Abstract
Hypothyroidism is among the most frequent chronic diseases in the elderly, and levothyroxine (l-T4) is worldwide within the 10 drugs more prescribed in the general population. Hypothyroidism is defined by increased serum thyroid-stimulating hormone (TSH) values and reduced circulating free thyroid hormones, whereas subclinical hypothyroidism (sHT) is characterized by free hormone fractions within the normal ranges and has been divided into two classes, depending on circulating TSH levels (above or below 10 mIU/L). Given that during aging, a natural trend toward higher values of circulating TSH has been reported, it is necessary to verify carefully the diagnosis of sHT to tailor an appropriate follow-up and ad hoc therapy, avoiding unnecessary or excessive treatment. In the current review, we evaluate the state of the art on hypothyroidism in the elderly with special focus on the effect of sHT on cognition and the cardiovascular system function. We also summarize the recommendations for a correct diagnostic workup and therapeutic approach to older people with an elevated TSH value, with special attention to the presence of frailty, comorbidities, and poly therapy. In conclusion, personalized therapy is crucial in good clinical practice, and in the management of older patients with sHT, multiple factors must be considered, including age-dependent TSH cutoffs, thyroid autoimmunity, the burden of comorbidities, and the possible presence of frailty. l-T4 is the drug of choice for the treatment of hypothyroid older people, but the risk of overtreatment, potential adverse drug reactions, and patient compliance should always be considered and thyroid status periodically reassessed.
Keywords: hypothyroidism, subclinical hypothyroidism, elderly, therapy, frailty, l-T4
The demographic growth in Western countries in the last decades has led to more people >65 years of age. It is estimated that in Italy (the second-oldest world population after Japan), subjects aged >65 years represent 22%, and >80 years are 6.7% of the overall population, with numbers expected to double in 25 years [1]. Thyroid diseases are frequent among the several chronic illnesses observed in the elderly, and in this review, we analyze the impact of hypothyroidism and its management in older people, especially the oldest old (>80 years).
Hypothyroidism is defined by the increase in thyroid-stimulating hormone (TSH) values, accompanied by reduced circulating free triiodothyronine (FT3) and free thyroxine (FT4). Such a condition is associated with a global mortality increase and an increased incidence of cardiovascular events; therefore, replacement therapy with levothyroxine (l-T4) is advisable and necessary [2]. However, sometimes, thyroid failure is of a mild degree, and caution needs to be taken in planning the diagnostic and therapeutic approach, particularly in the oldest old patient [3]. Subclinical hypothyroidism (sHT) is characterized by increased TSH values in the face of circulating FT3 and FT4 values within the normal range [3]. According to the results of large trials and meta-analyses available in scientific literature [4–8], the 2013 European Thyroid Association guideline split sHT into two distinct categories, based on cutoffs of circulating TSH values, respectively, between 4 and 10 mIU/L or >10 mIU/L [9].
The prevalence of overt hypothyroidism in the general population varies between 0.2% and 5.3% in Europe and between 0.3% and 3.7% in the United States, possibly in relation to different iodine intake [10]. According to the data from the National Health and Nutrition Examination Survey (NHANES III), the overall prevalence of hypothyroidism is 4.6%, with 0.3% for the overt and 4.3% for the subclinical type. The NHANES III study confirms hypothyroidism as the most frequent thyroid disease among the older population, with a greater prevalence in women [11]. Longitudinal studies conducted in the United Kingdom show an incidence of hypothyroidism ∼3.5% to 5% [12]. sHT has a variable incidence, depending on different cohorts [13]: 7.5% in the Wickham study [12]; ∼21% in women; and 16% in men in the Colorado study [14]. The NHANES III data demonstrate that circulating TSH levels increase with aging, as well as antithyroid autoantibodies; the percentage of subjects with TSH levels >4.5 mIU/L rises to 14% in the population aged 85 and above, confirming the higher prevalence in white women [11]. It is worth noting that in a British cohort of 6000 subjects aged >65 years, the prevalence of overt hypothyroidism was ∼2%, but the prevalence of sHT was lower than that generally reported in literature, at ∼2.9% [15]. Data from the same group have previously shown a prevalence of sHT of 11.6% in women and 2.9% in men >60 years in the same geographic area [16]. The authors explain the gap in the data, only 10 years apart, with a possible better screening campaign and wider information, as well as with an earlier treatment of sHT [17]. Indeed, the large prescription of l-T4 therapy in Western countries may reduce the crude prevalence of sHT in epidemiological surveys, evaluating only serum TSH levels of the examined population. In this setting, data from the Medicines Utilization Monitoring Centre have demonstrated that l-T4 is within the 10 drugs more prescribed among the general population in Italy, in line with worldwide projections [18].
To differentiate sHT from the age-related physiological modifications of the hypothalamus-pituitary-thyroid axis, it is crucial to consider the most frequent pathogenetic mechanisms of thyroid failure in older people. Although hypothyroidism, secondary to surgical and medical procedures (thyroidectomy or radioiodine treatment) with suboptimal l-T4 replacement therapy, should be not overlooked, chronic autoimmune thyroiditis (Hashimoto thyroiditis) is the most frequent cause of (subclinical) hypothyroidism in the elderly [19, 20]. Hashimoto thyroiditis is characterized in almost 90% of the cases by the presence of antithyroglobulin (TgAb) and antithyroid peroxidase (TPOAb) autoantibodies. These antibodies are not cytotoxic themselves; therefore, the thyroid cellular damage is supposed to be driven by T-lymphocytes (CD8+) [21]. The dosage of TPOAb is the most sensitive test to determine the presence of antithyroid autoimmunity and gives information about the progression toward overt hypothyroidism, which shows a higher incidence in patients who are TPOAb positive (4.3% per year) compared with the negative ones (2.6% per year) [21, 22]. However, when the presence of serum-positive TPOAb titers has been demonstrated, repeated measurements do not add much useful information in the monitoring of individual patients, as levels of TPOAb vary in parallel with TSH [23]. In the NHANES III study, 13,000 healthy subjects have been followed up and monitored by consecutive dosage of circulating FT3, FT4, TSH, TgAb, and TPOAb; among them, 10% resulted in TgAb and 11% in TPOAb positive [11]. However, in almost 20% of sHT subjects, the dosage of both TPOAb and TgAb may be negative; in these cases, thyroid ultrasound examination, showing the typical tissue inhomogeneity and hypoechogenicity, is supportive for the diagnosis [24]. Another important cause of hypothyroidism in the elderly is iatrogenic. Indeed, several classes of drugs contribute to reduce thyroid function, such as the following: thyrostatic medications (methimazole, perchlorate, and propylthiouracil), tyrosin-kinase inhibitors, β-blockers, interferon-α, IL-2, lithium, and ethionamide. Usually, iatrogenic damage is transient and reversible when the drug is withdrawn. In these cases, periodic monitoring of thyroid function is recommended, at least twice per year. Drugs interfering with l-T4 absorption are also a frequent cause of hypothyroidism, despite proper replacement therapy, especially in the elderly [19].
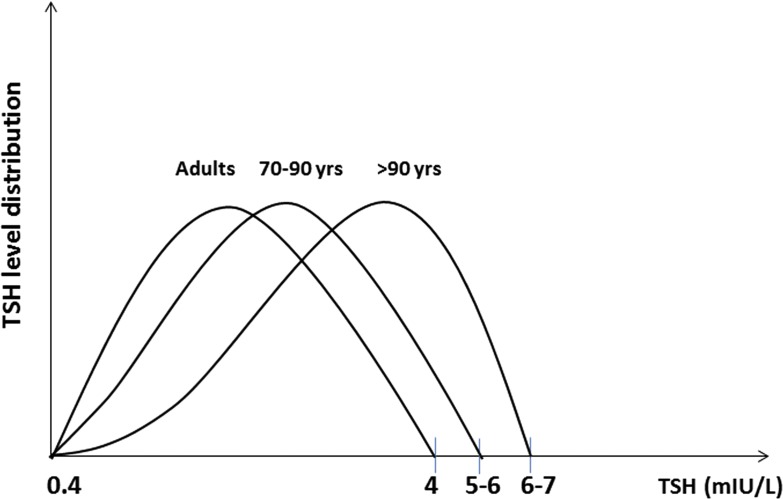
1. The Hypothalamus/Pituitary/Thyroid Axis in the Elderly
Circulating TSH levels in healthy subjects vary according to the circadian rhythm and respond with logarithmically amplified variation to minor changes in serum FT4 and FT3 values. Thus, abnormal serum TSH may indicate that serum FT4 and FT3 are not normal for an individual [25]. In light of that, serum TSH is a sensitive marker of thyroid function, with increased values indicating a reduced function and vice versa for lower TSH levels [20]. However, complex modifications of the hypothalamus/pituitary/thyroid axis and of the peripheral thyroid hormone metabolism have been described in euthyroid older people [26–28]. With the exclusion of pathologic conditions, the aging process is characterized by a reduced iodine absorption and organification and a lower response to TSH, leading to reduced thyroid hormone production [29–31]. Moreover, in the oldest olds, the nocturnal surge of TSH is partially or completely lost, and the inhibitory effect of corticosteroids is attenuated [25, 28, 32]. However, there are conflicting results regarding the age-dependent TSH variation: few previous studies (generally case control) have shown a tendency toward lower TSH circulating levels in individuals >75 to 80 years of age and in centenarians [26, 33]. On the other hand, further population studies have demonstrated an opposite tendency, with circulating TSH levels rising with the aging process: in the population >80 years of age, the upper limit at the 95% interval of confidence was >6.0 mIU/L, reaching 8.0 mIU/L in over-90s [34–36] (Fig. 1). Initially, the reduced TSH levels in centenarians were interpreted as a centrally mediated way to avoid excessive catabolism and promote “physiological aging.” However, it is important to distinguish this condition from the “low triiodothyronine (T3) syndrome” or “euthyroid sick syndrome” (not rare in the elderly as a result of the high prevalence of comorbidities), where circulating levels of TSH and T3 are reduced, whereas reverse T3 is increased as a response to acute and systemic diseases. Indeed, the low T3 syndrome is associated with reduced physical function and worse prognosis, both quoad vitam and quoad valetudinem [28, 37]. It is widely accepted that some of the aging effects are similar to those seen in the case of mild thyroid failure and are associated with reduction of the basal metabolism [38]. However, it is still a matter of debate whether the reduced thyroid function in the elderly (as a result of either reduced pituitary gland secretion or reduced hormone production) is just a consequence of a reduced metabolic request or instead represents a real protective condition against the increased catabolism seen in the aging process [38]. On that matter, a study conducted in North Europe has documented the potential role of genetics in longevity, which correlated with thyroid function. In particular, the offspring of 90-year-old subjects showed reduced FT3 circulating values and a better metabolic profile compared with their partners with less long-lived parents [31]. Likewise, a cohort study, carried out in a population of Southern Italy, has documented reduced levels of TSH and FT3 in centenarians’ grandchildren compared with an age-matched control group [39]. Moreover, several huge cohort studies have demonstrated that the distribution of serum TSH levels shows a tendency to increase in relation to the age and that the extreme longevity is associated with a further increase of TSH levels, at least in the Ashkenazy population [40, 41]. Nonetheless, a recent report from the Rotterdam study, carried out on >9000 healthy home-dwelling subjects, does not confirm this trend, showing instead a progressive reduction of mean serum TSH and a concomitant rising of TPOAb values with increasing age [42]. To understand better those apparent contradictions, it is worth noticing that with the aging process, some complex changes in the thyroid functional status may occur, which cannot always be spotted just from a TSH dosage, i.e., changing in the TSH bioactivity, in the thyrocyte sensitivity to TSH, in thyroid hormone metabolism, as well as in the receptors and cofactors modulating the response to T3 input. Moreover, compared with young adults, the elderly presents a reduced response to TRH, as well as a reduced TSH circadian variation and an attenuated steroidal-mediated inhibitory effect [27, 43]. Finally, with the aging process, a reduced cellular ability to capture iodine and secrete thyroxine (T4) and a reduced hormonal clearance rate have been described [26]. Overall, these data show how the aging process is characterized by a downregulation of the hypothalamus-pituitary-thyroid-peripheral-tissues axis, although it is yet to be established whether these changes reflect an adaptive mechanism to reduced metabolic functions or instead, represent a protective mechanism to preserve the body from excessive catabolism. Finally, the circulating TSH variability, besides being a consequence of the aging process, could be the expression of actual thyroid pathology, especially in the case of positive TPOAb and TgAb titers. With the knowledge of the extreme variability of thyroid hormone physiopathology in the elderly, it is crucial to avoid a superficial diagnosis and therefore, inappropriate treatment [44]. In light of that, the clinical relevance of sHT is still under debate, especially in the case of a slight serum TSH increase (<10 mIU/L) that is particularly relevant in the therapeutic management of the older population [44].
Figure 1.
Approximate changes of serum TSH values with aging.
2. sHT and the Cardiovascular System
Thyroid hormones exert an important metabolic function across adulthood and in the aging phases [45]. Thyroid hormones are involved in the regulation of the oxygen consumption and heat production; moreover, they facilitate cellular glucose uptake, enhancing glycogenolysis and glycogenosis with a specific contrainsular activity [45]. The cardiovascular system is partly influenced by thyroid function: thyroid hormones are necessary for a correct protein synthesis, modulate the adrenergic system activity, and regulate the vascular peripheral resistance [20]. However, the effects and the clinical significance of sHT on the cardiovascular system are still under debate; they are usually recognized in young adults, whereas more conflicting results have been obtained in the elderly [4, 13, 46, 47]. Unfortunately, no randomized clinical trials evaluating the effect of l-T4 therapy on cardiovascular outcome in old and very old patients with sHT are available yet, leaving the decision about such a treatment without a clear and widely approved consensus [4, 48]. Overt hypothyroidism determines changes in myocardial contractility, vascular resistance, and endothelial function, which are usually reversible with adequate treatment [4]. sHT exerts similar but milder effects; thus, a potential role of such a condition in the trajectory of cardiovascular diseases could be not excluded a priori [4]. In a recent large study, the possible relationship between sHT in the elderly and the metabolic syndrome, either in prevalence or incidence, has been evaluated longitudinally [5]. Over 2100 patients have been included in the study; among them, 687 had metabolic syndrome at baseline. For over 6 years of follow-up, 239 further subjects developed metabolic syndrome; the presence of serum TSH levels >10 mIU/L was associated with higher odds of prevalent metabolic syndrome [5]. The relationship between mild thyroid disorder and heart failure (HF) has been evaluated too, and an increased risk, particularly for TSH levels >10 mIU/L, has been shown in several studies [4]. The Prospective Study of Pravastatin in the Elderly at Risk study evaluated a cohort of subjects aged 70 to 82 years with known cardiovascular risk factors; an association between sHT and HF was detected over 3.2 years of follow-up (only at TSH threshold >10 mIU/L), whereas no association was found with cardiovascular events or mortality except in those with TSH higher than 10 mIU/L and not taking pravastatin [6]. Likewise, a meta-analysis of six prospective cohort studies, with individual data on >25,000 patients with 216,248 person-years of follow-up, showed an increased risk of HF events for patients with both higher and lower TSH levels, particularly for TSH ≥10 and <0.10 mIU/L [7].
sHT and coronary heart disease are also linked, although with conflicting results, and a positive association is generally observed in the youngest population only [4]. Indeed, a large, prospective cohort study on 3233 subjects older than 65 years did not show any relationship between cardiovascular disorders and sHT, apart from an association with the development of atrial fibrillation [43]. Accordingly, a large meta-analysis showed increased mortality and risk of ischemic cardiac disease in patients with sHT younger than 65 years only [49]. However, another large meta-analysis of 11 prospective studies, with collected data from 55,287 subjects over 35 years of follow-up, after adjusting for sex and age, still demonstrated a higher risk of coronary heart disease events and mortality in sHT subjects, particularly in those with serum TSH >10 mIU/L [5].
3. sHT and Cognition
The involvement of thyroid hormones in the correct brain development and cognitive performance is well known. The role of hypothyroidism on cognition in the elderly has been widely questioned, and few studies have been conducted to evaluate the effect of overt and sHT on cognitive function with inconsistent results [50]. Data from the Framingham study, aiming to relate serum TSH levels to the risk of developing Alzheimer’s disease (AD) over an average of 12.7 years of follow-up, showed that women in the lowest (<1.0 mIU/L) and highest (>2.1 mIU/L) tertiles of serum TSH concentration were at increased risk for AD [multivariate-adjusted hazard ratio, 2.39 (95% confidence interval, 1.47 to 3.87)], whereas no increased risk was found in men [51]. In analyses limited to participants with serum TSH levels of 0.1 to 10.0 mIU/L, the U-shaped relationship between thyrotropin level and AD risk was maintained in women but not when analyses were limited to those with TSH levels of 0.5 to 5.0 mIU/L.
Other studies have reported rather negative results, instead. In a longitudinal study conducted on a population of 1077 elderly subjects (aged 60 to 90 years), cognitively normal, no association was identified between TSH or thyroid hormones and hippocampal atrophy or risk of developing AD [52]. In the Health, Aging and Body Composition Study, an elderly population (70 to 79 years of age) was assessed to evaluate the incident-adjudicated dementia and change in the cognitive performance; among the whole population, over 9 years of follow-up, the risk to develop dementia was found increased in sHT but not in sHT [53]. Accordingly, a recent meta-analysis aiming to evaluate the risk of cognitive impairment and dementia in mild thyroid dysfunction showed an association between sHT and risk of developing dementia, whereas such risk was not found in sHT [54]. Interestingly, another recent meta-analysis from 13 studies demonstrates a substantial relationship between sHT and cognitive impairment only in individuals younger than 75 years of age and those with higher TSH concentrations, whereas no correlation was found while considering all of the studies as a whole [55]. The authors conclude that the lack of use of age-related serum TSH reference ranges and consequent potential misdiagnosis of sHT in older people may account for this. Thus, with the consideration of the important role of thyroid function in the central nervous system and the lack of homogenous results, it would be interesting and crucial to widen the age range of the studies or set up long-term longitudinal studies to evaluate better if, when, and how mild thyroid failure could increase the risk of cognitive impairment, allowing clinicians to evaluate the therapeutic approach better.
4. Treatment of Hypothyroidism and sHT
The fundamental clinical principle in the case of glandular deficiency is replacement therapy, while available; in the case of overt hypothyroidism, the treatment of choice is l-T4 replacement therapy, also in the elderly [56]. It has been widely demonstrated that the resolution of hypothyroidism leads to release of related symptoms (i.e., fatigue, increased sensitivity to cold, constipation, dry skin, weight gain, puffy face, hoarseness, muscle weakness, etc.), as well as the improvement of cardiovascular, executive, and cognitive functions [44]. More caution should be considered while discussing the clinical management of sHT, especially in patients older than 75 to 80 years. The results of many studies have shown that l-T4 replacement therapy could be indicated in patients with TSH values >10 mIU/L, as they are at an increased risk of developing health disorders [4–8]. Beside the serum TSH cutoff value, clinicians should be advised to decide on a case-by-case basis, particularly if patients have other risk factors for cardiovascular disease and encompass signs and symptoms possibly associated with sHT. In this setting, it should be considered that sHT is seldom symptomatic in the elderly, and symptoms are generally nonspecific and poor predictors of actual thyroid status. Moreover, with prescriptions of new drugs, older people should be evaluated also in terms of the presence of frail syndrome and comorbidity [44]. In fact, the goal of treatment and the balance between benefits and risks may change depending on the specific needs of the patients, and also, the prescription of l-T4 replacement therapy should be tailored on the patient’s clinical characteristics.
Overall, in managing older patients with sHT, we should consider the values of TSH and their trend over time, and international guidelines suggest a cutoff of 10 mIU/L and a double check of TSH level (within 3 and 6 months) before treatment is recommended [9, 19]. Beside serum TSH cutoff levels, the presence of symptoms and signs of hypothyroidism should be checked before starting any treatment [9]. Unfortunately, the identification of the clinical pattern of hypothyroidism in the elderly is quite challenging for the presence of unspecific symptoms (i.e., fatigue, cognitive alterations, sleep disturbances, constipations, etc.), especially in frail older patients with comorbidity [57]. According to these considerations, a comprehensive, multidimensional geriatric assessment [58] may be helpful in the clinical approach to older patients with increased serum TSH values [44]. Moreover, it is conceivable to perform a complete thyroid function assessment, including either laboratory tests (FT3, FT4, TgAb, TPOAb) or ultrasound examination, to recognize an actual thyroid disease (e.g., autoimmune thyroiditis, gland atrophy, etc.), which may imply permanent TSH elevation or an increasing trend. Moreover, it is crucial to collect an accurate pharmacological history and evaluate whether the patient is receiving treatments affecting thyroid function (e.g., amiodarone, etc.), especially in the elderly with several comorbidities. However, it is worth noting that the “physiological” age-related increase of serum TSH values usually does not reach levels >7 to 8 mIU/L [59].
In this setting, a double-blind, randomized, placebo-controlled, parallel-group trial, involving 737 older patients with persistent sHT (mean age 74.4 years, 53.7% women), was recently performed to verify the efficacy and safety of l-T4 therapy [47]. The two primary outcomes were the change in the Hypothyroid Symptoms score and Tiredness score on a thyroid-related, quality-of-life questionnaire at 1 year. The mean entry TSH level was 6.40 ± 2.01 mIU/L. At 12 months, the mean TSH level was 5.48 ± 2.48 in the placebo group compared with 3.63 ± 2.11 mIU/L in the treatment group. No differences in terms of symptoms of hypothyroidism and quality of life (QoL) were observed between the treatment and placebo group. On the other hand, no substantial excess of serious adverse events prespecified as being of special interest was reported. The authors conclude that treatment with l-T4 in older persons with sHT provided no symptomatic benefits. However, few participants had a baseline TSH level >10 mIU/L, the symptom levels at trial entry were low, and the trial was underpowered to detect any effect of l-T4 on the incidence of cardiovascular events or mortality. Finally, the circulating antithyroid autoantibody titer was not assessed, and antibody-positive patients are more likely to have progressive hypothyroidism; therefore, they may be more likely to have a benefit from long-term l-T4 therapy [47, 60]. However, data on large, randomized trials on l-T4 treatment in antibody-positive older patients are not available yet, and the presence of autoimmunity may lose its clinical impact in frail older patients in whom l-T4 therapy is not recommended at all (Table 1). Nonetheless, in our opinion, in the clinical management of older people with sHT, physicians should take into account not only the TSH cutoff value but also the presence of positive antithyroid autoantibody titers, as well as chronic comorbidities, especially if potentially impaired by hypothyroidism, such as HF [48]. Moreover, particular attention should be given to the presence of frailty, a well-described clinical entity affecting the prognosis of older patients and their QoL [57, 61]. Indeed, frail patients are more vulnerable to drug side effects for several reasons, and the prescription of l-T4 should be carefully balanced with the risk of overtreatment (e.g., excessive intake of l-T4 as a result of insufficient compliance, no proper titration of dosage at the start of treatment, etc.).
Table 1.
Suggested Strategy of Care According to Either TSH Value or the Patient Clinical Features
| Serum TSH Valuea | Fit Patient | Frail Patient | ||
|---|---|---|---|---|
| 65–75 Y | >75 Y | 65–75 Y | >75 Y | |
| >10 mIU/L | Treatb | Treatc/observe | Observe/treatc | Observe |
| 6–10 mIU/L | Observe/treatc,d | Observe/treatc,d | Observe | Observe |
| 4–6 mIU/L | Observe/treatc,d | Observe | Observe | Observe |
Elevation of serum TSH value should be confirmed by at least a second measurement at 3- to 6-mo follow-up.
l-T4 dosage starting from 0.3 to 0.4 µg/kg/d; increments by 10% to 15% after 6 to 8 wk, if necessary. Optimal TSH target value for patients >75 y receiving l-T4 therapy: 2.5 to 3.5 mIU/L.
In the presence of positive antithyroid autoantibody titres, symptoms of hypothyroidism, concomitant diseases potentially impaired by mild thyroid failure (i.e., HF), also according to patients’ willing.
In the case of progressive increase of serum TSH value up to ≥10 mIU/L.
In conclusion, we could generalize that l-T4 replacement therapy is recommended for older patients (aged 65 to 75 years) in good health status with serum TSH values >10 mIU/L [9, 19]. l-T4 therapy should also be considered in clinically “fit” patients older than 75 years with actual thyroid disease and clear signs and symptoms of hypothyroidism, considering the presence of specific comorbidities (such as HF or cognitive impairment); otherwise, a proper strategy could simply be TSH value monitoring to check a possible increasing trend during time [9]. Conversely, in frail patients with serum TSH levels >10 mIU/L, more caution is recommended regardless of the age, and a wait-and-see strategy is recommended. In case of fit patients with circulating TSH value >6 and <10 mIU/L and actual risk factors for disease progression (women and positive antithyroid autoantibody titers), the possibility of a low-dose l-T4 replacement trial could be considered; otherwise, close monitoring of thyroid function every 3 to 6 months is recommended, and l-T4 therapy could be considered in the case of a progressive increase of a serum TSH value up to 10 mIU/L and above. In frail patients younger than 75 years, with serum TSH levels between 6 and 10 mIU/L, l-T4 replacement should be generally avoided; it could be hypothesized, with extreme caution, only in the case of a progressive TSH increase up to 10 mIU/L, in the presence of positive antithyroid autoantibody titers, signs and symptoms of hypothyroidism, and concomitant diseases potentially impaired by sHT (i.e., HF). In patients with serum TSH values between 4 and 6 mIU/L, a wait-and-see strategy is generally recommended. However, in fit patients younger than 75 years, with positive antithyroid autoantibody titres, signs and symptoms of hypothyroidism, and concomitant diseases potentially impaired by mild thyroid failure (i.e., HF), a low-dose l-T4 replacement trial could be considered (Table 1).
In any case, l-T4 dosage should be titrated starting from ∼0.4 to 0.5 µg/kg/d with increments by 10% to 15% after 6 to 8 weeks, if necessary, and an optimal TSH target value should be ∼2.5 to 3.5 mIU/L, according to international guidelines [9, 19]. It is highly recommended, especially in the oldest olds, to monitor thyroid function over time (e.g., every 6 months) to avoid over treatment and the consequent negative impact on cardiovascular and osteomuscular systems [9, 19].
5. l-T4 Administration
When orally administrated, l-T4 gets absorbed in the jejunum and ileus, 2 to 3 hours after ingestion. It is important to take into account that the administration of food and other drugs (i.e., proton pump inhibitor), together with l-T4, impairs its absorption; therefore, it is recommended to administer l-T4 either 1 hour before breakfast or 3 or more hours after dinner [9, 19].
There are different ways of administration, depending on the pharmaceutical formulation, such as tablet, liquid, or soft gel. The formulations differ for the excipient, and the liquid or soft gel formulation could be preferred when the traditional tablets fail to normalize the TSH values. The gastrointestinal absorption is the critical event to be considered in the therapy, and it can be influenced by multiple factors of a different nature, from the patient’s compliance to the therapy, to the presence of gastrointestinal diseases (i.e., gastric atrophy, Helicobacter pylori infection, lactose intolerance, or celiac disease), and/or the concomitant intake of drugs or food [62]. The most used pharmaceutical formulation for the therapy is the l-T4 tablet, and it is the first choice in the elderly patient without a swallowing problem; it is indeed possible to swap to a different pharmaceutical formulation if the normalization of the TSH values is not met, even after a careful evaluation of the current therapy. Liquid and soft gel formulations have been developed over the last years to overcome the issue as a result of the gastric absorption, even when administered together with the food. The liquid formulation seems to have a better bioavailability compared with the l-T4 tablets, whereas the soft gel is equivalent. In a recent review by Virili et al. [62], several studies evaluating the use of liquid and soft gel formulation in different populations were considered. Few studies and case reports demonstrated the higher efficacy of the soft gel formulation compared with the tablets in patients with gastric disorders, impaired absorption, even iatrogenic, or during pregnancy [62]. These data, although interesting and promising, are not confirmed by long-term, controlled studies; therefore, some caution is necessary to tailor properly the therapy according to a patient’s needs.
Whereas some studies suggest that the liquid formulation gives less variability in the TSH level under treatment, both in adults and elderly, it is important to consider that the timing of ingestion of the liquid formulation is different from the tablet; therefore, the results need to be interpreted carefully [55]. Indeed, there are patients who certainly would benefit from the use of a liquid formulation, such as those who underwent bariatric surgery, have enteric-tube nutrition, have drug-drug interference (in particular, proton pump inhibitors), or are less keen to comply with the timings of administration of the tablets in relation to breakfast or coffee [55]. In this respect, it has been demonstrated that circulating FT4 levels, 3 and 4 hours after administration of liquid l-T4, are comparable with the levels obtained with fasting administration of l-T4 tablets [63].
The use of combined therapy with T4 + T3 is still a matter of debate. A recent review of the literature examined several studies comparing different outcomes from the combined therapy compared with the classic approach [64]. When evaluating QoL, mood, and cognitive performance, few studies reported an improvement in those outcomes, but in the majority of them, no advantages were seen, and a possible placebo effect was considered worth noticing [64]. A meta-analysis conducted on 1216 subjects and evaluating several outcomes (pain, depression, fatigue, anxiety, cognitive performance, and QoL) did not find any difference between T4 + T3 and T4 in monotherapy [65]. Another meta-analysis evaluating 10 randomized double-blinded studies confirmed the same outcome of no advantage with the combined therapy [66]. A limitation of the available studies is that they are all short term, not exceeding 1-year duration [64]. However, a 17-year follow-up study analyzed multiple outcomes in a population receiving T3, in most cases, associated with T4, compared with patients receiving only T4. The outcomes in terms of cardiovascular events, diabetes, fractures, atrial fibrillation, or death were comparable; the only difference was an increased use of antipsychotic drugs in the combined therapy population, presumably as a result of the use of T3 [67]. Accordingly, the 2012 European Thyroid Association guidelines suggest that the combined therapy T4/T3 should be used only in patients with normal TSH values reached with l-T4 alone, but with persistent complaints, and only after having educated patients about the chronic nature of their disease [68]. Moreover, the combination therapy should be avoided in pregnant women, as well as in subjects with cardiac arrhythmias, and extreme caution is required in the elderly. In the treated population, it should be stopped if there is no clinical improvement achieved after 3 months [68].
6. Conclusions
Hypothyroidism is frequently observed in the elderly with an increasing trend with age. A “natural” trend in a slight TSH increase has been documented in the older population, even in subjects without documentable thyroid diseases, but an age-related TSH reference range is not available yet. Thus, it is worth the performance of an extensive thyroid evaluation in older subjects with a circulating TSH rise, especially in the oldest olds, including either laboratory tests (free thyroid hormone levels and antithyroid autoantibody titers) or thyroid ultrasound examination. This diagnostic process is aimed to assess the presence of an actual thyroid disease (Hashimoto thyroiditis, gland atrophy, etc.), which may lead to the diagnosis of sHT rather than a physiological age-related TSH elevation, although circulating TSH values >10 mIU/L should be considered clinically relevant. The choice of treatment should also depend on the presence of clinical signs and symptoms consistent with hypothyroidism, as well as concomitant comorbidities and patient compliance. Nonetheless, several data from literature warn the clinician to be extremely cautious in treating older patients, especially the oldest olds (>80 years).
In conclusion, in the clinical management of older people with sHT, not only TSH value cutoffs need to be considered, but also, the presence of an actual thyroid disease, as well as chronic comorbidities and frailty, should be taken into account. Good clinical practice implies the prescription of l-T4 on a case-by-case manner, carefully balanced by the risk of overtreatment (e.g., excessive intake of the drug as a result of mistakes in administration, no proper titration of dosage at the start of treatment, etc.). In any case, l-T4 dosage should be titrated starting from ∼0.3 to 0.4 µg/kg/d, with increments by 10% to 15% after 6 to 8 weeks, if necessary, and optimal TSH target value should be ∼2.5 to 3.5 mIU/L.
Acknowledgments
Disclosure Summary: The authors have nothing to disclose.
Glossary
Abbreviations:
- AD
Alzheimer’s disease
- FT3
free triiodothyronine
- FT4
free thyroxine
- HF
heart failure
- l-T4
levothyroxine
- NHANES III
National Health and Nutrition Examination Survey
- QoL
quality of life
- sHT
subclinical hypothyroidism
- T3
triiodothyronine
- T4
thyroxine
- TgAb
antithyroglobulin
- TPOAb
antithyroid peroxidase
- TSH
thyroid-stimulating hormone
References and Notes
- 1.Eurostat. EUROPOP13 database, main scenario. Available at: ec.europa.eu/eurostat/web/population-demography-migration-projections/population-projections-data. Accessed 10 April 2018.
- 2. Pearce EN. Hypothyroidism is associated with increased mortality risk in individuals 65 years of age or older. Clin Thyroidol. 2018;30(8):350–352. [Google Scholar]
- 3. Biondi B, Cooper DS. The clinical significance of subclinical thyroid dysfunction. Endocr Rev. 2008;29(1):76–131. [DOI] [PubMed] [Google Scholar]
- 4. Pasqualetti G, Tognini S, Polini A, Caraccio N, Monzani F. Is subclinical hypothyroidism a cardiovascular risk factor in the elderly? J Clin Endocrinol Metab. 2013;98(6):2256–2266. [DOI] [PubMed] [Google Scholar]
- 5. Waring AC, Rodondi N, Harrison S, Kanaya AM, Simonsick EM, Miljkovic I, Satterfield S, Newman AB, Bauer DC; Health, Ageing, and Body Composition (Health ABC) Study . Thyroid function and prevalent and incident metabolic syndrome in older adults: the Health, Ageing and Body Composition Study. Clin Endocrinol (Oxf). 2012;76(6):911–918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Nanchen D, Gussekloo J, Westendorp RG, Stott DJ, Jukema JW, Trompet S, Ford I, Welsh P, Sattar N, Macfarlane PW, Mooijaart SP, Rodondi N, de Craen AJ; PROSPER Group . Subclinical thyroid dysfunction and the risk of heart failure in older persons at high cardiovascular risk. J Clin Endocrinol Metab. 2012;97(3):852–861. [DOI] [PubMed] [Google Scholar]
- 7. Gencer B, Collet TH, Virgini V, Bauer DC, Gussekloo J, Cappola AR, Nanchen D, den Elzen WP, Balmer P, Luben RN, Iacoviello M, Triggiani V, Cornuz J, Newman AB, Khaw KT, Jukema JW, Westendorp RG, Vittinghoff E, Aujesky D, Rodondi N; Thyroid Studies Collaboration . Subclinical thyroid dysfunction and the risk of heart failure events: an individual participant data analysis from 6 prospective cohorts. Circulation. 2012;126(9):1040–1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Rodondi N, den Elzen WP, Bauer DC, Cappola AR, Razvi S, Walsh JP, Asvold BO, Iervasi G, Imaizumi M, Collet TH, Bremner A, Maisonneuve P, Sgarbi JA, Khaw KT, Vanderpump MP, Newman AB, Cornuz J, Franklyn JA, Westendorp RG, Vittinghoff E, Gussekloo J; Thyroid Studies Collaboration . Subclinical hypothyroidism and the risk of coronary heart disease and mortality. JAMA. 2010;304(12):1365–1374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Pearce SH, Brabant G, Duntas LH, Monzani F, Peeters RP, Razvi S, Wemeau JL. 2013 ETA Guideline: management of subclinical hypothyroidism. Eur Thyroid J. 2013;2(4):215–228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Taylor PN, Albrecht D, Scholz A, Gutierrez-Buey G, Lazarus JH, Dayan CM, Okosieme OE. Global epidemiology of hyperthyroidism and hypothyroidism. Nat Rev Endocrinol. 2018;14(5):301–316. [DOI] [PubMed] [Google Scholar]
- 11. Hollowell JG, Staehling NW, Flanders WD, Hannon WH, Gunter EW, Spencer CA, Braverman LE. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2002;87(2):489–499. [DOI] [PubMed] [Google Scholar]
- 12. Vanderpump MP, Tunbridge WM, French JM, Appleton D, Bates D, Clark F, Grimley Evans J, Hasan DM, Rodgers H, Tunbridge F, Young ET. The incidence of thyroid disorders in the community: a twenty-year follow-up of the Whickham Survey. Clin Endocrinol (Oxf). 1995;43(1):55–68. [DOI] [PubMed] [Google Scholar]
- 13. Cooper DS, Biondi B. Subclinical thyroid disease. Lancet. 2012;379(9821):1142–1154. [DOI] [PubMed] [Google Scholar]
- 14. Canaris GJ, Manowitz NR, Mayor G, Ridgway EC. The Colorado thyroid disease prevalence study. Arch Intern Med. 2000;160(4):526–534. [DOI] [PubMed] [Google Scholar]
- 15. Wilson S, Parle JV, Roberts LM, Roalfe AK, Hobbs FD, Clark P, Sheppard MC, Gammage MD, Pattison HM, Franklyn JA; Birmingham Elderly Thyroid Study Team . Prevalence of subclinical thyroid dysfunction and its relation to socioeconomic deprivation in the elderly: a community-based cross-sectional survey. J Clin Endocrinol Metab. 2006;91(12):4809–4816. [DOI] [PubMed] [Google Scholar]
- 16. Parle JV, Franklyn JA, Cross KW, Jones SC, Sheppard MC. Prevalence and follow-up of abnormal thyrotrophin (TSH) concentrations in the elderly in the United Kingdom. Clin Endocrinol (Oxf). 1991;34(1):77–83. [DOI] [PubMed] [Google Scholar]
- 17. Franklyn JA. The thyroid--too much and too little across the ages. The consequences of subclinical thyroid dysfunction. Clin Endocrinol (Oxf). 2013;78(1):1–8. [DOI] [PubMed] [Google Scholar]
- 18. The Medicines Utilization Monitoring Centre National Report on Medicines Use in Italy 2016. Rome, Italy: Italian Medicines Agency; 2017:39. [Google Scholar]
- 19. Jonklaas J, Bianco AC, Bauer AJ, Burman KD, Cappola AR, Celi FS, Cooper DS, Kim BW, Peeters RP, Rosenthal MS, Sawka AM; American Thyroid Association Task Force on Thyroid Hormone Replacement . Guidelines for the treatment of hypothyroidism: prepared by the American Thyroid Association Task Force on Thyroid Hormone Replacement. Thyroid. 2014;24(12):1670–1751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Braverman LE, Utiger RD. Werner and Ingbar’s, The Thyroid, a Fundamental and Clinical Text. Philadelphia, PA: Lippincott Williams & Wilkins; 2005:697. [Google Scholar]
- 21. Mariotti S, Caturegli P, Piccolo P, Barbesino G, Pinchera A. Antithyroid peroxidase autoantibodies in thyroid diseases. J Clin Endocrinol Metab. 1990;71(3):661–669. [DOI] [PubMed] [Google Scholar]
- 22. Huber G, Staub JJ, Meier C, Mitrache C, Guglielmetti M, Huber P, Braverman LE. Prospective study of the spontaneous course of subclinical hypothyroidism: prognostic value of thyrotropin, thyroid reserve, and thyroid antibodies. J Clin Endocrinol Metab. 2002;87(7):3221–3226. [DOI] [PubMed] [Google Scholar]
- 23. Karmisholt J, Andersen S, Laurberg P. Variation in thyroid function in subclinical hypothyroidism: importance of clinical follow-up and therapy. Eur J Endocrinol. 2011;164(3):317–323. [DOI] [PubMed] [Google Scholar]
- 24. Pedersen OM, Aardal NP, Larssen TB, Varhaug JE, Myking O, Vik-Mo H. The value of ultrasonography in predicting autoimmune thyroid disease. Thyroid. 2000;10(3):251–259. [DOI] [PubMed] [Google Scholar]
- 25. Andersen S, Pedersen KM, Bruun NH, Laurberg P. Narrow individual variations in serum T(4) and T(3) in normal subjects: a clue to the understanding of subclinical thyroid disease. J Clin Endocrinol Metab. 2002;87(3):1068–1072. [DOI] [PubMed] [Google Scholar]
- 26. Mariotti S, Franceschi C, Cossarizza A, Pinchera A. The aging thyroid. Endocr Rev. 1995;16(6):686–715. [DOI] [PubMed] [Google Scholar]
- 27. Monzani F, Del Guerra P, Caraccio N, Del Corso L, Casolaro A, Mariotti S, Pentimone F. Age-related modifications in the regulation of the hypothalamic-pituitary-thyroid axis. Horm Res. 1996;46(3):107–112. [DOI] [PubMed] [Google Scholar]
- 28. van den Beld AW, Visser TJ, Feelders RA, Grobbee DE, Lamberts SW. Thyroid hormone concentrations, disease, physical function, and mortality in elderly men. J Clin Endocrinol Metab. 2005;90(12):6403–6409. [DOI] [PubMed] [Google Scholar]
- 29. Wiersinga WM, van den Berghe G. Nonthyroidal illness syndrome In: Braverman LE, Cooper D, eds. Werner and Ingbar’s, The Thyroid, a Fundamental and Clinical Text. 9th edPhiladelphia, PA: Lippincott Williams & Wilkins; 2004:246–263. [Google Scholar]
- 30. Tognini S, Polini A, Pasqualetti G, Ursino S, Caraccio N, Ferdeghini M, Monzani F. Age and gender substantially influence the relationship between thyroid status and the lipoprotein profile: results from a large cross-sectional study. Thyroid. 2012;22(11):1096–1103. [DOI] [PubMed] [Google Scholar]
- 31. Rozing MP, Houwing-Duistermaat JJ, Slagboom PE, Beekman M, Frölich M, de Craen AJ, Westendorp RG, Van Heemst D. Familial longevity is associated with decreased thyroid function. J Clin Endocrinol Metab. 2010;95(11):4979–4984. [DOI] [PubMed] [Google Scholar]
- 32. Iovino M, Steardo L, Monteleone P. Impaired sensitivity of the hypothalamo-pituitary-thyroid axis to the suppressant effect of dexamethasone in elderly subjects. Psychopharmacology (Berl). 1991;105(4):481–484. [DOI] [PubMed] [Google Scholar]
- 33. Mariotti S, Barbesino G, Caturegli P, Bartalena L, Sansoni P, Fagnoni F, Monti D, Fagiolo U, Franceschi C, Pinchera A. Complex alteration of thyroid function in healthy centenarians. J Clin Endocrinol Metab. 1993;77(5):1130–1134. [DOI] [PubMed] [Google Scholar]
- 34. Sawin CT, Chopra D, Azizi F, Mannix JE, Bacharach P. The aging thyroid. Increased prevalence of elevated serum thyrotropin levels in the elderly. JAMA. 1979;242(3):247–250. [DOI] [PubMed] [Google Scholar]
- 35. Surks MI, Hollowell JG. Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: implications for the prevalence of subclinical hypothyroidism. J Clin Endocrinol Metab. 2007;92(12):4575–4582. [DOI] [PubMed] [Google Scholar]
- 36. Hennessey JV, Espaillat R. Diagnosis and management of subclinical hypothyroidism in elderly adults: a review of the literature. J Am Geriatr Soc. 2015;63(8):1663–1673. [DOI] [PubMed] [Google Scholar]
- 37. Tognini S, Marchini F, Dardano A, Polini A, Ferdeghini M, Castiglioni M, Monzani F. Non-thyroidal illness syndrome and short-term survival in a hospitalised older population. Age Ageing. 2010;39(1):46–50. [DOI] [PubMed] [Google Scholar]
- 38. Piers LS, Soares MJ, McCormack LM, O’Dea K. Is there evidence for an age-related reduction in metabolic rate? J Appl Physiol (1985). 1998;85(6):2196–2204. [DOI] [PubMed] [Google Scholar]
- 39. Corsonello A, Montesanto A, Berardelli M, De Rango F, Dato S, Mari V, Mazzei B, Lattanzio F, Passarino G. A cross-section analysis of FT3 age-related changes in a group of old and oldest-old subjects, including centenarians’ relatives, shows that a down-regulated thyroid function has a familial component and is related to longevity. Age Ageing. 2010;39(6):723–727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Boucai L, Hollowell JG, Surks MI. An approach for development of age-, gender-, and ethnicity-specific thyrotropin reference limits. Thyroid. 2011;21(1):5–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Atzmon G, Barzilai N, Hollowell JG, Surks MI, Gabriely I. Extreme longevity is associated with increased serum thyrotropin. J Clin Endocrinol Metab. 2009;94(4):1251–1254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Chaker L, Wolters FJ, Bos D, Korevaar TI, Hofman A, van der Lugt A, Koudstaal PJ, Franco OH, Dehghan A, Vernooij MW, Peeters RP, Ikram MA. Thyroid function and the risk of dementia: The Rotterdam Study. Neurology. 2016;87(16):1688–1695. [DOI] [PubMed] [Google Scholar]
- 43. Bremner AP, Feddema P, Leedman PJ, Brown SJ, Beilby JP, Lim EM, Wilson SG, O’Leary PC, Walsh JP. Age-related changes in thyroid function: a longitudinal study of a community-based cohort. J Clin Endocrinol Metab. 2012;97(5):1554–1562. [DOI] [PubMed] [Google Scholar]
- 44. Ruggeri RM, Trimarchi F, Biondi B. Management of endocrine disease: l-thyroxine replacement therapy in the frail elderly: a challenge in clinical practice. Eur J Endocrinol. 2017;177(4):R199–R217. [DOI] [PubMed] [Google Scholar]
- 45. Solini A, Monzani F. Hypothyroidism and intermediate metabolism: a complex relationship. Thyroid. 2010;20(8):837–839. [DOI] [PubMed] [Google Scholar]
- 46. Huang HK, Wang JH, Kao SL. Association of hypothyroidism with all-cause mortality: a cohort study in an older adult population. J Clin Endocrinol Metab. 2018;103(9):3310–3318. [DOI] [PubMed] [Google Scholar]
- 47. Stott DJ, Rodondi N, Kearney PM, Ford I, Westendorp RGJ, Mooijaart SP, Sattar N, Aubert CE, Aujesky D, Bauer DC, Baumgartner C, Blum MR, Browne JP, Byrne S, Collet TH, Dekkers OM, den Elzen WPJ, Du Puy RS, Ellis G, Feller M, Floriani C, Hendry K, Hurley C, Jukema JW, Kean S, Kelly M, Krebs D, Langhorne P, McCarthy G, McCarthy V, McConnachie A, McDade M, Messow M, O’Flynn A, O’Riordan D, Poortvliet RKE, Quinn TJ, Russell A, Sinnott C, Smit JWA, Van Dorland HA, Walsh KA, Walsh EK, Watt T, Wilson R, Gussekloo J; TRUST Study Group . Thyroid hormone therapy for older adults with subclinical hypothyroidism. N Engl J Med. 2017;376(26):2534–2544. [DOI] [PubMed] [Google Scholar]
- 48. Pasqualetti G, Tognini S, Polini A, Caraccio N, Monzani F. Subclinical hypothyroidism and heart failure risk in older people. Endocr Metab Immune Disord Drug Targets. 2013;13(1):13–21. [DOI] [PubMed] [Google Scholar]
- 49. Sun J, Yao L, Fang Y, Yang R, Chen Y, Yang K, Tian L. Relationship between subclinical thyroid dysfunction and the risk of cardiovascular outcomes: a systematic review and meta-analysis of prospective cohort studies. Int J Endocrinol. 2017;2017:8130796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Szlejf C, Suemoto CK, Santos IS, Lotufo PA, Haueisen Sander Diniz MF, Barreto SM, Benseñor IM. Thyrotropin level and cognitive performance: baseline results from the ELSA-Brasil Study. Psychoneuroendocrinology. 2018;87:152–158. [DOI] [PubMed] [Google Scholar]
- 51. Tan ZS, Beiser A, Vasan RS, Au R, Auerbach S, Kiel DP, Wolf PA, Seshadri S. Thyroid function and the risk of Alzheimer disease: the Framingham Study. Arch Intern Med. 2008;168(14):1514–1520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. de Jong FJ, den Heijer T, Visser TJ, de Rijke YB, Drexhage HA, Hofman A, Breteler MM. Thyroid hormones, dementia, and atrophy of the medial temporal lobe. J Clin Endocrinol Metab. 2006;91(7):2569–2573. [DOI] [PubMed] [Google Scholar]
- 53. Aubert CE, Bauer DC, da Costa BR, Feller M, Rieben C, Simonsick EM, Yaffe K, Rodondi N; Health ABC Study . The association between subclinical thyroid dysfunction and dementia: the Health, Aging and Body Composition (Health ABC) Study. Clin Endocrinol (Oxf). 2017;87(5):617–626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Rieben C, Segna D, da Costa BR, Collet TH, Chaker L, Aubert CE, Baumgartner C, Almeida OP, Hogervorst E, Trompet S, Masaki K, Mooijaart SP, Gussekloo J, Peeters RP, Bauer DC, Aujesky D, Rodondi N. Subclinical thyroid dysfunction and the risk of cognitive decline: a meta-analysis of prospective cohort studies. J Clin Endocrinol Metab. 2016;101(12):4945–4954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Pasqualetti G, Pagano G, Rengo G, Ferrara N, Monzani F. Subclinical hypothyroidism and cognitive impairment: systematic review and meta-analysis. J Clin Endocrinol Metab. 2015;100(11):4240–4248. [DOI] [PubMed] [Google Scholar]
- 56. Garber JR, Cobin RH, Gharib H, Hennessey JV, Klein I, Mechanick JI, Pessah-Pollack R, Singer PA, Woeber KA; American Association of Clinical Endocrinologists and American Thyroid Association Taskforce on Hypothyroidism in Adults . Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr Pract. 2012;18(6):988–1028. [DOI] [PubMed] [Google Scholar]
- 57. Pasqualetti G, Calsolaro V, Bernardini S, Linsalata G, Bigazzi R, Caraccio N, Monzani F. Degree of peripheral thyroxin deiodination, frailty, and long-term survival in hospitalized older patients. J Clin Endocrinol Metab. 2018;103(5):1867–1876. [DOI] [PubMed] [Google Scholar]
- 58. Parker SG, McCue P, Phelps K, McCleod A, Arora S, Nockels K, Kennedy S, Roberts H, Conroy S. What is comprehensive geriatric assessment (CGA)? An umbrella review. Age Ageing. 2018;47(1):149–155. [DOI] [PubMed] [Google Scholar]
- 59. Hennessey JV, Espaillat R. Subclinical hypothyroidism: a historical view and shifting prevalence. Int J Clin Pract. 2015;69(7):771–782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Javed Z, Sathyapalan T. Levothyroxine treatment of mild subclinical hypothyroidism: a review of potential risks and benefits. Ther Adv Endocrinol Metab. 2016;7(1):12–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, Seeman T, Tracy R, Kop WJ, Burke G, McBurnie MA; Cardiovascular Health Study Collaborative Research Group . Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56(3):M146–M156. [DOI] [PubMed] [Google Scholar]
- 62. Virili C, Giovanella L, Fallahi P, Antonelli A, Santaguida MG, Centanni M, Trimboli P. Levothyroxine therapy: changes of TSH levels by switching patients from tablet to liquid formulation. A systematic review and meta-analysis. Front Endocrinol (Lausanne). 2018;9:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Marina M, Ceda GP, Aloe R, Gnocchi C, Ceresini G. Circulating concentrations of free thyroxine after an oral intake of liquid LT4 taken either during fasting conditions or at breakfast. Acta Biomed. 2017;87(3):247–252. [PMC free article] [PubMed] [Google Scholar]
- 64. Hennessey JV, Espaillat R. Current evidence for the treatment of hypothyroidism with levothyroxine/levotriiodothyronine combination therapy versus levothyroxine monotherapy. Int J Clin Pract. 2018;72(2):e13062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Grozinsky-Glasberg S, Fraser A, Nahshoni E, Weizman A, Leibovici L. Thyroxine-triiodothyronine combination therapy versus thyroxine monotherapy for clinical hypothyroidism: meta-analysis of randomized controlled trials. J Clin Endocrinol Metab. 2006;91(7):2592–2599. [DOI] [PubMed] [Google Scholar]
- 66. Ma C, Xie J, Huang X, Wang G, Wang Y, Wang X, Zuo S. Thyroxine alone or thyroxine plus triiodothyronine replacement therapy for hypothyroidism. Nucl Med Commun. 2009;30(8):586–593. [DOI] [PubMed] [Google Scholar]
- 67. Leese GP, Soto-Pedre E, Donnelly LA. Liothyronine use in a 17 year observational population-based study—the tears study. Clin Endocrinol (Oxf). 2016;85(6):918–925. [DOI] [PubMed] [Google Scholar]
- 68. Wiersinga WM, Duntas L, Fadeyev V, Nygaard B, Vanderpump MP. 2012 ETA Guidelines: the use of L-T4 + L-T3 in the treatment of hypothyroidism. Eur Thyroid J. 2012;1(2):55–71. [DOI] [PMC free article] [PubMed] [Google Scholar]