Abstract
Background: Genetic testing is performed for different purposes, such as identifying carriers, predicting a disease onset in presymptomatic individuals or confirming a diagnosis. However, these tests may have notable psychological effects, such as generating anxiety and depression. These effects may depend on people's perception of risk, severity, and controllability of the disease; and the availability of treatments. To date, there are no reports that analyze these factors specifically, and their role in influencing genetic test users' experience.
Methods: We performed a systematic review of the psychological implication of undergoing genetic testing for cardiovascular, neurodegenerative and cancer diseases. Articles were searched on PubMed, Google Scholar, and PsychInfo.
Results: 47 studies were included, 9 concerning cardiovascular disease, 18 neurodegenerative disorders, and 20 for cancer disease. According to the reviewed studies, people experience no significant increase in distress and anxiety, or adverse impacts on quality of life, except the Huntington disease, which is characterized by depressive symptoms, suicidal ideations, and hopelessness in gene carriers. People tend to consider genetic tests as valid information to take important preventive decisions. Genetic risk for cardiovascular disease is perceived to be manageable; genetic analysis for some neurodegenerative diseases (e.g., Alzheimer) or cancer (breast cancer in particular) is considered useful because the problem could be addressed in advance with preventive behaviors.
Conclusions: Genetic tests should be proposed along with proper psychological support and counseling focused on users' genetic health literacy; perception of risk, beliefs about disease controllability, in order to foster fruitful medical decisions.
Keywords: genetic testing, genetic risk, chronic disease, psychological implication, quality of life, health psychology
Introduction
Cancer, cardiovascular diseases and dementia are among the main causes of mortality and morbidity in Europe. Since they will have even larger economic implications in the future, policy-makers have increasingly focused their attention on them (Abegunde et al., 2007; Désesquelles et al., 2014; Mackenbach et al., 2014; Suzman et al., 2015). These conditions affect many people worldwide often causing an impairment of the quality of life and psychosocial well-being, thus they require the attention of the scientific community. In 2015 cardiovascular disease death were 17.92 million, with 422.7 million of cases worldwide (Roth et al., 2017). The WHO estimated that 17.7 million people died from cardiovascular disease last year. Alzheimer disease (AD) affects approximately 24 million people globally (Erkkinen et al., 2018) and this number could quadruple by 2050. Currently, dementia is reported to be the leading cause of mortality in England and Wales (Office for National Statistics ONS), and in 2015 the Eurostat (the Directorate-General of the European Commission) reported 213,000 deaths in Europe caused by nervous system diseases including Alzheimer's.
Regarding cancer data, 14.1 million new cancer cases and 8.2 million cancer deaths occurred in 2012 worldwide (Ferlay et al., 2015), which in 2015 grew to 8.8 million (WHO).
These complex conditions usually need long-term treatments and care, involving different health professionals, expensive drugs, access to medical equipment, putting a large burden on society.
The growing diagnoses of many chronic diseases are associated with an aging population, but also with lifestyle choices such as smoking, diet and exercise, and genetic predisposition (WHO and FAO, 2003; World Health Organisation, 2014).
In the last decades, there have been considerable investments in genomics (DNA-based) research to study susceptibility to cancer and other chronic diseases and to promote new preventive interventions (Walter and Emery, 2012). Currently, the use of family health history and multiplex genetic tests to identify an individual's risk for multiple diseases simultaneously is a frequent clinical practice (Yang et al., 2003; Yoon and Scheuner, 2003; Khoury et al., 2004; Yoon, 2005). Many associations between single-nucleotide polymorphisms (SNPs) and risks for common complex diseases have been identified. Genetic testing generally provides information about the presence of these genetic variants (SNPs), which could represent an increased risk of developing the disease. Their clinical utility depends on how much the knowledge about this genetic variant could give additional information concerning diagnosis, prognosis or contribute to disease management. Available types of testing include for instance diagnostic, carrier, predictive and susceptibility tests. Diagnostic tests confirm a diagnosis when a particular condition is suspected, based on physical symptoms. Carrier testing identifies people who carry one copy of a gene mutation that can be inherited by their offspring. Predictive and susceptibility testing identifies mutations that increase a person's risk of developing disorders with a genetic basis. These tests may help people making decisions about their daily habits or medical care. For instance, discovering the susceptibility for breast cancer, or stroke could allow people to change their lifestyle, nutrition and “take steps to reduce those risks for which interventions are or will be available” (Collins and McKusick, 2001). Nonetheless, not all kinds of genetic testing are useful for clinical management or outcomes improvement, either because of a lack of treatments, uselessness for the personal decision, or absence of scientific evidence for the genetic predisposition. Proven clinical utility and cost-effectiveness need to be carefully evaluated when considering the implementation of genetic testing in healthcare (Cornel et al., 2014), even when considering the recent discoveries which have underlined the heterogeneity of chronic diseases and the importance of gene-environment interaction in modulating disease onset and responses to preventive interventions (Curtis et al., 2012).
In spite of these premises, a recent study, published by the Market Research Future (the “Global Genetic Testing Market - Forecasts from 2018 to 2023” see https://www.marketresearchfuture.com/), reported the amount of genetic tests performed in the European and non-European countries based on the subdivision of pathologies or on the type of genetic test (diagnostic, predictive, etc.). The study estimated a global growth of genetic testing market at a Compounded Average Growth Rate (CAGR) of 12.94% by 2023. This exponential growth should go hand in hand with an appropriate genetic counseling practice, but to date genetic information is often given to people with poor genetic literacy without a specific psychological assessment (Burke et al., 2002) or genetic testing users do not receive a specific pre and post-test counseling (Janssens et al., 2017). For instance, in Italy, only 12% of all genetic analyses had been accompanied by pre or post-test counseling (Giardino et al., 2016).
The genetic counseling is usually provided by trained professionals, mainly geneticists, who explain the genetic aspects of illnesses and the risk of developing or passing an illness to their offspring (WHO). It should address patients' concerns, and one of their families, to help with the decision-making process. Nevertheless, this not always happened in recent years especially with the introduction of direct to consumers genetic testing (DTC), genetic tests sold to people without a medical intermediate (Oliveri and Pravettoni, 2016).
Starting from these premises, we could infer that genetic risk communication might deeply affect people's lives and habits. To date, studies on the psychological impact of genetic tests mainly focused on “harmful” reactions, such as anxiety, distress, and depression, when receiving genetic risk information, obtaining discordant results and without any explanation/discussion for this discrepancy (Oliveri et al., 2016a). Previous reviews revealed that DNA based disease risk has little or no effect on health-related behaviors (Heshka et al., 2008; Hollands et al., 2016). We should consider that genetic testing impact also depends on how people perceive their risk, severity, and controllability related to specific categories of disease (Cameron and Muller, 2009; Wang et al., 2009; Wade et al., 2012); on the genetic tests predictability or nature of the diseases (from monogenic to genetic susceptibility factors), and on the presence/absence of treatments (Cameron and Muller, 2009).
People's emotional reactions to genetic testing and how risk perceptions vary from diseases to disease are fundamental aspects to be investigated on, in order to correlate preventive behaviors they may, or may not, adopt (DiLorenzo et al., 2006; Shiloh et al., 2013).
For this reason, we aim to provide a comprehensive overview of studies realized in the last two decades (2000–2016), which investigated psychological and behavioral issues after having undergone genetic testing for different categories of chronic diseases. The purpose is to identify a limited number of overarching psychological reactions for each condition. In particular, we chose to compare neurodegenerative, cardiovascular and cancer diseases, because of their differences in treatments availability and preventive options (e.g., there are fewer preventive options for neurodegenerative disorders compared to cancer or cardiovascular diseases, where risk is in some cases manageable with screenings or healthier lifestyles). Moreover, people have different beliefs and perception of the controllability for these diseases which could affect their reaction to a positive genetic test result.
Methods
Study Design and Search Strategy
Potential eligible articles were systematically searched on PubMed, Google Scholar and PsychInfo using the following combinations of terms: “psychological outcomes,” “psychological impact,” “genetic test,” “genetic risk,” “neurodegenerative disorders,” “cancer,” and “cardiovascular disease.” Depending on the disease for which the genetic test was performed, we allocated the collected articles into three general categories: Cancer (C), Cardiovascular diseases (CV), and Neurodegenerative disorders (N) (see Table 1). Following criteria were considered to include articles:
Table 1.
Characteristics of studies evaluating psychological impact of genetic testing for cardiovascular, neurodegenerative and cancer diseases.
| Dc | Author | Year/Country | Population | Subject of evaluation (disease specific) | Design | Used instruments behavioral changes, coping, QoL, wellbeing and symptoms | Used instruments psychological impact | Main findings | |
|---|---|---|---|---|---|---|---|---|---|
| CARDIOVASCULAR DISEASES | |||||||||
| 1 | CV | Hickey et al. (a) | 2014 (USA) | ♀♂adults (n = 58) with cardiac genetic diagnoses | Cardiac genetic testing | After testing | SF-36 | HADS-A, HADS-D, IPQ-R | Positive genetic results did not negatively impact patient well-being with the exception of the bodily pain domain of the SF-36 |
| 2 | CV | Hickey et al. (b) | 2014 (USA) | ♀♂adults (n = 31) gc, nc | Cardiac genetic testing | After testing | SF-36 | Physical components of the SF-36 were within normal limits (46.2 ± 6.6) but elevated for mental components (59.9 ± 5.3) | |
| 3 | CV | Christiaans et al. | 2009 (GERMANY) | ♀♂adults (n = 228) MYBPC3, MYH7, TPM1, TNNT2, TNNI3, PRKAG2 and GLA gc with and without symptoms | Hypertrophic cardiomyopathy y (HCM) | After testing | SF-36; IPQ-R Perceived risks 10-point response options (“very small” to “very large”) | HADS-A, HADS-D | QoL and distress were worst in gc with manifest HCM before DNA testing and best in predictively tested gc without HCM Illness and risk perception related variables were major determinants of QoL and distress |
| 4 | CV | Jones & Clayton | 2012 (USA) | ♀♂adults (n = 70) affected or at-risk BMPR2 mutations | Primary pulmonary arterial hypertension | Before/After testing | IES | Test acceptors evidenced dramatic changes in levels of distress, often meeting levels associated with PTSD prior to testing, and evidencing dramatically lower levels of distress following testing Not observed differences in levels of distress between gc and nc | |
| 5 | CV | van Maarle et al. | 2001 (NETHERLANDS) | ♀♂adults(n = 677) gc, nc | (FH) Familial hypercholesterolaemia | Before/After testing | SF-36, EuroQol | HADS | Effects on mood were minimal to absent, as were general QoL effects |
| 6 | CV | Marteau et al. | 2004 (UK) | ♀♂adults (n = 341) at risk and adult relatives (n = 128) | (FH) Familial hypercholesterolaemia | Before/After testing | Items from RIPQ; Self-reports about smoking, diet, activity level and cholesterol-lowering medication adherence | STAI short form, HADS | Finding a mutation had no impact on perceived control or adherence to risk-reducing behavior gc believed less strongly in the efficacy of diet in reducing their cholesterol level and showed a trend in believing more strongly in the efficacy of cholesterol-lowering medication |
| 7 | CV | Hietaranta-Luoma et al. | 2015 (FINLAND) | ♀♂adults and young adults (n = 107) high-risk vs. low-risk group vs. control group (n = 56) | APOE susceptibility testing for Cardio-vascular disease | Before/After testing | Ad hoc questionnaire (Diet, alcohol consumption), HTAS | STAI | Psychological effects of personal genetic risk information were shown to be short-term Slightly higher levels of state anxiety and threat experienced in the high-risk group compared to baseline Information on the ApoE genotype impacted the experience of cardiovascular threat; this effect was most intense immediately after genetic feedback was received |
| 8 | CV | Hendriks et al. | 2008 (NETHERLANDS) | ♀♂adults (n = 134), relatives of LQTS patients, with abnormal, uncertain, or normal ECG | Long qt syndrome (LQTS) | Before/After testing | IES, BDI | Individuals with an abnormal ECG (i.e., clinical diagnosis of LQTS) expressed a moderate level of anxiety that was not affected by mutation carrier status Individuals with a normal or uncertain ECG likewise displayed a moderate level of anxiety at baseline Individuals with an initial uncertain ECG who were later identified as genotype-positive maintained moderate disease-related anxiety over time, although depression scores declined to a level comparable to the general population | |
| 9 | CV | Legnani et al. | 2006 (ITALY) | ♀♂ adults and relatives (n = 140) gc, nc, control group | Thrombophilia alterations | Before/After testing | Perceived health status, perceived well-being, and perceived daily-life stress | CBA-H anxiety, health fears, depressive reactions, moods | For both groups gc and nc, none of the psychological variable scores showed significant worsening, at pre and post-test Anxiety significantly decreased at post-test in adults with thrombophilia alterations Diagnosis of thrombophilia alterations seemed to be well accepted in the short term |
| NEURODEGENERATIVE DISORDERS | |||||||||
| 10 | N | Decruyenaere et al. | 2003 (BELGIUM) | ♀♂adults (n = 57) gc and nc | HD | Before/After testing | STAI, BDI, SCL-90, IES, HOS, MMPI | 5 years after the test, mean distress scores of both gc and nc were within the normal range gc had significantly less positive feelings and were more consciously avoiding HD-related situations and thoughts (Avoidance) Compared with baseline level, mean depression, general and specific anxiety had significantly decreased 1 year and 5 years post-test Persons who asked the test to get rid of the uncertainty, without being able to specify implications for substantial life areas, had more psychological distress before and after the test than those who wanted the test for specific reasons | |
| 11 | N | Licklederer et al. | 2008 (GERMANY) | ♀♂ adults (n = 121) gc, nc, and patients with HD | HD | After testing | SF-12, German Social Support Questionnaire, SOC-L9, BFS | BDI-II, BSI, GSI | Comparable mental health and QoL in nc and gc without symptoms of HD Patients with manifest HD showed a higher level of depression and lower QoL than nc and gc without symptoms In nc increased depression and low mental QoL were due to: low perceived social support, no intimate relationship, female sex and younger age For gc increased depression and low mental QoL were due to: low perceived social support, the expectation of an unfavorable genetic test result before the testing procedure and being childless |
| 12 | N | Almqvist et al. | 2003 (CANADA) | ♀♂ adults (n = 106) gc, nc | HD | Before/After testing | GWS | BDI, SCL-90-R | Adverse events, e.g., suicide, clinical depression occurring in a part of both groups but more frequently in gc Overall improvement in psychological distress compared to baseline in both groups More depressive symptoms in gc than in nc 5 years after testing Adverse events, e.g., suicide, clinical depression occurring in a part of both groups but more frequently in gc Overall improvement in psychological distress compared to baseline in both groups More depressive symptoms in gc than in nc 5 years after testing Adverse events, e.g., suicide, clinical depression occurring in a part of both groups but more frequently in gc Overall improvement in psychological distress compared to baseline in both groups More depressive symptoms in gc than in nc 5 years after testing -Adverse events, e.g., suicide, clinical depression occurred in both groups gc and nc, but more frequently in gc-Overall improvements in psychological distress compared to baseline in both groups were registered -More depressive symptoms in gc than in nc 5 years after testing |
| 13 | N | Timman et al. | 2004 (NETHERLANDS) | ♀♂ adults (n = 142) gc, nc | HD | Before/After testing | GHQ-28 | BHS, IES | Long-term increase in hopelessness in gc compared to baseline No long-term improvement in hopelessness in nc compared to baseline Improvement in long-term in avoidance and intrusions in both groups compared to baseline but more pronounced for nc |
| 14 | N | Larsson et al. | 2006 (SWEDEN) | ♀♂ adults (n = 93) gc, nc | HD | Before/After testing | Well-being: 150-millimeter- long line on a visual analog scale. GHQ-30, SIBS, LSI, LSA | BDI | Both gc and nc showed high suicidal ideation before the predictive testing Depression scores and frequency of suicidal thoughts increased in gc over time No differences regarding life satisfaction and life style |
| 15 | N | Horowitz et al. | 2001 (USA) | ♀♂ adults (n = 59) nc, gc with and without HD neurological symptoms | HD | Before/After testing | IES, BDI | gc with neurological impairment had significantly higher psychological symptom scores at baseline than gc without neurological impairment or nc For nc and gc without HD neurological symptoms the news of genetic testing for the HD gene had limited detrimental impact | |
| 16 | N | Gargiulo et al. | 2009 (FRANCE) | ♀♂ adults (n = 119) gc, nc | HD | After testing | BDI,BHS, STAI, IES | Depression was frequent in asymptomatic gc (58%) 27% of nc did not cope well with a favorable result, and a significant percentage of nc (24%) were depressed during follow-up A previous episode of depression was predictive of depression after genetic testing in both gc and nc | |
| 17 | N | Robins Wahlin et al. | 2000 (SWEDEN) | ♀♂ adults (n = 34) gc, nc | HD | Before/After testing | GHQ-30, SIBS, Risk perception (VAS); LSI, LSA | BDI | Both groups showed high suicidal ideation and self-injurious behavior nc had a very high frequency of attempted suicide, and both groups had similarly pronounced psychiatric dysfunction |
| 18 | N | Witjes-Ané et al. | 2002 (NETHERLANDS) | ♀♂ adults (n = 134) gc, nc | HD | Before/After testing | UHDRS | gc complained more than nc about sadness, low self-esteem, aggressive behavior, and compulsions At 18-months follow-up gc still complained about aggression, while complaints about mood and low self-esteem had disappeared | |
| 19 | N | Surampalli et al. | 2015 (USA) | ♀♂adults (n = 29) gc, nc | VCP gene for Hereditary myopathies (HIBM), Paget's disease of bone, Fronto-temporal dementia | Before/After testing | Risk Perception and Symptom Specific Concern (five point Likert scale), RBD | HADS | At baseline, one quarter of the participants had high levels of anxiety and nobody was depressed Scores were in normal range one year following testing Mean risk perception at baseline was 50.1% |
| 20 | N | Gonzalez et al. | 2012 (ISLAND OF FLORES AND S. MIGUEL) | ♀♂adults (n = 47) gc, nc | Pre-symptomatic testing for Machado-Joseph disease (MJD) | After testing | PGWB | Scores indicating moderate or severe stress were found in half of participants The average score in the PGWB scale was lower in symptomatic than in asymptomatic subjects Impact of the appearance of first symptoms on the psychological state | |
| 21 | N | Gooding et al. | 2006 (USA) | ♀♂adults (n = 60) and parents affected with AD | APOE susceptibility testing for AD | After testing | Interview -perceived likelihood of developing AD; -beliefs about the causes of AD; perceived control over AD; | Interview -reactions to receiving results | Most participants viewed genetic testing as providing valuable information that could help direct future health care decisions, to seek information about health threats, and need to feel in control of their health |
| 22 | N | Linnenbringer et al. | 2010 (USA) | ♀♂adults (n = 246) and parents affected with AD | APOE susceptibility testing for AD. | Before/After testing | Perceived personal risk to develop AD: a scale of 0–100%. Understanding of risk results, AD concern, severity of AD, AD treatment optimism, personal control (5-point Likert scales) | BAI, CESD | 69.3% of participants, who accurately recalled their AD risk assessment 6 weeks after risk disclosure, believed that their AD risk was higher than risk estimate they were given This group of people had higher scores in AD control and anxiety at baseline Overall, anxiety scores were below clinically significant levels |
| 23 | N | Cassidy et al. | 2008 (USA) | ♀♂ adults (n = 123) APOE vs. dominant mutations carriers | APOE susceptibility testing vs. autosomal dominant mutations (presenilin-1, presenilin-2, or TAU genotype) for AD or frontotemporal dementia | After testing | IES | The test-related distress experienced by those receiving positive results for a deterministic mutation was similar to the distress experienced by those receiving positive results from genetic susceptibility testing The majority of participants receiving genotype disclosure did not experience clinically significant distress 1 year after learning of their test results | |
| 24 | N | Green et al. | 2009 (USA) | ♀♂asymptomatic adults (n = 162) and parents with AD REVEAL STUDY | APOE for AD, Disclosure group vs. non-disclosure group | Before/After testing | BAI, CESD, IES | The disclosure of APOE genotyping results to adult children of patients with Alzheimer's disease did not result in significant short-term psychological risks Test-related distress was reduced among those who learnt that they were APOE ε4–negative Persons with high levels of emotional distress before undergoing genetic testing were more likely to have emotional difficulties after disclosure | |
| 25 | N | Chao et al. | 2008 (USA) | ♀♂ asymptomatic adults (n = 162) and parents with AD REVEAL STUDY | APOE for AD, Disclosure group vs. non-disclosure group | After testing | Yes/no questions about changes in: Any behavior specific to AD prevention Medications/vitamins Diet Exercise | 12 months after disclosure, APOE-positive participants reported changes in any one of the domains of diet, physical exercise and medication or vitamin intake, more often (52%) than ε4-negative participants (24%) or the nondisclosure group (30%) Within each domain, there were no significant differences between the groups (disclosure vs. non-disclosure group) | |
| 26 | N | Vernarelli et al. | 2010 (USA) | ♀♂ adults (n = 272) REVEAL STUDY | APOE for AD | After testing | Yes/no questions with free-text field on changes in: Overall diet use of dietary supplements exercise | Genetic susceptibility testing for AD was positively associated with dietary supplement use after risk disclosure. Such changes occurred despite the absence of evidence that supplement use reduces the risk of AD | |
| 27 | N | Romero et al. | 2005 (USA) | ♀♂ adults (n = 76) gc, nc | APOE | After testing | Self-developed questionnaire | nc did not feel worried or depressed, but they felt relieved 15–30% of gc felt depressed and 11–22% felt worried. A little percentage also felt relieved | |
| CANCER | |||||||||
| 28 | C | Aspinwall et al. | 2013 (USA) | ♀♂ adults (n = 60) unaffected nc (n = 27),unaffected gc (n = 15), affected gc (n = 18) | CDKN2A/p16 mutations risk for melanoma and pancreatic cancer | Before/After testing | Open-ended questions on costs and benefits of Genetic Testing | HADS, MICRA, 3-item melanoma and pancreatic cancer worry | Low reported anxiety and depression For gc and nc, anxiety decreased significantly throughout the 2-year period Depression and melanoma worry showed short-term decreases Worry about pancreatic cancer was low and decreased significantly In all groups, test-related distress and uncertainty (MICRA) were low, regret was absent, and positive experiences were high All participants reported at least one perceived benefit of genetic testing |
| gc reported increased knowledge about melanoma risk and prevention and increased prevention and screening behaviors for self and family nc reported increased knowledge and emotional benefits | |||||||||
| 29 | C | Lammens et al. | 2010 (NETHERLANDS) | ♀♂ adults (n = 119) gc, nc | p53 germline mutation Li-Fraumeni Syndrome (LFS) | After testing | SF36, Questionnaires perceived risk, social support, motivations to undergo or not undergo genetic testing and regrets | IES, Social Constraint Questionnaire, CWS | Uptake of pre-symptomatic testing was 55% 23% reported clinically relevant levels of LFS-related distress. - gc were not significantly more distressed than nc or than those with a 50% risk who did not undergo genetic testing Those with a lack of social support were more prone to report clinically relevant levels of distress |
| 30 | C | Di Prospero et al. | 2001 (CANANDA) | ♀ adults (n = 24) unaffected | BRCA 1/2 B/Ov | After testing | Ad Hoc questionnaire: Communication with family members, attitudes toward surveillance, prevention options, satisfaction with clinical services, need for additional support, and satisfaction with decision to undergo genetic testing | Focus group and ad hoc questionnaire for perception of cancer risk and worry assessment | Cancer risk perception and worry increased after receipt of the test results Participants did not regret their decision to undergo testing Confidence in the efficacy of cancer surveillance was high Prophylactic oophorectomy was much more acceptable than prophylactic mastectomy 38% of the participants felt they would benefit from a support group |
| 31 | C | Ertmanski et al. | 2009 (POLAND) | ♀ adults (n = 3,524) affected and unaffected | BRCA1/2 B/Ov | Before/After testing | Patient satisfaction | STAI, BHI-12, IES | Anxiety does not increase in women positive for BRCA1 Women who experience high levels of anxiety before genetic testing continue to experience high levels of anxiety up to 1 year post testing BRCA1 gc with a previous diagnosis of cancer had significantly higher levels of cancer-related distress at 1 month post-test than those without cancer |
| 32 | C | Hamilton et al. | 2009 (USA and CANADA) | ♀ adults (n = 7) non symptomatic individuals | BRCA1/2 B/Ov | After testing | Follow-up interviews Impact on daily life, health behaviors in the intervening years. | Participants accepted recommended surveillance and preventative measures to maximize a healthy lifestyle and reported both the benefits of knowing their mutation status as well as challenges they had encountered since testing Over time, awareness of genetic risk does not appear to diminish | |
| 33 | C | Vos et al. | 2012 (NETHERLANDS) | ♀ adults (n = 248) gc, unclassified-variants and uninformative-results | BRCA1/2 B/Ov | After testing | IPQ-R, COPE Ad hoc questionnaire: perception of risk, medical outcomes, familial and psychological contexts | RSPWB | The actually communicated cancer risks did not directly predict any outcomes (lifestyle changes, medical intentions and emotions) The counselees' perception of risk and heredity likelihood predicted medical intentions, behaviors, physical and psychological life-changes, stigma, mastery, negativity and cancer-worries Short-term distress was related to the perception not only of their own risks, but also of their relatives' risks and heredity-likelihood |
| 34 | C | Katapodi et al. | 2011 (USA) | ♀ adults (n = 372) affected probands and relatives not tested | BRCA1/2 B/Ov | After testing | IPQ-R, DCS Ad hoc questionnaire Perceived risk (scale 0-100), knowledge risk factors and modes of gene inheritance, family relationships, family communication | Psychological distress (scale 0–10) | Probands perceived higher risk and had more psychological distress associated with breast cancer Probands had more knowledge regarding risk factors and gene inheritance, and greater decisional conflict regarding genetic testing Relatives reported higher perceived severity and controllability No differences were observed in family relationships and family communication between probands and relatives |
| 35 | C | Lodder et al. | 2000 (NETHERLANDS) | ♀ adults (n = 78) healthy subjects, nc, gc | BRCA1/2 B/Ov | Before/After testing | Interview transcripts about intentions concerning risk management, reported impact of the test outcome | HADS, IES | High post-test anxiety was reported by 20% of the gc women and by 35% of their partners 11% of women nc and 13% of their partners reported high post-test anxiety levels nc who had a sister identified as a gc had higher post-test levels of depression than the other nc |
| 36 | C | Meiser et al. | 2002 (AUSTRALIA) | ♀ adults (n = 143) nc, gc and not tested | BRCA1/2 B/Ov | Before/After testing | MBSS, satisfaction with the decision to undergo testing | BAI, STAI-S, IES | Compared with women not offered testing, gc had significantly higher distress 7–10 days and 12 months post results nc showed a significant decrease in state anxiety 7–10 days post-notification and in depression 4 months post-notification compared with women not offered testing |
| 37 | C | Rini et al. | 2009 (USA) | ♀ adults (n = 182) affected probands | BRCA1/2 B/Ov | After testing | Ad hoc questionnaire (Likert scale): decision status, perceived risk for relapse benefits/barriers to mammography benefits of and barriers to risk-reducing mastectomy | MICRA, BSI, IES, DCS | Substantial numbers of women reported elevated decisional conflicts 1-month and 12-months post disclosure, health beliefs and emotional factors predicted decisional conflict at different time points, with health beliefs more important 1 month after test disclosure and emotional factors more important 1 year later |
| 38 | C | Smith et al. | 2008 (USA) | ♀ adults (n = 126) gc and women elected not to be tested | BRCA1/2 B/Ov | Before/After testing | Perceived risk, SF36 | SCL-90-R, GSI, IES, PSC, CESD, STAI-Form Y1 | Results indicated no systematic effects of testing based on personal cancer history gc and women elected not to be tested reported greater perceived risk and intrusive and avoidant thoughts at follow-up time points than did women who received negative (uninformative) or variant results gc reported more distress at the 3-month follow-up but by 6 months the effects of test result on distress dissipated and groups were comparable |
| 39 | C | Dougall et al. | 2009 (USA) | ♀ adults (n = 126) | BRCA1/2 B/Ov | Before/After testing | COPE | SCL-90-Revised, IES, PSC, STAI CESD | Coping was relatively stable over time and did not vary as a function of genetic test results Active coping strategies were used more often by women with a personal cancer history than by women without cancer Use of avoidant coping was reliably and positively associated with distress over time, independently of cancer history and test result |
| 40 | C | Samson et al. | 2014 (CANADA) | ♀ adults (n = 6) with family history of B/Ov | BRCA1/2 B/Ov | After testing | Grounded theory interview | Main topics: Physical Task: attempting to limit the impact of the test result; Psychological Task: living with uncertainty; Social Task: finding effective support | |
| 41 | C | Arver et al. | 2004 (CANADA) | ♀ adults (n = 87) healthy women, gc, nc | BRCA1, BRCA2, MLH1, and MSH2 B/Ov and colon cancer susceptibility genes | Before/After testing | SF-36, satisfaction with decision making and testing (5-graded scale) | HADS | Significant decrease in anxiety scores over time The levels of depression in gc decreased over time while, surprisingly the levels in nc increased, however, still within the normative range Vitality was the only domain that was statistically significantly influenced by gene testing. In gc of colon cancer genes the scores dropped two to 6 months after the result disclosure, followed by increased levels, while the levels in nc were more stable |
| 42 | C | Claes et al. | 2004 (SWEDEN) | ♀ adults (n = 62) gc, nc, and patients with an inconclusive (no specific familial mutation is found) genetic test result | BRCA1/2 B/Ov | After testing | Semi-structured interviews about personal experiences with cancer, family history of B/Ov cancer, genetic testing. Questions on willingness for testing, perceived seriousness and perceived control | UCL, STAI, SCL-90, IES | Gc felt more in control, but they also reported negative emotional impact and being concerned about their children Nc were relieved No differences among groups in general and cancer-specific distress No differences among groups regarding perceived seriousness of breast and ovarian cancer and perceived control of breast cancer Perceived control of ovarian cancer was highest in the inconclusive group |
| 43 | C | Manchanda et al. | 2015 (UK) | ♀♂ adults (n = 1,034) family history based testing (FH) vs. population-based genetic testing (PS) | BRCA1/2 B/Ov | After testing | SF-12 | HADS, HAI, MICRA | There were no statistically significant differences on anxiety, depression, distress, uncertainty and quality-of-life between the FH and PS arms, at 7 days or 3 months after genetic testing. Overall anxiety and uncertainty associated with genetic testing decreased Positive experience scores increased |
| 44 | C | Shiloh et al. | 2013 (ISRAEL) | ♂ adults (n = 81) gc, nc | BRCA1/2 B/Ov | After testing | Brief IPQ, Questions about health behavior, questions about risk perception | MICRA | Up to 4 years post genetic testing, 48% of those who tested positively reported that the test increased their perceptions of risk, and 74% of them increased surveillance for cancer nc did not report increased perceived risk and relatively few increased surveillance (31%) gc were significantly more distressed from testing |
| 45 | C | Claes et al. | 2005 (BELGIUM) | ♀ adults (n = 68) unaffected gc, nc | B/Ov | Before/After testing | UCL, Questions about Perceived impact of genetic test, Questions about illness representation, SCL-90 | IES, STAI | Mean levels of distress were within normal ranges in both gc and nc Cancer-specific distress and state-anxiety significantly decreased in nc from pre to post- test while general distress remained about the same There were no significant changes in distress in the group of gc except for ovarian cancer distress which significantly decreased from pre- to post test The study did not reveal adverse effects of predictive testing when offered in the context of a multidisciplinary approach |
| 46 | C | Andrews et al. | 2004 (UK) | ♀ adults (n = 43) gc, nc | BRCA1/2 B/Ov | Before/After testing | Ad hoc questionnaire: satisfaction with genetic testing perceived Risk of developing breast cancer accuracy of perceived risk | IES, STAI, BDI | Women who chose to learn their results had significantly higher baseline breast cancer anxiety, compared to those who choose not to learn their results Unaffected women who choose to learn their results showed a significant decrease in breast cancer anxiety 4 months and 12 months post-notification compared to baseline Genetic testing does not lead to adverse psychological outcomes |
| 47 | C | Reichelt et al. | 2004 (NORWAY) | ♀ adults (n = 395) with or without personal history of disease | BRCA1/2 B/Ov | Before/After testing | GHQ-28 | HADS, IES, BHS | No significant changes were found in psychological distress from baseline to follow-up in any groups Women with cancer were significantly more distressed at baseline and at 6 weeks |
Dc, Disease category; CV, Cardiovascular disease and other vascular pathologies; C, Cancer; N, Neurological disorders.
Used abbreviations:
♀, female; ♂, male; AD, Alzheimer disease; HD, Huntington disease; B/Ov, breast and/or ovarian cancer; QoL, Quality of life; gc, gene carrier/mutation carriers; nc, non-carriers.
Abbreviations of used instruments:
BAI, Beck Anxiety Inventory; BDI, Beck's Depression Inventory; BFS, Benefit Finding Scale; BHI-12, Basic Hope Inventory. BHS, Becks Hopelessness Scale; BSI, Brief Symptom Inventory; CBA-H, Cognitive Behavioral Assessment Hospital Form; CESD, Center for Epidemiologic Studies Depression Scale; COPE, inventory of coping responses; CWS, Cancer Worry Scale; DCS, Decisional Conflict Scale; GHQ-28- GHQ-30, General health questionnaire 28-items/30-items;GSI, Global Severity Index; GWS, General Wellbeing Scale; HADS, Hospital Anxiety and Depression Scale; HAI, Health Anxiety Inventory; HOS, Health Orientation Scale; HTAS, Health and taste attitude; IES, Impact of Event Scale; IPQ-R, Illness Perception Questionnaire – Revised; LSA, Life-Styles Assessment; LSI, Life Satisfaction Index; MBSS, Miller Behavioral Style Scale; MICRA, The Multidimensional Impact of Cancer Risk Assessment; MMPI, Minnesota Multiphasic Personality Inventory; PGWB, Psychological General Well-Being Index; PSC, Perceived Stress Scale; RBD, Risk Behavior Diagnostic Scale; RIPQ, Revised Illness Perceptions Questionnaire; RSPWB, The Ryff Scales of Psychological Well-Being; SCL-90, The Symptom Checklist; SF-36/SF-12, Short-Form Health Questionnaire 36-Item/12-Item; SIBS, Self-Injurious Behavior Scale; SOC-L9, Leipzig Sense of Coherence Scale; STAI, State Trait Anxiety Inventory; UCL, Utrecht Coping List; UHDRS, Unified Huntington′s Disease Rating Scale.
studies in which a psycho-behavioral and/or quality of life evaluation after having received genetic test results was performed;
studies in which subjects tested were adults.
Exclusion criteria were determined by the aim to analyze the “raw” impact genetic test information can have on people's psychological reactions and/or quality of life, without the mediation of interventions (e.g., counseling), or specific population (e.g., children) or phenomenon (e.g., Direct to Consumer genetic testing).
Exclusion criteria were as follow:
investigation of prenatal screening, or childhood and adolescent genetic testing;
investigation of genetic testing for psychiatric disorders;
investigation of family dynamics, the efficacy of psychological or other kind of educational and counseling intervention;
hypothetical situations in undergoing genetic testing;
direct to consumer genetic testing.
Recent reviews, meta-analyses or narrative accounts of knowledge were excluded.
For each study, we identified the implemented design, the number, and composition of participants, the psychological instruments employed and the main findings regarding psycho-behavioral outcomes and quality of life after testing.
Because of substantial heterogeneity among research studies, no attempt at formal meta-analysis was made in this contribution.
Results
Studies Selection and Characteristics
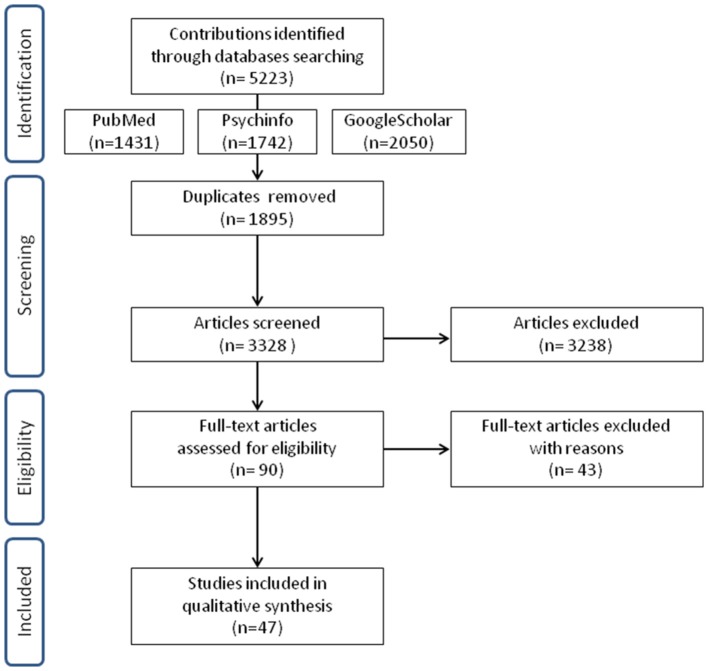
Three thousand and three hundred twenty-eight manuscripts, published between 2000 and 2016, were assessed for eligibility by reading title and abstract. In total 90 studies were potentially eligible. After reading the full text, articles were included for qualitative synthesis only if they met inclusion/exclusion criteria. As a consequence, 43 studies could not be included, mostly because they investigated prenatal screening or childhood diseases, psychiatric disorders, family dynamics, the efficacy of the psychological intervention, genetic counseling effects or hypothetical situations in undergoing genetic testing. We considered these studies suitable to be treated as a separate topic.
Finally, a set of 47 studies met our inclusion criteria and were completely assessed (see Figure 1 for study selection and Table 1 for a summary of selected studies). Nine studies are concerned with cardiovascular diseases, 18 neurodegenerative disorders, and 20 cancer diseases. They had been conducted in the United States and Canada (n = 22), Europe (n = 22), Australia (n = 1), Israel (n = 1) and the islands of Flores and S.Miguel (n = 1). Included studies have been classified according to the disease for which patients were tested (Table 1 first column), while the study design is reported in Table 2.
Figure 1.
Flow diagram describing the study selection.
Table 2.
Moments of evaluation, before and after the genetic test, per study.
| Before genetic test | After genetic test | |||||
|---|---|---|---|---|---|---|
| Author | Year | Before disclosure of results | < or = 6 weeks after disclosure genetic test result | >6 weeks after disclosure genetic test result | 1 year after disclosure genetic test result | >1 year after disclosure genetic test result |
| Hickey et al. | 2014 | Not defined | ||||
| Hickey et al. (b) | 2014 | Not defined | ||||
| Christiaans et al. | 2009 | + | ||||
| Jones & Clayton | 2012 | + | + (18 months) | |||
| van Maarle et al. | 2001 | + | + | |||
| Marteau et al. | 2004 | + | + | + (6 months) | ||
| Hietaranta-Luoma et al. | 2015 | + | + (10 weeks and 6 months) | + | ||
| Hendriks et al. | 2008 | + | + | + (18 months) | ||
| Legnani et al. | 2006 | + | + | |||
| Decruyenaere et al. | 2003 | + | + | + (5 years) | ||
| Licklederer et al. | 2008 | Not defined | ||||
| Almqvist et al. | 2003 | + | + | + | + | + (2 and 5 years) |
| Timman et al. | 2004 | + | + | + | + (1.5, 3, 7–10 years) | |
| Larsson et al. | 2006 | + | + | + | + (2 years) | |
| Horowitz et al. | 2001 | + | + (3 and 6months) | + | ||
| Gargiulo et al. | 2009 | + | + | |||
| Wahlin et al. | 2000 | + | + (2 and 6 months) | + | + (2 years) | |
| Witjes-Ané et al. | 2002 | + | + (18 months) | |||
| Surampalli et al. | 2015 | + | + | |||
| Gonzalez et al. | 2012 | + (5 years) | ||||
| Gooding et al. | 2006 | Not defined | ||||
| Linnenbringer et al. | 2010 | + | + | |||
| Cassidy et al. | 2008 | + | + | |||
| Green et al. | 2009 | + | + | + (6 months) | + (1 year) | |
| Chao et al. | 2008 | + | ||||
| Vernarelli et al. | 2010 | + | ||||
| Romero et al. | 2005 | + (1 month and 4 months) | ||||
| Aspinwall et al. | 2013 | + | + | + | + | + (2 years) |
| Lammens et al. | 2010 | – | Not defined | |||
| Di Prospero et al. | 2001 | Not defined | ||||
| Ertmanski S et al. | 2009 | + | + | + | ||
| Hamilton et al. | 2009 | |||||
| Vos et al. | 2012 | + | + (3- to 4-years) | |||
| Katapodi et al. | 2011 | Not defined | ||||
| Lodder et al. | 2000 | + | Not defined | |||
| Meiser et al. | 2002 | + | + | + | + | |
| Rini et al. | 2009 | + | + | + | ||
| Smith et al. | 2008 | + | + | + (3 and 6 months) | ||
| Dougall et al. | 2009 | + | + | + (3 and 6 months) | ||
| Samson et al. | 2014 | + (6 months) | ||||
| Arver et al. | 2004 | + | + | + (2 and 6 months) | + | |
| Claes et al. | 2004 | Not defined | ||||
| Manchanda et al. | 2014 | Not defined | + | +(2, 3 years) | ||
| Shiloh et al. | 2011 | +(Up to 4 years after) | + | |||
| Claes et al. | 2005 | + | + | |||
| Andrews et al. | 2004 | + | + | + | + | |
| Reichelt et al. | 2004 | + | + | |||
Cardiovascular Disease
Genetic testing for cardiovascular diseases is usually performed to detect users' susceptibility to conditions that affect the heart muscle, inherited heart conditions that might cause arrhythmias or risk factors which could cause a heart attack. Some of these conditions may require changes in lifestyle or medical therapy. Studies taken into examination evaluated the impact of genetic testing related to different clinical conditions (Hickey et al., 2014a,b), such as Long qt syndrome (Hendriks et al., 2008), thrombophilia (Legnani et al., 2006), cardiomyopathy (Christiaans et al., 2009), arterial hypertension (Jones and Clayton, 2012), and familial hypercholesterolaemia (Van Maarle et al., 2001; Marteau et al., 2004). In general, these studies used similar scales for the assessment of the quality of life and perception of risk (SF-36, IPQ-R) and for evaluating the psychological impact and wellbeing (STAI, HADS, IES). Psychological aspects mainly concerned the anxiety-depressive symptoms and the subjective distress caused by the “traumatic event” of genetic risk communication.
Results showed that no negative impacts on quality of life and no serious increase in distress or anxiety levels were registered after receiving genetic test results (Van Maarle et al., 2001; Marteau et al., 2004; Legnani et al., 2006; Hickey et al., 2014a,b). Anxiety levels were overall moderate and tended to last over time only if the genetic test result was associated with uncertain physiological data (Hendriks et al., 2008) or in case of marked clinical conditions, such as in patients with symptoms of hypertrophic cardiomyopathy before DNA testing (Christiaans et al., 2009). Hietaranta-Luoma et al. (2015) evaluated the impact of genetic testing for the Apolipoprotein E (ApoE, a protein involved in Alzheimer's disease and cardiovascular disease and mapped to chromosome 19). They reported that in high-risk subjects the genetic information combined with personal health status influenced the levels of anxiety, and promoted the short-term reduction of risk factors for cardiovascular disease. One study investigated arterial hypertension (Jones and Clayton, 2012) and detected distress symptoms in patients before undergoing genetic testing, comparable to PTSD symptoms. These symptoms significantly decreased after the reception of genetic results, in both carriers and non-carriers.
Even the quality of life after genetic test results was influenced by patients' difficulties in managing mental states, compared to other aspects. Hickey et al. (2014b) found that mental difficulties, assessed by the SF-36, were higher (59.9 ± 5.3) if compared to physical components, which resulted within normal ranges (46.2 ± 6.6), whereas Christiaans et al. (2009) clarified that levels of distress and impact on mental components did not significantly differ from the average of the general population who never underwent a genetic test. Finally, Marteau et al. (2004) showed that genetic testing for patients, already aware of their risk, does not affect their sense of control over the condition (hypercholesterolemia) but influenced their beliefs on how effectively achieve control on their health (e.g., with drug assumption).
Neurodegenerative Disorders
Genetic testing for neurodegenerative disorders are usually performed: (a) for diagnostic purposes, (b) to determine if a person, who has a family history of disease, is a mutation carrier and thus he/she is at risk to develop the disorder or could have an affected offspring. Currently, no therapies exist for complete remission of these pathologies. Studies we have collected primarily investigated risk related to Alzheimer disease (AD) and Huntington disease (HD). Two studies devoted attention to other neurodegenerative disorders, such as the Machado-Joseph disease (MJD)(Gonzalez et al., 2012) (which causes symptoms like spasticity, difficulty with speech and swallowing, weakness in arms and legs, frequent urination) and mutation to VCP gene (Surampalli et al., 2015) (which along with the inclusion body myopathy it causes frontotemporal dementia).
Huntington Disease (HD) is a dominantly transmitted neurodegenerative disorder: genetic analysis detects, with 100% of certainty, the presence of the mutation gene, confirming the status as a carrier of the condition (Evers-Kiebooms and Decruyenaere, 1998). The outcomes of gene testing can rarely fall within the reduced penetrance range (36–39 CAG repeats), whereby individuals may or may not develop symptoms of the disease; or individuals may be carriers of intermediate alleles (27–35 CAG repeats) and will not develop symptoms of the disease themselves, but their children will be at-risk of HD (Myers, 2004).
Tools used to evaluate the psychological impact of genetic testing for HD predominantly measured anxiety and depression (STAI, BDI), the traumatic impact of the event “genetic test results communication” (IES) or severe psychological symptoms, up to suicidal ideations (see Table 1).
Most authors highlighted the presence of depression and suicidal ideation in a significant percentage of participants, even before undergoing genetic testing (Robins Wahlin et al., 2000; Horowitz et al., 2001; Larsson et al., 2006; Gargiulo et al., 2009), with higher psychological suffering and negative impact on QoL for those with neurological symptoms (Horowitz et al., 2001). Licklederer et al. (2008) found that patients, with mutation and already manifesting HD symptoms, had higher levels of depression and lower levels in QoL indexes, compared to gene carriers without symptoms and non-carriers. Moreover, they showed that depression values in HD gene carriers were related to unfavorable genetic test result in conjunction with negative social and relational conditions (e.g., low perceived social support and being childless). Summarizing, higher levels of depression and lower quality of life were registered in patients with manifest HD or neurological impairments (Horowitz et al., 2001; Licklederer et al., 2008).
Interestingly, Gargiulo et al. (2009) found that 27% of non-carriers (asymptomatic) do not positively elaborate the favorable genetic results whereas Robins Wahlin et al. (2000) showed that non-carriers had a very high frequency of suicide ideations. Another study has also shown that non-carriers tended to develop avoidant or intrusive styles as a reaction to the stressful event (genetic test results) over time (Timman et al., 2004).
Considering the long-term impact of genetic tests for HD, several studies revealed the presence or the increase in depressive symptoms, suicidal ideations, hopelessness, and aggressive reactions in gene carriers (Robins Wahlin et al., 2000; Witjes-Ané et al., 2002; Almqvist et al., 2003; Timman et al., 2004; Larsson et al., 2006; Gargiulo et al., 2009), except for the study of Decruyenaere et al. (2003), showing a significant decrease of depressive symptoms after 1 year, both in gene carriers and non-carriers.
Concerning Alzheimer disease (AD), currently ApoE testing is used in clinical settings to identify people who may have an increased risk of developing AD, whereas other genetic tests investigate the presence of autosomal dominant mutations (in genes PSEN1, PSEN2, and APP which are more predictive for disease development (Goldman et al., 2011).
The REVEAL studies (Chao et al., 2008; Green et al., 2009; Vernarelli et al., 2010) showed that ApoE carriers were not more anxious, depressed, or test-related distressed than people who did not receive any information about their genotype (Green et al., 2009). The levels of anxiety, depression, and distress were below clinical thresholds both in carriers and non-carriers, with a significant distress reduction among those who learned that they were ApoE negative. People who were highly distressed before undergoing genetic testing were more vulnerable to emotional difficulties after outcome disclosure, but distress values were well below clinical thresholds for clinical concern (Green et al., 2009). Romero et al. (2005) described that a small percentage of ApoE gene carriers felt depressed (15–30%) or worried (11–22%). A small percentage also felt relieved.
The study by Cassidy et al. (2008) found that participants who received a positive result for a deterministic mutation experienced the same levels of distress experienced by those receiving positive results for genetic susceptibility testing (ApoE). The same study reported that after 1 year from result disclosure the majority of participants did not experience clinically significant distress.
Concerning more in detail long-term results or changes in health-related behaviors, 12 months after ApoE results, carriers reported changes in lifestyle (diet, physical exercise, and medication or vitamin intake) more often than non-carriers or the nondisclosure group (Chao et al., 2008). A positive correlation between genetic susceptibility testing for AD (an APOE epsilon4+ genotype status) and changes in vitamin intake was also confirmed by Vernarelli et al. (2010), despite there is no evidence that supplement use reduces the risk of AD.
Finally, Linnenbringer et al. (2010) showed that people who accurately recalled their AD disease risk assessment (the risk percentage) tended to perceive their risk higher than the percentage of risk they were given (below clinical thresholds).
Finally, Gooding et al. (2006) interviewed a group of people at high risk for AD (because of relatives affected by AD), and genetic testing was estimated valuable information to improve personal control on health and guide future decisions.
In rare pathologies such as Machado Joseph disease (Gonzalez et al., 2012), anxiety levels were from moderate to severe in half of the participants (52.6%). Five years later quality of life was significantly more compromised in symptomatic people, confirming an impact of the appearance of first symptoms on the psychological state. Meanwhile in VCP genetic testing (Surampalli et al., 2015) were found similar results in anxiety levels as for Alzheimer disease.
Cancer
Genetic testing for cancer is usually performed in pre-symptomatic conditions (the user never developed any symptom related to the cancer disease), or after an episode of cancer diagnosis, to know if there is a hereditary cancer syndrome and/or a risk of relapse.
Most of the articles focused on the risk of developing ovarian and breast cancer, by examining the presence of BRCA1 and BRCA2 mutations (see Table 1). Three studies, respectively, investigated the impact of genetic mutations responsible for pancreatic cancer and melanoma (Aspinwall et al., 2013), colon cancer along with breast cancer (Arver et al., 2004), and Li-Fraumeni Syndrome (Lammens et al., 2010). Studies on BRCA testing used several tools (STAI, IES, SCL-90, BDI, HADS the most used ones) assessing anxiety, post-traumatic stress disorders, psychopathological symptoms and depression, and showed quite heterogeneous results.
Breast and ovarian cancer were overall perceived as having the same seriousness independently by genetic test results (Claes et al., 2004). Many authors revealed the poor influence of genetic tests on anxiety and distress (distress levels within normal ranges), without significant difference between gene carriers and non-carriers (Andrews et al., 2004; Claes et al., 2005; Ertmanski et al., 2009). These results suggest that genetic testing for BRCA does not cause adverse psychological reactions. Four studies reported slightly greater levels of anxiety and negative psychological outcomes in gene carriers (Lodder et al., 2001a; Meiser et al., 2002; Katapodi et al., 2011; Shiloh et al., 2013) whereas Vos et al. (2012) specified that these anxiety levels would be mediated by individual risk perception and concerns about their own relatives' heredity-likelihood. Gene carriers and probands showed to be more distressed and negatively influenced by genetic test results, even because they were concerned about their offspring and experienced decisional conflicts toward their relatives (Claes et al., 2004; Rini et al., 2009; Katapodi et al., 2011). Three studies (Reichelt et al., 2004; Ertmanski et al., 2009; Manchanda et al., 2015) investigated the experience of genetic testing and risk perception in people with a family history or personal history of illness, comparing them with healthy people or people without previous family experience of disease, and they found conflicting results. Manchanda et al. (2015) demonstrated that there were no differences in levels of anxiety and distress based on the presence/absence of a family history of disease, while Reichelt et al. (2004) and Ertmanski et al. (2009) reported higher levels of distress in people who have already had a diagnosis and/or cancer experience. Women with a personal cancer history tended to enact concrete coping strategies more than women without previous experience with cancer (Dougall et al., 2009).
Finally, there are studies which reported satisfaction and positive consequences of having carried out the genetic test for breast/ovarian cancer susceptibility and thus discovering something about the presence of a mutation. In particular, receiving a positive result increased the perception of risk (Di Prospero et al., 2001; Claes et al., 2005; Katapodi et al., 2011; Vos et al., 2012), which correlated with more frequent screenings and checkups, and with a sense of self-efficacy (Di Prospero et al., 2001; Hamilton et al., 2009; Shiloh et al., 2013).
Long-term results showed that levels of test-related distress decreased in the first 4/6 months (Andrews et al., 2004; Arver et al., 2004; Smith et al., 2008), then enduring at low levels after years (Andrews et al., 2004; Manchanda et al., 2015), with an impact on surveillance actions up to 4 years after test disclosure (Shiloh et al., 2013).
Concerning other cancers, in Aspinwall et al. (2013) gene carriers for pancreatic cancer and melanoma increased preventive screening for themselves and their families, thanks to informative genetic test results. A small percentage of patients reported clinically relevant levels of distress related to genetic testing for p53 germline mutation (Lammens et al., 2010). Distress was higher for patients with a lack of social support, as was the case for Huntington Disease (Licklederer et al., 2008).
Discussion
In the last decades, clinical application of genetic testing for diagnosis and prevention has gained more importance to such an extent as to create a market where patients can obtain information on genetic risk in complete autonomy (Su, 2013; Oliveri et al., 2015, 2016b; Oliveri and Pravettoni, 2016). In this framework, there are many possible psychological reactions and related issues worthy of consideration, such as risk perception and perceived controllability after a positive result for a mutation, or concerns about transmitting susceptibility for a disease to future generations.
With this contribution, we aimed to sound out possible differences in psychological reactions to predictive genetic testing based on different disease categories. To date, there are no reports that compare the psychological impact of genetic testing for cardiovascular, neurodegenerative and cancer diseases.
Our review shows that there is no significant increase in distress levels or adverse impact on the quality of life in subjects who undergo a genetic test for cardiovascular diseases; when higher distress is present it does not exceed the clinically significant threshold (Van Maarle et al., 2001; Marteau et al., 2004; Legnani et al., 2006; Hickey et al., 2014b). The psychological distress is related to a full-blown clinical condition in addition to a positive genetic result (Hendriks et al., 2008; Christiaans et al., 2009). Overall people maintain confidence in being able to cope with their risk, even though they modify the opinion on how to address this risk: they tend to believe that lifestyle might be useless to face their “genetic predisposition,” and they need other “more concrete” methods of prevention, such as drug therapies (Marteau et al., 2004). In our opinion, these trends arise from a “deterministic” interpretation of genetic data, and the lack of evidence concerning the effects of lifestyle modifications in the disease course. Changes in lifestyle only concern people who already have physical symptoms (Marteau et al., 2004; Hietaranta-Luoma et al., 2015) and are at higher risk of adverse heart conditions, although these lifestyle changes have short duration (Hietaranta-Luoma et al., 2015). We hypothesize that people with full-blown symptoms are motivated to gather all possible health-related information, including genetic risk information, in order to manage their risk of developing the disease. In general, our review shows that genetic risk for cardiovascular disease is perceived to be manageable, and this might also be due to the existence of screenings to prevent it and possible treatments.
Concerning neurodegenerative disorders, studies put more attention on anxiety and depression symptoms, since these disorders usually have relevance on complex emotions such as embarrassment and social withdrawal (Levenson et al., 2014), affect family relations and put carriers at risk of social discrimination (Perry, 1981; Craufurd and Harris, 1986). Our review describes marked negative psychological impact after positive genetic results for Huntington Disease in patients who have depressive symptoms already before undergoing genetic testing, including suicidal ideation, which are increased also by the presence of adverse relational/family situations (Robins Wahlin et al., 2000; Horowitz et al., 2001; Larsson et al., 2006; Licklederer et al., 2008; Gargiulo et al., 2009). Differently from other chronic diseases, a negative genetic result for HD does not reassure, but it causes negative emotions. This reaction might be due to the uncertainty of results and a lack of “response” for the etiology of cognitive symptoms, when present, or sense of guilty toward family members that have the diseases (Robins Wahlin et al., 2000; Timman et al., 2004; Gargiulo et al., 2009). However, we believe that these results for Huntington disease should be taken with the due caution called by the fact that, often, the evidence was based on participants with previous psychiatric history (Robins Wahlin et al., 2000; Almqvist et al., 2003; Larsson et al., 2006; Gargiulo et al., 2009). Thus, we cannot rule out that, for example, a manifestation of suicidal ideation can be ascribable to this previous psychiatric history rather than to the positive or negative genetic result. Future studies should settle this issue.
Negative psychological impact of genetic testing in gene carriers for HD persists over time (Almqvist et al., 2003; Timman et al., 2004; Larsson et al., 2006), and it might be due to the regret for having undergone the test, anticipating life change limitations (Hagberg et al., 2011).
We argue that in addition to the regret for getting such genetic information, negative reactions may be understandable in light of the certainty these people have to develop HD in the future, the perception of something uncontrollable and fatal, alongside the complete absence of valid therapies and inevitable cognitive decline (Gooding et al., 2006). The decision in undergoing predictive genetic testing, in this case, could be a coping strategy (Gooding et al., 2006)acted to redirect important life decisions.
Slightly different seems to be the impact of genetic analysis for Alzheimer disease. Effects of genetic test results are comparable to those described for cardiovascular diseases, since distress anxiety and depression are below clinically significant thresholds, even for gene carriers, and these results concern both APOE and autosomal dominant mutation testing (Cassidy et al., 2008; Green et al., 2009; Linnenbringer et al., 2010). Therefore, the test is overall experienced as something useful to achieve a good degree of awareness and immediately act preventive behaviors to address the risk. Anyway, for Alzheimer's prevention behavioral changes are not always positive: for example, an increasing assumption of dietary supplements harmless, such as vitamin E, could give people a false perception of control on the health without any significant scientific evidence (Morris et al., 2002). For this reason, it would be beneficial to provide people with more information on how to effectively prevent AD, before providing the opportunity to undergo genetic testing.
Finally, in rare diseases, such as Machado Joseph, anxiety levels were prominent after genetic testing in at least half of patients studied (Gonzalez et al., 2012). These results are understandable in the light of an immediate impairment of daily life (e.g., spasticity, difficulty with speech and swallowing, weakness in arms and legs, frequent urination) and the fact that symptoms get worse over time.
Results on the impact of gene testing in cancer raise more complex and heterogeneous issues than in previous cases. From the emotional point of view, the levels of anxiety and depression decrease significantly after having received test results (Andrews et al., 2004; Arver et al., 2004; Reichelt et al., 2004; Claes et al., 2005; Smith et al., 2008; Ertmanski et al., 2009; Lammens et al., 2010; Aspinwall et al., 2013; Manchanda et al., 2015), and a positive effect emerges as regards screening behaviors as well (Hamilton et al., 2009; Shiloh et al., 2013). We must consider, in order to give an interpretation to the previous results, that breast and ovarian cancer are potentially preventable and early detection can guarantee to heal with effective treatments (Shaw and Bassi, 2001). If results are positive, screening or surgery could help patients reduce their risks, and immediate communication to family members about genetic risk can be crucial to prevent the “danger” of disease development. Deciding on how to address the risk means being able to “recommend” a pathway for prevention to their families (Lodder et al., 2001b; Katapodi et al., 2011; Vos et al., 2012). Preventive and prophylactic decisional pathways are no easy nor straight: risk-reducing prophylactic mastectomy on healthy breast goes along the risk of surgical side effects, body image modification, regrets in women who decided for this solution. Periodic screening is instead potentially accompanied by frequent negative thoughts and emotions (anxiety components).
People who have already had an experience of illness tend to actively cope with the risk of disease onset, although sometimes this is accompanied by higher levels of distress (Dougall et al., 2009). These levels of arousal, however, should not be necessarily perceived in a negative sense; on the contrary emotional arousal could be the engine for the decisional process in cancer care and for acting on coping strategies. Even for cancer, as already found for APOE and Alzheimer's, studies indicate that there are positive aspects reported by patients about having undergone genetic testing. These findings are related to an increase in screening behaviors and an increased sense of self-efficacy in managing the risk (Hamilton et al., 2009; Aspinwall et al., 2013; Shiloh et al., 2013). Summarizing, in cancer, if people get important information on time they can manage their risky or healthy behaviors enhancing the perception of control over their lives and direct it as they wish (e.g., surgery vs. screening).
This is not completely true for cardiovascular disease and Huntington disease, because cardiovascular disease genetic information and evidence about preventive medicine efficacy are completely uncertain and say “something less” about predisposition and prevention options; for HD, deterministic implications of genetic testing give a piece of information that is likely to be “too much information,” and thus perceived as uncontrollable.
Before to conclude, it is important to point out some limitations of the reviewed studies. The first limitation is that gender was not well balanced in all the selected studies and was not investigated as a factor that could influence the psycho-behavioral impact of genetic testing. We exclude papers on breast cancer (which investigated women samples), Li-Fraumeni Syndrome (LFS) (where men and female were equally balanced and female gender was associated with heightened levels of LFS-related distress), and on the risk for developing diseases which disproportionately affect women (e.g., the primary pulmonary arterial hypertension)(Jones and Clayton, 2012). In many studies about neurodegenerative diseases, women were overrepresented (Cassidy et al., 2008; Green et al., 2009; Vernarelli et al., 2010; Gonzalez et al., 2012) and we cannot exclude that the results could be affected by general gender bias. Chao et al. (2008), for example, claimed that the REVEAL study participants were mainly women. Therefore the results may not be generalized to all population who might qualify for APOE genotype testing in the future.
In most studies concerning cardiovascular disease, males and female samples seemed to be well balanced, except for Hietaranta-Luoma et al. (2015) who evaluated more females. Nevertheless, gender differences, when investigated (Legnani et al., 2006; Jones and Clayton, 2012; Hickey et al., 2014a) did not relate to any of the measures of patient well-being.
A second limit was that not all the studies considered participants' educational level as a factor that could correlate with the decision to undergo a genetic test and its psycho-behavioral impact. In the selected studies participants had at least high school education (10–12 years of education completed)(Lodder et al., 2001b; Van Maarle et al., 2001; Witjes-Ané et al., 2002; Almqvist et al., 2003; Claes et al., 2004; Gooding et al., 2006; Legnani et al., 2006; Licklederer et al., 2008; Smith et al., 2008; Christiaans et al., 2009; Rini et al., 2009; Vernarelli et al., 2010; Gonzalez et al., 2012; Vos et al., 2012; Hickey et al., 2014b) or were predominantly highly educated (Andrews et al., 2004; Claes et al., 2005; Dougall et al., 2009; Vernarelli et al., 2010; Aspinwall et al., 2013; Shiloh et al., 2013; Manchanda et al., 2015). Educational level was associated to a better recall of disease risk information (Linnenbringer et al., 2010), to a higher response rates in the follow up (Almqvist et al., 2003), or was not an influential predictor of the psycho-behavioral measures (Robins Wahlin et al., 2000; Meiser et al., 2002; Andrews et al., 2004; Claes et al., 2005; Legnani et al., 2006; Licklederer et al., 2008; Smith et al., 2008; Green et al., 2009; Rini et al., 2009; Jones and Clayton, 2012; Shiloh et al., 2013). Other studies included in our review did not perform analysis based on the educational level, and future studies should address this issue.
Finally, some of the results presented in these studies are based on small sample sizes (Robins Wahlin et al., 2000; Di Prospero et al., 2001; Andrews et al., 2004; Hamilton et al., 2009; Gonzalez et al., 2012; Hickey et al., 2014b; Samson et al., 2014; Surampalli et al., 2015) and are not cross-cultural. Therefore, it is possible that ethnicity and cultural aspects may play a role in determining the psychological implications of genetic testing.
Conclusions
This review presented a comprehensive overview of the psychological impact of genetic testing across the most common chronic adults' diseases. The information level of genetic data varies according to the type of test. Along with this aspect, each of us has a specific perception of disease categories, for which genetic testing is available. Risk perception, worry, and other psychological reactions depend, for instance, on the perceived controllability and existing therapies to manage the illness; it is essential to proceed with an assessment of such factors along with the provision of genetic information.
Over the last 20 years we have witnessed a proliferation of investments in genomics research in order to study disease prevention, disease treatments, better drug therapies, and genetic paths to cure, and, thanks to media coverage such as Angelina Jolie's case (Evans et al., 2014), the psychological impact of these discoveries has gradually become more and more important.
For these reasons, in the present review, we tried to understand better how genetic testing users' perceptions of developing specific diseases affect their psychological well-being and lifestyle. Understand psycho-behavioral reactions could be an important starting point for an effective clinical application of genetic testing and to organize personalized care plans, which can drive patients to self-determination of a healthy lifestyle and to make appropriate decisions for their health.
Author Contributions
SO and FF contributed to the design and implementation of the research, to the analysis of the results and the writing of the manuscript. AM contributed to the writing of the manuscript, and GP supervised all the process, was in charge of overall direction and planning.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This work is supported by a grant to the project Mind the Risk from The Swedish Foundation for Humanities and Social Sciences which did not influence the content of this paper. Grant nr. M13-0260:1.
References
- Abegunde D. O., Mathers C. D., Adam T., Ortegon M., Strong K. (2007). The burden and costs of chronic diseases in low-income and middle-income countries. Lancet 370, 1929–1938. 10.1016/S0140-6736(07)61696-1 [DOI] [PubMed] [Google Scholar]
- Almqvist E. W., Brinkman R. R., Wiggins S., Hayden M. R. (2003). Psychological consequences and predictors of adverse events in the first 5 years after predictive testing for Huntington's disease. Clin. Genet. 64, 300–309. 10.1034/j.1399-0004.2003.00157.x [DOI] [PubMed] [Google Scholar]
- Andrews L., Meiser B., Apicella C., Tucker K. (2004). Psychological impact of genetic testing for breast cancer susceptibility in women of Ashkenazi Jewish background: a prospective study. Genet. Test. 8, 240–247. 10.1089/gte.2004.8.240 [DOI] [PubMed] [Google Scholar]
- Arver B., Haegermark A., Platten U., Lindblom A., Brandberg Y. (2004). Evaluation of psychosocial effects of pre-symptomatic testing for breast/ovarian and colon cancer pre-disposing genes: a 12-month follow-up. Fam. Cancer 3, 109–116. 10.1023/B:FAME.0000039863.89137.f9 [DOI] [PubMed] [Google Scholar]
- Aspinwall L. G., Taber J. M., Leaf S. L., Kohlmann W., Leachman S. A. (2013). Genetic testing for hereditary melanoma and pancreatic cancer: a longitudinal study of psychological outcome. Psychooncology. 22, 276–289. 10.1002/pon.2080 [DOI] [PubMed] [Google Scholar]
- Burke W., Atkins D., Gwinn M., Guttmacher A., Haddow J., Lau J., et al. (2002). Genetic test evaluation: Information needs of clinicians, policy makers, and the public. Am. J. Epidemiol. 156, 311–318. 10.1093/aje/kwf055 [DOI] [PubMed] [Google Scholar]
- Cameron L. D., Muller C. (2009). Psychosocial aspects of genetic testing. Curr. Opin. Psychiatry 22, 218–223. 10.1097/YCO.0b013e3283252d80 [DOI] [PubMed] [Google Scholar]
- Cassidy M. R., Roberts J. S., Bird T. D., Steinbart E. J., Cupples L. A., Chen C. A., et al. (2008). Comparing test-specific distress of susceptibility versus deterministic genetic testing for Alzheimer's disease. Alzheimer's Dement. 4, 406–413. 10.1016/j.jalz.2008.04.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chao S., Roberts J. S., Marteau T. M., Silliman R., Cupples L. A., Green R. C. (2008). Health behavior changes after genetic risk assessment for Alzheimer disease: The REVEAL Study. Alzheimer Dis. Assoc. Disord. 22, 94–97. 10.1097/WAD.0b013e31815a9dcc [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christiaans I., Van Langen I. M., Birnie E., Bonsel G. J., Wilde A. A. M., Smets E. M. A. (2009). Quality of life and psychological distress in hypertrophic cardiomyopathy mutation carriers: a cross-sectional cohort study. Am. J. Med. Genet. Part A 149, 602–612. 10.1002/ajmg.a.32710 [DOI] [PubMed] [Google Scholar]
- Claes E., Evers-Kiebooms G., Boogaerts A., Decruyenaere M., Denayer L., Legius E. (2004). Diagnostic genetic testing for hereditary breast and ovarian cancer in cancer patients: women's looking back on the pre-test period and a psychological evaluation. Genet. Test. 8, 13–21. 10.1089/109065704323015996 [DOI] [PubMed] [Google Scholar]
- Claes E., Evers-Kiebooms G., Denayer L., Decruyenaere M., Boogaerts A., Philippe K., et al. (2005). Predictive genetic testing for hereditary breast and ovarian cancer: psychological distress and illness representations 1 year following disclosure. J. Genet. Couns. 14, 349–363. 10.1007/s10897-005-1371-4 [DOI] [PubMed] [Google Scholar]
- Collins F. S., McKusick V. A. (2001). Implications of the Human Genome Project for medical science. JAMA 285, 540–544. 10.1001/jama.285.5.540 [DOI] [PubMed] [Google Scholar]
- Cornel M. C., Van El C. G., Borry P. (2014). The challenge of implementing genetic tests with clinical utility while avoiding unsound applications. J. Community Genet. 5, 7–12. 10.1007/s12687-012-0121-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craufurd D. I., Harris R. (1986). Ethics of predictive testing for Huntington's chorea: the need for more information. Br. Med. J. (Clin. Res. Ed). 293, 249–251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curtis C., Shah S. P., Chin S.-F., Turashvili G., Rueda O. M., Dunning M. J., et al. (2012). The genomic and transcriptomic architecture of 2,000 breast tumours reveals novel subgroups. Nature 486, 346–352. 10.1038/nature10983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decruyenaere M., Evers-Kiebooms G., Cloostermans T., Boogaerts A., Demyttenaere K., Dom R., et al. (2003). Psychological distress in the 5-year period after predictive testing for Huntington's disease. Eur. J. Hum. Genet. 11, 30–38. 10.1038/sj.ejhg.5200913 [DOI] [PubMed] [Google Scholar]
- Désesquelles A., Demuru E., Pappagallo M., Salvatore M. A., Frova L., Meslé F., et al. (2014). Mortality from Alzheimer' s disease, Parkinson' s disease, and dementias in france and Italy : a comparison using the multiple approach. J. Aging Health 26, 283–315. 10.1177/0898264313514443 [DOI] [PubMed] [Google Scholar]
- Di Prospero L. S., Seminsky M., Honeyford J., Doan B., Franssen E., Meschino W., et al. (2001). Psychosocial issues following a positive result of genetic testing for BRCA1 and BRCA2 mutations: findings from a focus group and a needs-assessment survey. CMAJ 164, 1005–1009. 10.1002/ijc.23340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiLorenzo T. A., Schnur J., Montgomery G. H., Erblich J., Winkel G., Bovbjerg D. H. (2006). A model of disease-specific worry in heritable disease: The influence of family history, perceived risk and worry about other illnesses. J. Behav. Med. 29, 37–49. 10.1007/s10865-005-9039-y [DOI] [PubMed] [Google Scholar]
- Dougall A. L., Smith A. W., Somers T. J., Posluszny D. M., Rubinstein W. S., Baum A. (2009). Coping with genetic testing for breast cancer susceptibility. Psychosom. Med. 71, 98–105. 10.1097/PSY.0b013e318190d7b4 [DOI] [PubMed] [Google Scholar]
- Erkkinen M. G., Kim M.-O., Geschwind M. D. (2018). Clinical neurology and epidemiology of the major neurodegenerative diseases. Cold Spring Harb. Perspect. Biol. 10:a033118. 10.1101/cshperspect.a033118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ertmanski S.ł., Metcalfe K., TrempaŁa J., GŁowacka M. D., Lubinski J., Narod S. A., et al. (2009). Identification of patients at high risk of psychological distress after BRCA1 genetic testing. Genet. Test. Mol. Biomarkers 13, 325–330. 10.1089/gtmb.2008.0126 [DOI] [PubMed] [Google Scholar]
- Evans D. G., Barwell J., Eccles D. M., Collins A., Izatt L., Jacobs C., et al. (2014). The Angelina Jolie effect: how high celebrity profile can have a major impact on provision of cancer related services. Breast Cancer Res. 16, 442. 10.1186/s13058-014-0442-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evers-Kiebooms G., Decruyenaere M. (1998). Predictive testing for Huntington's disease: a challenge for persons at risk and for professionals. Patient Educ. Couns. 35, 15–26. [DOI] [PubMed] [Google Scholar]
- Ferlay J., Soerjomataram I., Dikshit R., Eser S., Mathers C., Rebelo M., et al. (2015). Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 136, E359–E386. 10.1002/ijc.29210 [DOI] [PubMed] [Google Scholar]
- Gargiulo M., Lejeune S., Tanguy M.-L., Lahlou-Laforêt K., Faudet A., Cohen D., et al. (2009). Long-term outcome of presymptomatic testing in Huntington disease. Eur. J. Hum. Genet. 17, 165–171. 10.1038/ejhg.2008.146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giardino D., Mingarelli R., Lauretti T., Amoroso A., Larizza L., Dallapiccola B. (2016). Survey of medical genetic services in Italy: year 2011. BMC Health Serv. Res. 16:96. 10.1186/s12913-016-1340-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman J. S., Hahn S. E., Catania J. W., LaRusse-Eckert S., Butson M. B., Rumbaugh M., et al. (2011). Genetic counseling and testing for Alzheimer disease: joint practice guidelines of the American College of Medical Genetics and the National Society of Genetic Counselors. Genet. Med. 13, 597–605. 10.1097/GIM.0b013e31821d69b8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez C., Gomes E., Kazachkova N., Bettencourt C., Raposo M., Kay T. T., et al. (2012). Psychological well-being and family satisfaction levels five years after being confirmed as a carrier of the Machado-Joseph disease mutation. Genet. Test. Mol. Biomarkers 16, 1363–1368. 10.1089/gtmb.2011.0370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gooding H. C., Linnenbringer E. L., Burack J., Roberts J. S., Green R. C., Biesecker B. B. (2006). Genetic susceptibility testing for Alzheimer disease: Motivation to obtain information and control as precursors to coping with increased risk. Patient Educ. Couns. 64, 259–267. 10.1016/j.pec.2006.03.002 [DOI] [PubMed] [Google Scholar]
- Green R. C., Roberts J. S., Cupples L. A., Relkin N. R., Whitehouse P. J., Brown T., et al. (2009). Disclosure of APOE genotype for risk of Alzheimer's disease. N. Engl. J. Med. 361, 245–254. 10.1056/NEJMoa0809578 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagberg A., Bui T. H., Winnberg E. (2011). More appreciation of life or regretting the test? Experiences of living as a mutation carrier of Huntington's disease. J. Genet. Couns. 20, 70–79. 10.1007/s10897-010-9329-6 [DOI] [PubMed] [Google Scholar]
- Hamilton R., Williams J. K., Skirton H., Bowers B. J. (2009). Living with genetic test results for hereditary breast and ovarian cancer. J. Nurs. Scholarsh. 41, 276–283. 10.1111/j.1547-5069.2009.01279.x [DOI] [PubMed] [Google Scholar]
- Hendriks K. S. W. H., Hendriks M. M. W. B., Birnie E., Grosfeld F. J. M., Wilde A., van den Bout J., et al. (2008). Familial disease with a risk of sudden death: a longitudinal study of the psychological consequences of predictive testing for long QT syndrome. Heart Rhythm 5, 719–724. 10.1016/j.hrthm.2008.01.032 [DOI] [PubMed] [Google Scholar]
- Heshka J. T., Palleschi C., Howley H., Wilson B., Wells P. S. (2008). A systematic review of perceived risks, psychological and behavioral impacts of genetic testing. Genet. Med. 10, 19–32. 10.1097/GIM.0b013e31815f524f [DOI] [PubMed] [Google Scholar]
- Hickey K. T., Sciacca R. R., Biviano A. B., Whang W., Dizon J. M., Garan H., et al. (2014a). The effect of cardiac genetic testing on psychological well-being and illness perceptions. Hear. Lung J. Acute Crit. Care 43, 127–132. 10.1016/j.hrtlng.2014.01.006 [DOI] [PubMed] [Google Scholar]
- Hickey K. T., Taylor J. Y., Sciacca R. R., Aboelela S., Gonzalez P., Castillo C., et al. (2014b). Cardiac genetic testing: a single-center pilot study of a Dominican population. Hisp. Health Care Int. 12, 183–188. 10.1891/1540-4153.12.4.183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hietaranta-Luoma H.-L., Luomala H. T., Puolijoki H., Hopia A. (2015). Using ApoE genotyping to promote healthy lifestyles in Finland–psychological impacts: randomized controlled trial. J. Genet. Couns. 24, 908–921. 10.1007/s10897-015-9826-8 [DOI] [PubMed] [Google Scholar]
- Hollands G. J., French D. P., Griffin S. J., Prevost A. T., Sutton S., King S., et al. (2016). The impact of communicating genetic risks of disease on riskreducing health behaviour: Systematic review with meta-analysis. BMJ 352. 10.1136/bmj.i1102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horowitz M. J., Field N. P., Zanko A., Donnelly E. F., Epstein C., Longo F. (2001). Psychological impact of news of genetic risk for Huntington disease. Am. J. Med. Genet. 103, 188–192. 10.1002/ajmg.1538 [DOI] [PubMed] [Google Scholar]
- Janssens S., Chokoshvili D., Vears D. F., De Paepe A., Borry P. (2017). Pre- and post-testing counseling considerations for the provision of expanded carrier screening: Exploration of European geneticists' views. BMC Med. Ethics 18:46. 10.1186/s12910-017-0206-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones D. L., Clayton E. W. (2012). The role of distress in uptake and response to predisposition genetic testing: the BMPR2 experience. Genet. Test. Mol. Biomarkers 16, 203–209. 10.1089/gtmb.2011.0059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katapodi M. C., Northouse L., Pierce P., Milliron K. J., Liu G., Merajver S. D. (2011). Differences between women who pursued genetic testing for hereditary breast and ovarian cancer and their at-risk relatives who did not. Oncol. Nurs. Forum 38, 572–581. 10.1188/11.ONF.572-581 [DOI] [PubMed] [Google Scholar]
- Khoury M. J., Yang Q., Gwinn M., Little J., Flanders W. D. (2004). An epidemiologic assessment of genomic profiling for measuring susceptibility to common diseases and targeting interventions. Genet. Med. 6, 38–47. 10.1097/01.GIM.0000105751.71430.79 [DOI] [PubMed] [Google Scholar]
- Lammens C. R. M., Aaronson N. K., Wagner A., Sijmons R. H., Ausems M. G. E. M., Vriends A. H. J. T., et al. (2010). Genetic testing in Li-Fraumeni syndrome: Uptake and psychosocial consequences. J. Clin. Oncol. 28, 3008–3014. 10.1200/JCO.2009.27.2112 [DOI] [PubMed] [Google Scholar]
- Larsson M. U., Luszcz M., a Bui, T.-H., Wahlin T.-B. R. (2006). Depression and suicidal ideation after predictive testing for Huntington's disease: a two-year follow-up study. J. Genet. Couns. 15, 361–374. 10.1007/s10897-006-9027-6 [DOI] [PubMed] [Google Scholar]
- Legnani C., Razzaboni E., Gremigni P., Bitti P. E. R., Favaretto E., Palareti G. (2006). Psychological impact of testing for thrombophilic alterations. Thromb. Haemost. 96, 348–355. 10.1160/TH06-01-0015 [DOI] [PubMed] [Google Scholar]
- Levenson R. W., Sturm V. E., Haase C. M. (2014). Emotional and behavioral symptoms in neurodegenerative disease: a model for studying the neural bases of psychopathology. Annu. Rev. Clin. Psychol. 10, 581–606. 10.1146/annurev-clinpsy-032813-153653 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Licklederer C., Wolff G., Barth J. (2008). Mental health and quality of life after genetic testing for huntington disease: a long-term effect study in Germany. Am. J. Med. Genet. Part A 146, 2078–2085. 10.1002/ajmg.a.32423 [DOI] [PubMed] [Google Scholar]
- Linnenbringer E., Roberts J. S., Hiraki S., Cupples L. A., Green R. C. (2010). “I know what you told me, but this is what I think:” perceived risk of Alzheimer disease among individuals who accurately recall their genetics-based risk estimate. Genet. Med. 12, 219–227. 10.1097/GIM.0b013e3181cef9e1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodder L., Frets P. G., Trijsburg R. W., Meijers-Heijboer E. J., Klijn J. G. M., Duivenvoorden H. J., et al. (2001a). Psychological impact of receiving a BRCA1/BRCA2 test result. Am. J. Med. Genet. Part A 98, 15–24. [PubMed] [Google Scholar]
- Lodder L., Frets P. G., Willem Trijsburg R., Johanna Meijers-Heijboer E., Klijn J. G. M., Duivenvoorden H. J., et al. (2001b). Psychological impact of receiving a BRCA1/BRCA2 test result. Am. J. Med. Genet. 98, 15–24. [DOI] [PubMed] [Google Scholar]
- Mackenbach J. P., Karanikolos M., Looman C. W. N. (2014). The rise of mortality from mental and neurological diseases in Europe, 1979-2009: observational study. BMC Public Health 14:840. 10.1186/1471-2458-14-840 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manchanda R., Loggenberg K., Sanderson S., Burnell M., Wardle J., Gessler S., et al. (2015). Population testing for cancer predisposing BRCA1/BRCA2 mutations in the ashkenazi-jewish community: a randomized controlled trial. J. Natl. Cancer Inst. 107. 10.1093/jnci/dju379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marteau T., Senior V., Humphries S. E., Bobrow M., Cranston T., Crook M., a, et al. (2004). Psychological impact of genetic testing for familial hypercholesterolemia within a previously aware population: a randomized controlled trial. Am. J. Med. Genet. A 128A, 285–293. 10.1002/ajmg.a.30102 [DOI] [PubMed] [Google Scholar]
- Meiser B., Butow P., Friedlander M., Barratt A., Schnieden V., Watson M., et al. (2002). Psychological impact of genetic testing in women from high-risk breast cancer families. Eur. J. Cancer 38, 2025–2031. 10.1016/S0959-8049(02)00264-2 [DOI] [PubMed] [Google Scholar]
- Morris M. C., Evans D., a Bienias, J. L., Tangney C. C., Bennett D., a, Aggarwal N., et al. (2002). Dietary intake of antioxidant nutrients and the risk of incident Alzheimer disease in a biracial community study. JAMA 287, 3230–3237. 10.1001/jama.287.24.3230 [DOI] [PubMed] [Google Scholar]
- Myers R. H. (2004). Huntington' s Disease Genetics. Am. Soc. Exp. Neurother. 1, 255–262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveri S., Howard H. C., Renzi C., Hansson M. G., Pravettoni G. (2016a). Anxiety delivered direct-to-consumer: are we asking the right questions about the impacts of DTC genetic testing? J. Med. Genet. 53, 798–799. 10.1136/jmedgenet-2016-104184 [DOI] [PubMed] [Google Scholar]
- Oliveri S., Pravettoni G. (2016). The disclosure of direct-to-consumer genetic testing: sounding out the psychological perspective of consumers. Biol. Med. 8, 1–4. 10.4172/0974-8369.1000316 [DOI] [Google Scholar]
- Oliveri S., Pravettoni G., Fioretti C., Hansson M. G. (2016b). Let the individuals directly concerned decide: a solution to tragic choices in genetic risk information. Public Health Genomics 19, 307–313. 10.1159/000448913 [DOI] [PubMed] [Google Scholar]
- Oliveri S., Renzi C., Masiero M., Pravettoni G. (2015). Living at risk: factors that affect the experience of direct-to-consumer genetic testing. Mayo Clin. Proc. 90, 1323–1326. 10.1016/j.mayocp.2015.06.014 [DOI] [PubMed] [Google Scholar]
- Perry T. L. (1981). Some ethical problems in Huntington's chorea. Can. Med. Assoc. J. 125, 1098. [PMC free article] [PubMed] [Google Scholar]
- Reichelt J. G., Heimdal K., Møller P., Dahl A. A. (2004). BRCA1 testing with definitive results: a prospective study of psychological distress in a large clinic-based sample. Fam. Cancer 3, 21–28. 10.1023/B:FAME.0000026820.32469.4a [DOI] [PubMed] [Google Scholar]
- Rini C., O'Neill S. C., Valdimarsdottir H., Goldsmith R. E., Jandorf L., Brown K., et al. (2009). Cognitive and emotional factors predicting decisional conflict among high-risk breast cancer survivors who receive uninformative BRCA1/2 results. Health Psychol. 28, 569–578. 10.1037/a0015205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robins Wahlin T. B., Backman L., Lundin A., Haegermark A., Winblad B., Anvret M. (2000). High suicidal ideation in persons testing for Huntington's disease. Acta Neurol Scand 102, 150–161. 10.1034/j.1600-0404.2000.102003150.x [DOI] [PubMed] [Google Scholar]
- Romero L. J., Garry P. J., Schuyler M., Bennahum D. A., Qualls C., Ballinger L., et al. (2005). Emotional responses to APO E genotype disclosure for Alzheimer disease. J. Genet. Couns. 14, 141–150. 10.1007/s10897-005-4063-1 [DOI] [PubMed] [Google Scholar]
- Roth G. A., Johnson C., Abajobir A., Abd-Allah F., Abera S. F., Abyu G., et al. (2017). Global, regional, and national burden of cardiovascular diseases for 10 causes, 1990 to 2015. J. Am. Coll. Cardiol. 70, 1–25. 10.1016/j.jacc.2017.04.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samson A., DiMillo J., Thériault A., Lowry S., Corsini L., Verma S., et al. (2014). Living with the BRCA1 and BRCA2 genetic mutation: Learning how to adapt to a virtual chronic illness. Psychol. Health Med. 19, 103–114. 10.1080/13548506.2013.779729 [DOI] [PubMed] [Google Scholar]
- Shaw J. S., Bassi K. L. (2001). Lay attitudes toward genetic Testing for susceptibility to inherited diseases. J. Health Psychol. 6, 405–423. 10.1177/135910530100600404 [DOI] [PubMed] [Google Scholar]
- Shiloh S., Dagan E., Friedman I., Blank N., Friedman E. (2013). A follow-up study on men tested for BRCA1/BRCA2 mutations: impacts and coping processes. Psychooncology. 22, 417–425. 10.1002/pon.2106 [DOI] [PubMed] [Google Scholar]
- Smith A. W., Dougall A. L., Posluszny D. M., Somers T. J., Rubinstein W. S., Baum A. (2008). Psychological distress and quality of life associated with genetic testing for breast cancer risk. Psychooncology 17, 767–773. 10.1002/pon.1291 [DOI] [PubMed] [Google Scholar]
- Su P. (2013). Direct-to-consumer genetic testing: a comprehensive view. Yale J. Biol. Med. 86, 359–365. [PMC free article] [PubMed] [Google Scholar]
- Surampalli A., Khare M., Kubrussi G., Wencel M., Tanaja J., Donkervoort S., et al. (2015). Psychological impact of predictive genetic testing in VCP inclusion body myopathy, paget disease of bone and frontotemporal dementia. J. Genet. Couns. 24, 842–850. 10.1007/s10897-015-9819-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzman R., Beard J. R., Boerma T., Chatterji S. (2015). Health in an ageing world—what do we know? Lancet 385, 484–486. 10.1016/S0140-6736(14)61597-X [DOI] [PubMed] [Google Scholar]
- Timman R., Roos R., Maat-Kievit A., Tibben A. (2004). Adverse effects of predictive testing for Huntington disease underestimated: long-term effects 7-10 years after the test. Health Psychol. 23, 189–197. 10.1037/0278-6133.23.2.189 [DOI] [PubMed] [Google Scholar]
- Van Maarle M. C., Stouthard M. E. A., Marang -V, d. Mheen P. J., Klazinga N. S., Bonsel G. J. (2001). How disturbing is it to be approached for a genetic cascade screening programme for familial hypercholesterolaemia?: psychological impact and screenees' views. Community Genet. 4, 244–252. 10.1159/000064200 [DOI] [PubMed] [Google Scholar]
- Vernarelli J. A., Roberts J. S., Hiraki S., Chen C. A., Cupples L. A., Green R. C. (2010). Effect of Alzheimer disease genetic risk disclosure on dietary supplement use. Am. J. Clin. Nutr. 91, 1402–1407. 10.3945/ajcn.2009.28981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vos J., Oosterwijk J. C., Gomez-Garcia E., Menko F. H., Collee M. J., van Asperen C. J., et al. (2012). Exploring the short-term impact of DNA-testing in breast cancer patients: The counselees' perception matters, but the actual BRCA1/2 result does not. Patient Educ. Couns. 86, 239–251. 10.1016/j.pec.2011.04.017 [DOI] [PubMed] [Google Scholar]
- Wade C. H., Shiloh S., Woolford S. W., Roberts J. S., Alford S. H., Marteau T. M., et al. (2012). Modelling decisions to undergo genetic testing for susceptibility to common health conditions: an ancillary study of the Multiplex Initiative. Psychol. Health 27, 430–444. 10.1080/08870446.2011.586699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walter F. M., Emery J. D. (2012). Genetic advances in medicine: has the promise been fulfilled in general practice? Br. J. Gen. Pract. 62, 120–121. 10.3399/bjgp12X629955 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C., O'Neill S. M., Rothrock N., Gramling R., Sen A., Acheson L. S., et al. (2009). Comparison of risk perceptions and beliefs across common chronic diseases. Prev. Med. (Baltim). 48, 197–202. 10.1016/j.ypmed.2008.11.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO and FAO (2003). Diet, Nutrition and the Prevention of Chronic Diseases. WHO and FAO. [PubMed] [Google Scholar]
- Witjes-Ané M. N. W., Zwinderman A. H., Tibben A., van Ommen G. J. B., Roos R. A. C. (2002). Behavioural complaints in participants who underwent predictive testing for Huntington's disease. J. Med. Genet. 39, 857–862. 10.1136/jmg.39.11.857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organisation (2014). Diet, Nutrition and the Prevention of Chronic Diseases. WHO Tech. Rep. Ser. No. 916. Available online at: http://www.who.int/dietphysicalactivity/publications/trs916/summary/en/ [PubMed]
- Yang Q., Khoury M. J., Botto L., Friedman J. M., Flanders W. D. (2003). Improving the prediction of complex diseases by testing for multiple disease-susceptibility genes. Am. J. Hum. Genet. 72, 636–649. 10.1086/367923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon P. (2005). CDC's family history public health initiative: 2005 update in Genomics and Population Health 2005, eds Gwinn M., Bedrosian S., Ottman D., Khoury M. J. (Atlanta, GA: Centers for Disease Control and Prevention, Office of Genomics and Disease Prevention; ), 11–13. [Google Scholar]
- Yoon P., Scheuner M. (2003). The family history public health initiative in Genomics and Population Health: United States 2004, eds Gwinn M., Bedrosian S., Ottman D., Khoury M. J. (Atlanta, GA: Centers for Disease Control and Prevention, Office of Genomics and Disease Prevention; ), 39–45. [Google Scholar]