Abstract
There is a clear need to establish and validate new methodologies to more quickly and efficiently screen chemicals for potential toxic effects, particularly on development. The emergence of alternative animal systems for rapid toxicology screens presents valuable opportunities to evaluate how systems complement each other. In this article, we compare a chemical library of 87-compounds in 2 such systems, developing zebrafish and freshwater planarians, by screening for developmental neurotoxic effects. We show that the systems’ toxicological profiles are complementary to each other, with zebrafish yielding more detailed morphological endpoints and planarians more behavioral endpoints. Overall, zebrafish was more sensitive to this chemical library, yielding 86/87 hits, compared with 50/87 hits in planarians. The difference in sensitivity could not be attributed to molecular weight, log Kow, or the bioconcentration factor. Of the 87 chemicals, 28 had previously been evaluated in mammalian developmental neuro- (DNT), neuro-, or developmental toxicity studies. Of the 28, 20 were hits in the planarian, and 27 were hits in zebrafish. Eighteen of the 28 had previously been identified as DNT hits in mammals and were highly associated with activity in zebrafish and planarian behavioral assays in this study. Only 1 chemical (of 28) was a false negative in both zebrafish and planarian systems. The differences in endpoint coverage and system sensitivity illustrate the value of a dual systems approach to rapidly query a large chemical-bioactivity space and provide weight-of-evidence for prioritization of chemicals for further testing.
Keywords: alternative animal models, planarian, zebrafish, developmental neurotoxicity
This article is published as part of the NTP Neurotoxicology Screening Strategies Initiative.
There is increasing recognition in the field of toxicology that the high expense, low throughput, and uncertainty of traditional rodent testing are inadequate to evaluate the ever-growing number of environmental chemicals. Alternative integrated systems, such as zebrafish, nematodes, and planarians, have emerged to fill these gaps because they are small, easy to breed and maintain, comparatively inexpensive, and develop quickly (Boyd et al., 2012, 2015; Hagstrom et al., 2016; Hill et al., 2005; Tejeda-Benitez and Olivero-Verbel, 2016; Truong et al., 2014). The use of these models, with their own unique strengths and limitations, is an effective bridge between in vitro and whole animal/mammalian testing methods, allowing for rapid exploration of adverse outcome pathways. Comparative screening in multiple such animal models covers more chemical-biological space, thus providing greater weight of evidence for prioritization of chemical hazards.
Recently, the NIEHS National Toxicology Program (NTP) initiated a collaboration wherein different labs studying a variety of alternative models, including in vitro cell culture systems, zebrafish, and planarians, screened a library of 87 unique compounds, allowing for cross-system comparisons (Behl et al., 2018). This library consisted of known and suspected developmental neurotoxicants and 5 designated negative control chemicals, expanded from a previously tested library of 80 compounds (Ryan et al., 2016). Here we compare the results of screening this library in a developmental zebrafish model and freshwater planarian system.
Zebrafish embryos and larvae have become a popular animal model for developmental (Linney et al., 2004) and neurotoxicity studies (Bailey et al., 2013; He et al., 2014) because this system strikes an optimal balance among model complexity and tractability. This system is amenable to high-throughput whole animal screening (Geier et al., 2018; Noyes et al., 2015; Truong et al., 2014) and maintains strong developmental and physiological similarity to higher vertebrates. In particular, the external development and optical transparency of zebrafish embryos allows for detailed morphological studies. Moreover, 84% of genes that have been associated with a human disease have an obvious orthologue in zebrafish (Howe et al., 2013).
The asexual freshwater planarian Dugesia japonica has recently emerged as a high-throughput, alternative whole animal model for developmental neurotoxicology (Hagstrom et al., 2015, 2016). Planarians have a high capacity to regenerate, which allows them to reproduce asexually by binary fission (Malinowski et al., 2017) and fully regenerate a centralized nervous system (Cebrià, 2007; Rink, 2013). In this asexual species, neurodevelopment is achieved through neuroregeneration and can be induced by decapitation. Because of the similar sizes of adult and regenerating/developing animals, the planarian system offers the unique ability to screen both life stages in parallel with the same assays to identify potential effects specific to development. Moreover, planarians possess a large repertoire of quantifiable behaviors, many of which can be assayed in a fully automated fashion, including thermotaxis, phototaxis, and a characteristic escape response, providing distinct readouts of neuronal function (Cochet-Escartin et al., 2015; Hagstrom et al., 2016; Inoue et al., 2014; Paskin et al., 2014; Zhang et al., 2018).
Because the systems differ in their developmental timeline, we compared the chemical effects at 2 different stages: (1) early development (24 hours post fertilization [hpf] in the zebrafish and day 7 in the planarian), when the animals begin to form major anatomical structures and (2) late development (day 5 or 120 hpf in the zebrafish and day 12 in the planarian), when the animals have essentially completed development. We assayed for developmental delays, morphological abnormality, mortality, and altered behavioral endpoints. Eighty-six of 87 chemicals were bioactive in the zebrafish, whereas 50 were bioactive in regenerating planarians. Physicochemical properties (eg, molecular weight [MW], log octanol/water partition coefficient [log Kow], and bioconcentration factor [BCF]) did not account for bioactivity differences between the models. Of the 87 chemicals, 28 were previously associated with high-quality mammalian toxicity data in Toxicity Reference Database (ToxRefDB). The regenerating planarian responses to 20 of these 28 chemicals (∼71%) and the zebrafish responses to 27 of the 28 (96%) were concordant with mammalian outcomes. By utilizing both model systems, the field might rapidly query more chemical-bioactivity space for guided prioritization of testing in mammals using mechanistic insight gained from the breadth of morphological and behavioral endpoints jointly provided by the 2 systems.
MATERIALS AND METHODS
Chemical library
Supplementary Table 1 lists the chemicals composing the NTP 87-compound library and their purity, consisting of 5 chemical classes (drugs, industrial chemicals, flame retardants, polycyclic aromatic hydrocarbons [PAHs], and pesticides) as well as 5 intended negative control chemicals. More details on the composition of the library can be found in Behl et al. (2018), the introduction of this special issue. Chemicals were provided as ∼20 mM stocks, dissolved in dimethyl sulfoxide (DMSO). Four duplicate chemicals were provided in the 91-compound library tested in the zebrafish study but were not provided in the planarian library.
Planarian studies
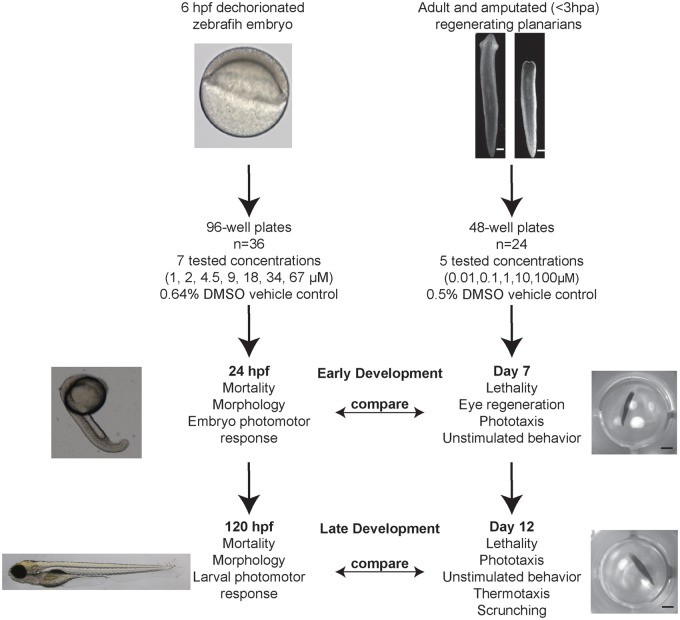
Asexual D. japonica were used for the planarian screen. Details of the experimental procedures and data analysis, as well as the analyzed data for each endpoint can be found in Zhang et al. (2018). In brief, 3 replicates of n = 8 (n = 24 in total) developing/regenerating and adult/intact planarians were each screened for every chemical of the library, generally at 10 nM, 100 nM, 1 μM, 10 μM, and 100 μM. Exposure began on day 1, after the amputation of regenerating planarians, and the planarians were kept in sealed 48-well plates (Genesee Scientific, San Diego, CA) for 12 days, with screening occurring on days 7 and 12 (Figure 1). The DMSO levels were kept constant at 0.5% and n = 8 control populations exposed to the 0.5% DMSO solvent control were included in every plate. We have previously shown this DMSO concentration has no effect on planarian morphology or behaviors (Hagstrom et al., 2015). All raw data associated with the screen are available on the Dryad Digital Repository (doi: 10.5061/dryad.mk6m608).
Figure 1.
Comparison of screening schemes in the zebrafish and planarian systems. Details of the 2 screens including testing conditions and endpoints by time-point. Table 1 summarizes the different morphological endpoints assayed in the zebrafish system. Hpa: hours post-amputation. Scales are as follows: white scale bars: 0.5 mm; black scale bars: 2 mm; Zebrafish 6 hpf embryo is ∼0.7 mm diameter; 24 hpf is 1.9 mm long; 120 hpf is 3.9 mm long (https://zfin.org/zf_info/zfbook/stages/index.html).
Zebrafish studies
Tropical 5 D wildtype adult zebrafish were housed at Oregon State University, Sinnhuber Aquatic Research Laboratory (SARL) in standard 14 h light/10 h dark–light cycle. Embryos were collected, cleaned, and staged prior to dechorionation at 4 hpf. The chorions were enzymatically removed and at 6 hpf, 1 embryo was placed per well in round bottom 96-well plates prefilled will 100 µl of embryo media. The chemicals were digitally dispensed directly from the 20 mM stocks into the test wells using a Hewlett Packard D300e, and all wells normalized to 0.64% DMSO (vol/vol). Up to 1% DMSO has no adverse effects to developing zebrafish (Maes et al., 2012). Each chemical was tested at 0, 1, 2, 4.5, 9, 18, 34, and 67 µM and sealed with parafilm to minimize evaporation and shaken gently overnight at 235 rpm (Truong et al., 2016). Ten chemicals (2,3,7,8-tetrachlorodibenzo-p-dioxin, bis(tributyltin)oxide, benzo[g,h,i]perylene, hexachlorophene, rotenone, tetraethylthiuram disulfide, deltamethrin, methyl mercury (II) chloride, saccharin sodium salt hydrate, and valinomycin) were retested at lower concentrations as the first assessment resulted in 100% mortality or morbidity in all concentrations. A list of the concentrations can be found in Supplementary Table 2. Developmental toxicity was assessed by evaluating mortality, developmental progression, spontaneous movement, and notochord distortion at 24 hpf. Behavioral assessments were conducted at 24 hpf (Embryo photomotor response, EPR) using the Photo-motor Response Assessment Tool (Reif et al., 2016) and at 120 hpf using Viewpoint LifeScience Zebraboxes (Truong et al., 2014; Zhang et al., 2017). At 120 hpf, additional developmental and morphological toxicity endpoints were assessed including: mortality, yolk sac edema, curved or bent body axis in either direction, missing or smaller/larger than normal eye, shortened or malformed snout, malformed jaw, malformed or missing otic vesicle, pericardial edema, malformation or necrosis of the brain, malformed, missing or disorganized somites, malformed or missing pectoral fin, malformed or missing caudal fin, lack of pigment or over pigmentation, lack of circulation, truncated body, failure of swim bladder to inflate, bent notochord and/or tail, and response to touch. After chemical exposure, embryos were not exposed to visible light until administration of the EPR test at 24 hpf (Reif et al., 2016). The test consisted of 30 s of darkness (IR light, Background); first 1 s pulse of intense VIS light, 9 s darkness (Excitation); second pulse of VIS light, 10 s darkness (Refractory) (Truong et al., 2014). Statistical significance was calculated for each interval using a Kolmogorov-Smirnov test (K-S) with a threshold of p < .01. The 120 hpf larval photomotor response (LPR) assay was conducted just prior to morphological evaluation. In the LPR, larvae experience a total of 4 light cycles, each cycle consisting of 3 min of alternating light and dark. Wells with mortality or malformed animals were excluded from the subsequent analysis. An entropy score was calculated for each interval and compared with the control to compute a relative ratio, as described in Zhang et al. (2017). Statistical significance was determined using a K-S test (p < .01) and a relative ratio of >10% or <10%. All analyses were conducted using custom R scripts previously described (R Core Team, 2016). All zebrafish morphological concentration response plots are available in Supplementary Figure 2.
Meta-analysis
Supplementary File 1 contains the lowest effect levels (LELs) identified for each endpoint in both the zebrafish and planarian screens. LELs which did not pass an additional criterion of having at least 2 consecutive concentration hits for an endpoint (statistically significant for zebrafish, and statistically and biologically significant for planarians—see Zhang et al. (2018) for more details) are marked in red. In parenthesis is the new LEL after applying the criteria (ie, excluding concentration-independent hits). Incidences of endpoint abnormality (hit detection) in the 2 systems were compared based on whether the chemical was deemed active or not, irrespective of concentration. Each chemical was scored in MATLAB (Mathworks, Nattick, MA) as either active in planarians, active in zebrafish, active in both, or inactive. We did not consider the concentration which elicited a response due to differences in the concentrations tested and because of the unknown relationships between the nominal aquatic concentrations and the internal concentration within the animals. As such, we have no available information to determine the actual amount of toxicants inside the animals, making direct comparisons of concentration inappropriate. Therefore, the data were converted to binary data: hit or no hit (Supplementary Table 3). The endpoints were organized into classes (Table 1) to compare similar endpoints between the 2 systems. Furthermore, to compare similar developmental stages, endpoints at 24 hpf in the zebrafish studies were compared with endpoints at day 7 in the planarian studies and 120 hpf (day 5) zebrafish endpoints with day 12 planarian endpoints. Comparisons were also made at the system level, considering any affected endpoint.
Table 1.
Classes of Endpoints Used in the 2 Systems
| Endpoint Class | Zebrafish Endpoints | Planarian Endpoints |
|---|---|---|
| Mortality | Mortality (24 hpf, 120 hpf) | Lethality (day 7, day 12) |
| Morphology | Developmental progress (24 hpf) | Eye regeneration (day 7) |
| Spontaneous movement (24 hpf) | ||
| Notochord distortion (24 hpf) | ||
| Curved/bent axis (120 hpf) | ||
| Brain malformation or necrosis (120 hpf) | ||
| Malformed or missing caudal fin (120 hpf) | ||
| No circulation or blood flow (120 hpf) | ||
| Eyes malformed, missing or abnormal sized (120 hpf) | ||
| Heart malformation, pericardial edema (120 hpf) | ||
| Malformed jaw (120 hpf) | ||
| Malformed or missing otic (120 hpf) | ||
| Malformed or missing pectoral fin (120 hpf) | ||
| Lack of pigmentation or over pigmentation (120 hpf) | ||
| Behavior |
|
|
The physicochemical and in vivo animal data analysis was completed using custom R scripts (R Core Team, 2016). The chemical-physical property log Kow and BCF were obtained using EPISuite v4.11 (http://www.epa.gov/opptintr/exposure/pubs/episuite.htm; last accessed July 18, 2018). The CAS was input into EPISuite and the experimental log Kow and BCF values were used when available. The MW and CAS were provided by the NTP.
To compare the results of the zebrafish and planarian screens with in vivo animal data, the publicly available animal data were downloaded from the US Environmental Protection Agency’s ToxRefDB (https://www.epa.gov/chemical-research/toxicity-forecaster-toxcasttm-data; last accessed July 18, 2018). The August 2014 data release of the study treatment file (“toxrefdb_study_tg_effect_endpoint_AUG2014_FOR_PUBLIC_RELEASE”) and the summary file (“toxrefdb_nel_lel_noael_loael_summary_AUG2014_FOR_PUBLIC_RELEASE”) were used. The studies used in this analysis were those that fit the usability criteria of “Acceptable guideline (post-1998),” “Acceptable Guideline (pre-1998),” and “Acceptable Non-guideline.” As the 87 compounds were selected to be potential developmental or developmental neurotoxicants, the study types were filtered to developmental (DEV), multigenerational (MGR), neurotoxicity (NEU), and developmental neurotoxicity (DNT). There was no filter on the species used. One point to consider is that the current version of ToxRefDB only houses chemicals that cause adverse effects in animal studies. The 59 other chemicals found in this 87-compound library could either have been in ToxRefDB but with studies that did not follow guideline protocols (which we deemed not usable for these analyses) or were negatives. For these reasons, the concordance study was benchmarked to the 28 active chemicals. Concordance analysis was conducted using a custom R script, and the R package, circlize. DEV and MGR study types were mapped to zebrafish and planarian mortality and morphology, whereas NEU and DNT were mapped to zebrafish EPR and LPR and planarian early and late behavior.
RESULTS
Screening the NTP 87-Compound Library in Both Systems
Each of the chemicals of the NTP 87-compound library (Behl et al., 2018; Ryan et al., 2016) was classified as either generally developmentally toxic, developmentally neurotoxic, neurotoxic, or unknown due to limited data (https://sandbox.ntp.niehs.nih.gov/neurotox/; last accessed July 18, 2018). The chemicals were structure and use classified as drug, flame retardant, industrial, PAH, pesticide, or inactive (as defined by the NTP). As Supplementary Figure 1 illustrates, the largest class in the library consisted of drugs (19 of 87; 22%). There were 5 chemicals selected as inactive negative controls by the NTP library curators.
Figure 1 shows an overview of the different experimental schemes used for the zebrafish and planarian screens. In both studies, developing animals (either dechorionated 6 hpf zebrafish embryos or amputated planarian tails) were statically exposed to multiple concentrations of each chemical in multi-well plates. For planarian studies, both adult (intact) animals and decapitated animals regenerating a new brain (regenerating tail pieces) were assayed. For ease of comparison to the developing zebrafish, we focused on data associated with the regenerating planarians. The comparison with adult worms can be found in Supplementary Figure 3. Chemical bioactivity was assayed in early development (24 hpf zebrafish and day 7 planarian) and late development (120 hpf zebrafish and day 12 planarian) (Figure 1). Dual system screening yielded a significantly larger coverage of endpoints, with zebrafish contributing most of the morphological endpoints (13 vs. 1 in planarians) and the planarian system contributing more behavioral endpoints, covering different behavioral stimuli (light, temperature, noxious heat) and general locomotion compared with photoresponse behaviors (6 vs. 2 in zebrafish).
Concordance of Active Chemicals Between Zebrafish and Planarians
Considering any assay endpoint, zebrafish were more sensitive indicators of bioactivity, ie, 86 of 87 (99%) unique chemicals were bioactive (Figure 2A). Thirty-two chemicals were hits in morphological endpoints, 49 in the EPR, and 66 in the LPR. Fifteen chemicals were bioactive in all 4 assays. Five chemicals for morphological endpoints, 3 in EPR, and 6 in LPR did not show concentration-dependence (marked in red in Supplementary File 1). Collectively, this did not change our identification of bioactive chemicals, as they were bioactive in the other assays under these new criteria.
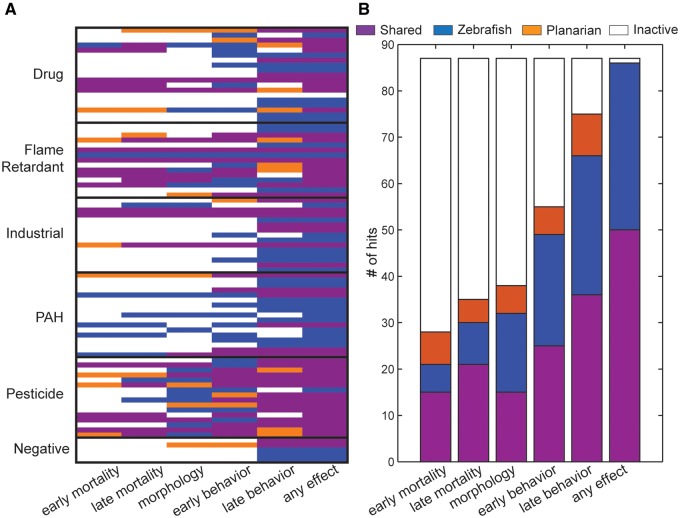
Figure 2.
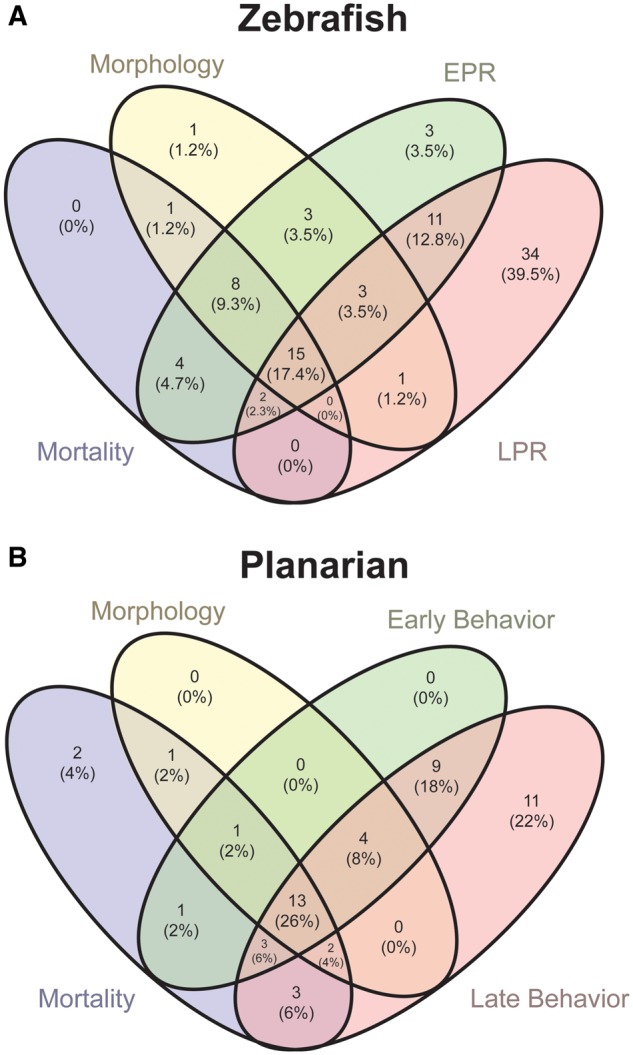
Summary of (A) zebrafish and (B) planarian hits in each endpoint class. Both model systems were exposed to the 87 chemicals and assessed in 4 assays: morphology, mortality, (A) EPR and LPR in zebrafish, and (B) early and late behaviors in planarians.
Additionally, 50 of the 87 chemicals (57%) were bioactive at any endpoint in regenerating (developing) planarians and 48 (55%) in adult worms (see Supplementary Figure 3). In regenerating planarians, 21 chemicals were hits for eye regeneration (morphology), 31 for at least 1 early behavior, and 45 for at least 1 late behavior (Figure 2B). Thus, the majority of bioactivity in the planarian system (47 of 50 chemicals, 90%) was detected by behavioral endpoints and almost one-fourth (12 of 50) with behavior alone. Five chemicals showed concentration-independent effects (ie, active at a lower but not higher concentrations) on unstimulated behavior in regenerating planarians (marked in red in Supplementary File 1). However, for 4 of these, effects were still seen at higher concentrations in other endpoints. For 1 chemical, Chrysene, the only observed effect was concentration-independent hyper-activity in the unstimulated behavior assay. All other hits showed concentration-dependence or only caused effects at the highest concentration tested. Thirteen chemicals were active in all 4 endpoint categories in planarians. All of the developing planarian bioactivity hits were also hits in zebrafish, accounting for 58% of the zebrafish bioactivity hits. The only chemical inactive in both screens was the drug hydroxyurea.
Similar endpoints—mortality, morphology, behavior—were assessed in each model; thus, we compared the chemical hit rate for each endpoint class (Table 1 and Figure 3). Because of the similar developmental timelines, early and late endpoints were compared across models and concordance was based on the number of shared hits out of the total in zebrafish. Similar numbers of chemicals were found to be lethal in the 2 systems, with approximately 70% of these mortality hits being concordant (15/21 and 21/30 for early and late time points, respectively). In addition, 15/32 chemicals were concordant for morphological effects (47%). In both systems, the majority of chemical hits were detected in the behavior endpoints with 25/49 (51%) and 36/66 (55%) chemicals concordant at the early and late time-points, respectively. Similar trends were also found when comparing developing zebrafish to adult planarians, albeit with slightly less concordance, particularly for behavioral endpoints (Supplementary Figure 3).
Figure 3.
Comparison of active hits in the zebrafish and regenerating planarian screens. A, Classification of hits for each chemical (rows), organized by chemical class, whether it was active in both systems, zebrafish only, regenerating planarians only, or inactive in each endpoint class (columns). See Table 1 for a description of the endpoints within classes. B, Number of hits in each endpoint classification used in (A).
Comparison to Physicochemical Properties
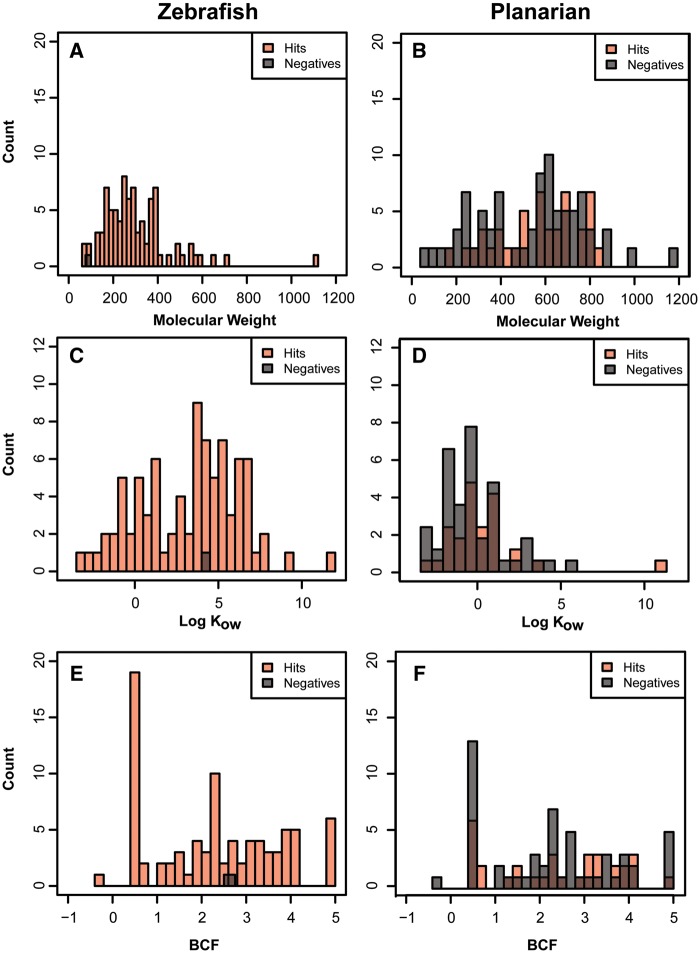
The NTP 87-compound library consists of chemicals with a range of physicochemical properties. We focused on properties of putative high relevance to a waterborne exposure paradigm: MW, log Kow, and BCF. We found that neither MW, log Kow, nor BCF was entirely predictive of a response for both model systems. The single inactive chemical in the zebrafish screen, hydroxyurea, was not due to high MW (Figure 4A), log Kow (Figure 4C), or BCF (Figure 4D). Similarly, high MW did not explain the instances of negative chemicals in the planarian model (Figure 4B) as they were all below 600 g/mol. Log Kow and BCF (Figs. 4D and 4F, respectively) were also not readily associated with instances of chemical inactivity. Thus, the overall association of physicochemical parameters with whole animal chemical bioactivity was weak.
Figure 4.
Physicochemical properties of the NTP 87-compound library. Comparing the (A, B) molecular weight, (C, D) log Kow, and (E, F) BCF of the inactives and the biological actives in zebrafish (left) and planarian (right).
Concordance With Available Animal Data
The US EPA ToxRefDB houses in vivo studies from over 1000 chemicals and thousands of animal toxicity studies in rat, rabbit, mouse, primate, dog, guinea-pig, hamster, and mink. We found that 28 chemicals in ToxRefDB (Supplementary Table 4) were also common to the NTP 87-compound library. Of note, this shared chemical set mainly consisted of pesticides (12 chemicals), drugs (8 chemicals), and industrial chemicals (5 chemicals) as well as 2 of the designated negative controls and 1 PAH.
By way of dataset comparison, we filtered the 28 ToxRefDB chemicals by adverse response category: 16 were identified in ToxRefDB as developmentally toxic (DEV), 18 as developmentally neurotoxic (DNT), 1 as a neurotoxic (NT), and 15 as multi-generationally toxic (MGR), in their respective studies (Supplementary Table 4).
Among the 28 chemicals common to this study and ToxRefDB, the overall, any effect, hit concordance was 27 of 28 (96%) for zebrafish bioactivity (Figure 5A) and 20 of 28 (71%) for regenerating planarian bioactivity (Figure 5B). For the 16 chemicals associated with general developmental toxicity in ToxRefDB, using morphology and mortality endpoints, 11 (69%) were hits in zebrafish, 7 (44%) were hits in the regenerating planarian, and 6 (37.5%) were hits in both. Four developmentally toxic chemicals did not show activity in our dual screen in either morphology or mortality: 2-methyloxyethanol, captan, di(2-ethylhexyl) phthalate, and naphthalene. For the 15 multigenerationally toxic chemicals, 12 (80%) were hits in zebrafish, 6 (40%) were hits in the regenerating planarian, and 6 (40%) were hits in both systems when considering only morphology and mortality. Three of the 15 were inactive: 6-propyl-2-thiouracil, acetaminophen, and di(2-ethylhexyl)phthalate. Eighteen of the 28 shared chemicals had DNT studies, indicating that the 28 chemicals (and the NTP 87-compound library itself) were enriched with developmental neurotoxicants. For the 18 developmentally neurotoxic chemicals, 17 (95%) were also hits in zebrafish behavior (both EPR and LPR), and 10 (56%) were hits in early and/or late planarian behavior, with all 10 of these being hits in both models. Neither model in the present study detected bioactivity for hydroxyurea. Only 1 chemical, carbamic acid, had a neurotoxicity study and was a hit in both the zebrafish and planarian. We note that 5 chemicals of the NTP 87-compound library were previously classified as negatives by the library’s curators, but 2, acetaminophen and acetylsalicylic acid, were hits in ToxRefDB in MGR and DEV studies, respectively.
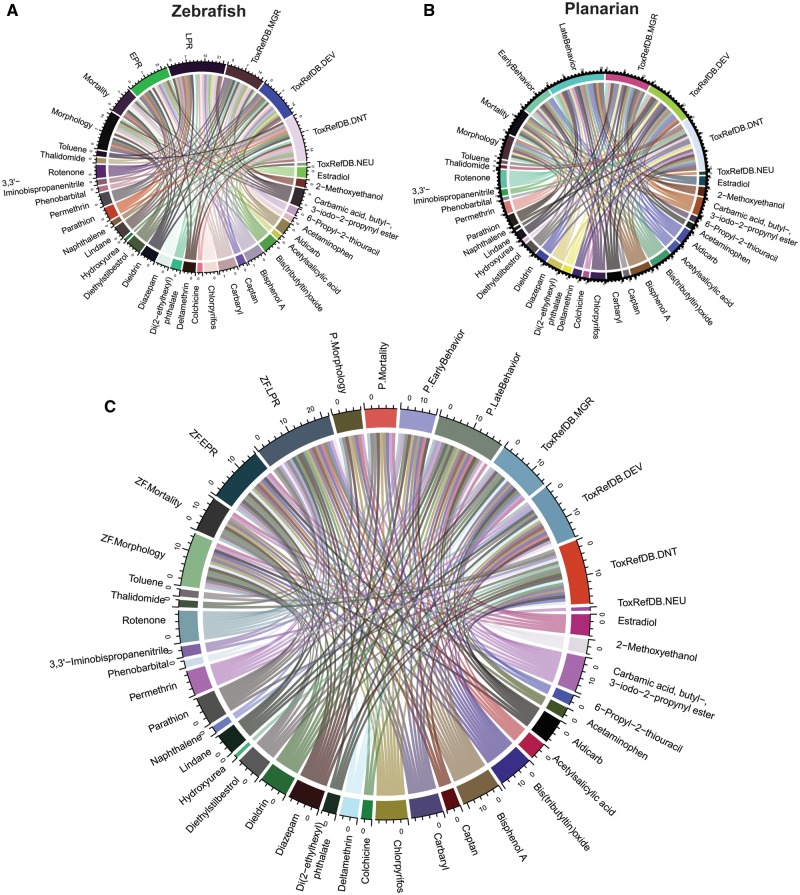
Figure 5.
Inter-relationship between 28 chemicals, zebrafish and planarian assay endpoints, and study types in ToxRefDB. A total of 28 chemicals had in vivo animal studies and were linked to (A) 4 zebrafish endpoints, (B) 4 planarian endpoints, and (C) study types in ToxRefDB (DEV: Developmental; MGR: multigeneration; DNT: developmental neurotoxicity; or NEU: neurotoxicity), zebrafish (morphology, mortality, EPR, or LPR) and planarian assay endpoints (morphology, mortality, early and late behaviors). Each color represents one of these parameters, and the line indicates the relationship between 2 parameters. The width of each parameter is a count of the number of relationships. Thus, a relatively longer width represents that chemical/endpoint has more identified links to the other chemical/endpoints.
For a summary perspective of how the 28 chemicals interacted with the zebrafish and planarian endpoints, we created a chord diagram (Figure 5), which links the chemicals to ToxRefDB study types and zebrafish/planarian endpoints. The width of each endpoint or chemical indicates the number of interactions. In the zebrafish chord plot, LPR and morphology had the most interactions and were on par with the ToxRefDB DNT study type (Figure 5A). For the planarian, this trend is similar with late behavior being a highly linked endpoint (Figure 5B). The chord diagram for both models’ endpoint classes (4 each) and the 4 ToxRefDB toxicity types is shown in Figure 5C. Both the zebrafish LPR and planarian late behavior endpoints had the most interactions (associated bioactivity with the largest width) with the subset of 28 chemicals, supporting the utility and predictivity of the systems’ behavioral endpoints for classifying DNT.
DISCUSSION
Trends by Chemical Class
The differences were observed in the sensitivity of the 2 systems to the various chemical classes in the NTP 87-compound library. Because almost all chemicals were bioactive in the zebrafish screen, concordance was based on whether a zebrafish hit was also a planarian hit. Concordance (from most to least): pesticides (15/16, planarian/zebrafish; 94%), flame retardants (10/15, 67%), drugs (10/18, 56%), industrial chemicals (7/15, 47%), and PAHs (6/17, 35%). The class of PAHs had the lowest concordance between the 2 models, which may be due to the absence of known PAH targets and pathways in planarians. Some PAHs activate the aryl hydrocarbon receptor (AHR) to produce toxicity and cancer (Choi et al., 2010; Garcia et al., 2018; Geier et al., 2018; Knecht et al., 2017b; Qiao et al., 2017). Benzo[a]pyrene, a hit in zebrafish, but not planarians, produces developmental and neurobehavioral deficits dependent on the presence of the AHR2 (Incardona et al., 2011; Knecht et al., 2017a). Thus, lack of conservation of the AHR pathway in planarians may explain the observed insensitivity to PAH exposure.
MW, log Kow, and BCF values are physicochemical properties proposed to be the most predictive for water exposure. In this study, we found that not to be true as no clear trends emerged for actives and inactives in the planarian system (Figure 4). As 86 of the 87 chemicals were hits in the zebrafish model, it was not feasible to assess this trend. However, the one negative, hydroxyurea, did not have any extreme values, supporting the conclusion from the planarian system that the three parameters are weakly predictive of bioactivity.
The differences in chemical sensitivity could be due to a variety of factors: route of exposure/chemical uptake, metabolic activity, etc. It is worth noting that although exposure in both systems is mainly achieved through epidermal diffusion, other routes (eg, planarian pharynx) can also be involved, the extent to which may depend on the life-stage of the animal or the chemical itself. Additionally, planarians are covered in a protective mucus barrier, important for defense against infection and injury (Cochet-Escartin et al., 2015; Pedersen, 1963), which may impede uptake of some chemicals.
The NTP 87-compound library curators classified 5 chemicals as inactive in the toxicological screens performed to date under the range of test conditions used: acetaminophen, acetylsalicylic acid (aspirin), d-glucitol, l-ascorbic acid, and saccharin sodium salt hydrate. Two of these (acetaminophen and acetylsalicylic acid) were identified as hits in animal guideline studies (MGR and DEV, respectively). Additionally, they were also found to be bioactive in the zebrafish LPR and in regenerating planarian late behavior (acetaminophen) and morphology and early/late behaviors (acetylsalicylic acid). However, we note that the regenerating planarian behavioral effects of acetaminophen were very mild, being just outside the noise level (biological cutoffs) of the controls (Zhang et al., 2018). Other studies have also observed this bioactivity (Marques et al., 2004; Prášková et al., 2012; Weigt et al., 2010). Both the zebrafish and planarian detected bioactivity for these 2 misclassified DNTs and did so in less than 12 days. The remaining 3 NTP-inactives had either limited data (d-glucitol), were a developmental toxicant (l-ascorbic acid), or a known carcinogen (saccharin sodium salt hydrate) (https://sandbox.ntp.niehs.nih.gov/neurotox/; last accessed July 18, 2018). Although these 3 chemicals were not DNT compounds, they were bioactive in the zebrafish assays, likely due to the sensitivity of the developing zebrafish as a biosensor and the fact that highly diverse chemical insults during vertebrate development often manifest as common endpoint readouts. Of note, none of the negative controls were active in adult planarians (Supplementary Figure 3). However, it is difficult to classify chemicals as being negative when dosimetry is unknown, in any model system. Therefore, the differences in classification could be due to different databases and criteria.
The Battery of Models Approach to Screening and Its Predictive Power
Whatever the end goal of a chemical screen might be, the principles of the 3Rs (Replacement, Reduction, and Refinement [Dix et al., 2007]) and good scientific practice collectively necessitate that the simplest yet most informative model that minimizes the number of false negatives and false positives is the preferred choice. However, in reality, no one model is likely to be sufficient to capture all necessary biological space in a time- and cost-efficient manner. Thus, battery testing relying on comparative analysis across a range of complementary models (including both in vitro and in vivo systems) may provide the best option for efficient testing, particularly during early hazard identification and prioritization.
In this study, we showed that the zebrafish and planarian models provide a complementary assessment of biological space making them well-suited for battery-approach screening. The optical transparency of developing zebrafish allows for a wide range of morphological assessments to monitor proper developmental milestones and organ formation, exemplified by the high concordance of bioactivity in zebrafish morphological endpoints and ToxRefDB DEV studies. Additionally, their rapid development allows for integration of the central nervous system and assessment for developmental toxicants. On the other hand, the breadth of quantifiable planarian behaviors, some of which are known to be controlled through distinct neuronal subpopulations (Currie and Pearson, 2013; Inoue et al., 2014; Nishimura et al., 2010), provides insight in the mechanisms of (developmental) neurotoxicity. Moreover, planarians are uniquely suited to allow for direct comparisons between adult and developing animals to be able to distinguish developmental effects from general (neuro-)toxicity. In fact, we found that 13 of the 50 chemicals active in regenerating planarians were developmentally selective, ie, toxicity was not found in adult planarians or was found at a higher concentration (Zhang et al., 2018).
Moreover, the zebrafish and planarian models were concordant in their bioactivity readouts across the diverse chemical space captured in this NTP 87-compound library (Figure 3). When comparing actives in the different endpoint classes, both systems contained hits that were not captured by the other. Most chemicals of this library, 86/87 (∼99%), were bioactive in the developing zebrafish, which may be due to the large number (13) of morphological endpoints evaluated. It may appear that the zebrafish model is oversensitive and some of these hits may be false positives. However, the NTP 87-compound library was specifically designed to consist primarily of developmental, developmental neurotoxic, or neurotoxic chemicals, and the zebrafish early life stage system is sensitive to all of these defined endpoints, plus others, such as carcinogenicity. Thus, the high hit rate may be a direct consequence of this particular library. The unique endpoint hits in planarians (particularly late behavior, consisting of 4 different endpoints testing both stimulated [in response to light, thermal gradients, and noxious heat stimuli] and unstimulated behaviors) may provide additional insight into the phenotypic profiling and mechanisms of neurotoxicity for some chemicals.
Even combined, the zebrafish and planarians models are likely not sufficient to capture all realms of possible human health hazards. For example, both models are aquatic organisms relying on chemical exposure in their aquatic environment. This may lead to inconsistencies in toxic outcomes when compared with the breadth of possible routes of exposure in other systems. Moreover, the relationship between the nominal chemical concentrations and the internal concentrations found in the animals is often lacking and could be affected by various factors (solubility issues, absorption by the plastic, absorption into the animal, instability in water over the course of the screen, metabolism, etc.). The understanding of these factors and the pharmacokinetic/pharmacodynamics in these systems will be essential for further validation. This will be particularly important to compare and connect activity in these models with the relevant concentrations/exposure seen in mammals and humans.
The appropriateness and effectiveness of gold standard in vivo mammalian, developmental neurotoxicity studies, which can take 130+ days from exposure to evaluation of the neurobehavioral development of the offspring through adulthood (Dubovický et al., 2008; Virginia Moser et al., 2016) is fiercely debated. The time and expense costs of such guideline studies make them inadequate to evaluate the growing list of chemicals of concern (Tsuji and Crofton, 2012). Moreover, there is uncertainty how to accurately extrapolate data from rats to humans. By adapting high throughput alternative models, we can streamline the toxicology pipeline to efficiently prioritize which chemicals should be tested in guideline studies. These alternative models will likely not completely replace guideline studies, which may still be required for decision making, but can provide rapid guidance of which studies are worth pursuing and which toxicants are of the greatest concern. Libraries, such as the NTP 87-compound library tested here, which are enriched in chemicals with known toxicity, are useful tools for model validation to determine whether effects in alternative models are predictive of mammalian, and ultimately, human toxicity. Twenty-seven of the 28 compounds (96%) in this library which had quality guideline studies associated with them in ToxRefDB were bioactive in either the planarian and/or zebrafish screens, with 20 (71%) bioactive in both, validating the predictivity and relevancy of these models for mammalian toxicity. Using both systems, we are able to provide necessary information to prioritize the chemicals of highest concern, such as in Behl et al. (2015), and help fill the data gaps of under-represented toxicant classes with potential hazards in a relatively quick manner. We thus envision using the zebrafish and planarian models as primary screening tools for vast swathes of chemical space, building big structure-bioactivity datasets from which to prioritize chemicals for further evaluation in the current testing pipeline and potentially predict chemical hazard in the future.
Thus, although a lot of work remains to be done to understand how these and other alternative systems compare to the standard toxicology models, what this and the other studies of the same chemical library in this special issue demonstrate is the added value alternative models are bringing to modern toxicology. A battery approach that harvests the strengths of each of these systems in combination will ultimately transform the toxicology pipeline.
SUPPLEMENTARY DATA
Supplementary data are available at Toxicological Sciences online.
FUNDING
Hellman Foundation, Burroughs Wellcome CASI award, Alfred P. Sloan Foundation (to E.-M.S.C.), and National Institutes of Health [P42 ES016465]. D.H. was partially supported by the National Institutes of Health Cell and Molecular Genetics Training Grant (5T32GM007240-37) and Marye Anne Fox Endowed Fellowship.
Supplementary Material
ACKNOWLEDGMENTS
The authors would like to thank the National Toxicology Program for providing the 87-compound library, Christina Rabeler for help with data compilation, and the staff at Oregon State University Sinnhuber Aquatic Research Laboratory for their assistance in the zebrafish screening.
REFERENCES
- Bailey J., Oliveri A., Levin E. D. (2013). Zebrafish model systems for developmental neurobehavioral toxicology. Birth Defects Res. C. Embryo Today 99, 14–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behl M., Hsieh J. H., Shafer T. J., Mundy W. R., Rice J. R., Boyd W. A., Freedman J. H., Hunter E. S., Jarema K. A., Padilla S. (2015). Use of alternative assays to identify and prioritize organophosphorus flame retardants for potential developmental and neurotoxicity. Neurotoxicol. Teratol.52, 181–193. [DOI] [PubMed] [Google Scholar]
- Behl M., Ryan K., Hsieh J.-H., Parham F., Shapiro A., Collins B. J., Birnbaum L. S., Bucher J. R., Walker N. J., Foster P. M., et al. (2018). Screening for developmental neurotoxicity (DNT) at the National Toxicology Program: The future is now! Toxicol. Sci. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyd W., Smith M., Freedman J. (2012). Caenorhabditis elegans as a model in developmental toxicology In Developmental Toxicology (Harris C., Hansen J. M., Eds.), pp. 15–24. Totowa, NJ: Humana Press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyd W. A., Smith M. V., Co C. A., Pirone J. R., Rice J. R., Shockley K. R., Freedman J. H. (2015). Developmental effects of the ToxCastTM phase I and phase II chemicals in Caenorhabditis elegans and corresponding responses in zebrafish, rats, and rabbits. Environ. Health Perspect. 124, 586–593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cebrià F. (2007). Regenerating the central nervous system: how easy for planarians! Dev. Genes Evol. 217, 733–748. [DOI] [PubMed] [Google Scholar]
- Choi H., Harrison R., Komulainen H., Delgado Saborit J. M. (2010). Polycyclic aromatic hydrocarbons In WHO Guidelines for Indoor Air Quality: Selected Pollutants, pp. 289–345. Bonn, Germany: World Health Organization. [PubMed] [Google Scholar]
- Cochet-Escartin O., Mickolajczyk K. J., Collins E.-M. S. (2015). Scrunching: a novel escape gait in planarians. Phys. Biol. 12, 056010.. [DOI] [PubMed] [Google Scholar]
- Currie K. W., Pearson B. J. (2013). Transcription factors lhx1/5-1 and pitx are required for the maintenance and regeneration of serotonergic neurons in planarians. Development 140, 3577–3588. [DOI] [PubMed] [Google Scholar]
- Dix D. J., Houck K. A., Martin M. T., Richard A. M., Setzer R. W., Kavlock R. J. (2007). The Toxcast program for prioritizing toxicity testing of environmental chemicals. Toxicol. Sci. 95, 5–12. [DOI] [PubMed] [Google Scholar]
- Dubovický M., Kovačovský P., Ujházy E., Navarová J., Brucknerová I., Mach M. (2008). Evaluation of developmental neurotoxicity: some important issues focused on neurobehavioral development. Interdiscip. Toxicol. 1, 206–210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia G. R., Bugel S. M., Truong L., Spagnoli S., Tanguay R. L. (2018). AHR2 required for normal behavioral responses and proper development of the skeletal and reproductive systems in zebrafish. PLoS One 13, e0193484.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geier M. C., Chlebowski A. C., Truong L., Massey Simonich S. L., Anderson K. A., Tanguay R. L. (2018). Comparative developmental toxicity of a comprehensive suite of polycyclic aromatic hydrocarbons. Arch. Toxicol. 92, 571–586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagstrom D., Cochet-Escartin O., Zhang S., Khuu C., Collins E.-M. S. (2015). Freshwater planarians as an alternative animal model for neurotoxicology. Toxicol. Sci. 147, 270–285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagstrom D., Cochet-Escartin O., Collins E.-M. S. (2016). Planarian brain regeneration as a model system for developmental neurotoxicology. Regeneration 3, 65–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He J.-H., Gao J.-M., Huang C.-J., Li C.-Q. (2014). Zebrafish models for assessing developmental and reproductive toxicity. Neurotoxicol. Teratol. 42, 35–42. [DOI] [PubMed] [Google Scholar]
- Hill A. J., Teraoka H., Heideman W., Peterson R. E. (2005). Zebrafish as a model vertebrate for investigating chemical toxicity. Toxicol. Sci. 86, 6–19. [DOI] [PubMed] [Google Scholar]
- Howe K., Clark M. D., Torroja C. F., Torrance J., Berthelot C., Muffato M., Collins J. E., Humphray S., McLaren K., Matthews L., et al. (2013). The zebrafish reference genome sequence and its relationship to the human genome. Nature 496, 498–503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Incardona J. P., Linbo T. L., Scholz N. L. (2011). Cardiac toxicity of 5-ring polycyclic aromatic hydrocarbons is differentially dependent on the aryl hydrocarbon receptor 2 isoform during zebrafish development. Toxicol. Appl. Pharmacol. 257, 242–249. [DOI] [PubMed] [Google Scholar]
- Inoue T., Yamashita T., Agata K. (2014). Thermosensory signaling by TRPM is processed by brain serotonergic neurons to produce planarian thermotaxis. J. Neurosci. 34, 15701–15714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knecht A. L., Truong L., Simonich M. T., Tanguay R. L. (2017). Developmental benzo[a]pyrene (B[a]P) exposure impacts larval behavior and impairs adult learning in zebrafish. Neurotoxicol. Teratol. 59, 27–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knecht A. L., Truong L., Marvel S. W., Reif D. M., Garcia A., Lu C., Simonich M. T., Teeguarden J. G., Tanguay R. L. (2017). Transgenerational inheritance of neurobehavioral and physiological deficits from developmental exposure to benzo[a]pyrene in zebrafish. Toxicol. Appl. Pharmacol. 329, 148–157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linney E., Upchurch L., Donerly S. (2004). Zebrafish as a neurotoxicological model. Neurotoxicol. Teratol. 26, 709–718. [DOI] [PubMed] [Google Scholar]
- Maes J., Verlooy L., Buenafe O. E., de Witte P. A. M., Esguerra C. V., Crawford A. D. (2012). Evaluation of 14 organic solvents and carriers for screening applications in zebrafish embryos and larvae. PLoS One 7, e43850.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malinowski P. T., Cochet-Escartin O., Kaj K. J., Ronan E., Groisman A., Diamond P. H., Collins E.-M. S. (2017). Mechanics dictate where and how freshwater planarians fission. Proc. Natl. Acad. Sci. U. S. A. 114, 10888–10893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marques C. R., Abrantes N., Gonçalves F. (2004). Life-history traits of standard and autochthonous cladocerans: I. Acute and chronic effects of acetylsalicylic acid. Environ. Toxicol. 19, 518–526. [DOI] [PubMed] [Google Scholar]
- Nishimura K., Kitamura Y., Taniguchi T., Agata K. (2010). Analysis of motor function modulated by cholinergic neurons in planarian Dugesia japonica. Neuroscience 168, 18–30. [DOI] [PubMed] [Google Scholar]
- Noyes P. D., Haggard D. E., Gonnerman G. D., Tanguay R. L. (2015). Advanced morphological-behavioral test platform reveals neurodevelopmental defects in embryonic zebrafish exposed to comprehensive suite of halogenated and organophosphate flame retardants. Toxicol. Sci. 145, 177–195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paskin T. R., Jellies J., Bacher J., Beane W. S. (2014). Planarian phototactic assay reveals differential behavioral responses based on wavelength. PLoS One 9, e114708.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedersen K. J. (1963). Sliime-secreting cells of planarians. Ann. NY Acad. Sci. 106, 424–443. [DOI] [PubMed] [Google Scholar]
- Prášková E., Živná D., Štěpánová S., Ševčíková M., Blahová J., Maršálek P., Široká Z., Voslářová E., Svobodová Z. (2012). Acute toxicity of acetylsalicylic acid to juvenile and embryonic stages of Danio rerio. Neuroendocrinol. Lett. 33, 72–76. [PubMed] [Google Scholar]
- Qiao Y., Li Q., Du H.-Y., Wang Q.-W., Huang Y., Liu W. (2017). Airborne polycyclic aromatic hydrocarbons trigger human skin cells aging through aryl hydrocarbon receptor. Biochem. Biophys. Res. Commun. 488, 445–452. [DOI] [PubMed] [Google Scholar]
- R Core Team. (2016). R: A Language Environment for Statistical Computing. Vienna, Austria. [Google Scholar]
- Reif D. M., Truong L., Mandrell D., Marvel S., Zhang G., Tanguay R. L. (2016). High-throughput characterization of chemical-associated embryonic behavioral changes predicts teratogenic outcomes. Arch. Toxicol. 90, 1459–1470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rink J. C. (2013). Stem cell systems and regeneration in planaria. Dev. Genes Evol. 223, 67–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryan K. R., Sirenko O., Parham F., Hsieh J. H., Cromwell E. F., Tice R. R., Behl M. (2016). Neurite outgrowth in human induced pluripotent stem cell-derived neurons as a high-throughput screen for developmental neurotoxicity or neurotoxicity. Neurotoxicology 53, 271–281. [DOI] [PubMed] [Google Scholar]
- Tejeda-Benitez L., Olivero-Verbel J. (2016). Caenorhabditis elegans, a biological model for research in toxicology In Reviews of Environmental Contamination and Toxicology (de Voogt W., Ed.), pp. 1–35. Springer, Cham. [DOI] [PubMed] [Google Scholar]
- Truong L., Reif D. M., St Mary L., Geier M. C., Truong H. D., Tanguay R. L. (2014). Multidimensional in vivo hazard assessment using zebrafish. Toxicol. Sci. 137, 212–233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Truong L., Bugel S. M., Chlebowski A., Usenko C. Y., Simonich M. T., Simonich S. L. M., Tanguay R. L. (2016). Optimizing multi-dimensional high throughput screening using zebrafish. Reprod. Toxicol. 65, 139–147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuji R., Crofton K. M. (2012). Developmental neurotoxicity guideline study: Issues with methodology, evaluation and regulation. Congenit. Anom. (Kyoto) 52, 122–128. [DOI] [PubMed] [Google Scholar]
- Virginia Moser, Raffaele K., Crofton K., Gilbert M., Bowers W., Bailey F. (2016). NAFTA Technical working group on pesticides (TWG): Developmental neurotoxicity study (DNT) guidance document.
- Weigt S., Huebler N., Braunbeck T., von Landenberg F., Broschard T. H. (2010). Zebrafish teratogenicity test with metabolic activation (mDarT): Effects of phase I activation of acetaminophen on zebrafish Danio rerio embryos. Toxicology 275, 36–49. [DOI] [PubMed] [Google Scholar]
- Zhang G., Roell K. R., Truong L., Tanguay R. L., Reif D. M. (2017). A data-driven weighting scheme for multivariate phenotypic endpoints recapitulates zebrafish developmental cascades. Toxicol. Appl. Pharmacol. 314, 109–117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang S., Hagstrom D., Hayes P., Graham A., Collins E.-M. S. (2018). Multi-behavioral endpoint testing of an 87-chemical compound library in freshwater planarians. Toxicol. Sci. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.