Abstract
Previous studies have revealed that miR-142-5p serves a critical role in human cancer progression. However, the biological function of miR-142-5p in osteosarcoma (OS) development remains unclear. In the present study, the role of miR-142-5p in human OS HOS cells was determined, and the underlying mechanism involved was examined. Compared with the adjacent healthy tissues, the expression level of miR-142-5p was downregulated and the expression level of group XVI phospholipase A2 (PLA2G16) protein was upregulated in human OS tissues. The aforementioned results were also indicated in human OS HOS cells when compared with human fetal osteoblastic hFOB1.19 cells. Additionally, the results demonstrated that PLA2G16 was a direct target of miR-142-5p. miR-142-5p transfection upregulated the expression level of miR-142-5p and suppressed the expression level of PLA2G16 protein in HOS cells. MTT assays indicated a time-dependent decrease by miR-142-5p transfection in the proliferation of HOS cells. 5-bromo-2′-deoxyuridine incorporation assays confirmed that miR-142-5p transfection inhibited DNA synthesis in HOS cells. In addition, miR-142-5p transfection increased the Caspase-3 (CASP3) activity and apoptotic rate. Western blot analysis indicated that miR-142-5p transfection reduced BCL2, apoptosis regulator expression and upregulated the expression of CASP3 and BCL2 associated X, apoptosis regulator in HOS cells. Furthermore, miR-142-5p transfection decreased the expression levels of phosphorylated (p)-proto-oncogene, serine/threonine kinase, p-mitogen-activated protein kinase kinase, and p-extracellular signal-regulated kinase (ERK) 1/2 proteins in HOS cells. PLA2G16 overexpression restored the expression level of p-ERK 1/2 protein, which was reduced by miR-142-5p overexpression. MTT and CASP3 activity assays indicated that restoration of PLA2G16 reversed the tumour-suppressive role of miR-142-5p transfection in HOS cells. In conclusion, the results of the present study indicated that miR-142-5p suppressed proliferation and promoted apoptosis in human OS HOS cells by targeting PLA2G16 through ERK1/2 signaling pathway.
Keywords: miR-142-5p, Group XVI phospholipase A2, osteosarcoma, proliferation, apoptosis
Introduction
Osteosarcoma (OS) is an aggressive primary bone sarcoma, which occurs mainly in adolescents and young adults. OS has been reported to comprise 2.4% of all types of malignancy in paediatric patients and ~20% of all primary types of bone cancer (1). As the most common primary malignant bone tumour, OS has been reported to have a high metastatic rate and patients with metastasis have poorer prognosis with a lower 5-year survival rate (2). The prognosis and treatment of OS remains a challenge due to local relapse or metastases following surgical resection and intensive-chemotherapy (3). Therefore, it is essential to illustrate the underlying mechanism of OS progression regulation, to improve the prognosis, and to identify novel therapeutic targets and approaches for OS treatment.
microRNAs (miRNAs) are endogenous single-stranded, 19–22 nucleotide-long, noncoding RNAs that regulate post-transcriptional silencing by binding with partial complementarity to the 3′-untranslated region (3′-UTR) of the target mRNA (4). It has been reported that miRNAs are highly conserved and are able regulate ~30% of human genes (5). miRNAs have been demonstrated to be involved in multiple fundamental cell processes, including stem cell development, autophagy, transformation, proliferation control, cell differentiation, regulation of the cell cycle and cell apoptosis, as well as tumorigenesis (6,7). It has been validated that dysregulation of miRNAs can induce a number of human diseases and the abnormal expression of miRNAs has been closely associated with tumour progression, making them potential markers for cancer prognosis and diagnosis (8). Particularly, downregulation of miR-142-5p expression has been observed in OS human samples and compared with controls (9).
Group XVI phospholipase A2 (PLA2G16) expression, also known as H-REV-107, Ha-RAS like suppressor 3 (HRASLS3) and adipose specific PLA2 (AdPLA2), has been reported to be observed in the majority of healthy tissues (10) and was first identified as a class II tumour suppressor by its inhibition of Ras-mediated transformation (11). Furthermore, PLA2G16 overexpression suppresses proliferation and promotes apoptosis (12). PLA2G16 expression has been reported to be absent in various types of human cancer, including breast, ovary, kidney and testicular germ cells (13). Nevertheless, increased levels of PLA2G16 expression have been observed in lung, colon, stomach and rectal cancer, suggesting that PLA2G16 may serve an oncogenic role in the aforementioned tumours (14). It has also been reported that PLA2G16 is an important prognostic factor in patients with primary OS, predicting the development of metastases and poor survival and is associated with pulmonary metastasis and poor prognosis in human OS (15). Therefore, in the present study, the regulatory role of miR-142-5p was examined in proliferation and apoptosis, as well as its association with PLA2G16 expression, in the human OS cell line, HOS.
Materials and methods
Tissue samples and cell lines
OS tissue samples (n=21) and adjacent noncancerous tissues (n=21) were collected from patients at Honghui Hospital of Xi'an Jiaotong University (Xi'an, China) with written informed consent, in accordance with the Declaration of Helsinki of the World Medical Association. Diagnosis was confirmed by immunohistochemical analysis by pathologists from Honghui Hospital of Xi'an Jiaotong University (Xi'an, China) prior to the experimentation of the present study. The present study was approved by the Institutional Review Board and Human Ethics Committee of Xi'an Jiaotong University (Xi'an, China).
The human OS cell line, HOS, the human fetal osteoblastic cell line, hFOB1.19 and the 293T cell line were obtained from the American Type Culture Collection (Manassas, VA, USA). The hFOB1.19 cells were cultured in Dulbecco's modified Eagle's medium/F12 (DMEM/F12; Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) and HOS cells were cultured in DMEM medium (Gibco; Thermo Fisher Scientific, Inc.) all of which were supplemented with 10% fetal bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.) at 37°C with 5% CO2. The 293T cells were cultured in DMEM medium supplemented with 10% FBS (Gibco; Thermo Fisher Scientific, Inc.), high glucose (Gibco; Thermo Fisher Scientific, Inc.), L-glutamine and sodium pyruvate (all from Thermo Fisher Scientific, Inc.) at 37°C with 5% CO2.
Cell transfection
The miR-NC mimics (5′-GUGUAACACGUCUAUACGCCCA-3′) and miR-142-5p mimics (5′-CAUAAAGUAGAAAGCACUACU-3′) were designed and chemically synthesized (Shanghai GenePharma Co., Ltd., Shanghai, China). Cultured cells (4×105 cells/well) were seeded into 6-well plates (Corning Incorporated, Corning, NY, USA) for 24 h and transfected with 50 nM miR-NC or miR-142-5p mimics using Lipofectamine RNAiMAX (Invitrogen; Thermo Fisher Scientific, Inc.), according to the manufacturer's protocol. The transfection efficiency was measured using reverse transcription-quantitative polymerase chain reaction (RT-qPCR) assays. Cells were then subjected to further analysis 72 h following transfection as indicated in the results section. In addition, PLA2G16 coding sequence without the 3′-UTR was selected and cloned into pcDNA3.1 (Invitrogen; Thermo Fisher Scientific, Inc.) to overexpress PLA2G16. The plasmids were transfected into HOS cells using Lipofectamine® 2000, according to the manufacturer's protocol.
MTT assay
Proliferation was determined using the MTT assay. A total of 5×103 cells/well were seeded into 96-well plates (Corning Incorporated) following transfection. The cells were grown for 24, 48 and 72 h, and subsequently 20 µl of modified tetrazolium salt (5 mg/ml) was added to each well and incubated for 4 h at 37°C. The supernatant was subsequently removed and 100 µl dimethyl sulfoxide (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was added to dissolve the formazan crystals. The absorbance was evaluated at a wavelength of 490 nm with a Bio-Rad Benchmark micro-plate reader (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
5-Bromo-2′-deoxyuridine (BrdU) incorporation assay
BrdU incorporation during DNA synthesis was determined by ELISA using the BrdU kit (cat. no., C0088L; Beyotime Institute of Biotechnology, Shanghai, China) 72 h following transfection, according to the manufacturer's protocol. The assay was conducted in triplicate and the absorbance was detected at a wavelength of 450 nm with a microplate reader.
RT-qPCR
Total RNA, including miRNAs, from tissue samples or cells, was isolated by the miRNA Isolation kit (Ambion; Thermo Fisher Scientific, Inc.), according to the manufacturer's protocol. The miR-142-5p expression level was measured using the TaqMan miRNA assay (Applied Biosystems; Thermo Fisher Scientific, Inc.), according to the manufacturer's protocol. The miRNA expression levels were quantified to RNU48 expression. The relative miR-142-5p expression level was calculated by the 2−ΔΔCq method (16). The RT-qPCR reaction condition was as follows: Pre-degeneration at 95°C for 3 min, denaturation at 95°C for 30 sec, annealing at 60°C for 30 sec and extension at 72°C for 30 sec with a total of 40 cycles. The sequence of miR-142-5p primer was designed as follows: Forward, 5′-GGCCCATAAAGTAGAAAGC-3′ and reverse, 5′-TTTGGCACTAGCACATT-3′. Endogenous control RNU48 was amplified using forward, 5′-TGATGATGACCCCAGGTAACTC-3′ and reverse, 5′-GAGCGCTGCGGTGATG-3′. The above experiment was performed in triplicate. The sample that lacked cDNA was used as a negative control.
Caspase-3 (CASP3) activity assay
CASP3 activity was measured by the CASP3 kit (Beyotime Institute of Biotechnology), according to the manufacturer's protocols. Aliquots of protein were incubated with corresponding CASP3 substrate Ac-DEVD-pNA for 2 h at 37°C. The colorimetric release of phosphorylated (p)-nitroaniline from the Ac-DEVD-pNA substrate was recorded at wavelength of 405 nm.
Luciferase reporter assay
The potential binding site between miR-142-5p and PLA2G16 was initially obtained and analyzed online (http://www.targetscan.org/). The 3′-UTR of human PLA2G16, containing the miR-142-5p targeting sequence, was inserted into the pMIR-REPORT™ miRNA Expression Reporter Vector system (Ambion; Thermo Fisher Scientific, Inc.). Subsequently, the reporter plasmids, PLA2G16-wt 3′-UTR and PLA2G16-mut 3′-UTR, were co-transfected with miR-NC or miR-142-5p mimics into 293T cells using Lipofectamine® 2000. Cultured cells were collected following 48 h to measure reporter activity in comparison with Renilla luciferase activity using a Renilla Promega luciferase assay kit (Promega Corperation, Madison, WI, USA), according to the manufacturer's protocols.
Flow cytometry
The apoptotic rate was detected by flow cytometry analysis using the Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) apoptosis detection kit (R&D Systems Europe, Ltd., Abingdon, UK), according to the manufacturer's protocols. Briefly, cultured HOS cells were collected and suspended in annexin-binding buffer and then exposed to AnnexinV-FITC and PI for 12 min in the dark. The apoptotic rate was analyzed and quantified using a FACSCalibur flow cytometer equipped with CellQuest 6.0 software (BD Biosciences, Franklin Lakes, NJ, USA).
Western blot analysis
Cells were lysed using radioimmunoprecipitation assay buffer [50 mM Tris hydrochloride, pH 7.4, 1% Nonidet-P40, 0.25% sodium deoxycholate, 150 mM NaCl, 1 mM EDTA, 1 mM NaVO4, 2 mM NaF, Complete protease inhibitor cocktail (Roche Diagnostics, Indianapolis, IN, USA)], and the protein concentrations were examined by the Pierce BCA Protein Assay kit (Thermo Fisher Scientific, Inc.), according to the manufacturer's protocols. Equal amounts of protein (50 µg) were subsequently separated by 10% SDS-PAGE and transferred onto polyvinylidene fluoride membranes (EMD Millipore, Billerica, MA, USA). Subsequent to blocking using 5% BSA (Sangon Biotech Co. Ltd., Shanghai, China). for 1 h at room temperature, the membranes were incubated with primary antibodies against PLA2G16 (cat. no. 10337, Cayman Chemical Company, Ann Arbor, MI, USA) at a 5 ug/ml dilution; BCL2 associated X, apoptosis regulator (Bax; cat. no. sc20067), BCL2, apoptosis regulator (BCL2; cat. no. sc23960), proto-oncogene, serine/threonine kinase 1 (Raf-1; cat. no. sc373722) and p-Raf-1 (cat. no. sc271928) (dilution, 1:500; all from Santa Cruz Biotechnology, Inc., Dallas, TX, USA); CASP3 (cat. no. 9662), p-extracellular signal-regulated kinase (ERK) 1/2 (cat. no. 4370), ERK1/2 (cat. no. 4695), p-mitogen-activated protein kinase kinase (MEK; cat. no. 9154), MEK (cat. no. 4694) and β-actin (cat. no. 4970) (dilution, 1:1,000; all from Cell Signaling Technology, Inc., Danvers, MA, USA) at 4°C overnight. Following incubation with the secondary antibodies, including anti-rabbit (cat. no. A0208) or anti-mouse (cat. no. A0216) immunoglobulin G conjugated to horseradish peroxidase (dilution, 1:5,000; Beyotime Institute of Biotechnology) at 37°C for 1 h, the protein bands were visualized by Enhanced Chemiluminescent (ECL) kit (Beyotime Institute of Biotechnology), according to manufacturer's protocols. Band intensity was quantified and analyzed with Image J software 1.6 (National Institutes of Health, Bethesda, MD, USA).
Statistical analysis
The results of the present study were obtained from three independent experiments and analyzed using GraphPad Prism 5.0 software (GraphPad Software, Inc., La Jolla, CA, USA). The results are presented as the mean ± standard deviation. Statistical analysis was conducted by the conventional Student's t-test or paired Student's t-test. Multiple comparisons were performed with one-way analysis of variance followed by Least Significant Difference test. P<0.05 was considered to indicate a statistically significant difference compared with the respective control.
Results
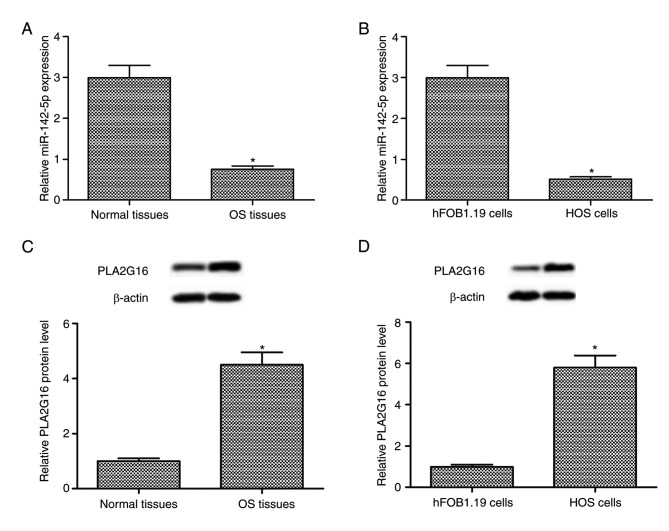
miR-142-5p downregulation and PLA2G16 upregulation in human OS tissues and HOS cells
The results of RT-qPCR and western blot analysis indicated that the expression level of miR-142-5p was downregulated and the expression level of PLA2G16 protein was upregulated in human OS tissues when compared with the adjacent healthy tissues (P<0.05; Fig. 1A and C). Compared with the human fetal osteoblastic hFOB1.19 cells, a similar expression pattern was observed in the human OS HOS cells (P<0.05; Fig. 1B and D). The aforementioned findings suggested that miR-142-5p downregulation and PLA2G16 upregulation was associated with the progression of human OS.
Figure 1.
The expression level of PLA2G16 protein and miR-142-5p in human OS tissues and adjacent healthy tissues, in addition to human hFOB1.19 cells and HOS cells. (A) miR-142-5p expression level in OS tissues compared with adjacent healthy tissues. (B) miR-142-5p expression level of human hFOB1.19 cells compared with HOS cells. (C) PLA2G16 protein expression level in OS tissues compared with adjacent healthy tissues. (D) PLA2G16 protein expression level of human hFOB1.19 cells compared with HOS cells. *P<0.05 compared with normal tissues or hFOB1.19 cells. OS, osteosarcoma; miR, microRNA; PLA2G16, group XVI phospholipase A2.
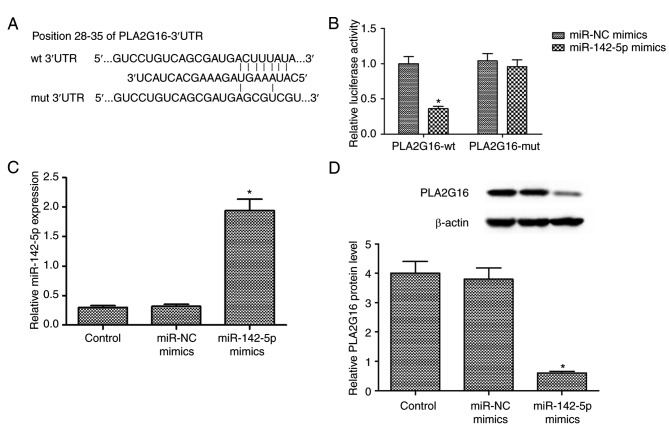
PLA2G16 is a direct target of miR-142-5p
The potential underlying mechanism between miR-142-5p and PLA2G16 was investigated by bioinformatics prediction (http://www.targetscan.org). The results indicated one putative binding site between miR-142-5p and 3′-UTR of PLA2G16 (Fig. 2A). Luciferase reporter assays revealed that miR-142-5p overexpression significantly reduced the luciferase activity of the PLA2G16-wt 3′-UTR-transfected cells, whereas PLA2G16-mut 3′-UTR blocked this effect (P<0.05; Fig. 2B). Following transfection with miR-142-5p mimics and miR-NC, RT-qPCR analysis revealed that miR-142-5p transfection upregulated the expression of mature miR-142-5p in HOS cells (P<0.05; Fig. 2C). Furthermore, miR-142-5p overexpression significantly reduced the expression level of PLA2G16 protein in HOS cells (Fig. 2D). The aforementioned data illustrated that PLA2G16 was a direct target of miR-142-5p.
Figure 2.
PLA2G16 is a direct target gene of miR-142-5p. (A) A microRNA database search predicted an miR-142-5p binding site on the PLA2G16 3′-UTR. (B) miR-142-5p overexpression in 293T cells reduced the luciferase activity of cells transfected with pMIR-PLA2G16-wt 3′-UTR, whereas pMIR-PLA2G16-mut 3′-UTR blocked this effect. (C) Following transfection with miR-142-5p mimics or miR-NC, miR-142-5p transfection induced an elevation in the expression level of mature miR-142-5p in HOS cells. (D) The expression level of PLA2G16 protein in HOS cells was detected and measured 72 h following transfection with miR-142-5p and miR-NC mimics. *P<0.05 compared with control. miR, microRNA; PLA2G16, group XVI phospholipase A2; NC, negative control; UTR, untranslated region; wt, wild type; mut, mutated.
miR-142-5p overexpression suppresses proliferation and promotes apoptosis in HOS cells
The effects of miR-142-5p on proliferation and apoptosis were examined in HOS cells transfected with miR-NC or miR-142-5p mimics. MTT assays demonstrated that miR-142-5p overexpression significantly inhibited in a time-dependent manner the proliferation of HOS cells (P<0.05; Fig. 3A). The results of BrdU incorporation assays confirmed that miR-142-5p overexpression inhibited DNA synthesis in HOS cells (P<0.05; Fig. 3B). A significant increase of CASP3 activity was observed following miR-142-5p overexpression (P<0.05; Fig. 4A). Additionally, miR-142-5p overexpression significantly elevated the percentage of apoptotic cells (P<0.05; Fig. 4B). Western blot analysis indicated that miR-142-5p overexpression significantly promoted apoptosis, along with BCL2 reduction and elevation of CASP3 and Bax expression level in HOS cells (P<0.05; Fig. 4C). Therefore, the results revealed that miR-142-5p overexpression suppressed proliferation and promoted apoptosis in HOS cells.
Figure 3.
Effects of miR-142-5p on the proliferation in HOS cells. (A) Proliferation of HOS cells transfected with miR-142-5p mimics or miR-NC at 24, 48 and 72 h by MTT assay. (B) BrdU incorporation assays confirmed that miR-142-5p overexpression inhibited DNA synthesis in HOS cells transfected with miR-142-5p and miR-NC mimics. *P<0.05 compared with control. BrdU, 5-Bromo-2′-deoxyuridine; miRNAs, microRNAs; NC, negative control; OD, optical density.
Figure 4.
Effect of miR-142-5p on apoptosis of HOS cells. (A) Increased CASP3 activity was observed following miR-142-5p overexpression in HOS cells transfected with miR-142-5p and miR-NC mimics. (B) Apoptotic percentage measured by flow cytometry in HOS cells transfected with miR-142-5p and miR-NC mimics. (C) The expression level of CASP3, BCL2, Bax and β-actin proteins were detected and measured. miR-142-5p overexpression promoted apoptosis, reduced BCL2 expression and elevated CASP3 and Bax expression levels in HOS cells. *P<0.05 compared with control. Bax, BCL2-associated X, apoptosis regulator; BCL2, BCL2, apoptosis regulator; CASP3, Caspase-3; miR, microRNA; Annexin V-FITC, Annexin V-fluorescein isothiocyanate; PI, propidium iodide; NC, negative control.
Restoration of PLA2G16 reverses the tumour-suppressive roles of miR-142-5p overexpression in HOS cells through ERK1/2 signaling pathway
The results of the western blot analysis demonstrated that miR-142-5p transfection significantly decreased the expression levels of p-Raf-1, p-MEK, and p-ERK1/2 proteins in HOS cells (P<0.05; Fig. 5A). To further examine whether the role of miR-142-5p in OS progression was regulated through PLA2G16, the pCDNA3.1-PLA2G16 vector was transfected into HOS cells. The results demonstrated that PLA2G16 expression was significantly enhanced in HOS cells following transfection with pCDNA3.1-PLA2G16 and PLA2G16 overexpression rescued the reduced expression level of p-ERK1/2 protein associated with miR-142-5p transfection in HOS cells (P<0.05; Fig. 5B). MTT and CASP3 activity assays were conducted in HOS cells following transfection with miR-142-5p mimics either with or without pCDNA3.1-PLA2G16. The results revealed that restoration of PLA2G16 reversed the tumour-suppressive roles of miR-142-5p overexpression in HOS cells (P<0.05; Fig. 5C and D). The aforementioned findings suggest that miR-142-5p overexpression suppressed proliferation and promoted apoptosis in HOS cells by the ERK1/2 signaling pathway.
Figure 5.
Effects of miR-142-5p and PLA2G16 on p-ERK1/2 protein expression, proliferation and apoptosis of HOS cells. HOS cells were transfected with miR-142-5p, miR-NC mimics and pcDNA3.1-PLA2G16 plasmids. (A) The expression level of p-Raf-1, Raf-1, p-MEK, MEK, p-ERK1/2, ERK1/2 and β-actin proteins. miR-142-5p transfection decreased the expression levels of p-Raf-1, p-MEK, and p-ERK1/2 proteins in HOS cells. (B) The expression levels of PLA2G16, p-ERK1/2, ERK1/2 and β-actin proteins were detected and measured. The PLA2G16 expression level was enhanced in HOS cells following transfection with pCDNA3.1-PLA2G16 and PLA2G16 overexpression rescued the reduced expression level of p-ERK1/2 protein associated with miR-142-5p transfection in HOS cells. (C) Apoptotic rate of HOS cells following transfection with miR-142-5p mimics either with or without pCDNA3.1-PLA2G16 at 24, 48 and 72 h. (D) CASP3 activity assays were conducted in HOS cells following transfection with miR-142-5p mimics either with or without pCDNA3.1-PLA2G16. *P<0.05 compared with control. #P<0.05 compared with miR-142-5p mimics. OD, optical density; p, phosphorylated; Raf-1, proto-oncogene, serine/threonine kinase 1, CASP3, Caspase-3; ERK1/2, extracellular signal-regulated kinase 1/2; MEK, mitogen-activated protein kinase kinase; NC, negative control; PLA2G16, group XVI phospholipase A2.
Discussion
Despite OS being an uncommon malignant mesenchymal neoplasm, it is the third most common cancer-associated disease among children and adolescents (17). An increasing number of novel miRNAs have been identified, which mediate complex processes, including osteogenic differentiation and osteoblastic bone formation (18). However, the dysregulation and abnormal expression of miRNAs have been reported to accelerate the development of OS (19,20). Therefore, it is essential to investigate the abnormal expression of miRNAs as the first step to characterize the miRNA-mediated pathways involved in human OS progression.
Accumulating evidence has indicated that miR-142-5p expression levels are upregulated in a number of pathological contexts, including small bowel inflammation (21), Hashimoto's thyroiditis (22), simian immunodeficiency virus encephalitis (23) and atherosclerosis (24). It has been demonstrated that miR-142-5p is a potential tumour suppressor gene in numerous types of cancer, including colon cancer (25). Reduced miR-142-5p expression levels have been reported to be associated with a high frequency of recurrence and a poor survival rate in patients with gastric cancer (26). It has been demonstrated that miR-142-5p expression is downregulated in transgenic lung cancer and that miR-142-5p overexpression inhibits proliferation in lung cancer (27). It has also been reported that miR-142-5p overexpression suppresses hepatocellular carcinoma proliferation and promotes apoptosis by regulating forkhead box O (28). Furthermore, reduced miR-142-5p expression has been observed in osteosarcomagenesis, as it is used as an miRNA signature to indicate the expression identity of OS (9). In addition, miR-142-5p has been reported to contribute to bone repair by sustaining osteoblast activity (29). In the present study, the observed levels of miR-142-5p expression were downregulated in human OS tissues and HOS cells when compared with adjacent healthy tissues and human fetal osteoblastic hFOB1.19 cells. In addition, it was indicated that miR-142-5p overexpression suppressed proliferation and promoted apoptosis in human osteosarcoma HOS cells.
Evidence suggests that PLA2G16 is associated with tumour progression (14), and that it may contribute to tumour progression through altered metabolic pathways (30). As a phospholipase, PLA2G16 produces lysophosphatidic acid (LPA) and free fatty acid (FFA) from phosphatidic acid, which has been reported to promote proliferation, migration and metastasis in cancer progression (31). It has been demonstrated that LPA promotes tumour progression by modulating cytoskeletal changes, cell-cell contacts, cell survival, proliferation, invasion and metastasis through activating multiple signaling pathways, including HRAS, MAPK, Rac family small GTPase, rhodopsin, phospholipase C, AKT, serine/threonine kinase and Hippo-YAP pathways (32,33). It has been demonstrated that FFAs contribute to the production of prostaglandin Es, which serves an important role in cancer pathogenesis (10). Furthermore, PLA2G16 has been reported to promote OS progression and metastasis in mouse and human OS cells (15). Similarly, the findings of the present study indicated that compared to adjacent healthy tissues and human fetal osteoblastic hFOB1.19 cells, the expression levels of the PLA2G16 protein were upregulated in human OS tissues and HOS cells. Furthermore, it was demonstrated that PLA2G16 was a direct target of miR-142-5p and that miR-142-5p overexpression suppressed proliferation and promoted apoptosis by targeting PLA2G16 in human osteosarcoma HOS cells.
It has been demonstrated that the MAPK/ERK pathway is involved in the processes of proliferation, differentiation, migration, senescence and apoptosis (34). As a key signal transduction pathway, the ERK signaling pathway has been reported to be frequently aberrantly activated in cancer cells and considered central to a number of signaling pathways (35). miR-142-5p has been reported to mediate certain target genes in numerous oncogenic signaling pathways, including MAPK (26). The underlying mechanism of miR-142 function in mediating ERK phosphorylation has been illustrated and decreased miR-142 expression levels lead to enhanced levels of ERK phosphorylation, contributing to cell differentiation in mouse embryonic stem cells (36,37). It has also been demonstrated that enhanced PLA2G16 expression promotes OS metastasis through activating the MAPK pathway and may be a novel therapeutic target for various types of cancer (38). The findings of the present study indicated that miR-142-5p transfection decreased the phosphorylation of Raf-1/MEK/ERK1/2 proteins in HOS cells, suggesting that miR-142-5p may act by the Raf/MEK/ERK1/2 signaling pathway. Additionally, PLA2G16 overexpression restored the expression levels of p-ERK1/2 protein reduced by miR-142-5p overexpression. PLA2G16 restoration reversed the tumour-suppressive roles of miR-142-5p overexpression in HOS cells.
In conclusion, the results of the present study confirmed the tumor-suppressive role of miR-142-5p in human OS progression. The aforementioned findings suggested that miR-142-5p suppressed proliferation and promoted apoptosis in human osteosarcoma HOS cells by targeting PLA2G16 by the ERK1/2 signaling pathway. Therefore, the present study provided novel insight into the underlying mechanisms of OS progression, suggesting that miR-142-5p may be used as a novel candidate therapeutic target in OS. Considering that only a single OS cell line was used to elucidate the effects of miR-142-5p, further studies are required, in order to fully elucidate the roles of miR-142-5p and PLA2G16 in proliferation and apoptosis of OS cells. Additional research is also required to determine the potential of miR-142-5p in clinical applications.
Acknowledgements
Not applicable.
Glossary
Abbreviations
- OS
Osteosarcoma
- miRNAs
microRNAs
- 3′UTR
3′-untranslated region
- DMEM
Dulbecco's modified Eagle's medium
- PLA2G16
Group XVI phospholipase A2
- HRASLS3
Ha-RAS like suppressor 3
- AdPLA2
adipose specific PLA2
- ATCC
American Type Culture Collection
- MTT
modified tetrazolium salt 3-(4,5-dimethyl-2-thiazolyl)-2,5-dipheny-2 H-tetrazolium-bromide
- DMSO
dimethyl sulfoxide
- BrdU
5-Bromo-2′-deoxyuridine
- PVDF
polyvinylidene fluoride membranes
- RT-qPCR
reverse transcription-quantitative polymerase chain reaction
- SD
standard deviation
- LPA
lysophosphatidic acid
- FBS
fetal bovine serum
- FFA
free fatty acid
- ELISA
enzyme-linked immunosorbent assay
- MAPK/ERK
mitogen-activated protein kinases/extracellular signal-regulated kinase
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The Institutional Review Board and Human Ethics Committee of Xi'an Jiaotong University (Xi'an, China) approved the study. All patients signed written informed consent for participation in the present study, which was conducted according to the Declaration of Helsinki.
Authors' contributions
DC and LH designed the study, collected the literature, analyzed and interpreted the data, and prepared the manuscript. JL collected, analyzed and interpreted the data, and prepared the manuscript. LZ and LH collected the literature, and analyzed and interpreted the data. All authors approved the final manuscript.
Patient consent for publication
Patient consent was obtained for the publication of the present study.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Longhi A, Errani C, De Paolis M, Mercuri M, Bacci G. Primary bone osteosarcoma in the pediatric age: State of the art. Cancer Treat Rev. 2006;32:423–436. doi: 10.1016/j.ctrv.2006.05.005. [DOI] [PubMed] [Google Scholar]
- 2.Aljubran AH, Griffin A, Pintilie M, Blackstein M. Osteosarcoma in adolescents and adults: Survival analysis with and without lung metastases. Ann Oncol. 2009;20:1136–1141. doi: 10.1093/annonc/mdn731. [DOI] [PubMed] [Google Scholar]
- 3.Ferreira CG, de Melo AC, Nogueira-Rodrigues A. The adolescent and young adult with cancer: State of the art-epithelial cancer. Curr Oncol Rep. 2013;15:287–295. doi: 10.1007/s11912-013-0322-8. [DOI] [PubMed] [Google Scholar]
- 4.Bartel DP. MicroRNAs: Target recognition and regulatory functions. Cell. 2009;136:215–233. doi: 10.1016/j.cell.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell. 2005;120:15–20. doi: 10.1016/j.cell.2004.12.035. [DOI] [PubMed] [Google Scholar]
- 6.Li S, Wang L, Fu B, Berman MA, Diallo A, Dorf ME. TRIM65 regulates microRNA activity by ubiquitination of TNRC6. Proc Natl Acad Sci USA. 2014;111:6970–6975. doi: 10.1073/pnas.1322545111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu S, Yin F, Zhang J, Wicha MS, Chang AE, Fan W, Chen L, Fan M, Li Q. Regulatory roles of miRNA in the human neural stem cell transformation to glioma stem cells. J Cell Biochem. 2014;115:1368–1380. doi: 10.1002/jcb.24786. [DOI] [PubMed] [Google Scholar]
- 8.Cheng G. Circulating miRNAs: Roles in cancer diagnosis, prognosis and therapy. Adv Drug Deliv Rev. 2015;81:75–93. doi: 10.1016/j.addr.2014.09.001. [DOI] [PubMed] [Google Scholar]
- 9.Jones KB, Salah Z, Del Mare S, Galasso M, Gaudio E, Nuovo GJ, Lovat F, LeBlanc K, Palatini J, Randall RL, et al. miRNA signatures associate with pathogenesis and progression of osteosarcoma. Cancer Res. 2012;72:1865–1877. doi: 10.1158/0008-5472.CAN-11-2663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Duncan RE, Sarkadi-Nagy E, Jaworski K, Ahmadian M, Sul HS. Identification and functional characterization of adipose-specific phospholipase A2 (AdPLA) J Biol Chem. 2008;283:25428–25436. doi: 10.1074/jbc.M804146200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hajnal A, Klemenz R, Schafer R. Subtraction cloning of H-rev107, a gene specifically expressed in H-ras resistant fibroblasts. Oncogene. 1994;9:479–490. [PubMed] [Google Scholar]
- 12.Nazarenko I, Schafer R, Sers C. Mechanisms of the HRSL3 tumor suppressor function in ovarian carcinoma cells. J Cell Sci. 2007;120:1393–1404. doi: 10.1242/jcs.000018. [DOI] [PubMed] [Google Scholar]
- 13.Siegrist S, Feral C, Chami M, Solhonne B, Mattei MG, Rajpert-De Meyts E, Guellaen G, Bulle F. hH-Rev107, a class II tumor suppressor gene, is expressed by post-meiotic testicular germ cells and CIS cells but not by human testicular germ cell tumors. Oncogene. 2001;20:5155–5163. doi: 10.1038/sj.onc.1204658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nazarenko I, Kristiansen G, Fonfara S, Guenther R, Gieseler C, Kemmner W, Schafer R, Petersen I, Sers C. H-REV107-1 stimulates growth in non-small cell lung carcinomas via the activation of mitogenic signaling. Am J Pathol. 2006;169:1427–1439. doi: 10.2353/ajpath.2006.051341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liang S, Ren Z, Han X, Yang J, Shan L, Li L, Wang B, Zhang Q, Mu T, Chen K, et al. PLA2G16 expression in human osteosarcoma is associated with pulmonary metastasis and poor prognosis. PLoS One. 2015;10:e0127236. doi: 10.1371/journal.pone.0127236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 17.Wolf NK, Largaespada DA, Moriarity BS. Coping with cancer genes altered by copy number. Oncotarget. 2015;6:35155–35156. doi: 10.18632/oncotarget.6215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wei J, Shi Y, Zheng L, Zhou B, Inose H, Wang J, Guo XE, Grosschedl R, Karsenty G. miR-34s inhibit osteoblast proliferation and differentiation in the mouse by targeting SATB2. J Cell Biol. 2012;197:509–521. doi: 10.1083/jcb.201201057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang J, Yan YG, Wang C, Zhang SJ, Yu XH, Wang WJ. MicroRNAs in osteosarcoma. Clin Chim Acta. 2015;444:9–17. doi: 10.1016/j.cca.2015.01.025. [DOI] [PubMed] [Google Scholar]
- 20.Sampson VB, Yoo S, Kumar A, Vetter NS, Kolb EA. MicroRNAs and potential targets in osteosarcoma: Review. Front Pediatr. 2015;3:69. doi: 10.3389/fped.2015.00069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schaefer JS, Montufar-Solis D, Vigneswaran N, Klein JR. Selective upregulation of microRNA expression in peripheral blood leukocytes in IL-10-/- mice precedes expression in the colon. J Immunol. 2011;187:5834–5841. doi: 10.4049/jimmunol.1100922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhu J, Zhang Y, Zhang W, Fan L, Wang L, Liu Y, Liu S, Guo Y, Wang Y, Yi J, et al. MicroRNA-142-5p contributes to hashimoto's thyroiditis by targeting CLDN1. J Transl Med. 2016;14:166. doi: 10.1186/s12967-016-0917-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chaudhuri AD, Yelamanchili SV, Marcondes MC, Fox HS. Up-regulation of microRNA-142 in simian immunodeficiency virus encephalitis leads to repression of sirtuin1. FASEB J. 2013;27:3720–3729. doi: 10.1096/fj.13-232678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Xu R, Bi C, Song J, Wang L, Ge C, Liu X, Zhang M. Upregulation of miR-142-5p in atherosclerotic plaques and regulation of oxidized low-density lipoprotein-induced apoptosis in macrophages. Mol Med Rep. 2015;11:3229–3234. doi: 10.3892/mmr.2015.3191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shi D, Zhai B, Zheng Y, Ren R, Han M, Wang X. Transcatheter arterial infusion chemotherapy increases expression level of miR-142-5p in stage III colorectal cancer. Indian J Cancer. 2015;52(Suppl 2):e47–e55. doi: 10.4103/0019-509X.172513. [DOI] [PubMed] [Google Scholar]
- 26.Zhang X, Yan Z, Zhang J, Gong L, Li W, Cui J, Liu Y, Gao Z, Li J, Shen L, Lu Y. Combination of hsa-miR-375 and hsa-miR-142-5p as a predictor for recurrence risk in gastric cancer patients following surgical resection. Ann Oncol. 2011;22:2257–2266. doi: 10.1093/annonc/mdq758. [DOI] [PubMed] [Google Scholar]
- 27.Liu X, Sempere LF, Galimberti F, Freemantle SJ, Black C, Dragnev KH, Ma Y, Fiering S, Memoli V, Li H, et al. Uncovering growth-suppressive MicroRNAs in lung cancer. Clin Cancer Res. 2009;15:1177–1183. doi: 10.1158/1078-0432.CCR-08-1355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lou K, Chen N, Li Z, Zhang B, Wang X, Chen Y, Xu H, Wang D, Wang H. MicroRNA-142-5p overexpression inhibits cell growth and induces apoptosis by regulating FOXO in hepatocellular carcinoma cells. Oncol Res. 2017;25:65–73. doi: 10.3727/096504016X14719078133366. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 29.Tu M, Tang J, He H, Cheng P, Chen C. MiR-142-5p promotes bone repair by maintaining osteoblast activity. J Bone Miner Metab. 2017;35:255–264. doi: 10.1007/s00774-016-0757-8. [DOI] [PubMed] [Google Scholar]
- 30.Jaworski K, Ahmadian M, Duncan RE, Sarkadi-Nagy E, Varady KA, Hellerstein MK, Lee HY, Samuel VT, Shulman GI, Kim KH, et al. AdPLA ablation increases lipolysis and prevents obesity induced by high-fat feeding or leptin deficiency. Nat Med. 2009;15:159–168. doi: 10.1038/nm.1904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nomura DK, Long JZ, Niessen S, Hoover HS, Ng SW, Cravatt BF. Monoacylglycerol lipase regulates a fatty acid network that promotes cancer pathogenesis. Cell. 2010;140:49–61. doi: 10.1016/j.cell.2009.11.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee SC, Fujiwara Y, Tigyi GJ. Uncovering unique roles of LPA receptors in the tumor microenvironment. Receptors Clin Investig. 2015;2:e440. doi: 10.14800/rci.440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang H, Liu W, Wei D, Hu K, Wu X, Yao Y. Effect of the LPA-mediated CXCL12-CXCR4 axis in the tumor proliferation, migration and invasion of ovarian cancer cell lines. Oncol Lett. 2014;7:1581–1585. doi: 10.3892/ol.2014.1926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sun Y, Liu WZ, Liu T, Feng X, Yang N, Zhou HF. Signaling pathway of MAPK/ERK in cell proliferation, differentiation, migration, senescence and apoptosis. J Recept Signal Transduct Res. 2015;35:600–604. doi: 10.3109/10799893.2015.1030412. [DOI] [PubMed] [Google Scholar]
- 35.Yu Z, Ye S, Hu G, Lv M, Tu Z, Zhou K, Li Q. The RAF-MEK-ERK pathway: Targeting ERK to overcome obstacles to effective cancer therapy. Future Med Chem. 2015;7:269–289. doi: 10.4155/fmc.14.143. [DOI] [PubMed] [Google Scholar]
- 36.Sladitschek HL, Neveu PA. The bimodally expressed microRNA miR-142 gates exit from pluripotency. Mol Syst Biol. 2015;11:850. doi: 10.15252/msb.20156525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shrestha A, Mukhametshina RT, Taghizadeh S, Vasquez-Pacheco E, Cabrera-Fuentes H, Rizvanov A, Mari B, Carraro G, Bellusci S. MicroRNA-142 is a multifaceted regulator in organogenesis, homeostasis, and disease. Dev Dyn. 2017;246:285–290. doi: 10.1002/dvdy.24477. [DOI] [PubMed] [Google Scholar]
- 38.Li L, Liang S, Wasylishen AR, Zhang Y, Yang X, Zhou B, Shan L, Han X, Mu T, Wang G, Xiong S. PLA2G16 promotes osteosarcoma metastasis and drug resistance via the MAPK pathway. Oncotarget. 2016;7:18021–18035. doi: 10.18632/oncotarget.7694. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.