Abstract
The association between oral health and systemic disease is recognized in the literature. The present study aimed to clarify the association between oral bacteria on the tongue dorsum and factors associated with oral health and systemic disease in middle-aged and elderly patients. The association between bacterial numbers, oral health status and systemic disease was preliminarily investigated in 70 patients (mean age, 69.5 years; range, 45–92 years) who visited the Department of Oral Health, Hiroshima University Hospital (Hiroshima, Japan). The bacterial 16S ribosomal RNA gene was employed to quantitate bacterial numbers using real-time polymerase chain reaction (PCR). PCR was also performed to detect the DNA of periodontal disease-related bacteria. Oral bacterial numbers were marginally negatively correlated with moisture levels on the tongue surface [Spearman's rank correlation coefficient (R)=−0.131, P=0.28). Subjects with bleeding on probing (BOP) or a ≥4 mm probing depth (PD) exhibited higher Porphyromonas gingivalis (P. gingivalis)-positive rates (50.0 and 51.1%, respectively) than those without BOP or a <4 mm PD (39.5 and 30.4%, respectively). Subjects with medical histories of hypertension, diabetes, stroke and heart disease exhibited a trend toward higher P. gingivalis-positive rates than those without such disorders. These findings indicated that the tongue moisture level may be associated with bacterial numbers on the tongue surface, while P. gingivalis on the tongue surface may be associated with systemic and periodontal diseases. Further investigation in a larger number of participants is necessary to clarify the correlation between bacterial numbers and systemic disease.
Keywords: 16S ribosomal RNA gene, bacterial number, periodontal disease-related bacteria, real-time polymerase chain reaction, tongue dorsum
Introduction
A relationship between oral pathogenic bacteria and systemic and oral diseases has been recognized in elderly and hospitalized patients (1,2). In particular, elderly patients exhibit high incidence rates of aspiration pneumonia, which is considered to be associated with poor oral hygiene and impaired oral function (1). Additionally, dry mouth is common among the elderly, and it may lead to oral diseases including dental caries, oral candidiasis and swallowing disorders (3). Importantly, oral health care can aid to remove dental plaques, moisturize the oral cavity, relieve mucosal inflammation, and promote oral hygiene by reducing oral bacterial numbers (2,4). Thus, regular oral care serves a vital role in the maintenance of oral health, which may result in a decreased risk of systemic disease in elderly subjects.
In a previous study, our group prospectively investigated the changes in oral bacterial numbers in colorectal cancer patients during the perioperative period (4). Notably, bacterial numbers on the tongue dorsum changed less markedly than those of the gingiva of the upper anterior teeth and palatoglossal arch, even when perioperative oral health care had been performed (5). Bacteria of the tongue dorsum may be less susceptible to oral health care practices such as tongue cleaning with a sponge or tongue brushes (5,6). This may be because the anatomical features of the tongue dorsum, namely the papillae, creates reservoirs for pathogenic bacteria, which may be associated with oral and systemic disease (7). To date, the association of between bacteria on the tongue surface with oral health status and systemic disease remains unclear. Thus, the correlation between bacteria on the tongue surface and oral and general health status should be clarified.
In taxonomic studies, sequence analysis of the 16S ribosomal RNA (rRNA) gene is widely used to identify bacterial species (8–10). This method enables the measurement of numerous bacterial strains without specialized culturing conditions. Bacterial 16S rRNA genes generally contain 9 hypervariable regions that demonstrate considerable sequence diversity among bacterial species and can be used for species identification (9,10). Conserved regions of the gene can be used to design universal primers to amplify various bacteria (9,10), thus allowing total bacterial numbers to be estimated using a bacterial DNA sample. Herein, the present study performed real-time polymerase chain reaction (PCR) using universal primers for the bacterial 16S rRNA gene to calculate oral bacterial numbers.
Further clinical investigation is required to clarify the relationship between oral bacteria and general health status. Therefore, this study preliminary investigated bacteria on the tongue surface using PCR amplification of the bacterial 16S rRNA gene, and examined the correlation between tongue bacteria and systemic disease in middle-aged and elderly patients.
Materials and methods
Subjects
The current study investigated 70 patients (23 males, 47 females; mean age, 69.5 years; range, 45–92 years) who visited the Department of Oral Health of Hiroshima University Hospital, Hiroshima, Japan from March 2018 to April 2018. A 0–3 mm probing pocket depth was considered to indicate mild or no periodontitis (11). Moderate periodontal disease was defined as the presence of ≥4 and <6 mm probing pocket depth (11) Severe periodontitis was defined as the presence of ≥6 mm pocket (11). All patients exhibited a mild to moderate periodontal status. No patients exhibited symptoms of acute periodontal disease. Regarding toothbrushing frequency, all participants brushed their teeth more than twice daily. None used a tongue scraper to clean the surface area of the tongue. The study design was approved by the Ethics Committee of Hiroshima University, and all participants signed an informed consent agreement. Clinical data were obtained regarding age, sex, smoking status, alcohol consumption and medical history.
Oral sample processing and DNA extraction
Samples were collected from the tongue dorsum using a Orcellex® Brush (Rovers Medical Devices, Oss, The Netherlands) which allows collection of cells or bacteria from oral epithelial layers. The tongue surface was swabbed prior to tooth brushing using the Orcellex® 10 times. DNA was extracted and purified using a PureLink™ Microbiome DNA Purification kit (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA). For PCR analysis, 1 µl was used from a total of 50 µl of oral sample DNA.
Quantification of bacterial numbers
The bacterial 16S rRNA gene was used to quantify bacterial numbers using real-time PCR. Since 16S rRNA gene copy numbers differ among bacteria, it is difficult to use them in the accurate quantification of bacteria. Many bacteria have 1 to 10 16S rRNA gene copies, while some have >10 (12,13). Here, 16S rRNA gene copy numbers were used from Staphylococcus aureus (5 copies per cell) to quantify approximate oral bacterial numbers.
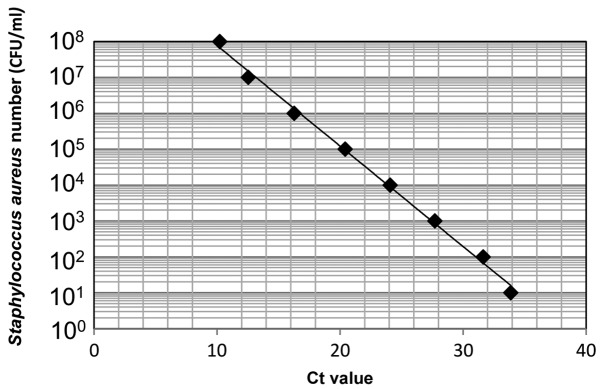
First, DNA was extracted from Staphylococcus aureus [1.0×108 colony forming units (CFU)/ml] and used for real-time PCR. Serial 10-fold dilutions of the DNA sample (ranging from 101 to 108 CFU/ml) were prepared to make a standard curve (Fig. 1). DNA levels were quantified using a CFX connect real-time PCR detection system (Bio-Rad Laboratories, Inc., Hercules, CA, USA) and SYBR-Green PCR Master Mix (Toyobo Life Science, Osaka, Japan), with a reaction mixture containing 1 µl DNA, 9 µl SYBR-Green Mix, and 10 µmol of each oligonucleotide primer pair. Amplifications were performed with initial melting at 95°C for 5 min, followed by 40 cycles of denaturing at 95°C for 30 sec, annealing at 56°C for 30 sec, and extension at 72°C for 1 min. The 16S rRNA primer sequences were forward, 5′-CGTTAGTAATCGTGGATCAGAATG-3′ and reverse, 5′-TGTGACGGGCGGTGTGTA-3′ (14). A standard curve indicating the cycle threshold value versus the 16S rRNA gene was obtained to estimate the bacterial number per sample (15).
Figure 1.
Standard curve indicating Ct values vs. Staphylococcus aureus numbers (CFU/ml). Serial 10-fold dilutions of Staphylococcus aureus DNA (ranging from 101 to 108 CFU/ml) were used to make a standard curve. CFU, colony forming units.
Detecting periodontal disease-related bacteria by PCR
For PCR, 1 µl DNA samples (bacterial numbers: 1.0×103−1.0×106 CFU/ml) were used. Each mixture was amplified with 10X PCR buffer, dNTPs, Taq DNA polymerase (both from Toyobo Life Science) and primers. The following previously reported PCR primer sets were used: For Porphyromonas gingivalis (P. gingivalis) forward, 5′-AGGCAGCTTGCCATACTGCG-3′ and reverse, 5′-ACTGTTAGCAACTACCGATGT-3′; For Tannerella forsythia (T. forsythia) forward, 5′-GCGTATGTAACCTGCCCGCA-3′ and reverse, 5′-TGCTTCAGTGTCAGTTATACCT-3′; and for Treponema denticola (T. denticola) forward, 5′-TAATACCGAATGTGCTCATTTACAT-3′ and reverse, 5′-TCAAAGAAGCATTCCCTCTTCTTCTTA-3′ (16). The PCR program included initial melting at 95°C for 5 min, followed by 35 cycles of 95°C for 30 sec, 56°C for 1 min and 72°C for 30 sec. Following the reaction, 10 µl of the PCR product was electrophoresed on 2% agarose gels with ethidium bromide staining.
Oral examination
Oral examination including periodontal examination [to assess probing depth (PD) and bleeding on probing (BOP)] was performed by dentists at Hiroshima University Hospital. Remaining teeth and denture use were recorded. Oral wetness was evaluated using an oral moisture-checking device (Moisture Checker Mucus®; Scalar Corporation, Tokyo, Japan) as previously described (17). The oral moisture level was measured at the tongue dorsum 10 mm from the apex linguae and expressed as a percentage, with mean values calculated from three independent measurements; a value of >30% was defined as high moisture level and a value of ≤30% as low moisture level as in previous study (17). The tongue coating was classified as none, light-thin (visibly pink underneath) or heavy-thick (no pink observed). The presence of a light-thin and/or heavy-thick coating was considered positive (18).
Statistical analysis
Statistical analysis was performed using SPSS software, version 24.0 (IBM Corp, Armonk, NY, USA). Determination of Spearman's rank correlation coefficient was performed to examine the correlation of bacterial number with age and moisture level. The Mann-Whitney U test with Bonferroni correction or the Kruskal-Wallis test was used to evaluate significant differences in bacterial numbers and clinical factors. All Kruskal-Wallis tests indicated no significant difference and therefore post-hoc tests were not applicable. The χ2 test or Fisher's exact test was used to evaluate significant differences between positive rates of periodontal bacteria and clinical factors. P<0.05 was considered to indicate statistical significance.
Results
Correlation between bacterial numbers on the tongue surface and clinical factors
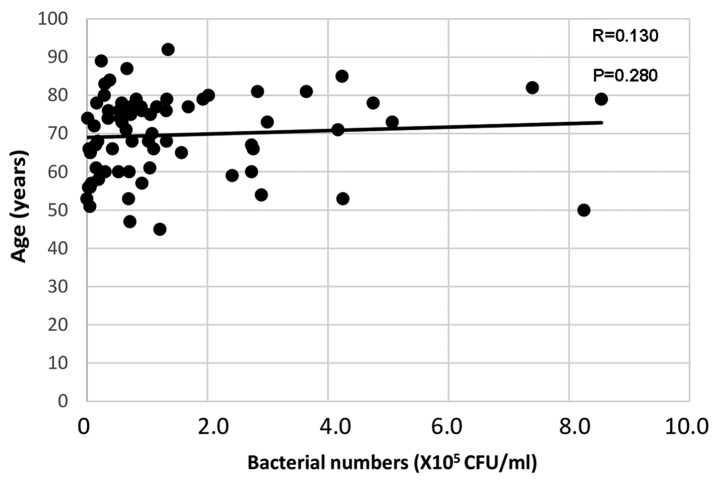
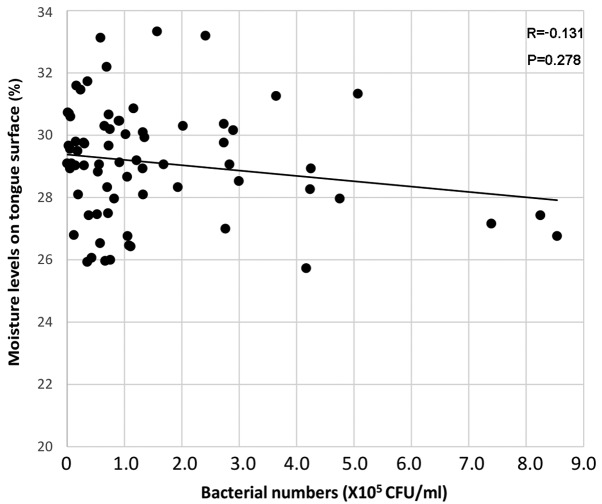
Age was weakly correlated with bacterial numbers on the tongue surface [Spearman's rank correlation coefficient (R)=0.130, P=0.280; Fig. 2]. Meanwhile, the oral bacterial numbers were weakly negatively correlated with moisture levels (R=−0.131, P=0.278; Fig. 3). Table I summarizes the associations between bacterial counts detected by real-time PCR and clinical factors. Oral bacterial numbers appeared to be increased in individuals with fewer than 10 remaining teeth and in denture users; however, bacterial numbers did not differ significantly according to remaining teeth or denture use (Table I). Furthermore, no significant difference was identified between bacterial numbers on the tongue surface and oral hygiene status [BOP and PD (≥4 mm)]. Individuals with a medical history of stroke exhibited increased bacteria compared with those without, but the difference was not significant (Table I).
Figure 2.
Correlation between age and bacterial numbers on the tongue surface. Age was weakly positively correlated with bacterial numbers on the tongue surface.
Figure 3.
Correlation between moisture levels and bacterial numbers on the tongue surface. A weak negative relationship was indicated between oral bacterial numbers and moisture levels.
Table I.
Associations between bacterial numbers on the tongue surface and clinical factors.
| Bacterial numbers | ||
|---|---|---|
| Factor (n) | Mean ± SD (×105) | P-value |
| Sex | ||
| Male (23) | 1.53±1.84 | 0.66 |
| Female (47) | 1.62±2.17 | |
| Age (years) | ||
| ≥65 (50) | 1.58±1.39 | 0.52 |
| <65 (20) | 1.63±2.01 | |
| Alcohol consumption | ||
| Non-drinker (42) | 1.54±1.93 | 0.06 |
| Social drinker (15) | 1.00±1.13 | |
| Every day (13) | 2.45±2.39 | |
| Smoking status | ||
| Non-smoker (62) | 1.66±2.10 | 0.93 |
| Smoker (8) | 1.06±0.85 | |
| Stroke | ||
| No (66) | 1.54±1.91 | 0.14 |
| Yes (4) | 2.54±2.14 | |
| Heart disease | ||
| No (64) | 1.61±1.95 | 0.69 |
| Yes (6) | 1.54±1.65 | |
| Hypertension | ||
| No (50) | 1.53±1.85 | 0.39 |
| Yes (20) | 1.74±2.13 | |
| Diabetes | ||
| No (59) | 1.70±2.03 | 0.51 |
| Yes (11) | 1.00±0.99 | |
| Hyperlipidemia | ||
| No (60) | 1.65±1.99 | 0.77 |
| Yes (10) | 1.27±1.44 | |
| Remaining teeth | ||
| 0–9 (3) | 2.23±2.46 | 0.73 |
| 10–19 (13) | 1.31±1.39 | |
| ≥20 (54) | 1.63±2.02 | |
| Denture use | ||
| Non-user (54) | 0.88±1.72 | 0.12 |
| User (16) | 1.70±1.70 | |
| Oral wetness | ||
| Low (47) | 1.74±2.16 | 0.11 |
| High (23) | 1.30±1.29 | |
| Tongue coating | ||
| Negative (66) | 1.57±1.92 | 0.53 |
| Positive (4) | 1.92±2.12 | |
| Probing depth | ||
| <4 mm (23) | 1.55±1.83 | 0.93 |
| ≥4 mm (47) | 1.61±1.98 | |
| Bleeding on probing | ||
| No (38) | 1.72±2.07 | 0.98 |
| Yes (32) | 1.44±1.74 | |
SD, standard deviation.
Detecting periodontal disease-related bacteria by PCR
Red complex bacteria (e.g., T. denticola, T. forsythia and P. gingivalis) serve major roles in periodontal disease etiology (19). Therefore, the present study performed PCR to detect DNA from these bacteria (Fig. 4), to examine the relationship between periodontal bacteria of the tongue dorsum and clinical factors. A total of 31 out of the 70 participants (57.1%) were P. gingivalis-positive (Table II), with the rate appearing higher in people aged 65 years and over (50.0%) compared with in those aged under 65 (30.0%). Additionally, smokers exhibited higher P. gingivalis-positive rates than nonsmokers. Subjects with medical histories of hypertension, diabetes, stroke and heart disease exhibited higher P. gingivalis-positive rates than those without these disorders. Subjects with BOP and ≥4 mm PD exhibited higher P. gingivalis-positive rates (50.0 and 51.1%, respectively) than those with no BOP and <4 mm PD (39.5 and 30.4%, respectively), but these differences were not significant.
Figure 4.
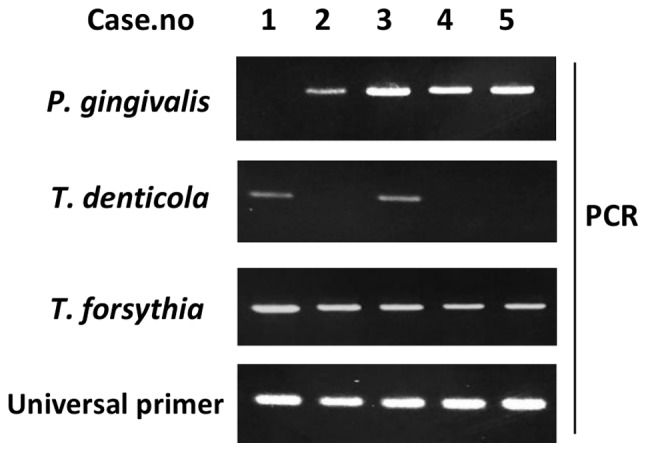
PCR detection of periodontal disease-related bacteria on the tongue surface. PCR was performed to detect T. denticola, T. forsythia and P. gingivalis DNA. 10 µl of each 20-µl PCR product was separated on a 2% agarose gel. P. gingivalis, Porphyromonas gingivalis; T. forsythia, Tannerella forsythia; T. denticola, Treponema denticola; PCR, polymerase chain reaction.
Table II.
Associations between positive rate of P. gingivalis and clinical factors.
| P. gingivalis | |||
|---|---|---|---|
| Factor (n) | (−) | (+) | P-value |
| Sex | |||
| Male (23) | 11 (47.8%) | 12 (52.2%) | 0.44 |
| Female (47) | 28 (59.6%) | 19 (40.4%) | |
| Age (years) | |||
| ≥65 (50) | 25 (50.0%) | 25 (50.0%) | 0.18 |
| <65 (20) | 14 (70.0%) | 6 (30.0%) | |
| Alcohol consumption | |||
| Non-drinker (42) | 26 (61.9%) | 16 (38.1%) | 0.14 |
| Social drinker (15) | 9 (60.0%) | 6 (40.0%) | |
| Every day (13) | 4 (30.8%) | 9 (69.2%) | |
| Smoking status | |||
| Non-Smoker (62) | 37 (59.7%) | 25 (40.3%) | 0.13 |
| Smoker (8) | 2 (25.0%) | 6 (75.0%) | |
| Stroke | |||
| No (66) | 38 (57.6%) | 28 (42.4%) | 0.32 |
| Yes (4) | 1 (25.0%) | 3 (75.0%) | |
| Heart disease | |||
| No (64) | 37 (57.8%) | 27 (42.2%) | 0.40 |
| Yes (6) | 2 (33.3%) | 4 (66.7%) | |
| Hypertension | |||
| No (50) | 31 (62.0%) | 19 (38.0%) | 0.12 |
| Yes (20) | 8 (40.0%) | 12 (60.0%) | |
| Diabetes | |||
| No (59) | 36 (61.0%) | 23 (39.0%) | 0.05 |
| Yes (11) | 3 (27.3%) | 8 (72.7%) | |
| Hyperlipidemia | |||
| No (60) | 35 (58.3%) | 25 (41.7%) | 0.30 |
| Yes (10) | 4 (40.0%) | 6 (60.0%) | |
| Remaining teeth | |||
| 0–9 (3) | 1 (33.3%) | 2 (66.7%) | 0.26 |
| 10–19 (13) | 5 (38.5%) | 8 (61.5%) | |
| ≥20 (54) | 33 (61.1%) | 21 (38.9%) | |
| Denture user | |||
| Non-user (54) | 32 (59.3%) | 22 (40.7%) | 0.41 |
| User (16) | 7 (43.8%) | 9 (56.2%) | |
| Oral wetness | |||
| Low (47) | 25 (53.2%) | 22 (46.8%) | 0.73 |
| High (23) | 14 (60.9%) | 9 (39.1%) | |
| Tongue coating | |||
| Negative (66) | 37 (56.0%) | 29 (44.0%) | 1.00 |
| Positive (4) | 2 (50.0%) | 2 (50.0%) | |
| Probing depth | |||
| <4 mm (23) | 16 (69.6%) | 7 (30.4%) | 0.17 |
| ≥4 mm (47) | 23 (48.9%) | 24 (51.1%)) | |
| Bleeding on probing | |||
| No (38) | 23 (60.5%) | 15 (39.5%) | 0.47 |
| Yes (32) | 16 (50.0%) | 16 (50.0%) | |
P. gingivalis, Porphyromonas gingivalis.
In contrast to the results for P. gingivalis, T. forsythia exhibited no apparent differences in positive rates between those with and without factors associated with systemic and oral disease (Table III). All smokers were T. forsythia-positive, but no significant difference was identified in positive rate between smokers and never-smokers.
Table III.
Associations between positive rate of T. forsythia and clinical factors.
| T. forsythia | |||
|---|---|---|---|
| Factor (n) | (−) | (+) | P-value |
| Sex | |||
| Male (23) | 4 (17.4%) | 19 (82.6%) | 0.17 |
| Female (47) | 16 (34.0%) | 31 (66.0%) | |
| Age (years) | |||
| ≥65 (50) | 12 (24.0%) | 38 (76.0%) | 0.24 |
| <65 (20) | 8 (40.0%) | 12 (60.0%) | |
| Alcohol consumption | |||
| Non-drinker (42) | 15 (26.3%) | 42 (73.7%) | 0.57 |
| Social drinker (15) | 4 (40.0%) | 6 (60.0%) | |
| Every day (13) | 1 (33.3%) | 2 (66.7%) | |
| Smoking status | |||
| Non-smoker (62) | 20 (32.3%) | 42 (67.7%) | 0.05 |
| Smoker (8) | 0 (0.0%) | 8 (100.0%) | |
| Stroke | |||
| No (66) | 19 (28.8%) | 47 (71.2%) | 1.00 |
| Yes (4) | 1 (25.0%) | 3 (75.0%) | |
| Heart disease | |||
| No (64) | 37 (57.8%) | 27 (42.2%) | 0.40 |
| Yes (6) | 2 (33.3%) | 4 (66.7%) | |
| Hypertension | |||
| No (50) | 16 (32.0%) | 34 (68.0%) | 0.39 |
| Yes (20) | 4 (20.0%) | 16 (80%) | |
| Diabetes | |||
| No (59) | 17 (32.2%) | 42 (67.8%) | 1.00 |
| Yes (11) | 3 (27.3%) | 8 (72.7%) | |
| Hyperlipidemia | |||
| No (60) | 17 (28.3%) | 43 (71.7%) | 1.00 |
| Yes (10) | 3 (30.0%) | 7 (70.0%) | |
| Remaining teeth | |||
| 0–9 (3) | 0 (0.0%) | 3 (100.0%) | 0.68 |
| 10–19 (13) | 3 (23.1%) | 10 (76.9%) | |
| ≥20 (54) | 17 (31.5%) | 37 (68.5%) | |
| Denture use | |||
| Non-user (54) | 17 (31.5%) | 37 (68.5%) | 0.36 |
| User (16) | 3 (18.8%) | 14 (81.2%) | |
| Oral wetness | |||
| Low (47) | 13 (27.7%) | 34 (72.3%) | 1.00 |
| High (23) | 7 (30.4%) | 16 (69.6%) | |
| Tongue coating | |||
| Negative (66) | 20 (30.3%) | 46 (69.7%) | 0.32 |
| Positive (4) | 0 (0.0%) | 4 (100.0%) | |
| Probing depth | |||
| <4 mm (23) | 9 (39.1%) | 14 (60.9%) | 0.26 |
| ≥4 mm (47) | 23 (48.9%) | 24 (51.1%) | |
| Bleeding on probing | |||
| No (38) | 13 (34.2%) | 25 (65.8%) | 0.30 |
| Yes (32) | 7 (21.9%) | 25 (78.1%) | |
T. forsythia, Tannerella forsythia.
For T. denticola, men were indicated to exhibit significantly higher positive rates than women (Table IV). T. denticola positivity did not differ between those with and without a medical history of hypertension, diabetes, stroke or heart disease. Interestingly, there appeared to be a significant association between the T. denticola-positive rate and the number of remaining teeth. All individuals with fewer than nine teeth exhibited T. denticola-positivity, while the positive rates in those with 10 or more teeth were <40%. These results indicate that T. denticola on the tongue surface may be associated with tooth loss.
Table IV.
Associations between positive rate of T. denticola and clinical factors.
| T. denticola | |||
|---|---|---|---|
| Factor (n) | (−) | (+) | P-value |
| Sex | |||
| Male (23) | 11 (47.8%) | 12 (52.2%) | 0.03 |
| Female (47) | 36 (76.6%) | 11 (23.4%) | |
| Age (years) | |||
| ≥65 (50) | 32 (64.0%) | 18 (36.0%) | 0.42 |
| <65 (20) | 15 (75.0%) | 5 (25.0%) | |
| Alcohol consumption | |||
| Non-drinker (42) | 32 (56.1%) | 10 (43.9%) | 0.05 |
| Social drinker (15) | 10 (66.7%) | 5 (33.3%) | |
| Every day (13) | 5 (38.5%) | 8 (61.5%) | |
| Smoking status | |||
| Non-smoker (62) | 43 (69.4%) | 19 (30.6%) | 0.43 |
| Smoker (8) | 4 (50.0%) | 4 (50.0%) | |
| Stroke | |||
| No (66) | 44 (66.7%) | 22 (33.3%) | 1.00 |
| Yes (4) | 3 (75.0%) | 1 (25.0%) | |
| Heart disease | |||
| No (64) | 42 (65.6%) | 22 (34.4%) | 0.66 |
| Yes (6) | 5 (83.3%) | 1 (16.7%) | |
| Hypertension | |||
| No (50) | 35 (70.0%) | 15 (30.0%) | 0.58 |
| Yes (20) | 12 (60.0%) | 8 (40.0%) | |
| Diabetes | |||
| No (59) | 41 (69.5%) | 18 (30.5%) | 0.49 |
| Yes (11) | 6 (54.5%) | 5 (45.5%) | |
| Hyperlipidemia | |||
| No (60) | 41 (68.3%) | 19 (31.7%) | 0.72 |
| Yes (10) | 6 (60.0%) | 4 (40.0%) | |
| Remaining teeth | |||
| 0–9 (3) | 0 (0.0%) | 3 (100.0%) | 0.04 |
| 10–19 (13) | 8 (61.6%) | 5 (38.4%) | |
| ≥20 (54) | 39 (72.2%) | 15 (27.8%) | |
| Denture use | |||
| Non-user (54) | 37 (68.5%) | 17 (31.5%) | 0.88 |
| User (16) | 10 (62.5%) | 6 (37.5%) | |
| Oral wetness | |||
| Low (47) | 30 (63.8%) | 17 (36.2%) | 0.43 |
| High (23) | 17 (73.9%) | 6 (26.1%) | |
| Tongue coating | |||
| Negative (66) | 45 (68.2%) | 21 (31.8%) | 0.59 |
| Positive (4) | 2 (50.0%) | 2 (50.0%) | |
| Probing depth | |||
| <4 mm (23) | 17 (73.9%) | 6 (26.1%) | 0.43 |
| ≥4 mm (47) | 30 (63.8%) | 17 (36.2%) | |
| Bleeding on probing | |||
| No (38) | 25 (65.8%) | 13 (34.2%) | 1.00 |
| Yes (32) | 22 (68.8%) | 10 (31.2%) | |
T. denticola, Treponema denticola.
Discussion
Real-time PCR has advantages in calculating total bacterial numbers in terms of sensitivity and rapidity compared with other methods (e.g., DNA hybridization and flow cytometry) (20,21). Therefore, in the present study real-time PCR methods were employed using universal primers for the 16S rRNA gene to quantitate approximate oral bacterial numbers. Bacterial counters use a dielectrophoretic impedance measurement method via a microelectrode chip on which bacteria from liquids are captured by dielectrophoresis (22). Initially, the current study compared oral bacteria numbers obtained from real-time PCR using 16S rRNA gene copy numbers with those obtained from a bacterial counter (Panasonic Healthcare Co., Ltd., Tokyo, Japan) and identified a significant positive association between these methods (data not shown). This confirmed that real-time PCR targeting the 16S rRNA gene can be used to estimate approximate oral bacterial numbers.
Bacterial numbers were weakly negatively correlated with moisture levels on the tongue surface. This indicated that low moisture levels of the tongue may be associated with bacterial growth on the tongue surface. Kobayashi et al (6) previously demonstrated that moisturizing the tongue is important for reducing bacterial numbers on the tongue surface. They determined that combining tongue cleaning with a mouthwash and moisturizing gel was most effective for reducing bacterial numbers on the tongue (6). Thus, moisturizing the tongue surface may serve an important role in inhibiting oral bacterial growth.
Regarding associations between systemic disease and oral health, all diabetic subjects exhibited ≥4-mm periodontal pockets (not shown). Although no significant association was observed between diabetes and oral health status, periodontitis likely serves a role in developing diabetes (23). Periodontal disease-associated cytokines may induce systemic inflammation leading to insulin resistance in type 2 diabetic patients (24). Indeed, the host inflammatory response may be involved in the mechanisms of diabetes (23,24).
In the present study, P. gingivalis infection was more common among elderly (≥65 years old) subjects. Previously, the P. gingivalis detection rate was observed to significantly increase with age, while that of T. denticola and T. forsythia was comparably high across all age groups (25). In the current study, participants with ≥4-mm pockets exhibited higher positive rates of the red complex bacteria P. gingivalis and T. denticola compared with those with <4-mm pockets. Faveri et al (26) reported that the tongue surface is an important reservoir for periodontal pathogens including P. gingivalis. These results indicate that periodontal disease-associated bacteria (i.e., P. gingivalis and T. denticola) on the tongue surface may be associated with deep periodontal pockets and periodontal tissue inflammation.
Regarding the association between systemic disease and red complex bacteria, subjects with medical histories of hypertension, diabetes, stroke and heart disease exhibited higher P. gingivalis-positive rates than those without such diseases. P. gingivalis may be associated with heart disease by inducing endothelial cell dysfunction and atherosclerosis via increased cytokine production, direct bacterial infection and induction of an immune response to bacterial heat-shock proteins (27,28). Additionally, P. gingivalis is likely involved in both diabetes and oral health conditions (29). Among participants with systemic disease, diabetic patients exhibited increased P. gingivalis-positive rates (72.7%) than those without (39.0%), indicating that there is an important association between P. gingivalis and diabetes. These findings and other previous results (29) suggest that P. gingivalis on the tongue surface serves a crucial role in the development of diabetes. However, it remains unclear whether there is a significant association between other red complex bacteria including T. forsythia and T. denticola and systemic disease.
Interestingly, males exhibited a greater T. denticola-positive rate than females, although the reason for this is unknown. Additionally, people with fewer than 10 teeth exhibited a higher T. denticola-positive rate than those with 10 teeth or more. T. denticola has been detected more frequently and in higher counts in people with recurring periodontal disease (30). Thus, T. denticola may serve an important role in deteriorating periodontal tissue, which may be associated with greater tooth loss.
In conclusion, 16S rRNA gene-targeted PCR using swab samples from the tongue dorsum may be useful for counting approximate oral bacterial numbers and detecting periodontal disease-associated bacteria to predict the condition of periodontal tissue. The number of participants in the current study was relatively small; therefore, further investigation in a large-scale study is necessary to clarify the correlations between oral bacterial numbers and systemic disease and factors associated with oral health, particularly in elderly subjects.
Acknowledgements
Not applicable.
Glossary
Abbreviations
- 16S rRNA
16S ribosomal RNA
- BOP
bleeding on probing
- PD
probing depth
- CFU
colony forming units
- P. gingivalis
Porphyromonas gingivalis
- T. forsythia
Tannerella forsythia
- T. denticola
Treponema denticola
- PCR
polymerase chain reaction
Funding
Not applicable.
Availability of data and materials
All data generated or analyzed in this study are included in this article.
Authors' contributions
CYS designed the study and performed experiments, analyzed and interpreted the data and was involved in manuscript writing. HS performed experiments, analyzed and interpreted the data and aided to write the paper. RN performed experiments. KO analyzed and interpreted the data. MS designed the study, analyzed and interpreted the data and aided to write the paper.
Ethics approval and consent to participate
The study design was approved by the Ethics Committee of Hiroshima University and all participants signed an informed consent agreement prior to participation.
Patient consent for publication
All participants consented to the publication of relevant data following anonymization of personal information.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Terpenning M. Geriatric oral health and pneumonia risk. Clin Infect Dis. 2005;40:1807–1810. doi: 10.1086/430603. [DOI] [PubMed] [Google Scholar]
- 2.Shigeishi H, Ohta K, Fujimoto S, Nakagawa T, Mizuta K, Ono S, Shimasue H, Ninomiya Y, Higashikawa K, Tada M, et al. Preoperative oral health care reduces postoperative inflammation and complications in oral cancer patients. Exp Ther Med. 2016;12:1922–1928. doi: 10.3892/etm.2016.3532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Turner MD, Ship JA. Dry mouth and its effects on the oral health of elderly people. J Am Dent Assoc. 2007;138:15S–20S. doi: 10.14219/jada.archive.2007.0358. [DOI] [PubMed] [Google Scholar]
- 4.Kawano T, Shigeishi H, Fukada E, Yanagisawa T, Kuroda N, Takemoto T, Sugiyama M. Changes in bacterial number at different sites of oral cavity during perioperative oral care management in gastrointestinal cancer patients: Preliminary study. J Appl Oral Sci. 2018;26:e20170516. doi: 10.1590/1678-7757-2017-0516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hayashida S, Funahara M, Sekino M, Yamaguchi N, Kosai K, Yanamoto S, Yanagihara K, Umeda M. The effect of tooth brushing, irrigation, and topical tetracycline administration on the reduction of oral bacteria in mechanically ventilated patients: A preliminary study. BMC Oral Health. 2016;16:67. doi: 10.1186/s12903-016-0224-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kobayashi K, Ryu M, Izumi S, Ueda T, Sakurai K. Effect of oral cleaning using mouthwash and a mouth moisturizing gel on bacterial number and moisture level of the tongue surface of older adults requiring nursing care. Geriatr Gerontol Int. 2017;17:116–121. doi: 10.1111/ggi.12684. [DOI] [PubMed] [Google Scholar]
- 7.Danser MM, Gómez SM, Van der Weijden GA. Tongue coating and tongue brushing: A literature review. Int J Dent Hyg. 2003;1:151–158. doi: 10.1034/j.1601-5037.2003.00034.x. [DOI] [PubMed] [Google Scholar]
- 8.Segata N, Haake SK, Mannon P, Lemon KP, Waldron L, Gevers D, Huttenhower C, Izard J. Composition of the adult digestive tract bacterial microbiome based on seven mouth surfaces, tonsils, throat and stool samples. Genome Biol. 2012;13:R42. doi: 10.1186/gb-2012-13-6-r42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Horz HP, Vianna ME, Gomes BP, Conrads G. Evaluation of universal probes and primer sets for assessing total bacterial load in clinical samples: General implications and practical use in endodontic antimicrobial therapy. J Clin Microbiol. 2005;43:5332–5337. doi: 10.1128/JCM.43.10.5332-5337.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Suzuki N, Yoshida A, Nakano Y. Quantitative analysis of multi-species oral biofilms by TaqMan Real-Time PCR. Clin Med Res. 2005;3:176–185. doi: 10.3121/cmr.3.3.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cutress TW, Ainamo J, Sardo-Infirri J. The community periodontal index of treatment needs (CPITN) procedure for population groups and individuals. Int Dent J. 1987;37:222–233. [PubMed] [Google Scholar]
- 12.Kembel SW, Wu M, Eisen JA, Green JL. Incorporating 16S gene copy number information improves estimates of microbial diversity and abundance. PLOS Comput Biol. 2012;8:e1002743. doi: 10.1371/journal.pcbi.1002743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Perisin M, Vetter M, Gilbert JA, Bergelson J. 16Stimator: Statistical estimation of ribosomal gene copy numbers from draft genome assemblies. ISME J. 2016;10:1020–1024. doi: 10.1038/ismej.2015.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yoshida A, Suzuki N, Nakano Y, Oho T, Kawada M, Koga T. Development of a 5 fluorogenic nuclease-based real-time PCR assay for quantitative detection of Actinobacillus actinomycetemcomitans Porphyromonas gingivalis. J Clin Microbiol. 2003;41:863–866. doi: 10.1128/JCM.41.9.4438-4441.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 16.Yasukawa T, Ohmori M, Sato S. The relationship between physiologic halitosis and periodontopathic bacteria of the tongue and gingival sulcus. Odontology. 2010;98:44–51. doi: 10.1007/s10266-009-0114-7. [DOI] [PubMed] [Google Scholar]
- 17.Takahashi F, Koji T, Morita O. Oral dryness examinations: Use of an oral moisture checking device and the usefulness of an oral moisture checking device and a modified cotton method. Prosthodont Res Pract. 2006;5:26–30. doi: 10.2186/prp.5.26. [DOI] [Google Scholar]
- 18.Winkel EG, Roldán S, Van Winkelhoff AJ, Herrera D, Sanz M. Clinical effects of a new mouthrinse containing chlorhexidine, cetylpyridinium chloride and zinc-lactate on oral halitosis. A dual-center, double-blind placebo-controlled study. J Clin Periodontol. 2003;30:300–306. doi: 10.1034/j.1600-051X.2003.00342.x. [DOI] [PubMed] [Google Scholar]
- 19.Socransky SS, Haffajee AD, Cugini MA, Smith C, Kent RL., Jr Microbial complexes in subgingival plaque. J Clin Periodontol. 1998;25:134–144. doi: 10.1111/j.1600-051X.1998.tb02419.x. [DOI] [PubMed] [Google Scholar]
- 20.Veal DA, Deere D, Ferrari B, Piper J, Attfield PV. Fluorescence staining and flow cytometry for monitoring microbial cells. J Immunol Methods. 2000;243:191–210. doi: 10.1016/S0022-1759(00)00234-9. [DOI] [PubMed] [Google Scholar]
- 21.Socransky SS, Smith C, Martin L, Paster BJ, Dewhirst FE, Levin AE. ‘Checkerboard’ DNA-DNA hybridization. Biotechniques. 1994;17:788–792. [PubMed] [Google Scholar]
- 22.Hamada R, Suehiro J, Nakano M, Kikutani T, Konishi K. Development of rapid oral bacteria detection apparatus based on dielectrophoretic impedance measurement method. IET Nanobiotechnol. 2011;5:25–31. doi: 10.1049/iet-nbt.2010.0011. [DOI] [PubMed] [Google Scholar]
- 23.Genco RJ, Borgnakke WS. Risk factors for periodontal disease. Periodontol 2000. 2013;62:59–94. doi: 10.1111/j.1600-0757.2012.00457.x. [DOI] [PubMed] [Google Scholar]
- 24.Nishimura F, Iwamoto Y, Mineshiba J, Shimizu A, Soga Y, Murayama Y. Periodontal disease and diabetes mellitus: The role of tumor necrosis factor-alpha in a 2-way relationship. J Periodontol. 2003;74:97–102. doi: 10.1902/jop.2003.74.1.97. [DOI] [PubMed] [Google Scholar]
- 25.Chigasaki O, Takeuchi Y, Aoki A, Sasaki Y, Mizutani K, Aoyama N, Ikeda Y, Gokyu M, Umeda M, Ishikawa I, et al. A cross-sectional study on the periodontal status and prevalence of red complex periodontal pathogens in a Japanese population. J Oral Sci. 2018;60:293–303. doi: 10.2334/josnusd.17-0223. [DOI] [PubMed] [Google Scholar]
- 26.Faveri M, Feres M, Shibli JA, Hayacibara RF, Hayacibara MM, de Figueiredo LC. Microbiota of the dorsum of the tongue after plaque accumulation: An experimental study in humans. J Periodontol. 2006;77:1539–1546. doi: 10.1902/jop.2006.050366. [DOI] [PubMed] [Google Scholar]
- 27.Ford PJ, Gemmell E, Hamlet SM, Hasan A, Walker PJ, West MJ, Cullinan MP, Seymour GJ. Cross-reactivity of GroEL antibodies with human heat shock protein 60 and quantification of pathogens in atherosclerosis. Oral Microbiol Immunol. 2005;20:296–302. doi: 10.1111/j.1399-302X.2005.00230.x. [DOI] [PubMed] [Google Scholar]
- 28.Seymour GJ, Ford PJ, Cullinan MP, Leishman S, Yamazaki K. Relationship between periodontal infections and systemic disease. Clin Microbiol Infect. 2007;13(Suppl 4):3–10. doi: 10.1111/j.1469-0691.2007.01798.x. [DOI] [PubMed] [Google Scholar]
- 29.Li H, Yang H, Ding Y, Aprecio R, Zhang W, Wang Q, Li Y. Experimental periodontitis induced by Porphyromonas gingivalis does not alter the onset or severity of diabetes in mice. J Periodontal Res. 2013;48:582–590. doi: 10.1111/jre.12041. [DOI] [PubMed] [Google Scholar]
- 30.Meyer-Bäumer A, Eick S, Mertens C, Uhlmann L, Hagenfeld D, Eickholz P, Kim TS, Cosgarea R. Periodontal pathogens and associated factors in aggressive periodontitis: Results 5-17 years after active periodontal therapy. J Clin Periodontol. 2014;41:662–672. doi: 10.1111/jcpe.12255. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed in this study are included in this article.