15–20%, i.e., much lower than the baculovirus transduction efficiencies of around 60% at MOI = 1 (Supplementary Fig. 2b). Furthermore, relative transgene expression levels were significantly higher by baculovirus transduction than plasmid DNA transfection (Supplementary Fig. 2c).
Functional analysis of various baculovirus-incorporated promoters and their efficiencies in C6/36 cells
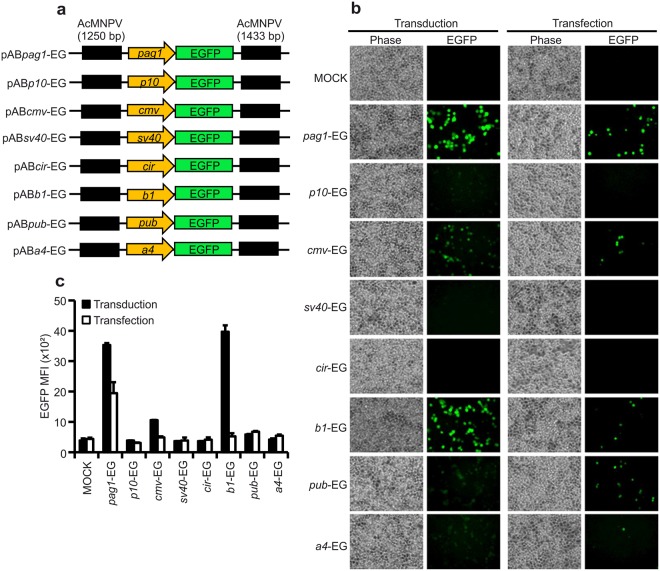
Next, we tested the functionality and efficiency of various promoters by inserting their DNA sequences into the baculovirus genome. As reported earlier34, mosquito densovirus expression cassettes suffer from a size limitation for inserts. However, baculovirus does not have such size limitations so we constructed transfer vectors with different promoter sequences driving EGFP expression, as shown in Fig. 4a. After packaging of each transfer vector into baculovirus, C6/36 cells were transduced at MOI = 1 so that the majority of cells contained one or fewer viral genomic copies per cell. For comparison, C6/36 cells were also transfected with 500 ng plasmid of the same transfer vector so that each cell was transfected with an average of 1.15 × 105 copies of plasmid DNA. EGFP images were taken at 48 h post-transduction (Fig. 4b).
Figure 4.
Efficiency of baculovirus-incorporated promoters in C6/36 cells. (a) Schematic representation of transfer vectors used to generate the recombinant baculoviruses expressing EGFP driven by several baculovirus, mammalian viral, and mosquito host promoters from the following genes: pag1, a HzNV-1 viral early expressing gene23; p10, baculovirus late gene46; cmv, cytomegalovirus47; sv40, simian virus 4024; cir, chimeric internal ribosome entry site (IRES) of RhPV virus and EV71 virus48,49; b1, cecropin b1 gene50; pub, polyubiquitin gene42 and a4, defensin a4 gene51. (b) Fluorescence images. C6/36 cells were transduced with recombinant viruses expressing EGFP at an MOI = 1 or transfected with 500 ng of the recombinant plasmid DNA constructs used to generate recombinant baculoviruses. The images were taken fluorescence microscopy at 48 h post-transduction. (c) Flow cytometry analysis of EGFP fluorescence driven by the promoters of interest. Recombinant baculovirus-transduced or plasmid DNA-transfected C6/36 cells were collected after 48 h and mean EGFP fluorescence intensities were measured by flow cytometry to determine promoter efficiency. Data represent the average ± SD of three biological replicates (n = 3).
We evaluated the strength of each promoter in C6/36 cells by quantifying mean fluorescence intensities of EGFP-positive cells by flow cytometry. We found pag1 and b1 promoters are consistently stronger among others, visually looked similar but the quantification data showed that b1 driven EGFP expression was slightly better than pag1 driven EGFP expression. In contrast, the p10, sv40, cir, pub, and a4 promoters exhibited little or no fluorescence, and the cmv promoter only showed minimal activity. In our plasmid transfection system, pag1 promoter showed better activity while the activity of other promoters remained basal or below the detection limit. Furthermore, under plasmid transfection, the p10, sv40, cir, and a4 promoters exhibited similar results to baculovirus transduction (i.e., no fluorescence), and the cmv promoter only presented basal activity. However, the pub promoter functions better by plasmid transfection than baculovirus transduction. As expected, the baculovirus very late gene, p10 promoter was not functional, further supporting that baculovirus is not a replicative virus in mosquito C6/36 cells (Fig. 4c).
We then introduced b1 promoter-EGFP sequence into an established bi-cistronic baculovirus transfer vector35 containing 5′UTR internal ribosome entry site (IRES) of the Rhopalosiphum padi virus (RhPV) fused to EGFP. The b1-EGFP is reserved as a site for future foreign gene insertion in the BacMos to be expressed in mosquito hosts, and IRES-EGFP will serve as reporter protein in insect Sf21 host and facilitated the isolation of the recombinant AcMNPV. The resultant baculovirus, vBacb1EG-irEG showed significant enhancement in the EGFP expression compared to vABb1EG virus in C6/36 cells (Supplementary Fig. 3a,b). Furthermore, we transduced vBacb1EG-irEG in other mosquito cell lines derived from A. aegypti (CCL-125) and A. pseudoscutellaris (AP-61) in addition to C6/36 cells. We found C6/36 has better transduction efficiency compared to CCL-125 and AP-61 cell lines transduced with different viral doses (Supplementary Fig. 3c,d) indicating baculovirus transduction is not cell-type specific. Our results demonstrate that the baculovirus-inserted b1 promoter is the strongest among the tested promoters in mosquito C6/36 cells.
Application of baculovirus mediated gene expression in mosquito C6/36 cell line
Besides establishing the BacMos system using florescent reporter genes (mCherry and EGFP), we aimed to demonstrate the application of BacMos system by expressing the gene of interest which has functional assay that can be performed. In this study, we wished to express neuraminidase (NA) of influenza A virus H5N3 (NA3) and examine its enzymatic activity from the cell lysates. C6/36 cells were transduced with vABb1p10-empty or vABb1p10-NA3 baculovirus expressing b1 promoter driven influenza NA3 (Fig. 5a). The EGFP florescence pictures revealed that these two viruses has similar transduction efficiency (Fig. 5b) and the cell lysates harvested at 48 h post-transduction were subjected to SDS-PAGE and Western blotting to examine the NA3 expression (Fig. 5c). The cell lysates were tested in florescence based assay that uses 4-methyl umbelliferone N-acetyl neuraminic acid (4-MUNANA) as a substrate to examine the neuraminidase activity (NA)36. We observed cell lysates from baculovirus mediated expression of NA3 showed a strong enzymatic activity in converting the substrate to the final products almost comparable to the activity resulted by the purified NA (H5N9 strain) protein, while the readings remained at basal level for the mock or vABb1p10-empty virus transduced cell lysates. We also ensured the specificity of neuraminidase functions of NA by treating with well-known drug inhibitors such as Oseltamivir or Zanamivir (10 µM) which inhibited NA activity (Fig. 5d). This data demonstrates the application of baculovirus mediated gene expression for the desired genes in the mosquito model systems.
Figure 5.
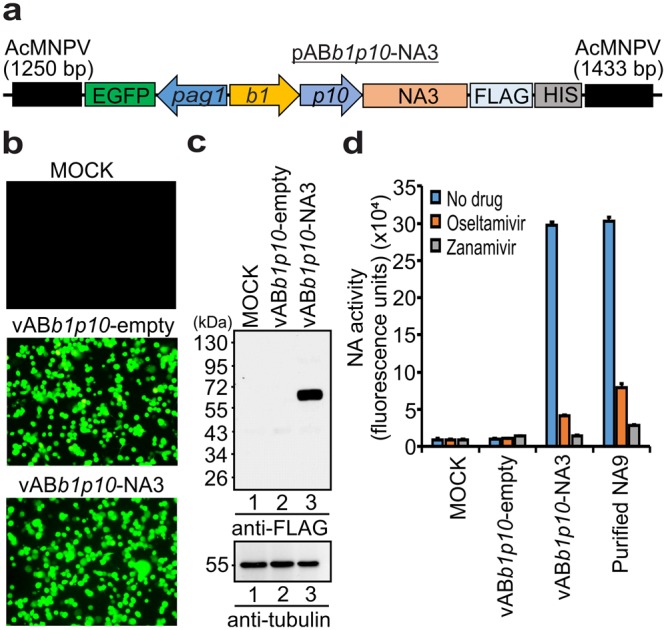
Enzymatic analysis of influenza virus NA expressed in mosquito C6/36 cells by recombinant baculovirus. (a) Schematic representation of a pABb1p10-NA3 transfer vector for generating recombinant baculovirus. The transfer vector contains the EGFP reporter gene driven by the pag1 promoter; NA (H5N3) gene under the control of dual host b1 and p10 promoters for mosquito C6/36 and insect Sf21 cells; FLAG and HIS tags were inserted at c-terminal; AcMNPV lateral ends flanking the expression cassette for homologous recombination. (b) C6/36 cells were transduced with vABb1p10-empty (virus backbone without NA3) and vABb1p10-NA3 baculovirus and the EGFP florescent pictures were taken at 48 h. (c) Western blot analysis. The cell lysates of MOCK, vABb1p10-empty and vABb1p10-NA3 were harvested at 48 h, analyzed by SDS-PAGE, followed by Western detection of NA3 protein in C6/36 cells. (d) Neuraminidase activity and Neuraminidase inhibition assays: The cell lysates of MOCK, vABb1p10-empty and vABb1p10-NA3 were mixed with or without (10 µM) NA inhibitors (Oseltamivir or Zanamivir) in the presence of 4-MUNANA substrate. Purified NA9 protein (H5N9 strain) was used as a positive control in the experiment. The experiment was performed three times and the data shown is the representative of one independent experiment done in triplicate. The error bars indicate the standard deviations.
Baculovirus-mediated gene expression in larvae and adults of various mosquito species
Next, we examined the transduction ability of baculovirus into larvae of different mosquito species. A. aegypti (the yellow fever mosquito) and A. albopictus (Asian tiger mosquito or forest mosquito) are responsible for the transmission of various potentially fatal viruses in humans including dengue, chikungunya, zika, yellow fever, and Mayaro, as well as several filarial nematodes such as Dirofilaria immitis and other diseases. Culex (Culex) tritaeniorhynchus is the main vector of Japanese encephalitis and Anopheles sinensis transmits malaria, lymphatic filariasis3. We microinjected a dose of 1 × 105 PFU recombinant vBacb1EG-irEG baculovirus into larvae of these four mosquito species and detected EGFP expression 2 days post-transduction. We observed strong EGFP expression in A. aegypti and A. albopictus, but weak expression in C. tritaeniorhynchus and A. sinensis (Fig. 6a).
Figure 6.
Analysis of in vivo baculovirus transduction of mosquitoes. (a) Transduction of baculovirus into mosquito larvae. Four different mosquito species (A. aegypti, A. albopictus, C. tritaeniorhynchus, and A. sinensis) were microinjected with 1 × 105 PFU of vBacb1EG-irEG baculovirus. EGFP expression was visualized by fluorescence microscopy at 2 days post-transduction. (b) Dose-dependent expression of a baculovirus-mediated transgene in adult mosquitoes. Four different mosquito species were microinjected intra-thoracically with several doses (1 × 103, 104 or 105 PFU) of vBacb1EG-irEG baculovirus. EGFP expression was visualized by fluorescence microscopy at 6 days post-transduction. (c) Transduction of baculovirus into adult mosquitoes. Four different mosquito species were microinjected intra-thoracically with 1 × 105 PFU of vBacb1EG-irEG baculovirus. EGFP expression was visualized by fluorescence microscopy as a function of time over several days as shown.
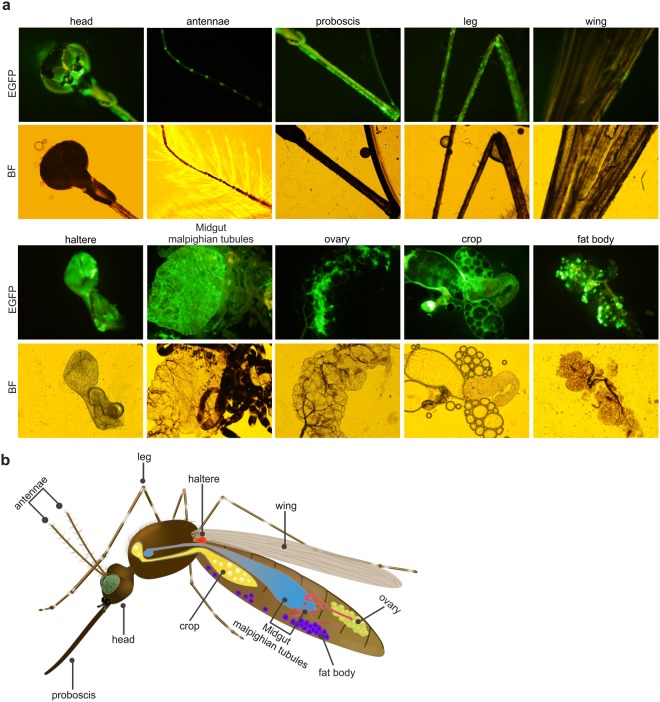
We were also curious to examine the transduction ability of baculovirus into adult mosquitoes, so we assessed dose-dependent expression by intrathoracically microinjecting the same recombinant baculovirus (vBacb1EG-irEG). Interestingly, baculovirus-mediated EGFP expression in adult mosquitoes was higher with increasing viral dosages at 6 days post-microinjection. Again, we observed relatively weaker EGFP expression in A. sinensis compared to other three mosquito species (Fig. 6b). We also examined whether or not baculovirus replicates in vivo by performing qPCR on total genomic DNA isolated from transduced adult mosquitoes. We found that baculovirus does not replicate in adult mosquitoes (data not shown), corroborating our in vitro experiments. Next, we conducted a time-course experiment to repeatedly examine microinjected mosquitoes at regular time-points. EGFP expression was weak at 1 day post-injection, but increased thereafter throughout the experiment until 12 days post-injection (Fig. 6c). To determine the tissue tropism of baculovirus-mediated foreign gene expression, we dissected the transduced mosquitoes. EGFP expression was strongly observed in most of the tissues including head, proboscis, leg, haltere, midgut, ovary, crop, and fat body, but somewhat weak expression was observed in the Malpighian tubules, antennae, and wings (Fig. 7a). The schematic representation of the respective adult mosquito tissues are indicated (Fig. 7b). These results suggest that baculovirus can successfully transduce and express transgenes in the larvae and adults of the mosquito species tested here with little or no tissue barriers.
Figure 7.
Tissue tropism of baculovirus in adult A. aegypti. (a) Dissection of baculovirus transduced adult mosquito. A. aegypti adult mosquitoes were microinjected intra-thoracically with 1 × 105 PFU of vBacb1EG-irEG baculovirus and observed at 15 days post-transduction. EGFP fluorescence was observed in head, antennae, proboscis, leg, wing, haltere, midgut Malpighian tubules, ovary, crop, and fat body tissues of adult mosquitoes. EGFP: green fluorescence, and BF: bright field images. (b) Schematic representation of adult mosquito tissues. For simplicity, cartoon of adult mosquito showing various tissues representing respective images of (a) is shown.
Discussion
Mosquitoes are perhaps the most deadly animals for humans, transmitting disease to millions annually. Efficient gene delivery technologies are essential to advance studies of mosquito/pathogen interactions and mosquito biology. In this study, we have developed baculovirus as an efficient tool for gene delivery in mosquitoes.
Baculovirus has been widely used in various biotechnological applications such as protein production, gene transfer and also as a microbial insecticide37,38. Baculovirus was demonstrated possible for gene delivery into hepatocyte since the discovery of baculovirus as a gene delivery tool into non-host mammalian cell system6,14, after long year developments, it is now used in many mammalian cells for various fields of study and applications, including the transduction in many other cells, miRNA delivery, vaccine delivery, and for tissue engineering in mammals21. In this study, we show that baculovirus transduction by a dosage of MOI = 1 is enough to exhibit gene expression in about 55–60% of the cells (Fig. 1d) and it does not significantly affect the cellular proliferation rate but slightly a delay in the early time-points was observed. However, such slightly delay is quite nature due to baculovirus loading into the cells, and strong foreign protein expressions in these cells. Also, we observed that transduced cells were viable for up to 12 days at lower viral doses, with only mild effects being observed at MOI = 100. Our mCherry fluorescence images indicate that mCherry is persistently expressed up to 8 days post-transduction, and then slightly declined by 12 days post-transduction (Fig. 2f). We think this result is likely due to the transduced viral DNA being degraded or diluted upon cellular replication over time.
Although baculovirus is capable of transducing many mammalian cell lines, relatively high MOIs (usually 100–1000) are needed19,39. We found that baculovirus transduction efficiency was much higher in C6/36 cells compared to mammalian Vero-E6 and HEK-293T cells (Supplementary Fig. 1a,b). This is a significant finding because low MOIs decrease possible interference with cellular physiology by the viral transduction process. Further, we found that the efficiency of baculovirus transduction is greater than that of plasmid transfection and that protein expression was also much higher than that achieved through classical transfection approaches in C6/36 cells (Supplementary Fig. 2). Although we began our experiments with recombinant baculovirus (vABspmC, pag1-mCherry) containing a reasonably strong promoter, we discovered that the mosquito cecropin b1 gene promoter is the strongest among our set of tested promoters through systematic comparison of transduction and transfection approaches in C6/36 cells (Fig. 4). The pub promoter consistently showed less fluorescence in the baculovirus transduction system but remained functional in the plasmid transfection system. We think this result could be due to the baculovirus-mediated cellular response suppressing pub promoter efficiency, or the promoter may not be well exposed in the context of viral genome. Similarly, this cellular response might have upregulated b1 promoter activity in the baculovirus transduction system because cecropin b1 is an immune-related gene; though the a4 promoter is also from an immune-related gene, but its activity remained weak. Apart from C6/36 cells, the mosquito b1 and pub promoters were also functional in insect Sf21 cells (data not shown), implying that these promoters could be functionally conserved to some extent in these evolutionarily divergent host cells.
In this study, we expressed NA of influenza H5N3 and analyzed its enzymatic activity from the baculovirus transduced C6/36 cell lysates as an example to demonstrate the application of BacMos system besides establishing using florescent reporter genes (Fig. 5). Baculovirus-mediated gene expression persisted for up to 12 days post-transduction, and the adult mosquitoes remained healthy and expressed transgenes in almost all tissues with little or no tissue barrier (Fig. 7a). Hence, it will be possible to study the function and regulation of the genes in different tissues using tissue specific promoters. It is noteworthy that baculovirus transduction could achieve maximum transgene expression at a low viral dose (1 × 105 PFU) compared to the gene expression at a high viral dose (1 × 107 PFU) by mosquito densoviruses (MDVs)7 and to plasmid injection methods40. In contrast to MDVs7,41, baculovirus likely does not exhibit species or tissue barriers, so construction of a single recombinant baculovirus is sufficient to study gene regulation in various mosquito species. Moreover, baculovirus will be possible in the future to deliver genes or DNA encoding small regulatory sequences into mosquito cells and organisms to study gene function, regulation, protein production and interactions, drug assays, virus/host interactions, and many other experiments in the cells or organisms with a flexibility whenever gene transduction is needed. BacMos system gives a flexibility to introduce or block a gene to analyze its function in a particular developmental stages from embryo to adults which is difficult to achieve by the conventional germ line engineering. Because, the deletion or over expression of many genes by germ line engineering could be lethal in the earlier developmental stages, therefore, limits the functional study of many such important genes in the later developmental stages. In these cases, baculovirus will be a novel alternative for the blocking or introducing of these genes transiently in different suitable developmental stages to study gene functions.
In summary, we have developed a BacMos system, a simple and efficient way to introduce foreign genes into the cells, larvae, and adults of many different mosquito genus. In contrast to transgenic mosquitoes, BacMos allows expression of a gene that may be lethal in early developmental stages but functions at other stages. Therefore, this robust BacMos system offers flexibility in the time of introduction, high level in vivo and in vitro gene expression, non-lytic transduction, broad host range, large DNA cargo capacity, and finally, with biosafety. Thus, our BacMos system exhibits considerable potential for the advancements of gene regulation studies in mosquitoes and associated viruses.
Methods
Cells and viruses
A. albopictus C6/36 and A. aegypti CCL-125 cell clones (ATCC, Taiwan) were maintained in RPMI-1640 medium (Invitrogen) supplemented with 10% fetal bovine serum (FBS), 2 mM L-glutamine, 0.1 mM non-essential amino acids, 1 mM sodium pyruvate, and 2% penicillin-streptomycin at 28 °C in a 5% CO2 humidified incubator. A. pseudoscutellaris AP-61 cell clone were maintained in L-15 medium (Leibovitz; GIBCO) + L-glutamine supplemented with 20% FBS and 2% penicillin-streptomycin at 28 °C in a 5% CO2 humidified incubator. The Spodoptera frugiperda IPLB-Sf21 (Sf21) cells (ThermoFisher Scientific) were grown at 26 °C in TC100 insect medium containing 10% FBS. HEK-293T and Vero-E6 cells (ATCC, Taiwan) were maintained in Dulbecco’s modified Eagle’s medium (DMEM; GIBCO) supplemented with 10% FBS and 2% penicillin-streptomycin at 37 °C in a 5% CO2 humidified incubator. All cell lines were routinely checked for mycoplasma every 2 months. The Autographa californica nucleopolyhedrovirus (AcMNPV) baculovirus (gene bank accession number NC_001623.1) genome (flashBACULTRA) was used to generate recombinant baculoviruses in this study. Viral frozen stocks in respective culture mediums were stored at −80 °C.
Construction of transfer vectors and recombinant baculoviruses
The PCR amplified TriEx promoter from pTriEx™-3 vector (Novagen) was subcloned into pBacPAK8 (Clontech) replacing the polyhedrin promoter locus naming shortly as pAB transfer vector (p; plasmid, A; AcMNPV lateral ends, B; pBacPAK8 backbone vector) used for further modifications. In this plasmid, we have inserted mammalian sv40 and HzNV-1 pag1 gene promoters upstream to the TriEx promoter. The PCR-amplified mCherry gene from pJET-mCherry vector was subcloned into downstream of the pag1 promoter to generate the final transfer vector, named pABspmC. In this composite vector, mCherry expression is driven by the sv40 and pag1 promoters in the mammalian and insect systems, respectively. For promoter analysis, we constructed several transfer vectors as listed in Fig. 3a. Originally, the PCR amplified fragments of hsp70 promoter and EGFP from different sources was subcloned into the EcoRV restriction site of pBluescript II SK (+) Phagemid (Agilent Technologies) to obtain pks/hsp70-EGFP plasmid. Then, the complete DNA fragment of hsp70 promoter plus EGFP was subcloned in reverse orientation into the EcoRV restriction site of pBacPAK8 (Clontech), named as pABhEG (h; hsp70 promoter and EG; EGFP). The PCR-amplified various promoter regions were cloned upstream of EGFP into the pABhEG plasmid replacing the hsp70 promoter. The promoter regions of pag1, baculovirus early gene; p10, baculovirus late gene; cmv, cytomegalovirus; sv40, simian virus 40; cir, chimeric internal ribosome entry site (IRES) of RhPV virus and EV71 virus; as well as two mosquito genes cecropin b1 and defensin a4 (GenBank IDs: HQ285957.1, and HQ285959.1) both amplified by PCR from the A. aegypti genome; and the commercially-synthesized Polyubiquitin gene (GenBank ID: GU179018) promoter region (565 bp upstream of Pub gene)42 (named respectively as the pABpag1EG, pABp10EG, pABcmvEG, pABsv40EG, pABcirEG, pABb1EG, pABpubEG, and pABa4EG transfer vectors). The transfer vector pABspmC was modified in sequential manner to obtain the final pABb1p10-NA3 plasmid used in Fig. 5; firstly the sv40 promoter and mCherry gene was replaced by pag1 promoter and EGFP gene upstream of TriEx promoter in the reverse orientation followed by replacing cmv and T7 promoter by b1 promoter upstream of p10 promoter in the TriEx promoter. Next, the Neuraminidase (NA) of H5N3 (A/duck/Taiwan/a180/2015) influenza strain43, (named as NA3) was cloned in the multiple cloning site. At N-terminus of NA3, the signal peptide (SP) and cytoplasmic domain (CTD) of baculovirus envelope GP64 protein and FLAG and HIS tags at the C-terminus of NA3 were cloned respectively. These transfer vectors were constructed by following the standard protocols of the In-Fusion Cloning Kit (Clontech). The transfer vector pBacb1EG-irEG was constructed according to the traditional cloning methods. Briefly, a NcoI–XbaI EGFP fragment was subcloned into the NcoI and XbaI sites of pGL3 to create the plasmid, pGL3-EGFP. Both NheI–BglII promoter fragments of cecropin b1 were amplified and subcloned into pGL3-EGFP to create pGL3-b1EGFP. The restriction enzyme-digested region between NheI–NotI from pGL3-b1EGFP was subcloned into pBac-CHIKV-26S-Rhir-E44 replacing CHIKV-26S region. The large NheI-BglII fragment of pBac-b1EGFP-RhirE was fill-in and self-ligated to create the basic vector (shortly; pBacb1EG-irEG). All the above transfer vectors were verified by sequencing. Briefly, these transfer vectors were co-transfected with flashBACULTRA, a viral DNA of AcMNPV containing lateral fragments of 1250 bp and 1433 bp to facilitate homologous recombination, into Sf21 cells by using Cellfectin (Life Technologies). Homologous recombination occurs at the site of the polyhedron gene and produces a baculovirus lacking this gene. The resulting recombinant baculoviruses were isolated through end-point dilutions using florescent protein as the marker, amplifications in T75 flasks, and virus titers were determined by TCID50 analysis in Sf21 cells.
Baculovirus transduction and infection
Mosquito C6/36 cells or mammalian cells were plated at a density of 4 × 105 cells or 1 × 105 cells (unless stated) per well in 24-well plates 1 day before transduction. The cells were washed with 1X DPBS and baculovirus was added at different MOIs as indicated in respective medium without any FBS or antibiotics. Plates were centrifuged at 2000 rpm for 32 min at room temperature (RT). Transduced mosquito or mammalian cells were incubated at 28 °C or RT, respectively, for 2 h. The plates were washed twice with 1X DPBS containing 0.1% trypsin to remove unattached viruses. Then complete 10%-FBS medium was added and cultured for various time-points as indicated. For baculovirus infection assays, Sf21 cells were plated at a density of 2 × 105 cells per well in 24-well plates 1 day before infection and baculovirus was added at different MOIs. The plates were centrifuged at 2000 rpm for 32 min at RT and then incubated at 28 °C for various timeframes as indicated.
Baculovirus neutralization assay
The vABspmC baculovirus (4 × 105 PFU) at MOI = 1 was treated with 1 µg of neutralizing (AcV1, Santa Cruz Biotechnology) or non-neutralizing monoclonal antibodies (AcV5, Santa Cruz Biotechnology) against GP64 protein in a total volume of 300 µl with 1X DPBS and centrifuged at 300 rpm for 1 h at RT. The virus + antibody mixture was then added to C6/36 cells for 2 h and kept at 28 °C in a 5% CO2 humidified incubator. The virus + antibody mixture was then removed and washed three times with 1X DPBS containing 0.1% trypsin to remove unattached virus particles. Complete 10%-FBS medium was then added, and the cells were cultured for 2 days before examining mCherry fluorescence.
Cell proliferation dye and cell viability analyses
C6/36 cells (4 × 105) were seeded in 12-well culture dish, and the next day cells were transduced with recombinant vABspmC baculovirus at an MOI = 1, 10 and 100 for 2 hours. The unattached virus was removed, washed twice with 1X DPBS containing 0.1% trypsin, and 1 ml 10 µM cell proliferation dye (CPD) eFluor450 (eBioscience, catalog number: 65-0842) in 1X DPBS was added per well. Then, incubated at 28 °C in the dark for 10 min and the labeling was stopped by adding 3 ml of cold complete media keeping on ice for 5 min. Cells were washed 3 times with complete media and then cultured in complete media at 28 °C incubator. The experiment was performed in three biological replicates, three wells of cells were harvested immediately (time zero), followed by harvesting of samples 24, 48, 72 and 96 h. Cells were harvested by trypsinization, washed twice in 1X DPBS and prepared cells for flow cytometry analysis. For cell viability analysis, relative cell metabolic activity was determined by adding 10% v/v AlamarBlue to the cells and incubating for 4 h. Reductions in AlamarBlue concentrations were measured with a fluorescence reader (EnSpire, PerkinElmer) at an excitation wavelength of 560 nm and an emission wavelength of 590 nm.
Plasmid DNA transfection
Mosquito C6/36 cells were plated at a density of 4 × 105 cells per well in 24-well plates. The next day, various doses of recombinant plasmid DNA (1 µg, 2 µg and 3 µg in Supplementary Fig. 2 and 0.5 µg in Fig. 3) were mixed with respective amounts of transfection reagent (TransIT-Insect Transfection Reagent; Mirus) as per manufacturer recommendations and incubated for 30 min at RT, before the DNA + transfection reagent mixture was added to the cells. After 5 h in a 28 °C incubator, the cells were removed and washed with 1X DPBS, before 10%-FBS complete medium was added. The cells were then incubated at 28 °C for 2 days before being collected for flow cytometry analyses.
Preparation of cells for flow cytometry analyses
Mosquito C6/36 cells were transduced with baculovirus in 24-well plates for various timeframes as shown in the figures. The medium was removed from the wells, cells were washed twice with 1X DPBS, and then treated with 100 µl of pre-warmed trypsin-EDTA for 5 min. Then 100 µl of RPMI-1640 medium (Invitrogen) was added to neutralize the trypsin-EDTA and cells were collected in 1 ml of 1X DPBS in 1.5 ml Eppendorf tubes. The cells were centrifuged at 5000 rpm for 5 min to remove the DMEM and the cell pellet was washed at least three times with 1X DPBS. Finally, cells were resuspended in 1 ml of fixing buffer (1% FBS + 1% formaldehyde diluted in 1X DPBS) and analyzed by flow cytometry. The numbers of mCherry- or EGFP-positive cells and the mean fluorescence intensities (MFI) were determined using flow cytometry and FlowJo software.
Quantification of baculovirus DNA by real-time qPCR
Baculovirus-infected Sf21 or baculovirus-transduced C6/36 cells were extensively washed three-times with 1X DPBS containing 0.1% trypsin to remove free virus particles from cells. Total cellular DNA was extracted using the High Pure PCR Template Preparation Kit (Roche). The quantity and quality of DNA was checked using a NanoDrop ND-1000 spectrophotometer (Thermo Fisher Scientific). Real time-qPCR was performed in a 96-well plate, with each well containing 2 μl DNA (100 ng/µl), 0.6 μl 10 μM specific primers (final concentration, 300 nM), 6.8 μl H2O, and 10 μl 2X SYBR Green Master Mix (ABI). Samples were run in triplicate. Real-time PCR was conducted in the ABI 7500 FAST system for over 40 cycles with an annealing temperature of 60 °C. Assessment of the expression of each target gene was based on relative quantification (RQ) using the comparative critical threshold (CT) value method. The RQ of a specific gene was evaluated in each reaction by normalization to the CT obtained for the endogenous control genes (Actin gene in C6/36 cells and GAPDH in Sf21 cells). Three independent transfection experiments were conducted, and the data presented represent the results from one independent infection experiment. The primers for quantitative real-time PCR used in this study were as follows: primers for endogenous internal control: ACTIN forward (5′-CACGAACTGGGACGATATGG A-3′) and ACTIN reverse (5′- CGGTTAGCCTTCGGGTTCAG-3′); primers for baculovirus DNA amplification: IE-1 forward (5′-TTCGAATCCCTTGAGCAGC-3′) and IE-1 reverse (5′-TGCCGATGGTTGGTTCACA -3′); primers for endogenous internal control: GAPDH forward (5′-ATCAAGCAGAAGGTCAAGGAG-3′) and GAPDH reverse (5′-GCAGCAGCATCGAAGATAGA-3′).
Western blot analysis
Mosquito C6/36 cells were transduced with vABb1p10-empty (virus backbone) or vABb1p10-NA3 at MOI = 10 in 24-well plates and cultured for 48 h. The culture medium was removed and cell lysate was harvested in 100 µl of 1X Radioimmunoprecipitation assay buffer (RIPA buffer, Thermoscientific, product#89901) and protein concentration was quantified by Coomassie Plus Bradford assay kit (Thermoscientific). 50 μg of total protein was mixed with 5X sample buffer and heated at 95 °C for 10 min, and proteins were resolved by 10% SDS-PAGE and subjected to Western detection. Western transfer was performed on nitrocellulose membranes. After blocking with 5% skim milk, the blot was incubated with mouse monoclonal anti-FLAG antibody (1:5,000; GeneTex) and rabbit polyclonal anti-Tubulin (1:5,000; GeneTex), followed by horseradish peroxidase (HRP)-conjugated mouse or goat anti-rabbit IgG (1:5,000). The blot was processed for detection with a commercial T-Pro Lumilong Plus Chemiluminescence Detection Kit (T-Pro Biotechnology, Cat. No: JT96-K004M).
Neuraminidase activity (NA) and Neuraminidase inhibition (NI) assays
Neuraminidase activity (NA) and Neuraminidase inhibition (NI) assays was performed using 4-MUNANA assay36. 4-MUNANA assay is based on the cleavage of a fluorescent substrate, 2′-(4-methylumbelliferyl)-α-D-N-acetylneuraminic acid sodium salt hydrate (4-MUNANA; Sigma-Aldrich, Cat: M8639) by influenza NA; and the yield of free 4-methylumbelliferone (4-MU) fluorescent product is then quantitated based on its fluorescence intensity. In the NA assay, 5 µg of total cellular protein was mixed with 30 µl of 1 × 4-MUNANA substrate (final concentration of 100 µM) by pipetting in 96-well plate. In the NI assay, 10 µM of NA inhibitors (Oseltamivir or Zanamivir) were added to the cell lysates and incubated at 37 °C for 30 min before adding the 4-MUNANA substrate. Samples in 96-well plate were then incubated at 37 °C for 1 h to catalyze the 4-MUNANA substrate. The reaction was terminated by adding 150 µl of stop solution (0.14 M NaOH in 83% ethanol). The fluorescence due to release of 4-methylumbelliferone was read using an excitation wavelength of 365 nm and an emission wavelength of 450 nm.
Mosquito rearing
The A. aegypti (Kaoshiung strain), A. albopictus (Chung-Ho strain), C. tritaeniorhynchus (Peitou strain), and Anopheles sinensis (Yunlin strain) were reared at 27 °C and at 80% humidity under a 12-h light/dark cycle. Larval stages were fed on pulverized fish food, and adults were provided with 5% sucrose ad libitum. Mosquitoes were maintained and performed experiments in full compliance with the Ethical Committee Guidelines and Regulations set by the National Defense Medical Center, Taipei 114, Taiwan, ROC.
Mosquito virus intrathoracic inoculation
Frozen stocks of viruses were thawed at 37 °C, and four or five 10-fold serial dilutions of virus were made in RPMI1640 medium. Stocks of viruses with appropriate RPMI 1640 medium dilution were injected with 0.98 nl volume into 4–5 day post-eclosion female A. aegypti, A. albopictus, C. tritaeniorhynchus, and A. sinensis mosquitoes (starved for 24 h). Cold-anesthetized mosquitoes were intra-thoracically inoculated with virus according to the previous reported protocol45.
Statistics
Data were analyzed by Student’s t-test and two tailed P value of <0.05 was considered significant.
Accession codes
Nucleotide sequences have been deposited in GenBank under the following accession codes: pABpag1-EG: MH290746; pABp10-EG: MH290747; pABcmv-EG: MH290748; pABsv40-EG: MH290749; pABcir-EG: MH290750; pABb1-EG: MH290751; pABpub-EG: MH290752; pABa4-EG: MH290753; pABb1p10-NA3: MH290754; pBacb1EG-irEG: MH290755.
Electronic supplementary material
Acknowledgements
This research was funded by grants MOST 106-2321-B-001-027, MOST-2811-B-001-016, MOST 106-3114-B-033-001 and MOST105-2321-B-001-040 from the Ministry of Science and Technology, NHRI-106A1-MRCO-0217171, NHRI-106A1-MRCO-1117171 and NHRI-106A1-MRCO-1117172, MR-107-PP-22 and MR-107-PP-26 from The National Health Research Institutes, MAB-107-046 from The National Defense Medical Center, and 022361 from Academia Sinica, Taiwan, ROC. We would like to thank Dr. W. C. Du for providing mosquito Anopheles sinensis, Dr. Paul Hsu of the Academia Sinica IMB computer room facility for drawing the mosquito cartoon, Dr. John O’Brien of the Academia Sinica IMB Editing Core for editing this manuscript.
Author Contributions
N.G.N., C.-C.L., S.-C.K., and Y.-C.C. conceived and designed research; N.G.N. and Y.-W.L. performed research; N.G.N., T.-Y.W., C.-C.L., S.-C.K., and Y.-C.C. analyzed the data; N.G.N. and Y.-C.C. wrote the paper.
Data availability
The authors declare that the data supporting the findings of this study are available from the authors upon reasonable request.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-35463-8.
References
- 1.Cheng G, Liu Y, Wang P, Xiao X. Mosquito Defense Strategies against Viral Infection. Trends Parasitol. 2016;32:177–186. doi: 10.1016/j.pt.2015.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Guzman A, Isturiz RE. Update on the global spread of dengue. Int J Antimicrob Agents. 2010;36(Suppl 1):S40–42. doi: 10.1016/j.ijantimicag.2010.06.018. [DOI] [PubMed] [Google Scholar]
- 3.Huang YJ, Higgs S, Horne KM, Vanlandingham DL. Flavivirus-mosquito interactions. Viruses. 2014;6:4703–4730. doi: 10.3390/v6114703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.WHO. Malaria Fact Sheet. World Health Organization (WHO) (2017).
- 5.Allen ML, O’Brochta DA, Atkinson PW, Levesque CS. Stable, germ-line transformation of Culex quinquefasciatus (Diptera: Culicidae) J Med Entomol. 2001;38:701–710. doi: 10.1603/0022-2585-38.5.701. [DOI] [PubMed] [Google Scholar]
- 6.Xlao-Guang Chen GM, Anthony A. James. Gene Expression Studies in Mosquitoes. Adv Genet. 2008;64:19–50. doi: 10.1016/S0065-2660(08)00802-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Suzuki, Y., Niu, G. D., Hughes, G. L. & Rasgon, J. L. A viral over-expression system for the major malaria mosquito Anopheles gambiae. Scientific Reports4 (2014). [DOI] [PMC free article] [PubMed]
- 8.Ledermann JP, Suchman EL, Black WC. t. & Carlson, J. O. Infection and pathogenicity of the mosquito densoviruses AeDNV, HeDNV, and APeDNV in Aedes aegypti mosquitoes (Diptera: Culicidae) J Econ Entomol. 2004;97:1828–1835. doi: 10.1093/jee/97.6.1828. [DOI] [PubMed] [Google Scholar]
- 9.O’Neill SL, et al. Insect densoviruses may be widespread in mosquito cell lines. J Gen Virol. 1995;76(Pt 8):2067–2074. doi: 10.1099/0022-1317-76-8-2067. [DOI] [PubMed] [Google Scholar]
- 10.Afanasiev BN, Ward TW, Beaty BJ, Carlson JO. Transduction of Aedes aegypti mosquitoes with vectors derived from Aedes densovirus. Virology. 1999;257:62–72. doi: 10.1006/viro.1999.9621. [DOI] [PubMed] [Google Scholar]
- 11.Kost TA, Condreay JP, Jarvis DL. Baculovirus as versatile vectors for protein expression in insect and mammalian cells. Nat Biotechnol. 2005;23:567–575. doi: 10.1038/nbt1095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Possee RD, Thomas CJ, King LA. The use of baculovirus vectors for the production of membrane proteins in insect cells. Biochem Soc Trans. 1999;27:928–932. doi: 10.1042/bst0270928. [DOI] [PubMed] [Google Scholar]
- 13.Ayres MD, Howard SC, Kuzio J, Lopez-Ferber M, Possee RD. The complete DNA sequence of Autographa californica nuclear polyhedrosis virus. Virology. 1994;202:586–605. doi: 10.1006/viro.1994.1380. [DOI] [PubMed] [Google Scholar]
- 14.Hofmann C, et al. Efficient gene transfer into human hepatocytes by baculovirus vectors. Proc Natl Acad Sci USA. 1995;92:10099–10103. doi: 10.1073/pnas.92.22.10099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Laakkonen JP, et al. Baculovirus-mediated immediate-early gene expression and nuclear reorganization in human cells. Cell Microbiol. 2008;10:667–681. doi: 10.1111/j.1462-5822.2007.01074.x. [DOI] [PubMed] [Google Scholar]
- 16.Lee HP, Chen YL, Shen HC, Lo WH, Hu YC. Baculovirus transduction of rat articular chondrocytes: roles of cell cycle. J Gene Med. 2007;9:33–43. doi: 10.1002/jgm.994. [DOI] [PubMed] [Google Scholar]
- 17.Sherman KE, McIntosh AH. Baculovirus replication in a mosquito (dipteran) cell line. Infect Immun. 1979;26:232–234. doi: 10.1128/iai.26.1.232-234.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Becnel JJ. Prospects for the mosquito baculovirus CuniNPV as a tool for mosquito control. J Am Mosq Control Assoc. 2006;22:523–526. doi: 10.2987/8756-971X(2006)22[523:PFTMBC]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 19.Mansouri M, et al. Highly efficient baculovirus-mediated multigene delivery in primary cells. Nat Commun. 2016;7:11529. doi: 10.1038/ncomms11529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Boyce FM, Bucher NL. Baculovirus-mediated gene transfer into mammalian cells. Proc Natl Acad Sci USA. 1996;93:2348–2352. doi: 10.1073/pnas.93.6.2348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Airenne KJ, et al. Baculovirus: an insect-derived vector for diverse gene transfer applications. Mol Ther. 2013;21:739–749. doi: 10.1038/mt.2012.286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee DF, Chen CC, Hsu TA, Juang JL. A baculovirus superinfection system: efficient vehicle for gene transfer into Drosophila S2 cells. J Virol. 2000;74:11873–11880. doi: 10.1128/JVI.74.24.11873-11880.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wu YL, et al. The early gene hhi1 reactivates Heliothis zea nudivirus 1 in latently infected cells. J Virol. 2010;84:1057–1065. doi: 10.1128/JVI.01548-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Byrne BJ, Davis MS, Yamaguchi J, Bergsma DJ, Subramanian KN. Definition of the simian virus 40 early promoter region and demonstration of a host range bias in the enhancement effect of the simian virus 40 72-base-pair repeat. Proc Natl Acad Sci USA. 1983;80:721–725. doi: 10.1073/pnas.80.3.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hefferon KL, Oomens AG, Monsma SA, Finnerty CM, Blissard GW. Host cell receptor binding by baculovirus GP64 and kinetics of virion entry. Virology. 1999;258:455–468. doi: 10.1006/viro.1999.9758. [DOI] [PubMed] [Google Scholar]
- 26.Salem TZ, Zhang F, Xie Y, Thiem SM. Comprehensive analysis of host gene expression in Autographa californica nucleopolyhedrovirus-infected Spodoptera frugiperda cells. Virology. 2011;412:167–178. doi: 10.1016/j.virol.2011.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Carinhas N, et al. Quantitative proteomics of Spodoptera frugiperda cells during growth and baculovirus infection. PLoS One. 2011;6:e26444. doi: 10.1371/journal.pone.0026444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pollizzi KN, et al. Asymmetric inheritance of mTORC1 kinase activity during division dictates CD8(+) T cell differentiation. Nat Immunol. 2016;17:704–711. doi: 10.1038/ni.3438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Granados RR, Derksen AC, Dwyer KG. Replication of the Trichoplusia ni granulosis and nuclear polyhedrosis viruses in cell cultures. Virology. 1986;152:472–476. doi: 10.1016/0042-6822(86)90150-9. [DOI] [PubMed] [Google Scholar]
- 30.Kost TA, Condreay JP. Recombinant baculoviruses as mammalian cell gene-delivery vectors. Trends Biotechnol. 2002;20:173–180. doi: 10.1016/S0167-7799(01)01911-4. [DOI] [PubMed] [Google Scholar]
- 31.Liu CY, Wang CH, Wang JC, Chao YC. Stimulation of baculovirus transcriptome expression in mammalian cells by baculoviral transcriptional activators. J Gen Virol. 2007;88:2176–2184. doi: 10.1099/vir.0.82664-0. [DOI] [PubMed] [Google Scholar]
- 32.Sollerbrant K, et al. A novel method using baculovirus-mediated gene transfer for production of recombinant adeno-associated virus vectors. J Gen Virol. 2001;82:2051–2060. doi: 10.1099/0022-1317-82-9-2051. [DOI] [PubMed] [Google Scholar]
- 33.Kenoutis C, et al. Baculovirus-mediated gene delivery into Mammalian cells does not alter their transcriptional and differentiating potential but is accompanied by early viral gene expression. J Virol. 2006;80:4135–4146. doi: 10.1128/JVI.80.8.4135-4146.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Afanasiev BN, Kozlov YV, Carlson JO, Beaty BJ. Densovirus of Aedes aegypti as an expression vector in mosquito cells. Exp Parasitol. 1994;79:322–339. doi: 10.1006/expr.1994.1095. [DOI] [PubMed] [Google Scholar]
- 35.Chen YJ, Chen WS, Wu TY. Development of a bi-cistronic baculovirus expression vector by the Rhopalosiphum padi virus 5′ internal ribosome entry site. Biochem Biophys Res Commun. 2005;335:616–623. doi: 10.1016/j.bbrc.2005.07.116. [DOI] [PubMed] [Google Scholar]
- 36.Leang, S. K. & Hurt, A. C. Fluorescence-based Neuraminidase Inhibition Assay to Assess the Susceptibility of Influenza Viruses to The Neuraminidase Inhibitor Class of Antivirals. J Vis Exp (2017). [DOI] [PMC free article] [PubMed]
- 37.Makkonen KE, Airenne K, Yla-Herttulala S. Baculovirus-mediated gene delivery and RNAi applications. Viruses. 2015;7:2099–2125. doi: 10.3390/v7042099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Merryweather AT, et al. Construction of genetically engineered baculovirus insecticides containing the Bacillus thuringiensis subsp. kurstaki HD-73 delta endotoxin. J Gen Virol. 1990;71(Pt 7):1535–1544. doi: 10.1099/0022-1317-71-7-1535. [DOI] [PubMed] [Google Scholar]
- 39.Kost TA, Kemp CW. Fundamentals of Baculovirus Expression and Applications. Adv Exp Med Biol. 2016;896:187–197. doi: 10.1007/978-3-319-27216-0_12. [DOI] [PubMed] [Google Scholar]
- 40.Wilke ABB, Scaife S, Alphey L, Marrelli MT. DsRed2 transient expression in Culex quinquefasciatus mosquitoes. Memorias Do Instituto Oswaldo Cruz. 2013;108:529–531. doi: 10.1590/S0074-02762013000400023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Suzuki, Y., Barik, T. K., Johnson, R. M. & Rasgon, J. L. In vitro and in vivo host range of Anopheles gambiae densovirus (AgDNV). Scientific Reports5 (2015). [DOI] [PMC free article] [PubMed]
- 42.Anderson MAE, Gross TL, Myles KM, Adelman ZN. Validation of novel promoter sequences derived from two endogenous ubiquitin genes in transgenic Aedes aegypti. Insect Molecular Biology. 2010;19:441–449. doi: 10.1111/j.1365-2583.2010.01005.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lee MS, et al. Highly pathogenic avian influenza viruses H5N2, H5N3, and H5N8 in Taiwan in 2015. Vet Microbiol. 2016;187:50–57. doi: 10.1016/j.vetmic.2016.03.012. [DOI] [PubMed] [Google Scholar]
- 44.Kuo SC, et al. Cell-based analysis of Chikungunya virus membrane fusion using baculovirus-expression vectors. Journal of Virological Methods. 2011;175:206–215. doi: 10.1016/j.jviromet.2011.05.015. [DOI] [PubMed] [Google Scholar]
- 45.Gubler DJ, Rosen L. A simple technique for demonstrating transmission of dengue virus by mosquitoes without the use of vertebrate hosts. Am J Trop Med Hyg. 1976;25:146–150. doi: 10.4269/ajtmh.1976.25.146. [DOI] [PubMed] [Google Scholar]
- 46.Min MK, Bishop DH. Transcriptional analyses of baculovirus polyhedrin and foreign gene expression relative to baculovirus p10 mRNA levels. J Gen Virol. 1991;72(Pt 10):2551–2556. doi: 10.1099/0022-1317-72-10-2551. [DOI] [PubMed] [Google Scholar]
- 47.Kerry JA, Priddy MA, Stenberg RM. Identification of sequence elements in the human cytomegalovirus DNA polymerase gene promoter required for activation by viral gene products. J Virol. 1994;68:4167–4176. doi: 10.1128/jvi.68.7.4167-4176.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wu YJ, et al. Internal ribosome entry site of Rhopalosiphum padi virus is functional in mammalian cells and has cryptic promoter activity in baculovirus-infected Sf21 cells. Acta Pharmacol Sin. 2008;29:965–974. doi: 10.1111/j.1745-7254.2008.00820.x. [DOI] [PubMed] [Google Scholar]
- 49.Thompson SR, Sarnow P. Enterovirus 71 contains a type I IRES element that functions when eukaryotic initiation factor eIF4G is cleaved. Virology. 2003;315:259–266. doi: 10.1016/S0042-6822(03)00544-0. [DOI] [PubMed] [Google Scholar]
- 50.Sun D, Fallon AM. Characterization of genomic DNA encoding cecropins from an Aedes albopictus mosquito cell line. Insect Mol Biol. 2002;11:21–30. doi: 10.1046/j.0962-1075.2001.00305.x. [DOI] [PubMed] [Google Scholar]
- 51.Gao Y, Hernandez VP, Fallon AM. Immunity proteins from mosquito cell lines include three defensin A isoforms from Aedes aegypti and a defensin D from Aedes albopictus. Insect Mol Biol. 1999;8:311–318. doi: 10.1046/j.1365-2583.1999.83119.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The authors declare that the data supporting the findings of this study are available from the authors upon reasonable request.