Abstract
Cytochrome P4502E1 (CYP2E1) is involved in the biotransformation of several low molecular weight chemicals and plays an important role in the metabolic activation of carcinogens and hepatotoxins such as CCl4. Induction of CYP2E1 is exerted mainly at posttranscriptional levels through mRNA and protein stabilization, and there is little evidence of xenobiotic induction at the transcriptional level. Previously, we reported microarray analysis data suggesting a decrease in Cyp2e1 gene expression on Ahr-null livers when compared to wild-type mouse livers.
The goal of the present study was to determine whether 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) increased mouse CYP2E1 levels in an AhR-dependent manner and the impact on CCl4-induced hepatotoxicity.
TCDD treatment induced CYP2E1 mRNA and protein levels in mouse liver, and this effect was aryl hydrocarbon receptor (AhR)-dependent.
Moreover, TCDD pre-treatment increased the CCl4-induced alanine aminotransferase (ALT) activity, the extent of CCl4-induced necrosis, and the number of sinusoidal cells in wild-type animals, while this potentiating effect was not observed in Ahr-null mice.
In conclusion, this study revealed that TCDD, probably in an AhR-dependent manner, exacerbated CCl4-induced hepatotoxicity through induction of CYP2E1.
Keywords: TCDD, AhR, CYP2E1, CCl4
Introduction
Cytochrome P4502E1 (CYP2E1) is involved in the biotransformation of several low molecular weight chemicals, many of which are organic solvents. This enzyme has been extensively studied because it plays an important role in the metabolic activation of precarcinogens, such as benzene and nitrosamines, as well as hepatotoxins, including carbon tetrachloride (CCl4) and acetaminophen (Wong et al., 1998; Cheung et al., 2005). Several endogenous CYP2E1 substrates also have been identified, including lipid peroxidation products, ketones, and fatty acids; and it was suggested that they play a role in gluconeogenesis (Lieber, 2004).
CYP2E1 is constitutively expressed in the liver and several other tissues. CYP2E1 induction by various chemicals is exerted mainly at post-transcriptional level, and there is little evidence of xenobiotic induction at the transcriptional level. Mouse, rat, and human CYP2E1 transcription is mainly under the control of hepatocyte nuclear factor 1α (HNF1α) (Cheung et al., 2003). Other studies have shown that mouse CYP2E1 expression is also regulated by β-catenin (Sekine et al., 2006) and leptine (Leclercq et al., 2000). On the other hand, aryl hydrocarbon receptor (AhR) ligands such as 3-methylcholanthrene, β-naphthoflavone and pyridine increase CYP2E1 transcription and activity (Anderson and Angel, 1980; Mostafa et al., 1981; Kim et al., 1988; Sinal et al., 1999; Iba et al., 2000), suggesting a role for this transcription factor in the regulation of CYP2E1 expression. More recently, it was reported that TCDD induce CYP2E1 expression in H9c2 cells (Zordoky and El-Kadi, 2010).
AhR, a member of the basic helix-loop-helix-Per-Arnt-Sim (bHLH-PAS) transcription factor family, is a ligand-activated receptor that mediates the toxicity of environmental pollutants, such as 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) (Okey et al., 1994; Whitlock, 1999). Upon binding to TCDD, AhR translocate to the nucleus, dimerises with AhR nuclear-translocator protein (ARNT), binds dioxin-responsive elements (DREs, also known as xenobiotic-responsive elements or XREs), and upregulates the expression of a battery of genes encoding xenobiotic-metabolizing enzymes, such as cytochrome P450s (e.g. CYP1A1, CYP1A2, CYP1B1), NAD(P)H quinone oxidoreductase, and a form of UDP-glucoronosyl-transferase-6 (Gonzalez and Fernandez-Salguero, 1998). Although AhR may function as part of an adaptive chemical response, several studies have suggested that this transcription factor could have important functions in liver and cardiac development, cell proliferation, circadian rhythmicity, cholesterol and glucose metabolism (Fernandez-Salguero et al., 1996; Gonzalez and Fernandez-Salguero, 1998; Elizondo et al., 2000; Sato et al., 2008), as well as in the ubiquitin-proteasome system (Reyes-Hernandez et al., 2010). Previously, we reported microarray analysis data suggesting that several genes encoding for metabolizing enzymes are under AhR regulation (Reyes-Hernandez et al., 2010). Several years ago, some of these were identified as part of the AhR battery, including cyp1a1 and cyp1a2. The microarray data also indicated that akr1c13, akr1e1, cyp39a1, cyp4a12a and cyp2e1 gene expression was down regulated in Ahr-null mouse livers when compared to wild-type livers.
Given the role of CYP2E1 in the metabolism of several toxins and carcinogens, the effect of xenobiotics on CYP2E1 liver expression is of significant interest. Therefore, the goal of the present study was to determine whether TCDD increased mouse CYP2E1 levels in an AhR-dependent manner and the impact on CCl4-induced hepatotoxicity.
Materials and methods
Materials
TCDD (99.1% purity) was purchased from AccuStandard (New Haven, CT), carbon tetrachloride (CCl4, 99.9% purity) was purchased from J.T. Baker (Austin, TX), Tris-HCl and sodium dodecylsulfate were purchased from Invitrogen (Camarillo, CA), Tween 20 and Nonidet P-40 were purchased from Sigma-Aldrich (St. Louis, MO), and glycine was purchased from Usbio (Marblehead, MA).
Animals
The Ahr-null mice (C57BL/6N) described previously (Fernandez-Salguero et al., 1995) were genotyped by PCR as reported elsewhere (Rodriguez-Sosa et al., 2005). Wild-type (WT) littermates were used as control mice. Animals were randomly distributed (n = 5), housed in a pathogen-free facility, fed with autoclaved Purina rodent chow (St. Louis, MO), and provided with water ad libitum. All animal studies were performed according to the Guide for the Care and Use of Laboratory Animals, as adopted and enforced by the US National Institutes of Health and the Mexican Regulation of Animal Care and Maintenance (NOM-062-ZOO-1999, 2001).
Treatments
Three treatment groups were formed (male mice, aged 6–8 weeks, n = 5). Group 1 mice were given a single intraperitoneal (i.p.) dose of TCDD at 80 μg/kg body weight and were sacrificed 72 h later. Group 2 mice were given a single i.p. dose of CCl4 at 1.59 g/kg body weight and were sacrificed 24 h later. Group 3 mice were given a single i.p. dose of TCDD at 80 μg/kg body weight and 48 h later were given a single i.p. dose of CCl4 at 1.59 g/kg; the mice were sacrificed 24 h later. Controls for each group were injected with corn oil vehicle only. After treatments, mice were euthanized by cervical dislocation, and livers were removed, frozen in liquid nitrogen, and stored at −70°C. For histopathology analysis, representative liver fragments were fixed with 10% formaldehyde in phosphate-buffered saline, pH 7.2–7.4.
Real-time quantitative PCR (rtPCR) analysis
Briefly, total RNA was prepared from mouse liver using the TRIzol reagent according to the manufacturer’s instructions (Invitrogen; Camarillo, CA). RNA was quantified spectrophotometrically at OD260. Subsequently, RNA integrity was evaluated by electrophoresis of RNA samples on 1% agarose gels. cDNA for the rtPCR assay was prepared from 2 μg of total RNA using the SuperScript First-Strand Synthesis reagent (Invitrogen; Camarillo, CA) and oligo dT (Invitrogen; Camarillo, CA). PCR experiments were conducted using a Step One Real Time PCR System (Applied Biosystems; Branchburg, NJ) and analysed using the comparative threshold cycle (Ct) method, as described previously (Medina-Diaz and Elizondo, 2005). The mRNAs encoding CYP2E1 or 18S ribosomal RNA (rRNA, endogenous) were amplified in a single PCR experiment to allow for normalization of the mRNA data. The PCR mixture contained 2 μl of cDNA, 0.9 μM primers, and 0.25 μM probes in 1X TaqMan Universal PCR Master Mix (Applied Biosystems; Foster City, CA). The primer and probe sequences used for CYP2E1 were as follows: 5′-CCAAAGAGAGGCACACTTCCT-3′ (forward), 5′-GCCAATCAGAAAGGTAGGGTCAAAA-3′ (reverse), and CTGGCCTTTGGTCTTTT (FAM probe). The primers and probe for CYP1A1 were: 5′- GCCTTCATTCTGGAGACCTTCC-3′ (forward), 5′-CAATGGTCTCTCCGATGC-3′ (reverse), and CAGGACATTTGAGAAGGGCCACATC (FAM probe).
In silico analysis
The mouse cyp2e1 gene promoter was analysed using the following web-based bioinformatic tools: Genomatix (http://www.genomatix.de/cgi-bin/matinspector_prof/mat_fam.pl?s=eabd9a3f188f5b276aaa00a3445cdcbe, 2009) and CBRC (http://www.cbrc.jp/research/db/TFSEARCH.html, 2009).
Western blot analysis
Liver fragments were homogenised in buffer containing 10 mM Tris-HCl (pH 7.4), 1% Nonidet P-40, 10 μg/ml aprotinin, 10 μg/ml leupeptin, 10 μg/ml soybean trypsin inhibitor, and 1 mM PMSF. Protein concentrations were determined using the Bradford reaction (BioRad; Hercules, CA). Aliquots (20 μg) were solubilised in sample buffer [60 mM Tris-HCl, pH 6.8; 2% sodium dodecylsulfate (SDS); 20% glycerol; 2% mercaptoethanol; 0.001% bromophenol blue] and subjected to 10% SDS-polyacrylamide gel electrophoresis. Protein extracts were transferred to a nitrocellulose membrane using a mini trans-blot (Bio-Rad; Hercules, CA). The transfer was performed at a constant voltage of 80 V for 2 h in transfer buffer (48 mM Tris-HCl, 39 mM glycine, pH 8.3; 20% methanol). Following the transfer, membranes were blocked overnight at 4°C in the presence of 2% nonfat dry milk and 0.5% bovine serum albumin (BSA) in blocking buffer (25 mM Tris-HCl, pH 7.5; 150 mM NaCl) and subsequently incubated at 4°C for 3 h with goat polyclonal anti-CYP2E1 (1:10,000; Abcam; Cambridge, MA), or actin (1:5000; Zymed; San Francisco, CA) diluted in buffer (25 mM Tris-HCl, pH 7.5; 150 mM NaCl; 0.1% Tween 20; 0.05% nonfat dry milk; 0.05% BSA). After washing, the membranes were incubated with the corresponding horseradish peroxidase (HRP)-conjugated secondary antibody, HRP-rabbit anti-goat IgG (1:10,000; Pierce; Rockford, IL), for 2 h at 4°C. The membrane was washed, and the immunoreactive protein was detected using an ECL western blotting detection kit (Amersham; Arlington Heights, IL). The integrated optical density of the bands was quantified using scanning densitometry (GS-800 Calibrated Densitometer; BioRad; Hercules, CA).
Determination of serum aminotransferase activities
Serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities were determined enzymatically by the rate of NADPH oxidation using Randox reagents according to the manufacturer’s instructions (Randox; Antrim, UK).
Liver histology
For each treatment group, a representative section was excised from the liver. Tissues were fixed in 10% formaldehyde in phosphate-buffered saline for 48 h, washed in water, dehydrated in alcohol, and embedded in paraffin. Liver sections (5 μm in thickness) were stained with hematoxylin-eosin (H&E) for histological examination under a light microscope (Nikon H550L).
Statistical analysis
Results are presented as the mean values ± the standard deviation (SD) or the standard error (SE). The statistical significance of the data was evaluated using the Mann–Whitney’s U–Wilcoxon rank or the Student’s t-test, as appropriate. In all cases, the differences between animal groups were considered to be statistically significant when the p value was less than 0.05.
Results
Recently, DNA microarray analysis revealed a decrease in CYP2E1 expression on Ahr-null livers when compared to wild-type mouse livers (Reyes-Hernandez et al., 2010) (Table 1). In the present study, we determined whether Ahr plays a role in the modulation of CYP2E1 expression levels. In order to measure the efficacy of TCDD treatment, CYP1A1 mRNA levels were determined. As expected, an induction of CYP1A1 expression was observed only in the wild-type mouse (Figure 1A). No differences were observed on basal CYP2E1 mRNA levels, between wild-type and Ahr-null mice. However, TCDD treatment resulted in a five-fold induction of CYP2E1 mRNA levels only in the wild-type mice (Figure 1B). These data suggest that the induction of cyp2e1 gene expression after TCDD treatment is Ahr-dependent. In order to identify putative XREs in the mouse cyp2e1 gene promoter, in silico analysis was performed. Common regulatory sequences, such as the TATA box, HNF-3b, GATA-2, Oct-1, AP1, and C/ EBP, were located between position-2046 and the putative translation start site. However, no XREs were identified (data not shown).
Table 1. Basal gene expression in Ahr-null mice livers as compared to wild-type livers*.
Metabolism of xenobiotics – Mus musculus (mouse).
| Accession No. | Symbol | Gene name | Z score** |
|---|---|---|---|
| Phase I | |||
| NM_010750 | cyp1a1 | cytochrome P450, family 1, subfamily a, polypeptide 1 | −2.248441 |
| NM_010759 | cyp1a2 | cytochrome P450, family 1, subfamily a, polypeptide 2 | −1.873759 |
| NM_023371 | cyp2e1 | cytochrome P450, family 2, subfamily e, polypeptide 1 | −1.854343 |
| NM_018887 | cyp39a1 | cytochrome P450, family 39, subfamily a, polypeptide 1 | −2.076368 |
| Z12223 | cyp4a12a | cytochrome P450, family 4, subfamily a, polypeptide 12a | −1.705495 |
| NM_013777 | akr1c13 | aldo-keto reductase family 1, member c13 | −2.625138 |
| NM_018859 | akr1e1 | aldo-keto reductase family 1, member e1 | −2.025381 |
| Phase II | |||
| NM_008487 | gstm2 | glutathione S-transferase, mu 2 | −2.585971 |
| NM_008510 | gstt1 | glutathione S-transferase, theta 1 | −3.387056 |
| NM_013873 | sult4a1 | sulfotransferase family 4a, member 1 | −2.071361 |
Figure 1.
TCDD increases CYP2E1 mRNA levels. Wild-type or Ahr-null mice were treated with TCDD (80 μg/kg) or vehicle. After 72 h, liver samples were removed and the relative expression of CYP1A1 (A) or CYP2E1 (B) mRNA levels were determined. mRNA levels were normalised with 18S ribosomal RNA. Data represent mean values ± SD. *p < 0.05.
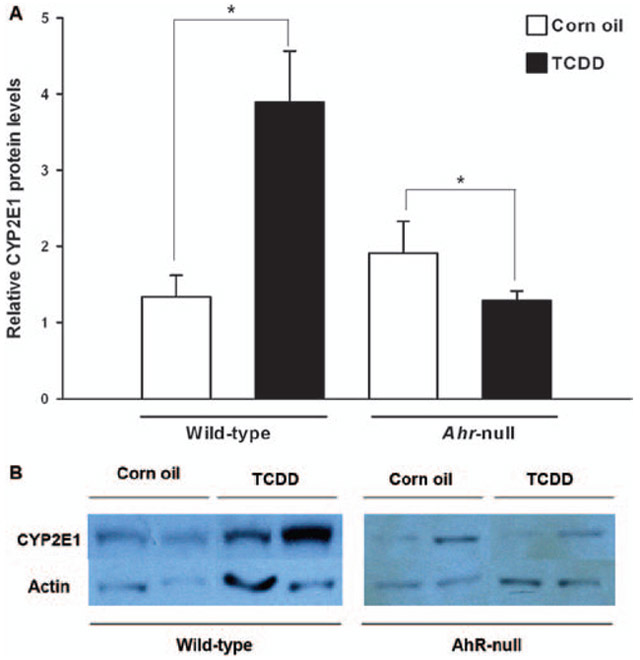
Next, we examined whether the induction of CYP2E1 mRNA causes increased protein levels. After TCDD treatment, a four-fold induction of CYP2E1 protein levels was observed in wild-type mice. In contrast, this effect was not detected in the livers of Ahr-null mice. Moreover, TCDD treatment resulted in a light but significant decrease of the immunoreactive CYP2E1 protein level in Ahr-null livers (Figure 2). In addition, TCDD treatment is known to result in typical wasting syndrome and loss of body weight, which may cause increased CYP2E1 expression due to the accumulation of ketone bodies. Therefore, the body weight of the animals was monitored throughout TCDD treatment. No change was observed during the study, suggesting that the observed induction of CYP2E1 mRNA might be a direct result of AhR activation (Figure 3).
Figure 2.
Effect of TCDD on CYP2E1 protein levels. Wild-type or Ahr-null mice were treated with TCDD (80 μg/kg) or vehicle. After 72 h, liver samples were removed and CYP2E1 protein levels were determined. (A) Relative values for CYP2E1 protein signals were normalised with actin. (B) Representative immuno-blot image. Data represent mean values ± SE. *p < 0.05.
Figure 3.

Body weights during TCDD treatment. Both wild-type and Ahr-null mice were treated with TCDD (80 μg/kg) as described in the Materials and Methods section, and the body weight was evaluated every day for 4 days. Data represent mean values ± SE from five animals of each genotype.
It is well established that CYP2E1 mediates the toxicity of several agents, such as acetaminophen and CCl4. Therefore, we explored whether TCDD pretreatment magnifies the CCl4 toxicity. Initially, TCDD hepatotoxicity was determined. In both wild-type and Ahr-null mice, increased serum aminotransaminase activities were observed after TCDD treatment. For both ALT and AST, the increased activity was higher in wild-type mice compared to Ahr-null mice (Figure 4). Histopathological analysis showed that after TCDD treatment, the wild-type liver presents pyknotic cells and mild necrosis. In contrast, Ahr-null mice did not exhibit apparent histopathological changes (Figure 5). Next, the effect of TCDD pretreatment on CCl4 hepatotoxicity was determined (Figure 6). Treatment with CCl4 alone resulted in marked increases of serum ALT and AST activities in both wild-type and Ahr-null mice. There were approximately 800- and 113-fold increases in ALT and AST activities, respectively, in wild-type mice when compared to their corn oil-treated counterparts (Figure 4). In contrast, after CCl4 treatment, Ahr-null mice presented lower increases in ALT and AST activities (580-fold and 101-fold, respectively). In addition, TCDD pretreatment increased the CCl4-induced ALT activity by 1643-fold in wild-type animals compared to the corn oil control group, while this potentiating effect was not observed in Ahr-null mice. Although TCDD pretreatment increased CCl4-induced AST activity in wild-type mice, this effect was slightly significant. No differences were observed in Ahr-null animals (Figure 6).
Figure 4.
Serum aminotransaminase activity after TCDD treatment. Wild-type or Ahr-null mice were treated with TCDD (80 μg/kg) or vehicle. After 72 h, serum levels of AST (A) or ALT (B) were determined. Data represent mean values ± SE. *p < 0.05.
Figure 5.

Histopathological analysis of livers of mice treated with TCDD. Representative H&E-stained liver sections are shown from wild-type and Ahr-null mice treated with corn oil or TCDD (80 μg/kg) for 72 h.
Figure 6.
Aminotransaminase activity after CCl4 and TCDD treatments. Wild-type or Ahr-null mice were treated with CCl4 (1.59 g/kg) for 24 h, or co-treated with TCDD (80 μg/kg) and CCl4 (1.59 g/kg) as indicated in Materials and methods section. After treatments, serum levels of AST (A) or ALT (B) were determined. Data represent mean values ± SE. *p < 0.05. Wildtype-CCl4 vs. AhR-null-CCl4, §p < 0.05.
It is known that CCl4 induces hepatoxicity and is characterized by centrilobular necrosis. Histophatological evaluation of liver sections from wild-type mice treated with CCl4 showed severe centrilobular necrosis when compared to corn oil-treated wild-type animals (Figures 5 and 7). Pretreatment with TCDD increased the extent of CCl4-induced necrosis and the number of sinusoidal cells. Although necrosis was observed in liver sections from Ahr-null mice treated with CCl4, the extent of the damage was lower when compared to wild-type mice. Pretreatment with TCDD did not potentiate the CCl4-induced necrosis or produce altered nuclei and sinusoidal damage in Ahr-null liver samples (Figure 7).
Figure 7.

Histopathological analysis of livers of mice after TCDD and CCl4 treatments. Representative H&E-stained liver sections are shown from wild-type and Ahr-null mice treated with CCl4 (1.59 g/kg) for 24 h, or co-treated with TCDD (80 μg/kg) and CCl4 (1.59 g/kg) as indicated in Materials and methods section.
Discussion
Transcriptional induction of CYP2E1 is complex and not well understood. The present study shows that AhR activation by TCDD enhanced CCl4-induced hepatotoxicity, at least in part, by increasing CYP2E1 mRNA and protein levels. Previous reports have suggested that AhR mediates CYP2E1 expression. In addition, β-naphthoflavone treatment has been shown to increase the CYP2E1 transcript levels in rat liver, lung, heart, and kidney (Sinal et al., 1999). On the other hand, pyridine, a major constituent of tobacco smoke induces CYP2E1 levels (Kim et al., 1988). This agent also induces cyp1a1 gene expression in an AhR-dependent manner (Iba et al., 2000).
Similar to the present report, hematotoxicity induced by benzene, a CYP2E1 substrate, is mediated by AhR. Moreover, benzene treatment increases CYP2E1 expression in wild-type but not in Ahr-null mouse livers (Yoon et al., 2002). These reports together with the present data suggest that AhR participates in CYP2E1 induction.
However, in silico analysis has failed to identify XREs at the cyp2e1 gene promoter. While the presence of a nonconsensus XREs cannot rule out, it is likely that AhR acts through a novel, XRE-independent mechanism. Indeed, several studies have demonstrated that the cyp2e1 gene transcription control is under control by transcription factors such as HNF1α (Cheung et al., 2003). On the other hand, a 24-h rhythm of CYP2E1 protein and mRNA levels in mouse liver has also been reported (Matsunaga et al., 2008). The same authors have already elucidated the nature of this daily variation. Transcription of cyp2e1 gene is rhythmically controlled by HNF1α. This activation is negatively regulated by CRY1, a component of the circadian timing system, through its interaction with HNF1α transcriptional complexes. More recently, others demonstrated that AhR activation disrupts CLOCK-BMAL1 transcriptional activity (Xu et al., 2010), which positively regulates cry1 gene expression (Gekakis et al., 1998). The latter data allows us to hypothesize that the TCDD effect on CYP2E1 expression, observed in the present study, is the result of CLOCK-BMAL1 complex disruption leading to decreased cry1 expression. Therefore, induction of CYP2E1 mRNA and protein levels occurs. It also cannot be ruled out that other transcription factors control cyp2e1 gene expression, such as β-catenin (Sekine et al., 2006). Recently, an interaction between the AhR and the β-catenin pathway has been shown (Braeuning et al., 2011; Prochazkova et al., 2011).
Alternatively, TCDD may modify CYP2E1 mRNA levels at the post-transcriptional level. It was reported that TCDD increases stabilization of several mRNAs. In particular, TCDD induces transforming growth factor α and urokinase plasminogen activator mRNA stabilization in the human keratinocyte cell line SCC-12F (Gaido et al., 1992; Gaido and Maness, 1995). Moreover, Henley and collaborators provided evidence that AhR is involved in TCDD-stimulated IL-1β expression through a posttranscriptional mechanism, specifically by increasing the steady state of the mRNA (Henley et al., 2004). According to a genome-wide analysis of nuclear and cytoplasmic RNA levels, the impact of TCDD-activated AhR at the posttranscriptional level includes tens, maybe hundreds, of genes (Schwanekamp et al., 2006).
Regardless of the level of control, AhR activation by TCDD resulted in increased CYP2E1 protein levels and, therefore, a potential increase in CYP2E1 activity. To assess this last possibility, the role of TCDD-activated AhR on CCl4 toxicity was investigated. Although the magnitude of the change was low compared to other hepatotoxins, TCDD treatment increased AST and ALT activities in wild-type serum mice (2.6- and 2.2-fold induction, respectively). Surprisingly, similar results were observed, but to a lesser extent in Ahr-null samples. This data suggest that there are AhR-independent pathways for the toxic actions of TCDD. Consistent with this idea, Yoshioka and collaborators recently reported antiproliferative effects of TCDD in an AhR-independent way (Yoshioka et al., 2012). In agreement with previous studies (Fernandez-Salguero et al., 1996; Smith et al., 2001), liver histology showed that TCDD treatment resulted in necrosis and pycnotic nuclei, suggesting the presence of apoptotic cells in wild-type mice. In contrast, livers from Ahr-null mice were unaffected. As expected, CCl4 treatment resulted in increased AST and ALT activities in both wild-type and Ahr-null serum. However, the serum ALT activity was potentiated by TCDD pretreatment by two-fold only in wild-type livers. In agreement with this observation, histopathology analysis showed that CCl4 toxicity was exacerbated by TCDD treatment. These results suggest that AhR activation intensified the CCl4 hepatotoxicity by increasing the CYP2E1 levels. Thus, exposure to AhR ligands may alter the capacity of organisms to metabolize CCl4 into reactive metabolites that account for severe toxicity. The latter applies for all toxic compounds that are metabolized by CYP2E1, such as benzene. On the other hand, it has been shown that co-treatment with TCDD and CCl4 results in the loss of CYP1A2 accompanied with a possible generation of oxidative species (Kuroki et al., 1986; De Matteis et al., 1991). This may contribute to the hepatotoxicity observed in the present study.
CYP2E1 metabolizes benzene into metabolites that induce hematopoietic toxicity, aplastic anemia, and leukemia (Cronkite et al., 1984; Farris et al., 1997). Similar to the present study, Yoon and collaborators (Yoon et al., 2002) reported that Ahr-null mice are less susceptible to benzene toxicity than wild-type animals. Moreover, benzene treatment results in induction of CYP2E1 expression in wild-type mice, while no significant changes were observed in Ahr-null mouse livers.
Evidence from other laboratories and the present results suggest that AhR activation induces CYP2E1 levels. However, it is not clear whether the underlying molecular mechanism is at the transcriptional or posttranscriptional level or both.
In conclusion, the present study showed that TCDD, probably in an AhR-dependent manner, exacerbated CCl4-induced hepatotoxicity by inducing CYP2E1 levels. Future studies are necessary to elucidate the molecular mechanism whereby AhR mediates CYP2E1 induction.
Acknowledgments
Declaration of interest
This work was supported by CONACYT grant 24275.
References
- Anderson LM, Angel M. (1980). Induction of dimethylnitrosamine demethylase activity in mouse liver by polychlorinated biphenyls and 3-methylcholanthrene. Biochem Pharmacol 29:1375–1383. [DOI] [PubMed] [Google Scholar]
- Braeuning A, Köhle C, Buchmann A, Schwarz M. (2011). Coordinate regulation of cytochrome P450 1a1 expression in mouse liver by the aryl hydrocarbon receptor and the beta-catenin pathway. Toxicol Sci 122:16–25. [DOI] [PubMed] [Google Scholar]
- Cheadle C, Vawter MP, Freed WJ, Becker KG. (2003). Analysis of microarray data using Z score transformation. J Mol Diagn 5:73–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheung C, Akiyama TE, Kudo G, Gonzalez FJ. (2003). Hepatic expression of cytochrome P450s in hepatocyte nuclear factor 1-alpha (HNF1alpha)-deficient mice. Biochem Pharmacol 66:2011–2020. [DOI] [PubMed] [Google Scholar]
- Cheung C, Yu AM, Ward JM, Krausz KW, Akiyama TE, Feigenbaum L, Gonzalez FJ. (2005). The cyp2e1-humanized transgenic mouse: role of cyp2e1 in acetaminophen hepatotoxicity. Drug Metab Dispos 33:449–457. [DOI] [PubMed] [Google Scholar]
- Cronkite EP, Bullis J, Inoue T, Drew RT. (1984). Benzene inhalation produces leukemia in mice. Toxicol Appl Pharmacol 75:358–361. [DOI] [PubMed] [Google Scholar]
- De Matteis F, Dawson SJ, Boobis AR, Comoglio A. (1991). Inducible bilirubin-degrading system of rat liver microsomes: role of cytochrome P450IA1. Mol Pharmacol 40:686–691. [PubMed] [Google Scholar]
- Elizondo G, Fernandez-Salguero P, Sheikh MS, Kim GY, Fornace AJ, Lee KS, Gonzalez FJ. (2000). Altered cell cycle control at the G(2)/M phases in aryl hydrocarbon receptor-null embryo fibroblast. Mol Pharmacol 57:1056–1063. [PubMed] [Google Scholar]
- Farris GM, Robinson SN, Gaido KW, Wong BA, Wong VA, Hahn WP, Shah RS. (1997). Benzene-induced hematotoxicity and bone marrow compensation in B6C3F1 mice. Fundam Appl Toxicol 36:119–129. [DOI] [PubMed] [Google Scholar]
- Fernandez-Salguero P, Pineau T, Hilbert DM, McPhail T, Lee SS, Kimura S, Nebert DW, Rudikoff S, Ward JM, Gonzalez FJ. (1995). Immune system impairment and hepatic fibrosis in mice lacking the dioxin-binding Ah receptor. Science 268:722–726. [DOI] [PubMed] [Google Scholar]
- Fernandez-Salguero PM, Hilbert DM, Rudikoff S, Ward JM, Gonzalez FJ. (1996). Aryl-hydrocarbon receptor-deficient mice are resistant to 2,3,7,8-tetrachlorodibenzo-p-dioxin-induced toxicity. Toxicol Appl Pharmacol 140:173–179. [DOI] [PubMed] [Google Scholar]
- Gaido KW, Maness SC. (1995). Post-transcriptional stabilization of urokinase plasminogen activator mRNA by 2,3,7,8-tetrachlorodibenzo-p-dioxin in a human keratinocyte cell line. Toxicol Appl Pharmacol 133:34–42. [DOI] [PubMed] [Google Scholar]
- Gaido KW, Maness SC, Leonard LS, Greenlee WF. (1992). 2,3,7,8-Tetrachlorodibenzo-p-dioxin-dependent regulation of transforming growth factors-alpha and -beta 2 expression in a human keratinocyte cell line involves both transcriptional and post-transcriptional control. J Biol Chem 267:24591–24595. [PubMed] [Google Scholar]
- Gekakis N, Staknis D, Nguyen HB, Davis FC, Wilsbacher LD, King DP, Takahashi JS, Weitz CJ. (1998). Role of the CLOCK protein in the mammalian circadian mechanism. Science 280:1564–1569. [DOI] [PubMed] [Google Scholar]
- Gonzalez FJ, Fernandez-Salguero P. (1998). The aryl hydrocarbon receptor: studies using the AHR-null mice. Drug Metab Dispos 26:1194–1198. [PubMed] [Google Scholar]
- Henley DV, Bellone CJ, Williams DA, Ruh TS, Ruh MF. (2004). Aryl hydrocarbon receptor-mediated posttranscriptional regulation of IL-1beta. Arch Biochem Biophys 422:42–51. [DOI] [PubMed] [Google Scholar]
- Iba MM, Fung J, Giannone JV, Okey AB. (2000). Comparative induction of CYP1A1 expression by pyridine and its metabolites. Arch Biochem Biophys 378:299–310. [DOI] [PubMed] [Google Scholar]
- Kim SG, Williams DE, Schuetz EG, Guzelian PS, Novak RF. (1988). Pyridine induction of cytochrome P-450 in the rat: role of P-450j (alcohol-inducible form) in pyridine N-oxidation. J Pharmacol Exp Ther 246:1175–1182. [PubMed] [Google Scholar]
- Kuroki J, Koga N, Yoshimura H. (1986). High affinity of 2,3,4,7,8-pentachlorodibenzofuran to cytochrome P-450 in the hepatic microsomes of rats. Chemosphere, 15, 731–738. [Google Scholar]
- Leclercq IA, Field J, Enriquez A, Farrell GC, Robertson GR. (2000). Constitutive and inducible expression of hepatic CYP2E1 in leptin-deficient ob/ob mice. Biochem Biophys Res Commun 268:337–344. [DOI] [PubMed] [Google Scholar]
- Lieber CS. (2004). The discovery of the microsomal ethanol oxidizing system and its physiologic and pathologic role. Drug Metab Rev 36:511–529. [DOI] [PubMed] [Google Scholar]
- Matsunaga N, Ikeda M, Takiguchi T, Koyanagi S, Ohdo S. (2008). The molecular mechanism regulating 24-hour rhythm of CYP2E1 expression in the mouse liver. Hepatology 48:240–251. [DOI] [PubMed] [Google Scholar]
- Medina-Díaz IM, Elizondo G. (2005). Transcriptional induction of CYP3A4 by o,p’-DDT in HepG2 cells. Toxicol Lett 157:41–47. [DOI] [PubMed] [Google Scholar]
- Mostafa MH, Ruchirawat M, Weisburger EK. (1981). Comparative studies on the effects of various microsomal enzyme inducers on the N-demethylation of dimethylnitrosamine. Biochem Pharmacol 30:2007–2011. [DOI] [PubMed] [Google Scholar]
- Okey AB, Riddick DS, Harper PA. (1994). The Ah receptor: mediator of the toxicity of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) and related compounds. Toxicol Lett 70:1–22. [DOI] [PubMed] [Google Scholar]
- Procházková J, Kabátková M, Bryja V, Umannová L, Bernatík O, Kozubík A, Machala M, Vondrácek J. (2011). The interplay of the aryl hydrocarbon receptor and β-catenin alters both AhR-dependent transcription and Wnt/β-catenin signaling in liver progenitors. Toxicol Sci 122:349–360. [DOI] [PubMed] [Google Scholar]
- Reyes-Hernández OD, Mejía-García A, Sánchez-Ocampo EM, Cabañas-Cortés MA, Ramírez P, Chávez-González L, Gonzalez FJ, Elizondo G. (2010). Ube2l3 gene expression is modulated by activation of the aryl hydrocarbon receptor: implications for p53 ubiquitination. Biochem Pharmacol 80:932–940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodríguez-Sosa M, Elizondo G, López-Durán RM, Rivera I, Gonzalez FJ, Vega L. (2005). Over-production of IFN-gamma and IL-12 in AhR-null mice. FEBS Lett 579:6403–6410. [DOI] [PubMed] [Google Scholar]
- Sato S, Shirakawa H, Tomita S, Ohsaki Y, Haketa K, Tooi O, Santo N, Tohkin M, Furukawa Y, Gonzalez FJ, Komai M. (2008). Low-dose dioxins alter gene expression related to cholesterol biosynthesis, lipogenesis, and glucose metabolism through the aryl hydrocarbon receptor-mediated pathway in mouse liver. Toxicol Appl Pharmacol 229:10–19. [DOI] [PubMed] [Google Scholar]
- Schwanekamp JA, Sartor MA, Karyala S, Halbleib D, Medvedovic M, Tomlinson CR. (2006). Genome-wide analyses show that nuclear and cytoplasmic RNA levels are differentially affected by dioxin. Biochim Biophys Acta 1759:388–402. [DOI] [PubMed] [Google Scholar]
- Sekine S, Lan BY, Bedolli M, Feng S, Hebrok M. (2006). Liver-specific loss of beta-catenin blocks glutamine synthesis pathway activity and cytochrome p450 expression in mice. Hepatology 43:817–825. [DOI] [PubMed] [Google Scholar]
- Sinal CJ, Webb CD, Bend JR. (1999). Differential in vivo effects of alpha-naphthoflavone and beta-naphthoflavone on CYP1A1 and CYP2E1 in rat liver, lung, heart, and kidney. J Biochem Mol Toxicol 13:29–40. [DOI] [PubMed] [Google Scholar]
- Smith AG, Clothier B, Carthew P, Childs NL, Sinclair PR, Nebert DW, Dalton TP. (2001). Protection of the Cyp1a2(−/−) null mouse against uroporphyria and hepatic injury following exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Toxicol Appl Pharmacol 173:89–98. [DOI] [PubMed] [Google Scholar]
- Whitlock JP Jr. (1999). Induction of cytochrome P4501A1. Annu Rev Pharmacol Toxicol 39:103–125. [DOI] [PubMed] [Google Scholar]
- Wong FW, Chan WY, Lee SS. (1998). Resistance to carbon tetrachlorideinduced hepatotoxicity in mice which lack CYP2E1 expression. Toxicol Appl Pharmacol 153:109–118. [DOI] [PubMed] [Google Scholar]
- Xu CX, Krager SL, Liao DF, Tischkau SA. (2010). Disruption of CLOCKBMAL1 transcriptional activity is responsible for aryl hydrocarbon receptor-mediated regulation of Period1 gene. Toxicol Sci 115:98–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon BI, Hirabayashi Y, Kawasaki Y, Kodama Y, Kaneko T, Kanno J, Kim DY, Fujii-Kuriyama Y, Inoue T. (2002). Aryl hydrocarbon receptor mediates benzene-induced hematotoxicity. Toxicol Sci 70:150–156. [DOI] [PubMed] [Google Scholar]
- Yoshioka H, Hiromori Y, Aoki A, Kimura T, Fujii-Kuriyama Y, Nagase H, Nakanishi T. (2012). Possible aryl hydrocarbon receptorindependent pathway of 2,3,7,8-tetrachlorodibenzo-p-dioxininduced antiproliferative response in human breast cancer cells. Toxicol Lett 211:257–265. [DOI] [PubMed] [Google Scholar]
- Zordoky BN, El-Kadi AO. (2010). 2,3,7,8-Tetrachlorodibenzo-p-dioxin and beta-naphthoflavone induce cellular hypertrophy in H9c2 cells by an aryl hydrocarbon receptor-dependant mechanism. Toxicol In Vitro 24:863–871. [DOI] [PubMed] [Google Scholar]