Abstract
Introduction
Past research has shown relationships between stress during pregnancy, and related psychosocial health measures such as anxiety and depressive symptoms, with infant, child, and adult outcomes. However, most research is from high-income countries. We conducted a scoping review to identify research studies on prenatal stress and outcomes of the pregnancy or offspring in low- and middle-income countries (LMICs), and to synthesize the stress measures and outcomes assessed, the findings observed, and directions for future research.
Methods
We searched PubMed, Scopus, and PsycINFO for English-language abstracts published from Jan 1960-Jan 2017. Search terms were related to stress and psychosocial health; pregnancy; infant or child development; and LMICs.
Results
48 articles were identified. Sixty percent of studies were in upper-middle, 25% in lower-middle, and 15% in low income countries. Most studies used questionnaires, either existing or tailor-made, to assess stress. Eight assessed cortisol. Most studies (n = 31) assessed infant outcomes at birth, particularly gestational age or preterm birth (n = 22, 12 showing significant relationships), and birthweight (n = 21, 14 showing significant relationships). Five studies analyzed outcomes later in infancy such as temperament and motor development, all showing significant results; and nine in childhood such as behavioral development, asthma, and physical growth, with eight showing significant results.
Conclusions
Results highlight the importance of prenatal stress on infant and child outcomes in LMICs. Methods used in high-income countries are successfully employed in LMICs, but tailored tools remain necessary. Careful assessment of covariates is needed to foster analyses of interactive effects and pathways. Studies including longer-term follow-up should be prioritized.
Introduction
Research in the Developmental Origins of Health and Disease has highlighted effects of maternal stress during pregnancy on long-term health outcomes among offspring [1–6]. This reflects in part the effects of maternal stress hormones such as cortisol, which is important in promoting fetal neurodevelopment but which, at high levels, might have adverse effects that vary by infant sex and the timing of exposure during gestation [7–10]. Furthermore, prenatal stress is associated with epigenetic changes in the placenta and fetus that affect both early and later development [11]. Human studies have shown relationships between prenatal stress and birth outcomes, such as low birthweight and preterm birth [1, 10, 12]. Prenatal stress also predicts later cognitive, behavioral, and psychomotor development [10, 13, 14], linguistic development [5], immune function [5], and metabolic outcomes in childhood and persisting throughout life [2].
Health researchers and international organizations such as the World Health Organization often distinguish between high-income countries and low and middle-income countries (LMICs), based on differences in economic indicators that also capture differences in common health risks and burdens [15, 16]. Countries falling below thresholds defined by the World Bank [17] (detailed below) are classified as low, lower-middle, or upper-middle income countries and are together referred to as LMICs. The prenatal stress reviews cited above [1–14] synthesize evidence primarily from industrialized or high-income countries, with little information on developing countries or LMICs. This reflects the fewer number of studies in LMICs [1, 3, 18], difficulty comparing studies because of varied methods [18], and the likelihood of different relationships between prenatal stress and developmental outcomes in LMICs compared to high-income countries, which complicates the synthesis of results [6].
Reviews that do include LMICs highlight the importance of maternal psychosocial health on child developmental outcomes, but also the need for a broader review. For example, one review summarized relationships between perinatal distress and preterm birth in LMICs, with an emphasis on allostatic load [18]. The review provides an excellent overview of allostatic load as a stress indicator, but the focused search yielded only three studies from LMICs. Another review synthesized research on perinatal mental disorders and child outcomes [19], but focused on disorders (anxiety, depression) and did not include search terms for stress. Furthermore, the seven studies from LMICs all focused on postnatal rather than prenatal exposure [19]. Finally, a review of depression during pregnancy [20] identified several studies from LMICs. Whereas some of these also included maternal stress, the aim of the review was to synthesize research specifically on depression, and thus a scoping review of prenatal stress remains necessary.
Although there is wide variability among countries, developing countries and LMICs share many similar challenges in healthcare funding [15] and provision of services [16], as well as maternal and infant health burdens such as low birthweight and prematurity. For example, prevalence of low birthweight is 18.6% in least developed countries and 16.5% in less developed, compared to only 7.0% in more developed countries [21]. Given these persistent disparities in outcomes associated with prenatal stress, there is a growing interest in incorporating prenatal stress into global health research [22] and better integrating maternal psychosocial health into prenatal care in LMICs [23]. A review that synthesizes the literature more broadly could highlight existing datasets, commonly assessed stress and outcome measures, research challenges, and gaps in knowledge to guide future research. Thus, our objective was to review research in LMICs on stress and related maternal psychosocial health measures during pregnancy, and child developmental outcomes, in order to summarize the stress measures and outcomes assessed, the general relationships observed, and priorities for future research.
Materials and methods
The search strategy targeted studies assessing relationships between stress and related psychosocial health outcomes during pregnancy, and any child developmental outcome. S1 Table shows the full search strategy. We searched PubMed, Scopus, and PsycINFO for English-language abstracts published between Jan 1, 1960, and Jan 31, 2017. Search terms included those related to stress, distress, stressful life events, and psychosocial health; pregnancy; infant or child development; and developing countries or LMICs, including countries classified by the World Bank [17] in 2017 as low income (Gross National Income per capita<$1,025 USD), lower-middle income ($1,026–$4,035), or upper-middle income ($4,036–$12,475). The full list of country classifications is available in S1 Table. Reviewed articles were limited to English. Articles were excluded if they did not include a stress or psychosocial health measure, did not assess outcomes of the pregnancy or offspring, were not in LMICs, presented animal models, or focused on in vitro fertilization.
S1 Appendix presents the PRISMA checklist for the current study.
Results
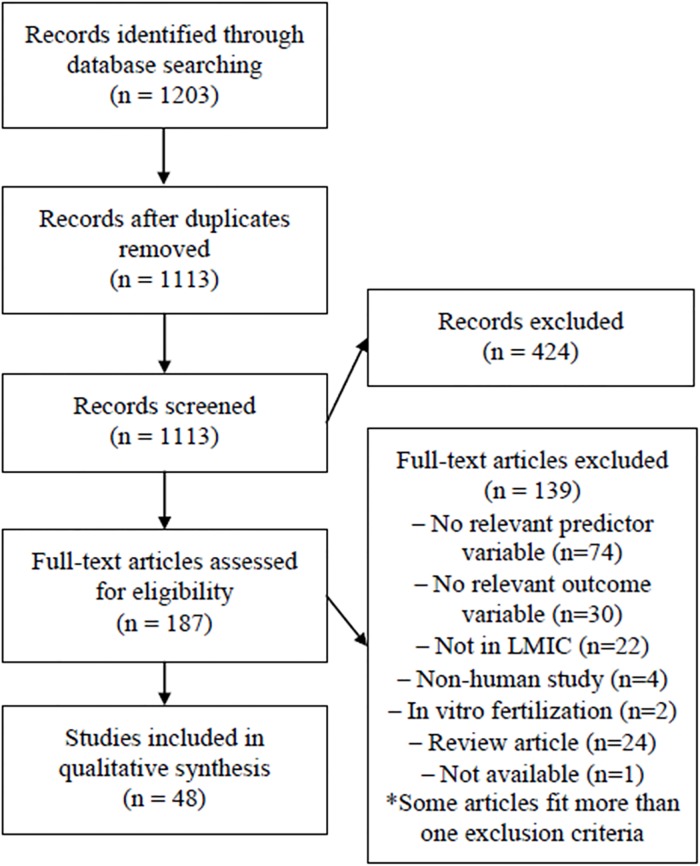
The search strategy yielded 1113 abstracts after removal of duplicates (Fig 1). Initial screening of descriptors resulted in exclusion of 424 abstracts. Review for keywords in titles and abstracts yielded 187 articles for complete review (Fig 1). One article deemed likely pertinent [24] was not available despite attempts to contact the journal and authors. Thus, 186 articles were reviewed in detail by at least one author and discussed by at least two to establish consensus of pertinence. Articles were excluded if they did not include a stress or psychosocial health measure (n = 74). This was the case for many studies of stress following war (n = 21) and natural disasters (n = 10) in which, without a measure of psychosocial health, it is not possible to isolate the effects of stress from those of other factors such as infectious diseases or undernutrition that might be more prevalent during such events and that might also affect child development. Other studies were excluded because they did not assess outcomes of the pregnancy or offspring (n = 30); they were not in LMICs (n = 22); they presented animal models (n = 4); or they focused on in vitro fertilization (n = 2), which was deemed not pertinent to the current review. Some articles fit more than one exclusion category. Finally, 24 reviews were excluded.
Fig 1. Flow diagram of search strategy and article selection.
The review process highlighted the use of questionnaires to assess common mental disorders, such as anxiety or depression, as a construct of stress and psychosocial health more generally. This reflects the difficulty of distinguishing between constructs that share many similar underlying mechanisms of influence, which might be particularly challenging in the context of LMICs lacking detailed and validated tools to evaluate stress specifically. We thus chose to retain articles that used measures of common mental disorders as a generalized conceptualization of stress or distress. However, we group results by the measure employed, for distinction.
Forty-eight articles were retained, and were read and summarized by at least two authors. Table 1 summarizes the country in which each study was conducted, measures of stress examined, outcomes, and results. S2 Table includes these information as well as detailed descriptions of the sample, data collection, analyses, and discussion points highlighting strengths and limitations. S2 Appendix presents methodological quality assessment for individual studies, based on the Quality Assessment Tool for Quantitative Studies developed by the Effective Public Health Practice Project.
Table 1. Summary of studies of prenatal stress and pregnancy and child health outcomes in LMICs.
| Article | Country | Sample | Stress measure | Outcome measures | Results |
|---|---|---|---|---|---|
| Abeysena et al. 2010 (35) | Sri Lanka (South Asia, LM income) | 737 pregnant women | Modified Life Events Inv. (MLEI), General Health Quest. (GHQ) | PTB | GHQ and MLEI scores were not significant predictors of PTB. Multivariate analyses indicated a trend for exposure to MLEIs during the 2ndT and PTB (p = 0.09, OR 1.80). |
| Abramson et al. 1961 (62) | South Africa (Sub-Saharan Africa, UM income) | 101 pregnant women | Interview: Feelings about pregnancy, life events, family relationships | Neuromotor development 3, 32, and 93 days after delivery | Excluding babies with BW>8.0 pounds, 42.5% in the high-stress group had motor scores <3, compared to 15.4% in the low-stress group. Results were similar at the 2nd but not 3rd evaluation. |
| Arffin et al. 2012 (56) | Malaysia (East Asia & Pacific, UM income) | 33 pregnant women with history of miscarriage and PTB (n = 20) or controls (n = 13) | Salivary cortisol | History of miscarriages, PTB | Mean salivary cortisol levels did not differ between the test (1.016±SEM 0.182 lg/ml) and control (0.978±0.298 lg/ml) groups (p = 0.392). |
| Arteaga-Guerra et al. 2010 (29) | Colombia (Latin Am. & Caribbean, UM income) | 46 perinatal women | Perceived Stress Scale | PTB, LBW, PTB & LBW combined (PLBW) | Perceived stress was not associated with PTB or LBW. The combination of periodontitis but no elevated stress predicted PLBW (OR = 10.3, CI = 1.1, 93.2, p = 0.01). |
| Baig et al. 2013 (43) | Pakistan (South Asia, LM income) | 600 women with preterm (n = 300) or term (n = 300) births | Edinburgh Postnatal Depression Scale | PTB | Prevalence of emotional stress was 69% among cases and 55% among controls (p<0.01). Risk factors for PTB included maternal weight <50 kg, periodontal diseases, low haemoglobin, history of PTB, and poor nutritional status. |
| Barrios et al. 2014 (31) | Peru (Latin Am. & Caribbean, UM income) | 959 women with preterm (n = 479) or term (n = 480) births | Interview: Life events | PTB | PTB was associated with severe life events including death of a relative (OR = 2.10, CI = 1.38, 3.20), divorce/separation (OR = 2.09, CI = 1.10, 4.00), financial troubles (OR = 2.70, CI = 1.85, 3.94), and fights with partner (OR = 2.40, CI = 1.78, 3.17). Risk increased with # of life events. |
| Başgül et al. 2011 (69) | Turkey (Europe & Central Asia, UM income) | 309 children age 3–5 yrs: 204 controls, 105 with psychiatric complaints | Investigator-prepared form: Stressors experienced by the mother | Early Childhood Inventory-4 Parent Form, DSM-IV interviews | No differences in stress or other predictor variables were found between children with psychiatric conditions and those without. |
| Bhat et al. 2015 (59) | India (South Asia, LM income) | 100 pregnant women | General Health Quest. (GHQ-28) | Early Infancy Temperament Questionnaire at 1–4 months of age | GHQ scores were not correlated with temperament. Mean adaptability and approach scores were higher among infants of mothers with GHQ<7 (3.11 and 2.70) compared to those with GHQ≥7 (2.67, 2.06) (p = 0.03, p = 0.05). Other temperament dimensions did not differ between groups. |
| Bindt et al. 2013 (40) | Ghana & Cote d’Ivoire (Sub-Saharan Africa, LM income) | 719 pregnant women | Patient Health Quest., Generalized Anxiety Disorder Scale | BW, LBW, head circumference, GA, PTB, Apgar score | Anxiety and depression scores were weakly correlated with Apgar scores (r = -0.106 and -0.102, respectively) but not with BW or GA. Depression and anxiety were not predictive of BW or PTB. |
| Brittain et al. 2015 (41) | South Africa (Sub-Saharan Africa, UM income) | 726 pregnant women | Beck Depression Inv., World Mental Health Life Events Quest. | BW, HC, WAZ, HCAZ, SGA | No associations were observed between antenatal depression and PTB. Antenatal depression predicted smaller WAZ (OR = 0.2, CI = 0.02, 0.4) and HCAZ (OR = 0.3, CI = 0.1, 0.6). Relationships between depression and WAZ did not persist when controlling for stressful life events. |
| Cerón-Mireles et al. 1996 (44) | Mexico (Latin Am. & Caribbean, UM income) | 2623 perinatal women | Karasek’s Job Content Quest. | GA, PTB, BW, SGA | Conflicts at work predicted SGA only among women who delivered at the public assistance hospital, which typically sees the poorest women (OR = 4.93, CI = 2.09, 11.66). |
| Chen et al. 2000 (53) | China (East Asia & Pacific, UM income) | 792 pregnant women exposed to benzene (n = 354) or not exposed (n = 438) | Perceived work stress: single questionnaire item | BW | Adjusted mean BW was 3445g among the group with neither work stress nor benzene exposure, 3426g with work stress alone, 3430g with benzene exposure alone, and 3262g with both exposures. |
| Christian et al. 2016 (36) | Nepal (South Asia, low income) | 737 pregnant women assigned to one of 5 supplement groups | Serum cortisol | GA, LBW, PTB | Cortisol predicted PTB (OR = 1.04, CI = 1.00, 1.08), but not BW (β = 4.2, p = 0.17) or LBW (OR = 0.98, CI = 0.95, 1.01). |
| Fan et al. 2016 (70) | China (East Asia & Pacific, UM income) | 216 pregnant women | Hamilton Anxiety Scale, Hamilton Rating Scale for Depression | Resting blood pressure (BP) and heart rate (HR) at age 7–9 years | Maternal anxiety predicted all child HR and BP measurements in multivariate analyses. Maternal depression predicted children’s resting HR, stress systolic BP, and recovery systolic and diastolic BP. |
| Frith et al. 2015 (37) | Bangladesh (South Asia, LM income) | 1041 women assigned to “early” or “usual start” supplementation during pregnancy | Salivary cortisol | BW, birth length (BL), head circumference (HC), GA | Male (but not female) infants of mothers with higher cortisol had smaller BW, HC, and GA. In general linear models, relationships between cortisol and BW and HC differed by supplementation group. In the “usual start” group, greater cortisol predicted smaller values. In the “early start” group, cortisol did not predict outcomes. |
| Hanlon et al. 2009 (48) | Ethiopia (Sub-Saharan Africa, low income) | 1065 pregnant women, 521 singleton infants | Self-Reporting Quest. (SRQ-20) of common mental disorders (CMDs), List of Threatening Experiences Quest. | BW, stillbirth, neonatal mortality, prolonged labor, time to initiation of breast-feeding | There were no associations between BW, stillbirth, or neonatal death and CMD symptoms, stressful life events, or worry in pregnancy. Prolonged labor was associated with CMD symptoms (Low, RR = 1.4, CI = 1.0, 1.9; High, RR = 1.6, CI = 1.0, 2.6) and worry about the delivery (RR = 1.5, CI = 1.0, 2.1). Analyses did not support a linear effect of SRQ score. |
| Isaksson et al. 2015 (64) | Nicaragua (Latin Am. & Caribbean, LM income) | 147 pregnant women, 70 children | Salivary cortisol, Self- Reporting Quest. (SRQ-20) | Child’s psychiatric symptoms at age 9 years (CBCL), child’s salivary cortisol | Morning cortisol during pregnancy was associated with total CBCL scores (r = 0.31, p = 0.009), and with internalizing (r = 0.28, p = 0.020) and externalizing symptoms (r = 0.35, p = 0.003). Cortisol during pregnancy did not correlate with children’s cortisol at age 9. SRQ was not associated with CBCL scores or children’s cortisol. |
| Karamoozian & Askarizaden 2015 (54) | Iran (Middle East & North Africa, UM income) | 29 pregnant women with anxiety or depression assigned to stress management intervention (n = 14) or controls (n = 15) | Edinburgh Postnatal Depression Scale, Pregnancy-Related Anxiety Quest. | Apgar scores | Mean one-minute Apgar scores were higher in the experimental (8.93) than control (8.07) group (p = 0.01). Similar results were observed at 5 minutes (9.71 and 9.27, respectively; p = 0.05). |
| Kertes et al. 2016 (57) | Democratic Republic of Congo (Sub-Saharan Africa, low income) | 24 mother-newborn dyads | Semi-structured ethnographic interviews: Stressful events during pregnancy | Methylation of CRH, CRHBP, NR3C1, and FKBP5, from maternal venous blood, placenta, and umbilical cord blood | 18 CpG sites were associated with either chronic stress (n = 11), war trauma (n = 14), or both (n = 6). Correcting for multiple testing, 8 CpG sites remained, all associated with war trauma. Methylation levels at four CpG sites, situated at transcription factor binding sites in NR3C1 and CRH, collectively explained 55% of the variance in BW. |
| Koen et al. 2016 (45) | South Africa (Sub-Saharan Africa, UM income) | 544 pregnant women | Childhood Trauma Quest., Mini Int. Neuropsychiatric Interview (MINI) to assess PTSD | WAZ, HCAZ, SGA, PTB | PTSD did not predict WAZ, HCAZ, SGA, or PTB. Lifetime trauma exposure (MINI) predicted a 0.3 unit reduction in HCAZ (p = 0.026). Trauma did not predict most outcomes. |
| Meghea et al. 2014 (25) | Romania (Europe & Central Asia, UM income) | 474 pregnant women | Perceived Stress Scale (PSS-4) | BW, GA, SGA, PTB | High stress predicted a 113g reduction in BW (CI = -213, -11), and predicted PTB (OR = 2.81, CI = 1.17, 6.76). |
| Mirabzadeh et al. 2013 (34) | Iran (Middle East & North Africa, UM income) | 550 pregnant women | Depression Anxiety Stress Scales (DASS-21), Stressful Life Events Quest. | PTB | GA was negatively correlated with depression, anxiety, stress, and DASS scores. Path analysis showed direct relationships among DASS and GA (β = -0.18). |
| Mulligan et al. 2012 (58) | Democratic Republic of Congo (Sub-Saharan Africa, low income) | 25 mother-newborn dyads | Ethnographic interviews: deprivation, “mundane stressors”, war stressors; Peritraumatic Distress Inv. | Methylation of NR3C1 promoter, from maternal venous and umbilical cord blood; BW | BW was correlated with maternal deprivation (r = -0.484), mundane stress (r = -0.521), and war stress (r = -0.620). War stress was correlated with cord blood methylation levels of the NR3C1 gene (r = 0.586) and BW (r = -0.449). Relationships between war stress and maternal methylation, and between maternal methylation and BW, were not significant. |
| Nasiri et al. 2010 (38) | Iran (Middle East & North Africa, UM income) | 600 pregnant women | Spielberger State-Trait Anxiety Inv. | PTB, LBW | State anxiety score ≥45 was associated with PTB (RR = 3.1, CI = 2.1, 4.7) and LBW (RR = 2.6, CI = 1.6, 4.2). |
| Nasreen et al. 2010 (49) | Bangladesh (South Asia, LM income) | 583 pregnant and postpartum women | Edinburgh Postnatal Depressive Scale, State Trait Anxiety Inv. | LBW | Depressive symptoms predicted LBW (OR = 2.24, CI = 1.37, 3.68). Results were similar for anxiety (OR = 2.08, CI = 1.30, 3.25). |
| Nepomnaschy et al. 2006 (55) | Guatemala (Latin Am. & Caribbean, LM income) | 61 women of reproductive age | Urinary cortisol | Pregnancy: successful or unsuccessful | Mean cortisol levels were higher in unsuccessful than in successful pregnancies. Increased cortisol predicted pregnancy loss (OR = 2.7, CI = 1.2, 6.2). Unsuccessful pregnancies presented a larger proportion of cortisol peaks than successful ones. |
| Pires et al. 2013 (65) | Brazil (Latin Am. & Caribbean, UM income) | 370 children ages 6–13 years | Questionnaire: Mothers were asked “whether the pregnancy was a peaceful time or marked by discord and arguments” | CBCL Teacher Report Form of ADHD symptoms | Discord during pregnancy predicted mother-reported ADHD (OR = 4.54, CI = 2.16, 9.57), but not teacher reported ADHD. Other predictors for mother-reported ADHD included family functioning, social support, and life events in the past year. |
| Qiao et al. 2012 (42) | China (East Asia & Pacific, UM income) | 463 pregnant women | Hospital Anxiety and Depression Scale | Obstetric outcomes | Prevalence of prolonged pregnancy was higher in the Symptom (n = 3, 8.3%) than in the Symptomless group (n = 5, 1.6%) (RR = 4.08, CI = 1.25, 13.33). Other obstetric and neonatal outcomes did not differ among groups. |
| Qu et al. 2016 (46) | China (East Asia & Pacific, UM income) | 2189 pregnant women with PTB (n = 130) or term births (n = 2059) | Revised Pregnancy Stress Rating Scale | PTB | High pregnancy specific stress predicted PTB (RR = 2.92, CI = 1.12, 7.58). Low and medium levels of pregnancy-specific stress were not significant predictors of PTB. |
| Ramchandani et al. 2010 (67) | South Africa (Sub-Saharan Africa, UM income) | 953 pregnant women | Interviews: Marital, family, economic, and societal stress and violence | Richman Behaviour Screening Questionnaire at ages 2 and 4 | Child behavior scores at age 4 were higher among children in the high (5.4) compared to low (4.4) prenatal stress group. Prenatal stress predicted behavioral problems at age 4 (OR = 2.66, CI = 1.28, 5.54). |
| Rondó et al. 2003 (27) | Brazil (Latin Am. & Caribbean, UM income) | 865 pregnant women | Perceived Stress Scale (PSS), General Health Quest. (GHQ), State Trait Anxiety Inv. (STAI) | LBW, PTB, intra-uterine growth restriction (IUGR) | GHQ >3 in the 2nd interview predicted LBW (RR = 1.97, CI = 1.12, 3.47), and GHQ >3 in the 3rd interview predicted PTB (RR = 2.32, CI = 1.18, 4.60). Maternal psychosocial health did not predict IUGR. STAI and PSS scores did not predict any outcomes. |
| Rondó et al. 2013 (71) | Brazil (Latin Am. & Caribbean, UM income) | 409 pregnant women and their children ages 5–8 years | Perceived Stress Scale (PSS), General Health Quest. (GHQ), State Trait Anxiety Inv. | BMI Z-scores at ages 5–8 years | PSS scores 5–8 years postpartum (β = -0.04) and 2ndT GHQ scores (β = -0.09) predicted children’s BMIZ scores. Thus, greater maternal stress and distress both during pregnancy and postpartum predicted lower child BMIZ scores. |
| Rosa et al. 2016 (72) | Mexico (Latin Am. & Caribbean, UM income) | 417 pregnant women | Crisis in Family Systems Revised survey of negative life events (NLEs) | Asthma, wheeze | NLEs predicted risk of ever wheeze (RR = 1.08, CI = 1.00, 1.16) and wheeze in the past 12 months (RR = 1.12, CI = 1.00, 1.26). |
| Ross et al. 2011 (63) | Ethiopia (Sub-Saharan Africa, low income) | 954 perinatal women | Self-Reporting Quest. of common mental disorders (CMDs) | Infant illness episodes since birth | Persistent CMD symptoms predicted infant diarrhoea (RR = 2.15, CI = 1.39, 3.34). Univariate analyses showed relationships between persistent CMD and acute respiratory infection (crude RR = 2.24, CI = 1.52, 3.30) and fever (crude RR = 1.61, CI = 1.10, 2.35), but results did not persist in multivariate analyses. |
| Rothberg et al. 1991 (47) | South Africa (Sub-Saharan Africa, UM income) | 1197 perinatal women, delivering at Johannesburg or Baragwanath Hospital | Social Readjustment Rating Scale (SRRS) of life events | BW | Among the Johannesburg group, greater SRRS predicted BW. SRRS scores did not predict BW in the Baragwanath group. Maternal perception of stress and psychosocial support did not predict BW. |
| Ruwanpathirana & Fernando 2014 (50) | Sri Lanka (South Asia, LM income) | 835 pregnant women and their infants: 167 SGA, 668 controls | General Health Quest. (GHQ-30) | SGA | High 2ndT stress levels predicted SGA in univariate (OR = 2.17, CI = 1.43, 3.30) and multivariate (OR = 1.92, CI = 1.17, 3.14) analyses. |
| Sanchez et al. 2013 (39) | Peru (Latin Am. & Caribbean, UM income) | 959 pregnant women: 479 with PTB, 480 with term births | Patient Health Quest. (PHQ-9), Depression Anxiety Stress Scales (DASS-21) | PTB | Predictors of PTB included PHQ-9 Depression scores (OR for moderate-severe group = 3.67, CI = 2.09, 6.46), and DASS-21 Depression (OR = 2.90, CI = 1.66, 5.04), Anxiety (OR = 2.76, CI = 1.83, 4.16), and Stress (OR = 11.07, CI = 5.64, 21.71) scores. |
| Sanguanklin et al. 2014 (52) | Thailand (East Asia & Pacific, UM income) | 175 pregnant women | Center for Epidem. Studies Depression Scale | BW, LBW, PTB | Depression had no main effects on BW. |
| Santos et al. 2014 (68) | Brazil (Latin Am. & Caribbean, UM income) | 4231 perinatal women (2004 Pelotas Birth Cohort) | Perinatal interview: “During pregnancy, did you feel depressed or have any nervous condition?” | Dev. & Well-Being Assessment questionnaire (psychiatric disorders) at age 6 | Mood symptoms during pregnancy predicted psychiatric disorders among children (OR = 1.97, CI = 1.60, 2.41). Results were similar for mood symptoms at 3 months postpartum (OR = 2.29 CI = 1.86, 2.81). |
| Sasaluxnanon & Kaewpornsawan 2005 (66) | Thailand (East Asia & Pacific, UM income) | 241 children age 6–12: 122 with ADHD, 119 controls | Questionnaire: “Emotional distress” during pregnancy, reported as “yes” or “no”. | Attention Deficit Hyperactivity Disorder (ADHD) | Emotional distress predicted ADHD in univariate (OR = 4.49, CI = 2.37, 8.45) and multivariate analyses (OR = 2.99, CI = 1.43, 5.40). |
| Shaikh et al. 2011 (30) | Pakistan (South Asia, LM income) | 125 pregnant women | A-Z stress scale, Center for Epidem. Studies Depression scale, serum cortisol | PTB | There were no relationships between PTB and cortisol (OR per 10-point increase in cortisol = 0.78, p = 0.507), high stress (OR = 0.60, p = 0.519) or depression (OR = 1.4, p = 0.697) |
| Stewart et al. 2015 (26) | Malawi (Sub-Saharan Africa, low income) | 1391 pregnant women enrolled into one of 3 supplement groups | Perceived Stress Scale (PSS-10), salivary cortisol | BW, BL, HC, arm circumference, WAZ, LAZ, HCZ | Greater cortisol at enrolment and 36 wks predicted shorter GA. Greater cortisol at 28 and 36 wks predicted lower BW. Cortisol was not associated with WAZ, LAZ, or HCZ at any time. PSS scores at 28 and 36 wks predicted shorter LAZ. PSS did not predict other outcomes. |
| Tran et al. 2014 (60) | Vietnam (East Asia & Pacific, LM income) | 378 pregnant women | Edinburgh Postnatal Depression Scale to assess common mental disorders (CMDs) | Bayley Scales of Infant and Toddler Development Social-Emotional Questionnaire | Antenatal CMDs predicted Bayley social-emotional scores (β = -3.30, CI = -6.32, -2.89). Path analyses revealed no direct effects, but a significant indirect effect (estimate = -0.61, CI = -1.17, -0.04): antenatal CMDs predicted postnatal CMDs, which then predicted social-emotional scores. |
| Valladares et al. 2009 (28) | Nicaragua (Latin Am. & Caribbean, LM income) | 147 pregnant women | Salivary cortisol, intimate partner violence, emotional distress, social resources | LBW, PTB, SGA | Increased cortisol predicted LBW and SGA, but not PTB. Women who reported violence had greater prevalence of LBW and SGA, but not PTB. Path analyses suggested that violence predicted LBW through: 1) increased cortisol; 2) increased cortisol and subsequent lower GA; and 3) direct abdominal trauma, and subsequent lower GA. |
| Wado et al. 2014 (51) | Ethiopia (Sub-Saharan Africa, low income) | 537 pregnant women | Edinburgh Postnatal Depression Scale | LBW | Incidence of LBW was 17.9% and was higher among women with depressive symptoms (26.2%) compared to non-symptomatic women (15.8%) (p = 0.01). Antenatal depression predicted LBW (OR = 1.87, CI = 1.09, 3.21). |
| Zhang et al. 2012 (32) | China (East Asia & Pacific, UM income) | 2782 pregnant women: 1391 with PTB, 1391 controls | Questionnaire: Life events (details not provided) | PTB | Stressful life events predicted PTB (OR = 5.54, CI = 2.34, 13.23). |
| Zhu et al. 2014 (61) | China (East Asia & Pacific, UM income) | 152 pregnant women: 38 exposed to severe life events, 114 controls | 19-item Prenatal Life Events Checklist | Bayley Mental (MDI) & Psychomotor Dev. Index (PDI), Toddler Temperament Scale (TTS) | Exposure to life events in the 1stT predicted lower MDI scores (adjusted mean = 103.11 among cases, 110.09 among controls). Life events did not predict PDI scores. 1stT life events predicted higher TTS regularity (adjusted means = 2.77 vs. 2.52) and persistence and attention span scores (adjusted means = 3.61 vs. 3.35). |
| Zhu et al. 2010 (33) | China (East Asia & Pacific, UM income) | 1800 pregnant women | 19-item Prenatal Life Events Checklist | PTB, BW | Life events predicted PTB in the 1stT (RR = 2.60, CI = 1.29, 5.22) and 2ndT (RR = 2.86, CI = 1.32, 6.22), but not the 3rdT. Life events during the 1stT, but not the 2nd and 3rd, predicted BW (RR = -122.97, CI = –166.64, -79.29). |
Given the variability in samples, stress measures, and outcomes, calculation of effect sizes for direct comparison among studies was not possible. Measures provided by the authors such as group means, odds ratios, risk ratios, or percentage of variability explained are included for contextualization.
Countries represented
Twenty-four countries were represented, including four in East Asia and Pacific region (China = 7 studies, Malaysia = 1, Thailand = 2, Vietnam = 1), two in Europe and Central Asia (Romania = 1, Turkey = 1), six in Latin America and the Caribbean (Brazil = 4, Columbia = 1, Guatemala = 1, Mexico = 2, Nicaragua = 2, Peru = 2), one in Middle East and North Africa (Iran = 3), five in South Asia (Bangladesh = 2, India = 1, Nepal = 1, Pakistan = 2, Sri Lanka = 2), and six in Sub-Saharan Africa (Côte d’Ivoire = 1, Democratic Republic of Congo = 2, Ethiopia = 3, Ghana = 1, Malawi = 1, South Africa = 5). One study included samples from two countries. Sixty percent of studies were in upper-middle, 25% in lower-middle, and 15% in low income countries.
Stress measures analyzed
The stress measures varied widely. Most authors used questionnaires, either tailor-made or existing, such as the Perceived Stress Scale, the Modified Life Events Inventory, or the Depression Anxiety Stress Scales. Others used questionnaires of common mental disorders or affective state such as the Patient Health Questionnaire, Self-Reporting Questionnaire, General Health Questionnaire, Edinburgh Postnatal Depression Scale, Beck Depression Inventory, Center for Epidemiological Studies Depression Scale, and State-Trait Anxiety Inventory. These were often presented as a construct of stress or psychological distress, and were thus retained in the review. Stress measures are categorized below into perceived stress (n = 11), life events (n = 13), depression, anxiety, and common mental disorders (n = 23), occupational stress (n = 2), and trauma (n = 3). Eight others analyzed cortisol, a stress hormone. Many included multiple measures.
Outcomes analyzed
Most studies (n = 31) assessed infant outcomes at birth, particularly gestational age or preterm birth (n = 22), and birth weight, low birthweight, or small for gestational age (n = 21). Six analyzed other outcomes of the pregnancy or at birth, such as Apgar scores, miscarriage, or methylation of maternal and fetal tissues. Finally, 14 studies analyzed outcomes later in infancy (n = 5) and childhood (n = 9), such as behavioral development, illnesses, and growth measures.
Gestational age and preterm birth
Twenty-two studies (46%) analyzed gestational age or preterm birth (PTB). Twelve of these (55%) showed significant associations with at least one stress measure.
Perceived stress: One of six studies showed significant relationships. Studies of 474 Romanian women [25] analyzed Perceived Stress Scale scores within the context of a smoking cessation program. In multivariate regressions, high stress predicted PTB (OR = 2.81, CI = 1.17–6.76). Three others showed no effect of perceived stress, but highlighted other important relationships. For example, studies of 1391 women in Malawi [26] showed relationships between cortisol and PTB (discussed below), but no relationships between Perceived Stress Scale scores and cortisol or gestational age. Similarly, studies of 865 Brazilian women [27] showed that Perceived Stress Scale scores did not predict PTB, but highlighted relationships between General Health Questionnaire scores and PTB (discussed below). Finally, studies of 147 women from Nicaragua [28] showed that perceived stress, based on tailored interviews, predicted increased cortisol, but this was not related to PTB.
Two other studies that did not show significant results suffered from small sample sizes. Studies of 46 women from Columbia [29] analyzed combinations of periodontitis and Perceived Stress Scale scores 48 hours postpartum, and found that Perceived Stress Scale scores did not predict PTB, but the small sample limits analyses. Similarly, case-control studies of 125 women from Pakistan [30] showed no relationships between high stress defined by A-Z Stress Scale scores, and PTB (OR = 0.60, p = 0.519), but the small number of preterm births (n = 15) limits analyses.
Life events: Four of five studies showed significant relationships. Case-control studies in Peru [31] showed that life events such as death of a relative, marital discord, and financial troubles predicted PTB, with ORs ranging from 2.09–2.70. Similar case-control studies in China [32] showed that stressful life events such as hospitalization, death of a relative, and family conflict predicted PTB (OR = 5.54). A second study in China [33] showed that stressful life events predicted PTB in the 1stT (RR = 2.60) and 2ndT (RR = 2.86), but not the 3rdT. Finally, studies of 550 women in Iran [34] showed that whereas anxiety and depression had the greatest direct effects on PTB, stressful life events predicted anxiety and depression and were thus indirectly related to PTB. A fifth study from Sri Lanka [35] failed to show significant relationships, likely because it was underpowered. Results nevertheless showed a trend between 2ndT life events and PTB.
Cortisol: Three of five studies showed significant relationships. Studies in Nepal [36] demonstrated that 3rdT cortisol predicted PTB (OR = 1.04). Similarly, studies in Bangladesh [37] showed that high 3rdT cortisol predicted shorter gestational age among male but not female infants. Finally, studies of 1391 women in the International Lipid-based Nutrient Supplement study in Malawi [26] showed that greater cortisol at <20 and 36 weeks pregnancy predicted smaller gestational age. Two others showed no significant relationships. Studies of 125 women in Pakistan [30] showed no differences in blood cortisol levels from 28–30 weeks pregnancy between women who delivered preterm or at term, and the number of PTBs was insufficient for detailed analyses. Studies in Nicaragua [28] showed that cortisol predicted low birthweight and small for gestational age (described below), but not PTB.
Anxiety, Depression, and Common Mental Disorders: Four of ten studies showed significant relationships. Studies of 550 women in Iran [34] showed that Depression Anxiety Stress Scales scores at 24–32 weeks pregnancy predicted gestational age (β = -0.18). Studies of 600 women in Iran [38] showed that State-Trait Anxiety Inventory scores were higher among women with PTB compared to term deliveries, and that state anxiety scores predicted PTB (RR = 3.1, CI = 2.05–4.7). One study of 865 women in Brazil [27] showed that General Health Questionnaire scores >3 at 30–36 weeks predicted PTB (RR = 2.32, CI = 1.18–4.60), controlling for confounders. In contrast, Perceived Stress Scale (discussed above) and State-Trait Anxiety Inventory scores were not significant predictors. Finally, case-control studies of 959 women in Peru [39] showed that Patient Health Questionnaire and Depression Anxiety Stress Scales scores predicted PTB, with ORs for the moderate-severe symptomatic group ranging from 2.76 for the anxiety subscale (CI = 1.83–4.16) to 11.07 (CI = 5.64–21.71) for stress.
Six other studies showed no significant relationships. Studies of 719 women in Ghana and Cote d’Ivoire [40] showed that 3rdT Patient Health Questionnaire and Generalized Anxiety Disorder Scale scores did not predict gestational age or PTB. The sample represented uncomplicated pregnancies; thus, the authors suggest that results might highlight confounding effects of pregnancy complications in the relationship between maternal depression and anxiety, and birth outcomes. Studies of 726 women from South Africa [41] showed that depression, based on Beck Depression Inventory scores at 20–28 weeks pregnancy, did not predict PTB, although relationships with other outcomes are discussed below.
Of studies showing no significant relationships, two are limited by sample size. Studies of Hospital Anxiety and Depression Scale scores among 463 women in China [42] showed that PTB risk did not differ between the Symptom and Symptomless groups. However, only 13 women (4.2%) delivered preterm, all in the symptomless group. Similarly, case-control studies of 125 women from Pakistan [30] showed no relationships between depression, based on the Center for Epidemiological Studies Depression Scale, and PTB (OR = 1.4, p = 0.697), but the small number of preterm births (n = 15) limits analyses. Finally, two studies with large samples might benefit from more detailed analyses. Studies among 737 women in Sri Lanka [35] showed no relationships between General Health Questionnaire scores and PTB. Scores were dichotomized; more detailed analyses might be possible. Similarly, studies of 600 women from Pakistan [43] showed that prevalence of “emotional stress” based on the Edinburgh Postnatal Depression Scale was 69% among cases who delivered preterm and 55% among controls (p<0.01), but the cutoffs were not well described, and stress did not predict PTB in logistic regressions including covariates.
Other stress measures: Three final studies of PTB assessed occupational stress [44], trauma [45], and pregnancy-related stress [46], with mixed results. Case-control studies among 2189 women in China [46] showed that high stress based on the Pregnancy Stress Rating Scale predicted PTB (RR = 2.92, CI = 1.12–7.58), but low and medium levels were not significant predictors. Studies among 2623 women in Mexico [44] showed that occupational stress did not predict PTB, but highlighted other occupational risk factors. Finally, among 544 women in South Africa [45], prevalence of childhood (34%) and lifetime trauma (67%) was high, but trauma did not predict PTB. However, trauma predicted infant head circumference, discussed below.
Birthweight, low birthweight, and small for gestational age
Twenty-one studies (44%) analyzed birthweight, low birthweight (LBW), and small for gestational age. Fourteen (67%) of these showed significant associations with at least one stress measure.
Perceived stress: Two of three studies showed significant relationships. Studies of Perceived Stress Scale scores among 474 Romanian in a smoking cessation program [25], discussed above with PTB and gestational age, showed that high stress predicted a 113g reduction in birthweight (CI = -213–-11). Studies in Nicaragua [28] showed that interviewer-assessed perceived stress predicted increased cortisol, which then predicted LBW and small for gestational age. These results are discussed more in the section on cortisol. In contrast, studies of 46 women from Columbia [29] analyzing periodontitis and Perceived Stress Scale scores found that perceived stress did not predict PTB. However, the small sample limits the analyses.
Life events: Two of four studies showed significant relationships. Studies of 1800 women in China [33] showed that life events during the 1stT, but not 2ndT or 3rdT, predicted birthweight (RR = -122.97, CI = -166.64–-79.29), even adjusting for covariates. Similarly, studies of 1197 women in South Africa [47] used the Social Readjustment Rating Scale to assess life events in the year before delivery. Results in one cohort showed that greater life events scores predicted lower birthweight. Another study of 726 women in South Africa [41] showed no direct effects of life events on birthweight, but highlighted relationships between life events and antenatal depression. Finally, studies of 1065 women in Ethiopia [48] showed no relationships between life events and birthweight.
Cortisol: Three of four studies showed significant relationships. Studies of 1041 women from Bangladesh [37] demonstrated that higher 3rdT cortisol predicted smaller birthweight among male but not female infants. Studies of 1391 women from Malawi [26] showed that greater cortisol at 28 and 36 weeks pregnancy predicted lower BW, particularly among primiparous women. Studies of 147 women in Nicaragua [28] showed that low social resources and high perceived stress predicted increased cortisol, and that increased cortisol then predicted LBW and small for gestational age. In contrast, studies of 737 women in a nutrient supplement study from Nepal [36] showed that 3rdT cortisol did not predict birthweight (β = 4.2, p = 0.17) or LBW (OR = 0.98, CI = 0.95–1.01).
Anxiety, Depression, and Common Mental Disorders: Six of ten studies showed significant relationships. These all had relatively large samples and considered covariates in analyses. Studies of 600 women in Iran [38] showed that high state anxiety (State-Trait Anxiety Inventory score ≥45) predicted LBW (RR = 2.6, CI = 1.6–4.2). Studies assessing 3rdT Edinburgh Postnatal Depression Scale and State-Trait Anxiety Inventory scores among 583 mothers in Bangladesh [49] showed that depressive symptoms (OR = 2.24, CI = 1.37–3.68) and anxiety (OR = 2.08, CI = 1.30–3.25) predicted LBW. Studies of 865 women in Brazil [27] showed that General Health Questionnaire scores >3 at 30–36 weeks pregnancy predicted LBW (RR = 1.97, CI = 1.12–3.47). Similarly, case-control studies of 835 women in Sri Lanka [50] showed that 2ndT General Health Questionnaire scores predicted small for gestational age (OR = 1.92, CI = 1.17–3.14). Studies of 537 women in Ethiopia [51] showed that depressive symptoms assessed with the Edinburgh Postnatal Depression Scale predicted LBW (OR = 1.87, CI = 1.09–3.21). Finally, studies of 726 women in South Africa [41] showed that depression, based on the Beck Depression Inventory at 20–28 weeks pregnancy, predicted weight for age Z-scores (OR = 0.2, CI = 0.02–0.4), although relationships did not persist when controlling for life events.
Four studies (including three discussed above with gestational age and PTB) failed to show significant relationships. Studies of 719 women with healthy, uncomplicated pregnancies in Ghana and Cote d’Ivoire [40] showed that 3rdT Patient Health Questionnaire and Generalized Anxiety Disorder Scale scores did not predict birthweight. Similarly, studies of 521 women in Ethiopia [48] showed that 3rdT Self-Reporting Questionnaire scores did not predict BW. Studies of 175 women in Thailand who completed the Center for Epidemiological Studies Depression Scale at 26–38 weeks pregnancy showed no main effects of antenatal depression on BW [52]. Finally, studies of 463 women in China [42] showed no associations between Hospital Anxiety and Depression Scale scores and birthweight but, as noted above, analyses suffer from small sample sizes.
Other stress measures: Other studies of birthweight, LBW, and small for gestational age assessed occupational stress, trauma, and intimate partner violence. Two studies of occupational stress showed significant effects. Studies among 2623 women in Mexico [44] showed that conflicts at work predicted small for gestational age, but only among women who delivered at the public assistance hospital, which typically sees the poorest women (OR = 4.93, CI = 2.09–11.66). Studies of 792 women working at a chemical plant in China [53] showed interactive effects of benzene exposure and work stress. Adjusted mean birthweight was 183g lower among women with both work stress and benzene exposure compared to those with neither. Three studies of trauma and intimate partner violence showed no direct effects, but highlighted other important relationships. Studies of 726 women from South Africa [41] showed that childhood and past-year trauma predicted antenatal depression, but did not directly predict weight for age Z-scores. Similarly, among 544 women in South Africa [45], prevalence of childhood (34%) and lifetime trauma (67%) were high, but did not predict weight outcomes. Finally, studies of 147 women from Nicaragua [28] demonstrated that intimate partner violence during pregnancy predicted cortisol, which then predicted lower BW. Intimate partner violence might affect both physical and psychosocial health, which complicates analyses. However, the study from Nicaragua [28] highlights maternal stress as a potential mechanism through which intimate partner violence influences birthweight.
Other infant outcomes at birth
Anthropometric measurements: Six studies analyzed other anthropometric measures at birth, including head circumference, head circumference Z-scores, head circumference for age Z-scores, birth length, and length for age Z-scores. Five showed significant results. Studies of 726 women from South Africa [41] showed that antenatal depression, based on Beck Depression Inventory scores at 20–28 weeks pregnancy, predicted smaller head circumference for age Z-scores (OR = 0.3, CI = 0.1–0.6). Similarly, among 544 women in South Africa [45], lifetime trauma exposure predicted a 0.3 unit reduction in head circumference for age Z-scores (p = 0.026). Studies of 1391 women from Malawi [26] showed that perceived stress at 28 and 36 weeks pregnancy predicted shorter length for age Z-scores, but not head circumference Z-scores. In contrast, studies of 461 women in China [42] showed that head circumference and birth length did not differ among infants of women symptomatic and asymptomatic for anxiety and depression. Finally, studies of 719 women in Ghana and Cote d’Ivoire [40] showed that mean head circumference did not differ among low-obstetric risk women with and without anxiety and depression in the 3rdT, although more detailed analyses are needed.
Two studies analyzed relationships between these measures and cortisol. Studies of 1041 women from Bangladesh [37] demonstrated that higher 3rdT cortisol predicted smaller head circumference among male but not female infants, mirroring results for birthweight (discussed above). In contrast, studies of 1391 women from Malawi [26] (discussed above) showed that cortisol at 28 and 36 weeks pregnancy did not predict length for age Z-scores or head circumference Z-scores.
Apgar scores: Studies in Iran [54] of 29 pregnant women with anxiety or depression assigned to a stress management intervention (n = 14) or control group (n = 15) showed that mean 1-minute Apgar scores were higher in the intervention (8.93) than control (8.07) group (p = 0.01), with similar results at five minutes. Two other previously-described studies also assessed Apgar scores. Studies of 719 women in Ghana and Cote d’Ivoire [40] showed that Apgar scores were weakly correlated with Patient Health Questionnaire (r = -0.106, p<0.05) and Generalized Anxiety Disorder Scale scores (r = -0.102, p<0.05). In contrast, studies of 461 women in China [42] showed that Apgar scores did not differ among those who were symptomatic or asymptomatic for anxiety and depression.
Other outcomes of the pregnancy
Six articles examined other outcomes of the pregnancy or at birth (three of these also analyzed other birth outcomes, discussed above). Studies of 1065 pregnant women in Ethiopia [48] showed no associations between stillbirth or neonatal death and 3rdT common mental disorder symptoms, life events, or worry in pregnancy. However, common mental disorder symptoms were associated with prolonged labor (Low symptoms, RR = 1.4, CI = 1.0–1.9; High, RR = 1.6, CI = 1.0–2.6). Similarly, analyses of 461 women in China [42] showed that prevalence of prolonged pregnancy was higher among women symptomatic for anxiety and depression (n = 3, 8.3%) compared to symptomless women (n = 5, 1.6%) (RR = 4.08, CI = 1.25–13.33), although several other outcomes did not differ among groups.
Two studies assessed miscarriage, with one showing significant results. Studies of 22 women in rural Guatemala [55] followed in detail from before pregnancy showed that increased urinary cortisol predicted pregnancy loss (OR = 2.7, CI = 1.2–6.2), and highlighted that placentation might be a particularly sensitive period to maternal stressors. In contrast, case-control studies of 33 women in Malaysia [56] showed no differences in mean morning cortisol at 24–32 weeks pregnancy between women with a history of PTB and miscarriage, and those with no such history. However, it is unclear how many women were assigned to the case group because of previous miscarriages, PTB, or both.
Finally, two studies assessed methylation patterns following exposure to severe stress and trauma during pregnancy. Studies of 24 mother-infant dyads in Democratic Republic of Congo [57] show differential methylation of maternal venous blood, placental tissue, and umbilical cord blood among women exposed to chronic stress and war trauma. Correcting for multiple testing, four CpG sites were differentially methylated in at least one tissue, all associated with war trauma. Methylation at four sites predicted birthweight, collectively explaining 55% of variance. Studies by the same group [58] of 25 women in Democratic Republic of Congo showed that BW was correlated with maternal deprivation (r = -0.484), mundane stress (r = -0.521), and war stress (r = -0.620). Of the war stressors, rape was the most important predictor, explaining 31% of variance. War stress was correlated with birthweight (r = -0.449), and with cord blood methylation of the NR3C1 gene (r = 0.586), which might have long-term effects on the child’s stress response.
Outcomes later in infancy and childhood
Among the 48 studies, 14 (29%) analyzed outcomes later in infancy (n = 5) and childhood (n = 9). These are grouped by age period and outcome below rather than stress measure because of the limited number of studies.
Infancy–Temperament: Three studies assessed temperament, all showing significant results. Studies of 100 women in India [59] showed no linear relationships between 3rdT General Health Questionnaire scores and temperament at 1–4 months of age. However, adaptability and approach scores were higher among infants whose mothers had low (score <7) compared to high distress. Studies of 378 women in Vietnam [60] showed that 2ndT Edinburgh Postnatal Depression Scale scores predicted Bayley Scales of Infant Development social-emotional scores at age 6 months (β = -3.30, CI = -6.32–-2.89). Path analyses showed that antenatal common mental disorders predicted postnatal common mental disorders, which then predicted social-emotional scores. Studies in China of 38 women with healthy, uncomplicated pregnancies [61] compared Bayley and Toddler Temperament Scale scores at age 16 and 18 months among infants of 38 mothers exposed to 1stT life events with 114 unexposed controls. Controlling for covariates, exposure to life events predicted lower infant mental development scores, and higher regularity and persistence and attention span scores (on which lower scores indicate better responses).
Infancy–Motor Development: The study of 38 women in China discussed above [61] showed no relationships between maternal life events and infant motor development. Others showed mixed results. Studies in South Africa [62] categorized women into low and high stress groups based on feelings about pregnancy, life events, and family relationships, and analyzed infants’ neuromotor development shortly after birth, and at 1 and 3 months. Motor scores did not differ between groups. However, excluding babies with birthweight >8 pounds (n = 35), significant differences were noted: 42.5% of infants in the high maternal stress group had motor scores <3, compared to 15.4% in the low-stress group. The limited analyses restrict generalization, and the exclusion of larger babies is not clearly justified. However, the rigorous assessments of neuromotor development are an important contribution to the literature.
Infancy–Illness: Studies in Ethiopia [63] analyzed 3rdT and postpartum Self-Reporting Questionnaire scores among 954 women, and maternal-reported infant illness at age 2 months. Multivariate analyses showed that persistent (during both pregnancy and postpartum) common mental disorder symptoms predicted infant diarrhoea (RR = 2.15, CI = 1.39–3.34). Relationships with other illnesses were inconsistent.
Childhood–Behavioral Outcomes: Six studies analyzed behavioral and psychiatric outcomes, with five showing significant relationships. Studies in Nicaragua [64] analyzed 2ndT–3rdT salivary cortisol and Self-Reporting Questionnaire scores among 147 women. Children were assessed at age 9 using the Child Behavior Checklist, and their cortisol was analyzed. Maternal cortisol predicted total Child Behavior Checklist scores (r = 0.31), and internalizing (r = 0.28) and externalizing symptoms (r = 0.35), but not children’s cortisol. Self-Reporting Questionnaire scores did not predict any child outcomes. Researchers in Brazil [65] assessed discord during pregnancy using a single retrospective interview question, and Attention Deficit Hyperactivity Disorder (ADHD) symptoms among 370 children age 6–13 with the Child Behavior Checklist and Teacher Report Form. Discord during pregnancy predicted mother-reported ADHD (OR = 4.54, CI = 2.16–9.57), controlling for covariates. Similarly, studies of 241 women in Thailand [66] assessed emotional distress during pregnancy using a single retrospective question, and compared results between 122 6-12-year-old children with ADHD, and 119 controls with no behavioral problems. Maternal distress predicted ADHD in multivariate analyses (OR = 2.99, CI = 1.43–5.40). Studies in South Africa [67] assessed 3rdT exposure to 16 stressors, divided into high and low exposure, among 953 women, and children’s behavioral problems at ages 2 and 4 using the Behaviour Screening Questionnaire. Controlling for covariates, maternal stress exposure predicted behavioral problems among children at age 4 (OR = 2.66, CI = 1.28–5.54). Finally, researchers in Brazil [68] used a single questionnaire item, administered shortly after delivery, to assess mood symptoms during pregnancy. Psychiatric disorders were assessed among 3581 children at age 6 using the Development and Well-Being Assessment. Controlling for confounders, maternal mood symptoms predicted psychiatric disorders among children (OR = 1.97, CI = 1.60–2.41). In contrast, studies in Turkey [69] retrospectively assessed stressors experienced during pregnancy using a tailor-made form and showed no differences in exposure between 105 3-5-year-old children with psychiatric complaints, and 205 controls.
Childhood–Other Outcomes: Other studies analyzed heart rate [70], physical growth [71], and asthma [72]. Studies in China analyzed depression and anxiety among 216 pregnant women [70], and children’s heart rate and blood pressure before, during, and after a video game stressor at age 7–9. Heart rate and blood pressure stress responses were higher among children of mothers with anxiety, and anxiety predicted all heart rate and blood pressure outcomes in multivariate analyses. Results for depression were significant but less consistent. Studies in Brazil [71] analyzed body mass index Z-scores at age 5–8 among 409 children whose mothers completed the Perceived Stress Scale, General Health Questionnaire, and State-Trait Anxiety Inventory three times during pregnancy. In multivariate regressions, 2ndT General Health Questionnaire scores predicted children’s body mass index Z-scores (β = -0.09). Finally, studies in Mexico [72] analyzed relationships between 2ndT–3rdT life events and child asthma symptoms at age 4. Controlling for confounders, negative life events during pregnancy predicted ever wheeze among children (RR = 1.08, CI = 1.00–1.16) and wheeze in the past 12 months (RR = 1.12, CI = 1.00–1.26).
Discussion
This scoping review highlights a number of patterns in prenatal stress research already observed in other studies. For example, two-thirds of studies that assessed relationships between prenatal stress and birthweight showed modest but significant associations, consistent with the general patterns observed in past research from mostly high-income countries [1, 12]. Similarly, effects vary based on timing of exposure, which has been widely observed in other studies [5, 9, 10, 73]. Effects were evident even later in childhood, consistent with long-term programming effects of prenatal stress observed by others [2–4, 8–10].
The synthesis also highlights wide variability that has already been observed in the literature. For example, relationships between life events and birthweight were not significant in two of four studies, which likely reflects variability in the number and type of life events assessed, complications with retrospective assessment in many studies, and confounding by other variables, as in past research from mostly high-income countries [1]. Similarly, relationships between prenatal stress and gestational age were significant in only about half of studies. Researchers in high-income countries have observed wide heterogeneity in relationships between prenatal stress and gestational age, reflecting variability in relationships based on the timing of stress exposure during pregnancy, lack of distinction between the particular components of the stress response that might predict gestational age, and interactive relationships with other factors such as maternal infection or nutrition [73]. In particular, gestational age seems to be more strongly linked to prenatal anxiety than to stress or depression [74], and results might be masked in studies that cannot distinguish between these constructs.
Finally, our synthesis highlights broader challenges to research on prenatal stress and child development in LMICs. For example, although many studies from high-income countries have shown relationships between perceived stress or common mental disorders and gestational age [1, 12, 73], results here were significant in only one of six studies assessing perceived stress, and only four of ten studies assessing anxiety, depression, and common mental disorders. The interacting effects of confounding variables might be a particularly important source of heterogeneity in this context. Furthermore, measurement of gestational age is a challenge in many LMICs where women might not receive early prenatal care [75]. Thus, inconsistencies in relationships between prenatal stress and gestational age or preterm birth might be further exacerbated by methodological challenges. Similarly, as noted above, distinguishing between constructs such as stress, distress, anxiety, and depression is important, but this might not be possible in LMICs lacking detailed evaluation tools. Some studies specifically compared effects of stress, anxiety, and depression on developmental outcomes [39], but others focused more on similarities between the constructs of stress and common mental disorders in the context. For example, authors noted that family life stressors [49] and stress due to poverty or ill health [40] might exacerbate or underlie part of the relationship between common mental disorders and perinatal outcomes, and thus discussed these concepts together rather than distinguishing between them.
Synthesis of these studies highlights other important steps for future research. First, existing research is biased toward upper-middle income countries (60% of studies). In particular, three upper-middle income countries (China, South Africa, and Brazil) accounted for 16 of 48 studies reviewed (33%). Studies in low income countries [26, 36, 48, 51, 57, 58, 63] represented only 15%, and lower-middle income [28, 30, 35, 37, 40, 43, 49, 50, 55, 59, 60, 64] only 25%. This likely reflects limited research and healthcare infrastructure that complicates data collection and follow-up. However, our reviews show that, in general, researchers were able to collect sufficient data during pregnancy and up to birth to conduct solid analyses of relationships. Of the 19 studies in low and lower-middle income countries, 12 [26, 28, 36, 37, 49–51, 55, 57, 58, 60, 64] showed significant relationships. Four others [40, 48, 59, 63] were less robust, but showed relationships with some outcomes. Although low, lower-middle, and upper-middle income countries share many similar challenges in maternal, infant, and psychosocial health, studies from diverse regions remain necessary because of differences in public health burdens such as communicable diseases, maternal and perinatal conditions and nutritional deficiencies [76] and infant mortality [77], as well as different public health spending patterns [15], maternal care services [75], and intervention priorities [78]. These factors might particularly affect the application of prenatal stress research findings to healthcare. More research in low and lower-middle income countries is warranted to foster generalization of major findings and their eventual application to public health.
This review also indicates a bias for studies of outcomes at birth, with limited follow-up beyond, across all three income groups. Although studies of relationships between prenatal stress and birth outcomes remain necessary, longer-term follow-up is essential to assess how patterns seen at birth might translate into child and adult well-being, and to identify relationships with outcomes not measurable at birth. Limited infrastructure likely hinders long-term follow-up of mothers and infants in LMICs.
Assessment of the varied stress measures employed shows the applicability of many instruments used in high-income countries across all three income groups, but also the usefulness of and need for tailored measures. Existing surveys of stress, anxiety, depression, and life events were used in 32 studies, including four from low income countries [26, 48, 51, 63], nine from lower-middle income [30, 35, 40, 43, 49, 50, 59, 60, 64], and 19 from upper-middle income countries [25, 27, 29, 33, 34, 38, 39, 41, 42, 44–47, 52, 54, 61, 70–72]. Cortisol was analyzed in eight, including two from low-income countries [26, 36], five from lower-middle income [28, 30, 37, 55, 64], and one from upper-middle income [56]. The use of these existing measures facilitates comparisons across studies. However, collecting biological samples is not always feasible where infrastructure is limited, and existing questionnaires might be difficult to employ because of a lack of validated translations, high illiteracy rates, and because women might not be familiar with self-report surveys. Thus, tailor-made questionnaires are sometimes the only option. Five studies [31, 32, 62, 67, 69] used tailor-made surveys to assess life events and perceived stress. Two of these [32, 69] do not describe the instruments in detail, but the others provide enough information to allow basic comparisons across studies. Three others [28, 57, 58] used ethnographic interviews with structured questionnaires to assess trauma and violence. Although this limits generalization, the authors highlight that culturally sensitive interview methods might allow for more complete and accurate ascertainment of stressors than existing or unfamiliar questionnaires [57], and the development of rapport with the interviewer might help to minimize underreporting [28]. Finally, four studies [53, 65, 66, 68] used responses to a single questionnaire item to assess stress, all showing significant results. While this again limits generalization, it demonstrates the usefulness of simple measures that might pave the way for more detailed future evaluations.
These studies also highlighted the importance of the broader social and environmental context in studies of prenatal stress in LMICs. For example, in one study [62], women’s reports of difficulties predicted outcomes, but results were less consistent for situations that might objectively appear stressful such as poor diet or household crowding, highlighting the importance of women’s own perceptions of stress. Similarly, researchers suggested that the lack of relationship between common mental disorders and birth outcomes in one study [48] might reflect interrelationships among factors such as illness, undernutrition, and socioeconomic disadvantage that were common in the population and might overwhelm the effects of common mental disorders. Qualitative interviews [79] from the same group indicated that worry about the delivery was an important source of distress, reflecting the reality of high maternal and infant morbidity and mortality in the region. Other themes highlighted in articles reviewed here such as unwanted pregnancy and intimate partner violence are not unique to LMICs, but lack of resources in these settings might exacerbate women’s associated distress.
The conclusions that can be drawn from several studies are limited because of small sample size and limited statistical analyses, which was observed across the three income categories. Twelve studies with particularly limited samples and analyses were identified (see Discussion points in S2 Table and S2 Appendix), and several others would benefit from more rigorous assessment of covariates. Stress during pregnancy is associated with numerous covariates such as maternal age, socioeconomic status, nutrition, marital status, physical health, and pregnancy history that might all affect outcomes independently or in interaction with stress, but these were not considered in many studies. However, careful consideration of these variables might highlight pathways underlying relationships between prenatal stress and child development. Three studies used path analyses [28, 34, 60] to address such relationships; these methods might be particularly important for guiding future interventions. Studies of interactive effects should also be prioritized. For example, research in Bangladesh [37] showed interactive effects of stress and nutrition, suggesting that early nutrient supplementation might ameliorate the negative effects of prenatal stress on infant growth. When exposure to stress cannot be changed, other interacting variables might provide targets to alleviate its effects. More rigorous assessment of interactions and pathways is needed to identify these targets.
Strengths and limitations
This is a broad study aiming to synthesize available research related to prenatal stress and child development from LMICs. The inclusion of multiple measures of stress and developmental outcomes resulted in a large body of literature for analysis, allowing us to highlight the wide diversity in prenatal stress research in LMICs. However, given the breadth of studies included, with important differences in samples, predictor measures, and outcomes, calculation and direct comparison of effect sizes was not conducted. Furthermore, detailed comparison of study results from LMICs and high income countries is beyond the scope of this review. Direct comparison of the direction and strength of relationships from LMICs and high income countries represents an important area for future reviews focusing on specific stress measures, such as life events or perceived stress, or developmental outcomes, such as low birthweight or motor development.
Conclusions
Many current studies show relationships between prenatal stress and outcomes of the pregnancy, infant, or child in LMICs. Synthesis of these reviews highlights the applicability in LMICs of methods used in high-income countries, but also the need for tailored tools adapted to the cultural context. These should include careful assessment of covariates, especially those that might have interactive effects with maternal stress. Studies including longer-term follow-up should be prioritized. The development and validation of brief and simple survey measures that could be incorporated into broader studies of maternal and child health might be an important avenue to guide future research. Direct comparison of studies analyzing specific stress measures or developmental outcomes from LMICs and high income countries represents an important area for future review.
Supporting information
(DOCX)
(DOCX)
(DOC)
(DOCX)
Acknowledgments
We are grateful to Anne-Marie Turcotte-Tremblay for insightful discussions on the development of this project, and her contribution to preliminary versions of the search strategy.
Abbreviations
- 1stT, 2ndT, 3rdT
first, second, and third trimester
- ADHD
Attention Deficit Hyperactivity Disorder
- BL
Birth length
- BMI
Body Mass Index (kg/m2)
- BMIZ
Body mass index Z-score
- BP
Blood pressure
- BW
Birth weight
- CBCL
Child Behavior Checklist
- CI
Confidence interval
- CMD
Common Mental Disorder
- DASS
Depression Anxiety Stress Scales
- DSM
Diagnostic and Statistical Manual of Mental Disorders
- GA
Gestational age
- GHQ
General Health Questionnaire
- HC
Head circumference
- HCAZ
Head circumference for age Z-score
- HCZ
Head circumference Z-score
- HR
Heart rate
- IUGR
Intra-uterine growth restriction
- LAZ
Length for age Z-score
- LBW
Low birthweight
- LM income
Lower-middle income
- LMIC
Low- and middle-income countries
- MLEI
Modified Life Events Inventory
- OR
Odds ratio
- PHQ
Patient Health Questionnaire
- PSS
Perceived Stress Scale
- PTB
Preterm birth
- RR
Relative risk
- SGA
Small for gestational age
- SRQ
Self-Reporting Questionnaire
- SRRS
Social Readjustment Rating Scale
- STAI
State-Trait Anxiety Inventory
- TTS
Toddler Temperament Scale
- UM income
Upper-middle income
- WAZ
Weight for age Z-score
Data Availability
All relevant data are within the paper and its Supporting Information files. The full history of the protocol and bibliographic research strategy is available via the PROSPERO International prospective register of systematic reviews, # CRD42016048634.
Funding Statement
Kelsey Dancause was supported by a salary award from Fonds de recherche du Québec – Santé while working on this manuscript: http://www.frqs.gouv.qc.ca/partenariat/nos-projets-de-recherche/projet?id=3zjix07l1442500387658.
References
- 1.Beydoun H, Saftlas AF. Physical and mental health outcomes of prenatal maternal stress in human and animal studies: A review of recent evidence. Paediatr Perinat Epidemiol. 2008;22(5):438–66. 10.1111/j.1365-3016.2008.00951.x [DOI] [PubMed] [Google Scholar]
- 2.Entringer S, Buss C, Wadhwa PD. Prenatal stress and developmental programming of human health and disease risk: Concepts and integration of empirical findings. Curr Opin Endocrinol Diabetes Obes. 2010;17(6):507–16. 10.1097/MED.0b013e3283405921 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Glover V, O’Connor TG, O’Donnell K. Prenatal stress and the programming of the HPA axis. Neurosci Biobehav Rev. 2010;35(1):17–22. 10.1016/j.neubiorev.2009.11.008 [DOI] [PubMed] [Google Scholar]
- 4.Hocher B. Fetal programming of cardiovascular diseases in later life: Mechanisms beyond maternal undernutrition. J Physiol. 2007;579(Pt 2):287–8. 10.1113/jphysiol.2007.127738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.King S, Dancause KN, Turcotte-Tremblay A-M, Veru F, Laplante DP. Using natural disasters to study the effects of prenatal maternal stress on child health and development. Birth Defects Res. C: Embryo Today. 2012;96:273–88. 10.1002/bdrc.21026 [DOI] [PubMed] [Google Scholar]
- 6.Kingston D. The effects of prenatal and postpartum maternal psychological distress on child development: A systematic review. Alberta Centre for Child, Family, and Community Research; 2011. [Google Scholar]
- 7.Lazinski MJ, Shea AK, Steiner M. Effects of maternal prenatal stress on offspring development: A commentary. Arch Womens Ment Health. 2008;11:363–75 10.1007/s00737-008-0035-4 [DOI] [PubMed] [Google Scholar]
- 8.Kapoor A, Dunn E, Kostaki A, Andrews MH, Matthews SG. Fetal programming of hypothalamo-pituitary-adrenal function: Prenatal stress and glucocorticoids. J Physiol. 2006;572(Pt 1):31–44. 10.1113/jphysiol.2006.105254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sandman CA, Davis EP, Buss C, Glynn LM. Prenatal programming of human neurological function. Int J Pept. 2011;2011:837596 10.1155/2011/837596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sandman CA, Davis EP, Buss C, Glynn LM. Exposure to prenatal psychobiological stress exerts programming influences on the mother and her fetus. Neuroendocrinology. 2012;95(1):7–21. 10.1159/000327017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Monk C, Spicer J, Champagne FA. Linking prenatal maternal adversity to developmental outcomes in infants: The role of epigenetic pathways. Dev Psychopathol. 2012;24(4):1361–76. 10.1017/S0954579412000764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hobel CJ, Goldstein A, Barrett ES. Psychosocial stress and pregnancy outcome. Clin Obstet Gynecol. 2008;51(2):333–48. 10.1097/GRF.0b013e31816f2709 [DOI] [PubMed] [Google Scholar]
- 13.Kingston D, Tough S, Whitfield H. Prenatal and postpartum maternal psychological distress and infant development: A systematic review. Child Psychiatry Hum Dev. 2012;43(5):683–714. 10.1007/s10578-012-0291-4 [DOI] [PubMed] [Google Scholar]
- 14.Kingston D, Tough S. Prenatal and postnatal maternal mental health and school-age child development: A systematic review. Matern Child Health J. 2014;18(7):1728–41. 10.1007/s10995-013-1418-3 [DOI] [PubMed] [Google Scholar]
- 15.WHO (World Health Organization). New perspectives on global health spending for universal health coverage. Geneva: WHO, 2017. http://www.who.int/health_financing/topics/resource-tracking/new-perspectives/en/ [Google Scholar]
- 16.WHO (World Health Organization). Maternal mortality. Geneva: WHO, 2018. http://www.who.int/news-room/fact-sheets/detail/maternal-mortality. [Google Scholar]
- 17.World Bank. World bank country and lending groups 2018. https://datahelpdesk.worldbank.org/knowledgebase/articles/906519-world-bank-country-and-lending-groups.
- 18.Premji S. Perinatal distress in women in low- and middle-income countries: Allostatic load as a framework to examine the effect of perinatal distress on preterm birth and infant health. Matern Child Health J. 2014;18(10):2393–407. 10.1007/s10995-014-1479-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stein A, Pearson RM, Goodman SH, Rapa E, Rahman A, McCallum M, et al. Effects of perinatal mental disorders on the fetus and child. Lancet. 2014;384(9956):1800–19. 10.1016/S0140-6736(14)61277-0 [DOI] [PubMed] [Google Scholar]
- 20.Gelaye B, Rondon MB, Araya R, Williams MA. Epidemiology of maternal depression, risk factors, and child outcomes in low-income and middle-income countries. Lancet Psychiatry. 2016;3(10):973–82. 10.1016/S2215-0366(16)30284-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.UNICEF. Low birthweight: country, regional, and global estimates. New York: UNICEF, 2004. https://www.unicef.org/publications/index_24840.html [Google Scholar]
- 22.Rondo PH, Ferreira RF, Nogueira F, Ribeiro MC, Lobert H, Artes R. Maternal psychological stress and distress as predictors of low birth weight, prematurity and intrauterine growth retardation. Eur J Clin Nutr. 2003;57(2):266–72. 10.1038/sj.ejcn.1601526 [DOI] [PubMed] [Google Scholar]
- 23.WHO (World Health Organization). Maternal mental health and child health and development in low and middle income countries. Geneva: WHO, 2008. www.who.int/mental_health/prevention/suicide/mmh_jan08_meeting_report.pdf [Google Scholar]
- 24.Kumari S, Joshi S, Jain M. Religiosity, anxiety and pregnancy outcomes in Indian Women. J Indian Academy Applied Psychology. 2013;39(1):110–6. [Google Scholar]
- 25.Meghea CI, Rus IA, Chereches RM, Costin N, Caracostea G, Brinzaniuc A. Maternal smoking during pregnancy and birth outcomes in a sample of Romanian women. Cent Eur J Public Health. 2014;22(3):153–8. 10.21101/cejph.a3947 [DOI] [PubMed] [Google Scholar]
- 26.Stewart CP, Oaks BM, Laugero KD, Ashorn U, Harjunmaa U, Kumwenda C, et al. Maternal cortisol and stress are associated with birth outcomes, but are not affected by lipid-based nutrient supplements during pregnancy: An analysis of data from a randomized controlled trial in rural Malawi. BMC Pregnancy Childbirth. 2015;15(1). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rondó PHC, Ferreira RF, Nogueira F, Ribeiro MCN, Lobert H, Artes R. Maternal psychological stress and distress as predictors of low birth weight, prematurity and intrauterine growth retardation. Eur J Clin Nutr. 2003;57(2):266–72. 10.1038/sj.ejcn.1601526 [DOI] [PubMed] [Google Scholar]
- 28.Valladares E, Peña R, Ellsberg M, Persson LÅ, Högberg U. Neuroendocrine response to violence during pregnancy: Impact on duration of pregnancy and fetal growth. Acta Obstet Gynecol Scand. 2009;88(7):818–23. 10.1080/00016340903015321 [DOI] [PubMed] [Google Scholar]
- 29.Arteaga-Guerra JJ, Cerón-Souza V, Mafla AC. Dynamic among periodontal disease, stress, and adverse pregnancy outcomes. Rev Salud Publica. 2010;12(2):276–86. [DOI] [PubMed] [Google Scholar]
- 30.Shaikh K, Premji SS, Rose MS, Kazi A, Khowaja S, Tough S. The association between parity, infant gender, higher level of paternal education and preterm birth in Pakistan: A cohort study. BMC Pregnancy Childbirth. 2011;11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Barrios YV, Esanchez S, Qiu C, Gelaye B, williams MA. Risk of spontaneous preterm birth in relation to maternal experience of serious life events during pregnancy. Int J Womens Health. 2014;6(1):249–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhang YP, Liu XH, Gao SH, Wang JM, Gu YS, Zhang JY, et al. Risk factors for preterm birth in five Maternal and Child Health hospitals in Beijing. PLoS One. 2012;7(12):e52780 10.1371/journal.pone.0052780 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhu P, Tao F, Hao J, Sun Y, Jiang X. Prenatal life events stress: Implications for preterm birth and infant birthweight. Am J Obstet Gynecol. 2010;203(1):34.e1–.e8. [DOI] [PubMed] [Google Scholar]
- 34.Mirabzadeh A, Dolatian M, Forouzan AS, Sajjadi H, Majd HA, Mahmoodi Z. Path analysis associations between perceived social support, stressful life events and other psychosocial risk factors during pregnancy and preterm delivery. Iran Red Crescent Med J. 2013;15(6):507–14. 10.5812/ircmj.11271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Abeysena C, Jayawardana P, Seneviratne RDA. Effect of psychosocial stress and physical activity on preterm birth: A cohort study. J Obstet Gynaecol Res. 2010;36(2):260–7. 10.1111/j.1447-0756.2009.01160.x [DOI] [PubMed] [Google Scholar]
- 36.Christian P, Nanayakkara-Bind A, Schulze K, Wu L, Leclerq SC, Khatry SK. Antenatal micronutrient supplementation and third trimester cortisol and erythropoietin concentrations. Matern Child Nutr. 2016;12(1):64–73. 10.1111/mcn.12138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Frith AL, Naved RT, Persson LA, Frongillo EA. Early prenatal food supplementation ameliorates the negative association of maternal stress with birth size in a randomised trial. Matern Child Nutr. 2015;11(4):537–49. 10.1111/mcn.12047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nasiri Amiri F, Mohamadpour RA, Salmalian H, Ahmadi AM. The association between prenatal anxiety and spontaneous preterm birth and low birth weight. Iran Red Crescent Med J. 2010;12(6):650–4. [Google Scholar]
- 39.Sanchez SE, Puente GC, Atencio G, Qiu C, Yanez D, Gelaye B, et al. Risk of spontaneous preterm birth in relation to maternal depressive, anxiety, and stress symptoms. J Reprod Med. 2013;58(1–2):25–33. [PMC free article] [PubMed] [Google Scholar]
- 40.Bindt C, Guo N, Te Bonle M, Appiah-Poku J, Hinz R, Barthel D, et al. No association between antenatal common mental disorders in low-obstetric risk women and adverse birth outcomes in their offspring: Results from the CDS study in Ghana and Côte D’Ivoire. PLoS One. 2013;8(11). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Brittain K, Myer L, Koen N, Koopowitz S, Donald KA, Barnett W, et al. Risk factors for antenatal depression and associations with infant birth outcomes: Results from a South African birth cohort study. Paediatr Perinat Epidemiol. 2015;29(6):504–14. [DOI] [PubMed] [Google Scholar]
- 42.Qiao Y, Li J, Wang J. Effects of depressive and anxiety symptoms during pregnancy on pregnant, obstetric and neonatal outcomes: A follow-up study. J Obstet Gynaecol. 2012;32(3):237–40. 10.3109/01443615.2011.647736 [DOI] [PubMed] [Google Scholar]
- 43.Baig SA, Khan N, Baqai T, Fatima A, Karim SA, Aziz S. Preterm birth and its associated risk factors. A study at tertiary care hospitals of Karachi, Pakistan. J Pak Med Assoc. 2013;63(3):414–8. [PubMed] [Google Scholar]
- 44.Cerón-Mireles P, Harlow SD, Sánchez-Carrillo CI. The risk of prematurity and small-for-gestational-age birth in Mexico City: The effects of working conditions and antenatal leave. Am J Public Health. 1996;86(6):825–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Koen N, Brittain K, Donald KA, Barnett W, Koopowitz S, Maré K, et al. Psychological trauma and posttraumatic stress disorder: Risk factors and associations with birth outcomes in the Drakenstein Child Health Study. Eur J Psychotraumatology. 2016;7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Qu XL, Zhu WJ, Chen WQ, Cui YY, He P, He ZH, et al. Effect of pregnancy-specific stress on spontaneous preterm birth among Chinese people. Clin Exp Obstet Gynecol. 2016;43(1):103–8. [PubMed] [Google Scholar]
- 47.Rothberg AD, Shuenyane E, Lits B, Strebel PM. Effect of stress on birth weight in two Johannesburg populations. S Afr Med J. 1991;79(1):35–8. [PubMed] [Google Scholar]
- 48.Hanlon C, Medhin G, Alem A, Tesfaye F, Lakew Z, Worku B, et al. Impact of antenatal common mental disorders upon perinatal outcomes in Ethiopia: The P-MaMiE population-based cohort study. Trop Med Int Health. 2009;14(2):156–66. 10.1111/j.1365-3156.2008.02198.x [DOI] [PubMed] [Google Scholar]
- 49.Nasreen HE, Kabir ZN, Forsell Y, Edhborg M. Low birth weight in offspring of women with depressive and anxiety symptoms during pregnancy: Results from a population based study in Bangladesh. BMC Public Health. 2010;10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ruwanpathirana T, Fernando DN. Risk factors for ‘small for gestational age babies’. Indian J Pediatr. 2014;81(10):1000–4. 10.1007/s12098-014-1382-y [DOI] [PubMed] [Google Scholar]
- 51.Wado YD, Afework MF, Hindin MJ. Effects of maternal pregnancy intention, depressive symptoms and social support on risk of low birth weight: A prospective study from southwestern Ethiopia. PLoS ONE. 2014;9(5):e96304 10.1371/journal.pone.0096304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sanguanklin N, McFarlin BL, Park CG, Giurgescu C, Finnegan L, White-Traut R, et al. Effects of the 2011 Flood in Thailand on birth outcomes and perceived social support. J Obstet Gynecol Neonatal Nurs. 2014;43(4):435–44. 10.1111/1552-6909.12466 [DOI] [PubMed] [Google Scholar]
- 53.Chen D, Cho SI, Chen C, Wang X, Damokosh AI, Ryan L, et al. Exposure to benzene, occupational stress, and reduced birth weight. Occup Environ Med. 2000;57(10):661–7. 10.1136/oem.57.10.661 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Karamoozian M, Askarizadeh G. Impact of prenatal cognitive-behavioral stress management intervention on maternal anxiety and depression and newborns’ Apgar scores. Iranian J Neonatology. 2015;6(2):14–23. [Google Scholar]
- 55.Nepomnaschy PA, Welch KB, McConnell DS, Low BS, Strassmann BI, England BG. Cortisol levels and very early pregnancy loss in humans. Proc Natl Acad Sci U S A. 2006;103(10):3938–42. 10.1073/pnas.0511183103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Arffin F, Al-Bayaty FH, Hassan J. Environmental tobacco smoke and stress as risk factors for miscarriage and preterm births. Arch Gynecol Obstet. 2012;286(5):1187–91. 10.1007/s00404-012-2417-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kertes DA, Kamin HS, Hughes DA, Rodney NC, Bhatt S, Mulligan CJ. Prenatal maternal stress predicts methylation of genes regulating the hypothalamic-pituitary-adrenocortical system in mothers and newborns in the Democratic Republic of Congo. Child Dev. 2016;87(1):61–72. 10.1111/cdev.12487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Mulligan CJ, D'Errico NC, Stees J, Hughes DA. Methylation changes at NR3C1 in newborns associate with maternal prenatal stress exposure and newborn birth weight. Epigenetics. 2012;7(8):853–7. 10.4161/epi.21180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bhat A, Chowdayya R, Selvam S, Khan A, Kolts R, Srinivasan K. Maternal prenatal psychological distress and temperament in 1–4 month old infants: A study in a non-western population. Infant Behav Dev. 2015;39:35–41. 10.1016/j.infbeh.2014.12.002 [DOI] [PubMed] [Google Scholar]
- 60.Tran TD, Biggs BA, Tran T, Simpson JA, De Mello MC, Hanieh S, et al. Perinatal common mental disorders among women and the social and emotional development of their infants in rural Vietnam. J Affective Disord. 2014;160:104–12. [DOI] [PubMed] [Google Scholar]
- 61.Zhu P, Sun MS, Hao JH, Chen YJ, Jiang XM, Tao RX, et al. Does prenatal maternal stress impair cognitive development and alter temperament characteristics in toddlers with healthy birth outcomes? Dev Medicine Child Neurol. 2014;56(3):283–9. [DOI] [PubMed] [Google Scholar]
- 62.Abramson JH, Singh AR, Mbambo V. Antenatal stress and the baby’s development. Arch Dis Child. 1961;36(185):42–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ross J, Hanlon C, Medhin G, Alem A, Tesfaye F, Worku B, et al. Perinatal mental distress and infant morbidity in Ethiopia: A cohort study. Arch Dis Child Fetal Neonatal Ed. 2011;96(1):F59–F64. 10.1136/adc.2010.183327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Isaksson J, Lindblad F, Valladares E, Högberg U. High maternal cortisol levels during pregnancy are associated with more psychiatric symptoms in offspring at age of nine: A prospective study from Nicaragua. J Psychiatr Res. 2015;71:97–102. 10.1016/j.jpsychires.2015.09.016 [DOI] [PubMed] [Google Scholar]
- 65.Pires TDO, da Silva CMFP, de Assis SG. Association between family environment and attention deficit hyperactivity disorder in children: Mothers’ and teachers’ views. BMC Psychiatry. 2013;13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Sasaluxnanon C, Kaewpornsawan T. Risk factor of birth weight below 2,500 grams and attention deficit hyperactivity disorder in Thai children. J Med Assoc Thailand. 2005;88(11):1514–8. [PubMed] [Google Scholar]
- 67.Ramchandani PG, Richter LM, Norris SA, Stein A. Maternal prenatal stress and later child behavioral problems in an urban South African setting. J Am Acad Child Adolesc Psychiatry. 2010;49(3):239–47. [PubMed] [Google Scholar]
- 68.Santos IS, Matijasevich A, Barros AJD, Barros FC. Antenatal and postnatal maternal mood symptoms and psychiatric disorders in pre-school children from the 2004 Pelotas Birth Cohort. J Affective Disord. 2014;164:112–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Başgül SS, Etiler N, Çakin-Memik N, Coşkun A, Aǧaoǧlu B. Frequency and correlates of psychiatric disorders in early childhood: A study of population and clinical samples in Turkey. Turk J Pediatr. 2011;53(6):666–79. [PubMed] [Google Scholar]
- 70.Fan F, Zou Y, Tian H, Zhang Y, Zhang J, Ma X, et al. Effects of maternal anxiety and depression during pregnancy in Chinese women on children’s heart rate and blood pressure response to stress. J Hum Hypertens. 2016;30(3):171–6. 10.1038/jhh.2015.64 [DOI] [PubMed] [Google Scholar]
- 71.Rondó PHC, Rezende G, Lemos JO, Pereira JA. Maternal stress and distress and child nutritional status. Eur J Clin Nutr. 2013;67(4):348–52. 10.1038/ejcn.2013.28 [DOI] [PubMed] [Google Scholar]
- 72.Rosa MJ, Just AC, Tamayo y Ortiz M, Schnaas L, Svensson K, Wright RO, et al. Prenatal and postnatal stress and wheeze in Mexican children: Sex-specific differences. Ann Allergy Asthma Immunol. 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Wadhwa PD, Entringer S, Buss C, Lu MC. The contribution of maternal stress to preterm birth: Issues and considerations. Clin Perinatol. 2011;38(3):351–84. 10.1016/j.clp.2011.06.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Dunkel Schetter C, Tanner L. Anxiety, depression and stress in pregnancy: Implications for mothers, children, research, and practice. Curr Opin Psychiatry. 2012;25(2):141–8. 10.1097/YCO.0b013e3283503680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Moller AB, Petzold M, Chou D, Say L. Early antenatal care visit: A systematic analysis of regional and global levels and trends of coverage from 1990 to 2013. Lancet Glob Health. 2017;5(10):e977–e83. 10.1016/S2214-109X(17)30325-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.WHO (World Health Organization). Chronic diseases in low and middle income countries. Geneva: WHO, 2005. http://www.who.int/chp/chronic_disease_report/media/Factsheet3.pdf [Google Scholar]
- 77.Nations United. World mortality report. New York: United Nations, 2017. http://www.un.org/en/development/desa/population/publications/pdf/mortality/WMR2015/WMR2015_Highlights.pdf [Google Scholar]
- 78.Chersich M, Blaauw D, Dumbaugh M, Penn-Kekana L, Thwala S, Bijlmakers L, et al. Mapping of research on maternal health interventions in low- and middle-income countries: A review of 2292 publications between 2000 and 2012. Global Health. 2016;12(1):52 10.1186/s12992-016-0189-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hanlon C, Whitley R, Wondimagegn D, Alem A, Prince M. Between life and death: Exploring the sociocultural context of antenatal mental distress in rural Ethiopia. Arch Womens Ment Health. 2010;13(5):385–93. 10.1007/s00737-010-0149-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)
(DOCX)
(DOC)
(DOCX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files. The full history of the protocol and bibliographic research strategy is available via the PROSPERO International prospective register of systematic reviews, # CRD42016048634.