Publisher's Note: There is a Blood Commentary on this article in this issue.
Key Points
BCL6 expression is necessary in donor marrow B and T cells for a GC reaction in cGVHD mice.
A small-molecule inhibitor of BCL6-mediated transcriptional repression presents a novel therapeutic strategy for cGVHD.
Abstract
Patient outcomes for steroid-dependent or -refractory chronic graft-versus-host diesease (cGVHD) are poor, and only ibrutinib has been US Food and Drug Administration (FDA) approved for this indication. cGVHD is often driven by the germinal center (GC) reaction, in which T follicular helper cells interact with GC B cells to produce antibodies that are associated with disease pathogenesis. The transcriptional corepressor B-cell lymphoma 6 (BCL6) is a member of the Broad-complex, Tramtrack, and Bric-abrac/poxvirus and zinc finger (BTB/POZ) transcription factor family and master regulator of the immune cells in the GC reaction. We demonstrate that BCL6 expression in both donor T cells and B cells is necessary for cGVHD development, pointing to BCL6 as a therapeutic cGVHD target. A small-molecule BCL6 inhibitor reversed active cGVHD in a mouse model of multiorgan system injury with bronchiolitis obliterans associated with a robust GC reaction, but not in cGVHD mice with scleroderma as the prominent manifestation. For cGVHD patients with antibody-driven cGVHD, targeting of BCL6 represents a new approach with specificity for a master GC regulator that would extend the currently available second-line agents.
Visual Abstract
Introduction
Chronic graft-versus-host disease (cGVHD) is a leading cause of long-term morbidity and mortality after allogeneic hematopoietic stem cell transplantation.1 Animal models have allowed for greater understanding of the pathology of disease and have been instrumental in developing therapeutic interventions for patients. No 1 model faithfully recapitulates the entire range of clinical, pathophysiological, and immune-mediated events seen in human cGVHD; thus, several preclinical models have been developed to represent various patient characteristics including cGVHD with or without scleroderma (generally antibody mediated).2
For cGVHD without scleroderma, bone marrow (BM) with low T-cell doses given to conditioned allogeneic recipients can cause chronic T-cell–mediated antigenic stimulation and coordinated interactions of T follicular helper (TFH) cells, germinal center (GC) B cells, and T follicular regulatory (TFR) cells.3 The net effects are GC formation, plasma cell generation with antibody deposition, and subsequent lung, liver, and gut, but not skin, fibrosis with bronchiolitis obliterans (BO) as a prominent manifestation.3 This model simulates active cGVHD patients who have circulating TFH cells with an activated phenotype, increased CXCL13 indicative of TFH cells, and the capacity to promote B-cell maturation.4
The transcriptional repressor B-cell lymphoma 6 (BCL6) is a master regulator of GC reactions, essential for development and function of TFH, TFR, and GC B cells.5-10 BCL6 has unique roles in each cell type. BCL6 allows GC B cells undergoing somatic hypermutation and DNA double-stranded breaks during class-switch recombination to better tolerate this stress by suppressing DNA damage responses and checkpoint genes.11 BCL6 also regulates pathways in the B-cell receptor (BCR) and CD40 signal transduction cascades in mature B cells.12 In TFH cells, BCL6 represses promoters involved in T-cell function, specifically controlling cell migration and alternative cell-fate inhibition.13 Mice deficient in Bcl6 are unable to form GCs and therefore do not produce high-affinity antibodies.14
We assessed the requirement of BCL6 expression in both donor T cells and B cells, as sources of BM-derived GC and splenic-derived TFH precursors, respectively, in a murine BO cGVHD model.3 Furthermore, we used a small-molecule, peptidomemitic BCL6 inhibitor, 79-6, for treating established disease in both BO and sclerodermatous cGVHD models.
Study design
Mice and transplantation
C57BL/6 (B6; Charles River) and B10.BR mice were housed in a pathogen-free facility and used with institutional animal care committee approval. B6→B10.BR (BO cGVHD) and B10.D2→Balb/c (scleroderma cGVHD) models, including disease severity assessments, were used as described.3,15,16 For BO cGVHD, cyclophosphamide-treated (120 mg/kg per day, days −3 and −2), irradiated (8.3 Gy by radiograph, day −1) recipients received, on day 0, B6 T-cell–depleted (TCD) BM ± 0.75 × 105 purified splenic T cells. Where indicated, BM or splenic T cells from BCL6fl/fl × CD19-Cre or BCL6 knockout (KO) mice was compared with wild-type (WT) cells. For scleroderma cGVHD, irradiated (7 Gy, day −1) recipients received B10.D2 BM ± 1.8 × 106 CD4+ and 0.9 × 106 CD8+ T cells on day 0.
cGVHD analyses
Pulmonary function tests assessing cGVHD-associated BO were performed as described.3 Flow cytometry, fluorescent microscopy, trichrome staining,3,15 histopathology,17 and skin scoring for the scleroderma model18 were performed as described.
Results and discussion
BCL6 expression is required in both donor T and B cells for BO cGVHD
Given the importance of BCL6 in regulating GC reactions in response to foreign antigen exposure, we sought to determine whether BCL6 expression in donor T or B cells is required for the GC reactions in murine cGVHD. B10.BR mice were transplanted with WT BM and WT or BCL6 KO T cells. Recipients of KO T cells did not develop BO pulmonary dysfunction (Figure 1A) and had reduced GC B-cell and TFH cell frequencies (Figure 1B). Pulmonary macrophage infiltration along with antibody deposition in the lung results in pulmonary fibrosis and BO.19,20 Pulmonary collagen and immunoglobulin deposition were reduced in mice receiving BCL6 KO T cells (Figure 1C-D). These results agree with earlier findings that interleukin-21 (IL-21) KO and ICOS-KO donor T cells do not cause cGVHD,15 consistent with donor TFH support of GC formation as disease initiators.
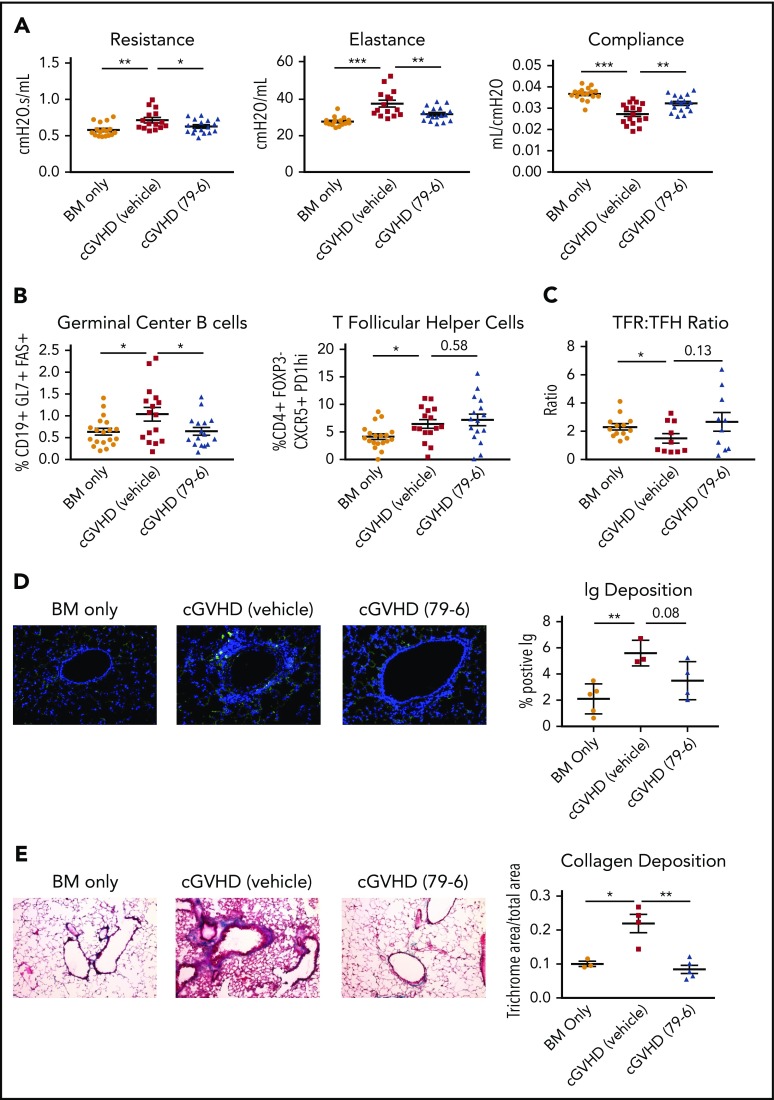
Figure 1.
BCL6 expression in donor T and B cells is necessary for development of BO cGVHD. B10.BR mice were conditioned with cyclophosphamide (120 mg/kg per day intraperitoneally) on days −3 and −2, and with irradiation (8.3 Gy by radiograph) on day −1 before transplantation. On day 0, mice were transplanted with either TCD B6 BM (BM only) or B6 TCD BM with purified B6 T cells (cGVHD) to establish cGVHD. (A-C) An additional group received TCD BM with BCL6 KO T. (A) Pulmonary function tests, measuring airway resistance, total lung elastance, and compliance were performed on day 56 after transplant. Mice receiving BCL6 KO T cells did not develop pulmonary dysfunction like that of the cGVHD controls. (B) Frequency of splenic GC B cells and TFH cells were analyzed by flow cytometry on day 56 posttransplant. GC B cells and TFH cell frequencies were decreased in the mice that received the BCL6 KO T cells. (C) Collagen deposition as identified by trichrome staining (original magnification ×20) of lung sections harvested day 56 posttransplant. Mice that received KO T cells had reduced collagen deposition in their lungs. (D) Immunoglobulin G (IgG) deposition in lungs of mice that received BCL6 KO T cells was significantly reduced (IgG in Fitc and Dapi; original magnification ×20). (E-H) Transplant was performed as described above with the addition of mice that received either TCD, BCL6fl/fl × CD19-Cre BM (BCL6 KO BM) alone or with purified B6 T cells. cGVHD was assessed as above. (E) Mice that received BCL6 KO BM with WT T cells did not develop pulmonary dysfunction. (F) Frequency of TFH and GC B cells from spleens of mice that received BCL6 KO BM with WT T cells was reduced. (G) Collagen deposition in the lung was decreased in mice that received the BCL6 KO BM with WT T cells (Masson‘s trichrome stain; original magnification ×20). (H) Deposition of IgG in lungs (IgG in Fitc and Dapi; original magnification ×20). Data from 1 experiment with 4 to 6 mice per group. An unpaired Student t test was used when comparing 2 groups. Error bars indicate standard error of the mean (SEM). Significance: *P < .05; **P < .01; ***P < .001.
To examine the role of BCL6 in donor BM-derived B cells, B10.BR mice were given WT or BCL6fl/fl × CD19-cre (BCL6 KO) BM plus or minus WT T cells. Mice receiving BCL6 KO BM and WT T cells did not develop pathogenic pulmonary dysfunction (Figure 1E). GC B-cell and TFH frequencies (Figure 1F) and lung collagen deposition (Figure 1G) were significantly decreased. In addition, there was a trend toward statistically reduced (P = .065) immunoglobulin deposition in the lungs (Figure 1H). These data indicate that BCL6 in T and B cells is required for development of BO cGVHD and that inhibiting either population could be beneficial for therapy.
A small-molecule inhibitor of BCL6 ameliorates established murine cGVHD with BO
The finding that BCL6 expression is necessary in donor cells for cGVHD development suggests that targeting BCL6 might be therapeutically advantageous. In diffuse large B-cell lymphomas (DLBCLs), BCL6 expression is often upregulated and the gene signature is similar to that of a GC B cell. Computer-aided drug design led to the development of compound 79-6, which binds to the Broad-complex, Tramtrack, and Bric-abrac (BTB) domain of BCL6, the site of corepressor (silencing mediator of retinoid or thyroid hormone receptors, nuclear receptor corepressor, BCL6 corepressor) binding critical for repressor activity and DLBCL cell survival.18 As expected by highly specific BCL6 vs non-BCL6 BTB domain binding, 79-6 kills DLBCL cells in vitro and in vivo.21
B10.BR mice received WT BM and WT T cells. On day 28, after cGVHD was established, mice were treated with 79-6 or vehicle. Mice treated with 79-6 had improved pulmonary function (Figure 2A). Treatment with 79-6 also resulted in significantly decreased splenic GC B-cell frequencies (Figure 2B), and plasma cell frequencies in the lung (data not shown). Correspondingly, there was a trend toward decreased immunoglobulin deposition in the lungs of mice treated with 79-6 (P = .08; Figure 2D). Pulmonary collagen deposition was less in 79-6–treated mice (Figure 2E). However, treatment did not result in the improvement in damage observed in liver and colon associated with disease, as assessed by histological scoring17 (Figure 2F). Splenic TFH frequencies were not significantly reduced in 79-6–treated mice, although TFR-to-TFH ratios were improved in some mice (Figure 2C). Thus, despite the dependency on BCL-6 expression for donor TFH and TFR generation,22 the major effect of 79-6 appears to be directly on the GC B cells in this model.
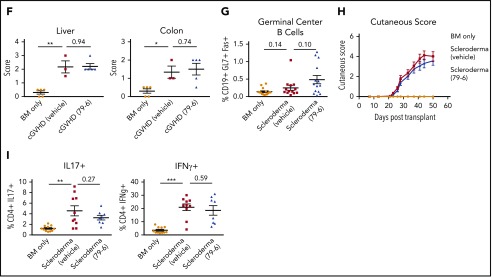
Figure 2.
Inhibition of BCL6 corepression activity can treat established BO cGVHD not sclerodermatous cGVHD. (A-E) BO cGVHD was established and assessed as described above. On days 28 to 56, mice were treated with vehicle or 50 mg/kg 79-6. (A) Pulmonary function test results demonstrate that 79-6 is able to improve pulmonary function compared with cGVHD control. (B-C) Frequency of GC B cells not TFH is decreased in mice treated with 79-6. (D) Immunoglobulin deposition in the lung had a trend toward being decreased (IgG in Fitc and Dapi; original magnification ×20). (E) Collagen deposition in the lung was reduced with 79-6 treatment (Masson's trichrome stain; original magnification ×20). (F) Histopathology scores of hemotoxylin and eosin–stained tissue sections demonstrated that 79-6 did not improve organ damage in liver and colon. (A-B) Data are pooled from 3 experiments, 4 to 7 mice per group per experiment. (C) Pooled from 2 experiments, 5 to 8 mice per group per experiment. (D-F) Representative from 1 experiment, 3 to 5 mice per group. (G-I) Sclerodermatous cGVHD was induced in Balb/c recipients treated on day 21 with irradiation (7 Gy by radiograph) and infusion of B10.D2 BM alone (BM only) or with B10.D2 purified T cells (scleroderma). With the appearance of significant difference in skin scores (∼day 21), mice began treatment with 50 mg/kg 79-6. (G) Analysis of frequency of GC B cells in spleens taken day 50 after transplant. Frequency of GC B cells in scleroderma mice demonstrates that the GC center is not a significant contributor to this disease. (H) Skin scores were determined twice weekly as described.18 Treatment with 79-6 was unable to improve skin manifestations of disease. (I) Cytokine analysis from pooled (2 mice per sample) lymph node harvested at day 50 after transplant. Cells were stimulated with phorbol myristate acetate (PMA)/ionomycin for 5 hours and then stained. 79-6 treatment did not change the frequencies of IL-17A+ or IFNg+ CD4+ cells from that of scleroderma controls. (G-I) Data are pooled from 2 experiments, 7 to 10 mice per group per experiment. The unpaired Student t test was used when comparing 2 groups. Error bars indicate SEM. Significance: *P < .05; **P < .01; ***P < .001.
To assess 79-6 efficacy in sclerodermatous cGVHD, BALB/c recipients were given B10.D2 BM ± 2.7 × 106 T cells.16,23 The systemic and cutaneous inflammatory response caused by pSTAT3+CD4+ T cells results in a Th1-dependent and Th17-associated sclerodermatous cGVHD.23 The skin is infiltrated with transforming growth factor β1 (TGF-β1)-expressing mononuclear cells, elevated C-C chemokines, and macrophage infiltration.16 Our findings indicate that a GC reaction appears unnecessary (Figure 1G) for cGVHD development in this model, agreeing with recent findings.24 No improvement in outcome, as determined by cutaneous score, was seen following 79-6 (Figure 2H). IL-17A and interferon-γ (IFNγ), significant contributors to skin cGVHD in this model, remained increased in treated mice (Figure 2I).
In this study, we have shown that mice receiving BCL6 KO T or B cells failed to develop BO cGVHD. Importantly, a small-molecule BCL6 inhibitor (79-6) was effective in limiting cGVHD in this GC-dependent model. In contrast, in a cytokine-dependent scleroderma model with systemic and cutaneous inflammation, 79-6 was ineffective in treating cGVHD. This provides support that BCL6 inhibition could be a novel treatment or prophylaxis for cGVHD and potentially other immune diseases in which the GC has a role.
Acknowledgments
The authors thank Marc Jenkins for providing BCL6 KO cells used for T-cell studies.
This work was supported by National Institutes of Health grants P01-CA142106 (National Cancer Institute), PO1 AI 056299 (National Institute of Allergy and Infectious Diseases), and T32 CA009138 (National Cancer Institute) as well as grants from the Leukemia & Lymphoma Society of America (Translational Research Grants 6458-15 and 6462-15).
Footnotes
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: K.P. designed experiments and wrote the paper; R.F. designed and performed experiments, discussed results, and edited the paper; J.Q. and J.E.B. were responsible for 79-6 synthesis; J.D., L.L., I.M., K.P.M., G.R.H., J.S.S., W.J.M., P.T.S., A.H.S, D.M., C.S.C., J.K., J.H.A., R.J.S., and J.R. discussed results and edited the paper; A.M.M. provided BCL6fl/fl × CD19-cre BM; and B.R.B. contributed to experimental design, discussed results, and edited the paper.
Conflict-of-interest disclosure: J.E.B. is now an executive and shareholder of Novartis AG, and has been a founder and shareholder of SHAPE (acquired by Medivir), Acetylon (acquired by Celgene), Tensha (acquired by Roche), Syros, Regency, and C4 Therapeutics. The remaining authors declare no competing financial interests.
Correspondence: Bruce R. Blazar, University of Minnesota, MMC 109, 420 Delaware St SE, Minneapolis, MN 55455; e-mail: blazar001@umn.edu.
REFERENCES
- 1.Arai S, Jagasia M, Storer B, et al. . Global and organ-specific chronic graft-versus-host disease severity according to the 2005 NIH Consensus Criteria. Blood. 2011;118(15):4242-4249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zeiser R, Blazar BR. Pathophysiology of chronic graft-versus-host disease and therapeutic targets. N Engl J Med. 2017;377(26):2565-2579. [DOI] [PubMed] [Google Scholar]
- 3.Srinivasan M, Flynn R, Price A, et al. . Donor B-cell alloantibody deposition and germinal center formation are required for the development of murine chronic GVHD and bronchiolitis obliterans. Blood. 2012;119(6):1570-1580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Forcade E, Kim HT, Cutler C, et al. . Circulating T follicular helper cells with increased function during chronic graft-versus-host disease. Blood. 2016;127(20):2489-2497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dent AL, Shaffer AL, Yu X, Allman D, Staudt LM. Control of inflammation, cytokine expression, and germinal center formation by BCL-6. Science. 1997;276(5312):589-592. [DOI] [PubMed] [Google Scholar]
- 6.Fukuda T, Yoshida T, Okada S, et al. . Disruption of the Bcl6 gene results in an impaired germinal center formation. J Exp Med. 1997;186(3):439-448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ye BH, Cattoretti G, Shen Q, et al. . The BCL-6 proto-oncogene controls germinal-centre formation and Th2-type inflammation. Nat Genet. 1997;16(2):161-170. [DOI] [PubMed] [Google Scholar]
- 8.Nurieva RI, Chung Y, Martinez GJ, et al. . Bcl6 mediates the development of T follicular helper cells. Science. 2009;325(5943):1001-1005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yu D, Rao S, Tsai LM, et al. . The transcriptional repressor Bcl-6 directs T follicular helper cell lineage commitment. Immunity. 2009;31(3):457-468. [DOI] [PubMed] [Google Scholar]
- 10.Wu H, Chen Y, Liu H, et al. . Follicular regulatory T cells repress cytokine production by follicular helper T cells and optimize IgG responses in mice. Eur J Immunol. 2016;46(5):1152-1161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hatzi K, Melnick A. Breaking bad in the germinal center: how deregulation of BCL6 contributes to lymphomagenesis. Trends Mol Med. 2014;20(6):343-352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Basso K, Dalla-Favera R. Roles of BCL6 in normal and transformed germinal center B cells. Immunol Rev. 2012;247(1):172-183. [DOI] [PubMed] [Google Scholar]
- 13.Hatzi K, Nance JP, Kroenke MA, et al. . BCL6 orchestrates Tfh cell differentiation via multiple distinct mechanisms. J Exp Med. 2015;212(4):539-553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Huang C, Melnick A. Mechanisms of action of BCL6 during germinal center B cell development. Sci China Life Sci. 2015;58(12):1226-1232. [DOI] [PubMed] [Google Scholar]
- 15.Flynn R, Du J, Veenstra RG, et al. . Increased T follicular helper cells and germinal center B cells are required for cGVHD and bronchiolitis obliterans. Blood. 2014;123(25):3988-3998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang Y, McCormick LL, Desai SR, Wu C, Gilliam AC. Murine sclerodermatous graft-versus-host disease, a model for human scleroderma: cutaneous cytokines, chemokines, and immune cell activation. J Immunol. 2002;168(6):3088-3098. [DOI] [PubMed] [Google Scholar]
- 17.Blazar BR, Taylor PA, McElmurry R, et al. . Engraftment of severe combined immune deficient mice receiving allogeneic bone marrow via In utero or postnatal transfer. Blood. 1998;92(10):3949-3959. [PubMed] [Google Scholar]
- 18.Le Huu D, Matsushita T, Jin G, et al. . IL-6 blockade attenuates the development of murine sclerodermatous chronic graft-versus-host disease. J Invest Dermatol. 2012;132(12):2752-2761. [DOI] [PubMed] [Google Scholar]
- 19.Du J, Paz K, Flynn R, et al. . Pirfenidone ameliorates murine chronic GVHD through inhibition of macrophage infiltration and TGF-β production. Blood. 2017;129(18):2570-2580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Alexander KA, Flynn R, Lineburg KE, et al. . CSF-1-dependant donor-derived macrophages mediate chronic graft-versus-host disease. J Clin Invest. 2014;124(10):4266-4280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cerchietti LC, Ghetu AF, Zhu X, et al. . A small-molecule inhibitor of BCL6 kills DLBCL cells in vitro and in vivo. Cancer Cell. 2010;17(4):400-411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fu W, Liu X, Lin X, et al. . Deficiency in T follicular regulatory cells promotes autoimmunity. J Exp Med. 2018;215(3):815-825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Radojcic V, Pletneva MA, Yen HR, et al. . STAT3 signaling in CD4+ T cells is critical for the pathogenesis of chronic sclerodermatous graft-versus-host disease in a murine model. J Immunol. 2010;184(2):764-774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Deng R, Hurtz C, Song Q, et al. . Extrafollicular CD4+ T-B interactions are sufficient for inducing autoimmune-like chronic graft-versus-host disease. Nat Commun. 2017;8(1):978. [DOI] [PMC free article] [PubMed] [Google Scholar]