Abstract
The ability to recognize and appreciate from a reproductive standpoint that males and females possess different attributes has been long standing. Only more recently have we begun to look more deeply into both the similarities and differences between men and women, as well as between boys and girls, with respect to the structure and function of other organ systems. This article focuses on the cardiovascular system, with examples of sex differences in the control of coronary function, blood pressure, and volume. Recognizing the differences between the sexes with respect to cardiovascular function facilitates understanding of the mechanisms whereby homeostasis can be achieved using different contributions or components of the living system. Furthermore, recognition of the differences as well as the similarities permits the design of appropriate diagnostic instruments, recognition of sex-specific pathophysiology, and implementation of appropriate treatment of cardiovascular disease in men and women.
Keywords: heart rate, blood pressure, volume homeostasis, microvascular function
sometimes, differences are, simply, annoying. Sometimes, they are illuminating and provide insight into hitherto hidden mechanisms. So it appears to be when considering the cardiovascular function and dysfunction of women and men. The purpose of this article is to highlight the features of cardiovascular function (e.g., pumping of the heart, blood flow control, pressure regulation, and solute delivery) and manifestation of dysfunction (e.g., symptomology, prevalence, and comorbitities) in men and women with the dual intent of learning more about how the integrated system functions and identifying fundamental questions yet to be answered.
It comes as little surprise to learn that the size of the heart and major blood vessels of women are smaller than those of men of the same race and age (39). The anatomic location of the major vessels in the heart, lung, and most organs are indistinguishable among the sexes. Similarly, the cardiovascular systems of males and females have the same formed elements circulating in the blood, the vessels are composed of the same cell types, and the union of the elements perform the same functions. Healthy men and women are in homeostasis. Consequently, it makes sense to conclude that the functions and functional responses of all of these common elements are the same in men and women, right? Well, no, not necessarily, and that is where the tale becomes interesting. The take-home message of this monograph will be that while men and women are indeed in homeostasis and they possess the same structural elements, how those components function to achieve homeostasis with respect to the cardiovascular system differs (from subtly to profoundly). The manifestation of these differences has a real outcome: how and when cardiovascular disease occurs, how disease can be prevented, and what can be done to ameliorate or treat its manifestations effectively. As physiology students, the functional differences as well as the similarities are of utmost importance as they provide fundamental insight into the mechanisms regulating the integrated functions of the intact organism.
At the core, few students of physiology are surprised that men and women can differ. At the very least, males produce testosterone and females produce the sex hormones estrogen and progesterone. What has become evident, with respect to the cardiovascular system, is that the differences and similarities involve more than the sex hormones, per se. It is essential to realize that every cell has a “sex” thanks to the presence of either an XX or an XY chromosome, that this sex is manifested in the womb, and that, beyond behavioral differences, prepubertal differences exist between the cardiovascular systems of boys and girls.
History
Before going further, it is important to understand historically how we have failed to recognize more widely that the physiology of males and females might differ. Part of the reason is that the “women’s cycle,” e.g., the sex hormone status of females, changes in a cyclical fashion over the lunar month; thus, when experiments are designed, the question has been how to make observations against a changing background. The status of the reproductive hormone testosterone was viewed as relatively constant; therefore, males provided a “cleaner model” for the study of cardiovascular function. In fact, serum testosterone levels rise to 20% above mean levels (500 ng/dl), peaking at 20:00 hours, and fall to 35% below mean levels, at 7:00 hours, in healthy men daily (16). While a circadian rhythm is maintained with age, the mean levels of younger men (~25 yr) are ~100 ng/dl higher than those of healthy elderly (70 yr) men, and the excursion from the nadir to peak is greater at the younger age. In men, a state similar to menopause (e.g., andropause) has not been demonstrated convincingly. Thus, men not only cycle, but cycle faster, for longer than women.
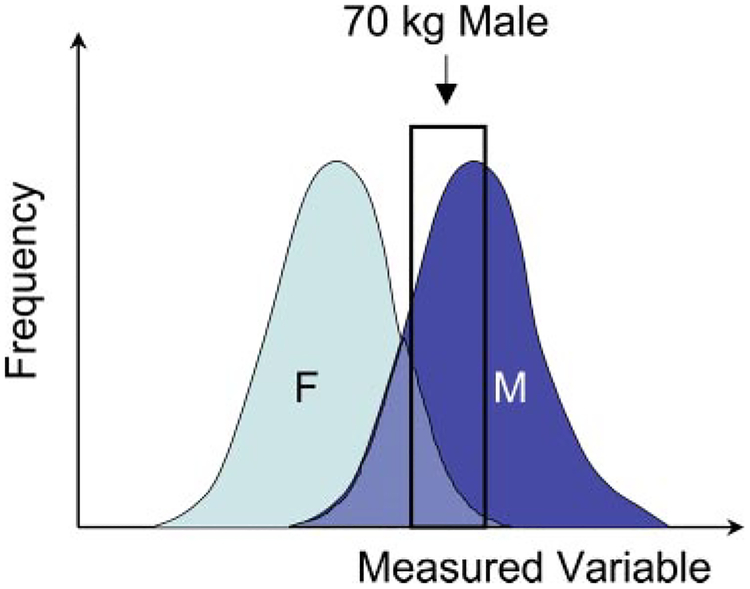
We need to also think further on the characteristics of the subjects tested whose data occupy the tables and graphs in handbooks, medical “scut” books, textbooks, encyclopedias, and internet files. Much of the human data found in medical texts represent the environment in which the testing was conducted. Medical schools and military institutions located largely in Europe and North America have provided the largest numbers of “healthy” individuals in which to make measures of function. Consequently, most of the data represent young (18–22 yr), healthy, 70-kg Caucasian males. The majority of studies of human cardiovascular disease have been carried out, largely from the end of the 1800s through the 1970s. Until recently, these studies were conducted on males to “eliminate the confounding problem of cycling;” similarly, when cardiovascular adaptations to exercise, altitude, or space were the subjects, young Caucasian males who were now endurance-trained athletes again populated the data set. As shown in Fig. 1, the actual distribution of humans is broader than that represented by these subsets, and the distribution of females likely overlaps, but not coincides, with the distribution for males. While the data obtained from these subjects have greatly furthered our knowledge of physiology and pathophysiology, this information has been used to represent the behavior of humans as a collective rather than a selective. The consequences, as we will see, have led to a limited view of the mechanisms controlling organ and tissue function and to limitations in the diagnosis and care of humans with disease.
Fig. 1.
Sampling a subset of a population may represent only a portion of the population. Many of the data on healthy humans came from studies of Caucasians men of 18–22 yr of age that were part of the military, athletes, or European medical students. Depending on the variable measured, the degree of overlap between females (F) and males (M) could range from negligible to complete, and, a priori, it cannot be predicted whether they accurately reflect the responses of both sexes, even of the same age and race.
Sexual Dimorphism With Respect to Cardiac Function
The dual-chambered pump sending blood into the lungs for gas exchange and into the body for gas, water, and solute exchange appears, grossly, to just be smaller in women than in men (9, 17, 20a). In the living state, it is evident that this translates into a smaller stroke volume (SV) and thus a lower cardiac output (CO) (see Eq. 1). Less evident, until the living human is observed, is that the rate of pumping [heart rate (HR)] is greater in females than in males (38). Contrary to common belief, though, the HRs of male and female fetuses do not differ in utero (29). Given (as shown in Eq. 1) that the frequency of beats (HR; beats/time) multiplied by the amount ejected with each beat (SV; amount) is what determines CO (amount/time),
| (1) |
In studies of large numbers of normal healthy adults, even after accounting for the larger body surface area of men than women, the SV was ~10% smaller and HR similarly greater, resulting in no difference in cardiac index (volume·time−1·surface area−1)(7). The gradient in pressure across the systemic circulation between the aorta [or mean arterial pressure outside the left heart (Pa)] and the vena cava [coming into the right heart (Pv)] is a function of the amount of blood pumped into the systemic vasculature (CO) and the resistance to blood flow offered by the vessels of the tissues [total peripheral resistance (TPR)], as shown by Eq. 2:
| (2) |
In the normal circulatory system, Pv is on the order of 5% of Pa and is therefore considered negligible; under these conditions, the relationship can be rewritten to indicate that Pa is determined by the product of CO and TPR, as shown by Eq. 3:
| (3) |
Under conditions of cardiovascular stress (e.g., exercise, loud noises, or psychological stress) men respond by increasing mainly vascular resistance (TPR in Eqs. 2 and 3), which is manifested as an increase in mean Pa (blood pressure) (2), whereas women predominantly increase HR (Eq. 1), thereby increasing CO. In both cases, there is an appropriate cardiovascular response, but there are potentially different outcomes.
Recall further that the control of blood pressure involves actions of the autonomic nervous system. On the one hand, there is a sympathetic drive (by analogy, the accelerator) to the heart and periphery. Increases in sympathetic activity in the heart results in both elevated contractility, which leads to increases in SV, and increases in HR. Either or both actions lead to an increase in CO. In the periphery, increases in sympathetic activity results in resistance vessel constriction with an increase in TPR. Reduction or withdrawal of sympathetic activity is analogous to taking one’s foot off of the accelerator, thereby reducing SV, HR, and TPR. On the other hand, the parasympathetic (vagal) system (by analogy, the brake) slows the heart; withdrawal of parasympathetic activity unmasks the sympathetic drive.
Recent studies of blood pressure control and cardiac function in healthy men and women have demonstrated that women and men use the two arms of the baroreflex system differently. At all ages, women were found to have reduced sympathetic activity (reflected by lower TPR and Pa) and enhanced para-sympathetic activity relative to men. Similarly, men were found to have higher plasma norepinephrine levels than women(12). The consequence, though, was that in response to changes in body position (e.g., in response to fluid shifts), women appeared to be more vulnerable to orthostatic hypotension and fainting (2).
Should the stresses be maintained and the system adapts or remodels, pathophysiology will develop. Given that the different mechanisms put strains on different components of the cardiovascular system, the long-term consequences of repeated and prolonged exposure to stress in the conduit and resistance vessels of males is vessel remodeling, resulting in sustained hypertension with less tissue perfusion. The consequences of reductions in tissue perfusion can be appreciated when recalling that metabolically active cells of an organ are downstream of the constricting arterial vessels. In women, it is the heart rather than the large arterial vessels that takes the burden (32).
As mentioned above, women also respond to changes in body position, application of lower body negative pressure, prolonged standing, or return to gravity from outer space with fainting (orthostatic hypotension) as a consequence of the reduced ability to maintain venous return to the heart and CO (orthostatic intolerance) (5, 10, 27, 40). Exactly which mechanisms account for the sex differences in orthostatic intolerance or even normal control of blood pressure, while widely studied, are not known (11). It should be evident, though, that if men and women use different components of the cardiovascular system to achieve homeostasis, then a single approach to the treatment of dysfunction will not be optimal for both men and women.
In the general population, women are less likely to experience heart failure than men (14). The situation reverses itself following myocardial infarction, when it is women who are at greater risk than men to experience heart failure (36). From studies to determine left ventricular geometry and function in normal and hypertensive males and females by echocardiography while also controlling for differences in body size, HR, and age, it was confirmed that left ventricular systolic function (as deduced from measures of ejection fraction and fractional shortening by echocardiography) are influenced by the anatomic geometry of the left ventricle and that left ventricle geometry differs between men and women (7). Works of this type provided sex-specific data to define normal and patho-physiological cardiac function that can be used to correctly identify otherwise asymptomatic patients. In addition to the anatomic size differences between men’s and women’s hearts (31), the electrical activity of cardiac myocytes differ (35). In the intact beating heart, the ECG of women demonstrates a longer QT interval than that of men; prior to sexual maturity, the QT interval is similar, and it is after puberty that the interval in boys shortens to the times observed in adult males. There are also differences in the patterns of cardiac repolarization between males and females. It has been postulated that females have a lower density of subclasses of repolarizing potassium currents (Kr and Ks currents) and more variance in L-type calcium current in the different layers of the heart. The implications are that these differences, in turn, account for the sex-specific incidence in cardiac arrhythmias and differences in responses to drugs used to treat heart disease in men and women (1).
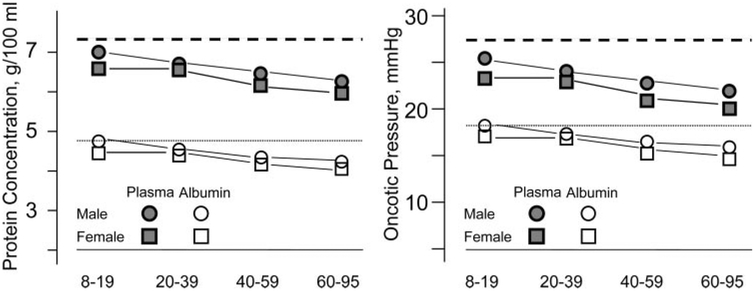
The composition of the blood circulating in the body also displays sex-specific differences in the levels of the formed elements, the most obvious being that lower numbers of circulating red blood cells per unit volume of plasma in females than in males (20a). This is manifested as lower hematocrit in women than in men. Both lipid and plasma protein compositions demonstrate sexual dimorphism. With respect to lipids, high-density lipoprotein (HDL) is higher and triglycerides are lower in females than in males; this “antiathrogenic” blood lipid profile is also associated with a lower incidence of cardiovascular disease. Following menopause, the lipid profile of females becomes more athrogenic and is correlated with the higher incidence of heart disease in that population (6). Less obvious is the lower plasma protein levels in females than in males (Fig. 2); the implications of this are discussed below.
Fig. 2.
Total plasma protein and plasma albumin concentrations for males and females at 4 ages. Data are redrawn from Ciba-Giegy data tables (20a) (A). In B, the data shown in A were used to construct a plot of the net plasma oncotic pressures for the same groups. Dashed lines represent the “normal” values for humans (20a).
With respect to hemodynamic responses, while most of the studies have been conducted on males, reexamination appears to be underway in several areas. One example is the response of the cardiovascular system to ingestion of a meal. Whereas mean arterial pressure is lower in females than in males, ingestion of a meal results in no change in females and a fall (reduced diastolic pressure) in males. Blood flow into the superior mesenteric artery is higher in females than in males; and, following a meal, it increases to a similar extent in both sexes, thereby maintaining the difference. In the portal venous system, no apparent differences exist for males and females, and for both sexes, venous flow increased to a similar extent in males and females. Although the control of blood flow within the splanchnic circulation for males was reflected in the systemic circulation by a drop in peripheral resistance, the same was not true in females.
With respect to cardiovascular disease, men are at greater risk, and their incidence of hypertension is greater than for premenopausal women of the same age (26). Ambulatory blood pressure measurements have consistently found that men have higher blood pressures than premenopausal, age-matched women (20). After menopause, the sex difference in the incidence of hypertension is lost (34). In animal models of hyper-tension, studies have also shown males to have higher blood pressure than females. For example, in Dahl salt sensitive-hypertensive rats, spontaneously hypertensive rats, and rats with reduced renal mass fed high-salt diets, males have higher blood pressures than females (18).
While the mechanisms underlying sex differences are poorly understood, there is significant evidence for a role for both testosterone in the development of and estrogen in the protection against high blood pressure (18). Evidence supporting the hypothesis of estrogen involvement in the differential expression of coronary vascular disease (CVD) and hypertension between men and women has been shown by several laboratories (18). While estrogen replacement therapy has been shown to reduce low-density lipoprotein-cholesterol and increase HDL levels, (18) these protective findings contrast with the findings of the Women’s Health Initiative (6, 22) and Heart and Estrogen/Progestin Replacement Studies I and II (15, 19), two clinical trials that failed to find a protective effect for hormone replacement therapy (HRT) against CVD. It has been suggested that the type of estrogen used in the replacement trials, the fact that a significant number of participants were obese, and that many of the women had already gone through menopause could account for these outcome discrepancies (34,37). Furthermore, it has been suggested that HRT started during perimenopause rather than after menopause may have provide CVD protection (28).
Having suggested that sex hormones play a role in the development of hypertension, it should be recognized that 24-h monitoring of blood pressures and HRs in pediatric patients has demonstrated differences between boys and girls. Systolic blood pressure was normally distributed for girls and skewed toward higher levels in boys; median levels only became statistically different at the onset of puberty. Diastolic blood pressures also tended to be skewed toward higher levels at night than during the day and showed a movement to low or very low levels in children approaching puberty (41). What is notable is that there were not only differences in absolute values between boys and girls but the time of day when those differences were apparent (day vs. night, for example) was significant. Overall, HR decreased almost linearly with age and to a greater extent in boys than in girls. In a study of children in Italy, HR and blood pressure were positively correlated in boys and girls, with HR being consistently higher in girls than boys. Having said that, HR was independently correlated age, sexual maturation, height, physical activity, and “parental sociocultural level” (31).
In response to exercise, 11-yr-old children showed circulatory responses similar to those of adults, but, compared with adult men, boys have lower CO and higher HR for a given maximal oxygen output than men. At each work level, CO was ~0.5 higher than the values for boys. In boys, the increase in CO was achieved by an increase in HR, whereas girls achieved their increase in CO by increasing SV and HR to a similar extent. Consistent with the previous studies on children and adults, girls had a higher HR than boys; the difference was maintained with increasing exercise, although the age, height, weight, and surface area of the children studied were not different between boys and girls (3).
Physiological Consequences of Sexual Dimorphism With Respect to Volume Balance
First of all, we know that healthy males and females are in volume balance. According to Starling’s law of filtration, volume balance between the vascular and tissue spaces is determined by two set of forces existing across the walls of the exchange vasculature. These forces expressed per unit surface area are given as pressures: hydrostatic and oncotic. The hydrostatic pressure of the vascular space is generated by the force of the beating of the heart (Pcap) and that of interstitial space between the cells of the tissue (Pint) is a product of the fluid captured within the organ. Given that the oncotic pressure is determined from van’t Hoff’s Law,
| (4) |
where n is the number of osmotically active particles, ϕ is an correction factor for the nonideality of particles in solution, and π is a function of the protein concentration (C) in the plasma (πp) and interstitial spaces (πi), respectively. The modern statement of Starling’s law is that volume flux (Jv; volume/time) occurs when there is a gradient in the forces across a barrier with hydraulic conductivity (Lp; “leakiness” to water, distance·time−1·unit pressure−1), surface area (S, distance squared), and reflection coefficient (σ) to protein as follows:
| (5) |
In a perfectly semipermeable membrane, σ to the primary protein constituent of plasma (albumin) is 1 (e.g., the full oncotic pressure is expressed); in organs such as the spleen or liver, where the spaces between endothelial cells are macroscopic, σ is 0 and no oncotic pressures are expressed as the protein concentrations in the vascular and tissue spaces are equal.
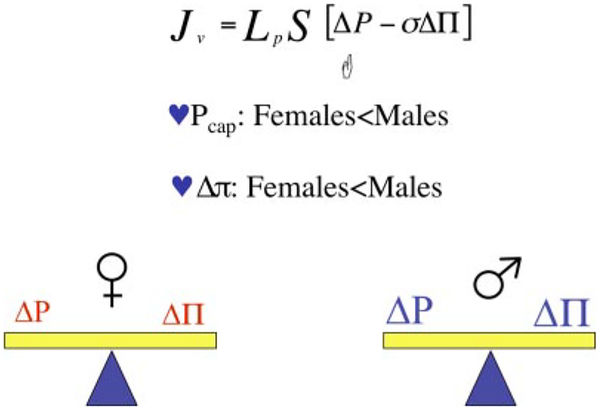
As shown in Fig. 2, there is also a difference in the protein content of plasma between women and men (20a). The discussion above should lead the reader to the conclusion that the net plasma oncotic pressure of blood in women will be less than in men, yet they are both in volume balance. As it happens, there are good physiological data demonstrating that not only is the mean arterial pressure of women less than that of men but that pressure in the exchange microvessels is also lower in women than men (33). The outcome is that in females, relative to males, there is a lower absorbing force (plasma oncotic pressure) in the exchange vessels that is offset by a lower filtering force (capillary hydrostatic pressure). In both cases, assuming that the hydrodraulic conductivities and surface areas of the vasculatures are equal, volume balance is maintained (Fig. 3). The other potential variable that may offset the pressures with respect to volume flux is the surface area for exchange, which, in turn, is a function of the size, number, and length of the microvessels in a whole organ. The actual pressures in those vessels is also a function of how the vessels are arranged in a network. In normal animals, it appears that microvessel density is not remarkably different between males and females.
Fig. 3.
Is the Starling balance the same for males and females? In normal males and females, yes, the Starling balance is maintained; apparently, in females, the lower oncotic pressure (Δπ) is offset by the lower capillary hydrostatic pressure (Pcap) relative to that of healthy normal males.
Remodeling of the network architecture has been observed in male, but not female, models of hypertension. In this pathophysiological state, males have fewer microvessels (rarefaction), resulting in changes in exchange surface area and TPR. In hypertensive humans, without respect to sex, forearm skin capillary density is reduced by ~20% compared with normotensive subjects and correlates inversely with blood pressure. Rarefaction of this magnitude, if present in all vascular beds, would result in an ~20% increase in TPR (30).
The contribution of sex in this process was studied in a hypertensive rat model of reduced renal mass plus feeding of a high-salt diet. In males, mean arterial pressure increased by >50% and skeletal muscle capillary density decreased by 33%, results similar to those observed in male normotensive and spontaneously hypertensive rats on a high-salt diet. In contrast, mean arterial pressures of females rose only slightly, and no changes in microvessel density were observed (29).
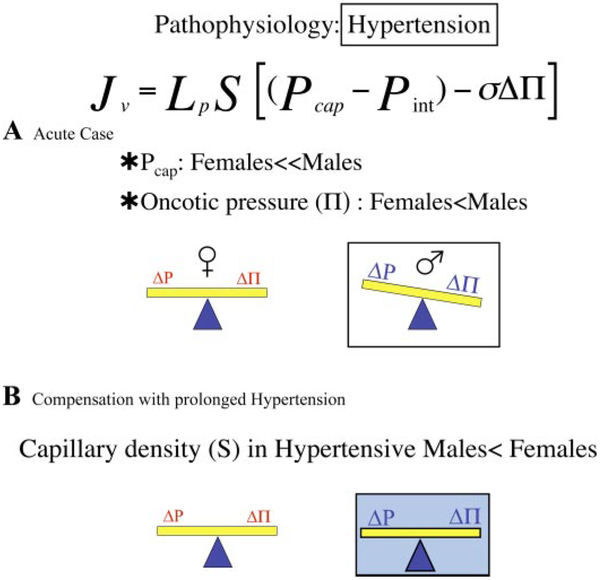
Returning to the discussion of fluid movement into tissue (volume flux, Eq. 5), fewer vessels implies reductions in both surface area and capillary pressure (or unchanged capillary pressure) relative to normotensive males, serving to maintain a constant flux of water in the face of an increase in arterial pressure. The modification in the network architecture appears to “protect” the exchange vasculature from an elevation in driving force. The problem with the compensatory mechanism is that “rebuilding” of the vasculature means that it is difficult to return blood pressure to “normal” as the variable resistance elements, the arterioles, have been removed. In addition, elevation of Pcap alone would be expected to result in an increase in filtration from the exchange microvasculature (leading to an increase in tissue water, edema) (Fig. 4A). Of interest, measures of the capillary filtration coefficient (CFC, the product of LpS) in the forearm of hypertensives averaged 41% of controls, and the relation between CFC and arterial blood pressure values is inversely nonlinear (30). In aggregate, whether there is a net change in fluid filtration in hypertensive relative to normal subjects will depend on the combination of the changes in driving force (capillary pressure), capillary area (rarefaction), and permeability properties (Fig. 4B). The mechanisms controlling these changes and the role of the sex hormones are poorly understood.
Fig. 4.
In hypertension, the Pcap of females, while raised, is still less than that of males, as is Δπ. Without any changes in the system [e.g., hydraulic conductivity (Lp) and surface area (S) remain as in health], the forces favor fluid movement into the tissues of males relative to females (A). While Δπ remains, there is a reduction in the total number of skeletal muscle microvessels in hypertensive males, resulting in a reduction in S in the face of a rise in Pcap, bringing the Starling forces back into balance in males and females (B).
Sexual Dimorphism in the Presentation of Cardiovascular Disease
We are all taught that there are three hallmarks of a heart attack:
Chest discomfort or uncomfortable pressure, fullness, squeezing, or pain in the center of the chest that lasts longer than a few minutes or that comes and goes.
Spreading pain to one or both arms, back, jaw, or stomach.
Cold sweats and nausea.
It turns out that these three hallmarks are experienced more often by men than by women, and, in fact, only one in three women have these symptoms when experiencing a myocardial infarction. Instead, women experience shortness of breath, nausea, vomiting, sleeplessness, back or jaw pain, and/or a feeling of generalized fatigue or weakness weeks prior to an acute myocardial infarction! The serious consequence is that appropriate treatment for women is delayed, inappropriate, or incorrect, leading to preventable deaths.
Current studies of disease processes and diagnostic procedures are demonstrating that there is a need not only to recognize that differences exist between men and women but also to perform appropriate studies documenting normal and pathophysiological parameters, especially as they relate to diagnostic procedures. Studies of this nature are leading to the evaluation of diagnostic modalities, such as exercise ECGs and cardiac-imaging modalities, with respect to sex differences to improve the diagnosis and risk assessment of women and men, specifically, with suspected cardiovascular disease (25).
Summary
Over the past 10 years, differences between the physiology of women and men have been recognized with respect to normal cardiovascular physiology as well as with respect to the incidence, severity, and comorbidities of obesity, diabetes, and cancer of cardiovascular disease. What is important, in addition to knowing that these differences exist, is the elucidation of the physiological mechanisms responsible for the differences as well as the similarities. With these data, appropriate treatments can be designed to maintain function as well as to treat disease.
Acknowledgments
GRANTS
This work was supported by National Institutes of Health Grant RO1-HL-078816, RO1-HL-075186, 1-CO6-RR-017353 and by National Aeronautics and Space Administration Grant NNJ05HF37G.
Footnotes
The costs of publication of this article were defrayed in part by the payment of page charges. The article must therefore be hereby marked “advertisement” in accordance with 18 U.S.C. Section 1734 solely to indicate this fact.
REFERENCES
- 1.Anthony M Male/female differences in pharmacology: safety issues with QT-prolonging drugs. J Womens Health 14: 47–52, 2005. [DOI] [PubMed] [Google Scholar]
- 2.Barnett SR, Morin RJ, Kiely DK, Gagnon M, Azhar G, Knight EL, Nelson JC, Lipsitz LA. Effects of age and gender on autonomic control of blood pressure dynamics. Hypertension 33: 1195–1200, 1999. [DOI] [PubMed] [Google Scholar]
- 3.Bar-or O, Shephard RJ, Allen C. Cardiac output of 10- to 13-year-old boys and girls during submaximal exercise. J Appl Physiol 30: 219–223, 1971. [DOI] [PubMed] [Google Scholar]
- 4.Bevegård S, Shepherd JT. Regulation of the circulation during exercise in man. Physiol Rev 47: 178–213, 1967. [DOI] [PubMed] [Google Scholar]
- 5.Buckey JC Jr, Lane LD, Levine BD, Watenpaugh DE, Wright SJ, Moore WE, Gaffney FA, Blomqvist CG. Orthostatic intolerance after spaceflight. J Appl Physiol 81: 7–18, 1996. [DOI] [PubMed] [Google Scholar]
- 6.Burry KA. Risks and benefits of estrogen plus progestin in healthy postmenopausal women. Principal results from the Women’s Health Initiative randomized controlled trial. Curr Women’s Health Rep 2: 331–332, 2002. [DOI] [PubMed] [Google Scholar]
- 7.Celentano A, Palmieri V, Arezzi E, Mureddu GF, Sabatella M, Di Minno G, De Simone G. Gender differences in left ventricular chamber and midwall systolic function in normotensive and hypertensive adults. J Hypertens 21: 1415–1423, 2003. [DOI] [PubMed] [Google Scholar]
- 8.Convertino VA. Gender differences in autonomic functions associated with blood pressure regulation. Am J Physiol Regul Integr Comp Physiol 275: R1909–R1920, 1998. [DOI] [PubMed] [Google Scholar]
- 9.De Simone G, Devereux RB, Roman MJ, Ganau A, Chien S, Alderman MH, Atlas S, Laragh JH. Gender differences in left ventricular anatomy, blood viscosity and volume regulatory hormones in normal adults. Am J Cardiol 68: 1704–1708, 1991. [DOI] [PubMed] [Google Scholar]
- 10.Frey MA, Hoffler GW. Association of sex and age with responses to lower-body negative pressure. J Appl Physiol 65: 1752–1756, 1988. [DOI] [PubMed] [Google Scholar]
- 11.Fu Q, Arbab-Zadeh A, Perhonen MA, Zhang R, Zuckerman JH, Levine BD. Hemodynamics of orthostatic intolerance: implications for gender differences. Am J Physiol Heart Circ Physiol 286: H449–H457, 2004. [DOI] [PubMed] [Google Scholar]
- 12.Geelen G, Laitinen T, Hartikainen J, Bergstrom K, Niskanen L. Gender influence on vasoactive hormones at rest and during a 70 degree head-up tilt in healthy humans. J Appl Physiol 92: 1401–1408, 2002. [DOI] [PubMed] [Google Scholar]
- 14.Gottdiener GS, Arnold AM, Aurigemma GP, Polak JF, Tracy RP, Kitzman DW, Gardin JM, Rutledge JE, Boineau RC. Predictors of congestive heart failure in the elderly: the cardiovascular health study. J Am Coll Cardiol 35: 1628–1637, 2000. [DOI] [PubMed] [Google Scholar]
- 15.Grady D, Herrington D, Bittner V, Blumenthal Davidson M, Hlatky M, Hsia J, Hulley S, Herd A, Khan S, Newby LK, Waters D, Vittinghoff E, Wenger N; for the HERS Research Group. Cardiovascular disease outcomes during 68 years of hormone therapy: Heart and Estrogen/Progestin Replacement Study follow-up (HERS II). JAMA 288: 49–57, 2002. [DOI] [PubMed] [Google Scholar]
- 16.Gupta SK, Lindemulder EA, Sathyan G. Modeling of circadian testosterone in healthy men and hypogonadal men. J Clin Pharmacol 40: 731–738, 2000. [DOI] [PubMed] [Google Scholar]
- 17.Hammond IW, Devereux RB, Alderman MH, Laragh JH. Relation of blood pressure and body build to left ventricular mass in normotensive and hypertensive employed adults. J Am Coll Cardiol 12: 996–1004, 1988. [DOI] [PubMed] [Google Scholar]
- 18.Hay M, Huxley VH. Sexual dimorphism in hypertension In: Molecular Mechanisms of Hypertension, edited by DiPette D, Schiffrin E, Re R, Sowers JR. Philadelphia, PA: Taylor & Francis, 2006, chapt. 42, p. 407–414. [Google Scholar]
- 19.Herrington DM. The HERS trial results: paradigms lost? Ann Intern Med 131: 463–466, 1999. [DOI] [PubMed] [Google Scholar]
- 20.Khoury S, Yarows SA, O’Brien TK, Sowers JR. Ambulatory blood pressure monitoring in a nonacademic setting. Effects of age and sex. Am J Hypertens 5: 616–623, 1992. [DOI] [PubMed] [Google Scholar]
- 20a.Lentner C (editor). Geigy Scientific Tables (8th ed.). Basel, Switzerland:Ciba-Geigy, vol. 3, 1982.21. [Google Scholar]
- 21.Levy D, Savage DD, Garrison RJ, Anderson KM, Kannel WB, Castelli WP. Echocardiographic criteria for left ventricular hypertrophy: the Framingham Heart Study. Am J Cardiol 59: 956–960, 1987 [DOI] [PubMed] [Google Scholar]
- 22.Manson J, Hsia J, Johnson KC, Rossouw JE, Assaf AR, Lasser NL, Trevisan M, Black HR, Heckbert SR, Detrano R, Strickland OL, Wong ND, Crouse JR, Stein E, Cushman M. Estrogen plus progestin and the risk of coronary heart disease. N Engl J Med 349: 523–534, 2003. [DOI] [PubMed] [Google Scholar]
- 23.Malyusz M, Ehrens HJ, Wrigge P. Effect of castration on the experimental renal hypertension in the rat. Blood pressure, nephrosclerosis, long-chain fatty acids, and N-acetylation of PAH in the kidney. Nephron 40: 96–99, 1985. [DOI] [PubMed] [Google Scholar]
- 24.McKenna DS, Ventolini G, Neiger R, Downing C. Gender-related differences in fetal heart rate during first trimester. Fetal Diagn Ther 21: 144–147, 2006. [DOI] [PubMed] [Google Scholar]
- 25.Mieres JH, Shaw LJ, Arai A, Budoff MJ, Flamm SD, Hundley WG, Marwick TH, Mosca L, Patel AR, Quinones MA, Redberg RF, Taubert KA, Taylor AJ, Thomas GS, Wenger NK,. Role of noninvasive testing in the clinical evaluation of women with suspected coronary artery disease. Circulation 111: 682–696, 2005. [DOI] [PubMed] [Google Scholar]
- 26.Messerli FH, Garavaglia GE, Schmieder RE, Sundgaard-Riise K, Nunez BD, Amodeo C. Disparate cardiovascular findings in men and women with essential hypertension. Ann Intern Med 107: 158–161, 1987. [DOI] [PubMed] [Google Scholar]
- 27.Montgomery LD, Kirk PJ, Payne PA, Gerber RL, Newton SD, Williams BA. Cardiovascular responses of men and women to lower body negative pressure. Aviat Space Environ Med 48: 138–145, 1977. [PubMed] [Google Scholar]
- 28.Naftolin F, Taylor H, Karas R, Brinton E, Newman I, Clarkson TB, Mendelsohn M, Lobo RA, Judelson DR, Nachtigall LE, Heward CB, Hecht H, Jaff MR, Harman SR. The Women’s Health Initiative could not have detected cardioprotective effects of starting hormone therapy during the menopausal transition. Fertil Steril 81: 1498–1501, 2004. [DOI] [PubMed] [Google Scholar]
- 29.Papanek PE, Rieder MJ, Lombard JH, Greene AS. Gender-specific protection from microvessel rarefaction in female hypertensive rats. Am J Hypertens 11: 998–1005, 1998. [DOI] [PubMed] [Google Scholar]
- 30.Prasad A, Dunnill GS, Mortimer PS, MacGregor GA. Capillary rarefaction in the forearm skin in essential hypertension. J Hypertens 13: 265–268, 1995. [PubMed] [Google Scholar]
- 31.Rabbia F, Grosso T, Cat Genova G, Conterno AA, De Vito B, Mulatero P, Chiandussi L, Veglio F. Assessing resting rate in adolescents: determinants and correlates. J Hum Hypertens 16: 327–332, 2002. [DOI] [PubMed] [Google Scholar]
- 32.Saba MM, Ibrahim MM, Rizk HH. Gender and the relationship between resting heart rate and left ventricular geometry. J Hypertens 19: 367–373, 2001. [DOI] [PubMed] [Google Scholar]
- 33.Shore AC, Sandeman DD, Tooke JE. Capillary pressure, pulse pressure amplitude, and pressure waveform in healthy volunteers. Am J Physiol Heart Circ Physiol 268: H147–H154, 1995. [DOI] [PubMed] [Google Scholar]
- 34.Staessen J, Bulpitt CJ, Fagard R, Lijnen P, Amery A. The influence of menopause on blood pressure. J Hum Hypertens 3: 427–433, 1989. [PubMed] [Google Scholar]
- 35.Surawicz B, Parikh SR. Differences between ventricular repolarization in men and women: description, mechanism and implications. Ann Noninvasive Electrocardiol 8: 333–340, 2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tofler GH, Stone PH, Muller JE, Willich SN, Davis VG, Poole WK, Strauss HW, Willerson JT, Jaffe AS, Robertson T. Effects of gender and race on prognosis after myocardial infarction: adverse prognosis for women, particularly black women. J Am Coll Cardiol 9: 473–482, 1987. [DOI] [PubMed] [Google Scholar]
- 37.Turgeon JL, McDonnell DP, Martin KA, Wise PM,. Hormone therapy: physiological complexity belies therapeutic simplicity. Science 304: 1269–1273, 2004. [DOI] [PubMed] [Google Scholar]
- 38.Umetani K, Singer Dh Mccraty R, Atkinson M. Twenty-four hour time domain heart rate variability and heart rate: relations to age and gender over nine decades. J Am Coll Cardiol 31: 593–601, 1998. [DOI] [PubMed] [Google Scholar]
- 39.Vasan RS, Larson MG, Levy D, Evans JC, Benjamin EJ. Distribution and categorization of echocardiographic measurements in relation to reference limits: The Framingham Heart Study. Formulation of a height-and sex-specific classification and its prospective validation. Circulation 96: 1863–1873, 1997. [DOI] [PubMed] [Google Scholar]
- 40.Waters WW, Ziegler MG, Meck JV. Postspaceflight orthostatic hypo-tension occurs mostly in women and is predicted by low vascular resistance. J Appl Physiol 92: 586–594, 2002. [DOI] [PubMed] [Google Scholar]
- 41.Wühl E, Witte K, Soergel M, Mehis O, Schaefer F; German Working Group on Pediatric Hypertension. Distribution of 24-h ambulatory blood pressure in children: normalized reference values and role of body dimensions. J Hypertens 20: 1995–2007, 2002. [DOI] [PubMed] [Google Scholar]