Abstract
Purpose
To determine whether prostate-specific antigen (PSA) levels adjusted by prostate and zonal volumes estimated from magnetic resonance imaging (MRI) improve the diagnosis of prostate cancer (PCa) and differentiation between patients who harbor high-Gleason-sum PCa and those without PCa.
Materials and Methods
This retrospective study was Health Insurance Portability and Accountability Act (HIPAA)-compliant and approved by the Institutional Review Board of participating medical institutions. T2-weighted MR images were acquired for 61 PCa patients and 100 patients with elevated PSA but without PCa. Computer methods were used to segment prostate and zonal structures and to estimate the total prostate and central-gland (CG) volumes, which were then used to calculate CG volume fraction, PSA density, and PSA density adjusted by CG volume. These quantities were used to differentiate patients with and without PCa. Area under the receiver operating characteristic curve (AUC) was used as the figure of merit.
Results
The total prostate and CG volumes, CG volume fraction, and PSA density adjusted by the total prostate and CG volumes were statistically significantly different between patients with PCa and patients without PCa (P ≤ 0.007). AUC values for the total prostate and CG volumes, and PSA density adjusted by CG volume, were 0.68 ± 0.04, 0.68 ± 0.04, and 0.66 ± 0.04, respectively, and were significantly better than that of PSA (P < 0.02), for differentiation of PCa patients from patients without PCa.
Conclusion
The total prostate and CG volumes estimated from T2-weighted MR images and PSA density adjusted by these volumes can improve the effectiveness of PSA for the diagnosis of PCa and differentiation of high-Gleason-sum PCa patients from patients without PCa.
Prostate cancer (PCa) is the most commonly diagnosed nonskin cancer in men in the US.1 PCa screening is driven by serum prostate-specific antigen (PSA) values and digital rectal examination (DRE), with a definitive diagnosis based on histologic analysis of tissue obtained by random transrectal ultrasound (TRUS)-guided biopsies. However, only about one-third of the patients with elevated PSA have positive biopsies.2–6 This low detection rate is in part due to the blind approach of the TRUS-guided biopsy and the limitations of the PSA test should not be overlooked. Because elevated PSA can also be caused by benign prostatic conditions such as benign prostatic hyperplasia (BPH) and prostatitis, identifying those patients with benign conditions can potentially help avoid a large number of “unnecessary” prostate biopsies. On the other hand, PSA also tends to be elevated in patients with biologically aggressive PCa, which may be characterized by Gleason sums ≥7.7,8 Obviously, identification of patients likely to harbor aggressive PCa is clinically important for better-informed counseling and treatment selection.
Anatomically, the prostate is composed of several zonal structures developed from different embryological origins.9,10 Prostate diseases also tend to correlate with this zonal structure, eg, the central gland (CG), including transition zone and central zone, is the typical site of BPH, whereas the peripheral zone (PZ) accounts for 75–85% of all PCa.11 We hypothesize that, due to different cancer risk in the CG and PZ, prostate zonal volumes can provide additional information in the assessment of personalized risk of PCa.
Because PSA is produced by both normal prostate tissue and prostate cancer, it follows that adjusting the PSA value by prostate volume may improve the effectiveness of PSA for identification of patients who harbor PCa. This idea has its origin in the well-known PSA “density,” whereby the PSA value is divided by the prostate volume, estimated from either DRE or TRUS. Both the total prostate and zonal prostate volumes have been used and several studies have shown that PSA adjusted by prostate volumes have advantages over the PSA level alone in different settings.12–19 However, determination of prostate volume with DRE or TRUS is notoriously inaccurate. On the other hand, estimation of prostate volumes from magnetic resonance imaging (MRI) is potentially much more accurate.20–25 Furthermore, with MRI-based segmentation, it is possible to differentiate the total prostate volume from the volume of the CG. The purpose of this study was, therefore, to determine whether elevated PSA levels adjusted by prostate and zonal volumes estimated from MRI improve the diagnosis of PCa and differentiation between patients who harbor high-Gleason-sum (Gleason sum ≥7) PCa and those without PCa.
Materials and Methods
Patients
This retrospective study was approved by the Institutional Review Board of participating medical institutions where patient data were collected with a waiver of informed consent and was in compliance with the Health Insurance Portability and Accountability Act. Two groups of patients were included in this study. The PCa patient group included 61 consecutive patients diagnosed with PCa by TRUS-guided prostate biopsy between September 2008 and February 2010 who: 1) underwent multiparametric MRI with endorectal coil on 1.5T or 3.0T scanners, 2) subsequently underwent radical prostatectomy, and 3) did not receive radiation or hormonal therapy prior to MRI. The non-PCa patient group included 100 consecutive patients with elevated PSA and/or abnormal DRE who: 1) had a negative 12-core TRUS-guided prostate biopsy, and 2) subsequently had a negative MR-ultrasound-fusion guided targeted prostate biopsy.
The time interval between the PSA measurement and the MR study for all the patients was in the range of 7–69 days with an average of 28.6 days. The PCa patients and non-PCa patients were not intentionally matched in terms of the age, prostate volume, and PSA level.
The prostatectomy specimens of the 61 PCa patients were reviewed and Gleason sums were determined based on the histopathological analysis of the specimens by a genitourinary pathologist (T.A., 8 years experience in genitourinary pathology).
The patients diagnosed with PCa have been reported in a previous study,26 and the patients with elevated PSA and/or abnormal DRE but no PCa have been reported previously in another study with a larger patient cohort.27 However, the current study investigated the characteristics of the patients for a different purpose.
MRI Protocols
All MR scans were performed with both an endorectal coil (Medrad, Warrendale, PA) and a phased-array surface coil by using either a 1.5T or 3.0T Achieva MR scanner (Philips Medical Systems, Eindhoven, Netherlands [n = 131]), or a 1.5T Excite MR scanner (GE Healthcare, Waukesha, WI [n = 30]). Immediately before the MR scan, 1 mg glucagon (Lilly, Indianapolis, IN) was injected intramuscularly to reduce peristalsis of the rectum. We imaged the entire prostate and oriented axial images to be perpendicular to the rectal wall, based on the sagittal images. A parallel imaging factor of 2 was utilized in all sequences. The following image types were obtained: axial, coronal, and sagittal T2-weighted fast spin echo (FSE), axial T1-weighted FSE, axial free-breathing diffusion-weighted imaging (DWI), and axial free-breathing dynamic contrast enhanced-MR (DCE-MR) imaging. Acquisition of DCE-MR images (of the entire prostate) started 30 seconds before intravenous bolus administration of 0.1 mmol/kg Gadodiamide (Omniscan, GE Healthcare, Princeton, NJ), which was followed immediately by a 20-mL saline flush at the rate of 2.0 mL/s. The total image acquisition time was ~45 minutes. Due to logistic reasons, MRI protocols were not identical for patients with PCa and patients without PCa. Detailed T2-weighted imaging acquisition protocols are given in the Appendix.
Prostate Volume Estimation
The method for estimation of the total prostate, and CG, volumes from T2-weighted MR images was reported previously.26 Briefly, the entire prostate and the CG was semiautomatically segmented on T2-weighted MR images28 and a radiologist (A.O., 10 years experience in prostate MRI) reviewed and manually corrected the segmentation results, especially at the base and the apex of the prostate, to ensure accuracy. This method was used in this study for analysis of the patients diagnosed with PCa. For logistic reasons, the total prostate and CG volumes of the non-PCa patients were estimated with different automated prostate segmentation software.29 However, both methods were validated using prostatectomy specimens as the reference.26,29 For all patients, the total prostate and the CG volumes were estimated by multiplying MRI voxel size (in cm3) by the number of voxels within a segmented volume. Subsequently, the PZ volume was estimated by subtracting the CG volume from the total prostate volume. We defined the CG volume fraction as the CG volume divided by the total prostate volume.
Volume-Adjusted PSAs (PSA Densities)
Volume-adjusted PSA, or PSA density (PSAD), was estimated by dividing PSA value by the total prostate volume, and CG volume-adjusted PSA (PSAD-CG) was estimated by dividing PSA value by the CG volume.
Statistical Analysis
Prostate volumes and PSAD were compared by using the Mann–Whitney U-test between patients who harbor high-Gleason-sum (Gleason sum ≥7) PCa and those without PCa, and between patients who harbor PCa and those without PCa. The Pearson correlation coefficient (r) between prostate and its zonal volumes was estimated. We used receiver operating characteristic (ROC) analysis to evaluate the effectiveness of prostate volume and volume-adjusted PSAD for the identification of PCa from non-PCa patients.30 Proper binormal ROC curves were estimated with maximum likelihood estimation 31 and the area under the ROC curve (AUC) was used as the figure of merit. The conventional bivariate binormal model was used to compare two AUC values statistically.32 Statistical significance was assessed by comparing two-tailed P-values with both the critical value of α = 0.05, and the critical value corrected with the Bonferroni method for each set of the comparisons.
Results
The median age was 64 (average 63.3, range 52–75) years and 61 (average 60.6, range 44–77) years, respectively, for the 61 PCa patients and the 100 non-PCa patients. For the PCa patients, 13% (8/61), 66% (40/61), 11% (7/61), and 10% (6/61) had Gleason sums of 6, 7, 8, and 9, respectively.
In Fig. 1 we show typical segmentation results of the whole prostate and the CZ for a PCa patient.
FIGURE 1.
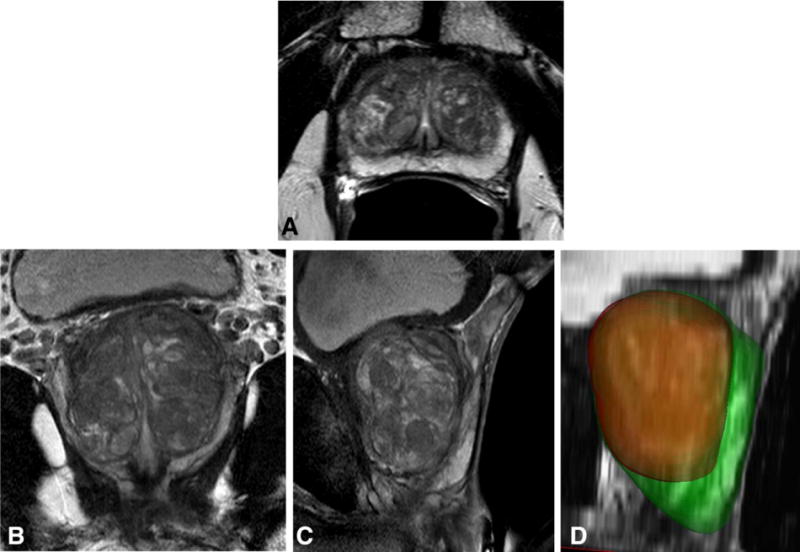
Segmentation results for a 66-year-old patient with PSA level of 6.92 ng/mL, a negative 12-core transrectal ultrasond-guided biopsy, and a negative MR-ultrasound-fusion guided targeted prostate biopsy. Shown are the transverse (A), coronal (B), and sagittal (C) T2-weighted MR images, and the segmentation results of the whole prostate and the central gland (D) in green and red colors, respectively. Based on the segmentation results, the central gland and the peripheral zone are measured 42 and 27 cc in volume, respectively.
Total Prostate and Zonal Volumes and PSA
Comparisons of the total prostate and zonal volumes are listed in Table 1 between the 61 PCa and 100 non-PCa patients, and in Table 2 between the 53 high-Gleason-sum (Gleason sum ≥7) PCa and 100 non-PCa patients. The differences in the total prostate volume and in the CG volume between the patient groups in both comparisons were statistically significant, but the differences in the PZ volume were not statistically significant (Tables 1 and 2). The correlation between the total prostate volume and the CG volume was strong (r = 0.96, P < 0.0001 among PCa patients and r = 0.99, P < 0.0001 among non-PCa patients), but the correlation between the CG volume and the PZ volume was weaker (r = 0.16, P = 0.2 among PCa patients and r = 0.44, P < 0.0001 among non-PCa patients). The CG ratio was statistically significantly different between PCa and non-PCa patients (Table 1) and between high-Gleason-sum (Gleason sum ≥7) PCa and non-PCa patients (Table 2).
TABLE 1.
Comparison of the Average Estimated Prostate Volumes and PSA Densities Between 100 Non-PCa and 61 PCa Patients
| Variable | Non-PCa | PCa | P-valuea |
|---|---|---|---|
| Total prostate volume (cm3) | 67.0 ± 42.3 | 46.4 ± 21.2 | 0.0001 |
| PZ volume (cm3) | 21.5 ± 6.7 | 19.5 ± 6.1 | 0.05 |
| CG volume (cm3) | 45.5 ± 39.0 | 26.9 ± 19.3 | 0.00003 |
| CG ratio | 0.62 ± 0.13 | 0.54 ± 0.12 | 0.00005 |
| PSA (ng/mL) | 8.7 ± 6.3 | 10.2 ± 13.1 | 0.13 |
| PSAD (ng/mL/cm3) | 0.15 ± 0.12 | 0.23 ± 0.24 | 0.007 |
| PSAD-CG (ng/mL/cm3) | 0.26 ± 0.24 | 0.47 ± 0.53 | 0.0005 |
PSA: prostate-specific antigen; PCa: prostate cancer; PZ: peripheral zone; CG: central gland; PSAD: PSA density; PSAD-CG: CG volume-adjusted PSA.
A critical value of 0.05 was used for statistical significance; after Bonferroni correction, the critical value was adjusted to 0.05/7 = 0.007.
TABLE 2.
Comparison of the Average Estimated Prostate Volumes and PSADs Between 100 Non-PCa and 53 High-Gleason-Sum (Gleason Score ≥ 7) PCa Patients
| Variable | Non-PCa | High-Gleason-sum PCa | P-valuea |
|---|---|---|---|
| Total prostate volume (cm3) | 67.0 ± 42.3 | 44.2 ± 19.5 | 0.00002 |
| PZ volume (cm3) | 21.5 ± 6.7 | 19.7 ± 6.0 | 0.07 |
| CG volume (cm3) | 45.5 ± 39.0 | 24.5 ± 17.2 | < 0.00001 |
| CG ratio | 0.62 ± 0.13 | 0.52 ± 0.11 | < 0.00001 |
| PSA (ng/mL) | 8.7 ± 6.3 | 10.9 ± 13.9 | 0.2 |
| PSAD (ng/mL/cm3) | 0.15 ± 0.12 | 0.25 ± 0.25 | 0.001 |
| PSAD-CG (ng/mL/cm3) | 0.26 ± 0.24 | 0.51 ± 0.55 | 0.00002 |
PSA: prostate-specific antigen; PCa: prostate cancer; PZ: peripheral zone; CG: central gland; PSAD: PSA density; PSAD-CG: CG volume-adjusted PSA.
A critical value of 0.05 was used for statistical significance; after Bonferroni correction, the critical value was adjusted to 0.05/7 = 0.007.
Similar comparisons of PSA and PSADs are also shown in Tables 1 and 2. PSA was not significantly different between PCa and non-PCa patients or between high-Gleason-sum PCa and non-PCa patients, but both PSAD and PSAD-CG were statistically significantly different between the patient groups in both comparisons, demonstrating improved discrimination effectiveness.
Classification Performance
The AUC values, for differentiation of PCa from non-PCa patients and differentiation of high-Gleason-sum PCa from non-PCa patients, of the total prostate and zonal volumes, PSA, and PSADs are listed in Table 3. Figures 2 and 3 show the ROC curves for differentiation of PCa from non-PCa patients and differentiation of high-Gleason-sum PCa from non-PCa patients, respectively. For differentiation of PCa from non-PCa patients, the AUC values of the total prostate volume (P = 0.01) and CG volume (P = 0.01) were statistically significantly greater than that of PSA, at the critical level of 0.05. For differentiation of high-Gleason-sum PCa from non-PCa patients, the AUC values of the total prostate volume (P < 0.001), the CG volume (P < 0.001) and PSAD-CG (P = 0.02) were statistically significantly greater than that of PSA, at the critical level of 0.05. After the Bonferroni correction, the critical level was adjusted to 0.05/12 = 0.004. The significance was then diminished, except the total prostate and CG volumes for differentiation of high-Gleason-sum PCa from non-PCa patients.
TABLE 3.
Classification Performance (in Terms of AUC Values) of the Total Prostate and Zonal Volumes, PSA, and PSADs for Differentiation Between Non-PCa and PCa Patients and for Differentiation Between Non-PCa and High-Gleason-Sum (Gleason Sum ≥ 7) PCa Patients
| Variable | Classification performance (AUC) | |
|---|---|---|
| Non-PCa vs. PCa | Non-PCa vs. high-Gleason-sum PCa |
|
| Total prostate volume (cm3) | 0.68 ± 0.04 | 0.70 ± 0.04 |
| PZ volume (cm3) | 0.58 ± 0.05 | 0.57 ± 0.05 |
| CG volume (cm3) | 0.68 ± 0.04 | 0.72 ± 0.04 |
| CG ratio | 0.66 ± 0.04 | 0.72 ± 0.04 |
| PSA (ng/mL) | 0.53 ± 0.05 | 0.52 ± 0.05 |
| PSAD (ng/mL/cm3) | 0.62 ± 0.05 | 0.65 ± 0.05 |
| PSAD-CG (ng/mL/cm3) | 0.66 ± 0.04 | 0.70 ± 0.04 |
AUC: area under the receiver operating characteristic curve; PSA: prostate-specific antigen; PCa: prostate cancer; PZ: peripheral zone; CG: central gland; PSAD: PSA density; PSAD-CG: CG volume-adjusted PSA.
FIGURE 2.
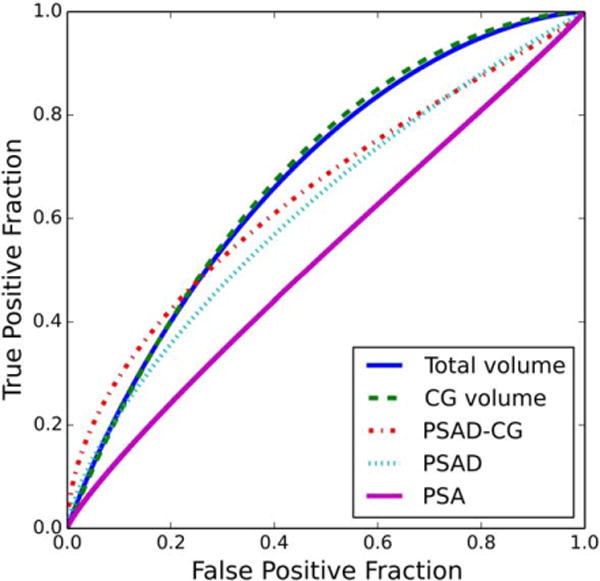
ROC curves for the total prostate volume, CG volume, CG-adjusted PSA density, PSA density, and PSA concentration in the classification of PCa patients and non-PCa patients with elevated PSA level.
FIGURE 3.
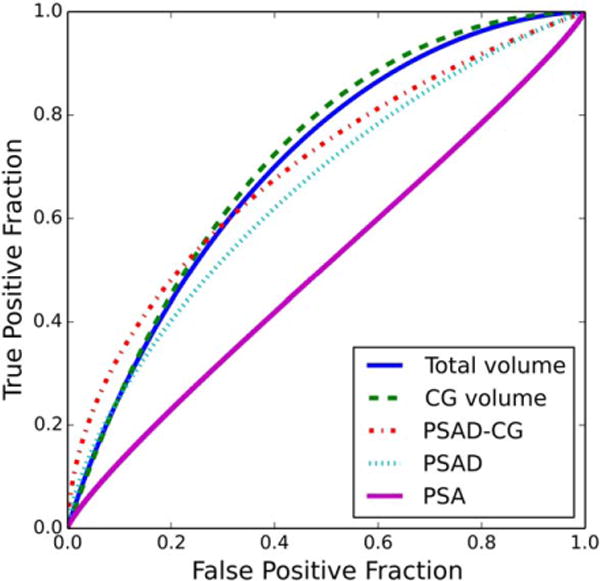
ROC curves for the total prostate volume, CG volume, CG-adjusted PSA density, PSA density, and PSA concentration in the classification of high-Gleason-sum PCa patients and non-PCa patients with elevated PSA level.
Discussion
Because of well-known limitations of PSA screening,33 there has been considerable effort to improve the accuracy of PSA-based screening by adjusting PSA for prostate volume (PSA density) or for the rate of PSA increase (PSA velocity). For example, PSAD adjusted by the CG volume has been shown to be more accurate than PSAD adjusted by the total prostate volume, using sonography for volume estimation.14 Similarly, we show in this study that PSAD adjusted by MRI-based total prostate and CG volumes improves the differentiation of patients who harbor higher-grade PCa from those who do not have PCa. This conclusion is supported by a previous study with fewer patients (n = 73).13 Other studies have also shown that PSAD adjusted by the total prostate or CG volumes improves specificity of PCa detection and reduces the number of biopsy recommendations.12,15,17,18 In a recent study of 129 patients who underwent prostatectomy, PSA adjusted by the CG volume was found significantly different between patients with indolent versus high-Gleason-sum PCa.16 These results, taken together, support the notion that PSAD adjusted for MRI-derived total prostate and zonal volumes is potentially useful for identification of men with high-Gleason-sum PCa.
Studies of large numbers of patients have shown that the CG volume (or the transition zone volume) is smaller in PCa patients than in patients with BPH but not PCa.34–36 In a study of 69 patients, the CG volume was found smaller in PCa patients than in BPH patients, whereas the total prostate volume was found similar in the two groups.36 In another study, the CG volume was found to be a better determinant of BPH size than the total prostate volume, and the ratio of CG volume to total prostate volume was also found to be correlated with clinical parameters of BPH.37 In a third study of 235 patients with intermediate PSA levels (4.1–10 ng/mL), the total prostate and the CG volumes were found to have significantly greater predictive values than PSA for the detection of PCa.17 The results of these studies indicate that the total prostate and zonal volumes can help improve the effectiveness of PSA in differentiation of PCa from BPH.
It is well known, however, that TRUS has limited reproducibility, and inaccurate volume estimation from TRUS hinders widespread clinical use of prostate volume as a diagnostic variable.21 With TRUS, the total prostate and CG volumes were estimated based on an approximation of an ellipsoid—volume = π / 6 × length × width × height—with the three linear dimensions estimated using TRUS images. However, the prostate is not an ellipsoid, and this assumption can lead to large errors in the volume estimation.21 Furthermore, small errors in each of the three linear dimensions can aggregate into large errors due to the effects of the multiplication. Moreover, TRUS-based estimation of linear dimensions is known to vary in patients with large prostates and vary with observer experience.21
Compared with TRUS, MRI has higher spatial resolution and better soft tissue contrast, and thus it should provide more accurate and more robust volume estimation.13,22,24 Furthermore, computer segmentation methods can be applied to MR images for automatic or semiautomatic segmentation of the prostate,20,23 and for segmentation of the CG and PZ.10,38–40
This retrospective study has several limitations. Due to the selection bias, direct extrapolation of the conclusions to other patient groups may not be appropriate. Verification bias exists between the PCa and non-PCa patient groups: All patients with PCa underwent prostatectomy, while negative MR-TRUS-guided biopsy cannot rule out PCa completely for the “non-PCa” patients since the follow-up data for the “non-PCa” patients was not available. Because of the multicenter study setting, the patients were imaged on several different MR scanners and the method of volume estimation from MRI was not exactly the same for all patients. Because the DRE and TRUS prostate volumes were not routinely measured in our institutions, we did not compare the MRI-based prostate volumes with the TRUS prostate volumes. In addition, due to the subjective nature of the Gleason grading system, Gleason score has its own limitations in reproducibility and sampling effect. Finally, there were large variations in patient age and, hence, the size of their prostates.
In conclusion, this study indicates that the total prostate and CG volumes estimated from T2-weighted MR images can help the diagnosis of PCa and differentiate patients with PCa from patients with elevated PSA but without PCa. The tools for segmentation of the prostate and its zonal structures from MRI are highly sophisticated. As MRI is used more and more frequently for the management of PCa, the clinical utility of MRI-based prostate volume estimates and PSADs deserve further studies to explore their potential for identification of patients at high risk for PCa.
Acknowledgments
Contract grant sponsor: US Army Medical Research and Materiel Command Prostate Cancer Research Program; Contract grant number: Idea Development Award PC093485; Contract grant sponsor: Beijing Jiaotong University Talent Fund; Contract grant number: 2014RC044.
APPENDIX: MRI Acquisition Protocols
GE Healthcare Unit Protocol
An array spatial sensitivity encoding technique (ASSET) (parallel imaging) factor of two was used in all sequences. T2-weighted imaging parameters were as follows: repetition time / echo time, 3200–3500/90–100 msec; matrix, 192 × 256; echo train length, 19; number of signals acquired, 4; section thickness, 3 mm; intersection gap, 0 mm; and field of view, 14–16 cm.
Philips Healthcare Unit Protocol
For prostate cancer patients, T2-weighted imaging parameters were as follows: repetition time / echo time, 4300–5000/120 msec; matrix, 204 × 256; echo train length, 24; number of signals acquired, 4; section thickness, 3 mm; intersection gap, 0 mm; field of view, 14–18 cm; and spatial resolution, 0.8 × 0.8 × 3 mm. For nonprostate cancer patients, T2-weighted imaging parameters were as follows: repetition time / echo time, 8869/120 msec; matrix, 304 × 234; flip angle, 90°; section thickness, 3 mm; intersection gap, 0 mm; field of view, 14 cm; and spatial resolution, 0.27 × 0.27 × 3 mm.
References
- 1.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. doi: 10.3322/caac.21166. [DOI] [PubMed] [Google Scholar]
- 2.Catalona WJ, Richie JP, Ahmann FR, et al. Comparison of digital rectal examination and serum prostate-specific antigen in the early detection of prostate cancer: results of a multicenter clinical trial of 6,630 men. J Urol. 1994;151:1283–1290. doi: 10.1016/s0022-5347(17)35233-3. [DOI] [PubMed] [Google Scholar]
- 3.Keetch DW, Catalona WJ, Smith DS. Serial prostatic biopsies in men with persistently elevated serum prostate specific antigen values. J Urol. 1994;151:1571–1574. doi: 10.1016/s0022-5347(17)35304-1. [DOI] [PubMed] [Google Scholar]
- 4.Djavan B, Ravery V, Zlotta A, et al. Prospective evaluation of prostate cancer detected on biopsies 1, 2, 3 and 4: when should we stop? J Urol. 2001;166:1679–1683. [PubMed] [Google Scholar]
- 5.Roehl KA, Antenor JAV, Catalona WJ. Serial biopsy results in prostate cancer screening study. J Urol. 2002;167:2435–2439. [PubMed] [Google Scholar]
- 6.Elabbady AA, Khedr MM. Extended 12-core prostate biopsy increases both the detection of prostate cancer and the accuracy of Gleason score. Eur Urol. 2006;49:49–53. doi: 10.1016/j.eururo.2005.08.013. [DOI] [PubMed] [Google Scholar]
- 7.Bill-Axelson A, Holmberg L, Ruutu M, et al. Radical prostatectomy versus watchful waiting in early prostate cancer. N Engl J Med. 2011;364:1708–1717. doi: 10.1056/NEJMoa1011967. [DOI] [PubMed] [Google Scholar]
- 8.Cooperberg MR, Carroll PR, Klotz L. Active surveillance for prostate cancer: progress and promise. J Clin Oncol. 2011;29:3669–3676. doi: 10.1200/JCO.2011.34.9738. [DOI] [PubMed] [Google Scholar]
- 9.McNeal JE. Regional morphology and pathology of the prostate. Am J Clin Pathol. 1968;49:347–357. doi: 10.1093/ajcp/49.3.347. [DOI] [PubMed] [Google Scholar]
- 10.Sommer FG, McNeal JE, Carrol CL. MR depiction of zonal anatomy of the prostate at 1.5 T. J Comput Assist Tomogr. 1986;10:983–989. doi: 10.1097/00004728-198611000-00017. [DOI] [PubMed] [Google Scholar]
- 11.Augustin H, Erbersdobler A, Hammerer PG, Graefen M, Huland H. Prostate cancers in the transition zone. Part 2. Clinical aspects. BJU Int. 2004;94:1226–1229. doi: 10.1111/j.1464-410X.2004.05147.x. [DOI] [PubMed] [Google Scholar]
- 12.Benson MC, Whang IS, Olsson CA, Mcmahon DJ, Cooner WH. The use of prostate specific antigen density to enhance the predictive value of intermediate levels of serum prostate specific antigen. J Urol. 1992;147:817–821. doi: 10.1016/s0022-5347(17)37394-9. [DOI] [PubMed] [Google Scholar]
- 13.Hardman RL, Liang Y, Ware S, et al. Correlation of PSA density to prostate cancer based on prostate volume by 3.0 T MRI. Open J Urol. 2011;1:28–36. [Google Scholar]
- 14.Kalish J, Cooner WH, Graham SD. Serum PSA adjusted for volume of transition zone (PSAT) is more accurate than PSA adjusted for total gland volume (PSAD) in detecting adenocarcinoma of the prostate. Urology. 1994;43:601–606. doi: 10.1016/0090-4295(94)90170-8. [DOI] [PubMed] [Google Scholar]
- 15.Kang SH, Bae JH, Park HS, et al. Prostate-specific antigen adjusted for the transition zone volume as a second screening test: a prospective study of 248 cases. Int J Urol. 2006;13:910–914. doi: 10.1111/j.1442-2042.2006.01439.x. [DOI] [PubMed] [Google Scholar]
- 16.Ohigashi T, Kanao K, Mizuno R, Kikuchi E, Nakashima J, Oya M. Predicting the probability of significant prostate cancer in Japanese men with serum prostate-specific antigen less than 10 ng/mL: development of a novel pre-biopsy nomogram. Int J Urol. 2010;17:274–279. doi: 10.1111/j.1442-2042.2010.02453.x. [DOI] [PubMed] [Google Scholar]
- 17.Tanaka N, Fujimoto K, Chihara Y, et al. Prostatic volume and volume-adjusted prostate-specific antigen as predictive parameters for prostate cancer patients with intermediate PSA levels. Prostate Cancer Prostatic Dis. 2007;10:274–278. doi: 10.1038/sj.pcan.4500957. [DOI] [PubMed] [Google Scholar]
- 18.Zlotta AR, Djavan B, Petein M, Susani M, Marberger M, Schulman CC. Prostate specific antigen density of the transition zone for predicting pathological stage of localized prostate cancer in patients with serum prostate specific antigen less than 10 ng/ml. J Urol. 1998;160:2089–2095. doi: 10.1097/00005392-199812010-00040. [DOI] [PubMed] [Google Scholar]
- 19.Aksoy Y, Oral A, Aksoy H, Demirel A, Akcay F. PSA density and PSA transition zone density in the diagnosis of prostate cancer in PSA gray zone cases. Ann Clin Lab Sci. 2003;33:320–323. [PubMed] [Google Scholar]
- 20.Bulman JC, Toth R, Patel AD, et al. Automated computer-derived prostate volumes from MR imaging data: comparison with radiologist-derived MR imaging and pathologic specimen volumes. Radiology. 2012;262:144–151. doi: 10.1148/radiol.11110266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Choi YJ, Kim JK, Kim HJ, Cho KS. Interobserver variability of transrectal ultrasound for prostate volume measurement according to volume and observer experience. AJR Am J Roentgenol. 2009;192:444–449. doi: 10.2214/AJR.07.3617. [DOI] [PubMed] [Google Scholar]
- 22.Jeong CW, Park HK, Hong SK, Byun SS, Lee HJ, Lee SE. Comparison of prostate volume measured by transrectal ultrasonography and MRI with the actual prostate volume measured after radical prostatectomy. Urol Int. 2008;81:179–185. doi: 10.1159/000144057. [DOI] [PubMed] [Google Scholar]
- 23.Jia G, Baudendistel KT, von Tengg-Kobligk H, et al. Assessing prostate volume by magnetic resonance imaging: a comparison of different measurement approaches for organ volume analysis. Invest Radiol. 2005;40:243–248. doi: 10.1097/01.rli.0000156312.24604.7c. [DOI] [PubMed] [Google Scholar]
- 24.Lee JS, Chung BH. Transrectal ultrasound versus magnetic resonance imaging in the estimation of prostate volume as compared with radical prostatectomy specimens. Urol Int. 2007;78:323–327. doi: 10.1159/000100836. [DOI] [PubMed] [Google Scholar]
- 25.Milosevic M, Voruganti S, Blend R, et al. Magnetic resonance imaging (MRI) for localization of the prostatic apex: comparison to computed tomography (CT) and urethrography. Radiother Oncol. 1998;47:277–284. doi: 10.1016/s0167-8140(97)00232-6. [DOI] [PubMed] [Google Scholar]
- 26.Karademir I, Shen D, Peng Y, et al. Prostate volumes derived from MRI and volume-adjusted serum prostate-specific antigen: correlation with Gleason score of prostate cancer. AJR Am J Roentgenol. 2013;201:1041–1048. doi: 10.2214/AJR.13.10591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rais-Bahrami S, Siddiqui MM, Turkbey B, et al. Utility of multiparametric magnetic resonance imaging suspicion levels for detecting prostate cancer. J Urol. 2013;190:1721–1727. doi: 10.1016/j.juro.2013.05.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liao S, Gao Y, Shen D. Sparse patch based prostate segmentation in CT images. MICCAI; Nice, France: 2012. pp. 385–392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Turkbey B, Fotin SV, Huang RJ, et al. Fully automated prostate segmentation on MRI: comparison with manual segmentation methods and specimen volumes. AJR Am J Roentgenol. 2013;201:W720–729. doi: 10.2214/AJR.12.9712. [DOI] [PubMed] [Google Scholar]
- 30.Metz CE. ROC methodology in radiologic imaging. Invest Radiol. 1986;21:720–733. doi: 10.1097/00004424-198609000-00009. [DOI] [PubMed] [Google Scholar]
- 31.Pan X, Metz CE. The “proper” binormal model: parametric receiver operating characteristic curve estimation with degenerate data. Acad Radiol. 1997;4:380–389. doi: 10.1016/s1076-6332(97)80121-3. [DOI] [PubMed] [Google Scholar]
- 32.Metz CE, Wang P-L, Kronman H. A new approach for testing the significance of differences between ROC curves measured from correlated data. In: Deconinck F, editor. Information processing in medical imaging. The Hague, Netherlands: Martinus Nijhoff; 1984. pp. 432–445. [Google Scholar]
- 33.Moyer VA, Force UPST. Screening for prostate cancer: US Preventive Services Task Force Recommendation Statement. Ann Intern Med. 2012;157:120–134. doi: 10.7326/0003-4819-157-2-201207170-00459. [DOI] [PubMed] [Google Scholar]
- 34.Djavan B, Zlotta AR, Byttebier G, et al. Prostate specific antigen density of the transition zone for early detection of prostate cancer. J Urol. 1998;160:411–418. discussion 418–419. [PubMed] [Google Scholar]
- 35.Djavan B, Remzi M, Zlotta AR, et al. Complexed prostate-specific antigen, complexed prostate-specific antigen density of total and transition zone, complexed/total prostate-specific antigen ratio, free-to-total prostate-specific antigen ratio, density of total and transition zone prostate-specific antigen: results of the prospective multicenter European trial. Urology. 2002;60:4–9. doi: 10.1016/s0090-4295(02)01896-4. [DOI] [PubMed] [Google Scholar]
- 36.Ohi M, Ito K, Suzuki K, Yamamoto T, Yamanaka H. Diagnostic significance of PSA density adjusted by transition zone volume in males with PSA levels between 2 and 4 ng/ml. Eur Urol. 2004;45:92–97. doi: 10.1016/j.eururo.2003.07.001. [DOI] [PubMed] [Google Scholar]
- 37.Kaplan SA, Te AE, Pressler LB, Olsson CA. Transition zone index as a method of assessing benign prostatic hyperplasia — correlation with symptoms, urine flow and detrusor pressure. J Urol. 1995;154:1764–1769. [PubMed] [Google Scholar]
- 38.Schiebler ML, Tomaszewski JE, Bezzi M, et al. Prostatic-carcinoma and benign prostatic hyperplasia — correlation of high-resolution mr and histopathologic findings. Radiology. 1989;172:131–137. doi: 10.1148/radiology.172.1.2472644. [DOI] [PubMed] [Google Scholar]
- 39.Ishida J, Sugimura K, Okizuka H, et al. Benign prostatic hyperplasia — value of MR-imaging for determining histologic type. Radiology. 1994;190:329–331. doi: 10.1148/radiology.190.2.7506836. [DOI] [PubMed] [Google Scholar]
- 40.Oto A, Kayhan A, Jiang YL, et al. Prostate cancer: differentiation of central gland cancer from benign prostatic hyperplasia by using diffusion-weighted and dynamic contrast-enhanced MR imaging. Radiology. 2010;257:715–723. doi: 10.1148/radiol.10100021. [DOI] [PMC free article] [PubMed] [Google Scholar]


