Abstract
Intrahepatic cholestasis of pregnancy (ICP) is a pregnancy-specific liver disorder which typically commences in the late second or third trimester and resolves within 48 hours after delivery. It is characterized by mild to severe pruritus, without any specific dermatologic features, elevated liver enzymes and increased serum bile acids (BA). The etiology of ICP is still not completely explicit. Pathogenesis includes a combination of hormonal and environmental factors superimposing on a genetic predisposition. During recent years increasingly ICP is recognized to be associated with an abnormal metabolic profile, including glucose intolerance and dyslipidemia, although it is considered to be secondary to maternal aberrant BA homeostasis. This article reviews the recent literature data and current concepts for ICP, with emphasis on a possibility of metabolic disorders being primary causative factors in ICP pathogenesis.
Keywords: intrahepatic cholestasis, pregnancy, bile acid, metabolic syndrome, irisin
Introduction
Intrahepatic cholestasis of pregnancy (ICP), also described as obstetric cholestasis or hepatosis gestationalis, is the most common pregnancy-specific liver disease. This reversible form of cholestasis typically presents in the late second or third trimester with otherwise unexplained itching and elevated serum total bile acid (TBA) level, as well as abnormal serum liver tests. All of the symptoms cannot be explained by other liver disease and usually dissolve within 48 hours following the delivery of the fetus [1, 2]. It is the second most frequent cause of jaundice in pregnancy following viral hepatitis [3]. The frequency of ICP varies widely with ethnicity and geographic location between 0.1% and 25%. It is considerably more frequent in South Asian (0.8-1.46%) and South American populations with the greatest prevalence up to 25% in women of Araucanian Indian [2, 4]. More recently, this has fallen to approximately 1.5-4% due to revision of the diagnostic criteria [5]. In Europe, the prevalence is 0.5% to 1.5% of all pregnancies, with a higher incidence in Scandinavian countries (1.5% in Sweden) [6]. The recurrence rate has been reported to be between 40-60%.
Several studies mention such risk factors related to ICP as hepatitis C virus (HCV) infection, advanced age, multiple gestations, low selenium (Se) and vitamin D levels [7]. Higher risk of development of ICP has been extensively described among HCV-positive women. Locatelli et al. [8] improved significantly higher incidence rate of cholestasis in women whose HCV antibodies were positive compared to those who were negative (15.9% vs. 0.8%, p < 0.001). Hepatitis C virus infection was also associated with earlier onset of ICP symptoms. More recently, similar conclusions were presented in study analyzing data of women with births between 1973 and 2009, registered in the Swedish Medical Birth Registry [9]. Wijarnpreecha et al. [10] demonstrated in meta-analysis not only a higher risk of ICP among HCV-infected pregnant women but also an increased risk of later HCV infection among ICP patients suggesting potential benefits of screening for hepatitis C in women with signs of ICP.
Also women of relatively advanced age (over 35 years) were at increased risk of developing intrahepatic cholestasis [11]. There is a higher incidence of ICP in women with a multiple pregnancies (14% in twin and 43% in triplet pregnancies). This condition is explained by higher levels of estrogens in multiple pregnancies in comparison with singleton pregnancies [12]. Wikström et al. [13] found that ICP women had significantly lower levels of 1,25-D3 in serum than healthy women (76.4 vs. 112.0 ng/l, p = 0.004) at delivery in spite of the fact that parathormone levels did not differ between this two groups.
It has also been suggested that ICP patients had significantly lower Se plasma levels than non-ICP pregnant women. Additionally copper plasma levels correlated with the biochemical severity of the disease [14]. The definite role of Se bile secretion has yet to be elucidated, although the role of Se as a co-factor of enzymes in oxidative metabolism in the liver may explain the impact on ICP pathogenesis. ICP is considered to be more common in the winter months. This conclusion is based on studies including women populations of Finland, Sweden or Chile [14-16] and it is strictly related to natural lower levels of selenium and vitamin D during winter.
The etiology of ICP is still not completely explicit. Most of the publications emphasize complex and multifactorial character of this condition. Pathogenesis includes a combination of hormonal and environmental factors superimposing on a genetic predisposition [17].
Maternal course of ICP is mostly mild, except an aggravating pruritus, without any severe long-range complications. However, it is necessary to highlights the possibility of postpartum bleeding as the leading causes of maternal morbidity. Contrarily, fetal outcomes can be much worse, including a 3- to 5-fold increased risk of fetal death in utero [18]. Furthermore, ICP is associated with increased risk of such fetal morbidities as meconium staining of amniotic fluid, cardiotocography (CTG) abnormalities, spontaneous preterm labor and respiratory distress syndrome that is independent risk factor of premature birth [2]. The fetal complications in intrahepatic cholestasis of pregnancy are believed to be related to high levels of bile acids (BA) in the fetal serum. Glantz et al. [19] found increase in fetal risk in ICP patients with BA levels > 40 µmol/l. Obtained results confirmed this hypothesis. Moreover, one of the studies presented direct evidence that ICP can program metabolic disease in the offspring, showing altered metabolic phenotypes in 16-year-old teenagers from ICP mothers who were not diabetic or lean/obese. Papacleovoulou et al. [20] analyzed the Northern Finland birth cohort 1985-1986 database to learn whether ICP affects metabolic health of children. They found significantly increased BMI and fasting insulin in males compared with their contemporaries of normal pregnancies. In females, in relation to females from normal pregnancies, increased hip girth and waist girth with decreased fasting HDL cholesterol were noticed.
The present article reviews the recent literature data and current concepts for ICP, with emphasis on an association between metabolic disorders and pathogenesis of ICP.
Clinical presentation
Intrahepatic cholestasis of pregnancy is characterized by mild to severe pruritus (an unpleasant sensation of the skin that provokes the desire to scratch) starting in the late second or third trimester, with up to 80% of women presenting after 30 weeks of gestation. It is the most common symptom and sometimes the only clinical manifestation of cholestasis. The pruritus classically affects the palms and soles, and gets worse at night, leading to insomnia and fatigue afterwards. There are no specific dermatologic features associated with ICP, apart from excoriations due to scratching, which are rather common [2, 17].
Clinical jaundice is rare, affecting approximately 14-25% of pregnant women with ICP, and if it does occur, it tends to be mild [21].
Women with ICP may have systemic symptoms of cholestasis, including dark urine and steatorrhea due to fat malabsorption.
Diagnosis
The diagnosis of ICP is partially a diagnosis of exclusion, because it is essential to exclude other conditions which are included in the differential diagnosis of cholestasis and hepatic disease. Liver function tests are to be performed in every pregnant woman who experiences pruritus. The increase of serum BA levels in combination with typical pruritus is highly suggestive of the diagnosis of ICP.
The most sensitive and specific marker for diagnosis is the serum TBA level, with increase concentration > 11 µmol/l to be indicative of ICP. Furthermore, characteristic rise in cholic acid (CA) and a decline in chenodeoxycholic acid (CDCA) levels leading to a marked elevation in the CA/CDCA ratio may be detected. Reduction of glycine/taurine ratio may also be present [17, 22]. Another laboratory abnormality which may occur even before the rise of TBA is the elevation of liver transaminase activities. A mild elevation in liver enzymes may be detected in up to 60% of the subjects. Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities rarely exceed two times the upper limits of norm in pregnancy, but it may also raised up to 30-fold [2, 17]. An elevated γ-glutamyltransferase (GGT) activity occurs in less than one-third of patients with ICP and may give insight into the genetic etiology of disease. Serum bilirubin levels may be elevated (rarely reaches 6 mg/dl), alkaline phosphatase, physiologically raised during pregnancy, is higher than expected (around four times of normal). Liver biopsy is not routinely required and it is pursued only in cases of jaundice with no pruritus, the beginning of symptoms before 20 week of gestation, and sustained abnormal laboratory findings beyond 8 weeks after delivery [23].
Etiology and pathophysiology
The etiology of ICP is influenced by a combination of genetic, endocrine, and environmental factors. The evidence, that reproductive hormones are included in the etiology of ICP, is provided in several studies. The disease is more frequent in the case of higher circulating estrogen and progesterone levels, e.g. multiple pregnancies (20.9% in multiple vs. 4.7% in singleton pregnancies) [23]. Additionally, Williamson et al. [24] presented that some women with a history of ICP developed pruritus on consumption of the combined oral contraceptive pill, which leads to similar conclusion. Estrogen role in the development of cholestasis is associated with reduction of hepatic biliary transport proteins expression and through internalization of the bile salt export pump (BSEP) [2]. Also progesterone is of considerable importance in the etiology of ICP. One of the studies improves that sulfated progesterone metabolites are supraphysiologically raised in the serum of ICP patients and as partial agonists of farnesoid X receptor (FXR) reduce the function of the main hepatic bile acid receptor [25].
Genetic defects in at least six canalicular transporters have been found to be associated with ICP. The most extensively studied mutations refer to ATP binding cassette subfamily B member 4 (ABCB4) gene, which encodes the multidrug resistance protein 3 (MRP3), acting in biliary phosphatidylcholine excretion. Genetic variation in ATP8B1, which encodes the phosphatidylserine flippase (FIC1), has been found in some cases of ICP, however its role in pathogenesis is vague, because one of the expanded study showed no significant evidence for association with ATP8B1. Heterozygous mutations of ABCB11 have been recognized as having a smaller but important role in the etiology of ICP. In particular, the European study identified six single nucleotide polymorphisms (SNIPs) in ABCB11gene, significantly associated with risk for ICP [26]. Also canicular transporters such as ABCC2, NR1H4 and fibroblast growth factor 19 (FGF19) have been described in molecular pathogenesis of cholestasis [7]. In addition, many other loci have been examined, usually in small studies, but they require further investigations [27].
In the recent years the role of immune system abnormalities have been considered as an important etiological factor of ICP [28-30]. Zhang et al. [31] presented the higher expressions of peroxisome proliferator-activated receptor γ (PPAR-γ) and nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) in ICP placenta and then induced abnormal serum levels of cytokines interleukin 4 (IL-4), IL-6, IL-12 and tumor necrosis factor α (TNF-α). This condition might disturb the balance of inflammatory and immune reaction, placental BA and serum lipids transport, finally resulting in fatal cholestasis and probably one of the mechanism of ICP. Such findings may conceive a potential target for future therapy.
Environmental factors, such as seasonal variations, related to low maternal vitamin D levels or reduced dietary Se intake, and HCV co-infection were described wider above.
Intrahepatic cholestasis of pregnancy and metabolic disorders
As it was mentioned before, the mechanisms responsible for the development of ICP in otherwise healthy pregnancies are still not fully understood. During recent years increasingly ICP is recognized to be associated with an abnormal metabolic profile. Several studies confirmed ICP to be characterized by glucose intolerance and dyslipidemia, consistent with the changes observed in the metabolic syndrome. This adverse effect was considered to be secondary to maternal aberrant bile acid homeostasis.
An association between glucose intolerance and dyslipidemia in ICP has previously been reported [32, 33]. Martineau et al. [34] performed a retrospective case-control study to evaluate the association between ICP and gestational diabetes mellitus (GDM). Of the 57 724 pregnancies, between February 2005 and June 2011, 143 were complicated by ICP (0.25%) and 4880 by GDM (8.5%). Nineteen ICP cases had GDM. The incidence of GDM in pregnancies complicated by ICP was 13.6% (OR 1.68, 95% CI: 1.04-2.72, p = 0.03). Among women with ICP the incidence of GDM was 13.4% in cases screened before they developed ICP (OR 1.66, 95% CI: 0.89-3.10, p = 0.11), rising to 30% (OR 4.69, 95% CI: 1.98-11.1, p = 0.0002) following the onset of cholestasis. The results clearly showed higher risk of GDM among patients with ICP and proved association between this two conditions. The same study presented that overweight was more common in ICP women than in not complicated pregnancies, although the difference was not statistically significant (p = 0.11) and probably did not have impact on GDM prevalence.
Subsequently, another study by Martineau et al. [35] confirmed these results and suggested not only higher prevalence of GDM, but also higher postprandial plasma glucose levels among pregnancies complicated by ICP comparing to control group. The mean maternal peak of postprandial glucose concentrations was significantly higher in women with ICP compared with uncomplicated pregnancy (7.2 vs. 6.4 mmol/l, p ≤ 0.005) measured by continuous glucose monitoring (CGM) system. Similar results were presented using a standard oral glucose tolerance test (OGTT). Furthermore, analysis of plasma glucagon like peptide (GLP-1) levels revealed its concentrations at 60 minutes following glucose ingestion to be significantly decreased in ICP.
The mechanism of changes in glucose metabolism may potentially be related to reduced activity of FXR, which influences glucose, lipid and BA homeostasis. As it was mentioned above ICP patients had downregulated FXR function due to antagonism of sulphated progesterone metabolites which level is elevated. Physiologically, primary bile acids – CA and CDCA, suppress the expression of key enzymes involved in gluconeogenesis by interaction with FXR [36]. Additionally, BA have recently been reported to act synergistically with glucose to promote β cell insulin secretion [37, 38], as well as induce expression of the insulin-regulated glucose transporter GLUT-4, through FXR-mediated pathways [39]. Disruption in these homeostatic pathways may promote impaired glucose tolerance observed in ICP.
Enteric BA also stimulate the G protein-coupled receptor TGR5, resulting in GLP-1 release and further stimulating pancreatic endocrine activity, increasing insulin and decreasing glucagon release. It is therefore probable that the insulinotropic effect will be attenuated in ICP, both directly and indirectly, due to reduction of the enterohepatic BA circulation [40, 41].
From the 12th week of pregnancy, lipid parameters, including total cholesterol (TC), triglycerides (TG), low-density lipoprotein-cholesterol (LDL-C) and high-density lipoprotein-cholesterol (HDL-C) are increased, especially in the second and third trimesters [42]. These physiological differences in the lipid profile were excessive in women with ICP. Moreover, according to the study of Jin et al. [42] hypertriglyceridemia could be considered as significant predictor of ICP. High TG at the second trimester were significantly associated with the morbidity of ICP (p = 0.006, AOR = 1.50, 95% CI: 1.12-2.00). Additionally, every mmol/l elevation in maternal third-trimester TG concentration was associated with an increased risk of this condition (p = 0.002, AOR = 1.28, 95% CI: 1.09-1.51).
Dann et al. [43] assessed plasma lipid concentrations in non-fasting blood samples from 132 pregnant women (63 women with ICP) during pregnancy and at 4-6 weeks postpartum. In the ICP group TC concentrations were significantly elevated compared to controls either during the gestational or postnatal period (p = 0.001). No significant difference in very-low-density lipoprotein (VLDL) cholesterol concentrations were found. Plasma LDL-C was consistently elevated from 16 weeks (p = 0.001) over whole pregnancy and even postpartum in the ICP women compared to control groups. Almost all (92%) of ICP cases had maximal LDL-C level over 3.4 mmol/l (120 mg/dl), whereas only 42% of controls exceeded this level. Apolipoprotein B-100 level was also raised, but the difference was not significant between mentioned groups. HDL-C was slightly lower in the ICP group. No significant differences in HDL-C levels were detected postnatally between analyzed groups. Consequently, cardiovascular risk indices LDL/HDL-C cholesterol and TC/HDL-C were consistently higher in the gestational period and in postnatal samples in the ICP group compared to controls.
Subsequently, two studies confirmed, beside such a character of lipid profile changes, TG concentration to be significantly higher in ICP women when compared to uncomplicated pregnancy [35, 42]. The lipid changes associated with ICP could be the consequence of the modulation of lipid metabolism by BA similar to changes of glucose metabolism described above. Several studies presented almost identical modification of the lipid profiles in FXR-deficient mice [44, 45]. The only difference was observed in HDL-C concentration, which level was decreased among ICP patients compared to FXR-deficient mice. Expression of PPAR-α, a nuclear receptor regulating expression of apolipoprotein A1 (a major protein component of HDL), is enhanced by FXR. Interestingly, either HDL-C or apolipoprotein A1 levels are decreased in ICP with advancing gestation due to suppression of FXR, associated with elevated reproductive hormone excretion [35].
Human studies have shown that the intrauterine environment is one of the principal factor having an impact on development of metabolic disease in adults. As mentioned before, the study by Papacleovoulou et al. [20] showed that 16-year-old children of mothers with ICP had altered lipid profiles in comparison to control group. Males had increased BMI (23.36 kg/m<sup>2</sup> vs. 21.07 kg/m<sup>2</sup>, p < 0.05) and insulin level (23.60 mU/l vs. 10.76 mU/l, p < 0.05), while females exhibited increased waist (77.5 cm vs. 71.8 cm, p < 0.05) and hip (96.7 cm vs. 92.9 cm, p < 0.05) girth compared with the offspring of uncomplicated pregnancies. These results were also validated in the mouse model. The metabolic profiles of both male and female offsprings of ICP mothers were associated with a preobese and prediabetic phenotype as well as altered biochemical profiles. The potential mechanism of programming effect in the offspring is linked to excessive BA in utero, which escalates cholesterol storage and transport in the fetoplacental unit.
The role of ICP in pathogenesis of metabolic disorders, such as glucose intolerance and dyslipidemia, is incontrovertible, although is it possible that these conditions are primary causative factors in ICP?
According to Dann et al. [43], ICP related dyslipidemia presented in their study could be the effect of the modulation of lipid metabolism by BA. Though, BA (and other liver function markers, e.g. ALT, AST, GGT) were only raised from 28 weeks gestation, whereas LDL-C was shown to be elevated from 16 weeks of gestation. That several-weeks interval confirms that the changes of LDL-C profile may be essential in ICP pathogenesis, rather than a result of the disease process. Contrarily, BA may be responsible for the modification of HDL-C among ICP women.
A population-based study from China [42] suggested that maternal high TG concentration during the second and third trimester were independently and significantly associated with a greater risk of ICP.
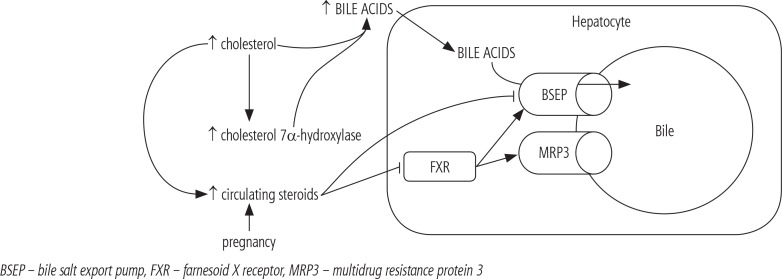
A primary role for altered lipid metabolism in the pathophysiology of ICP is valid. Lipoproteins redound to oxidative stress and influence cell membrane fluidity, permeability of canalicular epithelium, and the function of hepatobiliary transporters and receptors. Lipids also promote synthesis of cholestatic metabolites of placental steroid hormones [43]. Another evidence of essential role of dyslipidemia in ICP pathogenesis, is the case of a 26-year-old woman with a history of familial combined hyperlipidaemia [46]. She also presented with a recurring ICP, with an early onset of symptoms (16 weeks of gestation) and a severe clinical trial (serum BA level up to 207 µmol/l). Co-existing diseases, which modify lipid metabolism, such as familial combined hyperlipidemia, may not only be a trigger of ICP, but also escalate its severity. Cholesterol is also the precursor for the synthesis of steroid hormones and BA. This process of BA synthesis is one of the prevalent ways of excretion of cholesterol excess. The primary route to BA is initiated by 7α-hydroxylation of cholesterol by cholesterol 7α-hydroxylase, a microsomal cytochrome P450-dependent enzyme. Post et al. [47] proved that lipoprotein cholesterol induces the expression of cholesterol 7α-hydroxylase gene directly at the level of its transcription and, in consequence, up-regulates BA synthesis. As we can see, hyperlipidemia presents multiple effect on ICP pathogenesis. This complex mechanism is shown in Figure 1.
Fig. 1.
The association between hyperlipidemia and intrahepatic cholestasis of pregnancy, and excretion of bile acids into the biliary canaliculus
Metabolic pathways may be regulated by some muscle, adipose tissue and hepatic related factors termed myokines, adipokines and hepatokines, respectively. Up-to now the data regarding the meaning of these factors in ICP is explained very sparsely or not at all.
Irisin is a novel exercise-induced myokine, which is able to regulate glucose and lipid levels, improving insulin sensitivity in experimental animal models [48]. Obese and diabetic patients have significantly decreased serum irisin level [49]. Kirbas et al. [50] analyzed maternal serum irisin concentrations in 58 pregnant women with ICP and 30 healthy women with uncomplicated pregnancies. The study presented that serum irisin levels were significantly higher in the severe ICP comparing to mild ICP and control groups (p = 0.005 and p < 0.001, respectively). There was also a significant negative correlation between irisin and fasting blood glucose levels (r = −0.399, p = 0.021). According to those results the elevation of serum irisin concentration among ICP women potentially could be a compensatory reaction to pre-existing metabolic disorders.
Conclusions
Intrahepatic cholestasis of pregnancy is a relatively common hepatic disorder in pregnancy. Whereas environmental, endocrine and genetic factors are well known to be involved in pathogenesis of ICP, the role of metabolic disorders in this process is still poorly recognized. The relevance of myokines and adipokines which influence metabolic pathways has been hardly explained. Large-scale clinical trials are required to discover which metabolic pathways are altered in ICP. These knowledge is indispensable to establish new, more effective management strategies for ICP and as a consequence to improve both maternal, and especially fetal prognosis.
Disclosure
Authors report no conflict of interest.
References
- 1.Williamson C, Geenes V. Intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2014;124:120–133. doi: 10.1097/AOG.0000000000000346. [DOI] [PubMed] [Google Scholar]
- 2.Geenes V, Williamson C. Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2009;15:2049–2066. doi: 10.3748/wjg.15.2049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Saleh MM, Abdo KR. Intrahepatic cholestasis of pregnancy: review of the literature and evaluation of current evidence. J Womens Health (Larchmt) 2007;16:833–841. doi: 10.1089/jwh.2007.0158. [DOI] [PubMed] [Google Scholar]
- 4.Reyes H, Gonzalez MC, Ribalta J, et al. Prevalence of intrahepatic cholestasis of pregnancy in Chile. Ann Intern Med. 1978;88:487–493. doi: 10.7326/0003-4819-88-4-487. [DOI] [PubMed] [Google Scholar]
- 5.Reyes H. Sex hormones and bile acids in intrahepatic cholestasis of pregnancy. Hepatology. 2008;47:376–379. doi: 10.1002/hep.22139. [DOI] [PubMed] [Google Scholar]
- 6.Arrese M, Reyes H. Intrahepatic cholestasis of pregnancy: a past and present riddle. Ann Hepatol. 2006;5:202–205. [PubMed] [Google Scholar]
- 7.Floreani A, Gervasi MT. New insights on intrahepatic cholestasis of pregnancy. Clin Liver Dis. 2016;20:177–189. doi: 10.1016/j.cld.2015.08.010. [DOI] [PubMed] [Google Scholar]
- 8.Locatelli A, Roncaglia N, Arreghini A, et al. Hepatitis C virus infection is associated with a higher incidence of cholestasis of pregnancy. Br J Obstet Gynaecol. 1999;106:498–500. doi: 10.1111/j.1471-0528.1999.tb08305.x. [DOI] [PubMed] [Google Scholar]
- 9.Marschall H-U, Shemer EW, Ludvigsson JF, et al. Intrahepatic cholestasis of pregnancy and associated hepatobiliary disease: a population based cohort study. Hepatology. 2013;58:1385–1391. doi: 10.1002/hep.26444. [DOI] [PubMed] [Google Scholar]
- 10.Wijarnpreecha K, Thongprayoon C, Sanguankeo A, et al. Hepatitis C infection and intrahepatic cholestasis of pregnancy: A systematic review and meta-analysis. Clin Res Hepatol Gastroenterol. 2017;41:39–45. doi: 10.1016/j.clinre.2016.07.004. [DOI] [PubMed] [Google Scholar]
- 11.Heinonen S, Kirkinen P. Pregnancy outcome with intrahepatic cholestasis. Obstet Gynecol. 1999;94:189–193. doi: 10.1016/s0029-7844(99)00254-9. [DOI] [PubMed] [Google Scholar]
- 12.Savander M, Ropponen A, Avela K, et al. Genetic evidence of heterogeneity in intrahepatic cholestasis of pregnancy. Gut. 2003;52:1025–1029. doi: 10.1136/gut.52.7.1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wilkstrom Shemer E, Marshall HU. Decreased 1,25 dihydroxy vitamin D levels in women with intrahepatic cholestasis of pregnancy. Acta Obstet Gynecol Scand. 2010;89:1420–1423. doi: 10.3109/00016349.2010.515665. [DOI] [PubMed] [Google Scholar]
- 14.Reis H, Kauppila A, Korpela H, et al. Low serum selenium concentration and glutathione peroxidase activity in intrahepatic cholestasis of pregnancy. Br Med J. 1987;294:150–152. doi: 10.1136/bmj.294.6565.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Berg B, Helm G, Petersohn L, et al. Cholestasis of pregnancy. Clinical and laboratory studies. Acta Obstet Gynecol. 1996;175:957–960. doi: 10.3109/00016348609158363. [DOI] [PubMed] [Google Scholar]
- 16.Laatikainen T, Ikonen E. Fetal prognosis in obstetric hepatosis. Ann Chir Gynaecol Fenn. 1975;64:155–164. [PubMed] [Google Scholar]
- 17.Ozkan S, Ceylan Y, Ozkan OV, et al. Review of a challenging clinical issue: Intrahepatic cholestasis of pregnancy. World J Gastroenterol. 2015;21:7134–7141. doi: 10.3748/wjg.v21.i23.7134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Reid R, Ivery KJ, Rencoret RH, et al. Fetal complications of obstetric cholestasis. Br Med J. 1976;1:870–872. doi: 10.1136/bmj.1.6014.870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Glantz A, Marschall HU, Mattsson LA. Intrahepatic cholestasis of pregnancy: Relationships between bile acid levels and fetal complication rates. Hepatology. 2004;40:467–474. doi: 10.1002/hep.20336. [DOI] [PubMed] [Google Scholar]
- 20.Papacleovoulou G, Abu-Hayyeh S, Nikolopoulou E, et al. Maternal cholestasis during pregnancy programs metabolic disease in offspring. J Clin Invest. 2013;123:3172–3181. doi: 10.1172/JCI68927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kondrackiene J, Kupcinskas L. Intrahepatic cholestasis of pregnancy-current achievements and unsolved problems. World J Gastroenterol. 2008;14:5781–5788. doi: 10.3748/wjg.14.5781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Boregowda G, Shehata HA. Gastrointestinal and liver disease in pregnancy. Best Pract Res Clin Obstet Gynaecol. 2013;27:835–853. doi: 10.1016/j.bpobgyn.2013.07.006. [DOI] [PubMed] [Google Scholar]
- 23.Gonzalez MC, Reyes H, Arrese M, et al. Intrahepatic cholestasis of pregnancy in twin pregnancies. J Hepatol. 1989;9:84–90. doi: 10.1016/0168-8278(89)90079-2. [DOI] [PubMed] [Google Scholar]
- 24.Williamson C, Hems LM, Goulis DG, et al. Clinical outcome in a series of cases of obstetric cholestasis identified via a patient support group. BJOG. 2004;111:676–681. doi: 10.1111/j.1471-0528.2004.00167.x. [DOI] [PubMed] [Google Scholar]
- 25.Abu-Hayyeh S, Papacleovoulou G, Lövgren-Sandblom A, et al. Intrahepatic cholestasis of pregnancy levels of sulfated progesterone metabolites inhibit FXR resulting in a pro-cholestatic phenotype. Hepatology. 2013;57:716–726. doi: 10.1002/hep.26055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dixon PH, Wadsworth A, Chambers J, et al. A comprehensive analysis of common genetic variation around six candidate loci for intrahepatic cholestasis of pregnancy. Am J Gastroenterol. 2014;109:76–84. doi: 10.1038/ajg.2013.406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dixon PH, Williamson C. The pathophysiology of intrahepatic cholestasis of pregnancy. Clin Res Hepatol Gastroenterol. 2016;40:141–153. doi: 10.1016/j.clinre.2015.12.008. [DOI] [PubMed] [Google Scholar]
- 28.Larson SP, Kovilam O, Agrawal DK. Immunological basis in the pathogenesis of intrahepatic cholestasis of pregnancy. Expert Rev Clin Immunol. 2016;12:39–48. doi: 10.1586/1744666X.2016.1101344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ozler A, Ucmak D, Evsen MH, et al. Immune mechanisms and the role of oxidative stress in intrahepatic cholestasis of pregnancy. Cent Eur J Immunol. 2014;39:198–202. doi: 10.5114/ceji.2014.43723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yayi H, Danqing W, Shuyun L, et al. Immunologic abnormality of intrahepatic cholestasis of pregnancy. Am J Reprod Immunol. 2010;63:267–273. doi: 10.1111/j.1600-0897.2009.00798.x. [DOI] [PubMed] [Google Scholar]
- 31.Zhang Y, Hu L, Cui Y, et al. Roles of PPARγ/NF-κB signaling pathway in the pathogenesis of intrahepatic cholestasis of pregnancy. PLoS One. 2014;9:e87343. doi: 10.1371/journal.pone.0087343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee RH, Kwok KM, Ingles S, et al. Pregnancy outcomes during an era of aggressive management for intrahepatic cholestasis of pregnancy. Am J Perinatol. 2008;25:341–345. doi: 10.1055/s-2008-1078756. [DOI] [PubMed] [Google Scholar]
- 33.Baliutavičienė D, Zubruvienė N, Zalinkevičius R. Pregnancy outcome in cases of intrahepatic cholestasis of pregnancy. Int J Gynaecol Obstet. 2011;112:250–251. doi: 10.1016/j.ijgo.2010.11.002. [DOI] [PubMed] [Google Scholar]
- 34.Martineau MG, Raker C, Powrie R, et al. Intrahepatic cholestasis of pregnancy is associated with an increased risk of gestational diabetes. Eur J Obstet Gynecol Reprod Biol. 2014;176:80–85. doi: 10.1016/j.ejogrb.2013.12.037. [DOI] [PubMed] [Google Scholar]
- 35.Martineau MG, Raker C, Dixon PH, et al. The metabolic profile of intrahepatic cholestasis of pregnancy is associated with impaired glucose tolerance, dyslipidemia, and increased fetal growth. Diabetes Care. 2015;38:243–248. doi: 10.2337/dc14-2143. [DOI] [PubMed] [Google Scholar]
- 36.Ma K, Saha PK, Chan L, et al. Farnesoid X receptor is essential for normal glucose homeostasis. J Clin Invest. 2006;116:1102–1109. doi: 10.1172/JCI25604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Renga B, Mencarelli A, Vavassori P, et al. The bile acid sensor FXR regulates insulin transcription and secretion. Biochim Biophys Acta. 2010;1802:363–372. doi: 10.1016/j.bbadis.2010.01.002. [DOI] [PubMed] [Google Scholar]
- 38.Seyer P, Vallois D, Poitry-Yamate C, et al. Hepatic glucose sensing is required to preserve β cell glucose competence. J Clin Invest. 2013;123:1662–1676. doi: 10.1172/JCI65538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Shen H, Zhang Y, Ding H, et al. Farnesoid X receptor induces GLUT4 expression through FXR response element in the GLUT4 promoter. Cell Physiol Biochem. 2008;22:1–14. doi: 10.1159/000149779. [DOI] [PubMed] [Google Scholar]
- 40.Roberts RE, Glicksman C, Alaghband-Zadeh J, et al. The relationship between postprandial bile acid concentration, GLP-1, PYY and ghrelin. Clin Endocrinol. 2011;74:67–72. doi: 10.1111/j.1365-2265.2010.03886.x. [DOI] [PubMed] [Google Scholar]
- 41.Parker HE, Wallis K, le Roux CW, et al. Molecular mechanisms underlying bile acid-stimulated glucagon-like peptide-1 secretion. Br J Pharmacol. 2012;165:414–423. doi: 10.1111/j.1476-5381.2011.01561.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Jin WY, Lin SL, Hou RL, et al. Associations between maternal lipid profile and pregnancy complications and perinatal outcomes: apopulation-based study from China. BMC Pregnancy Childbirth. 2016;16:60. doi: 10.1186/s12884-016-0852-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dann AT, Kenyon AP, Wierzbicki AS, et al. Plasma lipid profiles of women with intrahepatic cholestasis of pregnancy. Obstet Gynecol. 2006;107:106–114. doi: 10.1097/01.AOG.0000189096.94874.9c. [DOI] [PubMed] [Google Scholar]
- 44.Zhang Y, Lee FY, Barrera G, et al. Activation of the nuclear receptor FXR improves hyperglycemia and hyperlipidemia in diabetic mice. Proc Natl Acad Sci USA. 2006;103:1006–1011. doi: 10.1073/pnas.0506982103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cariou B, van Harmelen K, Duran-Sandoval D, et al. The farnesoid X receptor modulates adiposity and peripheral insulin sensitivity in mice. J Biol Chem. 2006;281:11039–11049. doi: 10.1074/jbc.M510258200. [DOI] [PubMed] [Google Scholar]
- 46.Ajala T, Rafi J, Wray R, et al. There may be a link between intrahepatic cholestasis of pregnancy and familial combined hyperlipidaemia: a case report. Cases J. 2009;2:8679. doi: 10.4076/1757-1626-2-8679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Post SM, Twisk J, van der Fits L, et al. Lipoprotein cholesterol uptake mediates up-regulation of bile-acid synthesis by increasing cholesterol 7alpha-hydroxylase but not sterol 27-hydroxylase gene expression in cultured rat hepatocytes. Biochem J. 1999;341:339–346. [PMC free article] [PubMed] [Google Scholar]
- 48.Polyzos SA, Mathew H, Mantzoros CS. Irisin: a true, circulating hormone. Metabolism. 2015;64:1611–1618. doi: 10.1016/j.metabol.2015.09.001. [DOI] [PubMed] [Google Scholar]
- 49.Moreno-Navarrete JM, Ortega F, Serrano M, et al. Irisin is expressed and produced by human muscle and adipose tissue in association with obesity and insulin resistance. J Clin Endocrinol Metab. 2013;98:769–778. doi: 10.1210/jc.2012-2749. [DOI] [PubMed] [Google Scholar]
- 50.Kirbas A, Daglar K, Timur H, et al. Maternal circulating levels of irisin in intrahepatic cholestasis of pregnancy. J Matern Fetal Neonatal Med. 2016;22:1–5. doi: 10.3109/14767058.2015.1132694. [DOI] [PubMed] [Google Scholar]