Abstract
Fragile X syndrome (FXS) is an X-chromosome-linked disease leading to severe intellectual disabilities. FXS is caused by inactivation of the fragile X mental retardation 1 (FMR1) gene, but how FMR1 inactivation induces FXS remains unclear. Using human neurons generated from control and FXS patient-derived induced pluripotent stem cells (iPSCs) or from embryonic stem cells carrying conditional FMR1 mutations, we show here that loss of FMR1 function specifically abolished homeostatic synaptic plasticity without affecting basal synaptic transmission. We demonstrated that in human neurons, homeostatic plasticity induced by synaptic silencing was mediated by retinoic acid, which regulated both excitatory and inhibitory synaptic strength. FMR1 inactivation impaired homeostatic plasticity by blocking retinoic acid-mediated regulation of synaptic strength. Repairing the genetic mutation in the FMR1 gene in an FXS patient cell line restored FMRP expression and fully rescued synaptic retinoic acid signaling. Thus, our study reveals a robust functional impairment caused by FMR1 mutations that might contribute to neuronal dysfunction in FXS. In addition, our results suggest that FXS patient iPSC-derived neurons might be useful for studying the mechanisms mediating functional abnormalities in FXS.
One sentence summary
Inactivation of the Fmr1 gene that is mutated in Fragile X syndrome leads to loss of retinoic-acid mediated homeostatic plasticity in human neurons.
Introduction
Fragile-X syndrome (FXS) is the most common genetic form of mental retardation and predisposes patients to autism-spectrum disorders (ASDs) (1). Symptoms of FXS include intellectual disability, hyperactivity, attention deficit, and autistic-like behaviors (2). FXS is caused by mutations in the FMR1 gene that encodes for the fragile-X mental retardation protein (FMRP) (3). Expansion of CGG repeats in the FMR1 gene causing hypermethylation-induced FMRP silencing is the most frequent cause of FXS (4, 5). Rare cases of single-nucleotide mutations in FMR1 also cause FXS (6). FMRP, which is highly expressed in the brain (7–9), is a RNA-binding protein that binds to a specific set of mRNAs and regulates their translation (10–12). Studies using a mouse model of FXS induced by constitutive deletion of the Fmr1 gene have contributed much to our understanding of the function of FMRP and of the pathophysiology of FXS. Consistent with FMRP’s role in regulating protein translation, Fmr1 KO neurons exhibit dysregulated protein translation and elevated basal protein synthesis (13, 14), which affects neuronal development and function. Many genes whose mRNAs are targets of FMRP encode for proteins involved in synaptic functions (12), pointing to a potential contribution of synaptic dysfunction to FXS pathophysiology. In agreement with this hypothesis, Fmr1 KO mice show altered excitatory and inhibitory synaptic transmission at specific subsets of synapses (15–17), phasic and/or tonic GABAergic inhibition (18, 19), impairments in certain forms of long-term potentiation (LTP) (20, 21), enhanced metabotropic glutamate receptor (mGluR)-mediated long-term depression (LTD) (22), and a general dysfunction in G protein-coupled receptor (GPCR) responses (23–26) (for comprehensive reviews, see (27, 28)).
In addition to altered Hebbian plasticity, recent reports indicate that defects in synaptic retinoic acid (RA) signaling and homeostatic synaptic plasticity may contribute to network instability and cognitive impairment in Fmr1 KO mice (29, 30). Homeostatic synaptic plasticity is a unique form of neuronal plasticity that operates to optimize network activity (31). For example, prolonged network silencing triggers a compensatory increases in excitatory synaptic strength and decreases in inhibitory synaptic strength (32). A key molecular player in homeostatic synaptic plasticity is RA. Different from its developmental morphogen function, which regulates gene transcription (33), RA modulates synaptic transmission in mature neurons through a transcription-independent action (34–36). RA synthesis is triggered by reduced excitatory synaptic transmission and decreased dendritic calcium concentrations (37, 38). Acting through a protein synthesis-dependent pathway, newly synthesized RA promotes insertion of α-amino-3-hydroxy-5-methyl-4isoxazolepropionic acid (AMPA) receptors into excitatory synapses and removal of gamma-aminobutyric acid A (GABAA) receptors from inhibitory synapses, thus quickly altering the synaptic E/I balance and changing network excitability (29, 34, 39). In the Fmr1 KO mouse, RA signaling is severely impaired despite its normal synthesis (29, 30, 34, 39), opening the possibility that impaired homeostatic plasticity may contribute to network instability and cognitive impairment in FXS.
One of the missing links between mouse FXS studies and effective therapeutic intervention in human patients is whether phenotypes discovered from mouse studies can be validated in human neurons. Recent exciting advancements in stem cell biology provide the basis for an alternative ex-vivo approach of disease models. The generation of human pluripotent stem cells (hPS cells), including human embryonic stem cells (ES cells) and human induced pluripotent stem cells (iPS cells), and their in vitro differentiation into neurons, allows direct comparison of neuronal function between human neurons derived from control subjects and from patients (40–45). Using this approach, a number of studies have already attempted to examine FXS pathogenesis in ES cells isolated from embryos carrying FXS mutations (46–49), and from iPS cells generated from somatic cells of FXS individuals (50–56). Studies on FXS iPS cell-derived neurons differentiated using variations of the dual SMAD inhibition protocol (53–55, 57–60) reported defects in neuronal differentiation, maturation, neurite outgrowth and presynaptic vesicle recycling, recapitulating some the phenotypes described in the FXS mouse models. However, the synaptic plasticity phenotype has not been fully elucidated in FXS human neurons. Additionally, albeit powerful and informative, the approach of phenotypic characterization based on comparisons of neurons derived from different individuals (control and affected patients) carries an inherent limitation - the variability between different cell lines and between individual patients provides a challenge that cannot easily be addressed and introduces uncertainty in assessing the applicability of results to understanding a disease. Several approaches have been suggested to overcome these limitations. One common approach is to use several clones from each individual and cell clones from multiple different individuals in order to identify consistent phenotypes (61). Another approach is to generate isogenic lines by repairing a mutation using constitutive genome editing (62). The latter approach, although genetically better defined, still runs the risk of clonal variation due to newly generated mutations during clone selection (63).
In the present study, we first established that in ES cell-derived human neurons from control subjects co-cultured with mouse neurons, RA-dependent homeostatic synaptic plasticity induced by prolonged synaptic silencing is robustly expressed at both excitatory and inhibitory synapses. We then generated two conditional KO ES cell lines that allowed us to create FMR1-null mutations in a conditional manner without clonal variation. We showed that loss of FMRP expression impaired RA signaling and homeostatic synaptic plasticity in human neurons, while leaving most other neuronal and synaptic properties intact. Using multiple human iPS cell lines from control subjects and FXS patients, we validated that RA signaling and homeostatic synaptic plasticity is indeed absent in neurons derived from FXS patients but not in neurons derived from control individuals. Moreover, Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)/CRISPR-associated protein 9 (Cas9)-mediated genetic repairing of the mutant FMR1 gene in one of the patient iPS cell line fully rescued synaptic RA signaling. Thus, we established that defective synaptic RA signaling and homeostatic synaptic plasticity constitutes a major synaptic dysfunction in the FXS.
Results
RA-dependent homeostatic plasticity in Human neurons
In mouse neurons, retinoic acid (RA) is a key molecule involved in homeostatic synaptic plasticity (34). In murine FXS model, RA-dependent homeostatic plasticity process is impaired; therefore, we asked whether loss of homeostatic plasticity may also occur in human iPS cell-derived neurons from FXS patients.
To this end, we first assessed whether cultured human neurons were responsive to RA. We converted human ES cells into a homogeneous population of excitatory induced neuronal (iN) cells using forced expression of Neurogenin 2 (Ngn2) (42), and co-cultured these neurons with mouse glia to promote synapse formation (Fig. 1A). Application of RA to control human neurons co-cultured with mouse glia produced a great increase in excitatory synaptic strength, monitored via the amplitude of miniature excitatory postsynaptic currents (mEPSCs) (Fig. 1B and fig. S1A). The increase in mEPSC amplitude occurred across all amplitude ranges (5–50 pA), reflected in a relatively uniform right shift of the amplitude histogram (fig. S1A). The average mEPSC frequency and interevent interval were not significantly changed (fig. S1B). These findings suggest that Ngn2-induced human neurons co-cultured with glia have the ability to change synaptic strength in response to RA.
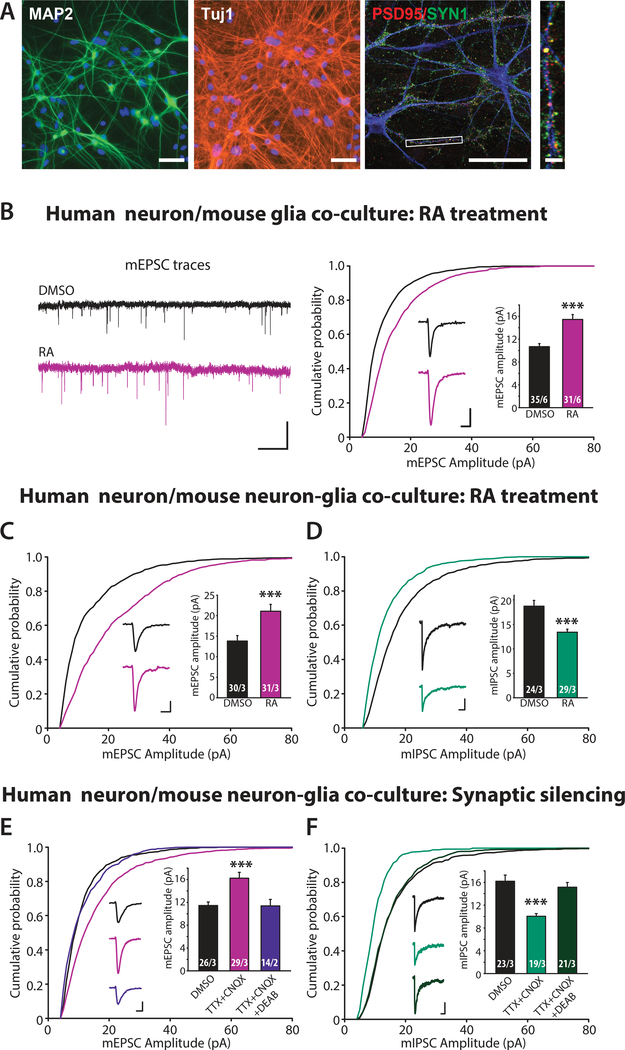
Figure 1. RA-dependent homeostatic synaptic plasticity in human neurons (iN cells) differentiated from human H1 ES cell line.
(A) Representative images showing expression of pan neuronal markers (MAP2 and Tuj1) as well as pre (PSD95) and post synaptic (Synapsin1, SYN1) markers in iN cells generated from control human ES (H1) cells and co-cultured with mouse glia for four weeks. Scale bars: 50 μm (MAP2 and Tuj) and 5 μm (PSD95/SYN1). (B) Example traces (left) and quantification (right) for mEPSC amplitudes recorded from iN cells co-cultured with mouse glia and treated with DMSO or RA (30–45 minutes plus 90 minutes washout) (***, p < 0.001; student’s t-test). Scale bars: left, 20 pA, 1s; right, 5 pA, 5 ms. (C) Quantification for mEPSC amplitudes recorded from iN cells co-cultured with mouse cortical neurons and treated with DMSO or RA (***, p < 0.001; student’s t-test). Scale bars: 5 pA, 5 ms. (D) Quantification for mIPSC amplitudes recorded from iN cells cocultured with mouse cortical neurons (***, p < 0.001; student’s t-test). Scale bars: 5 pA, 5 ms. (E-F) Quantification for mEPSC amplitudes (E) and mIPSCs (F) recorded from iN cells co-cultured with mouse cortical neurons treated with DMSO (24 h), TTX+CNQX (24 h), or TTX+CNQX+DEAB (24 h) (***, p < 0.001; one-way ANOVA). Scale bars: 5 pA, 50 ms. n/N = # of neurons/# of independent experiments. In all graphs, data represent average values ± SEM.
Ngn2-induced human neurons are purely glutamatergic (42). In order to assess inhibitory synapse responsiveness to RA and their function in homeostatic synaptic plasticity, we introduced inhibitory interneurons by co-culturing the human neurons with a mixed population of neurons and glia dissociated from P0 WT mouse brain. Under these conditions, RA induced a strong enhancement of excitatory synaptic strength and a concomitant reduction in inhibitory synaptic strength of the human neurons (identified via co-expressed EGFP), indicated by the altered mEPSC and mIPSC amplitudes but not their frequencies (Fig. 1C, and 1D, fig. S1C-S1F). Mirroring the changes in mEPSC amplitudes (fig. S1C), the decrease in mIPSC amplitudes was represented by a relatively uniform left shift in the amplitude histogram (fig. S1E).
Having established robust RA-responsiveness at both excitatory and inhibitory synapses, we next asked whether synaptic silencing-induced homeostatic synaptic plasticity also occurs in these human neurons. Prolonged 24-hr treatment of the voltage-gated sodium channel blocker tetrodotoxin (TTX) and the AMPA receptor antagonist 6-cyano-7nitroquinoxaline-2,3-dione (CNQX) (TTX+CNQX) robustly changed excitatory and inhibitory strength in opposite directions (Fig. 1E, and 1F, fig. S1G-S1J), mimicking the effects of a direct RA treatment. The increase in excitatory synaptic strength and the decrease in inhibitory synaptic strength that were induced by synaptic silencing were both blocked by 4-diethylamino-benzaldehyde (DEAB), an inhibitor of the RA synthesizing enzyme retinal dehydrogenase (RALDH) (34, 38) (Fig. 1E and 1F).
Our results thus show that Ngn2-induced human neurons co-cultured on mouse glia/neurons showed RA-dependent homeostatic plasticity at both excitatory and inhibitory synapses. Next, we investigated potential impairments in homeostatic plasticity in human fragile-X neurons under this experimental framework.
Generation of human ES cell lines carrying the FMR1 conditional KO (cKO) allele
Aiming to explore whether FMR1 inactivation alters homeostatic plasticity in human neurons, we designed conditional knockout (cKO) experiments that allow complete control of the genetic background (64). Using homologous recombination, we derived male human H1 ES cells in which exon 5 of the FMR1 gene is flanked with loxP sites; in addition, we introduced a neomycin resistance cassette (NEO) surrounded by flp-recombination sites as a selectable marker for homologous recombination events (Fig. 2A). Two independent ES cell clones (F17 and F33) were analyzed in parallel to control for possible additional mutations introduced by clonal selection (Fig. 2B, fig. S2A and S2B). The presence of the neomycin cassette effectively blocked the expression of FMRP, as illustrated for the F33 clone in Fig. 2C. Removal of the neomycin cassette with Flp recombinase (for example, in sub-clones F33.F3 and F17.F2) converted the KO allele to a cKO allele, and reactivated normal FMRP expression (FMR1 cKO; Fig. 2C). Deletion of exon 5 by Cre-recombinase created an out-of-frame junction between exon 4 and exon 6, thus producing a null allele and converting the FMR1 cKO ES cells into FMR1 KO cells (Fig. 2D). In the following, all experiments were performed with FMR1 cKO ES cell clones from which the neomycin resistance cassette had been removed using Flp recombinase, with parallel analysis of two independently derived clones (F33.F3 and F17.F2). Inactivation of FMR1 by Cre-recombinase occurred only after neuronal trans-differentiation had been initiated.
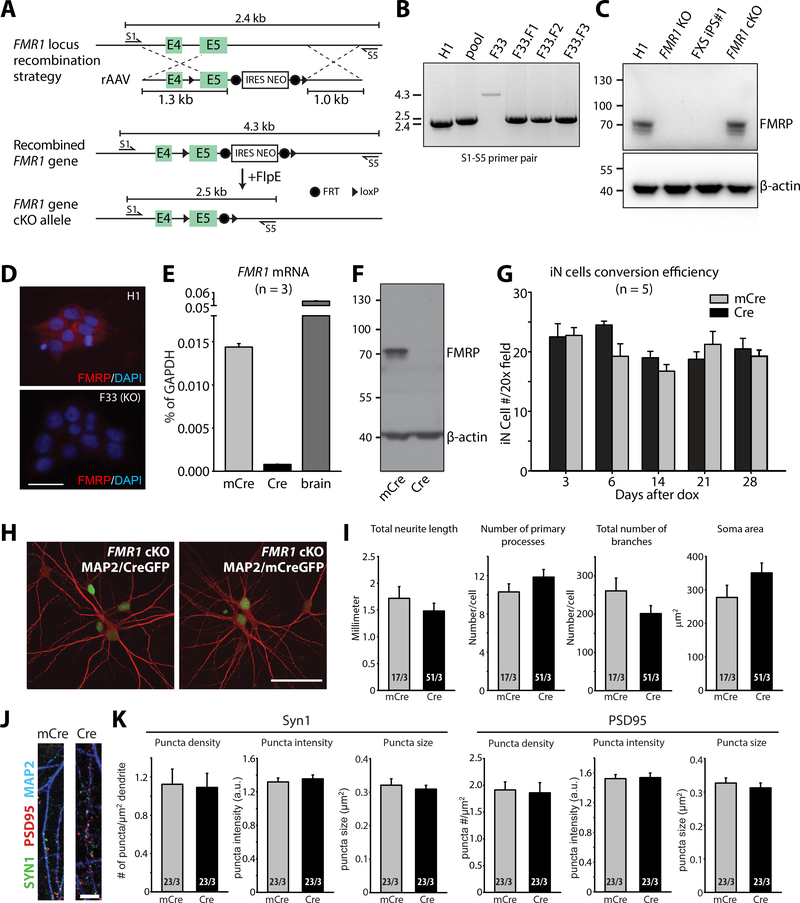
Figure 2. Generation and characterization of FMR1 conditional knock-out human neurons.
(A) Schematics of the gene targeting strategy used to generate FMR1 cKO hES cells. (B) Homologous recombination validation by PCR using S1 and S5 external primers. F33 is the original targeted clone, F33.F1, F33.F2 and F33.F3 are the subclones generated after Flp-mediated loop out of the selection cassette. (C) Immunoblot analysis of FMRP protein in H1, FMR1 KO (F33), FXS iPS cell #1 (SC135), and FMR1 cKO (F33.F3) cells. (D) Immunostaining of FMRP in H1 and FMR1 KO (F33)cells. Scale bar: 50 μm. (E) qPCR analysis of FMR1 mRNA expression in iN cells differentiated from F33.F3 cKO ES cells transduced with mCre (WT, control) or Cre (KO), and in brain tissues. (F) Immunoblot of FMRP protein in iN cells differentiated from F33.F3 cKO ES cells transduced with mCre or Cre. (G) Quantification of the iN conversion efficiency of F33.F3 cKO ES cells after doxycycline induction. (H) Representative immunofluorescence images of F33.F3 cKO ES cells transduced with mCre-GFP (WT, control; right) or Cre-GFP (KO; left) (red: MAP2). Scale bars: 50 μm. (I) Quantification of general neuronal morphology in WT (mCre) and FMR1 KO (Cre) iN cells. (J) Images of dendrites immunolabeled with Synapsin-1, PSD95 and MAP2 from WT (mCre) and FMR1 KO (Cre) iN cells. Scale bar: 5 μm. (K) Quantification of puncta density, intensity and size for SYN1 and PSD95 immunofluorescent signals. n/N = # of neurons/# of independent experiments. In all graphs, data represent average values ± SEM.
A large number of diverse functional impairments, including major changes in neuronal development, were suggested for FMR1-mutant human neurons (53, 55, 57, 59). Taking advantage of the precise control of genetic background afforded by FMR1 cKO neurons, we examined whether the loss of FMR1 function would impair neuronal development or dendritic arborization. We converted FMR1 cKO ES cells into neurons with Ngn2, and co-expressed either mutant inactive Cre-recombinase (mCre, as a control) or wild-type active Cre-recombinase. Cre recombinase eliminated FMRP mRNA and protein expression in human FMR1 cKO neurons as expected (Fig. 2E, 2F, and fig. S2C). FMR1 cKO cells transduced with Cre and mCre had similar conversion efficiencies, suggesting that FMRP did not affect Ngn2-mediated neuronal reprogramming (Fig. 2G).
Morphological analyses of FMR1 mutant and control neurons co-cultured with mouse glia revealed no differences in total neurite growth, number of processes, number of neurite branches, and neural soma size (Fig. 2H, 2I), indicating that the lack of FMRP in mature human neurons in vitro had no effect on overall neuronal morphology. We next examined synapse formation by immunostaining these neurons for the pre- and post-synaptic markers synapsin-1 and postsynaptic density protein 95 (PSD95), respectively (Fig. 2J, 2K, and fig. S2D). Dense excitatory synapses were formed on dendrites of both mCre (control) and Cre (FMR1 KO) neurons (Fig. 2J). Quantifications of synapsin-1 and PSD95 puncta density, intensity and size uncovered no difference between control and FMR1 KO neurons (Fig. 2K), suggesting normal synapse development in vitro in the absence of FMR1 expression.
As the next step, we examined the basic membrane properties of control and FMR1-KO neurons using whole-cell patch-clamp recordings. Membrane capacitance and resistance was not altered by the lack of FMR1 expression (fig. S2E), which is consistent with their unaltered soma size. Active membrane properties and excitability were analyzed under current-clamp mode with steps of current injection. All groups of neurons readily fired action potentials in response to current injections (fig. S2F). Resting membrane potentials and action potential amplitudes were comparable between control and FMR1 KO neurons (fig. S2G and S2H). FMR1 KO neurons differentiated from the F33 FMR1 cKO ES cell line but not from the F17 FMR1 cKO ES cell line exhibited increased excitability (fig. S2F), lower action potential threshold (fig. S2I) and narrower action potential width (fig. 2J) compared to the respective controls. The same FMR1 KO neurons also displayed larger voltage-activated Na+ currents with normal voltage-activated potassium (K+) currents (fig. S2K and S2L).
These data show that deletion of FMRP expression from developing human neurons did not impair neuronal differentiation induced by Ngn2 expression, dendritic arborization, synapse formation, or development of normal electric properties, but may affect neuronal excitability.
Impaired RA signaling and homeostatic synaptic plasticity in human neurons with FMR1 inactivation
We next investigated synaptic properties and plasticity in the conditional FMR1 KO neurons by measuring mEPSCs to monitor synaptic strength and quantal size. In cocultures of human neuron differentiated from either F17 or F33 FMR1 cKO cells with mouse glia, Cre-induced FMR1 deletion in FMR1 cKO neurons had no effect on baseline mEPSC amplitudes and frequencies (Fig. 3A, fig. S3A, S4A and S4B). RA treatment enhanced mEPSC amplitudes in control neurons, but not in FMR1 KO neurons differentiated from both FMR1 cKO cell lines (Fig. 3A and S4A). Thus, similar to what was observed in mouse neurons (30), the FMR1 mutation blocked the effect of RA at excitatory synapses.
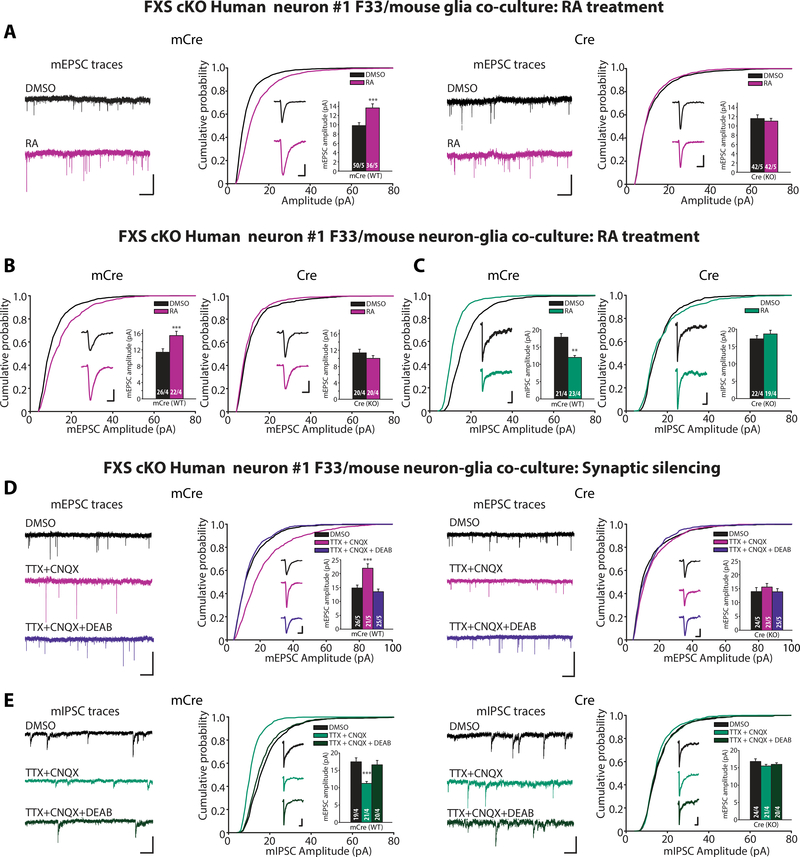
Figure 3. Impaired RA synaptic signaling and homeostatic synaptic plasticity in human neurons lacking FMR1 expression.
(A) Example traces and quantification for mEPSC amplitudes recorded from iN cells differentiated from FXS cKO #1 F33 ES cells, infected with mCre (WT) or Cre (KO) viruses, co-cultured with mouse glia and treated with DMSO or RA (**, p < 0.01; student’s t-test). Scale bars: top traces, 10 pA, 1s; inset traces, 5 pA, 5 ms. (B) Quantification for mEPSC amplitudes recorded from WT or KO iN cells differentiated from FXS cKO #1 F33 ES cells, co-cultured with mouse cortical neurons, and treated with DMSO or RA (**, p < 0.01; student’s t-test). Scale bars: 5 pA, 5 ms. (C) Quantification for mIPSC amplitudes recorded from WT or KO iN cells treated with DMSO or RA (**, p < 0.01; student’s t-test). Scale bars: 5 pA, 50 ms. (D) Example traces and quantification for mEPSC amplitudes recorded from WT or KO iN cells differentiated from FXS cKO #1 F33 ES cells treated with DMSO (24 h), TTX+CNQX (24 h), or TTX+CNQX+DEAB (24 h) (***, p < 0.001; oneway ANOVA). Scale bars: long traces, 20 pA and 1s; single response traces, 5 pA and 5 ms. (E) Example traces and quantification for mIPSC amplitudes recorded from WT or KO iN cells treated with DMSO (24 h), TTX+CNQX (24 h), or TTX+CNQX+DEAB (24 h) (***, p < 0.001; one-way ANOVA). Scale bars: left, 20 pA and 1s; right, 5 pA and 50 ms. n/N = # of neurons/# of independent experiments. In all graphs, data represent average values ± SEM.
We next examined synaptic RA signaling in co-cultures of human neurons with mixtures of mouse neurons and glia (as opposed to glia alone), in which both excitatory and inhibitory synapses are present. Again, blocking FMR1 expression in the human neurons did not significantly alter their baseline mEPSC amplitude and frequency, nor did it affect their baseline miniature inhibitory postsynaptic current (mIPSC) amplitude and frequency (Fig. 3B, 3C, and fig. S3B, S3C, S4C-F). However, whereas RA robustly increased mEPSC amplitudes and decreased mIPSC amplitudes in control neurons, RA had no effect on either mEPSC or mIPSC amplitudes in FMR1 KO neurons (Fig. 3B, 3C, and fig. S4C and S4E). Thus, blocking FMR1 expression blocked RA signaling at both excitatory and inhibitory synapses.
To inquire whether the block of synaptic RA signaling by the FMR1 deletion inhibited RA-dependent homeostatic plasticity, we directly assayed homeostatic plasticity induced by synaptic silencing. In control neurons differentiated from either F17 or F33 FMR1 cKO cells, synaptic silencing (induced by a 24-hour TTX+CNQX treatment) increased mEPSC amplitudes and decreased mIPSC amplitudes without affecting the mEPSC or mIPSC frequencies. Both homeostatic changes were abolished by the RA-synthesis inhibitor DEAB, indicating that they were mediated by RA (Fig. 3D, 3E, and fig. S3D, S3E, S4GJ). Ablation of FMR1 expression by Cre-recombinase blocked the homeostatic changes induced by synaptic silencing (Fig. 3D, 3E, and fig. S3D, S3E, S4G-J).
Therefore, we established in human neurons differentiated from two independent FMR1 cKO ES cell clones that FMR1 expression is critical for synaptic RA signaling and RA-dependent homeostatic plasticity.
Generation of human neurons from FXS patients
The profound but selective changes in human neurons resulting from conditional inactivation of the FMR1 gene raises the question whether these changes are representative of pathogenic events in human FXS. To address this question, we generated human neurons using forced expression of Ngn2 from iPS cells derived from two FXS patients and two unrelated control individuals.
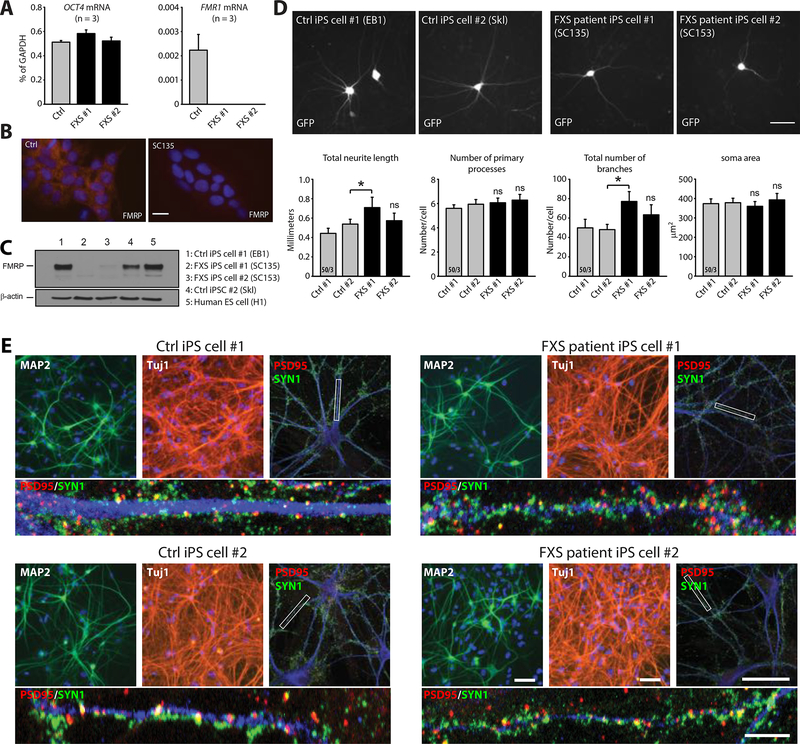
We analyzed neurons generated from two FXS patient-derived iPS cell lines (SC135 and SC153 (51), referred to as FXS iPS cells #1 and #2, respectively), and two control iPS cell lines. FXS iPS cell lines #1 and #2 have expanded CGG repeats in the full mutation range. FMR1 mRNA expression was inactivated in both FXS iPS cell lines, and FMR1-encoded FMRP protein was undetectable in neurons differentiated from both lines as expected (Fig. 4A-4C). Consistent with our FMR1 cKO analyses, iPS cell-derived neurons co-cultured with mouse glia exhibited a typical neuronal morphology without major changes in neuronal differentiation, but with extensive synapse formation (Fig. 4D and 4E). The only morphological change observed was a modest increase in neurite growth and number of dendritic branches in neurons differentiated from iPS cell line #1, which however was absent from neurons differentiated from iPS cell line #2 and from neurons with conditional FMR1 deletions (Fig 4D).
Figure 4. Biochemical and morphological characterization of human neurons differentiated from control and FXS patient iPS cell lines.
(A) Quantification of OCT4 and FMR1 mRNA expression in H1 ES cell and FXS iPS cells. (B) Immunostaining images of FMRP showing lack of FMRP expression in control and FXS iPS cell lines. Scale bar: 20 μm. (C) Western blot analysis of FMRP expression in iN cells differentiated from H1 ES cell, FXS iPS cells #1 and #2 and control iPS cells #1 and #2. (D) Representative images and quantification of morphological aspects of iN cells differentiated from both control and FXS iPS cell lines (Ctrl #1 EB1, Ctrl #2 Skl, FXS #1 SC135, FXS #2 SC153) (*, p < 0.05; one-way ANOVA). Scale bar: 50 μm. n/N = # of neurons/# of independent experiments. (E) Representative images showing expression of pan neuronal markers (MAP2 and Tuj1) as well as pre (Synapsin1, SYN1) and post synaptic (PSD95) markers in iN cells generated from control human iPS cells (Ctrl #1 EB1, Ctrl #2 Skl) and FXS human iPS cells (FXS #1 SC135, FXS #2 SC153) and cocultured with mouse glia for four weeks. Scale bars: 50 μm for the top images and 5 μm for the bottom higher magnification images. In all graphs, data represent average values ± SEM.
Electrophysiological recordings showed that the FXS-derived and the control neurons exhibited a similar membrane capacitance and resistance, suggesting similar neuronal size between the two groups (fig. S5A). We then analyzed active membrane properties and neuronal excitability of the iN cells in current-clamp mode. Neurons differentiated from all four iPS cell lines (from both control and FXS patients) exhibited similar resting membrane potentials, and generated action potentials in response to current injections with similar firing thresholds and amplitudes (fig. S5B and S5C). However, similar to what we observed in one conditional KO line, neurons generated from both FXS iPS cell lines produced more action potentials in response to increasing current injections, suggesting increased excitability (fig. S5B). Moreover, the action potential width (measured at 50% of the action potential amplitude) was smaller in FXS neurons than in control neurons (fig. S5C). Accordingly, Na+-currents were significantly increased in the FXS neurons, whereas K+-currents were unchanged (fig. S5D and S5E).
Blocked RA-induced synaptic potentiation in neurons derived from FXS patients
We next examined RA-induced changes in synaptic transmission. Human neurons differentiated from all iPS cell lines and co-cultured with mouse glia exhibited similar mEPSC amplitudes and frequencies (Fig. 5A, 5B, and fig. S6A-D). Control neurons responded robustly to incubation with 2 μM RA with an increase in mEPSC amplitude (Fig. 5A and fig. S6B). On the contrary, neurons from FXS iPS cell lines exhibited no changes in mESPC amplitudes following RA incubation (Fig. 5B and fig. S6D). Similarly, human neurons from all iPS cell lines co-cultured with mouse neurons and glia exhibited comparable mEPSC and mIPSC properties (Fig. 5C, 5D, and fig. S6E-L). Again, RA induced a robust increase in mEPSC amplitudes and also a strong decrease in mIPSC amplitudes in the mixed neuron-glia co-cultures (Fig. 5C, 5D, and fig. S6F and S6J) whereas mEPSCs and mIPSCs were not modulated by RA in FXS neurons cocultured with mouse neurons and glia (Fig. 5C, 5D, and fig. S6H, and S6L).
Figure 5. Impaired synaptic RA signaling in human neurons derived from FXS patients co-cultured with mouse glia or mouse neurons.
(A-B) Example traces and quantification for mEPSC amplitudes recorded from iN cells differentiated from control #1 iPS cells (EB1) (A) and FXS iPS cell #1 (SC135) (B), cocultured with mouse glia, and treated with DMSO or RA (***, p < 0.001; student’s t-test). Scale bars: left, 20 pA, 1s; right, 5 pA, 5 ms. (C) Quantification for mEPSC amplitudes recorded from iN cells differentiated from control iPS cell #1 (left) or FXS iPS cell #1 (right), co-cultured with mouse neurons, and treated with DMSO or RA (***, p < 0.001; student’s t-test). Scale bars: 5 pA and 5 ms. (D) Quantification for mIPSC amplitudes recorded from control and FXS iN cells treated with DMSO or RA (***, p < 0.001; student’s t-test). Scale bars: 5 pA and 50 ms. (E) Representative traces and quantification of evoked EPSCs recorded from iN cells differentiated from control iPS cell #1 and FXS iPS cell #1 lines after DMSO or RA treatment. Scale bars: 200 pA, 10 ms. (F) Representative traces and quantification of evoked IPSCs recorded from control and FXS iN cells after DMSO or RA treatment. Scale bars: 200 pA, 20 ms. n/N = # of neurons/# of independent experiments. In all graphs, data represent average values ± SEM.
A potential concern about these experiments is that measuring only spontaneous miniature synaptic responses might not be a precise reflection of synaptic strength (65). To address this concern, we monitored evoked synaptic responses using extracellular stimulations and human neurons co-cultured with mixed mouse neurons and glia. Similar to the results from miniature synaptic response recordings, RA potentiated the evoked EPSC amplitude in control neurons (Fig. 5E and fig. S6M), but had no effect on evoked EPSCs in FXS neurons (Fig. 5E, and fig. S6N). Furthermore, RA strongly suppressed evoked IPSC amplitudes in control neurons (Fig. 5F and fig. S6O), but had no effect on evoked IPSCs in FXS neurons carrying FMR1 mutations (Fig. 5F and fig. S6P). Taken together, these results showed that synaptic RA signaling was blocked in human FXS neurons. Moreover, because the co-cultured mouse glia and mouse neurons are wildtype with normal Fmr1 expression, these results suggest that the defect in RA signaling reflected a cell-autonomous function of FMRP in synaptic RA signaling.
Properties of RA-mediated up-regulation of EPSCs in neurons derived from control and FXS patients
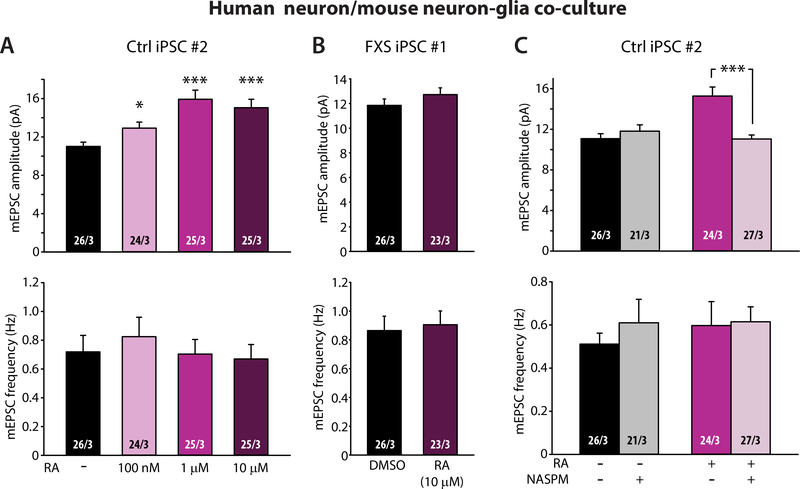
To investigate the possibility that impaired RA responsiveness in FXS neurons was due to reduced sensitivity to RA, we applied different concentrations of RA and found that 12 μM was a saturating dose for the RA-induced mEPSC increase in control neurons (Fig. 6A). In FXS neurons, however, even 10 μM RA yielded no response (Fig. 6B), indicating that lack of RA signaling in FXS neurons was likely not due to reduced RA sensitivity, but corresponded to a lack of signaling due to the loss of FMRP.
Figure 6. Properties of RA signaling in human neurons differentiated from control patient iPS cells.
(A) RA dose-dependent responses of mEPSC in iN cells differentiated from control iPS cell #2 line (*, p < 0.05; ***, p < 0.001; one-way ANOVA). (B) Quantification of mEPSCs recorded from iN cells differentiated from FXS iPS cell #1 line and treated with maximum dose of RA (10 μM). (C) Quantification of mEPSC amplitudes and frequencies in iN cells differentiated from control iPS cell #2 line treated with RA and NASPM (***, p < 0.001; student’s t-test). n/N = # of neurons/# of independent experiments. In all graphs, data represent average values ± SEM.
Next, we investigated whether RA treatment altered synapse density in control neurons. Consistent with lack of changes in mEPSC and mIPSC frequencies, we did not observe any changes in excitatory or inhibitory synapse density, as assessed morphologically by staining excitatory and inhibitory synapses for PSD95 and VGAT1, respectively (fig. S7). Thus, RA’s effect on synaptic strength was not mediated by modification of synapse numbers. In mouse neurons, RA increases excitatory synaptic strength by promoting synthesis and synaptic insertion of homomeric GluA1 AMPARs (34). In our system, 1Naphthylacetyl spermine trihydrochloride (NASPM), a polyamine blocker specific for GluA2-lacking AMPARs (66), completely reversed the RA-induced increase in mEPSC amplitude in control neurons without affecting basal amplitude in untreated neurons (Fig. 6C), indicating a similar mechanism involving synaptic incorporation of homomeric GluA1 AMPARs at play in human neurons.
Absence of homeostatic synaptic plasticity in neurons derived from FXS patients
Synaptic RA-signaling is a key component in the molecular pathway mediating homeostatic synaptic plasticity (67). To confirm that the block of synaptic RA-signaling in FXS results in a loss of RA-dependent homeostatic plasticity, we again examined the effect of chronic synaptic silencing (24 hr TTX and CNQX treatment), but now studied neurons differentiated from control and FXS-patient iPS cells co-cultured with mouse neurons and glia. As a further control to ensure that we were monitoring an RA-dependent response, we examined the effect of synaptic silencing in both the absence and presence of the RA-synthesis blocker DEAB. We monitored both spontaneous mEPSCs and mIPSCs as well as evoked EPSCs and IPSCs. In control iPS but not in FXS iPS cells, synaptic silencing increased the mEPSC amplitude (Fig. 7A, 7B, fig. S8A - S8D), decreased the mIPSC amplitude (Fig. 7C, 7D, fig. S8E – 8H). Consistent with changes in miniature synaptic responses, in control but not FXS iPS cells, synaptic silencing increased the evoked EPSC amplitude (Fig. 7E – 7G, fig. S8I and S8J), and decreased the evoked IPSC amplitude (Fig. 7H – 7J, fig. S8K and S8L). All of these changes were blocked by co-incubation with DEAB. Additionally, in control iPS cells, 24-hr TTX+CNQX treatment occluded further response to RA treatment in these neurons (fig. S9), demonstrating that synaptic silencing-induced mEPSC and mIPSC changes are mediated by RA. Thus, homeostatic synaptic plasticity is similarly abolished in human neurons in which the FMR1 gene was inactivated either conditionally or via a pathogenic event in FXS patients.
Figure 7. Impaired homeostatic synaptic plasticity induced by synaptic silencing i in human neurons differentiated from FXS patient iPS cells.
(A-B) Example traces and quantification for mEPSC amplitudes recorded from iN cells differentiated from control iPS cell #1 line (A) or FXS iPS cell #1 line (B), co-cultured with mouse cortical neurons, and treated with DMSO (24 h), TTX+CNQX (24 h), or TTX+CNQX+DEAB (24 h) (***, p < 0.001; one-way ANOVA). Scale bars: left, 20 pA and 1s; right, 5 pA and 5 ms. (C-D) Example traces and quantification for mIPSC amplitudes recorded from iN cells differentiated from control iPS cell #1 line (C) and FXS iPS cell #1 line (D) treated with DMSO (24 h), TTX+CNQX (24 h), or TTX+CNQX+DEAB (24 h) (**, p < 0.01; one-way ANOVA). Scale bars: left, 50 pA and 1s; right, 5 pA and 50 ms. (E) Example traces of evoked EPSCs recorded from control and FXS iN cells under various treatment conditions. Scale bars: 200 pA and 10 ms. (F-G) Quantification of eEPSC amplitudes from control (F) and FXS (G) iN cells (**, p < 0.01; one-way ANOVA). (H) Example traces of evoked IPSCs recorded from control and FXS iN cells under various treatment conditions. Scale bars: 200 pA and 20 ms. (I-J) Quantification of eIPSC amplitudes from control (I) and FXS (J) iN cells (**, p < 0.01; one-way ANOVA). n/N = # of neurons/# of independent experiments. In all graphs, data represent average values ± SEM.
Restoring synaptic RA signaling by removing pathogenic expanded CGG repeats from the FMR1 gene of an FXS patient
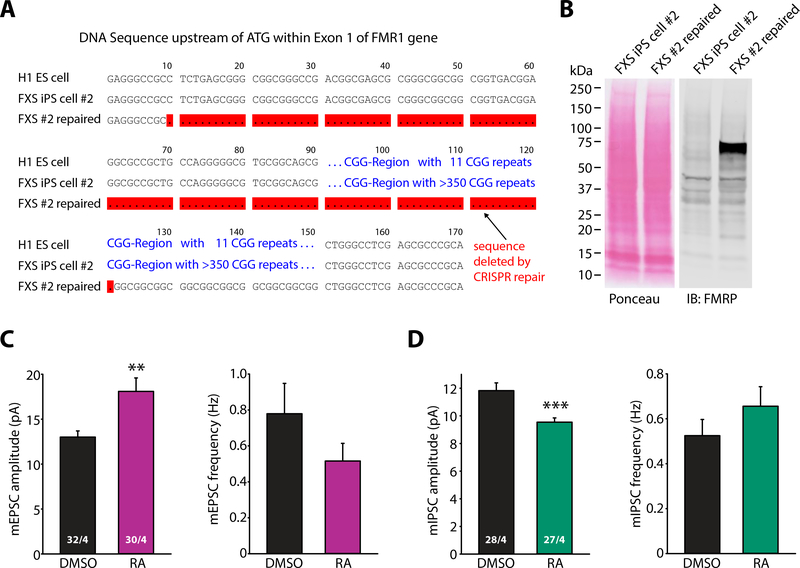
Our data using FMR1 cKO ES cell lines thus provide strong evidence that lack of FMRP expression directly underlies impaired RA-mediated homeostatic synaptic plasticity seen in human FXS neurons. Therefore, we hypothesized that RA signaling in FXS neurons may be restored by repairing the defective FMR1 gene. We thus applied CRISPR/Cas9mediated genome editing methods to one of the FXS patient iPS cell lines (FXS #2, SC153) in order to excise the expanded CGG repeats. DNA sequencing indicated successful removal of the CGG repeats upstream of the initiator ATG in exon 1 of the FMR1 gene (Fig. 8A), and recovery of FMRP expression was confirmed by immunoblotting (Fig. 8B).
Figure 8. Rescues of synaptic RA signaling in human neurons induced from FXS iPS cells by CRISPR/Cas9-mediated removal of excessive CGG repeats upstreams of FMR1 gene.
(A) Sequence alignment diagram showing the region within exon 1 flanking the CGG repeats upstream of ATG of FMR1 gene from a control human ES cell line (H1) and a FXS patient iPS cell line before and after the CRISPR/Cas9-mediated removal of excessive CGG repeats (red-highlighted). (B) Immunoblot of FMRP expression in FXS iPS cell line before and after the repair. (C-D) Quantification of mEPSC (C) and mIPSC (D) amplitudes and frequencies in neurons derived from repaired FXS iPS cells treated with DMSO or RA (**, p < 0.01; ***, p < 0.001; student’s t-test). n/N = # of neurons/# of independent experiments. In all graphs, data represent average values ± SEM.
In the repaired FXS neurons, responsiveness to RA at both excitatory and inhibitory synapses was fully restored, indicated by a robust increase in mEPSC amplitude (Fig. 8C) and a decrease in mIPSC amplitude upon RA treatment (Fig. 8D). This result, together with the data from the FMR1 cKO neurons, suggest a direct causal relationship between silenced FMR1 gene function and defective synaptic RA signaling in FXS.
Discussion
This study examines the impairments induced by FMR1 mutations in human neurons. In view of the widely divergent results from prior studies on this issue (53, 57, 59, 68) (see discussion below), we analyzed the effects of both genetically engineered conditional FMR1 mutations and of disease-associated FMR1 mutations in patient-derived iPS cells, and additionally confirmed the role of the FMR1 mutation by repairing a pathogenic mutation in an FXS patient-derived iPS cell line. In this manner, we were able to investigate the effects of FMR1 mutations both with precise control of genetic background in the conditional FMR1 mutations, and with a clinically relevant approach in the patient-derived FMR1 mutations. Our study builds on previous results obtained in rodent models bearing Fmr1 mutations, which showed that such mutations specifically impair homeostatic plasticity via interfering with synaptic RA signaling (29, 30). Since homeostatic plasticity and synaptic RA signaling in human neurons have not been previously studied, we first examined whether these processes are present in human neurons and could be reproduced with neurons derived from ES or iPS cells. Thus, we first described the properties of RA-dependent homeostatic plasticity in human neurons with functionally mature synapses, and then analyzed the effect of both conditionally engineered and patient-associated FMR1 mutations on such plasticity, demonstrating that FMR1 mutations induced a robust and universal block of homeostatic plasticity in human neurons.
Our data allow the following overall conclusions. First, RA-dependent homeostatic plasticity in human neurons operates in a two-stage process, an induction of the RA signal via synaptic silencing, and the transduction of the RA signal into a synaptic response (fig. S10). DEAB, an RA synthesis enzyme blocker, inhibited homeostatic synaptic plasticity by abolishing RA synthesis in response to synaptic silencing. Direct RA treatment mimics the effect of synaptic silencing-induced at both excitatory and inhibitory synapses, and synaptic silencing occludes RA-induced synaptic changes. These results show that in human neurons co-cultured with mouse neurons, which form both excitatory and inhibitory synapses, the RA-dependent homeostatic plasticity mechanism is conserved.
Second, deletion of FMR1 in mature neurons has no major effects on the principal properties of human neurons, including their basic synaptic properties as assayed in vitro. This conclusion holds true for both conditional and patient-associated FMR1 mutations, and might partially help to explain why FXS patients, although affected by intellectual disability, are often still able to speak and in some cases to act semi-independently. Compared to our study, previous iPS cell analysis reported more dramatic morphological effects from FMR1 deletion, which might represent neurogenesis defects that may be compensated in vivo.
Third, FMR1 mutations selectively block the synaptic transduction of a homeostatically produced RA signal, and thereby impair homeostatic plasticity. This observation –the core of our results, confirmed independently with multiple lines of evidence– identifies a robust impairment in human FXS neurons that might contribute to the synaptic and circuit abnormalities underlying the cognitive impairments in FXS, and that could potentially form the basis for in vitro drug screening.
Our results differ from previous studies that reported a divergent range of other phenotypes produced by FMR1 mutations in human neurons. These phenotypes can be summarized to affect three main processes: neuronal differentiation, neuronal maturation, and synaptic functional maturation. In terms of neuronal differentiation, unlike a previously reported shift to gliogenic development (57), our results show that the neuronal conversion efficiency is comparable between WT (mCre-infected) and FMR1 KO (Cre-infected) ES cells, suggesting that neurogenesis is normal despite silenced FMRP expression. With regards to neuronal maturation, poor neuronal maturation with defects in initial neurite outgrowth (53) as well as reduced neuronal excitability (inability to discharge trains of APs), reduced inward Na+ currents, and decreased outward K+ currents were observed in neurons from FXS patients (59). By contrast, our observations on neurons differentiated from either FXS iPS cells or FMR1 cKO ES cells showed that these neurons exhibited normal neuronal morphology and no change in synapse numbers. Moreover, in our hands, human neurons differentiated from FMR1 cKO ES cells or FXS patient iPS cells respond to current injections with robust action potential firing. In fact, we observed changes opposite to those previously reported – that is increased neuronal excitability and enhanced voltage-activated Na+ currents in FXS neurons derived from FXS iPS cells and in FXS neurons derived from one of the two FMR1 cKO ES cell lines compared to WT controls. In terms of functional maturation of synapses, previous studies using FXS neurons differentiated from human FXS ES cells reported a complete absence of spontaneous synaptic activity in FXS neurons, which the authors attributed to fewer synaptic vesicles (59), whereas another study reported immature responses to GABA, which was thought to be caused by impaired GABAergic synapse development during neurogenesis (68). Complete absence of synaptic activity is likely not compatible with the clinical presentation of FXS. Our results showing no difference in basal synaptic transmission at either excitatory or inhibitory synapses in iPS cell-derived or ES cell-derived FXS neurons suggest that these cells maintain a certain degree of functionality and are compatible with the clinical manifestation of FXS.
What may account for these apparently contradictory observations on neurons generated from FXS patients? In previous studies ES cells derived from FXS blastocysts show no methylation of the FMR1 locus and proper FMRP expression. Neurons differentiated from such ES cells retain FMRP expression in the very early stages as epigenetic silencing of the FMR1 gene by DNA methylation and histone modification only occurs upon differentiation (47) (but see (48)). In one extreme case, expression of FMR1 mRNA and FMRP were found normal until 48 days after neuronal differentiation in neurons derived from FXS hES cells derived from FXS blastocysts (69). By contrast, our FXS neurons are differentiated from FXS iPSC cells where FMRP expression is already silenced before the differentiation step. Thus, FMRP expression immediately before neuronal differentiation may contribute to phenotypic differences observed between our study and others. However, our results with neurons differentiated from FMR1 cKO human ES cells argue differently. In these experiments, FMRP expression is normal in ES cells and silencing occurs at the same time of neuronal differentiation because we transduced ES cells with Ngn2-expressing and Cre-expressing viruses simultaneously. Some of the previously reported defect in neurons differentiated from patient ES or iPS cells may thus simply reflect clonal variability rather than a direct consequence of lack of FMRP expression.
Another potential contributing factor to experimental differences is the culture condition. Most previous studies on human FXS neurons were performed using pure human neurons cultured in supplemented media on surfaces coated with various substrates (53, 57, 68). Our human neurons were co-cultured with mouse glia shortly after differentiation and with mouse cortical neurons that were added two weeks later. Contribution of glia cells to neuronal maturation and synapse formation has been demonstrated in numerous studies (70, 71). In the current study, we further added mouse cortical neurons to provide inhibitory synaptic inputs to these otherwise completely excitatory human neurons, thus allowing the establishment of a synaptic network that has at least some resemblance to balanced synaptic excitation and inhibition, which may be key to the induction of homeostatic synaptic plasticity in human neurons. Thus, culture conditions, which dictate the stages of neuronal and synaptic maturation, may contribute to some of the discrepancies observed between our study and the previous ones.
A third factor potentially contributing to the differences in reports is the difference in neuronal differentiation protocols (forced Ngn2 expression versus dual SMAD inhibition protocol involving intermediate progenitors (60)), which might produce different types of neurons and contribute to the different phenotypes observed in various studies. It is possible that the various seemingly contradicting phenotypes reflect the diverse phenotypic manifestation of FMR1 mutations in different neuronal types in FXS patients. However, the grossly unaltered morphology of the brain and the normal basic behaviors (moving, feeding) observed in FXS patients and in FXS mouse models lead us to speculate that FXS affects preferentially higher functions of the brain, represented at physiological level by subtler neurological abnormalities rather than severely impaired basal synaptic transmission and neuronal development.
Our study raises multiple questions. Although the synaptic effects of RA inhibited in FXS are very robust, our results do not exclude the possibility that FMR1 inactivation causes additional major phenotypes besides blocking homeostatic plasticity. In our experiments, we observed a modest increase in neuronal excitability in FMR1-mutant neurons derived from both patient-derived iPS cell lines and from one of the two cKO ES cell lines, suggesting that increased neuronal excitability may be a general feature of FXS, but may have been lost for inexplicable reasons from one of the two cKO lines. Indeed, approximately 10–20% FXS patients with full mutation exhibit childhood seizures (72–74), suggesting that increased neuronal excitability may be associated with FXS. Similarly, we observed in neurons derived from one of the FXS patient iPS cell lines but not from cKO ES cells a small change in dendritic arborization, which may either be due to genetic background effects (which are not controlled in the patient-derived neurons), or reflect an additional feature of iPS cell-derived neurons. Both of these observations require further study, but are clearly not as universal as the changes in homeostatic plasticity uniformly observed in all FMR1-mutant neurons.
Finally, previous work in mice showed that Fmr1 mutations caused an increase in mGluR5-mediated LTD (22). Both LTD and homeostatic synaptic plasticity phenotypes involve changes in postsynaptic receptor trafficking (29, 34, 67, 75, 76), and the final pathways regulating such trafficking that are activated by synaptic RA-signaling may overlap with those used in LTD. Thus, there may be functional interactions between different types of synaptic plasticity that could be altered in the absence of FMRP. Future studies will have to pursue this exciting possibility.
By necessity, our work in human neurons was entirely performed in culture. This approach, however, includes limitations in that we could not analyze the effects of the FMR1 mutations on a physiological neural circuit in a living brain. Because cultured neurons lack the full scope of synaptic properties observed in situ in the brain and are for example unable to undergo classical NMDA-receptor dependent long-term potentiation or long-term depression, our study cannot make any conclusions about the impact of FMR1 mutations on these forms of plasticity, and cannot analyze the circuit consequences of changes in homeostatic plasticity in a human brain. A second limitation of our study is that due to technical conditions, we did not examine inhibitory human neurons, which may have exhibited additional phenotypes induced by FMR1 mutations. Thus, although our results uncovered a loss of homeostatic plasticity as a robust impairment in FMR1 mutant human neurons that could potentially be a major contributor to the overall clinical presentation of fragile-X syndrome, we cannot - and do not wish to - exclude the possibility that FMR1 mutations cause additional major phenotypes in human neurons that were not accessible to our experimental approach.
How may defective synaptic RA signaling and homeostatic synaptic plasticity contribute to the clinical presentation of FXS? Identified and characterized as a developmental morphogen, RA has been primarily studied as a transcriptional regulator during development. However, the link between RA and neuropsychiatric disease also has a surprisingly long history (77–84), supporting RA’s role in cognitive function in adults. Homeostatic plasticity is a form of synaptic plasticity that is distinct from Hebbian plasticity. Although it has always been thought that the primary functional significance of homeostatic synaptic plasticity is to maintain neural network stability by dynamically regulating neuronal excitability, the biochemical events involved in the homeostatic adjustment of synaptic activity may well directly influence the ability of a neuron to undergo Hebbian-type plasticity. Acting through a distinct molecular mechanism, RA is capable of rapidly changing excitatory and inhibitory synaptic strength (29, 34). Thus, RA acts as a candidate ‘metaplasticity molecule’ capable of changing the state of a neuron (its excitatory/inhibitory balance) and influencing Hebbian plasticity (85). Indeed, earlier studies showed that vitamin A deficiency (which depletes RA) leads to impaired hippocampal Hebbian plasticity and learning (86–88). A more recent study using a dominant negative form of RARα expressed in adult forebrain demonstrated impairments in AMPAR-mediated synaptic transmission, hippocampal LTP, hippocampal-dependent social recognition, and spatial memory (89). In our hands, RA-induced increases in excitatory synaptic transmission significantly impaired subsequent induction of LTP, a phenomenon that can be reversed by acute genetic deletion of RARα or inhibiting protein synthesis during RA treatment (75). These findings suggest that the functional impact of RA may go beyond homeostatic plasticity, and affects performances of neural circuits underlying cognitive functions. Thus, impaired RA signaling may contribute to various aspects of synaptic and circuit dysfunction in FXS patients.
EXPERIMENTAL METHODS
Study design
This study was designed to thoroughly characterize the basic morphological and functional properties as well as retinoic acid (RA)-mediated synaptic silencing-induced homeostatic synaptic plasticity in human neurons differentiated from human ES cells or iPS cells, and to investigate whether RA-dependent homeostatic plasticity is absent in human neurons lacking FMRP expression. We generated FMR1 cKO hES cells to test the necessity of FMRP expression for synaptic RA signaling and homeostatic plasticity, then verified the clinical relevance of the results in human neurons derived from control and FXS patient iPS cells. Additionally, we performed CRISPR/Cas9-mediated repair of the defective FMR1 gene in FXS iPS cells to further investigate the causality between FMRP expression and RA-mediated homeostatic synaptic plasticity. All human samples and animal work were approved by the Institutional Animal Care and Use Committee (IACUC) and Institutional Review Board (IRB) of Stanford University. For all experiments, three to six biological replicates (independent experiments) were performed and all results were analyzed by experimenters who were blinded to the experimental groups.
Statistical Analysis
Data were presented as mean ± standard SEM. Sample sizes were estimated with power set at 0.8 and α value at 0.05. Statistical significances were tested with two-tailed t-test or ANOVA. Interactions between factors were tested with two-way ANOVA. Tests were performed with Microsoft Excel and GraphPad Prism 6 software. p-Values are indicated where tests have been performed.
Supplementary Material
Acknowledgment:
We thank members of the Chen, Wernig and Südhof labs for comments and help during the course of the study. We also thank the Stanford Neuroscience Microscopy Service for technical assistance. Fragile X Syndrome (FXS) induced pluripotent stem (iPS) cells are available from the Autism Spectrum Disorders Stem Cell Resource at Children’s Hospital of Orange County, CA, USA under a material transfer agreement with Dr. Philip H. Schwartz.
Funding: The work was supported by NIH grants MH086403 (L.C.), MH091193 (L.C.), HD084215 (L.C.), R01MH092931 (to M.W. and T.C.S.), the Department of Defense grant PR100175 (to L.C. and M.W.), the FRAXA Research Foundation Award (S.G.M), and the New York Stem Cell Foundation-Robertson Stem Cell Award (M.W.). M.W. is a Tashia and John Morgridge Faculty Scholar, Child Health Research Institute at Stanford and New York Stem Cell Foundation Robertson Investigator. T.C.S. is a Howard Hughes Medical Institute Investigator.
Footnotes
Competing interests: The authors do not have competing interests.
Data availability: All data associated with this study are present in the paper or supplementary materials.
References
- 1.Santoro MR, Bray SM, Warren ST, Molecular mechanisms of fragile X syndrome: a twentyyear perspective. Annu Rev Pathol 7, 219 (2012). [DOI] [PubMed] [Google Scholar]
- 2.Bagni C, Tassone F, Neri G, Hagerman R, Fragile X syndrome: causes, diagnosis, mechanisms, and therapeutics. J Clin Invest 122, 4314 (December, 2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Verkerk AJ et al. , Identification of a gene (FMR-1) containing a CGG repeat coincident with a breakpoint cluster region exhibiting length variation in fragile X syndrome. Cell 65, 905 (May 31, 1991). [DOI] [PubMed] [Google Scholar]
- 4.Fu YH et al. , Variation of the CGG repeat at the fragile X site results in genetic instability: resolution of the Sherman paradox. Cell 67, 1047 (December 20, 1991). [DOI] [PubMed] [Google Scholar]
- 5.Heitz D, Devys D, Imbert G, Kretz C, Mandel JL, Inheritance of the fragile X syndrome: size of the fragile X premutation is a major determinant of the transition to full mutation. J Med Genet 29, 794 (November, 1992). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Suhl JA, Warren ST, Single-Nucleotide Mutations in FMR1 Reveal Novel Functions and Regulatory Mechanisms of the Fragile X Syndrome Protein FMRP. J Exp Neurosci 9, 35 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Laggerbauer B, Ostareck D, Keidel EM, Ostareck-Lederer A, Fischer U, Evidence that fragile X mental retardation protein is a negative regulator of translation. Hum Mol Genet 10, 329 (February 15, 2001). [DOI] [PubMed] [Google Scholar]
- 8.Ashley CT Jr., Wilkinson KD, Reines D, Warren ST, FMR1 protein: conserved RNP family domains and selective RNA binding. Science 262, 563 (October 22, 1993). [DOI] [PubMed] [Google Scholar]
- 9.Hinds HL et al. , Tissue specific expression of FMR-1 provides evidence for a functional role in fragile X syndrome. Nat Genet 3, 36 (January, 1993). [DOI] [PubMed] [Google Scholar]
- 10.Chen L, Yun SW, Seto J, Liu W, Toth M, The fragile X mental retardation protein binds and regulates a novel class of mRNAs containing U rich target sequences. Neuroscience 120, 1005 (2003). [DOI] [PubMed] [Google Scholar]
- 11.Miyashiro KY et al. , RNA cargoes associating with FMRP reveal deficits in cellular functioning in Fmr1 null mice. Neuron 37, 417 (February 6, 2003). [DOI] [PubMed] [Google Scholar]
- 12.Darnell JC et al. , FMRP stalls ribosomal translocation on mRNAs linked to synaptic function and autism. Cell 146, 247 (July 22, 2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bassell GJ, Warren ST, Fragile X syndrome: loss of local mRNA regulation alters synaptic development and function. Neuron 60, 201 (October 23, 2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li Z et al. , The fragile X mental retardation protein inhibits translation via interacting with mRNA. Nucleic Acids Res 29, 2276 (June 1, 2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gibson JR, Bartley AF, Hays SA, Huber KM, Imbalance of neocortical excitation and inhibition and altered UP states reflect network hyperexcitability in the mouse model of fragile X syndrome. J Neurophysiol 100, 2615 (November, 2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tyzio R et al. , Oxytocin-mediated GABA inhibition during delivery attenuates autism pathogenesis in rodent offspring. Science 343, 675 (February 7, 2014). [DOI] [PubMed] [Google Scholar]
- 17.Centonze D et al. , Abnormal striatal GABA transmission in the mouse model for the fragile X syndrome. Biol Psychiatry 63, 963 (May 15, 2008). [DOI] [PubMed] [Google Scholar]
- 18.Curia G, Papouin T, Seguela P, Avoli M, Downregulation of tonic GABAergic inhibition in a mouse model of fragile X syndrome. Cereb Cortex 19, 1515 (July, 2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Olmos-Serrano JL et al. , Defective GABAergic neurotransmission and pharmacological rescue of neuronal hyperexcitability in the amygdala in a mouse model of fragile X syndrome. J Neurosci 30, 9929 (July 21, 2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Larson J, Jessen RE, Kim D, Fine AK, du Hoffmann J, Age-dependent and selective impairment of long-term potentiation in the anterior piriform cortex of mice lacking the fragile X mental retardation protein. J Neurosci 25, 9460 (October 12, 2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Li J, Pelletier MR, Perez Velazquez JL, Carlen PL, Reduced cortical synaptic plasticity and GluR1 expression associated with fragile X mental retardation protein deficiency. Mol Cell Neurosci 19, 138 (February, 2002). [DOI] [PubMed] [Google Scholar]
- 22.Huber KM, Gallagher SM, Warren ST, Bear MF, Altered synaptic plasticity in a mouse model of fragile X mental retardation. Proc Natl Acad Sci U S A 99, 7746 (May 28, 2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Coghlan S et al. , GABA system dysfunction in autism and related disorders: from synapse to symptoms. Neurosci Biobehav Rev 36, 2044 (October, 2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fung LK, Quintin EM, Haas BW, Reiss AL, Conceptualizing neurodevelopmental disorders through a mechanistic understanding of fragile X syndrome and Williams syndrome. Curr Opin Neurol 25, 112 (April, 2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Paluszkiewicz SM, Martin BS, Huntsman MM, Fragile X syndrome: the GABAergic system and circuit dysfunction. Dev Neurosci 33, 349 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang T, Bray SM, Warren ST, New perspectives on the biology of fragile X syndrome. Curr Opin Genet Dev 22, 256 (June, 2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Contractor A, Klyachko VA, Portera-Cailliau C, Altered Neuronal and Circuit Excitability in Fragile X Syndrome. Neuron 87, 699 (August 19, 2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pfeiffer BE, Huber KM, The state of synapses in fragile X syndrome. Neuroscientist 15, 549 (October, 2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sarti F, Zhang Z, Schroeder J, Chen L, Rapid suppression of inhibitory synaptic transmission by retinoic acid. J Neurosci 33, 11440 (July 10, 2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Soden ME, Chen L, Fragile X protein FMRP is required for homeostatic plasticity and regulation of synaptic strength by retinoic acid. J Neurosci 30, 16910 (December 15, 2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Turrigiano G, Homeostatic synaptic plasticity: local and global mechanisms for stabilizing neuronal function. Cold Spring Harb Perspect Biol 4, a005736 (January, 2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Turrigiano GG, Leslie KR, Desai NS, Rutherford LC, Nelson SB, Activity-dependent scaling of quantal amplitude in neocortical neurons. Nature 391, 892 (February 26, 1998). [DOI] [PubMed] [Google Scholar]
- 33.Chambon P, A decade of molecular biology of retinoic acid receptors. Faseb J 10, 940 (July, 1996). [PubMed] [Google Scholar]
- 34.Aoto J, Nam CI, Poon MM, Ting P, Chen L, Synaptic signaling by all-trans retinoic acid in homeostatic synaptic plasticity. Neuron 60, 308 (October 23, 2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sarti F, Schroeder J, Aoto J, Chen L, Conditional RARalpha knockout mice reveal acute requirement for retinoic acid and RARalpha in homeostatic plasticity. Front Mol Neurosci 5, 16 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Poon MM, Chen L, Retinoic acid-gated sequence-specific translational control by RARalpha. Proc Natl Acad Sci U S A 105, 20303 (December 23, 2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Arendt KL et al. , Calcineurin mediates homeostatic synaptic plasticity by regulating retinoic acid synthesis. Proc Natl Acad Sci U S A 112, E5744 (October 20, 2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang HL, Zhang Z, Hintze M, Chen L, Decrease in calcium concentration triggers neuronal retinoic acid synthesis during homeostatic synaptic plasticity. J Neurosci 31, 17764 (December 7, 2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Maghsoodi B et al. , Retinoic acid regulates RARalpha-mediated control of translation in dendritic RNA granules during homeostatic synaptic plasticity. Proc Natl Acad Sci U S A 105, 16015 (October 14, 2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hanna JH, Saha K, Jaenisch R, Pluripotency and cellular reprogramming: facts, hypotheses, unresolved issues. Cell 143, 508 (November 12, 2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Okita K, Yamanaka S, Induced pluripotent stem cells: opportunities and challenges. Philos Trans R Soc Lond B Biol Sci 366, 2198 (August 12, 2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Zhang Y et al. , Rapid single-step induction of functional neurons from human pluripotent stem cells. Neuron 78, 785 (June 5, 2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pang ZP et al. , Induction of human neuronal cells by defined transcription factors. Nature 476, 220 (August 11, 2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pasca SP, Panagiotakos G, Dolmetsch RE, Generating human neurons in vitro and using them to understand neuropsychiatric disease. Annu Rev Neurosci 37, 479 (2014). [DOI] [PubMed] [Google Scholar]
- 45.Marchetto MC, Gage FH, Modeling brain disease in a dish: really? Cell Stem Cell 10, 642 (June 14, 2012). [DOI] [PubMed] [Google Scholar]
- 46.Verlinsky Y et al. , Human embryonic stem cell lines with genetic disorders. Reprod Biomed Online 10, 105 (January, 2005). [DOI] [PubMed] [Google Scholar]
- 47.Eiges R et al. , Developmental study of fragile X syndrome using human embryonic stem cells derived from preimplantation genetically diagnosed embryos. Cell Stem Cell 1, 568 (November, 2007). [DOI] [PubMed] [Google Scholar]
- 48.Avitzour M et al. , FMR1 epigenetic silencing commonly occurs in undifferentiated fragile Xaffected embryonic stem cells. Stem Cell Reports 3, 699 (November 11, 2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gerhardt J et al. , The DNA replication program is altered at the FMR1 locus in fragile X embryonic stem cells. Mol Cell 53, 19 (January 9, 2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bar-Nur O, Caspi I, Benvenisty N, Molecular analysis of FMR1 reactivation in fragile-X induced pluripotent stem cells and their neuronal derivatives. J Mol Cell Biol 4, 180 (June, 2012). [DOI] [PubMed] [Google Scholar]
- 51.Brick DJ et al. , The Autism Spectrum Disorders Stem Cell Resource at Children’s Hospital of Orange County: Implications for Disease Modeling and Drug Discovery. Stem Cells Transl Med 3, 1275 (November, 2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.de Esch CE et al. , Epigenetic characterization of the FMR1 promoter in induced pluripotent stem cells from human fibroblasts carrying an unmethylated full mutation. Stem Cell Reports 3, 548 (October 14, 2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Doers ME et al. , iPSC-derived forebrain neurons from FXS individuals show defects in initial neurite outgrowth. Stem Cells Dev 23, 1777 (August 1, 2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Halevy T, Czech C, Benvenisty N, Molecular mechanisms regulating the defects in fragile X syndrome neurons derived from human pluripotent stem cells. Stem Cell Reports 4, 37 (January 13, 2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sheridan SD et al. , Epigenetic characterization of the FMR1 gene and aberrant neurodevelopment in human induced pluripotent stem cell models of fragile X syndrome. PLoS One 6, e26203 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Urbach A, Bar-Nur O, Daley GQ, Benvenisty N, Differential modeling of fragile X syndrome by human embryonic stem cells and induced pluripotent stem cells. Cell Stem Cell 6, 407 (May 7, 2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Telias M, Segal M, Ben-Yosef D, Neural differentiation of Fragile X human Embryonic Stem Cells reveals abnormal patterns of development despite successful neurogenesis. Dev Biol 374, 32 (February 1, 2013). [DOI] [PubMed] [Google Scholar]
- 58.Niedringhaus M et al. , Transferable neuronal mini-cultures to accelerate screening in primary and induced pluripotent stem cell-derived neurons. Sci Rep 5, 8353 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Telias M, Kuznitsov-Yanovsky L, Segal M, Ben-Yosef D, Functional Deficiencies in Fragile X Neurons Derived from Human Embryonic Stem Cells. J Neurosci 35, 15295 (November 18, 2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Chambers SM et al. , Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat Biotechnol 27, 275 (March, 2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.The Hd iPsc Consortium, Induced Pluripotent Stem Cells from Patients with Huntington’s Disease Show CAG-Repeat-Expansion-Associated Phenotypes. Cell Stem Cell 11, 264 (2012/08/03/, 2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Soldner F et al. , Generation of Isogenic Pluripotent Stem Cells Differing Exclusively at Two Early Onset Parkinson Point Mutations. Cell 146, 318 (2011/07/22/, 2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Patzke C et al. , Analysis of conditional heterozygous STXBP1 mutations in human neurons. J Clin Invest 125, 3560 (September, 2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Patzke C, Sudhof TC, The conditional KO approach: Cre/Lox technology in human neurons. Rare Dis 4, e1131884 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kavalali ET, The mechanisms and functions of spontaneous neurotransmitter release. Nat Rev Neurosci 16, 5 (January, 2015). [DOI] [PubMed] [Google Scholar]
- 66.Nelson SB, Turrigiano GG, Synaptic depression: a key player in the cortical balancing act. Nat Neurosci 1, 539 (November, 1998). [DOI] [PubMed] [Google Scholar]
- 67.Chen L, Lau AG, Sarti F, Synaptic retinoic acid signaling and homeostatic synaptic plasticity. Neuropharmacology (in press), (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Telias M, Segal M, Ben-Yosef D, Immature Responses to GABA in Fragile X Neurons Derived from Human Embryonic Stem Cells. Front Cell Neurosci 10, 121 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Colak D et al. , Promoter-bound trinucleotide repeat mRNA drives epigenetic silencing in fragile X syndrome. Science 343, 1002 (February 28, 2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Huang YA, Zhou B, Wernig M, Sudhof TC, ApoE2, ApoE3, and ApoE4 Differentially Stimulate APP Transcription and Abeta Secretion. Cell 168, 427 (January 26, 2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ullian EM, Christopherson KS, Barres BA, Role for glia in synaptogenesis. Glia 47, 209 (August 15, 2004). [DOI] [PubMed] [Google Scholar]
- 72.Incorpora G, Sorge G, Sorge A, Pavone L, Epilepsy in fragile X syndrome. Brain Dev 24, 766 (December, 2002). [DOI] [PubMed] [Google Scholar]
- 73.Berry-Kravis E, Epilepsy in fragile X syndrome. Dev Med Child Neurol 44, 724 (November, 2002). [DOI] [PubMed] [Google Scholar]
- 74.Berry-Kravis E et al. , Seizures in fragile X syndrome: characteristics and comorbid diagnoses. Am J Intellect Dev Disabil 115, 461 (November, 2010). [DOI] [PubMed] [Google Scholar]
- 75.Arendt KL et al. , Retinoic Acid and LTP Recruit Postsynaptic AMPA Receptors Using Distinct SNARE-Dependent Mechanisms. Neuron 86, 442 (April 22, 2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Waung MW, Huber KM, Protein translation in synaptic plasticity: mGluR-LTD, Fragile X. Curr Opin Neurobiol 19, 319 (June, 2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Muenter MD, Perry HO, Ludwig J, Chronic vitamin A intoxication in adults. Hepatic, neurologic and dermatologic complications. Am J Med 50, 129 (January, 1971). [DOI] [PubMed] [Google Scholar]
- 78.Hull PR, D’Arcy C, Acne, depression, and suicide. Dermatol Clin 23, 665 (October, 2005). [DOI] [PubMed] [Google Scholar]
- 79.Bremner JD, McCaffery P, The neurobiology of retinoic acid in affective disorders. Prog Neuropsychopharmacol Biol Psychiatry 32, 315 (February 15, 2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Scher JM, Psychotic reaction to disulfiram. Jama 201, 1051 (September 25, 1967). [DOI] [PubMed] [Google Scholar]
- 81.Martensen-Larsen O, Psychotic phenomena provoked by tetraethylthiuram disulfide. Q J Stud Alcohol 12, 206 (June, 1951). [PubMed] [Google Scholar]
- 82.Liddon SC, Satran R, Disulfiram (Antabuse) psychosis. Am J Psychiatry 123, 1284 (April, 1967). [DOI] [PubMed] [Google Scholar]
- 83.Guris DL, Duester G, Papaioannou VE, Imamoto A, Dose-dependent interaction of Tbx1 and Crkl and locally aberrant RA signaling in a model of del22q11 syndrome. Dev Cell 10, 81 (January, 2006). [DOI] [PubMed] [Google Scholar]
- 84.Ceylan ME, Turkcan A, Mutlu E, Onal O, Manic episode with psychotic symptoms associated with high dose of disulfiram: a case report. J Clin Psychopharmacol 27, 224 (April, 2007). [DOI] [PubMed] [Google Scholar]
- 85.Yee AX, Hsu YT, Chen L, A metaplasticity view of the interaction between homeostatic and Hebbian plasticity. Philos Trans R Soc Lond B Biol Sci 372, (March 5, 2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Chiang MY et al. , An essential role for retinoid receptors RARbeta and RXRgamma in long-term potentiation and depression. Neuron 21, 1353 (December, 1998). [DOI] [PubMed] [Google Scholar]
- 87.Cocco S et al. , Vitamin A deficiency produces spatial learning and memory impairment in rats. Neuroscience 115, 475 (2002). [DOI] [PubMed] [Google Scholar]
- 88.Misner DL et al. , Vitamin A deprivation results in reversible loss of hippocampal long-term synaptic plasticity. Proc Natl Acad Sci U S A 98, 11714 (September 25, 2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Nomoto M et al. , Dysfunction of the RAR/RXR signaling pathway in the forebrain impairs hippocampal memory and synaptic plasticity. Mol Brain 5, 8 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Sebastiano V et al. , In situ genetic correction of the sickle cell anemia mutation in human induced pluripotent stem cells using engineered zinc finger nucleases. Stem Cells 29, 1717 (November, 2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Xu C et al. , Feeder-free growth of undifferentiated human embryonic stem cells. Nat Biotechnol 19, 971 (October, 2001). [DOI] [PubMed] [Google Scholar]
- 92.Chen C, Okayama H, High-efficiency transformation of mammalian cells by plasmid DNA. Mol Cell Biol 7, 2745 (August, 1987). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lisowski L et al. , Selection and evaluation of clinically relevant AAV variants in a xenograft liver model. Nature 506, 382 (February 20, 2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Sanjana NE, Shalem O, Zhang F, Improved vectors and genome-wide libraries for CRISPR screening. Nature methods 11, 783 (August, 2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Park CY et al. , Reversion of FMR1 Methylation and Silencing by Editing the Triplet Repeats in Fragile X iPSC-Derived Neurons. Cell Rep 13, 234 (October 13, 2015). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.