Abstract
Objective
To investigate the effect of MDA-19 on progression of melanoma, and explore the relevant mechanism.
Methods
The melanoma cell lines, M14 and UACC257, were treated with different concentrations of MDA-19, then CCK8, clone formation assay, Transwell and flow cytometry assays were performed to examine cell proliferation, migration, invasion and apoptosis, respectively. The expression of apoptosis-related proteins (Bcl-2, Bax and caspase 3 P17), EMT and signaling pathway-related proteins were also detected by Western blot.
Results
MDA-19 inhibited melanoma cells in a dose-dependent manner. Compared to the NC group, MDA-19 significantly inhibited cell growth capacity, migration and invasion of M14 and UACC257 cells, and accelerated cell apoptosis in a mitochondrial pathway through regulating Bcl-2/Bax and Caspase 3 in M14 and UACC257 cells. Moreover, MDA-19 was observed to up-regulate the expression of E-cad and down-regulate the expression of N-cad, Vimentin and Slug in melanoma cells in vitro. Furthermore, MDA-19 could inhibit the PI3K/Akt pathway by blocking Akt phosphorylation (p-Akt) and downstream proteins, P70 and Cyclin D1 in M14 and UACC257 cells.
Conclusion
Our data demonstrate that MDA-19 could inhibit progression of melanoma by suppressing the PI3K/Akt pathway, suggesting that MDA-19 is a potential anti-cancer agent for therapy of melanoma.
Keywords: MDA-19, progression, EMT, PI3K/Akt
1. Introduction
With high morbidity and mortality, melanoma has been shown to account for over 85% of skin cancer deaths, resulting in a serious socio-economic problem in the world [1, 2, 3]. In recent years, the incidence of malignant melanoma has been increasing with an annual growth rate of 3% - 7% [4]. Early melanoma can be cured by surgical resection, however, metastatic melanoma could be resistant to radiotherapy and chemotherapy in the event of dissemination, resulting in a high mortality in advanced patients [3, 5, 6, 7]. Therefore, it is necessary to find new therapeutic targets and strategies to effectively block the progression and metastasis of melanoma.
Cannabinoids have been revealed to have anti-tumorogenic activity for inhibiting tumor cell proliferation and promoting apoptosis [8, 9, 10]. Thence, cannabinoids has been forced as a potential chemotherapeutic agent for the therapy of cancer. The function of cannabinoids is carried out through cannabinoid receptor 1 (CB1) and CB2, G protein-coupled receptors. CB1 is primarily expressed in the brain, while CB2 is exclusively expressed in immune cells [10, 11]. CB1 and CB2 have been found to express in some tumor cells, including melanoma [12, 13, 14]. MDA-19, N'-[(3Z)-1-(1-hexyl)-2-oxo-1,2-dihydro-3H-indol-3-ylidene] benzohydrazide, a novel CB2 agonist, has been demonstrated to have the function of relieving neuropathic pain without potential adverse effect on the central nervous system [15]. Some synthetic CB1 and CB2 agonist are found to play a role in inhibiting the tumor growth and metastasis [12, 16, 17]. However, there are no studies available to explore the effect of MDA-19 on melanoma progression.
In the present study, we observed that MDA-19 treatment inhibited melanoma cells in a dose-dependent manner, and effectively inhibited cell proliferation, migration and invasion of melanoma M14 and UACC257 cells and promoted their apoptosis. Furthermore, our study found that the expression of marker proteins of epithelial-to-mesenchymal transition (EMT) was also modulated by MDA-19 in melanoma cells in vitro. More importantly, we substantiated that MDA-19 modulates the biological behavior of melanoma cells by suppressing the PI3K/Akt pathway. Based on these results, MDA-19 is a potentially effective anti-cancer agent for therapy of melanoma.
2. Materials and methods
2.1. Cells culture
The melanoma cell lines, M14 and UACC257, were obtained from the Cell Bank of the Chinese Academy of Sciences (China), and cultured in RPMI-1640 (HYCLONE, USA) medium containing 10% FBS (Gibco, USA) at 37°C with 5% CO2.
2.2. Dose-dependent assay
M14 and UACC257 cells were treated with different concentrations of MDA-19 (0, 1, 2, 4, 6, 8, 10, 20, 40, 60, 80, 100, and 200 μM), and CCK8 kit (Beijing solarbio science & technology, China) was performed to detect the absorbance after 24 h of incubation. Cells were cultured with CCK8 reagent (10 μl/well) at 37°C for 90 min before the detection.
2.3. Proliferation and viability assay
CCK8 and colony formation assays were performed to evaluate cell proliferation and viability of M14 and UACC257 cells. For CCK8 assay, about 1×103 cells were seeded in each well of 96-well plate and cultured for 24h. Cells were treated with MDA-19 for treatment group or DMSO for negative control (NC). Cell proliferation was detected by CCK8 kit every 24 h. For the colony formation assay, treated cells were seeded about 500 cells in each medium to culture at 37°C with 5% CO2 until cells formed sufficiently large colonies. Cells were fixed with 1 ml 4% paraformaldehyde for 30 min followed by staining with crystal violet for 30 min. The number of colonies were counted and pictures were taken.
2.4. Migration and invasion assay
For invasion assay, Transwell chamber coated with Matrigel were used for the cell invasion assay. Cells were treated with MDA-19 for treatment group or DMSO for NC and cultured for 24h. Treated cells were trypsinized and re-suspended in serum-free culture medium, and about 1×104 cells were transferred to the upper chamber, and complete medium was added to the lower chamber. After incubation for 24h, non-invading cells were removed and cells were fixed with 4% paraformaldehyde. Then, 0.1% crystal violet was used to stain cells for 20 min. The invaded cells were imaged and counted under the microscope.
For the migration assay, the method of migration assay was similar to the invasion assay except for the absence of Matrigel and the number of cells were 5×103.
2.5. Apoptosis assay
Flow cytometry was performed to examine cell apoptosis using Annexin V-FITC- PI apoptosis detection kit (4A Biotech, China). Briefly, after incubation with MDA-19 or DMSO for 24h, cells were treated with serum-free medium for 24h with starvation. After that, cells were re-suspended in 1× binding buffer (10 mM HEPES/NaOH [pH 7.4], 140 mM NaCl, 2.5 mM CaCl2) at a density of 1-5×106 cells/ml. 100 μl cell suspension was stained with 5 μl of Annexin V-FITC for 5 min incubation in the dark, and then cultured with 10 μl of PI for 2min. The apoptosis rate was analyzed by a flow cytometer (BD FACSC anto II, BD Biosciences, USA), and calculated by BD FACSDiva software.
2.6. Western blot assay
After incubation with MDA-19 or DMSO for 48h, cells were collected and lysed using ice-cold RIPA Lysis Buffer (CWBIO, China), then BCA Protein Assay Kit (Beyotime, China) was performed to detect protein concentration. 20 μg of protein of each sample was loaded to each lane and resolved by 10% 24 SDS-PAGE gel. Thereafter, protein was transferred to a PVDF membrane (Millipore, Bedford, USA) and blocked with 5% non-fat milk for 1 h at room temperature. Then, the PVDF membrane was incubated with primary antibodies at 4°C overnight. After washing with TBST 3 times, the membrane was incubated with the secondary antibody for 1h. After the membrane was washed, chemiluminescence detection kit was used for signal development. The primary antibodies used were as followed: anti-Bcl-2 (1:1000), anti-Bax (1:1000), anti-Caspase3 P17 (1:1000), anti-E-cad (1:1000), anti-N-cad (1:1000), anti-Vimentin (1:1000), anti-Slug (1:1000), anti-VEGF (1:1000), anti-p-P70 (1:1000), anti-Cyclin D1 (1:1000) and anti-GAPDH (1:5000) were obtained from PTG Company (USA); anti-Akt (1:1000) and anti-p-Akt (1:1000) were obtained from Cell Signaling Technology (USA). All secondary antibodies (1:5000) were obtained from PTG Company (USA).
2.7. Statistical analysis
In this study, SPSS 18.0 statistical software was used to carry out statistical analysis. The experiment was conducted at least three times. Student’s t test was used to analyze differences between two groups, and differences were considered statistically significant for values of P<0.05.
3. Results
3.1. MDA-19 inhibits melanoma cells in a dose-dependent manner
In order to identify whether MDA-19 affects the cellular functions of melanoma cells, different concentrations of MDA-19 were used to treat melanoma cell lines, M14 and UACC257. Our results showed that MDA-19 had no effect on survival of M14 cells at the concentration lower than 10 μM. At the concentration of 20 μM or higher, MDA-19 had a significant inhibitory effect on the viability of M14 cells in a dose-dependent manner (Figure 1A). Similar results were also shown in UACC257 cells (Figure 1B). The IC50 of MDA-19 was 36.3 μM for M14 cells, and was 24.2 μM for UACC257 cells, and 20 μM or 10 μM of MDA-19 was used to treat M14 and UACC257 cells for the rest experiments, respectively. The results suggested that MDA-19 might have an inhibitory effect on melanoma cells.
Figure 1.
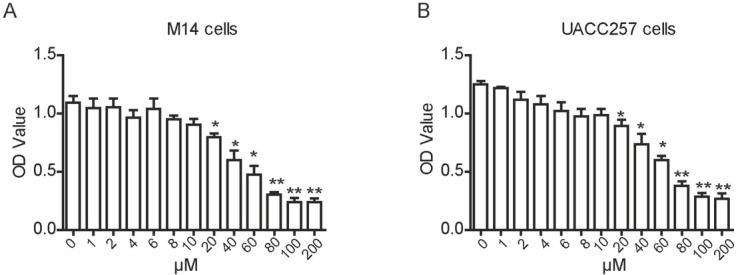
MDA-19 inhibits melanoma cells in a dose-dependent manner. (A) (B) M14 cell (A) and UACC257 cell (B) were treated with different concentrations of MDA-19 for 24h, the absorbance was detected using CCK8 kit. Data are expressed as the mean ± SD. *P<0.05, **P<0.01.
3.2. MDA-19 inhibits the viability and proliferation of melanoma cells
To substantiate the inhibition of MDA-19 on the growth of melanoma, CCK8 and colony formation assays were performed after MDA-19 treatment. We found that compared with the NC group, the proliferation rate was significantly decreased in M14 and UACC257 cells which was treated with MDA-19 for 48 h (Figure 2A and C). The inhibitory effects of MDA-19 on the proliferation of M14 and UACC257 cells were still significant with the treatment of 72 h (P<0.05, Figure 2A and C). Moreover, results of colony formation assay showed that MDA-19 treatment significantly reduced the number of colonies compared with the negative control (P<0.05, Figure 2C and D). Above all, these results suggested that MDA-19 treatment could inhibit the viability and proliferation of melanoma cells in vitro.
Figure 2.
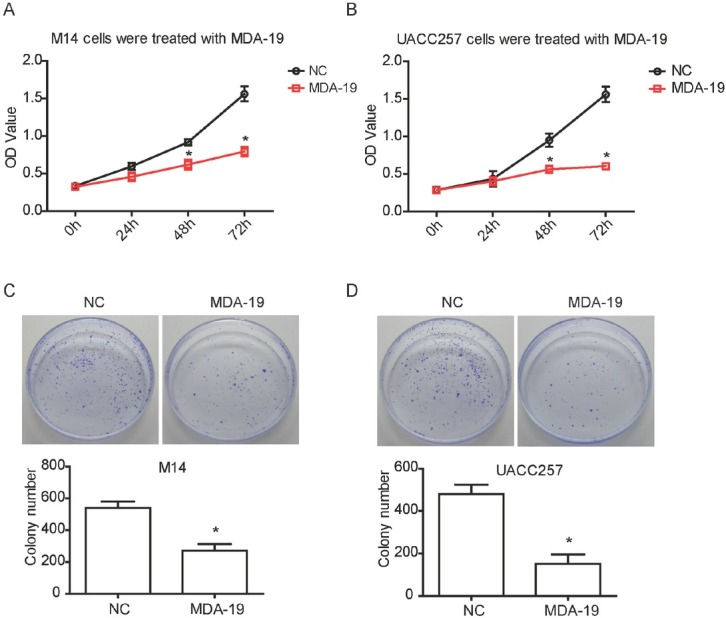
MDA-19 inhibits the viability and proliferation of melanoma cells in vitro. M14 cells was treated with 20 μM of MDA-19, and UACC257 cells was treated with 10 μM of MDA-19. (A) (B) M14 cell (A) and UACC257 cell (B) were treated with MDA-19 for 0, 24, 48, 72 h, and the absorbance was detected using CCK8 kit. (C) (D) M14 cell (C) and UACC257 cell (D) were treated with MDA-19 for colony formation assay. Data are expressed as the mean ± SD. MDA-19: MDA-19 treated group; NC: DMSO treated group, negative control. *P<0.05.
3.3. MDA-19 attenuates migration and invasion of melanoma cells
To further assess the effect of MDA-19 on the metastasis of melanoma cell, 20μM or 10μM of MDA-19 was used to treat M14 and UACC257 cells, respectively. Transwell assay
revealed that migratory capacity of M14 and UACC257 cells were both significantly decreased by MDA-19 treatment compared to non-treated cells (P<0.05, Figure 3A and B). A significant decrease of invasion capacity was also validated in MDA-19 treated M14 and UACC257 cells by Transwell invasion assay (P<0.05, Figure 3A and B). Thus, it was concluded that treatment of MDA-19 may reduce cell mobility of melanoma.
Figure 3.
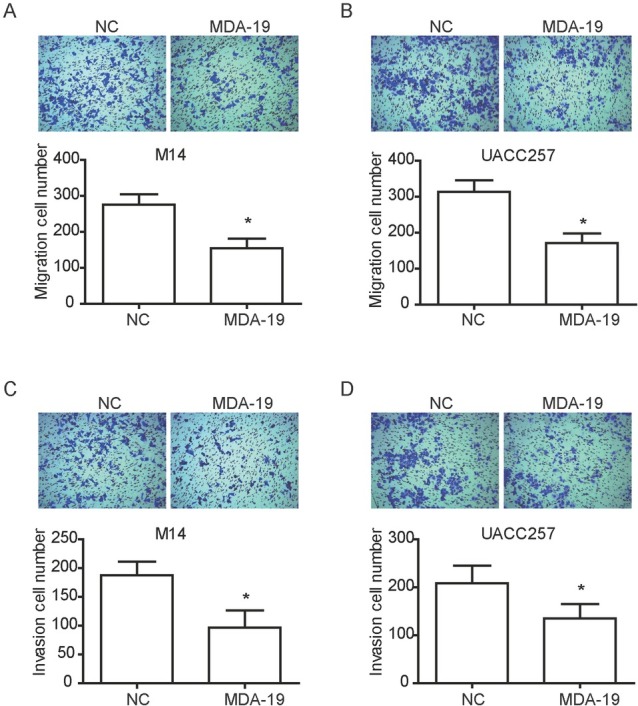
MDA-19 attenuates migration and invasion of melanoma cells in vitro. M14 cells was treated with 20 μM of MDA-19, and UACC257 cells was treated with 10 μM of MDA-19. (A) (B) After treatment with MDA-19 for 24h, Transwell assay was performed to examine cell migration capability in M14 cell (A) and UACC257 cell (B). (C) (D) After treatment with MDA-19 for 24h, Transwell assay was performed to examine cell invasion capability in M14 cell (C) and UACC257 cell (D). Data are expressed as the mean ± SD. MDA-19: MDA-19 treated group; NC: DMSO treated group, negative control. *P<0.05.
3.4. MDA-19 induces apoptosis of melanoma cells
In order to further determine the effect of MDA-19 on cell survival of melanoma, cell apoptosis was examined by using flow cytometry in M14 and UACC257 cells. As shown in Figure 4A, MDA-19 significantly promoted apoptosis in M14 and UACC257 cells compared to non-treated cells (P<0.05).
Figure 4.
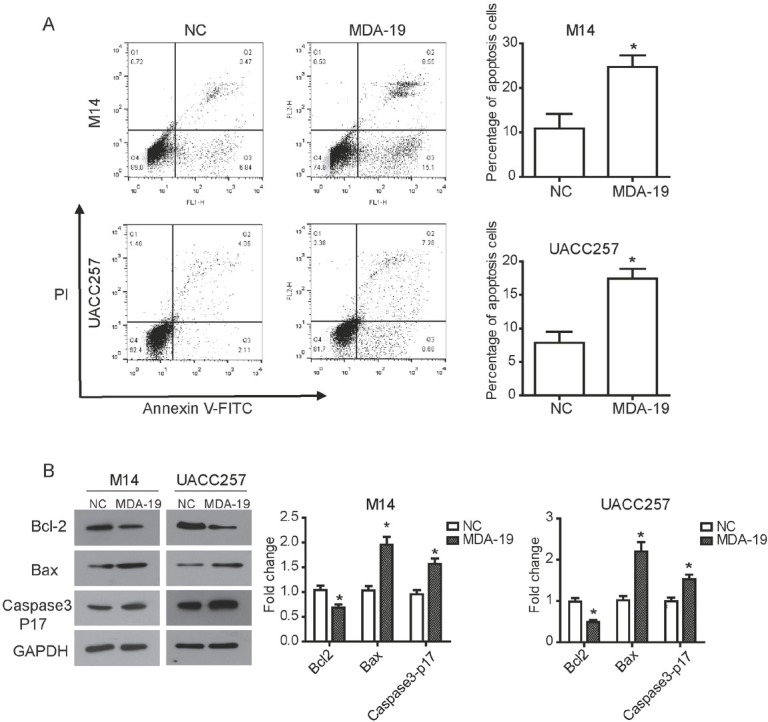
MDA-19 induces apoptosis of melanoma cells in vitro. M14 cells was treated with 20 μM of MDA-19, and UACC257 cells was treated with 10 μM of MDA-19. (A) After treatment with MDA-19 for 24h, flow cytometry assay was performed to detect cell apoptosis in M14 cell (upper) and UACC257 cell (below) using Annexin V-FITC- PI apoptosis detection kit. (B) After treatment with MDA-19 for 48h, Western blot was used to examine expression of related proteins in M14 cell (left) and UACC257 cell (right). The results showed that expression of Bcl-2 was decreased by MDA-19, expression of Bax and Caspase3 P17 were significantly up-regulated. Data are expressed as the mean ± SD. MDA-19: MDA-19 treated group; NC: DMSO treated group, negative control. *P<0.05.
The expression of apoptosis-related proteins were examined by western blot assay to investigate the relevant mechanism of increased apoptosis mediated by MDA-19. As expected, the expression of Bcl-2 (an anti-apoptotic protein) was decreased in MDA-19 treated M14 cells, while the expression of Bax (a pro-apoptotic protein) and Caspase 3 P17 (an apoptotic executioner) were up-regulated by MDA-19 treatment in M14 cells (P<0.05, Figure 4B). Similar results were found in UACC257 cells. Collectively, these data suggested that MDA-19 could promote cell apoptosis by regulating apoptosis-related proteins in melanoma.
3.5. MDA-19 impairs mesenchymal properties of melanoma cells
EMT is a pivotal mechanism involved in modulating cell migration and invasion. Therefore, various EMT markers were detected to explore the mechanism of the suppression induced by MDA-19. Our data revealed that the expression of E-cad was significantly up-regulated by MDA-19 in M14 and UACC257 cells (P<0.05, Figure 5A). MDA-19 treatment also led to a significant decreased expression of N-cad, Vimentin and Slug in M14 and UACC257 cells (P<0.05, Figure 5A). These results suggested that MDA-19 could impair mesenchymal properties of melanoma cells in vitro.
Figure 5.
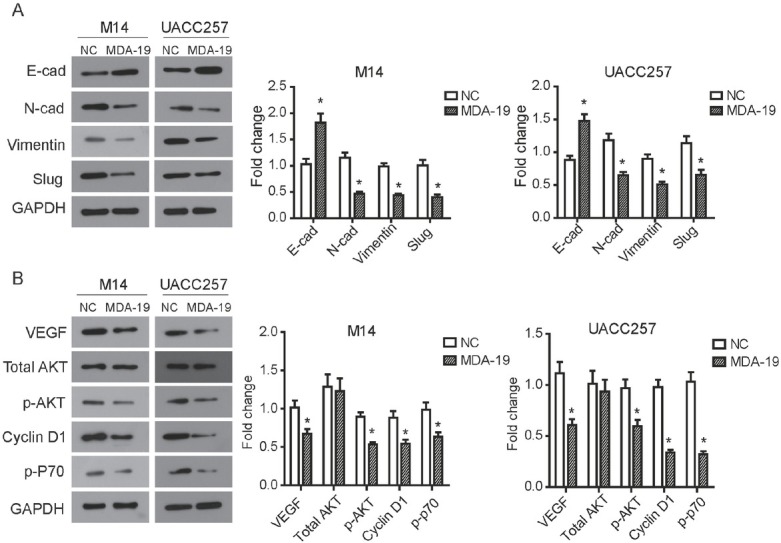
MDA-19 suppresses EMT process and PI3K/Akt pathway in melanoma cells. M14 cells was treated with 20 μM of MDA-19, and UACC257 cells was treated with 10 μM of MDA-19. After treatment with MDA-19 for 48h, Western blot was used to examine expression of related proteins in M14 cell (left) and UACC257 cell (right). (A) The results showed that expression of E-cad was significantly increased, while expression of N-cad, Vimentin and Slug were decreased by MDA-19. (B) The results showed that MDA-19 reduced phosphorylation levels of Akt (p-Akt), and the expression of the downstream proteins Cyclin D1 and p-P70 were also decreased. Data are expressed as the mean ± SD. MDA-19: MDA-19 treated group; NC: DMSO treated group, negative control. *P<0.05.
3.6. MDA-19 inactivates PI3K/Akt pathway in melanoma cells
It is widely held that the phosphatidylinositol-3-kinase/protein kinase B (PI3K/Akt) is involved in tumor cell proliferation, apoptosis, migration and malignant transformation [18]. Herein, PI3K/Akt pathway was examined to explore the relevant mechanism of MDA-19 blocking
proliferation and mobility of M14 and UACC257 cells. We noticed that the phosphorylation levels of Akt (p-Akt) was significantly inhibited by MDA-19 treatment (P<0.05), while the amount of total Akt was not changed both in M14 and UACC257 cells (Figure 5B). The western blot results also demonstrated that the expression of the important proteins Cyclin D1 and p-P70 in the downstream of PI3K/Akt was also reduced accordingly with the treatment of MDA-19 (P<0.05, Figure 5B). In addition, we found that treatment with MDA-19 obviously suppressed the expression of vascular endothelial growth factor (VEGF) (P<0.05, Figure 5B). Taken together, our data indicated that MDA-19 regulates the biological behavior of melanoma cells through interfering with the activation status of PI3K/Akt pathway.
4. Discussion
Melanoma is one of the most aggressive skin cancers, characterized by locoregional invasiveness and early distant dissemination [19, 20]. Therefore, blocking tumor metastasis and invasion is an effective method for the therapy of melanoma. Recent studies have focused on the role of CB receptors in antitumor progression. Above all, we validated that MDA-19, a small-molecule CB2 agonist, had a dose-dependent inhibition for melanoma cells in vitro. Then, the anti-cancer effect of MDA-19 on progression and invasion of melanoma was examined. As expected, MDA-19 significantly inhibited cell viability and proliferation, migration and invasion of melanoma cells M14 and UACC257, and induced their apoptosis, suggesting that MDA-19 plays the role of anti-cancer agent in the progression and motility of melanoma. In addition to MDA-19, other CB receptors agonist have also been reported to exert suppression on tumor growth and metastasis [12, 16, 17]. Blazquez C et al. reports that THC and WIN-55,212–2, the CB1 and CB2 agonist, obviously inhibit cell proliferation of melanoma cell lines B16 and A375 [12]. In breast cancer, the CB2 synthetic agonist JWH-133 and WIN-55,212–2 also have a similar inhibitory effect on the suppression of cell proliferation and migration [16]. Based on these research, CB agonists might be an effective way for melanoma therapy, and MDA-19 is a useful small molecular anti-cancer agent.
Deregulated apoptosis is one of the hallmarks of tumor cells and regulation of apoptosis is one of the mechanisms for survival of malignant melanoma cells. We found that MDA-19 treatment significantly promoted rate of apoptosis in M14 and UACC257 cells, further study was performed to elucidate the mechanism underlying the elevated apoptosis. Apoptosis involves a series of proteolytic events, which mainly mediated by the important family of cysteine proteases, including the activated caspase-3, a pivotal executioner to trigger apoptosis [21]. The Bcl-2 family plays an essential role in apoptosis induction by triggering the mitochondrial pathway which is one of apoptosis mechanisms [22, 23]. Bcl-2 inhibits apoptosis by combining with Bax, and up-regulation of Bcl-2 has been revealed in a variety of tumors, including melanoma. The present study revealed that the expression of Bcl-2 was down-regulated by MDA-19, meanwhile the expression of Bax and Caspase 3 P17 were up-regulated, suggesting that apoptosis promoted by MDA-19 is triggered by the mitochondrial pathway.
EMT is a crucial mechanism to promote tumor cell invasion and metastasis. The reduction of E-cad, a significant marker of epithelial cell, is an effective hallmark of EMT. Reduced E-cad accumulation has been confirmed frequently in melanoma [20]. Apart from this, N-cad, Vimentin and Snail are three mesenchymal markers commonly used to reflect EMT progression [24]. Slug, also known as Snail2, is a member of Snail family and a key transcription factor in EMT progress. Slug plays a role as repressor in E-cad transcription by binding the E-box element in the E-cad promoter [25, 26]. Thus, further investigation of EMT markers was carried out in the present study. We observed that MDA-19 could up-regulate the expression of epithelial marker E-cad and down-regulate the expression of mesenchymal markers N-cad, Vimentin and Slug in melanoma cells in vitro, suggesting that MDA-19 is involved in the regulation of the EMT process, and MDA-19 might be a potential small molecule agent that inhibits tumor metastasis.
The relevant mechanism underlying the anit-cancer effects of MDA-19 on growth, metastasis and survival of melanoma cells need to be elucidated. It is demonstrated that PI3K/Akt pathway is a vital pathway function in various tumor cellular processes, including cell proliferation, apoptosis, survival and metastasis [27, 18]. Aberrant excessively activated PI3K/Akt pathway is frequently observed in cancers, even in melanoma [28]. There is growing evidence that inhibition of PI3K/Akt pathway achieves anti-tumor activity and improves cell sensitivity to cisplatin and temozolomide [29, 30, 31], suggesting inhibition of PI3K/Akt pathway is a novel strategy for therapy of melanoma. Hence, further study was focused on the PI3K/Akt pathway, and the effect of MDA-19 on PI3K/Akt pathway was examined in melanoma cells. Our data showed that MDA-19 suppressed PI3K/Akt pathway by inhibiting Akt phosphorylation in melanoma cells. Moreover, the downstream proteins were subsequently down-regulated by MDA-19. Cyclin D1 is a key regulator in cell cycle, its overexpression has been confirmed to be associated with vigorous cell division of tumor cells [32, 33]. P70 (p70 ribosomal S6 kinase, p70S6K) possess ribosome protein kinase activity. Activated P70 by mTOR could promote the synthesis of ribosome and other regulatory proteins, mediating cell growth and proliferation. It has been demonstrated that overexpression of P70 could promote tumor cell proliferation and angiogenesis [34, 35]. Furthermore, we also found that MDA-19 also led to a significant decrease in the expression of VEGF, a pivotal regulator in tumor angiogenesis at upstream of the PI3K/Akt pathway. Based on these studies, it is the suppression effect of MDA-19 on the PI3K/Akt pathway that contributes to the inhibition on proliferation in melanoma in vitro.
Together, the present study underscore MDA-19 as a potential anti-cancer agent for therapy of melanoma through inhibiting the PI3K/Akt pathway.
Footnotes
Conflict of interest
Conflict of interest statement: Authors state no conflict of interest.
Funding: This study was supported by China Postdoctoral Science Foundation (CPSF) (No.2014M550370, 2015T80740) and Shandong Provincial Natural Science Foundation, China No. ZR2017MH074).
Reference
- [1].Mackie RM, Hauschild A, Eggermont AMM. Epidemiology of invasive cutaneous melanoma. Annals of Oncology Official Journal of the European Society for Medical Oncology. 2009;20(6) doi: 10.1093/annonc/mdp252. (suppl_6):vi1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Rastrelli M, Tropea S, Rossi CR, Alaibac M. Melanoma: epidemiology, risk factors, pathogenesis, diagnosis and classification. Vivo. 2014;28(12):1005–1011. [PubMed] [Google Scholar]
- [3].Wang Y, Zhang G, Jin J, Degan S, Tameze Y, Zhang JY. MALT1 promotes melanoma progression through JNK/c-Jun signaling. Oncogenesis. 2017;6(7):e365. doi: 10.1038/oncsis.2017.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Hagen B, Trinh VA. Managing Side Effects of Vemurafenib Therapy for Advanced Melanoma. 2014;5(6):400–410. [PMC free article] [PubMed] [Google Scholar]
- [5].Gorayski P, Burmeister B, Foote M. Radiotherapy for cutaneous melanoma: current and future applications. Future Oncology. 2015;11(3):525–534. doi: 10.2217/fon.14.300. [DOI] [PubMed] [Google Scholar]
- [6].Lancet T. Melanoma research gathers momentum. Lancet. 2015;385(9985):2323. doi: 10.1016/S0140-6736(15)61087-X. [DOI] [PubMed] [Google Scholar]
- [7].Chen X, Hao D, Sha L, Li Y, Yan D, Yao X. Long noncoding RNA MHENCR promotes melanoma progression via regulating miR-425/489-mediated PI3K-Akt pathway. American Journal of Translational Research. 2017;9(1):90. [PMC free article] [PubMed] [Google Scholar]
- [8].Carracedo A, Gironella M, Lorente M, Garcia S, Guzmán M, Velasco G. Cannabinoids induce apoptosis of pancreatic tumor cells via endoplasmic reticulum stress-related genes. Cancer Research. 2006;66(13):6748–6755. doi: 10.1158/0008-5472.CAN-06-0169. [DOI] [PubMed] [Google Scholar]
- [9].Casanova ML, Blázquez C, Martínez-Palacio J, Villanueva C, Fernández-Aceñero MJ, Huffman JW. Inhibition of skin tumor growth and angiogenesis in vivo by activation of cannabinoid receptors. Journal of Clinical Investigation. 2003;111(1):43–50. doi: 10.1172/JCI16116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Sarfaraz S, Adhami VM, Syed DN, Afaq F, Mukhtar H. Cannabinoids for cancer treatment: progress and promise. Cancer Research. 2008;68(2):339–342. doi: 10.1158/0008-5472.CAN-07-2785. [DOI] [PubMed] [Google Scholar]
- [11].Guzmán M.. Cannabinoids: potential anticancer agents. Nature Reviews Cancer. 2003;3(10):745. doi: 10.1038/nrc1188. [DOI] [PubMed] [Google Scholar]
- [12].Blázquez C, Carracedo A, Barrado L, Real PJ, Fernándezluna JL, Velasco G. Cannabinoid receptors as novel targets for the treatment of melanoma. Faseb Journal Official Publication of the Federation of American Societies for Experimental Biology. 2006;20(14):2633. doi: 10.1096/fj.06-6638fje. [DOI] [PubMed] [Google Scholar]
- [13].Sarfaraz S, Afaq F, Adhami VM, Malik A, Mukhtar H. Cannabinoid Receptor Agonist-induced Apoptosis of Human Prostate Cancer Cells LNCaP Proceeds through Sustained Activation of ERK1/2 Leading to G1 Cell Cycle Arrest. Journal of Biological Chemistry. 2006;281(51):39480. doi: 10.1074/jbc.M603495200. [DOI] [PubMed] [Google Scholar]
- [14].Xu X, Liu Y, Huang S, Liu G, Xie C, Zhou J. Overexpression of cannabinoid receptors CB1 and CB2 correlates with improved prognosis of patients with hepatocellular carcinoma. Cancer Genetics & Cytogenetics. 2006;171(1):31–38. doi: 10.1016/j.cancergencyto.2006.06.014. [DOI] [PubMed] [Google Scholar]
- [15].Xu JJ, Diaz P, Astrucdiaz F, Craig S, Munoz E, Naguib M. Pharmacological characterization of a novel cannabinoid ligand, MDA19, for treatment of neuropathic pain. Anesthesia & Analgesia. 2010;111(1):99–109. doi: 10.1213/ANE.0b013e3181e0cdaf. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Qamri Z, Preet A, Nasser MW, Bass CE, Leone G, Barsky SH. Synthetic cannabinoid receptor agonists inhibit tumor growth and metastasis of breast cancer. Molecular Cancer Therapeutics. 2009;8(11):3117. doi: 10.1158/1535-7163.MCT-09-0448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Sarnataro D, Pisanti S, Santoro A, Gazzerro P, Malfitano AM, Laezza C. The cannabinoid CB1 receptor antagonist rimonabant (SR141716) inhibits human breast cancer cell proliferation through a lipid raft-mediated mechanism. Molecular Pharmacology. 2006;70(4):1298–306. doi: 10.1124/mol.106.025601. [DOI] [PubMed] [Google Scholar]
- [18].Lassen A, Atefi M, Robert L, Wong DJ, Cerniglia M, Cominanduix B. Effects of AKT inhibitor therapy in response and resistance to BRAF inhibition in melanoma. Molecular Cancer. 2014;13(1):83. doi: 10.1186/1476-4598-13-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Lazovich D, Choi K, Vogel RI. Time to get serious about skin cancer prevention. Cancer epidemiology, biomarkers & prevention : a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2012;21(11):1893. doi: 10.1158/1055-9965.EPI-12-0327. [DOI] [PubMed] [Google Scholar]
- [20].Tang L, Yi XM, Chen J, Chen FJ, Lou W, Gao YL. Ubiquitin ligase UBE3C promotes melanoma progression by increasing epithelial-mesenchymal transition in melanoma cells. Oncotarget. 2016;7(13):15738–15746. doi: 10.18632/oncotarget.7393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Gui D, Guo Y, Feng W, Wei L, Chen J, Chen Y. Astragaloside IV, a Novel Antioxidant, Prevents Glucose-Induced Podocyte Apoptosis In Vitro and In Vivo. Plos One. 2012;7(6):e39824. doi: 10.1371/journal.pone.0039824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Martinou JC, Youle RJ. Mitochondria in Apoptosis: Bcl-2 Family Members and Mtochondrial Dynamics. Developmental Cell. 2011;21(1):92. doi: 10.1016/j.devcel.2011.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Wu R, Tang S, Wang M, Xu X, Yao C, Wang S. MicroRNA-497 Induces Apoptosis and Suppresses Proliferation via the Bcl-2/Bax-Caspase9-Caspase3 Pathway and Cyclin D2 Protein in HUVECs. Plos One. 2016;11(12):e0167052. doi: 10.1371/journal.pone.0167052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Xiao C, Wu CH, Hu HZ. LncRNA UCA1 promotes epithelial-mesenchymal transition (EMT) of breast cancer cells via enhancing Wnt/beta-catenin signaling pathway. European Review for Medical & Pharmacological Sciences. 2016;20(13):2819. [PubMed] [Google Scholar]
- [25].Bolós V, Peinado H, Pérez-Moreno MA, Fraga MF, Esteller M, Cano A.. The transcription factor Slug represses E-cadherin expression and induces epithelial to mesenchymal transitions: a comparison with Snail and E47 repressors. Journal of Cell Science. 2003;116(3):499–511. doi: 10.1242/jcs.00224. [DOI] [PubMed] [Google Scholar]
- [26].Yi P, Jing L, Zhang Y, Nan W, Liang H, Yuan L. Slug-up- regulated miR-221 promotes breast cancer progression through suppressing E-cadherin expression. Scientific Reports. 2016;6:25798. doi: 10.1038/srep25798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Kwong LN, Davies MA. Navigating the therapeutic complexity of PI3K pathway inhibition in melanoma. Clinical Cancer Research An Official Journal of the American Association for Cancer Research. 2013;19(19):5310–5319. doi: 10.1158/1078-0432.CCR-13-0142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Davies MA. The role of the PI3K-AKT pathway in melanoma. Cancer Journal. 2012;18(2):142–147. doi: 10.1097/PPO.0b013e31824d448c. [DOI] [PubMed] [Google Scholar]
- [29].Niessner H, Schmitz J, Tabatabai G, Schmid A, Calaminus C, Sinnberg T. PI3K pathway inhibition achieves potent antitumor activity in melanoma brain metastases in vitro and in vivo. Clinical Cancer Research. 2016;22(23) doi: 10.1158/1078-0432.CCR-16-0064. [DOI] [PubMed] [Google Scholar]
- [30].Aziz SA, Jilaveanu LB, Zito C, Camp RL, Rimm DL, Conrad P. Vertical targeting of the phosphatidylinositol-3 kinase (PI3K) pathway as a strategy for treating melanoma. Clinical Cancer Research An Official Journal of the American Association for Cancer Research. 2010;16(24):6029. doi: 10.1158/1078-0432.CCR-10-1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Sinnberg T, Lasithiotakis K, Niessner H, Schittek B, Flaherty KT, Kulms D. Inhibition of PI3K-AKT-mTOR signaling sensitizes melanoma cells to cisplatin and temozolomide. Journal of Investigative Dermatology. 2009;129(6):1500–1515. doi: 10.1038/jid.2008.379. [DOI] [PubMed] [Google Scholar]
- [32].Diehl JA. Cycling to Cancer with Cyclin D1. Cancer Biology & Therapy. 2002;1(3):226. doi: 10.4161/cbt.72. [DOI] [PubMed] [Google Scholar]
- [33].Qie S, Diehl JA. Cyclin D1, cancer progression, and opportunities in cancer treatment. Journal of Molecular Medicine. 2016;94(12):1–14. doi: 10.1007/s00109-016-1475-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Tavares MR, Pavan IC, Amaral CL, Meneguello L, Luchessi AD, Simabuco FM. The S6K protein family in health and disease. Life Sciences. 2015;131:1. doi: 10.1016/j.lfs.2015.03.001. [DOI] [PubMed] [Google Scholar]
- [35].Bian CX, Shi Z, Meng Q, Jiang Y, Liu LZ, Jiang BH. P70S6K 1 regulation of angiogenesis through VEGF and HIF-1alpha expression. Biochemical & Biophysical Research Communications. 2010;398(3):395–399. doi: 10.1016/j.bbrc.2010.06.080. [DOI] [PMC free article] [PubMed] [Google Scholar]


