Abstract
Particle therapy can achieve excellent dose localization but is sensitive to range uncertainty. Therefore, online in-vivo range verification before treatment is critical for treatment safety and quality assurance. We introduce a novel range-probing technique that uses mid-range treatment spots selected from the treatment plan as probing beams to be delivered before other treatment spots in pencil beam scanning. The probing spot signal can be acquired by an in-beam positron emission tomography (PET) scanner, and the reconstructed spot positions are compared with pre-calculated positions to measure the range shift. Mid-range probing ensures that the Bragg peaks stay inside the tumor even with significant range variation from the plan. Single-layered spots enable easier spot detection than multi-layered spots without cross-layered spot smearing. With therapeutic dose, the probing beam offers higher positron activities and range detectability than the low-dose imaging beam by up to two orders of magnitude, without exposing patients to extra radiation. Higher positron activities allow sufficient signal statistics in shorter acquisition time, therefore reducing metabolic washout of positron emitters. Thus, range shifts from the plan can be measured easily. We also describe two online range-compensated plan modification methods. We apply correction, if the range shift is above a certain tolerance.
We studied feasibility using simulated particle treatment plans with online anatomical changes. For illustration, we demonstrate range shift measurement using simulated probing dose. The proposed range probing and correction effectively handled range shifts in the simulated cases. Both range-compensated adaptation and optimization accounted for online changes so that the delivered dose matched the planned dose.
With a dedicated online in-beam PET scanner and phantom and clinical studies, which are currently being developed, this novel strategy may open up a range-guided particle therapy (RGPT1) paradigm.
Keywords: mid-range probing, intra-treatment adaptation, online range verification, range-guided particle therapy
1. Introduction
Particle beams, such as proton beams or carbon ion beams, have a characteristic Bragg peak along the depth dose curve with low entrance dose and steep dose falloff after the peak. Such characteristic allows particle therapy treatments to deliver highly localized dose but may strongly impact delivery accuracy. In clinical beams, a 5-mm shift of the Bragg peak can result in 50% underdosing at the distal part of the target (Verburg 2015) or significant overdosing to normal tissues distal to the target. The beam range (distal 80% of the Bragg peak) is indeed associated with considerable uncertainties, which can be ascribed to image conversion uncertainty and degeneracy, stopping power formula inaccuracy, minor setup errors, online anatomical changes, beam energy uncertainties, and dose calculation algorithms (Paganetti 2012). A major challenge in current particle therapy is to verify the beam range in the patient before treatment to ensure treatment safety and quality.
Image guidance such as online cone beam computed tomography (CBCT) has become a standard practice in radiotherapy treatment. Patient setup alignment may be achieved using online CBCT, but beam energy or modulator-related range uncertainties, and also anatomical changes-induced range uncertainties cannot be resolved through target alignment (Dong et al. 2012).
Positron emission tomography (PET) is a powerful tool for in situ and non-invasive dose monitoring in particle therapy (Enghardt et al. 2004) (Attanasi et al. 2009). Pre-treatment range verification using stable beams with low dose and PET imaging was pioneered in the 1970s at the Lawrence Berkeley National Laboratory (LBL), USA (Enghardt et al. 2004). Low radiation doses (~cGy or less) are required for non-therapeutic deliveries to minimize patient exposure to radiation. However, with low positron activity from low dose particle beams, PET images cannot be acquired with sufficient SNR within a short time to verify beam range accurately (Shao et al. 2014). Low dose radioactive beams 11C or 10C (Iseki et al. 2004) were used at the Heavy Ion Medical Accelerator in Chiba (HIMAC), Japan, but since the production rate of secondary radioactive ions is only in the order of 0.1–1% (Enghardt et al., 2004), their clinical application is impractical.
Recent studies using PET to detect short-lived isotopes (Dendooven et al. 2015) and improvements in PET scanners for online verification (Shao et al. 2014) with on-the-fly reconstruction (Ferrero et al. 2018) have shown the potential of PET imaging for timely online range measurement.
Other imaging techniques, including prompt gamma imaging (Krimmer et al. 2017), proton interaction vertex imaging (Henriquet et al. 2012), and thermo-acoustic imaging (Assmann et al. 2015), are also attractive particle beam delivery verification tools but challenged by signal statistic issues as well.
Techniques that use much higher energy proton beams (> 250 MeV) for range probing (Mumot et al. 2010) or proton radiography (Schneider et al. 1995) may be used for range verification but only provide total range change through the whole patient without range information at the target (Knopf et al. 2013). Proton CT, mainly studied for offline treatment planning, is of relevant interest, as it directly provides the stopping power values of the tissues; but it has major disadvantages, which are common to all proton transmission imaging techniques, in the requirement of high energy protons to penetrate the patient and challenges in spatial resolution (Knopf et al. 2013). Dual energy CT (Yang et al. 2010) is also competitive for offline treatment planning to reduce range uncertainty. However, all these techniques would involve exposing the patient to additional imaging dose.
The state-of-the-art techniques are insufficient for managing ranging uncertainty, indicating the need for new modalities with high signal statistics and spatial resolution for online pre-treatment range verification in particle therapy.
We propose a novel approach that exploits mid-range therapeutic beams directly for range probing in pencil beam scanning (Kanai et al. 1980) (Pedroni et al. 1995) (Lomax 1999). With this approach, the induced positron activities are stronger than those induced by pre-treatment imaging beams by up to two orders of magnitude, improving signal statistics and enabling shorter acquisition time when used in conjunction with a dedicated in-beam PET system (Shao et al. 2014). Therefore, the effect of complicated biological decay and washout of positron emitters can be largely reduced.
Currently, we are developing a dedicated PET system for mid-range probing. In this paper, we used probing beam dose as a surrogate of the detector image to illustrate the mid-range therapeutic probing beam method, and described range correction strategies and the workflow for range-guided particle therapy (RGPT).
2. Method
Since each treatment beam direction will be handled independently in a treatment plan, without loss of generality, we assume a treatment plan has only a single beam direction. The RGPT workflow (figure 1) includes the following automated steps in addition to treatment planning and patient setup: (1) after treatment planning, select a layer of mid-range spots from the planned spots as the probing spots and pre-calculate the probing spot positions for all spots in the mid-range layer, (2) after patient setup, deliver the probing spots, detect the activation signal, and reconstruct the probing spot positions, (3) compare detection with pre-calculation to delineate the range shift, and (4) deliver the remaining spots if the range shift is within tolerance, otherwise activate range correction and deliver range-compensated spots. Step (1) is completed offline and steps (2)-(4) are completed online.
Figure 1.
RGPT workflow. The shaded blocks indicate automatic steps unique to RGPT.
2.1. Mid-range therapeutic probing beams
Particle treatment delivery by pencil beam scanning can be realized in discrete (spot scanning), continuous (line scanning), or in-between (raster scanning) modes (Paganetti et al. 2005). For convenience, the descriptions are in spot scanning but can be translated directly to the other two modes.
A treatment plan consists of multiple layers of treatment spots, each layer corresponding to single beam energy and each spot specified by its transverse position (perpendicular to the beam direction) and intensity (spot weight). The beam energy has one-to-one correspondence with the beam range in water and can be expressed in water equivalent path length (WEPL). The treatment spots form a 3-dimensional grid in the tumor target and may also be grouped according to the beam path (figure 2 and figure 3). The mid-range spots refer to the spots of middle energy values on each beam path. For range-probing, a single mid-range spot is selected from each ray-tracing path based on safety, convenience, and high intensity criteria.
Figure 2.
Planned mid-range spots and online pre-treatment probing spots. Left: planned situation. The circles in the tumor represent the planned spots with the shaded ones indicating the mid-range spots. Right: online situation. Mid-range probing spots become anterior and posterior to the planned positions due to an unexpected bony object and air pocket, respectively, on the beam path. Nevertheless, the midrange spots remain inside the tumor, but other spots may shift outside the tumor.
Figure 3.
Crescent-shaped target with the treatment spots depicted by circles. Mid-range spots are indicated by shaded circles. The long blue arrow lines indicate beam paths.
In terms of safety, the range values before and after the selected mid-range create a buffer zone for range uncertainty (figure 2). The planned mid-range spots are located near the middle of the target. During online probing, the mid-range spots land away from the planned positions due to an unexpected change in anatomy by a bony object and air pocket on the beam path, but remain inside the tumor.
In terms of convenience, we select the mid-range so that the need to switch beam energy is minimal. For example, using the mean value of the mid-ranges of all beam paths would require only single energy probing beams. In terms of high intensity, the spot with higher intensity in the candidate list of midrange would be chosen. Two practical selections, the anatomical mid-range and single energy mid-range, are described below.
Anatomical mid-range spots
A simple way to select mid-ranges is to use those that agree with WEPL in the middle of the target. Then, the mid-range spots will roughly land in the middle of the target (figure 3); the planned spots are depicted by circles inside the target with beams coming from the left side of the image, and the midrange spots are depicted by shaded circles (figure 3).
Single energy mid-range spots
Since the mid-range may correspond to different energies for different beam paths, we use the mean value of the mid-ranges of all beam paths for delivery convenience. Such mid-ranges require only a single energy, and the spots would remain within the target with enough buffer. This is most convenient with respect to probing spot delivery and range detection: only spots of the same energy are delivered, and they would land near a predicted surface to facilitate range measurement.
2.2. Range measurement
After delivering the probing spots, we can use online in-beam PET (Shao et al. 2014) to acquire probing spot signals. The images reconstructed from the detected signals are compared with the pre-calculated images to verify range or identify range shift. For illustration purposes, we used probing beam dose instead of PET images for range measurement.
2.3. Range correction
Range compensated adaptation
A particle beam treatment plan can be represented as an intensity map I of the following parameters: E, x, y, and φ, where E denotes the spot energy, and x and y denote the coordinates on the fluence plane for the beam angle φ. For convenience, we will use the Bragg peak position R in the unit of WEPL instead of the beam energy E in our formulation, i.e., I = (R, x, y, φ).
Suppose there is a shift (ΔR ≠ 0) between the planned mid-range spot and the measured probing spot (figure 4). Positive difference ΔR > 0 and negative difference ΔR < 0 indicate that the measured spot is distal and proximal to the planned spot, respectively. A simple correction strategy is to apply the range shift to the plan. Let I0 denotes the original plan, then the new intensity I1 at (R, x, y, φ) is I0(R + ΔR, x, y, φ) as in Eq. (1). This range correction is based on the assumption that the relative WEPL in the target remains unchanged.
Figure 4.
Range shift ΔR measured in WEPL between the probing spot and planned mid-range spot.
| (1) |
Accounting for the probing beam dose
To account for the probing beam dose, we subtract the probing beam intensity I0(R0, x, y, φ) from the shifted intensity but zero out negative values as in Eq. (2), since the intensity value can’t be negative.
| (2) |
where (R0, x, y, φ) is the planned mid-range spot for range-probing and the subscript + indicates.
Range compensated optimization
Alternatively, we may re-optimize the intensity using the range-shifted beamlet dose matrix based on the range difference identified through the probing beams. Let BR,x,y,φ denote the original beamlet dose indexed by (R, x, y, φ) as described above. The beamlet dose is a 3-dimensional distribution, i.e., a function of the spatial coordinates (px, py, pz): BR,x,y,φ(px, py, pz). Here, positive pz indicates the forward beam direction φ. The shifted beamlet dose is defined as in Eq. (3).
| (3) |
The probing beam dose d∗, which is already delivered, can be accounted for by using complementary treatment planning (Lu et al. 2010). Specifically, the objective in re-optimization is to minimize deviation of the new dose from the planned dose dp after adding the probing beam dose d∗ as in Eq. (4).
| (4) |
where Iopt denotes the optimized intensity. Note that the norm squared objective in equation (4) is a sum of squared deviation of delivery dose from planning dose summing over all voxels, which are omitted here for brevity. This optimization compensates for both beam range shift and probing beam dose so that the total delivered dose approaches the planned dose without time-consuming plan objective tuning. Simple gradient-based box-constrained optimizers, such as limited-memory Broyden–Fletche–Goldfarb–Shanno with box constraints (L-BFGS-B) (Nocedal et al. 2006) can be used to solve the optimization problem (4).
2.4. Phantoms and simulations
We will illustrate the range probing approach through examples of anatomical changes, though the approach can be applied for other types of range uncertainties (Paganetti 2012), such as those caused by range modulator errors, beam energy errors, and stopping power ratios. Anatomical changes cause larger range uncertainties than the beam energy or systematic factors by an order of magnitude (Lomax 2016).
We designed a digital phantom to illustrate the probing beams and probing dose. The phantom is represented as a circle with a crescent-shaped target at the center embracing a small distal circle designated as the organ at risk (OAR). A treatment plan was optimized for the phantom, and both anatomical mid-range probing and single energy mid-range probing were demonstrated.
Also, we studied a brain case for range correction for two simulated online scenarios: one with an air pocket and the other with a bony structure on the beam path. First, a treatment plan was optimized. Then, the mid-range probing beams were delivered to online CT. Both online scenarios impacted the beam ranges substantially. We calculated the probing dose and delineated the range shift. We tested both range-compensated adaptation and optimization that use range-shifted beamlets, and compared the results to the treatment plan.
3. Results
The digital phantom case is shown in figure 5. The first row shows the planning CT, WEPL calculations, and plan dose. The circular phantom has a crescent-shaped target outlined in pink, an OAR outlined in blue, and a body outlined in green (figure 5(a)). The WEPL is overlaid on the target, and the red curve depicts the mid-values (figure 5(b)). The WEPL calculation was based on the beam direction, which goes from the left to the right side of the CT image. The plan dose is overlaid on the CT, and the red curve depicts the peak dose values along the beam direction (figure 5(c)). The three horizontal lines indicate the positions of the dose profiles plotted in the inset.
Figure 5.
Range probing with anatomical mid-ranges or single energy mid-ranges and their respective probing beam dose. Top row (left to right): (a) CT with contours of target (crescent shape), body (large outer circle), and OAR (small inner circle); (b) WEPL in target according to horizontal beam direction from negative to positive position, and red curve indicating the mid-values of WEPL; (c) plan dose with red curve indicating the dose peaks on the beam paths, and three horizontal lines (dashed, dash-dot, and dotted) indicating the positions of three dose profiles shown in the inset. Middle row (left to right): (d) treatment plan (beam intensity map) with red curve indicating the anatomical mid-range; (e) mid-range probing beam intensity extracted from the treatment plan; (f) probing dose with red curve indicating the dose peaks along the beam paths, and three horizontal lines (dashed, dash-dot, and dotted) indicating the positions of three probing beam dose profiles shown in the inset. Bottom row ((g), (h), and (i)): same as the middle row, except for the single-energy mid-range.
The second row shows the treatment plan and mid-ranges (figure 5(d)), probing beam intensity (figure 5(e)), and probing beam dose (figure 5(f)) for anatomical mid-range probing. The third row (figure 5(g), (h), and (i)) is the same as the second row, except for single-energy mid-range probing.
The treatment plan is shown as an intensity image, where the horizontal axis indicates the spot range and the vertical axis indicates the spot vertical position (figure 5(d) and (g)). The red curve on the treatment plan indicates the mid-ranges. The single-energy mid-ranges are on a straight line, since they have the same range values.
The probing beam intensities, which are part of the treatment plan, are shown in figure 5(e) and (h). The probing beam dose is overlaid on the CT (figure 5(f) and (i)), with the red curve depicting the peak values and the three horizontal lines marking the positions of the dose profiles in the insets.
We used the peaks of the probing beam dose along the beam paths to measure the beam range. Through anatomical mid-range probing, the peaks were in the middle of the target and were consistent with the middle WEPL in the target. Through single energy mid-range probing, the peaks were closer to the boundary but still within the target. Also, peak locations were consistent with the WEPL of the single energy mid-range inside the target. While the single energy mid-ranges are on a straight line in the plan, this was not the case for the peaks of the probing dose because of the curved body contour at the beam entrance.
For the brain case, because the results of range-compensated optimization were similar to those of range-compensated adaptation, only the latter will be shown. The dose superimposed on CT in transverse, coronal, and sagittal (TCS) views is shown in figure 6.
Figure 6.
Dose overlay on online CT in TCS views with countoured place holder (in pink), tumor (in red), and OAR (in yellow) along the beam path. (a) Plan dose on planning CT. (b) and (c) Doses of the same plan delivered to the online images with an air pocket and bony structure, respectively, at the place holder.
The original treatment plan delivered to the planning CT is used as reference (figure 6(a)). A place holder, a tumor, and an OAR, contoured in pink, red, and yellow, respectively, are encountered along the beam path in that order. For comparison, we show the delivered dose without treatment plan modification (figure 6(b) and (c)).
When the place holder was filled with air, the delivered dose overshot causing a proximal cold spot and an overdose to the distal OAR (figure 6(b)). When the place holder was filled with bony material, the delivered dose undershot and caused a distal cold spot (figure 6(c)).
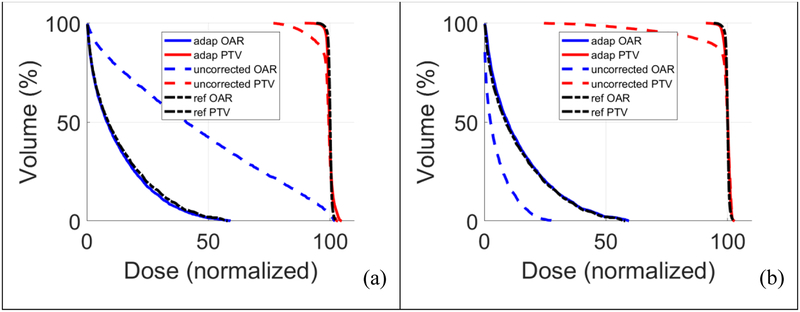
With range-probing and range-compensated adaptation, the modified plans exhibit the same plan quality as the original one. The doses of the modified plans shown in figure 7(a) and (b) for the cases with the air pocket and bony material, respectively, are very similar to the reference dose (figure 6(a)). The similarity in plan quality can also be seen in the dose volume histograms (DVHs) (figure 8). The DVHs of the range-compensated adaptation (solid curves), online delivery without range correction (dashed curves), and reference plans (dash-dotted curves) are presented for the air pocket and bony structure cases in figure 8(a) and (b), respectively.
Figure 7.
Dose of modified treatment plan based on range-compensated adaptation with (a) air and (b) bony material at the placeholder. The modified plan doses are similar to that of the original plan shown in figure 6(a).
Figure 8.
DVH comparison for two online scenarios: (a) an air pocket at the place holder and (b) a bony structure at the place holder. Solid curves: Range-compensated adaptation. Dashed curves: online delivery of the unmodified plan. Dash-dotted curves: the reference DVH. Range-compensated adapation produced similar results to those of the original plan.
The unmodified plan resulted in underdosing to the tumor in both scenarios. Through simple range-compensated adaptation, the DVHs approached the original plan’s DVHs. Both online range-compensated adaptation and optimization were completed within 2 min.
4. Discussion
In the event of online range variation, the mid-range probing spots will likely remain within the target. However, if different beam paths have different mid-ranges, the beam energy may need to be switched for probing beam delivery. The single energy mid-range, a good proxy for anatomical midranges, could be implemented to reduce the need to switch the beam energy and still allows a good buffer within the planning target.
The range shift may be identified by comparing the online measured PET with the offline Monte Carlo simulated PET for single layered spots. PET signals are known to correlate with dose distributions (Fourkal et al. 2009), and single-layered spots would improve detection of the Bragg peak without cross-layered spot smearing.
A quantitative study on mid-range probing with PET simulation is currently being investigated. Our preliminary results showed that in 2–3 min signal acquisition and PET reconstruction, the range measurement errors were within 0.4 ± 1.6 mm (Zhong et al. 2017). A similar time scale has also been reported for online monitoring in PET signal detection (Ferrero et al. 2018). PET reconstruction can start during signal acquisition using iterative algorithms, such as ordered subset expectation maximization (Hudson et al. 1994), and it can be focused on limited ROI along the target to further reduce data processing time. Such time scale and precision would be acceptable for online pre-treatment range measurement and subsequent treatment adaptation.
Other imaging techniques, such as prompt gamma imaging (Krimmer et al. 2017), proton interaction vertex imaging (Henriquet et al. 2012), and thermo-acoustic imaging (Assmann et al. 2015), can potentially be used to detect probing beam signals in real-time. However, similar to PET, they are all challenged by signal statistic issues and complicated system design.
For range correction, we assumed that the target shape and density have not changed from the plan, which is a good approximation for most solid tumors. Changes in the target would typically be caught in the offline re-imaging and require re-planning. Based on this assumption, we propose two online correction strategies: (1) range-compensated adaptation and (2) range-compensated optimization using range-shifted beamlets. A similar adaptation strategy was previously described in (Zhang et al. 2011), where the authors re-determined beam energy based on online CT for adaptive proton therapy.
In our phantom study, we simulated examples with air and bone pockets on the beam paths. True anatomical variations would be more complicated but less extreme. However, these simple but drastic range variations may simulate the effects of true changes, such as bladder filling, nasal cavity changes, and weight loss, as well as range modulator errors or beam energy errors. Even with large range variations, the examples were successfully handled by the proposed approach. As shown in the simulations, the simple range-compensated adaptation strategy accounted for online anatomical changes, which cannot be addressed by setup alignment, and achieved similar plan quality to that of the original plan. The results of range-compensated optimization were similar to those of simple adaptation.
In general, optimization may take advantage of online anatomical variations. Furthermore, a potential advantage of optimization is that it allows for increased probing beam intensity for enhanced mid-range measurement accuracy, as the probing beam dose could be accounted for in optimization.
Treatment planning based on radiobiological effectiveness (RBE) is a bit more favorable to midrange probing. For the same dose, the distal spots have higher RBE than proximal spots, since RBE increases with respect to linear energy transfer (Paganetti 2014). Thus, RBE-based planning would require less distal spot weight, or equivalently, higher mid-range spot weight to achieve uniform target coverage than dose-based planning. That is, RBE-based planning would increase probing beam activities and improve range detection.
Quality assurance (QA) is a common challenge to all online adaptive treatments. Online QA poses an additional time demand than offline QA, as the patient is lying on the treatment couch waiting to be treated. Specific QA challenges in this situation include verification of the range shift measurement and range correction. Since the shift measurement is based on PET images, an independent calculation could be used to verify that the shift amount is the correct range shift. One way to do this is to re-simulate a PET image with spot range equal to the mid-range plus the range shift and to verify its agreement with the measured probing beam image. This QA strategy will be tested extensively in our future work.
The advantages of our proposed techniques are summarized as follows:
Therapeutic probing beams provide stronger signals than pre-treatment imaging beams. The β+ activities along the probing spots are comparable to the measurements conducted after treatment. Therefore, better statistics can be achieved with less marginal physical and biological decay, resulting in equal or better Bragg peak delineation than imaging beams or measurements conducted after treatment.
Single layered spots enable the Bragg peaks to be delineated more easily than multi-layered spots. A dedicated online in-beam PET image device can be designed with high accuracy and low cost to focus only on a narrow region around the mid-plane.
The Bragg peaks would be detected more easily in a short amount of time. Signal contamination due to other decay activities that increase over time can then be kept to a minimum, which is critical for online range measurement.
Mid-range probing is safe. The mid-range probing spots will remain in the tumor region, even with various anatomical changes. The strategy ensures sparing of sensitive structures as in the treatment plan.
Patients will not be exposed to additional radiation for pre-treatment range probing. If the range is verified (in normal scenarios, the measured range shift is most likely within the predefined tolerance), the remaining spots will be delivered as planned. Otherwise, range deviation will be incorporated in subsequent delivery modification or re-planning, taking into account the probing dose that was already delivered.
RGPT-specific procedures are automated and hence do not increase the clinical workload. Probing beam selection and pre-calculation are part of offline procedures in treatment planning. Simulations of online range measurement are in progress, and we anticipate the time of online PET signal detection and reconstruction to be 2–3 min. Calculations of range-compensated adaptation or optimization can be completed in the order of seconds. Since the time increase is marginal, we do not expect the RGPT procedures to interfere with the clinical workflow.
The simulations demonstrated the feasibility of our proposed mid-range probing and improvements achieved by range-guided online adaptation.
5. Conclusion
We propose a novel online in vivo pre-treatment range probing technique using mid-range therapeutic beams and range correction strategies for particle therapy.
The approach is safe, effective, and practical, as it mitigates range uncertainty challenged by beam energy and anatomical variations to ensure patient safety and treatment quality. The proposed range probing and corrections may open a new era in range-guided particle therapy (RGPT).
Acknowledgement
This work was partially funded by CPRIT RP120326, R21CA18771703, and R01CA218402–01A1. The authors would like to thank Dr. Damiana Chiavolini for editing the manuscript.
Footnotes
Part of this work was presented at ICCR 2016 and ASTRO 2017.
References
- Assmann W, Kellnberger S, Reinhardt S, Lehrack S, Edlich A, Thirolf PG, Moser M, Dollinger G, Omar M, Ntziachristos V and Parodi K 2015. Ionoacoustic characterization of the proton Bragg peak with submillimeter accuracy Med Phys 42 (2) 567–574. [DOI] [PubMed] [Google Scholar]
- Attanasi F, Belcari N, Del Guerra A, Enghardt W, Moehrs S, Parodi K, Rosso V and Vecchio S 2009. Comparison of two dedicated ‘in beam’ PET systems via simultaneous imaging of C-12-induced beta(+)-activity Physics in Medicine and Biology 54 (2) N29–N35. [DOI] [PubMed] [Google Scholar]
- Dendooven P, Buitenhuis HJ, Diblen F, Heeres PN, Biegun AK, Fiedler F, van Goethem MJ, van der Graaf ER and Brandenburg S 2015. Short-lived positron emitters in beam-on PET imaging during proton therapy Phys Med Biol 60 (23) 8923–8947. [DOI] [PubMed] [Google Scholar]
- Dong L, Cheung JP and Zhu RX (2012) Image-Guided Proton and Carbon Ion Therapy Proton and Carbon Ion Therapy. Ma C and Lomax T, CRC Press: 127–149. [Google Scholar]
- Enghardt W, Crespo P, Fiedler F, Hinz R, Parodi K, Pawelke J and Ponisch F 2004. Charged hadron tumour therapy monitoring by means of PET Nuclear Instruments & Methods in Physics Research Section a-Accelerators Spectrometers Detectors and Associated Equipment 525 (1–2) 284–288. [Google Scholar]
- Ferrero V, Fiorina E, Morrocchi M, Pennazio F, Baroni G, Battistoni G, Belcari N, Camarlinghi N, Ciocca M, Del Guerra A, Donetti M, Giordanengo S, Giraudo G, Patera V, Peroni C, Rivetti A, Rolo M, Rossi S, Rosso V, Sportelli G, Tampellini S, Valvo F, Wheadon R, Cerello P and Bisogni MG 2018. Online proton therapy monitoring: clinical test of a Silicon-photodetector-based in-beam PET Sci Rep 8 (1) 4100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fourkal E, Fan J and Veltchev I 2009. Absolute dose reconstruction in proton therapy using PET imaging modality: feasibility study Physics in Medicine and Biology 54 (11) N217–N228. [DOI] [PubMed] [Google Scholar]
- Henriquet P, Testa E, Chevallier M, Dauvergne D, Dedes G, Freud N, Krimmer J, Letang JM, Ray C, Richard MH and Sauli F 2012. Interaction vertex imaging (IVI) for carbon ion therapy monitoring: a feasibility study Phys Med Biol 57 (14) 4655–4669. [DOI] [PubMed] [Google Scholar]
- Hudson HM and Larkin RS 1994. Accelerated image reconstruction using ordered subsets of projection data IEEE Trans Med Imaging 13 (4) 601–609. [DOI] [PubMed] [Google Scholar]
- Iseki Y, Kanai T, Kanazawa M, Kitagawa A, Mizuno H, Tomitani T, Suda M and Urakabe E 2004. Range verification system using positron emitting beams for heavy-ion radiotherapy Physics in Medicine and Biology 49 (14) 3179–3195. [DOI] [PubMed] [Google Scholar]
- Kanai T, Kawachi K, Kumamoto Y, Ogawa H, Yamada T, Matsuzawa H and Inada T 1980. Spot scanning system for proton radiotherapy Med Phys 7 (4) 365–369. [DOI] [PubMed] [Google Scholar]
- Knopf AC and Lomax A 2013. In vivo proton range verification: a review Physics in Medicine and Biology 58 (15). [DOI] [PubMed] [Google Scholar]
- Krimmer J, Dauvergne D, Letang JM and Testa E 2017. Prompt-gamma monitoring in hadrontherapy: A review Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment. [Google Scholar]
- Lomax A 1999. Intensity modulation methods for proton radiotherapy Phys Med Biol 44 (1) 185–205. [DOI] [PubMed] [Google Scholar]
- Lomax T 2016. SP-0502: In-vivo range estimation and adaptive particle therapy Radiotherapy and Oncology 119 S238. [Google Scholar]
- Lu W and Ruchala KJ (2010) System and method of optimizing a heterogeneous radiation dose to be delivered to a patient https://www.google.com/patents/US20100228116
- Mumot M, Algranati C, Hartmann M, Schippers JM, Hug E and Lomax AJ 2010. Proton range verification using a range probe: definition of concept and initial analysis Physics in Medicine and Biology 55 (16) 4771–4782. [DOI] [PubMed] [Google Scholar]
- Nocedal J and Wright SJ (2006) Numerical optimization New York, Springer. [Google Scholar]
- Paganetti H 2012. Range uncertainties in proton therapy and the role of Monte Carlo simulations Physics in Medicine and Biology 57 (11) R99–R117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paganetti H 2014. Relative biological effectiveness (RBE) values for proton beam therapy. Variations as a function of biological endpoint, dose, and linear energy transfer Physics in Medicine and Biology 59 (22) R419–R472. [DOI] [PubMed] [Google Scholar]
- Paganetti H and Bortfeld T (2005) Proton Beam Radiotherapy - The State of the Art New Technologies in Radiation Oncology. Schlegel W, Bortfeld T and Grosu A-L. Heidelberg, Springer Verlag. [Google Scholar]
- Pedroni E, Bacher R, Blattmann H, Bohringer T, Coray A, Lomax A, Lin S, Munkel G, Scheib S, Schneider U and et al. 1995. The 200-MeV proton therapy project at the Paul Scherrer Institute: conceptual design and practical realization Med Phys 22 (1) 37–53. [DOI] [PubMed] [Google Scholar]
- Schneider U and Pedroni E 1995. Proton radiography as a tool for quality control in proton therapy Med Phys 22 (4) 353–363. [DOI] [PubMed] [Google Scholar]
- Shao Y, Sun X, Lou K, Zhu XR, Mirkovic D, Poenisch F and Grosshans D 2014. In-beam PET imaging for on-line adaptive proton therapy: an initial phantom study Phys Med Biol 59 (13) 3373–3388. [DOI] [PubMed] [Google Scholar]
- Verburg JM (2015). Reducing range uncertainty in proton therapy. Doctoral degree, Technische Universiteit Eindhoven. [Google Scholar]
- Yang M, Virshup G, Clayton J, Zhu XR, Mohan R and Dong L 2010. Theoretical variance analysis of single- and dual-energy computed tomography methods for calculating proton stopping power ratios of biological tissues Phys Med Biol 55 (5) 1343–1362. [DOI] [PubMed] [Google Scholar]
- Zhang M, Westerly DC and Mackie TR 2011. Introducing an on-line adaptive procedure for prostate image guided intensity modulate proton therapy Phys Med Biol 56 (15) 4947–4965. [DOI] [PubMed] [Google Scholar]
- Zhong Y, Lu W, Chen M and Shao Y 2017. PET Image Based Intrabeam Range Verification and Delivery Optimization for Charged Particle Radiation Therapy International Journal of Radiation Oncology*Biology*Physics 99 (2, Supplement) S92. [Google Scholar]