Abstract
Background & Aims:
Neutrophil extracellular traps (NETs) are an important strategy utilized by neutrophils to immobilize and kill invading microorganisms. Here we studied NET formation and its clearance by macrophages (MØ) (efferocytosis) in acute sepsis following binge drinking.
Methods:
Healthy volunteers consumed 2 mL of vodka/kg body weight and blood endotoxin and 16s rDNA was measured. Peripheral neutrophils were isolated and exposed to alcohol followed by phorbol 12-myristate 13-acetate (PMA) stimulation. Mice were treated with three alcohol binges and i.p. LPS to assess the dynamics of NET formation and efferocytosis. In vivo, anti-Ly6G antibody (IA8) was used for neutrophil depletion.
Results:
Inducers of NETs (endotoxin and bacterial DNA) significantly increased in the circulation after binge alcohol drinking in humans. Ex vivo, alcohol alone increased NET formation but attenuated NET formation upon PMA stimulation. Binge alcohol in mice in vivo resulted in a biphasic response to LPS. Initially, binge alcohol reduced LPS-induced NET formation and resulted in a diffuse distribution of neutrophils in the liver compared to alcohol-naïve mice. Moreover, indicators of NET formation including citrullinated histone H3, neutrophil elastase, and neutrophil myeloperoxidase were decreased at an early time point after LPS challenge in mice with alcohol binge suggesting decreased NET formation. However, in the efferocytosis phase (15 h after LPS) citrullinated histone-H3 was increased in the liver in alcohol binge mice, suggesting decreased clearance of NETs. In vitro alcohol treatment reduced efferocytosis and phagocytosis of NETosing neutrophils and promoted expression of CD206 on MØ. Finally, depletion of neutrophils prior to binge alcohol ameliorated LPS-induced systemic inflammation and liver injury in mice.
Conclusions:
Dysfunctional neutrophil NETosis and efferocytosis after binge drinking exacerbates liver injury associated with sepsis.
Keywords: Sepsis, binge drinking, alcoholic hepatitis, neutrophil elastase, neutrophil depletion
Lay Abstract:
Disease severity in alcoholic liver disease (ALD) is associated with significant liver neutrophil presence. It remains unknown how alcohol affects the capacity of neutrophils to control infection, a major hallmark of ALD. We found that binge alcohol drinking impaired important strategies used by neutrophils to contain and resolve infection resulting in increased liver injury during ALD.
Graphical Abstract
Introduction
Alcoholic liver disease (ALD) affects millions of people worldwide. The multifaceted disease spectrum is characterized by increased liver inflammation and steatosis as a direct effect of alcohol, alcohol metabolites and increased hepatic oxidative stress [1–3]. Sustained dysregulated hepatic inflammation during ALD is mediated by increased mobilization and recruitment of inflammatory cells into the liver. This occurrence normally precedes the breakdown of the gut barrier integrity and increased serum endotoxin levels, leading to prolonged hepatic inflammation and cell death [3, 4]. During alcoholic hepatitis (AH), innate immune cells play a crucial role not only in recognizing and responding to pathogen-associated molecular patterns (PAMPs) but also in contributing to the activation of the inflammatory cascade that correlates with disease severity in AH [5–7]. Most studies on innate immune cells in AH and ALD have focused on the dysregulated migration, phagocytosis, and inflammatory cytokine release properties of macrophages (MΦ) and neutrophils [8–10]. During ALD associated with clinical sepsis, neutrophils are recruited to the liver within hours, adhering to activated blood vessels or migrating to the parenchyma. Neutrophil recruitment is increased under systemic inflammatory conditions [1, 11]. Increased liver neutrophil infiltration correlates with mortality in acute AH; however, little is known about the functional capacity of neutrophils in AH. Mechanisms of neutrophil activation, recruitment, and innate immune functions, as well as their contribution to hepatic inflammation and injury during AH/ALD also remain incompletely understood.
Neutrophils represent the most abundant innate immune cell type accounting for about 40–75% of all circulating white blood cells in humans [12]. They have a short lifespan in the circulation of a few hours which is dramatically increased to days when they leave the circulation and migrate into tissues [13, 14]. Despite their short lifespans, neutrophils are constantly replenished from bone marrow stem cells [15]. When released from the bone marrow, neutrophils can phagocytose and kill pathogens [15]. During infection or sterile tissue injury, neutrophils are the first responder cell type to be recruited to the site providing immune protection and contributing to healing and recovery. When exposed to pathogens or damage-associated molecular patterns (DAMPs), neutrophils engulf and degrade them in phagolysosomes by oxidative and non-oxidative mechanisms[16]. The migratory, phagocytic capacity of PAMPs/DAMPs is significantly reduced in ALD but oxidative burst is increased. In addition to this pathogen killing strategy, in 2004, Brinkmann et al demonstrated the ability of bacteria-killing by neutrophils through neutrophil extracellular traps (NETs)[17]. NETs are networks of extracellular ‘fibers in a spider-web like’ formation composed of DNA, citrullinated histones and antimicrobial peptides capable of directly killing some bacterial species. The functional role of NETs in ALD has not been described.
NET formation is dependent on PAMPs (LPS) and DAMPs (HMGB1) which can activate neutrophils leading to the release of NETs. We found that ALD patients had significantly higher levels of blood LPS and HMGB1 protein expression. Ex vivo stimulation of neutrophils from ALD patients showed reduced NET formation compared to healthy controls. In vitro and in vivo mouse experiments demonstrated that alcohol significantly suppressed hepatic NET formation and significantly decreased NETosis neutrophil clearance by macrophages. Taken together, our observations suggest that increased hepatic injury during ALD associated with acute sepsis might be exacerbated by reduced NET formation and their impaired clearance by macrophages.
Materials and Methods
Mice
Experiments presented used Female 10–12-week old wild type C57BL/6J mice obtained from Jackson Laboratories (Bar Harbor, ME, USA) and maintained on chow Rodent Diet, AIN-76A(BioServe). All mice were housed in MicroVENT IVC Systems cages at the University of Massachusetts Medical School (UMMS) animal facility and treated humanely according to the criteria outlined in the “Guide for the Care and Use of Laboratory Animals” prepared by the National Academy of Sciences and published by the National Institutes of Health. All experiments were performed under a UMMS Institutional Animal Care and Use Committee approved protocol number 1154. All mice had free and unlimited access to water at all times throughout the entire experimental period. A 12hours light and 12hours dark cycle was maintained for all experiments.
Acute binge alcoholic hepatitis model
For assessing the effect of alcohol consumption on NET formation in vivo, mice received a daily oral gavage of alcohol at 5 mg/kg body weight (20% vol/vol in sterile phosphate buffer saline) for three days as previously described [18]. On day three following the final alcohol gavage, mice received a single i.p. injection of LPS or saline. Mice were sacrificed 12h and 15h after the injection. At the end of all animal experiments, blood samples were obtained from mice by cheek bleed into serum collection tubes (BD Biosciences, San Jose, CA, USA) and processed within the hour. Following blood collections, mice were euthanized and liver samples harvested and stored appropriately for further analysis.
Neutrophil isolation from blood samples
Total neutrophils from whole blood from humans or mice were obtained by negative selection using the EasySep Mouse or Human Neutrophil Enrichment Kit from STEMCELL technologies (Vancouver, Canada) according to the manufacturer’s specifications. For the NETosis assay giving the need for more neutrophils, we employed a modified ficoll isolation method. Briefly, whole blood was separated by ficoll separation method as similarly described. The red blood cell-neutrophil containing fraction was recovered. This fraction was treated with red blood cell lysis buffer (Sigma-Aldrich cat. #11814389001) and washed three times with 1x phosphate buffer saline. The recovered neutrophils were assessed to ascertain non-activation using the Cellular Reactive Oxygen Species Detection Assay Kit (Abcam cat. #186029). Neutrophils were used for subsequent NETosis experiments as detailed below.
NET trap formation and analysis assay
In vitro NET formation by neutrophils was assessed by microscopy and by ELISA. For microscopy analysis, neutrophils (105 cells) from healthy donors were plated on poly-L-lysine-coated coverslips. Cells were allowed to settle for 1h, then treated with 50mM ethanol or untreated, then stimulated with 0.2nM PMA (phorbol 12-myristate 13-acetate) for 4h. Cells were then fixed in 25 formol-saline for 10min at room temperature, then stained with DAPI and visualized by fluorescent microscopy for extracellular DNA. Quantitative neutrophil elastase, which forms part of NETs, was measured using the Cayman Chemical NETosis assay kit (cat #: 601010, Cayman Chemical, Ann Arbor, MI, USA) according to the manufacturer’s specifications.
Healthy subjects and patients diagnosed with alcoholic hepatitis
The use of human samples for this research was approved by the UMMS Committee for the Protection of Human Subjects in Research and conformed to the ethical guidelines of the 1975 Declaration of Helsinki. All apparently healthy blood donor volunteers were recruited from the laboratory, hospital staff, graduate and medical students at UMMS. Signed informed written consents were obtained from all participants and the study was approved by the UMMS Institutional Review Board for the Protection of Human Subjects in Research (IRB #2284). Healthy individuals were defined as being free of any systemic and non-systemic diseases, and either abstained from alcohol use or reported only occasional social alcohol use based on patients’ history and routine laboratory findings identified by their primary care physician and at interviews. Subjects for binge alcohol use were recruited following a strict criterion. Males were expected to never have had more than 12 drinks/week and females less than 9 drinks/week. All study participants had to abstain from alcohol use for at least 48 h prior to participating in the study. Study participants received 2 ml of vodka 40%v/v ethanol/kg body weight in 300 ml of orange or strawberry juice similar to previous reports [19]. Control subjects received 300 ml of orange or strawberry juice.
Additional methods
Please see the supplementary methods section for additional methodology used in this study.
Statistical analysis
All graphical results are expressed as the mean + S.E.M. for data analyzed with an unpaired two-tailed student’s t-test and checked by analysis of variance (ANOVA) with p<0.05 considered to be statistically significant.
Results
Binge drinking increases blood endotoxins and significantly reduces NET formation following ex vivo stimulation of neutrophils in humans
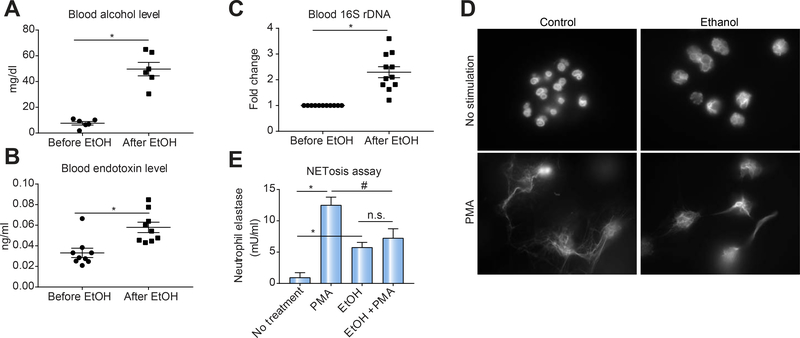
Despite significant neutrophil infiltration into the liver, sepsis-related organ failure represents the major cause of mortality in ALD patients [1, 20–22]. While studies have shown that alcohol use can compromise neutrophil oxidative burst and phagocytosis [23], the mechanism of neutrophil NET formation and clearance following binge alcohol use remains unknown. We found that binge alcohol consumption in healthy subjects resulted in a significant increase in blood alcohol (Fig. 1A), serum endotoxins (LPS) (Fig. 1B) and bacterial 16s rDNA levels (Fig. 1C). To directly assess the effect of binge alcohol consumption on NET formation, neutrophils from healthy subjects were treated with ethanol (50 mM) and/or PMA stimulation ex vivo. Our neutrophil isolation method did not lead to cellular activation based on measurement of cellular ROS compared to positive activation control (Sup. Fig. 1). Alcohol-treated neutrophils exhibited increased release of extracellular DNA (Fig. 1D) and increased release of neutrophil elastase (Fig. 1E). PMA stimulation alone also induced both extracellular DNA (Fig. 1D) and neutrophil elastase release in neutrophils (Fig 1E). However, acute alcohol treatment of neutrophils prior to PMA stimulation resulted in significantly lower release of extracellular DNA (Fig. 1D) and released less NETs [NETosis assay] (Fig. 1E) by neutrophils compared to PMA only stimulation suggesting a suppressive or exhaustive effect of acute alcohol on NET formation.
Figure 1: Binge drinking induces increases in blood alcohol content and serum endotoxin but acute alcohol reduces ex vivo NET formation in human neutrophils.
Following binge drinking by healthy human subjects, blood samples were analyzed for alcohol content (A), serum endotoxin (B) and bacterial 16s rDNA (C). Ex vivo NETosis assay of neutrophils with and without acute alcohol exposure followed by PMA stimulation was assessed for extracellular DNA (D) and quantitative NETosis assay (E) methods, respectively. Samples are representative of 7–15 healthy subjects. A p < 0.05 compared to baseline no treatment control (*) or PMA with no alcohol treatment group (#) was considered statistically significant by analysis of variance. NS, not significant.
Alcohol binge inhibits LPS-induced neutrophil NET formation in the mouse liver
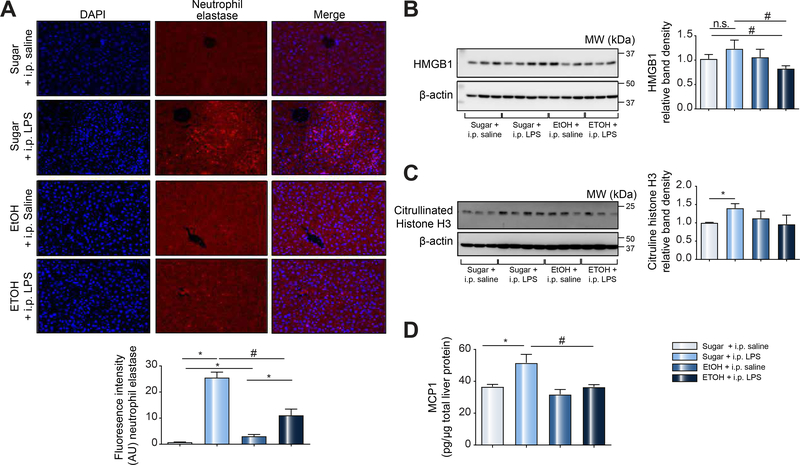
We sought to further characterize the effects of binge-alcohol on neutrophil NET formation using mouse disease models. To assess hepatic neutrophil infiltration and trap formation, mice received a single i.p. injection of LPS. Mice were then sacrificed 12, 15 and 20 h post LPS injections. We found significant neutrophil infiltration at 12 h post LPS injection in trap-like cluster formations in the hepatic parenchyma as demonstrated by H&E and neutrophil elastase immunohistology (Sup. Fig. 2). These neutrophil clusters were decreased by 15 h and were completely cleared by 20 h post LPS injections (Sup. Fig. 2). Based on the neutrophil infiltration kinetics in alcohol-naïve mice, we next assessed the effect of alcohol binge on hepatic neutrophil infiltration and cluster formation. We found that alcohol binge or LPS resulted in a significant increase in neutrophil elastase expression (indicative of hepatic neutrophil infiltration) in the liver compared to sugar controls (Fig. 2A). However, neutrophil recruitment to the liver in response to LPS was significantly lower in mice that received binge alcohol compared to sugar control (Fig. 2A). Further, binge alcohol treatment with and without LPS injection resulted in either no change or only a slight reduction in the hepatic levels of the NET activator, HMGB1 (Fig. 2B). However, binge alcohol significantly attenuated hepatic LPS-induced HMGB1 increase (Fig. 2B). Another marker of NET, citrullinated histone H3 protein levels, was significantly increased in control LPS-treated mice compared to sugar controls, but was not significantly changed in binge alcohol-treated mice (Fig. 2C). These observations suggest that binge alcohol suppressed LPS-induced hepatic NET formation in mice. Because an encounter with a pathogen by innate immune cells leads to an initial inflammatory response characterized by cytokine release [24], we assessed MCP-1 expression, an early marker of the inflammatory response. We found that while LPS induced a significant increase in MCP-1 protein expression, alcohol binge significantly reduced MCP-1 induction in the liver (Fig. 2D).
Figure 2: Reduced hepatic neutrophil infiltration and NET formation following acute alcohol use associated with sepsis.
C57BL/6 mice received sugar or ethanol gavage daily for three days followed by a single i.p. LPS injection (0.05mg/kg). Mice were sacrificed 12h after LPS injection and liver samples analyzed by immunohistology for neutrophil elastase (A). Liver samples from mice were additionally analyzed by western blotting for HMGB1 (B) and citrullinated histone H3 (C) using β-Actin as a protein loading control. ELISA was performed on total liver protein lysate for MCP1 (D). A p < 0.05 compared to baseline sugar gavage (*) or sugar plus i.p. LPS treatment (#) was considered statistically significant using ANOVA for 5–6 mice per experimental group. NS, not significant.
Alcohol binge significantly reduces hepatic NET clearance in LPS-induced sepsis in mice
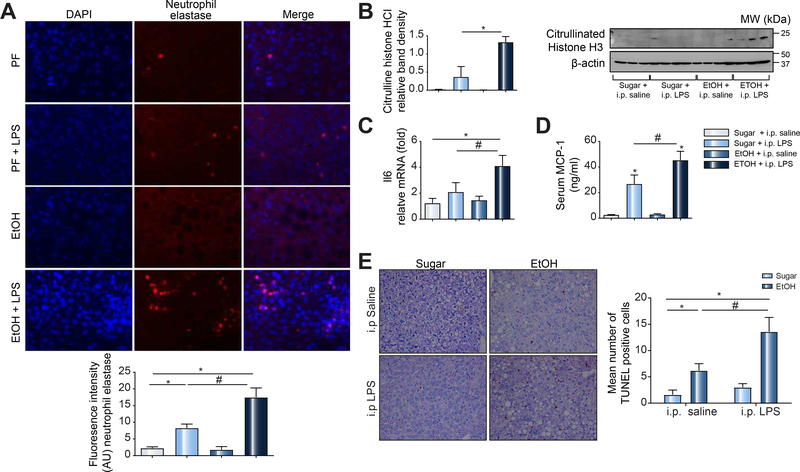
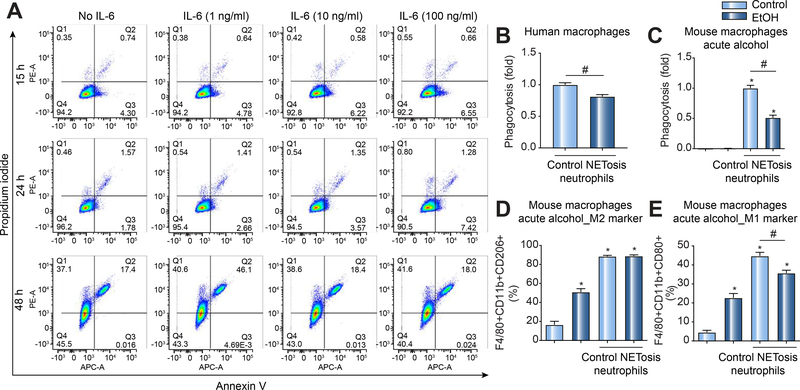
To assess the effects of binge alcohol consumption on MΦ clearance of NETosis neutrophils, mice received three alcohol binges followed by a single i.p. LPS injection. Based on our preliminary findings that showed neutrophil recruitment followed by neutrophil clearance 15 h after LPS-induced acute sepsis in mice (Sup. Fig. 2), we next tested the effects of alcohol 15 h after LPS challenge. We found that alcohol binge prior to LPS-induced sepsis resulted in sustained presence of hepatic neutrophils at 15 h post LPS injection (Fig. 3A). Western blot analysis revealed significantly higher citrullinated histone H3 expression in livers from mice with alcohol binge compared to control mice after LPS injection suggestive of reduced clearance of NETosis neutrophils (Fig. 3B). Further, in LPS-induced sepsis, mice with binge alcohol treatment had significantly higher levels of liver inflammation compared to control mice as demonstrated by increased IL-6 total liver mRNA expression and increased serum MCP-1 levels (Fig. 3C&D). In the liver, we found increased numbers of apoptotic cells after alcohol binge alone (Fig. 3E). Furthermore, sepsis in alcohol binge mice was associated with significant hepatocyte cell death compared to sepsis in alcohol-naïve mice (Fig. 3E). Given these observations, we surmised that persistent neutrophil hepatic survival which can be mediated by increased IL-6 [25] and or reduced NETosis neutrophil clearance of DAMPs could be responsible for worsening disease outcomes in the event of acute infection following binge drinking. To address the effect of IL6 on neutrophil viability, we carried out additional experiments. In-vitro, cultured neutrophils spontaneously undergo apoptosis over time[26, 27]. We found that dose dependent IL6 treatment in vitro did not impact cellular apoptosis of human neutrophils (Fig. 4A) similar to a previous report[28]. These observations suggest that IL6 might predominantly mediate hepatic neutrophil recruitment as previously reported[29].
Figure 3: Binge drinking associated with sepsis is characterized by the sustained hepatic presence of neutrophils, NETosis molecules, and increased cell death in mice.
C57BL/6 mice received three alcohol or sugar gavages with i.p. LPS or saline treatment. Mice were sacrificed after 15h and liver/serum samples analyzed by immunohistology for neutrophil elastase (A), western blot analysis for citrullinated histone H3 (B), RT-qPCR for IL-6 (C) and ELISA for MCP-1(D). TUNEL assay was performed on liver samples from C57BL/6 binge alcohol or sugar gavage mice with or without i.p. LPS or saline treatment as indicated (E). A p < 0.05 compared to baseline sugar gavage (*) or sugar plus i.p. LPS treatment (#) was considered statistically significant using ANOVA for 3–6 mice per experimental group.
Figure 4: IL6 does not impact neutrophil viability but alcohol use suppresses effective phagocytosis of NETotic neutrophils and promotes macrophage differentiation to a pro-inflammatory phenotype.
(A) Human blood neutrophils were treated with varying doses of IL6 and maintained in culture over 48h. Neutrophil apoptosis was assessed over time using flow cytometry as indicated. (B-E) Human primary macrophages and RAW 264.7 cells were acutely exposed to alcohol (50 mM, 4 h) and cocultured with CFSE labeled NETotic neutrophils (7:1 ratio of dead [NETotic] neutrophils to human macrophages or RAW 264.7) for 3 h or 48 h. Three hours after coculture, RAW 264.7 were harvested and analyzed by flow cytometry for MΦ phagocytosis (B & C). (D & E) Following 48h post co-culture, acute alcohol-exposed RAW 264.7 cells were analyzed by flow cytometry for MΦ differentiation to an M1 (D) versus M2 (E) phenotype. A p < 0.05 compared to baseline sugar gavage controls (*) or sugar plus LPS stimulation (#) was considered statistically significant using ANOVA for three independent repeat experiments.
Alcohol binge impairs clearance of NETosis neutrophils and attenuates efferocytosis by macrophages
Apoptotic cell death by neutrophils (NETosis) is an integral part of the immune response following NET formation to control microbial infection [30, 31]. To prevent further inflammation following NETosis, NETs (DAMPs) are cleared by macrophages (MΦ) in the process of efferocytosis, a vital process for maintaining tissue homeostasis [32]. MΦ clearance of neutrophils following NETosis can also lead to MΦ polarization into an M1 or M2 phenotype that can sustain or suppress inflammation, respectively [33]. It remains unclear, however, how acute alcohol use affects MΦ phagocytosis and polarization following clearance of NETosis DAMPs. To assess this gap in knowledge, we co-cultured NETosis neutrophils (Fig. 4B-E) with acute alcohol-exposed macrophages (Human monocyte-derived macrophages[34] and RAW 264.7 MΦ) to determine how acute alcohol modulated efferocytosis of neutrophils. We found that acute alcohol treatment significantly decreased phagocytosis of NETosis neutrophils by human (Fig. 4B) and mouse(Fig. 4C) MØ. We revealed that alcohol treatment alone of RAW 264.7 cells induced expression of both M1 (Fig. 4D) and M2 (Fig. 4E) MØ surface markers. Phagocytosis of NETosis neutrophils by MΦ in the absence of alcohol exposure increased both M1 and M2 surface markers on MØ (Fig. 4D&E). Strikingly, alcohol suppressed NETosis neutrophil-induced M1 markers, but did not suppress NETosis neutrophil-induced M2 markers following phagocytosis of NETosis neutrophils (Fig. 4D&E).
In vivo neutrophil depletion attenuates binge alcohol and/or LPS-induced liver damage
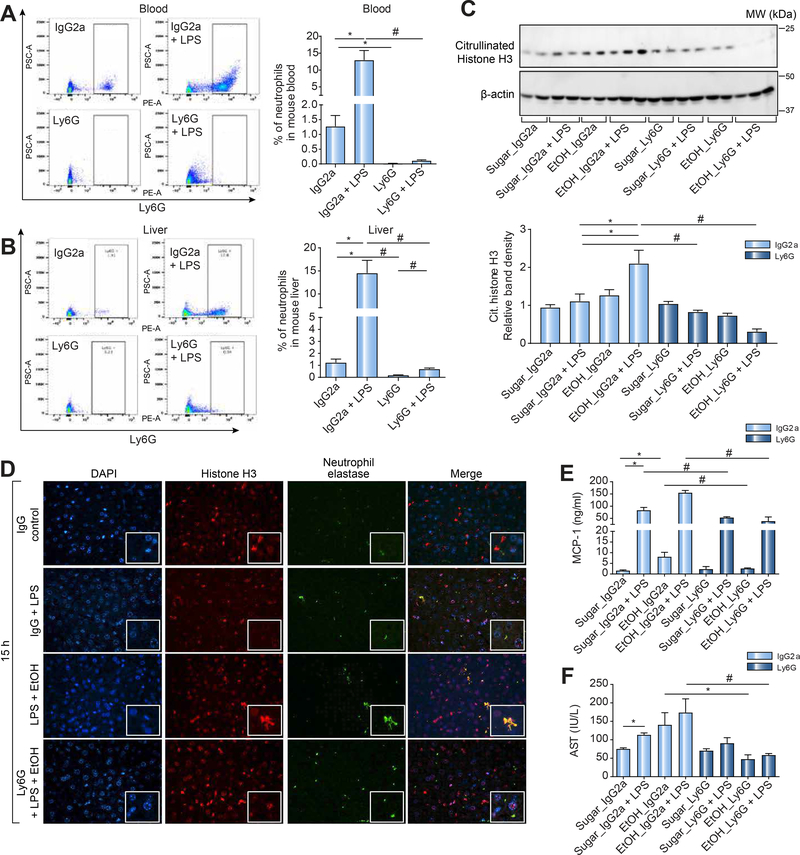
Altogether, previous studies and our novel observations suggest a role for neutrophils in the liver manifestation of sepsis and in ALD [10, 35]. Thus, we hypothesized that transient depletion of neutrophils might provide therapeutic benefits in alcohol-induced liver injury. To test this hypothesis, we first examined if a specific Ly6G antibody could deplete circulating and hepatic neutrophils in mice. We found that, compared to an IgG control antibody, the Ly6G antibody could specifically deplete circulating (Fig. 5A) and hepatic (Fig. 5B) neutrophils and this depletion prevented the LPS-induced increase in circulating (Fig. 5A) and hepatic (Fig. 5B) neutrophil numbers. The minimal increase in circulating and hepatic neutrophils after LPS challenge was suggestive of potential bone marrow recruitment.
Figure 5: Neutrophil depletion in mice alleviates binge alcohol-induced hepatic inflammation and injury.
Mice were untreated or pretreated with anti-mouse Ly6G or IgG control antibody followed by i.p. LPS injection as indicated. Blood (A) and liver (B) samples were collected after 15 h of LPS injection and analyzed for neutrophil content by flow cytometry. Mice were untreated or pretreated with anti-mouse Ly6G or IgG control antibody followed by three alcohol binges or sugar gavage as indicated. Some mice received additional i.p. LPS (0.05mg/kg) as indicated. Mice were sacrificed 15 h after i.p. LPS injection and serum/liver samples collected. Liver samples were analyzed by western blotting for citrullinated histone H3 and β-actin was used as a loading control (C). Liver samples were analyzed by fluorescent immunobiology for neutrophil elastase and Histone H3 as NET markers with dapi as nuclear stain. Serum samples collected from mice were analyzed for MCP-1 (D) and AST (E). A p < 0.05 compared to baseline sugar gavage IgG controls (*) or sugar IgG plus LPS stimulation (#) was considered statistically significant using ANOVA for 3–6 mice per experimental group.
Next, we assessed the in vivo therapeutic benefit of neutrophil depletion in the context of an alcohol binge. In the control antibody-treated mice 15 h post LPS injection, we found significantly increased levels of citrullinated histone H3 (NET component) in the alcohol binge plus LPS mice consistent with previous findings (Fig. 5C). These observations suggested possible reduced clearance of NETosis neutrophils or prolonged neutrophil persistence during AH in mice. Neutrophil depletion with the Ly6G antibody significantly reduced citrullinated histone H3 in both LPS alone and in binge alcohol plus LPS-treated mice compared to control antibody-treated mice (Fig. 5C). To determine the in-vivo presence of NETs and the effects of Ly6G neutrophil depletion, we performed fluorescent liver immunobiology probing for neutrophil elastase and histone H3. We found that binge-alcohol associated with sepsis resulted in persistent hepatic NET presence (Fig. 5D). In addition, we found that circulating neutrophil depletion using a Ly6G antibody in mice with alcohol binge plus LPS (AH) significantly attenuated systemic inflammation indicated by serum MCP-1 levels compared to controls (Fig. 5E). More importantly, neutrophil depletion prevented liver injury induced by LPS, alcohol binge or their combination as indicated by normal AST serum levels in alcohol or alcohol plus LPS challenged mice compared to IgG-control (Fig. 5F). These results suggested that neutrophil leukocyte depletion ameliorated liver injury and pro-inflammatory cytokine induction in the liver in LPS-induced sepsis with and without binge alcohol exposure.
Discussion
Neutrophils represent the first line of innate immune defense to clear or contain invading pathogens. In AH, characterized by increased serum endotoxins, an increase in hepatic neutrophils is associated with poor clinical outcomes [1, 22]. Mortality in AH is closely linked to the development of sepsis and multi-organ failure [36, 37]. Recent studies have shown that neutrophils can exacerbate hepatic injury in ischemia or surgical injury models [38, 39]. The capacity for neutrophils to cause or exacerbate liver injury was linked to their NET formation properties [38, 39]. While neutrophils have been implicated in AH pathogenesis, their NET formation properties and functions after alcohol binge and sepsis have not been assessed. Here, we show for the first time that acute-alcohol can induce spontaneous release of NETs by neutrophils. Subsequent stimulation or antigenic challenge of alcohol-exposed neutrophils however suppresses further NET formation and impairs efferocytosis (their clearance by MΦ) which can contribute to prolonged liver inflammation and injury.
Both ALD and binge drinking are associated with a significant increase in serum endotoxins (LPS) and DAMPs (HMGB1 and histones) due to compromised gut integrity and increased hepatocyte cell death, respectively [1, 2, 5, 19]. LPS and HMGB1 are important activators of neutrophil NET formation [39, 40]. Neutrophil NETs are composed of neutrophil elastase, citrullinated histones, and extracellular DNA. We found that ex vivo stimulation of acute alcohol-exposed neutrophils from healthy donors released significantly lower amounts of NETs compared to alcohol-naïve neutrophils. It is possible that this reduced NET production in addition to decreased migratory and phagocytic properties of neutrophils demonstrated in previous studies and our current study can significantly impair neutrophil functions during binge drinking associated with sepsis. Importantly, we found spontaneous NET formation by neutrophils following acute alcohol treatment without additional in vitro activation. Given that the serum milieu following binge drinking is enriched in LPS which can induce NET formation, it is possible that blood neutrophils are activated in the circulation in binge drinking and become ‘anergic’ to further stimulation. Alternatively, our observation that alcohol alone can induce spontaneous NET formation while simultaneously suppressing further NET formation in response to ex vivo stimulation suggest complex alcohol effects on NET formation. Strikingly, our in vivo mouse studies demonstrated that alcohol binge significantly reduced hepatic neutrophil infiltration in an LPS-induced sepsis model. These observations suggest that in addition to suppressing migration and phagocytosis of neutrophils [11], alcohol can also suppress NET formation properties of neutrophils.
To maintain organ homeostasis following suicide lytic NETosis [30], proper clearance of NETosis neutrophils by MΦs is indispensable [32, 41, 42]. Our novel data suggest that acute alcohol use could significantly impair the clearance of NETosis neutrophils by MΦs. Following phagocytosis of NETosis neutrophils, MΦs can polarize towards an M1 or M2 phenotype [42, 43]. We found that acute alcohol exposure of MΦs, in addition to their reduced phagocytosis of NETosis neutrophils, was associated with a significantly lower increase in MΦ polarization to an M1 phenotype. During initial phagocytosis, an M1 MΦ phenotype is necessary for the initial respiratory burst to clear bacteria, which we observed to be suppressed with acute alcohol use. Further, acute alcohol exposure of MΦs did not suppress their M2 phenotype, which increased significantly following phagocytosis of NETosis neutrophils similar to the alcohol-free condition. MΦs with an M2 phenotype decrease inflammation and encourage tissue repair, which was not diminished with binge alcohol use. These observations prompted us to speculate that alcohol use suppresses the initial phagocytic and respiratory burst functions of MΦs to metabolize arginine to the “killer” molecule nitric oxide needed for the initial killing of bacteria [44–46]. Additionally, binge alcohol can also suppress M2 MΦ polarization following NETosis neutrophil phagocytosis, which can hinder their ability to metabolize arginine to the “repair” molecule ornithine further exacerbating liver inflammation and injury [44, 46]. We confirmed with in vivo experiments that binge alcohol use associated with acute sepsis was associated with higher systemic inflammation and hepatocyte cell death.
In addition to serum endotoxins, NET components were increased in both the circulation and liver of AH patients and a mouse model of alcohol binge and can act as DAMPs to exacerbate liver injury [39]. Further, histones and HMGB1 contained in NETs may also play a role in additional hepatic recruitment and activation of neutrophils to form NETs thereby sustaining inflammation and injury [47] Our observations reveal that the acute sepsis in mice is associated with significantly increased hepatic HMGB1 protein plus NET formation. However, acute sepsis associated with binge alcohol is characterized by significantly lower hepatic HMGB1 and NET, suggesting a potential mechanistic role of HMGB1 in the disease process. However, we did not completely assess all NET inducers. As such HMGB1 might not be the only factor. Other NET proteins like neutrophil elastase, a serine protease, induce liver injury in rats following ischemic reperfusion injury [48]. Given that NETs have been associated with sterile hepatic injury [48], further investigations to determine if NETs directly trap damaged hepatocytes and increase inflammatory responses while impairing their clearance are needed.
Therapeutically, neutrophil depletion has been shown to provide beneficial effects during ischemia-reperfusion injury and during bacterial infections [49, 50]. Given the important role of neutrophils in the pathomechanism of AH, we assessed if transient neutrophil depletion could provide similar therapeutic benefits during binge drinking. We found that a Ly6G-specific mAb [51], could deplete both circulating and hepatic neutrophils in mice. Neutrophil depletion alleviated alcohol-induced liver injury during acute alcohol binge, LPS induced sepsis, or a combination of both as demonstrated by significantly reduced systemic inflammation and hepatic injury. Given that neutrophils form an important immune cell type in host defense, transient neutrophil depletion might be feasible as a therapeutic option for patients with AH.
In conclusion (Fig. 6), our study demonstrates that alcohol use impairs circulating and hepatic NET formation. Further, binge alcohol significantly suppresses the phagocytic clearance of NETosis DAMPs and favors polarization of MΦs to a pro-inflammatory phenotype. Our observations suggest that transient neutrophil depletion could become a promising new approach in the treatment of patients following binge drinking associated with sepsis.
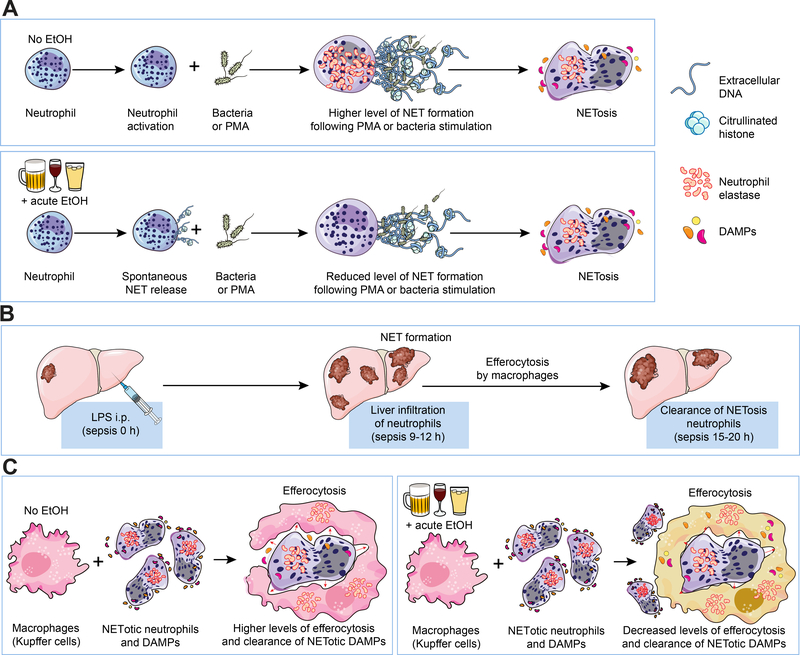
Figure 6: Illustrations summarizing our findings on the hepatic effects of alcohol on NET formation and clearance during binge-alcohol drinking associated with sepsis.
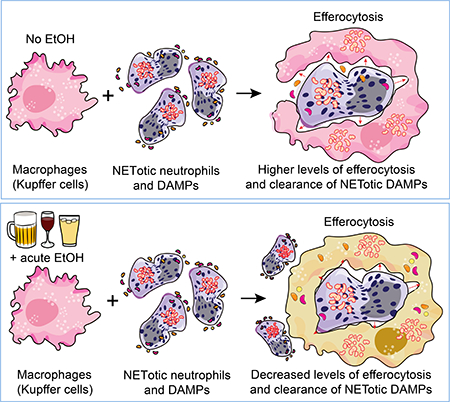
(A) Acute alcohol induces spontaneous NET formation by neutrophils. In the same cells subsequent PMA stimulation or antigenic challenge (LPS) results in significantly reduced NET formation compared to stimulations/activation of alcohol naïve neutrophils. (B) Acute sepsis in wild type C57BL/6 mice is associated with significant neutrophil hepatic infiltration and NET formation which occurs 9–12h post i.p LPS. Following NET formation, NETosis occurs followed by clearance of NETotic Neutrophils and NETotic DAMPs which occurs 15–20 post LPS injection in mice(C) Acute alcohol treatment of macrophages or binge alcohol use in mice significantly lowers the efferocytosis capacity of macrophages and clearance of NETotic neutrophils-DAMPs. Schematic illustrations made use of some motifolio templates (www.motifolio.com).
Supplementary Material
Acknowledgments
Financial Support: This work was supported by National Institutes of Health Grant R37 AA014372 (to G.S.).
Footnotes
Conflict of Interest: The authors declare that they have no competing interests.
References
- [1].Gao B, Bataller R. Alcoholic liver disease: pathogenesis and new therapeutic targets. Gastroenterology 2011;141:1572–1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Szabo G, Mandrekar P. Focus on: Alcohol and the liver. Alcohol Res Health 2010;33:87–96. [PMC free article] [PubMed] [Google Scholar]
- [3].Szabo G Gut-liver axis in alcoholic liver disease. Gastroenterology 2015;148:30–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Wang HJ, Gao B, Zakhari S, Nagy LE. Inflammation in alcoholic liver disease. Annual review of nutrition 2012;32:343–368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Gao B, Seki E, Brenner DA, Friedman S, Cohen JI, Nagy L, et al. Innate immunity in alcoholic liver disease. American journal of physiology Gastrointestinal and liver physiology 2011;300:G516–525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Szabo G, Petrasek J, Bala S. Innate immunity and alcoholic liver disease. Dig Dis 2012;30 Suppl 1:55–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Nagy LE. The Role of Innate Immunity in Alcoholic Liver Disease. Alcohol Res 2015;37:237–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Jaeschke H Neutrophil-mediated tissue injury in alcoholic hepatitis. Alcohol 2002;27:23–27. [DOI] [PubMed] [Google Scholar]
- [9].Mookerjee RP, Stadlbauer V, Lidder S, Wright GA, Hodges SJ, Davies NA, et al. Neutrophil dysfunction in alcoholic hepatitis superimposed on cirrhosis is reversible and predicts the outcome. Hepatology 2007;46:831–840. [DOI] [PubMed] [Google Scholar]
- [10].Bertola A, Park O, Gao B. Chronic plus binge ethanol feeding synergistically induces neutrophil infiltration and liver injury in mice: a critical role for E-selectin. Hepatology 2013;58:1814–1823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Xu R, Huang H, Zhang Z, Wang FS. The role of neutrophils in the development of liver diseases. Cellular & molecular immunology 2014;11:224–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Hirz T, Dumontet C. Neutrophil Isolation and Analysis to Determine their Role in Lymphoma Cell Sensitivity to Therapeutic Agents. J Vis Exp 2016:e53846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Tak T, Tesselaar K, Pillay J, Borghans JA, Koenderman L. What’s your age again? Determination of human neutrophil half-lives revisited. J Leukoc Biol 2013;94:595–601. [DOI] [PubMed] [Google Scholar]
- [14].Sabroe I, Prince LR, Jones EC, Horsburgh MJ, Foster SJ, Vogel SN, et al. Selective roles for Toll-like receptor (TLR)2 and TLR4 in the regulation of neutrophil activation and life span. J Immunol 2003;170:5268–5275. [DOI] [PubMed] [Google Scholar]
- [15].Navarini AA, Lang KS, Verschoor A, Recher M, Zinkernagel AS, Nizet V, et al. Innate immune-induced depletion of bone marrow neutrophils aggravates systemic bacterial infections. Proc Natl Acad Sci U S A 2009;106:7107–7112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Miyasaki KT, Wilson ME, Brunetti AJ, Genco RJ. Oxidative and nonoxidative killing of Actinobacillus actinomycetemcomitans by human neutrophils. Infect Immun 1986;53:154–160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Brinkmann V, Reichard U, Goosmann C, Fauler B, Uhlemann Y, Weiss DS, et al. Neutrophil extracellular traps kill bacteria. Science 2004;303:1532–1535. [DOI] [PubMed] [Google Scholar]
- [18].Bukong TN, Iracheta-Vellve A, Gyongyosi B, Ambade A, Catalano D, Kodys K, et al. Therapeutic Benefits of Spleen Tyrosine Kinase Inhibitor Administration on Binge Drinking-Induced Alcoholic Liver Injury, Steatosis, and Inflammation in Mice. Alcohol Clin Exp Res 2016;40:1524–1530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Bala S, Marcos M, Gattu A, Catalano D, Szabo G. Acute binge drinking increases serum endotoxin and bacterial DNA levels in healthy individuals. PLoS One 2014;9:e96864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].O’Brien JM Jr, Lu B, Ali NA, Martin GS, Aberegg SK, Marsh CB, et al. Alcohol dependence is independently associated with sepsis, septic shock, and hospital mortality among adult intensive care unit patients. Crit Care Med 2007;35:345–350. [DOI] [PubMed] [Google Scholar]
- [21].Wong F, Bernardi M, Balk R, Christman B, Moreau R, Garcia-Tsao G, et al. Sepsis in cirrhosis: report on the 7th meeting of the International Ascites Club. Gut 2005;54:718–725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Taylor NJ, Nishtala A, Manakkat Vijay GK, Abeles RD, Auzinger G, Bernal W, et al. Circulating neutrophil dysfunction in acute liver failure. Hepatology 2013;57:1142–1152. [DOI] [PubMed] [Google Scholar]
- [23].Stadlbauer V, Mookerjee RP, Hodges S, Wright GA, Davies NA, Jalan R. Effect of probiotic treatment on deranged neutrophil function and cytokine responses in patients with compensated alcoholic cirrhosis. J Hepatol 2008;48:945–951. [DOI] [PubMed] [Google Scholar]
- [24].Pullamsetti SS, Savai R, Janssen W, Dahal BK, Seeger W, Grimminger F, et al. Inflammation, immunological reaction and role of infection in pulmonary hypertension. Clin Microbiol Infect 2011;17:7–14. [DOI] [PubMed] [Google Scholar]
- [25].Biffl WL, Moore EE, Moore FA, Barnett CC Jr. Interleukin-6 delays neutrophil apoptosis via a mechanism involving platelet-activating factor. J Trauma 1996;40:575–578; discussion 578–579. [DOI] [PubMed] [Google Scholar]
- [26].Majewska E, Sulowska Z, Baj Z. Spontaneous apoptosis of neutrophils in whole blood and its relation to apoptosis gene proteins. Scand J Immunol 2000;52:496–501. [DOI] [PubMed] [Google Scholar]
- [27].Kirschnek S, Vier J, Gautam S, Frankenberg T, Rangelova S, Eitz-Ferrer P, et al. Molecular analysis of neutrophil spontaneous apoptosis reveals a strong role for the pro-apoptotic BH3-only protein Noxa. Cell Death Differ 2011;18:1805–1814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Wright HL, Cross AL, Edwards SW, Moots RJ. Effects of IL-6 and IL-6 blockade on neutrophil function in vitro and in vivo. Rheumatology (Oxford) 2014;53:1321–1331. [DOI] [PubMed] [Google Scholar]
- [29].Fielding CA, McLoughlin RM, McLeod L, Colmont CS, Najdovska M, Grail D, et al. IL-6 regulates neutrophil trafficking during acute inflammation via STAT3. J Immunol 2008;181:2189–2195. [DOI] [PubMed] [Google Scholar]
- [30].Yipp BG, Kubes P. NETosis: how vital is it? Blood 2013;122:2784–2794. [DOI] [PubMed] [Google Scholar]
- [31].Steinberg BE, Grinstein S. Unconventional roles of the NADPH oxidase: signaling, ion homeostasis, and cell death. Sci STKE 2007;2007:pe11. [DOI] [PubMed] [Google Scholar]
- [32].Farrera C, Fadeel B. Macrophage clearance of neutrophil extracellular traps is a silent process. Journal of immunology 2013;191:2647–2656. [DOI] [PubMed] [Google Scholar]
- [33].Wang M, You Q, Lor K, Chen F, Gao B, Ju C. Chronic alcohol ingestion modulates hepatic macrophage populations and functions in mice. J Leukoc Biol 2014;96:657–665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Bukong TN, Hall WW, Jacque JM. Lentivirus-associated MAPK/ERK2 phosphorylates EMD and regulates infectivity. J Gen Virol 2010;91:2381–2392. [DOI] [PubMed] [Google Scholar]
- [35].Agraz-Cibrian JM, Segura-Ortega JE, Delgado-Rizo V, Fafutis-Morris M. Alterations in neutrophil extracellular traps is associated with the degree of decompensation of liver cirrhosis. J Infect Dev Ctries 2016;10:512–517. [DOI] [PubMed] [Google Scholar]
- [36].Fukui H Relation of endotoxin, endotoxin binding proteins and macrophages to severe alcoholic liver injury and multiple organ failure. Alcohol Clin Exp Res 2005;29:172S–179S. [DOI] [PubMed] [Google Scholar]
- [37].Michelena J, Altamirano J, Abraldes JG, Affo S, Morales-Ibanez O, Sancho-Bru P, et al. Systemic inflammatory response and serum lipopolysaccharide levels predict multiple organ failure and death in alcoholic hepatitis. Hepatology 2015;62:762–772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Tohme S, Yazdani HO, Al-Khafaji AB, Chidi AP, Loughran P, Mowen K, et al. Neutrophil Extracellular Traps Promote the Development and Progression of Liver Metastases after Surgical Stress. Cancer research 2016;76:1367–1380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Huang H, Tohme S, Al-Khafaji AB, Tai S, Loughran P, Chen L, et al. Damage-associated molecular pattern-activated neutrophil extracellular trap exacerbates sterile inflammatory liver injury. Hepatology 2015;62:600–614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Tadie JM, Bae HB, Jiang S, Park DW, Bell CP, Yang H, et al. HMGB1 promotes neutrophil extracellular trap formation through interactions with Toll-like receptor 4. American journal of physiology Lung cellular and molecular physiology 2013;304:L342–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Kaplan MJ, Radic M. Neutrophil extracellular traps: double-edged swords of innate immunity. Journal of immunology 2012;189:2689–2695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Nakazawa D, Shida H, Kusunoki Y, Miyoshi A, Nishio S, Tomaru U, et al. The responses of macrophages in interaction with neutrophils that undergo NETosis. J Autoimmun 2016;67:19–28. [DOI] [PubMed] [Google Scholar]
- [43].Martinez FO, Gordon S. The M1 and M2 paradigm of macrophage activation: time for reassessment. F1000Prime Rep 2014;6:13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Mills CD. Anatomy of a discovery: m1 and m2 macrophages. Front Immunol 2015;6:212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Thomas AC, Mattila JT. “Of mice and men”: arginine metabolism in macrophages. Front Immunol 2014;5:479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Rath M, Muller I, Kropf P, Closs EI, Munder M. Metabolism via Arginase or Nitric Oxide Synthase: Two Competing Arginine Pathways in Macrophages. Front Immunol 2014;5:532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Pittman K, Kubes P. Damage-associated molecular patterns control neutrophil recruitment. J Innate Immun 2013;5:315–323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Uchida Y, Freitas MC, Zhao D, Busuttil RW, Kupiec-Weglinski JW. The inhibition of neutrophil elastase ameliorates mouse liver damage due to ischemia and reperfusion. Liver Transpl 2009;15:939–947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].Carr KD, Sieve AN, Indramohan M, Break TJ, Lee S, Berg RE. Specific depletion reveals a novel role for neutrophil-mediated protection in the liver during Listeria monocytogenes infection. Eur J Immunol 2011;41:2666–2676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Martinez-Mier G, Toledo-Pereyra LH, McDuffie JE, Warner RL, Ward PA. Neutrophil depletion and chemokine response after liver ischemia and reperfusion. J Invest Surg 2001;14:99–107. [DOI] [PubMed] [Google Scholar]
- [51].Daley JM, Thomay AA, Connolly MD, Reichner JS, Albina JE. Use of Ly6G-specific monoclonal antibody to deplete neutrophils in mice. J Leukoc Biol 2008;83:64–70. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.