Highlights
-
•
β-1,4-d-Xylanohydrolase from Geobacillus stearothermophilus KIBGE-IB29 was purified and characterized.
-
•
The catalytic properties revealed significant stability over broad pH and temperature range.
-
•
Native-PAGE and In-gel activity assay were carried out.
-
•
Various organic solvents and detergents significantly improved the enzyme activity.
-
•
β-1,4-d-Xylanohydrolase showed excellent storage stability for prospective industrial use.
Chemical compounds studied in this article: Xylan (Simson Laboratories, UK) Xylose (PubChem CID: 135191); 3′5′ dinitrosalicylic acid (PubChem CID: 11873); Sodium potassium tartrate (PubChem CID: 9357); Dipotassium hydrogen phosphate (PubChem CID: 24450); Dihydrogen potassium phosphate (PubChem CID: 516951); Ammonium sulfate (PubChem CID: 6097028); Glycine (PubChem CID: 750); Citric acid (PubChem CID: 311)
Keywords: Xylanohydrolase, Purification, Characterization, Catalysis, Thermal stability, Industrial use
Graphical abstract
Abstract
The β-1,4-d-xylanohydrolase is an industry valuable catalytic protein and used to synthesize xylooligosaccharides and xylose. In the current study, β-1,4-d-xylanohydrolase from Geobacillus stearothermophilus KIBGE-IB29 was partially purified up to 9.5-fold with a recovery yield of 52%. It exhibited optimal catalytic activity at pH-7.0 and 50 °C within 5 min. Almost 50% activity retained at pH-4.0 to 9.0 however, 70% activity observed within the range of 40 °C to 70 °C. The β-1,4-d-xylanohydrolase showed a significant hydrolytic pattern with 48.7 kDa molecular mass. It was found that the enzymatic activity improved up to 160% with 1.0 mM ethanol. Moreover, the activity of enzyme drastically increased up to 2.3 and 1.5 fold when incubated with Tween 80 and Triton X-100 (1.0 mM), respectively. The β-1,4-d-xylanohydrolase also retained 72% activity at −80 °C after 180 days. Such a remarkable biochemical properties of β-1,4-d-xylanohydrolase make it possible to forecast its potential use in textile and food industries.
1. Introduction
Xylan is the major component of hemicellulose and extensively found in wheat, barley, rice and many other plants. It is a highly branched heteropolysaccharide composed of 1,4-linked β-d-xylopyranosyl units with different side groups [1]. Synergistic action of different enzymes is mostly required to completely hydrolyze the xylan polymer due to its heterogeneity and structural complexity. Among them, β-1,4-d-xylanohydrolase [E.C.3.2.1.8, endo-β-1,4-xylanase] is a key enzyme that catalyzes the endohydrolysis of 1,4-β-d-xylosidic linkages in xylan yielding xylooligosaccharides, xylobiose and xylose as end products [2]. Owing to the valuable catalytic properties, β-1,4-d-xylanohydrolase has broad potential to be used in different biotechnological processes and clarification of fruit juices with combinations of pectinase and cellulase. β-1,4-d-Xylanohydrolase is extensively used in baking industry for improving the elasticity and strength of dough, bread volume and texture. Xylooligosaccharides synthesized during bread-making have profound effect on health and are used as prebiotics [3]. Biscuits manufacturing industry uses β-1,4-d-xylanohydrolase for making cream crackers lighter and enhancing the texture, palatability and homogeneity of wafers. β-1,4-d-Xylanohydrolase is also used in animal feed processing industry for increasing the digestibility and nutritive value of feed, in the extraction of plant oils and coffee as well as it is employed in bleaching process of paper and pulp [[4], [5], [6], [7]].
Considering the broad potential of β-1,4-d-xylanohydrolase for different biotechnological applications, researchers are focusing on the exploration of functionally stable enzymes that can survive harsh industrial conditions. Industrial processes are usually hostile in terms of high pH, high temperature and different inhibitors such as metal ions etc. Therefore, the enzyme must have ability to tolerate such extreme conditions [8]. Several reported β-1,4-d-xylanohydrolases do not fulfill this requirement and has developed the challenge for the synthesis of more stable enzyme with maximum catalytic efficiency [9,10]. Thermophilic Geobacillus species have been reported to produce high quantity of thermozymes that do not only survive high temperature but also reveal more tolerance to broad pH range, different metal ions, solvents and detergents due to their specific three dimensional structure as compared to mesophilic counterpart [11]. Efficient catalytic properties of β-1,4-d-xylanohydrolase with cost effective production approach are necessary in order to meet the escalating demand for its commercialization [12].
In the current study, thermo-tolerant β-1,4-d-xylanohydrolase produced from Geobacillus stearothermophilus KIBGE-IB29 was partially purified, dialyzed and biochemically characterized with reference to its maximum catalytic efficiency. Moreover, the effect of different solvents and detergents was analyzed on the catalytic activity of β-1,4-d-xylanohydrolase. Thermal and storage stability of β-1,4-d-xylanohydrolase was also examined for its prospective industrial applications.
2. Material and methods
2.1. Production of β-1,4-d-xylanohydrolase
Highly thermophilic Geobacillus stearothermophilus KIBGE-IB29 [GenBank Accession: KF241865.1] was isolated earlier from soil collected around the blast furnace area of a steel processing industry. Bacterial isolate was cultivated into modified growth medium containing (gL−1): xylan, 5.0; yeast extract, 2.0; meat extract, 1.0; peptone, 5.0; KH2PO4, 0.5; K2HPO4, 2.5; (NH4)2SO4, 0.5; CaCl2, 0.1 with pH-6.0 and incubated at 60 °C for 24 h to achieve high production yield of β-1,4-d-xylanohydrolase [13]. The cells were harvested after 24 h through centrifugation at 13,440 ☓ g for 15 min at 4 °C. Supernatant was passed through 0.22 μm filter membrane (Nitrocellulose, Millipore, Germany) in order to obtain cell free filtrate and subjected to next partial purification step.
2.2. Partial purification and dialysis of β-1,4-d-xylanohydrolase
After biomass removal, the cell free filtrate was partially purified by salt precipitation method. For this purpose, gradient precipitation approach was followed by using 30%–50% concentration of ammonium sulfate. It was added gradually in cell free filtrate with constant stirring at 4 °C and left overnight at same temperature to attain complete equilibration. The precipitated enzyme fraction was collected by centrifugation at 13,440 ☓ g for 15 min at 4 °C and re-dissolved in 25.0 mM phosphate buffer with pH-7.0. Finally, dialysis was performed against the same buffer for 18 h by using dialysis tubing (12,000 kDa cut-off, Servapor®) and dialyzed protein was kept at 4 °C for further enzyme analysis.
2.3. Enzyme assay for β-1,4-d-xylanohydrolase
The catalytic activity of β-1,4-d-xylanohydrolase was determined by measuring the amount of released end product (xylose) after xylan hydrolysis using 3′5′ dinitrosalicylic acid (DNS) method [14]. The enzyme (0.1 ml) was mixed with 20.0 g L−1 xylan (1.0 ml) dissolved in potassium phosphate buffer (25.0 mM, pH-7.0) and kept at 50 °C for 5 min. Afterwards, DNS reagent (1.0 ml) was incorporated into reaction mixture to cease the enzymatic activity and subjected to boiling for 5 min. The reaction mixture was allowed to cool at room temperature and optical density was measured by spectrophotometer at 546 nm. The enzyme activity was calculated by plotting the xylose calibration curve. One unit of β-1,4-d-xylanohydrolase is defined as “the amount of enzyme required to produce 1.0 μmol of xylose per minute under abovementioned assay conditions”. The β-1,4-d-xylanohydrolase units (Unit−1 ml−1 min−1) were calculated by the formula given below:
2.4. Quantification of total protein
The protein concentration of cell free filtrate before and after enzyme precipitation and dialysis was estimated through Lowry’s method using Bovine serum albumin (BSA) as a standard [15].
2.5. Impact of reaction time on the hydrolytic property of β-1,4-d-xylanohydrolase
The time course for maximum enzyme-substrate reaction was examined by performing the enzyme assay for different time intervals (5–60 min), by keeping all other conditions constant (pH: 7.0; temperature: 50 °C; substrate: 20.0 g L−1).
2.6. Influence of temperature on the hydrolytic property of β-1,4-d-xylanohydrolase
Temperature is a critical parameter to be analyzed for maximum catalytic activity of any enzyme. In this study, impact of different temperatures was examined on β-1,4-d-xylanohydrolase by carrying out enzyme-substrate reaction at temperature range of 30 °C–80 °C, after maintaining other parameters at constant rate (pH: 7.0; substrate: 20.0 g L−1).
2.7. Effect of pH on the hydrolytic activity of β-1,4-d-xylanohydrolase
The effect of pH on the hydrolytic activity of β-1,4-d-xylanohydrolase was determined by conducting the reaction with xylan (20.0 g L−1) dissolved in different pH buffers ranging from 4.0 to 10.0, keeping same ionic strength (25.0 mM). Buffers used in this experiment were citrate-phosphate buffer having pH-4.0 to 6.0, potassium phosphate buffer having pH-7.0 to 8.0 and glycin-NaOH buffer having pH-9.0 to 10.0.
2.8. Effect of ionic strength of selected buffer on the activity of β-1,4-d-xylanohydrolase
In order to examine the effect of ionic strength of selected buffer on β-1,4-d-xylanohydrolase, the enzyme assay was performed in different ionic concentrations (25.0–200.0 mM) of selected potassium phosphate buffer with a constant pH-7.0.
2.9. Effect of various solvents, surfactants and metal ions chelator on the activity of β-1,4-d-xylanohydrolase
β-1,4-d-Xylanohydrolase was incubated with 1.0 mM and 5.0 mM concentration of various organic solvents (ethanol, methanol, isopropanol, formaldehyde, chloroform, DMSO), surfactants (sodium dodecyle sulfate, triton X-100, tween 80) and metal ions chelator (ethylenediaminetetraacetic acid) in order to determine their effect on the activity of β-1,4-d-xylanohydrolase. The mixture was incubated at 37 °C for 60 min and the enzyme assay was performed at 50 °C. Percent relative activity was calculated and compared with the control. Control was taken as 100% and was not treated with any chemical.
2.10. Native-PAGE electrophoresis and In-gel activity assay of β-1,4-d-xylanohydrolase
Polyacrylamide gel electrophoresis (PAGE) was conducted to determine the molecular weight of β-1,4-d-xylanohydrolase. For this purpose, the migration rate of the partially purified β-1,4-d-xylanohydrolase was compared with known molecular weight standard protein (Bovine serum albumin: A8654-Sigma Aldrich). A slightly modified method of Ornstein [16] and Davis [17] was employed in this experiment. The partially purified enzyme was mixed in equal volume (1:1) with the sample diluting buffer and loaded in 12.5% polyacrylamide gel. After completion of the electrophoresis, gel was stained with Coomassie brilliant blue R-250 for 4.0 h and destained until a clear background was achieved. For In-gel activity assay, slightly modified method of Zheng et al. [18] was used by incorporating 1% xylan into the gel to detect the activity of β-1,4-d-xylanohydrolase. Initially, gel was washed with double deionized water and potassium phosphate buffer (25.0 mM, pH-7.0) with multiple changes. Afterwards, gel was placed in phosphate buffer for 1 h at 50 °C with slight shaking and washed again with double deionized water. It was stained with 0.5% Congo red dye for 45 min and destained with 1.0 M NaCl for 45 min with slight shaking at 37 °C A clear hydrolytic zone was examined against a dark red background on the gel. Gel documentation system (Gel Doc 2000, Universal Hood, BioRad Laboratories Inc., USA) was used to estimate the approximate molecular mass of β-1,4-d-xylanohydrolase with Quantity One Quantitative Software.
2.11. Thermal stability of β-1,4-d-xylanohydrolase
The enzyme tolerance to high temperature is an important requirement for industrial applications. By considering its significance, β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 was pre-incubated at various temperatures ranging from 40 °C to 60 °C for 2 h. During this period, enzyme aliquots were retrieved after 30 min and released reducing sugar (xylose) was estimated at optimum temperature (50 °C) by following the standard assay procedure.
2.12. Storage stability of β-1,4-d-xylanohydrolase
The storage stability of β-1,4-d-xylanohydrolase was analyzed by placing the enzyme at different temperatures (30 °C, 4 °C, −80 °C) up to 6 months and catalytic activity was monitored in terms of percent residual activity.
2.13. Statistical analysis
All the experiments were conducted in triplicates and the data presented are a mean value of three observations. The data was analyzed through Bonferroni test for the calculation of significant difference and the significant level was adjusted at P < 0.05.
3. Results and discussion
3.1. Purification of β-1,4-d-xylanohydrolase
Ammonium sulfate precipitation was opted as a purification tool to purify and concentrate enzymatic protein by altering their solubility within cell free filtrate. It was observed that 40% ammonium sulfate concentration gave maximum precipitation of β-1,4-d-xylanohydrolase with reference to specific activity. Fold purification of enzyme was investigated by using 40% ammonium sulfate concentration. It was observed that fold precipitation increased as activity increased and enzyme precipitates showed 6617 U mg−1 specific activity with 4.8 fold purification whereas, dialyzed precipitates showed 13,125 U mg−1 specific activity with 9.5 fold purification. Percent recovery of dialyzed precipitates was increased from 30% to 52%. It has been concluded that purification of β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 yields 9.5 fold purified enzyme with 52% recovery rate (Table 1).
Table 1.
Ammonium sulfate precipitation and Fold purification with percent recovery of β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29.
| Ammonium sulfate concentration* g dL−1 | Total volume (ml) | Enzyme activity (U ml−1 min-1) | Total protein (mg ml−1) | Specific activity (U mg−1) | Fold purification | Recovery (%) |
|---|---|---|---|---|---|---|
| Crude enzyme | 97.0 | 380240.0 ± 19012.0 | 276.4 ± 13.8 | 1375.0 ± 68.7 | 1.0 | 100.0 |
| 30.0 | 5.0 | 9000.0 ± 450.0 | 23.0 ± 1.15 | 385.0 ± 19.2 | --- | --- |
| 40.0 | 5.0 | 112500.0 ± 5625.0 | 17.0 ± 0.85 | 6617.0 ± 330.8 | 4.8 | 30.0 |
| 50.0 | 5.0 | 12500.0 ± 625.0 | 16.0 ± 0.8 | 806.0 ± 40.3 | --- | --- |
| Precipitates (40.0%) | 5.0 | 112500.0 ± 5625.0 | 17.0 ± 0.85 | 6617.0 ± 330.8 | 4.8 | 30.0 |
| Dialyzed precipitates | 7.5 | 196875.0 ± 9843.7 | 15.0 ± 0.7 | 13125.0 ± 656.2 | 9.5 | 52.0 |
Each quantity of ammonium sulfate was added gradually in cell free filtrate on ice bath and incubated overnight. All of the samples were obtained after the centrifugation at 13,440 ☓ g for 15.0 min. at 4 °C.
3.2. Impact of reaction time on the catalytic activity of β-1,4-d-xylanohydrolase
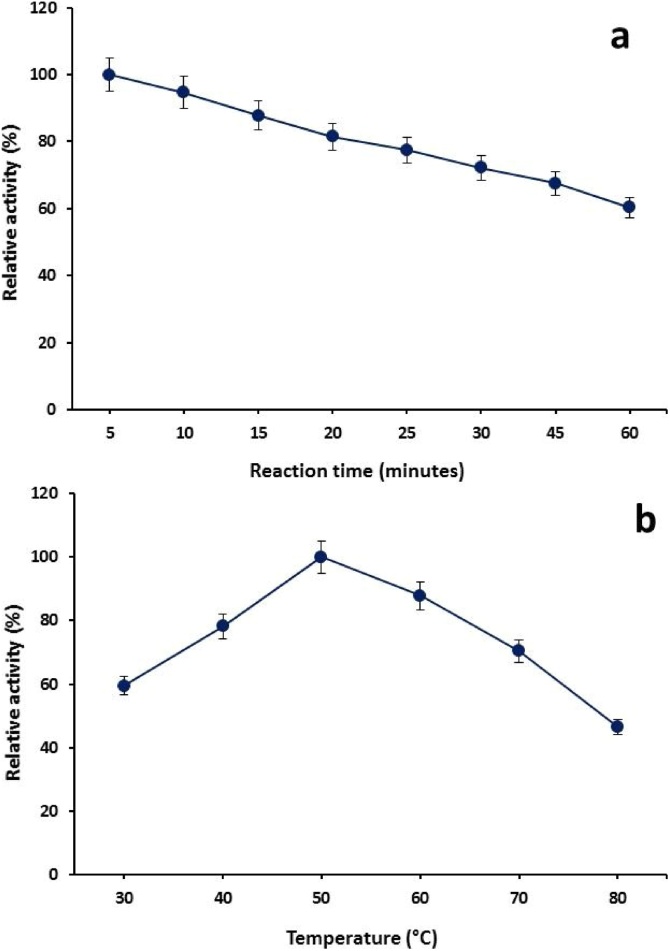
It was evident from the results that maximum enzymatic activity achieved after 5 min of reaction time and decline in activity was observed as time increased further (Fig. 1a). Above 80% and 70% relative activity were found after 20 and 30 min of incubation period, respectively while 60% relative activity also appeared after 60 min.
Fig. 1.
(a) Effect of reaction time and (b) temperature on the catalytic activity of β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 (Means ± S.E., n = 3).
3.3. Influence of temperature on the catalytic activity of β-1,4-d-xylanohydrolase
Selection of optimum temperature for the maximum catalytic activity of any enzyme is very important parameter for its characterization. It was observed that dialyzed β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 remained active in the broad range of temperatures (30 °C to 80 °C). The rate of xylan hydrolysis was increased as the temperature increased from 30 °C to 40 °C and exhibited more than 70% of its activity in the range from 40 °C to 60 °C. Optimum temperature was achieved at 50 °C with maximum catalytic efficiency (Fig. 1b). Further increase in temperature above optimum level resulted decline in activity and 88% relative activity was found at 60 °C. The β-1,4-d-xylanohydrolase activity was further dropped at temperature values of 70 °C and 80 °C that revealed 30% and 54% decreased activity, respectively. Temperature directly increased the kinetic energy of reactant until unless maximum catalytic activity achieved at optimum temperature (50 °C). Further increase in temperature caused the declined enzymatic activity by increasing more energy that ultimately broken the bonding and denatured the three dimensional structure of protein. Similar finding has been reported by investigating the xylanohydrolase from Jonesia denitrificans BN-13 that was stable at 50 °C for 9.34 h with maximum activity [19]. The optimal temperature of xylanohydrolase from Streptomyces sp. FA1 and Paenibacillus sp. NF1 were found to be 60 °C [20,21].
3.4. Effect of pH on the hydrolytic activity of β-1,4-d-xylanohydrolase
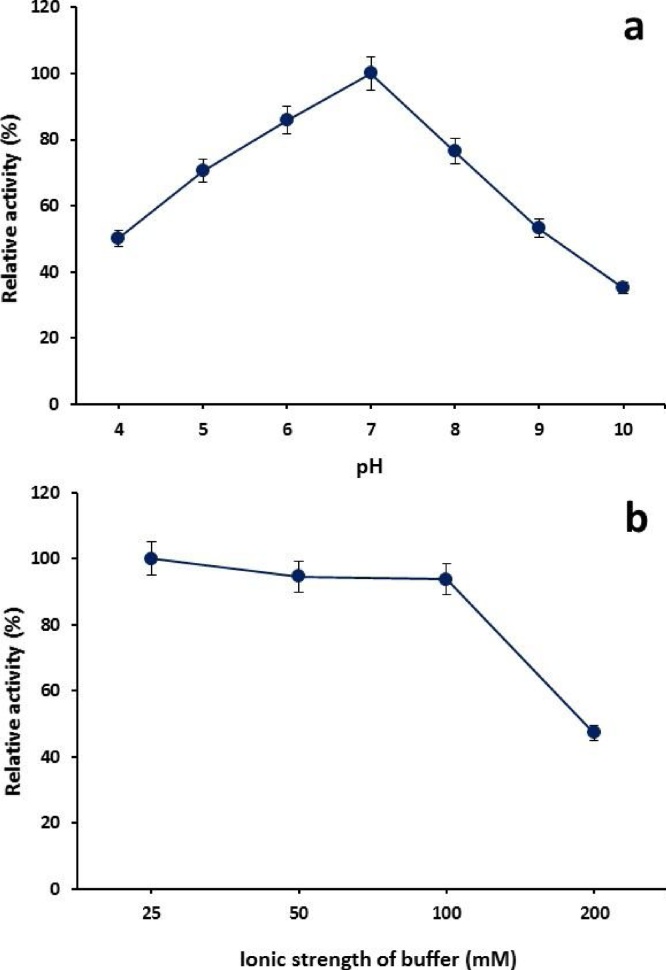
The enzymatic activity is markedly influenced by pH of reaction mixture that can cause alteration within three dimensional structure of protein. By investigating the effect of pH on β-1,4-d-xylanohydrolase, It was observed that β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE IB-29 exhibited its catalytic activity over broad range of pH and retained more than 70% relative activity in the pH range of 5.0 to 8.0. Maximum relative activity was attained at pH-7.0 (phosphate buffer, 25.0 mM) and pH above optimum declined the activity (Fig. 2a) as the enzyme activity and binding to substrate are mostly relied on charge distribution of both enzyme molecule and its substrate [22]. It was observed that increased pH (8.0) beyond optimum level showed 76% relative activity and when pH of reaction medium was further increased up to 9.0 and 10.0, the relative activity of enzyme reduced to 53% and 35%, respectively. In similar study, cellulase-free, thermo-alkali-solvent-stable xylanohydrolase from Bacillus altitudinis DHN8 and thermostable endo-type β-xylanase from streptomyces thermocarboxydus subspecies MW8 strain showed maximum residual activity at pH-7.0 [22,23]. Xylanohydrolase from Bacillus sp. PKD-9 was optimally active at pH-8.0 [24]. Majority of xylanohydrolases showed their catalytic activity at acidic or neutral pH [19,25]. Six recombinant xylanohydrolases from Acremonium cellulolyticus exhibited optimal activity at pH-4.0 to 4.5 [26].
Fig. 2.
(a) Effect of pH and (b) ionic strength of selected buffer on the catalytic activity of β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 (Means ± S.E., n = 3).
3.5. Effect of ionic strength of selected buffer on the hydrolytic activity of β-1,4-d-xylanohydrolase
Ionic strength of buffer is a very essential parameter to be analyzed for stabilization of an enzyme for maximum activity. Catalytic process is also influenced by the movement of charged molecules which ultimately affects the binding of charged substrate to enzymes and movement of the charged groups on the active site of any enzyme [27]. It was observed that β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 showed maximum catalytic activity when the buffer concentration was kept at 25.0 mM (Fig. 2b) and any alteration in optimum concentration led to the declined catalytic activity of enzyme. However, β-1,4-d-xylanohydrolase was also revealed to be active from 50.0 to 200.0 mM ionic strength, keeping pH-7.0 and 93% and 47% relative activity was found with buffer having ionic strength of 100.0 and 200.0 mM, respectively.
3.6. Effect of various solvents, surfactants and metal ion chelator on the activity of β-1,4-d-xylanohydrolase
In the current study, various organic solvents such as ethanol, methanol and isopropanol were found to enhance the enzymatic activity that showed 160%, 100% and 130% relative activity at 1.0 mM concentration, respectively. It was found that increase in concentration up to 5.0 mM stimulated the enzyme activity up to 3.5 fold in case of ethanol and 1.85 fold in methanol (Table 2). However, 5.0 mM concentration of isopropanol was found to be an inhibitor. These results suggested that the organic solvents generated specific effect on β-1,4-d-xylanohydrolase activity and stability. Similar finding has been reported with β-1,4-d-xylanohydrolase from Streptomyces sp. CS428 that indicated enhanced enzymatic activity with 1.49, 1.43, 1.45 fold increase by using 0.25% of ethanol, methanol and isopropanol, respectively [28]. It was also reported that xylanohydrolase from Bacillus altitudinis DHN8 revealed 1.08, 1.17 and 1.26 fold increase with ethanol, methanol and isopropanol, respectively [22]. Biocatalysis in organic solvents offers many advantages and increase the solubility of hydrophobic substrate, catalytic efficiency and decrease the microbial contamination [29].
Table 2.
Effect of various solvents, surfactants and metal ion chelator on the activity of β-1,4-d-xylanohydrolase produced from G. stearothermophilus KIBGE-IB29.
| Substances Used* |
Relative activity (%) |
|
|---|---|---|
| 1.0 mM | 5.0 mM | |
| Solvents | ||
| Control | 100 | 100 |
| Ethanol | 160.0 ± 8.0 | 350.0 ± 17.5 |
| Methanol | 100.0 ± 5.0 | 185.0 ± 9.2 |
| Isopropanol | 130.0 ± 6.5 | 7.0 ± 0.35 |
| DMSO | ND | ND |
| Formaldehyde | 260.0 ± 13.0 | 14.0 ± 0.7 |
| Chloroform | 220.0 ± 11.0 | 14.0 ± 0.7 |
| Surfactants | ||
| Control | 100 | 100 |
| Triton X-100 | 150.0 ± 7.5 | 35.0 ± 1.7 |
| Tween 80 | 230.0 ± 11.5 | 171.0 ± 8.5 |
| SDS | 290.0 ± 14.5 | 14.0 ± 0.7 |
| Metal ions chelator | ||
| Control | 100 | 100 |
| EDTA | ND | ND |
ND: Not detected.
The substances were incubated with β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 at 37 °C without substrate. The samples were retrieved after 60 min and the catalytic activity assay was performed at 50 °C for 5 min. All the experiments were conducted in triplicates and the data presented are a mean value of three observations.
Other organic solvents such as dimethyl sulfoxide (DMSO) caused complete loss of β-1,4-d-xylanohydrolase activity. Similar finding has been reported where thermostable cellulase free xylanohydrolase from Streptomyces sp. CS428 also showed inhibition in the presence of DMSO [28]. These results suggested that polarity of different solvents strongly affects the catalytic activity of enzymes. It has been observed that highly polarized solvents inactivate the enzymatic proteins by absorbing the water molecules surrounding the enzyme. Solvents that have a log P value <4 are considered as toxic due to higher degree of partitioning into aqueous layer. Enzymes that have capability to remain stable in organic phase can be used to catalyze different biochemical reactions and to develop bioremediation processes [30]. Furthermore, 1.0 mM concentration of formaldehyde and chloroform developed higher increase in catalytic activity of β-1,4-d-xylanohydrolase which showed 260% and 220% relative activity, respectively. However, 5.0 mM concentration generated inhibitory effect and ultimately declined the catalytic activity.
β-1,4-d-Xylanohydrolase activity was drastically increased in the presence of Tween 80 and Triton X-100 (1.0 mM) and up to 2.3 and 1.5 fold increase in catalytic activity was observed, respectively. It was observed that further increase in concentration (5.0 mM) of Tween 80 and Triton X-100 caused a negative effect on β-1,4-d-xylanohydrolase activity and only 1.71 fold increase was found in case of Tween 80. It was reported that Tween 80 and Triton X-100 do not play any role in the deactivation of β-1,4-d-xylanohydrolase and in the case of xylanohydrolase from Aspergillus awamori VTCC-F312, the catalytic activity was enhanced up to 28%and 16% in presence of Triton X-100 and Tween 80 [28,31]. The partially purified β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 showed 2.9 fold increase in its initial activity after mixing with 1.0 mM SDS and as the concentration increased up to 5.0 mM, a decline in activity was observed. It was reported that SDS showed its inhibitory effect on xylanohydrolase from Paenibacillus campinasensis G1-1 and it retained only 20% of its initial activity [32]. On the other hand, xylanohydrolase activity from Jonesia denitrificans BN-13 was stimulated up to 117% in the presence of 20.0 mM SDS [19]. It was found that EDTA completely inhibited the catalytic activity of β-1,4-d-xylanohydrolase as reported earlier [33].
3.7. Native-PAGE electrophoresis and In-gel activity assay of β-1,4-d-xylanohydrolase
Native-PAGE resulted a single protein band with molecular mass of about 48.7 kDa with reference to standard protein marker of Bovine serum albumin (BSA). Clear hydrolytic zone was also observed on zymogram which confirmed the presence of xylanolytic enzyme (Fig. 3). Molecular weight of different xylanohydrolases may vary from source to source. Bacillus licheniformis P11(C) produced two xylanohydrolase with 17.5 and 23 kDa molecular weight [3]. However, β-1,4-d-xylanohydrolase from Cellulosimicrobium sp. MTCC 10645 had a molecular mass of 78 kDa [34]. The molecular weight of β-1,4-d-xylanohydrolase (Xyn428) from Streptomyces sp. CS428 was estimated about 37 kDa with a single band [28].
Fig. 3.
Native-PAGE analysis and In-gel activity assay of β-1,4-d-xylanohydrolase. Lane M: Bovine serum albumin dimer and monomer protein marker. Lane 1: Dialyzed precipitates of enzyme. Lane 2: In-gel activity analysis of enzyme.
3.8. Thermal stability of β-1,4-d-xylanohydrolase
The heat tolerance or resistance to thermal denaturation of any enzyme is an important characteristic for different commercial applications [3]. It was found that 100% residual activity was achieved at 40 °C up to 120 min. Further increase in temperature up to 50 °C deactivated the enzyme which showed 95%, 83% and 63% residual activity after 30, 90 and 120 min, respectively (Fig. 4). The stability of β-1,4-d-xylanohydrolase was decreased as the temperature further increased up to 60 °C and 83%, 47% and 32% residual activity was found after 30, 90 and 120 min, respectively. The results indicated that β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 is substantially stable over high temperatures and could be of potential significance for different industrial processes. It was reported that β-1,4-d-xylanohydrolase from different bacterial sources showed their stability between 55 °C to 80 °C [35,36]. Bacillus sp. PKD-9 xylanohydrolase showed a half-life of 60 min at 45 °C [24]. Penicillium sp. endo-xylanase lost up to 63 to 68% activity at 60 °C after 90 min [37]. Thermal stability of enzyme encompasses kinetic stability, thermodynamics and highly dependent on the molecular interactions including disulfide bond, hydrogen bond, binding of metals as well as hydrophobic and electrostatic interactions present within the structure of an enzyme. By investigating aforementioned stability promoting characteristic different mesozymes can be re-engineered to thermozyme with enhanced thermo-tolerating efficiency [38,39].
Fig. 4.
Thermal stability of β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 (Means ± S.E., n = 3).
3.9. Storage stability of β-1,4-d-xylanohydrolase
The enzyme capability to retain its catalytic activity for long time period suggested its storage or shelf life. It was noticed that β-1,4-d-xylanohydrolase activity decreased gradually as time increased and higher storage efficiency of enzyme was noted at −80 °C where an enzyme showed 72% residual activity even after 180 days (Fig. 5). It was also found that enzyme retained 55% residual activity at 4 °C after 180 days. This declined catalytic activity might be due to the autocatalysis of enzyme at different temperatures. Storage at higher temperature decreases the chances of denaturation by ceasing the biochemical reaction or the alteration in three dimensional structure of enzyme. It was reported that the purified xylanohydrolase from Streptomyces cyaneus SN32 retained 100% activity up to 20 weeks at 4 °C [40]. β-1,4-d-Xylanohydrolase from Aspergillus foetidus showed its stability at −20 °C for 6 months and no activity lost was noted up to 15 days. It was found that on further increase in storage time only 5% to 10% loss in enzyme activity was observed [41].
Fig. 5.
Storage stability of β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 (Means ± S.E., n = 3).
4. Conclusion
It was concluded that partially purified β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 has an excellent catalytic properties. As most of the reported β-1,4-d-xylanohydrolase only show catalytic activity at the alkaline side and are limited to be used in specific industrial processes. However, β-1,4-d-xylanohydrolase from G. stearothermophilus KIBGE-IB29 exhibited broad pH profile from acidic to alkaline reaction environment. The enzyme also showed extended thermal and storage stability for long time period. These characteristics indicates the potential applications of β-1,4-d-xylanohydrolase for food technology in order to produce xylooligosaccharides as prebiotics and to clarify fruits and vegetable juices.
Competing interests
This publication is approved by all authors and they do not have any conflict of interest regarding any financial, personal or other relationships with any other people or organizations.
Acknowledgment
This research work was financially supported by the Karachi Institute of Biotechnology and Genetic Engineering (KIBGE), University of Karachi, Karachi-75270, Pakistan.
References
- 1.Spiridon I., Popa V.I. Hemicelluloses: Major sources, properties and applications. In: Belgacem M.N., Gandini J., editors. Monomers, Polymers and Composites from Renewable Resources. Elsevier; Amsterdam: 2008. pp. 289–304. [Google Scholar]
- 2.Collin T., Gerday C., Feller G. Xylanase, xylanase families and extremophilic xylanase. FEMS Microbiol. Rev. 2005;29:3–23. doi: 10.1016/j.femsre.2004.06.005. [DOI] [PubMed] [Google Scholar]
- 3.Bajaj B.K., Manhas K. Production and characterization of xylanase from Bacillus licheniformis P11(C) with potential for fruit juice and bakery industry. Biocatal. Agric. Biotechnol. 2012;1:330–337. [Google Scholar]
- 4.Bajaj B.K., Singh N.P. Production of xylanase from an alkalitolerant Streptomyces sp. 7b under solid-state fermentation, its purification and characterization. Appl. Biochem. Biotechnol. 2010;162:1804–1818. doi: 10.1007/s12010-010-8960-x. [DOI] [PubMed] [Google Scholar]
- 5.Butt M.S., Nadeem M.T., Ahmad Z., Sultan M.T. Xylanases and their applications in bakery industry. Food Technol. Biotechnol. 2008;46:22–31. [Google Scholar]
- 6.Polizeli M.L.T.M., Rizzatti A.C.S., Monti R., Terenzi H.F., Jorge J.A., Amorim D.S. Xylanase from fungi: properties and industrial applications. Appl. Microbiol. Biotechnol. 2005;67:577–591. doi: 10.1007/s00253-005-1904-7. [DOI] [PubMed] [Google Scholar]
- 7.Wong K.K.Y., Saddler J.N. Trichoderma xylanases, their properties and purification. Crit. Rev. Biotechnol. 1992;12:413–435. [Google Scholar]
- 8.Beg Q.K., Kapoor M., Mahajan L., Hoondal G.S. Microbial xylanases and their industrial applications: a review. Appl. Microbiol. Biotechnol. 2001;56:326–338. doi: 10.1007/s002530100704. [DOI] [PubMed] [Google Scholar]
- 9.Bochhini D.A., Gomes E., DaSilva R. Xylanase production using Bacillus circulans D1 using maltose as carbon source. Appl. Biochem. Biotechnol. 2008;146:29–37. doi: 10.1007/s12010-007-8051-9. [DOI] [PubMed] [Google Scholar]
- 10.Wu H., Li H., Xue Y., Luo G., Gan L., Liu J., Mao L., Long M. High efficiency co-production of ferulic acid and xylooligosaccharides from wheat bran by recombinant xylanase and feruloyl esterase. Biochem. Eng. J. 2017;120:41–48. [Google Scholar]
- 11.Anand A., Kumar V., Satyanarayana T. Characteristics of thermostable endoxylanase and β-xylosidase of the extremely thermophilic bacterium Geobacillus thermodenitrificans TSAA1 and its applicability in generating xylooligosaccharides and xylose from agro-residues. Extremophiles. 2013;17:357–366. doi: 10.1007/s00792-013-0524-x. [DOI] [PubMed] [Google Scholar]
- 12.Terrasan C.R.F., Guisan J.M., Carmona E.C. Xylanase and β-xylosidase from Penicillium janczewskii: purification, characterization and hydrolysis of substrates. Electron. J. Biotechnol. 2016;23:54–62. [Google Scholar]
- 13.Bibi Z., Ansari A., Zohra R.R., Aman A., Qader S.A.U. Production of xylan degrading endo-1,4-β-xylanase from thermophilic Geobacillus stearothermophilus KIBGE-IB29. J. Radiat. Res. Appl. Sci. 2014;7:478–485. [Google Scholar]
- 14.Miller G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugars. Anal. Chem. 1959;31:426–428. [Google Scholar]
- 15.Lowry O.H., Rosembrough N.J., Farr A.L., Randall R.J. Protein measurement with the folin phenol reagent. J. Biol. Chem. 1951;193:265–275. [PubMed] [Google Scholar]
- 16.Ornstein L. Disc electrophoresis I: background and theory. Ann. N. Y. Acad. Sci. 1964;121:321–349. doi: 10.1111/j.1749-6632.1964.tb14207.x. [DOI] [PubMed] [Google Scholar]
- 17.Davis B.J. Disc electrophoresis. II: method and application to human serum proteins. Ann. N. Y. Acad. Sci. 1964;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- 18.Zheng H.C., Liu Y.H., Liu X.G., Han Y., Wang J.L., Lu F.P. Overexpression of a Paenibacillus campinasensis xylanase in Bacillus megaterium and its applications to biobleaching of cotton stalk pulp and saccharification of recycled paper sludge. Bioresour. Technol. 2012;125:182–187. doi: 10.1016/j.biortech.2012.08.101. [DOI] [PubMed] [Google Scholar]
- 19.Boucherba N., Gagaoua M., Copinet E., Bettache A., Duchiron F., Benallaoua S. Purification and characterization of the xylanase produced by Jonesia denitrificans BN-13. Appl. Biochem. Biotechnol. 2014;172:2694–2705. doi: 10.1007/s12010-013-0709-x. [DOI] [PubMed] [Google Scholar]
- 20.He J., Su L., Sun X., Fu J., Chen J., Wu J. A novel xylanase from Streptomyces sp. FA1: purification, characterization, identification, and heterologous expression. Biotechnol. Bioprocess Eng. 2014;19:8–17. [Google Scholar]
- 21.Zheng H.C., Sun M.Z., Meng L.C., Pei H.S., Zhang X.G., Yan Z., Zeng W.H., Zhang J.S., Hu J.R., Lu F.P., Sun J.S. Purification and characterization of a thermostable xylanase from Paenibacillus sp. NF1 and its application in xylooligosaccharaides production. J. Microbiol. Biotechnol. 2014;24:489–496. doi: 10.4014/jmb.1312.12072. [DOI] [PubMed] [Google Scholar]
- 22.Adhyaru D.N., Bhatt N.S., Modi H.A. Enhanced production of cellulase-free, thermo-alkali-solvent-stable xylanase from Bacillus altitudinis DHN8, its characterization and application in sorghum straw saccharification. Biocatal. Agric. Biotechnol. 2014;3:182–190. [Google Scholar]
- 23.Chi W.J., Lim H.J., Park D.Y., Park J.S., Hong S.K. Production and characterization of a thermostable endo-type β-xylanase produced by a newly-isolated Streptomyces thermocarboxydus subspcies MW8 strain from jeju island. Process Biochem. 2013;48:1736–1743. [Google Scholar]
- 24.Panwar D., Sriyastaya P.K., Kapoor M. Production, extraction and characterization of alkaline xylanase from Bacillus sp. PKD-9 with potential for poultry feed. Biocatal. Agric. Biotechnol. 2014;3:118–125. [Google Scholar]
- 25.Kumar A., Gupta R., Shrivastava B., Khasa Y.P., Kuhad R.C. Xylanase production from an alkalophilic actinomycete isolate streptomyces sp. RCK-2010, its characterization and application in saccharification of second generation biomass. J. Mol. Catal., B Enzym. 2012;74:170–177. [Google Scholar]
- 26.Watanabe M., Inoue H., Inoue B., Yoshimi M., Fujii T., Ishikawa K. Xylanase (GH11) from Acremonium cellulolyticus: homologous expression and characterization. AMB Express. 2014;4:27. doi: 10.1186/s13568-014-0027-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rehman H.U., Aman A., Nawaz M.A., Qadar S.A.U. Characterization of pectin degrading polygalacturonase produced by Bacillus licheniformis KIBGE-IB21. Food Hydrocoll. 2015;43:819–824. [Google Scholar]
- 28.Pradeep G.C., Choi Y.H., Choi Y.S., Seong C.N., Cho S.S., Lee H.J., Yoo J.C. A novel thermostable cellulase free xylanase stable in broad range of pH from Streptomyces sp. CS428. Process Biochem. 2013;48:1188–1196. [Google Scholar]
- 29.Sardessai Y., Bhosle S. Industrial potential of organic solvent tolerant bacteria. Biotechnol. Prog. 2004;20:655–660. doi: 10.1021/bp0200595. [DOI] [PubMed] [Google Scholar]
- 30.Klibanov A.M. Improving enzymes by using them in organic solvents. Nature. 2001;409:241–246. doi: 10.1038/35051719. [DOI] [PubMed] [Google Scholar]
- 31.Do T.T., Quyen D.T., Dam T.H. Purification and characterization of an acid-stable and organic solvent-tolerant xylanase from Aspergillus awamori VTCC-F312. ScienceAsia. 2012;38:157–165. [Google Scholar]
- 32.Zheng H., Liu Y., Liu X., Wang J., Han Y., Lu F. Isolation, purification, and characterization of a thermostable xylanase from a novel strain, Paenibacillus campinasensis G1-1. J. Microbiol. Biotechnol. 2012;22:930–938. doi: 10.4014/jmb.1110.10060. [DOI] [PubMed] [Google Scholar]
- 33.Chapla D., Patel H., Singh A., Madawar D., Shah A. Production, purification and properties of a cellulase-free thermostable endoxylanase from newly isolated Paenibacillus sp. ASCD2. Ann. Microbiol. 2012;62:825–834. [Google Scholar]
- 34.Kamble R.D., Jadhav A.R. Production, purification and characterization of alkali stable xylanase from Cellulosimicrobium sp. MTCC 10645. Asian Pac. J. Trop. Biomed. 2012;2:1790–1797. [Google Scholar]
- 35.Dhiman S.S., Garg G., Sharma J., Mahajan R., Methoxy Characterization of statistically produced xylanase for enrichment of fruit juice clarification process. New Biotechnol. 2010;28:746–755. doi: 10.1016/j.nbt.2010.11.004. [DOI] [PubMed] [Google Scholar]
- 36.Kumar V., Satyanarayana T. Applicability of thermo-alkali-stable and cellulase-free xylanase from a novel thermo-halo-alkaliphilic Bacillus halodurans in producing xylooligosacchrides. Biotechnol. Lett. 2011;33:2279–2285. doi: 10.1007/s10529-011-0698-1. [DOI] [PubMed] [Google Scholar]
- 37.Bajaj B.K., Sharma M., Sharma S. Alkalistable endo-β-1,4-xylanase production from a newly isolated alkalitolerant Penicillium sp. SS1 using agro-residues. 3 Biotech. 2011;1:183–190. doi: 10.1007/s13205-011-0009-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bajaj B.K., Sharma P. An alkali-thermotolerant extracellular protease from a newly isolated Streptomyces sp. DP2. New Biotechnol. 2011;28:725–732. doi: 10.1016/j.nbt.2011.01.001. [DOI] [PubMed] [Google Scholar]
- 39.Li W.F., Zhou X.X., Lu P. Structural features of thermozymes. Biotechnol. Adv. 2005;23:271–281. doi: 10.1016/j.biotechadv.2005.01.002. [DOI] [PubMed] [Google Scholar]
- 40.Ninawe S., Kapoor M., Kuhad R.C. Purification and characterization of extracellular xylanase from Streptomyces cyaneus SN32. Bioresour. Technol. 2008;99:1252–1258. doi: 10.1016/j.biortech.2007.02.016. [DOI] [PubMed] [Google Scholar]
- 41.Shah A.R., Madamwar D. Xylanase production by a newly isolated Aspergillus foetidus strain and its characterization. Process Biochem. 2005;40:1763–1777. [Google Scholar]