Abstract
Cell-based technologies are used as a therapeutic strategy in spinal cord injury (SCI). Mesenchymal stem cells (MSCs), which secrete various neurotrophic factors and cytokines, have immunomodulatory, anti-apoptotic and anti-inflammatory effects, modulate reactivity/phenotype of astrocytes and the microglia, thereby promoting neuroregeneration seem to be the most promising. The therapeutic effect of MSCs is due to a paracrine mechanism of their action, therefore the survival of MSCs and their secretory phenotype is of particular importance. Nevertheless, these data are not always reported in efficacy studies of MSC therapy in SCI. Here, we provide a review with summaries of preclinical trials data evaluating the efficacy of MSCs in animal models of SCI. Based on the data collected, we have tried (1) to establish the behavior of MSCs after transplantation in SCI with an evaluation of cell survival, migration potential, distribution in the area of injured and intact tissue and possible differentiation; (2) to determine the effects MSCs on neuronal microenvironment and correlate them with the efficacy of functional recovery in SCI; (3) to ascertain the conditions under which MSCs demonstrate their best survival and greatest efficacy.
Keywords: spinal cord injury, mesenchymal stem cells, survival, migration, Rho/ROCK/PTEN, astrocytes, microglia, myelin-forming cells, axon growth, tissue integrity
Introduction
Spinal cord injury (SCI) leads to activation/inhibition of numerous cascades of intracellular and intercellular communication, and a change of transcription and translation levels of different genes involved in post-traumatic response (Mortazavi et al., 2015; Witiw and Fehlings, 2015; Quadri et al., 2018). Despite advances in understanding of the pathogenesis of a traumatic injury of the nervous system, current treatments for SCI are insufficient. There is a compelling need for novel treatment approaches based on the understanding of molecular and cellular mechanisms of SCI. Cell-based therapy is one of the approaches to prevent consequences of traumatic injury and to maintain the regeneration of nerve fibers. The use of stem and progenitor cells is aimed at restoring a tissue matrix with pathways to guide axonal growth, maintenance and re-myelination of axons, a trophic supply of injured and viable neurons, and the proliferation of the spinal cord’s own stem cells (Sabapathy et al., 2015; Assinck et al., 2017; Galieva et al., 2017).
At present mesenchymal stem cells (MSCs) are considered a promising material to stimulate neuroregeneration, related to their high biosafety and immunomodulatory properties; their ability to synthesize neurotrophic and proangiogenic factors by promoting the survival and regeneration of neurons and the growth of axons and angiogenesis (Laroni et al., 2015; Qu and Zhang, 2017; Khan et al., 2018). Various methods of MSC transplantation, even under conditions of multiple administrations, cause neither toxicity, nor induce tumor formation in animals and humans (Ra et al., 2011; Barkholt et al., 2013; Rengasamy et al., 2016). Preclinical studies demonstrate positive and promising results of MSC-based therapy in SCI. Attempts are being made to discover cellular and molecular mechanisms of MSC-mediated effects on reactivity/phenotype of astrocytes and microglia, the maintenance of a pool of myelin-forming cells and neurons, axonal growth and tissue integrity in general. Methods and sources for obtaining MSCs, as well as conditions of their culture might have a significant impact on the metabolic and secretory profile and the membrane expression profile of a resulting cell-based product. As the paracrine mechanism of MSCs action determines their therapeutic effect, special attention is paid to important aspects such as the preservation of MSCs viability and the optimal delivery of secreted therapeutic factors by them into the area of SCI. However, there is still a necessity to understand the cellular and molecular mechanisms by which MSCs ameliorate a SCI outcome (Park, 2018).
This review includes summaries of preclinical trials data evaluating the efficacy of MSCs in animal models of SCI. Based on the data collected we have tried (1) to establish the behavior of MSCs after transplantation in SCI with an evaluation of cell survival, migration potential, distribution in the area of injured and intact tissue and possible differentiation; (2) to determine effects MSCs on neuronal microenvironment and correlate them with the efficacy of functional recovery in SCI; (3) to ascertain the conditions under which MSCs demonstrate their best survival and greatest efficacy.
An electronic search of the Medline datebase for articles describing animal model of SCI from 1992 to 2018 was performed using the folowing conditions: SCI model (animal: rats, mice, pigs, dogs, non-human primates), MSC transplantation, animal behavior/physiology. The results were further screened by title/abstract and non-SCI experiments and review articles were excluded.
Behavior of MSCs in the Area of SCI
Essential cytogenetic processes such as the viability of MSCs, their migration potential, proliferative activity, self-maintenance and differentiation are controlled by numerous molecular signals from their microenvironment. These signals converge via specific receptor inputs on intracellular signaling pathways whose number is quite limited. Despite a large number of studies where MSC viability in the area of SCI was evaluated, to date there are still contradictory data. Additional Table 1 (86.4KB, pdf) contains the published data available on the duration of MSC survival in the area of SCI, their migration potential and possible differentiation.
Behavior of MSCs in the area of SCI based on preclinical trials data
The behavior of MSCs in the area of SCI depends on the route (intraspinal, intrathecal, intravenous and others) and type of cell transplantation, (xenogenic, allogenic), methods of cell labeling (green fluorescent protein-transgenic mice/rats, antibodies, green fluorescent protein-expressing viral vectors, fluorescent nanoparticles and other tracers of cells) and imaging techniques (confocal microscopy, in vivo imaging instruments (IVIS) system etc.). The SCI model (contusion, compression, ischemia, hemisection or transection) can also play a great role in homing and survival of MSCs. The latter is due to the different nature of nervous tissue alterations leading to disimilar pathophysiological mechanisms. Among SCI models, the most suitable for clinical use is the model of a dosed contused injury of spinal cord (Gruner, 1992). As a result of this contusion, the destruction of the nervous tissue occurs is similar to the damage observed in humans, including gliosis, demyelination and cyst formation.
MSCs demonstrate good survival (for at least 4–8 weeks) after an intraspinal injection of allogenic cells in the area of SCI (Ryu et al., 2012; Takahashi et al., 2018a). The results obtained are associated with an increased expression by implanted MSCs of the prosurvival signaling factors such as Akt and extracellular regulated protein kinase 1/2. However, intraspinal injections of MSCs were shown preferential distribution in the spinal cord lesion site (Ryu et al., 2012; Ribeiro et al., 2015; Takahashi et al., 2018a) and less often rostrally and caudally from the epicenter (Nakajima et al., 2012). Neirinckx et al. (Neirinckx et al., 2015) detected no Cell Tracker Green labeled MSCs in the area of SCI at 4 weeks after an intraspinal injection that might be due to low signal retention of fluorescent dye data. For example, it has been previously shown that Cell Tracker Green CMFDA cannot be reliably used to evaluate the migration of bone-marrow derived angiogenic cells (Beem and Segal, 2013).
Intravenously implanted MSCs are capable of targeted migration towards the SCI epicenter (Kim et al., 2015; Ramalho et al., 2018). At the same time, they demonstrated a better survival rostrally from the injury epicenter with distribution both in the grey and white matter of the spinal cord and around the cavitation area (Yang et al., 2018). The data obtained by Matsushita et al. (2015) stand out from the studies available, who found no migration of intravenously implanted MSCs into either contused or intact spinal cords. The authors based the evidence on stabilization of the blood-spinal cord barrier and improved locomotor functions and related this with indirect effects (by means of secreted factors) of MSCs implanted in the spinal cord vasculature.
Intrathecal (an intraventricular injection is its special case) and intra-arterial injections of MSCs in SCI are less often used in preclinical trials of their transplantation. The latter is more often associated with technical difficulties of reproducing these manipulations. With the intrathecal injection there is a low MSC migration into the lesion site (Urdzíková et al., 2014; Krupa et al., 2018). This might have caused the lack of available data on long-term survival of MSCs in the area of SCI after such administration.
The application of MSCs combined with scaffolds on top of the injury has become increasingly popular due to the possibilities of clinical use of this cell-based therapy in SCI. This type of transplantation demonstrates good MSC survival (for at least ten weeks) in the area of SCI (Mukhamedshina et al., 2017a; Sabino et al., 2018), that might be due to the possibility of maintaining matrix-enclosed cells which create a specific microenvironment similar to the natural one. It should be noted that the available data on MSCs survival in their xenotransplantation in SCI are not significantly different when compared to similar options of allogenic cells transplantation (Additional Table 1 (86.4KB, pdf) ). A comparative analysis of survival of MSCs derived from different sources (adipose tissue, bone marrow, umbilical cord blood and Wharton’s jelly) in the area of SCI showed a shorter survival of bone marrow derived-MSCs (Ryu et al., 2012; Takahashi et al., 2018a). This might be due to a low expression level of chemokine (C-X-C motif) ligand (CXCL)12 in bone marrow derived-MSCs which can improve MSC survival and proliferation in vitro (Liu et al., 2011; Takahashi et al., 2018a).
The possibilities of unorthodox MSC plasticity/transdifferentiation were shown in induction medium culture in vitro (Reyes and Verfaillie, 1999; Hermann et al., 2004) and in experimental models of various pathologies when these cells were administered in vivo, including in SCI (Ryu et al., 2012; Sabino et al., 2018). Previously investigators have most actively described the possibilities of MSC transdifferentiation into the neural lineage, using the capabilities of these cells to acquire specific morphology and the expression of neuron and glial cell markers. However, video time-lapse microscopy of the MSCs culture showed that specific morphological alterations (MSC’s assumption of a neural and glial cell shape) were not genuine transdifferentiation but resulted from degenerative changes when exposed to the cell culture medium used to induce the neural lineage (Bertani et al., 2005). The expression of neural markers detected with immunocytochemistry takes place very rapidly as a rule for genuine transformation to occur. For example, Sabino et al. (2018) showed that on day 7 after application of MSCs combined with a fibrin matrix in the site of transection the transplanted cells had already started expressing Nestin, NG2, β-III tubulin, Vimentin, neurofilament (NF) and neuron specific enolase. In another study the MSCs injected into the area of SCI began to express NF160, NeuN and glial fibrillary acidic protein (GFAP) at eight weeks after transplantation (Ryu et al., 2012). However, the specificity of neural markers such as βIII-tubuline and NeuN raises doubts and their production might result from the increased expression of MSC-specific genes (Pontius et al., 2003). Immunophenotyping which analyzes few markers in the cells taken out of their physiological “context” is not considered to be sufficient evidence to identify a particular cell type (Vladimirskaya, 2007; Vellosillo et al., 2017).
Gene expression profiling which makes it possible to analyze the transcriptome of hundreds and thousands of genes can be the most convincing evidence of MSC transdifferentiation into non-mesenchymal cell types. However, in this respect MSCs are also considered to be a complex object owing to the very high gene expression by these cells. For example, the number and diversity of MSC expressed genes are significantly higher than those of hematopoietic stem cells (Silva et al., 2003). Expression profiling of more than 21,000 genes performed with chemical induction of MSC neural differentiation in vitro demonstrated the lack of transcription of nervous tissue-specific genes and activation of the same genes as in MSC transformation into other cell types (Bertani et al., 2005). Thus, it was concluded that there is no absolutely reliable evidence of MSC transdifferentiation in vitro into non-mesenchymal cell types.
Rho/ROCK/PTEN Signaling Pathway in Mesenchymal Stem Cells
Rho/ROCK/PTEN (small Rho GTPases, Rho-associated kinase, phosphatase and the tensin homolog that is deleted on chromosome 10) is one of the key intracellular signaling pathways where numerous molecular signals from the microenvironment converge via special receptor inputs. Despite the significant interest of MSC researchers, the evidence disclosing the role the intracellular Rho/ROCK/PTEN signaling pathway plays in phenotype control, survival, proliferation and migration potential of MSCs is still lacking. ROCK inhibitors were shown to improve the physiological function of cryopreserved MSCs significantly within a cytoskeleton (Bit et al., 2017).
The effect of inhibiting the intracellular Rho/ROCK/PTEN signaling pathway on the phenotype and behavior of cells when transplanted in vivo in order to prevent neurodegeneration has not been studied. In this respect two approaches can be considered related. The first involves the management of neurodegeneration and stimulation of neuroregeneration using inhibitors of RhoА (Lord-Fontaine et al., 2008; McKerracher and Anderson, 2013; Drummond et al., 2014; Wu and Xu, 2016), ROCK (Furuya et al., 2009; Chiba et al., 2010; Yu et al., 2016; Li et al., 2017) and PTEN (Chen et al., 2015; Knafo et al., 2016) in different experimental models. The second targets the silencing of genes encoding for key molecules of the Rho/ROCK/PTEN signaling pathway through genetic constructions such as anti-sense oligonucleotides (Huang et al., 2015), microRNA (Lu et al., 2015), small interfering RNA (Wen et al., 2014; Ding et al., 2015; Gwak et al., 2017), and RNA spikes (Zukor et al., 2013; Haws et al., 2014; Lewandowski and Steward, 2014), inserted with viral vectors directly into spinal cord structures as well as using the Cre-Lox recombination technology (Willenberg et al., 2016).
There are data on a combined use of selective inhibitors of small GTPase, ROCK and PTEN with stem cell transplantation in order to prevent consequences of neurodegeneration. For example, the administration of fasudil, a ROCK selective inhibitor, for two weeks combined with transplantation of bone marrow-derived stromal cells significantly increased the number of regenerating axons in the corticospinal tract ingrowing through the area of SCI in rats but did not enhance the locomotor recovery (Chiba et al., 2010). However, another group of researchers managed to demonstrate improved locomotor rather than sense function, increased numbers of regenerating axons and serotonergic fibers in an area rostral to the injury epicenter as well as significantly reduced abnormal cavities with co-administration of fasudil intrathecally for 4 weeks and a single injection of bone marrow-derived stromal cells into the lesion site (Furuya et al., 2009). A positive synergistic effect of fasudil and transplantation of bone marrow-derived MSCs was demonstrated in experimental autoimmune encephalomyelitis in mice (Yu et al., 2016). In this case fasudil inhibited the effect of proinflammatory molecules such as TLR-4/MyD88, interferon (IFN)-γ, interleukin (IL)-1β and tumor necrosis factor-α (TNF-α), and activated the production of a glial-cell derived neurotrophic factor and a brain-derived neurotrophic factor (BDNF). It should be noted however that whether the ROCK inhibitor affects survival, phenotype characteristics and synthetic activity of the cells transplanted, MSCs in particular, remains unclear in all experiments of fasudil co-administration with cell-based therapy.
Cdc42 is a significant factor in extension of axonal growth cones and regulation of cell proliferation, and is involved in lipotoxic effects of palmitate in culturing Wharton’s jelly-derived MSCs. The delivery of constitutively active Cdc42 into the area of SCI reduces the number of reactive astrocytes and chondroitin sulfate proteoglycan deposition (Jain et al., 2011). At the same time, the use of shRNA against Cdc42 attenuated palmitate-induced synthesis of proinflammatory cytokines and cell death (Lu et al., 2017). These results indicate a mechanistic role of Cdc42 in Wharton’s jelly-derived MSCs proliferation and determine that the Cdc42 activity is a promising pharmacological target to reduce lipotoxic cell dysfunction and death.
Molecular and Cellular Mechanisms Mediating Impact of Mesenchymal Stem Cells on Neuronal Microenvironment in the Area of Spinal Cord Injury
Astrocytes
Traditionally, studies aimed to assess the efficacy of MSCs use for the purpose of neuroregeneration in SCI are focused on the state of reactive astrocytes and extracellular matrix in the glial scar. For this purpose, usually the expression level of GFAP and less often that of proteoglycans are determined in the area of SCI (Additional Table 2 (139.2KB, pdf) ). When the expression of these markers is decreased, it is often concluded that the glial barrier is reduced and axonal growth results (Liu et al., 2018; Yang et al., 2018). Such a judgement is not entirely objective, since the glial barrier is a complex and multicomponent structure. The glial barrier is composed not only of reactive astrocytes, but also macroglia precursors and reactive microglia/macrophages actively penetrating into a scar; there are also fibroblasts and perivascular cells present (Chelyshev et al., 2013; Yuan and He, 2013). Different post-traumatic periods are evaluated for changes in the glial scar and astrocyte activation in MSC transplantation. In this context the role of the glial barrier and reactive astrocytes is most often considered a negative one despite a known positive role in maintaining the spinal cord tissue structural integrity and the process of neuroregeneration during an early post-traumatic period (Adams and Gallo, 2018). Kim et al. (2015) demonstrated that an early intravenous administration of allogenic MSCs to dogs increased the expression of GFAP by 7 days after SCI. They consider the results obtained as positive taking into account the formation of a neuroprotective microenvironment by GFAP-expressing astrocytes for neurogenesis with transplanted MSCs.
Preclinical in vivo trials using MSCs transplantation after SCI and obtained results in the interpretation of the authors*
However, most studies with MSC transplantation in SCI give evidence to reduced astrocyte responsiveness both in acute (Liu et al., 2018), subacute (Ruppert et al., 2018), and chronic periods after injury (Ryu et al., 2012; Krupa et al., 2018; Mukhamedshina et al., 2018; Yang et al., 2018). The possibilities of preventing astrocytosis are attributed to the ability of MSCs to decrease cyclooxygenase-2 (COX-2) and IL-6 cytokine levels (Nakajima et al., 2012; Ryu et al., 2012; Liu et al., 2018; Sun et al., 2018) (Figure 1). It has previously been shown that reactive and hypertrophic astrocytes start expressing COX-2 within the area of ischemia in response to proinflammatory stimuli or an injection of neurotoxins (Hirst et al., 1999; Maślińska et al., 1999; Font-Nieves et al., 2012). Thus there are approaches involving the use of COX-2 inhibitors after SCI, that reduce oxidative stress and promote neuroprotection (Hakan et al., 2011; Hou et al., 2015; Yuksel et al., 2018). A decreased COX-2 level in MSC transplantation in SCI can be considered positive and results from reduced levels of prostaglandins which regulate MSC differentiation into osteoblastic cells (Zhang et al., 2002; Banovac et al., 2004). Heterotypic ossification is thereby prevented which is the most common orthopedic complication after SCI (Banovac et al., 2004).
Figure 1.
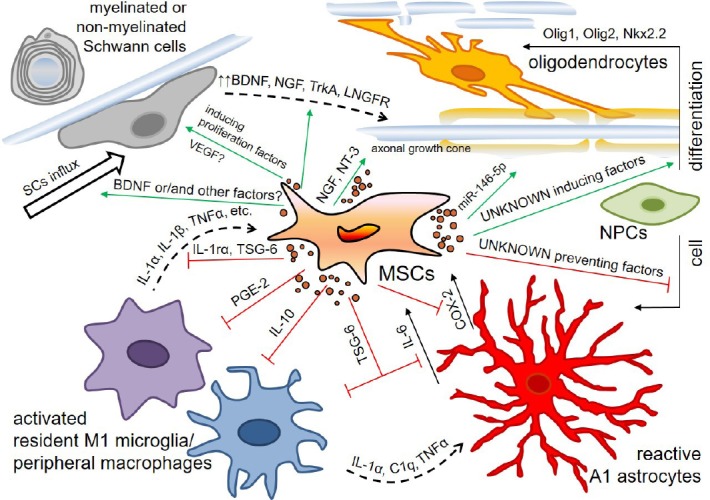
Schematic diagram for some effects of mesenchymal stem cells (MSCs) on the neuronal microenvironment in the area of spinal cord injury (SCI).
Activated resident microglia and peripheral macrophages attracted in the area of SCI produce proinflammatory cytokines such as interleukin (IL)-1α, IL-1β, tumor necrosis factor (TNF)-α, C1q and so on and activate A1 astrocytes and MSCs. In response to these stimuli and probably other signals the MSCs start to secret anti-inflammatory factors such as IL-1ra, TNF-stimulated gene-6 (TSG-6), prostaglandin E2 (PGE2), and IL-10 and modulate a phenotype of microglia/macrophages toward the anti-inflammatory M2 one and reduce the reactivity of astrocytes. The MSCs induce neural progenitor cells differentiation into oligodendrocytes and prevent differentiation into astrocytes. They facilitate myelination and axon growth by producing miR-146-5p and neurotrophic factors, lead not only to influx of Schwann cells in the area of SCI, but promote an increased expression of brain-derived neurotrophic factor (BDNF), nerve growth factor (NGF) and its high- and low-affinity receptors (TrkA and LNGFR) in these cells. COX-2: Cyclooxygenase-2; NT-3: neurotrophin-3.
IL-6 can play a double role, being involved in either a classic signaling pathway and thereby promoting a reduction of inflammation and enhanced regeneration, or a trans-signaling pathway important for cellular communication (Campbell et al., 2014; Scheller et al., 2014; Schaper and Rose-John, 2015). IL-6 is synthesized mainly by activated astrocytes in the central nervous system (Guptarak et al., 2013; Choi et al., 2014; Gruol, 2016). Decreased IL-6 levels are mediated by MSC secretion of TNF-stimulated gene-6 (TSG-6), released within exosomes (Wang et al., 2012; Qi et al., 2014; Song et al., 2017; Chaubey et al., 2018). Later TSG-6 reduces NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) signaling, thereby modulating a diminishing cascade of proinflammatory cytokines (TNF, IL-1β, IL-6, IL-1α, etc.).
It should be noted that astrocytes exist in at least two distinct reactive states such as A1 neuroinflammatory reactive astrocytes and ischemia-induced A2 neuroprotective reactive astrocytes (Liddelow and Barres, 2017). A1 reactive astrocytes induced by microglia activation quite rapidly respond to neurotrauma and lead to death of axotomized neurons (Liddelow and Barres, 2017). It was not until recently that it became possible to distinguish between different activation states of reactive astrocytes by means of complement component C3 which is specifically upregulated only in A1 neuroinflammatory reactive astrocytes (Liddelow and Barres, 2017). This resulted in the advent of publications evaluating a phenotype of reactive astrocytes in the area of SCI following MSC-based therapy. For example, Liu et al. (2018) were the first to demonstrate that an intravenous injection of MSCs-derived exosomes after SCI resulted in a decreased number of C3+/GFAP+-astrocytes in the lesion area as early as in 24 hours. The results obtained may be attributed to the secretion of TSG-6 by MSCs and consequently decreased NF-κB signaling, which induces А1 neuroinflammatory reactive astrocytes (Lian et al., 2015; Liddelow et al., 2017).
Despite the great progress in studying astrocytes in general and the MSC impact on them in particular, there is a need for communications between these cells in the setting of the pathological conditions in SCI to be more completely elucidated. Eventually this would provide a more complete understanding of the contribution of astrocytes during various post-traumatic periods and how we can modulate their inflammatory or neuroprotective potential with the use of MSCs.
Microglia and inflammation
The activation of microglia which are the first to respond to nervous tissue damage is one of the essential events of post-traumatic reactions. The phase of primary microglia activation peaks at post-traumatic day 7, its reactivation occurring in 2 weeks and lasting for up to 180 days (Kigerl et al., 2006; Beck et al., 2010). Activated microglia can synthesize not only trophic biomolecules such as neurotrophins, glutamate transporters and antioxidants, but also effectors that can be potentially neurotoxic, such as nitric oxide and pro-inflammatory cytokines (Persson et al., 2005; Lai and Todd, 2006; Hellwig et al., 2013). Owing to this dual nature the role that microglia play in regulating neuroregeneration at different port-traumatic time points is still controversial.
To date several states of microglia polarization are distinguished - they are classic activation (M1), alternative activation (M2a), alternative type II activation (M2b) and acquired deactivation (M2c). A number of investigators question whether the microglia can acquire a М3 phenotype (Walker and Lue, 2015). A number of proliferation studies showed which markers were specific for the microglia activated by a classic or alternative pathway (Martinez et al., 2006, 2013). Classically activated (M1) cells can produce reactive oxygen intermediates as well as proinflammatory cytokines such as TNF-α, IL-1β and IL-6, concurrently mediating inflammatory tissue damage. М2 microglia/macrophages are a phenotype of cells responding to IL-4 and IL-13, at present known as M2a. The phenotype M2a microglia is considered to have increased phagocytic activity and to produce growth factors such as an insulin-like growth factor-1 and proinflammatory cytokines such as IL-10 (Martinez and Gordon, 2014). The microglia of this type can dispose of cellular debris and stimulate tissue regeneration. An alternative activation was subdivided into two subcategories such as M2b and M2c. M2b is induced by ligation of immunoglobulin Fc-gamma-receptors that results in the IL-12 expression, increased IL-10 secretion and HLA-DR expression. This phenotype is also characterized by an increased expression of CD32 and CD64, which were found to be expressed by the cerebral microglia in Alzheimer’s disease (Peress et al., 1993) and to be related to increased phagocytosis activity. M2c (acquired deactivation) can be caused by the anti-inflammatory cytokine IL-10 or glucocorticoids, an increased expression of the transforming growth factor), sphingosine kinase (SPHK1) and CD163, a membrane-bound receptor for haptoglobin/hemoglobin complexes (Wilcock, 2014).
Some evidence indicates that MSCs can modulate a phenotype of microglia/macrophages toward the anti-inflammatory M2 phenotype in SCI (without detection of alternative activation) (Additional Table 2 (139.2KB, pdf) ). Previous studies in vitro may have attracted separate attention to the polarization of microglia/macrophages in MSC transplantation after SCI. MSCs co-cultured with macrophages showed a high expression level of soluble (IL-10) and surface (CD206) markers for M2 macrophages and increased phagocytic activity (Kim and Hematti, 2009). Subsequently it was shown in models of sepsis and peritonitis that mechanisms of MSC-mediated regulation on macrophages might depend upon PGE2 and TSG-6 secretion (Németh et al., 2009; Choi et al., 2011) (Figure 1). In SCI models transplantation of both MSCs and MSC-derived microvesicles increased the number of М2 microglia/macrophages on post-injury day 7–9, promoting a proregenerative environment (Nakajima et al., 2012; Caron et al., 2016; Sun et al., 2018). This effect of MSCs might be attributed to MSC-mediated increased level of IL-4, IL-13 and IL-10, which promote modulation of microglia/macrophages toward М2а and М2с neuroprotective phenotypes, respectively, as well as decreased levels of TNF-α and IFN-γ which promote polarization into the M1 phenotype. This assumption was confirmed by studies in vitro (Zhang et al., 2010; Pietilä et al., 2012; Shin et al., 2016) and in vivo (Nakajima et al., 2012; Sun et al., 2018).
In addition to polarization into the M2 phenotype MSC transplantation in SCI results in a decreased total number of microglia/macrophages in the area of injury. Pan markers such as Iba1 and CD68 are often used for this evaluation. There was a decreased total microglia/macrophages number in the area of SCI after MSC injection in acute (Liu et al., 2018), subacute (Zeng et al., 2011; Ruppert et al., 2018) and chronic periods of SCI (Neirinckx et al., 2015; Liu et al., 2018; Mukhamedshina et al., 2018; Yang et al., 2018). Only a few publications provide evidence for the lack of changes in total number of microglia/macrophages at a lesion site following transplantation of MSCs or their microvesicles (Neirinckx et al., 2015; Sun et al., 2018).
There is a direct relationship between the М2 microglia activation and anti-inflammatory effects in MSC transplantation in SCI. IL-1α and other molecules released within a focus of tissue destruction activate the resident microglia and migrating macrophages in the area of injury. The latter in turn activate MSCs by producing proinflammatory cytokines such as IL-1α, IL-1β or TNF-α. In response the MSCs start secreting anti-inflammatory factors such as TSG-6, PGE2 and IL-1ra, which both modulate the activation of M2 microglia/macrophages and reduce the effects of proinflammatory cytokines (Prockop and Oh, 2012). A subsequent decrease in IL-1β and TNF-α, which was observed in MSC transplantation in SCI and correlates with a functional outcome (Zeng et al., 2011, 2016; Urdzíková et al., 2014; Liu et al., 2018), is related to MSC-mediated suppression of NLRP3 inflammasome including by means of PGE2 action (Oh et al., 2014; Shin et al., 2016).
In conclusion, MSCs can act as cellular modulators of microglia/macrophages polarization by regulating the production of different cytokines and a proinflammatory response as a whole that results in attractive therapeutic outcomes for such type of stem cells.
Oligodendrocytes and Schwann cells
Demyelination is part of a general process of secondary degeneration in SCI. The number of demyelinated axons was shown in a rat model of SCI to peak 24 hours after injury, to decline over the next 1–2 weeks and to increase progressively by 450 days post injury (Totoiu and Keirstead, 2005). There were demyelinated axons at all experimental time points, indicating that post-traumatic processes were still ongoing. At the same time, not only oligodendrocytes but also Schwann cells were involved in the process of remyelination by the end of week 2. The latter were shown to migrate from peripheral nervous structures resulting from a disruption of the barrier integrity, to participate not only in remyelination of central axons in the area of injury, but also in the restoration of conduction (Franklin and Hinks, 1999; Jasmin et al., 2000; Shaymardanova et al., 2013; Mukhamedshina et al., 2017b; Galieva et al., 2018).
MSC-derived soluble factors have been previously shown to induce oligodendrogenesis by reducing the anti-oligodendrogenic determinant Id2 and increasing the pro-oligodendrogenic factor Olig2 expression in neural progenitor cells (Li et al., 2009; Steffenhagen et al., 2012). It was shown in vitro that MSCs could not only direct proliferating NPCs toward an oligodendrocyte fate but also induce oligodendrocyte differentiation (Rivera et al., 2006). The MSCs facilitate myelination by producing miR-146-5p and delivering them via exosomes (Lindsay et al., 2016). In co-culture the MSCs lead not only to improved survival and proliferation of Schwann cells, but promote an increased expression of BDNF, nerve growth factor (NGF) and its high- and low-affinity receptors (TrkA and LNGFR) in these cells (Wang et al., 2009) (Figure 1).
Not more than 1/3 of publications devoted to the evaluation of MSCs efficacy in SCI in vivo, carry out an analysis of post-traumatic changes in the population of myelin-producing cells and myelination as a whole (Additional Table 2 (139.2KB, pdf) ). Various MSC transplantations demonstrate an increase of myelin retention and the number of myelinated axons in the lesion site during a chronic post-injury period (Papa et al., 2018; Ramalho et al., 2018). Using electron microscopy Nakano et al. (2013) found in the same period that the number of Schwann cells associated with axons in the astrocyte-devoid lesion site increased following an intraventricular injection of MSCs. It was shown that MSCs could elicit the influx of Schwann cells into the site of injury and improve their survival (Ding et al., 2014). The influx and proliferation of Schwann cells can also be attributed to MSC secretion of BDNF, vascular endothelial growth factor (VEGF) and other unknown inducing factors. BDNF was shown to promote a significant expansion in the number of Schwann cells at three weeks after SCI, with VEGF stimulating their proliferation (Sondell et al., 2000; Blesch and Tuszynski, 2007). An intravenous injection of MSCs in the early post-injury period increased the expression of GalC, a marker of mature oligodendrocytes (Kim et al., 2015). However, the expression of an oligodendrocyte transcription factor (Olig2), which regulates oligodendrocyte differentiation does not seem to change, since the mRNA analysis of the Olig2 gene shows no significant differences with control groups (Urdzíková et al., 2014; Mukhamedshina et al., 2018).
Thus an increased number of functioning oligodendrocytes and Schwann cells in the area of demyelination in SCI is important for assessing the effectiveness of its regeneration following cell-based therapy that must maintain the survival and differentiation of not only the spinal cord cells but also endogenous migrants. The further elucidation of molecular mechanisms of MSC-derived activity in relation to myelin-producing cells will be essential for the treatment of not only neurotrauma, but also demyelinating diseases.
Axonal outgrowth
The issue of axon regeneration, which is normally low due to the small intrinsic capacity of central nervous system axons for regeneration as well as the synthesis of growth inhibitor molecules upon injury is pressing in SCI (Sakamoto and Kadomatsu, 2017). The MSC capacity to stimulate axonal outgrowth has been identified (Lin et al., 2018). However, molecular and cellular mechanisms of stimulating axonal growth and sprouting by MSCs are not completely clarified.
In order to evaluate possible effects of transplanted MSCs on axon regeneration usually the expression level of neurofilaments (NF200) and less often that of GAP-43 (axon growth associated protein) are determined in the area of SCI (Additional Table 2 (139.2KB, pdf) ). However, it must be remembered that the increased expression of these proteins cannot yet indicate successful axon regeneration. The process of axonal regeneration involves 5 stages and has to end in synapse formation and the restoration of conduction along the axons as a result (Sakamoto and Kadomatsu, 2017). Therefore, axonal regeneration following MSC therapy must be confirmed with electrophysiological studies.
Axon growth following MSC transplantation is most often associated with their ability to form bridges via a spinal cord cavity and reduce a glial scar (Zurita and Vaquero, 2006; Wright et al., 2007; Lin et al., 2018). In vitro studies demonstrate that MSCs can act as “cellular bridges” and stimulate neurite outgrowth by reducing inhibitory substrates (Wright et al., 2007). Krupa et al. (2018) found the expression of GAP-43 increased and that of GFAP decreased in a dose-dependent manner – the higher the number of cells transplanted, the more intense axonal sprouting was at 9 weeks post injury after an intrathecal injection of MSCs. It has previously been demonstrated in vitro that MSCs cultured with brain slices start secreting trophic factors such as NGF and NT-3 (Pisati et al., 2007). These results were translated in vivo, therefore some researchers attribute a positive effect of MSCs on axon growth to the increased expression of NGF and NT-3, which play an important role in this process (Li et al., 2016; Ramalho et al., 2018).
The search for a most effective promoter for axonal regeneration in SCI is still ongoing. MSC-based therapy can mediate axonal outgrowth that has been shown in some animal experiments. However, studies which evaluate post-traumatic changes in axonal regeneration following MSC transplantation in SCI have to be more detailed, and to reveal not only the potential for axonal growth and sprouting but also the functional competence of the changes observed, that is, the possibilities for the restoration of long-term conduction along these axons.
Neuroprotection
The anti-apoptotic effect of MSCs which is associated with the synthesis of bioactive molecules capable of inhibiting apoptosis is of a great importance in their gross neuroprotective effects. It was shown in experiments in vivo in a model of SCI that MSCs can affect a decrease in the expression of pro-apoptotic molecules and an increase of that of anti-apoptotic ones (Mukhamedshina et al., 2017a; Liu et al., 2018). However, when assessing the efficacy of MSCs in SCI most often terminal dexynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL)+-cells are generally assessed, where their decrease following therapy has been reported (Xiong et al., 2017; Liu et al., 2018; Takahashi et al., 2018b; Yang et al., 2018). Still, the molecular mechanisms of MSC-mediated anti-apoptotic effects have not yet been completely discovered.
The maintenance of adequate blood perfusion in the site of injury, in rostral and caudal regions of the spinal cord is essential for neuroprotection. Therefore the angiogenic activity of MSCs is significant for neuroregeneration. When detecting the MSCs in the walls of blood vessels an assumption was made that these cells might be involved in regulation of their growth and maintenance of a stable vessel. Subsequently the ability of MSCs to stimulate the growth of blood vessels including those regenerating after various injuries was established. The main mechanisms of angiogenic activity of MSCs are due to their ability to secrete pro-angiogenic factors and cytokines (VEGF, fibroblast growth factor-2, transforming growth factor-β, hepatocyte growth factor, etc.), as well as to express the adhesion receptor integrin α6β1, promote vessel sprouting, MSC proliferation and pericyte differentiation (Carrion et al., 2013). The transplantation of MSCs was shown to promote angiogenesis and reconstruction of the microvasculature network that enhances a functional recovery after SCI (Zeng et al., 2011; Zhou et al., 2016; Huang et al., 2017). This effect is most often interpreted when increased CD31+- and rat endothelial cell antigen‐1+-cell levels and enhanced VEGF expression are detected. An increased VEGF expression by MSCs is in turn related to the action of IL-8 (Hou et al., 2014). The MSCs transplanted closely associate with endothelial cells after SCI, that is due to performing a pericyte function and the possibility of contact stabilization of growing and formed blood vessels in later stages of angiogenesis (Zeng et al., 2011).
In general the neuroprotective effect of MSCs is due to their secretion of neurotrophic factors and cytokines. The latter have the above effects which MSCs exert through various signaling pathways, activated via specific receptors on target cells. Therefore the quality of MSCs derived from various sources is now top priority, being ahead of issues of delivery, dosage, etc.
Tissue integrity
The primary injury of the spinal cord associated with progressive tissue necrosis results in post-traumatic cavitation and triggers processes of a secondary injury which cause death of neurons and glial cells away from the injury epicenter (Priestley et al., 2012; Ward et al., 2014). The regulation of a cell response in an acute period is critical to inhibit rapid progression of a secondary injury after SCI. Therefore, MSC transplantation during the acute period of SCI may be targeted not only to replace the cells lost, but also to modulate a cell response, to enhance anti-oxidant and pro-inflammatory mechanisms preventing further injury.
The reduction of both a total area of abnormal cavities and lesion/cavity volumes were reported in numerous publications on MSC transplantation in SCI (Nakajima et al., 2012; Nakano et al., 2013; Neirinckx et al., 2015; Liu et al., 2018; Mukhamedshina et al., 2018; Sun et al., 2018). In this case tissue integrity also increases; however, there are controversial data on the integrity of the white and grey matter (Urdzíková et al., 2014; Krupa et al., 2018; Mukhamedshina et al., 2018). MSC-mediated anatomical improvement is due to their complex action by which the MSC graft induces tissue protection/repair in a manner unlike that in acute and chronic periods of SCI. For example, MSC transplantation in an acute period may have beneficial effects through their anti-inflammatory activity and microglia polarization, whereas in the subacute/chronic phase after SCI, the MSCs may be used for neurostimulatory, glial scar reducing and cell bridging effects (Wright et al., 2011; Nakajima et al., 2012).
Not all studies confirm the MSC ability to improve the retention of spinal cord injured tissue (Kim et al., 2015; Takahashi et al., 2018b). The contradictory results might be attributed to different approaches to MSC transplantation and a too early evaluation of their efficacy under these criteria (in post-injury week 1). The timing of cell transplantation and the cell number, routes of administration (intraspinal, intrathecal, intravenous, application to the area of injury as part of a matrix), immunosuppression, as well as the quality of MSCs generated in culture are certain to be relevant. In general, all these issues have a direct impact on the efficacy of MSC-mediated regulation of the neuronal microenvironment discussed above.
Conclusion
The use of MSCs is presently associated with possible advances of regenerative medicine. The number of advocates and opponents of this trend is more and more rising both in science and in society. The increased number of skeptics is largely due to the lack of a complete disclosure of mechanisms underlying the therapeutic effect of MSCs and data on long-term results of their use. Existing preclinical studies give evidence to the ability of MSCs to stimulate neuroregeneration, in SCI in particular. Nevertheless, it seems difficult to interpret the results obtained and draw a parallel between observed posttraumatic reactions, which often lack a complete and objective evaluation, and the resulting functional outcome. Sometimes observed controversial data on the efficacy evaluation of MSC-based therapy in SCI are primarily due to a different secretory profile of the cells obtained, and only then by differences in protocol details.
Additional files:
Additional Table 1 (86.4KB, pdf) : Behavior of MSCs in the area of SCI based on preclinical trials data.
Additional Table 2 (139.2KB, pdf) : Preclinical in vivo trials using MSCs transplantation after SCI and obtained results in the interpretation of the authors.
Additional file 1: Open peer review reports 1 (120.4KB, pdf) and 2 (134.9KB, pdf) .
Footnotes
Conflicts of interest: The authors declare that there is no conflict of interest regarding the publication of this paper.
Financial support: The work was supported by a grant from the Russian Foundation for Basic Research, No.16-34-60101 (to YOM), a grant from the Ministry of Education and Science of the Russian Federation, No. 20.5175.2017/6.7 (to AAR). This work was performed in accordance with the Program of Competitive Growth of the Kazan Federal University.
Copyright license agreement: The Copyright License Agreement has been signed by all authors before publication.
Plagiarism check: Checked twice by iThenticate.
Peer review: Externally peer reviewed.
Open peer reviewers: Alessandra Pacini, University of Florence, Italy; Gabriele Siciliano, Universita degli Studi di Pisa, Italy.
P-Reviewers: Pacini A, Siciliano G; C-Editors: Zhao M, Yu J; T-Editor: Liu XL
Funding: The work was supported by a grant from the Russian Foundation for Basic Research, No. 16-34-60101 (to YOM), a grant from the Ministry of Education and Science of the Russian Federation, No. 20.5175.2017/6.7 (to AAR). This work was performed in accordance with the Program of Competitive Growth of the Kazan Federal University.
References
- 1.Adams KL, Gallo V. The diversity and disparity of the glial scar. Nat Neurosci. 2018;21:9–15. doi: 10.1038/s41593-017-0033-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Assinck P, Duncan GJ, Hilton BJ, Plemel JR, Tetzlaff W. Cell transplantation therapy for spinal cord injury. Nat Neurosci. 2017;20:637–647. doi: 10.1038/nn.4541. [DOI] [PubMed] [Google Scholar]
- 3.Banovac K, Williams JM, Patrick LD, Levi A. Prevention of heterotopic ossification after spinal cord injury with COX-2 selective inhibitor (rofecoxib) Spinal Cord. 2004;42:707–710. doi: 10.1038/sj.sc.3101628. [DOI] [PubMed] [Google Scholar]
- 4.Barkholt L, Flory E, Jekerle V, Lucas-Samuel S, Ahnert P, Bisset L, Büscher D, Fibbe W, Foussat A, Kwa M, Lantz O, Mačiulaitis R, Palomäki T, Schneider CK, Sensebé L, Tachdjian G, Tarte K, Tosca L, Salmikangas P. Risk of tumorigenicity in mesenchymal stromal cell-based therapies--bridging scientific observations and regulatory viewpoints. Cytotherapy. 2013;15:753–759. doi: 10.1016/j.jcyt.2013.03.005. [DOI] [PubMed] [Google Scholar]
- 5.Beck KD, Nguyen HX, Galvan MD, Salazar DL, Woodruff TM, Anderson AJ. Quantitative analysis of cellular inflammation after traumatic spinal cord injury: evidence for a multiphasic inflammatory response in the acute to chronic environment. Brain. 2010;133:433–447. doi: 10.1093/brain/awp322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Beem E, Segal MS. Evaluation of stability and sensitivity of cell fluorescent labels when used for cell migration. J Fluoresc. 2013;23:975–987. doi: 10.1007/s10895-013-1224-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bertani N, Malatesta P, Volpi G, Sonego P, Perris R. Neurogenic potential of human mesenchymal stem cells revisited: analysis by immunostaining, time-lapse video and microarray. J Cell Sci. 2005;118:3925–3936. doi: 10.1242/jcs.02511. [DOI] [PubMed] [Google Scholar]
- 8.Bit A, Kumar A, Singh AK, Rizvanov AA, Kiassov AP, Patra PK, Kumar M, Bissoyi A. Crosstalk between substrates and Rho-associated kinase inhibitors in cryopreservation of tissue-engineered constructs. Stem Cells Int 2017. 2017 doi: 10.1155/2017/1380304. 1380304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Blesch A, Tuszynski MH. Transient growth factor delivery sustains regenerated axons after spinal cord injury. J Neurosci. 2007;27:10535–10545. doi: 10.1523/JNEUROSCI.1903-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Campbell IL, Erta M, Lim SL, Frausto R, May U, Rose-John S, Scheller J, Hidalgo J. Trans-signaling is a dominant mechanism for the pathogenic actions of interleukin-6 in the brain. J Neurosci. 2014;34:2503–2513. doi: 10.1523/JNEUROSCI.2830-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Caron I, Rossi F, Papa S, Aloe R, Sculco M, Mauri E, Sacchetti A, Erba E, Panini N, Parazzi V, Barilani M, Forloni G, Perale G, Lazzari L, Veglianese P. A new three dimensional biomimetic hydrogel to deliver factors secreted by human mesenchymal stem cells in spinal cord injury. Biomaterials. 2016;75:135–147. doi: 10.1016/j.biomaterials.2015.10.024. [DOI] [PubMed] [Google Scholar]
- 12.Carrion B, Kong YP, Kaigler D, Putnam AJ. Bone marrow-derived mesenchymal stem cells enhance angiogenesis via their alpha6beta1 integrin receptor. Exp Cell Res. 2013;319:2964–2976. doi: 10.1016/j.yexcr.2013.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chaubey S, Thueson S, Ponnalagu D, Alam MA, Gheorghe CP, Aghai Z, Singh H, Bhandari V. Early gestational mesenchymal stem cell secretome attenuates experimental bronchopulmonary dysplasia in part via exosome-associated factor TSG-6. Stem Cell Res Ther. 2018;9:173. doi: 10.1186/s13287-018-0903-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chelyshev YA, Shaymardanova GF, Mukhamedshina YO, Nigmetzyanova MV. Glial barriers in spinal cord injury as a target of gene-cell therapy. Neuroll Bull. 2013;14:87–93. [Google Scholar]
- 15.Chen Y, Luo C, Zhao M, Li Q, Hu R, Zhang JH, Liu Z, Feng H. Administration of a PTEN inhibitor BPV(pic) attenuates early brain injury via modulating AMPA receptor subunits after subarachnoid hemorrhage in rats. Neurosci Lett. 2015;588:131–136. doi: 10.1016/j.neulet.2015.01.005. [DOI] [PubMed] [Google Scholar]
- 16.Chiba Y, Kuroda S, Shichinohe H, Hokari M, Osanai T, Maruichi K, Yano S, Hida K, Iwasaki Y. Synergistic effects of bone marrow stromal cells and a Rho kinase (ROCK) inhibitor, fasudil on axon regeneration in rat spinal cord injury. Neuropathology. 2010;30:241–250. doi: 10.1111/j.1440-1789.2009.01077.x. [DOI] [PubMed] [Google Scholar]
- 17.Choi H, Lee RH, Bazhanov N, Oh JY, Prockop DJ. Anti-inflammatory protein TSG-6 secreted by activated MSCs attenuates zymosan-induced mouse peritonitis by decreasing TLR2/NF-kappaB signaling in resident macrophages. Blood. 2011;118:330–338. doi: 10.1182/blood-2010-12-327353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Choi SS, Lee HJ, Lim I, Satoh J, Kim SU. Human astrocytes: secretome profiles of cytokines and chemokines. PLoS One. 2014;9:e92325. doi: 10.1371/journal.pone.0092325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ding P, Yang Z, Wang W, Wang J, Xue L. Transplantation of bone marrow stromal cells enhances infiltration and survival of CNP and Schwann cells to promote axonal sprouting following complete transection of spinal cord in adult rats. Am J Transl Res. 2014;6:224–235. [PMC free article] [PubMed] [Google Scholar]
- 20.Ding S, Bao Y, Lin Y, Pan Y, Fan Y, Wan J, Jiang J. Neuroprotective effect of functionalized multi-walled carbon nanotubes on spinal cord injury in rats. Int J Clin Exp Pathol. 2015;8:15769–15777. [PMC free article] [PubMed] [Google Scholar]
- 21.Drummond ES, Rodger J, Penrose M, Robertson D, Hu Y, Harvey AR. Effects of intravitreal injection of a Rho-GTPase inhibitor (BA-210), or CNTF combined with an analogue of cAMP, on the dendritic morphology of regenerating retinal ganglion cells. Restor Neurol Neurosci. 2014;32:391–402. doi: 10.3233/RNN-130360. [DOI] [PubMed] [Google Scholar]
- 22.Font-Nieves M, Sans-Fons MG, Gorina R, Bonfill-Teixidor E, Salas-Perdomo A, Marquez-Kisinousky L, Santalucia T, Planas AM. Induction of COX-2 enzyme and down-regulation of COX-1 expression by lipopolysaccharide (LPS) control prostaglandin E2 production in astrocytes. J Biol Chem. 2012;287:6454–6468. doi: 10.1074/jbc.M111.327874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Franklin RJ, Hinks GL. Understanding CNS remyelination: clues from developmental and regeneration biology. J Neurosci Res. 1999;58:207–213. [PubMed] [Google Scholar]
- 24.Furuya T, Hashimoto M, Koda M, Okawa A, Murata A, Takahashi K, Yamashita T, Yamazaki M. Treatment of rat spinal cord injury with a Rho-kinase inhibitor and bone marrow stromal cell transplantation. Brain Res. 2009;1295:192–202. doi: 10.1016/j.brainres.2009.07.087. [DOI] [PubMed] [Google Scholar]
- 25.Galieva LR, Mukhamedshina YO, Arkhipova SS, Rizvanov AA. Human umbilical cord blood cell transplantation in neuroregenerative strategies. Front Pharmacol. 2017;8:628. doi: 10.3389/fphar.2017.00628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Galieva LR, Mukhamedshina YO, Akhmetzyanova ER, Gilazieva ZE, Arkhipova SS, Garanina EE, Rizvanov AA. Influence of genetically modified human umbilical cord blood mononuclear cells on the expression of Schwann cell molecular determinants in spinal cord injury. Stem Cells Int 2018. 2018:4695275. doi: 10.1155/2018/4695275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gruner JA. A monitored contusion model of spinal cord injury in the rat. J Neurotrauma. 1992;9:123–126. doi: 10.1089/neu.1992.9.123. discussion 126-128. [DOI] [PubMed] [Google Scholar]
- 28.Gruol DL. Impact of increased astrocyte expression of IL-6, CCL2 or CXCL10 in transgenic mice on hippocampal synaptic function. Brain Sci. 2016;6:E19. doi: 10.3390/brainsci6020019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Guptarak J, Wanchoo S, Durham-Lee J, Wu Y, Zivadinovic D, Paulucci-Holthauzen A, Nesic O. Inhibition of IL-6 signaling: A novel therapeutic approach to treating spinal cord injury pain. Pain. 2013;154:1115–1128. doi: 10.1016/j.pain.2013.03.026. [DOI] [PubMed] [Google Scholar]
- 30.Gwak SJ, Macks C, Jeong DU, Kindy M, Lynn M, Webb K, Lee JS. RhoA knockdown by cationic amphiphilic copolymer/siRhoA polyplexes enhances axonal regeneration in rat spinal cord injury model. Biomaterials. 2017;121:155–166. doi: 10.1016/j.biomaterials.2017.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hakan T, Toklu HZ, Biber N, Celik H, Erzik C, Oğünç AV, Çetinel S, Sener G. Meloxicam exerts neuroprotection on spinal cord trauma in rats. Int J Neurosci. 2011;121:142–148. doi: 10.3109/00207454.2010.537415. [DOI] [PubMed] [Google Scholar]
- 32.Haws ME, Jaramillo TC, Espinosa F, Widman AJ, Stuber GD, Sparta DR, Tye KM, Russo SJ, Parada LF, Stavarache M, Kaplitt M, Bonci A, Powell CM. PTEN knockdown alters dendritic spine/protrusion morphology, not density. J Comp Neurol. 2014;522:1171–1190. doi: 10.1002/cne.23488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hellwig S, Heinrich A, Biber K. The brain's best friend: microglial neurotoxicity revisited. Front Cell Neurosci. 2013;7:71. doi: 10.3389/fncel.2013.00071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hermann A, Gastl R, Liebau S, Popa MO, Fiedler J, Boehm BO, Maisel M, Lerche H, Schwarz J, Brenner R, Storch A. Efficient generation of neural stem cell-like cells from adult human bone marrow stromal cells. J Cell Sci. 2004;117:4411–4422. doi: 10.1242/jcs.01307. [DOI] [PubMed] [Google Scholar]
- 35.Hirst WD, Young KA, Newton R, Allport VC, Marriott DR, Wilkin GP. Expression of COX-2 by normal and reactive astrocytes in the adult rat central nervous system. Mol Cell Neurosci. 1999;13:57–68. doi: 10.1006/mcne.1998.0731. [DOI] [PubMed] [Google Scholar]
- 36.Hou XL, Chen Y, Yin H, Duan WG. Combination of fasudil and celecoxib promotes the recovery of injured spinal cord in rats better than celecoxib or fasudil alone. Neural Regen Res. 2015;10:1836–1840. doi: 10.4103/1673-5374.170314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hou Y, Ryu CH, Jun JA, Kim SM, Jeong CH, Jeun SS. IL-8 enhances the angiogenic potential of human bone marrow mesenchymal stem cells by increasing vascular endothelial growth factor. Cell Biol Int. 2014;38:1050–1059. doi: 10.1002/cbin.10294. [DOI] [PubMed] [Google Scholar]
- 38.Huang JH, Yin XM, Xu Y, Xu CC, Lin X, Ye FB, Cao Y, Lin FY. Systemic administration of exosomes released from mesenchymal stromal cells attenuates apoptosis, inflammation, and promotes angiogenesis after spinal cord injury in rats. J Neurotrauma. 2017;34:3388–3396. doi: 10.1089/neu.2017.5063. [DOI] [PubMed] [Google Scholar]
- 39.Huang SY, Sung CS, Chen WF, Chen CH, Feng CW, Yang SN, Hung HC, Chen NF, Lin PR, Chen SC, Wang HM, Chu TH, Tai MH, Wen ZH. Involvement of phosphatase and tensin homolog deleted from chromosome 10 in rodent model of neuropathic pain. J Neuroinflammation. 2015;12:59. doi: 10.1186/s12974-015-0280-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jain A, McKeon RJ, Brady-Kalnay SM, Bellamkonda RV. Sustained delivery of activated Rho GTPases and BDNF promotes axon growth in CSPG-rich regions following spinal cord injury. PLoS One. 2011;6:e16135. doi: 10.1371/journal.pone.0016135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jasmin L, Janni G, Moallem TM, Lappi DA, Ohara PT. Schwann cells are removed from the spinal cord after effecting recovery from paraplegia. J Neurosci. 2000;20:9215–9223. doi: 10.1523/JNEUROSCI.20-24-09215.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Khan S, Mafi P, Mafi R, Khan W. A systematic review of mesenchymal stem cells in spinal cord injury, intervertebral disc repair and spinal fusion. Curr Stem Cell Res Ther. 2018;13:316–323. doi: 10.2174/1574888X11666170907120030. [DOI] [PubMed] [Google Scholar]
- 43.Kigerl KA, McGaughy VM, Popovich PG. Comparative analysis of lesion development and intraspinal inflammation in four strains of mice following spinal contusion injury. J Comp Neurol. 2006;494:578–594. doi: 10.1002/cne.20827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kim J, Hematti P. Mesenchymal stem cell-educated macrophages: a novel type of alternatively activated macrophages. Exp Hematol. 2009;37:1445–1453. doi: 10.1016/j.exphem.2009.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kim Y, Jo SH, Kim WH, Kweon OK. Antioxidant and anti-inflammatory effects of intravenously injected adipose derived mesenchymal stem cells in dogs with acute spinal cord injury. Stem Cell Res Ther. 2015;6:229. doi: 10.1186/s13287-015-0236-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Knafo S, Sánchez-Puelles C, Palomer E, Delgado I, Draffin JE, Mingo J, Wahle T, Kaleka K, Mou L, Pereda-Perez I, Klosi E, Faber EB, Chapman HM, Lozano-Montes L, Ortega-Molina A, Ordóñez-Gutiérrez L, Wandosell F, Viña J, Dotti CG, Hall RA, et al. PTEN recruitment controls synaptic and cognitive function in Alzheimer's models. Nat Neurosci. 2016;19:443. doi: 10.1038/nn.4225. [DOI] [PubMed] [Google Scholar]
- 47.Krupa P, Vackova I, Ruzicka J, Zaviskova K, Dubisova J, Koci Z, Turnovcova K, Urdzikova LM, Kubinova S, Rehak S, Jendelova P. The effect of human mesenchymal stem cells derived from Wharton's jelly in spinal cord injury treatment is dose-dependent and can be facilitated by repeated application. Int J Mol Sci. 2018:19. doi: 10.3390/ijms19051503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lai AY, Todd KG. Microglia in cerebral ischemia: molecular actions and interactions. Can J Physiol Pharmacol. 2006;84:49–59. doi: 10.1139/Y05-143. [DOI] [PubMed] [Google Scholar]
- 49.Laroni A, de Rosbo NK, Uccelli A. Mesenchymal stem cells for the treatment of neurological diseases: Immunoregulation beyond neuroprotection. Immunol Lett. 2015;168:183–190. doi: 10.1016/j.imlet.2015.08.007. [DOI] [PubMed] [Google Scholar]
- 50.Lewandowski G, Steward O. AAVshRNA-mediated suppression of PTEN in adult rats in combination with salmon fibrin administration enables regenerative growth of corticospinal axons and enhances recovery of voluntary motor function after cervical spinal cord injury. J Neurosci. 2014;34:9951–9962. doi: 10.1523/JNEUROSCI.1996-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Li C, Chen X, Qiao S, Liu X, Liu C, Zhu D, Su J, Wang Z. Effects of Wharton's jelly cells of the human umbilical cord on acute spinal cord injury in rats, and expression of interleukin-1beta and nerve growth factor in spinal cord tissues. Artificial cells, nanomedicine, and biotechnology. 2016;44:1254–1258. doi: 10.3109/21691401.2015.1019671. [DOI] [PubMed] [Google Scholar]
- 52.Li QM, Fu YM, Shan ZY, Shen JL, Zhang XM, Lei L, Jin LH. MSCs guide neurite directional extension and promote oligodendrogenesis in NSCs. Biochem Biophys Res Commun. 2009;384:372–377. doi: 10.1016/j.bbrc.2009.04.147. [DOI] [PubMed] [Google Scholar]
- 53.Li YH, Yu JW, Xi JY, Yu WB, Liu JC, Wang Q, Song LJ, Feng L, Yan YP, Zhang GX, Xiao BG, Ma CG. Fasudil enhances therapeutic efficacy of neural stem cells in the mouse model of MPTP-induced Parkinson's disease. Mol Neurobiol. 2017;54:5400–5413. doi: 10.1007/s12035-016-0027-8. [DOI] [PubMed] [Google Scholar]
- 54.Lian H, Yang L, Cole A, Sun L, Chiang AC, Fowler SW, Shim DJ, Rodriguez-Rivera J, Taglialatela G, Jankowsky JL, Lu HC, Zheng H. NFkappaB-activated astroglial release of complement C3 compromises neuronal morphology and function associated with Alzheimer's disease. Neuron. 2015;85:101–115. doi: 10.1016/j.neuron.2014.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Liddelow SA, Barres BA. Reactive astrocytes: production, function, and therapeutic potential. Immunity. 2017;46:957–967. doi: 10.1016/j.immuni.2017.06.006. [DOI] [PubMed] [Google Scholar]
- 56.Liddelow SA, Guttenplan KA, Clarke LE, Bennett FC, Bohlen CJ, Schirmer L, Bennett ML, Munch AE, Chung WS, Peterson TC, Wilton DK, Frouin A, Napier BA, Panicker N, Kumar M, Buckwalter MS, Rowitch DH, Dawson VL, Dawson TM, Stevens B, et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature. 2017;541:481–487. doi: 10.1038/nature21029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lin L, Lin H, Bai S, Zheng L, Zhang X. Bone marrow mesenchymal stem cells (BMSCs) improved functional recovery of spinal cord injury partly by promoting axonal regeneration. Neurochem Int. 2018;115:80–84. doi: 10.1016/j.neuint.2018.02.007. [DOI] [PubMed] [Google Scholar]
- 58.Lindsay SL, Johnstone SA, McGrath MA, Mallinson D, Barnett SC. Comparative miRNA-based fingerprinting reveals biological differences in human olfactory mucosa- and bone-marrow-derived mesenchymal stromal cells. Stem Cell Reports. 2016;6:729–742. doi: 10.1016/j.stemcr.2016.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Liu W, Wang Y, Gong F, Rong Y, Luo Y, Tang P, Zhou Z, Zhou Z, Xu T, Jiang T, Yang S, Yin G, Chen J, Fan J, Cai W. Exosomes derived from bone mesenchymal stem cells repair traumatic spinal cord injury by suppressing the activation of A1 neurotoxic reactive astrocytes. J Neurotrauma. 2018 doi: 10.1089/neu.2018.5835. doi: 10.1089/neu.2018.5835. [DOI] [PubMed] [Google Scholar]
- 60.Liu X, Duan B, Cheng Z, Jia X, Mao L, Fu H, Che Y, Ou L, Liu L, Kong D. SDF-1/CXCR4 axis modulates bone marrow mesenchymal stem cell apoptosis, migration and cytokine secretion. Protein Cell. 2011;2:845–854. doi: 10.1007/s13238-011-1097-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lord-Fontaine S, Yang F, Diep Q, Dergham P, Munzer S, Tremblay P, McKerracher L. Local inhibition of Rho signaling by cell-permeable recombinant protein BA-210 prevents secondary damage and promotes functional recovery following acute spinal cord injury. J Neurotrauma. 2008;25:1309–1322. doi: 10.1089/neu.2008.0613. [DOI] [PubMed] [Google Scholar]
- 62.Lu J, Wang QH, Huang LH, Dong HY, Lin LJ, Tan JM. Correlation of CDC42 activity with cell proliferation and palmitate-mediated cell death in human umbilical cord Wharton's jelly derived mesenchymal stromal cells. Stem Cells Dev. 2017;26:1283–1292. doi: 10.1089/scd.2017.0032. [DOI] [PubMed] [Google Scholar]
- 63.Lu XC, Zheng JY, Tang LJ, Huang BS, Li K, Tao Y, Yu W, Zhu RL, Li S, Li LX. MiR-133b Promotes neurite outgrowth by targeting RhoA expression. Cell Physiol Biochem. 2015;35:246–258. doi: 10.1159/000369692. [DOI] [PubMed] [Google Scholar]
- 64.Martinez FO, Gordon S. The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep. 2014;6:13. doi: 10.12703/P6-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Martinez FO, Gordon S, Locati M, Mantovani A. Transcriptional profiling of the human monocyte-to-macrophage differentiation and polarization: new molecules and patterns of gene expression. J Immunol. 2006;177:7303–7311. doi: 10.4049/jimmunol.177.10.7303. [DOI] [PubMed] [Google Scholar]
- 66.Martinez FO, Helming L, Milde R, Varin A, Melgert BN, Draijer C, Thomas B, Fabbri M, Crawshaw A, Ho LP, Ten Hacken NH, Cobos Jimenez V, Kootstra NA, Hamann J, Greaves DR, Locati M, Mantovani A, Gordon S. Genetic programs expressed in resting and IL-4 alternatively activated mouse and human macrophages: similarities and differences. Blood. 2013;121:e57–69. doi: 10.1182/blood-2012-06-436212. [DOI] [PubMed] [Google Scholar]
- 67.Maślińska D, Woźniak R, Kaliszek A, Modelska I. Expression of cyclooxygenase-2 in astrocytes of human brain after global ischemia. Folia Neuropathol. 1999;37:75–79. [PubMed] [Google Scholar]
- 68.Matsushita T, Lankford KL, Arroyo EJ, Sasaki M, Neyazi M, Radtke C, Kocsis JD. Diffuse and persistent blood-spinal cord barrier disruption after contusive spinal cord injury rapidly recovers following intravenous infusion of bone marrow mesenchymal stem cells. Exp Neurol. 2015;267:152–164. doi: 10.1016/j.expneurol.2015.03.001. [DOI] [PubMed] [Google Scholar]
- 69.McKerracher L, Anderson KD. Analysis of recruitment and outcomes in the phase I/IIa Cethrin clinical trial for acute spinal cord injury. J Neurotrauma. 2013;30:1795–1804. doi: 10.1089/neu.2013.2909. [DOI] [PubMed] [Google Scholar]
- 70.Mortazavi MM, Verma K, Harmon OA, Griessenauer CJ, Adeeb N, Theodore N, Tubbs RS. The microanatomy of spinal cord injury: a review. Clin Anat. 2015;28:27–36. doi: 10.1002/ca.22432. [DOI] [PubMed] [Google Scholar]
- 71.Mukhamedshina YO, Zakirova EY, Galieva LR, Kostennikov AA, Akhmetzyanova ER, Rizvanov AA. Distribution and survival of transplanted adipose-derived mesenchymal stem cells in the spinal cord injury. BioNanoScience. 2017a;7:608–612. [Google Scholar]
- 72.Mukhamedshina YO, Akhmetzyanova ER, Kostennikov AA, Zakirova EY, Galieva LR, Garanina EE, Rogozin AA, Kiassov AP, Rizvanov AA. Adipose-derived mesenchymal stem cell application combined with fibrin matrix promotes structural and functional recovery following spinal cord injury in rats. Front Pharmacol. 2018;9:343. doi: 10.3389/fphar.2018.00343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Mukhamedshina YO, Gilazieva ZE, Arkhipova SS, Galieva LR, Garanina EE, Shulman AA, Yafarova GG, Chelyshev YA, Shamsutdinova NV, Rizvanov AA. Electrophysiological, morphological, and ultrastructural features of the injured spinal cord tissue after transplantation of human umbilical cord blood mononuclear cells genetically modified with the VEGF and GDNF genes. Neural Plast 2017. 2017b:9857918. doi: 10.1155/2017/9857918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Németh K, Leelahavanichkul A, Yuen PS, Mayer B, Parmelee A, Doi K, Robey PG, Leelahavanichkul K, Koller BH, Brown JM, Hu X, Jelinek I, Star RA, Mezey E. Bone marrow stromal cells attenuate sepsis via prostaglandin E(2)-dependent reprogramming of host macrophages to increase their interleukin-10 production. Nat Med. 2009;15:42–49. doi: 10.1038/nm.1905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Nakajima H, Uchida K, Guerrero AR, Watanabe S, Sugita D, Takeura N, Yoshida A, Long G, Wright KT, Johnson WE, Baba H. Transplantation of mesenchymal stem cells promotes an alternative pathway of macrophage activation and functional recovery after spinal cord injury. J Neurotrauma. 2012;29:1614–1625. doi: 10.1089/neu.2011.2109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Nakano N, Nakai Y, Seo TB, Homma T, Yamada Y, Ohta M, Suzuki Y, Nakatani T, Fukushima M, Hayashibe M, Ide C. Effects of bone marrow stromal cell transplantation through CSF on the subacute and chronic spinal cord injury in rats. PLoS One. 2013;8:e73494. doi: 10.1371/journal.pone.0073494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Neirinckx V, Agirman G, Coste C, Marquet A, Dion V, Rogister B, Franzen R, Wislet S. Adult bone marrow mesenchymal and neural crest stem cells are chemoattractive and accelerate motor recovery in a mouse model of spinal cord injury. Stem Cell Res Ther. 2015;6:211. doi: 10.1186/s13287-015-0202-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Oh JY, Ko JH, Lee HJ, Yu JM, Choi H, Kim MK, Wee WR, Prockop DJ. Mesenchymal stem/stromal cells inhibit the NLRP3 inflammasome by decreasing mitochondrial reactive oxygen species. Stem Cells. 2014;32:1553–1563. doi: 10.1002/stem.1608. [DOI] [PubMed] [Google Scholar]
- 79.Papa S, Vismara I, Mariani A, Barilani M, Rimondo S, De Paola M, Panini N, Erba E, Mauri E, Rossi F, Forloni G, Lazzari L, Veglianese P. Mesenchymal stem cells encapsulated into biomimetic hydrogel scaffold gradually release CCL2 chemokine in situ preserving cytoarchitecture and promoting functional recovery in spinal cord injury. J Control Release. 2018;278:49–56. doi: 10.1016/j.jconrel.2018.03.034. [DOI] [PubMed] [Google Scholar]
- 80.Park K. Functional recovery in spinal cord injury using mesenchymal stem cells. J Control Release. 2018;278:159. doi: 10.1016/j.jconrel.2018.04.044. [DOI] [PubMed] [Google Scholar]
- 81.Peress NS, Fleit HB, Perillo E, Kuljis R, Pezzullo C. Identification of Fc gamma RI, II and III on normal human brain ramified microglia and on microglia in senile plaques in Alzheimer's disease. J Neuroimmunol. 1993;48:71–79. doi: 10.1016/0165-5728(93)90060-c. [DOI] [PubMed] [Google Scholar]
- 82.Persson M, Brantefjord M, Hansson E, Rönnbäck L. Lipopolysaccharide increases microglial GLT-1 expression and glutamate uptake capacity in vitro by a mechanism dependent on TNF-alpha. Glia. 2005;51:111–120. doi: 10.1002/glia.20191. [DOI] [PubMed] [Google Scholar]
- 83.Pietilä M, Lehtonen S, Tuovinen E, Lähteenmäki K, Laitinen S, Leskelä HV, Nätynki A, Pesälä J, Nordström K, Lehenkari P. CD200 Positive human mesenchymal stem cells suppress TNF-alpha secretion from CD200 receptor positive macrophage-like cells. PLoS One. 2012;7:e31671. doi: 10.1371/journal.pone.0031671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Pisati F, Bossolasco P, Meregalli M, Cova L, Belicchi M, Gavina M, Marchesi C, Calzarossa C, Soligo D, Lambertenghi-Deliliers G, Bresolin N, Silani V, Torrente Y, Polli E. Induction of neurotrophin expression via human adult mesenchymal stem cells: implication for cell therapy in neurodegenerative diseases. Cell Transplant. 2007;16:41–55. doi: 10.3727/000000007783464443. [DOI] [PubMed] [Google Scholar]
- 85.Pontius JU, Wagner L, Schuler GD. The NCBI Handbook. Bethesda, MD, USA: National Library of Medicine; 2003. UniGene: A unified view of the transcriptome. [Google Scholar]
- 86.Priestley JV, Michael-Titus AT, Tetzlaff W. Limiting spinal cord injury by pharmacological intervention. Handb Clin Neurol. 2012;109:463–484. doi: 10.1016/B978-0-444-52137-8.00029-2. [DOI] [PubMed] [Google Scholar]
- 87.Prockop DJ, Oh JY. Mesenchymal stem/stromal cells (MSCs): role as guardians of inflammation. Mol Ther. 2012;20:14–20. doi: 10.1038/mt.2011.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Qi Y, Jiang D, Sindrilaru A, Stegemann A, Schatz S, Treiber N, Rojewski M, Schrezenmeier H, Vander Beken S, Wlaschek M, Bohm M, Seitz A, Scholz N, Durselen L, Brinckmann J, Ignatius A, Scharffetter-Kochanek K. TSG-6 released from intradermally injected mesenchymal stem cells accelerates wound healing and reduces tissue fibrosis in murine full-thickness skin wounds. J Invest Dermatol. 2014;134:526–537. doi: 10.1038/jid.2013.328. [DOI] [PubMed] [Google Scholar]
- 89.Qu J, Zhang H. Roles of mesenchymal stem cells in spinal cord injury. Stem Cells Int 2017. 2017:5251313. doi: 10.1155/2017/5251313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Quadri SA, Farooqui M, Ikram A, Zafar A, Khan MA, Suriya SS, Claus CF, Fiani B, Rahman M, Ramachandran A, Armstrong IIT, Taqi MA, Mortazavi MM. Recent update on basic mechanisms of spinal cord injury. Neurosurg Rev. 2018 doi: 10.1007/s10143-018-1008-3. doi: 10.1007/s10143-018-y1008.3. [DOI] [PubMed] [Google Scholar]
- 91.Ra JC, Shin IS, Kim SH, Kang SK, Kang BC, Lee HY, Kim YJ, Jo JY, Yoon EJ, Choi HJ, Kwon E. Safety of intravenous infusion of human adipose tissue-derived mesenchymal stem cells in animals and humans. Stem Cells Dev. 2011;20:1297–1308. doi: 10.1089/scd.2010.0466. [DOI] [PubMed] [Google Scholar]
- 92.Ramalho BS, Almeida FM, Sales CM, de Lima S, Martinez AMB. Injection of bone marrow mesenchymal stem cells by intravenous or intraperitoneal routes is a viable alternative to spinal cord injury treatment in mice. Neural Regen Res. 2018;13:1046–1053. doi: 10.4103/1673-5374.233448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Rengasamy M, Gupta PK, Kolkundkar U, Singh G, Balasubramanian S, SundarRaj S, Chullikana A, Majumdar AS. Preclinical safety &toxicity evaluation of pooled, allogeneic human bone marrow-derived mesenchymal stromal cells. Indian J Med Res. 2016;144:852–864. doi: 10.4103/ijmr.IJMR_1842_15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Reyes M, Verfaillie CM. Turning marrow into brain, generation of glial and neuronal cells from adult bone marrow mesenchymal stem cells. Blood. 1999;94:377a. [Google Scholar]
- 95.Ribeiro TB, Duarte AS, Longhini AL, Pradella F, Farias AS, Luzo AC, Oliveira AL, Olalla Saad ST. Neuroprotection and immunomodulation by xenografted human mesenchymal stem cells following spinal cord ventral root avulsion. Sci Rep. 2015;5:16167. doi: 10.1038/srep16167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Rivera FJ, Couillard-Despres S, Pedre X, Ploetz S, Caioni M, Lois C, Bogdahn U, Aigner L. Mesenchymal stem cells instruct oligodendrogenic fate decision on adult neural stem cells. Stem Cells. 2006;24:2209–2219. doi: 10.1634/stemcells.2005-0614. [DOI] [PubMed] [Google Scholar]
- 97.Ruppert KA, Nguyen TT, Prabhakara KS, Toledano Furman NE, Srivastava AK, Harting MT, Cox CS, Jr, Olson SD. Human mesenchymal stromal cell-derived extracellular vesicles modify microglial response and improve clinical outcomes in experimental spinal cord injury. Sci Rep. 2018;8:480. doi: 10.1038/s41598-017-18867-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ryu HH, Kang BJ, Park SS, Kim Y, Sung GJ, Woo HM, Kim WH, Kweon OK. Comparison of mesenchymal stem cells derived from fat, bone marrow, Wharton's jelly, and umbilical cord blood for treating spinal cord injuries in dogs. J Vet Med Sci. 2012;74:1617–1630. doi: 10.1292/jvms.12-0065. [DOI] [PubMed] [Google Scholar]
- 99.Sabapathy V, Tharion G, Kumar S. Cell therapy augments functional recovery subsequent to spinal cord injury under experimental conditions. Stem Cells Int 2015. 2015:132172. doi: 10.1155/2015/132172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Sabino L, Maria CA, Luca L, Valerio V, Edda F, Giacomo R, Gloria I, Juan GR, Antonio C. Engraftment, neuroglial transdifferentiation and behavioral recovery after complete spinal cord transection in rats. Surg Neurol Int. 2018;9:19. doi: 10.4103/sni.sni_369_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Sakamoto K, Kadomatsu K. Mechanisms of axon regeneration: The significance of proteoglycans. Biochim Biophys Acta Gen Subj. 2017;1861:2435–2441. doi: 10.1016/j.bbagen.2017.06.005. [DOI] [PubMed] [Google Scholar]
- 102.Schaper F, Rose-John S. Interleukin-6: Biology, signaling and strategies of blockade. Cytokine Growth Factor Rev. 2015;26:475–487. doi: 10.1016/j.cytogfr.2015.07.004. [DOI] [PubMed] [Google Scholar]
- 103.Scheller J, Garbers C, Rose-John S. Interleukin-6: from basic biology to selective blockade of pro-inflammatory activities. Semin Immunol. 2014;26:2–12. doi: 10.1016/j.smim.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 104.Shaymardanova GF, Mukhamedshina YO, Salafutdinov II, Rizvanov AA, Chelyshev YA. Usage of plasmid vector carrying vegf and fgf2 genes after spinal cord injury in rats. Bull Exp Biol Med. 2013;154:544–547. doi: 10.1007/s10517-013-1996-5. [DOI] [PubMed] [Google Scholar]
- 105.Shin TH, Kim HS, Kang TW, Lee BC, Lee HY, Kim YJ, Shin JH, Seo Y, Won Choi S, Lee S, Shin K, Seo KW, Kang KS. Human umbilical cord blood-stem cells direct macrophage polarization and block inflammasome activation to alleviate rheumatoid arthritis. Cell Death Dis. 2016;7:e2524. doi: 10.1038/cddis.2016.442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Silva WA, Jr, Covas DT, Panepucci RA, Proto-Siqueira R, Siufi JL, Zanette DL, Santos AR, Zago MA. The profile of gene expression of human marrow mesenchymal stem cells. Stem Cells. 2003;21:661–669. doi: 10.1634/stemcells.21-6-661. [DOI] [PubMed] [Google Scholar]
- 107.Sondell M, Sundler F, Kanje M. Vascular endothelial growth factor is a neurotrophic factor which stimulates axonal outgrowth through the flk-1 receptor. Eur J Neurosci. 2000;12:4243–4254. doi: 10.1046/j.0953-816x.2000.01326.x. [DOI] [PubMed] [Google Scholar]
- 108.Song WJ, Li Q, Ryu MO, Ahn JO, Ha Bhang D, Chan Jung Y, Youn HY. TSG-6 secreted by human adipose tissue-derived mesenchymal stem cells ameliorates dss-induced colitis by inducing m2 macrophage polarization in mice. Sci Rep. 2017;7:5187. doi: 10.1038/s41598-017-04766-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Steffenhagen C, Dechant FX, Oberbauer E, Furtner T, Weidner N, Kury P, Aigner L, Rivera FJ. Mesenchymal stem cells prime proliferating adult neural progenitors toward an oligodendrocyte fate. Stem Cells Dev. 2012;21:1838–1851. doi: 10.1089/scd.2011.0137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sun G, Li G, Li D, Huang W, Zhang R, Zhang H, Duan Y, Wang B. hucMSC derived exosomes promote functional recovery in spinal cord injury mice via attenuating inflammation. Mater Sci Eng C Mater Biol Appl. 2018;89:194–204. doi: 10.1016/j.msec.2018.04.006. [DOI] [PubMed] [Google Scholar]
- 111.Takahashi A, Nakajima H, Uchida K, Takeura N, Honjoh K, Watanabe S, Kitade M, Kokubo Y, Johnson WEB, Matsumine A. Comparison of mesenchymal stromal cells isolated from murine adipose tissue and bone marrow in the treatment of spinal cord injury. Cell Transplant. 2018a;27:1126–1139. doi: 10.1177/0963689718780309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Takahashi S, Nakagawa K, Tomiyasu M, Nakashima A, Katayama K, Imura T, Herlambang B, Okubo T, Arihiro K, Kawahara Y, Yuge L, Sueda T. Mesenchymal stem cell-based therapy improves lower limb movement after spinal cord ischemia in rats. Ann Thorac Surg. 2018b;105:1523–1530. doi: 10.1016/j.athoracsur.2017.12.014. [DOI] [PubMed] [Google Scholar]
- 113.Totoiu MO, Keirstead HS. Spinal cord injury is accompanied by chronic progressive demyelination. J Comp Neurol. 2005;486:373–383. doi: 10.1002/cne.20517. [DOI] [PubMed] [Google Scholar]
- 114.Urdzíková LM, Růžička J, LaBagnara M, Kárová K, Kubinová Š, Jiráková K, Murali R, Syková E, Jhanwar-Uniyal M, Jendelová P. Human mesenchymal stem cells modulate inflammatory cytokines after spinal cord injury in rat. Int J Mol Sci. 2014;15:11275–11293. doi: 10.3390/ijms150711275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Vellosillo L, Munoz MP, Paino CL. Adipose tissue-derived stromal cells (ADSC) express oligodendrocyte and myelin markers, but they do not function as oligodendrocytes. Histochem Cell Biol. 2017;148:503–515. doi: 10.1007/s00418-017-1588-y. [DOI] [PubMed] [Google Scholar]
- 116.Vladimirskaya EB. Mesenchymal stem cells in cellular therapy. Oncohematology. 2007;1:416. [Google Scholar]
- 117.Walker DG, Lue LF. Immune phenotypes of microglia in human neurodegenerative disease: challenges to detecting microglial polarization in human brains. Alzheimers Res Ther. 2015;7:56. doi: 10.1186/s13195-015-0139-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Wang J, Ding F, Gu Y, Liu J, Gu X. Bone marrow mesenchymal stem cells promote cell proliferation and neurotrophic function of Schwann cells in vitro and in vivo. Brain Res. 2009;1262:7–15. doi: 10.1016/j.brainres.2009.01.056. [DOI] [PubMed] [Google Scholar]
- 119.Wang N, Li Q, Zhang L, Lin H, Hu J, Li D, Shi S, Cui S, Zhou J, Ji J, Wan J, Cai G, Chen X. Mesenchymal stem cells attenuate peritoneal injury through secretion of TSG-6. PLoS One. 2012;7:e43768. doi: 10.1371/journal.pone.0043768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ward RE, Huang W, Kostusiak M, Pallier PN, Michael-Titus AT, Priestley JV. A characterization of white matter pathology following spinal cord compression injury in the rat. Neuroscience. 2014;260:227–239. doi: 10.1016/j.neuroscience.2013.12.024. [DOI] [PubMed] [Google Scholar]
- 121.Wen X, Wang L, Liu Z, Liu Y, Hu J. Intracranial injection of PEG-PEI/ROCK II-siRNA improves cognitive impairment in a mouse model of Alzheimer's disease. Int J Neurosci. 2014;124:697–703. doi: 10.3109/00207454.2013.877014. [DOI] [PubMed] [Google Scholar]
- 122.Wilcock DM. Neuroinflammatory phenotypes and their roles in Alzheimer's disease. Neurodegener Dis. 2014;13:183–185. doi: 10.1159/000354228. [DOI] [PubMed] [Google Scholar]
- 123.Willenberg R, Zukor K, Liu K, He Z, Steward O. Variable laterality of corticospinal tract axons that regenerate after spinal cord injury as a result of PTEN deletion or knock-down. J Comp Neurol. 2016;524:2654–2676. doi: 10.1002/cne.23987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Witiw CD, Fehlings MG. Acute spinal cord injury. J Spinal Disord Tech. 2015;28:202–210. doi: 10.1097/BSD.0000000000000287. [DOI] [PubMed] [Google Scholar]
- 125.Wright KT, El Masri W, Osman A, Chowdhury J, Johnson WE. Concise review: Bone marrow for the treatment of spinal cord injury: mechanisms and clinical applications. Stem Cells. 2011;29:169–178. doi: 10.1002/stem.570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Wright KT, El Masri W, Osman A, Roberts S, Chamberlain G, Ashton BA, Johnson WE. Bone marrow stromal cells stimulate neurite outgrowth over neural proteoglycans (CSPG), myelin associated glycoprotein and Nogo-A. Biochem Biophys Res Commun. 2007;354:559–566. doi: 10.1016/j.bbrc.2007.01.013. [DOI] [PubMed] [Google Scholar]
- 127.Wu X, Xu XM. RhoA/Rho kinase in spinal cord injury. Neural Regen Res. 2016;11:23–27. doi: 10.4103/1673-5374.169601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Xiong LL, Liu F, Lu BT, Zhao WL, Dong XJ, Liu J, Zhang RP, Zhang P, Wang TH. Bone marrow mesenchymal stem-cell transplantation promotes functional improvement associated with CNTF-STAT3 activation after hemi-sectioned spinal cord injury in tree shrews. Front Cell Neurosci. 2017;11:172. doi: 10.3389/fncel.2017.00172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Yang C, Wang G, Ma F, Yu B, Chen F, Yang J, Feng J, Wang Q. Repeated injections of human umbilical cord blood-derived mesenchymal stem cells significantly promotes functional recovery in rabbits with spinal cord injury of two noncontinuous segments. Stem Cell Res Ther. 2018;9:136. doi: 10.1186/s13287-018-0879-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Yu JW, Li YH, Song GB, Yu JZ, Liu CY, Liu JC, Zhang HF, Yang WF, Wang Q, Yan YP, Xiao BG, Ma CG. Synergistic and superimposed effect of bone marrow-derived mesenchymal stem cells combined with fasudil in experimental autoimmune encephalomyelitis. J Mol Neurosci. 2016;60:486–497. doi: 10.1007/s12031-016-0819-3. [DOI] [PubMed] [Google Scholar]
- 131.Yuan YM, He C. The glial scar in spinal cord injury and repair. Neurosci Bull. 2013;29:421–435. doi: 10.1007/s12264-013-1358-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Yuksel U, Bakar B, Dincel GC, Budak Yildiran FA, Ogden M, Kisa U. The investigation of the Cox-2 selective inhibitor parecoxib effects in spinal cord injury in rat. J Invest Surg. 2018:1–12. doi: 10.1080/08941939.2017.1423423. [DOI] [PubMed] [Google Scholar]
- 133.Zeng X, Ma YH, Chen YF, Qiu XC, Wu JL, Ling EA, Zeng YS. Autocrine fibronectin from differentiating mesenchymal stem cells induces the neurite elongation in vitro and promotes nerve fiber regeneration in transected spinal cord injury. J Biomed Mater Res A. 2016;104:1902–1911. doi: 10.1002/jbm.a.35720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Zeng X, Zeng YS, Ma YH, Lu LY, Du BL, Zhang W, Li Y, Chan WY. Bone marrow mesenchymal stem cells in a three-dimensional gelatin sponge scaffold attenuate inflammation, promote angiogenesis, and reduce cavity formation in experimental spinal cord injury. Cell Transplant. 2011;20:1881–1899. doi: 10.3727/096368911X566181. [DOI] [PubMed] [Google Scholar]
- 135.Zhang QZ, Su WR, Shi SH, Wilder-Smith P, Xiang AP, Wong A, Nguyen AL, Kwon CW, Le AD. Human gingiva-derived mesenchymal stem cells elicit polarization of m2 macrophages and enhance cutaneous wound healing. Stem Cells. 2010;28:1856–1868. doi: 10.1002/stem.503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Zhang X, Schwarz EM, Young DA, Puzas JE, Rosier RN, O’Keefe RJ. Cyclooxygenase-2 regulates mesenchymal cell differentiation into the osteoblast lineage and is critically involved in bone repair. J Clin Invest. 2002;109:1405–1415. doi: 10.1172/JCI15681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Zhou HL, Zhang XJ, Zhang MY, Yan ZJ, Xu ZM, Xu RX. Transplantation of human amniotic mesenchymal stem cells promotes functional recovery in a rat model of traumatic spinal cord injury. Neurochem Res. 2016;41:2708–2718. doi: 10.1007/s11064-016-1987-9. [DOI] [PubMed] [Google Scholar]
- 138.Zukor K, Belin S, Wang C, Keelan N, Wang X, He Z. Short hairpin RNA against PTEN enhances regenerative growth of corticospinal tract axons after spinal cord injury. J Neurosci. 2013;33:15350–15361. doi: 10.1523/JNEUROSCI.2510-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Zurita M, Vaquero J. Bone marrow stromal cells can achieve cure of chronic paraplegic rats: functional and morphological outcome one year after transplantation. Neurosci Lett. 2006;402:51–56. doi: 10.1016/j.neulet.2006.03.069. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Behavior of MSCs in the area of SCI based on preclinical trials data
Preclinical in vivo trials using MSCs transplantation after SCI and obtained results in the interpretation of the authors*


